1,2,3-Triazolylidene ruthenium(ii)-cyclometalated complexes and olefin selective hydrogenation catalysis†
Abstract
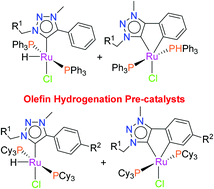
Silver(I) 1,2,3-triazol-5-ylidenes [(RCH2C2N2(NMe)Ph)2Ag][AgCl2] (R = Ph 3a, C6H2iPr33b, C6H2Me33c) and [(PhCH2C2N2(NMe)R)2Ag][AgCl2] (R = C6H4Me 3d, C6H4CF33e) were synthesized and subsequently treated with RuHCl(PPh3)3 and RuHCl(H2)(PCy3)2. The reaction of 3a with RuHCl(PPh3)3 gave RuHCl(PPh3)2(PhCH2C2N2(NMe)Ph) (4a1) as the minor product and the cyclometalated complex RuCl(PPh3)2(PhCH2C2N2(NMe)C6H4) (4a2) as the major product. However, similar reaction with 3b selectively formed the cyclometalated complex RuCl(PPh3)2((C6H2iPr3)CH2C2N2(NMe)C6H4) (4b2). Similarly the silver(I) triazolylidenes 3a and 3b were reacted with RuHCl(H2)(PCy3)2; gave RuHCl(PCy3)2(PhCH2C2N2(NMe)Ph) (5a1), RuCl(PCy3)2(PhCH2C2N2(NMe)C6H4) (5a2) and RuCl(PCy3)2((C6H2iPr3)CH2C2N2(NMe)C6H4) (5b2), respectively. Species 3c, 3d and 3e resulted in the cyclometalated complexes (5c2, 5d2 and 5e2) as the major products as well as the ruthenium-hydride complexes (5c1, 5d1 and 5e1) as the minor products. The cyclometalated species are derived from the ruthenium-hydride complexes via C(sp2)–H activation. These Ru-complexes were shown to act as hydrogenation catalyst precursors for olefinic substrates including those containing a variety of functional groups.

 Please wait while we load your content...
Please wait while we load your content...