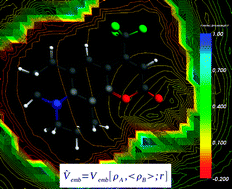
For nine solvents of various polarity (from cyclohexane to water), the solvatochromic shifts of the lowest absorption band of coumarin 153 are evaluated using a computational method based on frozen-density embedding theory [Wesolowski and Warshel, J. Chem Phys., 1993, 97, 9050, and subsequent articles]. In the calculations, the average electron density of the solvent 〈ρB(![[r with combining right harpoon above (vector)]](https://www.rsc.org/images/entities/i_char_0072_20d1.gif) )〉 is used as the frozen density. 〈ρB(
)〉 is used as the frozen density. 〈ρB(![[r with combining right harpoon above (vector)]](https://www.rsc.org/images/entities/i_char_0072_20d1.gif) )〉 is evaluated using the statistical-mechanical approach introduced in Kaminski et al., J. Phys. Chem. A, 2010, 114, 6082. The small deviations between experimental and calculated solvatochromic shifts (the average deviation equals to about 0.02 eV), confirm the adequacy of the key approximations applied: (a) in the evaluation of the average effect of the solvent on the excitation energy, using the average density of the solvent instead of averaging the shifts over statistical ensemble and (b) using the approximant for the bi-functional of the non-electrostatic component of the orbital-free embedding potential, are adequate for chromophores which interact with the environment by non-covalent bonds. The qualitative analyses of the origin of the solvatochromic shifts are made using the graphical representation of the orbital-free embedding potential.
)〉 is evaluated using the statistical-mechanical approach introduced in Kaminski et al., J. Phys. Chem. A, 2010, 114, 6082. The small deviations between experimental and calculated solvatochromic shifts (the average deviation equals to about 0.02 eV), confirm the adequacy of the key approximations applied: (a) in the evaluation of the average effect of the solvent on the excitation energy, using the average density of the solvent instead of averaging the shifts over statistical ensemble and (b) using the approximant for the bi-functional of the non-electrostatic component of the orbital-free embedding potential, are adequate for chromophores which interact with the environment by non-covalent bonds. The qualitative analyses of the origin of the solvatochromic shifts are made using the graphical representation of the orbital-free embedding potential.
You have access to this article
 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?
![[r with combining right harpoon above (vector)]](https://www.rsc.org/images/entities/i_char_0072_20d1.gif) )〉 is used as the frozen density. 〈ρB(
)〉 is used as the frozen density. 〈ρB(![[r with combining right harpoon above (vector)]](https://www.rsc.org/images/entities/i_char_0072_20d1.gif) )〉 is evaluated using the statistical-mechanical approach introduced in Kaminski et al., J. Phys. Chem. A, 2010, 114, 6082. The small deviations between experimental and calculated solvatochromic shifts (the average deviation equals to about 0.02 eV), confirm the adequacy of the key approximations applied: (a) in the evaluation of the average effect of the
)〉 is evaluated using the statistical-mechanical approach introduced in Kaminski et al., J. Phys. Chem. A, 2010, 114, 6082. The small deviations between experimental and calculated solvatochromic shifts (the average deviation equals to about 0.02 eV), confirm the adequacy of the key approximations applied: (a) in the evaluation of the average effect of the 

 Please wait while we load your content...
Please wait while we load your content...