Molecular complexes. Part 12.1 Dimeric toluene, torsional vibrations, dipoles and isomeric complexes in 1H NMR studies of weak arene complexes. Temperature dependence of CH signals
Abstract
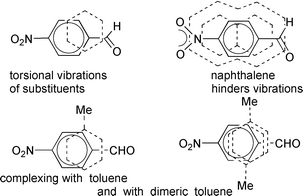
The refined (AUS concept, CCl4, external ref.) 1H NMR method provided association constants K and approximate complex shifts IK for stacking complexes of aromatic hydrocarbons D with A when A is 4-nitrobenzaldehyde (1) or is related to 1. D covers the benzene ring of A but torsionally vibrating substituents in A influence topology and K. In the absence of D, vibrations of A can be slowed by low temperatures making A signals go downfield in accord with an increased planarity of A. Vibration of CHO (a dipole) in complexes of 1 with benzene B or toluene T is made non-symmetric by interactions with the quadrupole of D. The large naphthalene (N) hinders vibrations thus enhancing the contact interface by a more planar 1 whose intramolecular deshielding of protons is increased providing small IK values. Aldehyde 1 forms complexes both with T and with its stacking dimer T2 since IK values for 1–T are significantly greater than for 1–B. Complexing with T2 is not found when the molecular dimensions of A allow a dipole–dipole interaction with T as in face-to-face complexes of 1-ethyltheobromine or of 1-chloro-2,4-dinitrobenzene (7). Different protons give slightly different values of K for 7–T and this points to an isomeric edge-on complex with both T and T2 where K for the two edge-on protons (shortest distance to D) is the sum of the binary K and the small K for the the ternary (7 + T2) complex. Compound 7 and 1,3,5-trimethylbenzene show some of the effect described for 1–N.

 Please wait while we load your content...
Please wait while we load your content...