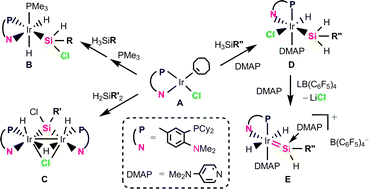
η2-Cyclooctene iridium and rhodium complexes bearing a P,N-chelate ligand C6H3Me-3-PCy2-4-NMe2 (abbreviated as PcyN), (PcyN-P,N)MCl(η2-coe) (1: M = Ir, 2: M = Rh), were synthesised and reactions toward several sterically hindered hydrosilanes were investigated to clarify their reactivity. The reaction of 1 with H2SiMes2 (Mes = mesityl = 2,4,6-trimethylphenyl) proceeded at 40 °C to give a di-iridium complex bridged by a chlorosilylene ligand (PcyN-P,N)2Ir2H2(μ-Cl)(μ-H)(μ-SiClMes) (5). The reaction of 1 with HSiMe2SiMes2Me occurred at room temperature to afford HSiMesMe2 and a silyl complex 6 formed by metallation of an ortho-methyl group of Mes, which slowly dimerised to give a dinuclear complex containing a chlorosilyl ligand (PcyN-P,N)2Ir2H(μ-Cl)(μ-H)[μ-SiMeCl(C6H3-2,4-Me2-6-CH)-Si,C,C] (7). The formation of complexes 5, 6 and 7 suggests that facile migration of Cl from Ir to Si occurs probably via silylene iridium intermediates. Treatment of 1 with HSiMe2SiMe2OMe at room temperature yielded a methoxy-bridged bis(silylene) complex (PcyN-P,N)IrHCl[SiMe2··O(Me)··SiMe2-Si,Si] (8) quantitatively, whose X-ray crystal structure was determined as the first iridium complex of this type. The reactions of 1 and 2 with a bulky trihydrosilane H3SiC(SiMe3)3 underwent an intramolecular γ-Si–Me activation to afford (PcyN-P,N)MHn[SiMe2C(SiMe3)2SiClMe-Si,Si] (9: M = Ir, n = 3; 10: M = Rh, n = 1). Complex 1 even reacted with an extremely sterically hindered terphenylhydrosilane H3SiDmp (Dmp = 2,6-dimesitylphenyl) in the presence of PMe3 to give a chlorosilyl complex (PcyN-P,N)IrH2(PMe3)(SiHClDmp) (11) without intramolecular bond activation. Complex 1 also reacted with H3SiTrip (Trip = 2,4,6-triisopropylphenyl) in the presence of excess 4-(dimethylamino)pyridine (DMAP) at room temperature to give a chlorido(dihydrosilyl) complex (PcyN-P,N)IrHCl(DMAP)(SiH2Trip) (12) instantaneously. Treatment of complex 12 with LiB(C6F5)4·2.5Et2O provided a cationic DMAP-stabilised silylene complex [(PcyN-P,N))IrH2(DMAP)(SiHTrip ← DMAP)][B(C6F5)4] (13).

 Please wait while we load your content...
Please wait while we load your content...