Open Access Article
Open Access ArticleThree wrongs make a right: a computational investigation of [4n]–[4n]–[4n] fused π-systems
Muhammad Usama Gul
Khan
a,
Katarzyna
Młodzikowska-Pieńko
 b,
Renana
Gershoni-Poranne
b,
Renana
Gershoni-Poranne
 *b and
Judy I.
Wu
*b and
Judy I.
Wu
 *a
*a
aDepartment of Chemistry, University of Houston, Houston, Texas 77204-5003, USA
bSchulich Faculty of Chemistry, The Resnick Sustainability Center for Catalysis, Technion—Israel Institute of Technology, Haifa 32000, Israel
First published on 23rd February 2026
Abstract
Fusing antiaromatic units to other antiaromatic units could afford delocalized [4n]–[4n]–[4n] π-systems with reduced paratropicity (or even weak diatropicity), while retaining narrow HOMO–LUMO gaps. Through a computational investigation of bis-annelated borole-fused cyclobutadiene, pentalene, s-indacene, and cyclooctatetraene isomers, we find that these effects are topology-dependent. Comparisons to analogous [4n + 2]–[4n]–[4n + 2] systems show that [4n]–[4n]–[4n] fusion represents a counterintuitive design strategy to access functional antiaromatic π-systems.
Introduction
Ring fusion is a powerful strategy to access π-expanded polycyclic (anti)aromatic hydrocarbons with diverse energetic, magnetic, and electronic properties, and the consequences of fusing [4n + 2] to [4n + 2] and [4n] units have been studied extensively.1–4 [4n + 2]–[4n + 2] annelation extends aromaticity; for example, fusing two benzene rings gives naphthalene.3 In contrast, [4n + 2]–[4n] fusion weakens both diatropicity in the [4n + 2] rings and paratropicity in the [4n] rings. For example, in biphenylene, the benzene rings exhibit pronounced bond length alternation while the four-membered ring resembles a [4]radialene.4,5 As such, [4n + 2]–[4n]–[4n + 2] annelation patterns have emerged as a popular approach for accessing synthetically viable antiaromatic π-systems, as exemplified by preparations of outer and core π-expanded cyclobutadienes,4,6 pentalenes,7–10 and s-indacenes,11–13 and extensive studies correlating structure and properties in these systems.13–16 Yet, the outcomes of combining [4n] and [4n] units are less intuitive.In this work, we investigate the effects of [4n]–[4n]–[4n] annelation. We explore whether fusing antiaromatic rings to antiaromatic rings could give rise to overall diatropic (aromatic) compounds while retaining desirable [4n] features, such as relatively narrow HOMO–LUMO gaps. The intriguing possibility that [4n]–[4n] fusion may generate an aromatic π-system with a [4n + 2] conjugation pathway was first considered by Breslow in the early 1960s17–19 and has since attracted much experimental and computational interest.20–22 Extended Hückel calculations suggest that planar [4n]–[4n] fused π-systems could exhibit resonance stabilization like the [4n + 2] annulenes.23 Indeed, butalene is planar and displays bond length equalization as well as large negative nucleus-independent chemical shifts (NICS) values, consistent with aromatic character.24,25 Nevertheless, attempts to prepare cyclooctatetraenocyclopentadienone (COT-CPD) (Fig. 1a),17 cyclooctatetraenocyclobutadiene (COT-CBD) (Fig. 1b),26–28 octalene (Fig. 1c and d),29,30 bicyclotropone (Fig. 1e),31 and butalene (Fig. 1f)32,33 generally yielded highly reactive or fleeting species, providing little evidence for aromatic stabilization.
 | ||
| Fig. 1 Synthetic examples of [4n]–[4n] fused systems: (a) COT-CPD,17 (b) COT-CBD,26–28 (c) benzooctalene,29 (d) octalene,30 (e) 1-bicyclotropone and 2-bicyclotropone,31 (f) butalene.33 | ||
Breslow et al. noted that “the high basicity [of COT-CPD] suggests that the bicyclic 10π-electron cation may be aromatic.”17 Proton NMR chemical shifts for COT-CBD (ranging between 6.18–7.32 ppm) were downfield shifted, indicating some diatropicity (Fig. 1b).28 Yet, 1H NMR chemical shifts for the eight-membered rings of COT-CPD (ranging between 5.50–6.50 ppm)17 (Fig. 1a) and benzooctalene (5.65–6.30 ppm)29 (Fig. 1c) are nearly identical to that of cyclooctatetraene (5.69 ppm). Bicyclotropone isomers reflect the intrinsic instability of their [4n] subunits rather than the peripheral [4n + 2] π-system; the 2-isomer polymerizes at room temperature, while the 1-isomer could not be isolated (Fig. 1e).31 Butalene could be generated separately from p-benzyne and trapped “somewhat below room temperature,” but has only fleeting existence (Fig. 1f).33 We note, however, that all of the examples shown in Fig. 1 contain either a nonplanar cyclooctatetraene unit (which precludes effective π-conjugation) or a strained cyclobutadiene unit34,35 (which competes with aromatic stabilization).
Here, we investigate the energetic, electronic, and magnetic properties of bis-annelated borole-fused cyclobutadiene, pentalene, s-indacene, and cyclooctatetraene isomers. By extending Breslow's original question to [4n]–[4n]–[4n] fused π-systems, we explore a counterintuitive design strategy in which antiaromatic units are fused to other antiaromatic units, rather than stabilized through conventional [4n + 2] annelation. We assess whether this largely unexplored region of chemical space can complement existing outer and core π-expanded strategies based on [4n + 2] annelation, and provide access to functional antiaromatic π-systems that retain narrow HOMO–LUMO gaps and desirable optoelectronic properties.36,37
Results and discussion
We investigated a series of [4n]–[4n]–[4n] bis-annelated borole-fused cyclobutadiene (1), pentalene (2), s-indacene (3), and cyclooctatetraene (4). We chose borole annelation because the five-membered borole rings are planar and relatively unstrained. Since the borole rings could be fused to the central [4n] components in different orientations, we also studied how placement of the boron atoms influences the overall tropicity of the fused π-system.38–40 We distinguished between molecules in which the boron atom is not adjacent to the fused bond (terminal) and those where it is; for the latter case, we further distinguished based on the relative orientation between the two borons (syn or anti). All structures were optimized at the (U)M11/6-311+G(d,p) level. 3-Syn (y0 = 0.40) and 3-terminal (y0 = 0.54) show modest diradicaloid character, while all others exhibit stable closed-shell wavefunctions (see details in the SI).NICS41,42 were computed at the (U)B97-2/6-311+G(d,p)//(U)M11/6-311+G(d,p) level to quantify the paratropic character of the annelated four- (4MR), five- (5MR), six- (6MR), or eight- (8MR) membered rings. The B97-2 functional was selected for NMR calculations as it has been shown to have the best performance based on test sets of a broad range of organic compounds.43 For planar systems, the NICS(1)zz index was applied and bqs were placed at 1 Å above the ring plane. For systems containing cyclooctatetraene subunits, a single NICS(0) value was computed; “0” is defined by the center of mass of the 8MR carbons. We adopted the NICS(0) index in this work to enable direct comparison with NICS(0) values computed for the non-planar species shown in Fig. 1, some of which do not have a defined “zz” direction (see data in the SI). NICS(1)zz-XY scans44 were computed for all planar systems, using the NICS(1)zz index; details of the scan paths are included in the SI. Current density plots (generated with the SYSMOIC package),45 containing only the contributions of the π-orbitals, were computed for all planar systems at 1 Å above the molecular plane at B97-2/6-311+G(d,p). Counter-clockwise currents indicate paratropicity and clockwise currents indicate diatropicity (see SI for details).
Across all four [4n] cores examined, a consistent pattern emerges: syn- and anti-fused topologies enable overlapping [4n + 2] circuits that weaken paratropicity of the [4n]–[4n]–[4n] π-system. Terminal-fusion precludes a local [4n + 2] circuit and therefore gives rise to π-systems with strong paratropicity. Interestingly, all of the borole-fused [4n]–[4n]–[4n] π-systems display notably narrower HOMO–LUMO gaps compared to their dibenzo-fused [4n + 2]–[4n]–[4n + 2] analogs. These findings suggest that [4n]–[4n]–[4n] may represent an alternative design strategy towards functional antiaromatic π-systems.
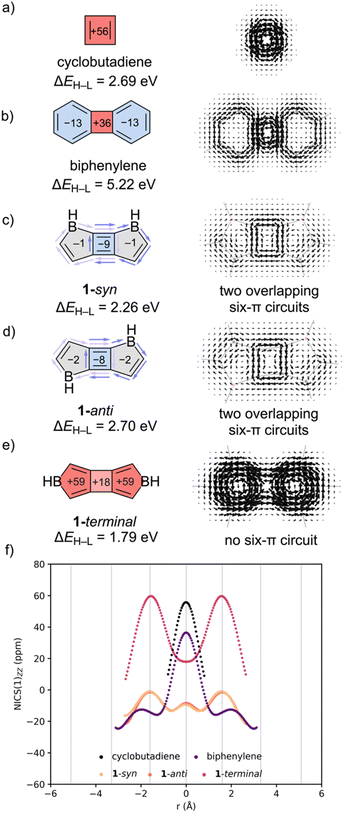
Computed NICS-XY scans for cyclobutadiene, biphenylene, and the bis-annelated borole-fused cyclobutadiene isomers: 1-syn, 1-anti, and 1-terminal (Fig. 2) reveal that cyclobutadiene is strongly paratropic (Fig. 2f, black curve), while biphenylene displays decreased paratropicity in the 4MR and weak diatropicity in the two 6MRs (purple). Bisborole-fusion in either a syn- (peach) or anti- (orange) fashion converts the 4MR ring into a modestly diatropic unit, while the two 5MRs become essentially non-aromatic. In 1-terminal (magenta), the 4MR shows reduced paratropicity compared to cyclobutadiene and biphenylene, while the two 5MRs are strongly paratropic.
SYSMOIC plots for cyclobutadiene and biphenylene show local paratropic ring currents around the 4MR and diatropic currents around the 6MR. However, in 1-syn (Fig. 2c) and 1-anti (Fig. 2d), there are two overlapping diatropic currents, each encompassing a 6π-electron perimeter of a pair of neighboring 4MR and 5MRs. The combined effect is an apparent weak diatropic current at the central 4MR (see light and dark purple arrows in Fig. 2c and d). In 1-terminal, an 8π-electron conjugation pathway gives rise to a strong paratropic current around the perimeter.
HOMO–LUMO gaps (ΔEH–L) for the borole-fused isomers were computed at TD-(U)M11/6-311+G(d,p) and compared to the dibenzo-fused [4n + 2]–[4n]–[4n + 2] analogs and parent [4n] systems. Surprisingly, all three [4n]–[4n]–[4n] systems, 1-syn (2.26 eV), 1-anti (2.70 eV), and 1-terminal (1.79 eV), exhibit narrower computed ΔEH–L values compared to the [4n + 2]–[4n]–[4n + 2] fused biphenylene (5.22 eV). This can be attributed in part to the low-lying LUMO orbitals of the borole-fused isomers (i.e., due to the empty p orbitals on boron), but nevertheless, demonstrate that heteroatom doping can be used to modulate [4n]–[4n]–[4n] π-systems and show reduced paratropicities while retaining narrow HOMO–LUMO gaps.
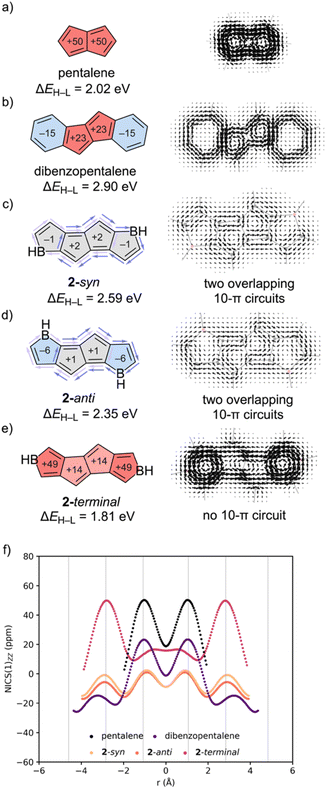
We next compared computed NICS-XY scans for pentalene, dibenzopentalene, and isomers: 2-syn, 2-anti, and 2-terminal (Fig. 3). Pentalene is strongly paratropic (Fig. 3f, black). In dibenzopentalene (purple), the 5MRs show reduced paratropicity and the 6MRs are modestly diatropic. Borole-fusion in either a syn- (peach) or anti- (orange) fashion completely alleviates the paratropicity of the pentalene core, while the fused borole rings are weakly diatropic. In 2-terminal (magenta), the pentalene rings are modestly paratropic and the borole rings are strongly paratropic. These findings corroborate prior work showing that fusion to heteroarenes can significantly alter the paratropicity of pentalene.46
SYSMOIC plots for pentalene (Fig. 3a) and dibenzopentalene (Fig. 3b) reveal strong paratropic ring currents around the pentalene unit and local diatropic currents around the 6MR, while those of the borole-fused isomers feature overlapping ring currents. Both 2-syn (Fig. 3c) and 2-anti (Fig. 3d) exhibit weak paratropic circuits at the pentalene core and two overlapping 10π-electron circuits, each encompassing the perimeter of a single borole subunit and its adjacent pentalene core (see light and dark purple arrows in Fig. 3c and d). As a result of these overlapping ring currents, the pentalene core is essentially non-aromatic. In contrast, 2-terminal features a 12π-electron conjugation pathway and a strong paratropic current around the perimeter. All three isomers: 2-syn (2.59 eV), 2-anti (2.35 eV), and 2-terminal (1.81 eV) exhibit narrower ΔEH–L gaps compared to the [4n + 2]–[4n]–[4n + 2] fused dibenzopentalene (2.90 eV).
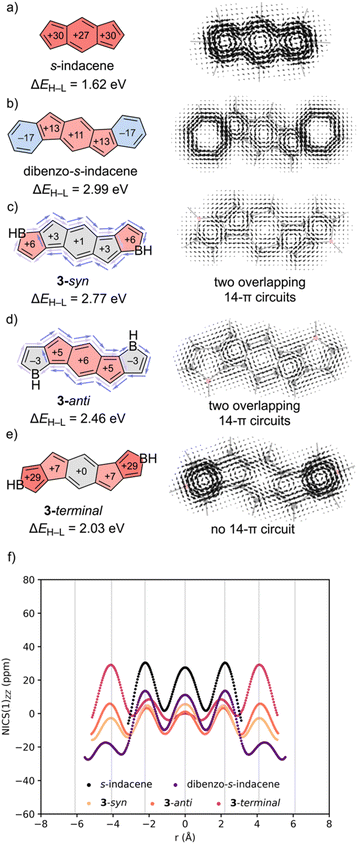
Next, we compared s-indacene and dibenzo-s-indacene to the bis-annelated borole-fused: 3-syn, 3-anti, and 3-terminal (Fig. 4). Computed NICS-XY scans show strong diatropicity in the 6MR and 5MRs of s-indacene (Fig. 4f, black). In dibenzo-s-indacene (purple), the s-indacene core shows significantly reduced paratropicity, while the two arenes are modestly diatropic. Bis-annelation to borole in either a syn- or anti-fashion further reduces paratropicity of the s-indacene core. Both 3-syn (peach) and 3-anti (orange) have nearly non-aromatic borole rings, while that of 3-anti shows weak diatropicity. In 3-terminal (magenta) the s-indacene core is essentially non-aromatic, but the two borole rings are strongly paratropic.
SYSMOIC plots reveal a strong global paratropic ring current for s-indacene (Fig. 4a), while dibenzo-s-indacene exhibits five local ring currents: two diatropic ones around the arenes and three paratropic ones in each of the rings in the s-indacene unit (Fig. 4b). Both 3-syn (Fig. 4c) and 3-anti (Fig. 4d) exhibit a weak global diatropic current, and weak paratropic currents in each of the component rings. Also visible from the current density plots are two overlapping 14π-electron circuits, each encompassing the perimeter of a pair of neighboring borole units and the s-indacene core (see light and dark purple arrows in Fig. 4c and d). In contrast, 3-terminal features an 18π-electron conjugation pathway and a paratropic current around the perimeter. In addition, the borole rings sustain strong local paratropic ring currents. All three bis-annelated borole-fused isomers: 3-syn (2.77 eV), 3-anti (2.46 eV), and 3-terminal (2.03 eV) exhibit narrower ΔEH–L gaps compared to the [4n + 2]–[4n]–[4n + 2] fused dibenzo-s-indacene (2.99 eV), and indicate heteroatom doping as a viable strategy for modulating [4n]–[4n]–[4n] π-systems.
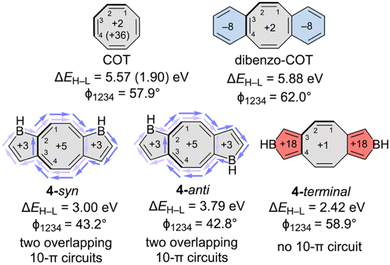
Finally, we examine [4n]–[4n]–[4n] annelated systems with a central cyclooctatetraene (COT) unit (Fig. 5). Planar annelated COTs have been studied extensively, both computationally and experimentally.47–53 Fusing small, strained rings to COT can enforce planarity,52 but the paratropicity of the planar COT core depends on the annelated ring(s); annelation to unconjugated units (e.g., bicyclo[2.1.1]hexeno and cyclobuteno) retains the paratropicity of planar COT, while annelation to conjugated units (e.g., cyclobutadieno) quenches paratropicity.53 Here, we compare dibenzo-COT to three isomers of bis-annelated borole-fused COTs: 4-syn, 4-anti, and 4-terminal (Fig. 5), all of which feature nonplanar COT rings.
Parent COT adopts a nonplanar tub-shape (D2d, ϕ1234 = 57.9°) to avoid ring strain and antiaromaticity that are inherent in the planar geometry,54 and the computed small positive NICS(0) value (+2.4 ppm) is consistent with a non-aromatic ring (Fig. 5). In contrast, planar COT (D4h) exhibits a large positive NICS(0) value (+36.0 ppm) indicating strong paratropicity. Dibenzo-COT retains a highly puckered tub shape (ϕ1234 = 62.0°), and computed NICS(0) values at the 8MR (+2.1 ppm) and 6MRs (−8.0 ppm, cf. −8.3 ppm for parent benzene) suggest a non-aromatic [4n] core flanked by two aromatic benzene rings.
Annelating COT to borole units in either a syn or anti fashion does not fully planarize the COT unit. Nevertheless, both 4-syn and 4-anti tend towards partial planarization exhibiting smaller ϕ1234 dihedral angles (43.2° and 42.8°, respectively) compared to dibenzo-COT. Computed NICS(0) values for the component rings in 4-syn (borole unit: +3.0 ppm, COT unit: +4.8 ppm) and 4-anti (borole unit: +2.9 ppm, COT unit: +4.8 ppm) are all small and positive, indicating essentially non-aromatic π-systems. The modestly flattened COT units in 4-syn and 4-anti suggest that some delocalization is possible.
Indeed, previous calculations of current densities for D2d COT geometries, progressively planarized from the tub-shaped parent structure, show that a paratropic ring current “survives almost all the way to the equilibrium tub-shaped D2d COT structure; the current vanishes at ca. 80% of the geometric change.”55 Hence, some delocalization is reasonable to expect for nonplanar 4-syn and 4-anti; specifically, the two overlapping 10π-conjugated circuits (see black and blue arrows in Fig. 5) can provide a driving force for planarization. 4-Terminal does not sustain a 10π-conjugated circuit, and therefore the COT unit is strongly puckered (ϕ1234 = 58.9°) like that of COT and dibenzo-COT. Computed NICS(0) values for 4-terminal indicate a non-aromatic COT unit (+1.2 ppm) flanked by two paratropic borole rings (+17.5 ppm). We note that all three isomers: 4-syn (2.00 eV), 4-anti (3.79 eV), 4-terminal (2.42 eV) have smaller ΔEH–L values compared to dibenzo-COT (5.88 eV).
Computed relative energies for the syn-, anti-, and terminal-isomers of 1–4 (ΔErel) support the conclusion that π-delocalization strongly stabilizes the syn- and anti-isomers relative to their terminal-analogs. As shown in Table 1, the syn- and anti-isomers for 2–4 are nearly thermoneutral with respect to one another or have only small relative energy differences, while the terminal-analogs lie 10–25 kcal mol−1 higher in energy. This trend does not hold for the isomers of 1, possibly due to strain associated with the 4MR unit. Note that all three isomers of 1 lie within 4 kcal mol−1 of each other. The small energy differences among 1-syn, 1-anti, and 1-terminal can be explained by the resonance forms of the 4MR unit. In 1-syn and 1-anti, the π-system is stabilized by having two overlapping [4n + 2] circuits, but the central 4MR unit resembles a strained cyclobutadiene. By contrast, in 1-terminal, the central 4MR unit resembles a [4]radialene.
We further studied the nature of the syn- and anti-fused isomers (relative to terminally fused isomers) by examining plots of their frontier molecular orbitals, by quantifying estimates of π-electron delocalization energies, and by analyzing their bond length alternation around the perimeter. Visualization of the highest occupied molecular orbitals (HOMOs) for the syn-, anti-, and terminal-isomers of 1–4 shows bonding MOs for the syn- and anti-isomers, which is characteristic of aromatic molecules. In contrast, the HOMOs of the terminal-isomers have much more antibonding character (Fig. 6), which is often seen in antiaromatic systems. This contrast is particularly noteworthy in light of the structural similarity between the systems (i.e., same number of atoms, rings, and extent of conjugated systems); it underlines the fundamental difference in electronic structure between the isomers. Further support was obtained from second-order orbital perturbation (E(2)) analyses, which quantify donor–acceptor interactions, and reveal consistently higher electron-delocalization energies for the syn- and anti-isomers compared to the terminal-analogs (see Table S1). Finally, from the geometrical aspect, the syn- and anti-isomers exhibit less pronounced bond length alternation around the perimeter, in agreement with a more globally delocalized π-system (see Fig. S4). These trends are consistent with a largely suppressed antiaromatic character for the syn- and anti-isomers, relative to the terminal-analogs, and demonstrate that the effects of [4n]–[4n]–[4n] fusion are topology-dependent.
 | ||
| Fig. 6 Computed HOMO (aSHOMO−1 for 3-syn) orbitals for the syn-, anti-, and terminal-isomers of 1–4. | ||
Conclusions
We have systematically investigated [4n]–[4n]–[4n] fused π-systems as an alternative and previously underexplored design strategy for functional antiaromatic molecules. By extending Breslow's original question beyond isolated [4n]–[4n] fusion to a broader set of topologically-controlled [4n]–[4n]–[4n] π-systems, we demonstrate that strategic bis-annelation of borole rings to cyclobutadiene, pentalene, s-indacene, and cyclooctatetraene frameworks can effectively “conceal antiaromaticity,”2 yielding non-aromatic or weakly aromatic π-systems. Remarkably, these [4n]–[4n]–[4n] π-systems exhibit narrower HOMO–LUMO gaps compared to analogous [4n + 2]–[4n]–[4n + 2] dibenzo-fused systems. Although the presence of the B atoms may play a role, the examples shown in Fig. 1 (which do not contain B atoms) make it clear that the [4n]–[4n]–[4n] topology itself results in attenuation of the paratropic current. Our computational investigation of [4n]–[4n]–[4n] fused π-systems demonstrates that “three wrongs can make a right”18 and provides topology-driven design principles that motivate future synthetic efforts in this region of chemical space.Author contributions
Conceptualization: JW; data curation and formal analysis: MUGK; funding acquisition: JW, RGP; investigation: MUGK, KMP; methodology: MUGK, RGP; project administration and resources: JW, RGP; supervision: JW, RGP; visualization: MUGK; writing – original draft: MUGK; writing – review and editing: MUGK, KMP, RGP, JW. All authors have given approval to the final version of the manuscript.Conflicts of interest
There are no conflicts of interest to declare.Data availability
The data supporting this article have been included as part of the supplementary information (SI). Supplementary information is available. See DOI: https://doi.org/10.1039/d5sc07395b.Acknowledgements
J. I. W. thanks the National Science Foundation (CHE-2303851) for financial support. We acknowledge the use of the Carya and Sabine clusters and support from the Research Computing Data Core at the University of Houston. K. M. P. is grateful to the Azrieli Foundation for financial support through a postdoctoral fellowship. R. G. P. gratefully acknowledges the support of the Israel Science Foundation (Grant #1745/23).Notes and references
- E. Vidal Jr., G. I. Warren, J. E. Barker and M. M. Haley, Chem. Sci., 2025, 16, 19026–19047 RSC.
- F. Glöcklhofer, Open Res. Eur., 2025, 5, 70 Search PubMed.
- E. Steiner and P. W. Fowler, Int. J. Quantum Chem., 1996, 60, 609–616 CrossRef CAS.
- S. Radenković, J. Tošović, R. W. A. Havenith and P. Bultinck, ChemPhysChem, 2015, 16, 216–222 CrossRef PubMed.
- J. Thiele, Adv. Cycloaddit., 1899, 306, 87–142 CAS.
- W. C. Lothrop, J. Am. Chem. Soc., 1941, 63, 1187–1191 CrossRef CAS.
- T. Kawase, T. Fujiwara, C. Kitamura, A. Konishi, Y. Hirao, K. Matsumoto, H. Kurata, T. Kubo, S. Shinamura, H. Mori, E. Miyazaki and K. Takimiya, Angew. Chem., Int. Ed., 2010, 49, 7728–7732 CrossRef CAS PubMed.
- K. Brand, Ber. Dtsch. Chem. Ges., 1912, 45, 3071–3077 CrossRef.
- T. Kawase, A. Konishi, Y. Hirao, K. Matsumoto, H. Kurata and T. Kubo, Chem.–Eur. J., 2009, 15, 2653–2661 CrossRef CAS PubMed.
- Z. U. Levi and T. D. Tilley, J. Am. Chem. Soc., 2009, 131, 2796–2797 CrossRef CAS PubMed.
- J. L. Marshall, K. Uchida, C. K. Frederickson, C. Schütt, A. M. Zeidell, K. P. Goetz, T. W. Finn, K. Jarolimek, L. N. Zakharov, C. Risko, R. Herges, O. D. Jurchescu and M. M. Haley, Chem. Sci., 2016, 7, 5547–5558 RSC.
- D. T. Chase, B. D. Rose, S. P. McClintock, L. N. Zakharov and M. M. Haley, Angew. Chem., Int. Ed., 2011, 50, 1127–1130 CrossRef CAS PubMed.
- J. Usuba and A. Fukazawa, Chem.–Eur. J., 2021, 27, 16127–16134 CrossRef CAS PubMed.
- G. I. Warren, K. Młodzikowska-Pieńko, S. Jalife, I. S. Demachkie, J. I. Wu, M. M. Haley and R. Gershoni-Poranne, Chem. Sci., 2025, 16, 575–583 RSC.
- S. Jalife and J. I. Wu, J. Org. Chem., 2025, 90, 4012–4017 CrossRef CAS PubMed.
- S. Jalife, A. Tsybizova, R. Gershoni-Poranne and J. I. Wu, Org. Lett., 2024, 26, 1293–1298 CrossRef CAS PubMed.
- R. Breslow, W. Vitale and K. Wendel, Tetrahedron Lett., 1965, 6, 365–368 CrossRef.
- R. Breslow, Chem. Rec., 2014, 14, 1174–1182 CrossRef CAS PubMed.
- M. E. Vol'pin, Russ. Chem. Rev., 1960, 29, 129–160 CrossRef.
- R. Breslow, Acc. Chem. Res., 1973, 6, 393–398 CrossRef CAS.
- R. G. Bergman, Acc. Chem. Res., 1973, 6, 25–31 CrossRef CAS.
- M. Oda, Pure Appl. Chem., 1986, 58, 7–14 CrossRef CAS.
- J. D. Roberts, A. Streitwieser Jr. and C. M. Regan, J. Am. Chem. Soc., 1952, 74, 4579–4582 CrossRef CAS.
- P. M. Warner and G. B. Jones, J. Am. Chem. Soc., 2001, 123, 10322–10328 CrossRef CAS PubMed.
- K. Ohta and T. Shima, Chem. Phys. Lett., 1994, 217, 7–12 CrossRef CAS.
- J. A. Elix, M. V. Sargent and F. Sondheimer, J. Am. Chem. Soc., 1967, 89, 180 CrossRef CAS.
- G. Schröder and H. Röttele, Angew Chem. Int. Ed. Engl., 1968, 7, 635–636 CrossRef.
- M. Oda and H. Oikawa, Tetrahedron Lett., 1980, 21, 107–110 CrossRef CAS.
- R. Breslow, W. Horspool, H. Sugiyama and W. Vitale, J. Am. Chem. Soc., 1966, 88, 3677–3678 CrossRef CAS.
- E. Vogel, H.-V. Runzheimer, F. Hogrefe, B. Baasner and J. Lex, Angew. Chem., Int. Ed. Engl., 1977, 16, 871–872 CrossRef.
- R. Breslow, M. Oda and T. Sugimoto, J. Am. Chem. Soc., 1974, 96, 1639–1640 CrossRef CAS.
- R. R. Jones and R. G. Bergman, J. Am. Chem. Soc., 1972, 94, 660–661 CrossRef CAS.
- R. Breslow, J. Napierski and T. C. Clarke, J. Am. Chem. Soc., 1975, 97, 6275–6276 CrossRef CAS.
- A. Fattahi, L. Lis, Z. Tian and S. R. Kass, Angew. Chem., Int. Ed., 2006, 45, 4984–4988 CrossRef CAS PubMed.
- J. I. Wu, Y. Mo, F. A. Evangelista and P. v. R. Schleyer, Chem. Commun., 2012, 48, 8437–8439 RSC.
- C. Hong, J. Baltazar and J. D. Tovar, Eur. J. Org Chem., 2022, 2022, e202101343 CrossRef CAS.
- R. Lavendomme and M. Yamashina, Chem. Sci., 2024, 15, 18677–18697 RSC.
- S. Chakraborty, E. M. Yanes and R. Gershoni-Poranne, Beilstein J. Org. Chem., 2024, 20, 1817–1830 CrossRef CAS PubMed.
- P. Finkelstein and R. R. Gershoni-Poranne, ChemPhysChem, 2019, 20, 1508–1520 CrossRef CAS PubMed.
- C. S. Anstöter and P. W. Fowler, ChemPhysChem, 2025, 26, e202401069 CrossRef PubMed.
- P. v. R. Schleyer, C. Maerker, A. Dransfeld, H. Jiao and N. J. R. van Eikema Hommes, J. Am. Chem. Soc., 1996, 118, 6317–6318 CrossRef CAS PubMed.
- C. Corminboeuf, T. Heine and J. Weber, Phys. Chem. Chem. Phys., 2003, 5, 246–251 RSC.
- D. Flaig, M. Maurer, M. Hanni, K. Braunger, L. Kick, M. Thubauville and C. Ochsenfeld, J. Chem. Theory Comput., 2014, 10, 572–578 CrossRef CAS PubMed.
- R. Gershoni-Poranne and A. Stanger, Chem.–Eur. J., 2014, 20, 5673–5688 CrossRef CAS PubMed.
- G. Monaco, F. F. Summa and R. Zanasi, J. Chem. Inf. Model., 2021, 61, 270–283 CrossRef CAS PubMed.
- T. Gazdag, P. J. Mayer, P. P. Kalapos, T. Holczbauer, O. El Bakouri and G. London, ACS Omega, 2022, 7, 8336–8349 CrossRef CAS PubMed.
- T. Ohmae, T. Nishinaga, M. Wu and M. Iyoda, J. Am. Chem. Soc., 2010, 132, 1066–1074 CrossRef CAS PubMed.
- T. C. W. Mak and W.-K. Li, J. Mol. Struct.:THEOCHEM, 1982, 89, 281–284 CrossRef.
- K. K. Baldridge and J. S. Siegel, J. Am. Chem. Soc., 2001, 123, 1755–1759 CrossRef CAS PubMed.
- A. Matsuura and K. Komatsu, J. Am. Chem. Soc., 2001, 123, 1768–1769 CrossRef CAS PubMed.
- T. Nishinaga, T. Ohmae and M. Iyoda, Symmetry, 2010, 2, 76–97 CrossRef CAS.
- K. K. Baldridge and J. S. Siegel, J. Am. Chem. Soc., 2002, 124, 5514–5517 CrossRef CAS PubMed.
- P. W. Fowler, R. W. A. Havenith, L. W. Jenneskens, A. Soncini and E. Steiner, Angew. Chem., Int. Ed., 2002, 41, 1558–1560 CrossRef CAS PubMed.
- C. S. Wannere, D. Moran, N. L. Allinger, B. A. Hess, L. J. Schaad and P. v. R. Schleyer, Org. Lett., 2003, 5, 2983–2986 CrossRef CAS PubMed.
- R. W. A. Havenith, P. W. Fowler and L. W. Jenneskens, Org. Lett., 2006, 8, 1255–1258 CrossRef CAS PubMed.
| This journal is © The Royal Society of Chemistry 2026 |