Open Access Article
Open Access ArticleStereospecific alkenylation of carboranes: copper-catalyzed access to pyridylcarboranyl alkenes
Ping
Li†
a,
Xiang
Li†
a,
Liyan
Wang†
b,
Mengmeng
Wang
b,
Deshuang
Tu
*b,
Hong
Yan
 *b,
Jian
Lu
*c and
Ju-You
Lu
*b,
Jian
Lu
*c and
Ju-You
Lu
 *a
*a
aSchool of Chemistry and Chemical Engineering, Hainan University, Haikou, 570228, China. E-mail: lujy@hainanu.edu.cn
bState Key Laboratory of Coordination Chemistry, School of Chemistry and Chemical Engineering, Nanjing University, Nanjing, 210023, China. E-mail: tudeshuang@nju.edu.cn; hyan1965@nju.edu.cn
cState Key Laboratory of Fluorine & Nitrogen Chemicals, Xi'an Modern Chemistry Research Institute, Xi'an, 710065, China. E-mail: lujian204@263.net
First published on 11th February 2026
Abstract
The direct functionalization of carbon vertices in polyhedral carboranes has received considerable attention because of their wide applications in functional materials, catalysis, and pharmaceuticals. However, achieving direct C–H bond activation of o-carboranes remains a challenge, as it typically requires strong bases to facilitate the deprotonation of the C–H bonds. Herein, we report a copper-catalyzed stereospecific C–H alkenylation of o-carboranes using Na2CO3 as a mild base. Remarkably, the reaction exhibits stereospecificity, allowing for switchable syn- or anti-selectivity simply by employing the E- or Z-configuration of the alkenyl halide coupling partner. Structurally diverse E- and Z-alkenylcarboranes are accessible by this protocol, among which the latter are difficult to access by existing methods. The synthetic utility of this methodology was demonstrated by the late-stage modification of amino acids, sugars, and drug molecules. Furthermore, the synthesized carboranylethylene scaffolds exhibit aggregation-induced emission and bright solid-state luminescence, with quantum yields reaching 67%. Thus, this work unveils a Cu(I)/Cu(III) catalytic model for the C–H activation of carboranes and establishes pyridylcarborane–alkene hybrids as a new class of functional, bioisosteric architectures.
Introduction
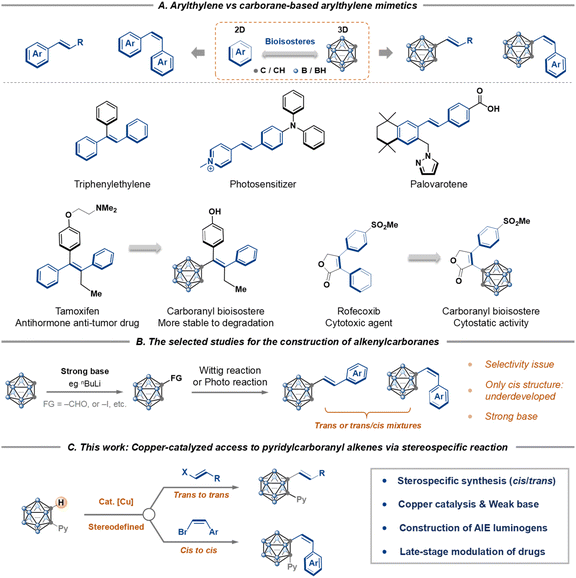
Carboranes, a class of three-dimensional icosahedral boron–carbon clusters, have emerged as unique and valuable building blocks in molecular design.1–4 Their spherical geometry, high boron content, and three-dimensional σ-aromaticity offer a compelling “escape from flatland,” positioning them as superior bioisosteric replacements for two-dimensional arenes in the development of pharmaceuticals, bioactive agents, and advanced materials (Scheme 1A).5Within this paradigm, the arylethylene motif is a ubiquitous and critical framework in functional molecules. It is a cornerstone of structures ranging from aggregation-induced emission (AIE)-active luminogens6 and phototherapeutic agents7 to numerous FDA-approved drugs (Scheme 1A).8 Inspired by this prevalence, we recognized a great opportunity: the development of carborane-based mimetics of these valuable arylethylene systems. Such a replacement, analogous to a carborane-based analogue of tamoxifen, holds immense promise for creating novel boron-cluster-functionalized molecules with tailored properties.9 Consequently, over the past decades, tremendous efforts have been made for the development of carborane C-alkenylation strategies, including condensation reactions,10a Ullmann-type coupling,10b coupling of metal–carboryne complexes,10c Wittig reactions,10d and photo-induced reactions.10e,f However, synthetic access to such carbon-substituted o-carborane architectures is limited, and stereochemical control represents a significant hurdle.10,11 In particular, a general and stereospecific route to Z-configured carboranylethylenes has been especially elusive (Scheme 1B).
To overcome this limitation, here we conceived a strategy based on a stereospecific reaction. Namely, the stereochemical information encoded in readily available Z/E-arylethylenes could be transferred12 through a catalytic cycle to dictate the geometry of the final carboranylethylene products (Scheme 1C). This approach would enable precise stereocontrol, a critical factor for function in both biological and materials contexts. Herein, we report the realization of this strategy through a copper-catalyzed stereospecific C–H alkenylation of o-carboranes. This method provides efficient access to a diverse range of pyridylcarboranyl alkenes with high fidelity in stereochemical transfer. The resulting C-alkenylcarborane scaffolds serve as versatile building blocks, which we have leveraged to construct AIE-active luminogens with bright and tunable emission and to develop novel drug candidates, thereby demonstrating their significant potential in both materials science and medicinal chemistry.
Results and discussion
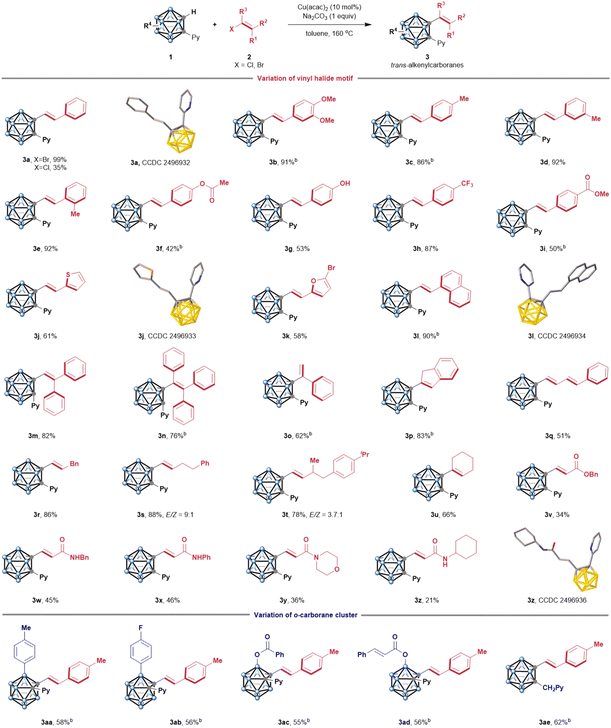
Initially, we investigated the C–H alkenylation of 1-(pyridin-2-yl)-o-carborane (1a) with (E)-1-(2-bromovinyl)naphthalene (2l) in the presence of various copper catalysts. After many attempts (see the SI for details), gratifyingly, with the use of a catalytic amount of Cu(acac)2, the cage C–H alkenylation provided the desired C-alkenylcarborane 3l in a 90% isolated yield.Next, we evaluated the substrate scope of the copper-catalyzed stereospecific C–H alkenylation of o-carboranes with vinyl electrophilics (Scheme 2). First, we tested various vinyl halides bearing diverse functional groups. The reaction of E-bromostyrene afforded alkenylcarborane 3a with exclusive retention of the configuration, indicating that a stereospecific process occurs. Several vinyl halides were compatible, including inert chlorides and reactive bromides. The former is a challenging substrate for conventional coupling reactions. Various aryl-conjugated β-vinyl halides possessing electron-donating (3b–3g) and electron-withdrawing (3h, and 3i) groups were tested, affording the desired products with excellent anti-selectivity and high yields. A wide variety of functional groups are tolerated, including valuable handles for further functionalization: aryl bromides, esters, and free hydroxyl groups. The cage C–H coupling was not limited to heterocyclic compounds, including thiophene (3j) and furan (3k). Naphthyl-conjugated vinyl bromide afforded 3l in 90% yield. In contrast, the conversion rate was significantly improved, and no syn-isomer product was detected,10e thereby greatly simplifying product isolation. Notably, polysubstituted olefins such as 1,1-diphenyl (3m) and 1,1,2-triphenyl (3n) scaffolds were well tolerated, providing boron cluster-based AIE-active luminogens (AIEgens, vide infra). Other vinyl electrophiles were coupled effectively, including sterically hindered α-bromostyrene (3o), 2-bromoindene (3p), and (4-bromo-1,3-butadien-1-yl)benzene (3q). In the previous report, substrates possessing aliphatic vinyl resulted in β′-H elimination to form a mixture of “Heck-type” and “ene-reaction-type” products.10c Here, several aliphatic bromoalkenes were exclusively converted to alkenylcarboranes (3r–3u). To investigate the robustness of this method, we also examined a mixture of Z- and E-haloalkenes, which we envisioned could perturb the Z/E-selectivity by competitive reactions. Notably, the maintenance of the Z![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) E ratio of products 3s and 3t further confirmed that the cage C–H alkenylation is a highly stereoselective process. Esters and amides conjugated with a double bond were viable for this protocol (3v–3z), among which amides are widely used as directing groups for C–H activation in organic synthesis.
E ratio of products 3s and 3t further confirmed that the cage C–H alkenylation is a highly stereoselective process. Esters and amides conjugated with a double bond were viable for this protocol (3v–3z), among which amides are widely used as directing groups for C–H activation in organic synthesis.
Next, we evaluated substituted o-carboranes. Various B(3)-substituted o-carboranes were compatible with this protocol. Sterically hindered B(3)-aryl carboranes bearing electron-donating (3aa) and electron-withdrawing (3ab) groups on the phenyl group all gave gratifying outcomes. Acyl-protected B(3)-hydroxyl o-carboranes worked well to provide the target products 3ac and 3ad, respectively. Additionally, picolyl group-directed cage C–H alkenylation was also achieved using copper catalysis (3ae). The reaction of 1-Ph-o-carborane (1f) or unsubstituted o-carborane (1g) and trans-β-bromostyrene (2a) did not proceed as the starting materials were fully recovered (Scheme S6). These results revealed that the pyridyl group, which acts as a directing group to facilitate the activation of C–H bonds, is essential and also provides a versatile platform for expanding the chemical space of boron cluster-based structural diversity.
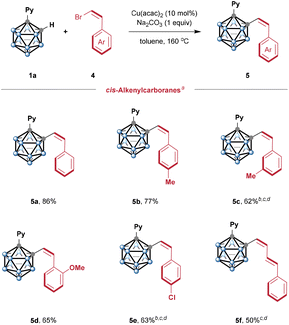
Encouraged by these results, we extended this method to bromoalkenes with a more challenging Z configuration (Scheme 3). It should be noted that modular synthesis of Z-alkenylcarborane through various methods has been a long-standing challenge. Using the Wittig reaction of C-formyl-o-carborane with a triphenyl phosphonium ylide produced alkenylcarborane as a 6![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 4 mixture of E/Z isomers.10f The photoalkenylation of iodocarborane with 2-vinylnaphthalene gave the alkenylated product in 22% yield with a 5.3
4 mixture of E/Z isomers.10f The photoalkenylation of iodocarborane with 2-vinylnaphthalene gave the alkenylated product in 22% yield with a 5.3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 E
1 E![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Z ratio.10e Nevertheless, these early studies prompted us to investigate whether this copper catalysis can achieve syn-selective alkenylation by cage C–H activation. Gratifyingly, employing (Z)-β-bromostyrene as a coupling partner exclusively afforded the desired syn-alkenylation product 5a with excellent yield. A diverse range of Z-bromoalkenes, featuring substituents at different positions on the phenyl ring, were generally well tolerated, affording Z-alkenylcarboranes (5b–5e) with excellent syn-selectivity and good yields. Additionally, the 1,3-dienyl substrate was also competent in this syn-selective process (5f).
Z ratio.10e Nevertheless, these early studies prompted us to investigate whether this copper catalysis can achieve syn-selective alkenylation by cage C–H activation. Gratifyingly, employing (Z)-β-bromostyrene as a coupling partner exclusively afforded the desired syn-alkenylation product 5a with excellent yield. A diverse range of Z-bromoalkenes, featuring substituents at different positions on the phenyl ring, were generally well tolerated, affording Z-alkenylcarboranes (5b–5e) with excellent syn-selectivity and good yields. Additionally, the 1,3-dienyl substrate was also competent in this syn-selective process (5f).
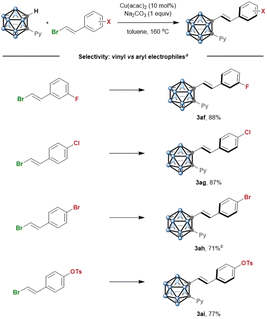
To probe the inherent selectivity in the competitive coupling of vinyl and aryl electrophiles, we further investigated the carboranylation of various electrophiles (Scheme 4). The Cu catalysis offers a selectivity that is complementary to those achieved by conventional coupling protocols. For example, our group has recently reported the C–H arylation of o-carborane with aryl electrophiles.13 Here, under Cu catalysis, employing electrophiles containing a vinyl bromide and an aryl electrophile, including the fluoride (3af), chloride (3ag), and even highly reactive bromide (3k and 3ah) and tosylate (3ai), provides completely exclusive selectivity for the C-alkenylated products.
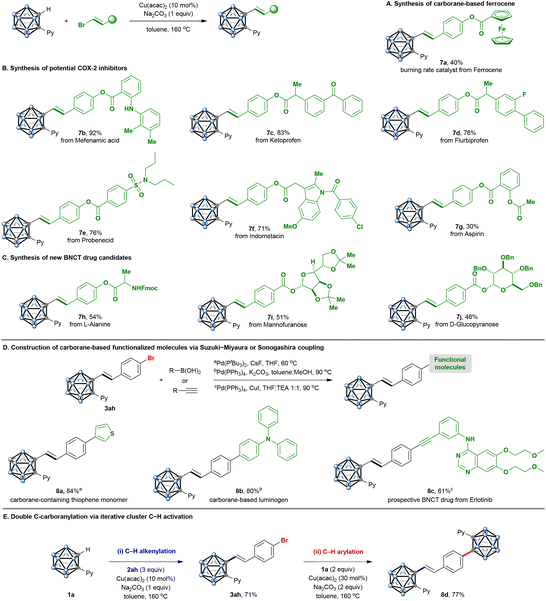
Given the fact that the current protocol offers a powerful reaction platform for the C–H alkenylation of o-carboranes, its synthetic applications were further investigated. First, carborane-containing ferrocenes are widely used in composite solid propellants due to their ability to regulate the burning rate.14 We were pleased to observe that a ferrocene-derived bromoalkene was successfully incorporated with o-carborane (7a) (Scheme 5A). Moreover, to underscore the utility of the cage C–H alkenylation for drug-discovery purposes, we examined a range of active pharmaceutical ingredients (Scheme 5B). Vinyl bromides bearing bioactive structures such as mefenamic acid (7b), ketoprofen (7c), flurbiprofen (7d), probenecid (7e), indometacin (7f), and aspirin (7g) were coupled to o-carborane in good to excellent yields. The compatibility of unprotected amines (7b) with the reaction conditions is noteworthy for medicinal chemistry, considering the ubiquity of this functionality among lead compounds.
The combination of carboranes and amino acid or glycoside fragments in one structure can serve as a basis for obtaining promising targeted boron delivery agents for cancer treatment by boron neutron capture therapy (BNCT).5L-Alanine-derived bromoalkene could be readily converted to a boronated amino acid (7h). Moreover, mannofuranose- and D-glucopyranose-containing bromoalkenes delivered boron clusters conjugated with glycosides (7i and 7j). The amenability of this new chemistry to complex drug molecules demonstrates the potential of this transformation to achieve late-stage diversification in pharmaceutically relevant settings (Scheme 5C).
To demonstrate the synthetic utility of the reaction, we performed the derivatization of 3ah (Scheme 5D). The metal-catalyzed cross-coupling toolkit can be employed for the structural elaboration of the aryl bromide group for the cage C-vinylated products. For example, the Suzuki–Miyaura reaction of 3ah proceeded smoothly, delivering the C–C coupling products 8a and 8b in useful to good yields. Specifically, o-carborane-containing triphenylamine is expected to be a potential photofunctional molecule.3 Moreover, to rapidly produce drug-like molecular architecture from native functionality, erlotinib, a popularly used lung cancer-targeting drug, was chosen to combine with 3ah by the facile Sonogashira reaction; thus, the BNCT drug candidate 8c could be prepared from o-carboranes in two steps.
Finally, successful iterative C–H activation of o-carborane was also demonstrated, providing a novel 2-fold C–H-functionalized bis(o-carborane) 8d in good yield (Scheme 5E). The tactical union of two powerful, recently invented transformations for chemical synthesis (catalytic cage C–H alkenylation and arylation) enables access to previously challenging molecular space.13
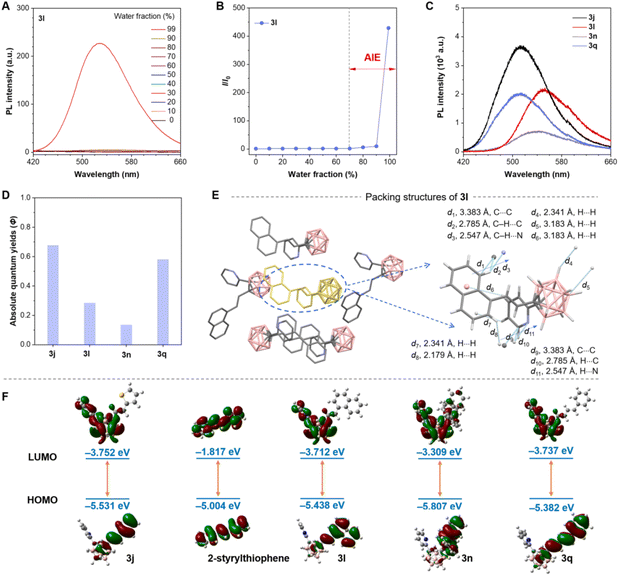
Carboranes have emerged as versatile building blocks for constructing luminescent materials. Herein, we present an efficient synthetic strategy to access hybrid conjugates integrating boron clusters with π-conjugated units. These hybrids are anticipated to exhibit efficient luminescence in the aggregate state, leveraging the synergistic steric bulk and electron-withdrawing nature of the carborane moiety.14,15 To validate this hypothesis, compounds 3j, 3l, 3n, and 3q were selected for photophysical characterization. As anticipated, photoluminescence (PL) studies in mixed solvents revealed pronounced AIE behavior (Fig. 1A and B). For instance, at water fractions below 70%, negligible emission intensity was observed—consistent with typical AIE luminogens. Upon increasing the water fraction beyond this threshold, a dramatic enhancement in PL intensity occurred, confirming AIE characteristics.16 Analogous behavior was observed for other carborane-based derivatives (Fig. S1–S3). Notably, these compounds exhibit bright, color-tunable emission in the solid state (Fig. 1C) with high absolute quantum yields (ϕ) of up to 67% (Fig. 1D and S4–S7), rivaling classical AIE luminogens such as tetraphenylethene (TPE).17
To elucidate the origin of efficient solid-state luminescence, we analyzed single-crystal X-ray structures of 3l (Fig. 1E). The incorporation of the carborane unit induces a V-shaped molecular geometry, which mitigates π–π stacking interactions between aromatic rings. Instead, multiple intermolecular interactions such as H⋯H and C–H⋯N contacts dominate the crystal packing. This structural motif suppresses the formation of non-emissive exciplex species, rationalizing the observed emission efficiency.18 Complementary theoretical calculations reveal that the highest occupied molecular orbitals (HOMOs) of these compounds reside predominantly on the arylvinyl moieties, while the lowest unoccupied molecular orbitals (LUMOs) significantly delocalize onto the o-carborane unit (Fig. 1F). This electronic separation strongly supports emission originating from charge-transfer (CT) states. Notably, the electronic structure of these carborane-based systems contrasts sharply with their phenyl-substituted counterparts (e.g., 2-styrylthiophene and 1-styrylnaphthalene, Fig. S8). Given the tunable properties imparted by the carborane moiety, these molecular frameworks present compelling opportunities for designing advanced photofunctional materials.
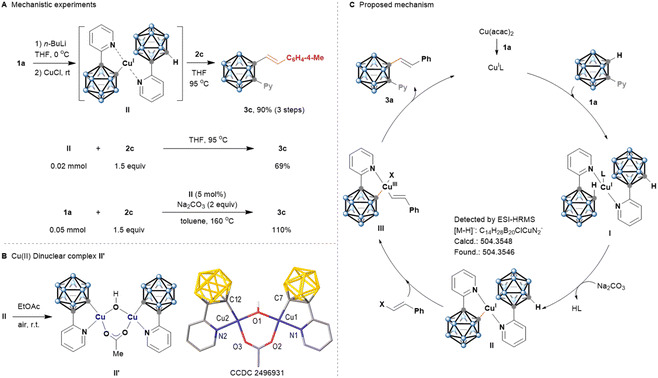
Preliminary mechanistic studies were conducted to gain further insight into the cage C–H alkenylation (Fig. 2A). First, the reaction of 1-(pyridin-2-yl)-o-carborane 1a with n-BuLi, followed by the addition of 1.5 equiv. of CuCl, afforded a key cyclometalated copper(I) intermediate (II), subsequent reaction with 1-[(1E)-2-bromoethenyl]-4-methylbenzene (2c) produced the C-alkenylated o-carborane 3c in 90% total yield. Trials to characterize this intermediate through NMR analysis were unsuccessful. To our delight, HRMS measurement successfully detected the elusive intermediate (II) (Fig. S9). Furthermore, complex (II), when subjected to the catalytic conditions with 1.5 equiv. of 2c as the coupling partner, afforded 3c in 69% yield as the sole product. Under standard conditions, complex (II) catalysed C–H alkenylation efficiently, leading to 3c with excellent yield. These results indicated that formation of a five-membered cyclometalated complex is a key step in the early stages of the catalytic cycle.
Notably, 2-phenylpyridine did not react with 2a under the optimal reaction conditions, indicating a significant difference in reactivity between the 3D delocalized σ aromatic o-carborane and the 2D aromatic benzene (Scheme S7). On the other hand, competition reactions between cis- or trans-bromoalkene substrates were performed, affording a mixture of cis- and trans-pyridylcarboranyl alkenes 5a and 3a in 92% yield with a 60![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 40 Z
40 Z![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) E ratio. These results suggest that cis-bromoalkenes are more reactive (Scheme S9). In addition, radical capture experiments with TEMPO (2,2,6,6-tetramethylpiperidyl-1-oxyl) were conducted. The reaction proceeded unaffected, thus excluding the radical mechanism (Scheme S11).
E ratio. These results suggest that cis-bromoalkenes are more reactive (Scheme S9). In addition, radical capture experiments with TEMPO (2,2,6,6-tetramethylpiperidyl-1-oxyl) were conducted. The reaction proceeded unaffected, thus excluding the radical mechanism (Scheme S11).
To find support for the proposed reaction mechanism, we sought to prepare the hypothesized cyclometalated complex. The isolation and characterization of copper–carborane complexes has been a long-standing challenge, possibly due to their high reactivity or low solubility.19 In the presence of weak base sodium carbonate, the stoichiometric reaction of 1a with copper salt did not generate an observable amount of copper species by 1H NMR. Finally, we found that the reaction of II in ethyl acetate at room temperature afforded a rare copper(II)-carborane complex (II'). The structure of this Cu(II) complex was confirmed by X-ray diffraction. Single-crystal analysis clearly indicated a Cu(II) dinuclear complex with an asymmetric double bridge formed by a µ-carboxylato and a µ-hydroxo ligand, resulting in an exopolyhedral C–Cu bond. The C(2)–Cu distance is 1.978(2) Å, which is consistent with recent reports on the related lithium o-carboranyl cuprate complex.20 The ancillary ligand on each copper center is the non-symmetric C, N chelate 1-pyridylcarboranyl bonded to the metal through a hard-donor nitrogen and a softer σ-bonded carbon atom (Fig. 2B). Surprisingly, complex (II') is the first known identifiable example of copper(II)-o-carborane dinuclear complexes containing an exopolyhedral C–Cu bond. Unfortunately, this complex is not catalytically active for this reaction.
On the basis of our observations and previous reports,13 a plausible reaction mechanism is proposed in Fig. 2C. Reduction of Cu(II) by o-carborane or a base forms Cu(I) species.21 A bidentate chelation of 1a with Cu(I) occurs to give the intermediate (I). Subsequently, in the presence of Na2CO3, cage C–H bond metalation of I generates a key cyclometalated intermediate copper(I)-o-carborane complex (II). Oxidative addition of Cu(I) by alkenyl halide affords the Cu(III) species (III), which undergoes C–C reductive elimination to give products 3a and regenerate the catalytically active Cu(I) complex. It is noteworthy that the double bond geometry of alkenyl halide reactants was fully retained in all of the stereospecific cage C–H alkenylation products.
Conclusions
In summary, we have developed an efficient Cu-catalyzed direct C–H alkenylation of o-carboranes by a stereospecific strategy. The use of easily accessible starting materials, a broad substrate scope, a good functional group tolerance, excellent stereospecific selectivity, mild base Na2CO3, and the versatile functionalization of the C-alkenylated o-carboranes render this method highly practical and appealing. This has enabled the fast construction of a new library of boron cluster-based functionalized molecules, including carborane-based luminogens and BNCT drug candidates. More importantly, these new boron cluster-based luminogens display interesting properties of enhanced solid-state luminous efficiency and AIE effects. This work offers an efficient method to construct boron cluster-based functional molecules, and may have great potential in biological applications and other related fields.Author contributions
J.-Y. L., D. T. and H. Y. conceived of the work. J.-Y. L., D. T., H. Y. and J. L. supervised the project. P. L., X. L., L. W. and M. W. ran the experiments. P. L., X. L., L. W., D. T. and J.-Y. L. analyzed the data. J.-Y. L. and D. T. wrote the manuscript. P. L., X. L., D. T. and J.-Y. L. provided revisions.Conflicts of interest
There are no conflicts to declare.Data availability
Data supporting this study are available in the supplementary information (SI) and further details are available from the authors on reasonable request. Supplementary information: experimental procedures and data for new compounds. See DOI: https://doi.org/10.1039/d5sc10093c.CCDC 2496931–2496934 and 2496936 contain the supplementary crystallographic data for this paper.22a–e
Acknowledgements
This work was supported by the National Natural Science Foundation of China (22161015) and Hainan Provincial Natural Science Foundation of China (221RC448). This work was also supported by the Fundamental Research Funds for the Central Universities (2024300362), the National Natural Science Foundation of China (92461308, 92261202, W2412072, 22401141 and 22571145) and the Natural Science Foundation of Jiangsu Province (BK20241229). The high-performance computing center of Nanjing University is acknowledged.Notes and references
- (a) R. N. Grimes, Carboranes, Elsevier, Oxford, 3rd edn, 2016, p. 283 Search PubMed; (b) J. Poater, C. Viñas, I. Bennour, S. Escayola, M. Solà and F. Teixidor, J. Am. Chem. Soc., 2020, 142, 9396–9407 CrossRef CAS PubMed; (c) J. Poater, C. Viñas, M. Solà and F. Teixidor, Nat. Commun., 2022, 13, 3844–3851 CrossRef CAS PubMed; (d) N. S. Hosmane and R. D. Eagling, Handbook of Boron Science: With Applications in Organometallics, Catalysis, Materials and Medicine, World Scientific, London, 2018 Search PubMed; (e) N. S. Hosmane, Boron Science: new Technologies and Applications, CRC Press, 1st edn, 2012, p. 878 Search PubMed; (f) M. Solà, Nat. Chem., 2022, 14, 585–590 CrossRef PubMed; (g) T. L. Chan and Z. Xie, Chem. Sci., 2018, 9, 2284–2289 RSC.
- (a) A. R. Popescu, F. Teixidor and C. Viñas, Coord. Chem. Rev., 2014, 269, 54–84 CrossRef CAS; (b) R. Núñez, M. Tarrés, A. Ferrer-Ugalde, F. F. de Biani and F. Teixidor, Chem. Rev., 2016, 116, 14307–14378 CrossRef PubMed; (c) P.-F. Cui, X.-R. Liu and G.-X. Jin, J. Am. Chem. Soc., 2023, 145, 19440–19457 CrossRef CAS PubMed; (d) P. Cui, X. Liu, Y. Lin, Z. Li and G. Jin, J. Am. Chem. Soc., 2022, 144, 6558–6565 CrossRef CAS PubMed; (e) S. Guo, P. Cui, X. Liu and G. Jin, J. Am. Chem. Soc., 2022, 144, 22221–22228 CrossRef CAS PubMed; (f) X. Liu, P. Cui, S. Guo, Y. Lin and G. Jin, J. Am. Chem. Soc., 2023, 145, 8569–8575 CrossRef CAS PubMed; (g) X.-R. Liu, P.-F. Cui, Y. García-Rodeja, M. Solà and G.-X. Jin, Chem. Sci., 2024, 15, 9274–9280 RSC; (h) A. Saha, E. Oleshkevich, C. Vinas and F. Teixidor, Adv. Mater., 2017, 29, 1704238 Search PubMed.
- (a) R. Nunez, M. Tarrés, A. Ferrer-Ugalde, F. F. de Biani and F. Teixidor, Chem. Rev., 2016, 116, 14307–14378 CrossRef CAS PubMed; (b) J. Ochi, K. Tanaka and Y. Chujo, Angew. Chem., Int. Ed., 2020, 59, 9841–9855 Search PubMed; (c) Z. Wu, J. Nitsch, J. Schuster, A. Friedrich, K. Edkins, M. Loebnitz, F. Dinkelbach, V. F. Stepanenko, V. Würthner, C. M. Marian, L. Ji and T. B. Marder, Angew. Chem., Int. Ed., 2020, 59, 17137–17144 CrossRef CAS PubMed; (d) L. Ji, S. Riese, A. Schmiedel, M. Holzapfel, M. Fest, J. Nitsch, B. F. E. Curchod, A. Friedrich, L. Wu, H. H. A. Mamari, S. Hammer, J. Pflaum, M. A. Fox, D. J. Tozer, M. Finze, C. Lambert and T. B. Marder, Chem. Sci., 2022, 13, 5205–5219 RSC; (e) D. K. You, H. So, C. H. Ryu, M. Kim and K. M. Lee, Chem. Sci., 2021, 12, 8411–8423 RSC; (f) X. Li, X. Tong, Y. Yin, H. Yan, C. Lu, W. Huang and Q. Zhao, Chem. Sci., 2017, 8, 5930–5940 RSC; (g) K. Yuhara and K. Tanaka, Chem. Sci., 2025, 16, 6495–6506 RSC; (h) F. Lerouge, A. Ferrer-Ugalde, C. Viñas, F. Teixidor, R. Sillanpää, A. Abreu, E. Xochitiotzi, N. Farfán, R. Santilland and R. Núñez, Dalton Trans., 2011, 40, 7541–7550 Search PubMed.
- (a) Y. Xu, Y. Yang, Y. Liu, Z. Li and H. Wang, Nat. Catal., 2023, 6, 16–22 Search PubMed; (b) Z. Lu, P. Qiu, H. Zhai, G.-G. Zhang, X.-W. Chen, Z. Lu, Y. Wu and X. Chen, Angew. Chem., Int. Ed., 2024, 63, e202412401 Search PubMed; (c) J. R. Riffle, C. L. Jowers, S. Luna, M. D. Smith and D. V. Peryshkov, Chem. Sci., 2025, 16, 15997–16003 Search PubMed; (d) B. J. Eleazer, M. D. Smith, A. A. Popov and D. V. Peryshkov, Chem. Sci., 2017, 8, 5399–5407 RSC; (e) P. Coburger, J. Leitl, D. J. Scott, G. Hierlmeier, I. G. Shenderovich, E. Hey-Hawkins and R. Wolf, Chem. Sci., 2021, 12, 11225–11235 RSC; (f) M. Keener, M. Mattejat, S.-L. Zheng, G. Wu, T. W. Hayton and G. Ménard, Chem. Sci., 2022, 13, 3369–3374 RSC; (g) J. Wang, L. Xiang, X. Liu, A. Matler, Z. Lin and Q. Ye, Chem. Sci., 2024, 15, 4839–4845 RSC; (h) S. Bairagi, D. K. Patel, D. Chatterjee, M. Kučeráková, J. Macháček, T. Base, T. Pradeep and S. Ghosh, Chem. Sci., 2025, 16, 14127–14139 RSC; (i) J. Xu, S. Yao, V. Postils, E. Matito, C. Lorent and M. Driess, Chem. Sci., 2025, 16, 10826–10832 RSC.
- (a) A. Marfavi, P. Kavianpour and L. M. Rendina, Nat. Rev. Chem., 2022, 6, 486–504 CrossRef PubMed; (b) R. J. Grams, W. L. Santos, I. R. Scorei, A. Abad-García, C. A. Rosenblum, A. Bita, H. Cerecetto, C. Viñas and M. A. Soriano-Ursua, Chem. Rev., 2024, 124, 2441–2511 CrossRef CAS PubMed; (c) P. Stockmann, M. Gozzi, R. Kuhnert, M. B. Sárosi and E. Hey-Hawkins, Chem. Soc. Rev., 2019, 48, 3497–3512 RSC; (d) F. Issa, M. Kassiou and L. M. Rendina, Chem. Rev., 2011, 111, 5701–5722 CrossRef CAS PubMed; (e) E. G. Tse, S. D. Houston, C. M. Williams, G. P. Savage, L. M. Rendina, I. Hallyburton, M. Anderson, R. Sharma, G. S. Walker, R. S. Obach and M. H. Todd, J. Med. Chem., 2020, 63, 11585–11601 CrossRef CAS PubMed; (f) H. Li, Y. L. Gao and J. J. Ma, Future Med. Chem., 2022, 14, 1681–1692 CrossRef CAS PubMed; (g) B. B. Jena and M. R. Pattanayak, Carborane Clusters for Promoting Medicinal Applications, Apple Academic Press: New Forms of Carbon, 2024, p. 219 Search PubMed; (h) D. A. Gruzdev, G. L. Levit, V. P. Krasnov and V. N. Charushin, Coord. Chem. Rev., 2021, 433, 213753 CrossRef CAS; (i) R. Otero, S. Seoane, R. Sigüeiro, A. Y. Belorusova, M. A. Maestro, R. Pérez-Fernández, N. Rochel and A. Mouriño, Chem. Sci., 2016, 7, 1033–1037 RSC; (j) W. Ma, Y. Wang, Y. Xue, M. Wang, C. Lu, W. Guo, Y.-H. Liu, D. Shu, G. Shao, Q. Xu, D. Tu and H. Yan, Chem. Sci., 2024, 15, 4019–4030 RSC; (k) M. Couto, M. F. García, C. Alamón, M. Cabrera, P. Cabral, A. Merlino, F. Teixidor, H. Cerecetto and C. Viñas, Chem. – Eur. J., 2018, 24, 3122–3126 CrossRef CAS PubMed.
- (a) Y. Wang, J. Nie, W. Fang, L. Yang, Q. Hu, Z. Wang, J. Z. Sun and B. Z. Tang, Chem. Rev., 2020, 120, 4534–4577 CrossRef CAS PubMed; (b) F. Würthner, Angew. Chem., Int. Ed., 2020, 59, 14192–14196 CrossRef PubMed; (c) C. Chen, X. Zhang, Z. Gao, G. Feng and D. Ding, Nat. Protoc., 2024, 19, 2408–2434 CrossRef CAS PubMed.
- D. Ma, H. Bian, M. Gu, L. Wang, X. Chen and X. Peng, Coord. Chem. Rev., 2024, 505, 215677 CrossRef CAS.
- W. Zhang, Y. Huang, Y. Chen, E. Zhao, Y. Hong, S. Chen, J. W. Y. Lam, Y. Chen, J. Hou and B. Z. Tang, ACS Appl. Mater. Interfaces, 2019, 11, 10567–10577 CrossRef CAS PubMed.
- (a) M. L. Beer, J. Lemon and J. F. Valliant, J. Med. Chem., 2010, 53, 8012–8020 CrossRef CAS PubMed; (b) A. Buzharevski, S. Paskaš, M.-B. Sárosi, M. Laube, P. Lönnecke, W. Neumann, B. Murganić, S. Mijatović, D. Maksimović-Ivanić, J. Pietzsch and E. Hey-Hawkins, Sci. Rep., 2020, 10, 4827 CrossRef CAS PubMed.
- (a) T. L. Heying, J. W. Ager Jr., S. L. Clark, R. P. Alexander, S. Papetti, J. A. Reid and S. I. Trotz, Inorg. Chem., 1963, 2, 1097–1105 CrossRef CAS; (b) L. I. Zakharkin, A. I. Kovderov and V. A. Ol'shevskaya, Russ. Chem. Bull., 1986, 35, 1260–1266 CrossRef; (c) Z. Qiu and Z. Xie, Angew. Chem., Int. Ed., 2008, 47, 6572–6575 CrossRef CAS PubMed; (d) A. Sousa-Pedrares, C. Viñas and F. Teixidor, Chem. Commun., 2010, 46, 2998–3000 RSC; (e) F. Zheng, T.-F. Leung, K.-W. Chan, H. H. Y. Sung, I. D. Williams, Z. Xie and G. Jia, Chem. Commun., 2016, 52, 10767–10770 RSC; (f) H. Ni, Z. Lu and Z. Xie, Dalton Trans., 2022, 51, 104–110 RSC.
- (a) V. I. Bregadze, Chem. Rev., 1992, 92, 209–223 CrossRef CAS; (b) D. Olid, R. Nunez, C. Vinas and F. Teixidor, Chem. Soc. Rev., 2013, 42, 3318–3336 RSC; (c) X. Zhang and H. Yan, Coord. Chem. Rev., 2019, 378, 466–482 CrossRef CAS; (d) Z. Qiu and Z. Xie, Acc. Chem. Res., 2021, 54, 4065–4079 CrossRef CAS PubMed; (e) M. Herberhold, H. Yan, W. Milius and B. Wrackmeyer, Angew. Chem., Int. Ed., 1999, 38, 3689–3691 CrossRef; (f) Z. Qiu, Y. Quan and Z. Xie, J. Am. Chem. Soc., 2013, 135, 12192–12195 CrossRef CAS PubMed; (g) X. Zhang, H. Zheng, J. Li, F. Xu, J. Zhao and H. Yan, J. Am. Chem. Soc., 2017, 139, 14511–14517 CrossRef CAS PubMed; (h) Y. Baek, K. Cheong, G. H. Ko, G. U. Han, S. H. Han, D. K. Kim, D. Lee and P. H. Lee, J. Am. Chem. Soc., 2020, 142, 9890–9895 CrossRef CAS PubMed; (i) H. A. Mills, J. L. Martin, A. L. Rheingold and A. M. Spokoyny, J. Am. Chem. Soc., 2020, 142, 4586–4591 CrossRef CAS PubMed; (j) Z. Wang, J. Yu, J. Zhang, D. Zhang, Z. Qiu and Z. Xie, Chem. Sci., 2025, 16, 3705–3712 RSC; (k) H.-J. Cao, X. Wei, F. Sun, X. Zhang, C. Lu and H. Yan, Chem. Sci., 2021, 12, 15563–15571 RSC; (l) Y.-N. Ma, Y. Gao, Y. Ma, Y. Wang, H. Ren and X. Chen, J. Am. Chem. Soc., 2022, 144, 8371–8378 CrossRef CAS PubMed; (m) Y. Ma, H. Ren, Y. Wu, N. Li, F. Chen and X. Chen, J. Am. Chem. Soc., 2023, 145, 7331–7342 CrossRef CAS PubMed; (n) M. Hamdaoui, F. Liu, Y. Cornaton, X. Lu, X. Shi, H. Zhang, J. Liu, B. Spingler, J. Djukic and S. Duttwyler, J. Am. Chem. Soc., 2022, 144, 18359–18374 CrossRef CAS PubMed; (o) W. Sun, Y. Jin, Y. Wang, Z. Wen, J. Sun, J. Yao, S. Duttwyler and H. Li, Chem. Sci., 2025, 16, 5942–5947 RSC; (p) H.-J. Cao, J.-X. Li, J.-H. Yan, M.-X. Liu, Q. Zhao, J. Zhang, J. Zhang and H. Yan, Chem. Sci., 2025, 16, 9406–9412 RSC; (q) X. Zhang and H. Yan, Chem. Sci., 2018, 9, 3964–3969 RSC; (r) H. Yang, Y. Guo, K. Cao, Q. Jiang, C. Teng, D. Zhu and S. Wang, Chem. Commun., 2024, 60, 1124–1127 RSC; (s) Y.-H. Liang, J.-X. Kang, Y. Wang, Y.-G. Li, H.-J. Cao, Q. Zhao, X. Chen and Y.-N. Ma, Chem, 2026, 12, 102788 Search PubMed; (t) Y.-F. Liang, L. Yang, B. B. Jei, R. Kuniyil and L. Ackermann, Chem. Sci., 2020, 11, 10764–10769 RSC; (u) L. Yang, B. B. Jei, A. Scheremetjew, B. Yuan, A. C. Stückl and L. Ackermann, Chem. Sci., 2021, 12, 12971–12976 RSC; (v) M. Zhu, P. Wang, Z. Wu, Y. Zhong, L. Su, Y. Xin, A. M. Spokoyny, C. Zou and X. Mu, Chem. Sci., 2024, 15, 10392–10401 RSC; (w) C. Tang and Z. Xie, Angew. Chem., Int. Ed., 2015, 54, 7662–7665 CrossRef CAS PubMed; (x) J.-Y. Lu, H. Wan, J. Zhang, Z. Wang, Y. Li, Y. Du, C. Li, Z.-T. Liu, Z.-W. Liu and J. Lu, Chem. – Eur. J., 2016, 22, 17542–17546 CrossRef CAS PubMed; (y) R. M. Dziedzic and A. M. Spokoyny, Chem. Commun., 2019, 55, 430–442 RSC; (z) M. Gazvoda, H. H. Dhanjee, J. Rodriguez, J. S. Brown, C. E. Farquhar, N. L. Truex, A. Loas, S. L. Buchwald and B. L. Pentelute, J. Am. Chem. Soc., 2022, 144, 7852–7860 CrossRef CAS PubMed.
- (a) Q. Song, J. Yang, K. Zheng, T. Zhang, C. Yuan, L.-M. Yuan and X. Hou, J. Am. Chem. Soc., 2024, 146, 7594–7604 CrossRef CAS PubMed; (b) Q. Zhao, H. J. Qi and T. Xie, Prog. Polym. Sci., 2015, 49–50, 79–120 CrossRef CAS.
- (a) M. Sun, L. Feng and J.-Y. Lu, Org. Lett., 2024, 26, 3697–3702 CrossRef CAS PubMed; (b) W. R. Gill, P. L. Herbertson, J. A. H. MacBride and K. Wade, J. Organomet. Chem., 1996, 507, 249–255 CrossRef CAS.
- J.-M. Gao, L. Wang, H.-J. Yu, A.-G. Xiao and W.-B. Ding, Pyrotechnics, 2011, 36, 404–409 CrossRef CAS.
- (a) D. Tu, P. Leong, S. Guo, H. Yan, C. Lu and Q. Zhao, Angew. Chem., Int. Ed., 2017, 56, 11370–11374 CrossRef CAS PubMed; (b) X. Wei, M.-J. Zhu, Z. Cheng, M. Lee, H. Yan, C. Lu and J. Xu, Angew. Chem., Int. Ed., 2019, 58, 3162–3166 CrossRef CAS PubMed.
- J. Zhong, W. Zhu, S. Shen, N. Zhou, M. Xi, K. Du, D. Wang and B. Z. Tang, Aggregate, 2025, 6, e70089 CrossRef CAS.
- J. Liu, H. Zhang, L. Hu, J. Wang, J. W. Y. Lam, L. Blancafort and B. Z. Tang, J. Am. Chem. Soc., 2022, 144, 7901–7910 CrossRef CAS PubMed.
- J. Mei, N. L. C. Leung, R. T. K. Kwok, J. W. Y. Lam and B. Z. Tang, Chem. Rev., 2015, 115, 11718–11940 CrossRef CAS PubMed.
- R. Coult, M. A. Fox, W. R. Gill, P. L. Herbertson, J. A. H. MacBride and K. Wade, J. Organomet. Chem., 1993, 462, 19–29 CrossRef CAS.
- Y. Hisata, D. Morishita and Y. Hoshimoto, J. Am. Chem. Soc., 2025, 147, 37677–37687 CrossRef CAS PubMed.
- M. J. Strauss, K. X. Liu, M. E. Greaves, J. C. Dahl, S.-T. Kim, Y.-J. Wu, M. A. Schmidt, P. M. Scola and S. L. Buchwald, J. Am. Chem. Soc., 2024, 146, 18616–18625 CrossRef CAS PubMed.
- (a) CCDC 2496931: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2pt85z; (b) CCDC 2496932: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2pt860; (c) CCDC 2496933: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2pt871; (d) CCDC 2496934: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2pt882; (e) CCDC 2496936: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2pt8b4.
Footnote |
| † P. L., X. L. and L. W. contributed equally. |
| This journal is © The Royal Society of Chemistry 2026 |