Open Access Article
Open Access ArticleInfluence of aliovalent substitution on structure and dynamics in sodium halide Na3−2xY1−xNbxCl6 solid electrolytes
Brian B.
Phan
 a,
Tso
Shuen
b,
Dmitry
Vrublevskiy
a,
Qingyu
Yan
a,
Tso
Shuen
b,
Dmitry
Vrublevskiy
a,
Qingyu
Yan
 b and
Vladimir K.
Michaelis
b and
Vladimir K.
Michaelis
 *a
*a
aDepartment of Chemistry, University of Alberta, Edmonton, Alberta T6G 2G2, Canada. E-mail: vladimir.michaelis@ualberta.ca
bSchool of Materials Science and Engineering, Nanyang Technological University, 639798, Singapore
First published on 26th February 2026
Abstract
Sodium halide solid electrolytes are garnering increased interest because of their synthetic flexibility to incorporate a variety of cations, thereby altering their structure and properties. Aliovalent substitution is said to increase ionic conductivity by promoting polyanion rotation. Herein, we synthesise and assess a series of Na3−2xY1−xNbxCl6, probing their complex structures using complementary powder X-ray diffraction and variable-temperature 1D and 2D solid-state nuclear magnetic resonance spectroscopy. The bond-valence energy landscapes of the end members are visualised to reveal potential sodium-ion transport pathways. A structural threshold is reached for Na2Y0.5Nb0.5Cl6, revealing a limit for niobium polyhedral distortion while the unit cell volume is retained up to 50% Nb substitution. Na2Y0.5Nb0.5Cl6 shows the greatest RT ionic conductivity enhancement in the series, from 10−11 S cm−1 to 10−5 S cm−1.
1. Introduction
The demand for energy security underscores the need to improve further battery technologies for long-term energy storage, especially those capable of supporting large-scale, grid-level storage using abundant, inexpensive elements. Lithium-ion batteries have greatly transformed portable electronics, electric vehicles, and the integration of renewable energy grids, helping to address the intermittency issues of solar and wind power. However, the rising demand for lithium and cobalt—key components in this transition—raises concerns about sustainable and ethical sourcing, as well as price volatility driven by their limited natural abundance.1–4 Sodium-ion batteries have emerged as a promising alternative to lithium-based systems and show compatibility with existing lithium infrastructure. Sodium is among the most prevalent elements in the Earth's crust; its sustainable extraction from both oceanic and terrestrial sources is a vital step toward reducing raw material costs and meeting the global demand for battery technologies.1,5To unlock the full potential of sodium-ion technology, researchers are shifting away from flammable organic liquid electrolytes and toward safer solid-state alternatives. The most common class of solid-state sodium electrolytes are sulphides, oxides, and, more recently, halides. Sodium sulphide electrolytes generally exhibit high ionic conductivity and can operate under mild synthetic conditions, but suffer from a narrow electrochemical stability window and are sensitive to moisture.6–9 In contrast, while oxide-based electrolytes offer superior thermal and electrochemical stability, their performance is significantly hindered by high grain-boundary resistance, which impedes ion flow at the interface and results in moderate ionic conductivities.10–14 Among these, sodium halide derivatives are attracting interest as candidates for sodium-ion batteries owing to their excellent interfacial contact with electrodes, broad electrochemical stability windows, defect tolerance, and extensive synthetic versatility, all of which are crucial for efficient ion transport during operation cycling.15–25 To date, several sodium halide derivatives have been reported with a general framework NanM6−nX6 (M = Al3+, Y3+, Sc3+, Ti3+, In3+, Cr3+, Er3+, Eu3+, Lu3+, Zr4+, Hf4+, V5+, Nb5+, Ta5+; X− = F, Cl, Br, I).15,17–20,22,26–33 For example, the role of polymorphism and cation disorder in Na3−xY1−xZrxCl6 was studied, and it was found that transition metal ordering influences ionic transport through polyanion rotation at elevated temperatures.18 A less-researched halide derivative is spinel-type sodium-based electrolytes, Na2MCl4 (M = Y2/3, Sc2/3, etc.), which are thought to have high ionic conductivities and a wide electrochemical window; however, these spinel compounds are largely theoretical.34–36 Analogous lithium halide electrolytes have demonstrated that halide frameworks can overcome some of the limitations described above, offering wide electrochemical stability windows, good compatibility with oxide cathodes, and tuneable ionic conductivities through compositional engineering.34,37–41 The electrochemical properties of these materials can be improved by tailoring their composition and structural framework through M and X ions and aliovalent substitutions that introduce vacancies via cation disorder. In addition to vacancies, aliovalent substitutions can force the material to undergo phase changes; however, the mechanisms by which ionic conductivity is influenced by these substitutions are not fully understood. Nonetheless, cationic substitution has been shown to improve the ionic conductivity of sodium halide electrolytes to 10−4 S cm−1, with the highest non-substituted conductivity reported in NaTaCl6 at 10−4 S cm−1.17,42,43 Hence, further research into aliovalent substitution in solid electrolyte frameworks may be an effective strategy to enhance ionic conductivity relative to their respective end members.44
While halide electrolytes are promising, their performance optimisation relies on understanding their atomic structure. Diffraction methods reveal the overall long-range structure, but it is typically the local chemical environment—including disorder, defects, and variations in chemical surroundings—that affects ionic transport. Differences in local environments can significantly affect activation energy barriers and alter sodium diffusion pathways. Solid-state nuclear magnetic resonance (NMR) spectroscopy is a robust analytical technique that provides structural and dynamic information on the impact of aliovalent substitution by probing local and medium-range structural components in ordered and disordered materials. Specifically, 23Na (nuclear spin, I = 3/2) NMR is highly sensitive due to its 100% natural abundance, small quadrupole moment (Q = 10.4 fm2), and moderate chemical shift range. These properties enable one- and two-dimensional NMR methods to analyse various components in batteries, including electrolytes, cathodes and anodes.45–50 Further extension of the polyanionic structure is accessible by studying the M-site using other NMR-active nuclei, such as 25Mg, 27Al, 89Y, 93Nb, and 125Te.12,51–59 Therefore, combining insights from both short- and long-range structures is essential for establishing precise structure–property relationships at the nanoscale and advancing the development of solid-state sodium electrolytes.
This study explores the impact of aliovalent niobium substitution in Na3−2x(Y1−xNbx)Cl6 on the crystal structure, sodium-ion dynamics, and resulting ionic conductivities. A series of compounds is synthesised using high-energy mechanochemistry and characterised by complementary powder X-ray diffraction (PXRD) and 23Na NMR spectroscopy to elucidate the long- and short-range structural evolutions induced by Y/Nb mixing and their influence on sodium-ion electrochemical properties. Subsequent 93Nb NMR experiments reveal the extent of local octahedral distortion of the [NbCl6]− polyanion. Across the series, the nominal Na2Y0.50Nb0.50Cl6 compound exhibits the highest ionic conductivity with potential Na-ion transport pathways visualised using bond valence sum energy calculations. These findings improve comprehension of the structure and the cation-dynamic role of aliovalent substitution in sodium-containing solid electrolytes based on metal halides.
2. Results and discussion
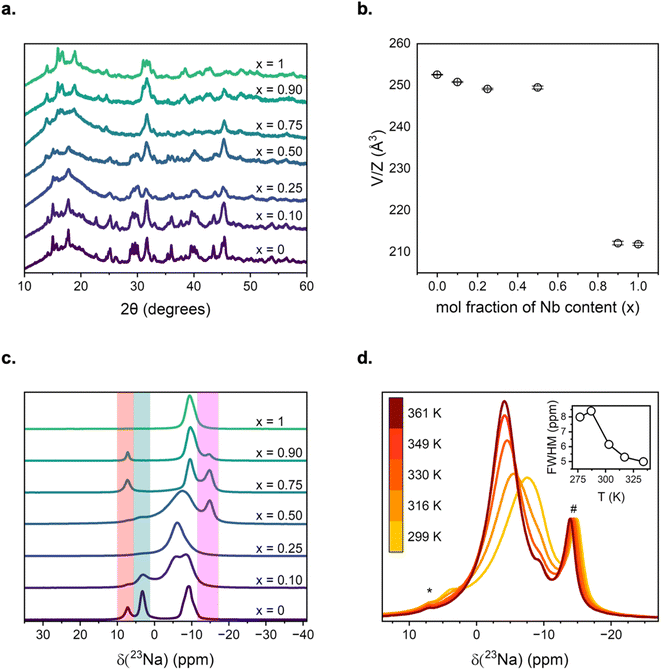
High-energy mechanochemistry offers an environmentally friendly method to create solid solutions with reduced crystallinity. This is achieved through site disorder and structural distortion, which enhance the ionic conductivity of the material electrolyte.60,61 This approach was used to synthesise a series of Na3−2xY1−xNbxCl6 compounds, with x = 0, 0.10, 0.25, 0.50, 0.75, 0.90, and 1.0, and the structure and ionic properties were characterised by PXRD, NMR, and EIS. Herein, the samples will be referred to as YNb0, YNb10, YNb25, YNb50, YNb75, YNb90, and YNb100, respectively, where the number indicates the percentage of Nb substitution at the Y position in the crystal structure.2.1 Long-range Na3−2xY1−xNbxCl6 periodic structure
![[3 with combining macron]](https://www.rsc.org/images/entities/char_0033_0304.gif) , or a monoclinic space group, P21/c.18,62 The trigonal polymorph is accessible through low-temperature, slow-cooling synthetic routes, whereas the high-temperature monoclinic polymorph can be obtained through high-energy ball-milling. While both parent phases crystallise in the space group no. 14, P21/c, Na3YCl6 is commonly depicted as P21/n such that β is closest to 90° and is related to the Na3AlF6 structure type, whereas NaNbCl6 is its own unique structure type. Both end members consist of alternating layers of their respective [YCl6]3− and [NbCl6]− octahedra but differ in their sodium environments. Na3YCl6 has two different Na crystallographic sites, Na1 (2b) and Na2 (4e), which are arranged in alternating layers of octahedral and prismatic sites along the a-axis, with coordination numbers of six and five, respectively. The octahedral and prismatic Na sites are connected via edge- and corner-sharing polyhedra, respectively. In contrast, NaNbCl6 contains only one distinct prismatic Na crystallographic site, Na2 (4e), which occupies every other layer along the c-axis.
, or a monoclinic space group, P21/c.18,62 The trigonal polymorph is accessible through low-temperature, slow-cooling synthetic routes, whereas the high-temperature monoclinic polymorph can be obtained through high-energy ball-milling. While both parent phases crystallise in the space group no. 14, P21/c, Na3YCl6 is commonly depicted as P21/n such that β is closest to 90° and is related to the Na3AlF6 structure type, whereas NaNbCl6 is its own unique structure type. Both end members consist of alternating layers of their respective [YCl6]3− and [NbCl6]− octahedra but differ in their sodium environments. Na3YCl6 has two different Na crystallographic sites, Na1 (2b) and Na2 (4e), which are arranged in alternating layers of octahedral and prismatic sites along the a-axis, with coordination numbers of six and five, respectively. The octahedral and prismatic Na sites are connected via edge- and corner-sharing polyhedra, respectively. In contrast, NaNbCl6 contains only one distinct prismatic Na crystallographic site, Na2 (4e), which occupies every other layer along the c-axis.
Na3YCl6 and NaNbCl6 can be seen as two members of a broader NanM6−nCl6 series where n = 1 (M = Nb) and n = 3 (M = Y), shown in Fig. 1c–e. The monoclinic Na-based series also includes a reported n = 2 member from Pearson's Crystal Data (PCD), Na2IrCl6 (K2TeBr6 type, 473 K), and a Na2ZrCl6 phase that coexists with its trigonal polymorph, although no structure is reported (no higher members, n > 3, have been found). Charge neutrality in the isostructural Na2IrCl6 necessitates sodium deficiency with respect to Na3YCl6. It is manifested as cation vacancies in the 2b sites, where octahedral Na atoms would otherwise be occupied, like in Na3YCl6. To relate NaNbCl6 to its Ir- and Y-bearing counterparts, both the 2b and half of the 4e sites can be imagined to be occupied by Na vacancies. The further removal of half of the 4e sites from Na2IrCl6 to NaNbCl6 seems to distort the structure severely, leading to the need for a larger cell to accommodate the new long-range ordering (e.g. the ordering of Na and “former” prismatic Na vacant sites) and crystallising with Z = 4 formula units (vs. Z = 2 in the other two structures). Overall, the main structural difference between Na3YCl6 and NaNbCl6 is the absence of octahedral and half of the prismatic Na sites, which occurs to maintain charge balance when Y(III) is replaced by Nb(V). The Na vacancy-driven structural relationship described suggests (1) the possible existence of a solid solution between the n = 1 and the n = 3 structures, in which a structure closely related to Na2IrCl6 could be mimicked by a 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 mixture of the end members, and (2) some degree of chemical control over Na vacancies in a wide range of concentrations.
1 mixture of the end members, and (2) some degree of chemical control over Na vacancies in a wide range of concentrations.
| Sample | Space group | V (Å3) | a (Å) | b (Å) | c (Å) | β (°) |
|---|---|---|---|---|---|---|
| a n.d. – not determined. | ||||||
| Na3YCl6 (YNb0) | P21/n | 505.1(1) | 10.145(2) | 7.257(1) | 6.860(1) | 90.79(2) |
| Na2.8Y0.90Nb0.10Cl6 (YNb10) | P21/n | 501.6(2) | 10.117(3) | 7.248(2) | 6.841(1) | 90.86(2) |
| Na2.5Y0.75Nb0.25Cl6 (YNb25) | P21/n | 498.3(3) | 10.105(4) | 7.227(3) | 6.823(2) | 91.01(2) |
| Na2Y0.50Nb0.50Cl6 (YNb50) | P21/n | 498.9(7) | 10.124(1) | 7.267(7) | 6.791(4) | 91.16(5) |
| Na1.5Y0.25Nb0.75Cl6 (YNb75) | P21/c | n.d. | n.d. | n.d. | n.d. | n.d. |
| Na1.2Y0.10Nb0.90Cl6 (YNb90) | P21/c | 849(1) | 6.414(7) | 6.905(8) | 19.16(1) | 90.96(7) |
| NaNbCl6 (YNb100) | P21/c | 847(1) | 6.411(5) | 6.911(6) | 19.13(1) | 90.88(3) |
2.2 Influence of aliovalent substitution on local sodium structure and dynamics using 23Na NMR spectroscopy
![[3 with combining macron]](https://www.rsc.org/images/entities/char_0033_0304.gif) phase can be observed between 2 and −1 ppm.64 The resonance at 7.2 ppm coincides with unreacted NaCl – a common impurity in Na3YCl6 systems due to its unreactive nature and the propensity of YCl3 to stick to the walls of the ball-mill jar during synthesis.60,64,65 Upon 10% substitution of Nb into the sample (YNb10), there is an emergence of a new 23Na peak with a δcgs of −6 ppm that is tentatively assigned to the sodium chemical environments interacting with both [NbCl6]− and [YCl6]3− octahedra. The octahedral sites are observed in the 23Na NMR spectra up to YNb50, consistent with PXRD data, suggesting a single phase with the Na3YCl6 unit cell volume. Likewise, three Na sites remain up to YNb50, where the ∼−6 ppm and prismatic resonances coalesce with small residual octahedral sites at 3.2 ppm for the YNb25 and YNb50 compounds. This coalescence is partly due to the averaging of the Na chemical environments during Na exchange, which appears to be at least on the millisecond time scale, vide infra, and to sensitivity to changes in the unit cell volume and to the influence of Y/Nb next-nearest neighbours. Upon equimolar mixing of Y and Nb, YNb50 reveals the broadest resonance attributed to the random disorder of [NbCl6]− and [YCl6]3− polyanions, inducing a large distribution of sodium chemical environments. YNb50 appears to be the critical threshold for three vs. one sodium sites, as higher Nb loading results in only a single prismatic site being resolved, consistent with the change in unit cell volume observed in XRD analysis.
phase can be observed between 2 and −1 ppm.64 The resonance at 7.2 ppm coincides with unreacted NaCl – a common impurity in Na3YCl6 systems due to its unreactive nature and the propensity of YCl3 to stick to the walls of the ball-mill jar during synthesis.60,64,65 Upon 10% substitution of Nb into the sample (YNb10), there is an emergence of a new 23Na peak with a δcgs of −6 ppm that is tentatively assigned to the sodium chemical environments interacting with both [NbCl6]− and [YCl6]3− octahedra. The octahedral sites are observed in the 23Na NMR spectra up to YNb50, consistent with PXRD data, suggesting a single phase with the Na3YCl6 unit cell volume. Likewise, three Na sites remain up to YNb50, where the ∼−6 ppm and prismatic resonances coalesce with small residual octahedral sites at 3.2 ppm for the YNb25 and YNb50 compounds. This coalescence is partly due to the averaging of the Na chemical environments during Na exchange, which appears to be at least on the millisecond time scale, vide infra, and to sensitivity to changes in the unit cell volume and to the influence of Y/Nb next-nearest neighbours. Upon equimolar mixing of Y and Nb, YNb50 reveals the broadest resonance attributed to the random disorder of [NbCl6]− and [YCl6]3− polyanions, inducing a large distribution of sodium chemical environments. YNb50 appears to be the critical threshold for three vs. one sodium sites, as higher Nb loading results in only a single prismatic site being resolved, consistent with the change in unit cell volume observed in XRD analysis.
Above 50% Nb substitution, a new resonance emerges at δcgs of −14.7 ppm and is attributed to the formation of an oxychloride impurity, NaNbOxCl6−2x (<15%), that is amorphous in nature. The chemical shift varies slightly across literature reports, and its range is shown in Fig. 2c (magenta). The variance in chemical shift is due to the tendency of Nb-rich phases to oxidise, forming a mixed anion phase; however, further refinement and elemental analysis are not possible due to the poor crystallinity and short coherence lengths.66–69 An attempt to form NaNbOCl4 is made; the PXRD and 23Na NMR are shown in Fig. S2. The main oxychloride resonance is observed close to the prismatic site for YNb100 at δcgs = −9.8 ppm, but smaller resonances are observed towards even lower frequency and are attributed to NaNbOxCl6−2x.
| k = Ae−Ea/RT | (1) |
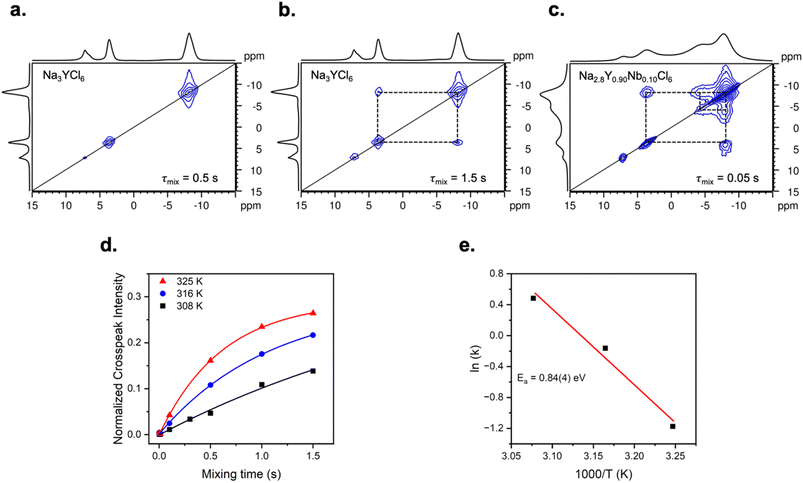
![[3 with combining macron]](https://www.rsc.org/images/entities/char_0033_0304.gif) phase does not influence the main P21/n phase, an EXSY experiment at 325 K and a maximum mixing time shows no cross-peaks between the two phases (i.e. the R
phase does not influence the main P21/n phase, an EXSY experiment at 325 K and a maximum mixing time shows no cross-peaks between the two phases (i.e. the R![[3 with combining macron]](https://www.rsc.org/images/entities/char_0033_0304.gif) phase does not contribute to the dynamics of the main phase), consistent with a previous report.64 Likewise, the residual NaCl peak at 7.2 ppm shows no cross peaks with the main phase and therefore does not contribute to Na-ion exchange.
phase does not contribute to the dynamics of the main phase), consistent with a previous report.64 Likewise, the residual NaCl peak at 7.2 ppm shows no cross peaks with the main phase and therefore does not contribute to Na-ion exchange.
2.3 Influence of aliovalent substitution on local niobium environments using 93Nb NMR
93Nb is a sensitive quadrupolar nucleus (I = 9/2) found in 100% natural abundance with a large quadrupole moment (Q = −32.0 fm2). While 100% abundance is highly advantageous, the quadrupolar moment often leads to significant second-order quadrupole broadening of the central transition. 93Nb NMR spectra were collected at 11.7 and 18.8 T under both spinning and non-spinning conditions to allow for accurate fitting of the NMR parameters. Fig. 4 shows the 93Nb NMR of the series synthesised in this work. The second-order quadrupole interaction in 93Nb NMR cannot be ignored even at high magnetic fields and under MAS conditions, Fig. 4b. Nonetheless, the quadrupole coupling constant is manageable at conventional magnetic field strengths used here for Nb-rich compounds. Table 2 contains the NMR parameters for the series, and the corresponding NMR simulations are shown in Fig. 4b–d and S5. Contrary to a previous report67 stating a 100 MHz CQ for NaNbCl6, the CQ of the same sample synthesized in this work is only 10.2(3) MHz with δiso of 30 ppm and a sizeable chemical shift anisotropy (Ω) of 160 ppm that dominates the central transition lineshape at 18.8 T. Our experimental findings were simulated at 9.4, 11.7 and 18.8 T solving for a unique fit. For comparison, the previously reported CQ of 100 MHz would result in a central transition spanning 3600 ppm at 9.4 T (Fig. S6), which does not agree with their published NMR spectra. Across the series, 93Nb NMR data reveal similar local Nb environments from YNb100 to YNb50 with approximately a 1 MHz increase in CQ as Nb is replaced with Y. That is, the Nb environment does not seem to be heavily distorted from 100 to 50% Nb content. This finding complements the 23Na NMR and PXRD data, which only show minor changes.| Sample | C Q (MHz) | η | δ iso (ppm) | Ω (ppm) | κ | α (°) | β (°) | γ (°) |
|---|---|---|---|---|---|---|---|---|
| a Computed GIPAW-DFT values from CASTEP. | ||||||||
| NaNbCl6 | 10.2(3) [11.1]a | 0.47(1) [0.67]a | 30(2) | 160(20) | −0.16(2) | 69(5) | 25(5) | 28(5) |
| Na1.2Y0.10Nb0.90Cl6 | 10.5(5) | 0.72(5) | 21(2) | 140(10) | 0.00 | 83(5) | 24(5) | 10(5) |
| Na1.5Y0.25Nb0.75Cl6 | 10.6(5) | 0.72(5) | 21(2) | 136(10) | 0.00 | 79(5) | 24(5) | 6(5) |
| Na2Y0.50Nb0.50Cl6 | 11.6(5) | 0.57(5) | 25(2) | 142(10) | −0.21(2) | 78(5) | 23(5) | 15(5) |
| Na2.5Y0.75Nb0.25Cl6 | 69.4(5) | 0.52(5) | −432(10) | 522(30) | 0.67(4) | 18(5) | 62(5) | 49(5) |
| NaNbOxCl6−2x | 80(5) | 0.15(5) | −541(50) | — | — | — | — | — |
| YNbO4 (ref. 72) | 82.2 | 0.38 | −840 | 307 | −0.49 | −1 | 17 | 95 |
| Na5NbO5 (ref. 73) | 11.1 | 0.01 | −903 | 144 | −0.33 | — | — | — |
| NaNbO3 (ref. 74) | 22.7 | — | −1073 | — | — | — | — | — |
| H-Nb2O5 (ref. 75) | 24 | — | −1204 | — | — | — | — | — |
As the niobium-rich compounds transition to yttrium-rich phases (<50% Nb), [NbCl6]− polyhedra appear to be heavily strained as a drastic change in the 93Nb central transition occurs, revealing a sizable second-order quadrupolar broadened central transition spanning over 500 ppm. While the exact cause of this is presently unknown, the M–Cl bond distances may provide some insight. Using crystal structure information derived from the parent phases, it is determined that the M–Cl bond distances differ significantly between the end members and may be inducing local- and medium-range distortions. Y–Cl bond distances are in a narrow range, varying from 2.62 to 2.63 Å, compared to Nb–Cl bond distances that are larger in range but more tightly packed, spanning from 2.25 to 2.48 Å. Although isolated, the Y-rich crystal structure will expand and require Na octahedral sites to return to maintain charge balance associated with replacing Nb5+ with Y3+. In addition, the ionic-covalent character of the different polyanions may also be impacted by higher Y3+ substitution.71 The observed reduction in sensitivity (signal-to-noise) of the Y-rich compounds (YNb10 and YNb25) in the NMR spectra is due to a combination of lower Nb concentration and the formation of a large quadrupole coupling constant of 69 MHz (Fig. S5). Finally, the chemical shift is also heavily influenced, shifting to lower frequency, to a δiso of −432 ppm for YNb25, for example. This result suggests that the stability of Y-rich phases may be more susceptible to oxidation of the niobium chemical environment. To further assess this possibility, we examine the synthesised sodium niobium oxychloride more closely.
2.4 Niobium oxychloride and niobate species
The observed 93Nb NMR chemical shifts for YNb25 and YNb10 lie between those of Nb-rich sodium chloride phases and other niobate species, where earlier studies on niobium oxide compounds typically report isotropic chemical shifts between −400 and −1600 ppm and are sensitive to their coordination environment.58,59,72–77 While our experimental observation suggests the formation of an oxychloride species, with an isotropic chemical shift found between the chloride and oxide-containing species, the 23Na NMR results (above) do not reveal any formation of a sodium-containing oxychloride (or oxides) in these two Y-rich compounds. Further work is needed to determine precisely how Y concentrations influence the Nb environment and the potential role of vacancies, polyanion disorder and oxidation in these solids. However, our assertion is further confirmed by 93Nb NMR (Fig. 5), where the chemical shifts and CQs of known sodium niobate and niobium oxide species do not agree with our experimental data.72–75 Interestingly, the NMR spectrum of the sodium oxychloride, NaNbOxCl6−2x, synthesised here, and that of yttrium niobate,72 YNbO4 (which excludes sodium), share some resemblance with our series. As such, the unique 93Nb NMR spectra may be caused by a Y3+ substitutional defect for the [NbCl6]− polyanion species, serving as an intermediary for the octahedral sodium site – their respective ionic sizes are less than 15% different, Y3+ (0.9 Å) vs. Na+ (1.02 Å). Finally, the 23Na NMR results confirm that a minor sodium oxychloride impurity is present in the Nb-rich compounds; however, it accounts for less than 15% of the total sample, which is a low % and, coupled with the large 80 MHz CQ, is unlikely to be significant. This prevents us from detecting any Nb signal related to this phase. While 93Nb NMR appears to be a sensitive probe nucleus for these types of chemical problems, further exploratory studies are needed into this emerging class of mixed anionic systems. | ||
| Fig. 5 93Nb NMR of samples synthesised in this work (black) compared to reported niobium oxides and niobates simulated in blue. The parameters used for simulations are listed in Table 2. | ||
2.5 Ionic conductivity of aliovalent substituted sodium metal halides
Electrochemical impedance spectroscopy (EIS) was performed on the series at temperatures ranging from 304 to 344 K to determine how their unique compositions and structures influence the bulk ionic conductivity of these sodium metal chlorides. The ionic conductivities (σ) were extracted from the Nyquist plots (Fig. 6a and S7) using Ohm's law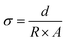 | (2) |
| Sample | E a EIS (eV) | σ (S cm−1) |
|---|---|---|
| a Activation energy determined from variable temperature 23Na EXSY NMR. | ||
| Na3YCl6 (YNb0) | 0.90(1) | 6.1 × 10−11 |
| 0.84(4)a | ||
| Na2.8Y0.90Nb0.10Cl6 (YNb10) | 0.39(3) | 5.7 × 10−8 |
| Na2.5Y0.75Nb0.25Cl6 (YNb25) | 0.44(5) | 2.7 × 10−6 |
| Na2Y0.50Nb0.50Cl6 (YNb50) | 0.28(9) | 9.0 × 10−5 |
| Na1.5Y0.25Nb0.75Cl6 (YNb75) | 0.31(0) | 6.0 × 10−5 |
| Na1.2Y0.10Nb0.90Cl6 (YNb90) | 0.29(0) | 2.3 × 10−5 |
| NaNbCl6 (YNb100) | 0.35(2) | 3.4 × 10−6 |
| NaNbOxCl6−2x | n.d. | 5.0 × 10−4 |
2.6 Bridging chemical structure, sodium mobility and electrochemical properties
Bond valence energy landscape calculations reveal several interstitial sites and saddle points. Briefly, interstitial sites are void spaces that represent a local minima, and saddle points are the transition states or hopping trajectory between two local minima. From the interstitial sites and saddle points, the Na-ion migration pathway for Na3YCl6 indicates a 2D diffusion network. A wide zigzag pattern, Na1-s1-Na0 to Na0-s2-i1-s3-Na1, is shown in Fig. 7b. Although Na1-s4-Na0 is shown as a potential migration pathway, it is not expected to be a primary pathway because s4 has a higher energy barrier (0.72 eV) than s1 (0.56 eV) for Na1-s1-Na0. Furthermore, s4 resides between edge-sharing octahedra, but s1 lies on the face of the octahedral Na. Assuming no vacancies in the structure, Na-ion migration between edge-sharing octahedra is clearly unfavourable even though the distance between Na1 and Na0 is shorter, 3.89 Å, than migration through s1, 4.14 Å. A 1D square zigzag pattern, Na0-s1-i1-s2-i2-Na0, is predicted for NaNbCl6 Na-ion migration along the a-axis shown in Fig. 7d. The migration barrier is predicted to be higher than Na3YCl6, 0.65 vs. 0.58 eV, but the opposite is observed experimentally. Incorrect prediction results can be due to 1D diffusion pathways being less efficient than 2D pathways, inductive effects (see below), and the inability to account for the role of vacancies. The bond valence energy landscapes here provide a first approximation for visualising the Na-ion diffusion network but require further justification to account for the experimental deviation from the predicted energy barriers, as this model cannot capture local disorder or the increased vacancies induced by ball milling.
3. Conclusion
An aliovalent series of Na3−2xY1−xNbxCl6 compounds is successfully synthesised using a solvent-free high-energy ball-milling method. Complementary PXRD and NMR reveal a structural threshold upon incorporation of a smaller, more electronegative Nb5+ at Na2Y0.50Nb0.50Cl6, where the unit cell volume shrinks significantly with higher Nb loading. This threshold at Na2Y0.50Nb0.50Cl6 indicates a single Na chemical environment isostructural with the parent end-member NaNbCl6 and corresponds to the optimal composition with the highest ionic conductivity. Variable-temperature 23Na NMR experiments reveal sodium dynamics on the millisecond (YNb10) and second (YNb0) timescales, demonstrating the importance of Nb substitution for improving sodium-ion conductivity. 93Nb NMR reveals similar chemical environments across Nb loadings; however, a clear shift occurs below YNb50, which correlates with the change in unit cell volume. It appears an intricate combination of interactions play a role in assisting ionic conductivity in these sodium metal halide materials including inductive effect, unit cell expansion enabling the smaller [NbCl6]− octahedra to exhibit polyanion rotation and ball-milling inducing structural disorder and the potential of inducing Na vacancies through aliovalent mixing, leading to a reduction in activation energy, thereby enabling more efficient Na+ conductivity as seen in Na2Y0.50Nb0.50Cl6.4. Experimental
4.1 Materials
NaCl (>99.0%, Millipore Sigma), NbCl5 (99.9%, Millipore Sigma), and YCl3 (99.99%, Millipore Sigma) were stored in an Ar-filled glovebox (O2 and H2O < 1 ppm) and used as is.4.2 Synthesis
Stoichiometric amounts of (3 − 2x)NaCl, xNbCl5, and (1 − x)YCl3 were combined to synthesise mixtures where x = 0, 0.10, 0.25, 0.50, 0.75, 0.90, and 1.0. The mixture was loaded into a 50 mL ZrO2 ball-mill jar with 5 mm ZrO2 balls occupying approximately one-third of the jar in an Ar-filled glovebox. The ball-mill medium was sealed in the glovebox, transferred to a Retsch PM 100 Planetary Mill, and subjected to 18 cycles at 650 rpm, with rotation reversal at each cycle. Each cycle consists of 45 minutes of milling and 15 minutes of rest.4.3 Powder X-ray diffraction
Measurements were performed on the Bruker D8 Advance Diffractometer (Cu-Kα1 (λ = 1.5406 Å) and Kα2 (λ = 1.5444 Å) radiation) operating in Bragg–Brentano (parafocusing) geometry. Samples were prepared in the glovebox with mineral oil and mounted on a zero-background sample holder rotating at 2 rpm. Diffraction patterns were scanned over a 2θ range of 5–90° with a step size of 0.01° s−1. Phase matching was done in Bruker DIFFRAC.EVA v. 7.2.0.2 against the Crystallography Open Database (COD). Pawley fitting and Rietveld refinements were done in the Topas Academic software package (v. 7.0.0.7).88 Zero error of the detector and instrumental contribution to peak broadening were refined from Al2O3 data, while sample displacement and lattice parameters were freely refined. Where appropriate, the NaCl model was included in the fit and refined using the Rietveld method (while the main phase was still refined in Pawley mode) due to the potential for NaCl peaks to overlap with the main phase. Rietveld allows constraining peak intensities, thus avoiding the possibility of fitting NaCl peaks with the 3-1-6/1-1-6 phase. For that, for NaCl, unit cell parameters, scale, and atomic displacement were fixed at published (unit cell) or manually adjusted values (scale, displacement) through a series of iterations.4.4 Solid-state nuclear magnetic resonance spectroscopy
The 23Na and 93Nb NMR were performed on a Bruker Avance NEO 500 (B0 = 11.75 T) and Bruker Advance NEO 800 (B0 = 18.8 T) spectrometer. A 4-mm triple-resonance (HXY) magic-angle spinning (MAS) probe operating in double-resonance mode was used on the NEO 500 spectrometer. Either a 3.2- or 4-mm double-resonance (HX) MAS probe was used on the NEO 800. All 23Na NMR spectra were referenced to 0.1 M NaCl in D2O at δ(23Na) = 0.00 ppm. All 93Nb NMR spectra were indirectly referenced to the 23Na frequency of 0.1 M NaCl in D2O at δ(23Na) = 0 ppm.Variable temperature (VT) 23Na NMR experiments at B0 = 11.75 T were acquired by heating the samples via N2 gas flow through a Bruker BCU II unit (289K ≤ T ≤ 360 K) under MAS (νr = 10 kHz). The samples were corrected for frictional heating and instrumental error using CH3NH3PbCl3 as a calibration standard.8923Na NMR spectra were acquired using a Bloch decay pulse sequence with a 4.0 µs π/2 pulse (νrf = 62.5 kHz), 64 co-added transients, and 15 s recycle delay. Spin–lattice relaxation (T1) experiments were performed using an inversion recovery pulse sequence with a 4.0 µs π/2 pulse, 16 scans, and 0.1 s recycle delay. The T1 values were calculated from the peak intensity using a single-exponential decay function: 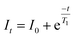 .
.
Two-dimensional 23Na EXchange SpectroscopY (EXSY) experiments were acquired at B0 = 18.8 T with gas flow through a Bruker BCU II unit (289 K ≤ T ≤ 325 K) under MAS (νr = 10 kHz) using mixing times (τmix) ranging from 50 µs to 2 s, 10 s recycle delay, 768 slices, and 8 co-added transients per slice. The samples were corrected for frictional heating and instrumental error using the same method described above. Spin–lattice relaxation (T1) experiments were performed before each EXSY experiment using an inversion recovery pulse sequence with a 2.0 µs π/2 pulse, 8 scans, and 0.1 s recycle delay to ensure mixing times were below the measured T1.
93Nb NMR measurements were acquired at B0 = 11.75 and 18.8 T. The B0 = 11.75 experiments were acquired using a Hahn echo sequence (νr = 0 and 14 kHz) with a 1.87 µs π/2 pulse (νrf = 133.7 kHz), 512 to 11![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 200 co-added transients, and 2 to 5 s recycle delays. The B0 = 18.8 T experiments were acquired using a Hahn echo sequence (νr = 0 and 14 kHz) with a 2.0 µs π/2 pulse (νrf = 125 kHz), 70k to 256k co-added transients, and 1 s recycle delay. All NMR spectra were processed with Bruker TopSpin 4.5.0. and fit using ssNake.90 NMR simulations were performed using WSolids1.91 Data were plotted and analysed in Origin 2024.
200 co-added transients, and 2 to 5 s recycle delays. The B0 = 18.8 T experiments were acquired using a Hahn echo sequence (νr = 0 and 14 kHz) with a 2.0 µs π/2 pulse (νrf = 125 kHz), 70k to 256k co-added transients, and 1 s recycle delay. All NMR spectra were processed with Bruker TopSpin 4.5.0. and fit using ssNake.90 NMR simulations were performed using WSolids1.91 Data were plotted and analysed in Origin 2024.
4.5 Powder electrochemical impedance spectroscopy
The synthesised powder sample was loaded into an electrically insulating Al2O3 cylindrical die with an inner diameter of 10 mm, then a stainless-steel electrode was subsequently inserted and compressed under 374 MPa for 10 minutes. The die was transferred to a rack with three screws and adjusted with a torque wrench to maintain a compressive pressure of 184 MPa for the following measurements. The amount of the specimen loaded for the measurement was 60 mg for each sample, resulting in ≈360 µm of thickness after compression. The EIS measurement was performed in the temperature range 303 K to 343 K using a potentiostat workstation BioLogic VP200 from 7 MHz to 1 Hz with an amplitude of 10 mV. All preparation for EIS measurements was conducted in a glovebox with humidity and oxygen levels below 0.1 ppm.4.6 Quantum chemical computations
Density functional theory (DFT) computations were performed using the Cambridge Serial Total Energy Package92 (CASTEP) with NMR components93–95 using the gauge-included projector augmented waves (GIPAW) method. NMR parameters are calculated using the Perdew–Burke–Ernzerhof (PBE) generalised gradient approximation (GGA) exchange–correlation functional with on-the-fly generated (OTFG) ultrasoft pseudopotentials via zeroth-order regular approximation (ZORA) relativistic effects. CIF files were obtained for NaCl (mp-22862) from Materials Project.71 CIF files for Na3YCl6 (COD ID: 2203822) and NaNbCl6 (COD ID: 8103950) were obtained from the Crystallography Open Database (COD). An initial Monkhorst–Pack (MP) grid was defined by setting the maximum distance between k-points to be 0.07 Å−1, corresponding to a 3 × 3 × 1 MP grid for NaNbCl6 and a 2 × 2 × 3 MP grid for Na3YCl6. k-spacing was stepped iteratively using an extreme basis set accuracy with a 783 eV plane wave cut-off energy until convergence of magnetic shielding parameters was reached within ∼1 ppm. A final MP grid of 6 × 5 × 2 (k-spacing of 0.03 Å−1) for NaNbCl6, 5 × 7 × 8 (k-spacing of 0.02 Å−1) for Na3YCl6P21/c, and 16 × 16 × 16 (k-spacing of 0.02 Å−1) for NaCl. Magnetic shielding and EFG tensor values were extracted from the final MP grid.Author contributions
B. B. Phan designed the project, performed the experiments and data analysis, and wrote the manuscript. T. Shuen conducted EIS experiments and data analysis. D. Vrublevskiy provided XRD data analysis and some manuscript review. Q. Yan provided resources and supervised the project. V. K. Michaelis helped design the project, provided experimental feedback, review & editing, and supervised the project.Conflicts of interest
There are no conflicts to declare.Data availability
The data supporting this article have been included as part of the supplementary information (SI). Supplementary information: Tables S1 and S2, NMR spectra and further experimental details. See DOI: https://doi.org/10.1039/d5sc10183b.Acknowledgements
The Natural Sciences and Engineering Research Council (NSERC) of Canada Discovery program, Canada Research Chairs program, and the NTU-UofA Seed program are acknowledged for research support. Q. Y. acknowledges Singapore ASTAR MTC program M23L9b0052. The Chemistry Centre Magnetic Resonance (C2MR) and X-ray diffraction facilities within the College of Natural and Applied Sciences is supported by the Canada Foundation for Innovation (CFI), the Government of Alberta and the Faculty of Science (University of Alberta). B. B. P. is supported by Future Energy Systems through the University of Alberta.References
- H. S. Hirsh, Y. Li, D. H. S. Tan, M. Zhang, E. Zhao and Y. S. Meng, Adv. Energy Mater., 2020, 10, 2001274 CrossRef.
- J. Huang, K. Wu, G. Xu, M. Wu, S. Dou and C. Wu, Chem. Soc. Rev., 2023, 52, 4933–4995 RSC.
- D. Searcey, E. Lipton and A. Gilbertson, N. Y. Times, 2021 Search PubMed.
- R. B. Kaunda, J. Energy Nat. Resour. Law, 2020, 38, 237–244 Search PubMed.
- K. M. Abraham, ACS Energy Lett., 2020, 5, 3544–3547 CrossRef.
- M. Chaudhary, A. Pominov, D. Mumbaraddi, B. Allen, J. Meyer, A. M. Kirchberger, G. M. Bernard, T. Nilges, A. Mar and V. K. Michaelis, Adv. Funct. Mater., 2024, 34, 2311829 CrossRef CAS.
- S. Yang, Y. Tang, Y. Yao, S. He, Z. Wu, Y. Yang, H. Pan, X. Rui and Y. Yu, Mater. Horiz., 2025, 12, 1058–1083 RSC.
- Y. Fujita, R. Yamanaka, D. Suehiro, K. Imai, K. Koga, T. Asakura, K. Motohashi, A. Sakuda and A. Hayashi, ACS Appl. Energy Mater., 2025, 8, 8216–8223 CrossRef.
- M. Chaudhary, M. Rawat, E. Springl, D. Weindl, D. Sarkar, A. Yu, D. Vrublevskiy, T. Nilges, A. Mar and V. K. Michaelis, ACS Mater. Lett., 2025, 7, 3427–3434 CrossRef.
- J. B. Goodenough, H. Y.-P. Hong and J. A. Kafalas, Mater. Res. Bull., 1976, 11, 203–220 CrossRef.
- L. E. Goodwin, P. Till, M. Bhardwaj, N. Nazer, P. Adelhelm, F. Tietz, W. G. Zeier, F. H. Richter and J. Janek, ACS Appl. Mater. Interfaces, 2023, 15, 50457–50468 CrossRef PubMed.
- D. Sarkar, A. Bhattacharya, J. Meyer, A. M. Kirchberger, V. Mishra, T. Nilges and V. K. Michaelis, J. Am. Chem. Soc., 2023, 145, 19727–19745 CrossRef PubMed.
- M. Akbar, I. Moeez, Y. H. Kim, M. Kim, J. Jeong, E. Lee, A. H. U. Bhatti, J.-H. Park and K. Y. Chung, Carbon Energy, 2025, 7, e717 CrossRef.
- C. Li, R. Li, K. Liu, R. Si, Z. Zhang and Y.-S. Hu, Interdiscip. Mater., 2022, 1, 396–416 Search PubMed.
- K. Motohashi, H. Tsukasaki, A. Sakuda, S. Mori and A. Hayashi, ACS Mater. Lett., 2024, 6, 1178–1183 CrossRef.
- S. J. Hussain, J. Liu, P.-H. Du, M. A. Nazir, Q. Sun and P. Jena, ACS Appl. Mater. Interfaces, 2024, 16, 14364–14370 CrossRef PubMed.
- Z. Huang, S. Yoshida, H. Akamatsu, K. Hayashi and S. Ohno, ACS Mater. Lett., 2024, 6, 1732–1738 CrossRef.
- E. Sebti, J. Qi, P. M. Richardson, P. Ridley, E. A. Wu, S. Banerjee, R. Giovine, A. Cronk, S.-Y. Ham, Y. Shirley Meng, S. Ping Ong and R. J. Clément, J. Mater. Chem. A, 2022, 10, 21565–21578 RSC.
- H. Kwak, J. Lyoo, J. Park, Y. Han, R. Asakura, A. Remhof, C. Battaglia, H. Kim, S.-T. Hong and Y. S. Jung, Energy Storage Mater., 2021, 37, 47–54 CrossRef.
- L. Wang, Z. Song, X. Lou, Y. Chen, T. Wang, Z. Wang, H. Chen, W. Yin, M. Avdeev, W. H. Kan, B. Hu and W. Luo, Small, 2024, 20, 2400195 CrossRef PubMed.
- H. Kwak, S. Wang, J. Park, Y. Liu, K. T. Kim, Y. Choi, Y. Mo and Y. S. Jung, ACS Energy Lett., 2022, 7, 1776–1805 CrossRef.
- Z. Wei, L. F. Nazar and J. Janek, Batter. Supercaps, 2024, 7, e202400005 CrossRef.
- S. Yu, K. Kim, B. C. Wood, H.-G. Jung and K. Yoon Chung, J. Mater. Chem. A, 2022, 10, 24301–24309 Search PubMed.
- Y. Yang, S. Yang, X. Xue, X. Zhang, Q. Li, Y. Yao, X. Rui, H. Pan and Y. Yu, Adv. Mater., 2024, 36, 2308332 Search PubMed.
- K. Tuo, C. Sun and S. Liu, Electrochem. Energy Rev., 2023, 6, 17 CrossRef.
- G. Meyer, S. Peter Ax, T. Schleid and M. Irmler, Z. Anorg. Allg. Chem., 1987, 554, 25–33 CrossRef.
- R. Li, K. Xu, K. Liu, R. Si and Z. Zhang, Chem. Mater., 2022, 34, 8356–8365 CrossRef.
- R. Schlem, A. Banik, M. Eckardt, M. Zobel and W. G. Zeier, ACS Appl. Energy Mater., 2020, 3, 10164–10173 CrossRef.
- E. A. Wu, S. Banerjee, H. Tang, P. M. Richardson, J.-M. Doux, J. Qi, Z. Zhu, A. Grenier, Y. Li, E. Zhao, G. Deysher, E. Sebti, H. Nguyen, R. Stephens, G. Verbist, K. W. Chapman, R. J. Clément, A. Banerjee, Y. S. Meng and S. P. Ong, Nat. Commun., 2021, 12, 1256 CrossRef PubMed.
- Y. Okada, T. Kimura, K. Motohashi, A. Sakuda and A. Hayashi, Electrochemistry, 2023, 91, 077009 Search PubMed.
- B. J. Park, J.-S. Kim, Y. Kim, J. Choe, C. Park, H.-J. Kim, H.-R. Cho, K. H. Shin, J. Lee, J. Choi, J. Kim, H.-W. Lee, Y.-J. Ko, J. Lee, D.-H. Seo and S.-K. Jung, ACS Energy Lett., 2025, 10, 4242–4251 Search PubMed.
- T. Zhao, A. N. Sobolev, R. Schlem, B. Helm, M. A. Kraft and W. G. Zeier, ACS Appl. Energy Mater., 2023, 6, 4334–4341 CrossRef.
- J. Park, J. P. Son, W. Ko, J.-S. Kim, Y. Choi, H. Kim, H. Kwak, D.-H. Seo, J. Kim and Y. S. Jung, ACS Energy Lett., 2022, 7, 3293–3301 CrossRef.
- L. Zhou, C. Y. Kwok, A. Shyamsunder, Q. Zhang, X. Wu and L. F. Nazar, Energy Environ. Sci., 2020, 13, 2056–2063 Search PubMed.
- F. Hussain, P. Yu, J. Zhu, H. Xia, Y. Zhao and W. Xia, Adv. Theory Simul., 2023, 6, 2200569 CrossRef.
- J. Liu, S. Wang, Y. Kawazoe and Q. Sun, ACS Mater. Lett., 2023, 5, 1009–1017 CrossRef.
- Y. Yang, K. Chen and H. Zhu, ACS Nano, 2025, 19, 35833–35841 Search PubMed.
- K. Kim, D. Park, H.-G. Jung, K. Y. Chung, J. H. Shim, B. C. Wood and S. Yu, Chem. Mater., 2021, 33, 3669–3677 Search PubMed.
- K. Tuo, F. Yin and C. Sun, ACS Sustain. Chem. Eng., 2024, 12, 7012–7025 Search PubMed.
- K. Tuo, F. Yin, F. Mi and C. Sun, J. Energy Chem., 2023, 87, 12–23 Search PubMed.
- K. Tuo, C. Sun, C. A. López, M. T. Fernández-Díaz and J. A. Alonso, J. Mater. Chem. A, 2023, 11, 15651–15662 RSC.
- F. Ling, Z. Wu, J. Feng, Z. Wang, Z. Li, R. Bai, W. Du, H. Huo, X. Rui, H. Pan, Y. Yao and Y. Yu, Adv. Mater., 2026, e21368 CrossRef.
- C. Fu, Y. Li, W. Xu, X. Feng, W. Gu, J. Liu, W. Deng, W. Wang, A. M. M. Abeykoon, L. Su, L. Zhu, X. Wu and H. Xiang, Nat. Commun., 2024, 15, 4315 CrossRef CAS.
- S. Ohno, A. Banik, G. F. Dewald, M. A. Kraft, T. Krauskopf, N. Minafra, P. Till, M. Weiss and W. G. Zeier, Prog. Energy, 2020, 2, 022001 Search PubMed.
- K. Gotoh, Batter. Supercaps, 2021, 4, 1267–1278 CrossRef CAS.
- B. Zhang, Y. Ji, L. Liang, Q. Zheng, K. Chen and G. Hou, Chem. Eng. J., 2024, 485, 149879 CrossRef CAS.
- D. L. Smiley and G. R. Goward, Chem. Mater., 2016, 28, 7645–7656 CrossRef CAS.
- P. M. Bayley, N. M. Trease and C. P. Grey, J. Am. Chem. Soc., 2016, 138, 1955–1961 CrossRef CAS.
- J. Billaud, R. J. Clément, A. R. Armstrong, J. Canales-Vázquez, P. Rozier, C. P. Grey and P. G. Bruce, J. Am. Chem. Soc., 2014, 136, 17243–17248 CrossRef CAS.
- M. Gabrijelčič, B. Tratnik, G. Kapun, E. Tchernychova, N. Z. Logar, A. Krajnc, R. Dominko and A. Vizintin, J. Mater. Chem. A, 2025, 13, 1042–1056 RSC.
- S. Zhang, Y. Xu, H. Wu, T. Pang, N. Zhang, C. Zhao, J. Yue, J. Fu, S. Xia, X. Zhu, G. Wang, H. Duan, B. Xiao, T. Mei, J. Liang, X. Sun and X. Li, Angew. Chem., Int. Ed., 2024, 63, e202401373 CrossRef CAS.
- E. Ruoff, S. Kmiec and A. Manthiram, Adv. Energy Mater., 2024, 14(37), 2402091 CrossRef CAS.
- X. Xu, Q. Gao, L. Tang and W. Chen, Appl. Phys. Lett., 2025, 126, 171903 CrossRef CAS.
- G. Wang, S. Zhang, H. Wu, M. Zheng, C. Zhao, J. Liang, L. Zhou, J. Yue, X. Zhu, Y. Xu, N. Zhang, T. Pang, J. Fu, W. Li, Y. Xia, W. Yin, X. Sun and X. Li, Adv. Mater., 2025, 37, 2410402 CrossRef CAS PubMed.
- B. Karasulu, S. P. Emge, M. F. Groh, C. P. Grey and A. J. Morris, J. Am. Chem. Soc., 2020, 142, 3132–3148 CrossRef CAS.
- M. A. Plass, S. Bette, N. Philipp, I. Moundrakovski, K. Küster, R. E. Dinnebier and B. V. Lotsch, J. Mater. Chem. A, 2023, 11, 13027–13038 RSC.
- L.-S. Du, R. W. Schurko, K. H. Lim and C. P. Grey, J. Phys. Chem. A, 2001, 105, 760–768 CrossRef CAS.
- K. E. Johnston, J. M. Griffin, R. I. Walton, D. M. Dawson, P. Lightfoot and S. E. Ashbrook, Phys. Chem. Chem. Phys., 2011, 13, 7565–7576 RSC.
- O. B. Lapina, D. F. Khabibulin, A. A. Shubin and V. V. Terskikh, Prog. Nucl. Magn. Reson. Spectrosc., 2008, 53, 128–191 CrossRef CAS.
- R. Schlem, S. Muy, N. Prinz, A. Banik, Y. Shao-Horn, M. Zobel and W. G. Zeier, Adv. Energy Mater., 2020, 10, 1903719 CrossRef CAS.
- C. Wang, J. Liang, J. T. Kim and X. Sun, Sci. Adv., 2022, 8, eadc9516 CrossRef CAS PubMed.
- A. Miura, K. Muraoka, K. Maki, S. Kawaguchi, K. Hikima, H. Muto, A. Matsuda, I. Yamane, T. Shimada, H. Ito, Y. Mizuguchi, C. Moriyoshi, H. Nakajima, S. Mori, H. Oike, A. Nakayama, W. Sun, N. C. Rosero-Navarro and K. Tadanaga, J. Am. Chem. Soc., 2024, 146, 25263–25269 CrossRef CAS.
- R. D. Shannon, Acta Crystallogr. A, 1976, 32, 751–767 CrossRef.
- E. Sebti, J. Qi, P. M. Richardson, P. Ridley, E. A. Wu, S. Banerjee, R. Giovine, A. Cronk, S.-Y. Ham, Y. S. Meng, S. P. Ong and R. J. Clément, J. Mater. Chem. A, 2022, 10, 21565–21578 RSC.
- E. Sebti, H. A. Evans, H. Chen, P. M. Richardson, K. M. White, R. Giovine, K. P. Koirala, Y. Xu, E. Gonzalez-Correa, C. Wang, C. M. Brown, A. K. Cheetham, P. Canepa and R. J. Clément, J. Am. Chem. Soc., 2022, 144, 5795–5811 CrossRef CAS.
- T. Zhao, B. Samanta, X. M. de Irujo-Labalde, G. Whang, N. Yadav, M. A. Kraft, P. Adelhelm, M. R. Hansen and W. G. Zeier, ACS Mater. Lett., 2024, 6, 3683–3689 CrossRef CAS.
- S. Kmiec, E. Ruoff and A. Manthiram, Angew. Chem., Int. Ed., 2025, 64, e202416979 CrossRef CAS.
- C. Ma, Z. Yu, J. Fang, Q. Shi, P. Wang, Y. Liu, Z. Liu, R. Monteiro, L. Parreira, H. Chen, Y. Liu, S. Lu, W. Feng, H. Zhu and Y. Zhao, Angew. Chem., Int. Ed., 2026, 65(2), e18183 CrossRef CAS.
- J. A. Newnham, A. G. Squires, M. A. Kraft, D. O. Scanlon and W. G. Zeier, Adv. Energy Mater., 2025, 15, e04067 CrossRef CAS.
- C. L. Perrin and T. J. Dwyer, Chem. Rev., 1990, 90, 935–967 CrossRef CAS.
- A. Jain, S. P. Ong, G. Hautier, W. Chen, W. D. Richards, S. Dacek, S. Cholia, D. Gunter, D. Skinner, G. Ceder and K. A. Persson, APL Mater., 2013, 1, 011002 Search PubMed.
- J. V. Hanna, K. J. Pike, T. Charpentier, T. F. Kemp, M. E. Smith, B. E. G. Lucier, R. W. Schurko and L. S. Cahill, Chem.–Eur. J., 2010, 16, 3222–3239 CrossRef CAS PubMed.
- O. B. Lapina, D. F. Khabibulin, K. V. Romanenko, Z. Gan, M. G. Zuev, V. N. Krasil’nikov and V. E. Fedorov, Solid State Nucl. Magn. Reson., 2005, 28, 204–224 CrossRef CAS PubMed.
- S. Prasad, P. Zhao, J. Huang, J. J. Fitzgerald and J. S. Shore, Solid State Nucl. Magn. Reson., 2001, 19, 45–62 CrossRef CAS.
- A. Flambard, L. Montagne, L. Delevoye and S. Steuernagel, Solid State Nucl. Magn. Reson., 2007, 32, 34–43 CrossRef CAS.
- I. V. Yakovlev, E. Papulovskiy, E. A. Paukshtis, V. M. Bondareva, A. V. Toktarev, V. I. Zaikovskii and O. B. Lapina, Appl. Magn. Reson., 2019, 50, 589–597 CrossRef CAS.
- L.-S. Du, R. W. Schurko, N. Kim and C. P. Grey, J. Phys. Chem. A, 2002, 106, 7876–7886 Search PubMed.
- H. Chen, L. L. Wong and S. Adams, Acta Crystallogr., Sect. B, 2019, 75, 18–33 Search PubMed.
- A. Manthiram and J. B. Goodenough, J. Power Sources, 1989, 26, 403–408 CrossRef CAS.
- C. Masquelier and L. Croguennec, Chem. Rev., 2013, 113, 6552–6591 CrossRef CAS.
- S. P. Culver, A. G. Squires, N. Minafra, C. W. F. Armstrong, T. Krauskopf, F. Böcher, C. Li, B. J. Morgan and W. G. Zeier, J. Am. Chem. Soc., 2020, 142, 21210–21219 CrossRef CAS PubMed.
- H. Fang and P. Jena, Nat. Commun., 2022, 13, 2078 CrossRef CAS PubMed.
- Z. Zhang, H. Li, K. Kaup, L. Zhou, P.-N. Roy and L. F. Nazar, Matter, 2020, 2, 1667–1684 CrossRef.
- M. Jansen, Angew Chem. Int. Ed. Engl., 1991, 30, 1547–1558 CrossRef.
- A. Lundén, Solid State Commun., 1988, 65, 1237–1240 CrossRef.
- Z. Zhang, P.-N. Roy, H. Li, M. Avdeev and L. F. Nazar, J. Am. Chem. Soc., 2019, 141, 19360–19372 CrossRef CAS PubMed.
- R. Li, K. Xu, S. Wen, X. Tang, Z. Lin, X. Guo, M. Avdeev, Z. Zhang and Y.-S. Hu, Nat. Commun., 2025, 16, 6633 CrossRef CAS PubMed.
- A. A. Coelho, TOPAS-Academic (version 7.0.0.7) Coelho Software, Brisbane, Austrailia, 2007 Search PubMed.
- G. M. Bernard, A. Goyal, M. Miskolzie, R. McKay, Q. Wu, R. E. Wasylishen and V. K. Michaelis, J. Magn. Reson., 2017, 283, 14–21 CrossRef CAS.
- S. G. J. van Meerten, W. M. J. Franssen and A. P. M. Kentgens, J. Magn. Reson., 2019, 301, 56–66 CrossRef CAS PubMed.
- K. Eichele, WSolids1 (version 1.21.7) 2021 Search PubMed.
- S. J. Clark, M. D. Segall, C. J. Pickard, P. J. Hasnip, M. I. J. Probert, K. Refson and M. C. Payne, Z. Kristallogr. -Cryst. Mater., 2005, 220, 567–570 CrossRef CAS.
- C. J. Pickard and F. Mauri, Phys. Rev. B: Condens. Matter Mater. Phys., 2001, 63, 245101 Search PubMed.
- M. Profeta, F. Mauri and C. J. Pickard, J. Am. Chem. Soc., 2003, 125, 541–548 CrossRef CAS.
- J. R. Yates, C. J. Pickard and F. Mauri, Phys. Rev. B: Condens. Matter Mater. Phys., 2007, 76, 024401 CrossRef.
| This journal is © The Royal Society of Chemistry 2026 |