Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Halogen-ion-driven polymorphs for high-performance nonlinear optical crystalline materials
Yuwei
Kang
ab,
Can
Yang
a,
Yunjie
Wang
a and
Qi
Wu
 *a
*a
aState Key Laboratory of New Textile Materials and Advanced Processing, Wuhan Textile University, Wuhan 430200, China. E-mail: wuqi2011@whu.edu.cn
bState Key Laboratory of Functional Crystals and Devices, Fujian Institute of Research on the Structure of Matter, Chinese Academy of Sciences, Fuzhou 350002, China
First published on 20th February 2026
Abstract
Noncentrosymmetric (NCS) crystalline materials are indispensable for nonlinear optical (NLO) applications, yet their rational design remains challenging due to thermodynamic preferences for centrosymmetric configurations. Herein, we demonstrate a halogen-driven strategy for obtaining NCS structures by leveraging competitive coordination between halide anions (X−) and stereochemically active Sn2+ centers. Precise halogen substitution induced the formation of two new polymorphs of [N(C2H5)4]SnBr3 (Cc and Cmc21 phase) using different halide sources (i.e., [N(C2H5)4]Cl or [N(C2H5)4]Br). Compared to the Cc phase (2 × KH2PO4), the resulting Cmc21 phase exhibits exceptional second-harmonic generation (SHG) efficiency (5.6 × KH2PO4). This performance ranks among the highest for all reported Sn-based organic–inorganic hybrid NLO materials to date. Theoretical calculations indicate that the [SnBr3]− unit is the primary source of the strong SHG response. This work establishes halogen-driven symmetry control as a viable strategy for achieving a dramatically enhanced SHG response, thereby providing a valuable reference for the rational design of high-performance NLO materials.
Introduction
Nonlinear optical (NLO) crystals are pivotal for applications such as laser frequency conversion, optical communications, and quantum technologies, all of which rely on their non-centrosymmetric (NCS) structure.1–7 Despite this, the rational design of such crystals faces a formidable challenge: approximately 80% of all inorganic compounds crystallize in centrosymmetric (CS) structures due to the thermodynamic preference for antiparallel dipole alignment.8–12 Although conventional design strategies utilize asymmetric building units, such as π-conjugated anions,13–22 d0 and d10 transition metals,23–28 and lone-pair-active cations to induce structural polarity,29–35 achieving precise control over the formation of NCS structures remains a significant challenge.By enabling diverse crystal structures under a fixed chemical composition, polymorphs significantly increase the probability of obtaining NCS compounds, thereby providing an effective route for designing high-performance NLO materials. It is this structural richness and tunable properties that make polymorphs a subject of extensive research interest.36 Wu et al. specifically reviewed NLO polymorphs and noted that, at present, most polymorphs are oxides and sulfides, obtained under different external conditions such as temperature, pressure, and the ratio of raw materials.37 Halides frequently exhibit polymorphism, and their synthesis, often conducted in solution, enables polymorphs' formation. Notable examples reported in recent years include α- and β-HgClBr,38 CsHgBr3,39 and β-RbCdI3·H2O.40 Tao et al. observed in 2016 that Sn2+ formed the crystal NH(CH3)3SnCl3 when only Cl− was present in the reaction system. However, upon introducing Br− into the system, Sn2+ preferentially coordinated with the softer Br− anion to yield NH(CH3)3SnBr3.41 This behavior aligns well with the hard–soft acid–base (HSAB) principle, as the relatively soft Sn2+ exhibits a greater affinity for the softer halide (Br− > Cl−) (Fig. 1a). The study clearly demonstrated the thermodynamic control exerted by the local ionic environment on product selectivity. Building on this, we further hypothesize that competitive coordination among halide ions and the central metal ion in mixed-halide systems may induce locally distorted intermediate states, which in turn can trigger the reorganization of anionic groups and ultimately drive the crystal structure toward a more thermodynamically stable phase.
 | ||
| Fig. 1 Visualization of halide-driven phase selection in NH(CH3)3SnX3 (X = Cl, Br) (a) and [N(C2H5)4]SnBr3 (b) based on the hard–soft acid–base principle. | ||
Here we designed a halide-driven synthesis strategy that leverages the competitive coordination between halide anions (X−) and stereochemically active Sn2+ centers to selectively obtain distinct structural polymorphs. Specifically, using [N(C2H5)4]Cl as the halide source yielded a monoclinic Cc phase (phase II), whereas [N(C2H5)4]Br produced an orthorhombic polar Cmc21 phase (phase I), demonstrating straightforward control over structural polymorphism in the [N(C2H5)4]SnX3 system. The stereochemical activity of the Sn2+ 5s2 lone-pair electrons induces pronounced distortion in the [SnX3]− units, a feature known to favor NCS structures. Phase selectivity aligns well with the HSAB principle: as a soft acid, Sn2+ exhibits a stronger thermodynamic preference for the softer base Br−, leading to the formation of stable [SnBr3]− units that subsequently assemble into the polar Cmc21 phase. Notably, when Cl− coexists with Br− in the system, competitive coordination arises between the relatively harder base Cl− and the softer Br−. This competition is likely to involve kinetically unstable mixed-halogen intermediates such as [SnBr3−xClx]−, which can alter the normal nucleation pathway and thereby direct crystallization toward either the lower-symmetry Cmc21 phase or the higher-symmetry Cc phase (Fig. 1b). Crucially, Br− owing to its greater polarizability and softer ligand character-further enhances the stereochemical activity of the Sn2+ lone pair within the [SnX3]− units, driving the structural evolution from the less polar Cc phase to the more polar Cmc21 phase. The highly ordered alignment of these distorted anions in the Cmc21 phase results in a remarkable second-harmonic generation (SHG) response, the highest reported to date among Sn-based organic–inorganic hybrid NLO crystals.
This work not only reaffirms the decisive role of the HSAB principle in governing product selectivity but, more importantly, reveals a new mechanism by which halide ions kinetically steer crystallization pathways through competitive coordination. This mechanism shows that halide ions can not only dictate chemical composition through thermodynamic preference but also influence intermediate structures and crystallization kinetics, enabling the directed optimization of both crystal symmetry and macroscopic performance. The findings provide a novel rationale for the rational design of high-performance NLO crystalline materials.
Results and discussion
The phase purity and high crystallinity of both compounds were confirmed by powder X-ray diffraction (PXRD), as shown in Fig. S1. Energy-dispersive X-ray spectroscopy (EDS) measurements yielded Sn![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Br molar ratios of 1
Br molar ratios of 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3.46 and 1
3.46 and 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3.01 for the respective phases. These values are in reasonable agreement with the expected stoichiometric ratio of 1
3.01 for the respective phases. These values are in reasonable agreement with the expected stoichiometric ratio of 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3 derived from single-crystal X-ray diffraction structural determinations, thus confirming the bulk composition. The crystallographic data have been deposited at the Cambridge Crystallographic Data Centre (CCDC) under deposition numbers 2518216 and 2518217.
3 derived from single-crystal X-ray diffraction structural determinations, thus confirming the bulk composition. The crystallographic data have been deposited at the Cambridge Crystallographic Data Centre (CCDC) under deposition numbers 2518216 and 2518217.
X-ray photoelectron spectroscopy (XPS) confirms the presence of C, H, N, Sn, and Br in both compounds I and II (Fig. S2 and S3). Furthermore, the high-resolution Sn 3d spectrum exhibits a well-defined doublet with binding energies of 484.9 eV (Sn 3d5/2) and 493.3 eV (Sn 3d3/2), characterized by a spin–orbit splitting of 8.4 eV, which is a definitive signature of the Sn2+ oxidation state. Critically, the lack of higher binding energy features confirms the absence of detectable Sn4+, indicating no significant oxidation. The XPS results confirm the Sn2+ oxidation state.41 This consistency suggests that the oxidation state assigned from the structure is correct and that no significant surface oxidation has occurred, thereby supporting the overall structural assignment.
Compounds I and II crystallize in the polar space groups Cmc21 and Cc, respectively. Their NCS nature is unambiguously confirmed by the observed SHG signals, despite slightly elevated Flack parameters in the structural refinements. Both compounds adopt zero-dimensional (0D) structures consisting of discrete [SnBr3]− anions and [N(C2H5)4]+ cations (Fig. 2b and c). The [SnBr3]− units exhibit distorted trigonal pyramidal geometries, as evidenced by Sn–Br bond lengths ranging from 2.679–2.682 Å in I and 2.653–2.710 Å in II, and Br–Sn–Br bond angles varying between 91.67–93.73° in I and 90.36–94.90° in II. These structural distortions play a crucial role in breaking symmetry. Although the two compounds share the same chemical composition, they exhibit markedly different crystal packing arrangements, which directly account for their distinct space group symmetries. In I, the [SnBr3]− units are aligned in a highly ordered fashion along the b-axis, forming a mirror-like arrangement (Fig. 2a and d). In the crystal structures of both compounds, the dipole moments of the [SnBr3]− units are arranged antiparallel along the b-axis, leading to mutual cancellation, while their polarities align cooperatively along the a and c-axes. This alignment results in the establishment of macroscopic non-centrosymmetry, which gives rise to a pronounced SHG response.
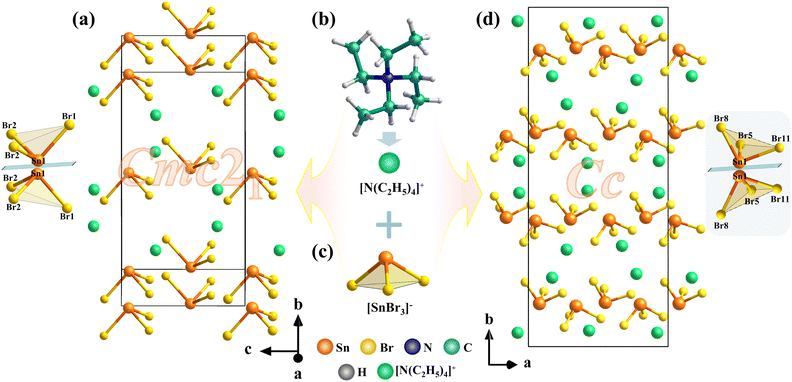 | ||
| Fig. 2 (a) Arrangement of the [SnBr3]− in I. (b) [N(C2H5)4]+ cations. (c) [SnBr3]− pyramid. (d) Arrangement of the [SnBr3]− in II. | ||
The infrared spectra reveal nearly identical absorption profiles for compounds I and II (Fig. S4, Table S8). Taking I as a representative example, the characteristic vibrational bands are unambiguously assigned as follows: the absorption at 2978 cm−1 corresponds to the asymmetric stretching vibration of C–H bonds (νas C–H), and the bands at 1457 cm−1 and 1391 cm−1 are attributed to the asymmetric bending vibrations of CH3 and CH2 groups (δas CH3 and δas CH2), while the features at 1183 cm−1 and 782 cm−1 are assigned to rocking modes of CH3 and CH2 moieties. All observed vibrational assignments align consistently with previously reported literature, thereby confirming the structural integrity of the organic components in the material.42
The optical properties of [N(C2H5)4]SnBr3 (compounds I and II) were further investigated using UV-vis absorption spectroscopy derived from the Kubelka–Munk function.43 As shown in Fig. S5, the optical band gaps of I and II were determined to be 3.40 eV and 3.41 eV, respectively. This mechanistic insight suggests that the fundamental electronic absorption is primarily dictated by the local [SnBr3]− coordination geometry rather than the global crystal packing.
Differential scanning calorimetry (DSC) analysis revealed a highly reversible thermally induced solid–solid phase transition in these compounds (Fig. S6). A sharp endothermic peak was observed at 49 °C/51 °C during heating, while a symmetrical exothermic peak appeared at a similar position during cooling. The narrow thermal hysteresis indicates an extremely low energy barrier for this phase transition, confirming its excellent thermal reversibility. This low-energy characteristic suggests that the transition is likely attributable to minor structural adjustments, such as distortions within the [SnBr3]− anion framework or conformational disordering of the organic cations, rather than a major reconstruction of the crystal lattice.
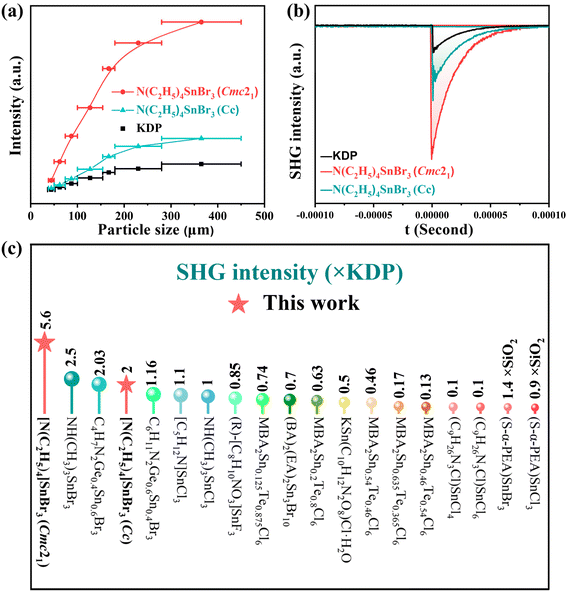
Since both compounds I and II crystallize in NCS space groups, their powder SHG effects were evaluated using the modified Kurtz–Perry method.44 As illustrated in Fig. 3a, under 1064 nm laser irradiation, the SHG intensities of I and II initially increase with particle size before plateauing, indicating phase-matching behavior. Quantitative measurements reveal that I and II exhibit SHG efficiencies approximately 5.6 and 2 times that of KH2PO4 (KDP), respectively (Fig. 3b). A survey of currently reported organic–inorganic hybrid Sn-based NLO crystals demonstrates that I possesses the highest SHG intensity among all such materials documented to date (Fig. 3c and Table S9).31,41,45–51
To gain deeper insights into the structure–property relationship, we performed systematic theoretical calculations. Band structure calculations reveal theoretical optical band gaps of 3.4 eV and 3.5 eV for compounds I and II, respectively, showing excellent agreement with the experimentally determined value of 3.4 eV. This close correspondence validates the reliability of our computational methodology. Density of states (DOS) analysis reveals that the valence band maximum (VBM) is dominated by contributions from the Sn-5p and Br-4s orbitals of the inorganic [SnBr3]− units. In contrast, the conduction band minimum (CBM) is primarily constituted by the Sn-5p orbitals, with a minor contribution from Br-4s states (Fig. 4a and b). This electronic configuration demonstrates that the formation of the optical band gap is principally governed by the inorganic [SnBr3]− trigonal pyramids. Such a well-defined electronic structure, predominantly localized on the anionic framework, provides critical insight into the origin of the SHG response, as the NLO activity can be directly correlated with these dominant transitions.
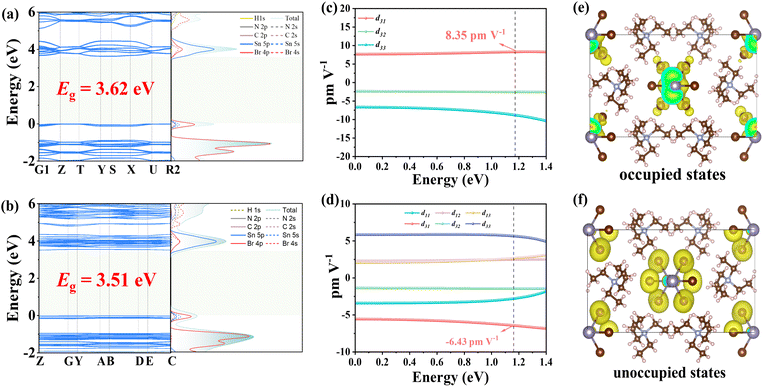 | ||
| Fig. 4 Band structure and density of states of I (a) and II (b). SHG tensor of I (c) and II (d). Occupied states (e) and unoccupied states (f) of I. | ||
To elucidate the origin of the SHG responses in compounds I and II, we calculated their NLO coefficients (Fig. 4c and d).52 The results show that the largest SHG tensor component of I is d31 = 8.35 pm V−1, with an effective nonlinear coefficient (deff) of 4.54 pm V−1. For II, the largest tensor component is d31 = −6.43 pm V−1, and its deff is 4.36 pm V−1. These computational results further confirm the validity of our experimental observations. Using I as an example, we further performed SHG density analysis, revealing that the primary contributions arise from the Br-4p nonbonding states (lone pairs) near the VBM and the Br antibonding orbitals at the CBM, which collectively modulate the electronic structure (Fig. 4e and f). Consequently, the pronounced SHG response of [N(C2H5)4]SnBr3 originates from the synergistic effects of the [SnBr3]− anionic framework. Additionally, the calculated shortest phase-matching wavelengths for the two compounds are 426 nm and 476 nm, respectively (Fig. S7), indicating that phase matching is achievable at 1064 nm, which further validates the accuracy of our experimental results.
Conclusions
In summary, this work demonstrates a halogen-mediated strategy for achieving controlled symmetry breaking and a significantly enhanced SHG response. We successfully obtained two distinct phases of [N(C2H5)4]SnBr3, the Cc and Cmc21 polymorphs. The Cmc21 phase exhibits a record SHG efficiency of 5.6 × KDP, a performance that underscores how the specific anionic arrangement is decisive in optimizing the macroscopic NLO response. Theoretical analyses confirm the [SnBr3]− unit as the fundamental NLO-active group. Consequently, our findings establish that metal centers with stereochemically active lone pairs, when integrated with competitive coordination, provide a foundation for precise symmetry control, offering a strategic route for designing NCS materials. This approach may be generalizable to other metal-halide systems, thereby paving a promising avenue for discovering future high-performance NLO materials.Author contributions
Yuwei Kang: formal analysis, writing-original draft, writing-review & editing. Can Yang: methodology, data curation. Yunjie Wang: data curation, formal analysis. Qi Wu: funding acquisition, supervision, conceptualization, writing-review & editing.Conflicts of interest
There are no conflicts to declare.Data availability
The data that support the findings of this study are available in the supplementary information (SI) of this article. Supplementary information: experimental section and additional tables and figures. See DOI: https://doi.org/10.1039/d5sc10064j.CCDC 2518216 and 2518217 contain the supplementary crystallographic data for this paper.53a,b
Acknowledgements
This work is supported by the National Natural Science Foundation of China (No. 22275052) and Department of Science and Technology of Hubei Province (2025AFA111 and 2024CSA076). The authors would like to thank Chensheng Lin for the help in the theoretical calculations.Notes and references
- W. Zeng, Y. Tian, H. Zeng, Z. Lin and G. Zou, Angew. Chem., Int. Ed., 2025, 64, e202422818 CrossRef CAS PubMed.
- Y. Zhu, J. Gou, C. Yang, Q. Zhu, Y. Xiong and Q. Wu, Angew. Chem., Int. Ed., 2025, 64, e202509290 CrossRef CAS PubMed.
- X. T. Zhang, X. X. Jiang, H. Gao, K. N. Duanmu, C. Wu, Z. S. Lin, Z. P. Huang, M. G. Humphrey and C. Zhang, Angew. Chem., Int. Ed., 2025, 64, e202521815 CrossRef.
- Q. Xia, X. Jiang, C. Jiang, H. Zhang, Y. Hu, L. Qi, C. Wu, G. Wei, Z. Lin, Z. Huang, M. G. Humphrey and C. Zhang, Angew. Chem., Int. Ed., 2025, 64, e202503136 CrossRef CAS PubMed.
- Y. Kang, C. Yang, J. Gou, Y. Zhu, Q. Zhu, W. Xu and Q. Wu, Angew. Chem., Int. Ed., 2024, 136, e202402086 CrossRef.
- J. Qu, Y. Wang, Z. Li, M. Li, T. Pei, N. Li, D. Jiang, B. Yang, B. Li, M. Ye, P. Zhu, Y. Wang, G. Liu, X. Wang and W. Yang, J. Am. Chem. Soc., 2025, 147, 6717–6726 CrossRef CAS PubMed.
- L. Qi, X. Jiang, K. Duanmu, C. Wu, Z. Lin, Z. Huang, M. G. Humphrey and C. Zhang, J. Am. Chem. Soc., 2024, 146, 9975–9983 CrossRef CAS PubMed.
- Y. Hu, X. Jiang, C. Wu, Z. Huang, Z. Lin, M. G. Humphrey and C. Zhang, Chem. Mater., 2021, 33, 5700–5708 CrossRef CAS.
- S. Yang, H. Wu, Z. Hu, J. Wang, Y. Wu and H. Yu, Inorg. Chem., 2024, 63, 1404–1413 CrossRef CAS PubMed.
- Y. Li, J. Luo and S. Zhao, Acc. Chem. Res., 2022, 55, 3460–3469 CrossRef CAS PubMed.
- H. Liu, H. Wu, Z. Hu, J. Wang, Y. Wu and H. Yu, Chem. Mater., 2022, 34, 3501–3508 CrossRef CAS.
- Y. Kang and Q. Wu, Coord. Chem. Rev., 2024, 498, 215458 CrossRef CAS.
- Y.-L. Deng, C.-L. Hu, X. Xu, M.-Z. Zhang and J.-G. Mao, J. Am. Chem. Soc., 2025, 147, 12858–12865 CrossRef CAS PubMed.
- G. Xu, X. Bai, Z. Yang, J. Han and S. Pan, Angew. Chem., Int. Ed., 2025, 64, e202510363 CrossRef CAS PubMed.
- W. Zhang, M. Arif, H. Zhou, R. An, Z. Yang, X. Hou, S. Pan and S. Han, Adv. Opt. Mater., 2025, 13, 2403372 CrossRef CAS.
- Y. Huang, T. Jiang, B.-P. Yang, C.-L. Hu, Z. Fang and J.-G. Mao, Inorg. Chem., 2022, 61, 3374–3378 CrossRef CAS PubMed.
- C. Wu, X. Jiang, Z. Wang, L. Lin, Z. Lin, Z. Huang, X. Long, M. G. Humphrey and C. Zhang, Angew. Chem., Int. Ed., 2021, 60, 3464–3468 CrossRef CAS.
- G. Peng, C. Lin, H. Fan, K. Chen, B. Li, G. Zhang and N. Ye, Angew. Chem., Int. Ed., 2021, 60, 17415–17418 CrossRef CAS PubMed.
- G. Peng, C. Lin, D. Zhao, L. Cao, H. Fan, K. Chen and N. Ye, Chem. Commun., 2019, 55, 11139–11142 RSC.
- H. Wu, Z. Wei, Z. Hu, J. Wang, Y. Wu and H. Yu, Angew. Chem., Int. Ed., 2024, 63, e202406318 CrossRef CAS.
- S. Li, W. Li, X. Li, G. Yang, N. Ye, Z. Hu, Y. Wu and C. Li, Chem. Sci., 2024, 15, 8959–8965 RSC.
- J.-N. Zhao, J. Chen and G.-Y. Yang, Cryst. Growth Des., 2025, 25, 7264–7269 CrossRef CAS.
- D. Lu, Y. Wang, X. Li, F. Liang, K. Wu, H. Yu and H. Zhang, Angew. Chem., Int. Ed., 2025, 64, e202503341 CrossRef CAS PubMed.
- H. Yu, M. L. Nisbet and K. R. Poeppelmeier, J. Am. Chem. Soc., 2018, 140, 8868–8876 CrossRef CAS PubMed.
- J. Chen, C. Hu, F. Mao, J. Feng and J. Mao, Angew. Chem., Int. Ed., 2019, 58, 2098–2102 CrossRef CAS PubMed.
- R. E. Sykora, K. M. Ok, P. S. Halasyamani and T. E. Albrecht-Schmitt, J. Am. Chem. Soc., 2002, 124, 1951–1957 CrossRef CAS PubMed.
- C. Yang, Y. Kang, X. Wang, J. Gou, Y. Xiong, Z. Zhu, L. Chen and Q. Wu, Chem. Sci., 2024, 15, 15725–15730 RSC.
- H. M. Ngo, Y. Kuk, J. Lee, J. Lu, Z. Bai, C. Hu and K. M. Ok, Adv. Opt. Mater., 2025, 13, 2403241 CrossRef CAS.
- P.-F. Li, J.-G. Mao and F. Kong, Mater. Today Phys., 2023, 37, 101197 CrossRef CAS.
- C.-L. Hu and J.-G. Mao, Coord. Chem. Rev., 2015, 288, 1–17 CrossRef CAS.
- Y. Kang, C. Yang, J. Gou, Y. Zhu, Q. Zhu and Q. Wu, Inorg. Chem., 2024, 63, 2725–2731 CrossRef CAS PubMed.
- D.-D. Zhou, C.-L. Hu, X.-W. Zhang, J.-G. Mao and F. Kong, Chem. Sci., 2024, 15, 19920–19927 RSC.
- J. Chen, C.-L. Hu, F.-F. Mao, X.-H. Zhang, B.-P. Yang and J.-G. Mao, Chem. Sci., 2019, 10, 10870–10875 RSC.
- B. Zhang, W. Huang, J. Zhang, X. Wu, H. Lin and Q. Zhu, Angew. Chem., Int. Ed., 2025, 64, e202508555 CrossRef CAS PubMed.
- J.-H. Wu, C.-L. Hu, Y.-F. Li, J.-G. Mao and F. Kong, Chem. Sci., 2024, 15, 8071–8079 RSC.
- C. A. López, C. Abia, J. Gainza, P. Kayser, N. N. Nemes, O. J. Dura, J. L. Martínez, M. T. Fernández-Díaz, C. Álvarez-Galván and J. A. Alonso, Mater. Adv., 2021, 2, 3620–3628 RSC.
- K. Wu, Y. Yang and L. Gao, Coord. Chem. Rev., 2020, 418, 213380 CrossRef CAS.
- Y. Dang, X. Meng, K. Jiang, C. Zhong, X. Chen and J. Qin, Dalton Trans., 2013, 42, 9893 RSC.
- S. Lv, Q. Wu, X. Meng, L. Kang, C. Zhong, Z. Lin, Z. Hu, X. Chen and J. Qin, J. Mater. Chem. C, 2014, 2, 6796–6801 RSC.
- Q. Wu, X. Liu, S. Xu, H. Pi, X. Han, Y. Liu and Y. Li, Dalton Trans., 2019, 48, 6787–6793 RSC.
- Y. Dang, C. Zhong, G. Zhang, D. Ju, L. Wang, S. Xia, H. Xia and X. Tao, Chem. Mater., 2016, 28, 6968–6974 CrossRef CAS.
- A. Ouasri, A. Rhandour, M. C. Dhamelincourt, P. Dhamelincourt, A. Mazzah and M. Taibi, Phase Transitions, 2003, 76, 701–709 CrossRef CAS.
- P. Kubelka and F. Munk, Tech. Phys., 1931, 12, 259–274 Search PubMed.
- S. K. Kurtz, T. T. Perry and J. G. Bergman, Appl. Phys. Lett., 1968, 12, 186–188 CrossRef CAS.
- X. Liu, C. Ji, Z. Wu, L. Li, S. Han, Y. Wang, Z. Sun and J. Luo, Chem.–Eur. J., 2019, 25, 2610–2615 CrossRef CAS PubMed.
- J. Wu, S.-F. Yan, W. Liu and S.-P. Guo, Inorg. Chem., 2024, 63, 17367–17371 CrossRef CAS PubMed.
- A. Jung, Y. Li and K. M. Ok, Dalton Trans., 2024, 53, 105–114 RSC.
- K. Li, X. Wang, X. Li, F. Wu, F. Zhang, Q. Wei, Z. Yue, J. Luo and X. Liu, Inorg. Chem., 2024, 63, 2275–2281 CrossRef CAS PubMed.
- Y.-H. Zhu, Z.-L. Geng, H.-X. Tang, R.-B. Fu, Z.-J. Ma and X.-T. Wu, Scr. Mater., 2024, 238, 115728 CrossRef CAS.
- K. Tao, Q. Li and Q. Yan, Adv. Opt. Mater., 2024, 12, 2400018 CrossRef CAS.
- X. Wen, G. Yi, Z. Zhang, C. Gou, H. Zeng, L. Huang, G. Zou and Z. Lin, Dalton Trans., 2024, 53, 13195–13200 RSC.
- X. Cheng, M. H. Whangbo, G. C. Gou, M. Hong and S. Deng, Angew. Chem., Int. Ed., 2018, 57, 3933–3937 CrossRef CAS.
- (a) CCDC 2518216: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2qjdsg; (b) CCDC 2518217: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2qjdth.
| This journal is © The Royal Society of Chemistry 2026 |