Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Al(II) transfer harnessing a well-defined cadmium precursor†
Dominic
Herle
 a,
Frerik
Wurm
bc,
Crispin
Lichtenberg
a,
Frerik
Wurm
bc,
Crispin
Lichtenberg
 bc and
Fabian
Dankert
bc and
Fabian
Dankert
 *a
*a
aInstitute of Chemistry, University of Kassel, Heinrich-Plett Str. 40, 34132 Kassel, Germany. E-mail: Fabian.Dankert@uni-kassel.de
bDepartment of Chemistry, Philipps-University Marburg, Hans-Meerwein Str. 4, 35032 Marburg, Germany. E-mail: crispin.lichtenberg@chemie.uni-marburg.de
cmar.quest | Marburg Center for Quantum Materials and Sustainable Technologies, 35032 Marburg, Germany
First published on 23rd February 2026
Abstract
Low-valent aluminum chemistry continues to expand the boundaries of main-group reactivity, yet the selective generation and transfer of Al(II) fragments remain underexplored. Controlled Al(II) transfer may establish a basis for selective substrate alumination and subsequent functionalization. More broadly, findings in the field offer conceptual guidance for the development of synthetically and potentially catalytically relevant main-group platforms. Here, we now show that heterometals can act as structural templates that tame and direct Al(II) reactivity within covalent Al/Cd frameworks. The trimetallic compound [({N(TMS)2})(Cp*)Al]2Cd (1tri; Cp* = C5Me5) proved as an excellent candidate, selectively transferring aluminum complex fragments through cadmium extrusion. This reactivity was verified through reactions with free radicals, dichalcogenides, and benzophenone (-derivatives), the latter representing spin-trapped versions of the fleeting Al(II) radical [({N(TMS)2})(Cp*)Al]˙. In contrast, [{N(TMS)2}(Cp*)Al–Cd{N(TMS)2}] (1bi) retains cadmium and instead promotes cadmium transfer, i.e. to form chalcogenophenolates, underscoring its nuclearity-dependent reactivity. Corroborating these experimental observations, DFT studies provide insight into the formation pathways, electronic structure, and stability of the resulting compounds.
Introduction
Aluminum chemistry has long been dominated by the +III oxidation state, whose classical Lewis acidic behavior is well established – i.e. in fundamental molecular chemistry to drive hydroelementation reactions or engage in FLP (= frustrated Lewis pair) chemistry.1,2 However, low-valent aluminum species are continuing to emerge as a rich source of unusual electronic structures and unconventional reactivity. Within modern main-group chemistry, aluminylenes (i.e., [R–Al]0), metal aluminyls (i.e. M+[R2Al]−, also referred to as “alumanyls”), and neutral dialumanes (-enes) (i.e., {[R2Al]2}0 and {[R–Al]2}0) have become central players in contemporary research.3–5 Their inherent capacity to serve as electron reservoirs for small-molecule fragments has strongly shaped current perspectives on bond-activation pathways. Consequently, the utilization of low-valent aluminum in reduction chemistry now occupies a substantial and steadily expanding chemical space. In recent years, an important subfield, namely heterometallic aluminum compounds, has rapidly gained momentum.6–9 Among these, alkali-metal aluminyls are widely recognized as [R2Al(I)]− transfer reagents. The landmark K2[Al(NON)]2 system (I) reported by Aldridge and Goicoechea (NON = 4,5-bis(2,6-diisopropylanilido)-2,7-di-tert-butyl-9,9-dimethylxanthene) stands out as one of the first potent examples of an aluminum-centered metallo-ligand.10 Since then, the groups of Yamashita (II),11–13 Coles (III),14–16 Hill (IV),17,18 Harder (V)19 or Hicks (VI)20 have steadily broadened the structural diversity and reactivity landscape of these systems (Scheme 1). These negatively charged Al-based metallo-ligands typically exhibit reactivity at the ambiphilic aluminum center, most prominently, among few other reactivity patterns,7 through single-center oxidative addition. Beyond this intrinsic behavior, however, they also provide access to a broader and highly attractive field of research: the formation of (unsupported) heterometallic partnerships. Such architectures are most commonly achieved through salt-metathesis pathways, which have become a defining strategy for generating novel heterometallic aluminum frameworks. A natural conceptual entry point into heterometallic aluminum chemistry is the fundamental molecular-orbital picture in which the aluminum fragment engages as an “X”-type ligand, operating within an electron-shared bonding regime (Scheme 1b). This scenario is prototypically realized in systems where significant covalency develops between aluminum and a second metal center. Under these conditions, a wide spectrum of metal partners were bound, spanning the s-block,10,12,15,18,20–28 p-block,29–32 transition metals—including early,11,33,34 mid-,35–38 and late35–54—as well as the f-block.55 Some representative examples are shown in Scheme 1b: VII–XI.25,31,40,44,47,55 An attractive feature of these systems is their ability to undergo synergistic bond activation, with the heterometallic partner often acting as a structure-directing unit in small molecule activation pathways concomitant with aluminum functioning Lewis-acidic.7,9 From a fundamental viewpoint, the heterometal may also serve as a template for constructing follow-up organometallic architectures.41Lately, Mankad's group has significantly expanded the accessible reactivity landscape of aluminum-based heterometallics toward radical chemistry under mild conditions by harnessing a [Al]–[Fe] platform.56 Typically, Al(II) radicals can arise via complementary pathways. One common approach is ligand-mediated, where redox-active ligands in the aluminum coordination sphere mimic Al(II) reactivity through low-lying acceptor orbitals.57–59 Another route involves alkali-metal reduction of suitable precursors, as demonstrated recently by Sekiguchi60 or Andrada and co-workers,61 which generates (fleeting) Al(II) radicals. In contrast, the generation of Al(II) via heterometallic frameworks, through direct electronic communication between aluminum and a transition or main-group metal, is far less explored. Notable examples hitherto include Yamashita's formal Al(II) transfer at vanadium,34 Aldridge's Al(II) transfer using Mg[Al(NON)]2,25 and Hill's Birch-type reduction of benzene with a silver bis-aluminyl metallate, where in situ EPR spectroscopy supports the involvement of Al(II).43 While photochemical activation is required for all examples, these studies collectively illustrate the potential of heterometallic frameworks as templates for accessing authentic Al(II) radicals. Despite the interest in Al(II), its migration or transfer between metal centers remains largely unexplored. Gaining insight into how an Al(II) fragment can be mobilized, stabilized, and transferred, particularly within heterometallic architectures, promises to reveal novel reactivity patterns and bonding motifs. From a synthetic perspective, harnessing Al(II) intermediates may as well allow selective substrate alumination and subsequent functionalization, providing entry points to substrate upgrading and diversification strategies.
Recently, we reported bi- (1bi = [{N(TMS)2}(Cp*)Al–Cd{N(TMS)2}]) and trimetallic (1tri = [({N(TMS)2})(Cp*)Al]2Cd) cadmium aluminyls, accessed through stoichiometric aluminylene insertion into Cd{N(TMS)2}2.53 These species exhibit a high degree of covalency between Al and Cd, are notably more stable than their zinc congeners,49,62 and have been shown to participate in incipient reactivity studies such as Al(I) transmetalation and heterocumulene insertion. While several bonding descriptions and oxidation state assignments are conceivable for these compounds, their overall covalent character suggested that they could serve as ideal candidates for Al(II) transfer reactions. Building on this concept, we demonstrate that Al/Cd frameworks can act as effective surrogates for Al(II) under mild, thermal conditions, enabling a range of Al(II) transfer processes, including spin-trap experiments. Remarkably, all transformations proceed at ambient temperature without the need for photochemical activation.
Results and discussion
Al(II) transfer employing a trimetallic cadmium aluminyl
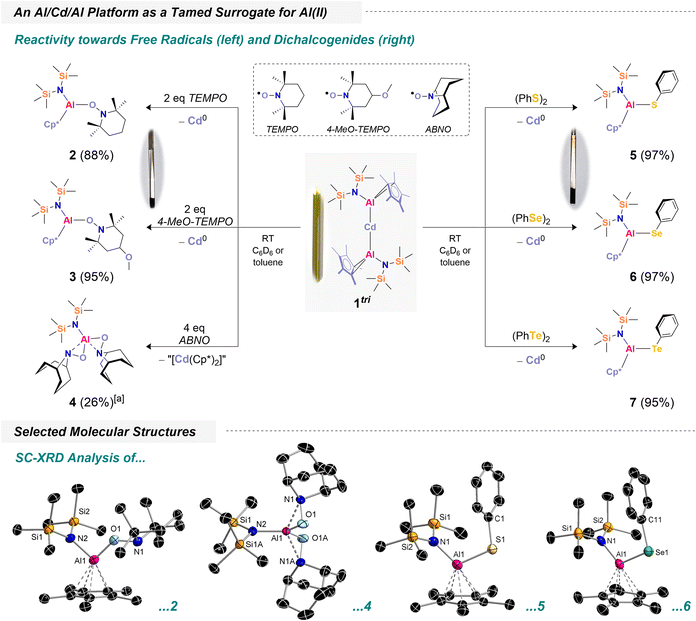
To explore Al(II) transfer, we began by reacting 1tri with two equivalents of TEMPO (TEMPO = 2,2,6,6-tetramethylpiperidine-1-oxyl). Upon addition of TEMPO to a yellow solution of 1tri, the mixture rapidly turned colorless, accompanied by the formation of large amounts of black precipitates (Scheme 2). 1H NMR spectroscopy revealed new resonances at 2.02 ppm and 0.31 ppm, corresponding to Cp* and HMDS moieties, consistent with the selective formation of a new species. Additional signals corresponding to –CH2 and CH3 groups were observed, and integration indicated a 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1
1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 ratio of {N(TMS)2}, Cp* and the associated TEMPO fragment (Fig. S4). Based on these observations, we proposed the formation of [({N(TMS)2})(Cp*)Al(TEMPO)] (2), with concomitant precipitation of elemental cadmium from the solution (see SI). Isolation of 2 was achieved by workup and evaporation from n-pentane, affording crystals suitable for X-ray diffraction (XRD) analysis. The structure confirms TEMPO coordination via its O atom, with an expectedly short Al–O bond of 1.740(5) Å.
1 ratio of {N(TMS)2}, Cp* and the associated TEMPO fragment (Fig. S4). Based on these observations, we proposed the formation of [({N(TMS)2})(Cp*)Al(TEMPO)] (2), with concomitant precipitation of elemental cadmium from the solution (see SI). Isolation of 2 was achieved by workup and evaporation from n-pentane, affording crystals suitable for X-ray diffraction (XRD) analysis. The structure confirms TEMPO coordination via its O atom, with an expectedly short Al–O bond of 1.740(5) Å.
Remarkably, the reaction proceeds with high selectivity: no side products were detected, the conversion of 1tri is quantitative, and 2 was isolated in 88% yield as a crystalline solid. Encouraged by this result, we investigated the scope of 1tri as a mild and controlled Al(II) transfer reagent. As a next example, we explored its reactivity with 4-MeO-TEMPO (MeO = –OCH3). Addition of 4-MeO-TEMPO to a yellow solution of 1tri led to observations analogous to those described for TEMPO. 1H NMR spectroscopy showed resonances consistent with selective formation of a new species, including a signal at 3.17 ppm corresponding to the –OCH3 group, confirming the formation of [({N(TMS)2})(Cp*)Al(4-MeO-TEMPO)] (3) (Fig. S7). After workup, 3 was isolated in 95% yield, and its molecular structure was determined (Fig. S65). Structural metrics are broadly comparable to those of 2. To further explore the scope of N-oxyl derivatives, 1tri was also reacted with ABNO (ABNO = 9-azabicyclo[3.3.1]nonan N-oxyl) following the same protocol. Initially, two equivalents of ABNO did not lead to complete consumption of 1tri (see Fig. S55). 1H NMR analysis revealed a new main species featuring an [{N(TMS)2}]− ligand resonance at 0.46 ppm, formed in a 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 ratio with unreacted 1tri. We therefore hypothesized that full conversion would require four equivalents of ABNO. Upon addition of the additional two equivalents, 1tri was fully consumed. Workup and careful crystallization of the resulting material afforded [({N(TMS)2})Al(ABNO)2] (4). Through meticulous washing and recrystallization from tetramethylsilane solutions, pure crystalline 4·Si(CH3)4 was obtained in 26% yield. The molecular structure of unsolvated 4, which was crystallized from n-pentane, confirms the formal addition of two radicals to the aluminum center with the Cp* ligands being fully replaced. Notably, 4 exhibits an unusual coordination mode: both the N- and O-atoms of the ABNO fragment coordinate to aluminum. This binding motif is reminiscent of [{C4H3N(2-CH2NtBu)}Al(ON = CMe2)]2, where an oximate ligand shows a similar coordination mode.63 Interestingly, a notable difference was observed during the reaction of 1tri with ABNO: only minimal black precipitate formed. This suggests that the fragmentation of 1tri may follow a distinct pathway here, likely involving a twofold one-electron transfer from each Al–Cd bond to four equivalents of ABNO, accompanied by formation of Cd(II)- and Cp*-containing side products. Attempts to identify these side products were unsuccessful, and the species consistently appeared as smeary greases. Beyond N-oxyl derivatives, we investigated dichalcogenides, (PhCh)2 (Ch = S, Se, Te), as potential radical scavengers owing to their relatively low bond dissociation energies. In all cases, the 1
1 ratio with unreacted 1tri. We therefore hypothesized that full conversion would require four equivalents of ABNO. Upon addition of the additional two equivalents, 1tri was fully consumed. Workup and careful crystallization of the resulting material afforded [({N(TMS)2})Al(ABNO)2] (4). Through meticulous washing and recrystallization from tetramethylsilane solutions, pure crystalline 4·Si(CH3)4 was obtained in 26% yield. The molecular structure of unsolvated 4, which was crystallized from n-pentane, confirms the formal addition of two radicals to the aluminum center with the Cp* ligands being fully replaced. Notably, 4 exhibits an unusual coordination mode: both the N- and O-atoms of the ABNO fragment coordinate to aluminum. This binding motif is reminiscent of [{C4H3N(2-CH2NtBu)}Al(ON = CMe2)]2, where an oximate ligand shows a similar coordination mode.63 Interestingly, a notable difference was observed during the reaction of 1tri with ABNO: only minimal black precipitate formed. This suggests that the fragmentation of 1tri may follow a distinct pathway here, likely involving a twofold one-electron transfer from each Al–Cd bond to four equivalents of ABNO, accompanied by formation of Cd(II)- and Cp*-containing side products. Attempts to identify these side products were unsuccessful, and the species consistently appeared as smeary greases. Beyond N-oxyl derivatives, we investigated dichalcogenides, (PhCh)2 (Ch = S, Se, Te), as potential radical scavengers owing to their relatively low bond dissociation energies. In all cases, the 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 reaction of 1tri with (PhCh)2 proceeded within seconds, accompanied by precipitation of elemental cadmium. For example, in the sulfur case, 1H NMR spectroscopy revealed diagnostic Cp* and HMDS resonances at 1.97 and 0.08 ppm, respectively, along with appropriate aromatic signals (see Fig. S14). Integration indicated a 1
1 reaction of 1tri with (PhCh)2 proceeded within seconds, accompanied by precipitation of elemental cadmium. For example, in the sulfur case, 1H NMR spectroscopy revealed diagnostic Cp* and HMDS resonances at 1.97 and 0.08 ppm, respectively, along with appropriate aromatic signals (see Fig. S14). Integration indicated a 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1
1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
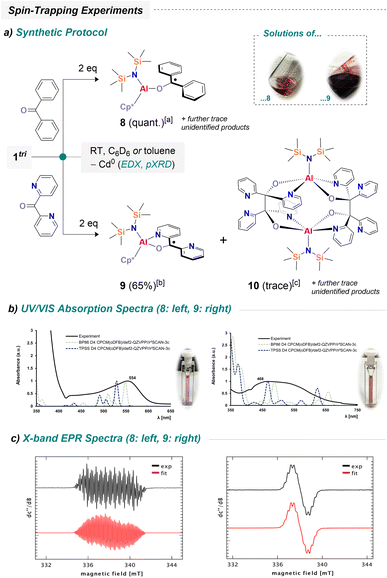
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 ratio of Ph-, Cp*-, and [{N(TMS)2}]-groups, consistent with selective formation of [({N(TMS)2})(Cp*)Al(S–Ph)] (5). X-ray crystallography confirmed that the structural motif mirrors the one observed for TEMPO addition. Similarly, reactions with the Se and Te congeners afforded [({N(TMS)2})(Cp*)Al(Se–Ph)] (6) and [({N(TMS)2})(Cp*)Al(Te–Ph)] (7). 7 is somewhat exceptional, displaying short Te⋯Te contacts in the solid state, resulting in a dimeric arrangement, [({N(TMS)2})(Cp*)Al(Te–Ph)]2 (see Fig. S69). Such contacts are known to influence crystal packing, as recently shown for other tellurium organometallics.64 We briefly note that we also tried to convert 1tri with a more complex trityl radical (i.e. via “Gomberg's dimer”),65 yet unsuccessful allegedly due to steric constraints (see Fig. S56). Collectively, the transformations furnishing compounds 2–7 represent authentic Al(II) transfer events from the Al/Cd framework. To further substantiate the plausibility of Al(II) transfer, we explored spin-trapping of the plausible radical [({N(TMS)2})(Cp*)Al]˙. For this purpose, 1tri was first reacted with two equivalents of benzophenone. The reaction rapidly produced black precipitates, analogous to the previous experiments. Using only one equivalent of benzophenone was insufficient for full conversion, whereas addition of a second equivalent drove the reaction to completion (Fig. S58). Filtration of the precipitate afforded a magenta-colored solution. UV/vis spectroscopy revealed absorption in the 450–600 nm region, with a distinct maximum at 554 nm (Scheme 3b) which almost perfectly matches related aluminum-based ketyl radicals: the absorption maximum of [(NON)Al(OC–Ph2)]˙, for instance, was found at 545 nm.561H NMR spectroscopic analysis of a freshly prepared sample in C6D6 solution showed only trace amounts of diamagnetic species (Fig. S58: top), confirming near-quantitative conversion of 1tri and supporting the formation of a radical product.
1 ratio of Ph-, Cp*-, and [{N(TMS)2}]-groups, consistent with selective formation of [({N(TMS)2})(Cp*)Al(S–Ph)] (5). X-ray crystallography confirmed that the structural motif mirrors the one observed for TEMPO addition. Similarly, reactions with the Se and Te congeners afforded [({N(TMS)2})(Cp*)Al(Se–Ph)] (6) and [({N(TMS)2})(Cp*)Al(Te–Ph)] (7). 7 is somewhat exceptional, displaying short Te⋯Te contacts in the solid state, resulting in a dimeric arrangement, [({N(TMS)2})(Cp*)Al(Te–Ph)]2 (see Fig. S69). Such contacts are known to influence crystal packing, as recently shown for other tellurium organometallics.64 We briefly note that we also tried to convert 1tri with a more complex trityl radical (i.e. via “Gomberg's dimer”),65 yet unsuccessful allegedly due to steric constraints (see Fig. S56). Collectively, the transformations furnishing compounds 2–7 represent authentic Al(II) transfer events from the Al/Cd framework. To further substantiate the plausibility of Al(II) transfer, we explored spin-trapping of the plausible radical [({N(TMS)2})(Cp*)Al]˙. For this purpose, 1tri was first reacted with two equivalents of benzophenone. The reaction rapidly produced black precipitates, analogous to the previous experiments. Using only one equivalent of benzophenone was insufficient for full conversion, whereas addition of a second equivalent drove the reaction to completion (Fig. S58). Filtration of the precipitate afforded a magenta-colored solution. UV/vis spectroscopy revealed absorption in the 450–600 nm region, with a distinct maximum at 554 nm (Scheme 3b) which almost perfectly matches related aluminum-based ketyl radicals: the absorption maximum of [(NON)Al(OC–Ph2)]˙, for instance, was found at 545 nm.561H NMR spectroscopic analysis of a freshly prepared sample in C6D6 solution showed only trace amounts of diamagnetic species (Fig. S58: top), confirming near-quantitative conversion of 1tri and supporting the formation of a radical product.
The most intuitive pathway involves replacement of cadmium by benzophenone, trapping the spin from the transient Al(II) center and generating [({N(TMS)2})(Cp*)Al(O–CPh2)]˙ (8). Unfortunately, all attempts to isolate 8 from the magenta solution were unsuccessful to date. Prolonged storage led to slow decomposition, producing black precipitates and colorless residues (see SI). Remarkably, even at −30 °C under inert conditions, the compound decomposed slowly. To gain further insight, the DFT-optimized structure of 8, combined with time-dependent DFT (TD-DFT) calculations, was utilized to investigate the absorption properties of 8 in the UV/vis region. Indeed, the theoretically predicted spectrum reproduces the experimentally observed UV/vis spectrum reasonably well (Scheme 3b). We note, however, a strong influence of Hartree–Fock contributions (see Table S6: top). NTO (= natural transition orbital) analysis indicates that the dominant excitation corresponds to promotion of the singly occupied molecular orbital into a ligand-centered π* orbital (see Table S6: bottom). Encouraged by these results, 1tri was also reacted with di(2-pyridyl) ketone. The pyridyl substituents were expected to provide additional stabilization through chelating N-coordination at aluminum. Upon addition of two equivalents of the ketone, large amounts of black precipitate formed, similar to previous reactions. Filtration of the resulting dark suspension yielded a deep red-to-purple solution, showing only trace amounts of diamagnetic impurities (see Fig. S59), suggesting formation of [({N(TMS)2})(Cp*)Al(OC–Py2)]˙ (9; Py = 2-pyridyl). Diluted solutions suitable for UV/vis spectroscopy appear amber-red, with the major absorption observed between 400–700 nm and a distinct maximum at 468 nm (Scheme 3b). Chelation by the pyridyl groups provides significant stabilization (vide infra), allowing 9 to be isolated as a dark purple powder in 65% (crude) yield, containing trace amounts of 10 and an additional unidentified species. The successful isolation of this material enabled further investigations such as FD mass spectrometry. The presence of 9 was unambiguously detected in the gas phase (m/z calcd 506.2598; found 506.2596; Fig. S29–S31, including high-resolution spectra). At this stage, all analytical data indicate the precipitation of elemental cadmium from the reaction solutions. To further substantiate this observation, energy-dispersive X-ray spectroscopy (EDX) as well as X-ray powder diffraction were performed on isolated precipitates. EDX analysis confirmed cadmium as the main component, with only trace amounts of aluminum and oxygen detected (Fig. S63; Table S1). The oxygen likely arises from sample preparation in air. Powder diffraction for which sample preparation was possible under inert conditions is consistent with cadmium as the sole component (Fig. S71). However, most eminently, the proposed structures of 8 and 9 have been investigated by X-band EPR spectroscopy. Both, 8 and 9 gave compound-specific couplings to different nuclei (Scheme 3c). For 8, the spectrum exhibits a reasonably resolved multiplet consistent with hyperfine couplings to ten 1H nuclei and one 27Al nucleus, indicating delocalization of the unpaired electron over the two phenyl groups of the ligand framework and (very) weak interaction with the aluminum center. It should be noted that these investigations indicate the inequivalence of the phenyl groups in 8 on the time scale of the EPR spectroscopic experiment, as previously observed for related species.56 For 9, the main resonance reveals coupling of the unpaired electron with two 14N nuclei and eight protons, consistent with spin delocalization over the two spectroscopically inequivalent pyridyl groups. In both cases, the experimental spectra are acceptably reproduced by assuming a single unpaired electron and the coupling constants noted in Fig. S60–S62. Corroborated by DFT calculations, the main contribution of spin density resides at the former carbonyl C atom (Fig. S79) yet is delocalized significantly across the aromatic groups of the ligand, with minor spin density at the aluminum center of 8. Through microscopic investigation of the material obtained during the isolation of 9, we could observe a few slight orange plates that had formed throughout work-up. To our delight, these were suitable for SC-XRD studies and revealed a peculiar decomposition product (Scheme 4a).
The structure refinement unveiled a dimeric structure, in which the Cp* ligands are (i) missing and (ii) are replaced by another equivalent of the former di(2-pyridyl) ketone (Scheme 4). As can further be seen here, the radical anions had undergone C–C coupling to form a dianionic chelate which holds together a dinuclear aluminum complex. As predicted from the spin density distributions in 8 and 9 C–C coupling occurs via the highest spin density location. The overall steric constraints of complex 10 force C–C bond elongation to 1.64 Å. Note that a standard C–C bond has a bond length of about 1.50–1.55 Å.66 Conclusively, the associated Wiberg bond index reveals a low value of 0.703 for each C–C bond, which may declare these bonds as weak. IBO analysis sheds further light on the nature of these bonds and reveals typical σ-bonding interactions (Scheme 4b). Both C atoms contribute equally according to IAO partial charge distributions of 0.95/0.95 in both C–C bonds of interest and are thus perfectly covalent.
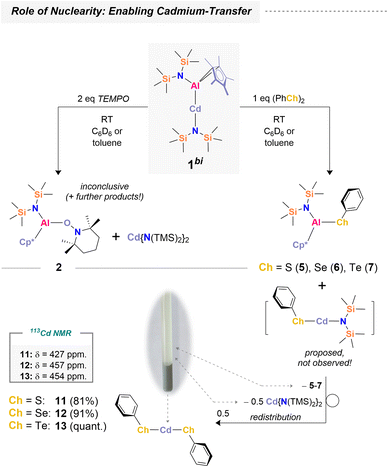
Influence of nuclearity
Having demonstrated selective Al(II) transfer via1tri, we next explored the bimetallic species 1bi to probe the role of nuclearity and investigate Cd(I) transfer – that is, retaining cadmium in the presence of a radical scavenger (Scheme 5). 1tri serves as an efficient reagent, selectively transferring aluminum complex fragments through cadmium extrusion, with convenient work-up by filtration leaving essentially only the desired products in solution (see SI). The bimetallic congener 1bi was expected to show similar aluminum transfer chemistry, but cadmium behavior was anticipated to differ. The absence of a second aluminum center may enhance cadmium stabilization via the strongly coordinating [{N(TMS)2}]− ligand,67 allowing cadmium to be maintained as Cd(I), paralleling the chemistry on the aluminum side. Reaction of 1bi with TEMPO proceeded inconclusively. Although 2 and [Cd({N(TMS)2})2] were observed as products (Fig. S55), further cadmium-containing species could not be identified. Given the limited success with TEMPO, we turned to dichalcogenides ((PhCh)2; Ch = S, Se, Te) as radical scavengers. In these cases, complete conversion of 1bi occurred within seconds, accompanied by formation of grey precipitates. 1H NMR spectroscopy confirmed clean formation of the corresponding [({N(TMS)2})(Cp*)Al(Ch-Ph)] species, 5–7. Interestingly, formation of [Cd({N(TMS)2})2] was similarly observed, but integration revealed only 0.5 equivalents, corresponding to a 2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 mixture of 5–7 and cadmium amide (Ch = S: Fig. S33–S35; Ch = Se: Fig. S39–S42; Ch = Te: Fig. S46–S50).
1 mixture of 5–7 and cadmium amide (Ch = S: Fig. S33–S35; Ch = Se: Fig. S39–S42; Ch = Te: Fig. S46–S50).
Analysis of the reaction stoichiometry indicates that 0.5 equivalents of Cd(ChPh)2 precipitate from solution. The formation of Cd(ChPh)2 is expected to proceed via heteroleptic precursors [Cd{N(TMS)2}(ChPh)], which rapidly redistribute to yield 0.5 equivalents each of [Cd(ChPh)2] and [Cd({N(TMS)2})2]. All three cadmium chalcogenophenolates (Cd(ChPh)2; Ch = S (11), Se (12), Te (13)) are poorly soluble in non-coordinating and even many coordinating solvents, due to formation of extended 3D network structures composed of adamantane units.68–70 Dissolution of isolated 11–13 in pyridine-d5 confirmed their formation by NMR spectroscopy (Scheme 5; Ch = S: Fig. S36–S38; Ch = Se: Fig. S43–S46; Ch = Te: Fig. S51–S54). In summary, the bimetallic congener 1bi exhibits similar aluminum transfer reactivity to 1tri, but cadmium remains bound in the form of cadmium amide or chalcogenide species, suppressing the formation of elemental Cd0. Isolation of the cadmium chalcogenides was achieved in >80% yield after workup.
Extended quantum chemical calculations
As previously demonstrated for 1bi and 1tri, intrinsic bond orbital (IBO) analysis provides an excellent tool to visualize the participation of each atom in bonding.53 These analyses reveal that the Al–Cd interaction is strongly covalent, yet its degree of covalency is tunable depending on the ligand environment. Experimentally (vide supra), we provided evidence for radical dissociation, confirmed through successful trapping. The plausibility for accessible and transferable Al(II) fragments within Al–Cd covalent bonds motivated us to further investigate the nature of the Al–Cd interactions computationally. We began with a computational fragmentation study of 1tri, following robust models that mimic both homo- and heterolytic bond cleavage. Calculations at the TPSS-D4 CPCM(benzene)/def2-QZVPP//r2SCAN-3c/CPCM(benzene)/H-opt level of theory (see SI for details), revealed that homolytic Al–Cd bond dissociation is by far most favorable (as compared to heterolytic [Al]–[Cd] bond cleavage yielding [Al]+ + [Cd]− or [Al]− + [Cd]+). The bond dissociation energies are favored by more than 250 kJ mol−1 (Scheme 6). These results suggest that fragmentation furnishing Al(II) may indeed yield thermodynamically viable pathways. This is only true though, when offering suitable reagents: a dissociation without radical scavenger is not expected due to the prohibitively high dissociation energy. To complement these thermodynamic considerations, we thus investigated the kinetic accessibility of the Al/Cd bonds in 1tri. As a representative system, we focused on the formation mechanism of 2, which (i) is computationally tractable and (ii) reflects a prototypical radical scavenging experiment. Additionally, thermodynamic considerations were extended to the formation of 3 and 5–9, providing a broader view of the energetic landscape associated with Al(II) transfer and trapping. Our calculations (TPSS-D4 CPCM(benzene)/def2-QZVPP//r2SCAN-3c) indicate that TEMPO can approach the trimetallic framework of 1trivia multiple pathways (Fig. 1).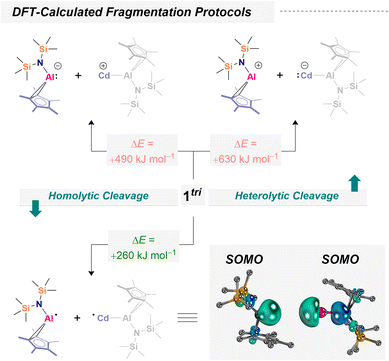 | ||
| Scheme 6 Quantum chemical assessment of vertical homo- and heterolytic cleavage energies of the Al–Cd bond in a trimetallic cadmium aluminyl. Energies and frontier orbitals at the TPSS-D4 CPCM(benzene) def2-QZVPP//r2SCAN-3c CPCM(benzene)/H-opt level of theory based on literature-extracted fragments of 1tri.53 Note: because all atoms except hydrogen were frozen during geometry relaxation, only electronic energies (E) were used for fragmentation comparisons; vibrational thermochemistry was not applied. These calculations furthermore shall not imply that homolytic cleavage takes place without a substrate. | ||
 | ||
| Fig. 1 DFT-derived mechanism for Al(II) transfer at 1tri using TEMPO as well as further thermodynamic considerations at the TPSS-D4 CPCM(benzene) def2-QZVPP//r2SCAN-3c level of theory. Indicated bond lengths at transition states and intermediates are given in Å. Cd0 was modelled as a small Cd8 metal cluster that was extracted from the solid-state structure71 of cadmium. | ||
The chemically intuitive route involves initial coordination of TEMPO to cadmium, which is sterically accessible, forming intermediate IM1A. This step is endergonic by +46 kJ mol−1. Subsequently, TEMPO attacks one of the aluminum atoms, activating the Al–Cd bond through a transition state on the doublet surface. This step is slightly endergonic (TS1A: ΔG‡ = +56 kJ mol−1) and leads to intermediate IM2A, which is slightly stabilized relative to the starting materials (ΔG = −6 kJ mol−1). Spin density analysis of IM2A shows that the unpaired electron is delocalized between both Al centers and Cd, suggestive of a three-electron three center (3e3c) interaction (Fig. S80). Although this intermediate could not be trapped experimentally due to rapid reaction completion, it highlights the potential for peculiar electron deficient bonding between two different metals as an exciting target for future synthetic investigations. Further attack by TEMPO, spin reorganization, and cadmium liberation complete the pathway. The overall energy gain for the addition of two equivalents of TEMPO to 1tri, forming two equivalents of 2, is substantial (ΔG = −190 kJ mol−1; −95 kJ mol−1 per equivalent). A second computationally accessible pathway involves a backside attack of TEMPO (IM1B, ΔG = +48 kJ mol−1) between the supporting ligands [Cp*]− and [{N(TMS)2}]−. Electron transfer to TEMPO in this scenario is kinetically less favorable, with a transition state associated with a Gibbs energy of ΔG‡ = +117 kJ mol−1 (TS1B), making the pathway inaccessible at ambient temperature. Formation of IM2B (ΔG = +47 kJ mol−1) is thus less relevant, although the structural motif of IM2B is similar to that of IM2A. Thermodynamic considerations for other experimentally observed products reveal consistent trends. Addition of benzophenone to form 8 yields ΔG = −56.5 kJ mol−1 per equivalent upon cadmium extrusion, which is the least energetically favored scenario and correlates with the observed decomposition of 8 during work-up. In contrast, formation of 9 is highly favorable (ΔG = −193.5 kJ mol−1 per equivalent), consistent with its stability in solution and successful isolation. The formation of 3 is energetically similar to that of 2 (ΔG = −89 kJ mol−1 per equivalent). The chalcogenide species 5–7 display decreasing thermodynamic stability from S (5) to Se (6) to Te (7), in agreement with their observed experimental stabilities: 5 remains stable for months under glovebox conditions at −30 °C, 6 decomposes over a few weeks, and 7 decomposes within approximately one week under the same conditions (see SI: Sections 3.4–3.6).
Conclusions
In summary, the Al/Cd platform 1tri unlocks an unprecedented regime of heterometal-guided Al(II) transfer chemistry under exceptionally mild, thermal conditions. The ability to generate and selectively deliver an authentic Al(II) fragment, without photochemical activation, marks a decisive expansion of reactivity accessible from aluminyl-based architectures. The broad substrate scope, spanning persistent nitroxides, dichalcogenides, and ketone-based spin traps, reveals a generalizable fragmentation manifold in which covalent Al–Cd linkages serve as controlled release points for reactive Al(II) species concomitant with heterometal extrusion. Together with corroborating EPR analyses and quantum-chemical insight, these findings are firmly established. Notably, the complementary reactivity of the bimetallic partner 1bi, yields molecular cadmium-based building blocks as well, underscoring that both metals embedded in the Al/Cd framework can be transferred when paired with an appropriate reaction partner. Overall, our results chart a path toward the broader use of Al(II) motifs in radical transformations and small-molecule activation chemistry in a predictable and chemoselective manner.Author contributions
D. H.: formal analysis, methodology, investigation, data curation, writing – review and editing. F. W.: formal analysis, data curation, writing – review and editing. C. L.: funding acquisition, methodology, validation, visualization, formal analysis, resources, writing – review and editing, data curation. F. D.: conceptualization, investigation, funding acquisition, writing – original draft, writing – review and editing, methodology, validation, visualization, formal analysis, project administration, data curation, supervision, resources. All authors contributed to drafting and revising the manuscript and have approved the final version for publication.Conflicts of interest
There are no conflicts to declare.Data availability
CCDC 2516482–2516488 contain the supplementary crystallographic data for this paper.100a–gSupplementary information (SI): ref. 72–99 have been cited in the SI. See DOI: https://doi.org/10.1039/d6sc00437g.
Acknowledgements
We gratefully thank Prof. Dr Dominik Munz for help with computations. Our thanks are extended to Fabian Schüler for his contributions to the computational chemistry part. Dr Denis Kargin and Dr Martin Maurer are gratefully acknowledged for their patience, help, and kind advice regarding the NMR spectroscopic characterization. Astrid Pilz and Dr Clemens Bruhn are acknowledged for their help with X-ray crystallography and measurement time. Martina Frühling-Schwalm is acknowledged for her help with elemental analysis. Philipp Kahler is gratefully acknowledged for his help with gathering EDX data. Prof. Dr Rudolf Pietschnig is gratefully acknowledged for providing the laboratory space, the required infrastructure to conduct this research, and his mentorship. F. D. gratefully acknowledges the FCI (Liebig Fellowship) and DFG (instrumentation grant; grant number 468464668: INST 159/146-1 FUGG) for funding. C. L. gratefully acknowledges funding by the Deutsche Forschungsgemeinschaft (DFG, grant number LI2860/5-1) and the LOEWE Program (LOEWE/4b//519/05/01.002(0002)/85). This project has received funding from the European Research Council (ERC) under the European Union's Horizon 2020 research and innovation program (grant agreement no. 946184).References
- C. Ni, X. Ma, Z. Yang and H. W. Roesky, Eur. J. Inorg. Chem., 2022, e202100929 Search PubMed.
- F. Krämer, Angew. Chem., Int. Ed., 2024, 63, e202405207 CrossRef PubMed.
- S. Nagendran and H. W. Roesky, Organometallics, 2008, 27, 457–492 CrossRef CAS.
- Y. Liu, J. Li, X. Ma, Z. Yang and H. W. Roesky, Coord. Chem. Rev., 2018, 374, 387–415 CrossRef CAS.
- K. Hobson, C. J. Carmalt and C. Bakewell, Chem. Sci., 2020, 11, 6942–6956 RSC.
- J. Hicks, P. Vasko, J. M. Goicoechea and S. Aldridge, Angew. Chem., Int. Ed., 2021, 60, 1702–1713 Search PubMed.
- M. P. Coles and M. J. Evans, Chem. Commun., 2023, 59, 503–519 RSC.
- S. Fernández, S. Fernando and O. Planas, Dalton Trans., 2023, 52, 14259–14286 RSC.
- M. P. Coles, Chem. Commun., 2025, 61, 15125–15144 RSC.
- J. Hicks, P. Vasko, J. M. Goicoechea and S. Aldridge, Nature, 2018, 557, 92–95 CrossRef CAS PubMed.
- S. Kurumada, S. Takamori and M. Yamashita, Nat. Chem., 2020, 12, 36–39 CrossRef CAS PubMed.
- G. Feng, K. L. Chan, Z. Lin and M. Yamashita, J. Am. Chem. Soc., 2022, 144, 22662–22668 CrossRef CAS PubMed.
- S. Kurumada, R. Yamanashi, K. Sugita, K. Kubota, H. Ito, S. Ikemoto, C. Chen, T. Moriyama, S. Muratsugu, M. Tada, T. Koitaya, T. Ozaki and M. Yamashita, Chem.–Eur. J., 2024, 30, e202303073 CrossRef CAS PubMed.
- R. J. Schwamm, M. D. Anker, M. Lein and M. P. Coles, Angew. Chem., Int. Ed., 2019, 58, 1489–1493 CrossRef CAS PubMed.
- M. J. Evans, M. D. Anker, M. G. Gardiner, C. L. McMullin and M. P. Coles, Inorg. Chem., 2021, 60, 18423–18431 CrossRef CAS PubMed.
- T. X. Gentner, M. J. Evans, A. R. Kennedy, S. E. Neale, C. L. McMullin, M. P. Coles and R. E. Mulvey, Chem. Commun., 2022, 58, 1390–1393 RSC.
- R. J. Schwamm, M. P. Coles, M. S. Hill, M. F. Mahon, C. L. McMullin, N. A. Rajabi and A. S. S. Wilson, Angew. Chem., Int. Ed., 2020, 59, 3928–3932 CrossRef CAS PubMed.
- K. G. Pearce, A. Morales, M. S. Hill and C. L. McMullin, Chem.–Eur. J., 2025, 31, e202502197 CrossRef CAS PubMed.
- S. Grams, J. Eyselein, J. Langer, C. Färber and S. Harder, Angew. Chem., Int. Ed., 2020, 59, 15982–15986 CrossRef CAS PubMed.
- R. A. Jackson, A. J. R. Matthews, P. Vasko, M. F. Mahon, J. Hicks and D. J. Liptrot, Chem. Commun., 2023, 59, 5277–5280 RSC.
- M. J. Evans, M. D. Anker, C. L. McMullin, S. E. Neale and M. P. Coles, Angew. Chem., Int. Ed., 2021, 60, 22289–22292 CrossRef CAS PubMed.
- A. Paparo, C. D. Smith and C. Jones, Angew. Chem., Int. Ed., 2019, 58, 11581–11585 CrossRef.
- J. T. Boronski, L. R. Thomas-Hargreaves, M. A. Ellwanger, A. E. Crumpton, J. Hicks, D. F. Bekiş, S. Aldridge and M. R. Buchner, J. Am. Chem. Soc., 2023, 145, 4408–4413 CrossRef CAS PubMed.
- J. T. Boronski, A. E. Crumpton, L. L. Wales and S. Aldridge, Science, 2023, 380, 1147–1149 CrossRef CAS PubMed.
- L. P. Griffin, M. A. Ellwanger, J. Clark, W. K. Myers, A. F. Roper, A. Heilmann and S. Aldridge, Angew. Chem., Int. Ed., 2024, 63, e202405053 CrossRef CAS PubMed.
- J. T. Boronski, L. P. Griffin, C. Conder, A. E. Crumpton, L. L. Wales and S. Aldridge, Chem. Sci., 2024, 15, 15377–15384 RSC.
- M. J. Evans, G. Iliffe, S. E. Neale, C. L. McMullin, J. R. Fulton, M. D. Anker and M. P. Coles, Chem. Commun., 2022, 58, 10091–10094 RSC.
- K. Koshino and R. Kinjo, J. Am. Chem. Soc., 2020, 142, 9057–9062 CrossRef CAS PubMed.
- T. Löwl, J. Maurer, N. Roig, J. Mai, J. Langer, M. Alonso and S. Harder, Eur. J. Inorg. Chem., 2025, e202500050 CrossRef.
- A. O'Reilly, A. M. S. Booth, G. W. A. Smith, M. J. Evans, L. Feng Lim, D. A. Pantazis, N. Cox, C. L. McMullin, J. R. Fulton and M. P. Coles, Chem.–Eur. J., 2025, 31, e202500358 CrossRef PubMed.
- G. W. A. Smith, S. E. Neale, M. J. Evans, X. Li, J. Wang, M. G. Gardiner, C. L. McMullin, J. R. Fulton and M. P. Coles, Chem.–Eur. J., 2025, 31, e202404206 CrossRef PubMed.
- G. Feng and M. Yamashita, J. Am. Chem. Soc., 2024, 146, 28653–28657 CrossRef CAS PubMed.
- K. Sugita and M. Yamashita, Chem.–Eur. J., 2020, 26, 4520–4523 CrossRef CAS PubMed.
- P. Zatsepin, T. Moriyama, C. Chen, S. Muratsugu, M. Tada and M. Yamashita, J. Am. Chem. Soc., 2024, 146, 3492–3497 CrossRef CAS PubMed.
- S. Morisako, S. Watanabe, S. Ikemoto, S. Muratsugu, M. Tada and M. Yamashita, Angew. Chem., Int. Ed., 2019, 58, 15031–15035 CrossRef CAS PubMed.
- B. N. Anand, I. Krossing and H. Nöth, Inorg. Chem., 1997, 36, 1979–1981 CrossRef CAS PubMed.
- S. Sinhababu, M. R. Radzhabov, J. Telser and N. P. Mankad, J. Am. Chem. Soc., 2022, 144, 3210–3221 CrossRef CAS PubMed.
- F. Kallmeier, A. J. R. Matthews, G. R. Nelmes, N. R. Lawson and J. Hicks, Dalton Trans., 2024, 53, 12450–12454 RSC.
- B. J. Graziano, M. V. Vollmer and C. C. Lu, Angew. Chem., Int. Ed., 2021, 60, 15087–15094 CrossRef CAS PubMed.
- J. Hicks, A. Mansikkamäki, P. Vasko, J. M. Goicoechea and S. Aldridge, Nat. Chem., 2019, 11, 237–241 CrossRef CAS PubMed.
- C. Yan, K. Koshino, L. Zhu and R. Kinjo, Nat. Synth., 2024, 3, 858–866 Search PubMed.
- H.-Y. Liu, S. E. Neale, M. S. Hill, M. F. Mahon and C. L. McMullin, Dalton Trans., 2022, 51, 3913–3924 RSC.
- H.-Y. Liu, J. Kenar, S. E. Neale, M. Garofalo, M. S. Hill, C. L. McMullin, M. F. Mahon and E. Richards, Organometallics, 2024, 43, 3074–3086 CrossRef CAS.
- C. McManus, J. Hicks, X. Cui, L. Zhao, G. Frenking, J. M. Goicoechea and S. Aldridge, Chem. Sci., 2021, 12, 13458–13468 RSC.
- H. Y. Liu, R. J. Schwamm, M. S. Hill, M. F. Mahon, C. L. McMullin and N. A. Rajabi, Angew. Chem., Int. Ed., 2021, 60, 14390–14393 Search PubMed.
- C. Bakewell, B. J. Ward, A. J. P. White and M. R. Crimmin, Chem. Sci., 2018, 9, 2348–2356 RSC.
- M. M. D. Roy, J. Hicks, P. Vasko, A. Heilmann, A. M. Baston, J. M. Goicoechea and S. Aldridge, Angew. Chem., Int. Ed., 2021, 60, 22301–22306 CrossRef CAS PubMed.
- F. Dankert and E. Hevia, Chem.–Eur. J., 2024, 30, e202304336 CrossRef CAS PubMed.
- J. Weßing, C. Göbel, B. Weber, C. Gemel and R. A. Fischer, Inorg. Chem., 2017, 56, 3517–3525 CrossRef.
- L. P. Griffin, M. A. Ellwanger, A. E. Crumpton, M. M. D. Roy, A. Heilmann and S. Aldridge, Angew. Chem., Int. Ed., 2024, 63, e202404527 CrossRef CAS PubMed.
- X. Zhang and L. L. Liu, Angew. Chem., Int. Ed., 2021, 60, 27062–27069 CrossRef CAS.
- F. Dankert, A. Kadriu and E. Hevia, ChemRxiv, 2024, preprint, DOI:10.26434/chemrxiv-2024-r1wz8.
- D. Herle, S. Sommer and F. Dankert, J. Am. Chem. Soc., 2025, 147, 33315–33323 Search PubMed.
- J. Hemsworth, J. Tomeček, A. Hasija, G. Whitehead, N. Kaltsoyannis, D. Mills and D. Willcox, Chem.–Eur. J., 2026, e03282 CrossRef PubMed , early view..
- G. Feng, K. L. Chan, Z. Lin and M. Yamashita, J. Am. Chem. Soc., 2024, 146, 7204–7209 CrossRef CAS.
- R. P. Singh, K. P. Quirion, J. Telser, D. H. Ess and N. P. Mankad, J. Am. Chem. Soc., 2025, 147, 12715–12721 CrossRef CAS PubMed.
- R. P. Singh and N. P. Mankad, JACS Au, 2025, 5, 2076–2088 CrossRef CAS.
- L. W. T. Parsons and L. A. Berben, Acc. Chem. Res., 2024, 57, 1087–1097 CrossRef CAS PubMed.
- L. A. Berben, Chem.–Eur. J., 2015, 21, 2734–2742 CrossRef CAS PubMed.
- M. Nakamoto, T. Yamasaki and A. Sekiguchi, J. Am. Chem. Soc., 2005, 127, 6954–6955 CrossRef CAS PubMed.
- D. Mandal, T. I. Demirer, T. Sergeieva, B. Morgenstern, H. T. A. Wiedemann, C. W. M. Kay and D. M. Andrada, Angew. Chem., Int. Ed., 2023, 62, e202217184 CrossRef CAS PubMed.
- F. Dankert, E. Sabater, P. Salvador and E. Hevia, ChemRxiv, 2024, preprint, DOI:10.26434/chemrxiv-2024-x5btd.
- Y. Chen, C. Lin, C. Li, J. Huang, L. Chang and T. Lee, Chem.–Eur. J., 2008, 14, 9747–9753 CrossRef CAS PubMed.
- J. Beckmann, J. Bolsinger and A. Duthie, Chem.–Eur. J., 2011, 17, 930–940 CrossRef CAS PubMed.
- M. Gomberg, J. Am. Chem. Soc., 1900, 22, 757–771 CrossRef.
- F. H. Allen, O. Kennard, D. G. Watson, L. Brammer, A. G. Orpen and R. Taylor, J. Chem. Soc., Perkin Trans. 2, 1987, S1–S19 RSC.
- A. Casnati and C. G. Werncke, Chem. Commun., 2025, 61, 14324–14339 Search PubMed.
- D. Craig, I. G. Dance and R. Garbutt, Angew. Chem., Int. Ed. Engl., 1986, 25, 165–166 CrossRef.
- K. S. Anjali and J. J. Vittal, Inorg. Chem. Commun., 2000, 3, 708–710 CrossRef.
- M. P. Campos and J. S. Owen, Chem. Mater., 2016, 28, 227–233 CrossRef CAS.
- D. A. Edwards, W. E. Wallace and R. S. Craig, J. Am. Chem. Soc., 1952, 74, 5256–5261 CrossRef CAS.
- A. M. Borys, Organometallics, 2023, 42, 182–196 CrossRef CAS.
- C. Ganesamoorthy, S. Loerke, C. Gemel, P. Jerabek, M. Winter, G. Frenking and R. A. Fischer, Chem. Commun., 2013, 49, 2858 RSC.
- H. Bürger, W. Sawodny and U. Wannagat, J. Organomet. Chem., 1965, 3, 113–120 CrossRef.
- S. Stoll and A. Schweiger, J. Magn. Reson., 2006, 178, 42–55 CrossRef CAS PubMed.
- T. Heurich, V. Nesterov, G. Schnakenburg, Z. Qu, S. Grimme, K. Hazin, D. P. Gates, M. Engeser and R. Streubel, Angew. Chem., Int. Ed., 2016, 55, 14439–14443 CrossRef CAS PubMed.
- R. D. Deslattes, E. G. Kessler, P. Indelicato, L. de Billy, E. Lindroth and J. Anton, Rev. Mod. Phys., 2003, 75, 35–99 CrossRef CAS.
- W. T. Elam, B. D. Ravel and J. R. Sieber, Radiat. Phys. Chem., 2002, 63, 121–128 CrossRef CAS.
- G. M. Sheldrick, Acta Crystallogr., Sect. A: Found. Adv., 2015, 71, 3–8 CrossRef.
- G. M. Sheldrick, Acta Crystallogr., Sect. C: Struct. Chem., 2015, 71, 3–8 Search PubMed.
- O. V. Dolomanov, L. J. Bourhis, R. J. Gildea, J. A. K. Howard and H. Puschmann, J. Appl. Crystallogr., 2009, 42, 339–341 CrossRef CAS.
- X-AREA, Integrated LANA, XRED32, Stoe & Cie, Darmstadt, Germany, 2020 Search PubMed.
- K. Brandenburg, Diamond, Version 4.6.8, Bonn, 2022 Search PubMed.
- F. Neese, Wiley Interdiscip. Rev.: Comput. Mol. Sci., 2012, 2, 73–78 CAS.
- F. Neese, Wiley Interdiscip. Rev.: Comput. Mol. Sci., 2022, 12, e1606 Search PubMed.
- S. Grimme, A. Hansen, S. Ehlert and J.-M. Mewes, J. Chem. Phys., 2021, 154, 064103 CrossRef CAS.
- J. Tao, J. P. Perdew, V. N. Staroverov and G. E. Scuseria, Phys. Rev. Lett., 2003, 91, 146401 CrossRef PubMed.
- F. Weigend and R. Ahlrichs, Phys. Chem. Chem. Phys., 2005, 7, 3297–3305 RSC.
- E. Caldeweyher, C. Bannwarth and S. Grimme, J. Chem. Phys., 2017, 147, 034112 CrossRef PubMed.
- R. Izsák and F. Neese, J. Chem. Phys., 2011, 135, 144105 CrossRef.
- F. Neese, F. Wennmohs, A. Hansen and U. Becker, Chem. Phys., 2009, 356, 98–109 CrossRef CAS.
- K. A. Peterson, D. Figgen, E. Goll, H. Stoll and M. Dolg, J. Chem. Phys., 2003, 119, 11113–11123 CrossRef CAS.
- G. L. Stoychev, A. A. Auer and F. Neese, J. Chem. Theory Comput., 2017, 13, 554–562 CrossRef CAS PubMed.
- A. V. Marenich, C. J. Cramer and D. G. Truhlar, J. Phys. Chem. B, 2009, 113, 6378–6396 CrossRef CAS PubMed.
- G. Knizia, IBOView – A Program for Chemical Analysis, http://www.iboview.org/ Search PubMed.
- G. Knizia and J. E. M. N. Klein, Angew. Chem., Int. Ed., 2015, 54, 5518–5522 CrossRef CAS PubMed.
- Chemcraft – Graphical Software for Visualization of Quantum Chemistry Computations, https://chemcraftprog.com Search PubMed.
- A. D. Becke, Phys. Rev. A: At., Mol., Opt. Phys., 1988, 38, 3098–3100 CrossRef CAS PubMed.
- T. Lu and F. Chen, J. Comput. Chem., 2012, 33, 580–592 CrossRef CAS PubMed.
- (a) CCDC 2516482: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2qglvn; (b) CCDC 2516483: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2qglwp; (c) CCDC 2516484: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2qglxq; (d) CCDC 2516485: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2qglyr; (e) CCDC 2516486: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2qglzs; (f) CCDC 2516487: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2qgm0v; (g) CCDC 2516488: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2qgm1w.
Footnote |
| † Dedicated to Prof. Udo Radius on the occasion of his 60th birthday. |
| This journal is © The Royal Society of Chemistry 2026 |