Impact of pseudohalide-based cesium formate salt as an additive or substituent on perovskite solar cell performance
Raja
Rajendran
ab,
Pei-Yu
Wang
a,
Ming-Hsien
Li
 *c,
Chen-Fu
Lin
a,
Farhan
Yousuf
a,
Cheng-Hung
Hou
d,
Jing-Jong
Shyue
*c,
Chen-Fu
Lin
a,
Farhan
Yousuf
a,
Cheng-Hung
Hou
d,
Jing-Jong
Shyue
 d and
Peter
Chen
d and
Peter
Chen
 *aefg
*aefg
aDepartment of Photonics, National Cheng Kung University, Tainan, 70101, Taiwan. E-mail: petercyc@ncku.edu.tw
bDepartment of Physics and Nanotechnology, SRM Institute of Science & Technology, Kattankulathur, Chennai-603203, Tamilnadu, India
cDepartment of Electro-Optical Engineering, National Formosa University, Yunlin 63201, Taiwan. E-mail: mhli1125@nfu.edu.tw
dResearch Center for Applied Sciences, Academia Sinica, Taipei 11529, Taiwan
eHierarchical Green-Energy Materials (Hi-GEM) Research Center, National Cheng Kung University, Tainan 70101, Taiwan
fProgram on Key Materials, Academy of Innovative Semiconductor and Sustainable Manufacturing, National Cheng Kung University, Tainan 70101, Taiwan
gResearch Center for Critical Issues (RCCI), Academia Sinica, Tainan 711010, Taiwan
First published on 24th October 2025
Abstract
The dual roles of pseudohalide formate anions (HCOO−), acting as both additives and substituents in perovskite solar cells (PSCs), are investigated and compared. An excess amount of CsHCOO added to the FA0.9Cs0.1PbI3 perovskite, serving as an additive, enhances the photovoltaic performance of PSCs. We observed a significant enlargement of perovskite grain size, improved perovskite crystallinity, preferred facet alignment perpendicular to the substrate, and reduced PbI2 formation after incorporating the CsHCOO additive into the perovskite film. This was achieved by slowing the perovskite growth rate through formate–iodide anion exchange. Moreover, the strong coordination between the formate anion and Pb2+ inhibits the formation of metallic Pb0 on the perovskite film surface, passivating surface defects. Benefiting from these improvements, reduced trap density and surface defect passivation lead to suppressed carrier recombination and longer carrier lifetimes. Incorporating 3 mol% CsHCOO additive in n–i–p structured PSCs improves the power conversion efficiency (PCE) from 17.12% to 18.57%, compared to PSCs without CsHCOO. Additionally, CsHCOO-added PSCs exhibited superior ambient stability, retaining 90% of their original PCE after 1000 hours in a 25 ± 5% relative humidity environment without encapsulation. For the p–i–n structured PSCs, 3 mol% CsHCOO additive enhanced the device PCE to 20.04%, outperforming the device without the CsHCOO additive (PCE = 18.01%). In contrast, substituting CsI with CsHCOO in the perovskite active layer, with a nominal formula of FA0.9Cs0.1PbI3−x(HCOO)x (x = 0.03 and 0.1), induced significant PbI2 formation and non-uniform perovskite films, resulting in reduced PSC performance. Our findings highlight the pivotal role of CsHCOO as an additive in enhancing PSC performance by addressing various challenges related to perovskite film quality and device stability.
1. Introduction
Halide-based perovskite materials have become promising optoelectronic materials due to their high absorption coefficiency,1 high carrier mobility, long carrier diffusion length,2 tunable bandgap and dimensionality,3–6 low exciton binding energy,7 and high defect tolerance.8,9 The power conversion efficiency (PCE) of perovskite solar cells (PSCs) has rapidly climbed from 3.8% to 27.0% since they were first reported in 2009,10 demonstrating their great potential for the photovoltaic (PV) market. Halide-based perovskite materials are based on the ABX3 structure, where A is a monovalent cation (e.g., CH3NH3+ (methylammonium, MA+), NH2CHNH2+ (formamidine, FA+), Cs+, Rb+, etc.), B is a divalent metal cation (Pb2+ and Sn2+), and X is a monovalent halide anion (Cl−, Br−, and I−).11 The perovskite film quality in terms of crystallinity, grain size, and film morphology is crucial to the device performance of PSCs.12 High-quality perovskite films can be produced using various methods, such as additive engineering,13 compositional engineering,14 and solvent engineering.15 Among these strategies, additive engineering is widely used for effectively controlling the crystal growth, film formation, defect passivation, and interfacial passivation.16,17 Large numbers of additives including halide or non-halide anions are being intensively investigated. Various non-halide anions, such as monovalent anions (e.g., HCOO−, CH3COO−, BF4−, SCN−, and NO3−), divalent anions (e.g., CO32− and SO42−), or trivalent anions (e.g., PO43−), serve as additives in PSCs to enhance the perovskite film quality and PCE of PSCs.18–23 However, the role of pseudohalides is not fully understood.Among the non-halide anions, pseudohalides, such as SCN− (ionic radius (r) of 2.17 Å), BF4− (r = 2.18 Å), and HCOO− (r < 2.0 Å), have recently attracted significant interest as additives due to their comparable ionic radius to iodide anions (r = 2.20 Å).18 Incorporating pseudohalides into the perovskite precursor has shown promise for fabricating high-quality perovskite films, particularly those regulating the perovskite film morphology and crystallinity.24–26 For instance, the SCN− anion can significantly improve film morphology27 and perovskite crystallinity,28 which increases the moisture tolerance of perovskite films. Furthermore, the addition of SCN− stabilizes the α-phase FAPbI3 perovskite by triggering the transformation of inorganic octahedral cages in the perovskite crystal from face-sharing (δ-phase) to point-sharing (α-phase) octahedra.29 In our previous studies, lead thiocyanate Pb(SCN)2 was employed as an additive in the FA0.9Cs0.1PbI3 perovskite to enhance perovskite grain size, extend carrier lifetime, and produce high perovskite crystallinity.30 Additionally, Pb(SCN)2 was employed to provide the lead source and reacts with MA(g)+ to form the perovskite film via the low pressure vapor-assisted process (LP-VASP).31 Furthermore, we introduced formamidinium thiocyanate (FASCN) as an additive to FA0.9Cs0.1PbI3-based perovskite solar cells to improve the perovskite film crystallinity and PCE of PSCs. Through systematic analysis, the presence of SCN− anion was demonstrated in the perovskite film, especially near the bottom interface, contributing to enhanced crystallinity and suppressed defect density of the matrix perovskite.32
Recently, the pseudohalide formate anion (HCOO−) has been extensively employed in PSCs to enhance surface coverage, improve film quality, and passivate defects at the perovskite active layer/electron transport layer (ETL) interface in n–i–p heterojunction PSCs.33 Various formate-based molecules, such as formic acid (HCOOH), methylammonium formate (MAHCOO), formamidine formate (FAHCOO), rubidium formate (RbHCOO), potassium formate (KHCOO), sodium formate (NaHCOO), cesium formate (CsHCOO), and ammonium formate (NH4HCOO) have been applied as additives in the perovskite films to improve crystallinity, grain size, and compactness. Reported results showed that the HCOO− pseudohalide anion could form a hydrogen bond with the FA+ cation, coordinate with Pb2+, passivate the iodide vacancy defects at grain boundaries and on the surface of perovskite films, and reduce bulk defects in the perovskite films. As a result, the non-radiative recombination loss could be suppressed, and device performance enhanced. Formate-based additives further enhance the ambient, thermal, and photo stability of PSCs.26,34–43
Gu et al. incorporated nonhalide lead formate (Pb(HCOO)2) as the lead source material, which reacted with methylammonium iodide (MAI) to prepare MAPbI3 perovskite. The results reveal that the PbI2 domains filling the perovskite grain boundaries effectively passivate defects at these grain boundaries, thereby suppressing carrier recombination and enhancing device PV performance.44 Baek et al. employed the sequential deposition method to fabricate the perovskite film by reacting a CsHCOO-incorporated PbI2 film with FAI. The addition of CsHCOO reduced the void density of the pre-deposited PbI2 film, leading to a smoother perovskite film surface accompanied by preferred crystalline facet alignment along the (001) facet, larger perovskite grain size, and reduced PbI2 residue due to the suppressed overcrystallization of PbI2 after adding CsHCOO. Furthermore, the CsHCOO treatment can reduce trap density, nonradiative recombination in the perovskite film, and improve stability against humidity and oxygen corrosion. Consequently, CsHCOO-treated PSCs deliver a higher PCE and superior long-term stability compared to pristine PSCs. Moreover, the CsHCOO treatment can enhance the reliability of PSCs fabricated by roll-to-roll processes, along with enhanced device performance and reduced hysteresis.45 Guo et al. fabricated a perovskite film by sequentially spin-coating two precursor solutions. In the first step, they replaced CsBr with CsHCOO in the precursor solution, creating a wet film on the substrate via spin-coating. A second-step precursor was then spin-coated onto this initial wet film, allowing the perovskite to form via interdiffusion between the stacked layers. The resulting complexes (HCOO·Pb+ and HCOOH·Cs+) and the volatilization of HCOOH retarded the perovskite growth rate. This kinetic control improved the film's crystallinity, grain size, phase stability against light soaking, and long-term stability. Furthermore, replacing CsBr by CsHCOO reduced the trap-state defect density, which ultimately enhanced device photovoltaic performance under AM 1.5G and dim-light illumination.41
FAHCOO was deposited on the surface of the SnO2 ETL as an interfacial modifier to passivate defects at the SnO2/perovskite buried interface. The strong coordination between the HCOO− anion and Sn4+ cation on the SnO2 surface passivates surface defects, thereby enhancing the conductivity of the SnO2 ETL. After FAHCOO modification, the upshifted energy level of the SnO2 ETL provides effective electron extraction from the perovskite layer to the SnO2 ETL. The FAHCOO-modified SnO2ETL further assists the crystallization process from PbI2 to the perovskite film via sequential deposition, yielding a highly crystalline and compact perovskite film, especially at the bottom side of the perovskite film, due to reduced unreacted PbI2. The suppressed carrier recombination and facilitated electron extraction at the SnO2/perovskite buried interface contribute to the high efficiency of PSCs.46,47 On the other hand, Deng et al. added CsHCOO to the SnO2 colloid precursor for preparing a CsHCOO pre-buried SnO2 ETL in the PSCs. The incorporation of CsHCOO into the SnO2 ETL enhances the conductivity of the SnO2 ETL to promote electron extraction and transport at the SnO2/perovskite interface. Moreover, after deposition of the perovskite film on the CsHCOO-added SnO2 ETL, the potential diffusion of Cs+ and HCOO− into the bottom side of the perovskite film can passivate defects, eliminate voids, release in-plane tensile strain on the bottom side, and enhance the crystallinity of the perovskite film. The PSCs with the CsHCOO-added SnO2 ETL exhibit superior PCE and operational stability compared to those without CsHCOO treatment.48 Beyond its application in PSCs, the CsHCOO additive has also been introduced in tin-based perovskite for the application of thin-film transistors (TFTs) and near-infrared light-emitting diodes (LEDs).49,50
Previous studies have focused primarily on introducing CsHCOO either into sequentially-processed perovskite films or as an agent to passivate interfacial defects. However, the application of CsHCOO in one-step-processed perovskite films remains unexplored. Given that one-step processing is a more facile and time-saving approach compared to two-step processing, we investigate two distinct roles of CsHCOO in the one-pot perovskite precursor solution: first, using CsCHOO as an additive; and second, completely replacing cesium iodide (CsI) with CsHCOO. Through these two approaches, we comprehensively investigate the effect of CsHCOO on the photovoltaic performance of PSCs. For CsHCOO as an additive, CsHCOO effectively improved PSC performance, primarily due to the HCOO−-assisted enhancement of perovskite film quality. From the analysis results, the CsHCOO additive can enhance the perovskite grain size, induce preferred crystalline orientation, promote the alignment of crystalline facets perpendicular to the substrate, suppress PbI2 formation, and passivate the surface defects. These improvements further reduce trap density, extend the photogenerated carrier lifetime in the perovskite film, and ultimately enhance the PV performance of PSCs. Specifically, n–i–p structured PSCs with 3% CsHCOO additive achieve a PCE of 18.57%, which outperforms PSCs without CsHCOO (PCE = 17.12%). Moreover, the 3%-CsHCOO-added PSCs retain 90% of their original PCE after 1000 hours in a 25 ± 5% relative humidity environment without encapsulation, demonstrating superior stability compared to PSCs without CsHCOO, which retain 79% of their initial PCE under the same test conditions. We also demonstrate p–i–n structured PSCs with 3% CsHCOO additive, which deliver a PCE of 20.04%. On the other hand, substitution of CsI with CsHCOO in the perovskite layer can induce the formation of significant amounts of PbI2 and a non-uniform perovskite film, thereby reducing the PCE of PSCs. Our findings provide an overall insight into the role of CsHCOO in PSCs and indicate the pivotal role of the CsHCOO as an additive in enhancing device performance.
2. Experimental section
2.1. Device fabrication
For fabrication of n–i–p structured devices, fluorine-doped tin oxide (FTO) coated glass substrates, sized at 2.5 × 2.5 cm2, were patterned using zinc powder and hydrochloric acid etching. The substrates were subsequently cleaned sequentially with detergent solution, acetone, isopropyl alcohol, and deionized water using ultrasonic treatment. A compact TiO2 (cp-TiO2) layer was then spray-coated onto the substrates using titanium diisopropoxide bis(acetylacetonate) in ethanol (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 39 weight ratio) at 480 °C for 30 minutes. The cp-TiO2-coated FTO substrates were treated with UV ozone for 30 minutes. The FTO/cp-TiO2 substrates were spin-coated with a tin oxide colloidal dispersion (SnO2, 15% in H2O) diluted with deionized water (1
39 weight ratio) at 480 °C for 30 minutes. The cp-TiO2-coated FTO substrates were treated with UV ozone for 30 minutes. The FTO/cp-TiO2 substrates were spin-coated with a tin oxide colloidal dispersion (SnO2, 15% in H2O) diluted with deionized water (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 weight ratio) and annealed at 150 °C for 30 minutes.
1 weight ratio) and annealed at 150 °C for 30 minutes.
In the first study using CsHCOO as an additive, the FA0.9Cs0.1PbI3 perovskite precursor solution was prepared by dissolving formamidinium iodide (FAI) (0.9 M), cesium iodide (CsI) (0.1 M), and lead iodide (PbI2) (1 M) in a solvent mixture of dimethyl sulfoxide (DMSO) and dimethylformamide (DMF) (3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 7 volume ratio of DMSO
7 volume ratio of DMSO![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) DMF). This solution was incorporated with 0, 3, 7, and 10 mol% of CsHCOO additive (with respect to 1 M of FA0.9Cs0.1PbI3). In the second study, where CsHCOO replaced a portion of CsI, the FA0.9Cs0.1PbI2.97(HCOO)0.03 (R3%) and FA0.9Cs0.1PbI2.9(HCOO)0.1 (R10%) perovskite precursor solutions were prepared similarly. Specifically, the FA0.9Cs0.1PbI2.97(HCOO)0.03 perovskite precursor was prepared by dissolving FAI (0.9 M), PbI2 (1 M), CsI (0.097 M), and CsHCOO (0.03 M) in the mixed solvent (3
DMF). This solution was incorporated with 0, 3, 7, and 10 mol% of CsHCOO additive (with respect to 1 M of FA0.9Cs0.1PbI3). In the second study, where CsHCOO replaced a portion of CsI, the FA0.9Cs0.1PbI2.97(HCOO)0.03 (R3%) and FA0.9Cs0.1PbI2.9(HCOO)0.1 (R10%) perovskite precursor solutions were prepared similarly. Specifically, the FA0.9Cs0.1PbI2.97(HCOO)0.03 perovskite precursor was prepared by dissolving FAI (0.9 M), PbI2 (1 M), CsI (0.097 M), and CsHCOO (0.03 M) in the mixed solvent (3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 7 volume ratio of DMSO
7 volume ratio of DMSO![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) DMF). The FA0.9Cs0.1PbI2.9(HCOO)0.1 perovskite precursor was prepared by dissolving FAI (0.9 M), PbI2 (1 M), CsI (0.09 M), and CsHCOO (0.1 M) in the same mixed solvent system (DMSO
DMF). The FA0.9Cs0.1PbI2.9(HCOO)0.1 perovskite precursor was prepared by dissolving FAI (0.9 M), PbI2 (1 M), CsI (0.09 M), and CsHCOO (0.1 M) in the same mixed solvent system (DMSO![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) DMF = 3
DMF = 3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 7 volume ratio).
7 volume ratio).
The perovskite light-absorber layer was spin-coated onto the FTO/cp-TiO2/SnO2 substrate using a one-step solution process with antisolvent engineering. The precursor solution was first deposited onto the substrate and spun at 1000 rpm for 10 seconds, followed by 5000 rpm for 30 seconds. During the final 10 seconds of the second step, 150 μL of chlorobenzene antisolvent was dropped onto the wet perovskite film. The as-coated films were immediately annealed at 150 °C for 30 min and subsequently cooled to room temperature. The hole-transporting layer (HTL) was deposited onto perovskite film by spin coating at 4000 rpm for 30 seconds. A 50 μL spiro-OMeTAD solution (0.06 M in chlorobenzene) containing two dopants (2.88 μL of TBP and 3.5 μL of 1.8 M lithium bis(trifluoromethylsulfonyl)imide (Li-TFSI) in acetonitrile) was spin-coated onto the perovskite film. The silver electrode was deposited using thermal evaporation under a vacuum of 5 × 10−6 Torr to a thickness of 60 nm. The final device structure was glass/FTO/cp-TiO2/SnO2/perovskite/spiro-OMeTAD/Ag, as schematically illustrated in Fig. S1(a). The corresponding top-view illustration of the device, featuring four solar cells on a single substrate, is shown in Fig. S1(b). The active area of each individual solar cell is approximately 4 mm × 7 mm, as indicated by the black dashed rectangle in Fig. S1(b).
For the fabrication of p–i–n structured devices, indium tin oxide (ITO) glass substrates (2.5 × 2.5 cm2) were patterned and cleaned using the same procedure as that employed for the n–i–p devices. The complete p–i–n structure, maintained at the same geometric size, consisted of glass/ITO/3PATAT-C3/perovskite/C60/BCP/Ag. A self-assembled monolayer (SAM) of 3PATAT-C3 (0.1 mM L−1 dissolved in DMF) was first deposited onto the ITO substrate by spin-coating at 3000![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) rpm for 30 s and then annealed at 100 °C for 10 min. The FA0.9Cs0.1PbI3 perovskite precursor solution was prepared by dissolving FAI (0.9 M), CsI (0.1 M), and PbI2 (1 M) in a DMSO and DMF solvent mixture (2
rpm for 30 s and then annealed at 100 °C for 10 min. The FA0.9Cs0.1PbI3 perovskite precursor solution was prepared by dissolving FAI (0.9 M), CsI (0.1 M), and PbI2 (1 M) in a DMSO and DMF solvent mixture (2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 8 volume ratio). This solution was incorporated with 5 mol% of MACl and 3 mol% CsHCOO, based on the 1 M concentration of FA0.9Cs0.1PbI3. The perovskite film was spin-coated using the same recipe as that for the n–i–p structured device. The as-coated films were immediately annealed at 150 °C for 30 min and subsequently cooled to room temperature. Finally, C60 (25 nm), BCP (6 nm), and a silver (Ag) top electrode (100 nm) were sequentially deposited onto the perovskite layer via thermal evaporation.
8 volume ratio). This solution was incorporated with 5 mol% of MACl and 3 mol% CsHCOO, based on the 1 M concentration of FA0.9Cs0.1PbI3. The perovskite film was spin-coated using the same recipe as that for the n–i–p structured device. The as-coated films were immediately annealed at 150 °C for 30 min and subsequently cooled to room temperature. Finally, C60 (25 nm), BCP (6 nm), and a silver (Ag) top electrode (100 nm) were sequentially deposited onto the perovskite layer via thermal evaporation.
2.2. Characterization
The surface and cross-sectional morphology of perovskite films were examined using scanning electron microscopy (Supra 55, Zeiss). Absorption spectra of the perovskite films were obtained using a UV-vis spectrometer (U-4100, Hitachi). A solar simulator system (SS-F5-3A, Enlitech) was used to measure the characteristic current density–voltage (J–V) curves of the devices under AM 1.5G illumination (100 mW cm−2), with a source meter (Keithley 2401) used to trace the J–V data using a scan rate of 2 V s−1. The one-sun light intensity (100 mW cm−2) was calibrated using a certified standard silicon solar cell (SRC-2020-KG3, Enlitech). During measurement, a metal mask with an area of 0.15 cm2 (3 mm × 5 mm), marked by a dashed blue rectangle in Fig. S1(b), was placed on the backside of the glass substrate to define the illuminated area. For incident photon-to-electron conversion efficiency (IPCE) measurements, a 300 W xenon light, a monochromator (Newport Cornerstone 260), and a source meter (Keithley 2401) were integrated. A PHI TRIFT V nanoTOF (ULVAC-PHI, Japan) instrument was used to collect all time-of-flight secondary-ion mass spectrometry (ToF-SIMS) depth profiles using the dual-beam slice-and-view analysis method. The main ion source during the analysis phase was a pulsed C60+ ion beam with a pulse frequency of roughly 8200 Hz and a pulse length of 15 ns. The C60+ had an acceleration voltage of 20 kV and a beam current of 0.15 nA-DC. Time-resolved photoluminescence (TRPL) measurements were conducted using a home-built micro-PL system equipped with a 375 nm pulsed diode laser (PicoQuant model: LDH-P-C-375B, minimum pulse width 49 ps, maximum repetition rate = 40 MHz) focused by a 10× objective. The time-correlated single photon counting system (TCSPC, PicoQuant model: PicoHarp 300) was used to integrate the PL signals captured using a photomultiplier tube (PMT, SouthPort CO).3. Results and discussion
In this work, CsHCOO was introduced into the FA0.9Cs0.1PbI3 perovskite precursor as an additive and a replacement for CsI. In the initial study, different molar ratios of 0%, 3%, 7%, and 10% CsHCOO relative to the 1 M FA0.9Cs0.1PbI3 perovskite precursor were added to the precursor. Fig. 1(a)–(d) show top-view scanning electron microscopy (SEM) images of FA0.9Cs0.1PbI3 perovskite films with varying additive levels of 0%, 3%, 7%, and 10% CsHCOO, deposited on FTO/cp-TiO2/SnO2 substrates, respectively. All perovskite films exhibit compact surfaces with densely packed grains on the substrate, with the corresponding average grain size shown in Fig. 1(e) based on a standard test method of ASTM E112. Briefly, following ASTM E112, we superimposed 20 reference lines randomly across the microstructure in the top-view SEM images. The length of each reference line was determined based on the scale bar provided in the SEM image. Using the linear intercept method, we counted the number of grain boundary intersections along these lines. By calculating the mean lineal intercept, which represents the average length of a line segment intercepted by a grain, we were able to estimate the average grain size.In Fig. 1(a) and (e), the pristine perovskite film shows an average grain size of 354.5 nm. The introduction of a 3% CsHCOO additive in the perovskite precursor, as seen in Fig. 1(b) and (e), effectively enlarges the average perovskite grain size to 593.1 nm. The addition of 3% CsHCOO to the FA0.9Cs0.1PbI3 perovskite can slow down the crystal growth rate, initially leading to the formation of smaller perovskite crystals. These smaller crystals can then dissolve and recrystallize, eventually forming larger grains through an Ostwald ripening process.51,52 As the additive level of CsHCOO increases to 7% and 10%, the perovskite films exhibit primary perovskite grains of comparable size to the 3% CsHCOO-added perovskite film. However, notably, small-sized grains form on and near the grain boundaries of the large-sized grains in the 7% and 10% CsHCOO-added perovskite films, as marked by dashed yellow circles. Consequently, due to these small-sized grains, the perovskite films with 7% and 10% CsHCOO additive presents average perovskite grain sizes of 472.5 nm and 428.9 nm, respectively. These average grain sizes are smaller than that of 3% CsHCOO-added perovskite film but still larger than that of the pristine perovskite film, as shown in Fig. 1(c), (d) and (e). The addition of more than 3% CsHCOO to the FA0.9Cs0.1PbI3 perovskite further decelerates the growth rate, resulting in even smaller perovskite crystals. While these smaller crystals could still facilitate recrystallization into larger grains, some may remain on the film surface before full coalescing with the larger grains during the post-annealing process.51
Fig. 2(a) shows the XRD profiles of the FA0.9Cs0.1PbI3 perovskite films with different additive levels of CsHCOO on the FTO/cp-TiO2/SnO2 substrate. The XRD peaks at 2θ = 13.9°, 19.7°, 24.3°, 28.0°, 31.6°, 40.1°, 42.7°, and 49.8° correspond to the (100), (110), (111), (200), (210), (220), (300), and (222) facets of the FA0.9Cs0.1PbI3 perovskite, respectively. This indicates that the FA0.9Cs0.1PbI3 perovskite has a cubic phase (α-phase) with a preferred orientation along the (100) facet.32 The XRD peak at 2θ = 12.6°, marked with ▼, indicates residual PbI2. Residual PbI2 is commonly detected in FA0.9Cs0.1PbI3 perovskite films due to incomplete transformation.53–57 This residual PbI2 can accelerate degradation of perovskite films upon light exposure, as it rapidly decomposes into metallic lead (Pb0) and iodine (I2) when illuminated.58–60 We found that adding 3%, 7%, and 10% CsHCOO to the perovskite precursor resulted in films exhibiting negligible XRD peaks corresponding to PbI2. This clearly reveals that incorporating CsHCOO into the FA0.9Cs0.1PbI3 composition promotes the full conversion of residual PbI2 into the desired perovskite phase. The HCOO− anion facilitates this by retarding the perovskite growth rate, which allows for a complete reaction between PbI2 and the CsI/FAI components. Similar to previous literature where formate additives (such as FAHCOO, RbHCOO, and KHCOO) have been shown to inhibit PbI2 formation in perovskite films, the CsHCOO additive is expected to enhance film stability against light and humidity due to the suppressed residual PbI2.37,38 Moreover, the intensity of the XRD peak corresponding to the (100) facet is significantly enhanced, indicating that the addition of CsHCOO in the perovskite precursor induces a preferred orientation of the (100) facet in the resultant perovskite films.
Grazing-incidence wide-angle X-ray scattering (GIWAXS) measurements were conducted to further elucidate the crystalline arrangement of FA0.9Cs0.1PbI3 perovskite films incorporating 0%, 3%, 7%, and 10% CsHCOO additives, as depicted in Fig. 2(b)–(e), respectively.61 All perovskite films exhibit a series of Debye–Scherrer rings corresponding to the characteristic (100), (110), (111), (200), (210), and (220) facets of FA0.9Cs0.1PbI3 perovskite films, as labeled in Fig. 2(a). The powder rings marked by rhombuses are attributed to the FTO/cp-TiO2/SnO2 substrate, while those corresponding to the characteristic facets of FA0.9Cs0.1PbI3 perovskite films imply a random arrangement of perovskite facets on the substrate. It is noted that the scattering intensity of the (100) facet, distributed on the rings, varies with the CsHCOO additive level in the perovskite films.
Fig. 2(e) illustrates the integrated azimuthal intensity profiles from GIWAXS of the (100) facet for the FA0.9Cs0.1PbI3 perovskite films with 0%, 3%, 7%, and 10% CsHCOO additives. The pristine FA0.9Cs0.1PbI3 perovskite film exhibits two prominent peaks at an azimuthal angle of ±35°, indicating that the (100) facet predominantly grows with a tilting angle of ±35° relative to the substrate.47 Upon adding 3% CsHCOO to the perovskite films, the integrated intensity of the two peaks at an azimuthal angle of ±35° is reduced. With further increases to 7% and 10% CsHCOO additives in the perovskite films, the integrated intensity concentrates in the meridian direction (azimuthal angle = 0°), revealing a shift in the preferred arrangement of the (100) facet toward a uniform alignment on the substrate. The evolution of the (100) facet arrangement in the perovskite films with CsHCOO additives suggests that the CsHCOO additive can regulate the crystalline arrangement to be perpendicular to the substrate, which is beneficial for carrier transport in the vertical direction within PSCs.
Based on the discussions above, we infer the effect of the CsHCOO additive on the final perovskite film formation, as illustrated in Fig. S2. In the perovskite precursor without the CsHCOO additive, perovskite ionic species, including the [PbI6]4− octahedra and the A-site Cs+ and FA+ cations, are formed. When the perovskite precursor is spin-coated onto the substrate and treated with an anti-solvent, the removal of the solvent leads to oversaturation, causing the precursor to crystallize as the [PbI6]4− octahedra self-assemble with Cs+ and FA+ cations to form the FA0.9Cs0.1PbI3 perovskite. When excess CsHCOO is added to the perovskite precursor, [PbI6−x(HCOO)x]4− (x ≤ 4) octahedral structures spontaneously form in the perovskite precursor due to their Gibbs free energy (ΔG) being less than zero.44 These octahedra gradually convert to [PbI6]4− octahedra through anion exchange of HCOO− by I− before forming the perovskite. This anion exchange process can slow down the perovskite growth rate and promote better crystallization,42 leading to a highly crystalline perovskite film with a preferred orientation along the (100) and (200) facets. The slower growth rate also facilitates the arrangement of these facets perpendicular to the substrate and enables recrystallization and grain-merging processes, thereby enlarging perovskite grain size.51
To examine the presence of the HCOO− anion in perovskite films after adding CsHCOO additive, Fourier transform infrared spectroscopy (FTIR) under attenuated total reflection (ATR) mode was implemented. Fig. S3 presents the full FTIR spectra of FA0.9Cs0.1PbI3 perovskite films with various CsHCOO additive levels, labeled with characteristic vibration modes ascribed to the FA+ cation. The FTIR spectrum of CsHCOO powder is included as a reference. Notably, all perovskite films with 0%, 3%, 7%, and 10% CsHCOO additives exhibit a similar FTIR profile in the full spectra. Fig. 3(a) shows magnified FTIR spectra, extracted from the light-yellow rectangle in Fig. S3. The FTIR spectrum of Pb(HCOO)2 powder is provided for comparison with that of CsHCOO powder. The perovskite films with 3% and 7% CsHCOO additives present FTIR profiles similar to the pristine perovskite film, showing no characteristic peaks assignable to CsHCOO because the remaining content in the annealed perovskite film is presumably below the FTIR detection limit. When the CsHCOO additive level increases to 10%, the perovskite film displays a characteristic peak at 1561 cm−1, which is close to the characteristic C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O stretch mode of the HCOO− anion, whose peak is at 1574 cm−1. The FTIR peak at 1561 cm−1 for the 10%-CsHCOO-added perovskite film is possibly attributed to the presence of the HCOO− anion in the perovskite films.37,45,46 The shift of the characteristic FTIR peak can be briefly explained using Hooke's law:
O stretch mode of the HCOO− anion, whose peak is at 1574 cm−1. The FTIR peak at 1561 cm−1 for the 10%-CsHCOO-added perovskite film is possibly attributed to the presence of the HCOO− anion in the perovskite films.37,45,46 The shift of the characteristic FTIR peak can be briefly explained using Hooke's law:
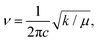 | (1) |
![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O stretch mode to a lower wavenumber in Pb(HCOO)2 compared to CsHCOO is primarily caused by the larger atomic mass of Pb relative to Cs. Consequently, the peak shift of the C
O stretch mode to a lower wavenumber in Pb(HCOO)2 compared to CsHCOO is primarily caused by the larger atomic mass of Pb relative to Cs. Consequently, the peak shift of the C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O mode in the 10%-CsHCOO-added perovskite film is presumably due to the interaction between the HCOO− anion and the Pb2+ cation.
O mode in the 10%-CsHCOO-added perovskite film is presumably due to the interaction between the HCOO− anion and the Pb2+ cation.
We further conducted X-ray photoelectron spectroscopy (XPS) to investigate the impact of CsHCOO additive on the chemical environment of the perovskite film. XPS is a surface-sensitive technique used to examine the chemical species of the top few nanometers of a material's surface, typically less than 10 nm. The survey scans of XPS spectra for perovskite films with 0% and 3% CsHCOO additives are shown in Fig. S4(a) and (b), respectively. Narrow scans of XPS spectra for the C 1s, O 1s, and Pb 4f orbitals in the perovskite films with 0%, 3%, and 10% CsHCOO additives are presented in Fig. 3(b)–(d), respectively. To identify the presence of CsHCOO in the perovskite film, the C 1s XPS spectra of perovskite films with 0%, 3%, and 10% CsHCOO additives is shown in Fig. 3(b) for comparison. In the C 1s narrow XPS spectra (Fig. 3(b)), both the pristine perovskite film and the 3%-CsHCOO-added perovskite film exhibit a similar profile with a characteristic peak at around 288 eV, which is attributed to the C–N bonding of the FA+ cation. After adding 10% CsHCOO to the perovskite film, two satellite peaks at ∼286.4 eV and ∼289.6 eV are detected, corresponding to C–O and C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O bonding in CsHCOO, respectively, as identified from the C 1s XPS spectrum of CsHCOO powder. Fig. 3(c) shows the O 1s narrow XPS spectra of perovskite films with 0%, 3%, and 10% CsHCOO additives. The perovskite films with 3% and 10% CsHCOO additives exhibit a prominent peak at a binding energy of around 532.7 eV, while the pristine perovskite film shows a negligible XPS peak at this binding energy. This suggests the presence of CsHCOO on the surface of the perovskite films.
O bonding in CsHCOO, respectively, as identified from the C 1s XPS spectrum of CsHCOO powder. Fig. 3(c) shows the O 1s narrow XPS spectra of perovskite films with 0%, 3%, and 10% CsHCOO additives. The perovskite films with 3% and 10% CsHCOO additives exhibit a prominent peak at a binding energy of around 532.7 eV, while the pristine perovskite film shows a negligible XPS peak at this binding energy. This suggests the presence of CsHCOO on the surface of the perovskite films.
The narrow XPS spectra in the Pb 4f level range, shown in Fig. 3(d), for the pristine perovskite films present two main peaks at 143.45 eV (Pb 4f5/2) and 138.55 eV (Pb 4f7/2), corresponding to Pb–I bonding, and two satellite peaks at 141.9 eV and 137 eV, which are indicative of metallic Pb (Pb0). After adding 10% CsHCOO to the perovskite film, the main peaks shift slightly to higher binding energies (143.58 eV for Pb 4f5/2 and 138.73 eV for Pb 4f7/2) due to the interaction of Pb2+ with the HCOO− anion. Specifically, the electron-rich oxygen atoms in the HCOO− anion (a Lewis base) form strong coordination bonds with the undercoordinated Pb2+ cations (Lewis acid sites).62 Notably, the XPS peaks corresponding to metallic Pb in the CsHCOO-added perovskite films vanish, suggesting that the HCOO− anion binds to the undercoordinated Pb2+ cation, thereby suppressing the formation of metallic Pb, which is responsible for surface defects.58
In our previous work, time-of-flight secondary ion mass spectrometry (ToF-SIMS) analysis of FA0.9Cs0.1PbI3 perovskite films containing the pseudohalide FASCN additive revealed the presence of thiocyanate near the film's bottom.32 Similarly, high-sensitivity ToF-SIMS depth profiling measurements were conducted on FA0.9Cs0.1PbI3 perovskite films with 0% and 3% CsHCOO additives to track the distribution of the HCOO− anion within the film. During depth profiling, a polyatomic C60+ ion beam was utilized to preserve the molecular structure, retaining the characteristic of organic species in the films. Fig. S4(c) and (d) display the ToF-SIMS depth profiles of perovskite films containing 0% and 3% CsHCOO additives, fabricated on FTO/cp-TiO2/SnO2 substrates to replicate the layer stacking structure of the actual device. The profiles revealed a uniform distribution of perovskite components (FA, Cs, Pb, and I) across the film for both additive concentrations (0% and 3% CsHCOO), indicating a consistent FA0.9Cs0.1PbI3 composition within the perovskite matrix. CsHCOO can potentially decompose into ionic species such as HCOO−, COO−, or CO molecules under ion beam sputtering. However, these species have masses comparable to the FA+ cation, hindering the detection of CsHCOO in the film using ToF-SIMS. Therefore, the oxygen signal was monitored to identify the presence of CsHCOO. The 3%-CsHCOO-added perovskite film exhibited no distinct O peaks throughout its depth, suggesting an absence of bulk HCOO− within the film, similar to the pristine perovskite film. These results imply that the 3% CsHCOO additive in the precursor introduces a negligible amount of HCOO− into the bulk of the perovskite film. Potentially, only a very low amount of HCOO− resides near the surface, where it might contribute to surface defect passivation. The negligible amount of HCOO− in the perovskite films is possibly due to the vaporization of HCOO− from the perovskite films during annealing.41,50
Fig. 4(a) presents the UV-Vis absorption spectra of FA0.9Cs0.1PbI3 perovskite films containing 0%, 3%, 7%, and 10% CsHCOO additives. All films exhibit a similar absorption profile with a cutoff wavelength around 810 nm, indicating that the primary composition of all films is FA0.9Cs0.1PbI3 perovskite. Fig. 4(b) displays the Tauc plots, derived from the UV-Vis absorption spectra, for the FA0.9Cs0.1PbI3 perovskite films with varying CsHCOO additive levels. The optical bandgap (Eg) determined from the cutoff energy is 1.54, 1.55, 1.55, and 1.56 eV for perovskite films with 0%, 3%, 7%, and 10% CsHCOO additives, respectively. This reveals that CsHCOO additives can increase the bandgap of FA0.9Cs0.1PbI3 perovskite films due to the reduced lattice spacing63 and bonding between O2− ions in HCOO− and Pb2+ ions.64 Fig. S5 presents a magnified view of the XRD profiles from Fig. 2(a) at 2θ ∼ 14°, focusing on the (100) facet peak for films with varying CsHCOO additive levels. A gradual shift of the (100) facet XRD peak to a higher angle is observed as the amount of CsHCOO additive increases, revealing a reduced lattice spacing. This reduction could be attributed to the smaller radius of the Cs+ cations (compared to FA+ cations) and/or the smaller radius of the pseudohalide HCOO− anions (compared to I− anions) upon CsHCOO incorporation into the perovskite lattice. However, the easy vaporization of HCOO− anions during post-annealing suggests that the reduced lattice spacing is primarily caused by the excess Cs+ cations incorporated into the perovskite lattice. Despite the easy vaporization of HCOO− anions during post-annealing, the added CsHCOO can still lead to a trace amount of HCOO− residuals within the perovskite film. Ultimately, the combined effects of the HCOO− residual (which enables the bonding between O2− in HCOO− and Pb2+) and the reduced lattice spacing are responsible for enhancing the perovskite's bandgap.
 | ||
| Fig. 4 (a) UV-Vis absorption spectra, (b) Tauc plots, (c) PL spectra, and (d) TRPL profiles of FA0.9Cs0.1PbI3 perovskite films with 0%, 3%, 7%, and 10% CsHCOO additives. | ||
To further characterize the band diagram of these perovskite films, their work function and valence band maximum (VBM) were measured using a Kelvin probe equipped with an ambient-pressure photoemission spectroscopy (APS) system. Table S1 in the SI provides the contact potential difference (CPD) between the perovskite films and the tip (work function of 4.576 eV), as well as the VBM of the FA0.9Cs0.1PbI3 perovskite films with different CsHCOO additive levels. Based on these results, the band diagrams of the FA0.9Cs0.1PbI3 perovskite films with 0%, 3%, 7%, and 10% CsHCOO additives are illustrated in Fig. S6. The band diagrams reveal that the Fermi level of FA0.9Cs0.1PbI3 perovskite films with 0%, 3%, 7%, and 10% CsHCOO additives is located close to the VBM, indicating p-type doping in these films. This result is similar to our previous work using FA0.9Cs0.1PbI3 perovskite film with FASCN additive.32 Furthermore, the Fermi level of the FA0.9Cs0.1PbI3 perovskite film added with CsHCOO is slightly lower than that of the pristine FA0.9Cs0.1PbI3 perovskite film. Consequently, the conductivity of CsHCOO-adding perovskite films is expected to be higher than that of the pristine perovskite film. It is also observed that the introduction of CsHCOO additives to the FA0.9Cs0.1PbI3 perovskite films results in an upward shift of both the conduction band minimum (CBM) and the VBM compared to the pristine perovskite. This upward shift of the VBM reduces the energy offset between the perovskite active layer and the spiro-OMeTAD HTL, potentially leading to more efficient hole extraction.
Steady-state photoluminescence (PL) measurements were conducted to elucidate the role of CsHCOO in FA0.9Cs0.1PbI3 perovskite films, as shown in Fig. 4(c). The FA0.9Cs0.1PbI3 perovskite film without CsHCOO additives exhibits a PL emission peak centered at approximately 802.2 nm, while the film with a 3% CsHCOO additive displays a blue shift in the PL emission peak to 798.5 nm. Upon further increasing the CsHCOO additive to 7% and 10%, the PL emission peaks of the perovskite films shift to 794.2 nm and 791 nm, respectively, accompanied by enhanced PL emission intensity compared to the pristine perovskite film. These blue-shifted peaks correlate with the increased bandgap observed in the Tauc plots after CsHCOO addition. The CsHCOO additives are believed to passivate defects of perovskite films, thereby augmenting PL emission intensity and reducing the full width at half maximum (FWHM) of PL emission profiles. Furthermore, the highest PL intensity achieved for the FA0.9Cs0.1PbI3 perovskite film with a 3% CsHCOO additive indicates that an optimal CsHCOO additive concentration can significantly reduce defect density.
Time-resolved photoluminescence (TRPL) measurements were conducted to investigate carrier dynamics in FA0.9Cs0.1PbI3 perovskite films with varying additive levels. Fig. S7 presents the normalized TRPL profiles of perovskite films containing 0%, 3%, 7%, and 10% CsHCOO additives, grown on FTO/cp-TiO2/SnO2 substrates and illuminated from the perovskite film surface using a UV laser (λ = 375 nm). Fig. 4(d) illustrates the corresponding fitted TRPL profiles. Given the UV laser penetration depth of less than 100 nm, this study focuses on carrier dynamics near the perovskite film surface. The normalized TRPL profiles were fitted with a bi-exponential decay equation: A1e−t/τ1 + A2e−t/τ2, where A1 and A2 are the corresponding amplitudes. The fitting revealed two distinct decay components for all samples: a fast decay time (τ1) attributed to trap-assisted recombination and a slower decay time (τ2) associated with bimolecular recombination.65Table 1 summarizes the characteristic TRPL decay parameters for the FA0.9Cs0.1PbI3 perovskite films with different additive levels. The pristine FA0.9Cs0.1PbI3 perovskite film exhibits τ1 = 8.16 ns and τ2 = 45.45 ns, resulting in an average lifetime (τavg = A1τ1 + A2τ2) of 33.94 ns. The 3%-CsHCOO-added FA0.9Cs0.1PbI3 film demonstrates extended lifetimes of τ1 = 10.08 ns and τ2 = 70.93 ns, leading to a longer average lifetime of 50.15 ns. This improvement is primarily attributed to the enhanced grain size, reduced grain boundaries, and a lower defect density. However, FA0.9Cs0.1PbI3 perovskite films with 7% and 10% CsHCOO additives show reduced τ1 values of 7.33 ns and 7.9 ns, respectively, compared to the 3% CsHCOO-added film. These findings suggest that high CsHCOO additive levels in FA0.9Cs0.1PbI3 perovskite films can increase trap-assisted recombination, potentially due to the formation of small debris on the perovskite film surface, as observed in Fig. 1(c) and (d). Furthermore, the trend in the average lifetime for the perovskite films with different additive level is consistent with the trend in average grain size shown in Fig. 1(e), indicating that perovskite grain size positively impacts carrier lifetime.
| Additive level of CsHCOO (%) | A 1 (%) | τ 1 (ns) | A 2(%) | τ 2 (ns) | τ avg (ns) |
|---|---|---|---|---|---|
| 0 | 30.87 | 8.16 | 69.13 | 45.45 | 33.94 |
| 3 | 34.16 | 10.08 | 65.84 | 70.93 | 50.15 |
| 7 | 31 | 7.33 | 69 | 53.63 | 39.28 |
| 10 | 37.64 | 7.9 | 62.36 | 47.59 | 32.66 |
We compared the photovoltaic performance of n–i–p structured PSCs with the device architecture FTO/cp-TiO2/SnO2/perovskite/Sprio-OMeTAD/Ag, employing FA0.9Cs0.1PbI3 perovskite films containing 0%, 3%, 7%, and 10% CsHCOO additives as light absorbers. Fig. S8 depicts the J–V curves of the champion-performing PSCs with 0%, 3%, 7%, and 10% CsHCOO additives, as measured under forward (from short circuit to open circuit) and reverse (from open circuit to short circuit) scans while exposed to standard one-sun AM 1.5G simulated solar illumination. Table 2 summarizes the corresponding photovoltaic parameters of these four devices. To clearly highlight the differences among devices with varying additive concentration, Fig. 5(a) specifically compares the J–V curves of these four champion-performing PSCs measured under a forward scan. The device without CsHCOO additive demonstrates an open-circuit voltage (VOC) of 1.03 V, a short-circuit current density (JSC) of 22.19 mA cm−2, and a fill factor (FF) of 74%, yielding a PCE of 17.12%. Incorporating 3%, 7%, and 10% CsHCOO additives into the PSCs achieves PCEs of 18.57%, 18.21%, and 18.09%, respectively, surpassing the performance of the control device without CsHCOO additive. Notably, the device with 3% CsHCOO additive exhibited the best PCE of 18.57%, accompanied by a VOC = 1.05 V, a JSC = 23.46 mA cm−2, and an FF of 75%. This significant PCE enhancement is primarily attributed to increased VOC, JSC, and FF. The improvements in VOC, JSC, and FF for the 3%-CsHCOO-containing devices are likely due to enhanced grain size, reduced grain boundaries (as observed in SEM images in Fig. 1), decreased defect density (as determined by SCLC measurements), and surface Pb0 defect passivation by the CsHCOO additive (as evidenced by XPS measurements in Fig. 4). However, when the CsHCOO additive concentration increased from 3% to 7% and 10% in the precursor solution, both VOC and JSC of the devices decreased slightly, leading to lower PCEs compared to the 3%-CsHCOO-added PSC. This reduction in performance is attributed to reduced grain size, the formation of small debris on the film surface, and increased defect density in the perovskite films with 7% and 10% CsHCOO additives. Fig. S9(a)–(d) present the statistical photovoltaic parameters of PSCs with 0%, 3%, 7%, and 10% CsHCOO additives. These data were derived from 20 devices measured under forward scan, indicating that the PSC incorporating with 3% CsHCOO exhibits the best performance and high reproducibility.
| Additive level of CsHCOO (%) | Scan direction | V OC (V) | J SC (mA cm−2) | FF (%) | PCE (%) | Integrated JSC (mA cm−2) |
|---|---|---|---|---|---|---|
| 0 | Forward | 1.03 | 22.19 | 74 | 17.12 | 21.81 |
| Reverse | 1.02 | 22.19 | 72 | 16.30 | ||
| 3 | Forward | 1.05 | 23.46 | 75 | 18.57 | 23.52 |
| Reverse | 1.04 | 23.00 | 73 | 17.36 | ||
| 7 | Forward | 1.05 | 23.03 | 75 | 18.21 | 22.82 |
| Reverse | 1.04 | 23.26 | 72 | 17.66 | ||
| 10 | Forward | 1.04 | 23.43 | 74 | 18.09 | 22.58 |
| Reverse | 1.02 | 22.69 | 71 | 16.50 |
Fig. 5(b) illustrates the IPCE spectra and integrated JSC of PSCs with different CsHCOO additive levels. The integrated JSC values for PSCs with 0%, 3%, 7%, and 10% CsHCOO additives are 20.81, 23.52, 22.82, and 22.58 mA cm−2, respectively, aligning with the JSC values obtained from the J–V curves. The IPCE values of PSCs with a 3% CsHCOO additive consistently exceeded those of the pristine PSCs over the entire wavelength range from 300 to 800 nm. Devices with 7% and 10% CsHCOO additives exhibited decreased IPCE values at longer wavelengths (highlighted by the light-yellow background), with a more pronounced decline observed in devices with 10% CsHCOO additive. As the perovskite film's absorption coefficient is inversely proportional to wavelength, long-wavelength light incident from the FTO substrate can penetrate through the perovskite film and reach the perovskite/spiro-OMeTAD interface. The formation of small debris on the surface of perovskite films with 7% and 10% CsHCOO additives enhances charge recombination at this interface, reducing charge extraction efficiency and leading to a decrease in IPCE values.
Fig. S9(e) presents the statistical hysteresis index (HI) distribution of devices incorporating 0%, 3%, 7%, and 10% CsHCOO additives. The HI is defined by the equation:
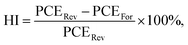 | (2) |
Fig. S9(f) compares the long-term stability of PSCs with different CsHCOO additive levels for unencapsulated devices stored under ambient conditions (RH = 25 ± 5%). Pristine devices retained 79% of their initial PCE after 1000 hours, while devices with 3%, 7%, and 10% CsHCOO additives maintained 90%, 97%, and 97% of their initial PCE values, respectively, after the same period. These results indicate that CsHCOO additives effectively suppress the formation of PbI2 in the perovskite film, which can degrade the performance and stability of PSCs.67,68 Additionally, the hydrogen bonding between FA+ and HCOO− can stabilize the perovskite structure, further enhancing the perovskite film's resistance to humidity.26
Devices incorporating CsHCOO additives exhibit superior PCE compared to those without CsHCOO, primarily due to enhanced values of VOC and JSC. This improvement is correlated with the HCOO− additive, which may effectively suppress trap-assisted recombination within the perovskite layer. To further investigate photogenerated charge extraction and recombination processes in PSCs with varying additive levels, we performed light-intensity-dependent J–V measurements, analyzing JSC and VOC as functions of illumination intensity ranging from 1 to 100 mW cm−2. The relationship between JSC and illumination light intensity (I) follows the equation: JSC ∝ Iα. Fig. 5(c) illustrates the power-law dependence of JSC on light intensity, displaying a linear relationship on a double logarithmic scale. The slopes (α values) of PSCs with 0%, 3%, 7%, and 10% CsHCOO are 1.02, 0.98, 0.97, and 0.98, respectively, which are comparable and close to 1. The slope of the logarithmic curve approaching 1 indicates low recombination in the devices, suggesting that photogenerated carriers in these devices are effectively extracted under short-circuit conditions.45
The relationship between VOC and light intensity can be expressed by the following formula for VOC ≫ kBT/q:
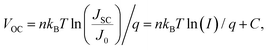 | (3) |
To elucidate the impact of CsHCOO on the suppression of trap-assisted recombination, we characterized the space-charge-limited current (SCLC) of devices with an FTO/perovskite/Ag architecture under varying bias voltages in the dark. Fig. S10 presents the dark current–voltage (I–V) curves of devices with different CsHCOO additive levels on a double logarithmic scale. The abrupt increase in dark current at the trap-filling limit voltage (VTFL), deduced from the intersection point of the trap-filling limited region and the trap-free SCLC region, indicates that all traps are occupied by charge carriers.71 The trap density (Ntrap) was calculated using the following equation:
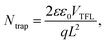 | (4) |
We also demonstrated the applicability of 3% CsHCOO additive in the p–i–n structured PSCs, which possess a device architecture of glass/ITO/3PATAT-C3/perovskite/C60/BCP/Ag.72 Fig. S11(a) compares the J–V curves of devices without and with the 3% CsHCOO additive under AM 1.5G solar illumination and both forward and reverse scan directions; the corresponding photovoltaic parameters are detailed in Table S2. The device containing the 3% CsHCOO additive achieves an optimized PCE of 20.04% under reverse scan, outperforming the PCE of 18.01% recorded for the control device. Furthermore, Fig. S11(b) presents the statistical PCE data derived from 20 devices measured under both scan directions. The device with 0% and 3% CsHCOO additive exhibits statistic PCEs of 15.39 ± 1.53% and 19.64 ± 0.14%, respectively. These results clearly indicate that the p–i–n structured PSC incorporating 3% CsHCOO not only exhibits the best performance but also demonstrates high reproducibility.
In the second study, we investigated the effect of CsHCOO on PSCs by substituting it for CsI in the FA0.9Cs0.1PbI3 perovskite precursor. Perovskite films were prepared using FA0.9Cs0.1PbI3 precursors containing 0%, 3%, and 10% molar ratios of CsHCOO replacing CsI, labeled as R0%, R3%, and R10%, respectively. The ideal stoichiometric compositions for the R0%, R3%, and R10% perovskite films are FA0.9Cs0.1PbI3, FA0.9Cs0.1PbI2.97HCOO0.03, and FA0.9Cs0.1PbI2.9HCOO0.1, respectively. Fig. 6(a)–(c) present top-view SEM images of the R0%, R3%, and R10% perovskite films deposited on FTO/cp-TiO2/SnO2 substrates, revealing compact surfaces with densely packed grains. These images demonstrate that the perovskite film grain size decreases as the CsHCOO substitution increases. Similar to the first study, some small debris (marked by yellow dashed circles) is observed in the R3% and R10% perovskite films, which are attributed to PbI2.32
 | ||
| Fig. 6 Top-view SEM images of perovskite films with (a) 0% (control, R0%), (b) 3% (R3%), and (c) 10% (R10%) molar ratios of CsHCOO replacing CsI. | ||
Fig. 7(a) presents the XRD profiles of the R0%, R3%, and R10% perovskite films deposited on FTO/cp-TiO2/SnO2 substrates. The primary XRD peaks correspond to the characteristic peaks of the α-phase FA0.9Cs0.1PbI3 perovskite. An XRD peak at 2θ = 12.6° (marked by ▼) is attributed to PbI2. Notably, the intensity of this PbI2 peak increases with the amount of CsHCOO replacing CsI. Unlike the first study, where excess CsHCOO as an additive in the perovskite precursor suppressed PbI2 formation, replacing CsI with CsHCOO in the perovskite precursor promotes PbI2 formation. As previously discussed, partially replacing CsI with CsHCOO leads to the formation of [PbI6−x(HCOO)x]4− octahedral in the precursor. These octahedral can convert into [PbI6]4− octahedral through anion exchange prior to perovskite formation. However, when the precursor contains insufficient CsI, the resulting I− deficiencies lead to incomplete HCOO− exchange. After the vaporization of HCOO− from the perovskite film by post-annealing, the non-exchanged corner-sharing [PbI6−x(HCOO)x]4− octahedra transform into face-sharing [PbI6]4− octahedra, ultimately resulting in the formation of PbI2.44 Although a moderate amount of residual PbI2 in the perovskite film is beneficial for defect passivation, excess PbI2 formation within the perovskite layer can severely hinder carrier transport.32 Following this PbI2 formation, an insufficient quantity of CsI is unable to fully convert the PbI2 into the perovskite phase. These results further suggest that the HCOO−–I− anion exchange occurred prior to perovskite formation.
Fig. 7(b) presents zoomed-in XRD profiles from Fig. 7(a) at 2θ ∼ 14°, focusing on the XRD peak of the (100) facet for the R0%, R3%, and R10% perovskite films. Similar to the first study, the results show a gradual shift of the (100) facet XRD peak to a higher angle. This shift, which occurs as the amount of CsHCOO replacing CsI in the perovskite precursor increases, indicates a reduced lattice spacing. Fig. 7(c) compares the steady-state PL spectra of CsHCOO-substituted perovskite films illuminated by a blue laser (λ = 405 nm, power = 2 mW), showing PL peaks at 802.2 nm, 801.1 nm, and 794.2 nm for the R0%, R3%, and R10% perovskite films, respectively. Just as observed with CsHCOO additives, films where CsHCOO completely replaces CsI show a PL peak shift toward shorter wavelengths as the CsHCOO concentration increases. This shift indicates a rising bandgap, which can be attributed to the incorporation of the smaller radius Cs+ cations (compared to FA+ cations) into the perovskite lattice. The resulting reduced perovskite lattice spacing, as confirmed in Fig. 7(b), directly leads to the increase in the film's bandgap. Fig. 7(d) displays the PL spectra of the R10% perovskite film excited by a blue laser at four different positions, revealing inconsistent PL peak intensities and slightly varying PL peak wavelengths. This variation is likely due to the uneven surface of the perovskite film, as observed in Fig. 6(c), and the non-uniform substitution of I− by HCOO−.
Fig. S12 compares the J–V curves of PSCs employing the device structure FTO/cp-TiO2/SnO2/perovskite/Sprio-OMeTAD/Ag, utilizing the R0%, R3%, and R10% perovskite films. These measurements were performed under standard one-sun AM 1.5G simulated solar illumination and both forward and reverse scan conditions. The corresponding photovoltaic parameters are summarized in Table 3. The J–V curves for all devices under forward scan direction are presented in Fig. 7(e) for clear comparison. The control device (R0% device), identical to the device without the CsHCOO additive presented in the first study, exhibits a PCE of 17.12%. After replacing 3% of CsI with CsHCOO in the perovskite precursor, the R3% device achieves a PCE of 16.23%, inferior to the R0% device primarily due to simultaneous reductions in JSC and VOC. The decrease in JSC and VOC for the R3% device is likely attributed to reduced grain size, increased grain boundaries (as observed in SEM images in Fig. 6(b)), and a higher PbI2 content. Further replacing CsI with CsHCOO in the R10% perovskite film reduces PCE due to an increase in defect states. The formation of small-sized debris on the perovskite film surface and a higher PbI2 content result in a reduced average grain size and an increased density of defect states. Consequently, severe charge recombination at the perovskite/spiro-OMeTAD interface significantly reduces JSC, VOC, and FF.
| Device | Scan direction | V OC (V) | J SC (mA cm−2) | FF (%) | PCE (%) | Integrated JSC (mA cm−2) |
|---|---|---|---|---|---|---|
| R0% PSCs (control device) | Forward | 1.03 | 22.19 | 74 | 17.12 | 21.81 |
| Reverse | 1.02 | 22.19 | 72 | 16.30 | ||
| R3% PSCs | Forward | 1.00 | 21.52 | 75 | 16.23 | 20.53 |
| Reverse | 0.98 | 21.56 | 74 | 15.53 | ||
| R10% PSCs | Forward | 0.98 | 20.13 | 71 | 13.99 | 15.10 |
| Reverse | 0.96 | 19.96 | 71 | 13.56 |
Fig. 7(f) presents the IPCE spectra and integrated JSC of the R0%, R3%, and R10% PSCs. The integrated JSC values for the R0%, R3%, and R10% PSCs are estimated to be 20.81, 20.53, and 15.10 mA cm−2, respectively. Notably, the difference between the measured JSC and integrated JSC for the R3% PSC is larger than that for the R0% PSC, and a significant difference of 5.03 mA cm−2 is observed for the R10% PSC. The high density of defect states, inducing severe recombination in the R10% PSC, leads to a notable discrepancy between the measured and integrated JSC, when the PSC is measured under low-intensity light illumination during IPCE measurement.
4. Conclusions
In summary, we investigated the effects of CsHCOO as both an additive and a substitute for CsI in the perovskite film of PSCs. CsHCOO addition effectively suppressed trap-assisted recombination in PSCs by promoting larger perovskite grain size and preferred facet alignment perpendicular to the substrate. The introduced HCOO− anion further passivated surface defects arising from uncoordinated Pb2+ cations. Consequently, an appropriate amount of CsHCOO additive (3%) in the perovskite film suppressed non-radiative recombination, facilitating charge carrier transport. Ultimately, 3%-CsHCOO-added PSCs achieved higher photovoltaic performance, with improved VOC and JSC compared to PSCs without CsHCOO. Conversely, substituting CsI with CsHCOO in the perovskite precursor negatively impacted the PV performance of PSCs. Poor perovskite film quality, characterized by reduced grain size, a rough film surface, and increased PbI2 residuals, was responsible for the reduced PV performance compared to control PSCs.Author contributions
The manuscript was written through contributions of all authors. All authors have given approval to the final version of the manuscript. Raja Rajendran: visualization, interpretation of data, and writing – original draft. Pei-Yu Wang: methodology, validation, formal analysis, investigation, data curation, and visualization. Ming-Hsien Li: visualization, interpretation of data, writing – original draft, and writing – review & editing. Chen-Fu Lin: interpretation of data, and writing – review & editing. Farhan Yousuf: methodology, validation, data curation, and visualization. Cheng-Hung Hou: resources, investigation, and data curation. Jing-Jong Shyue: resources, investigation, and data curation. Peter Chen: conceptualization, methodology, writing – review & editing, supervision, project administration, and funding acquisition.Conflicts of interest
There are no conflicts to declare.Data availability
The data supporting this article (including FTIR spectra, XPS spectra, TRPL profiles, and J–V curves) have been included as part of the supplementary information (SI). Supplementary information is available. See DOI: https://doi.org/10.1039/d5tc02615f.Acknowledgements
The authors are grateful to the research grant from the Ministry of Science and Technology, the National Science and Technology Council of Taiwan (MOST 110-2221-E-006-200, MOST 111-2221-E-006-061-MY2, NSTC 113-2223-E-006-011, NSTC 113-2112-M-150-002, and NSTC 114-2112-M-150-002). This work was financially supported by the Hierarchical Green-Energy Materials (Hi-GEM) Research Center, from The Featured Areas Research Center Program within the framework of the Higher Education Sprout Project by the Ministry of Education (MOE) in Taiwan. This research was supported in part by the Higher Education Sprout Project, Ministry of Education to the Headquarter of University Advancement at National Cheng Kung University (NCKU). The authors gratefully acknowledge the use of EM003600 of MOST 106-2731-M-006-001 equipment belonging to the Core Facility Center of National Cheng Kung University.References
- M. Habibi, F. Zabihi, M. R. Ahmadian-Yazdi and M. Eslamian, Renewable Sustainable Energy Rev., 2016, 62, 1012–1031 CrossRef CAS.
- S. Akel, A. Kulkarni, U. Rau and T. Kirchartz, PRX Energy, 2023, 2, 013004 CrossRef.
- M. H. Miah, M. U. Khandaker, M. B. Rahman, M. Nur-E-Alam and M. A. Islam, RSC Adv., 2024, 14, 15876–15906 RSC.
- D. Duan, C. Ge, M. Z. Rahaman, C.-H. Lin, Y. Shi, H. Lin, H. Hu and T. Wu, NPG Asia Mater., 2023, 15, 8 CrossRef CAS.
- T. Zhu and X. Gong, InfoMat, 2021, 3, 1039–1069 CrossRef CAS.
- S. J. Kim, S. Park, H. M. Cho and H. W. Jang, Mater. Today Electron., 2024, 9, 100111 CrossRef.
- F. Ruf, M. F. Aygüler, N. Giesbrecht, B. Rendenbach, A. Magin, P. Docampo, H. Kalt and M. Hetterich, APL Mater., 2019, 7, 031113 CrossRef.
- X. Zhang, M. E. Turiansky, J.-X. Shen and C. G. Van de Walle, J. Appl. Phys., 2022, 131 Search PubMed.
- N. P. Jasti, I. Levine, Y. Feldman, G. Hodes, S. Aharon and D. Cahen, Proc. Natl. Acad. Sci. U. S. A., 2024, 121, e2316867121 CrossRef CAS PubMed.
- M. A. Green, E. D. Dunlop, M. Yoshita, N. Kopidakis, K. Bothe, G. Siefer, D. Hinken, M. Rauer, J. Hohl-Ebinger and X. Hao, Prog. Photovoltaics Res. Appl., 2024, 32, 425–441 CrossRef.
- R. Sharif, A. Khalid, S. W. Ahmad, A. Rehman, H. G. Qutab, H. H. Akhtar, K. Mahmood, S. Afzal and F. Saleem, Nanoscale Adv., 2023, 5, 3803–3833 RSC.
- X. Xie, S. Zeng, C. Zhou and S. Xiao, Mater. Chem. Front., 2023, 7, 5309–5332 RSC.
- D.-K. Lee and N.-G. Park, Appl. Phys. Rev., 2023, 10, 011308 CAS.
- H. Lu, A. Krishna, S. M. Zakeeruddin, M. Grätzel and A. Hagfeldt, iScience, 2020, 23, 101359 CrossRef CAS PubMed.
- J. Jiao, C. Yang, Z. Wang, C. Yan and C. Fang, Results Eng., 2023, 18, 101158 CrossRef CAS.
- F. Zhang and K. Zhu, Adv. Energy Mater., 2020, 10, 1902579 CrossRef CAS.
- A. Mahapatra, D. Prochowicz, M. M. Tavakoli, S. Trivedi, P. Kumar and P. Yadav, J. Mater. Chem. A, 2020, 8, 27–54 RSC.
- B. Walker, G.-H. Kim and J. Y. Kim, Adv. Mater., 2019, 31, 1807029 CrossRef PubMed.
- P.-Y. Lin, A. Loganathan, I. Raifuku, M.-H. Li, Y.-Y. Chiu, S.-T. Chang, A. Fakharuddin, C.-F. Lin, T.-F. Guo, L. Schmidt-Mende and P. Chen, Adv. Energy Mater., 2021, 11, 2100818 CrossRef CAS.
- J. Chen and N.-G. Park, Small Methods, 2021, 5, 2100311 CrossRef CAS.
- L. Chu, Matter, 2021, 4, 1762–1764 CrossRef CAS.
- H. Kim, J. Lim, M. Sohail and M. K. Nazeeruddin, Solar RRL, 2022, 6, 2200013 CrossRef CAS.
- S. Wang, C. Wu, H. Yao, L. Ding and F. Hao, Mater. Chem. Front., 2023, 7, 789–805 RSC.
- H. Lu, Y. Liu, P. Ahlawat, A. Mishra, W. R. Tress, F. T. Eickemeyer, Y. Yang, F. Fu, Z. Wang, C. E. Avalos, B. I. Carlsen, A. Agarwalla, X. Zhang, X. Li, Y. Zhan, S. M. Zakeeruddin, L. Emsley, U. Rothlisberger, L. Zheng, A. Hagfeldt and M. Grätzel, Science, 2020, 370, eabb8985 CrossRef CAS PubMed.
- S. Bai, P. Da, C. Li, Z. Wang, Z. Yuan, F. Fu, M. Kawecki, X. Liu, N. Sakai, J. T.-W. Wang, S. Huettner, S. Buecheler, M. Fahlman, F. Gao and H. J. Snaith, Nature, 2019, 571, 245–250 CrossRef CAS.
- J. Jeong, M. Kim, J. Seo, H. Lu, P. Ahlawat, A. Mishra, Y. Yang, M. A. Hope, F. T. Eickemeyer, M. Kim, Y. J. Yoon, I. W. Choi, B. P. Darwich, S. J. Choi, Y. Jo, J. H. Lee, B. Walker, S. M. Zakeeruddin, L. Emsley, U. Rothlisberger, A. Hagfeldt, D. S. Kim, M. Grätzel and J. Y. Kim, Nature, 2021, 592, 381–385 CrossRef CAS PubMed.
- W. Ke, C. Xiao, C. Wang, B. Saparov, H.-S. Duan, D. Zhao, Z. Xiao, P. Schulz, S. P. Harvey, W. Liao, W. Meng, Y. Yu, A. J. Cimaroli, C.-S. Jiang, K. Zhu, M. Al-Jassim, G. Fang, D. B. Mitzi and Y. Yan, Adv. Mater., 2016, 28, 5214–5221 CrossRef CAS.
- M. K. Kim, T. Jeon, H. I. Park, J. M. Lee, S. A. Nam and S. O. Kim, CrystEngComm, 2016, 18, 6090–6095 RSC.
- D. Kim, H. J. Jung, I. J. Park, B. W. Larson, S. P. Dunfield, C. Xiao, J. Kim, J. Tong, P. Boonmongkolras, S. G. Ji, F. Zhang, S. R. Pae, M. Kim, S. B. Kang, V. Dravid, J. J. Berry, J. Y. Kim, K. Zhu, D. H. Kim and B. Shin, Science, 2020, 368, 155–160 CrossRef CAS.
- Y.-H. Chiang, M.-H. Li, H.-M. Cheng, P.-S. Shen and P. Chen, ACS Appl. Mater. Interfaces, 2017, 9, 2403–2409 CrossRef CAS PubMed.
- Y.-H. Chiang, H.-M. Cheng, M.-H. Li, T.-F. Guo and P. Chen, ChemSusChem, 2016, 9, 2620–2627 CrossRef CAS PubMed.
- P.-Y. Lin, C.-F. Lin, Y.-Y. Chiu, H.-H. Chen, M.-H. Li, R. Raja, C.-S. Wu, C.-H. Hou, S. Sung-Yun Hsiao, J.-J. Shyue, D.-C. Lee, S.-Z. Ho, Y.-C. Chen and P. Chen, ACS Appl. Energy Mater., 2023, 6, 79–88 CrossRef CAS.
- J. Lee, Y. S. Shin, E. Oleiki, J. Seo, J. Roe, D. Lee, Y. Lee, T. Song, H. Jang, J. W. Song, W. Lee, G. Lee, J. Y. Kim and D. S. Kim, Energy Environ. Sci., 2024, 17, 6003–6012 RSC.
- L. Meng, Q. Wei, Z. Yang, D. Yang, J. Feng, X. Ren, Y. Liu and S. Liu, J. Energy Chem., 2020, 41, 43–51 CrossRef.
- J.-Y. Seo, T. Matsui, J. Luo, J.-P. Correa-Baena, F. Giordano, M. Saliba, K. Schenk, A. Ummadisingu, K. Domanski, M. Hadadian, A. Hagfeldt, S. M. Zakeeruddin, U. Steiner, M. Grätzel and A. Abate, Adv. Energy Mater., 2016, 6, 1600767 CrossRef.
- W. Hui, L. Chao, H. Lu, F. Xia, Q. Wei, Z. Su, T. Niu, L. Tao, B. Du, D. Li, Y. Wang, H. Dong, S. Zuo, B. Li, W. Shi, X. Ran, P. Li, H. Zhang, Z. Wu, C. Ran, L. Song, G. Xing, X. Gao, J. Zhang, Y. Xia, Y. Chen and W. Huang, Science, 2021, 371, 1359–1364 CrossRef CAS PubMed.
- W. Song, X. Wang, T. Hou, X. Li, H. Chen, Y. Yu, X. Sun, A. Singh and M. Zhang, Chem. – Eur. J., 2023, 29, e202300576 CrossRef CAS PubMed.
- S. Thakur, S.-N. Kwon, D. S. Mann and S.-I. Na, J. Materiomics, 2022, 8, 1165–1171 CrossRef.
- M. J. Jeong, S. W. Jeon, S. Y. Kim and J. H. Noh, Adv. Energy Mater., 2023, 13, 2300698 CrossRef CAS.
- Z. Gan, L. Zhao, X. Sun, K. Xu, H. Li and J. Wei, Crystals, 2022, 12, 1194 CrossRef CAS.
- Q. Guo, Y. Ding, Z. Dai, Z. Chen, M. Du, Z. Wang, L. Gao, C. Duan, Q. Guo and E. Zhou, Phys. Chem. Chem. Phys., 2022, 24, 17526–17534 RSC.
- F. Yousuf, C.-F. Lin, M.-H. Li and P. Chen, ACS Appl. Energy Mater., 2025, 8, 11479–11489 CrossRef CAS.
- R. Ghayoor, F. Ghasemi, L. Shooshtari, F. Tajabadi, R. Mohammadpour and N. Taghavinia, Sustainable Energy Fuels, 2025, 9, 3374–3388 RSC.
- J. Gu, F. Li, Z. Wang, Y. Xie, L. Yan, P. Zeng, H. Yu and M. Liu, Adv. Sci., 2020, 7, 2002296 CrossRef CAS.
- D. Baek, G. Y. Park, J. Cha, H. Na, D. S. Ham and M. Kim, J. Mater. Sci. Technol., 2023, 165, 161–169 CrossRef CAS.
- Y. Wang, B. Zhou, M. Han, J. Zhao, R. Wang, J. Zhang, H. Ren, G. Hou, Y. Ding, Y. Zhao and X. Zhang, Nano Energy, 2023, 118, 108981 CrossRef CAS.
- Z. Zhou, J. Liang, Z. Zhang, Y. Zheng, X. Wu, C. Tian, Y. Huang, J. Wang, Y. Yang, A. Sun, Z. Chen and C.-C. Chen, ACS Appl. Mater. Interfaces, 2022, 14, 49886–49897 CrossRef CAS.
- L. Deng, H. Wang, S. Rafique, Y. Wang, T. Hu, K. Liu, Y. Wang, X. Li, Z. Xie, J. Tang, Z. Liu, J. Li, W. Yuan, J. Wang, A. Yu and Y. Zhan, Adv. Funct. Mater., 2023, 33, 2303742 CrossRef CAS.
- G. Park, W. Yang, A. Liu, H. Zhu, F. De Angelis and Y.-Y. Noh, Mater. Sci. Eng., R, 2024, 159, 100806 CrossRef.
- H. Yu, T. Zhang, Z. Zhang, Z. Liu, Q. Sun, J. Huang, L. Dai, Y. Shen, X. Li and M. Wang, Mater. Horiz., 2024, 11, 4730–4736 RSC.
- K.-W. Huang, M.-H. Li, P.-T. Hsieh, C.-F. Lin, R. Rajendran, Y.-L. Tung and P. Chen, J. Mater. Chem. C, 2022, 10, 16016–16027 RSC.
- Z. Wu, S. Sang, J. Zheng, Q. Gao, B. Huang, F. Li, K. Sun and S. Chen, Angew. Chem., Int. Ed., 2024, 63, e202319170 CrossRef CAS.
- B. Li, H. Mao, Y. Xing, X. Ma, L. Zhang, Z. Fang, L. Luo, A. Wang, N. Yuan and J. Ding, ACS Appl. Mater. Interfaces, 2025, 17, 22090–22097 CrossRef CAS.
- Y. Gao, H. Raza, Z. Zhang, W. Chen and Z. Liu, Adv. Funct. Mater., 2023, 33, 2215171 CrossRef CAS.
- L. Cai, C. W. Suen, Y. S. Lau, Z. Lan, J. Han and F. Zhu, ACS Appl. Energy Mater., 2022, 5, 8304–8312 CrossRef CAS.
- S. Shin, S. Seo, S. Jeong, A. S. Sharbirin, J. Kim, H. Ahn, N.-G. Park and H. Shin, Adv. Sci., 2023, 10, 2300798 CrossRef CAS PubMed.
- H. Kong, C. Zhao, H. Han, H. Liu, P. Zou, Y. Fu, K. Lang, F. Shen, X. Liu, J. Xu and J. Yao, Small, 2024, 20, 2401669 CrossRef CAS PubMed.
- J. Liang, X. Hu, C. Wang, C. Liang, C. Chen, M. Xiao, J. Li, C. Tao, G. Xing, R. Yu, W. Ke and G. Fang, Joule, 2022, 6, 816–833 CrossRef CAS.
- G. Tumen-Ulzii, C. Qin, D. Klotz, M. R. Leyden, P. Wang, M. Auffray, T. Fujihara, T. Matsushima, J.-W. Lee, S.-J. Lee, Y. Yang and C. Adachi, Adv. Mater., 2020, 32, 1905035 CrossRef CAS.
- L. Chen, J. Chen, C. Wang, H. Ren, Y.-X. Luo, K.-C. Shen, Y. Li, F. Song, X. Gao and J.-X. Tang, ACS Appl. Mater. Interfaces, 2021, 13, 24692–24701 CrossRef CAS.
- M. Qin, P. F. Chan and X. Lu, Adv. Mater., 2021, 33, 2105290 CrossRef CAS PubMed.
- G. Lau, H. Xu, Y. Li, K. Ding, Y. Zhang and W. Lin, Adv. Funct. Mater., 2025, 2503757 CrossRef CAS.
- J.-W. Lee, D.-H. Kim, H.-S. Kim, S.-W. Seo, S. M. Cho and N.-G. Park, Adv. Energy Mater., 2015, 5, 1501310 CrossRef.
- Y. Wang, M. Yang, Y. Tang and M. Zhao, Sol. Energy Mater. Sol. Cells, 2022, 246, 111941 CrossRef CAS.
- E. V. Péan, S. Dimitrov, C. S. De Castro and M. L. Davies, Phys. Chem. Chem. Phys., 2020, 22, 28345–28358 RSC.
- A. Kumar, A. Rana, N. Vashistha, K. K. Garg and R. K. Singh, Sol. Energy, 2020, 211, 345–353 CrossRef CAS.
- A. S. Yerramilli, Y. Chen, D. Sanni, J. Asare, N. D. Theodore and T. L. Alford, Org. Electron., 2018, 59, 107–112 CrossRef CAS.
- M. Mangrulkar, S. Y. Luchkin, A. F. Akbulatov, I. Zhidkov, E. Z. Kurmaev, P. A. Troshin and K. J. Stevenson, Synth. Met., 2021, 278, 116823 CrossRef CAS.
- D. Glowienka and Y. Galagan, Adv. Mater., 2022, 34, 2105920 CrossRef CAS PubMed.
- M. T. Sirtl, R. Hooijer, M. Armer, F. G. Ebadi, M. Mohammadi, C. Maheu, A. Weis, B. T. van Gorkom, S. Häringer, R. A. J. Janssen, T. Mayer, V. Dyakonov, W. Tress and T. Bein, Adv. Energy Mater., 2022, 12, 2103215 CrossRef CAS.
- F. Cai, L. Yang, Y. Yan, J. Zhang, F. Qin, D. Liu, Y.-B. Cheng, Y. Zhou and T. Wang, J. Mater. Chem. A, 2017, 5, 9402–9411 RSC.
- M. A. Truong, T. Funasaki, L. Ueberricke, W. Nojo, R. Murdey, T. Yamada, S. Hu, A. Akatsuka, N. Sekiguchi, S. Hira, L. Xie, T. Nakamura, N. Shioya, D. Kan, Y. Tsuji, S. Iikubo, H. Yoshida, Y. Shimakawa, T. Hasegawa, Y. Kanemitsu, T. Suzuki and A. Wakamiya, J. Am. Chem. Soc., 2023, 145, 7528–7539 CrossRef CAS.
| This journal is © The Royal Society of Chemistry 2026 |





