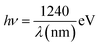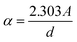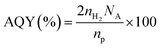Synergistic role of self-induced CdIn2S4 nanopyramid sacrificial layer for the enhanced photocatalytic activity of TiO2 nanotube-based chalcogenide heterostructures
Nilem
Khaliq
abc,
Habib
Ullah
 d,
Muhammad Asim
Rasheed
d,
Muhammad Asim
Rasheed
 *a,
Yi
Xie
*a,
Yi
Xie
 d,
Maaz
Khan
d,
Maaz
Khan
 e,
Muhammad
Imran
*f,
Patrik
Schmuki
e,
Muhammad
Imran
*f,
Patrik
Schmuki
 g and
Ghafar
Ali
g and
Ghafar
Ali
 *e
*e
aDepartment of Physics and Applied Mathematics, Pakistan Institute of Engineering and Applied Sciences (PIEAS), Islamabad 45650, Pakistan
bDepartment of Physics, Women University Swabi, KPK, Pakistan
cDepartment of Physics, University of Lakki Marwat, KPK, Pakistan
dState Key Laboratory of Silicate Materials for Architectures, Wuhan University of Technology, Wuhan 430070, P.R. China
eNanomaterials Research Group (NRG), Physics Division, PINSTECH, Islamabad 45650, Pakistan. E-mail: ghafarali@kaist.ac.kr; ghafarali@gmail.com
fDepartment of Physics, Faculty of Science, Islamic University of Madinah, Madinah 42351, Saudi Arabia. E-mail: masimr22@gmail.com; imranskku@gmail.com
gDepartment of Materials Science and Engineering, University of Erlangen-Nuremberg, Martensstrasse 7, D-91058 Erlangen, Germany
First published on 8th December 2025
Abstract
A simple treatment was employed to obtain a novel design of a highly stable visible light responsive In2S3/CdS/TiO2 nanotube-based photocatalyst for H2 production. At first, In2S3/CdS/TiO2 nanotubes were fabricated by incorporating CdS and In2S3 quantum dots (QDs) onto TiO2 nanotubes (TNTs) via a chemical bath deposition method. In the second step, a portion of the QDs layer (CdS and In2S3) was converted into a layer of CdIn2S4 pyramid-like structure by thermal treatment of In2S3/CdS/TNTs in an Ar environment (Ar-In2S3/CdS/TNTs). The photocatalytic performances of the different heterostructure assemblies (simple and Ar-treated samples) were tested. The incorporation of CdS and In2S3 QDs significantly improved the visible response, showing higher photocurrent density and H2 production rate (0.5694 mL h−1 cm−2) values. Furthermore, the outer layer of CdIn2S4 improved the photostability of In2S3/CdS/TNTs and further boosted the H2 evolution rate (0.6281 mL h−1 cm−2). The apparent quantum yields (AQYs) of Ar-In2S3/CdS/TNTs at 450 nm and 632 nm LED were 5.54% and 13.57%, respectively. The higher photocurrent density and lower charge transfer resistance suggest that the structure of the Ar-In2S3/CdS/TNTs photoanodes provides efficient charge carrier separation and interfacial charge transport properties. Dye degradation of methylene blue using Ar-In2S3/CdS/TNTs showed complete degradation of the dye within 22 min, which was 5 minutes lesser than that observed for In2S3/CdS/TNTs.
Introduction
The dearth of fossil fuel-based energy resources has led the world to seek alternative ways to harvest energy. Since the ground-breaking work of Fujishima and Honda on water splitting using titanium dioxide (TiO2),1 photocatalysis has gained much interest for harnessing solar energy to produce abundant and environmentally friendly hydrogen fuel. Semiconductor-mediated photocatalysis offers remarkable potential to produce hydrogen due to their suitable band structures. To date, in addition to TiO2, zinc oxide (ZnO) and tungsten oxide (WO3) have been the most extensively explored photocatalyst materials.2–6 However, TiO2-based photocatalysts offer advantages over others due to their corrosion resistance, biocompatibility and higher chemical stability.7–11 Furthermore, the one-dimensional (1D) morphology of TiO2 (e.g., TiO2 nanotubes (TNTs)) offers additional benefits over its bulk counterpart. For instance, the diffusion length of the photogenerated charge carriers is increased because the grain boundary effects, which typically act as recombination centers, are minimized due to the 1D, highly ordered structure of the nanotubes. The photogenerated electron–hole pair recombination is also prevented due to alternative charge transport pathways and differences in the diffusion lengths of electrons and holes.12,13 The hollow interior of the nanotubes boosts photoharvesting due to multi-light scattering and encourages surface-activated redox reactions on both sides of the walls. Despite all these benefits, the photocatalytic activity of simple TNTs is not significant due to their large band gap of ∼3.2 eV. This limits the light absorption by TNTs to the UV range in the solar spectrum. To overcome this issue, co-sensitization of TNTs with a photoactive material with a suitable band gap and position is expected to produce the desired results.Metal chalcogenides, such as CdS, have been seen as promising visible light responsive materials for coupling with TNTs. CdS offers complementary band edge positions for proton reduction. However, it tends to easily photo-corrode in aqueous electrolytes due to the capture of photogenerated holes. The photo-corrosion property of CdS hinders its extensive utilization for photocatalytic hydrogen generation and degradation of water contaminants. To resolve this problem, a corrosion-resistant passivation layer covering the sulfide surface was applied to prevent direct contact with the electrolyte. Efforts have also been made to facilitate hole transfer away from the sulfide surface. In some cases, a thin polymer coating has been used as a passivating layer to prevent photo-corrosion by shielding the sulfide nanostructures.14–17 However, the thickness of the passivation layer is the critical parameter. Furthermore, such a layer reduces the available active sites for photocatalysis, resulting in inferior efficiency. Meanwhile, the utilization of CdS in heterostructures with other semiconductors can significantly minimize these issues. CdS-based heterostructures have been synthesized by introducing metals and low band gap semiconductors with relatively higher chemical stability and cascade band alignment to reduce the photo-corrosion problem of CdS.18–20 Materials, such as CdS/rGO/MoS2, CdS/ZnIn2S4 core–shell, AgVO3/ZnIn2S4 and TiO2 nanowire-supported In2S3, have been reported. In such hetero-structures, easy hole transfer kinetics is achieved to prevent hole accumulation at the sulfide surface.21–24 CdS-based hetero-structures have been reported to show improved stability, with a subsequent increase in H2 production.18,21,25–27 The established potential gradient among the nanostructure interfaces of the two functional components of the heterostructure causes the rapid transfer of charge carriers. This, in turn, decreases the possibility of carrier recombination in such hybrid materials.28–31 Recently, sulfur vacancy-chalcogenide/Ti3C2 Schottky junctions have been studied to achieve a giant built-in electric field (BIEF) via defect-mediated heterocomponent anchorage.32 This technique yields a 13.34-fold higher efficiency than that of unmodulated chalcogenides. Other studies show that inclusion of In2S3 in CdS-based heterostructures is highly advantageous owing to its compatible band alignment. In addition, In2S3 is a more stable and environmentally friendly narrow band gap semiconductor compared to CdS.33–37 Ternary heterostructures based on TNTs can be utilized for achieving enhanced photocatalytic activity and improved photo-corrosion resistance of CdS. Moreover, the cascade bandgap positions of TNTs with CdS and In2S3 may also lead to a well-separated electron–hole pair.
There is a need to devise synthesis strategies to overcome the problem of photo-corrosion and increase the functional characteristics of sulfide-based photocatalysts.38 Due to the relatively unstable nature of binary chalcogenides, their conversion into highly stable ternary chalcogenides by chemical or physical means is highly useful to deal with the photo-corrosion issues.39,40 Recently, enhanced photo-conduction and photocatalytic efficiency were reported for In2S3 nanosheets doped with Cd and Zn.41 It would be interesting to fabricate a passivation layer composed of CdIn2S4, which may favor photogenerated charge separation and increase the stability of the sulfide layer due to its hole-capturing ability.
In this work, a novel design for a highly stable visible light-responsive ternary hetero-structure (In2S3/CdS/TNTs) was explored. First, a ternary hetero-structure based on TNTs incorporating CdS and In2S3 QDs was fabricated. The photocatalytic performance of the different hetero-structure assemblies (e.g., TNTs, CdS/TNTs, In2S3/TNTs, and In2S3/CdS/TNTs) was investigated. The synergistic effect of CdS and In2S3 QDs dramatically increases the visible response of the hetero-structure electrode. Furthermore, a simple treatment procedure was utilized to improve its photostability by introducing an outer sacrificial layer of CdIn2S4. The passivation layer composed of CdIn2S4 enhanced the H2 evaluation rate. In addition, the argon-treated hetero-structure (Ar-In2S3/CdS/TNTs) shows further enhancement in photocatalytic activity, as evidenced by the degradation of methylene blue (MB).
Materials and methods
Titanium (Ti) sheets (0.125 mm thickness and 99.6% purity) were acquired from ADVENT Research Materials Ltd, England. Ethylene glycol (EG, 99.9% purity), ammonium fluoride (NH4F), cadmium chloride (CdCl2, 99.99%), sodium sulfide (Na2S, 99.5%), indium fluoride (InF3) and methylene blue were obtained from Sigma-Aldrich, USA. Deionized (DI) water was used as per the requirements of the synthesis and photocatalytic experiments.Anodization of Ti
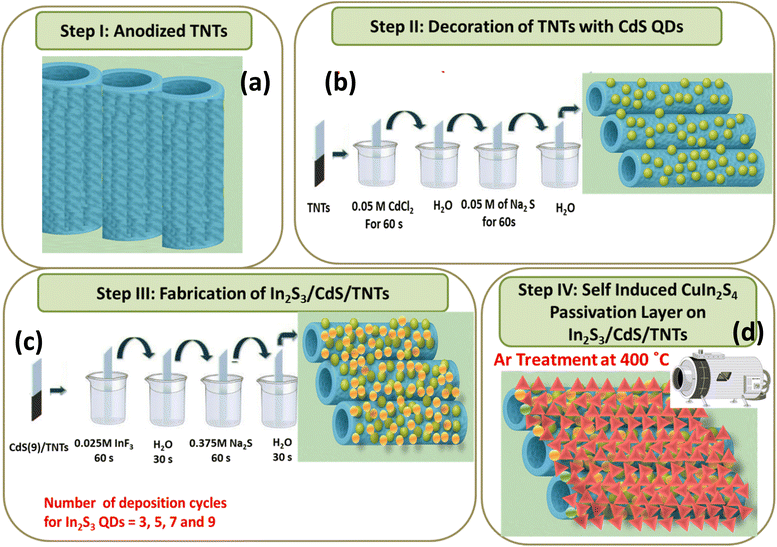
Commercial Ti sheets were cleaned with acetone, ethanol, and DI water successively and dried under a nitrogen (N2) stream. Anodization was performed in a two-electrode anodization cell (Ti sheet served as the anode and Pt sheet as the cathode) using a high-voltage potentiostat (Jaissle IMP 88 PC, Germany). Ti sheets were anodized at 50 V for 4 h in EG containing 0.25 wt% NH4F and 5 wt% DI water. The digital images of different stages of the sample are provided in Fig. S1(a–g). The TNTs grown on the Ti sheet were then removed by sonication in DI water for 15 min, resulting in a honeycomb-like patterned morphology on the surface (Digital image: Fig. S1b). The patterned Ti sheets were then used to obtain uniform arrays of nanotubes via the second step anodization under identical conditions to those used previously.42 After the second-step anodization, the samples were washed with ethanol and dried in a N2 stream (Digital image: Fig. S1c). The resulting amorphous TNTs were converted to anatase phase by annealing in air at 450 °C for 2 h, as shown in Scheme 1a (Digital image: Fig. S1d).Decoration of TNTs with CdS QDs
CdS QDs were decorated on TNTs using the conventional sonication-assisted successive ion layer adsorption and reaction (SILAR) method, as reported previously.43 Briefly, aqueous solutions of 0.05 M CdCl2 and 0.05 M Na2S were prepared separately. The annealed TNTs were sequentially immersed in both precursor solutions, each for 1 min, aided with a sonication process at room temperature. The excess amount of the precursors was removed after each step by washing with DI water. This four-step process (soaking and rinsing) is considered as one cycle, as shown in Scheme 1b (Digital image: Fig. S1e). This process was repeated for 3, 6, 9, and 12 cycles to increase the density of CdS QDs on TNTs, producing greenish-yellow samples. The samples were named as CdS(3) QDs/TNTs, CdS(6) QDs/TNTs, CdS(9) QDs/TNTs, and CdS(12) QDs/TNTs, corresponding to the number of deposition cycles.Fabrication of In2S3/CdS/TNTs photocatalysts
A layer of In2S3 QDS was deposited on CdS/TNTs using the facile sonication-assisted chemical bath deposition (CBD) method. The CdS deposition cycles were maintained at 9 cycles (CdS(9) QDs/TNTs). Briefly, the CdS/TNTs were sequentially dipped in a 0.025 M InF3 aqueous solution for 1 min, followed by rinsing with DI water for 30 s. The samples were then immersed in 0.375 M Na2S aqueous solution for another 1 min, followed by washing with DI water. The loading densities of In2S3 QDs were maintained by 3, 5, 7 and 9 deposition cycles, as shown in Scheme 1c (Digital image: Fig. S1f).Fabrication of CuIn2S4 passivation layer on In2S3/CdS/TNTs
The prepared In2S3/CdS/TNTs hetero-structure samples were annealed in an Ar environment at 400 °C for 1 h in order to obtain highly stable photo-electrodes (Digital image: Fig. S1g). Annealing induced a passivation layer of CdIn2S4 on In2S3/CdS/TNTs by thermal diffusion of Cd ions towards the In2S3 crystal structure. At first, CdS and In2S3 QDs form a compact thin layer, which subsequently changes into larger pyramid-like structures44 covering the nanotubes, as shown in Scheme 1d and can be seen from the TEM and SEM images (Fig. 3–5).Results and discussion
X-ray diffraction (XRD, X'pert Philips PMD, CuKα 1.54056 Å) was utilized for ascertaining the phase, purity, and crystal structure of the synthesized hetero-structures. Fig. 1a shows the XRD patterns of the pristine TNTs, CdS/TNTs, In2S3/TNTs, and In2S3/CdS/TNTs hetero-structures. The diffraction peaks for reflections from TNTs and CdS/TNTs crystal planes are indexed according to the tetragonal structure of anatase TiO2 (ICCD card # 01-071-1166) and CdS (ICCD card # 42-1411). The diffraction pattern of In2S3/TNTs shows the main reflection peaks at 23.33°, 27.43°, 33.24°, and 43.62° from (220), (311), (400) and (511) crystal planes of β-In2S3 (ICCD card # 01-084-1385), respectively, besides the characteristic reflections from anatase TiO2 (ref. 45) (Inset image). The qualitative analysis of the XRD data for the In2S3/CdS/TNTs hetero-structure shows all the characteristic peaks from its constituent materials without any significant changes in peak positions. The inset graph clearly shows the co-existence of reflections from both CdS and In2S3 crystal planes, designated by letters C and I, respectively, in their reflective planes, along with TNTs. | ||
| Fig. 1 XRD patterns of pristine TNTs, CdS/TNTS, In2S3/TNTs, and In2S3/CdS/TNTs (a) without and (b) with Ar-treatment at 400 °C. (Inset) (a) Shows the enlarge portion of XRD of In2S3/CdS/TNTs. | ||
Fig. 1b presents the XRD pattern of Ar-treated TNTs, CdS/TNTs, In2S3/TNTs, and In2S3/CdS/TNTs to determine any changes in the crystal structure and phase of the samples. XRD patterns of Ar-TNTs, Ar-CdS/TNTs, and Ar-In2S3/TNTs do not show any significant change in peak positions; however, the sharp peaks indicate improved crystallinity. An interesting result has emerged from the XRD measurement of the Ar-In2S3/CdS/TNTs heterojunction. Along with the crystalline phases of TNTs, a new diffraction peak appeared at 23.1° and 43.3°, corresponding to (220) and (511) related to CdIn2S4 at the expense of CdS. Moreover, the intensity of the apparent diffraction peaks of In2S3 at 23.3° and 27.4° (311) increases after thermal annealing of In2S3/CdS/TNTs in Ar as compared to the XRD pattern of Ar-In2S3/TNTs. This fact can be ascribed to the presence of cubic CdIn2S4 (ICCD card # 00-027-0060) sacrificial layer whose peaks lie in close proximity to those of In2S3 and thus overlap.
Morphological studies of various fabricated samples were conducted using a field emission scanning electron microscope (Hitachi FE-SEM S4800, Japan). Fig. 2 shows FESEM images of pristine TNTs, CdS/TNTs, In2S3/TNTs, and In2S3/CdS/TNTs systems. The top-view image of pristine TNTs (Fig. 2a) shows a regular array of nanotubes with open mouths, with an inner tube diameter of about 100 nm. The corresponding cross-sectional image (Fig. 2b) shows highly smooth tube walls. SEM images of CdS/TNTs (Fig. 2c and d) and In2S3/TNTs (Fig. 2e and f) show the homogeneous deposition of the CdS QDs or In2S3 QDs on the top as well as on the walls of the tube, respectively. The dimensions of CdS and In2S3 QDs estimated from SEM images are 5 nm and 4 nm, respectively. In the In2S3/CdS/TNTs heterogeneous system (Fig. 2g and h), more particles are deposited on the tube walls, but the top of the nanotubes remains open. It can also be seen (Fig. 2h) that the tube walls become rougher after co-sensitization of TNTs with CdS and In2S3 QDs. No significant increase in the QD size is observed for the co-sensitized system. This shows that no agglomeration occurs at these optimized experimental parameters, that is, the deposition cycle and solution concentration. The effect of different deposition cycles of In2S3 QDs on the morphology of In2S3/CdS/TNTs is presented in Fig. S2(a–h). The top and cross-sectional SEM images show the increased loading density of QDs on TNTs by increasing the deposition cycles from 3 to 7 (Fig. S2(a–f)). However, the QDs become agglomerated by further increasing the deposition cycles up to 9 (almost covering the nanotube's top as shown in Fig. S2g). Therefore, 7 cycles are considered an optimized value for the deposition cycle for In2S3 on In2S3/CdS/TNTs.
 | ||
| Fig. 2 FESEM top and cross-sectional images of (a and b) TNTs, (c and d) CdS/TNTs, (e and f) In2S3/TNTs, and (g and h) In2S3/CdS/TNTs hetero-structure. | ||
The argon (Ar) treatment of In2S3/CdS/TNTs heterogeneous structure was conducted in an Ar environment at 400 °C. The morphology of In2S3/CdS/TNTs was observed to change from uniformly deposited QDs on TNTs into randomly oriented three-dimensional pyramids aggregated on the surface of TNTs (Fig. 3a–h). To understand the formation process of CdIn2S4 pyramids on Ar-In2S3/CdS/TNTs (CdIn2S4/In2S3/CdS/TNTs), different loading densities of In2S3 QDs were utilized and controlled by the deposition cycles. FESEM images of Ar-treated In2S3/CdS/TNTs are presented in Fig. 3(a–h) for various deposition cycles of In2S3. For 3 cycles (Fig. 3(a and b)), after Ar annealing, a clearly rough surface appeared with a few small pyramids due to the insignificant amount of In2S3.
 | ||
| Fig. 3 FESEM top and cross-sectional images of Ar-In2S3/CdS/TNTs hetero-structures for (a and b) 3, (c and d) 5, (e and f) 7 and (g and f) 9 loading cycles of In2S3 on CdS/TNTs. | ||
By increasing the deposition cycles to 5 and 7 (Fig. 3(e and f)), the formation of interconnected pyramids increased and covered the whole TNTs surface. Any further increase in the loading density up to 9 cycles of In2S3 led to the deterioration of the pyramids into large agglomerates (Fig. 3g and h). This fact is attributed to the thermal diffusion of Cd from CdS into the In2S3 crystal structure, forming a new CdIn2S4 phase. Moreover, the loading density of In2S3 plays a vital role in the formation of these pyramids, which can be controlled by the deposition cycles. For comparison, Fig. S3 shows the cross-sectional FESEM images of Ar-annealed TNTs, CdS(9)/TNTs, and In2S3(7)/TNTs. Apart from their increased crystallinity as confirmed by XRD, the Ar-treatment brings no significant morphological change into the TNTs (Fig. S3a), CdS(9)/TNTs (Fig. S3b) and In2S3(7)/TNTs (Fig. S3c). Transmission electron microscopy (TEM) and high-resolution transmission electron microscopy (HRTEM) images of In2S3/CdS/TNTs and Ar-In In2S3/CdS/TNTs are presented in Fig. 4 and 5, respectively. Fig. 4a and b reveal hollow TiO2 nanotube (TNT) arrays with a diameter of about 100 nm and wall thickness of about 10–12 nm, which is in accordance with the FESEM findings. Fig. 4c and e show rough tube walls indicating the deposition of CdS and In2S3 on TNTs, which are uniformly decorated with CdS and In2S3 QDs, and appear as small spherical nanoparticles. The average sizes of CdS and In2S3 are 3 nm and 5 nm, respectively. Fig. 4d and f show the HRTEM images of In2S3/CdS/TNTs. The images clearly show well-defined lattice fringes. The interplanar spacing is determined using the ImageJ software and indexed in the figures. In Fig. 4d and f, the fringes corresponding to interplanar spacings of approximately 0.622 nm, 0.272 nm (attributed to (103) and (400) lattice planes of In2S3)46–48 and 0.358 nm, 0.336 nm (for (100) and (002) planes of CdS),49 confirm their existence and crystalline nature. The lattice fringes at 0.389 nm and 0.32 nm corresponding to the (220) and (311) crystal planes of CdIn2S4 are also observed, which indicate that the interdiffusion between Cd and In species occurred, leading to the formation of a homogeneous ternary CdIn2S4 phase on the TNTs surface after annealing in an Ar atmosphere.47,50,51 Moreover, the TNT walls remained crystalline, showing characteristic (101) planes with a spacing of ∼0.355 nm (Fig. 4d).52
Fig. 5(a–c) shows the TEM and HRTEM images of Ar-In2S3/CdS/TNTs. The TEM image in Fig. 5a shows a thin layer of CdS and In2S3 QDs on the TNT surface, indicating that the walls of TNTs are encapsulated by a thin layer of CdS and In2S3 QDS, forming a core-shell-like structure. In Fig. 5b, a continuous layer composed of deformed nano-pyramidal grains can be seen on the surface of TNTs, where the bottom of the nano-pyramids is interlaced with each other. The lattice fringes in the HRTEM image (Fig. 5c) show interplanar spacings of approximately 0.389 nm and 0.32 nm, which were indexed to the (220) and (311) crystal planes of CdIn2S4, respectively.
Energy-dispersive X-ray spectra (EDS) and elemental mapping of Ar-In2S3/CdS/TNTs further confirmed the structural evolution of the heterogeneous structure. Fig. 6a reveals the TEM image of the sample showing a rough surface between the walls of the nanotubes. This roughness of the walls can be due to the deposition of CdS and In2S3 QDS, along with a layer of deformed pyramids of CdIn2S4. The elemental mapping of the same image in Fig. (6b and c) shows Ti and O were localized within the tubular framework, while Fig. (6d–f) displays a homogeneous and overlapping distribution of Cd, In, and S elements along the nanotube walls, indicating core shell morphology along tube walls and interdiffusion of Cd into In2S3, leading to the formation of a non-uniform CdIn2S4 nano-pyramid layer. These observations confirm the partial transformation of CdS and In2S3 heterostructures into an outer layer of the crystalline CdIn2S4 phase with well-integrated interfaces on TNTs. To further validate the formation of CdIn2S4 after annealing In2S3/CdS/TNTs in the Ar environment, we obtained Raman spectra (LabRAM Odyssey, HORIBA France SAS, France) of the pristine TNTs, CdS/TNTs, In2S3/TNTs, In2S3/CdS/TNTs, and Ar-In2S3/CdS/TNTs, as shown in Fig. 6h. The Raman spectrum of the annealed TNTs displays five characteristic peaks at 143, 195, 398, 515, and 636 cm−1, corresponding to the Eg, B1g, A1g, and Eg modes of the anatase phase, respectively.50 The appearance of a sharp and broad peak at 297 cm−1 in CdS/TNTs indicates the presence of CdS QDs on the TNTs surface.53 Similarly, the In2S3/TNTs spectrum shows peaks at 306 and 933 cm−1, confirming the formation of an In2S3 layer on TNTs.46,48 For In2S3/CdS/TNTs, the peaks at 306 and 601 cm−1, along with those of TNTs, suggest the co-existence of In2S3 and CdS QDs. After the Ar annealing of In2S3/CdS/TNTs, two small but broad peaks appeared at 185 and 353 cm−1, attributed to the Eg and A1g modes, respectively, along with the main CdS and In2S3 peaks, confirming the appearance of CdIn2S4 on the In2S3/CdS/TNTs heterostructure.54,55 It is worth noting that the CdIn2S4/In2S3/CdS/TNTs electrode is referred to as Ar-In2S3/CdS/TNTs for comparison with other Ar-annealed samples, such as Ar-TNTs, Ar-CdS/TNTs, and Ar-In2S3/TNTs.
To investigate the surface composition and valence states of Ar-In2S3/CdS/TNTs, the XPS core level spectra of Ti 2p, O 1s, Cd 3d, In 3d, and S 2p were evaluated (Fig. 7(a–e)). The deconvoluted Ti 2p spectrum (Fig. 7a) exhibits a strong peak around 458.98 and 464.96 eV, which can be assigned to Ti 2p3/2 and Ti 2p1/2, respectively. These states demonstrate the presence of Ti4+ states in the sample. The high-resolution spectrum of oxygen is deconvoluted into two peaks, as shown in Fig. 7b. The main peak of O 1s at ∼529.8 eV can be assigned to the anionic oxygen in the TiO2 lattice. In addition, a small shoulder peak located at about 530.9 eV corresponds to the hydroxyl species. In Cd 3d spectra (Fig. 7c), the characteristic peaks located at 405.36 eV and 411.97 eV are attributed to Cd 3d5/2 and Cd 3d3/2, respectively, which verify the existence of Cd2+ in the system. In Fig. 7d (high scan spectrum of In 3d), the two peaks located at 445.16 eV and 452.76 eV are due to the In 3d5/2 and In 3d3/2 doublet states, respectively, which shows that In ions exist in the In3+ oxidation state. The S 2p spectrum is deconvoluted into three peaks. The peaks due to spin–orbit splitting for the S 2p core level (Fig. 7e) are located at 162.57 eV and 161.56 eV. Another small peak at 169.2 eV is attributed to S42−. Fig. S4 shows the comparison between high-resolution XPS spectra of In2S3/CdS/TNTs and Ar-In2S3/CdS/TNTs. Based on the high-resolution XPS elemental analysis of both annealed (treated) and un-annealed (un-treated) In2S3/CdS/TNTs samples, all the peaks (in Ti 2p, O 1s, Cd 3d, In 3d and S 2p spectra) match well with each other in both samples. However, a slight shift in the binding energies was observed, indicating the conversion of the binary chalcogenides CdS and In2S3 into the ternary chalcogenide CdIn2S4 crystal structure on the surface of TNTs.
 | ||
| Fig. 7 XPS core level spectra of (a) Ti 2p, (b) O 1s, (c) Cd 3d, (d) In 3d and (e) S 2p for Ar-In2S3/CdS/TNTs samples. | ||
The light absorption characteristics of the untreated and Ar-treated hetero-structures were investigated by UV-vis diffuse reflectance spectroscopy (DRS). Fig. 8a shows the DRS spectra of the untreated TNTs, CdS/TNTs, In2S3/TNTs, and In2S3/CdS/TNTs. An obvious absorption band at 368 nm in the UV range is observed for the pristine TNTs, which depicts the excitation of the electron from the valence band (VB) of TiO2 to the conduction band (CB) upon illumination with solar radiation. Incorporating CdS or In2S3 QDs on TNTs results in a red shift in the absorption edge of the TNTs. For a ternary heterogeneous structure, integrating In2S3 on the surface of CdS/TNTs, a well pronounced red shift in the absorption onset is observed. This can be due to the low band gap of In2S3 (about 2.3 eV), which further extends the absorption range of the heterogeneous sample (In2S3/CdS/TNTs) into the visible region. Fig. 8b shows the DRS spectra for Ar-TNTs, Ar-CdS/TNTs, Ar-In2S3/TNTs, and Ar-In2S3/CdS/TNTs. The Ar-TNTs, Ar-CdS/TNTs, and Ar-In2S3/TNTs samples do not show any significant changes in the spectra; however, Ar-In2S3/CdS/TNTs show an increase in the redshift with a flat tail, which depicts the emergence of a new phase of photoactive CdIn2S4via thermal treatment. These results indicate that the heterogeneous structure is also active in the visible range. The DRS data were employed to determine the optical bandgap of Ar-TNTs, Ar-CdS/TNTs, Ar-In2S3/TNTs, and Ar-In2S3/CdS/TNTs using Tauc analysis. The energy of the photons (hν) and absorption coefficient (α) were measured using the following relations, respectively,
where d is the thickness of the sample. Considering the direct allowed transition, the Tauc plot of (αhν)2vs. (hν) was constructed and is illustrated in Fig. S5. The linear region near the absorption edge was fitted and extrapolated to the energy axis. The intercept shows the value of the optical bandgap (Eg) of the material. Fig. S5(a–d) shows the values of optical bandgaps for Ar-TNTs, Ar-CdS/TNTs, Ar-In2S3/TNTs, and Ar-In2S3/CdS/TNTs, which are 3.2 eV, 2.6 eV, 2.4 eV and 1.9 eV, respectively. The obtained bandgap values are in close agreement with the values reported in the literature and confirm the expected band structure of Ar-In2S3/CdS/TNTs. CB and VB edge positions of all samples are measured by the following equations and are presented in Scheme S1.
| EVB = χ − 0.5Eg − Ec |
| ECB = EVB + Eg |
 | ||
| Fig. 8 (a and b) DRS spectra and (c and d) EIS plots of pristine TNTs, CdS/TNTs, In2S3/TNTs and In2S3/CdS/TNTs (a and c) without and (b and d) with Ar-treatment at 400 °C. | ||
Electrochemical impedance spectroscopy (EIS) was used to explore charge transfer characteristics at the surface of the photo-electrodes. Fig. 8c shows the Nyquist plots of TNTs, CdS/TNTs, In2S3/TNTs, and In2S3/CdS/TNTs (in 0.1 M PBS at 100 mV). The Nyquist plots clearly depict a distinctive smaller semicircle for the ternary heterogeneous structure (In2S3/CdS/TNTs). On the other hand, pristine TNTs and CdS QDs-sensitized TNTs do not show a semicircle and exhibit higher impedance values. The EIS of the Ar-treated samples (Fig. 8d) shows a remarkable decrease in the impedance for Ar-TNTs, Ar-In2S3/TNTs, and Ar-CdS/TNTs compared to untreated samples. This indicates that the charge transfer resistance has decreased tremendously. In the case of Ar-treated In2S3/CdS/TNTs, an obvious decrease in impedance and semicircle diameter (a low charge transfer resistance) is accompanied by interface-controlled charge carrier transport at low frequency values. It is evident from the DRS and EIS results that In2S3/CdS/TNTs ternary hetero-structure and Ar-In2S3/CdS/TNTs support efficient charge carrier separation and interfacial charge transport characteristics.
In order to demonstrate photoelectrochemical (PEC) activity, photocurrent density versus time responses were recorded. Fig. 9a shows photocurrent density vs. time curves for TNTs, CdS/TNTs, In2S3/TNTs, and In2S3/CdS/TNTs in 1 M KOH aqueous solution using a three-electrode system where Pt and Ag/AgCl electrodes served as the counter and reference electrodes, respectively. Transient photocurrent time curves are measured by illuminating the photocatalysts using a solar simulator (AM 1.5, 1000 W m−2) at regular time intervals. During illumination (the on cycle), the TNTs show a stable but smaller current density value (0.39 mA cm−2) compared to CdS/TNTs (1.57 mA cm−2) and In2S3/TNTs (2.48 mA cm−2). The current density of CdS/TNTs deteriorates with illumination time due to the lower photostability of the CdS QDs. However, the In2S3/TNTs electrode shows a more stable photocurrent response than the CdS/TNTs electrode due to the high photostability of the β-In2S3 QDs. In the case of the In2S3/CdS/TNTs ternary hetero-structure, a photocurrent density of 3.22 mA cm−2 (about 51% and 23% higher than those of CdS/TNTs and In2S3/TNTs electrodes, respectively) was observed. The enhancement of the PEC response is due to the staggered band structure of the constituent elements. On the other side, when illumination is cut off (the off cycle), a rapid drop in the photocurrent density to almost zero was observed. The photocurrent density regains its value for the In2S3/CdS/TNTs electrode and remains stable during several on–off cycles. In order to re-emphasize the choice of a particular number of deposition cycles for In2S3 QDs on the CdS/TNTs electrode, their PEC responses are recorded and shown in Fig. 9b. A sequentially higher photocurrent density was obtained by increasing the In2S3 deposition cycles, and an optimum value is achieved at 7 cycles. Furthermore, an abrupt decrease is observed when increasing the cycles to 9 due to the higher coverage of the QDs, which blocks the active sites for the photoelectrochemical process.
After heat treating In2S3/CdS/TNTs hetero-structures in an Ar-environment at 400 °C, photocurrent–time curves of the Ar-In2S3(3)/CdS/TNTs, Ar-In2S3(5)/CdS/TNTs, Ar-In2S3(7)/CdS/TNTs, and Ar-In2S3(9)/CdS/TNTs samples were recorded, as shown in Fig. 9c. The digit in parentheses indicates the number of different deposition cycles for In2S3 QDs on CdS/TNTs. These curves show a significant increase in the photocurrent density and stability of the treated samples. This is due to the formation of a stable layer (as a result of Ar annealing treatment) of ternary chalcogenide CdIn2S4 with compact pyramid-like nanostructures on the surface of the TNTs. Following the trend observed in Fig. 9b, the highest photocurrent density was observed for Ar-In2S3(7)/CdS/TNTs. Furthermore, this sample (Ar-In2S3(7)/CdS/TNTs) with the highest photo current density responses is compared with Ar-treated CdS/TNTs, In2S3/TNTs and pristine TNTs in Fig. 9d. It is evident from the figure that Ar-treatment of photo-electrodes leads to better crystallinity and stability.
The significantly enhanced photocurrent density of the ternary electrode system is due to the high surface area of the nanostructure, which offers more active sites for reactions. Integrating CdS and In2S3 simultaneously on TNTs gives the hetero-structure material a broader absorption window and increased photoresponse due to their reduced band gap energy. This fact was confirmed by the red shift in the DRS spectra. In parallel, the staggered band gap alignment formed at the interface of the conjugated active materials is desirable for the easy transfer and separation of photoexcited electron–hole pairs.
Photocatalytic H2 evolution on In2S3/CdS/TNTs
The photocatalytic ability of TNTs, CdS/TNTs, In2S3/CdS/TNTs, and In2S3/TNTs was recorded by monitoring the H2 evolution efficiency of the photocatalysts. The H2 evolution experiments were performed by irradiating each catalyst with visible light using a solar simulator (AM 1.5, 1000 W m−2) while immersing it in a water–methanol mixture solution within a quartz tube. The quartz cell was purged with N2 for 20 min before irradiation to remove the dissolved oxygen and air from the cell. The 200 µl gas was collected using a septum and injected into the gas chromatograph (GCMS-QO2010SE, Shimadzu, Japan) to analyze the amount of H2 evolved during the course of the reaction. Fig. S6a shows the average H2 evolution rate for different electrode configurations. The data clearly show a gradual increase in the H2 evolution rate by introducing the active materials one after another. The highest H2 generation rate (0.5694 mL h−1 cm−2) is achieved for the ternary In2S3/CdS/TNTs hetero-structure. The increased H2 generation activity is due to the existence of active heterojunctions between TNTs-CdS and CdS–In2S3. This allows efficient charge separation/transfer behavior at the interface of the junctions and increases light-harvesting ability.The consistency and re-usability of In2S3/CdS/TNTs for H2 production were tested for 8 consecutive cycles, as shown in Fig. S6b. After each cycle, the photocatalyst was thoroughly washed with DI water to remove any leftover residues of the reaction intermediates. These measurements (Fig. S6b) show a slight decline in photoresponse in the second cycle, and the H2 evolution efficiency reduces by 40% after the eighth cycle. The photocorrosion of the exposed CdS and In2S3 interface is the limiting factor for the reduced H2 evolution rate. Due to this reason, ternary chalcogenides formed by annealing In2S3/CdS/TNTs in an Ar atmosphere are highly useful to deal with the photo-corrosion issue.39,40 Introducing the CdIn2S4 nanostructure as a sacrificial layer on In2S3/CdS/TNTs not only favors photogenerated charge separation but also increases the stability of the sulfide layer due to its hole in-taking ability.58,59
Fig. 10a shows the H2 evolution rate for Ar-annealed samples, namely Ar-TNTs, Ar-CdS/TNTs, Ar-In2S3/TNTs, and Ar-In2S3/CdS/TNTs. It can be seen that Ar-In2S3/CdS/TNTs exhibits a significant increase in H2 evolution (0.6281 mL h−1 cm−2) in comparison to the un-annealed In2S3/CdS/TNTs (0.5694 mL h−1 cm−2). Ar annealing results in the formation of a thick layer of CdIn2S4 over TNTs due to the temperature-induced ion diffusion process. The band gap positions of CdIn2S4, along with those of In2S3, CdS, and TiO2, also favor the photocatalytic process. Interestingly, Ar-TNTs (0.0346 mL h−1 cm−2), Ar-CdS/TNTs (0.3808 mL h−1 cm−2), and Ar-In2S3/TNTs (0.3948 mL h−1 cm−2) also show a considerable increase in the H2 evolution rate compared to the non-Ar-treated samples. This is also due to the increased crystallinity of CdS and In2S3 QDS. Meanwhile, the H2 generation rate increases with increasing density of CdIn2S4 pyramids (Fig. 10b). The highest H2 evolution rate is observed for Ar-In2S3(7)/CdS/TNTs. Further increasing the density of CdIn2S4 reduces the H2 generation rate as CdIn2S4 no longer retains the pyramid nanostructure but is converted into big agglomerates (SEM Fig. 3g and h). Such agglomerates reduce the number of active interfaces and also inhibit the access of light to the photoactive sites. We have reported the average values of the H2 evolution rate by repeating all photocatalytic tests three times to ensure the reproducibility of the catalyst, and the data are presented in Fig. S(7–9). Fig. S7 and S8 show the H2 production rate for untreated and Ar-treated TNTs, CdS/TNTs, In2S3/TNTs and In2S3/CdS/TNTs. Fig. S9 shows the H2 production rate for Ar-In2S3/CdS/TNTs for different loading densities of In2S3 QDs.
Furthermore, to evaluate the stability of the photocatalysts for H2 evolution, 8 cycles of measurements were conducted on the same electrode. After each measurement, the electrode was cleaned with DI water and dried. A slight decrease in the H2 evolution rate is observed during the course of 3 cycles, and efficiency retention is 91% (Fig. 10c). The stability test was extended to eight consecutive cycles, showing decent retention of photocatalytic activity for Ar-In2S3/CdS/TNTs. The increased stability of the Ar-In2S3/CdS/TNTs sample is due to the considerably higher chemical and photostability of the outer CdIn2S4 layer. In order to reinforce the claim that CdIn2S4 suppresses photocorrosion, analysis using an inductively coupled plasma optical emission spectrometer (ICP-OES, LEEMAN, Prodigy 7, USA) of the reaction solution after H2 production by In2S3/CdS/TNTs and Ar-In2S3/CdS/TNTs is presented in Table S1. The electrolyte solution was collected and examined for Cd, In, and S ion traces. The results show the presence of significantly lower concentrations of Cd, S, and In ions in CdIn2S4-containing samples compared to unannealed samples, confirming reduced photocorrosion due to the induction of a CdIn2S4 layer. Furthermore, post-reaction XPS of Ti 2p, S 2p, O 1s, In 3d, and Cd 3d spectra are also presented in Fig. S10. By comparing the results with the XPS spectra of pre-reaction samples (Fig. 7), no major changes in Ti 2p, Cd 3d, In 3d, O 1s, and S 2p spectra are observed. The results further show fewer amounts of oxidized sulfur species, supporting photocorrosion suppression by CdIn2S4 passivation. The post-reaction SEM images of Ar-In2S3/CdS/TNTs were also checked and compared with the pre-reaction samples (Fig. S11) to analyze the stability of the CdIn2S4 structure and morphology. No major differences in the SEM images are observed, confirming the stable nature of CdIn2S4.
Further experiments were conducted by illuminating the Ar-In2S3/CdS/TNTs using 450 nm blue (90 mW cm−2) and 632 nm red LEDs (40 mW cm−2). Fig. 10d shows the average value of the H2 evolution rate obtained from 632 nm and 450 nm LED irradiation. The results show that the Ar-In2S3/CdS/TNTs heterostructure demonstrates wavelength-dependent photocatalytic behavior, with the highest H2 evolution rate obtained under 632 nm LED illumination. This behavior suggests that the material responds more efficiently to the red light than to blue or broad-spectrum illumination. The activity under 450 nm and the solar simulator was comparatively lower. This trend can be attributed to the unique optical response and charge transfer dynamics of the multi-sulfide heterostructure. Under 632 nm irradiation, the excitations match well with the absorption bands of CdIn2S4 and In2S3, leading to the creation of charge carriers and reducing recombination due to active charge transfer between the cascade band edge positions. For simulated sunlight, the broad-spectrum spreads energy over a wide wavelength range, leading to effective absorption of only a small portion and shows comparatively lower H2 production rates. These results indicate that CdS, In2S3, and CdIn2S4-based heterostructures have strong absorption ability towards a monochromatic visible source, as further confirmed by DRS measurements, which show higher absorption at 632 nm. AQY for photocatalytic H2 evolution was calculated by exciting the sample using a 450 nm LED lamp for 6 h. The sample is immersed in a solution of DI water having 12% methanol. The amount of evolved H2 production was measured using a gas chromatograph (GCMSQO2010SE, SHIMADZU, Japan) with a TCD detector. The power of the illumination source at the surface of the sample is 90 mW cm−2. AQY is determined using the following standard expression.
The photocatalytic hydrogen evolution in the absence of a scavenger was conducted in pure water. Ar-In2S3/CdS/TNTs was immersed in DI water and illuminated using a solar simulator (AM 1.5, 1000 W m−2). The results are presented in Fig. S12, which shows that a negligible amount of H2 is produced by Ar-annealed TNTs. For Ar-CdS/TNTs, Ar-In2S3/TNTs and Ar-In2S3/CdS/TNTs, about 24.5, 31.3 and 92.8 µL per h per cm2 per H2 evolution rates were obtained, respectively. In comparison to H2 production with sacrificial agents, about a 7-fold decrease is observed for Ar-In2S3/CdS/TNTs in pure water, indicating the necessity of sacrificial agents for efficient charge separation. The small amount of H2 production can be ascribed to the formation of a heterostructure, which effectively inhibit charge carrier recombination rates due to staggered band edge positions between CdIn2S4, In2S3, CdS, and TiO2. To demonstrate the effect of the annealing environment, we synthesized a control sample and annealed it in a N2 environment in a tube furnace under similar annealing conditions. Fig. S13 shows the cross-sectional SEM image of the N2-annealed sample, which is compared with the sample annealed in the Ar environment. The results demonstrate that Ar treatment yields a more defined CdIn2S4 nanostructure, particularly the formation of a distinctive nanopyramid structure observed on the walls of TNTs. The main phenomena in the formation of the CdIn2S4 phase are controlled interdiffusion of Cd2+ ions from CdS and In3+ ions from In2S3 into the sulfide lattice through solid-state cation exchange and anion reorganization, which results in nucleation and growth of CdIn2S4.47,54,60 An inert environment is required to prevent the oxidation of sulfide species that would otherwise disturb the stoichiometry. The localized interdiffusion at the In2S3/CdS interface promotes anisotropic crystal growth, resulting in the emergence of a faceted nano-pyramidal structure. These facets are related to low-energy crystallographic planes that are preferably stabilized under oxygen-free conditions provided by inert annealing. As shown in the figures, a noticeable difference in the morphology of CdIn2S4 formed on the surface of TNTs is observed for the sample annealed in a N2 environment compared to that in Ar. Although both gases offer an inert environment essential for the protection of sulfide stoichiometry, their elusive physical and chemical differences can influence crystal growth. Ar, being a completely inert noble gas, encourages uniform heat distribution and allows controlled solid-state diffusion between CdS and In2S3, which results in the formation of well-faceted pyramid-like CdIn2S4 nanocrystals. The lack of the ability of Ar to interact with surfaces ensures solely thermodynamically controlled atomic rearrangements. In contrast, N2 can interact weakly with surface defects and oxygen vacancies, particularly with TNTs, which can alter nucleation and growth kinetics. Consequently, the sample annealed in an N2 environment exhibits a smaller, less faceted and less textured CdIn2S4 layer. The photocatalytic H2 production for the N2 annealed sample is also shown in Fig. S14. The results exhibit low efficiency of H2 production for the N2 annealed sample as compared to the sample annealed in Ar. These findings depict that Ar annealing favors highly crystalline and well-defined pyramid structures compared to the N2 annealed sample.
The rate of H2 evolution in the present work was further compared with those reported in the literature.61–68 First, the H2 evolution rate for Ar-In2S3/CdS/TNTs is converted from mL h−1 cm−2 to mmol h−1 g−1, where the mass of the sample is 0.38 mg. From Table 1, it can be seen that among other photocatalysts, Ar-In2S3/CdS/TNTs exhibits the highest H2 evolution rate of 73.94 mmol h−1 g−1. This outstanding performance of Ar-In2S3/CdS/TNTs results from the separation of photogenerated charge carriers and higher utilization of the visible range of the solar spectrum, which are key for efficient photocatalysts. These two factors are supported by the staggered band alignment and low band gap of CdS and In2S3 and CdIn2S4.
| Sr. no. | Sample name | H2 evolution rate mmol h−1 g−1 | Illuminating source | Ref. |
|---|---|---|---|---|
| 1 | W-TiO2/Au hybrid | 24.000 | 300 W Xe lamp | 67 |
| 2 | Pt/TiO2 | 11.200 | AM 1.5 G solar simulator | 68 |
| 3 | F-TiO2 | 18.270 | 300 W Xe lamp | 66 |
| 4 | Co-TiO2 | 11.021 | Solar and UV (400 W Hg vapour lamp) | 65 |
| 5 | Gd/N-TiO2 | 10.764 | 150 W Xe lamp | 64 |
| 6 | GQDs/TiO2 nanocomposites (CTNP-3) | 29.548 | Natural solar light | 63 |
| 7 | CdIn2S4–In2O3–In2S3 | 2.004 | 225 W Xe arc lamp (320-780 nm) | 61 |
| 8 | Dibenzo[b,d]thiophene sulfone (P10-e) | 60.600 | 300 W Xe light source λ > 420 nm | 62 |
| 9 | P-Mo2CTx/CdS | 7.365 | Visible-light irradiation (λ = 420 nm) | 69 |
| 10 | F-doped TiO2/ZnIn2S4 | 1.58 | 300 W xenon lamp | 70 |
| 11 | CoP/ZIS-4 | 4.24 | 300 W xenon lamp with a 420 nm filter (λ ≥ 420 nm) | 71 |
| 12 | Ar-In2S3 QDs/CdS QDs/TNTs | 73.94 | 1.5 AM solar simulator (300 W Xe lamp) | Present work |
Photodegradation of MB on In2S3/CdS/TNTs
To evaluate the merits of ternary heterogeneous photocatalysts for wastewater decontamination, we adopted MB as a model dye in aqueous media and studied its degradation under simulated visible light illumination generated by a 300 W PLS-SXE300UV xenon lamp equipped with a UV cut-off filter (UVCCUT400). Prior to each experiment, samples were immersed in 50 ml of a 30 ppm dye solution for 40 min under dark conditions (with continuous stirring) to attain equilibrium between dye adsorption/desorption on the surface of the photocatalyst. The dye solution, along with the catalysts, was irradiated, and adequate amounts of aliquots (2 ml) were collected after a time interval of 3 min. The photo-degradation efficiency was examined by quantifying the variations in the characteristic absorption peak of the dye solution using the following relation.| D (%) = 100 × (C0 − C)/C0 | (1) |
Analogous experiments were performed in the dark and in the absence of any photocatalyst in order to validate that the degradation is exclusively due to the photocatalytic process initiated by incident light on the surface of the photocatalyst. The characteristic absorption edge of MB (at 668 nm) was utilized to observe the degradation process over time (Fig. S15a). Fig. S15b shows the dye degradation rate (C/C0) vs. the degradation time for pristine TNTs, CdS/TNTs, In2S3/TNTs, and In2S3/CdS/TNTs for MB. It is observed that the photocatalytic degradation performance of the In2S3/CdS/TNTs ternary heterostructure is much higher than that of other electrode configurations. In order to get a more appropriate representation of the dye degradation process, the data is fitted using a pseudo-first-order equation ln(C0/C) vs.t (Fig. S15c). The estimated linear fitting of the curves for all electrodes shows that all photocatalysts follow pseudo-first-order reaction kinetics. The comparison of the photocatalytic activities, the photocatalysis rate constants k (calculated from the slope of the linear plots of ln(C0/C) vs. irradiation time t) along with other experimental parameters for TNTs, CdS/TNTs, In2S3/TNTs, and In2S3/CdS/TNTs degradation of MB is listed in Table 2. The In2S3/CdS/TNTs heterogeneous electrode shows superior photocatalytic activity with a higher degradation rate constant than other electrodes. The photo-degradation efficiency block diagram for MB shown in Fig. S15d reveals that almost complete degradation of the dye was attained via heterogeneous nanostructures within 27 min, whereas for TNTs, about 25% of the dye remained for the same time interval and complete degradation occurred in 40 min.
| Sr. no. | Sample name | Photodegradation rate constant (k) min−1 | Photodegradation efficiency (%) for 27 min irradiation | Total degradation time (min) |
|---|---|---|---|---|
| 1 | TNTs | 0.0647 | 74 | 40 |
| 2 | CdS/TNTs | 0.886 | 85 | 36 |
| 3 | In2S3/TNTs | 0.1225 | 92 | 33 |
| 4 | In2S3/CdS/TNTs | 0.1661 | 99 | 27 |
The significantly enhanced photocatalytic efficiency of the ternary electrode system is attributed to the high surface area of the nanostructure, which offers more active sites and assists the adsorption of dye molecules on the catalyst surface. Another reason can be the utilization of appropriate active materials, which favor the utmost utilization of visible light. Integrating CdS and In2S3 on TNTs imparts an increased photoresponse due to their reduced band gap energy, as confirmed by the red shift in the DRS results. In parallel, the staggered band gap alignment formed at the interface of the conjugated active materials is desirable for facile transfer and separation of the photoexcited electron–hole pair.
To unfold the role of active species in the photodegradation of dyes under visible light illumination, a sequential addition of quenchers was utilized to determine the governing active species in photocatalysis. Benzoquinone (BQ), methanol (M), silver nitrate (AgNO3) and isopropanol (IP) were added separately to the dye solution before irradiation, which act as scavengers for O2−, h+, e− and ˙OH, respectively. Fig. S15e shows the photodegradation of MB, along with different scavengers. The presence of AgNO3, BQ and isopropanol considerably inhibits the photodegradation process for MB. However, the hole scavenger methanol slightly influences the photodegradation. The above findings illustrate that ˙OH radicals, which are also a subsequent product of photogenerated electrons and O2−, are the primary key element in the photodegradation of the dye, while a secondary role is played by the holes.
The consistency and re-usability of In2S3/CdS/TNTs for dye degradation were tested for 3 successive cycles, as shown in Fig. S15f. After each cycle, the photocatalyst was thoroughly washed with DI water and ethanol to remove any leftover residues of reaction intermediates. The results specify a minor decline in the photoactivity of the catalyst after 3 cycles, which could be due to photocorrosion of the exposed CdS QDs in the aqueous dye solution. These findings reveal improved stability and re-usability of In2S3/CdS/TNTs for potential dye degradation applications. These results suggest the significance of the morphological architecture (structural design), which favors an appropriate band structure that allows the transfer of holes to In2S3, thus preventing CdS photo-corrosion. This captivated approach to electrode fabrication lowers the operational cost of the purification process and leads to a new approach for designing novel heterostructures.
Degradation of MB on Ar-In2S3/CdS/TNTs
The dye degradation abilities of Ar-TNTs, Ar-CdS/TNTs, Ar-In2S3/TNTs, and Ar-In2S3/CdS/TNTs were measured by monitoring changes in the UV absorption spectra of MB with irradiation time. Fig. 11a shows the characteristic UV absorption spectra of MB complying with Ar-In2S3/CdS/TNTs. The characteristic absorption peaks seem to reduce effectively and turn into a nearly flat spectrum within a short time. The dye degradation rate (Fig. 11b) for all prepared structures shows that complete degradation for MB is observed within 22 min with Ar-In2S3/CdS/TNTs compared to Ar-TNTs, Ar-CdS/TNTs, and Ar-In2S3/TNTs. All electrodes follow the pseudo-first-order reaction as verified from the linear fitting of ln(C0/C) vs.t (Fig. 11c). Meanwhile, the corresponding rate constants derived from the slope of Fig. 11c for all electrodes are tabulated in Table 3. The Ar-In2S3/CdS/TNTs composite nanostructure displays the highest photocatalytic activity with higher photodegradation, where about 99.7% of MB is degraded in just 22 min (Fig. 11d). It can also be confirmed from the table that the value of the reaction rate constant for Ar-In2S3/CdS/TNTs is higher than that of In2S3/CdS/TNTs, while the values for annealed Ar-TNTs, Ar-CdS/TNTs, and Ar-In2S3/TNTs are better than that of the un-annealed samples. The higher degradation efficiency of Ar-In2S3/CdS/TNTs conjugated with ternary chalcogenides CdIn2S4 compared to the heterostructure of binary chalcogenides (In2S3/CdS/TNTs) can be ascribed to the higher catalytic sites on the surface of CdIn2S4 pyramids. Generally, the photocatalytic performance of a photocatalyst depends on its photoabsorption range, efficiency of electron–hole pair generation and higher separation to recombination ratio. In ternary chalcogenides, the metal ion insertion (Cd) in the structure of a binary chalcogenide (In2S3) leads to an increase in defect sites, which enables enhanced light harvesting and extra charge transport channels from the conduction band (CB).39,40| Sr. No. | Sample name | Photodegradation rate constant (k) min−1 | Photodegradation efficiency (%) for 27 min irradiation | Total degradation time (min) |
|---|---|---|---|---|
| 1 | Ar-TNTs | 0.0657 | 65 | 34 |
| 2 | Ar-CdS/TNTs | 0.1592 | 93.8 | 30 |
| 3 | Ar-In2S3/TNTs | 0.1724 | 94.9 | 24 |
| 4 | Ar-In2S3/CdS/TNTs | 0.2134 | 99.7 | 22 |
To get insight into the photocatalytic process of Ar-In2S3/CdS/TNTs, a schematic representation of the degradation process is shown in Scheme S1. Under irradiation, photogenerated electrons were produced in the CB of CdIn2S4 (due to its low band gap energy) and rapidly transferred to the CB of TiO2via In2S3 and CdS. This is due to the better interface between CdIn2S4–In2S3, In2S3–CdS, and CdS–TiO2 and favorable band edges. Meanwhile, the transfer of photogenerated holes from the VB of CdIn2S4 towards the VB of TiO2 is thermodynamically not favored. This is due to the more cathodic VB potential of CdIn2S4 as compared to those of In2S3, CdS and TiO2, enabling the separation of the electron–hole pair. These holes may further react with H2O molecules and produce high-activity hydroxyl radicals (˙OH). The electrons in the CB of TiO2 can be captured by dissolved oxygen in water to form superoxide radicals (O2−), which can effectively degrade the noxious molecules.
Various scavenging agents were used to test the predominant reactive species in the photodegradation of dyes. As evident from the results in Fig. 11e, a relatively small loss of photodegradation activity is observed for the hole scavenger, while others show a significant decrease. These results emphasize that the electrons transferred to the CB of TNTs are trapped by molecular oxygen in the electrolyte to form the reactive oxygen species (superoxide radical anion). They subsequently produce hydroxyl radicals, the dominant active species in the photodegradation process. The involvement of the photogenerated holes was also evident from the trapping experiments; thus, the combined effect of hydroxyl radicals and photogenerated holes determines the degradation of dyes. Furthermore, the photostability of Ar-In2S3/CdS/TNTs was tested by repeating the degradation experiments for MB. Fig. 11f shows the significant increase in the stability along with the degradation rate. These results show a comparatively enhanced anti-photocorrosion activity of the In2S3/CdS/TNTs due to induced CdIn2S4 sacrificial layer on In2S3/CdS/TNTs.
Conclusions
A ternary heterostructure based on TNTs incorporating CdS and In2S3 QDs was prepared and employed for photocatalysis of water and H2 generation (facile energy source). The photocatalytic performance of the different heterostructures was explored. The synergistic effect of CdS QDs on TNTs increases the visible light response of the heterostructure. Introducing In2S3 QDs on CdS/TNTs dramatically increases H2 evolution, which is due to the charge separation ability and cascade band gap positions of In2S3 with CdS and TiO2. A passivation layer composed of CdIn2S4 is formed by thermally treating In2S3/CdS/TNTs in Ar. This enhanced the chemical stability of the photoelectrode and significantly boosted the H2 production rate to 0.6281 mL h−1 cm−2. The AQY of Ar-In2S3/CdS/TNTs at 450 nm and 632 nm LED are 5.54% and 13.57% respectively. The higher photocurrent density and lower charge transfer resistance suggest that the structure of the Ar-In2S3/CdS/TNTs photoanodes supports more efficient charge carrier separation and interfacial charge transport properties. Ar-In2S3/CdS/TNTs shows complete degradation of MB within 22 min, as compared to In2S3/CdS/TNTs, where the dye is degraded within 27 min.Conflicts of interest
There are no conflicts to declare.Data availability
The data supporting this article have been included in the main text of the manuscript and in the supplementary information (SI). Supplementary information: the digital images of different stages of fabricated electrode, FESEM top and cross-sectional images of In2S3/CdS/TNTs hetero-structures for different loading cycles of In2S3 on CdS/TNTs, FESEM cross-sectional view of Ar-treated pristine TNTs, CdS/TNTs, and In2S3/TNTs, comparison of XPS core level spectra of Ti 2p, O 1s, Cd 3d, In 3d, and S 2p for In2S3/CdS/TNTs and Ar-In2S3/CdS/TNTs samples, Touc plots for Ar-TNTs, Ar-CdS/TNTs, Ar-In2S3/TNTs, and Ar-In2S3/CdS/TNTs. Band diagram with CB and VB edge positions of TNTs, CdS, In2S3 and CdIn2S4. H2 production rate for different electrodes and (b) H2 production rate of In2S3/CdS/TNTs for 8 consecutive cycles. H2 production reproducibility measurements for unannealed samples. H2 production reproducibility measurements for Ar annealed samples. H2 production reproducibility measurements for Ar-In2S3/CdS/TNTs of different thickness of the CdIn2S4 passivation layer controlled by In2S3 deposition cycles. Table indicating traces of Cd, S and In ions for In2S3/CdS/TNTs and Ar-In2S3/CdS/TNTs using Inductively Coupled Plasma Optical Emission Spectrometer. XPS core level spectra of Ti 2p, O 1s, Cd 3d, S 2p and In 3d for Ar-In2S3/CdS/TNTs after photocatalytic reactions. FESEM cross-sectional view of Ar-In2S3/CdS/TNTs samples (a) pre and (b) post photocatalytic reaction. H2 evolution rate by Ar-TNTs (Ar-T), Ar-CdS/TNTs (Ar-CT), Ar-In2S3/TNTs (Ar-IT) and Ar-In2S3/CdS/TNTs (Ar-ICT) in pure water in the absence of methanol. SEM cross-sectional image of In2S3/CdS/TNTs after N2 annealing and Ar annealing at 400 °C for 1 hour. H2 production rate for In2S3/CdS/TNTs after N2 annealing and Ar annealing. Absorption spectra of MB for different irradiation time on In2S3/CdS/TNTs, degradation rate, degradation reaction kinetics and degradation efficiency of MB with respect to irradiation time for different samples. Effect of different reactive species scavenges of photo-degradation of MB and degradation ratio plots for different repeating cycle on In2S3/CdS/TNTs. See DOI: https://doi.org/10.1039/d5ta05742f.Acknowledgements
The authors greatly acknowledge the Higher Education Commission (HEC) of Pakistan (grant no. IRSIP 36 PSc 31) for providing the financial support for this research work. Financial support from DFG, within the framework of its Excellence Initiative for the Cluster of Excellence “Engineering of Advanced Materials”, is thankfully acknowledged.References
- A. Fujishima and K. Honda, Electrochemical Photolysis of Water at a Semiconductor Electrode, Nature, 1972, 238, 37–38, DOI:10.1038/238037a0.
- J. Kegel, I. M. Povey and M. E. Pemble, Zinc Oxide for Solar Water Splitting: A Brief Review of the Material’s Challenges and Associated Opportunities, Nano Energy, 2018, 54, 409–428, DOI:10.1016/j.nanoen.2018.10.043.
- J. Joy, J. Mathew and S. C. George, Nanomaterials for Photoelectrochemical Water Splitting- Review, Int. J. Hydrogen Energy, 2018, 43(10), 4804–4817, DOI:10.1016/j.ijhydene.2018.01.099.
- R. Abe, M. Higashi and K. Domen, Overall Water Splitting under Visible Light through a Two- Step Photoexcitation between TaON and WO3 in the Presence of an Iodate – Iodide Shuttle Redox Mediator, ChemSusChem, 2011, 8656, 228–237, DOI:10.1002/cssc.201000333.
- M. Ge, Q. Li, C. Cao, J. Huang, S. Li, S. Zhang, Z. Chen, K. Zhang, S. S. Al-deyab and Y. Lai, One-Dimensional TiO2 Nanotube Photocatalysts for Solar Water Splitting, Adv. Sci., 2017, 4, 1–31, DOI:10.1002/advs.201600152.
- I. Paramasivam, H. Jha, N. Liu and P. Schmuki, A Review of Photocatalysis Using Self-Organized TiO2 Nanotubes and Other Ordered Oxide Nanostructures, Small, 2012, 8(20), 3073–3103, DOI:10.1002/smll.201200564.
- S. So, I. Hwang, J. E. Yoo, S. Mohajernia, M. Mackovic, E. Spiecker, G. Cha, A. Mazare and P. Schmuki, Inducing a Nanotwinned Grain Structure within the TiO2 Nanotubes Provides Enhanced Electron Transport and DSSC Efficiencies >10%, Adv. Energy Mater., 2018, 1800981, 1–8, DOI:10.1002/aenm.201800981.
- J. E. Yoo, A. Alshehri, S. Qin and M. Bawaked, Establishing High Photocatalytic H2 Evolution from Multiwalled Titanate Nanotubes, ChemCatChem, 2020, 12, 2951–2956, DOI:10.1002/cctc.202000281.
- N. Lakshmanareddy, V. Navakoteswara, K. Krishnan, E. Palanisamy and M. Venkatakrishnan, Pt/TiO2 Nanotube Photocatalyst Effect of Synthesis Methods on Valance State of Pt and Its Influence on Hydrogen Production and Dye Degradation, J. Colloid Interface Sci., 2019, 538, 83–98, DOI:10.1016/j.jcis.2018.11.077.
- N. Chi, T. Mai, T. Thi, T. Van and R. Juang, Separation and Purification Technology Enhanced Removal of Various Dyes from Aqueous Solutions by UV and Simulated Solar Photocatalysis over TiO2/ZnO/RGO Composites, Sep. Purif. Technol., 2020, 232, 115962, DOI:10.1016/j.seppur.2019.115962.
- M. R. Abhilash, G. Akshatha and S. Srikantaswamy, Photocatalytic Dye Degradation and Biological Activities of the Fe2O3/Cu2O Nanocomposite, RSC Adv., 2019, 9, 8557–8568, 10.1039/c8ra09929d.
- P. M. Perillo and D. F. Rodríguez, Formation of TiO2 Nanopores by Anodization of Ti-Films, OALib, 2014, 10, 1–9, DOI:10.4236/oalib.1100630.
- I. Paramasivam, H. Jha, N. Liu and P. Schmuki, A Review of Photocatalysis Using Self-Organized TiO2 Nanotubes and Other Ordered Oxide Nanostructures, Small, 2012, 8, 3073–3103, DOI:10.1002/smll.201200564.
- A. J. Frank and K. Honda, Visible-Light-Induced Water Cleavage and Stabilization of n-Type CdS to Photocorrosion with Surface-Attached Polypyrrole-Catalyst Coating, J. Phys. Chem., 1982, 83(4), 1933–1935, DOI:10.1021/j100208a005.
- W. Lee, H. Jung, D. Lee, B. Kim, H. Kim, K. Kim and M. Jae, Suppression of Photocorrosion in CdS/CdSe Quantum Dot-Sensitized Solar Cells : Formation of a Thin Polymer Layer on the Photoelectrode Surface, Synth. Met., 2013, 165, 60–63, DOI:10.1016/j.synthmet.2013.01.008.
- S. L. Lee and C. Chang, Recent Developments about Conductive Polymer Based Composite Photocatalysts, Polymers, 2019, 11, 206, DOI:10.3390/polym11020206.
- C. Wang, L. Wang, J. Jin, J. Liu, Y. Li, M. Wu, L. Chen, B. Wang, X. Yang and B.-L. Su, Probing Effective Photocorrosion Inhibition and Highly Improved Photocatalytic Hydrogen Production on Monodisperse PANI@CdS Core-Shell Nanospheres, Appl. Catal., B, 2016, 188, 351–359, DOI:10.1016/j.apcatb.2016.02.017.
- J. Su, Y. Wei and L. Vayssieres, Stability and Performance of Sulfide-, Nitride-, and Phosphide-Based Electrodes for Photocatalytic Solar Water Splitting, J. Phys. Chem. Lett., 2017, 5228–5238, DOI:10.1021/acs.jpclett.7b00772.
- P. Hsieh, Y. Chiu, T. Lai, M. Fang, Y. Wang and Y. Hsu, TiO2 Nanowire-Supported Sulfide Hybrid Photocatalysts for Durable Solar Hydrogen, ACS Appl. Mater. Interfaces, 2019, 11(3), 3006–3015, DOI:10.1021/acsami.8b17858.
- B. Weng, M. Qi, C. Han, Z. Tang and Y. Xu, Photocorrosion Inhibition of Semiconductor-Based Photocatalysts : Basic Principle, Current Development, and Future Perspective, ACS Catal., 2019, 9, 4642–4687, DOI:10.1021/acscatal.9b00313.
- M. Ben, W. Jo and H. Elhouichet, Reduced Graphene Oxide as an Efficient Support for CdS-MoS2 Heterostructures for Enhanced Photocatalytic H2 Evolution, Int. J. Hydrogen Energy, 2017, 1–10, DOI:10.1016/j.ijhydene.2017.05.225.
- W. Chen, R. Yan, G. Chen, Y. Chen, G. Huang and X. Liu, Hydrothermal Route to Synthesize Helical CdS@ZnIn2S4 Core-Shell Heterostructures with Enhanced Photocatalytic Hydrogeneration Activity, Ceram. Int., 2019, 45(2), 1803–1811, DOI:10.1016/j.ceramint.2018.10.067.
- C. Li, H. Che, Y. Yan, C. Liu and H. Dong, Z-Scheme AgVO3/ZnIn2S4 Photocatalysts: “One Stone and Two Birds” Strategy to Solve Photocorrosion and Improve the Photocatalytic Activity and Stability, Chem. Eng. J., 2020, 398, 125523, DOI:10.1016/j.cej.2020.125523.
- P. Hsieh, Y. Chiu, T. Lai, M. Fang, Y. Wang and Y. Hsu, TiO2 Nanowire-Supported Sulfide Hybrid Photocatalysts for Durable Solar Hydrogen Production, ACS Appl. Mater. Interfaces, 2019, 11, 3006–30115, DOI:10.1021/acsami.8b17858.
- L. Zheng, F. Teng, X. Ye, H. Zheng and X. Fang, Photo/Electrochemical Applications of Metal Sulfide/TiO2 Heterostructures, Adv. Energy Mater., 2019, 10(1), 1902355, DOI:10.1002/aenm.201902355.
- M. Mollavali, C. Falamaki and S. Rohani, Efficient Light Harvesting by NiS/CdS/ZnS NPs Incorporated in C , N-Co-Doped-TiO2 Nanotube Arrays as Visible-Light Sensitive Multilayer Photoanode for Solar Applications, Int. J. Hydrogen Energy, 2018, 1–20, DOI:10.1016/j.ijhydene.2018.03.102.
- X. Ning and G. Lu, Photocorrosion inhibition of CdS-based catalysts for photocatalytic overall water splitting, Nanoscale, 2020, 2020(12), 1213–1223, 10.1039/C9NR09183A.
- S. S. Reddy and B. Kotaiah, Decolorization of Simulated Spent Reactive Dye Bath Using Solar/TiO2/H2O2, Int. J. Environ. Sci. Technol., 2005, 2, 245–251, DOI:10.1007/BF03325883.
- T. Wo, T. Sno, C. Lin, C. Wu and Z. Onn, Degradation of 4-Chlorophenol in TiO2, WO3, SnO2, TiO2/WO3 and TiO2/SnO2 Systems, J. Hazard. Mater., 2008, 154, 1033–1039, DOI:10.1016/j.jhazmat.2007.11.010.
- M. Miyauchi, A. Nakajima, T. Watanabe and K. Hashimoto, Photoinduced Hydrophilic Conversion of TiO2/WO3 Layered Thin Films, Chem. Mater., 2002, 14, 4714–4720, DOI:10.1021/cm020355c.
- H. Tada, A. Kokubu, M. Iwasaki and S. Ito, Deactivation of the TiO2 Photocatalyst by Coupling with WO3 and the Electrochemically Assisted High Photocatalytic Activity of WO3, Langmuir, 2004, 20(11), 4665–4670, DOI:10.1021/la036104f.
- Y. W. Han, Y. T. Chu, L. Ye, T. J. Gong, X. B. Lu and Y. Fu, Enhancing Built-In Electric Field via Defect-Mediated Interfacial Chemical Bond Construction in Chalcogenide Heterojunction for Alcohol Photooxidation Coupled with H2 Production, Adv. Funct. Mater., 2025, 35(1), 1–13, DOI:10.1002/adfm.202411991.
- Y. He, D. Li, G. Xiao, W. Chen, Y. Chen, M. Sun, H. Huang and X. Fu, A New Application of Nanocrystal In2S3 in Efficient Degradation of Organic Pollutants under Visible Light Irradiation, J. Phys. Chem. C, 2009,(113), 5254–5262, DOI:10.1021/jp809028y.
- S. Khanchandani, S. Kundu, A. Patra and A. K. Ganguli, Band Gap Tuning of ZnO/In2S3 Core/Shell Nanorod Arrays for Enhanced Visible-Light-Driven Photocatalysis, J. Phys. Chem. C, 2013, 117(11), 5558–5567, DOI:10.1021/jp310495j.
- S. Kumar, N. Yadav, P. Kumar and A. K. Ganguli, Design and Comparative Studies of Z-Scheme and Type II Based Heterostructures of NaNbO3/CuInS2/In2S3 for Efficient Photoelectrochemical Applications, Inorg. Chem., 2018, 57, 15112–15122, DOI:10.1021/acs.inorgchem.8b02264.
- W. Dong, D. Wang, H. Wang, M. Li, F. Chen, F. Jia and Q. Yang, Facile Synthesis of In2S3/UiO-66 Composite with Enhanced Adsorption Performance and Photocatalytic Activity for the Removal of Tetracycline under Visible Light Irradiation, J. Colloid Interface Sci., 2019, 535, 444–457, DOI:10.1016/j.jcis.2018.10.008.
- S. Znin, C. S. Nanocrystals, S. K. Batabyal, S. E. Lu and J. J. Vittal, Synthesis, Characterization, and Photocatalytic Properties of In2S3, ZnIn2S4, and CdIn2S4 Nanocrystals, Cryst. Growth Des., 2016, 16, 2231–2238, DOI:10.1021/acs.cgd.6b00050.
- R. Malik, V. K. Tomer, M. Sain and Z. Chen, Dissimilar Dimensional Materials Based Tailored Heterostructures for Photocatalytic Hydrogen Production, Renew. Sustain. Energy Rev., 2023, 181, 113348, DOI:10.1016/j.rser.2023.113348.
- S. Wang, B. Y. Guan, Y. Lu, X. Wen and D. Lou, Formation of Hierarchical In2S3−CdIn2S4 Heterostructured Nanotubes for Efficient and Stable Visible Light CO2 Reduction, J. Am. Chem. Soc., 2017, 139, 17305–17308, DOI:10.1021/jacs.7b10733.
- Q. Zhang, J. Wang, X. Ye, Z. Hui, L. Ye, X. Wang and S. Chen, Self-Assembly of CdS/CdIn2S4 Heterostructure with Enhanced Photocascade Synthesis of Schiff Base Compounds in an Aromatic Alcohols and Nitrobenzene System with Visible Light, ACS Appl. Mater. Interfaces, 2019, 11(50), 46735–46745, DOI:10.1021/acsami.9b14450.
- S. K. Batabyal, S. E. Lu and J. J. Vittal, Synthesis, Characterization, and Photocatalytic Properties of In2S3, ZnIn2S4, and CdIn2S4 Nanocrystals, Cryst. Growth Des., 2016, 16(4), 2231–2238, DOI:10.1021/acs.cgd.6b00050.
- N. Khaliq, G. Ali, M. A. Rasheed, M. Khan, Y. Xie, P. Schmuki, S. O. Cho and S. Karim, Multifunctional Tunable Cu2O and CuInS2 Quantum Dots on TiO2 Nanotubes for Efficient Chemical Oxidation of Cholesterol and Ibuprofen, Nanoscale, 2024, 16(25), 12207–12227, 10.1039/d4nr00422a.
- N. Khaliq, M. A. Rasheed, M. Khan, M. Maqbool, M. Ahmad, S. Karim, A. Nisar, P. Schmuki, S. O. Cho and G. Ali, Voltage-Switchable Biosensor with Gold Nanoparticles on TiO2 Nanotubes Decorated with CdS Quantum Dots for the Detection of Cholesterol and H2O2, ACS Appl. Mater. Interfaces, 2021, 13(3), 3653–3668, DOI:10.1021/acsami.0c19979.
- L. Fan and R. Guo, Fabrication of Novel CdIn2S4 Hollow Spheres via a Facile Hydrothermal Process, J. Phys. Chem. C, 2010, 112, 10700–10706, DOI:10.1021/jp8022259.
- A. Raghavan, S. Sarkar, L. R. Nagappagari, S. Bojja, S. Muthukondavenkatakrishnan and S. Ghosh, Decoration of Graphene Quantum Dots on TiO2 Nanostructures : Photosensitizer and Cocatalyst Role for Enhanced Hydrogen Generation, Ind. Eng. Chem. Res., 2020, 59, 13060–13068, DOI:10.1021/acs.iecr.0c01663.
- R. Li, L. Tang, Q. Zhao, T. H. Ly, K. S. Teng, Y. Li, Y. Hu, C. Shu and S. P. Lau, In2S3 Quantum Dots: Preparation, Properties and Optoelectronic Application, Nanoscale Res. Lett., 2019, 14, 161, DOI:10.1186/s11671-019-2992-0.
- Y. Qi, G. Zhou, Y. Wu, H. Wang, Z. Yan and Y. Wu, In-Situ Construction of In2O3/In2S3-CdIn2S4 Z-Scheme Heterojunction Nanotubes for Enhanced Photocatalytic Hydrogen Production, J. Colloid Interface Sci., 2024, 664, 107–116, DOI:10.1016/j.jcis.2024.03.033.
- R. Verma, R. Rani, P. Choubey, A. Sharma and M. Basu, Au Nanoparticles on In2S3/In2O3 Nanopyramids Increase Photoanodic Activity in Photoelectrochemical Water Splitting, ACS Appl. Nano Mater., 2024, 7(23), 26434–26444, DOI:10.1021/acsanm.4c03123.
- L. Yang, C. McCue, Q. Zhang, E. Uchaker, Y. Mai and G. Cao, Highly Efficient Quantum Dot-Sensitized TiO2 Solar Cells Based on Multilayered Semiconductors (ZnSe/CdS/CdSe), Nanoscale, 2015, 7(7), 3173–3180, 10.1039/c4nr06935h.
- Y. Zhao, D. Wang, G. Sun, L. B. Wang, J. Zhang and B. Zhu, Rhodamine B Sensitization Promoting TiO2/CdIn2S4 S-Scheme Photocatalytic H2O2-Production Activity and Its Charge Transfer Mechanism, Chem. Eng. J., 2025, 518, 164759, DOI:10.1016/j.cej.2025.164759.
- E. K. Andreou, I. Vamvasakis and G. S. Armatas, Efficient Visible Light Photocatalytic Hydrogen Evolution by Boosting the Interfacial Electron Transfer in Mesoporous Mott-Schottky Heterojunctions of Co2P-Modified CdIn2S4 Nanocrystals, ACS Appl. Energy Mater., 2024, 7(11), 4891–4903, DOI:10.1021/acsaem.4c00710.
- X. Gao, D. Guan, J. Huo, J. Chen and C. Yuan, Free Standing TiO2 Nanotube Array Electrodes with an Ultra-Thin Al2O3 Barrier Layer and TiCl4 Surface Modification for Highly Efficient Dye Sensitized Solar Cells, Nanoscale, 2013, 5(21), 10438–10446, 10.1039/c3nr03198e.
- S. C. Nunes, N. J. O. Silva, J. Hümmer, R. A. S. Ferreira, P. Almeida, L. D. Carlos and V. De. Zea, Water-Mediated Structural Tunability of Alkyl/Siloxane Hybrid : From Amorphous material to Lamellar Structure or Bilamellar Superstructure, RSC Adv., 2012, 2087–2099, 10.1039/C2RA00702A.
- Z. Chen, L. Wang, X. Jiao, Y. Xia and D. Chen, Thermally Triggered Surface Reconstruction Enables Self-Protective Layer for Enhanced Charge Transfer in CdIn2S4 Photoelectrode, Chem. Eng. J., 2025, 522, 167707, DOI:10.1016/j.cej.2025.167707.
- M. A. Hamza, G. F. Metha and C. J. Shearer, Recent Advances and Future Directions of CdIn2S4-Based Photocatalysts: Properties, Synthesis, and Modifications for Energy and Environmental Applications, J. Mater. Chem. A, 2025, 13(27), 21292–21351, 10.1039/d5ta02047f.
- C. Zheng, Z. Wang, J. Yuan, Q. Xu, H. Li and X. Lu, A Facile Synthesis of Highly Efficient In2S3 Photocatalysts for the Removal of Cationic Dyes with Size-dependent Photocatalysis, RSC Adv., 2023, 13, 4173–4181, 10.1039/d2ra07108h.
- A. Prakash, M. Dan, S. Yu, S. Wei, Y. Li, F. Wang and Y. Zhou, In2S3/CuS Nanosheet Composite: An Excellent Visible Light Photocatalyst for H2 Production from H2S, Sol. Energy Mater. Sol. Cells, 2018, 180, 205–212, DOI:10.1016/j.solmat.2018.03.011.
- A. V. Kokate, M. R. Asabe, S. D. Delekar, L. V. Gavali and I. S. Mulla, Photoelectrochemical Properties of Electrochemically Deposited CdIn2S4 Thin Films, J. Phys. Chem. Solid., 2006, 67, 2331–2336, DOI:10.1016/j.jpcs.2006.05.022.
- B. B. B. Kale, J. O. Baeg, S. M. Lee, H. Chang, S. J. Moon and C. W. Lee, CdIn2S4 Nanotubes and “ Marigold ” Nanostructures : A Visible-Light Photocatalyst, Adv. Funct. Mater., 2006, 16, 1349–1354, DOI:10.1002/adfm.200500525.
- D. Sun, W. Wang, C. Liu, X. Li, J. Zhou, Y. Ma and S. Ruan, A Comprehensive Optimization of the Sensitivity of CdS/CdIn2S4 Composites to Triethylamine Gas, Sens. Actuators, B, 2022, 373, 132722, DOI:10.1016/j.snb.2022.132722.
- D. Ma, J.-W. Shi, Y. Zou, A. Z. Fan, J. Shi, L. Cheng, D. Sun, Z. Wang and C. Niu, Multiple Carrier-Transfer Pathways in Flower-like In2S3/CdIn2S4/In2O3 Ternary Heterostructure for Enhanced Photocatalytic Hydrogen Production, Nanoscale, 2018, 10, 7860–7870, 10.1039/C8NR00170G.
- C. M. Aitchison, R. S. Sprick and A. I. Cooper, Emulsion Polymerization Derived Organic Photocatalysts for Improved Light-Driven Hydrogen Evolution, J. Mater. Chem. A, 2019, 7, 2490–2496, 10.1039/C8TA11383A.
- A. Raghavan, S. Sarkar, L. R. Nagappagari, S. Bojja, S. M. Venkatakrishnan and S. Ghosh, Decoration of Graphene Quantum Dots on TiO2 Nanostructures: Photosensitizer and Cocatalyst Role for Enhanced Hydrogen Generation, Ind. Eng. Chem. Res., 2020, 59, 13060–13068, DOI:10.1021/acs.iecr.0c01663.
- K. Kumar, A. Kumar, R. Police, J. Yeon, M. Kang and C. Byon, Rare Earth Metal Gd Influenced Defect Sites in N Doped TiO2: Defect Mediated Improved Charge Transfer for Enhanced Photocatalytic Hydrogen Production, Int. J. Hydrogen Energy, 2018, 43, 2073–2082, DOI:10.1016/j.ijhydene.2017.12.050.
- G. Sadanandam, K. Lalitha and V. Durga, Cobalt Doped TiO2 : A Stable and Efficient Photocatalyst for Continuous Hydrogen Production from Glycerol : Water Mixtures under Solar Light Irradiation, Int. J. Hydrogen Energy, 2013, 38(23), 9655–9664, DOI:10.1016/j.ijhydene.2013.05.116.
- Y. Yang, K. Ye, D. Cao, P. Gao, M. Qiu, L. Liu and P. Yang, Efficient Charge Separation from F− Selective Etching and Doping of of Anatase-TiO2{001} for Enhanced Photocatalytic Hydrogen Production, ACS Appl. Mater. Interfaces, 2018, 10(23), 19633–19638, DOI:10.1021/acsami.8b02804.
- B. Liu, S. Su, W. Zhou, Y. Wang, D. Wei, L. Yao, Y. Ni, M. Cao and C. Hu, Photo-Reduction Assisted Synthesis of W-Doped TiO2 Coupled with Au Nanoparticles for Highly Efficient Photocatalytic Hydrogen Evolution, CrystEngComm, 2017, 19(4), 675–683, 10.1039/c6ce02417c.
- B. Banerjee, V. Amoli, A. Maurya, A. K. Sinha and A. Bhaumik, Green Synthesis of Pt-Doped TiO2 Nanocrystals with Exposed (001) Facets and Mesoscopic Void Space for Photo-Splitting of Water under Solar Irradiation, Nanoscale, 2015, 7(23), 10504–10512, 10.1039/c5nr02097b.
- X. Ke, P. Wang, X. Wang, F. Chen and H. Yu, Reducing Electron Release of Mo Sites in Mo2C MXene Cocatalyst for Efficient Photocatalytic H2 Generation, Appl. Catal. B Environ. Energy, 2025, 365, 124936, DOI:10.1016/j.apcatb.2024.124936.
- S. Farhan, A. H. Raza, L. Li, S. Yang and Y. Wu, Designed 2D/2D F-Doped TiO2@ZnIn2S4 Heterojunction for Efficient Photo-Utilization Hydrogen Generation, J. Colloid Interface Sci., 2025, 681, 1–15, DOI:10.1016/j.jcis.2024.11.124.
- S. Li, J. Ye, Z. Fan, Y. Dai, Y. Xie, Y. Ling, M. Xu and Y. Wang, CoP Co-Catalyst Modification ZnIn2S4 Driving Efficient H2 Evolution under Visible Light, Sep. Purif. Technol., 2025, 358, 130294, DOI:10.1016/j.seppur.2024.130294.
| This journal is © The Royal Society of Chemistry 2026 |