Open Access Article
Open Access ArticleVisible-light-driven valorization of 5-hydroxymethylfurfural over a hollow Co3O4@Zn3In2S6 nanocage in base-free water under an air atmosphere
Rong
Dang
a,
Kexin
Fan
a,
Yan-Ning
Wang
 b,
Guangsheng
Zhu
a,
Liu
Liu
c,
Zifu
Hu
a,
Taolian
Guo
b,
Guangsheng
Zhu
a,
Liu
Liu
c,
Zifu
Hu
a,
Taolian
Guo
 *a,
Wu
Chen
*a and
Fengxiang
Chen
*a,
Wu
Chen
*a and
Fengxiang
Chen
 *a
*a
aNational Local Joint Engineering Laboratory for Advanced Textile Processing and Clean Production, and State Key Laboratory of New Textile Materials and Advanced Processing, Wuhan Textile University, Wuhan 430200, China. E-mail: tlguo@wtu.edu.cn; wuchen@wtu.edu.cn; fxchen_czx@wtu.edu.cn
bCollege of Chemistry and Chemical Engineering, Xinyang Normal University, Xinyang, Henan 464000, China
cAnalytical and Testing Center, Wuhan Textile University, Wuhan 430200, China
First published on 23rd February 2026
Abstract
Visible-light-driven upgrading of biomass-derived 5-hydroxymethylfurfural (5-HMF) represents a green and sustainable strategy. However, photocatalytic selective oxidation of HMF to generate 2,5-diformylfuran (DFF), a highly promising biomass-derived molecule for pharmaceuticals and heterocyclic ligands, is extremely challenging under green and mild conditions. Herein, we construct a hollow Co3O4@Zn3In2S6 (ZC–x) nanocage photocatalyst, which exhibits a remarkable photocatalytic activity for the oxidation of HMF to DFF in base-free water under an air atmosphere. The optimal ZC-5 heterojunction achieves 95.4% HMF conversion and 94.1% DFF selectivity. The superior photocatalytic performance of the ZC-5 heterojunction is attributed to intimate interfacial contact between Co3O4 and Zn3In2S6, which is beneficial to generate strong interfacial interaction and facilitate the separation of electron–hole pairs. Characteristic analysis and density functional theory (DFT) calculations elucidate that the enhanced built-in electric field (IEF) and S-scheme charge transfer mechanism within the ZC heterojunction effectively drive the selective photo-oxidation of HMF to DFF. This work offers an efficient strategy to design heterojunction photocatalysts for highly effective oxidation of HMF under mild conditions.
Introduction
Photocatalytic upgrading of renewable biomass resources is a prospective avenue to produce high value-added chemicals and simultaneously relieve the energy crisis and environmental pollution.1–4 As an essential biomass-derived platform molecule, 5-hydroxymethylfurfural (HMF) can be converted into multiple value-added products due to its different functional groups including the alcohol hydroxyl group (–CH2OH) and aldehyde group (–CHO).5–8 In particular, 2,5-diformylfuran (DFF) generated by selective oxidation of HMF has been extensively utilized as a highly promising intermediate for synthesizing pharmaceuticals, resin and heterocyclic ligands.9,10 However, the highly selective oxidation of –CH2OH to produce DFF is extremely challenging during the photocatalytic HMF oxidation process, in which the appropriate oxidation capacity of photocatalysts is vital to avoid excessive oxidation.11–13 Therefore, it is crucial to design effective photocatalysts for highly selective oxidation of HMF to DFF.Transition metal sulfides, such as 2D layered Zn3In2S6, an n-type semiconductor, have attracted widespread attention for the selective oxidation of HMF to DFF due to its suitable band energy and outstanding visible-light-responsive capability.14 Based on the Zn3In2S6 semiconductor, many efforts (morphology engineering, heteroatom doping, defect engineering, and heterostructure construction) have been adopted to further improve its photocatalytic efficiency.15–19 Among them, the construction of the Zn3In2S6-based heterostructure has been proven to be an efficient strategy for facilitating charge carrier separation, regulating the photoelectric characteristics, and integrating the advantages of different components, eventually improving photocatalytic performance.20,21 Recently, the step scheme (S-scheme) heterojunction has emerged as an effective heterojunction system to achieve efficient electron–hole separation and retains their high redox capacities in semiconductor-based photocatalysts.22–25 Specifically, the S-scheme heterojunction based on a p–n heterojunction system favors the formation of a strong built-in electric field (IEF), significantly improving photocatalytic activity.26,27 As a typical p-type semiconductor photocatalyst, Co3O4 has aroused wide concern due to its non-poison, low cost, and tunable structural features.28–30 Accordingly, integrating Zn3In2S6 and Co3O4 to develop a heterojunction composite maybe indeed an effective approach for highly efficient photocatalytic oxidation of HMF into DFF.
In this work, we constructed a hollow Co3O4@Zn3In2S6 (ZC) heterojunction by uniformly coating Zn3In2S6 nanosheets (NSs) on the surface of ZIF-67-derived hollow Co3O4 nanocages for selective photo-oxidation of HMF to DFF.31 The hollow architecture of Co3O4 nanocages afford large surface area to form intimate interfacial contact with Zn3In2S6 NSs, which is beneficial to form strong interfacial interaction and facilitate the effective separation of electron–hole pairs. The optimal ZC-5 heterojunction delivers a remarkable HMF conversion rate of 95.4% with 94.1% DFF selectivity and a high yield of DFF (66.8%) under visible light irradiation and an air atmosphere in base-free water. Accordingly, characteristic analysis and density functional theory (DFT) calculations elucidate the charge transfer mechanism of S-scheme heterojunctions and the enhanced IEF effect, which improved interfacial charge separation and facilitated selective photo-oxidation of HMF to DFF. Moreover, a possible reaction mechanism for photocatalytic oxidation of HMF over the ZC S-scheme heterojunction has also been proposed. This work provides inspiring ideas to design heterojunction photocatalysts for upgrading biomass feedstock into higher-value chemicals under green and mild conditions.
Results and discussion
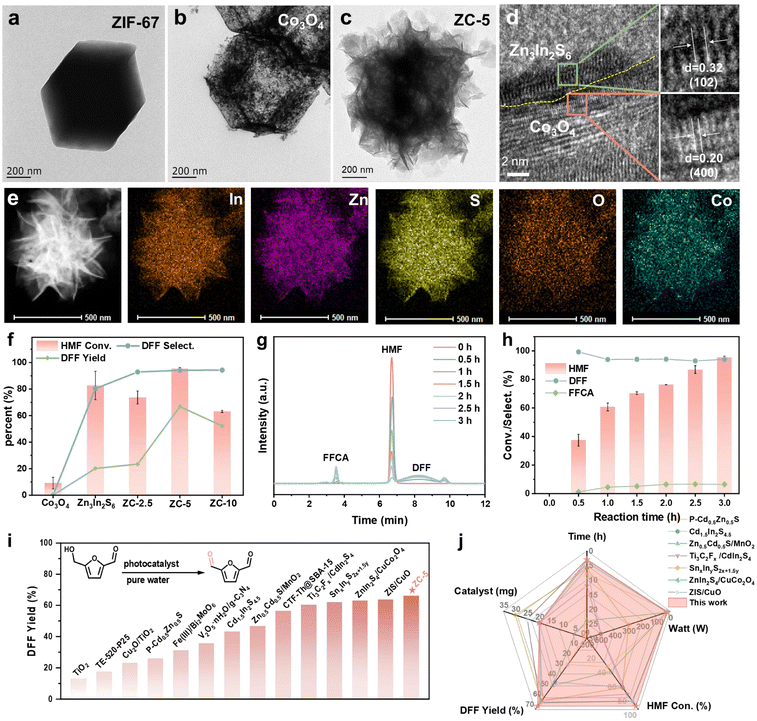
The synthetic process of the ZC–x S-scheme heterojunction is schematically illustrated in Fig. 1a. The high-quality Co3O4 nanocages were synthesized via the thermal treatment of ZIF-67 polyhedral precursors.32 Afterward, the hierarchical ZC heterostructure was constructed by in situ growing Zn3In2S6 NSs on the surface of the Co3O4 nanocages. The characteristic ZIF-67 peaks in the X-ray diffraction (XRD) pattern confirm its successful synthesis with a high degree of crystallinity (Fig. S1).33 As illustrated in Fig. 1b and S2, the derived Co3O4 displays distinct diffraction peaks at 2θ = 31.4°, 36.8°, 45°, 59.5°, and 65.3°, corresponding to the (220), (311), (400), (511), and (440) lattice planes of the spinel structure of Co3O4 (PDF#43-1003), respectively.31 The diffraction peaks of Zn3In2S6 observed at 20.7°, 28.4°, 47.2°, and 56.2° can be attributed to the (005), (102), (110), and (203) crystalline planes of hexagonal Zn3In2S6 (PDF#24-1453).34 Obviously, the ZC samples exhibit characteristic diffraction peaks of both Co3O4 and Zn3In2S6, and no impurity peaks were observed, further validating the successful synthesis of the highly crystalline ZC heterojunction. | ||
| Fig. 1 (a) Schematic illustration of the preparation process of ZC. (b) XRD patterns of Co3O4, Zn3In2S6, and ZC-5. (c) O 1s and (d) S 2p XPS spectra of Co3O4, Zn3In2S6, and ZC-5. | ||
X-ray photoelectron spectroscopy (XPS) was further measured to analyze the surface chemical compositions and states of the samples. The ZC-5 composite displays the presence of Co, O, S, Zn and In elements (Fig. S3). In the high-resolution O 1s spectrum of Co3O4, the peaks at approximately 529.91 eV, 531.16 eV, and 532.22 eV are corresponding to lattice oxygen (OL), oxygen vacancies (Ov), and hydroxyl species (O–H), respectively (Fig. 1c).31 For ZC-5, the corresponding peaks of O 1s shift to higher binding energies compared with those of individual Co3O4. In contrast, the peaks of S 2p, Zn 2p, and In 3d in ZC-5 shift toward the lower binding energy region compared with pure Zn3In2S6 (Fig. 1d, S4a and b).35–37 The peaks of Co 2p are difficult to observe due to their low surface content (Fig. S4c).33 The shifts of binding energies imply the occurrence of charge transfer within the ZC-5 heterojunction, confirming the formation of a strong interaction between Co3O4 and Zn3In2S6.
Scanning electron microscopy (SEM) and transmission electron microscopy (TEM) were performed to characterize the microstructures of as-prepared materials. ZIF-67 exhibits a dodecahedral morphology with a diameter of approximately 2 µm (Fig. 2a and S5). After calcination, Co3O4 was presented in the form of a hollow nanocage (Fig. 2b and S6). Zn3In2S6 exhibits a micro-flowerlike structure consisting of multiple nanosheets (Fig. S7). When Zn3In2S6 was in situ assembled on the surface of hollow Co3O4 nanocages, Zn3In2S6 NSs were uniformly and closely covered on Co3O4 nanocages (Fig. 2c and S8). The high-resolution TEM (HRTEM) images (Fig. 2d) of ZC-5 demonstrate an intimate interfacial contact between Zn3In2S6 and Co3O4, which can effectively promote the charge carrier transport. The lattice fringes observed at 0.20 and 0.32 nm are ascribed to the (400) plane of Co3O4 and the (102) plane of Zn3In2S6, respectively.38–40 Elemental mapping images indicate the presence of Co, O, S, Zn, and In elements within ZC-5, where S, Zn and In elements are uniformly distributed over the whole surface of Co3O4 (Fig. 2e). In addition, nitrogen adsorption–desorption measurements were conducted to analyse the porosity structure of these samples (Fig. S9 and Table S1). The ZC-5 heterojunction presents both the microporous structure of Co3O4 and the mesoporous feature of Zn3In2S6, which are advantageous for exposing more active sites.
Furthermore, the photocatalytic performance of the catalysts for HMF oxidation was evaluated under visible-light irradiation in base-free water and atmospheric oxygen. As shown in Fig. 2f, pure Zn3In2S6 exhibits a high HMF conversion rate of 82.7%, yet the yield of DFF is only 20.2%. When Co3O4 was alone used as the photocatalyst, minimal activity was displayed and no DFF was detected. Excitingly, the ZC heterojunctions enhanced catalytic activity and significantly improved the yield of DFF. The ZC heterojunctions with varying amounts of Co3O4 show distinct activity in the photocatalytic oxidation of HMF. Notably, ZC-5 with the optimal proportion (5% Co3O4) demonstrated exceptional performance, achieving 95.4% conversion of HMF with a 94.1% DFF selectivity and only trace amounts of FFCA detected as a byproduct (Fig. 2g, h and S10). More importantly, the yield of DFF (66.8%) was increased to three times that over Zn3In2S6, surpassing most reported photocatalysts under aqueous conditions (Fig. 2i and Table S3). In summary, the systematic comparison of HMF conversion rate, DFF yield, reaction time, catalyst amount and lamp power is presented in Fig. 2j, and ZC-5 exhibits superior photocatalytic performance compared to all control samples. Besides, the activity of ZC-5 showed almost no attenuation after five cycles, indicating excellent photocatalytic stability (Fig. S11), which was further verified by the XRD and XPS analyses of the used catalyst (Fig. S12 and S13).
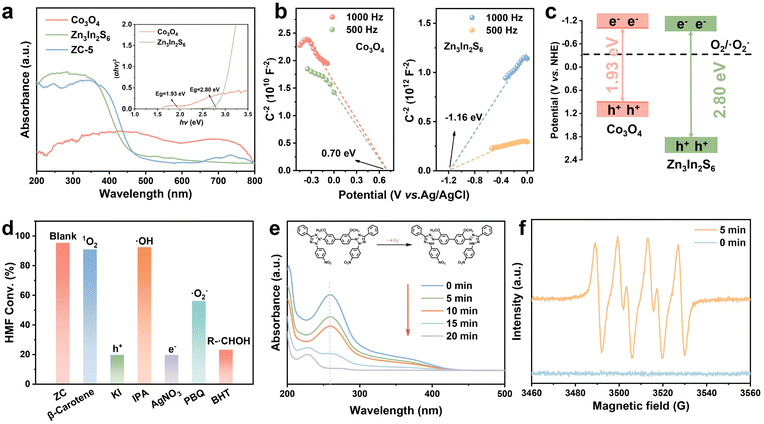
UV-vis diffuse reflectance spectroscopy (DRS, Fig. 3a) was used to investigate the optical properties and band structures of the samples, which is critical for selective photocatalytic oxidation of HMF. The ZIF-67-derived Co3O4 exhibits superior visible-light response capacity, which is ascribed to its rough, porous morphology and high surface area inherited from ZIF-67, promoting light scattering and internal reflection.41,42 Compared with pristine Zn3In2S6, the ZC composite shows obviously enhanced visible-light absorption after incorporation of Co3O4. The band gap energies (Eg) of Co3O4 and Zn3In2S6 were calculated to be 1.93 and 2.80 eV, respectively, which were determined from the corresponding Tauc plots (Fig. 3a inset).43 The Mott–Schottky (M−S) analysis indicates that Zn3In2S6 is an n-type semiconductor, while Co3O4 is a p-type semiconductor (Fig. 3b).31,44 The flat-band potentials Efb of Co3O4 and Zn3In2S6 were 0.70 and −1.16 eV based on the M−S plots. Accordingly, the valence band (VB) of Co3O4 is 0.92 eV and the conduction band (CB) of Zn3In2S6 is −0.94 eV, respectively, which were adjusted to account for the standard redox potential (0.22 eV) of the Ag/AgCl electrode.11,12 Hence, the calculated ECB of Co3O4 is −1.01 eV and the EVB of Zn3In2S6 is 1.86 eV. The above results demonstrate that the ZC heterojunction exhibits a suitable band structure for the photocatalytic oxidation of HMF (Fig. 3c).
To elucidate the photo-oxidation mechanism of HMF, scavenger tests were conducted through adding different quenchers.45 The roles of electron–hole pairs and reactive oxygen species (ROS) during the photocatalytic HMF oxidation over the ZC-5 heterojunction are illustrated in Fig. 3d. With the addition of KI and AgNO3 (h+ and e− quenchers, respectively), the conversion rate of HMF (19.8% and 19.7%) was significantly decreased.36,46 β-Carotene and isopropanol (IPA) were added to inhibit the generation of 1O2 and ˙OH, respectively, and resulted in a negligible variation in the conversion rate. When PBQ was added into the photocatalytic system, the HMF conversion rate was also reduced from 95.4% to 56.2%, suggesting that ˙O2− is the dominant ROS in the photo-oxidation of HMF over the ZC-5 heterojunction. Besides, the addition of 6-di-tert-butyl-4-methylphenol (BHT) can inhibit the generation of the R-˙CHOH radical and results in the reduction of HMF conversion, suggesting that R-CH2OH is oxidized by h+ to produce the R-˙CHOH radical during the photocatalytic oxidation process of HMF to DFF. Nitroblue tetrazolium (NBT) was further utilized as a probe to evaluate the ability of ZC-5 to generate ˙O2−.47,48 As shown in Fig. 3e and S14, upon irradiation, the conversion rate of NBT by the ZC-5 photocatalyst can reach 100% within 20 min, demonstrating that ˙O2− plays a crucial role in the catalytic process. Compared with Co3O4 and Zn3In2S6, the higher NBT degradation rate of ZC-5 demonstrates the enhanced generation of ˙O2−, thus promoting photocatalytic oxidation of HMF. Electron paramagnetic resonance (EPR) measurements were also performed using 5,5-dimethyl-1-pyrroline N-oxide (DMPO) as a trapping agent to accurately identify the generation of ˙O2− by the ZC-5 heterojunction. The distinct DMPO-˙O2− signals were detected upon irradiation, whereas no signals were detected without light irradiation (Fig. 3f).49 These findings illustrate the main ROS (˙O2−) and the formation pathway (R-˙CHOH) in the photocatalytic conversion of HMF to DFF.
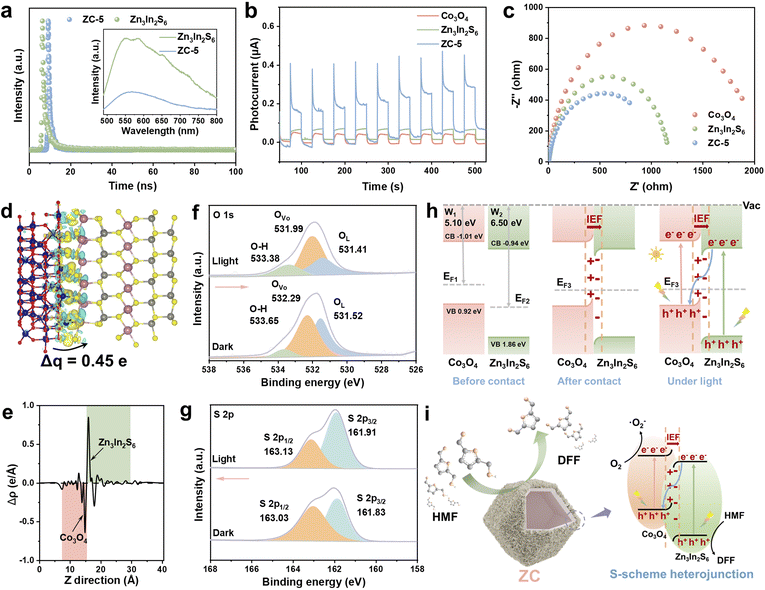
The efficiency of photogenerated carrier separation and migration is vitally important for achieving high photocatalytic performance. To elucidate the charge transfer kinetics on the ZC catalyst, photoluminescence (PL) and photo/electrochemical measurements were conducted. As shown in Fig. 4a, the ZC-5 composite exhibits significantly weaker photoluminescence (PL) intensity than that of Zn3In2S6, indicating that the construction of the ZC S-scheme heterojunction can inhibit charge recombination.33,50 In addition, the time-resolved photoluminescence (TR-PL) decay spectra display that the average fluorescence lifetime of ZC-5 (1.42 ns) is shorter than that of Zn3In2S6 (3.13 ns), demonstrating that the construction of the heterojunction significantly suppresses the recombination of photogenerated e−-h+ pairs (Fig. 4a and Table S2). The transient photocurrent curve (Fig. 4b) reveals that the ZC-5 heterojunction displays much higher photocurrent density than Co3O4 and Zn3In2S6, indicating that the ZC S-scheme heterojunction can efficiently facilitate the separation and transport of photogenerated charges. Furthermore, the electrochemical impedance spectroscopy (EIS, Fig. 4c) results show that ZC-5 exhibits a smaller semicircle and lower charge transfer resistance in the Nyquist plot compared to Co3O4 and Zn3In2S6, and thus electrons in ZC-5 can migrate more readily, resulting in a faster transfer efficiency of photogenerated charge carriers.
Furthermore, density functional theory (DFT) calculations were conducted to comprehend the detailed interface interaction and charge transfer mechanism of the ZC composite. Upon the formation of the heterojunction, the potential difference between the surfaces of Co3O4 and Zn3In2S6 facilitates the transport of free electrons, which can be reflected through work function determined by DFT calculation according to the equation Φ = Evac– EF.51,52 The work functions of Co3O4 and Zn3In2S6 are 5.1 and 6.5 eV, respectively (Fig. S15). Since the EF of Co3O4 is higher than that of Zn3In2S6, electrons spontaneously migrate from high EF (Co3O4) to low EF (Zn3In2S6) until the Fermi energy levels reach equilibrium, thereby establishing IEF.53,54 As illustrated in Fig. 4d and e, the charge density difference (CDF) of ZC shows that electron transfer occurs in the interface between Co3O4 and Zn3In2S6, where electrons accumulated on Zn3In2S6 and depleted on Co3O4, respectively. The redistribution of electrons resulted in the establishment of IEF (Fig. 4h). Under light irradiation, the IEF induces the recombination of electrons in the CB of Zn3In2S6 and holes in the VB of Co3O4, establishing an S-scheme electron transfer mechanism between Co3O4 and Zn3In2S6. In situ XPS spectroscopy measurements were conducted to further explore the transfer direction of photogenerated electrons in the ZC heterojunction during the photocatalytic process. As shown in Fig. 4f, g and S16, the binding energy of O 1s shifts toward lower values under light irradiation, while those of S 2p, In 3d, and Zn 2p in ZC-5 shift toward higher values. These observations indicate that the IEF drives charge transfer across the heterojunction interface. Specifically, photoexcited electrons in the conduction band of Zn3In2S6 are transferred to Co3O4 under the influence of the IEF, thereby promoting the formation of an S-scheme heterojunction within the ZC composite catalyst.
Based on the aforementioned characterization studies, the underlying mechanism for the improved photocatalytic performance of the ZC heterojunction for the oxidation of HMF to DFF is preliminarily proposed (Fig. 4i). Upon light illumination, the photoexcited e−−h+ pairs are generated at the first. Subsequently, the strong IEF within the ZC S-scheme heterojunction effectively drove the separation of photoexcited e− and h+ and facilitate their migration along the interface of Zn3In2S6 and Co3O4. Owing to the S-scheme electron transfer mechanism of the ZC heterojunction, photogenerated e− in the CB of Zn3In2S6 and photogenerated h+ in the VB of Co3O4 undergo recombination at the interface. Meanwhile, the photogenerated e− retained in the CB of Co3O4 efficiently reduce O2 to ˙O2− radicals, while the photogenerated h+ left in the VB of Zn3In2S6 oxidize HMF to generate active intermediates, which then react with ˙O2− to produce DFF.
Conclusions
In summary, we have successfully constructed a hollow Co3O4@Zn3In2S6 nanocage photocatalyst for enhancing photo-oxidation of HMF to DFF under visible light irradiation and an air atmosphere in pure water. The intimate interfacial contact between Co3O4 and Zn3In2S6 forms the strong interfacial interaction and accelerates the transfer of photogenerated charge carriers, resulting in remarkable photocatalytic performance. The optimal ZC-5 heterojunction achieves the highest HMF conversion rate (95.4%) and DFF selectivity (94.1%), as well as an increased DFF yield (66.8%). The systematic experiment analysis and DFT calculations elucidated that the formation of a strong IEF and an S-scheme charge transform mechanism within the ZC heterojunction can effectively drive the charge separation and migration, leading to enhanced photo-oxidation of HMF to DFF. This study provides an effective strategy for developing high-performance photocatalysts by designing a hollow nanocage heterojunction for oxidizing HMF to produce DFF under mild conditions.Author contributions
Rong Dang: validation, methodology, investigation, formal analysis, writing – original draft. Kexin Fan: data curation. Yan-Ning Wang: data curation. Guangsheng Zhu: data curation. Liu Liu: data curation. Zifu Hu: data curation. Taolian Guo: writing – review & editing, validation, methodology, supervision, funding acquisition, conceptualization. Wu Chen: writing – review & editing, supervision. Fengxiang Chen: writing – review & editing, supervision, funding acquisition. All authors analyzed and discussed the experimental results.Conflicts of interest
There are no conflicts to declare.Data availability
The data supporting this article have been included as part of the supplementary information (SI). Supplementary information is available. See DOI: https://doi.org/10.1039/d6sc00060f.Acknowledgements
This work was supported by grants from the National Natural Science Foundation of China (52373085, 52573090, U21A2095, and 52533017), Educational Commission of Hubei Province of China (Q20231711), Department of Science and Technology of Hubei Province (No. 2025CSA001 and 2024CSA076), Outstanding Young and Middle-aged Scientific and Technology Innovation Team of Higher Education Institutions of Hubei Province (No. T2024010), Innovative Team Program of Natural Science Foundation of Hubei Province (No. 2023AFA027), and Major Fundamental Research of Natural Science Foundation of Shandong Province (ZR2025ZD33). We thank the Analytical and Testing Center of Wuhan Textile University for the HPLC tests. The authors acknowledge Jinan Gengzi Supercomputing Cloud Tech Co., Ltd. for providing High Performance Computing resources that support the theoretical calculations.References
- N. Li, K. Yan, T. Rukkijakan, J. Liang, Y. Liu, Z. Wang, H. Nie, S. Muangmeesri, G. Castiella-Ona, X. Pan, Q. Zhou, G. Jiang, G. Zhou, J. Ralph, J. S. M. Samec and F. Wang, Nature, 2024, 630, 381–386 CrossRef CAS PubMed.
- F. Vidal, E. R. van der Marel, R. W. F. Kerr, C. McElroy, N. Schroeder, C. Mitchell, G. Rosetto, T. T. D. Chen, R. M. Bailey, C. Hepburn, C. Redgwell and C. K. Williams, Nature, 2024, 626, 45–57 CrossRef CAS PubMed.
- Z. Huang, Z. Zhao, C. Zhang, J. Lu, H. Liu, N. Luo, J. Zhang and F. Wang, Nat. Catal., 2020, 3, 170–178 CrossRef CAS.
- D. Shi, J. Zhang, Z. Qi, L. Duan, R. Yu, Q. Yue, D. Meng, T. Kang, L. Liu, K. Lan, W. Li, D. Zhao, L. Wu and Y. Ma, Adv. Mater., 2025, 37, 2510246 CrossRef CAS PubMed.
- L. Chico-Mesa, A. Rodes, R. M. Arán-Ais and E. Herrero, Nat. Commun., 2025, 16, 3349 CrossRef CAS PubMed.
- X. Cao, Y. Ding, D. Chen, W. Ye, W. Yang and L. Sun, J. Am. Chem. Soc., 2024, 146, 25125–25136 CrossRef CAS PubMed.
- T. Xia, W. Gong, Y. Chen, M. Duan, J. Ma, X. Cui, Y. Dai, C. Gao and Y. Xiong, Angew. Chem., Int. Ed., 2022, 61, e202204225 CrossRef CAS PubMed.
- Z. Li, M. Zhang, X. Xin and H. Lv, ChemCatChem, 2021, 13, 1389–1395 CrossRef CAS.
- W. Xue, J. Ye, Z. Zhu, R. Kumar and J. Zhao, Energy Environ. Sci., 2025, 18, 214–226 RSC.
- Y. Gao, L. Ge, H. Xu, K. Davey, Y. Zheng and S.-Z. Qiao, ACS Catal., 2023, 13, 11204–11231 CrossRef CAS.
- Y. Liu, W. Xue, J. Ye, R. Zhang, Y. Shao, A. P. Rangappa and J. Zhao, Chem. Eng. J., 2025, 515, 163620 CrossRef CAS.
- M. Zhang, Z. Li, X. Xin, J. Zhang, Y. Feng and H. Lv, ACS Catal., 2020, 10, 14793–14800 CrossRef CAS.
- S. Jiang, Z. Chen, S. Xiong, H. Zhao, X. Xiao and Z. Shen, J. Energy Chem., 2025, 109, 830–838 CrossRef CAS.
- S. Xiong, X. Liu, Z. Shen, Z. Hu, H. Yang, J. Hao, J. Cai and J. Yang, Inorg. Chem., 2023, 62, 20120–20128 CrossRef CAS PubMed.
- D. Jiao, C. Ding, M. Xu, X. Ruan, S. K. Ravi and X. Cui, Adv. Funct. Mater., 2024, 35, 2416753 CrossRef.
- J. Chen, M. Mu, Z. Wang, M. Ma, F. A. Qaraah, X. Yin and G. Bai, ACS Sustainable Chem. Eng., 2024, 12, 18161–18173 CrossRef CAS.
- T. Liu, Y. Xiong, X. Wang, Y. Xue, W. Liu and J. Tian, J. Colloid Interface Sci., 2023, 640, 31–40 CrossRef CAS PubMed.
- J. L. Zhou, Y. F. Mu, M. Qiao, M. R. Zhang, S. X. Yuan, M. Zhang and T. B. Lu, Angew. Chem., Int. Ed., 2025, 64, e202506963 CrossRef CAS PubMed.
- C. Li, X. Hu, Z. Chen and Z. Ding, Sep. Purif. Technol., 2025, 377, 134344 CrossRef CAS.
- Y. Che, K. Wang, C. Wang, B. Weng, S. Chen and S. Meng, J. Mater. Sci. Technol., 2026, 243, 228–236 CrossRef.
- X. Ruan, S. Zhao, M. Xu, D. Jiao, J. Leng, G. Fang, D. Meng, Z. Jiang, S. Jin, X. Cui and S. K. Ravi, Adv. Energy Mater., 2024, 14, 2401744 CrossRef CAS.
- X. Tan, S. Si, D. Xiao, X. Bao, K. Song, Z. Wang, Y. Liu, Z. Zheng, P. Wang, Y. Dai, B. Huang and H. Cheng, ACS Catal., 2023, 13, 14395–14403 CrossRef CAS.
- S. Wan, W. Wang, B. Cheng, G. Luo, Q. Shen, J. Yu, J. Zhang, S. Cao and L. Zhang, Nat. Commun., 2024, 15, 9612 CrossRef CAS PubMed.
- X. Deng, J. Zhang, K. Qi, G. Liang, F. Xu and J. Yu, Nat. Commun., 2024, 15, 4807 CrossRef CAS PubMed.
- Q. P. Huang, C. Yang, Q. Yin, A. A. Zhang, H. X. Liu, L. Li, M. M. Liu, Z. B. Fang and T. F. Liu, Angew. Chem., Int. Ed., 2025, 64, e202502009 CrossRef CAS PubMed.
- K. Huang, G. Liang, S. Sun, H. Hu, X. Peng, R. Shen and X. Li, J. Mater. Sci. Technol., 2024, 193, 98–106 CrossRef CAS.
- L. Shi, Y. Wu, Z. Chang, P. Jiang, Y. Hua, Q. Shi, S. Hou and H. Wang, Appl. Catal., B, 2025, 378, 125618 CrossRef CAS.
- L. Xu, Q. Jiang, Z. Xiao, X. Li, J. Huo, S. Wang and L. Dai, Angew. Chem., Int. Ed., 2016, 55, 5277–5281 CrossRef CAS PubMed.
- B. Yan, Q. Ruan, S. Wang, L. Kong, P. Zhang, H. Wang and Z. Sun, Adv. Funct. Mater., 2024, 34, 2408895 CrossRef CAS.
- S. Bai, W. Jing, G. He, C. Liao, F. Wang, Y. Liu and L. Guo, ACS Nano, 2023, 17, 10976–10986 CrossRef CAS PubMed.
- J. Fan, L. Shi, H. Ge, J. Liu, X. Deng, Z. Li and Q. Liang, Adv. Funct. Mater., 2024, 35, 2412078 CrossRef.
- D. Ding, K. Shen, X. Chen, H. Chen, J. Chen, T. Fan, R. Wu and Y. Li, ACS Catal., 2018, 8, 7879–7888 CrossRef CAS.
- C. Tang, T. Bao, S. Li, X. Li, H. Rao, P. She and J. Qin, Adv. Funct. Mater., 2025, 35, 2415280 CrossRef CAS.
- J. Luo, X. Wei, Y. Qiao, C. Wu, L. Li, L. Chen and J. Shi, Adv. Mater., 2023, 35, 2210110 CrossRef CAS PubMed.
- C. Yuan, H. Yin, J. Li, Y. Zhang, H. Chen, D. Xiao, Q. Wang, Y. Zhang and Q.-K. Xue, Nat. Commun., 2025, 16, 6607 CrossRef CAS PubMed.
- T. Guo, R. Dang, A. Zheng, S. Wang, X. Dong, Z. Xiong, Z. Chen, Z. Hu, W. Chen and C. Wang, Green Chem., 2025, 27, 1157–1168 RSC.
- Z. Zou, Y. Wang, T. Zhang, J. Lu, V. Turkevich, Y. Yu, J. Xu, H. Lin, Y. Li and L. Wang, Chem. Eng. J., 2025, 520, 165595 CrossRef CAS.
- Y. Liu, J. Wei, Z. Yang, L. Zheng, J. Zhao, Z. Song, Y. Zhou, J. Cheng, J. Meng, Z. Geng and J. Zeng, Nat. Commun., 2024, 15, 3619 CrossRef CAS PubMed.
- R. Chahal, R. Rathee, S. Dahiya, R. Punia, A. S. Maan, K. Singh, R. Tripathi, M. A. Manthrammel, M. Shkir and A. Ohlan, Chem. Eng. J., 2025, 520, 165715 CrossRef CAS.
- X. Li, G. Zhang, N. Li, Q. Xu, H. Li, J. Lu and D. Chen, Nano Energy, 2024, 126, 109671 CrossRef CAS.
- J. Fan, L. Shi, H. Ge, J. Liu, X. Deng, Z. Li and Q. Liang, Adv. Funct. Mater., 2025, 35, 2412078 CrossRef CAS.
- F. Xu, L. Zheng, J. Zhang, Y. He, H. Cao, X. Zheng, H. García and J. Yu, Nat. Catal., 2026, 9, 73–86 CrossRef CAS.
- P. Zhou, I. A. Navid, Y. Ma, Y. Xiao, P. Wang, Z. Ye, B. Zhou, K. Sun and Z. Mi, Nature, 2023, 613, 66–70 CrossRef CAS PubMed.
- B. Chen, Y. Wang, S. Shen, W. Zhong, H. Lu and Y. Pan, Small Methods, 2024, 8, 2301598 CrossRef CAS PubMed.
- J. Zhao, Y. Wang, H. Liu, R. Zhang, W. Jia, J. Zhang, Y. Sun and L. Peng, ACS Catal., 2025, 15, 3464–3474 CrossRef CAS.
- Y. Wang, H. Liu, T. Lv, W. Jia, R. Zhang, L. Peng and J. Zhang, Adv. Funct. Mater., 2024, 35, 2415842 CrossRef.
- Z. Xue, B. Zhang, Q. Guo, Y. Wang, Q. Li, K. Yang and S. Qiao, Adv. Mater., 2025, 37, e10201 CrossRef CAS PubMed.
- Y. Deng, W. Liu, R. Xu, R. Gao, N. Huang, Y. Zheng, Y. Huang, H. Li, X. Kong and L. Ye, Angew. Chem., Int. Ed., 2024, 63, e202319216 CrossRef CAS PubMed.
- W. Xue, Y. Liu, X. Ma, J. Ye, X. Bai, J. Zhao and B. Liu, ACS Catal., 2025, 15, 9621–9632 CrossRef CAS.
- X. Cheng, Q. Sun, G. Zhang, W. Xing, Z.-A. Lan, S. Wang and Z. Pan, ACS Catal., 2025, 15, 13167–13178 CrossRef CAS.
- Y. Fan, Z. Hu, X. Hao and Z. Jin, Carbon, 2024, 228, 119418 CrossRef CAS.
- H. Ding, R. Shen, K. Huang, C. Huang, G. Liang, P. Zhang and X. Li, Adv. Funct. Mater., 2024, 34, 2400065 CrossRef CAS.
- X. Ma, Z. Wu, Z. Zhou, S. Lu and Z. Jin, Appl. Catal., B, 2025, 278, 125614 CrossRef.
- H. L. He, L. L. Zhang, H. Zhou, Y. Nie, H. Wang, B. Tang, H. Li, T. Ma and H. Zhang, Adv. Energy Mater., 2024, 15, 2403168 CrossRef.
| This journal is © The Royal Society of Chemistry 2026 |