Open Access Article
Open Access ArticleA dual-function molecule enables stable four-electron conversion and Zn deposition for high-capacity aqueous Zn–I2 batteries
Huiquan
Zhang
a,
Xueying
Zhang
a,
Dongmin
Ma
a,
Xinxin
Cai
a,
Mochi
Lv
a,
Hongting
Yan
a,
Junbo
Niu
b and
Weixing
Song
 *a
*a
aDepartment of Chemistry, Capital Normal University, Beijing 100048, P. R. China. E-mail: songwx@cnu.edu.cn
bSchool of Materials Science and Engineering, Nanyang Technological University, 50 Nanyang Avenue, Singapore 639798, Singapore
First published on 5th February 2026
Abstract
Aqueous zinc–iodine batteries (ZIBs) are a promising energy storage technology due to the abundance of iodine, environmental friendliness, and low cost. This study introduces a multifunctional additive, L-lysine hydrochloride (LLH), designed to activate the four-electron transfer chemistry between I+ and I− species, significantly boosting energy density. LLH stabilizes I+via dual coordination from the amino groups and chloride ion, effectively suppressing hydrolysis and enabling reversible 2I−/I02/2I+ conversion. The preferential adsorption of the carboxyl group of protonated L-lysine at the zinc anode promotes uniform zinc deposition while inhibiting the hydrogen evolution reaction. Additionally, the incorporation of LLH effectively suppresses the shuttle effect by interacting with iodine species through its carboxyl and amino groups. LLH-modified Zn‖Zn symmetric batteries demonstrate extended cycling stability, operating beyond 4000 hours, while Zn‖I2 full batteries deliver a high specific capacity of 502 mAh g−1 at 1 A g−1. This additive strategy renders a facile and efficient approach to realizing high-capacity and durable ZIBs.
Introduction
Aqueous zinc–iodine batteries (ZIBs) have garnered significant attention due to their higher theoretical specific capacities and abundant resource potential compared to conventional intercalation-type electrodes.1–8 The redox reaction based on the 2I−/I02 couple is commonly employed at the cathode, offering a theoretical capacity of 211 mAh g−1. However, its energy density and output voltage in conventional two-electron aqueous zinc–iodine batteries (2eZIBs) remain suboptimal. The 2I−/I02 conversion generates water-soluble polyiodide intermediates, inducing a shuttle effect that causes irreversible active material loss and reduces I2 utilization.9–12 ZIBs also face limitations from conventional aqueous electrolytes, including the hydrogen evolution reaction (HER), corrosion, passivation at the Zn anode/electrolyte interface, and dendritic Zn growth due to inhomogeneous electric field distribution.13–18 Recently, four-electron aqueous zinc–iodine batteries (4eZIBs) based on sequential 2I−/I02/2I+ redox pairs have attracted extensive research interest, delivering enhanced specific capacities (422 mAh g−1) and higher operating voltage platforms to achieve superior energy density.19–21 Nevertheless, thermodynamic instability of I+ and unstable Zn plating/stripping in conventional electrolytes pose significant challenges. Issues such as active iodine dissolution and polyiodide shuttling further hinder the practical implementation of 4eZIBs.22Recent studies have reported some strategies to activate high-valent iodine redox reactions for high-theoretical-capacity aqueous Zn‖I2 batteries. These studies typically utilize relatively high concentrations of halide ions to form halides on the cathode side (such as introducing Cl− to combine and form ICl).22,23 A high-concentration electrolyte containing 19 M LiCl, 19 M ZnCl2, and 8 M acetonitrile was developed to suppress I+ hydrolysis while supplying abundant free Cl− for facilitating 2I−/I02 conversion.24 However, high Cl− concentrations corrode battery components, while insufficient Cl− fails to fully stabilize I+, leading to hydrolysis and limited capacity. A ternary hydrated eutectic electrolyte comprising dimethyl sulfone, nicotinamide, and Zn(ClO4)2·6H2O was used to stabilize electrophilic I+, which extends the electrochemical window.25 But high-concentration salts or eutectic electrolytes undermine the cost-effectiveness of aqueous systems and reduce their inherent ionic conductivity.
As an electrolyte additive distinct from conventional amino acid-based ones, L-lysine hydrochloride (LLH) is herein utilized to both stabilize the zinc anode and boost cycling performance, as well as anchor I+ species to realize reversible 2I−/I02/2I+ redox conversion. Compared to an electrolyte modified with HCl at an equivalent additive concentration, LLH exhibits stronger anchoring capability towards I+ species, which can effectively prevent its hydrolysis and enhance the cycling performance of Zn‖I2 batteries. Experimental analyses and density functional theory (DFT) calculations reveal that LLH anchors I+via the synergistic effect of the nucleophilic amino groups and chloride ion, suppressing hydrolysis, thermodynamically favoring iodine valence transitions, enhancing redox kinetics and reversibility, and mitigating the shuttle effect. Preferential adsorption of protonated L-lysine (LL+) on the Zn anode modulates the inner Helmholtz plane (IHP), extends the stable electrochemical window, and achieves highly reversible Zn plating/stripping with stable cycling over 4000 h. A typical Zn‖I2 battery with the LLH additive demonstrates an increased specific capacity of 502 mAh g−1 at 1 A g−1 and an average coulombic efficiency (CE) of 99.4% at 5 A g−1.
Results and discussion
Electrochemical performance of the LLH-optimized electrolyte for the 2I−/I02/2I+ reaction
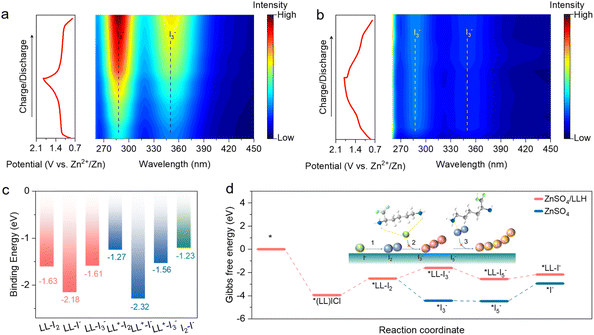
LLH is employed as an additive in 2 M ZnSO4 electrolyte (Fig. 1a) where the amino groups and chloride ion of LLH coordinate with I+ species to stabilize iodine intermediates at the cathode interface, thereby enabling stable four-electron transfer and an attendant enhancement in energy density. Simultaneously, LLH can coordinate with various iodine species, thereby suppressing polyiodide formation. Furthermore, preferential adsorption at the anode interface reduces reactive water population in the IHP, effectively suppressing the HER and passivation processes. The interfacial regulation further promotes homogeneous zinc deposition and inhibits dendritic growth. The optimal LLH concentration of 500 mM in ZnSO4 electrolyte (ZnSO4/LLH) is determined through anode electrochemical screening (Fig. S1). The performance degradation with shortened lifespan of the battery with more than 500 mM LLH in the ZnSO4 electrolyte is likely caused by increased Cl− concentration; therefore, 500 mM LLH was selected for further research.Distinct charge/discharge plateaus corresponding to I02/2I+ conversion are observed in ZnSO4/LLH electrolytes (Fig. 1b). Notably, a discharge capacity of 495 mAh g−1 is achieved at 2 A g−1. The specific capacity of the Zn‖I2 batteries exceeds the theoretical value of 422 mAh g−1 due to capacitive contributions from the activated carbon substrate in the cathode (Fig. S2). The observed capacity significantly exceeds that of the ZnSO4 electrolyte, which can be attributed to the four-electron redox conversion (2I−/I02/2I+). This result demonstrates the effectiveness of LLH-mediated suppression of I+ hydrolysis. Compared to the HCl-modified electrolyte at an equivalent concentration, LLH delivers higher capacity, demonstrating the superior I+ anchoring capability of LLH relative to HCl. Cyclic voltammetry (CV) curves exhibit two redox peak pairs (1.77/1.62 V and 1.27/1.14 V) for four-electron transfer reactions. These features align with galvanostatic charge/discharge (GCD) curve profiles and are assigned to I02/2I+ and 2I−/I02 conversions (Fig. S3).26 The I02/2I+ reduction peak in ZnSO4/LLH shows higher current density and a larger area than those in ZnSO4/HCl. Moreover, smaller redox potential gaps in ZnSO4/LLH indicate improved reversibility compared to other electrolytes. Lower Tafel slopes for I02/2I+ and 2I−/I02 conversions in ZnSO4/LLH confirm accelerated reaction kinetics (Fig. S4).9 A significant reduction in the Tafel slope of the R2 peak suggests highly reducibility and reversibility of the anchored I+ species.
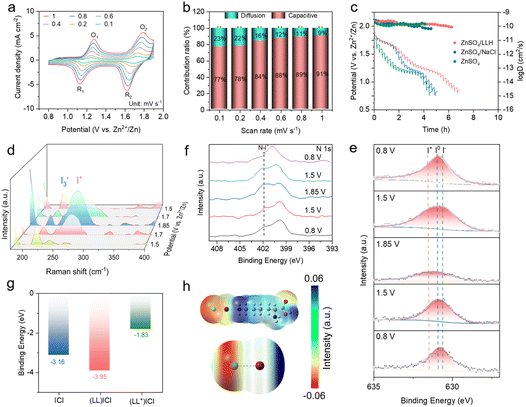
To further investigate the electrochemical reaction kinetics in the ZnSO4/LLH electrolyte, we conducted CV measurements at various scan rates (Fig. 2a). The contributions of surface-controlled capacitances can be qualitatively analyzed based on the relationship between the measured current (i) and scan rate (v) from the CV curves: i = avb. When b is close to 1.0, the electrochemical behavior is dominated by a surface capacitive process, whereas when b approaches 0.5, a diffusion-controlled process is observed. Through dynamic analysis of the log![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) i–log
i–log![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) v plot (peak current (ip) vs. scan rate (v)), it is found that the conversion process is mainly a redox reaction controlled by diffusion and capacitance. The fitted b values of two pairs of cathode and anode peaks are 0.93, 0.86, 0.90 and 0.99, respectively (Fig. S5). It indicates hybrid diffusion-capacitive control with rapid charge-carrier diffusion, thus enabling superior rate capability.27 The contributions of surface-controlled capacitances were quantified using i = k1v + k2v1/2, where k1v and k2v1/2 represent capacitive and diffusion-controlled components. The results show that at scan rates of 0.1, 0.2, 0.4, 0.6, 0.8, and 1.0 mV s−1, the surface-controlled capacitance contribution rates are 77%, 78%, 84%, 88%, 89%, and 91%, respectively. Compared with the HCl-modified electrolyte at an equivalent concentration, ZnSO4/LLH exhibits higher capacitance contribution (Fig. 2b and S6), indicating its improved rate performance and fast reaction kinetics of the 2I−/I02/2I+ conversion process. Galvanostatic intermittent titration technique (GITT) measurements revealed high ion diffusion coefficients (approximately 10−9 to 10−12 cm2 s−1) in the LLH-modified electrolyte (Fig. 2c).26,28 Diffusion coefficients during discharge are typically lower than those during charge, indicating that the I02 to 2I− conversion proceeds faster than the 2I+ to I02 conversion. This kinetic behavior is consistent with cyclic voltammetry (CV) analysis.
v plot (peak current (ip) vs. scan rate (v)), it is found that the conversion process is mainly a redox reaction controlled by diffusion and capacitance. The fitted b values of two pairs of cathode and anode peaks are 0.93, 0.86, 0.90 and 0.99, respectively (Fig. S5). It indicates hybrid diffusion-capacitive control with rapid charge-carrier diffusion, thus enabling superior rate capability.27 The contributions of surface-controlled capacitances were quantified using i = k1v + k2v1/2, where k1v and k2v1/2 represent capacitive and diffusion-controlled components. The results show that at scan rates of 0.1, 0.2, 0.4, 0.6, 0.8, and 1.0 mV s−1, the surface-controlled capacitance contribution rates are 77%, 78%, 84%, 88%, 89%, and 91%, respectively. Compared with the HCl-modified electrolyte at an equivalent concentration, ZnSO4/LLH exhibits higher capacitance contribution (Fig. 2b and S6), indicating its improved rate performance and fast reaction kinetics of the 2I−/I02/2I+ conversion process. Galvanostatic intermittent titration technique (GITT) measurements revealed high ion diffusion coefficients (approximately 10−9 to 10−12 cm2 s−1) in the LLH-modified electrolyte (Fig. 2c).26,28 Diffusion coefficients during discharge are typically lower than those during charge, indicating that the I02 to 2I− conversion proceeds faster than the 2I+ to I02 conversion. This kinetic behavior is consistent with cyclic voltammetry (CV) analysis.
 | ||
| Fig. 2 Four-electron 2I−/I02/2I+ conversion mechanism of Zn‖I2 batteries in ZnSO4/LLH electrolytes. (a) CV curves at different scan rates. (b) Contribution ratios of surface-controlled and diffusion-controlled capacities derived from fitting analysis at different scan rates. (c) GITT profiles and the corresponding diffusion coefficients of Zn‖I2 full batteries in different electrolytes.26 (d) In situ Raman spectroscopy of electrochemical processes. (e) Ex situ XPS spectra of I 3d for the cathode at different potentials during charge/discharge. (f) High-resolution N 1s spectra of the I2 cathode at different voltages. (g) The different coordination forms of fixed I+ and the corresponding binding energies. (h) Electrostatic potential of (LL)ICl (top) and ICl (bottom) (green spheres: Cl−, purple spheres: I+). | ||
Subsequently, in situ Raman spectroscopy was employed to investigate the four-electron (2I−/I02/2I+) conversion mechanism. A peak at 216 cm−1, assigned to I3−, weakens upon charging to 1.7 V, while a peak at 265 cm−1, corresponding to I+, intensified with increasing voltage (Fig. 2d). These observations confirm successful I+ anchoring. During discharge, the I+ peak diminishes as the voltage decreased, indicating reversible 2I−/I02/2I+ redox transitions. The X-ray photoelectron spectroscopy (XPS) analysis of cathodes at varying potentials reveals a gradual increase in binding energy during charging and a decrease during discharging (Fig. 2e and S7). This aligns with a valence state evolution during charging (2I− → I02 → 2I+) and the reverse during discharging (2I+ → I02 → 2I−), demonstrating the reversibility of 2I−/I02/2I+ redox reactions and effective I+ stabilization. Subsequently, further analysis of the 1s orbital of N reveals the appearance of an additional peak shape after charging to 1.85 V, which gradually disappears during discharge. This is speculated to be due to the coordination between N and I+ of LLH during charging, which also serves as an anchoring mechanism (Fig. 2f).29 DFT calculation results reveal that the (LL)ICl coordination mode possesses the highest binding energy, indicating the superior stability of this bidentate structure of ICl (Fig. 2h and S8). This configuration anchors I+ more effectively against hydrolysis compared to monodentate ICl coordination. Experimental observations correlate with these findings, where HCl-modified full batteries underperform LLH-based counterparts. Monodentate coordination enables four-electron reactions but provides inferior I+ anchoring capability relative to the (LL)ICl bidentate structure. Partial protonation occurs in the electrolyte, generating protonated L-lysine (LL+) species. Electrostatic interactions between LL+ and ICl2− constitute an alternative I+ anchoring mechanism. Calculated binding energies for this configuration remain comparatively low, suggesting limited stability. Electrostatic potential distributions demonstrate simultaneous I+ anchoring by N and Cl atoms (Fig. 2g). The (LL)ICl bidentate coordination thus establishes a stable I+ interaction mechanism, achieving enhanced theoretical specific capacity.
Moreover, in situ UV-vis spectroscopy further reveals two characteristic I3− peaks at 287 nm and 350 nm in ZnSO4 electrolytes upon charging to 1.2 V.30 The peak intensities increase with voltage (Fig. 3a), indicating continuous formation, accumulation, and diffusion of I3− from the cathode during cycling. Conversely, these peaks are significantly suppressed in ZnSO4/LLH, becoming nearly undetectable under identical conditions (Fig. 3b). Minimal intensity increase above 1.6 V demonstrates that LLH enhances reaction kinetics and suppresses polyiodide shuttling.31 Theoretical calculations elucidated interactions between the LL/LL+ and iodine species (I−, I2, I3−). Computational results reveal substantially reduced binding energy between both LL/LL+ and I−, indicating the preferential adsorption of I− towards LL+ among iodine species (Fig. 3c and S9). This I−–LL+ interaction (physical adsorption) during discharge suppresses the bonding between newly formed I− and residual I2, thereby inhibiting I3− formation at the cathode. Gibbs free energy profiles for iodine reduction reactions were computed, where the energy decrease (−3.95 eV) for the I+ → (LL)ICl transition demonstrates strong spontaneity and structural stability (Fig. 3d).25,32 Notably, LLH addition increased ΔG values for I2 → I3− conversion, indicating non-spontaneous reaction characteristics. Concurrently, reduced ΔG values for I5− → I− were observed relative to the ZnSO4 electrolyte, suggesting facilitated polyiodide conversion to I−. These results validate LLH's dual functionality: effective I+ anchoring and suppression of the shuttle effect.
Protection of the zinc anode by the LLH-optimized electrolyte
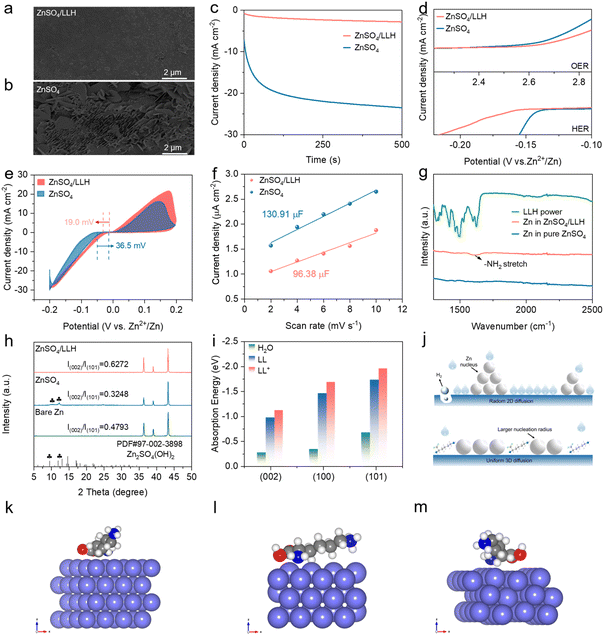
A comprehensive investigation combining DFT calculations and electrochemical characterization was conducted to assess the interfacial stabilization effects of LLH on zinc anodes. Zn‖Zn symmetric battery configurations with controlled LLH concentration gradients were employed for electrochemical analysis. The symmetric battery with 500 mM LLH in the electrolyte exhibits an enhanced cycling stability of 900 h at 5 mA cm−2, 2.5 mAh cm−2. The symmetric battery with ZnSO4/LLH electrolyte promotes smooth, uniform Zn deposits, indicating preferential (002)-oriented deposition, and suppresses corrosion/side reactions (Fig. 4a and S10). In contrast, the scanning electron microscopy (SEM) images of Zn anodes cycled for 50 h at 1 mA cm−2 and 1 mAh cm−2 reveal rough surfaces with dendrites, dead Zn fragments, and irregular by-products in ZnSO4 (Fig. 4b and S10). These features reduce the active surface area, degrade cycling stability and provide more reaction sites for further corrosion and side reactions. Chronoamperometry at −150 mV overpotential reveals a current density of ∼23.5 mA cm−2 after 500 s in ZnSO4, corresponding to 2D diffusion-driven Zn2+ aggregation and dendrite growth. In ZnSO4/LLH, the Zn2+ kinetics exhibits rapid transition to 3D diffusion with reduced current density (Fig. 4c). Linear sweep voltammetry (LSV) was subsequently employed to evaluate the electrochemical stability window, revealing that LLH incorporation suppresses the HER and oxygen evolution reaction (OER), while broadening the electrochemical stability window from −0.15 to 2.5 (V vs. Zn2+/Zn) (Fig. 4d, S11 and S12). This enhancement improves electrolyte stability during deposition/stripping cycles. In addition, CV tests on Zn‖Ti batteries were conducted to evaluate the nucleation potential of Zn. The nucleation overpotential in ZnSO4/LLH electrolyte (19.0 mV) is lower than that in ZnSO4/LLH electrolyte (36.5 mV) (Fig. 4e). These results demonstrate that LLH mitigates parasitic reactions (e.g., HER and passivation) and enables homogeneous Zn2+ deposition, effectively suppressing dendrite formation.IHP reconstruction consequently emerges as a principal strategy for optimizing zinc anode reversibility through interfacial engineering. Electrical double layer (EDL) capacitance measurements reveal a lower value for ZnSO4/LLH (96.38 µF cm−2) compared to ZnSO4 (130.91 µF cm−2), indicating LL adsorption at the Zn surface and IHP restructuring (Fig. 4f and S13).33 Molecular orbital analysis shows the highest occupied molecular orbital (HOMO) of LL+ (−0.351 eV) and LL (−6.544 eV) is higher than that of H2O (−8.701 eV), and the lowest unoccupied molecular orbital (LUMO) of LL+ (−0.205 eV) and LL (−0.473 eV) is lower, favoring electron transfer to zinc surfaces, thereby promoting LL+ adsorption (Fig. S14). This reduces the H2O molecule population density within the IHP, inducing IHP structural reconfiguration. Contact angle measurements also demonstrate improved wettability in ZnSO4/LLH (from 84° to 92°, Fig. S15), indicating enhanced zincophilic properties for uniform ion transport. LL+ alignment in the IHP suppresses H2O mobility/activity, mitigates parasitic reactions, and directs Zn2+ deposition via electronegative functional groups.34
The FTIR analysis of Zn foils soaked in ZnSO4/LLH reveals a bending vibration peak assigned to amino groups at 1600 cm−1, while the Zn foil immersed in ZnSO4 did not exhibit this peak, confirming LL+ surface adsorption (Fig. 4g). In addition, comparative energy-dispersive spectroscopy (EDS) mapping of zinc foils immersed in both electrolytes for 15 days reveals the homogeneous distribution of C, N, and O elements on ZnSO4-LLH immersed foils, confirming uniform LL+ adsorption (Fig. S16 and S17). In contrast, EDS mapping reveals spatially overlapping S, O, and Zn signals on zinc surfaces in pure ZnSO4 electrolyte. These observations, combined with XRD analysis after 15 day immersion, confirm the formation of the Zn4SO4(OH)6·xH2O byproduct (Fig. S18). LL+ incorporation attenuates H2O reactivity and reduced SO42− population within IHP, thereby preserving zinc anode integrity.
The XPS results provide additional evidence for the interfacial interaction between LL+ and the zinc metal surface. As shown in Fig. S19, the post-immersion zinc substrate exhibits distinct characteristic peaks in the high-resolution C 1s spectrum, corresponding to O–C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O (531.2 eV) and C–N/C–C (284.8 eV) functional groups derived from LL+. Concurrently, the O 1s spectrum reveals prominent peaks associated with O–H (532.1 eV) and O–C
O (531.2 eV) and C–N/C–C (284.8 eV) functional groups derived from LL+. Concurrently, the O 1s spectrum reveals prominent peaks associated with O–H (532.1 eV) and O–C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O (533.5 eV) species. These spectral features are absent in the bare zinc control samples. The O–C
O (533.5 eV) species. These spectral features are absent in the bare zinc control samples. The O–C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O peak exhibits a binding energy shift to 532.1 eV, indicative of carboxyl group adsorption on the Zn surface which reduces local electron density. XPS analysis reveals interfacial N–Zn bonding (399.4 eV) alongside LL+'s native N–C peak (398.2 eV), demonstrating nitrogen-mediated chemisorption on zinc.
O peak exhibits a binding energy shift to 532.1 eV, indicative of carboxyl group adsorption on the Zn surface which reduces local electron density. XPS analysis reveals interfacial N–Zn bonding (399.4 eV) alongside LL+'s native N–C peak (398.2 eV), demonstrating nitrogen-mediated chemisorption on zinc.
XRD analysis of Zn anodes was further carried out to elucidate the stabilizing effect of LLH on zinc anode morphology. Comparative XRD patterns of cycled Zn anodes of Zn‖Zn symmetric batteries in different electrolytes reveal increased I(002)/I(100) and I(002)/I(101) ratios in the ZnSO4/LLH electrolyte, demonstrating LLH-induced preferential (002)-oriented Zn deposition (Fig. 4h). DFT calculations were subsequently performed for verification, determining adsorption energies of H2O, LL, and LL+ on distinct zinc crystal facets. Both LL and LL+ exhibited consistently more negative adsorption energies than H2O on the (002), (100) and (101) planes, indicating stronger interactions with zinc metal anodes compared to water molecules, particularly for LL+ (Fig. 4i, S20 and S21). Competitive adsorption displaces H2O from zinc anode surfaces by LL+ molecules, forming a hydration-depleted IHP. LL+ cations are preferentially adsorbed onto the zinc surface, covering the high-charge-density active sites (e.g., tips and defects) that are susceptible to rapid Zn2+ deposition and subsequent dendrite formation. This preferential adsorption redirects Zn2+ deposition toward regions with sparser LL+ coverage and a more uniform electric field, thereby effectively suppressing zinc dendrite growth. This configuration suppresses parasitic reactions, while preferential adsorption hinders the lateral diffusion of Zn2+, thereby restricting dendritic formation (Fig. 4j). The nuclear magnetic resonance spectra reveal that the 2H peak of pure D2O at 4.69 ppm shifts to 4.721 ppm after the addition of 2 M ZnSO4, confirming strong Zn2+–D2O coordination. No significant peak shifts are observed with LLH addition, indicating LLH does not engage in solvation (Fig. S22).35 FTIR spectroscopy corroborates these findings, showing no notable shifts in SO42− vibrational bands, further confirming the absence of LLH-solvation interactions (Fig. S23).
Improved cycling performance with the LLH-optimized electrolyte
The pH stability of the electrolyte in ZIBs plays a critical role in suppressing zinc dendrite formation, mitigating the HER and enhancing cycling lifespan. Real-time monitoring of interfacial pH variations at the working electrode during charge/discharge was conducted using a custom-built in situ pH measurement system (Fig. S24). The addition of LLH results in attenuated pH fluctuations throughout the deposition/stripping cycles, with reduced pH variations observed before and after potential transitions at zinc foil interfaces. A substantial pH elevation occurs in ZnSO4 electrolyte during zinc deposition due to the vigorous HER. Subsequent pH fluctuations stem from dynamic changes in local H+ concentration at reaction sites. The initial pH rise during zinc stripping originates from the dissolution of the passivation layer formed during deposition, followed by gradual pH decrease induced by Zn2+ hydrolysis. The ZnSO4/LLH electrolyte exhibits considerably attenuated pH variations throughout deposition/stripping cycles, demonstrating pH buffering capacity and HER suppression (Fig. 5a). This mechanism arises from lysine adsorption reducing the population of active water molecules at the IHP, thereby suppressing the HER. As an amphoteric amino acid, LLH contains α-carboxyl, α-amino, and ε-amino side chain groups. In weakly acidic electrolytes, the carboxyl groups undergo partial deprotonation to release H+ while amino groups coordinate protons. Elevated pH triggers the deprotonation of –COOH and protonated amino groups to replenish H+ concentration. Under acidic conditions, –COO− groups and amino functionalities sequester H+ through protonation, establishing pH buffering capacity (Fig. S26). LLH can buffer the pH fluctuations induced by electrode reactions during charge/discharge cycles, thereby mitigating their impact on the bulk electrolyte pH. This effect reduces the occurrence of the HER and suppresses the formation of byproducts such as Zn4SO4(OH)6·xH2O.Linear polarization measurements show a positive shift in corrosion potential for ZnSO4/LLH (−0.966 V vs. Ag/AgCl) compared to pure ZnSO4 (−0.972 V vs. Ag/AgCl), indicating enhanced thermodynamic stability. The corrosion current density decreases from 13.18 mA cm−2 to 8.13 mA cm−2, confirming improved corrosion resistance. These findings suggest that LLH-mediated modification of the IHP effectively mitigates zinc anode corrosion (Fig. 5b).36
A comprehensive series of electrochemical evaluations was conducted to assess the synergistic effects of LL+ at the IHP on zinc anode reversibility and stability. Zn‖Zn symmetric batteries were subjected to varied current densities and areal capacities as primary evaluation parameters. As shown in Fig. 5c, batteries with the ZnSO4 electrolyte exhibit irreversible voltage drops after 550 hours under 3 mA cm−2 and 1 mAh cm−2, attributed to separator penetration by dendrite growth causing electrical shorting. In contrast, ZnSO4/LLH electrolyte-based batteries demonstrate extended cycling stability exceeding 4000 hours. This behavior originates from LL+ adsorption at the interface, which facilitates Zn2+ transport kinetics and promotes uniform zinc deposition. ZnSO4/LLH electrolyte batteries exhibited stable cycling for over 2850 hours at 5 mA cm−2 and 1 mAh cm−2, as well as 2250 hours at 1 mA cm−2 under identical capacity conditions (Fig. S27 and S28). Comparative analysis across current densities reveals enhanced cycling stability relative to pure ZnSO4 electrolyte systems.
As shown in Fig. 5d, Zn‖Zn symmetric batteries with pure ZnSO4 electrolyte exhibit unstable potential hysteresis at 50 mA cm−2 and undergo rapid failure after 130 hours. This behavior originates from accelerated Zn2+ migration under high current density. However, sluggish deposition kinetics cause Zn2+ accumulation at the interface, impeding subsequent ion transport and inducing severe concentration polarization. Simultaneously, solvated water molecules associated with accumulated Zn2+ promote parasitic reactions, further exacerbating electrolyte/electrode interfacial instability. The ZnSO4/LLH electrolyte demonstrates extended cycling stability exceeding 650 hours, attributed to accelerated reaction kinetics and ordered zinc deposition morphology. Reversibility analysis via symmetric batteries reveals superior rate capability and high reversibility for ZnSO4/LLH-based Zn‖Zn batteries across current densities from 1 to 50 mA cm−2 (Fig. 5e). This demonstrates enhanced compatibility of LLH-modified zinc-ion batteries with commercial fast-charging requirements of high-power charge/discharge capability.
CE of Zn‖Ti batteries in ZnSO4 electrolyte deteriorates rapidly beyond 113 plating/stripping cycles under 5 mA cm−2 and 2.5 mAh cm−2 conditions during reversibility testing (Fig. 5f). Degradation correlates with dendritic-induced micro-shorts. The ZnSO4/LLH electrolyte-based batteries demonstrate superior reversibility with stable cycling exceeding 350 cycles and an average CE of 99.2%. This improvement stems from LL+-induced homogeneous Zn2+ distribution and uniform zinc deposition. Fig. S29 and S30 illustrate stable cycling performance exceeding 800 cycles at 1 mAh cm−2 under 2 mA cm−2 and 1000 cycles at 1 mAh cm−2 under 5 mA cm−2, with average CE values of 98.8% and 99.4%, respectively. These performance metrics substantiate LL+'s critical role in establishing interfacial conditions enabling long-term zinc metal anode stabilization through IHP-mediated homogeneous deposition.
Performance of Zn‖I2 full batteries
Full-battery evaluations were performed on four-electron transfer Zn‖I2 batteries employing ZnSO4/LLH electrolyte. Rate capability across 1–15 A g−1 demonstrates specific capacities of 502, 472, 410, 362, 329, and 268 mAh g−1 for ZnSO4/LLH-based batteries (Fig. 6a and S2). A maximum net specific capacity of 409 mAh g−1 is achieved. The capacity retention is significantly enhanced when the current density is restored to 1 A g−1, delivering a specific capacity of 499 mAh g−1. Galvanostatic charge–discharge profiles (Fig. 6b) exhibit two distinct voltage plateaus at various current densities, attributed to effective I+ anchoring within the electrolyte. These values represent substantial capacity enhancement over pure ZnSO4 systems, highlighting the efficacy of 2I−/I02/2I+ four-electron redox chemistry. Comparative analysis with ZnSO4/HCl electrolyte systems reveals superior specific capacity at equivalent concentrations, indicating the enhanced stability of I+ and energy density provision by LLH versus HCl.Pure ZnSO4-based Zn‖I2 batteries exhibit capacity decay to 122.4 mAh g−1 after 1900 cycles at 2 A g−1, caused by separator piercing due to anode dendrite formation. HCl-modified batteries display rapid capacity fade (53.64% retention) after 1200 cycles with 98.94% average CE. This degradation is attributed to continuous cathode active material loss via iodine species shuttling, combined with I3−-mediated oxidation of zinc metal causing anode corrosion, which substantially reduces both specific capacity and CE. Furthermore, corrosion-induced anode surface heterogeneity disrupted subsequent uniform zinc deposition, while non-uniform electric field distribution triggered the HER and passivation. Additionally, competitive chlorine evolution reactions progressively depleted Cl− anions during cycling, ultimately compromising I+ stabilization capability, resulting in a 50% reduction in specific capacity. LLH-incorporating batteries exhibited 86.48% capacity retention (435.5 mAh g−1) with an average CE of 99.4% after 2300 cycles (Fig. 6c). GCD curves for LLH-based systems (Fig. S31) exhibit stable 2I+/I02 plateaus. The Zn‖I2 cell employing ZnSO4/LLH electrolyte achieved 6000 cycles at a high current density of 5 A g−1, retaining a discharge capacity of 323.9 mAh g−1 with a coulombic efficiency of 99.4%, which indicates dual stabilization of both the cathode and the zinc anode (Fig. 6d). The substantially enhanced cycling stability originates from LLH's superior I+ anchoring capability, optimized iodine conversion kinetics, and dual suppression of zinc dendrites and parasitic reactions. Practical viability was demonstrated through 4 × 4 cm2 pouch batteries employing ZnSO4/LLH electrolyte, which sustains 350 stable cycles at 0.5 A g−1 (Fig. 6e). Comparative analysis with prior Zn‖I2 systems demonstrates the superior performance of LLH-modified Zn‖I2 batteries in specific capacity and cycling stability among other parameters (Fig. 6f).23,25,27,29,37–42 Pouch batteries were subsequently evaluated under various operational conditions, where a single 4 × 4 cm2 battery powered a miniature fan and retained functionality after 180° bending deformation and mechanical restoration (Fig. S32). A series-connected array of three identical pouch batteries successfully powered a mobile device, as shown in Fig. 6g.
Conclusions
In summary, incorporating cost-effective LLH as an additive effectively enhances the performance of aqueous ZIBs. Spectroscopic characterization and theoretical simulations reveal LLH stabilizes intermediate I+ species. This enables a highly reversible four-electron redox conversion (2I−/I02/2I+), which enhances the specific capacity by 89%. Moreover, the functional groups of LLH suppress the iodine shuttle effect by interacting with I2 and I−. In addition, LL+ adsorption on the zinc anode surface restructures the IHP, promoting homogeneous zinc deposition and mitigating side reactions such as corrosion and passivation. This improves deposition reversibility, extending the anode's cycle life beyond 4000 hours. The synergistic modification of both electrodes results in outstanding overall performance: a high specific capacity of 502 mAh g−1 at 1 A g−1. Additionally, a practical 4 × 4 cm2 pouch cell demonstrates stable operation for over 350 cycles at 0.5 A g−1.Author contributions
H. Zhang designed and conducted the experiments, performed the data analysis, and drafted the manuscript; X. Zhang assisted with data discussion and analysis; D. Ma, X. Cai, M. Lv, and H. Yan participated in the data discussion and analysis; Junbo Niu participated in the theoretical calculation work; W. Song conceived and supervised the research, and contributed to methodology development, manuscript revision, and funding acquisition.Conflicts of interest
There are no conflicts to declare.Data availability
The data supporting this article have been included as part of the supplementary information (SI). Supplementary information: materials and experimental details, characterisation data, and electrochemical performance data (Fig. S1–S32 and, Tables S1 and S2). See DOI: https://doi.org/10.1039/d6sc00182c.Acknowledgements
This work was financially supported by the National Natural Science Foundation of China (No. 22172103).References
- Y. Li, M. Zhang, H. Lu, X. Cai, Z. Jiao, S. Li and W. Song, Boosting High-Performance Aqueous Zinc-Ion Hybrid Capacitors via Organic Redox Species on Laser-Induced Graphene Network, Adv. Funct. Mater., 2024, 34, 2400663 Search PubMed.
- Y. Li, H. Ba, Z. Wang, S. Wu, Y. Shang, S. Huang and H. Y. Yang, Electrolyte pH and operating potential: critical factors regulating the anodic oxidation and zinc storage mechanisms in aqueous zinc ion battery, Mater. Today Energy, 2024, 39, 101460 Search PubMed.
- J. Tao, X. Cai, Y. Li, L. Huang, X. Zhang, H. Zhang, D. Ma, L. Ran and W. Song, A Synergistic Zincophilic and Hydrophobic Supramolecule Shielding Layer for Actualizing Long-Term Zinc-Ion Batteries, Adv. Energy Mater., 2025, 15, 2403662 Search PubMed.
- T. Xiao, J.-L. Yang, D. Chao and H. J. Fan, Multimodal electrolyte architecting for static aqueous zinc-halogen batteries, Natl. Sci. Rev., 2025, 12, nwaf029 Search PubMed.
- F. Bu, Z. Sun, W. Zhou, Y. Zhang, Y. Chen, B. Ma, X. Liu, P. Liang, C. Zhong, R. Zhao, H. Li, L. Wang, T. Zhang, B. Wang, Z. Zhao, J. Zhang, W. Li, Y. S. Ibrahim, Y. Hassan, A. Elzatahry, D. Chao and D. Zhao, Reviving Zn0 Dendrites to Electroactive Zn2+ by Mesoporous MXene with Active Edge Sites, J. Am. Chem. Soc., 2023, 145, 24284–24293 Search PubMed.
- Y. Su, B. Chen, Y. Sun, Z. Xue, Y. Zou, D. Yang, L. Sun, X. Yang, C. Li, Y. Yang, X. Song, W. Guo, S. Dou, D. Chao, Z. Liu and J. Sun, Rationalized Electroepitaxy toward Scalable Single-Crystal Zn Anodes, Adv. Mater., 2023, 35, 2301410 Search PubMed.
- R. Hao, S. Gu, Z. Wang, J. Chen, W. Luo, J. Hu, C. Yan, H. Yuan, G. Liu, K. Liu, C. Liu, W. Wang and Z. Lu, Reconstructing the solvation structure and solid-liquid interface enables dendrite-free zinc-ion batteries, Mater. Today Energy, 2023, 33, 101279 Search PubMed.
- G. Li, Q. Cai, S. Zhang, J. A. Yuwono, L. Mao, H. Jin and Z. Guo, Decoupled dual-salt electrolyte for practical aqueous zinc batteries, Nat. Sustain., 2025, 8, 1349–1359 CrossRef.
- X. Liang, Q. Dong, S. Guo, C. Zeng, Z. Chen, B. Zhang, C. Zhou, J. Zhang, Z. Xing, X. Tian and X. Shi, Customized Design of R-SO3H-Containing Binders for Durable Iodine-Loading Cathode of Zinc–Iodine Batteries, Adv. Energy Mater., 2025, 15, 2500673 Search PubMed.
- X. Cai, X. Wang, Z. Bie, Z. Jiao, Y. Li, W. Yan, H. J. Fan and W. Song, A Layer-by-Layer Self-Assembled Bio-Macromolecule Film for Stable Zinc Anode, Adv. Mater., 2024, 36, 2306734 CrossRef CAS PubMed.
- X. Fu, Y. Pan, Z. Chen, F. Li, Y. Yang, M. Chen, H. Tu, T. Qiu, Z. Xing, P. Rao, Z. Kang, W. Zhang, X. Shi, L. Shan and X. Tian, Amino acid-based functional additive enables fast polyiodide conversion kinetics for durable Zn-I2 batteries, Energy Environ. Sci., 2026 10.1039/D5EE06668A.
- X. Chen, D. Feng, Y. Jiao and P. Wu, Dual-Electrode Synergistic Electrolyte Enabling Highly Reversible Multi-Electron Redox in Aqueous Zinc-Iodine Batteries, Adv. Mater., 2025, e13389 Search PubMed.
- H. Ge, X. Feng, D. Liu and Y. Zhang, Recent advances and perspectives for Zn-based batteries: Zn anode and electrolyte, Nano Res. Energy, 2023, 2, e9120039 CrossRef.
- M. Zhu, X. Li, W. Ni, C. Cai, J. Zhong, M. Zhong, L. Huang, Y. Tian and J. Zhang, Manipulating oriented crystal growth of Zn via lattice-matched and zincophilic MXene nanoarrays towards dendrite-free Zn batteries, J. Colloid Interface Sci., 2025, 691, 137433 CrossRef CAS PubMed.
- J. Wu, J.-L. Yang, B. Zhang and H. J. Fan, Immobilizing Polyiodides with Expanded Zn2+ Channels for High-Rate Practical Zinc-Iodine Battery, Adv. Energy Mater., 2024, 14, 2302738 Search PubMed.
- C. Xu, C. Lei, P. Jiang, W. Yang, W. Ma, X. He and X. Liang, Practical high-energy aqueous zinc-bromine static batteries enabled by synergistic exclusion-complexation chemistry, Joule, 2024, 8, 461–481 Search PubMed.
- X. He, X.-Y. Kong and L. Wen, A promising solution for highly reversible zinc metal anode chemistry: Functional gradient interphase, Nano Res. Energy, 2024, 3, e9120100 CrossRef.
- P. Jiang, Q. Du, C. Lei, C. Xu, T. Liu, X. He and X. Liang, Stabilized four-electron aqueous zinc–iodine batteries by quaternary ammonium complexation, Chem. Sci., 2024, 15, 3357–3364 Search PubMed.
- H. Zhao, L. Zhao, D. Yin, N. Gao, Y. Zhang, J. Feng, X. Chen, X. Liu, J. Wei, C. Fan, G. Gao, C. Xiao and S. Ding, Key Issues and Strategies in Aqueous Static Zinc–Halogen Battery Design, Adv. Mater., 2025, e15759 CrossRef PubMed.
- Z. Shi, G. Liu, H. Huang, Z. He, C. Lei, Z. Feng, M. Ye, Y. Zhang, Z. Wen, W. Du, X. Liu, Y. Wei, Q. Yang, Y. Tang and C. C. Li, Coordination-escorted organo-interhalogen conversion enables durable dual-deposition Zn‖I2 batteries with high areal capacities, Energy Environ. Sci., 2026 10.1039/D5EE06160A.
- K. Chen, Y. Xu, H. Li, Y. Li, L. Zhang, Y. Guo, Q. Xu, Y. Li and H. Xie, Design of cellulosic poly(ionic liquid)s with a hydrogen bond/ion dual regulation mechanism for highly reversible Zn anodes, Chem. Sci., 2025, 16, 8648–8660 RSC.
- T. Liu, C. Lei, H. Wang, J. Li, P. Jiang, X. He and X. Liang, Aqueous Electrolyte With Weak Hydrogen Bonds for Four-Electron Zinc–Iodine Battery Operates in a Wide Temperature Range, Adv. Mater., 2024, 36, 2405473 Search PubMed.
- X. Li, Y. Wang, Z. Chen, P. Li, G. Liang, Z. Huang, Q. Yang, A. Chen, H. Cui, B. Dong, H. He and C. Zhi, Two-Electron Redox Chemistry Enabled High-Performance Iodide-Ion Conversion Battery, Angew. Chem., Int. Ed., 2022, 61, e202113576 CrossRef CAS PubMed.
- Y. Zou, T. Liu, Q. Du, Y. Li, H. Yi, X. Zhou, Z. Li, L. Gao, L. Zhang and X. Liang, A four-electron Zn-I2 aqueous battery enabled by reversible I−/I2/I+ conversion, Nat. Commun., 2021, 12, 170 CrossRef CAS PubMed.
- W. Li, H. Xu, H. Zhang, F. Wei, T. Zhang, Y. Wu, L. Huang, J. Fu, C. Jing, J. Cheng and S. Liu, Designing ternary hydrated eutectic electrolyte capable of four-electron conversion for advanced Zn–I2 full batteries, Energy Environ. Sci., 2023, 16, 4502–4510 Search PubMed.
- J. Hu, Z. Zhang, T. Deng, F. C. Cui, X. Shi, Y. Tian and G. Zhu, Porous Aromatic Frameworks Enabling Polyiodide Confinement toward High Capacity and Long Lifespan Zinc–Iodine Batteries, Adv. Mater., 2024, 36, 2401091 Search PubMed.
- X. Li, S. Wang, D. Zhang, P. Li, Z. Chen, A. Chen, Z. Huang, G. Liang, A. L. Rogach and C. Zhi, Perovskite Cathodes for Aqueous and Organic Iodine Batteries Operating Under One and Two Electrons Redox Modes, Adv. Mater., 2023, 36, 2304557 Search PubMed.
- Z. Lian, W. Yang, Z. Wu, L. Zhong, Z. Liu, Z. Chen, G. Lian, E. Iwuoha, K. Ocakoglu, J. Lu, S. Zhang, G. Zhou and X. Peng, A Diffusion-Retarded Strategy for Practical Zn–I2 Batteries Under Harsh Conditions, Angew. Chem., Int. Ed., 2026, 65, e15756 CrossRef CAS PubMed.
- M. Wang, Y. Meng, M. Sajid, Z. Xie, P. Tong, Z. Ma, K. Zhang, D. Shen, R. Luo, L. Song, L. Wu, X. Zheng, X. Li and W. Chen, Bidentate Coordination Structure Facilitates High-Voltage and High-Utilization Aqueous Zn–I2 Batteries, Angew. Chem., Int. Ed., 2024, 63, e202404784 Search PubMed.
- Y. Wang, X. Jin, J. Xiong, Q. Zhu, Q. Li, R. Wang, J. Li, Y. Fan, Y. Zhao and X. Sun, Ultra-stable Electrolytic Zn-I2 Batteries Based on Nanocarbon Wrapped by Highly Efficient Single-Atom Fe-NC Iodine Catalysts, Adv. Mater., 2024, 36, 2404093 Search PubMed.
- H. Chen, X. Li, K. Fang, H. Wang, J. Ning and Y. Hu, Aqueous Zinc-Iodine Batteries: From Electrochemistry to Energy Storage Mechanism, Adv. Energy Mater., 2023, 13, 2302187 Search PubMed.
- L. Ran, X. Cai, D. Ma, J. Zhao, H. Zhang, S. Liang, J. Tao, X. Zhang, Y. Li, H. J. Fan and W. Song, Stable Four-Electron Zinc-Iodine Battery Realized by Polyacrylamide as Catalytic Binder, Angew. Chem., Int. Ed., 2025, 64, e15918 Search PubMed.
- X. Ma, Q. Wang, X. Zhang, Y. Lin, F. Zhang, J. Huang and Y. Wang, Highly reversible zinc anode enabled by a trace-amount additive with pH buffering capability, Energy Environ. Sci., 2025, 18, 982–990 Search PubMed.
- R. Jiang, T. Naren, Y. Chen, Z. Chen, C. Zhang, Y. Xie, L. Chen, Y. Qi, Q. Meng, W. Wei and L. Zhou, Enhanced Hydrogen Bonding Through Strong Water-Locking Additives for Long-Term Cycling of Zinc Ion Batteries, Adv. Funct. Mater., 2024, 34, 2411477 Search PubMed.
- N. Hu, W. Lv, W. Chen, H. Tang, X. Zhang, H. Qin, D. Huang, J. Zhu, Z. Chen, J. Xu and H. He, A Double-Charged Organic Molecule Additive to Customize Electric Double Layer for Super-Stable and Deep-Rechargeable Zn Metal Pouch Batteries, Adv. Funct. Mater., 2024, 34, 2311773 Search PubMed.
- Y. Wu, M. Xie, K. Fu, Z. Li, H. Shi, J. Zhang, L. Wang, Y. Jiang, C. Liu, D. Ma, H. Huang, F. Zeng, Y. Liao, H. Liu, Y. Ren and X. Liang, Realizing Lean-Electrolyte Zinc-Ion Batteries via An Ultrathin and Cost-Effective Separator, Adv. Funct. Mater., 2025, e27567 Search PubMed.
- X. Li, M. Li, Z. Huang, G. Liang, Z. Chen, Q. Yang, Q. Huang and C. Zhi, Activating the I0/I+ redox couple in an aqueous I2–Zn battery to achieve a high voltage plateau, Energy Environ. Sci., 2021, 14, 407–413 RSC.
- F. Zhu, Z. Li, Z. Wang, Y. Fu and W. Guo, From Inorganic to Organic Iodine: Stabilization of I+ Enabling High-Energy Lithium–Iodine Battery, J. Am. Chem. Soc., 2024, 146, 11193–11201 CAS.
- T. Hu, Y. Zhao, Y. Yang, H. Lv, R. Zhong, F. Ding, F. Mo, H. Hu, C. Zhi and G. Liang, Development of Inverse-Opal-Structured Charge-Deficient Co9S8@nitrogen-Doped-Carbon to Catalytically Enable High Energy and High Power for the Two-Electron Transfer I+/I− Electrode, Adv. Mater., 2024, 36, 2312246 CrossRef CAS PubMed.
- C. Li, H. Li, X. Ren, L. Hu, J. Deng, J. Mo, X. Sun, G. Chen and X. Yu, Urea Chelation of I+ for High-Voltage Aqueous Zinc–Iodine Batteries, ACS Nano, 2025, 19, 2633–2640 CrossRef CAS PubMed.
- X. Kong, J. Zhang, X. Zhang, Z. Wang and D. Wang, Boosting Reversible Four-Electron Redox in Aqueous Zn-Iodine Batteries with Two Halogen Ionic Additives and a N, F Codoped Carbon Cathode, ACS Appl. Energy Mater., 2025, 8, 601–610 Search PubMed.
- Y. Xia, W. Li, H. Xu, F. Wei, S. Ke, H. Chen, H. Zhang, G. Guo, L. Ma, J. Wang and S. Liu, Tunable Mesoporous Porphyrin-Based Conjugated Polymer Capable of Boosting Four-Electron Zn-I2 Batteries, Adv. Funct. Mater., 2025, 35, 2417283 CrossRef CAS.
| This journal is © The Royal Society of Chemistry 2026 |