Surface modification of supramolecular nanotubes and selective guest capture†
Abstract
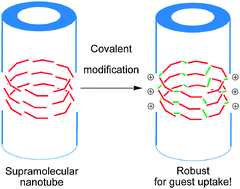
Post-modification of supramolecular assemblies via covalent capture is an important strategy for the fabrication of functional materials. This paper describes the synthesis of supramolecular nanotubes with appended pyridyl groups based on N,N′,N′′-tris(3-methylpyridyl)trimesic amide (TMPTA), and their successful post-modification by 1,4-bis(bromomethyl)benzene. The original tubular morphology of the TMPTA nanotubes was found to stay intact following the modification process. The modified nanotubes are resistant to attack by strong acids and bases. The continuous network of covalent bonds throughout the tubular network is responsible for this stability. The formation of pyridinium cations upon modification results in positively charged surfaces of nanotubes that were found to be highly effective adsorbents for a range of anionic dyes, demonstrating both high adsorption capacity and selectivities over cationic and neutral dye species. These materials can be reused for dye capture without losses in their capture ability.

 Please wait while we load your content...
Please wait while we load your content...