An efficient and durable electrocatalyst for electrochemical water splitting: WCuBP micro-leaf-clusters
Sumiya Akter
Dristy
 ,
Shusen
Lin
,
Shusen
Lin
 ,
Shalmali
Burse
,
Shalmali
Burse
 ,
Md Ahasan
Habib
,
Md Ahasan
Habib
 ,
Mehedi
Hasan Joni
,
Md
Najibullah
and
Jihoon
Lee
,
Mehedi
Hasan Joni
,
Md
Najibullah
and
Jihoon
Lee
 *
*
Department of Electronic Engineering, College of Electronics and Information, Kwangwoon University, Nowon-gu, Seoul, 01897, South Korea. E-mail: jihoonlee@kw.ac.kr
First published on 24th November 2025
Abstract
Electrochemical water splitting is a promising approach for green hydrogen production, which can significantly contribute to carbon neutrality by reducing reliance on fossil fuels and minimizing harmful emissions. Developing cost-effective, efficient and durable electrocatalysts is essential for the widespread implementation of sustainable green hydrogen. Herein, a WCuBP micro-leaf cluster (MLC) is demonstrated for overall water splitting via a single-step hydrothermal approach followed by vacuum annealing for the first time. The WCuBP MLC electrocatalyst exhibits outstanding hydrogen evolution reaction (HER) and oxygen evolution reaction (OER) activity, with 51 and 140 mV overpotentials at 50 mA cm−2 in 1 M KOH. The bifunctional WCuBP‖WCuBP system outperforms the benchmark system with a low cell voltage of 2.27 V at 1000 mA cm−2. Furthermore, bifunctional WCuBP demonstrates superior long-term stability, operating continuously for over 120 hours without noticeable performance degradation. The hybrid system of Pt/C‖WCuBP exhibits a lower cell voltage of 2.03 V at 1000 mA cm−2, outperforming both the bifunctional and benchmark systems, showing the successful integration of WCuBP MLC on the anodic side. The superior electrochemical performance of WCuBP can be attributed to the synergistic interactions among W, Cu, B and P along with its high active surface area and enhanced charge transfer kinetics.
1. Introduction
The globe is getting hotter and hotter and people from many countries are suffering from ever increasing temperatures and the effects of El Niño and La Niña.1,2 The global climate crisis and environmental pollution caused by excessive fossil fuel consumption are prompting the need for clean energy sources like hydrogen.3–5 Green hydrogen provides a zero-carbon energy source with an energy density nearly three times higher than that of gasoline, making it a renewable, environmentally friendly and versatile alternative fuel.6,7 Electrochemical water splitting (EWS), driven by two half-reactions, the hydrogen evolution reaction (HER) on the cathode and the oxygen evolution reaction (OER) on the anode, can offer an environmentally sustainable and effective approach to generate high-purity hydrogen.8–10 Precious-metal-based electrocatalysts such as Pt/C and RuO2/IrO2 are widely used as standard electrodes to facilitate the HER and OER.11 However, overcoming the challenges of high cost and limited availability of these catalysts is crucial for the successful implementation of EWS in large-scale industrial applications.12 On the other hand, most research focuses on achieving high catalytic activity at relatively low current density, which often results in low activity at high current density (HCD) due to the high polarization condition and excessive gas generation that can hinder the electron transfer process.13,14 The study of HCD is crucial for practical applications, considering both the efficiency and economic benefits. Therefore, more efforts should be directed toward developing affordable, highly efficient and stable electrocatalysts that can perform effectively under HCD and harsh environmental conditions.Recently, numerous transition metals such as Co, Ni, Mo, Mn, Cu, etc. have been widely explored in combination with non-metals such as phosphides, selenides, sulfides and oxides. Their high intrinsic properties make them potential candidates for efficient and cost-effective EWS electrocatalysts.15–17 Among various transition metals, the tungsten-based materials can be considered as effective water-splitting catalysts owing to their natural abundance, remarkable stability, high electron mobility, large hole diffusion length and strong electrocatalytic activity.18 For example, W-NiCoP catalysts have multiple active electrocatalytic sites for water dissociation and H2 formation.19 At the same time, the incorporation of copper (Cu) as a transition metal component can increase the electron density across the metal centers, promoting faster charge transfer and improving electrocatalytic performance.20 Moreover, Cu based catalysts may offer numerous accessible active sites, efficient bubble release channels and high conductivity, leading to low overpotential for both HER and OER.21 Additionally, the incorporation of boron (B) and phosphorus (P) together may improve the chemical bonding, promoting the stability and catalytic activity of Cu-based catalysts by adjusting their structural and surface properties, which can help in water molecule activation for splitting.22,23 B can be considered as an effective water splitting component due to its efficient charge transfer ability and electron-donating nature, providing an electron-enriched active surface area to enhance the adsorption and desorption of H+ and OH− ions.24 Furthermore, P can improve conductivity and prevent both corrosion and material degradation during redox reactions.25 Moreover, the interaction between transition metals and phosphorus can enhance H2 dissociation, facilitating the formation of stable catalytic intermediates and preventing catalyst deactivation.26
In this work, a novel WCuBP micro-leaf cluster (MLC) electrode is developed for the first time using a one pot deposition method. Herein, the combination of W, Cu, B and P, along with the unique properties of each element can be a rational design for electrochemical water splitting applications. The optimal reaction conditions are determined by varying the reaction duration and temperature. Additionally, a post-annealing treatment is also performed on the hydrothermally prepared WCuBP, as shown in the schematic diagram of WCuBP synthesis in Fig. S2. The optimized WCuBP MLC electrode exhibits low overpotential values of 51 and 140 mV at 50 mA cm−2 in 1 M KOH for HER and OER. Furthermore, the WCuBP electrode shows comparable performance to the benchmark electrode across all pH media and natural water. The bifunctional WCuBP demonstrates a low cell voltage of 2.27 V at 1000 mA cm−2 in 1 M KOH, suggesting its efficiency in reducing energy consumption for high current density water electrolysis. Additionally, the hybrid Pt/C‖WCuBP configuration exhibits improved two-electrode performance compared to the bifunctional and benchmark systems in alkaline electrolytes with an ultra-low potential value of 2.03 V at 1000 mA cm−2. Overall, the WCuBP MLC electrode can accelerate water-splitting activity and it holds strong potential for practical implementation in sustainable energy systems.
2. Materials and methods
The WCuBP MLC electrode is synthesized via a single-step hydrothermal reaction on a bare nickel foam (NF) substrate. The NF-based electrocatalyst can provide an increased active surface area and improved charge transfer for enhanced electrochemical water-splitting performance.27 For the synthesis of WCuBP, the reaction parameters, including W concentration, hydrothermal reaction temperature, and duration, were systematically varied to determine the optimal precursor composition. The precursor containing 1.2 mM sodium tungstate dihydrate (Na2O4W·2H2O), 3 mM copper(II) sulfate (CuSO4), 3.6 mM boric acid (H3BO3), 8.4 mM sodium hypophosphite monohydrate (NaH2PO2·H2O), 18 mM urea (CH4N2O) and 2 mM ammonium fluoride (NH4F) dissolved in 30 ml DI water was found to be the optimal composition for fabricating the WCuBP electrode. The reaction was performed at 160 °C for 4 hours, resulting in the highest activity of the WCuBP electrode compared to other variations of the WCuBP electrode. The corresponding experimental results are shown in Fig. S7–S12. Later, the WCuBP electrocatalyst was post-annealed at 50 °C for 30 min, which led to enhanced crystallinity and improved electrocatalytic performance. The detailed fabrication discussion is provided in SI Section S-1.1–S-1.4.3. Results and discussion
3.1. Optimization of the WCuBP micro-leaf cluster (MLC) electrocatalyst
Fig. 1 shows the morphological analysis of the WCuBP electrocatalyst with variations in post-annealing duration. Post-annealing treatment was conducted to optimize the electrocatalyst performance by refining the atomic arrangement and minimizing the defects via surface adatom diffusion, which can enhance the conductivity and crystallinity. Initially, the annealing temperature was varied from 50 to 200 °C with a fixed duration of 60 min as shown in Fig. S13 and S14. The pronounced micro-leaf structure on the Ni foam remained stable below 150 °C. The leaf edges became rougher at a higher temperature of 200 °C, leading to a decrease in the number of active sites and poor electrochemical performance. The 50 °C sample exhibited the most favorable electrochemical activity in this set as shown in Fig. S14. Higher annealing temperatures resulted in agglomeration and lower performance, likely due to increased adatom diffusion. Such alterations in structure can decrease the electrochemically active surface area and reduce the number of accessible redox-active sites.28 The catalytic performance of the electrocatalyst is strongly influenced by the interactions at active sites involved in hydrogen and oxygen evolution. The gas generation mechanism of the HER mainly depends on the coverage of adsorbed hydrogen (Had). At low Had coverage, Had tends to react with protons and electrons simultaneously because the reactive centers are adjacent to the adsorbed hydrogen on the cathode surface, leading to the Volmer–Heyrovsky reaction, i.e., H2O + e− → Had + OH− (Volmer) and H2O + e− → H2 + OH− (Heyrovsky). In contrast, when the coverage of Had is high, the numerous Had species tend to combine with each other, resulting in the Tafel step, i.e., 2Had → H2 (Tafel).29 In terms of the OER reaction, M–OH species form through the one-electron oxidation of hydroxide anions (OH−) adsorbed on the metallic active sites, which then transform into M–O after the removal of electrons. The reactions are as follows: OH− + M = M–OH + e− and M–OH + OH− = M–O + H2O + e−.30 The M–O species can then follow two different pathways to generate O2: 2M–O → 2M + O2 or M–O + OH− = M-OOH + e−; M-OOH + OH− → O2 + H2O + e− + M. The composition and optimization of catalyst materials can influence their activity, durability and overall effectiveness in hydrogen and oxygen evolution reactions. For example, W and Cu serve as active sites for hydroxide adsorption and oxidation during the oxygen evolution reaction. Tungsten can offer high conductivity, stable redox behavior, and high affinity for reaction intermediates and promote water dissociation on the catalyst surface.31,32 Copper can exhibit multiple oxidation states with low energy barriers, enabling efficient electron transport and influencing the adsorption of key reaction intermediates during both HER and OER.33 Furthermore, boron can introduce electron-deficient sites that promote water adsorption and support reactive oxygen species on the catalyst surface.34 Phosphorus can promote water dissociation by trapping protons and enhance catalytic activity through charge redistribution and surface reconstruction.35 These elements can establish a synergistic catalytic environment that may help to balance intermediate binding energies and accelerate both hydrogen and oxygen evolution reactions, enabling efficient overall water splitting.Furthermore, the post-annealing duration variation set exhibited the highest performance compared to other variations. The annealing duration was varied from 15 to 120 min, as shown in Fig. 1(a)–(d), where the WCuBP electrocatalyst exhibited a micro-leaf cluster morphology. The WCuBP electrode post-annealed for 30 min exhibited optimal performance. The Raman analysis of WCuBP demonstrated vibrational peaks at 144, 215, 291, 536 and 905 cm−1, as depicted in Fig. 1(e). Reference data for WCuBP are limited due to its unique composition. Literature reports suggest that the observed Raman peak at 144 cm−1 is commonly associated with lattice vibrations and the peak at 215 may correspond to O–W–O stretching.36 The Raman peak at 291 cm−1 might be closely associated with the Ag vibrational characteristic of CuO.37 The Raman band at 905 cm−1 might correspond to the [PO4]3− ligand.22 The intensity trends of the Raman peaks are presented as contour plots, as shown in Figures 1(e-1)–(e-3). The 30 min sample exhibited the highest peak intensity, indicating well-defined crystal structures compared to other samples in this set. Furthermore, EDS top-view phase mapping was performed on the optimal 30 min sample as shown in Fig. 1(f)–(f-4). This analysis clearly demonstrates the uniform distribution of W Lλ, Cu Kα, B Kα, and P Kα elements throughout the micro-leaf structure. Additionally, the line profiles in Fig. 1(g)–(g-4) also confirm the distribution of these elements across the micro-leaf cluster.
3.2. Characterization of the best WCuBP electrode
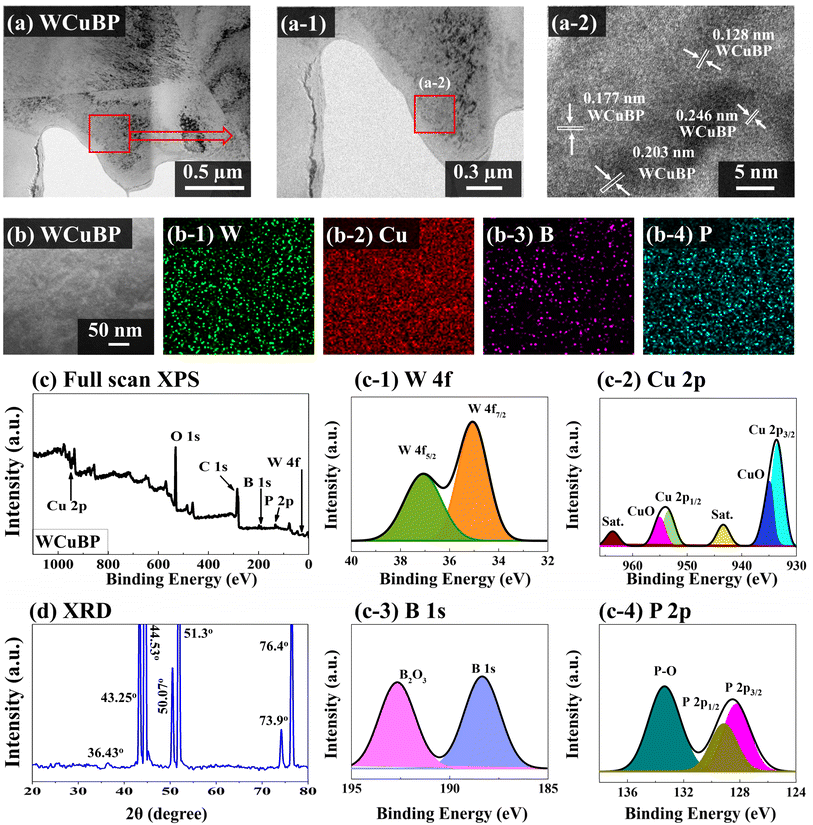
Fig. 2 presents the elemental composition, and structural and morphological analysis of the optimized WCuBP MLC electrodes. High-resolution transmission electron microscopy (HR-TEM) images of WCuBP are shown in Fig. 2(a)–(a-2), where the distinct lattice fringes with spacings of 0.128, 0.177, 0.203 and 0.246 nm correspond to the WCuBP composite. Multiple crystalline orientations confirm the polycrystalline nature of the WCuBP electrode. The TEM phase maps in Fig. 2(b)–(b-4) reveal a homogeneous distribution of W, Cu, B and P deep inside the micron-scale regions of the sample. The majority of the surface was composed of W and Cu with a significantly lower concentration of B and P as determined from the elemental percentage tables from the EDS analysis of WCuBP presented in Fig. S15.Furthermore, X-ray photoelectron spectroscopy (XPS) analysis was performed to investigate the surface elemental states and chemical composition of WCuBP. The full scan XPS spectrum in Fig. 2(c) confirms the presence of W, Cu, B and P on the catalyst surface. High-resolution deconvolution spectra of WCuBP are shown in Figures 2(c-1)–(c-4). All XPS peaks were compared with standard reference data from the XPS handbook to accurately determine the elemental states.38 In the W 4f spectrum, two strong peaks corresponding to W 4f7/2 and W 4f5/2 were observed at 35.0 and 37.1 eV, respectively, as shown in Figure 2(c-1). In the XPS handbook, pristine W 4f7/2 and W 4f5/2 peaks were identified at 31.4 and 33.58 eV.38 In comparison with the pristine peaks, the observed W 4f7/2 and W 4f5/2 peaks in Figure 2(c-1) shifted positively by 3.6 and 3.52 eV, indicating electron donation during the reaction. In a previous study, W 4f7/2 and W 4f5/2 peaks were observed at 34.84 and 37.03 eV for the WCoC2O4 electrocatalyst.39 In the Cu 2p spectrum (Figure 2(c-2)), the binding energies (BEs) at 933.4 and 953.2 eV are associated with Cu 2p3/2 and 2p1/2 peaks. The Cu 2p3/2 and Cu 2p1/2 peaks exhibited positive shifts of 0.7 and 0.71 eV, respectively, from their pristine values of 932.7 and 952.5 eV,38 indicating electron release from the Cu atoms. The peaks at 935.2 and 954.9 eV are linked to Cu2+ 2p3/2 and 2p1/2, suggesting the formation of CuO on the WCuBP surface due to air exposure.40 Previously Cu 2p3/2 and Cu 2p1/2 peaks were reported at 932.6 and 953.8 eV for B-CuCo2O4/NiO.41 The shakeup satellite peaks were found at 943.5 and 963.7 eV.22 In the B 1s spectrum (Figure 2(c-3)), the BEs at 188.33 and 192.66 eV were attributed to the B 1s elemental state and B–O bond formation. Here, B 1s peak exhibited a 1.03 eV shift towards higher binding energy from its initial position (187.3 eV),38 indicating electron transfer from B atoms to other elements. The interaction of B with other elements suggests orbital hybridization, leading to the formation of a metal–boron bond.42 The presence of the trivalent boron oxide (B2O3) peak at 192.66 eV can be due to surface oxidation.43 In the P 2p spectrum, the deconvoluted P 2p3/2 and 2p1/2 peaks were observed at 128.2 and 129.05 eV, respectively, as shown in Figure 2(c-4). The P 2p3/2 and P 2p1/2 peaks showed negative shifts of 1.7 and 1.69 eV, respectively, compared to their pristine positions at 129.9 and 130.74 eV, suggesting electron acceptance. In a previous study, the Ni-CoP/Co2P@NC electrocatalyst showed two strong P 2p3/2 and P 2p1/2 peaks at 129.3 and 130.2 eV.44The peak at 133.3 eV is associated with the formation of P–O bonds.45 Overall, the electron transfer between W, Cu, B and P suggests strong bond formation and successful synthesis of WCuBP composites.
The structural properties of the optimal WCuBP electrode were characterized by X-ray diffraction (XRD). No comparable XRD data for the WCuBP electrocatalyst were available in the current literature or databases, demonstrating its unique properties and polycrystalline phase, which aligns with TEM analysis. Multiple peaks at 36.43°, 43.25°, 50.07° and 73.9° were observed in the XRD analysis of WCuBP as shown in Fig. 2(d). The sharp peaks at 44.53°, 51.3° and 76.4° may correspond to (111), (200) and (220) planes of nickel foam.46 Notably, the polycrystalline phase can provide more active sites than the single-phase structure, making it particularly good for the high-rate water splitting process.47 Additionally, polycrystal structures can demonstrate improved stability as multiple grains can help distribute stress and strain more effectively.48
3.3. Electrochemical performance of the WCuBP MLC electrode
Fig. 3 shows the 3-electrode (3-E) electrochemical characterization of the post-annealing time variation set in 1 M KOH. The HER/OER polarization curves in Fig. 3(a) and (f) exhibit a performance trend of 30 > 15 > 60 > 120 min. The specific HER and OER overpotential values at 50 mA cm−2 are shown in the overpotential bar charts in Figures 3(a-1) and (f-1). The 30 min WCuBP electrode demonstrated optimal HER and OER activities, with overpotential values of only 51 and 140 mV, respectively, at 50 mA cm−2. The oxidation peak observed in the OER measurement can be associated with the transformation of Cu0/CuIII and multiple oxidation states of W.49,50 The detailed analysis is provided in Fig. S21 and SI Section S-1.8. The reaction kinetics of WCuBP were investigated using the Tafel slopes for HER and OER as shown in Fig. 3(b) and (g), based on the equation η = a + b![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) log|j|, where η is the overpotential, a is the Tafel constant, b is the Tafel slope and j is the current density.51 The 30 min WCuBP sample exhibited the lowest Tafel slope of 51 for HER and 180 mV dec−1 for OER, indicating excellent reaction kinetics. The HER Tafel slope value of WCuBP falls within the well-known range of 40 to 120 mV dec−1, implying the rate-determining step is the Heyrovsky step with the potential involvement of the Volmer step.52 The slope value exceeding 120 mV dec−1 for OER indicates the involvement of more complex processes, such as the formation of M–OH active sites or M–O species.22 Electrochemical impedance spectroscopy (EIS) was further conducted to analyze the charge transfer properties of HER and OER at applied potentials of 0.01 V and 1.34 V, respectively as shown in Fig. 3(c) and (h). Charge transfer resistance (Rct) represents the impedance at the catalyst–electrolyte interface, determined from the semicircle of the Nyquist plots.53 The 15, 30, 60 and 120 min WCuBP samples exhibited HER EIS values of 40.2, 29.8, 55.2 and 64.9 Ω and OER EIS values of 21.5, 18.8, 24.9 and 30.1 Ω, respectively. Among them, the 30 min sample showed the lowest impedance, indicating higher electrical conductivity. HER/OER CV measurements along with anodic and cathodic current density plots are shown in Fig. S16. The double-layer capacitance (Cdl) for HER and OER was calculated from the slope values of the anodic and cathodic current density plots for WCuBP post-annealing time variation electrodes, as shown in Fig. 3(d) and (i). The 30 min sample demonstrated the best electrochemical properties with Cdl values of 30.7 for HER and 43.4 mF cm−2 for OER in this set. Subsequently, the electrochemical surface area (ECSA) was determined using the Cdl values.54 The ECSA of the 30 min WCuBP was calculated to be 191.8 and 271.2 cm2 for the HER and OER, respectively based on the equation ECSA = A × Cdl/Cs.55 Here, A represents the geometrical surface area of CuBP (0.25 cm2) and Cs is the specific capacitance, typically taken as 0.04 mF cm−2 in 1 M KOH solution.56 The high ECSA values of the 30 min WCuBP sample in Fig. S18 indicate a larger number of active sites, likely due to the interconnected ultrathin micro-leaves and strong interactions between W and Cu sites. Turnover frequency (TOF) calculations were conducted to assess the intrinsic activity of WCuBP electrodes, considering all metallic W and Cu atoms as active sites, as shown in Fig. 3(e) and (j) and SI Section S-1.6. The 30-min WCuBP exhibited the highest HER/OER TOF values of 5.60/2.80 per site per s compared to the 15-, 60- and 120-min electrodes. To provide clearer insight into the effect of annealing, HER/OER LSV comparison of WCuBP before and after annealing is provided in Fig. S19, where the annealed sample exhibited better electrochemical activity. Additionally, the performance comparison of WCuBP and CuBP along with benchmark electrodes can be found in Fig. S20. The optimal WCuBP electrocatalyst demonstrated improved HER and OER activity compared to the CuBP electrocatalyst, which can be attributed to the synergistic contributions of each element.57
log|j|, where η is the overpotential, a is the Tafel constant, b is the Tafel slope and j is the current density.51 The 30 min WCuBP sample exhibited the lowest Tafel slope of 51 for HER and 180 mV dec−1 for OER, indicating excellent reaction kinetics. The HER Tafel slope value of WCuBP falls within the well-known range of 40 to 120 mV dec−1, implying the rate-determining step is the Heyrovsky step with the potential involvement of the Volmer step.52 The slope value exceeding 120 mV dec−1 for OER indicates the involvement of more complex processes, such as the formation of M–OH active sites or M–O species.22 Electrochemical impedance spectroscopy (EIS) was further conducted to analyze the charge transfer properties of HER and OER at applied potentials of 0.01 V and 1.34 V, respectively as shown in Fig. 3(c) and (h). Charge transfer resistance (Rct) represents the impedance at the catalyst–electrolyte interface, determined from the semicircle of the Nyquist plots.53 The 15, 30, 60 and 120 min WCuBP samples exhibited HER EIS values of 40.2, 29.8, 55.2 and 64.9 Ω and OER EIS values of 21.5, 18.8, 24.9 and 30.1 Ω, respectively. Among them, the 30 min sample showed the lowest impedance, indicating higher electrical conductivity. HER/OER CV measurements along with anodic and cathodic current density plots are shown in Fig. S16. The double-layer capacitance (Cdl) for HER and OER was calculated from the slope values of the anodic and cathodic current density plots for WCuBP post-annealing time variation electrodes, as shown in Fig. 3(d) and (i). The 30 min sample demonstrated the best electrochemical properties with Cdl values of 30.7 for HER and 43.4 mF cm−2 for OER in this set. Subsequently, the electrochemical surface area (ECSA) was determined using the Cdl values.54 The ECSA of the 30 min WCuBP was calculated to be 191.8 and 271.2 cm2 for the HER and OER, respectively based on the equation ECSA = A × Cdl/Cs.55 Here, A represents the geometrical surface area of CuBP (0.25 cm2) and Cs is the specific capacitance, typically taken as 0.04 mF cm−2 in 1 M KOH solution.56 The high ECSA values of the 30 min WCuBP sample in Fig. S18 indicate a larger number of active sites, likely due to the interconnected ultrathin micro-leaves and strong interactions between W and Cu sites. Turnover frequency (TOF) calculations were conducted to assess the intrinsic activity of WCuBP electrodes, considering all metallic W and Cu atoms as active sites, as shown in Fig. 3(e) and (j) and SI Section S-1.6. The 30-min WCuBP exhibited the highest HER/OER TOF values of 5.60/2.80 per site per s compared to the 15-, 60- and 120-min electrodes. To provide clearer insight into the effect of annealing, HER/OER LSV comparison of WCuBP before and after annealing is provided in Fig. S19, where the annealed sample exhibited better electrochemical activity. Additionally, the performance comparison of WCuBP and CuBP along with benchmark electrodes can be found in Fig. S20. The optimal WCuBP electrocatalyst demonstrated improved HER and OER activity compared to the CuBP electrocatalyst, which can be attributed to the synergistic contributions of each element.57
Faradaic efficiency (FE) is a key metric used to evaluate the efficiency of electron transfer during electrochemical reactions. It represents the ratio between the experimentally measured amount of H2 or O2 and the theoretically estimated amount. The water–gas displacement setup used for FE measurements is provided in Fig. S25. The FE of the WCuBP electrode was determined to be 96.0% for H2 and 95.74% for O2, as illustrated in Fig. 4(a). Detailed calculations for FE are provided in the SI Section S-1.7. The optimized WCuBP MLC electrode was compared with top-performing electrodes for both HER and OER as shown in Fig. 4(b) and (c). The performance comparison at 50 mA cm−2 demonstrates that the WCuBP MLC electrode is highly comparable to leading state-of-the-art catalysts. The overall HER/OER comparison can be found in Tables 1 and 2.
| Electrocatalysts | Overpotential [mV] at 50 mA cm−2 | Overpotential [mV] at 300 mA cm−2 | Reference |
|---|---|---|---|
| FeNiSe | 138 | 425 | 75 |
| WCuBP | 140 | 350 | This work |
| Ni-B-P | 150 | 360 | 76 |
| Cr/NiMoB | 158 | 386 | 77 |
| NiCoP | 202 | 240 | 78 |
| V-FeBP | 215 | 330 | 79 |
| Fe2P/Co2N | 220 | 268 | 80 |
| FeCoRuP | 222 | — | 81 |
| RuNiCoP/NF | 235 | — | 71 |
| W3CoB3 | 248 | — | 70 |
| Ru/CuMnBP | 262 | 421 | 22 |
3.4. WCuBP MLC performance in different pH media
Fig. 4 shows the 3-E and 2-E electrochemical performance of the best-performing WCuBP electrode in different pH electrolytes compared with the benchmark electrodes. The different pH solutions were prepared by dissolving 1 M KOH (alkaline), 0.5 M H2SO4 (acidic) and 1 M PBS (neutral) in 30 ml of DI water. Pt/C and RuO2 electrodes were used as benchmarks for HER and OER, respectively. The 3-E HER/OER performance was tested in different pH solutions as shown in Fig. 4(d)–(i). The WCuBP electrode demonstrated slightly lower performance than the HER benchmark (Pt/C) but significantly outperformed the OER benchmark electrode (RuO2) in all tested media. For HER, the WCuBP exhibited overpotential values of 251, 391 and 344 mV at 500 mA cm−2 in 1 M KOH, 0.5 M H2SO4 and 1 M PBS solutions while the Pt/C showed 208, 216 and 272 mV values. In the case of OER, the WCuBP MLC electrode demonstrated significantly lower OER overpotentials of 394, 864 and 801 mV at 500 mA cm−2 in alkaline, acidic and neutral media surpassing the 587, 905 and 1098 mV values of benchmark RuO2 as shown in Fig. 4(g)–(i). The alkaline environment exhibited the highest electrocatalytic activity due to its ability to facilitate the formation and stabilization of reactive intermediates during the reaction, enabling more efficient electron transfer processes and faster reaction rates.58 Additionally, the reaction pathways under alkaline conditions favor the participation of hydroxide ions (OH−) over protons (H+). This involvement of hydroxide ions results in more favorable reaction conditions and lower overpotentials for the overall water-splitting process.59 The electrocatalyst was less effective in acidic and neutral solutions compared to the alkaline solution. The degradation observed in the acidic solution is attributed to the strong corrosive nature of acidic electrolytes, which can damage the catalyst material and reduce the active surface area.60 In PBS solutions, the lack of conductive ions largely hinders the charger transfer and transport.61 The repeatability test of HER and OER performance in 1 M KOH is shown in Fig. S22, where no significant variation is observed even after 2000 CV cycles. Chronoamperometry (CA) measurements in alkaline media for HER and OER at different applied voltages are shown in Fig. S23 and S24. The steady-state current densities measured over extended periods demonstrate the excellent durability of WCuBP. Furthermore, the HER/OER activity of WCuBP was compared with that of bare NF in KOH as shown in Fig. S26, demonstrating the enhanced catalytic performance of the WCuBP electrocatalyst.Subsequently, the two-electrode (2-E) system was configured using the optimal WCuBP MLC electrode as both the anode and cathode, namely WCuBP‖WCuBP. The benchmark system was Pt/C‖RuO2 where Pt/C functioned as the cathode and RuO2 as the anode. The overall 2-E water splitting performance in different pH electrolytes is shown in Fig. 4(j)–(l). In alkaline media, WCuBP‖WCuBP exhibited a low cell voltage of 1.89 V compared to 2.04 V for the RuO2‖Pt/C benchmark system at 500 mA cm−2. Moreover, the 2-E system demonstrated better performance than the benchmark electrodes in both acidic and neutral media. Overall, the results demonstrate the robust electrocatalytic properties of WCuBP, showcasing its potential for applications under different pH conditions.
3.5. WCuBP MLC high current performance
Recently, significant efforts have been made to develop bifunctional electrocatalysts with superior electrocatalytic activity for overall water splitting applications. However, most of them operate at a relatively low current density. To meet the practical application requirements, an electrode with excellent performance at a high current density (HCD) (>500 mA cm−2) is essential, considering both efficiency and industrial demand. Moreover, the build-up of large amounts of H2/O2 gas at the electrode–electrolyte interface hinders the electrode transfer process and reduces the reactive surface area.62 Therefore, there is an urgent need to explore efficient electrocatalysts for overall water splitting under HCD conditions. In this work, the current density was extended up to 1000 mA cm−2 as seen in Fig. 5(a), to examine its performance at high current densities. The dual-functional WCuBP exhibited a cell voltage of 2.27 V at 1000 mA cm−2 in 1 M KOH. The electrocatalytic performance improved slightly to 2.21 V cell voltage at the same current density in 6 M KOH solution. Stability is another critical factor in determining the potential for practical application. Continuous operation of the WCuBP electrode for 120 hours demonstrated stable performance at 1000 mA cm−2 as shown in Fig. 5(b). Furthermore, the 2-E CA response was conducted at different potentials as shown in Fig. 5(c), indicating effective gas bubble release. Given the exceptionally high OER performance of WCuBP (Fig. 4(g)), a hybrid system was designed using Pt/C as the cathode and WCuBP as the anode. As expected, the Pt/C‖WCuBP configuration outperformed both the bifunctional WCuBP‖WCuBP and the benchmark Pt/C‖RuO2 systems in all pH media, as shown in Fig. 5(d)–(f). The hybrid system exhibited a lower cell voltage of 1.83 V at a current density of 500 mA cm−2 in KOH solution, compared to 2.04 V for the benchmark. In acidic and PBS solutions, the hybrid system demonstrated 2.31 V and 2.30 V, respectively at the same current density. The overall pH-dependent performance trend of the hybrid system is consistent with that of the 3-E and 2-E systems. Furthermore, the LSV results of the hybrid configuration were obtained up to 1000 mA cm−2 in both 1 M and 6 M KOH, as shown in Fig. 5(g). The hybrid system demonstrated improved energy efficiency in 1 and 6 M KOH, with only 1.98 and 2.03 V cell voltages, respectively, at a current density of 1000 mA cm−2. Additionally, the Pt/C‖WCuBP configuration showed stable CA measurements at various current densities such as 200, 600 and 1000 mA cm−2 in 6 M KOH at 60 °C as seen in Fig. 5(h), confirming the exceptional long-term durability of the WCuBP electrode. Fig. 5(i) shows a comparison of the bifunctional performance of WCuBP with leading electrocatalysts in 1 M KOH at a current density of 50 mA cm−2. A summary table of the 2-E performance compared with the state of the art electrodes can be seen in Table 3. Furthermore, the WCuBP electrode was tested in natural seawater (SW) and river water (RW) as shown in Fig. S27. In river water, both WCuBP and benchmark electrodes exhibited low activity due to the lack of conductive ions for the electrochemical reactions, which decreased the rate of redox reactions and negatively impacted the overall performance.63 In SW, the WCuBP electrode showed lower performance than the benchmark electrode at low current density but performed better at high current density. However, the presence of various ions and salts in SW can significantly impact electrochemical reactions.64 These ions may participate in the reactions or alter the reaction kinetics, resulting in a low electrocatalytic behavior.65 Moreover, chlorine evolution often competes with oxygen evolution during seawater electrolysis due to its lower electrode potential.66 To overcome these challenges, the addition of 1 M KOH in SW improves electrolyte conductivity and increases the availability of reactants for the water-splitting process, as shown in Fig. S27(b). Overall, WCuBP demonstrates effectiveness in natural water electrolysis, suggesting its suitability for practical water-splitting technologies.| Electrocatalysts | Electrolyte solution | Overpotential [V] at 50 mA cm−2 | Reference |
|---|---|---|---|
| FeNiSe | 1 M KOH | 1.36 | 75 |
| Ru/CoFe2O4 | 1 M KOH | 1.45 | 82 |
| WCuBP | 1 M KOH | 1.46 | This work |
| V/FeBP | 1 M KOH | 1.48 | 79 |
| NiMoO4 | 1 M KOH | 1.51 | 83 |
| W3CoB3 | 1 M KOH | 1.52 | 70 |
| Co/CoMoN | 1 M KOH | 1.52 | 84 |
| FeP/Ni2P | 1 M KOH | 1.55 | 85 |
| NiMoSe | 1 M KOH | 1.60 | 86 |
| NiMoB | 1 M KOH | 1.61 | 72 |
| CoP@NiFe | 1 M KOH | 1.63 | 87 |
| CoWBP | 1 M KOH | 1.69 | 88 |
Additionally, the post-stability characterization of the WCuBP electrode, including SEM, XRD, XPS and Raman analysis can be found in SI Fig. S28. Comprehensive post-stability analyses demonstrate that the WCuBP electrocatalyst exhibits strong and robust material properties, where surface oxidation resulting from extended redox processes can serve as active sites to enhance both stability and corrosion resistance. Detailed analysis is provided in SI Section S-1.9.
4. Conclusion
In summary, the WCuBP MLC electrocatalyst was successfully synthesized using a single-step hydrothermal method. The fabrication process involved precise adjustment of various parameters, including W concentration, reaction time, temperature and post-annealing treatment to ensure optimal performance. The WCuBP exhibited remarkable water splitting performance with overpotential values of 51 mV for HER and 140 mV for OER at 50 mA cm−2 in 1 M KOH. The bifunctional WCuBP electrode exhibited a low cell voltage of 2.27 V at a high current density of 1000 mA cm−2 for overall water splitting. Moreover, the WCuBP electrode maintained stable performance across all pH media and demonstrated enhanced stability under industrial electrolyte conditions (6 M KOH at 60 °C) for 120 hours. The hybrid Pt/C‖WCuBP system further exhibited improved performance, with a potential value of 2.03 V at 1000 mA cm−2 in alkaline solution. Overall, the superior electrocatalytic performance of the WCuBP electrode offers a promising solution for efficient water splitting and highlights its suitability for electrochemical energy system applications.Author contributions
Sumiya Akter Dristy: investigation, data curation, formal analysis, writing – original draft. Shusen Lin: investigation, data curation. Shalmali Burse: investigation, data curation. Md Ahasan Habib: investigation, data curation. Mehedi Hasan Joni: investigation, data curation. Md Najibullah: investigation, data curation. Jihoon Lee: supervision, methodology, writing – review & editing, project administration, funding acquisition, conceptualization.Conflicts of interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.Data availability
Data will be made available on request.Supplementary information (SI) is available. See DOI: https://doi.org/10.1039/d5ta07524f.
Acknowledgements
This research was supported by the Basic Science Research Program through the National Research Foundation of Korea (NRF) funded by the Ministry of Education (No. RS-2018-NR031063) and in part by a research grant from Kwangwoon University in 2025.References
- Z. Xu and Z.-S. Wu, eScience, 2025, 5, 100334 CrossRef.
- I. Ullah and M. Zaheer, Int. J. Hydrogen Energy, 2025, 105, 179–188 CrossRef CAS.
- H. Ma, Z. Chen, Z. Wang, C. V. Singh and Q. Jiang, Adv. Sci., 2022, 1–9 Search PubMed.
- C. Guo, Y. Shi, S. Lu, Y. Yu and B. Zhang, Chin. J. Catal., 2021, 42, 1287–1296 CrossRef CAS.
- C. Chen, Y. Tuo, Q. Lu, H. Lu, S. Zhang, Y. Zhou, J. Zhang, Z. Liu, Z. Kang, X. Feng and D. Chen, Appl. Catal., B, 2021, 287, 119953 CrossRef CAS.
- S. Chen, T. Zhang, J. Han, H. Qi, S. Jiao, C. Hou and J. Guan, Nano Res. Energy, 2024, 3, e9120106 CrossRef.
- W. Xiao, Y. Chen, Q. Zhao, D. Bukhvalov, C. Wang and X. Yang, Chin. Chem. Lett., 2024, 35, 110176 CrossRef CAS.
- R.-T. Liu, Z.-L. Xu, F.-M. Li, F.-Y. Chen, J.-Y. Yu, Y. Yan, Y. Chen and B. Y. Xia, Chem. Soc. Rev., 2023, 52, 5652–5683 RSC.
- K. Wang, Y. Guo, Z. Chen, D. Wu, S. Zhang, B. Yang and J. Zhang, InfoMat, 2022, 4, e12251 CrossRef CAS.
- B. Arumugam, E. E. Siddharthan, P. Mannu, R. Thapa, C.-L. Dong, A. A. Jeffery and S.-C. Kim, J. Mater. Chem. A., 2025, 13, 6749–6767 RSC.
- Q. Guo, X. Liu, C. Ke, W. Xiao and X. Yang, New J. Chem., 2023, 47, 21732–21742 RSC.
- M. Zhao, H. Li, W. Li, J. Li, L. Yi, W. Hu and C. M. Li, Chem.–Eur. J., 2020, 26, 17091–17096 CrossRef CAS.
- J. Wang, F. Xu, H. Jin, Y. Chen and Y. Wang, Adv. Mater., 2017, 29, 1605838 CrossRef PubMed.
- Y. Luo, Z. Zhang, M. Chhowalla and B. Liu, Adv. Mater., 2022, 34, 1–18 Search PubMed.
- J. Chen, J. Liu, J. Q. Xie, H. Ye, X. Z. Fu, R. Sun and C. P. Wong, Nano Energy, 2019, 56, 225–233 CrossRef CAS.
- C. Tang, R. Zhang, W. Lu, L. He, X. Jiang, A. M. Asiri and X. Sun, Adv. Mater., 2017, 29, 1602441 CrossRef.
- J. Cao, H. Li, J. Pu, S. Zeng, L. Liu, L. Zhang, F. Luo, L. Ma, K. Zhou and Q. Wei, Int. J. Hydrogen Energy, 2019, 44, 24712–24718 CrossRef CAS.
- S. Ghosh, S. R. Kadam, S. Kolatkar, A. Neyman, C. Singh, A. N. Enyashin, R. Bar-Ziv and M. Bar-Sadan, ACS Appl. Mater. Interfaces, 2022, 14, 581–589 CrossRef CAS.
- S. S. Lu, L. M. Zhang, Y. W. Dong, J. Q. Zhang, X. T. Yan, D. F. Sun, X. Shang, J. Q. Chi, Y. M. Chai and B. Dong, J. Mater. Chem. A, 2019, 7, 16859–16866 RSC.
- C. Luo, X. Liu, K. Yang, J. Xu, Z. Zhu, Z. Tang, S. Shen, J. Fan, D. Luo, N. A. H. Alshammari, Z. M. El-Bahy, X. Xu, Y. Xue and L. Pan, Coord. Chem. Rev., 2024, 516, 215936 CrossRef CAS.
- A. S. Sabir, E. Pervaiz, R. Khosa and U. Sohail, RSC Adv., 2023, 13, 4963–4993 RSC.
- S. Lin, R. Mandavkar, M. A. Habib, S. A. Dristy, M. H. Joni, J.-H. Jeong and J. Lee, J. Colloid Interface Sci., 2025, 677, 587–598 CrossRef CAS PubMed.
- L. Liu, L. Ge, Y. Sun, B. Jiang, Y. Cheng, L. Xu, F. Liao, Z. Kang and M. Shao, Nanoscale, 2019, 11, 6394–6400 RSC.
- S. Gupta, M. K. Patel, A. Miotello and N. Patel, Adv. Funct. Mater., 2019, 30, 1906481 CrossRef.
- S. A. Dristy, S. Lin, M. A. Habib, M. H. Joni, R. Mandavkar and J. Lee, Int. J. Hydrogen Energy, 2024, 96, 321–332 CrossRef.
- Y. Li, R. Li, D. Wang, H. Xu, F. Meng, D. Dong, J. Jiang, J. Zhang, M. An and P. Yang, Int. J. Hydrogen Energy, 2021, 46, 5131–5149 CrossRef CAS.
- W. Xiao, Y. Zhang, C. Ke, Q. Zhao, F. Han, J. Guo and X. Yang, Nanoscale, 2024, 16, 22350–22359 RSC.
- A. Alghannam, C. L. Muhich and C. B. Musgrave, Phys. Chem. Chem. Phys., 2017, 19, 4541–4552 RSC.
- A. Raveendran, M. Chandran and R. Dhanusuraman, RSC Adv., 2023, 13, 3843–3876 RSC.
- C. Hu, L. Zhang and J. Gong, Energy Environ. Sci., 2019, 12, 2620–2645 RSC.
- X. Duan, M. G. Sendeku, D. Zhang, D. Zhou, L. Xu, X. Gao, A. Chen, Y. Kuang and X. Sun, Acta Phys.-Chim. Sin., 2024, 40, 2303055 CrossRef.
- M. A. Habib, S. A. Dristy, S. Lin, M. H. Joni, M. Najibullah, R. Mandavkar and J. Lee, Adv. Mater. Technol., 2025, 10, 70021 CrossRef CAS.
- M. Devi, S. Hussain, M. Zimik, B. Moorthy and R. Thangavel, Int. J. Hydrogen Energy, 2025, 137, 333–341 CrossRef CAS.
- Y. Hong, J. Choi, E. Lee and Y. J. Hwang, Nanoscale, 2024, 16, 11564–11574 RSC.
- L. Kumar, B. Antil, A. Kumar, M. R. Das, O. López-Estrada, S. Siahrostami and S. Deka, ACS Appl. Mater. Interfaces, 2023, 15, 54446–54457 CrossRef CAS.
- H. Yoon, M. G. Mali, M. Kim, S. S. Al-Deyab and S. S. Yoon, Catal. Today, 2016, 260, 89–94 CrossRef CAS.
- M. Rashad, M. Rüsing, G. Berth, K. Lischka and A. Pawlis, J. Nanomater., 2013, 2013, 714853 CrossRef.
- D. Briggs, Handbook of Adhesion, 2nd edn, 2005, pp. 621–622 Search PubMed.
- K. Rajan, D. Thiruvengadam, M. Raj Kumar, K. Santhosh Kumar, J. Jayabharathi and M. Padmavathy, ACS Appl. Eng. Mater., 2025, 3, 1340–1355 CrossRef CAS.
- S. Zeng, D. Qu, H. Sun, Y. Chen, J. Wang, Y. Zheng, J. Pan, J. Cao and C. Li, ACS Appl. Mater. Interfaces, 2024, 16, 64797–64806 CrossRef CAS PubMed.
- G. Anandha babu, S. Perumal, M. K. A. Mohammed, M. Govindasamy, A. A. Alothman, M. Ouladsmane and R. Ganesan, Int. J. Hydrogen Energy, 2024, 54, 652–664 CrossRef CAS.
- R. Kulkarni, L. P. Lingamdinne, Z. A. Sheikh, V. D. Chavan, R. E. Ustad, S. R. Patil, J. R. Koduru and Y.-Y. Chang, Chem. Eng. J., 2024, 486, 150352 CrossRef CAS.
- H. Li, Y. Wang, C. Liu, S. Zhang, H. Zhang and Z. Zhu, Int. J. Hydrogen Energy, 2022, 47, 20718–20728 CrossRef CAS.
- H. Chu, P. Feng, B. Jin, G. Ye, S. Cui, M. Zheng, G.-X. Zhang and M. Yang, Chem. Eng. J., 2022, 433, 133523 CrossRef CAS.
- S. A. Dristy, S. Burse, M. A. Habib, S. Lin, M. H. Joni, M. Najibullah, R. Mandavkar, Y.-U. Chung and J. Lee, J. Environ. Chem. Eng., 2025, 13, 117400 CrossRef CAS.
- X. Hu, X. Tian, Y. W. Lin and Z. Wang, RSC Adv., 2019, 9, 31563–31571 RSC.
- S. Li, G. Zhang, X. Tu and J. Li, ChemElectroChem, 2018, 5, 701–707 CrossRef CAS.
- Q. Wen, Y. Zhao, Y. Liu, H. Li and T. Zhai, Small, 2022, 18, 2104513 CrossRef CAS PubMed.
- K. Ngamchuea, Curr. Opin. Electrochem., 2023, 37, 101193 CrossRef CAS.
- T. H. Wondimu, A. W. Bayeh, D. M. Kabtamu, Q. Xu, P. Leung and A. A. Shah, Int. J. Hydrogen Energy, 2022, 47, 20378–20397 CrossRef CAS.
- O. van der Heijden, S. Park, R. E. Vos, J. J. J. Eggebeen and M. T. M. Koper, ACS Energy Lett., 2024, 9, 1871–1879 CrossRef CAS.
- M. A. Habib, S. Lin, M. H. Joni, S. A. Dristy, R. Mandavkar, J.-H. Jeong and J. Lee, J. Energy Chem., 2025, 100, 397–408 CrossRef CAS.
- T. Munawar, S. Fatima, F. Iqbal, A. Bashir, M. Rafaqat, Z. Tu, L. Shen, S. A. Khan, M. Koc and C.-F. Yan, Fuel, 2025, 381, 133197 CrossRef CAS.
- R. Zhou, X. Han, Q. Chen, L. Peng, X. Qiu, P. Wang, C. Guo, J. Wang, Z. Wang and J. Hao, J. Mater. Chem. A, 2024, 12, 5719–5730 RSC.
- M. A. Habib, S. Burse, S. Lin, R. Mandavkar, M. H. Joni, J.-H. Jeong, S.-S. Lee and J. Lee, Small, 2024, 20, 2307533 CrossRef CAS.
- N. Rasool, A. S. Alqarni, K. Ahmad, A. G. Al-Sehemi, A. M. A. Henaish and S. Aman, Int. J. Hydrogen Energy, 2024, 81, 562–572 CrossRef CAS.
- S. Lin, R. Mandavkar, A. Habib, M. H. Joni, S. A. Dristy, S. Burse and J. Lee, Adv. Sustainable Syst., 2024, 1–11 Search PubMed.
- D. Jiang, W. Ma, R. Yang, B. Quan, D. Li, S. Meng and M. Chen, Electrochim. Acta, 2020, 329, 135121 CrossRef CAS.
- S. Lin, A. Habib, R. Mandavkar, R. Kulkarni, S. Burse, Y. Chung, C. Liu, Z. Wang, S. Lin, J. Jeong and J. Lee, Adv. Sustainable Syst., 2022, 1–14 Search PubMed.
- M. A. Habib, R. Mandavkar, S. Burse, S. Lin, R. Kulkarni, C. S. Patil, J.-H. Jeong and J. Lee, Mater. Today Energy, 2022, 101021 CrossRef CAS.
- S. Lin, M. A. Habib, R. Mandavkar, R. Kulkarni, S. Burse, Y.-U. Chung, C. Liu, Z. Wang, S. Lin, J.-H. Jeong and J. Lee, Adv. Sustainable Syst., 2022, 6, 2200213 CrossRef CAS.
- S. Li, E. Li, X. An, X. Hao, Z. Jiang and G. Guan, Nanoscale, 2021, 13, 12788–12817 RSC.
- S. Dresp, F. Dionigi, M. Klingenhof and P. Strasser, ACS Energy Lett., 2019, 4, 933–942 CrossRef CAS.
- J. Mohammed-Ibrahim and H. Moussab, Mater. Sci. Energy Technol., 2020, 3, 780–807 CAS.
- B. Xu, J. Liang, X. Sun and X. Xiong, Green Chem., 2023, 25, 3767–3790 RSC.
- T. Gao, W. Wang, Z. Zhang, W. Li, H. Gao, J. Liu, X. Zhao, Z. Liu and Y. Chen, Chem. Sci., 2025, 16, 15684–15696 RSC.
- D. Wang, Q. Li, C. Han, Q. Lu, Z. Xing and X. Yang, Nat. Commun., 2019, 10, 3899 CrossRef PubMed.
- A. M. Harzandi, S. Shadman, M. Ha, C. W. Myung, D. Y. Kim, H. J. Park, S. Sultan, W.-S. Noh, W. Lee, P. Thangavel, W. J. Byun, S. Lee, J. N. Tiwari, T. J. Shin, J.-H. Park, Z. Lee, J. S. Lee and K. S. Kim, Appl. Catal., B, 2020, 270, 118896 CrossRef CAS.
- S. A. Dristy, M. A. Habib, S. Lin, M. H. Joni, R. Mandavkar, Y.-U. Chung, M. Najibullah and J. Lee, Acta Phys.-Chim. Sin., 2025, 41, 100079 CrossRef.
- M. A. Habib, R. Mandavkar, S. Burse, S. Lin, R. Kulkarni, C. S. Patil, J. H. Jeong and J. Lee, Mater. Today Energy, 2022, 26, 101021 CrossRef CAS.
- J. Cen, P. K. Shen and Y. Zeng, J. Colloid Interface Sci., 2022, 610, 213–220 CrossRef CAS PubMed.
- R. Mandavkar, M. A. Habib, S. Lin, R. Kulkarni, S. Burse, J.-H. Jeong and J. Lee, Appl. Mater. Today, 2022, 29, 101579 CrossRef.
- H. Wang, J. Wu, S. Guo, H. Chen, R. Guo, D. Huan, D. Wang, W. Li, X. Li and D. Hou, J. Alloys Compd., 2025, 1022, 179796 CrossRef CAS.
- Y. Wang, Q. Ye, L. Lin, Y. Zhao and Y. Cheng, J. Colloid Interface Sci., 2023, 651, 1008–1019 CrossRef CAS PubMed.
- Y. Liu, J. Cao, Y. Chen, M. Wei, X. Liu, X. Li, Q. Wu, B. Feng, Y. Zhang and L. Yang, CrystEngComm, 2022, 24, 1704–1718 RSC.
- M. Ahasan Habib, R. Mandavkar, S. Lin, S. Burse, T. Khalid, M. Hasan Joni, J.-H. Jeong and J. Lee, Chem. Eng. J., 2023, 462, 142177 CrossRef CAS.
- M. H. Joni, A. Habib, S. Lin, S. A. Dristy, R. Mandavkar and J. Lee, J. Power Sources, 2025, 630, 236188 CrossRef CAS.
- X. Lv, S. Wan, T. Mou, X. Han, Y. Zhang, Z. Wang and X. Tao, Adv. Funct. Mater., 2022, 33, 2205161 CrossRef.
- S. Burse, R. Kulkarni, R. Mandavkar, M. A. Habib, S. Lin, Y.-U. Chung, J.-H. Jeong and J. Lee, Nanomaterials, 2022, 12, 3283 CrossRef CAS.
- X. Zhou, Y. Mo, F. Yu, L. Liao, X. Yong, F. Zhang, D. Li, Q. Zhou, T. Sheng and H. Zhou, Adv. Funct. Mater., 2023, 33, 2209465 CrossRef CAS.
- H. Liu, X. Li, L. Ge, C. Peng, L. Zhu, W. Zou, J. Chen, Q. Wu, Y. Zhang and H. Huang, Catal. Sci. Technol., 2020, 10, 8314–8324 RSC.
- Y.-T. Zeng, M.-Y. Xu, T. Wang, S.-Y. Wu, J. Zhang, S.-C. Mu and J. Yu, Electrochim. Acta, 2023, 444, 142004 CrossRef CAS.
- W. Xu, K. Wu, Y. Wu, Q. Guo, F. Fan, A. Li, L. Yang, F. Zheng, Y. Fan and W. Chen, Electrochim. Acta, 2023, 439, 141712 CrossRef CAS.
- H. Ma, Z. Chen, Z. Wang, C. V. Singh and Q. Jiang, Adv. Sci., 2022, 9, 1–9 Search PubMed.
- F. Yu, H. Zhou, Y. Huang, J. Sun, F. Qin, J. Bao, W. A. Goddard, S. Chen and Z. Ren, Nat. Commun., 2018, 9, 1–9 CrossRef PubMed.
- P. Zhang, L. Gong and Y. Tan, Sustainable Energy Fuels, 2021, 5, 5581–5593 RSC.
- Y. Li, S. Guo, T. Jin, Y. Wang, F. Cheng and L. Jiao, Nano Energy, 2019, 63, 103821 CrossRef CAS.
- G. Hu, W. Fu, Y. Lin, Y. Yue, M. Wang, P. Liu, Y. Li, J. Li, L. Wei, X. Zhao and Y. Wei, ACS Appl. Energy Mater., 2022, 5, 4259–4269 CrossRef CAS.
| This journal is © The Royal Society of Chemistry 2026 |