Open Access Article
Open Access ArticleXanthene-to-fluorene skeletal editing via oxygen deletion mediated by boron and aluminium radicals
Emily
Nahon
 a,
Gareth R.
Nelmes
a,
Gareth R.
Nelmes
 a,
Elena
Dallerba
a,
Elena
Dallerba
 b,
Li Feng
Lim
b,
Li Feng
Lim
 a,
Nicholas
Cox
a,
Nicholas
Cox
 a,
Claire L.
McMullin
a,
Claire L.
McMullin
 c,
Massimiliano
Massi
c,
Massimiliano
Massi
 b,
Fabian
Kallmeier
b,
Fabian
Kallmeier
 *a and
Jamie
Hicks
*a and
Jamie
Hicks
 *a
*a
aResearch School of Chemistry, Australian National University, Sullivans Creek Rd, Acton, ACT 2601, Australia. E-mail: fabian.kallmeier@anu.edu.au; jamie.hicks@anu.edu.au
bSchool of Molecular and Life Sciences (MLS), Curtin University, 500 Townsing Dr, Bentley, WA 6102, Australia
cDepartment of Chemistry, University of Bath, Claverton Down, Bath, BA2 7AY, UK
First published on 20th April 2026
Abstract
Single-atom skeletal editing via selective oxygen deletion from diarylethers remains an underdeveloped transformation, despite its potential to directly access new carbon frameworks. Here, we report a boron- and aluminium-mediated O-deletion reaction that converts xanthene and diphenylether motifs into fluorene and biphenyl architectures through concomitant C–C bond formation. Lithium metal reduction of diamido arylether boron halides affords lithium boryloxy complexes in high yield and on a multigram scale, with both the new C–C bond and terminal B–O− unit formed in a single step via a transient open-shell B(II) intermediate. Hydrolysis furnishes fluorene- and biphenyl-based [1,3,2]diazaborepin-2-ols, representing previously inaccessible boron-containing fluorophores that exhibit high photoluminescence quantum yields. Extension of this strategy to aluminium allows clean hydrolytic release of the organic scaffold and provides a concise, scalable synthesis of functionalised 4,5-diaminofluorenes. These results establish O-deletion as a viable skeletal editing strategy for arylethers and highlight the role of main-group radical intermediates in selective framework reorganisation.
Introduction
Skeletal editing, i.e., the selective alteration of a molecule's core motif, typically by a single atom, has recently experienced a surge in popularity and is now an invaluable tool in contemporary synthetic chemistry. Sarpong, Levin, and coworkers have recently suggested a classification scheme for single-atom skeletal edits.1 The methods currently in use typically revolve around privileged structures in medicinal chemistry,2,3e.g. pyridine to benzene4–7 and vice versa,8 isothiazole to pyrazole,9 or removing nitrogen atoms from dialkylamines.10 One example of single-atom skeletal editing that is less developed is the oxygen-deletion reaction in ethers, which would yield a new C–C bond. Such a reaction would be expected to proceed through a reductive pathway, eliminating “O2−”. However, the reduction of diarylethers to the corresponding biaryls (and cyclic analogues) is hampered by the non-selective reaction with alkali metals – including the low tolerance of functional groups – and the need for subsequent C–C coupling of the individual fragments (Fig. 1, top).11–16In 2024, Frenking, Driess and coworkers demonstrated how coordination chemistry could aid in this regard (Fig. 1, centre).17 The team showed that the 9,10-diboraphenanthrene motif in (II) could be prepared selectively via the reductive ring contraction of 4,5-([Si]B)2-xanthene ([Si] = {PhC(NtBu)2}(Me2N)Si) (I) by reduction with potassium graphite (KC8), with the oxygen atom suggested to be eliminated as K2O.17 Similarly, the same groups observed a related rearrangement upon treatment of xanthene-4,5-diyl-bis(dichloroborane) (III), in which an N-heterocyclic carbene is coordinated to each boron centre, with KC8. In this case, diboraoxirane (IV) could be obtained, which was shown to undergo ring expansion upon reaction with an array of small molecules, e.g., CO2, O2, and S8.18
In this work, we demonstrate a selective O-deletion reaction from diamido boron and aluminium compounds bearing a diarylether backbone (Fig. 1, bottom). The procedure remains selective even on a multigram scale and allows for the synthesis of diverse products, including synthetically challenging 4,5-substituted fluorenes and novel boron-based fluorescent dyes.
Results and discussion
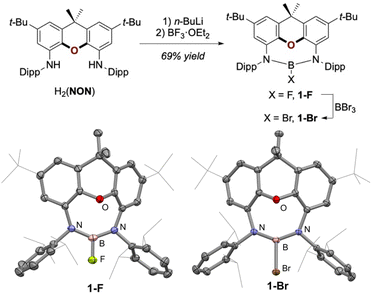
With the chemistry of I and III demonstrating general feasibility, we initially set out to synthesise diamidoxanthene-based diamido boron fluoride 1-F, as shown in Fig. 2. 1-F was obtained cleanly by deprotonation of H2(NON) (NON: 4,5-bis(2,6-diisopropylanilido)-2,7-di-tert-butyl-9,9-dimethylxanthene) using two equivalents of n-BuLi as the base, followed by subsequent salt metathesis with BF3·OEt2. The target compound, which is air- and moisture-stable, was obtained in 69% crystalline yield on a 5 g scale (see SI for synthetic details). 1-F was conveniently converted to the analogous bromide 1-Br in 72% yield by stirring with an excess of BBr3 in toluene at room temperature for 2 days.19 The compounds were characterised by multinuclear NMR spectroscopy, and their solid-state structures were confirmed by X-ray diffraction (Fig. 2, bottom).With suitable precursors in hand, our attention turned to their behaviour under reductive conditions (Fig. 3, top). A d8-THF solution of 1-F with a small piece of lithium ribbon was sonicated at ambient temperature in an air-tight NMR tube. Reaction progression was indicated by the appearance of a fluorescent yellow-coloured solution, which was complete after 24 hours, as indicated by NMR analysis. 11B-NMR spectroscopy showed a single new boron-containing product, with a slight shift compared to that of the starting material (20.5 ppm cf. 21.8 ppm for 1-F). In addition, the disappearance of a signal in 19F-NMR spectrum indicated the loss of the fluoride ligand. Multigram quantities of the same product were obtained by sonicating a flask containing 1-F and lithium ribbon overnight, followed by stirring the reaction mixture for 2 days at 60 °C, or by stirring a THF solution of 1-F over finely divided Li metal on LiCl (or LiBr) at room temperature overnight.20
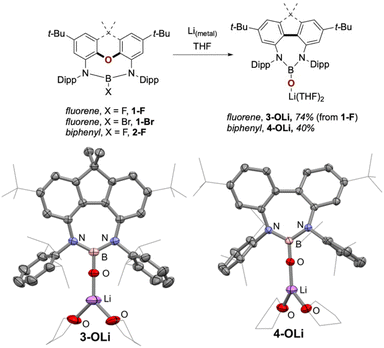
Crystals suitable for X-ray diffraction were grown from a concentrated benzene solution of the new product and showed it to be the lithium boryloxy 3-OLi (Fig. 3). Boryloxy compounds are boron(III) compounds featuring a B–O− moiety, and have gained significant attention over the last years as strong σ- and π-donor ligands.21–32 Complex 3-OLi consists of a 3-coordinate boron(III) centre, which, contrary to its progenitor 1-F, is contained within a 4,5-diamidofluorene scaffold. The oxygen atom, formerly residing within the xanthene backbone, completes the coordination sphere around the boron centre as an oxido ligand, which is bridging to the [Li(THF)2]+ cation. The N–B–N angle of 124.9° is indicative of an sp2 hybridised boron centre, with the short B–O bond length 1.313(4) Å indicating the B–O bond to possess some double bond character, which was confirmed by IR spectroscopy revealing the B–O stretch at 1320 cm−1 (calc. 1317 cm−1).
Reduction of the bromo starting material 1-Br with lithium at room temperature gave a near-identical result, with the same fluorene-based boryloxy isolated, but as the LiBr adduct (see SI). However, this reaction was lower-yielding and less selective than the reduction of 1-F, with multiple by-products observed by NMR spectroscopy.
With the O-deletion of 1-F and 1-Br confirmed, we wanted to investigate how important the cyclic nature of the xanthene-based ether was to this reaction. In recent work, we reported the synthesis of the “acyclic” substituted diphenylether-based equivalents of 1-F and 1-Br (2-F and 2-Br).19 Furthermore, we showed that the reduction of 2-Br with lithium metal at −80 °C leads to C,N-bond-cleavage and a 1,2-migration of one Dipp group to the boron atom.19 Gratifyingly, when the reduction of 2-F was carried out under the same conditions as for 1-F and 1-Br, the lithium boryloxy O-deletion product 4-OLi was isolated, albeit in lower yields than the fluorene analogue.
X-ray diffraction analysis of 4-OLi revealed a strikingly similar molecular structure to 3-OLi (Fig. 3). The biggest difference is found in the dihedral angle between the two aromatic rings of the fluorene motif in 3-OLi (3.3°) and those in the biphenyl motif in 4-OLi (39.8°), which aligns with the rigidity of the fluorene, forcing the backbone to adopt a planar geometry. To the best of our knowledge, this is the first report of an O-deletion reaction of amidoxanthene/diphenyl ethers, and the first report of diamidofluorene coordination complexes.33 Noteworthy is that the reverse reaction, O-insertion, has recently been reported to occur biochemically in insects on related substrates.34
Mechanistic studies
To shine light on the underlying mechanism of the O-deletion reaction, extensive computational studies were carried out on the xanthene system at the BP86-D3BJ-CPCM(THF)/6–311++G**//BP86-D3BJ/6-31G** level of theory. Taking inspiration from the mechanistic investigations of Frenking, Driess, and coworkers (vide supra),17,18 we initiated our mechanistic investigations by assuming that a closed-shell B(I) anion (boryl anion) was being generated from a two-electron reduction of the starting material (1-F or 1-Br). However, in all mechanisms calculated (monomeric and dimeric, with or without including a lithium cation), at least one implausible step with an extraordinarily high barrier was located (see SI for further details). This led us to reconsider our starting point and whether only a single-electron reduction of the starting material was responsible for the transformation. Boron(II) radical (boranyl) species have seen use in a number of different synthetic reactions, such as alkyl halide cross couplings and radical additions to multiple bonds.35 However they can be highly unstable, and so have only been isolated with coordination of an ancillary Lewis basic ligand.35Starting from the open-shell B(II) radical compound (NON)B˙, generated via a single-electron reduction of 1-F or 1-Br, a viable mechanism was calculated (Fig. 4). The first step involves coordination of the oxygen in the xanthene backbone to the B(II) centre (INT1), which proceeds through the low-energy transition state TS0-1 (4.4 kcal mol−1). In the next step, the radical is transferred from the boron(II) centre to one of the phenyl rings of the xanthene viaTS1-2 (15.3 kcal mol−1), breaking a C–O bond and forming the aryl-centred radical intermediate (INT2). After rotating the aryl radical behind the plane of the aryloxy ring in a wagging motion through another low-energy transition state TS2-3 (5.0 kcal mol−1), the stage is set within INT3 for the key C–C bond formation and C–O cleavage (TS3-4), which is the rate-determining step with a barrier of ca. 19.5 kcal mol−1. This yields the oxidaneylborane radical product (N,N)BO˙ in a strongly exergonic step (−92.2 kcal mol−1). Finally, in the reaction mixture, (N,N)BO˙ is converted into the isolated lithium boryloxy product 3-OLivia a further one-electron reduction with lithium metal.
This open-shell mechanism contrasts with the closed-shell mechanism reported for IV.18 However, in the reported study, an extraordinarily high barrier of +53.7 kcal mol−1 was calculated for one step, with the authors stating that the barrier does not agree with their experimental observations. In our case, an activation energy of ∼20 kcal mol−1 is in line with our experimental observations. Furthermore, additional evidence for this radical mechanism was acquired through EPR spectroscopy. The reduction of 1-Br was repeated, but after ca. 5 minutes of stirring at room temperature, an aliquot of the reaction mixture was removed and analysed by EPR spectroscopy. The EPR spectrum of the reaction solution revealed the presence of an open-shell species with hyperfine coupling consistent with the boryloxy radical (N,N)BO˙ (Fig. 4), further supporting the open-shell mechanism presented (see SI for further details).
[1,3,2]Diazaborepin-2-ol
The boryloxy compounds 3-OLi or 4-OLi are moisture-sensitive, with exposure to air yielding quantitative amounts of the protonated products 3-OH or 4-OH, rare examples of [1,3,2]diazaborepin-2-ol derivatives. Both compounds were crystallographically characterised (Fig. 5).3-OH or 4-OH were found to be remarkably stable, showing no signs of decomposition even when treated with hot triflic acid, boiling lithium hydroxide solution, or KHF2, amongst others. However, under forcing conditions, the OH group of 3-OH can be substituted with fluoride by treating the compound with neat BF3·OEt2 (10 equiv.) and heating to 100 °C over 48 hours, yielding the boron fluoride 3-F in a 50% isolated yield.
Photophysical properties
Working with these [1,3,2]diazaborepin compounds (3-OH, 3-F, and 4-OH), it was immediately evident that they were highly emissive compounds. Upon irradiation of a crystalline sample of 3-OH with a UV torch (365 nm), intense blue emission is observed (Fig. 5). As such, the photophysical properties of 3-OH, 3-F, and 4-OH were measured both in solution and in solid-state, and the results are summarised in Table 1. The absorption profiles measured from diluted toluene solutions (Fig. S30) reveal broad bands in the lower-energy UV region between 300 and 400 nm. These are ascribed to ππ* electronic transitions. Notably, the absorption spectra of 3-OH and 3-F appear more structured in the 350–400 nm region, consistent with the enhanced rigidity of the fluorene moiety compared with the more flexible biphenyl in 4-OH. Upon excitation at 350 nm, all three compounds appear blue/aqua luminescent in both solution and the solid state at room temperature. The emission spectra from diluted toluene solutions are very similar (Fig. S31), displaying broad bands between 400 and 550 nm, characterised by a vibronic structure typical of ππ* excited states. On the other hand, the emission profiles recorded from solid-state samples (Fig. S32) show a slight blueshift, attributed to the absence of solvent stabilisation of the excited states, without evidence of extensive aggregation. The excited state lifetime decays τ were satisfactorily fitted by monoexponential functions, revealing relatively short decays in the 5.70–7.03 ns range, typical of spin-allowed fluorescent emission (Table 1). Measurement of photoluminescent quantum yields (Φ) in the 40–59% range demonstrates that these compounds exhibit strong emission and are comparable to those of boron-dipyrromethene scaffolds (BODIPY).36 Given the wide range of applications that BODIPY-type species have found, we propose that these compounds could serve as a new, accessible class of fluorophores. Notably, the similarities of photophysical properties of 3-OH, 3-F, and 4-OH would suggest that this class of fluorophores are highly tunable, and that physico-chemical properties can be optimised independently from photophysical properties.| Absorption λabs/nm | λ em/nm | Emission τ/ns | Φ | |
|---|---|---|---|---|
| 3-OH | 357, 376 | 385, 407 | 5.70 | 0.59 |
| 3-F | 300, 358, 377 | 388, 409 | 7.03 | 0.40 |
| 4-OH | 339 | 422 | 6.37 | 0.50 |
Aluminium
The stability of the [1,3,2]diazaborepin compounds is beneficial for their potential future uses as fluorophores, but for the broader applicability of the O-deletion reaction in organic synthesis, removal of the organic fragment from the group 13 element would be beneficial. During this work, the removal of the boron centre from 3-OH, 3-F, and 4-OH to give the 4,5-substituted organic product was extensively investigated, but without success (see SI for details). Therefore, it was concluded that to liberate the newly C–C coupled organic diamine, an element other than boron would be required. Aluminium, being the element below boron in group 13, and with Al–N bonds much more susceptible to hydrolysis,37 was an obvious choice. However, the reduction of (NON)AlI (the aluminium analogue of 1-F/1-Br) has been extensively studied – producing Al(I) aluminyl anions and the Al(II) dialane – with no evidence of an O-deletion reaction.38 Aluminium(II) radicals have been reported several times, and can be capable of significant reactivity,39 however these results suggest that the Al(II) radical intermediate may be too stable to potentially achieve a similar O-deletion reaction. Therefore, to achieve comparable reactivity, the radical intermediate would need to be destabilised.In recent work, we reported tBuNON, where the flanking aryl groups of NON have been replaced with more electron-donating tert-butyl groups.38 Stirring a solution of AlI(tBuNON) (5) over excess KC8 at 50 °C overnight gives an analogous O-deletion reaction, from which the crystalline potassium ‘aloxy’ complex 6 can be isolated. The structure of 6 was confirmed by X-ray diffraction (Fig. 6) and consists of an Al2O2 core, in which each of the aluminium centres is coordinated by one diamidofluorene ligand. The charge is compensated by two potassium atoms that connect the two fluorene backbones via arene-π interactions. EPR spectroscopic analysis of the reaction solution indicates the presence of an open-shell intermediate, consistent with a radical mechanism analogous to that proposed for the boron system (see SI for details).
Exposure of 6 to air results in complete hydrolysis of the product, yielding the metal-free 4,5-diaminofluorene (7) in quantitative yield. To demonstrate the feasibility of this procedure in the large-scale synthesis of 4,5-diaminofluorenes, the reaction was repeated on a 10 g scale. A simple organic workup (in air) followed by recrystallisation gave the 4,5-diaminofluorene 7 in 63% yield (Fig. 6).
Finally, the N-bound tert-butyl groups on 7 can be easily removed by heating the compound in phosphoric acid (85%-wt) to 145 °C for 30 minutes. This gives the primary 4,5-diaminofluorene (8) in an 86% yield (Fig. 7). Conveniently, heating the same reaction for longer periods (16 hours) leads to complete de-tert-butylation of the compound, also removing the tert-butyl groups at the 2,7 positions of the fluorene, affording the unsubstituted 4,5-diaminofluorene (9) in 78% yield (Fig. 7). This represents the first high-yielding, concise, and reproducible synthesis of 4,5-diaminofluorenes, a class of compounds that have been barely studied due to synthetic inaccessibility,40–43 opening the door to broad future applications.
Conclusions
In summary, we have developed a selective and scalable O-deletion strategy for diarylether frameworks mediated by boron and aluminium radicals, enabling direct skeletal editing via formal oxygen removal and concomitant C–C bond formation. Reduction of diamido-xanthene and diphenylether boron complexes proceeds through a transient open-shell B(II) intermediate, furnishing lithium boryloxy species in high yield and on a multigram scale. Subsequent hydrolysis provides previously inaccessible fluorene- and biphenyl-based [1,3,2]diazaborepin-2-ols, which exhibit remarkable chemical robustness and high photoluminescence quantum yields, identifying them as a new, tunable class of boron-containing fluorophores.Extension of this chemistry to aluminium sees a similar O-deletion reaction, which is proposed to occur via an analogous Al(II) radical mechanism. In this case, the compound is readily hydrolysed, enabling the clean isolation of the metal-free N4,N5,2,7-tetra-tert-butyl-9,9-dimethyl-4,5-diaminofluorene (7). The compound can be partially or fully de-tert-butylated with hot phosphoric acid, giving the primary amine compounds 8 and 9. The compounds are expected to serve as versatile precursors to a previously inaccessible series of 4,5-diaminofluorene compounds in molecule synthesis and materials chemistry.
Author contributions
E. N. performed most of the synthetic work and more routine characterisation. G. R. N. performed the computational work under the supervision of C. L. M. E. D. performed the photophysical measurements under the supervision of M. M. L. F. L. was responsible for the EPR collection and modelling, supervised by N. C. F. K. performed some synthetic work and was responsible for project management. J. H. was responsible for funding acquisition and providing resources. All authors were involved in reviewing and editing the manuscript.Conflicts of interest
There are no conflicts to declare.Data availability
CCDC 2514982–2514992 and 2515042 contain the supplementary crystallographic data for this paper.44a–lThe data supporting this article have been included as part of the supplementary information (SI). Supplementary information: synthetic and characterisation data, including those for crystallographic, EPR, photophysical and computational studies. See DOI: https://doi.org/10.1039/d6sc01056c.
Acknowledgements
JH would like to thank the Australian Research Council (FT240100229) for the funding of this work. This research was undertaken with the assistance of resources from the National Computational Infrastructure (NCI Australia), an NCRIS enabled capability supported by the Australian Government, and the University of Bath's Research Computing Group (https://doi.org/10.15125/b6cd-s854) for their support of this work.Notes and references
- J. Jurczyk, J. Woo, S. F. Kim, B. D. Dherange, R. Sarpong and M. D. Levin, Nat. Synth., 2022, 1, 352 CrossRef CAS PubMed.
- K. R. Campos, P. J. Coleman, J. C. Alvarez, S. D. Dreher, R. M. Garbaccio, N. K. Terrett, R. D. Tillyer, M. D. Truppo and E. R. Parmee, Science, 2019, 363, 244 CrossRef.
- F.-P. Wu, J. L. Tyler and F. Glorius, Acc. Chem. Res., 2025, 58, 893 CrossRef CAS PubMed.
- A. R. Fout, B. C. Bailey, J. Tomaszewski and D. J. Mindiola, J. Am. Chem. Soc., 2007, 129, 12640 CrossRef CAS PubMed.
- A. R. Fout, B. C. Bailey, D. M. Buck, H. Fan, J. C. Huffman, M.-H. Baik and D. J. Mindiola, Organometallics, 2010, 29, 5409 CrossRef CAS.
- A. Conboy and M. F. Greaney, Chem, 2024, 10, 1940 CAS.
- Q. Cheng, D. Bhattacharya, M. Haring, H. Cao, C. Mück-Lichtenfeld and A. Studer, Nat. Chem., 2024, 16, 741 CrossRef CAS PubMed.
- T. J. Pearson, R. Shimazumi, J. L. Driscoll, B. D. Dherange, D.-I. Park and M. D. Levin, Science, 2023, 381, 1474 CrossRef CAS PubMed.
- A. Fanourakis, Y. Ali, L. Chen, P. Q. Kelly, A. J. Bracken, C. B. Kelly and M. D. Levin, Nature, 2025, 641, 646 CrossRef CAS PubMed.
- S. H. Kennedy, B. D. Dherange, K. J. Berger and M. D. Levin, Nature, 2021, 593, 223 CrossRef CAS PubMed.
- P. Schorigin, Ber. dtsch. Chem. Ges. A/B, 1923, 56, 176 CrossRef.
- P. Schorigin, Ber. dtsch. Chem. Ges. A/B, 1924, 57, 1627 CrossRef.
- P. A. Sartoretto and F. J. Sowa, J. Am. Chem. Soc., 1937, 59, 603 CrossRef CAS.
- F. C. Weber and F. J. Sowa, J. Am. Chem. Soc., 1938, 60, 94 CrossRef CAS.
- H. Gilman and J. Dietrich, J. Org. Chem., 1957, 22, 851 CrossRef CAS.
- M. Lissel, J. Kottmann, D. Tamarkin and M. Rabinovitz, Z. Naturforsch. B, 1988, 43, 1211 CrossRef CAS.
- J. Fan, J. Xu, Q. Ma, S. Yao, L. Zhao, G. Frenking and M. Driess, J. Am. Chem. Soc., 2024, 146, 20458 CrossRef CAS PubMed.
- J. Fan, S. Pan, S. Yao, C. Ding, G. Frenking and M. Driess, J. Am. Chem. Soc., 2025, 147, 6925 CrossRef CAS PubMed.
- E. E. Nahon, G. R. Nelmes, P. J. Brothers and J. Hicks, Chem. Commun., 2023, 59, 14281 RSC.
- J. Hicks, M. Juckel, A. Paparo, D. Dange and C. Jones, Organometallics, 2018, 37, 4810 CrossRef CAS.
- Y. K. Loh, L. Ying, M. Ángeles Fuentes, D. C. H. Do and S. Aldridge, Angew. Chem., Int. Ed., 2019, 58, 4847 CrossRef CAS PubMed.
- Y. K. Loh, K. Porteous, M. Á. Fuentes, D. C. H. Do, J. Hicks and S. Aldridge, J. Am. Chem. Soc., 2019, 141, 8073 CrossRef CAS PubMed.
- L. Weber, Coord. Chem. Rev., 2021, 431, 213667 CrossRef CAS.
- D. Sarkar, P. Vasko, T. Gluharev, L. P. Griffin, C. Bogle, J. Struijs, J. Tang, A. F. Roper, A. E. Crumpton and S. Aldridge, Angew. Chem., Int. Ed., 2024, 63, e202407427 CrossRef CAS PubMed.
- D. Sarkar, P. Vasko, A. F. Roper, A. E. Crumpton, M. M. D. Roy, L. P. Griffin, C. Bogle and S. Aldridge, J. Am. Chem. Soc., 2024, 146, 11792 CrossRef CAS PubMed.
- D. Sarkar, P. Vasko, L. Ying, J. J. C. Struijs, L. P. Griffin and S. Aldridge, Angew. Chem., Int. Ed., 2025, 64, e202502326 CrossRef CAS PubMed.
- R. Guthardt and C. Jones, Chem. Commun., 2024, 60, 1583 RSC.
- C. Helling, D. J. D. Wilson and C. Jones, J. Am. Chem. Soc., 2025, 147, 16620 CrossRef CAS PubMed.
- C. Wei, L. Guo, C. Zhu and C. Cui, Angew. Chem., Int. Ed., 2025, 64, e202414464 CrossRef CAS PubMed.
- L. R. Thomas-Hargreaves, D. Hunger, M. Kern, A. J. Wooles, J. Van Slageren, N. F. Chilton and S. T. Liddle, Chem. Commun., 2021, 57, 733 RSC.
- X. Dan, J. Du, S. Zhang, J. A. Seed, M. Perfetti, F. Tuna, A. J. Wooles and S. T. Liddle, Inorg. Chem., 2024, 63, 9588 CrossRef CAS PubMed.
- J. T. Boronski, L. R. Thomas-Hargreaves, M. A. Ellwanger, A. E. Crumpton, J. Hicks, D. F. Bekiş, S. Aldridge and M. R. Buchner, J. Am. Chem. Soc., 2023, 145, 4408 CrossRef CAS PubMed.
- Oxygen extrusion has been observed in the decamethylcobaltocene mediated reduction of strained dinaphthooxepine bisimides (a) M. Odajima, N. Fukui and H. Shinokubo, Org. Lett., 2023, 25, 282 CrossRef CAS PubMed and the reductive deoxygenation of ethers via nickel catalysis in the presence of reductants; (b) Z. C. Cao and Z. J. Shi, J. Am. Chem. Soc., 2017, 139, 6546 CrossRef CAS PubMed , through photochemical rearrangement of diphenyl ethers; (c) N. Haga and H. Takayanagi, J. Org. Chem., 1996, 61, 735 CrossRef CAS PubMed , and recently from oxepines using AFM tip chemistry; (d) S. Mishra, V. Malave, R. Svensson, H. Grönbeck, F. Albrecht, D. Peña and L. Gross, J. Am. Chem. Soc., 2025, 147, 44055 CrossRef CAS PubMed , and in the cobalt-catalysed transformation of various ethers into 1,2-diarylethanes:; (e) M. K. Sahu, S. Pattanaik, G. Joshi, E. D. Jemmis and C. Gunanathan, Angew. Chem., Int. Ed., 2026, 65, e20176 CrossRef CAS PubMed . Other examples are included in the perspective on unimolecular fragment coupling; (f) R. Shimazumi and M. Tobisu, JACS Au, 2024, 4, 1676 CrossRef CAS PubMed.
- A. Usami, H. Kono, V. Austen, Q. M. Phung, H. Shudo, T. Kato, H. Yamada, A. Yagi, K. Amaike, K. J. Fujimoto, T. Yanai and K. Itami, Science, 2025, 388, 1055 CrossRef CAS PubMed.
- Some reviews and select examples of boryl radicals and their reactivity (a) T. Taniguchi, Chem. Soc. Rev., 2021, 50, 8995 RSC; (b) X. Guo and Z. Lin, Chem. Sci., 2024, 15, 3060 RSC; (c) Y. Su and R. Kinjo, Coord. Chem. Rev., 2017, 352, 346 CrossRef CAS; (d) T. Taniguchi, Eur. J. Org Chem., 2019, 2019, 6308 CrossRef CAS; (e) Z. Zhang, M. J. Tilby and D. Leonori, Nat. Synth., 2024,(3), 1221 CrossRef.
- (a) A. Loudet and K. Burgess, Chem. Rev., 2007, 107, 4891 CrossRef CAS PubMed; (b) G. Ulrich, R. Ziessel and A. Harriman, Angew. Chem., Int. Ed., 2008, 47, 1184 CrossRef CAS PubMed; (c) H. Lu, J. Mack, Y. Yang and Z. Shen, Chem. Soc. Rev., 2014, 43, 4778 RSC; (d) Y. Ni and J. Wu, Org. Biomol. Chem., 2014, 12, 3774 RSC; (e) T. Kowada, H. Maeda and K. Kikuchi, Chem. Soc. Rev., 2015, 44, 4953 RSC; (f) J. Bañuelos-Prieto and R. Sola Llano, BODIPY Dyes - A Privilege Molecular Scaffold with Tunable Properties, IntechOpen, 2019 Search PubMed; (g) V. I. Martynov and A. A. Pakhomov, Russ. Chem. Rev., 2021, 90, 1213 CrossRef CAS; (h) N. A. Bumagina and E. V. Antina, Coord. Chem. Rev., 2024, 505, 215688 CrossRef CAS.
- D. R. Lide, CRC Handbook of Chemistry and Physics, CRC Press, Boca Raton, FL, 1974 Search PubMed.
- F. Kallmeier, A. J. R. Matthews, G. R. Nelmes, N. R. Lawson and J. Hicks, Dalton Trans., 2024, 53, 12450 RSC.
- Some reviews and select examples of aluminyl radicals and their reactivity (a) R. P. Singh and N. P. Mankad, JACS Au, 2025, 5, 2076 CrossRef CAS PubMed; (b) M. Nakamoto, T. Yamasaki and A. Sekiguchi, J. Am. Chem. Soc., 2005, 127, 6954 CrossRef CAS PubMed; (c) D. Mandal, T. I. Demirer, T. Sergeieva, B. Morgenstern, H. T. A. Wiedemann, C. W. M. Kay and D. M. Andrada, Angew. Chem., Int. Ed., 2023, 62, e202217184 CrossRef CAS PubMed.
- E. K. Weisburger and J. H. Weisburger, J. Org. Chem., 1955, 20, 1396 CrossRef CAS.
- E. Weisburger, J. Org. Chem., 1956, 21, 698 CrossRef CAS.
- H. A. Staab, T. Saupe and C. Krieger, Angew Chem. Int. Ed. Engl., 1983, 22, 731 CrossRef.
- O. V. Semidetko, L. A. Chetkina, V. K. Bel’skii, A. N. Poplavskii, A. M. Andrievskii and K. M. Dyumaev, J. Struct. Chem., 1988, 29, 330 CrossRef.
- (a) CCDC 2514982: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2qf1gp; (b) CCDC 2514983: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2qf1hq; (c) CCDC 2514984: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2qf1jr; (d) CCDC 2514985: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2qf1ks; (e) CCDC 2514986: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2qf1lt; (f) CCDC 2514987: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2qf1mv; (g) CCDC 2514988: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2qf1nw; (h) CCDC 2514989: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2qf1px; (i) CCDC 2514990: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2qf1qy; (j) CCDC 2514991: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2qf1rz; (k) CCDC 2514992: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2qf1s0; (l) CCDC 2515042: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2qf3dp.
| This journal is © The Royal Society of Chemistry 2026 |