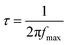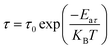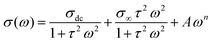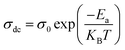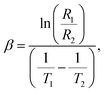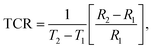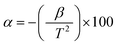Neodymium-engineered relaxor bismuth ferrite nanoparticles: structural tuning and dielectric enhancement for efficient sensor applications
Received
6th September 2025
, Accepted 30th October 2025
First published on 3rd November 2025
Abstract
This paper investigates the synthesis and properties of neodymium-doped bismuth ferrite (BiFeO3) nanoparticles, highlighting their enhanced functionality for advanced applications. The nanoparticles were successfully synthesized with a sol–gel method, where neodymium (Nd) was substituted into the A-site in concentrations ranging from 6% to 10%. A comprehensive analysis of the structural, morphological, and dielectric properties was conducted. X-ray diffraction (XRD) and Rietveld refinement confirmed that all samples maintained a rhombohedral crystal structure with the R3c space group. The incorporation of Nd cations was found to significantly alter the intrinsic distortion of the FeO6 octahedron within the lattice, which is identified as a primary mechanism for property enhancement. Morphological studies showed that the nanoparticles were uniform, with grain sizes between 160 nm and 195 nm. Furthermore, XPS confirmed the presence of Fe2+ ions, which are directly linked to the improved ferroelectric performance. An extensive study of the dielectric properties revealed a change in the electrical conduction mechanism with temperature and notable relaxor behavior. A reduction in the Néel temperature and increased thermal sensitivity were also detected. These remarkable findings demonstrate that Nd substitution is highly effective in tailoring the properties of bismuth ferrite, making these modified nanoparticles excellent candidates for next-generation devices.
I. Brief bibliography
The innovation potential is presented by modified relaxor bismuth ferrite nanoparticles, which find diverse applications.1 Exploration of their structural, morphological, and dielectric properties can lead us to new pathways toward technology. A variety of fields stand to benefit from these nanoparticles, which are becoming essential in areas like electronics and energy storage, among others.2 A substance that displays two or more ferroelectric properties at once, such as ferroelectricity and (anti)ferromagnetism, is known as a multiferroic.3 One of the most interesting multiferroic materials is BiFeO3 (BFO), which has a rhombohedral distortion along the [111] direction and crystallizes in the R3c space group.4 This material exhibits both a ferroelectric transition temperature (Tc = 1103 K) and an antiferromagnetic NEEL temperature (TN = 643 K),5 both of which are higher than the room temperature. However, achieving both electric (d0, lone pair) and magnetic order (dn, fn) at the same time is difficult. This is why single-phase multiferroics are rare.6 Producing pure BFO ceramics or thin films is difficult because the temperature range for stabilizing the phase is narrow.7,8 Researchers have shown that rare earth (RE) substitution at the A-site of the ABO3 perovskite materials is a proven method to address the aforementioned difficulties, improve the material's properties, and reduce impurity phases.9,10 Moreover, the integration of rare-earth elements has been demonstrated to enhance the dielectric characteristics of bismuth ferrite, thereby augmenting the dielectric constant and mitigating dielectric dissipation. Significant changes in impedance, modulus, and intrinsic conduction mechanisms are observed. These improvements are in line with, and have been documented well in, existing literature.11 We specifically selected neodymium among the various rare-earth elements considered because it has a proven ability to stabilize the host lattice and markedly enhance the overall functional properties, as our previous investigations confirmed.12 As perovskite materials are recognized for their relaxor behavior,13,14 which is advantageous for a broad spectrum of applications, they are considered a valuable asset in various fields. The introduction of these materials has created new possibilities for modern acoustic transducers,15 as well as for applications such as medical ultrasound, non-destructive sensing, marine seismic exploration, and energy harvesting.16
The present study aims to demonstrate the suitability of sol–gel synthesized samples as high-performance candidates for thermistor applications, while providing a comprehensive understanding of how neodymium incorporation governs the relaxor behavior of the system.
II. Methods for preparing and characterizing the samples
(1) Sol–gel process
High-purity precursors were used to ensure the production of high quality NdxBi1−xFeO3 nanoparticles (x = 0.06, x = 0.08 and x = 0.1). The precursors contain bismuth nitrate pentahydrate [Bi(NO3)3·5H2O], ferric nitrate nonahydrate [Fe(NO3)2·9H2O], neodymium nitrate hexahydrate [Nd (NO3)3·6H2O] and citric acid (C6H8O7). The other two reagents used included ethylene glycol (C2H6O2) and nitric acid (HNO3). These precursors were mixed in the right amounts. The resulting solution was heated to form a dark red mixture. Heating and stirring continuously took 2 hours. A clear brownish gel formed. The product was ground into powder and heated to 500 °C for 5 hours. The obtained nanoparticles were pressed into pellets. This was done using a hydraulic press. Then they were sintered at 600 °C for 4 hours. Fig. 1 illustrates the entire preparation process in detail.
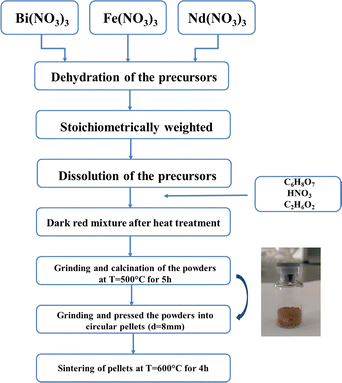 |
| | Fig. 1 Preparation process of NdBFO NPs using the sol–gel synthesis process. | |
(2) Characterization
To determine the crystal structure and phase analysis of the generated compounds, XRD measurements were taken at room temperature using a Bruker D8 instrument (source = Cu-Kα radiation, wavelength = 1.5418 Å). The analysis of the collected data was performed using the Rietveld refinement technique in the FullProf program (version January 2021). The morphology of the synthesized materials was examined using a JEOL JSM-1644 HR scanning electron microscope at the GREMAN laboratory in Tours, France. The surface chemical composition and oxidation states of the samples were investigated by X-ray photoelectron spectroscopy (XPS), employing a standard Al Kα radiation source (Omicron DAR 400, 1486.6 eV). The spectra were collected in constant analyzer energy (CAE) mode, and survey scans were recorded across a binding energy range of 0 to 1400 eV. Finally, the dielectric measurements were carried out over a frequency range of 100 Hz to 1 MHz at varying temperatures from room temperature up to 673 K.
III. Structural and morphological results
(1) Analysis of the sample's structure
As shown in Fig. 2(a), the XRD patterns of all the synthesized samples are depicted. The main indexed peaks indicate the presence of planes with Miller indices (hkl) belonging to the rhombohedral structure with the R3c space group. The experiment diffraction peaks showed good agreement with the standard crystal data [JCPDS No. 01-075-6667]. The FullProf software was utilized to model the peak profile of the NdBFO (x = 0.06, 0.08, and 0.10) samples. The Rietveld analysis was conducted, and the results are displayed in Fig. 2(b). During the refinement process, the pseudo-Voigt function was used to determine the peak shape, and linear interpolation was used to calculate the background. The observed and calculated patterns demonstrate excellent agreement, as confirmed by the satisfactory reliability factors.17Table 1 shows the extracted parameters and indicating that an increase in Nd content slightly influences the lattice parameters and the volume of the structure. These effects could be attributed to the difference between the ionic radii of Nd and Bismuth. In the same way, the electronic density of NdBFO nanostructured materials was obtained from the Rietveld refined XRD patterns. Electron density plots (x, y, z) are essential for understanding atomic-level interactions.18,19 These plots are derived from the reverse Fourier transform20,21 of the structure factors obtained from the Rietveld refinement of XRD information. As the neodymium concentration increased, an up-and-down shift of high-density contours around the neodymium sites along the c axis was observed in the 2D map of electronic density (Fig. 3(a)). The same trend was observed in the 3D map of density variation (Fig. 3(b)). These results reinforce the crystallographic findings and demonstrate a consistent trend across all studied lattice parameters. The influence of neodymium on the intrinsic distortion of the samples, particularly on the FeO6 octahedron in the structures, was investigated by studying parameter bonds using VESTA software. The results obtained are shown in Fig. 2(c) and summarized in Table 2. The Fe–O–Fe angle in the FeO6 octahedron plays a crucial role in the electrical, magnetic, and electronic properties of materials, particularly at the nanoscale. In a perfect structure, this angle would be 180°.22 However, if the angle is less than 145°, it can affect the materials’ electrical properties and if the angle is greater than 160°, the materials can become less magnetic.23,24 Our earlier tests showed that adding more neodymium could disrupt the structure of the FeO6 octahedron and reduce the Fe–O–Fe angle from 158.06° to around 156.29° (Fig. 4). In this case, a good balance between ferroelectricity and magnetism was achieved. The aforementioned properties make the samples NdxBi1−xFeO3 NPs (x = 0.06, x = 0.08, x = 0.1) suitable candidates for ferroelectric capacitors and ferroelectric random-access memory (FeRAM).25
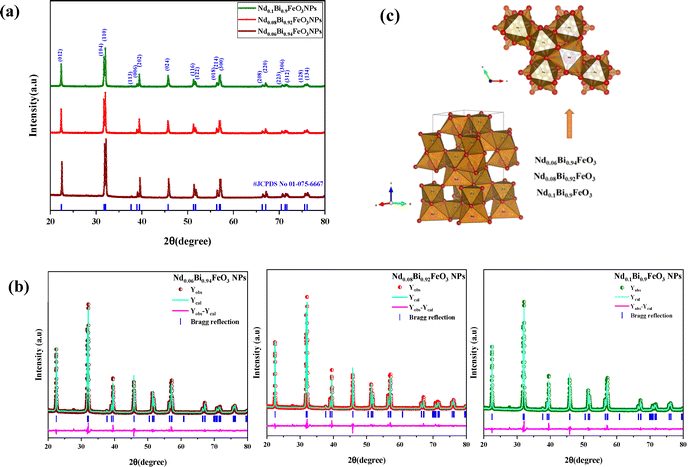 |
| | Fig. 2 (a) X-ray diffraction patterns for NdxBi1−xFeO3 (x = 0.06, 0.08, 0.1) nanoparticles. (b) NdBFO's FullProf Rietveld refinement simulation. (c) The visualization structure of the NdBFO NPs using Vesta software. | |
Table 1 Crystallographic parameters of NdBFO NPs
| Crystallographic parameters |
NdxBi1−xFeO3 |
|
x = 0.06 |
x = 0.08 |
x = 0.10 |
|
a = b (Å) (±0.0001) |
5.5748 |
5.5753 |
5.5748 |
|
c (Å) (±0.0002) |
13.8397 |
13.8479 |
13.8397 |
|
c/a |
2.4825 |
2.4837 |
2.4825 |
|
D
sch size (nm) |
51.45 |
52.35 |
47.29 |
| Volume (Å3) (±0.01) |
372.498 |
372.787 |
372.496 |
|
R
p
|
9.46 |
9.47 |
9.58 |
|
R
wp
|
9.67 |
9.75 |
9.75 |
|
χ
2
|
2.38 |
2.5 |
2.43 |
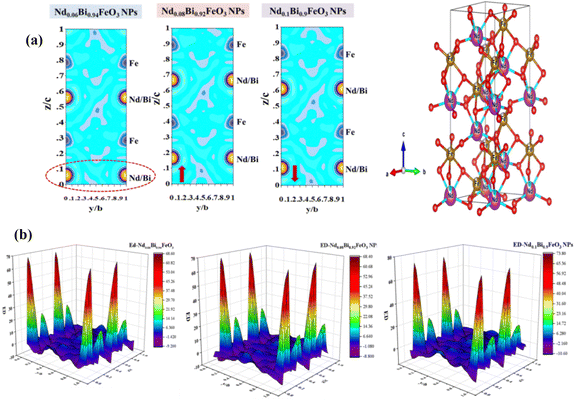 |
| | Fig. 3 (a) 2D contours for electron density of the NdBFO samples. (b) 3D Fourier map for electron density of NdBFO samples. | |
Table 2 NdBFO sample's parameters bond angle and bond length
| Molecular formula |
Space group |
Bond name |
Bond length (Å) |
Bond type |
Bond angle (deg) |
| Nd0.06Bi0.94FeO3 |
R3c |
Bi/Nd–O |
2.518, 2.367 |
Bi–O–Bi |
108.252 |
| Fe–O |
2.133, 1.899 |
Fe–O–Fe |
158.064 |
| Nd0.08Bi0.92FeO3 |
R3c |
Bi/Nd–O |
2.499, 2.383 |
Bi–O–Bi |
108.381 |
| Fe–O |
2.172, 1.869 |
Fe–O–Fe |
156.993 |
| Nd0.1Bi0.9FeO3 |
R3c |
Bi/Nd–O |
2.525, 2.358 |
Bi–O–Bi |
108.307 |
| Fe–O |
2.132, 1.902 |
Fe–O–Fe |
156.290 |
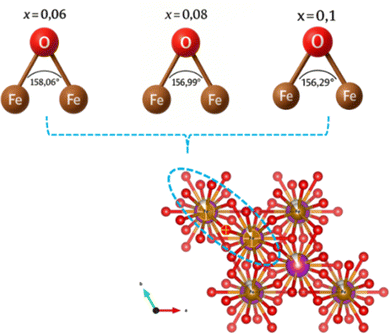 |
| | Fig. 4 Representation of the bond angle Fe–O–Fe of NdBFO NPs. | |
(2) Morphological analysis of synthetic samples
The samples’ microstructures, as revealed by SEM, show distinct spherical shapes and irregular agglomeration, forming stronger interconnections among them (Fig. 5). Thorough investigation of the size distribution histograms, employing ImageJ software, reveals that the synthesized samples, namely Nd0.06Bi0.94FeO3, Nd0.92Bi0.08FeO3, and Nd0.1Bi0.9FeO3, possessed particle sizes of 195 nm, 192 nm, and 160 nm, respectively.
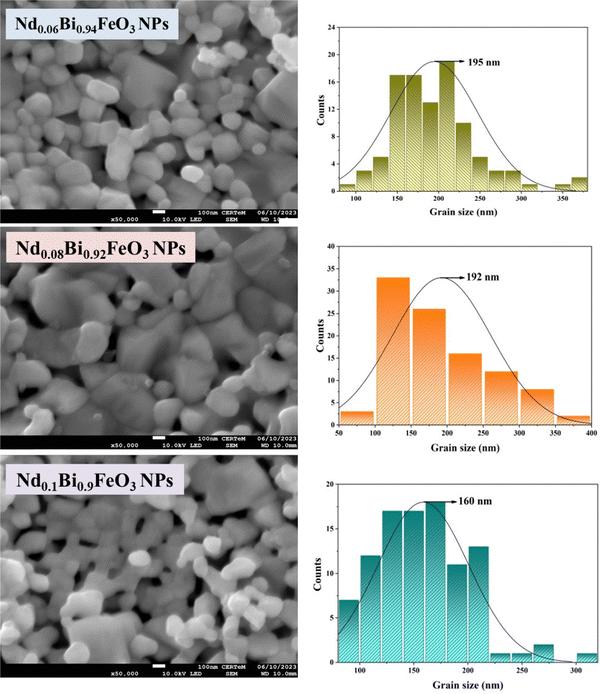 |
| | Fig. 5 SEM images of the Sol–gel synthesized samples and histograms of particle size distribution. | |
(3) XPS characterization of the nanoparticles
To verify the presence of the expected elements in each synthesized sample and to confirm the successful synthesis of high-quality samples free of residues or impurities, measurements using X-ray photoelectron spectroscopy (XPS) were performed. Fig. 6 illustrates the survey spectrum of the synthesized samples, confirming the presence of Nd, Bi, Fe and O, as indicated by their characteristic peaks. As shown in Fig. 7, two broad peaks correspond to Fe 2p3/2 and Fe 2p1/2. These peaks are located in the binding energy range of 700 eV to 730 eV. This indicates the presence of both Fe3+ and Fe2+ oxidation states. The XPS peak fitting was executed using the Thermo Avantage software, employing a mixed Gaussian–Lorentzian (GL) peak shape.26 The background subtraction and peak fitting parameters were carefully optimized within the software to ensure that accurate and reliable deconvolution results were achieved. The C 1s peak, at 284.7 eV, was used as a reference for the binding energy calibration. The deconvolution of the data reveals a decrease in the atomic percentage of Fe2+ ions compared to Fe3+ ions with increasing neodymium content (Table 3), confirming the significant role of neodymium in the structural configuration. This observation is consistent with previous research results.27
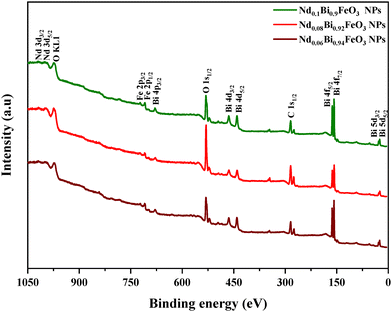 |
| | Fig. 6 XPS spectra of the NdBFO NPs. | |
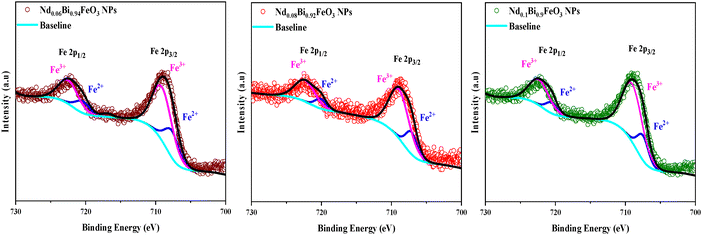 |
| | Fig. 7 Fe 2p XPS spectral fitting for NdBFO samples. | |
Table 3 Oxidation states of iron in the synthesized nanoparticles
| Samples |
Fe3+ (%) |
Fe2+ (%) |
| Nd0.06Bi0.94FeO3 |
73.51 |
26.49 |
| Nd0.08Bi0.92FeO3 |
79.41 |
20.59 |
| Nd0.1Bi0.9FeO3 |
80.46 |
19.54 |
Based on the structural and morphological analysis, it can be concluded that well-structured and impurity-free samples were successfully synthesized using the simple Sol–Gel method. The dielectric properties of the samples will now be explored.
IV. Examination of the nanoparticles’ dielectric qualities
(1) Frequency-dependent dielectric relaxation response of samples at a given temperature
The analysis of dielectric properties is a pivotal aspect of the study of ceramic materials. This investigation elucidates various phenomena, including the relaxation process, the degree of polarization, the impact of defects, and the underlying mechanisms contributing to dielectric loss. The electrodes were prepared by coating both surfaces of the pellets with a thin and uniform layer of silver lacquer to ensure good electrical contact during the measurements. The pellets were oval in shape, with an average diameter of approximately 8 mm and a thickness of about 1 mm. The measurements of dielectric parameters were performed over a frequency range from 100 Hz to 1 MHz, and within a specified temperature range between 537 K and 575 K. As depicted in Fig. 8, the dielectric parameters (ε′, ε′′, and tan![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) δ) of all samples demonstrate significantly elevated values in the low-frequency domain, a typical behavior for dielectrics, attributed to the contribution of various types of polarization, including dipolar, space-charge, electronic, and ionic polarizations. At elevated frequencies, the phenomena mainly involve atomic and electronic polarizations, which yield a decrease in the losses. Furthermore, the Maxwell–Wagner model, in alignment with Koops’ phenomenological theory, provides insight into the variations in dielectric parameters with frequency.28,29 This model suggests that in low-frequency regimes, grain boundaries have a significant impact, whereas grains become more pronounced at higher frequencies. In the low-frequency domain, the substantial presence of highly resistive grain boundaries facilitates significant space charge polarization by electrons. Conversely, at higher frequencies, the dielectric constant decreases as the electrons move between grains and grain boundaries. The curve depicting the tangent loss follows a similar trend to that of the dielectric constant, with high values at low frequencies due to the increased resistance of grain boundaries, which necessitates more energy for electron mobilization. As the frequency increases, the resistance decreases, resulting in a corresponding reduction in the tangent loss values.30 This low frequency dispersion effect is commonly seen in ferroelectric and dielectric materials.31,32 As the temperature increased, the dielectric constant of the two samples (Nd0.06Bi0.94FeO3 NPs and Nd0.08Bi0.92FeO3 NPs) exhibited an upward trend at low frequencies. However, a decline in the dielectric constant was observed with temperature at the high neodymium concentration (Nd0.1Bi0.9FeO3 NPs). The presence of neodymium disorder cations leads to the relaxor characteristics of this sample.33 Yadav et al. suggest another possible explanation for this: the electrons may be jumping between Fe3+ and Fe2+ ions.34 Similar types of behavior have been observed in other eco-friendly ABO3-type relaxor-ferroelectric ceramics.35–37
δ) of all samples demonstrate significantly elevated values in the low-frequency domain, a typical behavior for dielectrics, attributed to the contribution of various types of polarization, including dipolar, space-charge, electronic, and ionic polarizations. At elevated frequencies, the phenomena mainly involve atomic and electronic polarizations, which yield a decrease in the losses. Furthermore, the Maxwell–Wagner model, in alignment with Koops’ phenomenological theory, provides insight into the variations in dielectric parameters with frequency.28,29 This model suggests that in low-frequency regimes, grain boundaries have a significant impact, whereas grains become more pronounced at higher frequencies. In the low-frequency domain, the substantial presence of highly resistive grain boundaries facilitates significant space charge polarization by electrons. Conversely, at higher frequencies, the dielectric constant decreases as the electrons move between grains and grain boundaries. The curve depicting the tangent loss follows a similar trend to that of the dielectric constant, with high values at low frequencies due to the increased resistance of grain boundaries, which necessitates more energy for electron mobilization. As the frequency increases, the resistance decreases, resulting in a corresponding reduction in the tangent loss values.30 This low frequency dispersion effect is commonly seen in ferroelectric and dielectric materials.31,32 As the temperature increased, the dielectric constant of the two samples (Nd0.06Bi0.94FeO3 NPs and Nd0.08Bi0.92FeO3 NPs) exhibited an upward trend at low frequencies. However, a decline in the dielectric constant was observed with temperature at the high neodymium concentration (Nd0.1Bi0.9FeO3 NPs). The presence of neodymium disorder cations leads to the relaxor characteristics of this sample.33 Yadav et al. suggest another possible explanation for this: the electrons may be jumping between Fe3+ and Fe2+ ions.34 Similar types of behavior have been observed in other eco-friendly ABO3-type relaxor-ferroelectric ceramics.35–37
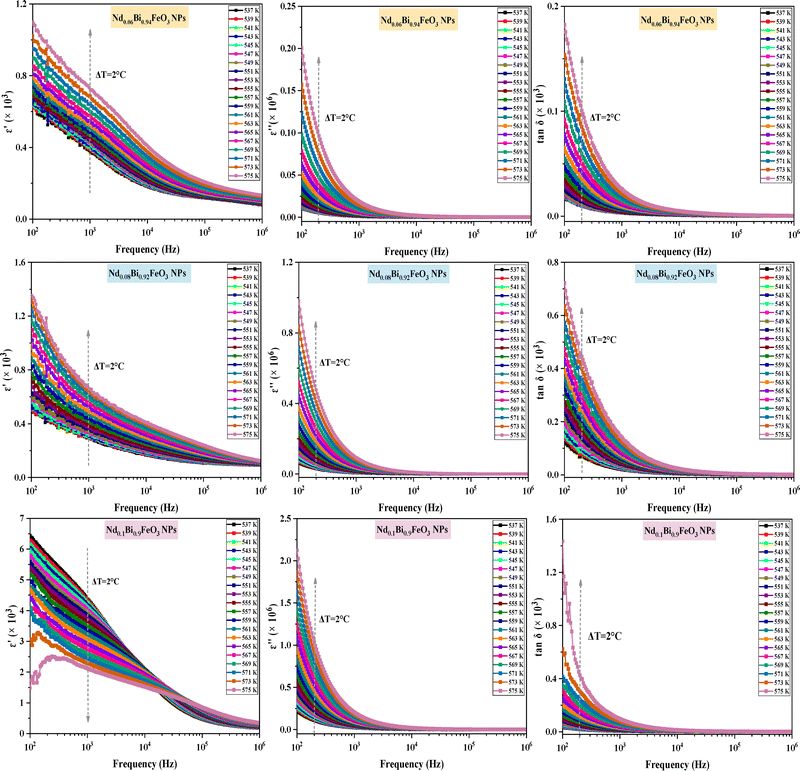 |
| | Fig. 8 Frequency dielectric parameters in a specific temperature range of NdxBi1xFeO3 (x = 0.06, x = 0.08 and x = 0.1) NPs. | |
(2) Thermal dielectric characteristics at a particular frequency
The temperature dependence of the dielectric permittivity and tangent loss at different frequencies (1 kHz, 10 kHz, 103 kHz, and 1 MHz) is presented in Fig. 9 for x = 0.06, 0.08, and 0.1, respectively. As the temperature increases, a notable rise in the dielectric parameters is observed, which can likely be attributed to the involvement of the thermally activated dipoles.38 Within the temperature range up to 373 K, the dielectric constant demonstrates consistent behavior across all frequencies. It exhibits a gradual increase, with a prominent peak observed around the NEEL temperature (TN). A distinctive feature of relaxing ferroelectrics is that the broad peak around the TN shifts toward higher temperatures and diminishes in intensity as the frequency increases from 1 kHz to 1 MHz for all samples. According to the literature,39 magnetoelectric coupling may directly cause this type of abnormal dielectric behavior. The observed peak in the temperature, ranging from 573 K to 650 K in Nd0.1Bi0.9FeO3 samples, confirms the progressive relaxor behavior induced by the addition of neodymium. This unusual behavior in magnetically ordered systems is predicted by Landau–Devonshire theory of phase transitions as an influence of the disappearance of the magnetic order on the electric order.40 As the temperature increases, tanδ shows a marked increase in the higher temperature region for different frequencies (across all compositions), which is due to the dominance of the conductance.41 Finally, studying the material's electric properties (see Fig. 10) showed that adding neodymium causes a drop in the NEEL temperature from TN = 619 K to TN = 520 K making these samples suitable for many applications.
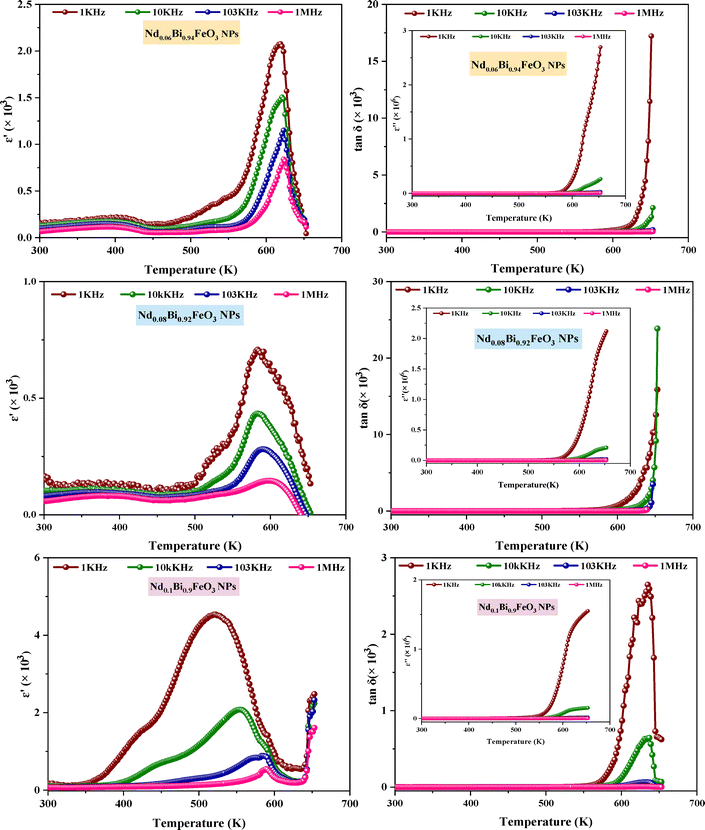 |
| | Fig. 9 Thermal dielectric parameters in a specific frequency range of NdxBi1xFeO3(x = 0.06, x = 0.08 and x = 0.1) NPs. | |
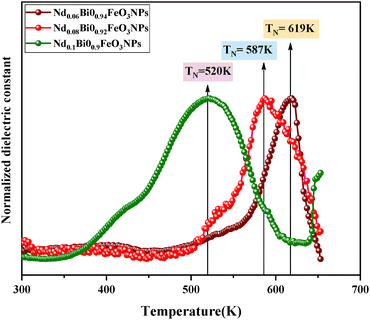 |
| | Fig. 10 Variation of the NEEL temperature of the NdBFO NPs. | |
(3) Modulus spectrum study
Electrical modulus spectroscopy is an advanced technique used to measure the properties of materials. These include the effects of grain boundaries, electrical conductivity, relaxation time, electrode polarization, conduction, and bulk properties. It can also be used to analyze the different electrical processes that occur within a material. To determine M′ and M′′, we used the formula given below:42| | 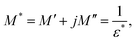 | (1) |
| | 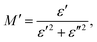 | (2) |
| | 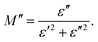 | (3) |
As shown in Fig. 11, the value of M′ increases as the frequency increases for all the samples. These results could be explained by the lack of a restoring force. This is the force that moves charges when an electric field is formed. These characteristics show that the material is not significant impacted by electrode polarization.43 The plot of the imaginary part M′′ of the modulus is characterized by the presence of distinct asymmetric peaks in the higher frequency range. As the temperature increased from 537 K to 575 K, the peak value of M′′ declines. This observation indicates that, as the temperature rises, the relaxation time decreases. The shift in the peak position further demonstrates that the samples undergo thermally driven relaxation processes, resulting in charge carrier hopping.44–46 The Cole–Cole diagram of the modulus was plotted, where a change in its shape was observed. Initially, the diagram showed an S-shaped curve for 6% neodymium, however it gradually transformed into an arc at 10% neodymium. The presence of a single arc indicates that the grain boundary is more important for conductivity than the individual grains.47 The plot shape changes when neodymium is substituted, and an incomplete arc appears at higher temperatures. This indicates that the material's relaxation behavior broadens, confirming the presence of non-Debye relaxation. These findings are consistent with those reported in several previous studies.48–50
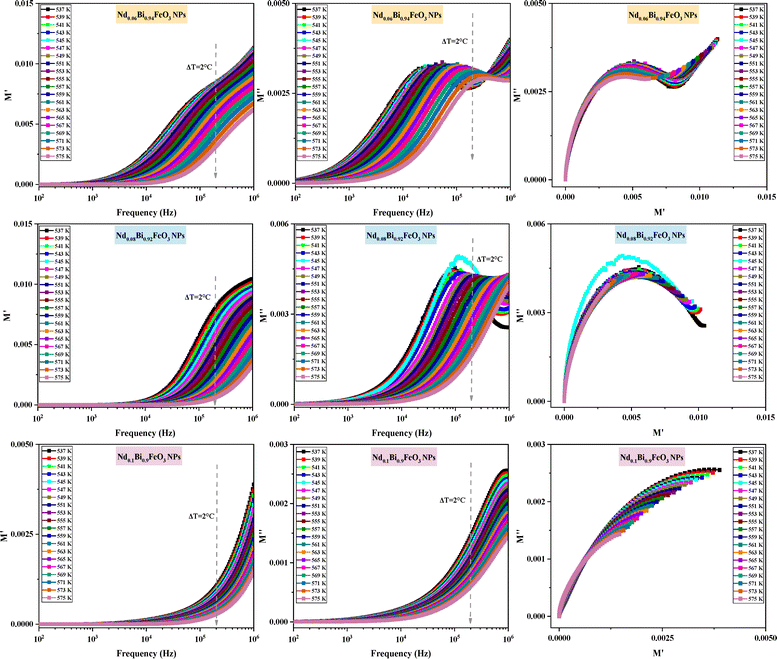 |
| | Fig. 11 Modulus spectra of NdxBi1−xFeO3 NPs. | |
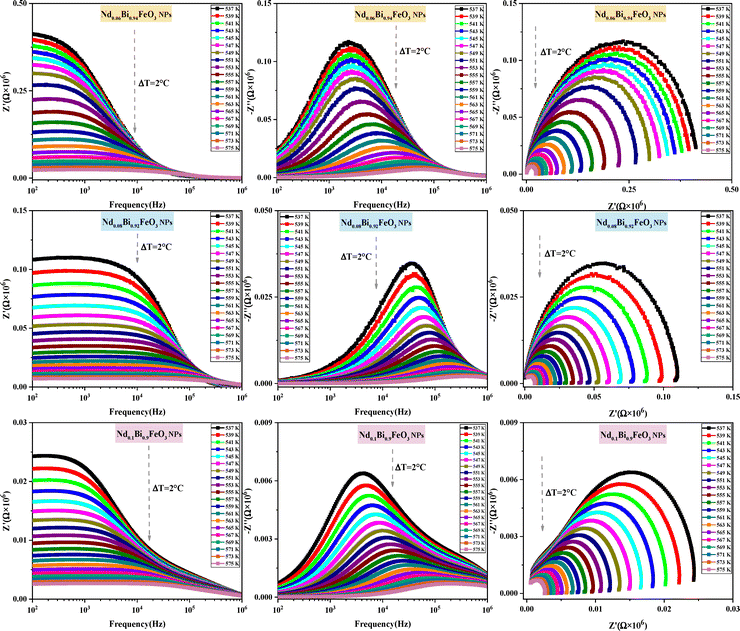
(4) Nanoparticles complex impedance analysis
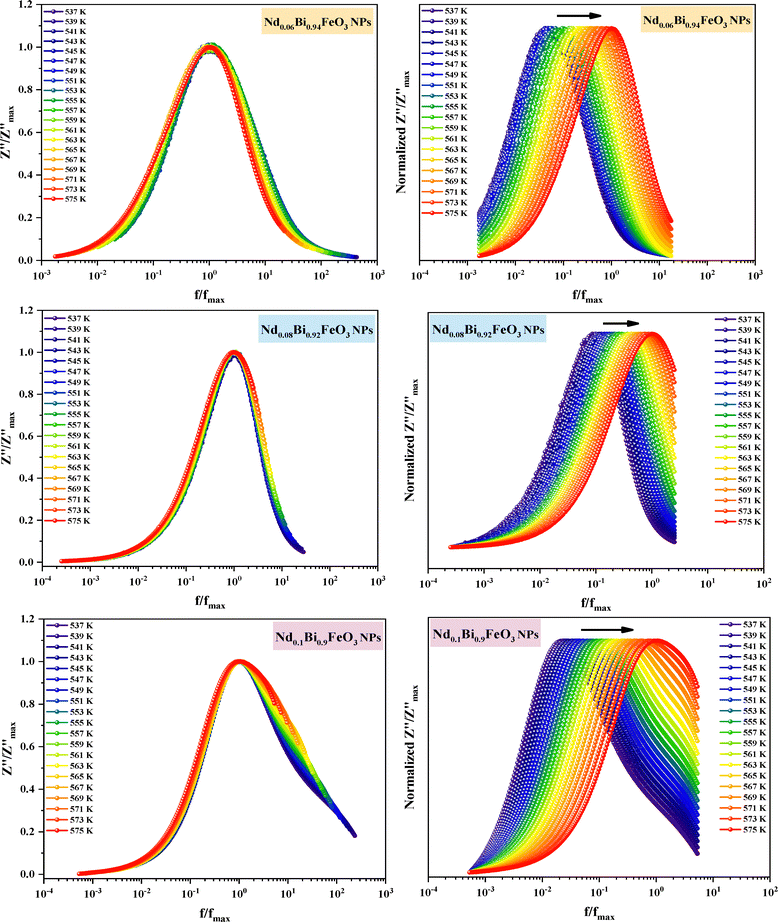
The study of how grains, grain boundaries, and electrode polarization affect the dielectric properties of a material is commonly performed using complex impedance spectroscopy (CIS). The real part, Z′, and the imaginary part, Z′′, of the complex impedance51 were measured at different temperatures (537–575 K) and frequencies (1 Hz–1 MHz). Fig. 12 shows the changes in the real Z′ and imaginary Z′′ parts of the complex impedance Z* and the Nyquist plots concerning frequency for the Nd0.06Bi0.94FeO3, Nd0.08Bi0.92FeO3, and Nd0.1Bi0.9FeO3 NPs. As can be seen in this figure, Z′ and Z′′ attain their maximum values at lower frequencies, indicating heightened polarization within the nanoparticles. The observed decline in Z′ and Z′′ values at higher frequencies and temperatures indicates of a decrease in resistance. This abrupt decline is influenced by three primary factors: grain–grain contact, grain boundaries, and electrode interfaces. Collectively, these factors result in a reduction in resistance. This phenomenon could be the underlying cause of the material's enhanced conductivity with temperature at higher frequencies, exhibiting a negative temperature coefficient of resistance (NTCR).52,53 A single arc is visible in samples containing 6% and 8% Nd, suggesting that grain resistance is the dominant factor. On the other hand, the formation of a double arc in the 537–575 K temperature range for 10% neodymium reveals simultaneous grain and grain boundary contributions to the conduction process. The change from a single to a double arc in the Nyquist diagram emphasizes non-Debye relaxation and is a relaxor effect confirmed to occur at high neodymium concentrations. According to existing literature,54 the phenomenon under investigation may be attributed to numerous factors, including, but not limited to, grain orientation and distribution, grain boundary presence, atomic defect distribution, and stress–strain phenomena. This behavior has been observed in rare earth-doped ferroelectric materials. To investigate the potential impact of temperature on the distribution of relaxation times in our samples, Z′′ data is plotted using scaled coordinates, specifically 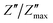 vs. f/fmax, where
vs. f/fmax, where 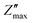 and fmax represent the peak value and it corresponding frequency of the peak in Z curves. As can be seen in Fig. 13, all peaks converge into a single master curve at different temperatures, suggesting that charges undergo dynamic processes with a consistent activation energy across various timescales. Additionally, plots of the
and fmax represent the peak value and it corresponding frequency of the peak in Z curves. As can be seen in Fig. 13, all peaks converge into a single master curve at different temperatures, suggesting that charges undergo dynamic processes with a consistent activation energy across various timescales. Additionally, plots of the 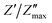 relaxation peak versus frequency show a gradual shift towards higher frequencies as the temperature increases. This further highlights the existence of multiple equilibrium states and the distribution of relaxation times in the prepared samples.55 Within the studied temperature range (537–575 K), neodymium does not alter the fundamental nature of the relaxation process, but it does affect the relaxation time value. This reflects the high homogeneity of the material in terms of the dynamics of the charge carriers or dipoles responsible for dielectric relaxation. The corresponding relaxation time τ is obtained through the following equation:
relaxation peak versus frequency show a gradual shift towards higher frequencies as the temperature increases. This further highlights the existence of multiple equilibrium states and the distribution of relaxation times in the prepared samples.55 Within the studied temperature range (537–575 K), neodymium does not alter the fundamental nature of the relaxation process, but it does affect the relaxation time value. This reflects the high homogeneity of the material in terms of the dynamics of the charge carriers or dipoles responsible for dielectric relaxation. The corresponding relaxation time τ is obtained through the following equation:| | 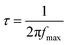 | (4) |
where fmax is the frequency at which Z′′ reaches its maximum value. To calculate the activation energy of the relaxation process, τ could be efficiently fitted using the Arrhenius equation:56| | 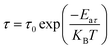 | (5) |
As shown in Fig. 14, the activation energies for the relaxation process of Nd0.06Bi0.94FeO3, Nd0.08Bi0.92FeO3, and Nd0.1Bi0.9FeO3 NPs are 1.18 eV, 0.8 eV, and 1.15 eV, respectively. The fluctuation in these values highlights the impact of neodymium on the relaxation process.
 |
| | Fig. 12 Impedance spectra of NdxBi1−xFeO3 NPs. | |
 |
| | Fig. 13 Frequency-dependent scaling behavior of Z′′ at varying temperatures. | |
 |
| | Fig. 14 Arrhenius plots of ln (τ) versus 1000/T for the nanoparticle samples. | |
(5) Transport characteristics of the synthetized NPs
A detailed analysis of the frequency-dependent electrical conductivity of the samples at various temperatures was conducted to investigate their conduction behavior and identify the parameters that influence the conduction process. Fig. 15 shows a plateau region at low frequencies and a dispersion region at higher frequencies. The plateau section indicates long-range DC conductivity, while the dispersion section indicates localized AC conductivity. The increase in conductivity (σ) is due to the presence of thermally activated carriers. The frequency dispersion of electrical conductivity is typically well described by the simple Jonscher equation. However, due to the modification of the relaxation process induced by neodymium, a modified form of the Jonscher equation was employed to accurately represent this behavior, as presented below:57| | 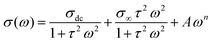 | (6) |
 |
| | Fig. 15 Variation in electrical conductivity with frequency at selected temperatures for the NPs. | |
The following is a list of the variables in this equation:
• σdc is the conductivity at low frequency
• σ∞ is the conductivity at high frequency
• τ is a characteristic relaxation time
• A is a temperature-dependent parameter
• n is the power law exponent factor (0 ≤ n ≤ 1)
Fig. 16(a) shows how the parameter n changes with temperature. This helps us to understand the nature of the materials’ electric conductivity. For the Nd 6% and Nd 8% composites, the behavior follows OLPT (overlapping large polaron tunneling). This means that n decreases at low temperatures and then increases at higher temperatures. By contrast, the third composite (Nd 10%) exhibits a conduction mechanism called NSPT (non-overlapping small polaron tunneling), as evidenced by a continuous increase in the n parameter with temperature. Increasing the neodymium (Nd) content leads to a beneficial structural distortion, stabilizing the crystal lattice and reducing defect concentration. This improved structural order decreases oxygen vacancies and defects, modifying the conduction mechanism. Consequently, the charge carriers become more localized due to the distortion, shifting the conduction from OLPT to NSPT. This change reflects a transition to a more thermally activated, hopping-type transport consistent with the stabilized and less defective Nd-doped structure. In addition, the activation energy was calculated using the following Arrhenius relation:
| | 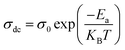 | (7) |
where
KB and
σ0 stand for the Boltzmann constant and pre-exponential factor, respectively. According to
Fig. 16(b), the decrease in the activation energy, observed with increasing neodymium, can be attributed to structural changes induced by the insertion of Nd
3+ into the lattice. The angles and distances of the Fe–O–Fe bonds observed in the previous section are affected by these changes. The hopping barriers for charge carriers are reduced by these changes, which facilitates their mobility and results in a gradual reduction in activation energy. This variation has been reported by several researchers.
58
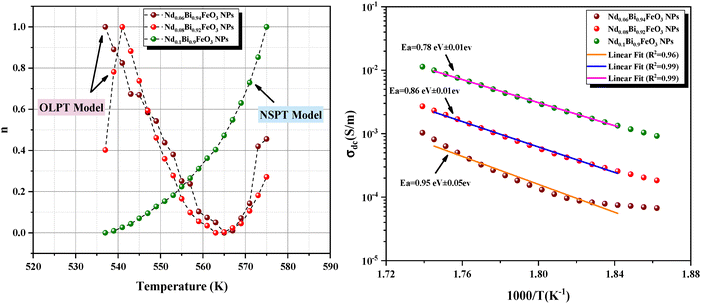 |
| | Fig. 16 (a) The mechanism of conductivity of NPs. (b) Activation energy from DC conductivity as a function of 1000/T. | |
In summary, this rise in conductivity dependent on temperature suggests that our samples show temperature-dependent hopping behavior. This indicates that the compounds have a negative temperature coefficient of resistance (NTCR).
(6) NTC thermistor behavior of the nanoparticles
In this section, we will demonstrate the reliability of our synthesized NdxBi1−xFeO3 NPs (x = 0.06, x = 0.08, x = 0.1) in NTC thermistor applications. Our previous dielectric results indicate that our samples are excellent candidates for thermal sensors, such as thermistors. In the following, we will assess their performance as NTC thermistor candidates by comparing them to traditional ones. Fig. 17 shows the resistance behavior at temperatures between 530 K and 580 K. We observed an exponential decrease in resistance with increasing temperature for each NPs. The development of a highly temperature-sensitive thermistor requires a high thermistor constant (β), a pronounced non-linearity in the temperature coefficient of resistance (TCR), and an extensive sensitivity range. These characteristics can be determined using the following formula:59| | 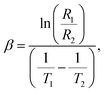 | (8) |
| | 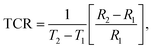 | (9) |
| | 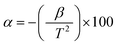 | (10) |
The obtained β values, for NdxBi1−xFeO3 (x = 0.06, x = 0.08, and x = 0.1) NPs, were around 574 K. This is higher than that published elsewhere60 (see Table 4 for comparison). This underscores their potential for use in advanced applications. In addition, the sensitivity parameter, calculated using the Steinhart–Hart equation (eqn (10)), revealed that all synthesized samples showed a promising value, including in the range between −1% and 12%. This wider range is more advantageous and surpasses that of conventional thermistors.
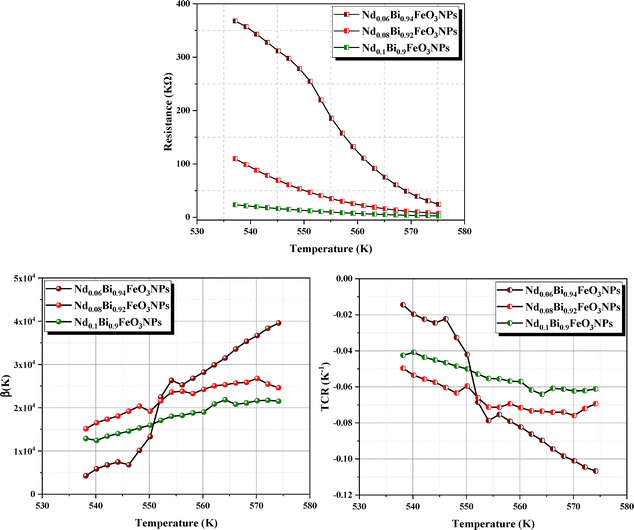 |
| | Fig. 17 Thermal sensors parameters of samples ranging in temperature (530–580 K). | |
Table 4 Comparison of sensitivity values with other materials at T = 574 K
| Compounds |
β (K) |
Ref. |
| NdxBi1−xFeO3 |
39![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 578 578 |
This work |
| Ba3Bi2WO9 |
8000 |
61
|
| ZnFe2O4 |
5820 |
62
|
| CaTiO3 |
10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 795 795 |
63
|
| Dimond thermistor |
8012 |
64
|
V. Conclusion
A cost-effective and straightforward sol–gel preparation technique was used to successfully synthesize environmentally friendly multiferroic materials based on neodymium-modified bismuth ferrite nanoparticles. Both experimental and simulation studies, including Rietveld refinement, confirmed the rhombohedral structure with the R3c space group and neodymium-induced intrinsic distortion modification. Our findings revealed that the relaxation behaviour was attributed to Maxwell–Wagner polarization in the dielectric properties. Analysis of the complex electric modulus indicated a conduction process, and impedance studies confirmed the negative temperature coefficient (NTC) thermistor behavior. The transport characteristics of the synthesized materials were observed with a transition from the OLPT mechanism to the NSPT. In summary, structural analysis revealed the enhancement and promotion of intrinsic distortion of the FeO6 octahedron by the substitution of neodymium, resulting in improved structural stability of the synthesized materials. These structural enhancements make the materials ideal candidates for use in ferroelectric capacitors and ferroelectric random-access memory (FeRAM). Additionally, dielectric measurements revealed a noticeable decline in the NEEL temperature and a modification in the conduction mechanism that renders these materials suitable for spintronic devices, magnetic refrigeration, and magnetic drug targeting. Finally, analysis of thermistor properties showed that the synthesized structures are well-suited for use in NTC thermistors and exhibit higher sensitivity than conventional thermistors. These unique characteristics make the synthesized samples highly promising for many different applications.
Conflicts of interest
There are no conflicts to declare.
Data availability
Detailed data for current research findings and studies is available from the corresponding author upon request.
References
- S.-Y. Chen, R. Montecillo and K.-C. Feng,
et al., High-efficiency energy storage in Nd-doped (1−x)BiFeO3–xBaTiO3 relaxor ferroelectric ceramics, Ceram. Int., 2023, 49(16), 26625–26634 CrossRef CAS.
- M. S. Bozgeyik, N. Kirkgecit and R. K. Katiyar,
et al., Monitoring structural variation on Gd ratio of La modified bismuth ferrite ceramics with enhanced magnetization, J. Alloys Compd., 2020, 819, 153050 CrossRef CAS.
- A. N. Ganie, I. Irshad and A. Tahir,
et al., Yb modification of bismuth Ferrite: Unveiling Optical, Dielectric, and electrochemical properties for advanced applications, Inorg. Chem. Commun., 2024, 168, 112905 CrossRef.
- M. M. Rhaman, M. S. Miah and T. Ahmad, Investigation of magnetic and electric properties of bismuth ferrite nanoparticles at different temperatures, Nano-Struct. Nano-Objects, 2024, 39, 101304 CrossRef CAS.
-
M. M. Rhaman, N. Rahman and S. Akther, et al., Optical and Structural Property Analysis of Samarium and Cobalt Co-doped Bismuth Ferrite Nanoparticles, in 2024 6th International Conference on Sustainable Technologies for Industry 5.0 (STI), IEEE, 2024, pp. 1–5 Search PubMed.
- N. V. Srihari, K. B. Vinayakumar and K. K. Nagaraja, Magnetoelectric coupling in bismuth ferrite—challenges and perspectives, Coatings, 2020, 10(12), 1221 CrossRef CAS.
- A. Taibi, S. Chaguetmi and P. E. Sanchez-Jimenez,
et al., Pure perovskite BiFeO3–BaTiO3 ceramics prepared by reaction flash sintering of Bi2O3–Fe2O3–BaTiO3 mixed powders, Ceram. Int., 2021, 47(19), 26947–26954 CrossRef CAS.
- X. Chen, P. Gao and C. Liu,
et al., Synthesis, structural stability and phase diagram of BiFeO3–CaSnO3 ceramics, Ceram. Int., 2023, 49(1), 1436–1443 CrossRef CAS.
- E. C. Singh and H. B. Sharma, Structural and dielectrical analysis of rare earth doped bismuth ferrite nanoparticle at high frequency, Mater. Today: Proc., 2022, 65, 2788–2793 Search PubMed.
- M. M. Rhaman, M. A. Matin and M. A. Hakim,
et al., Optical and electrical properties of impurity-less multiferroic bismuth ferrite nanoparticles, Mater. Sci. Eng., B, 2022, 275, 115501 CrossRef CAS.
- S. Pattanayak, Choudhary, R. N. P. Effect of Rare-earth Modification on Structural, Ferroelectric and Ferromagnetic Properties of BiFeO3 Ceramics at Room Temperature, Trans. Electr. Electron. Mater., 2025, 1–12 Search PubMed.
- I. Chiba, M. Zannen and N. Hamdaoui,
et al., Buckling effect of Neodymium on the Structural, Morphological, Optical, and Dielectric Properties of Bismuth Ferrite Nanoparticles, Ceram. Int., 2025, 1–19 Search PubMed.
- E. Lupi, R. B. Wexler and D. Meyers,
et al., Engineering relaxor behavior in (BaTiO3)n/(SrTiO3) n superlattices, Adv. Mater., 2023, 35(51), 2302012 CrossRef CAS.
- M. K. Rout and S. Keshri, Crossover to the negative dielectric constant in La3+ and Dy3+ doped Co-Zn spinel nanoferrites, J. Alloys Compd., 2023, 965, 171425 CrossRef CAS.
- A. Safari, E. K. Akdoğan and J. D. Leber, Ferroelectric ceramics and composites for piezoelectric transducer applications, Jpn. J. Appl. Phys., 2022, 61(SN), SN0801 CrossRef CAS.
- I. A. Parinov and A. V. Cherpakov, Overview: state-of-the-art in the energy harvesting based on piezoelectric devices for last decade, Symmetry, 2022, 14(4), 765 CrossRef CAS.
- V. K. Jha, A. K. Sijo and S. N. Alam,
et al., Effect of Nd doping on structural, electrical, thermal and magnetic properties of multifunctional BiFeO3 Ceramics, J. Supercond. Novel Magn., 2020, 33(2), 455–461 CrossRef CAS.
- S. Chauhan, R. Shweta and S. Pandit,
et al., Neodymium-doped zinc oxide nanoparticles catalytic cathode for enhanced efficiency of microbial desalination cells, Catalysts, 2023, 13(8), 1164 CrossRef CAS.
- S. Rani, O. Singh and S. Kaushik,
et al., Structural, dielectric, and magnetic properties of Dy-substituted BiFeO3 multiferroic ceramics, J. Mater. Sci.: Mater. Electron., 2023, 34(4), 258 CrossRef CAS.
- O. Singh, A. Agarwal and S. Sanghi,
et al., Variation of crystal structure, magnetization, and dielectric properties of Nd and Ba co-doped BiFeO3 multiferroics, Int. J. Appl. Ceram. Technol., 2019, 16(1), 119–129 CrossRef CAS.
- S. Kumari, N. Ortega and A. Kumar,
et al., Dielectric anomalies due to grain boundary conduction in chemically substituted BiFeO3, J. Appl. Phys., 2015, 117(11), 1–31 CrossRef.
- R. Tomar, N. Wadehra and V. M. Gaikwad,
et al., Low field manifestation of spiral ordering in sheet like BiFeO3 nanostructures, AIP Adv., 2018, 8(8), 1–7 Search PubMed.
- M. A. Matin, M. M. Rhaman and M. A. Hakim,
et al., Bi (1−y)SmyFeO3 as prospective photovoltaic materials, Bull. Mater. Sci., 2020, 43(1), 167 CrossRef CAS.
- M. M. Rhaman, M. A. Matin and M. A. Hakim,
et al., Bandgap tuning of samarium and cobalt co-doped bismuth ferrite nanoparticles, Mater. Sci. Eng., B, 2021, 263, 114842 CrossRef CAS.
- D. Carranza-Celis, A. Cardona-Rodríguez and J. Narváez,
et al., Control of Multiferroic properties in BiFeO3 nanoparticles, Sci. Rep., 2019, 9(1), 3182 CrossRef PubMed.
- I. B. Shameem Banu and S. Divya Lakshmi, Simultaneous enhancement of room temperature multiferroic properties of BiFeO3 by Nd doping at Bi site and Co doping at Fe site, J. Mater. Sci.: Mater. Electron., 2017, 28(21), 16044–16052 CrossRef CAS.
- Y. Zhang, Y. Wang and J. Qi,
et al., Structural, magnetic and impedance spectroscopy properties of Ho3+ modified BiFeO3 multiferroic thin film, J. Mater. Sci.: Mater. Electron., 2019, 30(3), 2942–2952 CrossRef CAS.
- H. Chouhan, S. Prasad and S. Parthasarathy,
et al., Unveiling the potential of a novel inorganic perovskite for NTC thermistor and energy-storage applications, Inorg. Chem. Commun., 2024, 170, 113127 CrossRef CAS.
- B. Dhanalakshmi, K. Pratap and B. Parvatheeswara Rao,
et al., Effects of Mn doping on structural, dielectric and multiferroic properties of BiFeO3 nanoceramics, J. Alloys Compd., 2016, 676, 193–201 CrossRef CAS.
- P. Naik, S. K. Patri and J. Nanda, Fabrication and characterization of Nd-doped BiFeO3-KNbO3 solid solution, J. Alloys Compd., 2025, 1010, 177601 CrossRef CAS.
- M. H. Ghozza, A. T. Mosleh and A. K. Elbadawy,
et al., Nanostructured bismuth ferrite nanoparticles: synthesis, characterization, electrical/magnetic properties and photocatalytic performance, Phys. Chem. Chem. Phys., 2025, 27(3), 1447–1458 RSC.
- I. Ragbaoui, S. Aydi and S. Chkoundali,
et al., Effect of substitution on the structural, electrical properties, and dielectric relaxor behavior in lead-free BiFeO3-based ceramics, RSC Adv., 2024, 14(2), 1330–1340 RSC.
- S. A. Bhat, M. A. Mushtaq and A. Rashid,
et al., Investigating the potential of Nd and Co modified bismuth ferrite (BiFeO3) nanoparticles for advanced energy storage and electronic applications, Ceram. Int., 2024, 50(21), 44347–44361 CrossRef CAS.
- M. Yadav, A. Agarwal and S. Sanghi,
et al., Crystal structure refinement, dielectric and magnetic properties of A-site and B-site co-substituted Bi0.90Nd0.10Fe1−xTixO3 (x = 0.00, 0.02, 0.05 & 0.07) ceramics, J. Alloys Compd., 2018, 750, 848–856 CrossRef CAS.
- F. Akram, J. Kim and S. A. Khan,
et al., Less temperature-dependent high dielectric and energy-storage properties of eco-friendly BiFeO3–BaTiO3-based ceramics, J. Alloys Compd., 2020, 818, 152878 CrossRef CAS.
- B. Khan, A. Kumar and P. Yadav,
et al., Structural, dielectric, magnetic and magneto-dielectric properties of (1−x)BiFeO3–(x)CaTiO3 composites, J. Mater. Sci.: Mater. Electron., 2021, 32(13), 18012–18027 CrossRef CAS.
- S. Joshi, A. Shukla and N. Kumar,
et al., Nd substitution response on structural, dielectric, and electrical features of bismuth iron titanate, Ceram. Int., 2024, 50(1), 1643–1654 CrossRef CAS.
- G. Singh, H. P. Bhasker and R. P. Yadav,
et al., Magneto-dielectric and multiferroic properties in Bi0.95Yb0.05Fe0.95Co0.05O3, Phys. Scr., 2019, 94(6), 065802 CrossRef CAS.
- S. Godara, N. Sinha and R. Geeta,
et al., Combined structural, electrical, magnetic and optical characterization of
bismuth ferrite nanoparticles synthesized by auto-combustion route, J. Asian Ceram. Soc., 2014, 2(4), 416–421 CrossRef.
- P. Uniyal and K. L. Yadav, Study of dielectric, magnetic and ferroelectric properties in Bi1−xGdxFeO3, Mater. Lett., 2008, 62(17–18), 2858–2861 CrossRef CAS.
- P. Acharya, R. N. P. Choudhary and S. K. Parida, Effect of lanthanum on structural, dielectric, electrical, and optical properties of the bismuth ferrite, Braz. J. Phys., 2024, 54(1), 17 CrossRef CAS.
- M. A. Nadir, M. Khalid and M. Younas,
et al., Impact of Neodymium substitution on electrical properties in mixed phase of Bismuth containing pyrochlore (Bi2Sn2O7 and Bi2O3–SnO2), J. Mater. Sci.: Mater. Electron., 2024, 35(26), 1708 CrossRef CAS.
- S. Iqbal, H. M. Rafique and G. Hussain,
et al., Investigation of multiferroic properties of dysprosium substituted bismuth ferrite (Bi1−xDyxFeO3) prepared by auto combustion technique, Digest J. Nanomater. Biostruct., 2022, 17(4), 1283–1295 CrossRef.
- T. Ahamed, A. Ahad and D. Mithun Kumar,
et al., Synthesis and characterization of dielectric, electric, and magnetic properties of vanadium doped-bismuth europium ferrites for multiferroic applications, Results Phys., 2023, 50, 106571 CrossRef.
- L. Thansanga, A. Shukla and N. Kumar,
et al., Study of effect of Dy substitution on structural, dielectric, impedance and magnetic properties of bismuth ferrite, J. Mater. Sci.: Mater. Electron., 2020, 31(13), 10006–10017 CrossRef CAS.
- A. Kumar, A. Panigrahi and M. Shekhar,
et al., Effect of Eu and Mn co-doping on temperature dependent dielectric relaxation behaviour and electric conduction mechanisms of bismuth ferrite, J. Electroceram., 2024, 52(2), 144–169 CrossRef CAS.
- D. D. Mathankar, N. S. Meshram and K. G. Rewatkar,
et al., Structural, magnetic, and dielectric studies of bismuth-doped nickel nanoferrites synthesized via sol–gel autocombustion method, J. Mater. Sci.: Mater. Electron., 2023, 34(27), 1845 CrossRef CAS.
- G. R. Gajula, L. R. Buddiga and K. N. Chidambara Kumar,
et al., An investigation on the conductivity, electric modulus and scaling behavior of electric modulus of barium titanate-lithium ferrite composite doped with Nb, Gd and Sm, Mater. Chem. Phys., 2020, 241, 122347 CrossRef CAS.
- M. Lohchab, S. Rohilla and S. K. Chaudhary,
et al., Investigating the structural, electrical, and magnetic characteristics of Nd-doped BiFeO3 synthesized via the solid-state reaction method, J. Mater. Res., 2024, 39(17), 2456–2468 CAS.
- M. Padhy, R. N. P. Choudhary and P. G. R. Achary, Electrical, magnetic and-optical-study of Gd3+ and Sm3+ ion doped at Fe-site in bismuth ferrite, Solid State Commun., 2022, 355, 114944 CrossRef CAS.
- F. Rehman, J. B. Li and I. Ahmad,
et al., Dielectric relaxation and electrical properties of Bi2.5Nd0.5Nb1.5Fe0.5O9 ceramics, Mater. Chem. Phys., 2019, 226, 100–105 CrossRef CAS.
- L. Thansanga, A. Shukla and N. Kumar,
et al., Studies of structural, electrical and ferroelectric characteristics of gadolinium and yttrium modified bismuth ferrite, Mater. Chem. Phys., 2021, 263, 124359 CrossRef CAS.
- A. K. Sahu, S. K. Satpathy and S. K. Rout,
et al., Dielectric and frequency dependent transport properties of gadolinium doped bismuth ferrite, Trans. Electr. Electron. Mater., 2020, 21(2), 217–226 CrossRef.
- I. Dmitrenko, K. Abdulvakhidov and A. Soldatov,
et al., Structure, dielectric, optical and magnetic properties of xBiFeO3–(1–x)PbZr0.9Ti0.1O3 composites, J. Mater. Sci.: Mater. Electron., 2023, 34(30), 2044 CrossRef CAS.
- M.-H. Yan, Z.-F. Zhang and H.-H. Huang,
et al., Multi-scale structural regulation of negative temperature coefficient effect and electrical conduction mechanism of xSm2O3−(1−x)CaCu3Ti4O12 ceramics, Ceram. Int., 2024, 50(24), 54390–54400 CrossRef CAS.
- Q. S. Fu, B. Meng and X. H. Chen,
et al., Dielectric response, electrical conductivity and scaling behavior of LuFe0.5Cr0.5O3 perovskite, J. Mater. Sci.: Mater. Electron., 2024, 35(33), 2135 CrossRef CAS.
- M. Seif Eddine, M. Horchani and H. Dhahri,
et al., Structural and High Dielectric Properties of La0.6Ba0.1Ce0.3Fe0.95Ni0.05O3 Multiferroic Perovskite for Electronic Applications, J. Inorg. Organomet. Polym. Mater., 2025, 1–14 Search PubMed.
- P. Siriprapa, A. Watcharapasorn and S. Jiansirisomboon, Electrical properties of Nd-doped BiFeO3 ceramics, Ferroelectrics, 2013, 451(1), 103–108 CrossRef CAS.
- S. Mishra and S. K. Parida, Investigation of structure, dielectric, thermistor, and optical properties study of an eco-friendly double perovskite for photovoltaic and optoelectronic applications: CaLiFeWO6, Curr. Appl. Phys., 2024, 65, 75–90 CrossRef.
- I. Chiba, M. Zannen and F. Hassen,
et al., Thermal Sensing Characteristics of Nd0.06Bi0.94FeO3 NPs for Thermistor Applications, J. Alloys Compd., 2025, 182028 CrossRef CAS.
- S. S. Hota, D. Panda and R. N. P. Choudhary, Studies of structural, dielectric, and electrical properties of polycrystalline barium bismuth tungstate for thermistor application, Inorg. Chem. Commun., 2023, 153, 110785 CrossRef.
- P. Mallick, A. U. Naik and D. Mohanty,
et al., Synthesis and analysis of structural, dielectric, and thermistor behaviour of zinc ferrite, Chem. Phys. Impact, 2023, 6, 100217 CrossRef.
- S. Sahoo, S. K. S. Parashar and S. M. Ali, CaTiO3 nano ceramic for NTCR thermistor-based sensor application, J. Adv. Ceram., 2014, 3(2), 117–124 CrossRef CAS.
- Q. Chen, Y. Zhao and J. Li,
et al., Enhanced thermal constant B of diamond films for ultrahigh sensitivity negative temperature coefficient thermistors, Sci. China Mater., 2024, 67(10), 3321–3329 CrossRef CAS.
|
| This journal is © The Royal Society of Chemistry 2026 |
Click here to see how this site uses Cookies. View our privacy policy here.  *a,
Ibtissem
Fraj
a,
Fredj
Hassen
*a,
Moneim
Zannen
b,
Cecile
Autret-Lambert
cd,
Mouldi
Zouaoui
e,
Thomas
Fourgassie
*a,
Ibtissem
Fraj
a,
Fredj
Hassen
*a,
Moneim
Zannen
b,
Cecile
Autret-Lambert
cd,
Mouldi
Zouaoui
e,
Thomas
Fourgassie
 cd,
Brahim
Dkhil
cd,
Brahim
Dkhil
 d,
Pierre-Eymeric
Janolin
d and
Omar
Ibder
d
d,
Pierre-Eymeric
Janolin
d and
Omar
Ibder
d

![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) δ) of all samples demonstrate significantly elevated values in the low-frequency domain, a typical behavior for dielectrics, attributed to the contribution of various types of polarization, including dipolar, space-charge, electronic, and ionic polarizations. At elevated frequencies, the phenomena mainly involve atomic and electronic polarizations, which yield a decrease in the losses. Furthermore, the Maxwell–Wagner model, in alignment with Koops’ phenomenological theory, provides insight into the variations in dielectric parameters with frequency.28,29 This model suggests that in low-frequency regimes, grain boundaries have a significant impact, whereas grains become more pronounced at higher frequencies. In the low-frequency domain, the substantial presence of highly resistive grain boundaries facilitates significant space charge polarization by electrons. Conversely, at higher frequencies, the dielectric constant decreases as the electrons move between grains and grain boundaries. The curve depicting the tangent loss follows a similar trend to that of the dielectric constant, with high values at low frequencies due to the increased resistance of grain boundaries, which necessitates more energy for electron mobilization. As the frequency increases, the resistance decreases, resulting in a corresponding reduction in the tangent loss values.30 This low frequency dispersion effect is commonly seen in ferroelectric and dielectric materials.31,32 As the temperature increased, the dielectric constant of the two samples (Nd0.06Bi0.94FeO3 NPs and Nd0.08Bi0.92FeO3 NPs) exhibited an upward trend at low frequencies. However, a decline in the dielectric constant was observed with temperature at the high neodymium concentration (Nd0.1Bi0.9FeO3 NPs). The presence of neodymium disorder cations leads to the relaxor characteristics of this sample.33 Yadav et al. suggest another possible explanation for this: the electrons may be jumping between Fe3+ and Fe2+ ions.34 Similar types of behavior have been observed in other eco-friendly ABO3-type relaxor-ferroelectric ceramics.35–37
δ) of all samples demonstrate significantly elevated values in the low-frequency domain, a typical behavior for dielectrics, attributed to the contribution of various types of polarization, including dipolar, space-charge, electronic, and ionic polarizations. At elevated frequencies, the phenomena mainly involve atomic and electronic polarizations, which yield a decrease in the losses. Furthermore, the Maxwell–Wagner model, in alignment with Koops’ phenomenological theory, provides insight into the variations in dielectric parameters with frequency.28,29 This model suggests that in low-frequency regimes, grain boundaries have a significant impact, whereas grains become more pronounced at higher frequencies. In the low-frequency domain, the substantial presence of highly resistive grain boundaries facilitates significant space charge polarization by electrons. Conversely, at higher frequencies, the dielectric constant decreases as the electrons move between grains and grain boundaries. The curve depicting the tangent loss follows a similar trend to that of the dielectric constant, with high values at low frequencies due to the increased resistance of grain boundaries, which necessitates more energy for electron mobilization. As the frequency increases, the resistance decreases, resulting in a corresponding reduction in the tangent loss values.30 This low frequency dispersion effect is commonly seen in ferroelectric and dielectric materials.31,32 As the temperature increased, the dielectric constant of the two samples (Nd0.06Bi0.94FeO3 NPs and Nd0.08Bi0.92FeO3 NPs) exhibited an upward trend at low frequencies. However, a decline in the dielectric constant was observed with temperature at the high neodymium concentration (Nd0.1Bi0.9FeO3 NPs). The presence of neodymium disorder cations leads to the relaxor characteristics of this sample.33 Yadav et al. suggest another possible explanation for this: the electrons may be jumping between Fe3+ and Fe2+ ions.34 Similar types of behavior have been observed in other eco-friendly ABO3-type relaxor-ferroelectric ceramics.35–37


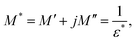
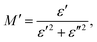
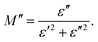
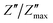 vs. f/fmax, where
vs. f/fmax, where 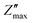 and fmax represent the peak value and it corresponding frequency of the peak in Z curves. As can be seen in Fig. 13, all peaks converge into a single master curve at different temperatures, suggesting that charges undergo dynamic processes with a consistent activation energy across various timescales. Additionally, plots of the
and fmax represent the peak value and it corresponding frequency of the peak in Z curves. As can be seen in Fig. 13, all peaks converge into a single master curve at different temperatures, suggesting that charges undergo dynamic processes with a consistent activation energy across various timescales. Additionally, plots of the 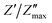 relaxation peak versus frequency show a gradual shift towards higher frequencies as the temperature increases. This further highlights the existence of multiple equilibrium states and the distribution of relaxation times in the prepared samples.55 Within the studied temperature range (537–575 K), neodymium does not alter the fundamental nature of the relaxation process, but it does affect the relaxation time value. This reflects the high homogeneity of the material in terms of the dynamics of the charge carriers or dipoles responsible for dielectric relaxation. The corresponding relaxation time τ is obtained through the following equation:
relaxation peak versus frequency show a gradual shift towards higher frequencies as the temperature increases. This further highlights the existence of multiple equilibrium states and the distribution of relaxation times in the prepared samples.55 Within the studied temperature range (537–575 K), neodymium does not alter the fundamental nature of the relaxation process, but it does affect the relaxation time value. This reflects the high homogeneity of the material in terms of the dynamics of the charge carriers or dipoles responsible for dielectric relaxation. The corresponding relaxation time τ is obtained through the following equation: