Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
A dual-responsive cationic acridinium nanohoop: redox activity and acid/base-controlled reversible guest capture and release
Jiao Minga,
Jin Zhangb,
Yan Hua,
Kai Lana,
Dongmei Zhanga,
Ping Lia,
Jiarong Miaoa,
Jiyong Jianga,
Xiaobo Zhanga,
Hanchi Zhonga,
Peiyuan Yu *b and
Chuyang Cheng
*b and
Chuyang Cheng *a
*a
aKey Laboratory of Green Chemistry & Technology, Sichuan University, Chengdu, 610064, China. E-mail: cycheng@scu.edu.cn
bDepartment of Chemistry, Guangming Advanced Research Institute, Southern University of Science and Technology, Shenzhen, 518055, China. E-mail: yupy@sustech.edu.cn
First published on 13th April 2026
Abstract
In this work, we report a fully conjugated, radially cationic nitrogen-doped cycloparaphenylene derivative, Ad[10]CPP+, constructed using the acridinium moiety as a functional building block. This compound exhibits remarkable reversibility in structural transformations under chemical stimuli and tunable redox behavior under electrochemical stimuli. Cyclic voltammetry reveals that Ad[10]CPP+ possesses multiple oxidation states and demonstrates significant redox versatility. Furthermore, Ad[10]CPP+ can reversibly transform from a near-circular configuration to a water-droplet shape upon hydroxide attack on the acridinium moiety when the acid concentration is lowered. The host–guest interaction between fullerene (C60) and Ad[10]CPP+ was confirmed by NMR spectroscopy and single-crystal X-ray diffraction. Significantly, the host–guest system exhibits excellent fatigue resistance, enabling at least five consecutive cycles of chemically triggered fullerene capture and release through reversible structural transformations.
Introduction
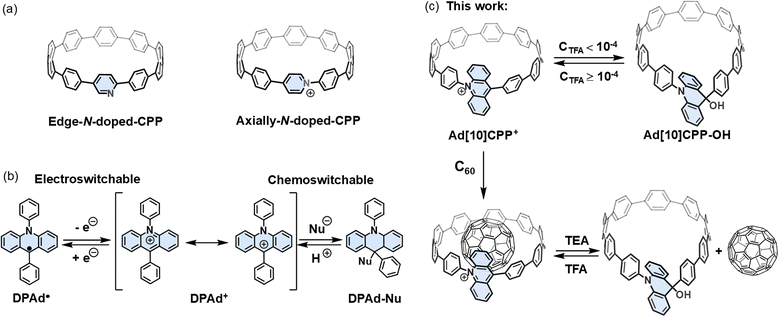
Cycloparaphenylenes (CPPs), aromatic macrocycles composed of para-phenylene units, have garnered considerable attention since their first synthesis.1–8 The interest is primarily attributed to their inherent ring strain9–15 and unique radial cyclic π-conjugated architectures.16–21 Over the past decade, the field has witnessed remarkable progress, as evidenced by the successful synthesis of CPPs and their derivatives of varying sizes.22–28 Despite these advancements, the incorporation of heteroatoms, particularly nitrogen, remains underexplored.8 Replacing phenylene units with nitrogen-containing aromatic rings can endow CPPs with acid–base responsiveness,29 metal coordination capabilities,30 and synthetic handles for constructing mechanically interlocked molecules.31–33 Subsequent alkylation of these nitrogen atoms can convert the neutral macrocycles into cationic ones, introducing redox activity.34,35 Both the amount and position of nitrogen doping can effectively modulate the photophysical36 and supramolecular properties of CPPs.37 Notably, in most reported cases, nitrogen atoms are incorporated at lateral positions of the CPP skeleton (Scheme 1a, left), as this approach aligns with conventional synthetic routes. In contrast, the direct fusion of nitrogen at the phenylene linkage sites, namely, axial doping, is far less common.38–42 Such a configuration is predicted to markedly influence the nanoring's photophysical profile, introduce multiple accessible redox states,38 and create a cavity with tunable size.39 However, the limited examples of axially N-doped carbon nanorings reported to date are, strictly speaking, not fully conjugated CPPs. Recently, we reported the first fully conjugated cationic CPPs bearing pyridinium units at the phenylene linkages43 (Scheme 1a, right), a long-standing synthetic challenge due to ring strain and cation instability under basic conditions. This synthetic challenge was effectively overcome by introducing an N-phenylpiperidine derivative strain-release unit followed by aromatization to construct a pyridinium moiety. In the present work, we extend this design by employing a π-expanded motif, in the form of an acridine fragment, as a strain-release unit. This approach not only enhances synthetic accessibility but also unlocks distinct functional advances. Specifically, we demonstrate that installing cationic nitrogen at the phenylene linkage sites in the form of acridinium enables reversible, stimuli-responsive switching of the macrocyclic framework between configurations in response to electrochemical and acid–base stimuli. This structural dynamism, in turn, facilitates controlled capture and release of guest molecules, advancing CPPs toward adaptive host–guest systems.The rigid and curved conjugated structures of CPPs serve as ideal host molecules for fullerenes.44–50 Their complementary shape and size facilitate strong π–π interactions with fullerenes, achieving binding constants of 105–106 M−1. The rigid and strained macrocyclic constitution, however, also poses a challenge for incorporating reversible guest capture and release functionality. Introducing stimuli-responsive motifs that can reversibly modulate the strained framework offers an effective strategy for achieving controlled guest capture and release. While several studies have incorporated stimuli-responsive motifs into CPPs to modulate the geometry of the strained macrocycle,16,51–54 to the best of our knowledge, no examples of stimuli-responsive guest capture and release for CPPs have been reported thus far.
The acridinium unit is known for its reversible structural transformation in response to electrochemical and chemical stimuli.55–63 Specifically, 9,10-diphenylacridinium (DPAd+) is highly susceptible to nucleophilic attack and can reversibly transition between its radical state and cationic form under electrochemical control (Scheme 1b). In this study, we incorporate a DPAd+ fragment into the CPP skeleton to construct a functionalized strain macrocycle similar in size to [10]CPP, namely, Ad[10]CPP+ (Scheme 1c). We carried out a comprehensive investigation into the synthesis and structural, optical, electronic, and computational properties of Ad[10]CPP+. Our findings revealed that Ad[10]CPP+ undergoes reversible structural transformations under electrochemical and acid/base stimulation. Furthermore, we performed extensive studies on the host–guest chemistry of Ad[10]CPP+ and successfully obtained the solid-state suprastructure of C60 captured by Ad[10]CPP+. Most notably, Ad[10]CPP+ demonstrated the ability to bind and release C60 in response to acid/base stimuli, exhibiting excellent fatigue resistance over multiple cycles. This approach offers significant potential for expanding the applications of CPPs in supramolecular chemistry and materials science.
Results and discussion
Synthesis and structure characterization
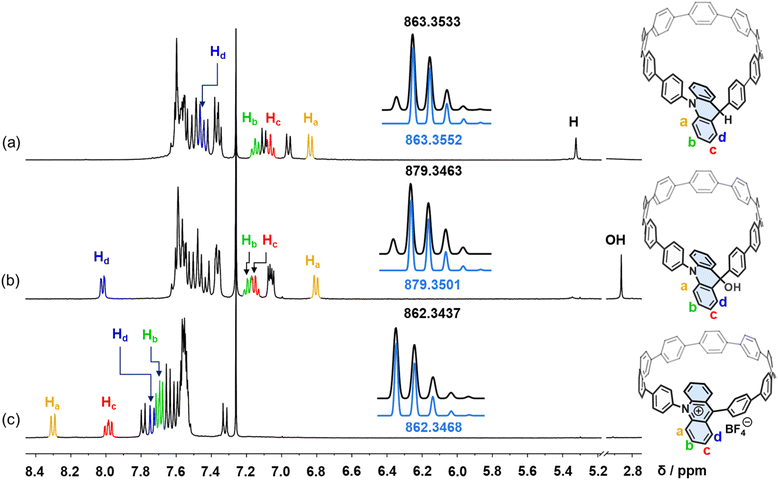
The synthesis of the target molecule commenced with a macrocyclization step through a double-site Suzuki–Miyaura cross-coupling reaction between the bent dibromoacridine derivative 3 and the C-shaped diboronate15,64 7 (Fig. 1a). Our initial strategy anticipated that the SnCl2-mediated reductive aromatization65 would yield the desired acridinium product. However, the compound obtained did not align with our expectations; the 1H NMR spectrum exhibited an unanticipated signal at 5.32 ppm, devoid of the characteristic deshielded aromatic signals above 8 ppm (Fig. 2a and S8). Moreover, the mass spectrometry data deviated by approximately one unit (Fig. S21). Single-crystal X-ray diffraction analysis ultimately confirmed that the product was a macrocycle with the acridinium unit hydrogenated (Ad[10]CPP-H), featuring an sp3-hybridized carbon within the acridine moiety, which imparted a teardrop shape to the macrocycle with dimensions of 16.7 Å along the long axis and 11.0 Å along the short axis (Fig. 1b). The over-reduction, while not the desired outcome, was not detrimental, as the product remained a macrocycle with a structure closely resembling that of our target. Subsequently, we endeavored to oxidize Ad[10]CPP-H using 2,3-dichloro-5,6-dicyano-1,4-benzoquinone (DDQ) to achieve the acridinium macrocycle. Despite our efforts, the product again closely resembled the expected macrocycle but exhibited an additional signal at 2.86 ppm in the 1H NMR spectrum, with the mass data offset by around 17 units (Fig. 2b, S11 and S22). Single-crystal X-ray diffraction revealed a macrocycle of similar teardrop shape (Ad[10]CPP-OH), with the only difference being the oxidation of a C–H bond to a C–OH group compared with Ad[10]CPP-H (Fig. 1c).With the macrocycle in the anticipated oxidative state, we proceeded to acidify the sample using HBF4 to protonate the hydroxy group and induce dehydration, aiming to convert the macrocycle into the cationic acridinium-functionalized final product (Ad[10]CPP·BF4). The 1H NMR spectrum demonstrated a complete transformation from Ad[10]CPP-OH to a new species, with the characteristic deshielded aromatic signal for Ha at 8.30 ppm and Hc at 7.99 ppm (Fig. 2c and S15). The high resolution MALDI-TOF MS spectrum confirmed the expected molecular weight of the acridinium macrocyclic cation (Fig. S23), thereby validating the successful synthesis of the desired macrocycle. Ad[10]CPP·TFA is also obtained by treating Ad[10]CPP-OH with excess trifluoracetic acid (TFA). The isolation of a solid Ad[10]CPP·BF4 or Ad[10]CPP·TFA sample proved to be elusive despite extensive efforts, attributed to the intrinsic instability of the cationic macrocycle under neutral conditions. Specifically, the acridinium moiety is highly vulnerable to nucleophilic attack, readily reverting to Ad[10]CPP-OH upon exposure to moisture. A TFA titration experiment revealed that the interconversion between Ad[10]CPP+ and Ad[10]CPP-OH occurs within the TFA range between 1.0 × 10−4 and 1.0 × 10−5 M in CH2Cl2 (Fig. S26). This highlights the dependency of the thermodynamic stability of the cationic macrocycle on acidic conditions. Strain energy calculations revealed a significant increase from 38.7 kcal mol−1 for Ad[10]CPP-OH to 56.2 kcal mol−1 for Ad[10]CPP+. This substantial increase in ring strain imposes a considerable thermodynamic penalty, thereby disfavoring the formation of the cationic species (Fig. S73 and Table S5). Furthermore, the instability also hinders the crystal growth of the cationic macrocycle. Specifically, the gradual evaporation of the volatile acid (e.g., TFA), which is essential for maintaining the cationic state, prevented the growth of diffraction-quality crystals over time. Despite months of dedicated efforts, including crystallization attempts in a nitrogen-filled glovebox, we were unable to obtain a single crystal suitable for X-ray diffraction analysis. Consequently, we employed density functional theory (DFT) calculations at the B3LYP-D3(BJ)/def2-SVP level to elucidate the structural characteristics of the acridinium macrocycle. The computational results depicted an elliptical nanoring with a long axis of 14.0 Å and a short axis of 13.6 Å. The acridinium aromatic plane is oriented at an angle of 61.0° relative to the macrocyclic plane (Fig. 1d). The torsion angles between the acridinium unit and its adjacent phenylene moieties were measured to be 51.5° and 67.3°, respectively. These values are notably larger than those between other phenylene rings (22.1° to 36.5°), a difference attributed to the steric hindrance between the close-contacted hydrogen atoms on acridinium and adjacent phenylene units (Fig. S76).
Photophysical properties
With the acridinium macrocycle in hand, we proceeded to investigate the photophysical properties of this compound together with its two precursor macrocycles (Fig. 3a and Table S1). The absorption spectra of Ad[10]CPP-H and Ad[10]CPP-OH were nearly identical, both exhibiting overlapping absorption bands between 300 and 440 nm and lacking a broad charge-transfer band. Both compounds showed an absorption maximum (λmax) at 326 nm, accompanied by a shoulder at approximately 378 nm. Time-dependent density functional theory (TD-DFT) calculations indicate that the major absorption at 326 nm arises from a combination of transitions: for Ad[10]CPP-H, these are HOMO−2 → LUMO, HOMO−1 → LUMO+1 and HOMO → LUMO+1 (Table S6); for Ad[10]CPP-OH, they are HOMO−2 → LUMO and HOMO → LUMO+1 (Table S7). In both Ad[10]CPP-H and Ad[10]CPP-OH, the HOMO is primarily confined to the oligoparaphenylene loop, indicating that the acridine segment acts as an electron donor in these neutral systems. A more pronounced spectroscopic change was observed upon protonating Ad[10]CPP-OH to form Ad[10]CPP+. The absorption maximum λmax of Ad[10]CPP+ was observed at 332 nm (Fig. 3a), which is slightly blue-shifted compared to [10]CPP (λmax = 341 nm)66 but red-shifted relative to Ad[10]CPP-OH (λmax = 326 nm). The main absorption band of Ad[10]CPP+ is attributed to HOMO−1 → LUMO+1 and HOMO → LUMO+2 transitions (Table S8). Additionally, a shoulder peak appeared at around 365 nm, along with a broad characteristic charge-transfer band spanning from 420 nm to nearly 700 nm. TD-DFT calculations attribute this broad band to several transitions, including HOMO−4 → LUMO, HOMO−2 → LUMO, HOMO−1 → LUMO, and HOMO → LUMO. The frontier molecular orbital calculations further reveal that the HOMO of Ad[10]CPP+ is localized on the oligoparaphenylene loop, while the LUMO is confined to the acridinium unit and its adjacent aromatic rings (Fig. 3b). This distinct spatial separation of orbitals is characteristic of a donor–acceptor system. Compared to Ad[10]CPP-OH, the HOMO and LUMO energy levels of Ad[10]CPP+ are lowered by approximately 2.11 eV and 0.2 eV (Fig. 3b). This decrease in frontier molecular orbital energy is attributed to the formation of a rigid cationic framework and electron delocalization within the acridinium fragment, which significantly stabilizes the entire π-system, particularly the HOMO.The fluorescence properties of Ad[10]CPP-H and Ad[10]CPP-OH were found to be similar. Both exhibited emission bands at nearly identical wavelengths (λem = 467 nm for Ad[10]CPP-H and 466 nm for Ad[10]CPP-OH; Fig. 3a) and high absolute fluorescence quantum yields (ΦF = 70.9% for Ad[10]CPP-H and 67.3% for Ad[10]CPP-OH; Fig. S32 and S33). Their singlet excited-state lifetimes (τ) were measured via time-resolved fluorescence spectroscopy to be 2.48 ns for Ad[10]CPP-H and 2.40 ns for Ad[10]CPP-OH, respectively (Fig. S35 and S36). A pronounced change was observed upon protonation of Ad[10]CPP-OH. The gradual addition of TFA to Ad[10]CPP-OH led to significant fluorescence quenching, with emission nearly completely suppressed upon full conversion to Ad[10]CPP+ (Fig. S27). The cationic species Ad[10]CPP+ itself displayed very weak fluorescence with an emission maximum blue-shifted to 435 nm (Fig. 3a). This weak emission further decreased in intensity and underwent an additional blue shift as the concentration of TFA was further elevated (Fig. S28). The stability of Ad[10]CPP+ is contingent on excess acid, a condition that inevitably modifies its photophysical behavior, resulting in a pronounced acid concentration-dependent blue shift emission. This phenomenon likely arises because the acidic environment alters the excited-state decay pathway, potentially favoring radiative transition from a higher-energy localized excited (LE) state on the acridinium moiety (Fig. S30). The absolute fluorescence quantum yield of Ad[10]CPP+ was determined to be only 0.95% (Fig. S34) and the singlet excited-state lifetime was measured to be 1.88 ns (Fig. S37). Based on these values, the nonradiative decay rate constant (knr = 5.3 × 108 s−1) is significantly higher than the radiative decay rate constant (kr = 5.1 × 106 s−1), indicating that the excited-state energy of Ad[10]CPP+ is primarily dissipated through non-radiative pathways. To gain further insight into the nature of the emissive state, we examined the emission spectra of Ad[10]CPP+ in solvents of varying polarity (CCl4, CHCl3, C2H2Cl4, CH2Cl2, and C2H4Cl2). A small negative solvatochromic effect of approximately 20 nm was observed (Fig. S29), suggesting that the emissive state is likely a LE state with only minor ICT character.67,68 Several donor–acceptor cycloparaphenylene derivatives have been reported to exhibit low quantum yields due to significant spatial HOMO–LUMO separation that promotes non-radiative deactivation of the excited state.34,35,69–77
Electrochemical properties
The electrochemical behavior of Ad[10]CPP+ was elucidated through cyclic voltammetry (CV) and differential pulse voltammetry (DPV), with DPAd+ serving as a comparative control (Fig. 3c, S41 and S42). The reference compound DPAd·BF4 revealed two distinct reduction waves at −0.49 V and −1.45 V (vs. Ag/AgCl), respectively.78 These waves correspond to two single-electron reduction processes, where DPAd·BF4 first undergoes a reversible one-electron reduction to a radical species, followed by an irreversible one-electron reduction to form an anionic intermediate, which then captures a proton to afford DPAd-H (Fig. S38a).79–81 Ad[10]CPP·BF4 displays similar two single-electron reduction processes with two notable distinctions. Firstly, the redox waves were shifted to a more negative potential by approximately 400 mV, appearing at −1.01 V and −1.94 V, respectively. Secondly, the transition to the radical state exhibited a semi-reversible nature (Fig. S38b). The calculated radical state Ad[10]CPP˙ shows a geometry between the teardrop shape of Ad[10]CPP-H/Ad[10]CPP-OH and the close-to-circle shape of Ad[10]CPP+, namely, the ratio of the long axis and short axis of the radical state falls between them (Fig. S74). It is postulated that a geometrical change occurs upon the formation of the radical species, which, when attempting to revert the acridinium form, requires the molecular framework to overcome significant ring strain. In our subsequent efforts, we attempted to generate and characterize Ad[10]CPP˙, the radical state of the macrocycle. As previously noted, the acridinium form Ad[10]CPP+ is only stable under moderately acidic conditions, which posed a challenge for generating radicals through reduction with active metals or cobaltocene, as these reagents tend to react with the acid first.To circumvent this issue, we tried an alternative strategy involving the oxidation of Ad[10]CPP-H to achieve the radical state by treating Ad[10]CPP-H with nitrosonium tetrafluoroborate (NOBF4), a well-known one-electron oxidizing agent. To our surprise, Ad[10]CPP-dimers were generated after quenching the solution with air, as indicated in the HR-MS spectra (Fig. S24). This result suggests that the generated radical species is highly prone to dimerization. We were able to isolate the dimers. The 1H NMR and 1H–1H COSY spectra (Fig. S52 and S53) indicated a mixture of two compounds in a ∼7![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3 ratio with very similar structures, as they were not differentiated by diffusion constants in the DOSY spectrum (Fig. S55) or the analytical HPLC trace (Fig. S56). This also explains the lack of hyperfine splitting patterns in the EPR spectrum (Fig. S47a). Further oxidizing the isolated dimers produces a broad near-infrared absorption band in the UV-vis-NIR spectrum ranging from 1000 nm to 2000 nm, with λmax at 1574 nm (Fig. S58), possibly coming from diradical dications of the dimers.82 A plausible mechanism for the formation of Ad[10]CPP-dimers is illustrated in Fig. S54. Oxidation of Ad[10]CPP-H generates a prochiral radical cation, which can dimerize by forming a new σ-bond at the para positions relative to the nitrogen atom. This process, accompanied by the loss of two protons, yields a meso dimer and a pair of racemic dimers. The inherent strain in Ad[10]CPP-H induces an electronic effect that governs a highly selective dimerization pathway, leading to the formation of giant double nanohoops.
3 ratio with very similar structures, as they were not differentiated by diffusion constants in the DOSY spectrum (Fig. S55) or the analytical HPLC trace (Fig. S56). This also explains the lack of hyperfine splitting patterns in the EPR spectrum (Fig. S47a). Further oxidizing the isolated dimers produces a broad near-infrared absorption band in the UV-vis-NIR spectrum ranging from 1000 nm to 2000 nm, with λmax at 1574 nm (Fig. S58), possibly coming from diradical dications of the dimers.82 A plausible mechanism for the formation of Ad[10]CPP-dimers is illustrated in Fig. S54. Oxidation of Ad[10]CPP-H generates a prochiral radical cation, which can dimerize by forming a new σ-bond at the para positions relative to the nitrogen atom. This process, accompanied by the loss of two protons, yields a meso dimer and a pair of racemic dimers. The inherent strain in Ad[10]CPP-H induces an electronic effect that governs a highly selective dimerization pathway, leading to the formation of giant double nanohoops.
Host and guest interactions
With the structure of Ad[10]CPP+ well characterized, we proceeded to evaluate its host capabilities for binding guest molecules, focusing on C60 due to the expected similarity in cavity size between Ad[10]CPP+ and [10]CPP.45 Significant chemical shift changes in the 1H NMR spectra of Ad[10]CPP·TFA upon the addition of C60 provided clear evidence of strong supramolecular interactions between the two species (Fig. S60). The binding constant of Ad[10]CPP·TFA and C60 was determined to be (2.18 ± 0.27) × 104 M−1 (Fig. S61) by NMR titration experiments, which is smaller than that of [10]CPP, (2.79 ± 0.03) × 106 M−1, for C60 characterized by fluorescence-quenching experiments.44After multiple attempts, we successfully obtained single crystals of the host–guest complex C60⊂Ad[10]CPP·TFA suitable for X-ray diffraction analysis. The crystal structure unambiguously confirms a 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 host–guest complex, with the C60 guest residing within the cavity of Ad[10]CPP·TFA (Fig. 4). The cycloparaphenylene host exhibits a slightly elliptical shape, with a long and short axis of 14.0 Å and 13.6 Å, respectively (Fig. 4a and b). Further structural analysis reveals more compact supramolecular interactions compared to C60⊂[10]CPP, as evidenced by the shorter average distance (3.21 Å) from the equatorial carbon atoms of C60 to the nearest phenylene plane of the host, relative to the 3.44 Å observed in the latter45 (Fig. S71). The torsion angles between the acridinium and its adjacent phenylene moieties are 53.8° and 75.8°, respectively, markedly larger than those between other phenylene rings (8.3°–29.0°), which can be attributed to steric repulsion from the hydrogen atoms at the acridinium–phenylene junctions (Fig. S72). Additionally, the acridinium fragment forms a dihedral angle of 57.2° with the macrocyclic plane (Fig. 4b). Collectively, we propose that this strained, elliptical structure imposes a certain compression upon C60 encapsulation, which diminishes the optimal host–guest complementarity achieved in the nearly circular [10]CPP complex, thereby accounting for the lower binding constant.
1 host–guest complex, with the C60 guest residing within the cavity of Ad[10]CPP·TFA (Fig. 4). The cycloparaphenylene host exhibits a slightly elliptical shape, with a long and short axis of 14.0 Å and 13.6 Å, respectively (Fig. 4a and b). Further structural analysis reveals more compact supramolecular interactions compared to C60⊂[10]CPP, as evidenced by the shorter average distance (3.21 Å) from the equatorial carbon atoms of C60 to the nearest phenylene plane of the host, relative to the 3.44 Å observed in the latter45 (Fig. S71). The torsion angles between the acridinium and its adjacent phenylene moieties are 53.8° and 75.8°, respectively, markedly larger than those between other phenylene rings (8.3°–29.0°), which can be attributed to steric repulsion from the hydrogen atoms at the acridinium–phenylene junctions (Fig. S72). Additionally, the acridinium fragment forms a dihedral angle of 57.2° with the macrocyclic plane (Fig. 4b). Collectively, we propose that this strained, elliptical structure imposes a certain compression upon C60 encapsulation, which diminishes the optimal host–guest complementarity achieved in the nearly circular [10]CPP complex, thereby accounting for the lower binding constant.
In order to assess the stabilizing effect of C60 encapsulation on the Ad[10]CPP+ host, we conducted systematic titration experiments using TFA. The protonation processes of Ad[10]CPP-OH by TFA were monitored via characteristic NMR signals, both in the presence and absence of C60 (Fig. S62 and S63). A change in solvent (CH2Cl2 to C2Cl4D2) lowered the concentration of TFA required for the complete conversion of Ad[10]CPP-OH to Ad[10]CPP+. At a TFA concentration of 1.0 × 10−4 M, the conversion of free Ad[10]CPP-OH was negligible, whereas partial conversion was achieved when C60 was present. This stabilization effect was further confirmed at a TFA concentration of 2.0 × 10−4 M, where Ad[10]CPP-OH alone underwent incomplete conversion, but full conversion was achieved in the presence of excess C60 (Fig. S64 and S65). These results demonstrate that host–guest complexation stabilizes the vulnerable structure of the host, thereby reducing the acid requirement for stabilizing the cationic host structure. This provides direct evidence that C60 substantially reinforces the molecular scaffold via π–π interactions. The encapsulation improves its stability against atmospheric moisture compared to the empty host, enabling the successful growth of single crystals suitable for X-ray diffraction, ultimately allowing us to resolve the structure of the C60⊂Ad[10]CPP·TFA complex.
Stimulus responsive system
With the binding affinity between Ad[10]CPP·TFA and C60 measured and the structure of host–guest complex characterized, we then explored the acid–base stimuli responsive properties of the host–guest complex (Fig. 5a). Gradually adding five equivalents of TFA to an NMR tube containing a CD2Cl2 solution of Ad[10]CPP-OH (Fig. 5b) caused a significant chemical shift change in the proton associated with the acridinium moiety. Specifically, Ha shifted from 6.78 ppm to 8.37 ppm, indicating complete transformation into Ad[10]CPP·TFA (Fig. 5c). Subsequently, adding 1.5 equivalents of C60 to the same NMR tube caused Ha to shift further downfield to 8.64 ppm, attributed to the formation of the host–guest complex C60⊂Ad[10]CPP·TFA (Fig. 5d). Adding five equivalents of triethylamine (TEA) to the solution caused the Ha signal to revert to 6.78 ppm, indicating that the macrocycle had completely transformed back into Ad[10]CPP-OH (Fig. 5e). Since the cavity of Ad[10]CPP-OH is not suitable for C60 (Fig. S59), the guest molecule is then released into the solution. The acid–base switch process is reversible for at least 5 cycles, as monitored by 1H NMR spectroscopy (Fig. 5f and S67).Conclusions
In summary, we successfully synthesized and characterized an acridinium-functionalized cycloparaphenylene derivative, Ad[10]CPP+. Structural characterization indicates that Ad[10]CPP+ adopts a nearly circular configuration similar to [10]CPP, with increased torsion angles in the acridinium moiety due to steric hindrance. Electrochemical studies reveal two well-separated reduction peaks for Ad[10]CPP+, suggesting its potential to form a radical species, Ad[10]CPP˙. However, attempts to generate Ad[10]CPP˙ through oxidation resulted in its dimerization, yielding giant double nanohoops. Ad[10]CPP+ also demonstrates strong host–guest interactions with electron-rich fullerenes, as evidenced by NMR spectroscopy and single-crystal X-ray analysis. Furthermore, Ad[10]CPP+ can undergo reversible structural transformations in response to chemical stimuli, enabling controlled capture and release of fullerene guests under alternating acidic and basic conditions for multiple cycles. The acid–base triggered guest capture and release of Ad[10]CPP+ represents a valuable addition to the cycloparaphenylene family. Its key advantage lies in the highly confined cavity and switchable cationic character. Our current efforts are focused on leveraging multi-acridinium motifs to develop advanced functional systems for applications in sensing wider guests.Author contributions
C. C. conceived and supervised the project. J. M. performed most of the synthesis and characterization experiments. J. Z. and P. Y. conducted the calculations. Y. H., J. J., X. Z. and H. Z. helped with synthesis of starting materials. K. L., D. Z., and P. L. helped with characterization. All authors contributed to manuscript writing.Conflicts of interest
There are no conflicts to declare.Data availability
CCDC 2449558, 2449559 and 2449561 contain the supplementary crystallographic data for this paper.83a–cSupplementary information (SI): additional experimental details, materials and methods, 1H and 13C NMR, (1H, 1H)-COSY NMR, MS, and UV-vis-IR spectra, EPR spectra, crystallographic data, and computational details (PDF). See DOI: https://doi.org/10.1039/d6sc01132b.
Acknowledgements
This work was supported by the Fundamental Research Funds for the Central Universities and NSFC 22171193. We thank Professor Lihua Yuan and Professor Xiaowei Li at Sichuan University for their help with UV-vis and fluorescence spectroscopy. We thank Yuxuan He, Professor Zhengyang Bin and Professor Jinsong You at Sichuan University for their help with the measurement of lifetimes of the singlet excited states and the fluorescence quantum yield. We thank Yimeng Hu, Professor Liangfang Zhu and Professor Changwei Hu at Sichuan University for UV-Vis-NIR spectroscopy. We thank Lingyan Sun and Professor Cheng Zhang at Sichuan University for their help with HPLC chromatograms. We thank Dr Jing Li, Dr Chunxia Wang and Dr Dongyan Deng from Sichuan University for HR-MS and NMR testing, respectively. We thank Dr Fucheng Leng at Westlake University for the single-crystal crystallography. Computational work was supported by the Center for Computational Science and Engineering and the CHEM High-Performance Supercomputer Cluster (CHEM-HPC) of the Department of Chemistry, Southern University of Science and Technology.References
- R. Jasti, J. Bhattacharjee, J. B. Neaton and C. R. Bertozzi, Synthesis, Characterization, and Theory of [9]-, [12]-, and [18]Cycloparaphenylene: Carbon Nanohoop Structures, J. Am. Chem. Soc., 2008, 130, 17646–17647 CrossRef CAS PubMed.
- S. E. Lewis, Cycloparaphenylenes and related nanohoops, Chem. Soc. Rev., 2015, 44, 2221–2304 RSC.
- Y. Segawa, A. Yagi, K. Matsui and K. Itami, Design and Synthesis of Carbon Nanotube Segments, Angew. Chem., Int. Ed., 2016, 55, 5136–5158 CrossRef CAS PubMed.
- Y. Xu and M. von Delius, The Supramolecular Chemistry of Strained Carbon Nanohoops, Angew. Chem., Int. Ed., 2020, 59, 559–573 CrossRef CAS PubMed.
- J. Wang, X. Zhang, H. Jia, S. Wang and P. Du, Large π-Extended and Curved Carbon Nanorings as Carbon Nanotube Segments, Acc. Chem. Res., 2021, 54, 4178–4190 CrossRef CAS PubMed.
- Y. Li, H. Kono, T. Maekawa, Y. Segawa, A. Yagi and K. Itami, Chemical Synthesis of Carbon Nanorings and Nanobelts, Acc. Mater. Res., 2021, 2, 681–691 CrossRef CAS.
- X. Li, S. Guo and H. Jiang, Recent advances in functionalized cycloparaphenylenes: from molecular design to applications, Chem. Commun., 2025, 61, 9836–9852 RSC.
- M. Hermann, D. Wassy and B. Esser, Conjugated Nanohoops Incorporating Donor, Acceptor, Hetero- or Polycyclic Aromatics, Angew. Chem., Int. Ed., 2021, 60, 15743–15766 CrossRef CAS PubMed.
- E. Kayahara, R. Qu and S. Yamago, Bromination of Cycloparaphenylenes: Strain-Induced Site-Selective Bis-Addition and Its Application for Late-Stage Functionalization, Angew. Chem., Int. Ed., 2017, 56, 10428–10432 CrossRef CAS PubMed.
- E. Kayahara, T. Hayashi, K. Takeuchi, F. Ozawa, K. Ashida, S. Ogoshi and S. Yamago, Strain-Induced Double Carbon–Carbon Bond Activations of Cycloparaphenylenes by a Platinum Complex: Application to the Synthesis of Cyclic Diketones, Angew. Chem., Int. Ed., 2018, 57, 11418–11421 CrossRef CAS PubMed.
- T. Terabayashi, E. Kayahara, Y. Zhang, Y. Mizuhata, N. Tokitoh, T. Nishinaga, T. Kato and S. Yamago, Synthesis of Twisted [n]Cycloparaphenylene by Alkene Insertion, Angew. Chem., Int. Ed., 2023, 62, e202214960 CrossRef CAS PubMed.
- J. M. Fehr, N. Myrthil, A. L. Garrison, T. W. Price, S. A. Lopez and R. Jasti, Experimental and theoretical elucidation of SPAAC kinetics for strained alkyne-containing cycloparaphenylenes, Chem. Sci., 2023, 14, 2839–2848 RSC.
- T. D. Clayton, J. M. Fehr, T. W. Price, L. N. Zakharov and R. Jasti, Pinwheel-like Curved Aromatics from the Cyclotrimerization of Strained Alkyne Cycloparaphenylenes, J. Am. Chem. Soc., 2024, 146, 30607–30614 CrossRef CAS PubMed.
- K. Lan, C. Zhang, Y. Li, C. Hu, Z. Su and C. Cheng, Strain Energy Prompted Tunable Aggregation-Induced Emission Property of Tetraphenylethylene, Angew. Chem., Int. Ed., 2025, 64, e202507763 CrossRef CAS PubMed.
- K. Lan, S. Zhang, Y. Lu, P. Yu, J. Chen and C. Cheng, Strain Energy Induced Rotary Speed Acceleration in a Light- Driven Molecular Motor, Angew. Chem., Int. Ed., 2025, 64, e202507487 CrossRef CAS PubMed.
- Z. A. Huang, C. Chen, X. D. Yang, X. B. Fan, W. Zhou, C. H. Tung, L. Z. Wu and H. Cong, Synthesis of Oligoparaphenylene-Derived Nanohoops Employing an Anthracene Photodimerization-Cycloreversion Strategy, J. Am. Chem. Soc., 2016, 138, 11144–11147 CrossRef CAS PubMed.
- Y. Segawa, M. Kuwayama and K. Itami, Synthesis and Structure of [9]Cycloparaphenylene Catenane: An All-Benzene Catenane Consisting of Small Rings, Org. Lett., 2020, 22, 1067–1070 CrossRef CAS PubMed.
- K. Li, S. Yoshida, R. Yakushiji, X. Liu, C. Ge, Z. Xu, Y. Ni, X. Ma, J. Wu, S. Sato and Z. Sun, Molecular cylinders with donor–acceptor structure and swinging motion, Chem. Sci., 2024, 15, 18832–18839 RSC.
- G. Li, X. Wang, L. L. Mao, J. N. Gao, X. Shi, H. Li, Z. T. Li, Z. Y. Huo, Y. Chen, H. B. Yang, J. Chen, C. H. Tung, L. Z. Wu and H. Cong, Tubular All-Benzene Nanocarbon with Evolving Excited-State Chirality, Angew. Chem., Int. Ed., 2025, e202518587 CAS.
- W. Shi, Y. Hu, L. Leanza, Y. Shchukin, P. A. Hoffmann, M.-H. Li, C. Ning, Z.-Y. Cao, Y.-Q. Xu, P. Du, M. von Delius, G. M. Pavan and Y. Xu, Ring-in-Ring Assembly Facilitates the Synthesis of a [12]Cycloparaphenylene ABC-Type [3]Catenane, Angew. Chem., Int. Ed., 2025, 64, e202421459 CrossRef CAS PubMed.
- L. Zhan, C. Dai, G. Zhang, J. Zhu, S. Zhang, H. Wang, Y. Zeng, C. H. Tung, L. Z. Wu and H. Cong, A Conjugated Figure-of-Eight Oligoparaphenylene Nanohoop with Adaptive Cavities Derived from Cyclooctatetrathiophene Core, Angew. Chem., Int. Ed., 2022, 61, e202113334 CrossRef CAS PubMed.
- P. Fang, M. Chen, N. Yin, G. Zhuang, T. Chen, X. Zhang and P. Du, Regulating supramolecular interactions in dimeric macrocycles, Chem. Sci., 2023, 14, 5425–5430 RSC.
- A. A. Kamin, T. D. Clayton, C. E. Otteson, P. M. Gannon, S. Krajewski, W. Kaminsky, R. Jasti and D. J. Xiao, Synthesis and metalation of polycatechol nanohoops derived from fluorocycloparaphenylenes, Chem. Sci., 2023, 14, 9724–9732 RSC.
- M. S. Mirzaei, S. Mirzaei, V. M. Espinoza Castro, C. Lawrence and R. Hernández Sánchez, Dual molecular tweezers extending from a nanohoop, Chem. Commun., 2024, 60, 14236–14239 RSC.
- K. Kovida, J. Malinčík, C. M. Cruz, A. G. Campaña and T. Šolomek, Role of exciton delocalization in chiroptical properties of benzothiadiazole carbon nanohoops, Chem. Sci., 2025, 16, 1405–1410 RSC.
- Y. Hu, T. Li, T. Su, W. Shi, Y. Yu, B. Li, M.-H. Li, S. Zhang, Y.-Q. Xu, Q. Liu, D. Wu and Y. Xu, Crown ether–cycloparaphenylene hybrid multimacrocycles: insights into supramolecular gas sensing and biological potential, Chem. Sci., 2025, 16, 13115–13121 RSC.
- T. Kato, D. Imoto, A. Yagi and K. Itami, Making non-emissive [6]cycloparaphenylene fluorescent by simple multiple methyl substitution, Chem. Sci., 2025, 16, 18952–18958 RSC.
- N. Liu, Y. Choi, Z. Zong, T. Feng, T. Kim and X.-S. Ke, BODIPY-Cycloparaphenylene Nanorings, CCS Chem., 2025, 7, 3650–3663 CrossRef CAS.
- M. Katsuma, S. Yasutomo and I. Kenichiro, Synthesis and Properties of Cycloparaphenylene-2,5-pyridylidene: A Nitrogen-Containing Carbon Nanoring, Org. Lett., 2012, 14, 1888–1891 CrossRef PubMed.
- J. M. Van Raden, S. Louie, L. N. Zakharov and R. Jasti, 2,2'-Bipyridyl-Embedded Cycloparaphenylenes as a General Strategy To Investigate Nanohoop-Based Coordination Complexes, J. Am. Chem. Soc., 2017, 139, 2936–2939 CrossRef CAS PubMed.
- Y. Y. Fan, D. Chen, Z. A. Huang, J. Zhu, C. H. Tung, L. Z. Wu and H. Cong, An isolable catenane consisting of two Mobius conjugated nanohoops, Nat. Commun., 2018, 9, 3037 CrossRef PubMed.
- J. H. May, J. M. Van Raden, R. L. Maust, L. N. Zakharov and R. Jasti, Active template strategy for the preparation of π-conjugated interlocked nanocarbons, Nat. Chem., 2023, 15, 170–176 CrossRef CAS PubMed.
- J. H. May, J. M. Fehr, J. C. Lorenz, L. N. Zakharov and R. Jasti, A High-Yielding Active Template Click Reaction (AT−CuAAC) for the Synthesis of Mechanically Interlocked Nanohoops, Angew. Chem., Int. Ed., 2024, 63, e202401823 CrossRef CAS PubMed.
- E. R. Darzi, E. S. Hirst, C. D. Weber, L. N. Zakharov, M. C. Lonergan and R. Jasti, Synthesis, Properties, and Design Principles of Donor-Acceptor Nanohoops, ACS Cent. Sci., 2015, 1, 335–342 CrossRef CAS PubMed.
- J. M. Van Raden, E. R. Darzi, L. N. Zakharov and R. Jasti, Synthesis and characterization of a highly strained donor-acceptor nanohoop, Org. Biomol. Chem., 2016, 14, 5721–5727 RSC.
- T. Drennhaus, D. Imoto, E. S. Horst, L. Lezius, H. Shudo, T. Kato, K. Bergander, C. G. Daniliuc, D. Leifert, A. Yagi, A. Studer and K. Itami, Cycloparaazine, a full-azine carbon nanoring, Nat. Commun., 2025, 16, 4643 CrossRef CAS PubMed.
- T. Wu, J. Yang, Y. Liu, C. Wang, J. Zhang, R.-H. Zheng, H. Yu, R. Liu and D. Lu, Size-controlled synthesis of N-doped [10]CPPs and investigation on the variations of their binding affinities with fullerene, Chin. Chem. Lett., 2025, 112199 CrossRef.
- S. Qiu, Y. Zhao, L. Zhang, Y. Ni, Y. Wu, H. Cong, D.-H. Qu, W. Jiang, J. Wu, H. Tian and Z. Wang, Axially N-Embedded Quasi-Carbon Nanohoops with Multioxidation States, CCS Chem., 2023, 5, 1763–1772 CrossRef CAS.
- X. Zhang, J. Jiang, K. Lan, D. Zhang and C. Cheng, Cycloparaphenylene-Type Hoops with Adaptive Cavities Derived from N,N'-diphenyldihydrodibenzo[a,c]phenazine, Org. Lett., 2025, 27, 10484–10488 CrossRef CAS PubMed.
- R. Frydrych, K. Senthilkumar, K. Ślusarek, M. Waliczek, W. Bury, P. J. Chmielewski, J. Cybińska and M. Stępień, Viologen–cycloparaphenylene hybrids: luminescent molecular nanocarbons for anion binding and specific vapor sorption, Org. Chem. Front., 2025, 12, 437–447 RSC.
- S. Qiu, L. Zhang, D.-H. Qu and Z. Wang, Supercycloalkanes: dihydropyrazine-embedded macrocycles with flexible conformations resembling cycloalkanes, Chem. Sci., 2025, 16, 20464–20472 RSC.
- L. Zhang, S. Qiu, S. Hu, H. Xu, X. Sun, W. Jiang, D.-H. Qu and Z. Wang, Cocrystals of axially N-embedded quasi-carbon nanohoops via modulating the electron affinity of guest acceptors, Sci. China Chem., 2025, 68, 3154–3161 CrossRef CAS.
- D. Zhang, J. Liu, J. Ming, K. Lan, J. Jiang, H. Zhong, X. Zhang, Z. Bin and C. Cheng, Incorporating a Pyridinium into a Cycloparaphenylene: A Cati-onic Donor–Acceptor Nanohoop with Reversible Redox Properties, Sci. China Chem., 2026 DOI:10.1007/s11426-11025-13313-11429.
- T. Iwamoto, Y. Watanabe, T. Sadahiro, T. Haino and S. Yamago, Size-Selective Encapsulation of C60 by [10]Cycloparaphenylene: Formation of the Shortest Fullerene-Peapod, Angew. Chem., Int. Ed., 2011, 50, 8342–8344 CrossRef CAS PubMed.
- J. Xia, J. W. Bacon and R. Jasti, Gram-scale synthesis and crystal structures of [8]- and [10]CPP, and the solid-state structure of C60@[10]CPP, Chem. Sci., 2012, 3, 3018–3021 RSC.
- T. Iwamoto, Y. Watanabe, H. Takaya, T. Haino, N. Yasuda and S. Yamago, Size- and Orientation- Selective Encapsulation of C70 by Cycloparaphenylenes, Chem.–Eur. J., 2013, 19, 14061–14068 CrossRef CAS PubMed.
- A. Stergiou, J. Rio, J. H. Griwatz, D. Arcon, H. A. Wegner, C. P. Ewels and N. Tagmatarchis, A Long-Lived Azafullerenyl Radical Stabilized by Supramolecular Shielding with a [10]Cycloparaphenylene, Angew. Chem., Int. Ed., 2019, 58, 17745–17750 CrossRef CAS PubMed.
- S. Wang, X. Li, X. Zhang, P. Huang, P. Fang, J. Wang, S. Yang, K. Wu and P. Du, A supramolecular polymeric heterojunction composed of an all-carbon conjugated polymer and fullerenes, Chem. Sci., 2021, 12, 10506–10513 RSC.
- E. Ubasart, O. Borodin, C. Fuertes-Espinosa, Y. Xu, C. García-Simón, L. Gómez, J. Juanhuix, F. Gándara, I. Imaz, D. Maspoch, M. von Delius and X. Ribas, A three-shell supramolecular complex enables the symmetry-mismatched chemo- and regioselective bis-functionalization of C60, Nat. Chem., 2021, 13, 420–427 CrossRef CAS PubMed.
- Y. Xu, F. Steudel, M. Y. Leung, B. Xia, M. von Delius and V. W. W. Yam, [n]Cycloparaphenylene- Pillar[5]arene Bismacrocycles: Their Circularly Polarized Luminescence and Multiple Guest Recognition Properties, Angew. Chem., Int. Ed., 2023, 62, e202302978 CrossRef CAS PubMed.
- P. J. Evans, L. N. Zakharov and R. Jasti, Synthesis of carbon nanohoops containing thermally stable cis azobenzene, J. Photochem. Photobiol., A, 2019, 382, 111878 CrossRef CAS.
- X. Li, L. Jia, W. Wang, Y. Wang, D. Sun and H. Jiang, A nonalternant azulene-embedded carbon nanohoop featuring anti-Kasha emission and tunable properties upon pH stimuli-responsiveness, J. Mater. Chem. C, 2023, 11, 1429–1434 RSC.
- T. Ide, W.-C. Huang and M. Horie, Tris-Azo Triangular Paraphenylenes: Synthesis and Reversible Interconversion into Radial π-Conjugated Macrocycles, J. Am. Chem. Soc., 2024, 146, 10246–10250 CrossRef CAS PubMed.
- S. Wu, J. Jie, L. Liu, L. Liu, S. Guo, X. Li, J. He, Z. Lian, Y. Wang, X. Xu, H. Su, X. Chen and H. Jiang, A Redox-Active π-Extended Tetrathiafulvalene-Based Carbon Nanohoop Featuring Unique Kasha/Anti-Kasha Dual Emissions: Structure, Photophysical Properties, and Photoconductivity, Angew. Chem., Int. Ed., 2025, 64, e202512167 CrossRef CAS PubMed.
- K. Kurihara, K. Yazaki, M. Akita and M. Yoshizawa, A Switchable Open/closed Polyaromatic Macrocycle that Shows Reversible Binding of Long Hydrophilic Molecules, Angew. Chem., Int. Ed., 2017, 56, 11360–11364 CrossRef CAS PubMed.
- J. Hu, J. S. Ward, A. Chaumont, K. Rissanen, J. M. Vincent, V. Heitz and H. P. Jacquot de Rouville, A Bis-Acridinium Macrocycle as Multi-Responsive Receptor and Selective Phase-Transfer Agent of Perylene, Angew. Chem., Int. Ed., 2020, 59, 23206–23212 CrossRef CAS PubMed.
- I. A. MacKenzie, L. Wang, N. P. R. Onuska, O. F. Williams, K. Begam, A. M. Moran, B. D. Dunietz and D. A. Nicewicz, Discovery and characterization of an acridine radical photoreductant, Nature, 2020, 580, 76–80 CrossRef CAS PubMed.
- H. P. Jacquot de Rouville, J. Hu and V. Heitz, N-Substituted Acridinium as a Multi-Responsive Recognition Unit in Supramolecular Chemistry, ChemPlusChem, 2021, 86, 110–129 CrossRef CAS PubMed.
- H.-P. Jacquot de Rouville, C. Gourlaouen, D. Bardelang, N. Le Breton, J. S. Ward, L. Ruhlmann, J.-M. Vincent, D. Jardel, K. Rissanen, J.-L. Clément, S. Choua and V. Heitz, Viridium: A Stable Radical and Its π-Dimerization, J. Am. Chem. Soc., 2024, 147, 1823–1830 CrossRef PubMed.
- S. Claude, J.-M. Lehn, F. Schmidt and J.-P. Vigneron, Binding of Nucleosides, Nucleotides and Anionic Planar Substrates by Bis-lntercaland Receptor Molecules, J. Chem. Soc., Chem. Commun., 1991, 1182–1185 RSC.
- H. Kawai, T. Takeda, K. Fujiwara, M. Wakeshima, Y. Hinatsu and T. Suzuki, Ultralong Carbon–Carbon Bonds in Dispirobis(10-methylacridan) Derivatives with an Acenaphthene, Pyracene, or Dihydropyracylene Skeleton, Chem.–Eur. J., 2008, 14, 5780–5793 CrossRef CAS PubMed.
- A. Petitjean, R. G. Khoury, N. Kyritsakas and J.-M. Lehn, Dynamic Devices. Shape Switching and Substrate Binding in Ion-Controlled Nanomechanical Molecular Tweezers, J. Am. Chem. Soc., 2004, 126, 6637–6647 CrossRef CAS PubMed.
- W. Abraham, K. Buck, M. Orda-Zgadzaj, S. Schmidt-Schäffer and U.-W. Grummt, Novel photoswitchable rotaxanes, Chem. Commun., 2007, 3094–3096 RSC.
- X. Zhang, K. Lan and C. Cheng, Figure-Eight Bismacrocycles Derived from a Tetraphenylmethane Core and Oligoparaphenylene Loops, Org. Lett., 2024, 26, 7853–7857 CrossRef CAS PubMed.
- V. K. Patel, E. Kayahara and S. Yamago, Practical Synthesis of [n]Cycloparaphenylenes (n=5, 7-12) by H2SnCl4-Mediated Aromatization of 1,4-Dihydroxycyclo-2,5-diene Precursors, Chem.–Eur. J., 2015, 21, 5742–5749 CrossRef CAS PubMed.
- T. Iwamoto, Y. Watanabe, Y. Sakamoto, T. Suzuki and S. Yamago, Selective and Random Syntheses of [n]Cycloparaphenylenes (n = 8-13) and Size Dependence of Their Electronic Properties, J. Am. Chem. Soc., 2011, 133, 8354–8361 CrossRef CAS PubMed.
- B. Carlotti, A. Cesaretti, C. G. Fortuna, A. Spalletti and F. Elisei, Experimental evidence of dual emission in a negatively solvatochromic push–pull pyridinium derivative, Phys. Chem. Chem. Phys., 2015, 17, 1877–1882 RSC.
- P. Sumsalee, P. Morgante, G. Pieters, J. Crassous, J. Autschbach and L. Favereau, Negative solvatochromism and sign inversion of circularly polarized luminescence in chiral exciplexes as a function of solvent polarity, J. Mater. Chem. C, 2023, 11, 8514–8523 RSC.
- T. Kuwabara, J. Orii, Y. Segawa and K. Itami, Curved Oligophenylenes as Donors in Shape-Persistent Donor-Acceptor Macrocycles with Solvatofluorochromic Properties, Angew. Chem., Int. Ed., 2015, 54, 9646–9649 CrossRef CAS PubMed.
- T. C. Lovell, Z. R. Garrison and R. Jasti, Synthesis, Characterization, and Computational Investigation of Bright Orange-Emitting Benzothiadiazole [10]Cycloparaphenylene, Angew. Chem., Int. Ed., 2020, 59, 14363–14367 CrossRef CAS PubMed.
- D. Chen, Y. Wada, Y. Kusakabe, L. Sun, E. Kayahara, K. Suzuki, H. Tanaka, S. Yamago, H. Kaji and E. Zysman-Colman, A Donor-Acceptor 10-Cycloparaphenylene and Its Use as an Emitter in an Organic Light-Emitting Diode, Org. Lett., 2023, 25, 998–1002 CrossRef CAS PubMed.
- V. Bliksted Roug Pedersen, T. W. Price, N. Kofod, L. N. Zakharov, B. W. Laursen, R. Jasti and M. Brøndsted Nielsen, Synthesis and Properties of Fluorenone-Containing Cycloparaphenylenes and Their Late-Stage Transformation, Chem.–Eur. J., 2023, 30, e202303490 CrossRef PubMed.
- P. Fang, Z. Cheng, W. Peng, J. Xu, X. Zhang, F. Zhang, G. Zhuang and P. Du, A Strained Donor-Acceptor Carbon Nanohoop: Synthesis, Photophysical and Charge Transport Properties, Angew. Chem., Int. Ed., 2024, 63, e202407078 CrossRef CAS PubMed.
- C. Brouillac, E. Dureau, O. Jeannin, J. Rault-Berthelot, C. Poriel and C. Quinton, Donor–Acceptor Nanohoops: Impact of the Ratio and Arrangement of the Fluorenone and Carbazole Moieties, J. Am. Chem. Soc., 2025, 147, 11267–11276 CrossRef CAS PubMed.
- S. Guo, L. Liu, L. Liu, Y. Fan, H. Yang, J. He, Y. Wang, Z. Bo, X. Xu, X. Chen and H. Jiang, Naphthalene Diimide-Embedded Donor-Acceptor Carbon Nanohoops: Photophysical, Photoconductive, and Charge Transport Properties, ACS Appl. Mater. Interfaces, 2025, 17, 5202–5212 CrossRef PubMed.
- X. Li, L. Liu, L. Jia, Z. Lian, J. He, S. Guo, Y. Wang, X. Chen and H. Jiang, Acceptor engineering of quinone-based cycloparaphenylenes via post-synthesis for achieving white-light emission in single-molecule, Nat. Commun., 2025, 16, 467 CrossRef CAS PubMed.
- T. C. Lovell, K. G. Fosnacht, C. E. Colwell and R. Jasti, Effect of curvature and placement of donor and acceptor units in cycloparaphenylenes: a computational study, Chem. Sci., 2020, 11, 12029–12035 RSC.
- T. P. Vaid, A. K. Lytton-Jean and B. C. Barnes, Investigations of the 9,10-Diphenylacridyl Radical as an Isostructural Dopant for the Molecular Semiconductor 9,10-Diphenylanthracene, Chem. Mater., 2003, 15, 4292–4299 CrossRef CAS.
- P. Hapiot, J. Moiroux and J.-M. Savéant, Electrochemistry of NADH/NAD+ Analogues. A Detailed Mechanistic Kinetic and Thermodynamic Analysis of the 10-Methylacridan/10-Methylacridinium Couple in Acetonitrile, J. Am. Chem. Soc., 1990, 112, 1337–1343 CrossRef CAS.
- D. T. Hogan and T. C. Sutherland, Modern Spin on the Electrochemical Persistence of Heteroatom-Bridged Triphenylmethyl-Type Radicals, J. Phys. Chem. Lett., 2018, 9, 2825–2829 CrossRef CAS PubMed.
- S. A. Jonker, F. Ariese and J. W. Verhoeven, Cation complexation with functionalized 9-arylacridinium ions: Possible applications in the development of cation-selective optical probes, Recl. Trav. Chim. Pays-Bas, 1989, 108, 109–115 CrossRef CAS.
- K. Kamada, S.-i. Fuku-en, S. Minamide, K. Ohta, R. Kishi, M. Nakano, H. Matsuzaki, H. Okamoto, H. Higashikawa, K. Inoue, S. Kojima and Y. Yamamoto, Impact of Diradical Character on Two-Photon Absorption: Bis(acridine) Dimers Synthesized from an Allenic Precursor, J. Am. Chem. Soc., 2012, 135, 232–241 CrossRef PubMed.
- (a) CCDC 2449558: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2n6z0w; (b) CCDC 2449559: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2n6z1x; (c) CCDC 2449561: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2n6z3z.
| This journal is © The Royal Society of Chemistry 2026 |