Open Access Article
Open Access ArticleZero-trigger ultrafast charge-transfer J-aggregates via Se/π-directed assembly enable synchronous ROS/heat amplification for NIR-II photoimmunotherapy
Dandan
Ma†
a,
Hui
Bian†
 b,
Fei
Pan
b,
Fei
Pan
 c,
Danhong
Zhou
c,
Danhong
Zhou
 d,
Zhi
Chen
d,
Zhi
Chen
 e,
Haoying
Ge
d,
Yuanlang
Guo
a,
Yingnan
Wu
a,
Xin
He
e,
Haoying
Ge
d,
Yuanlang
Guo
a,
Yingnan
Wu
a,
Xin
He
 a,
Panwang
Zhou
a,
Panwang
Zhou
 f,
Lei
Wang
f,
Lei
Wang
 a,
Xiaoqiang
Chen
*a and
Xiaojun
Peng
a,
Xiaoqiang
Chen
*a and
Xiaojun
Peng
 *ad
*ad
aCollege of Materials Science and Engineering, Shenzhen University, Shenzhen 518060, P.R. China. E-mail: chenxq@szu.edu.cn; pengxj@dlut.edu.cn
bDepartment of Chemistry and Nanoscience, Ewha Womans University, 03760 Seoul, Republic of Korea
cInstitute of Apicultural Research, Chinese Academy of Ag-ricultural Sciences, Beijing 100093, P. R. China
dState Key Laboratory of Fine Chemicals, Frontiers Science Center for Smart Materials, Dalian University of Technology, Dalian, 116024, P.R. China
eCollege of Chemistry and Environmental Engineering, Shenzhen University, Shenzhen, Guangdong 518060, P.R. China
fState Key Laboratory of Catalysis, National Laboratory for Clean Energy, Dalian Institute of Chemical Physics, Chinese Academy of Sciences, Dalian, 116023, P.R. China
First published on 20th February 2026
Abstract
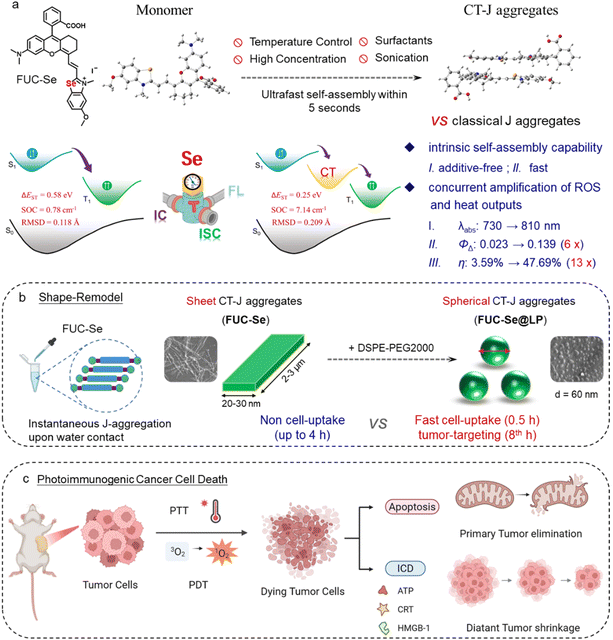
Supramolecular J-aggregation of π-conjugated photosensitizers provides a powerful strategy to optimize phototheranostic performance. However, integrating facile assembly with photodynamic and photothermal (PDT/PTT) synergy remains challenging. Herein, we report a charge transfer (CT)-state J-aggregation paradigm using a selenium-embedded molecular framework (FUC-Se). Driven by directional π–π stacking and Se⋯Se interactions, FUC-Se undergoes ultrafast (<5 s) self-assembly in water, forming stable CT-J aggregates without external triggers. Moreover, these aggregates enable concurrent amplification of reactive oxygen generation (ΦΔ = 0.139, 6 × monomer) and photothermal conversion (η = 47.69%, 13× monomer) through (i) reducing the singlet-triplet energy gap by 57% (ΔES1T1 = 0.25 eV vs. 0.58 eV) and enhancing spin–orbit coupling 9-fold (7.14 cm−1vs. 0.78 cm−1); (ii) promoting non-radiative decay with 77% surge in geometry reorganization (RMSDS1→S0 = 0.209 vs. 0.118) and 127% increase in the Huang–Rhys factor (λh = 0.25 eV vs. 0.11 eV). Phospholipid remodeling yields tumor-targeted FUC-Se@LP with hypoxia-tolerant ROS/PTT and NIR-II imaging. FUC-Se@LP achieves NIR-II-guided PDT/PTT, eradicating hypoxic tumors (IC50 = 2.47 µM) and suppressing metastasis via immunogenic cell death. This work establishes the first CT-J aggregate unifying exogenous-trigger-free ultrafast self-assembly, NIR-II imaging, and dual PDT/PTT enhancement, providing a promising strategy for precision oncology.
Introduction
Phototherapy, which spatiotemporally channels photon energy into cytotoxic signals, including reactive oxygen species (ROS) in photodynamic therapy (PDT) and localized hyperthermia in photothermal therapy (PTT), has significantly advanced oncotherapy.1–7 Despite significant anticancer advances, clinical translation remains hindered by intrinsic limitations of conventional π-conjugated photosensitizers (PSs), such as weak near-infrared absorption compromising tissue penetration, aggregation-caused quenching (ACQ) from uncontrolled stacking, and nonspecific biodistribution reducing tumor targeting efficiency.Supramolecular J-aggregation of π-conjugated PSs offers a promising strategy to modulate their optical properties and biodistribution profiles, endowing them with unique capabilities to address these challenges.8–16 Specifically, (i) redshifted absorption/emission via J-type exciton coupling enables deeper NIR photon penetration;17–23 (ii) enhanced extinction coefficients and exciton–exciton annihilation promote photothermal conversion;24–29 (iii) certain J-aggregate architectures may reduce singlet–triplet energy gaps (ΔEST), potentially enhancing intersystem crossing (ISC) for ROS generation;30–33 (iv) morphology-dependent biological behaviors (e.g., circulation time, cellular uptake, and organ distribution) can be tuned by stacking modes.34–36 Collectively, these advances position J-aggregation as a transformative platform to overcome the penetration–quenching–targeting trilemma of conventional PSs.
However, most existing J-aggregate phototheranostics rely on cumbersome assembly protocols, such as specific solvent,37–39 precise thermal40–43/ionic control,44–46 or carrier assistance47–56 for stable nanostructure formation. This complexity not only risks dissociation under physiological conditions (e.g., hemodilution-induced disassembly, thermal reversion at 37 °C, RES clearance) but also conflicts with clinical demands for simplified formulations. In this context, the concept of zero-trigger or exogenous-trigger-free assembly, where the aqueous environment itself serves as the sole thermodynamic driver without requiring any additional stimuli, becomes highly desirable yet remains largely unmet. Critically, most J-aggregates function exclusively as either PDT or PTT agents. Even the mere three reported J-aggregate systems demonstrating concurrent PDT/PTT effects fundamentally rely on exogenous stabilization methods, including solvent-triggered aggregation,57 sonication-induced crystallization,58 and surfactant-mediated phase transitions.37 Thus, the persistent failure of this field to achieve rapid, modulator-free aqueous J-aggregation with dual ROS/heat amplification constitutes a critical translational barrier.
Herein, we pioneer a selenium-directed charge-transfer J-aggregation paradigm through rational substitution of indole with selenazole in the well-established Changsha dye scaffold (Lin et al., 2012).59 The engineered FUC-Se intrinsically generates synergistic supramolecular forces, including cooperative π–π stacking (3.7 Å) and directional Se⋯Se contacts (4.0 Å), both of which contributed to drive exogenous-free, ultrafast (<5 s) J-aggregation in aqueous media. Crucially, unlike conventional J-aggregates limited to bathochromic shifts, the resulting CT-J architecture establishes interfacial charge-transfer states that reconfigure excited energy landscapes. This unique electronic reconstruction synergistically enhances intersystem crossing (ISC) while promoting vibronic thermalization, enabling concurrent amplification of singlet oxygen (ΦΔ = 0.139, 6 × monomer) and heat conversion (η = 47.69%, 13 × monomer) (Scheme 1a). Subsequent phospholipid remodeling further transforms the insoluble sheets into tumor-targeted nanospheres (FUC-Se@LP), which exhibit rapid cellular uptake (complete within 30 min), peak tumor accumulation at 8 h post-injection, dual hypoxia-tolerant ROS generation (Type I/II), and NIR-II imaging capability (Scheme 1b). Under NIR-II fluorescence guidance, FUC-Se@LP eradicates primary tumors while triggering immunogenic cell death via DAMP release, systemically inhibiting non-irradiated distant metastases (Scheme 1c). Overall, our J-aggregate system presents a promising strategy for solid tumor management by integrating three significant advantages, including facile and rapid assembly enabling off-the-shelf formulation, hypoxia-defying dual-mode therapy overcoming tumor resistance and phototherapy-initiated systemic immunity suppressing distant lesions. A quantitative comparison of key parameters between our system and other state-of-the-art J-aggregates60–62 is summarized in Table S1, highlighting the integrated advantages of our CT-J platform.
Results and discussion
Molecular design
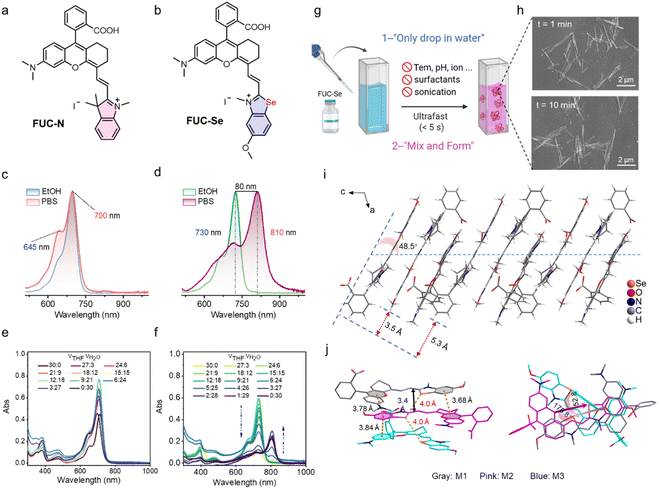
Hemicyanine dyes, characterized by a donor–π–acceptor (D–π–A) architecture, provide a versatile molecular platform for tailoring phototherapeutic and self-assembly properties through structural modularity.63–65 A landmark advancement came with Lin et al.’s rhodamine–hemicyanine hybrids (termed Changsha dyes CS1-6), comprising a rhodamine derivative and an indole/benzoindole segment connected by a vinyl bridge. This fusion synergizes the advantages of both parent dyes, NIR absorption/emission coupled with rhodamine-like fluorescence switching behavior, which cemented its status as a privileged and widely exploited framework in dye chemistry. Plenty of derivatization strategies, including carboxylic-acid-controlled spirocyclization, π-extension, and heterocycle replacement, have expanded this scaffold for bioimaging, sensing and diagnostics.66–72 Yet, despite these functional developments, the fundamental aggregation and assembly behavior of this hybrid skeleton remains largely unexplored. To transform this inert scaffold into a potent J-aggregation platform, we herein rationally replace indole with selenazole based on three design considerations: (i) Se's heavy-atom effect will boost ISC for ROS amplification;73,74 (ii) selenazole's intrinsic planar rigidity (vs. indole) will redshift absorption and facilitate ordered π-stacking; (iii) directional Se⋯Se interactions, well proven to facilitate interchain charge transfer in polymer photovoltaics,75–78 may induce and stabilize charge-separated states while acting as structural “zippers” to cooperatively guide π–π stacking toward ordered architectures. Thus, we speculate that such a transformation will simultaneously promote photosensitization and directional assembly arrangement. To validate this design, we synthesized FUC-N (indole-based control) and FUC-Se (selenazole-engineered analog). The detailed synthesis routes and structural characterization are shown in the SI (Fig. S21–S24).Optical properties
UV-Vis-NIR absorption spectra reveal that both dyes exhibit strong π–π* transitions with high extinction coefficients (>105 L mol−1 cm−1) in organic solvents (Fig. S1 and Table S2). And, FUC-Se displays a 30 nm redshift in absorption maxima (λabs = 730 nm) relative to FUC-N (λabs = 700 nm) due to larger planar rigidity of selenazole. Strikingly, they exhibited distinct spectral behaviors in PBS. FUC-N maintains its monomeric profile in PBS versus ethanol at concentrations up to 20 µM (Fig. 1c and S2a), whereas FUC-Se exhibits a pronounced red shift in water with λabs shifting from 730 to 810 nm, even at a dramatically lower concentration of 1 µM (Fig. 1d and S1–S2b). To exclude the effect of water polarity on the wavelength redshift, the absorption spectra in the THF/water binary system with varying water fractions (fw) were recorded. As shown in Fig. 1e, FUC-N exhibits only minor intensity fluctuations at 700 nm without peak displacement across all fw. Conversely, FUC-Se shows fw-dependent spectral evolution (Fig. 1f). Specifically, it maintained monomeric character (λabs = 730 nm) when fw < 80%, but exhibited a fixed 810 nm band at fw = 80%, which progressively intensified with increasing water (Fig. 1f). Moreover, similar spectral evolution was observed in EtOH/water and DMSO/water mixtures (Fig. S3), confirming that this aggregation behavior is independent of organic solvent identity. The combination of (i) fixed-wavelength redshift, (ii) threshold concentration dependence, and (iii) intensity growth of the new band provides solid evidence for J-aggregation of FUC-Se in water. Collectively, selenazole substitution successfully converts the inert scaffold into a J-aggregation platform, validating our molecular design.Notably, FUC-Se accomplishes spontaneous, ultra-fast and exogenous-trigger-free J-aggregation in aqueous media. This is rarely achieved in prior phototheranostic J-aggregates. Merely dispersing a 1 µM solution into water with brief pipette mixing (<5 s) immediately generates a characteristic J-aggregate absorption band at 810 nm (Fig. 1g and S2). Critically, this “mix-and-form” transition proceeds identically in both ultrapure water and PBS (Fig. 1d and S3) without any external triggers (temperature, pH, ultrasound, or surfactants). Scanning electron microscopy (SEM) confirms rapid nanostructure formation. The well-defined nanosheets with uniform dimensions (1–2 µm × 20–30 nm) emerge within 1 minute and maintain morphological integrity after 10 minutes (Fig. 1h). Atomic force microscopy (AFM) further confirmed the sheet-like morphology of the aggregates (Fig. S4). This combination of techniques confirms that barrierless molecular assembly generates macroscopic, two-dimensional architectures.
X-ray single-crystal structure analysis
The single-crystal structure unequivocally demonstrates that FUC-Se's remarkable J-aggregation stems from a hierarchically organized assembly process driven by π–π stacking and directional Se⋯Se bonding, orchestrated by precise molecular rotations (Fig. 1i–j and Tables S3–S4). In the asymmetric unit, there are three FUC-Se molecules (marked in gray, M1; pink, M2; and blue, M3). M2 undergoes a near-perfect 178° rotation relative to M1, enabling two face-to-face π–π interactions (3.68 Å and 3.78 Å). Concurrently, a directional Se⋯Se chalcogen bond (4.0 Å) bridges M1 and M2. This interaction acts as a “structural clamp”, rigidly fixing the 178° orientation and slip distance (3.5 Å). This corroborates our initial rationale that Se⋯Se contacts would act as “structural zippers” to cooperatively guide the assembly process. This M1–M2 dimer constitutes the fundamental excitonic unit, which is the primary structural basis for the large 80 nm J-band redshift (810 nm). M3 adopts a 58° rotation relative to M1, creating an asymmetric extension point. The tilted Se⋯Se bond enables the extension of the J-aggregate structure with controlled geometry. The cooperative interplay of Se⋯Se and π–π interactions, directed by specific molecular rotations, establishes the structural foundation for the rapid, exogenous-trigger-free J-aggregation of FUC-Se in aqueous media.Molecular dynamics simulation insights
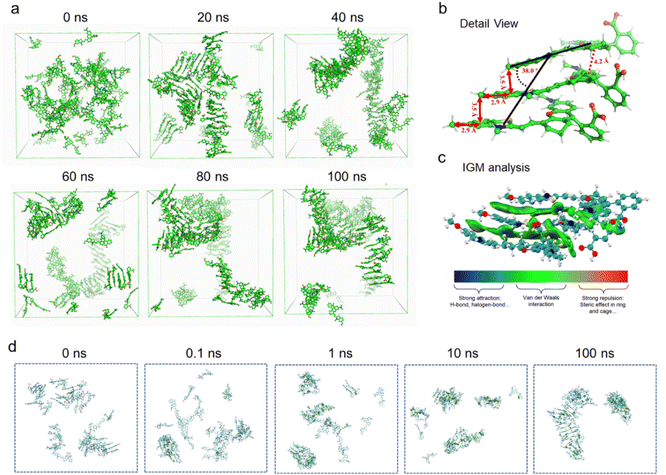
To further unravel the ultrafast assembly mechanism, molecular dynamics (MD) simulations were conducted using GROMACS 19.5 under periodic boundary conditions to model an infinite solution environment. As shown in Fig. 2a and Video S1, hydrophobic forces and directional Se⋯Se interactions initiate molecular clustering within 20 ns. Subsequent dominant π–π coupling drives rapid, oriented alignment, yielding stable J-aggregates by 100 ns with characteristic geometry, including 3.5 Å intermolecular spacing, 2.9 Å slide displacement, and slip angle 38.5° < 54.7° inclination (Fig. 2b). These parameters quantitatively match experimental crystal data (Fig. 1i and j). Complementary independent gradient model (IGM) analysis confirms π–π stacking (blue isosurfaces) as the primary driving force, with van der Waals (green) and hydrophobic interactions enabling long-range order (Fig. 2c and d). This synergy of chalcogen bonding initiation and π–π interaction rationalizes the sub-minute barrierless assembly observed experimentally.In vitro singlet oxygen and heat generation
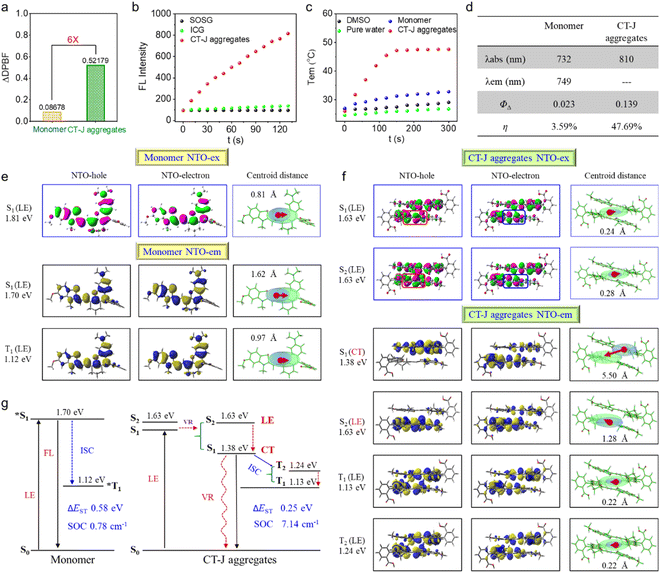
To quantify J-aggregation effects on photodynamic and photothermal performance, we compared singlet oxygen (1O2) generation and photothermal conversion between FUC-Se monomers and J-aggregates under strictly controlled identical conditions (solvent, absorption intensity, and irradiation parameters). Using DPBF as an 1O2 trap, J-aggregates induced sixfold greater DPBF degradation (Δabs = 0.52) than monomers within 7 min under 808 nm irradiation (Fig. 3a), a trend conserved across other solvent systems (Fig. S5). This significant enhancement was further validated using an alternative 1O2 detection method based on ABDA decomposition (Fig. S6), which yielded consistent results. Using ICG as a reference (ΦΔ = 0.008 in water),79,80 complementary SOSG assays further quantified ΦΔ of J-aggregates (0.139), 6× higher than that of monomers (ΦΔ = 0.023) (Fig. 3b, d and S7). Parallel evaluation of photothermal effects under 808 nm laser irradiation demonstrated that J-aggregates achieved rapid temperature elevation to 47.7 °C within 3 min (0.5 W cm−2, 5 min), contrasting with the negligible monomeric response (ΔT = 4.6 °C) (Fig. 3c and S8). Photothermal conversion efficiency (η), calculated via an established method,81 reached 47.69% for aggregates versus 3.59% for monomers (Fig. 3d and S9). Collectively, these data demonstrate that J-aggregation of FUC-Se simultaneously amplifies both photodynamic and photothermal efficacy.DFT calculation and natural transition orbital analysis
To decipher the photophysical origins of J aggregation-enhanced ROS and heat performance, we conducted systematic DFT calculations and NTO analysis. For monomeric FUC-Se, both excitation and emission processes involve conventional locally excited (LE) transitions, as evidenced by the compact electron–hole distribution (centroid distance < 1 Å) in NTOs (Fig. 3e, S9 and Tables S5–S6). This localized character results in a large S1–T1 energy gap (ΔEST = 0.58 eV) and weak spin–orbit coupling (SOC = 0.78 cm−1), thus limiting intersystem crossing efficiency. Strikingly, J-aggregation induces a paradigm shift in excited-state dynamics. Initial excitation generates degenerate S1/S2 states through HOMO → LUMO transitions (Fig. 3f, S10 and Tables S7–S8), which rapidly split into two distinct states: (1) a low-lying S1 state dominated by intermolecular charge-transfer excitation (CTE) with HOMO → LUMO transition across adjacent molecules (electron–hole separation, 5.5 Å), and (2) an S2 state retaining intramolecular LE character via HOMO-1 → LUMO transition. Notably, the formation of this intermolecular CT state fundamentally distinguishes the photophysics of FUC-Se J-aggregates from those driven solely by exciton delocalization in conventional J-aggregates. While exciton delocalization typically leads to bathochromic shifts and altered radiative rates, the key feature here is the spatially separated electron–hole distribution across molecular units. This CT character directly reduces ΔEST by 43% (0.25 eV vs. 0.58 eV) and enhances SOC nearly tenfold (7.14 cm−1vs. 0.78 cm−1), which are not inherently provided by excitonic coupling alone (Fig. 3g). Both synergistically promote ISC, accounting for the observed sixfold increase in 1O2 quantum yield, which is consistent with the El-Sayed rule:82,83| kISC∝∣〈S∣HSOC∣T〉∣2/ΔE2 |
Moreover, the CT-J architecture concurrently promotes efficient non-radiative decay, establishing a stronger photothermal pathway compared to the monomer. Firstly, the CT-J aggregates exhibit a significantly larger root-mean-square deviation (RMSD = 0.209 Å) between S0 and S1 geometries than the monomer (RMSD = 0.118 Å). This clearly suggested pronounced structural distortion in the aggregated state upon photoexcitation (Fig. S10 and S11). This distortion would enhance vibrational energy release during S1 → S0 relaxation, directly contributing to heat generation. Secondly, the reorganization energy of the CT-J aggregates (λh = 0.25 eV) is more than double that of the monomer (λh = 0.11 eV), demonstrating a stronger propensity for non-radiative decay over radiative transitions. Correspondingly, the kr/knr ratio of CT-J aggregates is drastically reduced to 0.003, far lower than that of the monomer (0.111). Additionally, the CTE state's low energy position (1.38 eV vs. 1.70 eV in monomer) creates a thermodynamic sink for rapid internal conversion, favoring absorbed photons into lattice vibrations.84
Ultrafast excited-state dynamics behaviors
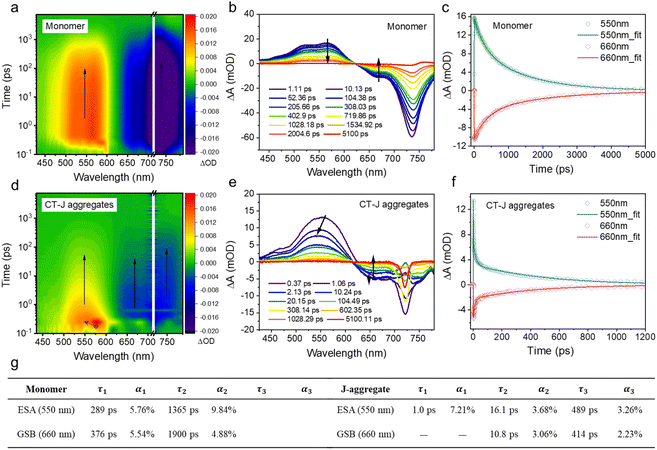
Following closely behind, femtosecond transient absorption spectroscopy further unraveled distinct excited-state dynamics between monomers and J-aggregates. For monomers, symmetric ground-state bleaching (GSB) (ΔA < 0 at 660 nm) and excited-state absorption (ESA) (ΔA > 0 at 550 nm) signals exhibited identical decay kinetics with matched lifetimes (τ1 ≈ 300 ps, τ2 ≈ 1.5 ns), indicative of direct S1 → S0 relaxation without intermediate states (Fig. 4a–c). In stark contrast, J-aggregates displayed a three-component ESA decay profile dominated by an ultrafast process (τ1 = 1.0 ps, α1 = 7.21%), absent in GSB dynamics (Fig. 4d–f). This unambiguously signals the formation of a charge-transfer intermediate state (CT) with near-unity yield (>95%), which is absent in the monomer and not a typical feature of excitonic J-aggregates, providing direct time-resolved evidence for a unique relaxation funnel induced by Se/π-directed assembly. This allows us to decouple the CT-mediated processes from generic aggregation effects. The CT state displays a biphasic relaxation: (i) initial vibrational cooling within 16.1 ps (τ2), followed by (ii) non-radiative decay to the ground state over 489 ps (τ3). The latter component's lifetime aligns with the enhanced SOC constant (7.14 cm−1) and reduced S1–T1 gap (0.25 eV), synergistically promoting ISC-mediated 1O2 generation. Concurrently, the ultrafast CT formation (1.0 ps) and vibronic coupling (τ2 = 16.1 ps) channel more absorbed energy into lattice vibrations, as evidenced by the negligible fluorescence quantum yield (Fig. 4g). Overall, the ultrafast CT formation and its subsequent relaxation (vibrational cooling and ISC-competent decay) specifically channel energy into the enhanced ROS and heat production observed, rather than through exciton delocalization pathways that would primarily affect radiative properties.Preparation and characterization of morphology-reshaped CT-J aggregates (FUC-Se@LP)
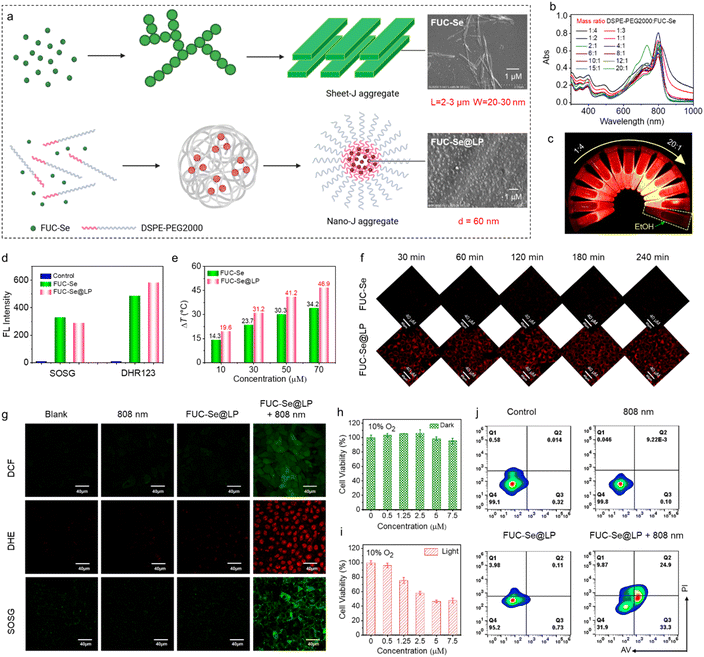
To address the incompatibility of micron-scale FUC-Se J-aggregates (sheet-like structures) in biological systems, we further restructured them into spherical nanoparticles using DSPE-PEG2K via a thin-film dispersion method (Fig. 5a). Systematic evaluation of DSPE-PEG2K/FUC-Se mass ratios (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 4–20
4–20![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) revealed distinct phase behaviors (Fig. 5b, c and Tables S9–S10). In detail, low ratios (1
1) revealed distinct phase behaviors (Fig. 5b, c and Tables S9–S10). In detail, low ratios (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 4–1
4–1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3) preserved J-aggregate absorption at ∼800 nm but retained oversized particles (>2000 nm); intermediate ratios (1
3) preserved J-aggregate absorption at ∼800 nm but retained oversized particles (>2000 nm); intermediate ratios (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2–2
2–2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) reduced size to >500 nm but still unsuitable for the EPR effect; high ratios (8
1) reduced size to >500 nm but still unsuitable for the EPR effect; high ratios (8![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1–20
1–20![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) induced obvious dissociation of J-aggregates. Optimal ratios (4
1) induced obvious dissociation of J-aggregates. Optimal ratios (4![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1–8
1–8![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) balanced J-aggregate integrity (∼800 nm) with a hydrodynamic diameter of ∼100 nm. NIR-II fluorescence analysis further showed enhanced emission at intermediate ratios (1
1) balanced J-aggregate integrity (∼800 nm) with a hydrodynamic diameter of ∼100 nm. NIR-II fluorescence analysis further showed enhanced emission at intermediate ratios (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2–8
2–8![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1), peaking at 8
1), peaking at 8![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 due to partial monomer dissociation. Finally, the 6
1 due to partial monomer dissociation. Finally, the 6![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 formulation emerged as ideal, exhibiting ∼80 nm hydrodynamic diameter (DLS), ∼60 nm SEM-measured size, and strong J-aggregate fluorescence, thereby enabling cellular uptake and EPR-driven tumor targeting. And, the preservation of the distinct spectral signature suggests that the core electronic structure of the CT-J aggregate remains intact after morphological reshaping. Furthermore, the optimized FUC-Se@LP formulation (6
1 formulation emerged as ideal, exhibiting ∼80 nm hydrodynamic diameter (DLS), ∼60 nm SEM-measured size, and strong J-aggregate fluorescence, thereby enabling cellular uptake and EPR-driven tumor targeting. And, the preservation of the distinct spectral signature suggests that the core electronic structure of the CT-J aggregate remains intact after morphological reshaping. Furthermore, the optimized FUC-Se@LP formulation (6![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 mass ratio) exhibited excellent colloidal stability, with negligible changes in hydrodynamic diameter and zeta potential over 8 days in PBS at 4 °C (Tables S9 and S10), meeting the prerequisite for in vivo applications. Notably, the FUC-Se@LP nanoparticles demonstrated stability in a physiologically relevant environment. The characteristic J-aggregate absorption at ∼800 nm remained intact after incubation in PBS and RMMI 1640 containing 10% fetal bovine serum at 37 °C for up to 24 hours (Fig. S12), confirming the robustness of the CT-J core against dissociation in media simulating in vivo conditions. In addition to spectral characterization, molecular dynamics simulations further visualized the DSPE-PEG-assisted aggregation and assembly process of FUC-Se, which still retained the characteristics of J-aggregation in nanoparticles (Video S2 and Fig. S13). The simulations explicitly show that the directional π–π stacking and Se⋯Se interactions, key to forming the CT state, are maintained within the nanoparticle core during the encapsulation process. This optimized FUC-Se@LP nanoparticle system was selected for subsequent biological studies.
1 mass ratio) exhibited excellent colloidal stability, with negligible changes in hydrodynamic diameter and zeta potential over 8 days in PBS at 4 °C (Tables S9 and S10), meeting the prerequisite for in vivo applications. Notably, the FUC-Se@LP nanoparticles demonstrated stability in a physiologically relevant environment. The characteristic J-aggregate absorption at ∼800 nm remained intact after incubation in PBS and RMMI 1640 containing 10% fetal bovine serum at 37 °C for up to 24 hours (Fig. S12), confirming the robustness of the CT-J core against dissociation in media simulating in vivo conditions. In addition to spectral characterization, molecular dynamics simulations further visualized the DSPE-PEG-assisted aggregation and assembly process of FUC-Se, which still retained the characteristics of J-aggregation in nanoparticles (Video S2 and Fig. S13). The simulations explicitly show that the directional π–π stacking and Se⋯Se interactions, key to forming the CT state, are maintained within the nanoparticle core during the encapsulation process. This optimized FUC-Se@LP nanoparticle system was selected for subsequent biological studies.
In vitro ROS generation and photothermal performance of FUC-Se@LP
Before evaluating the anticancer potential of FUC-Se@LP, we first examined whether morphological reshaping affected its ROS generation and photothermal properties. SOSG and DHR123 were used to detect 1O2 and superoxide anions (O2−˙), respectively. As shown in Fig. 5d and S14, FUC-Se (sheet J-aggregates) and FUC-Se@LP (spherical nanoparticles) showed comparable enhancement in either SOSG or DHR123 under identical conditions, suggesting that morphological remodeling has negligible impact on ROS generation. ESR spin-trapping experiments using DMPO (for O2−˙) and TEMP (for 1O2) confirmed the concurrent generation of both radical and non-radical ROS species upon irradiation (Fig. S15). Next, we further assessed their photothermal performance. As shown in Fig. 5e and S16, FUC-Se@LP exhibited significantly higher temperature elevations than FUC-Se across various concentrations and laser power densities. The photothermal conversion efficiency of FUC-Se@LP reached 52.6%, outperforming the 16% efficiency of ICG. Subsequently, photostability was further evaluated using ICG as a reference (Fig. S17). Within five minutes, ICG's absorption dropped below 0.1, indicating near complete degradation. FUC-Se retained absorbance above 0.3 at five minutes but declined to near zero after ten minutes. In contrast, FUC-Se@LP maintained absorbance above 0.45 throughout. Overall, compared to sheet J-aggregates (FUC-Se), FUC-Se@LP not only preserves comparable ROS generation capacity but also exhibits superior photostability and enhanced photothermal performance.Enhanced cellular uptake and mitochondrial targeting
Critically, the reshaped J-aggregates FUC-Se@LP exhibited significantly accelerated cellular internalization kinetics. Real-time confocal imaging revealed rapid intracellular accumulation of FUC-Se@LP, with intense red fluorescence saturation achieved within 30 minutes of treatment (Fig. 5f). This uptake kinetics starkly contrasted with negligible fluorescence in cells treated with non-engineered FUC-Se aggregates. Fluorescence imaging further demonstrated exceptional subcellular targeting precision. As shown in Fig. S18, FUC-Se@LP showed strong colocalization with MitoTracker TMRM (Pearson's coefficient = 0.85), confirming mitochondria-specific localization. This dual advantage of accelerated cellular uptake and organelle-level spatial precision creates favorable conditions for phototherapy, as mitochondrial targeting maximizes ROS-induced oxidative damage while minimizing off-target effects.85,86Dual-mode ROS generation and phototoxicity
Previous in vitro studies further established FUC-Se@LP as a dual-mode photosensitizer capable of simultaneously generating singlet oxygen and superoxide anions under 808 nm irradiation. To validate its cellular performance, we employed three fluorescent probes, including DCFH-DA for pan-ROS detection,7,80,87 SOSG for 1O2-specific tracking, and DHE for O2−˙ monitoring. As shown in Fig. 5g, neither FUC-Se@LP alone nor laser irradiation independently induced significant intracellular ROS production. However, laser-activated FUC-Se@LP triggered a synergistic ROS cascade, evidenced by concurrent fluorescence intensification across all probes, confirming efficient dual ROS generation. Crucially, this cooperative mechanism maintains ROS production efficiency under mild hypoxia, relieving the oxygen-dependent limitation of conventional photodynamic therapy. The cytotoxic effects correlated directly with ROS generation profiles. FUC-Se@LP exhibited exceptional biocompatibility under dark conditions, preserving >90% 4T1 cell viability even at 7.5 µM (Fig. 5h). Upon laser activation, it demonstrated concentration-dependent phototoxicity (10% O2, IC50 = 2.47 µM) (Fig. 5i). Importantly, this potent efficacy was retained even under severe hypoxia (2% O2), with a comparable IC50 value (Fig. S19). This robust hypoxia tolerance underscores the advantage of the CT-J aggregate-enabled dual-mode ROS mechanism, where the oxygen-independent Type-I pathway remains highly operative under oxygen-deficient conditions.To elucidate the mechanism of FUC-Se@LP-induced cell death, AV-FITC/PI staining was performed (Fig. 5j). Neither FUC-Se@LP alone nor 808 nm laser irradiation independently triggered apoptosis or necrosis (apoptotic rate < 5%). However, their combined application markedly elevated apoptotic cells to 58.2%. This apoptosis-dominant response aligns with the mitochondrial targeting specificity observed in prior mitochondrial colocalization experiments, indicating that localized ROS generation at mitochondrial sites preferentially activates intrinsic apoptotic pathways.80 These findings collectively position FUC-Se@LP as a hypoxia-tolerant phototherapeutic agent that synergizes dual ROS generation, mitochondrial precision targeting, and apoptosis-dominated cytotoxicity for solid tumor treatment.
In vivo distribution and anti-subcutaneous tumor treatment of FUC-Se@LP
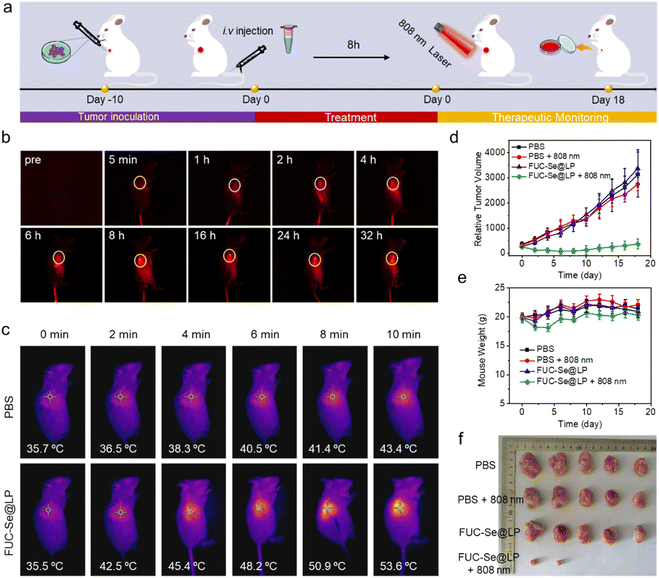
Prior to evaluating therapeutic efficacy, we first characterized the tumor-targeting capability of FUC-Se@LP through real-time NIR-II fluorescence imaging. As shown in Fig. 6a and b, following intravenous administration, time-lapsed imaging revealed progressive tumor accumulation commencing at 2 h post-injection, peaking at 8 h, and maintaining sustained retention through 24 h. This spatiotemporal profiling established the optimal therapeutic window at 8 h post-injection when maximum tumor accumulation was achieved. Capitalizing on that, we conducted phototherapy in 4T1 tumor-bearing mice under 808 nm irradiation (0.8 W cm−2). As shown in Fig. 6c, while PBS-injected controls exhibited a slight temperature increase, FUC-Se@LP-mediated photoconversion induced rapid tumor hyperthermia, reaching 46.6 °C within 4 min and 55.5 °C at 10 min, surpassing the critical threshold for protein denaturation. This localized thermal ablation translated to significant tumor regression. Phototherapy-treated tumors demonstrated significant volume reduction by Day 2 post-treatment, with complete eradication observed in 60% of subjects (3/5 mice) and only minimal residual tumors in the remaining cases (Fig. 6d and f). In stark contrast, control groups exhibited uninterrupted tumor progression. Crucially, all treatment groups maintained stable body weights throughout the observation period (Fig. 6e), confirming systemic biocompatibility.Immunogenic cell death and anti-distant tumor effects triggered by FUC-Se@LP
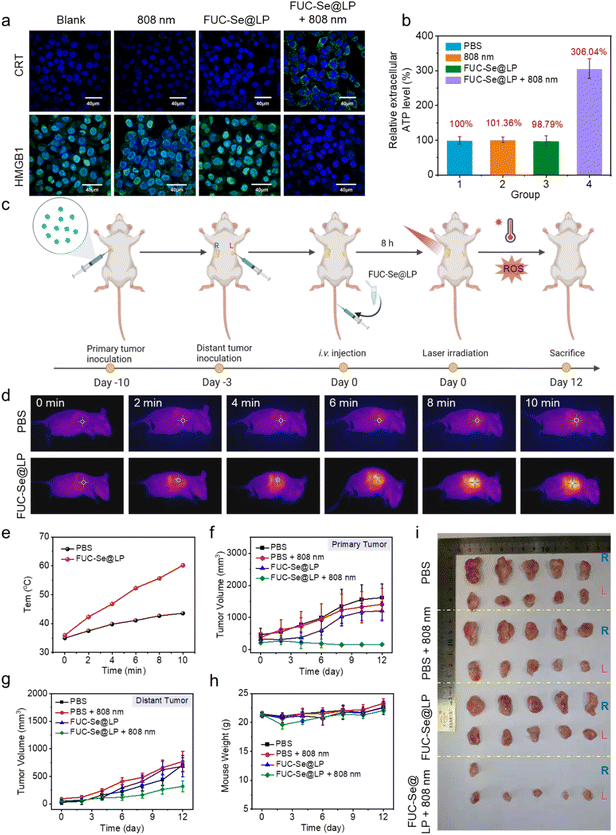
Beyond direct tumor ablation, FUC-Se@LP-mediated phototherapy demonstrates profound immunomodulatory effects through light-triggered immunogenic cell death (ICD).87–91 As a hallmark of ICD, the cascade release of damage-associated molecular patterns (DAMPs), including calreticulin (CRT) exposure, ATP secretion, and HMGB1 translocation, was systematically validated (Fig. 7). Confocal microscopy revealed intense CRT surface translocation exclusively in the FUC-Se@LP + laser group, compared to negligible signals in controls. This “eat me” signal primes antigen-presenting cells (APCs) for phagocytic activation, bridging innate and adaptive immunity. Concomitantly, HMGB1 (a nuclear DNA chaperone) underwent complete cytoplasmic relocalization and extracellular release in the treatment group, evidenced by the disappearance of nuclear green fluorescence (Fig. 7a). This spatial redistribution converts HMGB1 into a pro-inflammatory cytokine that activates Toll-like receptor 4 (TLR4) on dendritic cells, driving their maturation. The ICD cascade culminated 3-fold increase in ATP efflux (Fig. 7b), establishing a chemoattractant gradient for immune cell recruitment. These results indicate that FUC-Se@LP can effectively enhance the immunogenicity of 4T1 cells. Crucially, this immunogenic potential is strictly light-gated: FUC-Se@LP alone showed no DAMP release, ensuring spatiotemporal control over immune activation.The therapeutic implications of this ICD induction were validated in a bilateral 4T1 tumor model, where primary (right flank) and distant (left axilla) tumors were sequentially inoculated (Fig. 7c). Notably, temperature monitoring confirmed precise photothermal control: primary tumors reached ∼60 °C (sufficient for cell death) versus ∼40 °C in laser-only controls (Fig. 7d and e). Localized treatment of primary tumors (0.8 W cm−2 for 10 min) achieved complete ablation in 80% of subjects (4/5 mice) while suppressing distant tumor growth by 58% versus PBS controls (Fig. 7f–g and i). Systemic safety was concurrently demonstrated through stable body weight trajectories (Fig. 7h) and histologically confirmed organ integrity, with no hepatic inflammation, renal fibrosis, or pulmonary lesions observed (Fig. S20), demonstrating that the remodeled J-aggregate FUC-Se@LP exhibits excellent biocompatibility. These multimodal therapeutic advantages, combined with NIR-II imaging-guided treatment personalization, position FUC-Se@LP as a transformative candidate for clinical translation, particularly for treating hypoxic, immunotherapy-resistant malignancies.
Conclusions
In this study, we successfully developed a charge-transfer state J-aggregate photosensitizer (FUC-Se). By leveraging a unique CT-J aggregation mechanism, these aggregates exhibit a bathochromic shift of 80 nm (λabs = 810 nm) and reconfigured energy landscapes. This restructuring reduces the singlet–triplet gap to 0.25 eV (vs. 0.58 eV in monomers) and amplifies spin–orbit coupling to 7.14 cm−1 (9.2-fold enhancement). Concurrently, it also promotes a 77% surge in geometry reorganization (RMSDS1→S0 = 0.209 vs. 0.118) and a 127% increase in the Huang–Rhys factor (λh = 0.25 eV vs. 0.11 eV). Consequently, the ISC efficiency and vibronic heat conversion are significantly boosted, with a ΦΔ of 0.139 (a 6-fold increase over the monomer) and an η of 47.69% (a 13-fold improvement). Notably, FUC-Se achieves spontaneous aggregation at ultralow concentrations without requiring exogenous triggers. Subsequent phospholipid remodeling yields tumor-targeted nanospheres (FUC-Se@LP) capable of hypoxia-insensitive PDT/PTT and NIR-II fluorescence-guided imaging. In vivo, FUC-Se@LP eradicates primary tumors via ROS/thermal synergy (IC50 = 2.47 µM) and suppresses distant lesions by 58% through the induction of immunogenic death, all while demonstrating excellent biosafety. This work establishes CT-J aggregation as a new design strategy, offering potential for improved clinical metastasis management.Ethical statement
All the animal experiments were performed in accordance with the Guide for the Care and Use of Laboratory Animals published by the National Institutes of Health. The animal protocol was approved by the local research ethics review board of the Animal Ethics Committee of Dalian University of Technology (Certificate number//Ethics approval no. is 2018-043).Author contributions
D. M., H. B., X. C., and X. P. conceived and designed the experiments for this project. D. M. and H. B. performed the synthetic work, carried out the characterization of the compounds, and conducted spectroscopic measurements, other in vitro assays, cellular assays, and mouse experiments. F. P. was responsible for the molecular dynamics simulations, while D. Z. and P. Z. conducted the theoretical calculations. Z. C. and X. H. performed the crystal structure analysis. H. G., Y. G., and Y. W. assisted in the measurement and analysis of selected spectroscopic data. L. W. contributed to the data analysis and manuscript revision. X. C. and X. P. supervised the work.Conflicts of interest
The authors declare no competing financial interests.Data availability
CCDC 2431831 contains the supplementary crystallographic data for this paper.92The data supporting this article have been included as part of the supplementary information (SI). Supplementary information is available. See DOI: https://doi.org/10.1039/d5sc08871b.
Acknowledgements
This work was supported by the Major Program of the National Natural Science Foundation of China (Grant No. 22090010 and 22090011), the GuangDong Basic and Applied Basic Research Foundation (Grant No. 2023B1515120001), and the Shenzhen University 2035 Program for Excellent Research (Grant No. 00000208 and 00000225). D. M. acknowledges the support from the China Postdoctoral Science Foundation (Grant No. 2024M752118) and the Young Scientists Fund of the National Natural Science Foundation of China (Grant No. 22408232).Notes and references
- D. Ma, H. Bian, M. Gu, L. Wang, X. Chen and X. Peng, Recent advances in the design and applications of near-infrared II responsive small molecule phototherapeutic agents, Coord. Chem. Rev., 2024, 505, 215677 CrossRef CAS.
- X. Zhao, J. Du, W. Sun, J. Fan and X. Peng, Regulating Charge Transfer in Cyanine Dyes: A Universal Methodology for Enhancing Cancer Phototherapeutic Efficacy, Acc. Chem. Res., 2024, 57(17), 2582–2593 CrossRef CAS PubMed.
- X. Zhao, J. Liu, J. Fan, H. Chao and X. Peng, Recent progress in photosensitizers for overcoming the challenges of photodynamic therapy: from molecular design to application, Chem. Soc. Rev., 2021, 50(6), 4185–4219 RSC.
- H. Bian, D. Ma, F. Pan, X. Zhang, K. Xin, X. Zhang, Y. Yang, X. Peng and Y. Xiao, Cardiolipin-Targeted NIR-II Fluorophore Causes “Avalanche Effects” for Re-Engaging Cancer Apoptosis and Inhibiting Metastasis, J. Am. Chem. Soc., 2022, 144(49), 22562–22573 CrossRef CAS PubMed.
- X. Kang, Z. Du, S. Yang, M. Liang, Q. Liu and J. Qi, Smart molecular probes with controllable photophysical property for smart medicine, Smart Mol., 2024, 2(3), e20240033 CrossRef CAS PubMed.
- H. Kim, Y. Lee, H. Jeong, J. Lee, X. Wu, H. Li and J. Yoon, Photodynamic and photothermal therapies for bacterial infection treatment, Smart Mol., 2023, 1(1), e20220010 CrossRef.
- H. Gu, W. Sun, J. Du, J. Fan and X. Peng, Dual-acceptor engineering of donor-acceptor type molecules for all-round boosting anti-tumor phototherapy, Smart Mol., 2023, 2(2), e20230014 CrossRef PubMed.
- Y. Dai, J. Sun, X. Zhang, J. Zhao, W. Yang, J. Zhou, Z. Gao, Q. Wang, F. Yu and B. Wang, Supramolecular assembly boosting the phototherapy performances of BODIPYs, Coord. Chem. Rev., 2024, 517, 216054 CrossRef CAS.
- N. Dar and R. Ankari, Theoretical Models, Preparation, Characterization and Applications of Cyanine J-Aggregates: A Minireview, ChemistryOpen, 2022, 11(11), e20220010 CrossRef.
- X. Hu, C. Zhu, F. Sun, Z. Chen, J. Zou, X. Chen and Z. Yang, J-Aggregation Strategy toward Potentiated NIR-II Fluorescence Bioimaging of Molecular Fluorophores, Adv. Mater., 2023, 36(1), 2304848 CrossRef PubMed.
- J. H. Kim, T. Schembri, D. Bialas, M. Stolte and F. Würthner, Slip-Stacked J-Aggregate Materials for Organic Solar Cells and Photodetectors, Adv. Mater., 2021, 34(22), 2104678 CrossRef PubMed.
- P. Kumari, S. Arora, Y. Pan, I. Ahmed, S. Kumar and B. Parshad, Tailoring Indocyanine Green J-Aggregates for Imaging, Cancer Phototherapy, and Drug Delivery: A Review, ACS Appl. Bio Mater., 2024, 7(8), 5121–5135 CrossRef CAS PubMed.
- G. Sun, Y. Zhang, N. Zhou, F. Huo and C. Yin, Recent advances in the construction, imaging and phototherapy applications of NIR-II J-aggregates based on different fluorophores, Dyes Pigm., 2025, 243, 113054 CrossRef CAS.
- Y. Tian, D. Yin and L. Yan, J-aggregation strategy of organic dyes for near-infrared bioimaging and fluorescent image-guided phototherapy, Wiley Nanomed. Nanobiotechnology, 2022, 15(1), e1831 CrossRef PubMed.
- J. Xu, X. Zheng, T. Ren, L. Shi, X. Yin, L. Yuan and X. Zhang, Recent advances in near-infrared-II organic J-aggregates for bio-applications, Coord. Chem. Rev., 2025, 528, 216379 CrossRef CAS.
- S. Xu, H. Liu, S. Huan, L. Yuan and X. Zhang, Recent progress in utilizing near-infrared J-aggregates for imaging and cancer therapy, Mater. Chem. Front., 2021, 5(3), 1076–1089 RSC.
- K. Li, X. Duan, Z. Jiang, D. Ding, Y. Chen, G. Zhang and Z. Liu, J-aggregates of meso-[2.2]paracyclophanyl-BODIPY dye for NIR-II imaging, Nat. Commun., 2021, 12(1), 2376 Search PubMed.
- C. Sun, B. Li, M. Zhao, S. Wang, Z. Lei, L. Lu, H. Zhang, L. Feng, C. Dou, D. Yin, H. Xu, Y. Cheng and F. Zhang, J-Aggregates of Cyanine Dye for NIR-II in Vivo Dynamic Vascular Imaging beyond 1500 nm, J. Am. Chem. Soc., 2019, 141(49), 19221–19225 CrossRef CAS.
- C. Sun, X. Sun, P. Pei, H. He, J. Ming, X. Liu, M. Liu, Y. Zhang, Y. Xia, D. Zhao, X. Li, Y. Xie and F. Zhang, NIR-II J-Aggregates Labelled Mesoporous Implant for Imaging-Guided Osteosynthesis with Minimal Invasion, Adv. Funct. Mater., 2021, 31(23), 2100656 CrossRef CAS.
- S. Usama, D. Caldwell, P. Shrestha, M. Luciano, N. Patel, J. Kalen, J. Ivanic and M. Schnermann, Modified norcyanines enable ratiometric pH imaging beyond 1000 nm, Biosens. Bioelectron., 2022, 217, 114610 CrossRef CAS PubMed.
- C. Zhang, Y. Wu, F. Zeng, Y. Wen, J. Chen, G. Deng, L. Zhang, S. Zhao, S. Wu and Y. Zhao, Structurally Modulated Formation of Cyanine J-Aggregates with Sharp and Tunable Spectra for Multiplexed Optoacoustic and Fluorescence Bioimaging, Angew. Chem., Int. Ed., 2024, 63(34), e202406694 CrossRef CAS.
- Q. Zhang, P. Yu, Y. Fan, C. Sun, H. He, X. Liu, L. Lu, M. Zhao, H. Zhang and F. Zhang, Bright and Stable NIR-II J-Aggregated AIE Dibodipy-Based Fluorescent Probe for Dynamic in Vivo Bioimaging, Angew. Chem., Int. Ed., 2020, 60(8), 3967–3973 CrossRef PubMed.
- Y. Zhu, P. Wu, S. Liu, J. Yang, F. Wu, W. Cao, Y. Yang, B. Zheng and H. Xiong, Electron-Withdrawing Substituents Allow Boosted NIR-II Fluorescence in J-Type Aggregates for Bioimaging and Information Encryption, Angew. Chem., Int. Ed., 2023, 62(47), e202313166 CrossRef CAS PubMed.
- X. Bao, S. Zheng, L. Zhang, A. Shen, G. Zhang, S. Liu and J. Hu, Nitric-Oxide-Releasing aza-BODIPY: A New Near-Infrared J-Aggregate with Multiple Antibacterial Modalities, Angew. Chem., Int. Ed., 2022, 61(32), e202207250 CrossRef CAS PubMed.
- D. Chen, Y. Xu, Y. Wang, C. Teng, X. Li, D. Yin and L. Yan, J-aggregates of strong electron-donating groups linked Aza-BODIPY adjusting by polypeptide for NIR-II phototheranostics, Spectrochim. Acta, Part A, 2024, 322, 124789 CrossRef CAS PubMed.
- C. Ou, Z. Zhao, L. An, L. Zheng, F. Gao, Q. Zhu, W. Wang, J. Shao, L. Xie and X. Dong, J-Aggregate Promoting NIR-II Emission for Fluorescence/Photoacoustic Imaging-Guided Phototherapy, Adv. Healthcare Mater., 2024, 13(23), 2400846 CrossRef CAS PubMed.
- F. Tang, H. Yu, Y. Huang, X. Zhao, Z. Chen, H. Ma, B. Zheng, M. Ke, Y. Zhang, X. Li, J. Yoon and J. Huang, Phthalocyanine J-aggregate nanoparticles with enormous-redshifted and intense absorption: Potential structure-J-aggregation relationships and application in tumor-associated macrophages-targeted phototheranostics, Chem. Eng. J., 2024, 496, 154272 CrossRef CAS.
- Y. Wan, Y. Gao, W. Wei, K. Lee, J. Tan, C. Chen, H. Chen, S. Li, K. Wong and C. Lee, Facilely Achieving Near-Infrared-II J-Aggregates through Molecular Bending on a Donor-Acceptor Fluorophore for High-Performance Tumor Phototheranostics, ACS Nano, 2024, 18(41), 27949–27961 CrossRef CAS PubMed.
- Y. Xu, X. Meng, Y. Zhao, M. Jia, H. Zhu, J. Song, Y. Su, W. Qiao, J. Qi and Z. Y. Wang, Pyrrolopyrrole Cyanine J-Aggregate Nanoparticles with High Near-Infrared Fluorescence Brightness and Photothermal Performance for Efficient Phototheranostics, ACS Appl. Mater. Interfaces, 2024, 16(30), 39005–39020 CrossRef CAS PubMed.
- Y. Kang, W. Chen, K. Teng, L. Wang, X. Xu, L. Niu, G. Cui and Q. Yang, Aggregation Turns BODIPY Fluorophores into Photosensitizers: Reversibly Switching Intersystem Crossing On and Off for Smart Photodynamic Therapy, CCS Chem., 2022, 4(11), 3516–3528 CrossRef CAS.
- Y. Li, T. Ma, H. Jiang, W. Li, D. Tian, J. Zhu and Z. Li, Anionic Cyanine J-Type Aggregate Nanoparticles with Enhanced Photosensitization for Mitochondria-Targeting Tumor Phototherapy, Angew. Chem., Int. Ed., 2022, 61(24), e202203093 CrossRef CAS PubMed.
- Y. Liu, G. Liu, W. Zhou, G. Feng, Q. Ma, Y. Zhang and G. Xing, In Situ Self-Assembled J-Aggregate Nanofibers of Glycosylated Aza-BODIPY for Synergetic Cell Membrane Disruption and Type I Photodynamic Therapy, Angew. Chem., Int. Ed., 2023, 62(40), e202309786 CrossRef CAS PubMed.
- J. Xue, Q. Liang, R. Wang, J. Hou, W. Li, Q. Peng, Z. Shuai and J. Qiao, Highly Efficient Thermally Activated Delayed Fluorescence via J-Aggregates with Strong Intermolecular Charge Transfer, Adv. Mater., 2019, 31(28), 1808242 CrossRef PubMed.
- S. Li, X. Yan, J. Zhang, X. Guo, Y. Zhang, M. Su, C. Yang, H. Zhang and C. Li, J-Aggregation-Driven Supramolecular Assembly of Dye-Conjugated Block Polymers: From Morphological Tailoring to Anticancer Applications, Adv. Funct. Mater., 2021, 31(51), 2105189 CrossRef CAS.
- M. Su, Q. Han, X. Yan, Y. Liu, P. Luo, W. Zhai, Q. Zhang, L. Li and C. Li, A Supramolecular Strategy to Engineering a Non-photobleaching and Near-Infrared Absorbing Nano-J-Aggregate for Efficient Photothermal Therapy, ACS Nano, 2021, 15(3), 5032–5042 CrossRef CAS PubMed.
- Y. Tian, D. Yin, Q. Cheng, H. Dang, C. Teng and L. Yan, Supramolecular J-aggregates of aza-BODIPY by steric and π–π nteractions for NIR-II phototheranostics, J. Mater. Chem. B, 2022, 10(10), 1650–1662 RSC.
- X. Guo, W. Sheng, H. Pan, L. Guo, H. Zuo, Z. Wu, S. Ling, X. Jiang, Z. Chen, L. Jiao and E. Hao, Tuning Shortwave-Infrared J-aggregates of Aromatic Ring-Fused Aza-BODIPYs by Peripheral Substituents for Combined Photothermal and Photodynamic Therapies at Ultralow Laser Power, Angew. Chem., Int. Ed., 2024, 63(11), e202319875 Search PubMed.
- A. Sarbu, L. Biniek, J. Guenet, P. Mésini and M. Brinkmann, Reversible J- to H-aggregate transformation in thin films of a perylenebisimide organogelator, J. Mater. Chem. C, 2015, 3(6), 1235–1242 RSC.
- C. Shen and F. Würthner, NIR-emitting squaraine J-aggregate nanosheets, Chem. Commun., 2020, 56(68), 9878–9881 RSC.
- L. Dong, X. Shi, H. Li, Z. Liu, K. Teng, L. Niu, Z. Hu and Q. Yang, Light-Facilitated Reassembly to Generate J-Aggregates of Glucosyl aza-BODIPY with Second Near-Infrared Emission for Bioimaging, ACS Mater. Lett., 2024, 6(8), 3523–3532 CrossRef CAS.
- V. Grande, B. Soberats, S. Herbst, V. Stepanenko and F. Würthner, Hydrogen-bonded perylene bisimide J-aggregate aqua material, Chem. Sci., 2018, 9(34), 6904–6911 RSC.
- C. Sun, M. Zhao, X. Zhu, P. Pei and F. Zhang, One-Pot Preparation of Highly Dispersed Second Near-Infrared J-Aggregate Nanoparticles Based on FD-1080 Cyanine Dye for Bioimaging and Biosensing, CCS Chem., 2022, 4(2), 476–486 CrossRef CAS.
- W. Wagner, M. Wehner, V. Stepanenko, S. Ogi and F. Würthner, Living Supramolecular Polymerization of a Perylene Bisimide Dye into Fluorescent J-Aggregates, Angew. Chem., Int. Ed., 2017, 56(50), 16008–16012 CrossRef CAS PubMed.
- Z. He, Y. Gao, Z. Huang, M. Zhan, S. Tian, F. Fang, D. Zhao, Z. Li, F. Meng, B. Tang and L. Luo, Tuning the Near-Infrared J-Aggregate of a Multicationic Photosensitizer through Molecular Coassembly for Symbiotic Photothermal Therapy and Chemotherapy, ACS Nano, 2025, 19(10), 10220–10231 CrossRef CAS PubMed.
- X. Ma, Y. Huang, W. Chen, J. Liu, S. Liu, J. Yin and G. F. Yang, J-Aggregates Formed by NaCl Treatment of Aza-Coating Heptamethine Cyanines and Their Application to Monitoring Salt Stress of Plants and Promoting Photothermal Therapy of Tumors, Angew. Chem., Int. Ed., 2022, 62(3), e202216109 CrossRef PubMed.
- M. Wehner and F. Würthner, Supramolecular polymerization through kinetic pathway control and living chain growth, Nat. Rev. Chem., 2019, 4(1), 38–53 CrossRef.
- H. Alotaibi, T. Hatahet and W. Al-Jamal, Indocyanine green J-aggregate (IJA) theranostics: Challenges and opportunities, Int. J. Pharm., 2024, 661, 124456 CrossRef CAS PubMed.
- W. Chen, C. Cheng, E. Cosco, S. Ramakrishnan, J. Lingg, O. Bruns and J. Sletten, Shortwave Infrared Imaging with J-Aggregates Stabilized in Hollow Mesoporous Silica Nanoparticles, J. Am. Chem. Soc., 2019, 141(32), 12475–12480 CrossRef CAS PubMed.
- B. Du, R. Liu, C. Qu, K. Qian, Y. Suo, F. Wu, H. Chen, X. Li, Y. Li, H. Liu and Z. Cheng, J-aggregates albumin-based NIR-II fluorescent dye nanoparticles for cancer phototheranostics, Mater. Today Bio, 2022, 16, 100366 CrossRef CAS PubMed.
- X. Guo, M. Li, H. Wu, W. Sheng, Y. Feng, C. Yu, L. Jiao and E. Hao, Near-IR absorbing J-aggregates of a phenanthrene-fused BODIPY as a highly efficient photothermal nanoagent, Chem. Commun., 2020, 56(93), 14709–14712 RSC.
- X. Guo, J. Yang, M. Li, F. Zhang, W. Bu, H. Li, Q. Wu, D. Yin, L. Jiao and E. Hao, Unique Double Intramolecular and Intermolecular Exciton Coupling in Ethene-Bridged aza-BODIPY Dimers for High-Efficiency Near-Infrared Photothermal Conversion and Therapy, Angew. Chem., Int. Ed., 2022, 61(44), e202211081 CrossRef CAS PubMed.
- J. Liu, X. Zhang, M. Fu, X. Wang, Y. Gao, X. Xu, T. Xiao, Q. Wang and Q. Fan, A diketopyrrolopyrrole-based small molecule with an extended conjugated skeleton and J-aggregation behavior for 808 nm laser triggered phototheranostics, Biomater. Sci., 2023, 11(21), 7124–7131 RSC.
- C. Teng, H. Dang, S. Zhang, Y. Xu, D. Yin and L. Yan, J-aggregates of Br- and piperazine-modified cyanine dye with the assistance of amphiphilic polypeptides for efficient NIR-IIa phototheranostics under 1064 nm irradiation, Acta Biomater., 2022, 154, 572–582 CrossRef CAS PubMed.
- N. Yang, S. Song, M. Akhtar, C. Liu, L. Yao, J. Yu, Y. Li, Q. Li, D. He and C. Yu, J-Aggregation induced NIR-II fluorescence: an aza-BODIPY luminogen for efficient phototheranostics, J. Mater. Chem. B, 2023, 11(40), 9712–9720 RSC.
- F. Zhao, X. Zhang, F. Bai, S. Lei, G. He, P. Huang and J. Lin, Maximum Emission Peak Over 1500 nm of Organic Assembly for Blood–Brain Barrier-Crossing NIR-IIb Phototheranostics of Orthotopic Glioblastoma, Adv. Mater., 2023, 35(22), 2208097 CrossRef CAS PubMed.
- Y. Zhao, L. Li, Q. Ye, Y. Gong, R. Yang and H. Liu, Reaction-Activated Disassembly of the NIR-II Probe Enables Fast Detection and Ratiometric Photoacoustic Imaging of Glutathione in Vivo, Anal. Chem., 2023, 95(37), 14043–14051 CrossRef CAS PubMed.
- Y. Zhu, F. Wu, B. Zheng, Y. Yang, J. Yang and H. Xiong, Electron-Withdrawing Substituents Enhance the Type I PDT and NIR-II Fluorescence of BODIPY J Aggregates for Bioimaging and Cancer Therapy, Nano Lett., 2024, 24(27), 8287–8295 CrossRef CAS PubMed.
- Y. Liu, Y. Song, Z. Zhu, C. Ji, J. Li, H. Jia, Y. Shi, F. Hu, Z. Zhao, D. Ding, B. Tang and G. Feng, Twisted-Planar Molecular Engineering with Sonication-Induced J-Aggregation To Design Near-Infrared J-Aggregates for Enhanced Phototherapy, Angew. Chem., Int. Ed., 2024, 64(7), e202419428 CrossRef PubMed.
- L. Yuan, W. Lin, Y. Yang and H. Chen, A Unique Class of Near-Infrared Functional Fluorescent Dyes with Carboxylic-Acid-Modulated Fluorescence ON/OFF Switching: Rational Design, Synthesis, Optical Properties, Theoretical Calculations, and Applications for Fluorescence Imaging in Living Animals, J. Am. Chem. Soc., 2012, 134(2), 1200–1211 CrossRef CAS PubMed.
- Y. Tang, D. Xiang and Q. Li, In Situ Secondary Self-Assembly of Near-Infrared II J-Aggregates: A Novel Phototheranostic Strategy for Inducing Tumor Pyroptosis, Adv. Mater., 2025, 37, 2501184 CrossRef CAS PubMed.
- M. Cui, W. Li, Y. Chen, H. Liu, L. Liu, M. Ji and P. Wang, Aggregation-Enhanced Asymmetric Heptamethine Cyanine Nanoplatform for Imaging-Guided Synergistic Phototherapy and Ferroptosis, Aggregate, 2025, 6, e70211 CrossRef CAS.
- C. Sun, B. Li, M. Zhao, S. Wang, Z. Lei, L. Lu, H. Zhang, L. Feng, C. Dou, D. Yin, H. Xu, Y. Cheng and F. Zhang, J-Aggregates of Cyanine Dye for NIR-II in Vivo Dynamic Vascular Imaging beyond 1500 nm, J. Am. Chem. Soc., 2019, 141, 19221–19225 CrossRef CAS PubMed.
- H. Li, H. Kim, F. Xu, J. Han, Q. Yao, J. Wang, K. Pu, X. Peng and J. Yoon, Activity-based NIR fluorescent probes based on the versatile hemicyanine scaffold: design strategy, biomedical applications, and outlook, Chem. Soc. Rev., 2022, 51(5), 1795–1835 RSC.
- Q. Sun, D. He, L. Zhang, Z. Li, L. Qu and Y. Sun, Coumarin-hemicyanine-based far-red to near-infrared fluorescent probes: A new generation of fluorescent probe design platform, TrAC, Trends Anal. Chem., 2023, 167, 117272 CrossRef CAS.
- Z. Zeng, S. Liew, X. Wei and K. Pu, Hemicyanine-Based Near-Infrared Activatable Probes for Imaging and Diagnosis of Diseases, Angew. Chem., Int. Ed., 2021, 60(51), 26454–26475 CrossRef CAS PubMed.
- Y. Wei, X. Gao, J. Fang, Y. Xiao, J. Liu, Y. Liu, X. Zhang and B. Shen, Tailoring the pKa of Fluorescence Lifetime Imaging Probes to Visualize Aggrephagy and Resolve Its Microenvironmental Viscosity, Anal. Chem., 2024, 96(35), 14160–14167 CrossRef CAS PubMed.
- K. Wang, K. Gu, J. Cao, Y. Yang, H. Zhu, J. Shang and J. Zhou, Activatable Photoacoustic/Near-Infrared Probes for the Detection of Copper Ions of Cardiovascular Disease in Vivo and in Urine, ACS Sens., 2024, 9(9), 4898–4905 CrossRef CAS PubMed.
- Z. Chai, G. Pan, F. Qi, Y. Zhang, F. Dai, S. Zhang and B. Zhou, Cellular and Intravital ATP Imaging Using a Merocyanine-Hybrided Rhodamine Fluorescent Probe, Anal. Chem., 2025, 97(30), 16411–16420 CrossRef CAS PubMed.
- H. Pan, C. Wu, C. Lin, C. Hsu, Y. Tsai, P. Chowdhury, C. Wang, K. Chang, C. Yang, M. Liu, Y. Chen, S. Su, Y. Lee, H. Chiang, Y. Chan and P. Chou, Rational Design of Asymmetric Polymethines to Attain NIR(II) Bioimaging at >1100 nm, J. Am. Chem. Soc., 2022, 145(1), 516–526 CrossRef PubMed.
- X. Zhang, Y. Chen, H. He, S. Wang, Z. Lei and F. Zhang, ROS/RNS and Base Dual Activatable Merocyanine-Based NIR-II Fluorescent Molecular Probe for in Vivo Biosensing, Angew. Chem., Int. Ed., 2021, 60(50), 26337–26341 CrossRef CAS PubMed.
- Z. Qin, T. B. Ren, H. Zhou, X. Zhang, L. He, Z. Li, X. B. Zhang and L. Yuan, NIRII-HDs: A Versatile Platform for Developing Activatable NIR-II Fluorogenic Probes for Reliable in Vivo Analyte Sensing, Angew. Chem., Int. Ed., 2022, 134(19), e202201541 CrossRef.
- T. Ren, Z. Wang, Z. Xiang, P. Lu, H. Lai, L. Yuan, X. B. Zhang and W. Tan, A General Strategy for Development of Activatable NIR-II Fluorescent Probes for in Vivo High-Contrast Bioimaging, Angew. Chem., Int. Ed., 2020, 60(2), 800–805 CrossRef PubMed.
- R. Acharya, S. Cekli, C. J. Zeman, R. M. Altamimi and K. S. Schanze, Effect of Selenium Substitution on Intersystem Crossing in π-Conjugated Donor-Acceptor-Donor Chromophores: The LUMO Matters the Most, J. Phys. Chem. Lett., 2016, 7(4), 693–697 CrossRef CAS PubMed.
- T. Weng, G. Baryshnikov, C. Deng, X. Li, B. Wu, H. Wu, H. Ågren, Q. Zou, T. Zeng and L. Zhu, A Fluorescence-Phosphorescence-Phosphorescence Triple-Channel Emission Strategy for Full-Color Luminescence, Small, 2020, 16(7), 1906475 CrossRef CAS PubMed.
- B. Kim, H. Yeom, M. Yun, J. Kim and C. Yang, A Selenophene Analogue of PCDTBT: Selective Fine-Tuning of LUMO to Lower of the Bandgap for Efficient Polymer Solar Cells, Macromolecules, 2012, 45(21), 8658–8664 CrossRef CAS.
- W. Lee, S. Son, K. Kim, S. Lee, W. Shin, S. Moon and I. Kang, Synthesis and Characterization of New Selenophene-Based Donor-Acceptor Low-Bandgap Polymers for Organic Photovoltaic Cells, Macromolecules, 2012, 45(3), 1303–1312 CrossRef CAS.
- H. Saadeh, L. Lu, F. He, J. Bullock, W. Wang, B. Carsten and L. Yu, Polyselenopheno[3,4-b]selenophene for Highly Efficient Bulk Heterojunction Solar Cells, ACS Macro Lett., 2012, 1(3), 361–365 CrossRef CAS PubMed.
- Y. Wang, H. Masunaga, T. Hikima, H. Matsumoto, T. Mori and T. Michinobu, New Semiconducting Polymers Based on Benzobisthiadiazole Analogues: Tuning of Charge Polarity in Thin Film Transistors via Heteroatom Substitution, Macromolecules, 2015, 48(12), 4012–4023 CrossRef CAS.
- N. James, Y. Chen, P. Joshi, T. Ohulchanskyy, M. Ethirajan, M. Henary, L. Strekowsk and R. Pandey, Evaluation of polymethine dyes as potential probes for near infrared fluorescence imaging of tumors: part - 1, Theranostics, 2013, 3(9), 692–702 CrossRef PubMed.
- R. Wang, M. He, Z. Zhang, T. Qiu, Y. Xi, X. Zeng, J. Fan, W. Sun and X. Peng, Photodynamic therapy promotes hypoxia-activated nitrogen mustard drug release, Smart Mol., 2024, 2(3), e20240010 CrossRef PubMed.
- D. Ma, H. Bian, X. Zhang, K. Xin, Y. Yang, X. Peng and Y. Xiao, Tailored Engineering of Novel Xanthonium Polymethine Dyes for Synergetic PDT and PTT Triggered by 1064 nm Laser toward Deep-Seated Tumors, Small, 2021, 17(21), 2100398 CrossRef PubMed.
- C. Marian, Understanding and Controlling Intersystem Crossing in Molecules, Annu. Rev. Phys. Chem., 2021, 72, 617–640 CrossRef CAS PubMed.
- Y. Shimakura, THEORY OF INTERSYSTEM CROSSING IN AROMATIC COMPOUNDS: EXTENSION OF THE EL-SAYED RULE, Chem. Phys., 1977, 19, 155–163 CrossRef.
- K. Lee, Y. Gao, W. Wei, J. Tan, Y. Wan, Z. Feng, Y. Zhang, Y. Liu, X. Zheng, C. Cao, H. Chen, P. Wang, S. Li, K. Wong and C. Lee, Anti-Quenching NIR-II J-Aggregates of Benzo[c]thiophene Fluorophore for Highly Efficient Bioimaging and Phototheranostics, Adv. Mater., 2023, 35(20), e2211632 CrossRef PubMed.
- W. Chen, G. Luo and X. Zhang, Recent Advances in Subcellular Targeted Cancer Therapy Based on Functional Materials, Adv. Mater., 2018, 31(3), 1802725 CrossRef PubMed.
- T. Pham, V. Nguyen, Y. Choi, S. Lee and J. Yoon, Recent Strategies to Develop Innovative Photosensitizers for Enhanced Photodynamic Therapy, Chem. Rev., 2021, 121(21), 13454–13619 CrossRef CAS PubMed.
- J. Ding, X. Zhao, S. Long, W. Sun, J. Du, J. Fan and X. Peng, A photoactivatable tumor-targeting in situ nanovaccine for large-volume tumor therapy, Smart Mol., 2025, 3(3), e70014 CrossRef CAS PubMed.
- J. Ding, Y. Lu, X. Zhao, S. Long, J. Du, W. Sun, J. Fan and X. Peng, Activating Iterative Revolutions of the Cancer-Immunity Cycle in Hypoxic Tumors with a Smart Nano-Regulator, Adv. Mater., 2024, 36(29), 2400196 CrossRef CAS PubMed.
- H. Jia, J. Liu, M. Shi, M. Abbas, R. Xing and X. Yan, Microneedle delivery systems for vaccines and immunotherapy, Smart Mol., 2025, 3(3), e20240067 CrossRef CAS PubMed.
- Z. Lin, T. He, M. Kang, W. Xie, D. Wang and B. Tang, The Fast-Growing Field of Cancer Immunotherapy Based on Aggregation-Induced Emission, Aggregate, 2025, 6, e70189 CrossRef CAS.
- C. Zhang, X. Yin, L. Hao, Y. Wang, L. Dou, Q. Chen, J.-S. Lee, J. Wang, X. Peng, J. Yoon and H. Li, Integrin-Targeted, Activatable Nanophototherapeutics for Immune Modulation: Enhancing Photoimmunotherapy Efficacy in Prostate Cancer Through Macrophage Reprogramming, Aggregate, 2025, 6, e70189 CrossRef.
- CCDC 2431831: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2mmj5z.
Footnote |
| † These authors contributed equally. |
| This journal is © The Royal Society of Chemistry 2026 |