Establishing α-bromo-γ-butyrolactone as a platform for synthesis of functional aliphatic polyesters – bridging the gap between ROP and SET-LRP†
Abstract
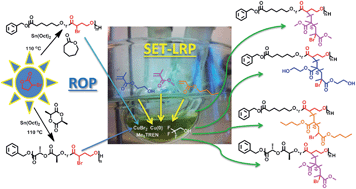
Utilizing α-bromo-γ-butyrolactone (αBrγBL) as a comonomer with ε-caprolactone (εCL) or L-lactide (LLA) produces copolymers with active and available grafting sites, e.g., for SET-LRP, where the choice of the grafting monomers is limited only by one's imagination. This was deduced by utilizing a wide range of different acrylates of varying polarities and was realized with the aid of a fluorinated alcohol, 2,2,2-trifluoroethanol, which acts as a universal solvent for both the hydrophobic macroinitiators and the grafting monomers. Using αBrγBL successfully provides a simple route to merge the two polymerization methodologies, ROP and SET-LRP. αBrγBL inherently meets all of the prerequisites to act as a platform monomer for the synthesis of functional aliphatic polyesters, i.e., it is inexpensive, available, and able to form isolated grafting sites along the polymer chain. The copolymerization of αBrγBL together with two of the most commonly used cyclic ester monomers, ε-CL and LLA, proceeds with a high degree of control and a linear relationship between the feed ratio of αBrγBL and its composition in the copolymer. The formation of isolated units of αBrγBL in the copolymer is visualized by the reactivity ratios of the copolymerization reactions and confirmed by 13C-NMR spectroscopy. The incorporation of isolated αBrγBL is the feature that makes this class of copolymers unique, and it can be considered to provide a route to the “perfect graft copolymer” with a degradable backbone.

 Please wait while we load your content...
Please wait while we load your content...