Realizing ultralow lattice thermal conductivity in CuInTe2 by controlled cation disorder
Songlin
Li†
ab,
Jianxiang
Zhai†
a,
Chenghao
Xie
a,
Minghao
Ye
a,
Guoqing
Ding
a,
Junxi
Mei
a,
Qingjie
Zhang
a,
Xinfeng
Tang
 a and
Gangjian
Tan
a and
Gangjian
Tan
 *ab
*ab
aState Key Laboratory of Advanced Technology for Materials Synthesis and Processing, Wuhan University of Technology, Wuhan, 430070, China. E-mail: gtan@whut.edu.cn
bHubei Longzhong Laboratory, Wuhan University of Technology Xiangyang Demonstration Zone, Xiangyang, 441000, China
First published on 17th December 2025
Abstract
Tailoring structural disorder enables phonon transport manipulation, providing a viable route toward ultralow lattice thermal conductivity and high thermoelectric performance. Here, CdTe alloying is used to modulate the cation-sublattice configuration of CuInTe2 and to investigate how atomic disorder influences heat and charge transport. The introduction of Cd promotes a gradual structural transition from an ordered tetragonal structure to a disordered cubic structure, accompanied by enhanced Cu–In cation intermixing. Such disorder, arising from the cooperative formation of Cd substitutional and Cu–In antisite defects together with intrinsically weaker Cd–Te bonds, strengthens point defect scattering and induces pronounced lattice softening. As a result, the lattice thermal conductivity decreases to 0.44 W m−1 K−1 at 873 K, approaching the amorphous limit. However, excessive disorder intensifies carrier scattering and deteriorates electrical transport, while moderate cation disorder achieves a favorable phonon–carrier trade-off, yielding a peak ZT of 0.75 at 873 K for (CuInTe2)0.9(2CdTe)0.1. This work demonstrates that controlled cation-sublattice disorder effectively modulates phonon scattering, providing insight into designing chalcopyrite thermoelectric materials with intrinsically low lattice thermal conductivity.
1. Introduction
Thermoelectric materials can directly convert thermal energy into electrical energy without any moving parts, making them attractive for waste heat recovery and solid-state cooling in sustainable energy technologies.1–3 The thermoelectric performance is commonly evaluated by using the dimensionless figure of merit ZT = S2σT/κtot, where S, σ, T, and κtot represent the Seebeck coefficient, electrical conductivity, absolute temperature, and total thermal conductivity, respectively.4,5 This expression couples electronic and thermal transport, serving as a central criterion for evaluating material performance.6,7 Numerous theoretical and experimental studies have shown that strategies for improving ZT fall into two major categories: (i) enhancing the power factor (PF = S2σ), for example through carrier concentration optimization,8 band convergence,9 or resonant levels10 to boost electronic transport; and (ii) reducing the κtot,11 such as by nanostructuring,12,13 point defect engineering,14,15 or alloy-induced disorder to strengthen phonon scattering.16,17 However, due to the intrinsic coupling between electron and phonon transport, these parameters often counteract each other. For instance, increasing the carrier concentration enhances σ but typically suppresses S and increases the electronic thermal conductivity (κele), making ZT optimization a complex trade-off process.Among all physical quantities governing thermoelectric performance, the lattice thermal conductivity (κlat) shows the weakest dependence on carrier concentration. Precisely for this reason, κlat can be readily decoupled from electronic transport, serving as one of the robust parameters for optimizing material properties.18 Moreover, since κlat is primarily governed by phonon transport, it can be effectively lowered through independent crystal structure or microstructure design, in some cases approaching the amorphous limit.19 Atomic-scale disorder has been widely utilized as an effective approach to suppress the κlat.20–22 A classical example is alloy-induced chemical disorder in solid solutions,23 where the random occupation of lattice sites creates local atomic randomness that acts as an efficient phonon scattering source, thereby reducing κlat.24 This phenomenon has been theoretically described by the classic point defect scattering models developed by Callaway and Klemens, which have been extensively applied to various solid solutions.25,26 In addition, cation-sublattice disorder represents an important pathway for attaining ultralow κlat in specific material systems.27–29 For example, the δ-phase of AgSbTe2 was long believed to crystallize in a cubic rock-salt structure with random Ag/Sb distribution on the cation sublattice.30 The differences in the local force constants of Ag+ and Sb3+ cations, along with the ground-state degeneracy, lead to the spontaneous formation of nanoscale cation-ordering domains, which significantly enhances phonon scattering and results in an ultralow κlat.31 Another form of disorder is manifested as antisite defects, in which atoms occupy non-native lattice sites.32 In the quaternary diamond-like compound Cu2HgSnTe4,33 CuHg and HgCu antisite defects generate exceptionally strong phonon scattering, primarily originating from the intrinsically distinct vibrational modes of the constituent elements.
CuInTe2, a promising ternary thermoelectric compound,34 suffers from a relatively high κlat (∼6 W m−1 K−1 at room temperature), which remains a major obstacle to further improving its thermoelectric performance.35 Building on the established effectiveness of cation sublattice disorder and antisite defects in reducing κlat, adopting similar strategies offers a practical route to suppress the κlat of CuInTe2 and enhance its overall thermoelectric performance. Structurally, CuInTe2 is derived from the cubic zinc-blende framework and features two distinct cation sublattices (Cu and In) that occupy the original cation positions in an ordered configuration.36 At elevated temperatures, CuInTe2 transforms into a cubic phase derived from the zinc-blende framework, in which Cu and In cations share a common sublattice, reflecting its intrinsic tolerance to cation disorder.37 Although this cubic phase is crystallographically distinct from the rock-salt structure of AgSbTe2, both systems exhibit pronounced cation disorder. This inherent tolerance of CuInTe2 toward cation disorder provides a natural framework for tuning phonon transport and suppressing lattice thermal conductivity via controlled cation disorder and antisite defects.
Consistent with this structural propensity, first-principles calculations for the CuInTe2 system reveal that under specific chemical potential conditions, InCu antisite defects possess low formation energies, indicating that such defects are thermodynamically favorable and can intrinsically promote cation disorder, while simultaneously enhancing phonon scattering.38 Furthermore, extrinsic doping (e.g., Zn substitution) has been experimentally shown to regulate the concentrations of different defects in CuInTe2,39 thereby exerting a pronounced influence on κlat and overall thermoelectric performance. Inspired by the intrinsic defect tunability and structural flexibility of CuInTe2, together with theoretical insights into defect-mediated phonon scattering, we adopted CdTe alloying as a practical means to induce controllable cation disorder and elucidate its role in governing the structural and thermoelectric transport properties.
In this work, a structural transition in CuInTe2 was achieved through CdTe alloying, accompanied by a pronounced reduction in κlat, as shown in Fig. 1a. The (CuInTe2)0.6(2CdTe)0.4 sample exhibits a κlat of 0.44 W m−1 K−1 at 873 K, approaching the amorphous limit (κmin = 0.35 W m−1 K−1, see the SI),40 primarily due to intense phonon scattering arising from cation-sublattice disorder. Defect formation energy calculations indicate that Cd can occupy both Cu and In sublattice sites, and its incorporation significantly lowers the formation energy of CuIn antisite defects, leading to the emergence of abundant CuIn and InCu antisite defects. The mutual occupation of Cu and In sites increases the degree of cation disorder, ultimately driving the transformation from the fully ordered tetragonal structure to a fully disordered cubic phase (Fig. 1b). Moreover, the introduction of Cd–Te weak bonding causes local lattice softening, which in turn reduces phonon velocities. The combination of CdTe alloying and antisite defect scattering effectively suppresses κlat. However, the increased cation disorder also enhances carrier scattering, leading to a degradation in electronic transport. As a result, the (CuInTe2)0.9(2CdTe)0.1 sample achieves a maximum ZT of 0.75 at 873 K. Therefore, rational regulation of cation disorder provides a practical route to optimize thermal transport in chalcopyrite compounds with inherently high lattice thermal conductivity.
2. Results and discussion
2.1 Crystal structure and phase homogeneity
Fig. 2a and b present the X-ray diffraction (XRD) patterns of the sintered (CuInTe2)1−x(2CdTe)x compounds. For compositions with x ≤ 0.2, all diffraction peaks can be well indexed to the tetragonal CuInTe2 phase (I![[4 with combining macron]](https://www.rsc.org/images/entities/char_0034_0304.gif) 2d). In contrast, when x > 0.2, the diffraction features are more consistent with a cubic structure (F
2d). In contrast, when x > 0.2, the diffraction features are more consistent with a cubic structure (F![[4 with combining macron]](https://www.rsc.org/images/entities/char_0034_0304.gif) 3m), indicating a gradual transition from the intrinsic tetragonal phase to a cubic phase with increasing Cd content. Specifically, the diffraction peak corresponding to the (221) plane of tetragonal CuInTe2 progressively weakens with increasing x and completely disappears when x reaches 0.25.
3m), indicating a gradual transition from the intrinsic tetragonal phase to a cubic phase with increasing Cd content. Specifically, the diffraction peak corresponding to the (221) plane of tetragonal CuInTe2 progressively weakens with increasing x and completely disappears when x reaches 0.25.
To verify the compositional homogeneity and Cd distribution, electron probe X-ray microanalysis (EPMA) was conducted on the x = 0.3 sample (Fig. 2c). The back-scattered electron (BSE) image shows no noticeable contrast variations, and the elemental compositions of the two selected regions (spots 1 and 2) closely match the nominal stoichiometry (Table S1). Moreover, energy dispersive spectroscopy (EDS) element mappings confirm that Cu, In, Cd, and Te are uniformly distributed across the matrix. These results collectively demonstrate that Cd atoms are homogeneously incorporated into the CuInTe2 lattice, forming a chemically uniform and structurally stable single-phase (CuInTe2)1−x(2CdTe)x solid solution.
2.2 Defect-mediated structural evolution and cation disorder in (CuInTe2)1−x(2CdTe)x
XRD analysis reveals that CdTe alloying induces a distinct structural transition in CuInTe2. Given the critical role of intrinsic defects in determining the crystal structure of CuInTe2, we conducted a detailed investigation into defect evolution during Cd incorporation to better understand the driving forces behind this structural change. As shown in Fig. 3a, defect formation energy calculations indicate that Cd substitution at both Cu and In sites yields negative formation energies under different chemical potential conditions (Fig. S1), with the most favorable values occurring under Cu-poor conditions.Upon Cd incorporation into the cation sublattice, the formation energies of Cu vacancies (VCu), CuIn, and InCu antisite defects were further evaluated under Cu-poor conditions and compared with that of pristine CuInTe2 (Fig. 3b). The formation energy of CuIn antisite defects near the Fermi level decreases markedly by approximately 0.65 eV, accompanied by a slight reduction for InCu. These concurrent changes indicate that Cd incorporation reduces the energetic disparity between Cu and In sublattices, thereby facilitating cation intermixing. As a result, the increased configurational disorder weakens the sublattice potential contrast, ultimately stabilizing the disordered cubic phase at higher Cd concentrations. In parallel, a moderate increase in the formation energy of VCu is observed, which suppresses the formation of the Cu vacancies and slightly diminishes the hole concentration.
To experimentally validate the defect-driven structural transition and quantify the evolution of cation disorder, Rietveld refinements were carried out on the XRD patterns for all compositions based on both tetragonal (I![[4 with combining macron]](https://www.rsc.org/images/entities/char_0034_0304.gif) 2d) and cubic (F
2d) and cubic (F![[4 with combining macron]](https://www.rsc.org/images/entities/char_0034_0304.gif) 3m) structural models (details of model construction and refinement results are provided in the SI). Within the tetragonal phase region, the refinements reveal a continuous increase in the Cu–In antisite defect concentration with increasing Cd content, indicating a progressive enhancement of cation disorder.
3m) structural models (details of model construction and refinement results are provided in the SI). Within the tetragonal phase region, the refinements reveal a continuous increase in the Cu–In antisite defect concentration with increasing Cd content, indicating a progressive enhancement of cation disorder.
As shown in Fig. 3c and S6, the evolution of the tetragonal distortion parameter (η = c/2a) and anion displacement parameter (u) obtained from Rietveld refinement provides deeper insight into the structural transition. The tetragonal distortion parameter η describes the global deviation of the lattice from cubic symmetry, with η ≈ 1 corresponding to an ideal pseudocubic structure.41 The anion displacement parameter u is an internal structural parameter that specifies the anion position at (u, 1/4, 1/8) within the tetrahedral coordination environment.42,43 Deviations of u from 0.25 indicate charge imbalance-induced bond-length asymmetry between different cation species and reflect local tetrahedral distortion.
With progressive Cd incorporation, u increases monotonically, indicating a gradual reduction in the bond-length mismatch and a continuous relaxation of the local tetrahedral distortion. In contrast, η remains close to 1 and exhibits only minor variation with increasing CdTe content. This contrast suggests that the macroscopic tetragonal distortion is relatively weak, whereas the local structural evolution is more sensitively captured by the anion displacement parameter u. In this system, CdTe alloying introduces Cd cations whose valence state and Cd–Te bonding characteristics lie between those of Cu and In, which progressively reduces the bond-length asymmetry in the local tetrahedral environment and drives u toward the ideal zinc-blende value.
As u approaches 0.25, the anion position becomes increasingly symmetric with respect to different cation species, indicating that Cu, In, and Cd experience progressively more equivalent local coordination environments. This trend is consistent with an enhanced degree of cation disorder on the cation sublattice and the structural evolution toward a cubic phase. Notably, at the critical composition (x = 0.25), Rietveld refinements based on the tetragonal chalcopyrite model yield anomalously elevated u values and apparently excessive antisite defect concentrations, suggesting that this structural model becomes increasingly limited in describing the average structure of the system.
Moreover, as illustrated in Fig. 3d, the refined lattice parameters exhibit a nearly linear increase across the entire solid–solution range, primarily attributed to the larger covalent radius of Cd compared with Cu and In. When the structure transforms into the cubic phase, the unit cell volume becomes approximately half that of the original tetragonal cell, and the lattice constants converge to equal values (a = b = c), consistent with the increased crystallographic symmetry. This continuous lattice expansion confirms the successful incorporation of Cd into the CuInTe2 lattice.
2.3 Lattice thermal conductivity suppression driven by point defect scattering and lattice softening
The temperature-dependent lattice thermal conductivity of (CuInTe2)1−x(2CdTe)x compounds is presented in Fig. 4a and b. The lattice contribution (κlat) was obtained from the relation κtot = κlat + κele, where κtot is the measured total thermal conductivity and κele is the electronic contribution. The κele was estimated using the Wiedemann–Franz law, κele = LσT, with the Lorenz number L evaluated from the empirical expression L = 1.5 + exp(−|S|/116).44 It is evident that κlat decreases markedly with increasing x. The κlat drops from 5.82 W m−1 K−1 (x = 0) to 1.83 W m−1 K−1 (x = 0.4) at room temperature, corresponding to a 69% reduction. Moreover, for compositions with x > 0.05, the κlat decreases to below 0.5 W m−1 K−1 at 873 K, approaching the amorphous limit, indicating that CdTe alloying effectively suppresses phonon heat transport.To elucidate the mechanisms responsible for the remarkable reduction in κlat, the Callaway–Klemens model was applied to evaluate the contribution of point defect scattering in tetragonal (CuInTe2)1−x(2CdTe)x compositions (Fig. 4c).45–47 This model primarily accounts for phonon scattering arising from mass and strain fluctuations (detailed calculation methods and fitting parameters are provided in the SI). As shown in Fig. S8, the large contrasts in atomic mass and covalent radius among Cd, Cu, and In produce strong mass and strain fluctuations that substantially depress κlat. However, a residual gap remains: calculations that include only point defect scattering (blue dashed line) overestimate the experimental results (red spheres), implying that additional mechanisms further reduce the κlat.
Ultrasonic pulse-echo data (Fig. 4d) show a nearly linear decrease in sound velocity (vs) with increasing Cd content, accompanied by a drop in Debye temperature from 190 K (x = 0) to 160 K (x = 0.5). The pronounced reductions in sound velocity and Debye temperature indicate the presence of significant lattice softening, which provides an additional pathway for the further suppression of κlat. The influence of lattice softening on κlat reduction was quantitatively evaluated using the measured sound velocity:48–50
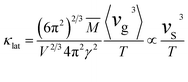 | (1) |
With the contribution of lattice softening incorporated into the theoretical model, the predicted κlat (green solid line in Fig. 4c) matches the experiment closely.
The crystal orbital Hamilton population (COHP) and integrated COHP (ICOHP) for Cu16In16Te32 and Cu12In12Cd8Te32 are shown in Fig. 4e and f. The COHP represents the density of states weighted by the Hamiltonian matrix elements and partitions the band energy into bonding, nonbonding, and antibonding contributions. Positive COHP values correspond to stabilizing bonding interactions, whereas negative values represent destabilizing antibonding states. The magnitude of ICOHP quantifies the bond strength, with larger absolute values indicating stronger bonds.51–53
In the Cu12In12Cd8Te32 compound, Cd–Te exhibits smaller ICOHP values (0.48 eV) than Cu–Te (0.60 eV) and In–Te (1.73 eV), evidencing weaker Cd–Te bonds. Such weakened bonding interactions are consistent with the experimentally observed reduction in sound velocity, reflecting pronounced lattice softening upon Cd incorporation. Notably, the monotonic decrease in sound velocity shows a similar compositional trend to the evolution of the anion displacement parameter u, highlighting the close correspondence between local structural evolution and the observed lattice softening.
In addition, Vickers hardness (HV) measurements (Fig. S9) offer supplementary mechanical insight into how Cd alloying is related to the structural rigidity of the samples. At low Cd contents (x ≤ 0.3), the hardness remains at a relatively high level and shows a slight increase, which may be associated with solid–solution strengthening, and the impact of Cd incorporation on the overall framework rigidity in this regime appears to be limited. At higher Cd contents (x > 0.3), the hardness shows a noticeable decrease. This trend is broadly consistent with the weakened Cd–Te bonding indicated by the ICOHP analysis and the reduction in sound velocity.
Collectively, these findings reveal that the pronounced reduction in κlat of CdTe-alloyed CuInTe2 arises from the cooperative effects of enhanced point defect scattering, induced by substitutional and antisite defects, together with lattice softening reflected by the sound velocity reduction and local structural evolution.
2.4 Electrical transport properties and figure of merit ZT
Fig. 5 presents the temperature-dependent electrical transport properties of (CuInTe2)1−x(2CdTe)x. The electrical conductivity (σ) initially increases and then decreases with Cd incorporation. All alloyed samples evolve from the intrinsic semiconducting behavior of the pristine compound to degenerate-semiconductor behavior, indicating a heavily doped state with high carrier concentration. At room temperature, σ increases from 22 S cm−1 for x = 0 to 251 S cm−1 for x = 0.1, and remains above 150 S cm−1 throughout the tetragonal composition range (x < 0.25). With further Cd addition, σ drops markedly, reaching 0.7 S cm−1 at x = 0.5. Conversely, the Seebeck coefficient (S) exhibits the opposite trend, decreasing from 424 µV K−1 (x = 0) to 132 µV K−1 (x = 0.1), staying nearly constant (∼150 µV K−1) in the tetragonal regime, and increasing to 523 µV K−1 for the x = 0.5 sample. This inverse correlation between S and σ is governed by carrier concentration modulation. Consequently, the power factor (PF) decreases monotonically with increasing Cd content.To elucidate the origin of the electrical transport behavior in (CuInTe2)1−x(2CdTe)x, carrier concentration (pH) and Hall mobility (µH) were analyzed (Fig. 6a). With increasing Cd content, pH initially increaes and then declines, while µH decreases monotonically. The sharp increase in pH (up to ∼5 × 1019 cm−3) at low Cd concentrations arises from defect-controlled carrier generation. Defect formation energy calculations indicate that both CdCu and CdIn substitutional defects can form, but CdIn is energetically more favorable, leading Cd to preferentially substitute In sites and generate acceptor-like defects that increase the hole concentration. At higher Cd contents, Cd also occupies Cu sites to form donor-like CdCu defects, which compensate for holes. Simultaneously, Cd incorporation markedly lowers the formation energy of CuIn antisite defects, creating additional donor-type centers that further neutralize holes. The concurrent increase in Cu-vacancy formation energy reduces intrinsic hole generation. These combined effects explain the subsequent decrease in pH with increasing Cd content. At room temperature, the Hall mobility decreases markedly from 89.5 cm2 V−1 s−1 for x = 0 to 27.9, 28.6, 18.1, and 17.4 cm2 V−1 s−1 for x = 0.05, 0.1, 0.15, and 0.2, respectively. With further Cd incorporation, the Hall mobility is reduced to an extremely low value of 0.6 cm2 V−1 s−1 at x = 0.5, primarily due to enhanced carrier–carrier scattering and disorder scattering induced by CdTe alloying.
As shown in Fig. 6b, the density-of-states effective mass 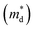 was estimated from the Seebeck coefficient and carrier concentration relationship:54–56
was estimated from the Seebeck coefficient and carrier concentration relationship:54–56
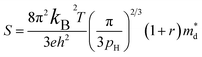 | (2) |
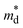 increases to approximately 0.7 me for all compositions and remains nearly constant across the solid-solution series. This finding indicates that the enhancement of the Seebeck coefficient originates primarily from the increased
increases to approximately 0.7 me for all compositions and remains nearly constant across the solid-solution series. This finding indicates that the enhancement of the Seebeck coefficient originates primarily from the increased 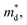 whereas the electronic band structure remains essentially unchanged with Cd incorporation.
whereas the electronic band structure remains essentially unchanged with Cd incorporation.
Temperature dependence of carrier concentration and Hall mobility is shown in Fig. 6c and d. The pH remains nearly constant over a wide temperature range for CdTe-alloyed samples, mainly due to their relatively high initial carrier concentration. This behavior aligns with our previous study,57 where it was shown to be governed by the position of the Fermi level and the temperature-dependent variation of the Cu vacancy formation energy. In contrast, (CuInTe2)1−x(2CdTe)x samples follow the µH–T−1 relationship, indicating a mixed scattering mechanism dominated by alloy scattering and acoustic phonon scattering. With increasing temperature, scattering strengthens further and mobility degrades accordingly.
Integrating the effects of CdTe alloying on the thermal and electrical transport, the temperature dependence of the thermoelectric figure of merit for (CuInTe2)1−x(2CdTe)x is presented in Fig. 6e and f. Within the tetragonal phase region, a modest improvement is achieved for the x = 0.1 sample, where ZT reaches 0.75 at 873 K. In the cubic phase compositions, despite the substantial reduction in κlat, severe carrier scattering induced by highly disordered cation sublattices deteriorates electrical transport, resulting in lower ZT values. These findings indicate that complete cation disorder does not represent the optimal configuration for maximizing thermoelectric performance in the CuInTe2 system. Rather, maintaining a moderate level of structural disorder that balances phonon scattering and carrier mobility enables a more favorable trade-off between electrical and thermal transport, thereby improving the overall thermoelectric performance.
3. Conclusion
In summary, we demonstrate that CdTe alloying effectively enhances the cation-sublattice disorder within the CuInTe2 system, driving a structural transformation from an ordered tetragonal lattice to a fully disordered cubic framework. Combined theoretical and experimental analyses reveal that the formation of Cd substitutional and Cu–In antisite defects induces strong point defect scattering, while the intrinsically weaker Cd–Te bonds promote lattice softening. Together, these effects suppress lattice thermal conductivity to 0.44 W m−1 K−1 at 873 K, approaching the amorphous limit. Meanwhile, severe carrier scattering induced by extensive cation disorder limits electrical transport, whereas compositions with moderate disorder achieve a more favorable phonon–carrier trade-off, yielding a peak ZT of 0.75 at 873 K in (CuInTe2)0.9(2CdTe)0.1. Although excessive disorder degrades carrier mobility, maintaining an optimal level of disorder achieves a favorable phonon–carrier balance. These results link atomic-scale disorder to macroscopic transport, underscoring the pivotal role of cation disorder in suppressing lattice thermal conductivity and offering a practical route to optimize thermal transport in thermoelectric materials that inherently possess high lattice thermal conductivity.4. Experimental section
4.1 Material synthesis
High-purity elemental Cu (99.999%), In (99.999%), Cd (99.999%), and Te (99.9999%) were weighed according to the nominal compositions (CuInTe2)1−x(2CdTe)x with x = 0, 0.05, 0.1, 0.15, 0.2, 0.25, 0.3, 1/3, 0.4, and 0.5. The mixtures were loaded into quartz ampoules, evacuated to high vacuum and flame-sealed, and then heated to 1273 K at 100 K h−1. After a 24 h dwell to ensure complete melting and homogenization, the melts were quenched in cold water. The solidified charges were subsequently brought to 823 K (same rate), annealed for 24 h, and cooled to ambient temperature. The ingots were pulverized in an agate mortar to obtain fine powders. Dense bulk specimens were then consolidated from these powders by spark plasma sintering at 773 K and 50 MPa for 5 min using a graphite die with an inner diameter of 13 mm under a vacuum below 20 Pa.4.2 Structural and compositional characterization
The phase compositions of the sintered samples were examined using powder X-ray diffraction (XRD, Empyrean; PANalytical) with Cu Kα radiation operated at 40 kV and 40 mA. Diffraction data were recorded within a 2θ range of 10°–90° at a step interval of 0.026°. The diffraction patterns were refined by the Rietveld method using the FullProf program to obtain the structural parameters. An electron probe microanalyzer (EPMA; JXA-8530F Plus; JEOL) equipped with an energy-dispersive X-ray spectrometer was used to acquire back-scattered electron images and elemental distribution mappings.4.3 Transport property measurements
The electrical conductivity and Seebeck coefficient were simultaneously tested using a four-probe system (CTA-3, Cryoall) in a helium atmosphere from 300 to 873 K using rectangular bar-shaped samples with typical dimensions of about 10 × 3 × 3 mm3. Thermal diffusivity (D) was evaluated by the Laser Flash Apparatus (LFA-467, Netzsch) under nitrogen over the same temperature range on disk-shaped samples with a diameter of approximately 12.7 mm and a thickness of ∼1.2 mm. The total thermal conductivity was obtained from the relation κtot = DρCp, where ρ represents the density determined via the Archimedes method, and Cp denotes the specific heat capacity estimated by using the Dulong–Petit law. The Hall coefficient (RH) was acquired using the van der Pauw configuration (Lakeshore-8404, Lakeshore) in an argon atmosphere between 300 and 773 K on square plate-shaped samples with lateral dimensions of approximately 10 × 10 mm2 and a thickness below 1 mm. Carrier concentration (pH = 1/eRH) and Hall mobility (µH = σRH) were calculated from the measured values.4.4 Physical property measurements
The ultrasonic pulse-echo method was used to determine the longitudinal and transverse sound velocities of the samples. The Vickers hardness (HV) was measured using a microhardness tester (Falcon 401AM, Innovatest Europe BV, Netherlands) under a load of 9.81 N with a dwell time of 15 s. The reported hardness values represent the average of at least five indentations.4.5 Theoretical calculations
All first-principles calculations presented in this paper were performed using density functional theory implemented in the Vienna Ab initio Simulation Package (VASP),58,59 with the projector augmented wave (PAW) method60 and the Perdew–Burke–Ernzerhof (PBE) functional within the generalized gradient approximation (GGA) for exchange–correlation interactions.61 For calculation of the primitive cell, the Brillouin zone was sampled using a 5 × 5 × 5 k-point grid generated by the Monkhorst–Pack scheme. The cut off energy was set to 500 eV, and all the atomic positions were fully relaxed until reaching a convergence criterion of 10−6 eV. Meanwhile, the force convergence criterion was set to 0.01 eV Å−1. To obtain the accurate defect energy, a 2 × 2 × 2 supercell was constructed using the VASPKIT code62 to ensure sufficient distance between defects. The defect formation energy calculation formula can be written as follows: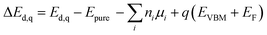 | (3) |
Author contributions
Gangjian Tan: funding acquisition, conceptualization, methodology, writing–review & editing and project supervision. Qingjie Zhang and Xinfeng Tang: resource provision and project supervision. Songlin Li: material synthesis, thermoelectric performance characterization, data curation, writing–original draft and manuscript revision. Jianxiang Zhai: theoretical calculations, data curation, manuscript revision and writing–review & editing. Chenghao Xie and Minghao Ye: theoretical calculations and manuscript revision. Junxi Mei and Guoqing Ding: data validation and manuscript revision.Conflicts of interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.Data availability
All data supporting the findings of this work are provided in the article and the supporting information (SI). Supplementary information is available. See DOI: https://doi.org/10.1039/d5ta09149g.Acknowledgements
We would like to acknowledge the financial support from the National Natural Science Foundation of China (Grant No. 52371235 and 52171221). The DFT calculations were performed at the Hefei advanced computing center.References
- X. Tang, Z. Li, W. Liu, Q. Zhang and C. Uher, Interdiscip. Mater., 2022, 1, 88–115 Search PubMed.
- Y. Qin, P. Qiu, R. Liu, Y. Li, F. Hao, T. Zhang, D. Ren, X. Shi and L. Chen, J. Mater. Chem. A, 2016, 4, 1277–1289 Search PubMed.
- D. Yang, Y. Xing, J. Wang, K. Hu, Y. Xiao, K. Tang, J. Lyu, J. Li, Y. Liu, P. Zhou, Y. Yu, Y. Yan and X. Tang, Interdiscip. Mater., 2024, 3, 326–337 CAS.
- G. Tan, F. Shi, H. Sun, L.-D. Zhao, C. Uher, V. P. Dravid and M. G. Kanatzidis, J. Mater. Chem. A, 2014, 2, 20849–20854 RSC.
- S. Luo, J. Liang, Z. Wei, Y. Du, L. Lv, Y. Jiang, S. Zheng, W. Song and Z. Zhang, J. Mater. Chem. A, 2025, 13, 4380–4389 Search PubMed.
- Y. Huang, T. Lyu, M. Zeng, M. Wang, Y. Yu, C. Zhang, F. Liu, M. Hong and L. Hu, Interdiscip. Mater., 2024, 3, 607–620 Search PubMed.
- H. Han, L. Zhao, X. Wu, B. Zuo, S. Bian, T. Li, X. Liu, Y. Jiang, C. Chen, J. Bi, J. Xu and L. Yu, J. Mater. Chem. A, 2024, 12, 24041–24083 RSC.
- C. Xie, Y. Fan, Z. Liu, M. Ye, J. Liu, G. Ding, J. Mei, Q. Zhang, X. Tang and G. Tan, Chem. Mater., 2025, 37, 4917–4924 CrossRef CAS.
- G. Tan, S. Hao, S. Cai, T. P. Bailey, Z. Luo, I. Hadar, C. Uher, V. P. Dravid, C. Wolverton and M. G. Kanatzidis, J. Am. Chem. Soc., 2019, 141, 4480–4486 CrossRef CAS PubMed.
- J. Sun, C. Xie, W. Cui, F. Yan, J. Li, J. Zhang, X. Sang, X. Tang and G. Tan, Chem. Mater., 2023, 35, 4366–4374 CrossRef CAS.
- J. Cui, C. Xie, W. Hu, H. Luo, Q. Mei, S. Li, W. Xu, Z. Gao, J. Wu, Q. Zhang, X. Tang and G. Tan, ACS Appl. Mater. Interfaces, 2024, 16, 39656–39663 CrossRef CAS PubMed.
- S. Acharya, J. Hwang, K. Kim, J. Kim, W. Hwang, A. Soon and W. Kim, Nano Energy, 2023, 112, 108493 CrossRef CAS.
- P. Ganesan, C. S. Gantepogu, S. Duraisamy, P. Wu, G.-T. Huang, M. Y. Fakhri, K.-H. Chen, Y.-Y. Chen and M.-K. Wu, J. Mater. Chem. A, 2025, 13, 8559–8570 RSC.
- J. Sun, R. Wang, W. Cui, S. Xie, T. Luo, H. Bai, X. Zhao, Z. Chen, X. Sang, X. Tan, X. Tang and G. Tan, Chem. Mater., 2022, 34, 6450–6459 CrossRef CAS.
- K. Suekuni, Y. Shimizu, E. Nishibori, H. Kasai, H. Saito, D. Yoshimoto, K. Hashikuni, Y. Bouyrie, R. Chetty, M. Ohta, E. Guilmeau, T. Takabatake, K. Watanabe and M. Ohtaki, J. Mater. Chem. A, 2019, 7, 228–235 RSC.
- Q. Mei, C. Liao, W. Xu, J. Cui, S. Li, J. Liu, G. Ding, J. Mei, X. Tang and G. Tan, Mater. Today Energy, 2025, 53, 101972 CrossRef CAS.
- J. Sun, X. Su, Y. Yan, W. Liu, G. Tan and X. Tang, ACS Appl. Energy Mater., 2020, 3, 2–8 CrossRef CAS.
- C. Xie, R. Li, W. Xu, B. Jin, X. Shi, X. Tang, G. Tan and Z. Chen, Adv. Funct. Mater., 2025, 35, 2421545 CrossRef CAS.
- G. Tan, L.-D. Zhao and M. G. Kanatzidis, Chem. Rev., 2016, 116, 12123–12149 CrossRef CAS PubMed.
- H. Wu, P. Chen, Z. Zhou, D. Zhang, X. Gong, B. Zhang, Y. Zhou, K. Peng, Y. Yan, G. Wang, J. Liu, D. Li, G. Han, G. Wang, X. Lu and X. Zhou, J. Mater. Sci. Technol., 2023, 154, 140–148 Search PubMed.
- E. Yang, G. Li, Z. Zhang, X. Shi, Y. Yan, M. Huang, Z. Chen, H. Kang, Z. Chen, E. Guo and T. Wang, ACS Appl. Electron. Mater., 2024, 6, 8116–8123 CrossRef CAS.
- D. Zhang, B. Zhang, Z. Zhou, K. Peng, H. Wu, H. Wang, G. Wang, G. Han, G. Wang, X. Zhou and X. Lu, Chem. Mater., 2021, 33, 9795–9802 CrossRef CAS.
- Z. Liu, T. Hong, L. Xu, S. Wang, X. Gao, C. Chang, X. Ding, Y. Xiao and L. Zhao, Interdiscip. Mater., 2023, 2, 161–170 Search PubMed.
- H. Wang, A. D. LaLonde, Y. Pei and G. J. Snyder, Adv. Funct. Mater., 2013, 23, 1586–1596 Search PubMed.
- J. Callaway and H. C. Von Baeyer, Phys. Rev., 1960, 120, 1149–1154 Search PubMed.
- P. G. Klemens, Phys. Rev., 1960, 119, 507–509 Search PubMed.
- Z. Feng, J. Zhang, Y. Yan, G. Zhang, C. Wang, C. Peng, F. Ren, Y. Wang and Z. Cheng, Sci. Rep., 2017, 7, 2572 CrossRef PubMed.
- S. Zhan, Y. Wen, B. Qin, S. Bai, Y. Qiu, S. Wang, T. Hong, D. Liu, L. Zheng, X. Gao, Y. Zhu, Q. Tan and L. Zhao, Adv. Energy Mater., 2025, 15, 2403194 CrossRef CAS.
- J. Jung, N. Ha and W. Yang, ACS Appl. Mater. Interfaces, 2025, 17, 16410–16420 CrossRef CAS PubMed.
- J. Ma, O. Delaire, A. F. May, C. E. Carlton, M. A. McGuire, L. H. VanBebber, D. L. Abernathy, G. Ehlers, T. Hong, A. Huq, W. Tian, V. M. Keppens, Y. Shao-Horn and B. C. Sales, Nat. Nanotechnol., 2013, 8, 445–451 CrossRef CAS PubMed.
- S. Roychowdhury, T. Ghosh, R. Arora, M. Samanta, L. Xie, N. K. Singh, A. Soni, J. He, U. V. Waghmare and K. Biswas, Science, 2021, 371, 722–727 Search PubMed.
- F. X. Zhang, C. Feng, Z. Guan, H. Song, S. Nan, Y. Tang, H. Yang, K. Zeng, X. Yuan, M. Xiao, Z. Lu, Y. Zhang, D. Ma and Y. Zhang, Acta Mater., 2025, 292, 121056 CrossRef.
- B. R. Ortiz, W. Peng, L. C. Gomes, P. Gorai, T. Zhu, D. M. Smiadak, G. J. Snyder, V. Stevanović, E. Ertekin, A. Zevalkink and E. S. Toberer, Chem. Mater., 2018, 30, 3395–3409 CrossRef CAS.
- R. Liu, L. Xi, H. Liu, X. Shi, W. Zhang and L. Chen, Chem. Commun., 2012, 48, 3818 RSC.
- H. Kim, S. K. Kihoi and H. S. Lee, J. Alloys Compd., 2021, 869, 159381 CrossRef CAS.
- H. Xie, L. Zhao and M. G. Kanatzidis, Interdiscip. Mater., 2024, 3, 5–28 CAS.
- P. Grima, M. Quintero, L. Nieves, H. Cabrera, E. Perez, I. Zumeta, J. Aitken and J. Brant, Rev. Latinoam. Metal. Mater., 2018, 38, 53–63 Search PubMed.
- J. M. Adamczyk, L. C. Gomes, J. Qu, G. A. Rome, S. M. Baumann, E. Ertekin and E. S. Toberer, Chem. Mater., 2021, 33, 359–369 Search PubMed.
- J. Yang, S. Chen, Z. Du, X. Liu and J. Cui, Dalton Trans., 2014, 43, 15228–15236 RSC.
- D. G. Cahill, S. K. Watson and R. O. Pohl, Phys. Rev. B: Condens. Matter Mater. Phys., 1992, 46, 6131–6140 CrossRef CAS.
- J. Zhang, R. Liu, N. Cheng, Y. Zhang, J. Yang, C. Uher, X. Shi, L. Chen and W. Zhang, Adv. Mater., 2014, 26, 3848–3853 Search PubMed.
- Y. Lu, S. Chen, W. Wu, Z. Du, Y. Chao and J. Cui, Sci. Rep., 2017, 7, 40224 Search PubMed.
- Y. Megdoud, Y. Benkrima, R. Meneceur, L. Tairi, A. Lakel, S. Ghemid and H. Meradji, East Eur. J. Phys., 2023, 231–243 Search PubMed.
- H.-S. Kim, Z. M. Gibbs, Y. Tang, H. Wang and G. J. Snyder, APL Mater., 2015, 3, 041506 CrossRef.
- S. Li, J. Sun, Y. Zhang, Y. Fan, X. Tang and G. Tan, Z. Anorg. Allg. Chem, 2022, 648, e202200038 CrossRef CAS.
- Y. Zhang, J. Li, W. Hu, X. Yang, X. Tang and G. Tan, Mater. Today Energy, 2022, 25, 100958 CrossRef CAS.
- H. Xie, Z. Li, Y. Liu, Y. Zhang, C. Uher, V. P. Dravid, C. Wolverton and M. G. Kanatzidis, J. Am. Chem. Soc., 2023, 145, 3211–3220 Search PubMed.
- W. Xu, W. Guo, L. Liao, C. Xie, J. Cui, Q. Mei, S. Li, J. Wu, Z. Luo, Q. Zhang, X. Tang and G. Tan, J. Am. Chem. Soc., 2025, 147, 34804–34812 Search PubMed.
- Q. Mei, C. Xie, J. Liu, Y. Wang, J. Cui, L. Liao, C. Liao, W. Xu, S. Li, Q. Zhang, X. Tang and G. Tan, Adv. Energy Mater., 2025, 15, 2500937 CrossRef CAS.
- E. S. Toberer, A. Zevalkink and G. J. Snyder, J. Mater. Chem., 2011, 21, 15843 RSC.
- V. L. Deringer, A. L. Tchougréeff and R. Dronskowski, J. Phys. Chem. A, 2011, 115, 5461–5466 CrossRef CAS.
- J. Cui, W. Xu, L. Liao, J. Cheng, S. Li, Q. Mei, C. Xie, C. Liao, J. Wu, Q. Zhang, X. Tang and G. Tan, ACS Appl. Mater. Interfaces, 2025, 17, 5133–5140 CrossRef CAS.
- H. Xie, E. S. Bozin, Z. Li, M. Abeykoon, S. Banerjee, J. P. Male, G. J. Snyder, C. Wolverton, S. J. L. Billinge and M. G. Kanatzidis, Adv. Mater., 2022, 34, 2202255 CrossRef CAS.
- M. Cutler, J. F. Leavy and R. L. Fitzpatrick, Phys. Rev., 1964, 133, A1143–A1152 CrossRef.
- H. Xie, Y. Liu, Y. Zhang, S. Hao, Z. Li, M. Cheng, S. Cai, G. J. Snyder, C. Wolverton, C. Uher, V. P. Dravid and M. G. Kanatzidis, J. Am. Chem. Soc., 2022, 144, 9113–9125 Search PubMed.
- H. Zhai, Y. Xiao, L.-D. Zhao, G. Tan and X. Tang, J. Alloys Compd., 2020, 826, 154154 CrossRef CAS.
- S. Li, C. Xie, K. Liu, J. Liu, J. Mei, G. Ding, J. Cui, W. Xu, C. Cao, Q. Zhang, X. Tang and G. Tan, J. Mater. Chem. A, 2025, 13, 27691–27700 RSC.
- G. Kresse and J. Hafner, Phys. Rev. B:Condens. Matter Mater. Phys., 1993, 48, 13115–13118 CrossRef CAS.
- G. Kresse and J. Furthmüller, Phys. Rev. B: Condens. Matter Mater. Phys., 1996, 54, 11169–11186 CrossRef CAS PubMed.
- P. E. Blöchl, Phys. Rev. B: Condens. Matter Mater. Phys., 1994, 50, 17953–17979 CrossRef.
- J. P. Perdew, K. Burke and M. Ernzerhof, Phys. Rev. Lett., 1996, 77, 3865–3868 CrossRef CAS PubMed.
- V. Wang, N. Xu, J. C. Liu, G. Tang and W.-T. Geng, Comput. Phys. Commun., 2021, 267, 108033 CrossRef CAS.
- S. Maintz, V. L. Deringer, A. L. Tchougréeff and R. Dronskowski, J. Comput. Chem., 2016, 37, 1030–1035 CrossRef CAS PubMed.
Footnote |
| † Songlin Li and Jianxiang Zhai contributed equally to this work. |
| This journal is © The Royal Society of Chemistry 2026 |