Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Deciphering the guest-free crystal structures and thermal breathing of the flexible metal–organic frameworks ZIF-7 and ZIF-9
Athanasios
Koutsianos
 a,
Erik
Svensson Grape†
a,
Erik
Svensson Grape†
 b,
Roman
Pallach
b,
Roman
Pallach
 a,
Julian
Keupp
a,
Julian
Keupp
 c,
Rochus
Schmid
c,
Rochus
Schmid
 c,
A. Ken
Inge
c,
A. Ken
Inge
 b and
Sebastian
Henke
b and
Sebastian
Henke
 *a
*a
aAnorganische Chemie, Fakultät für Chemie und Chemische Biologie, Technische Universität Dortmund, Otto-Hahn-Straße 6, 44227 Dortmund, Germany. E-mail: sebastian.henke@tu-dortmund.de
bWallenberg Initiative Materials Science for Sustainability, Department of Chemistry, Stockholm University, 10691 Stockholm, Sweden
cFakultät für Chemie und Biochemie, Ruhr-Universität Bochum, Universitätsstraße 150, 44801 Bochum, Germany
First published on 9th February 2026
Abstract
Owing to their dynamic phase behaviour and unique gas sorption properties, flexible metal–organic frameworks (MOFs) have emerged as a promising class of materials for applications in gas-related technologies and beyond. Resolving the crystal structures of the distinct phases is essential for understanding their transformation mechanisms and rational tuning framework responsiveness. Here, we revisit the prototypical flexible MOFs ZIF-7 (Zn(bim)2, bim− = benzimidazolate) and ZIF-9 (Co(bim)2) and resolve the long-standing ambiguity surrounding their guest-free narrow-pore (np) phases. Using microcrystal three-dimensional electron diffraction combined with powder X-ray diffraction (PXRD) and density functional theory calculations, we determine the crystal structures of both np phases. The results rectify previous structural models and incomplete structural descriptions of the np phase of ZIF-7 and establish the structure of the np phase of ZIF-9. In contrast to the high-symmetry, guest-accommodating large-pore (lp) phases, the np phases adopt distorted, densely packed frameworks with strongly deformed sodalite cages, reduced void fractions, and enhanced framework densities. Variable-temperature PXRD and differential scanning calorimetry further reveal metal-dependent anisotropic thermal expansion of the np phases and entropy-driven np–lp transitions.
Introduction
Zeolitic imidazolate frameworks (ZIFs) constitute an important subfamily of metal–organic frameworks (MOFs) that mimic network topologies of zeolites due to their structural resemblance.1–3 Their modular architecture, combined with exceptional thermal and chemical stability as well as facile synthesis, has driven intense research interest.1,4 Beyond their robustness, several ZIFs display framework flexibility,5–8 undergoing reversible structural transformations in response to external stimuli such as gas pressure, mechanical compression, or temperature.9,10 Notably, some ZIFs undergo gas-sorption-induced transitions, including gate-opening or breathing behaviour, whereby the pore size dynamically adapts upon gas sorption.6,11,12 This flexibility originates from a subtle balance between enthalpic (e.g. guest-framework and intra-framework interactions) and entropic (e.g. vibrational and configurational degrees of freedom) contributions.13–15ZIF-7 (Zn(bim)2, bim− = benzimidazolate) is a canonical example of a flexible MOF, known for its reversible guest-induced phase transition between a narrow-pore (np) and a large-pore (lp) phase.16 The as-synthesized form adopts the lp phase, stabilized by occluded solvent molecules. It features a rhombohedrally distorted sodalite (sod) topology, constructed from tetrahedrally coordinated Zn2+ centers interconnected by bim− linkers (space group: R![[3 with combining macron]](https://www.rsc.org/images/entities/char_0033_0304.gif) , Fig. 1 and Table 1).1 Upon desolvation, the framework transforms to a guest-free np phase. Besides guest removal, exposure to gases such as CO2, CH4, H2, and short-chain (C2–C3) hydrocarbons induces reopening of the structure, forming the lp phase at well-defined threshold pressures, as evidenced by sigmoidal gas sorption isotherms17,18 and in situ powder X-ray diffraction (PXRD) studies.19 In addition to guest-induced transformations, it has been shown that guest-free ZIF-7 also undergoes an np–lp transition upon heating.19 This thermally induced phase transition is driven by entropic stabilisation of the expanded lp phase, reflecting an adsorption-independent breathing mechanism. This responsive behaviour underpins ZIF-7's relevance for gas separation, storage and sensing applications.17,20,21 Specifically, the step-shaped isotherms associated with the np–lp transition render ZIF-7 particularly attractive for selective gas separation and pressure-swing adsorption processes.22,23
, Fig. 1 and Table 1).1 Upon desolvation, the framework transforms to a guest-free np phase. Besides guest removal, exposure to gases such as CO2, CH4, H2, and short-chain (C2–C3) hydrocarbons induces reopening of the structure, forming the lp phase at well-defined threshold pressures, as evidenced by sigmoidal gas sorption isotherms17,18 and in situ powder X-ray diffraction (PXRD) studies.19 In addition to guest-induced transformations, it has been shown that guest-free ZIF-7 also undergoes an np–lp transition upon heating.19 This thermally induced phase transition is driven by entropic stabilisation of the expanded lp phase, reflecting an adsorption-independent breathing mechanism. This responsive behaviour underpins ZIF-7's relevance for gas separation, storage and sensing applications.17,20,21 Specifically, the step-shaped isotherms associated with the np–lp transition render ZIF-7 particularly attractive for selective gas separation and pressure-swing adsorption processes.22,23
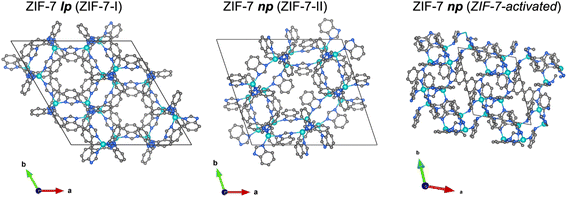 | ||
| Fig. 1 Illustrations of the crystal structures of the ZIF-7 lp phase (ZIF-7-I1) and the two ZIF-7 np phases (ZIF-7-II29 and ZIF-7 activated21) proposed in the literature. Zn, teal; N, blue; C, grey; hydrogen atoms are omitted for clarity. | ||
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) and the two ZIF-7 np phases (ZIF-7-II
1) and the two ZIF-7 np phases (ZIF-7-II![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 29 and ZIF-7 activated21) proposed in the literature. The void fractions have been calculated via Mercury software (probe radius 1.2 Å and grid spacing 0.2 Å)
29 and ZIF-7 activated21) proposed in the literature. The void fractions have been calculated via Mercury software (probe radius 1.2 Å and grid spacing 0.2 Å)
| Material | ZIF-7-I | ZIF-7-II | ZIF-7-activated |
|---|---|---|---|
| Phase model | lp phase | np phase | np phase |
| Space group |
R![[3 with combining macron]](https://www.rsc.org/images/entities/char_0033_0304.gif) |
P![[1 with combining macron]](https://www.rsc.org/images/entities/char_0031_0304.gif) |
P![[1 with combining macron]](https://www.rsc.org/images/entities/char_0031_0304.gif) |
| a/Å | 22.989(3) | 23.948(6) | 11.320(3) |
| b/Å | 22.989(3) | 21.354(6) | 14.158(4) |
| c/Å | 15.763(3) | 16.349(4) | 14.212(5) |
| α/° | 90 | 90.28(2) | 109.82(2) |
| β/° | 90 | 93.275(17) | 95.30(2) |
| γ/° | 120 | 108.412(14) | 109.459(19) |
| V/Å3 | 7214(2) | 7917(3) | 1965.5(11) |
| Z | 18 | 2 | 2 |
| Volume per Zn(bim)2 formula unit/Å3 | 400.77(11) | 439.85(17) | 327.6(2) |
| Void fraction/% | 26.1 | 34.4 | 1.2 |
| Density/g cm−3 | 1.24 | 1.13 | 1.51 |
Already more than 19 years ago, the crystal structure of the lp phase of ZIF-7 was determined by single-crystal X-ray diffraction (SCXRD), as large single crystals form under solvothermal synthesis conditions.1 In contrast, the guest-free np phase remains difficult to characterise structurally. Upon desolvation and formation of the np phase, the crystals fracture into small microparticles. Moreover, PXRD patterns of the np phase are characterised by substantial peak broadening, likely caused by size and strain effects, in combination with extensive Bragg reflection overlap due to low symmetry. These factors render structure determination from PXRD data highly challenging.
To realise gas separation and sensing applications under varying operational conditions, precise control of switching pressures is required.24–26 Metal substitution has emerged as an effective strategy to tune transition thermodynamics. In this context, isostructural analogues such as ZIF-9 (Co(bim)2) and CdIF-13 (Cd(bim)2) have been explored in comparison to ZIF-7 with regard to gas uptake and switching behaviour.27,28 Like ZIF-7, both materials exhibit reversible transitions between np and lp phases. Substituting Zn2+ with Co2+ or Cd2+ raises the threshold pressure for the gas-induced transition, emphasising the role of the metal node in modulating framework flexibility. Due to the similarity in PXRD patterns and gas uptake behaviour, structurally analogous np phases have been hypothesised for ZIF-7 and ZIF-9. However, the structure of guest-free ZIF-9 has remained unresolved.
Initial attempts to solve the np structure of the ZIF-7 relied on structural modeling and Rietveld refinement of PXRD data.29 The resulting model, ZIF-7-II (space group P![[1 with combining macron]](https://www.rsc.org/images/entities/char_0031_0304.gif) ), is inconsistent with key experimental and computational observations. Notably, it exhibits a lower crystallographic density and a higher pore volume than the lp phase (Fig. 1 and Table 1) – an implausible scenario for an enthalpically stabilized contracted phase. The fact that molecular dynamics simulations predicted a higher crystal density for the np phase,19 alongside the inability of independent groups to reproduce the ZIF-7-II model by Rietveld refinement of PXRD data,19,28 raises further doubts. Despite these inconsistencies, the ZIF-7-II model has nonetheless been employed in experimental and computational studies to rationalise ZIF-7's sorption behaviour.29,30
), is inconsistent with key experimental and computational observations. Notably, it exhibits a lower crystallographic density and a higher pore volume than the lp phase (Fig. 1 and Table 1) – an implausible scenario for an enthalpically stabilized contracted phase. The fact that molecular dynamics simulations predicted a higher crystal density for the np phase,19 alongside the inability of independent groups to reproduce the ZIF-7-II model by Rietveld refinement of PXRD data,19,28 raises further doubts. Despite these inconsistencies, the ZIF-7-II model has nonetheless been employed in experimental and computational studies to rationalise ZIF-7's sorption behaviour.29,30
In 2021 a study addressed this issue by fitting the powder neutron diffraction pattern of the np phase of ZIF-7 using a structural model derived from single-crystal X-ray diffraction (SCXRD) of the guest-free isoreticular analogue CdIF-13.21,27 The resulting structure, referred to as ZIF-7-activated (Fig. 1 and Table 1), represents a dense, chemically plausible triclinic phase (space group P![[1 with combining macron]](https://www.rsc.org/images/entities/char_0031_0304.gif) ), consistent with prior simulation data and thermodynamic expectations. Importantly, unlike the ZIF-7-II model, this structure is denser than the lp phase, aligning with the expected features of a guest-free contracted framework. However, the reliability of this structural model is limited by the inherent challenges of neutron powder diffraction for low-symmetry, poorly crystalline phases. Severe Bragg peak broadening and overlap precluded a detailed refinement, necessitating the use of rigid-body constraints. Moreover, key structural features, such as atomic anisotropic displacement parameters (ADPs), could not be determined.
), consistent with prior simulation data and thermodynamic expectations. Importantly, unlike the ZIF-7-II model, this structure is denser than the lp phase, aligning with the expected features of a guest-free contracted framework. However, the reliability of this structural model is limited by the inherent challenges of neutron powder diffraction for low-symmetry, poorly crystalline phases. Severe Bragg peak broadening and overlap precluded a detailed refinement, necessitating the use of rigid-body constraints. Moreover, key structural features, such as atomic anisotropic displacement parameters (ADPs), could not be determined.
To overcome these limitations, we employ three-dimensional electron diffraction (3DED), an emerging technique,31–41 to resolve the crystal structures of the guest-free np phases of ZIF-7 and ZIF-9 from single crystal microparticles. 3DED enables high-quality reciprocal space data collection from submicron-sized single crystals, allowing detailed atomistic models to be derived. Comparative analysis reveals pronounced structural resemblance between ZIF-7 and ZIF-9, which is also reflected in their analogous thermal expansion and thermo-responsive breathing behaviour as established by variable-temperature (VT-)PXRD and differential scanning calorimetry (DSC). Density functional theory (DFT) calculations further support the derived structural models and elucidate the enthalpic stabilization of the np phases by dispersion interactions.
Results and discussion
Structural characterization
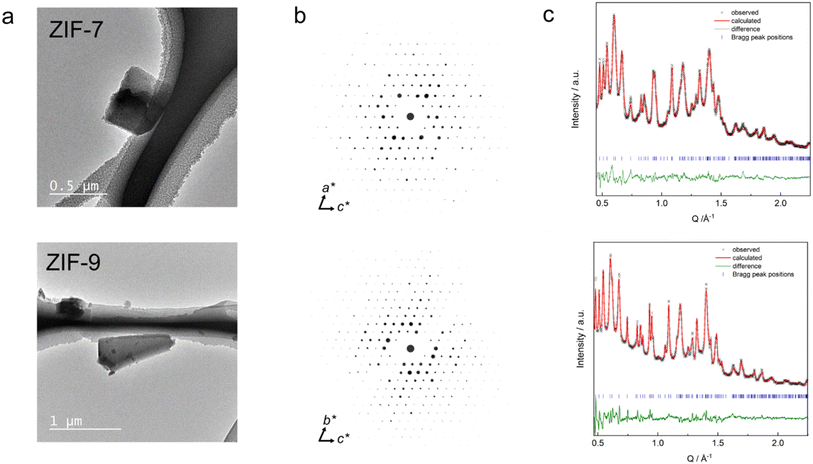
Following reported procedures,28,42 highly crystalline solvated crystals of ZIF-7 and ZIF-9 (lp phases) were synthesized, free of detectable impurities, as confirmed by PXRD (Fig. S1). Upon activation at 200 °C under dynamic vacuum, the materials converted into their guest-free np phases. These activated phases exhibited diminished crystallinity, broadened Bragg reflections, and extensive peak overlap, features consistent with previously reported PXRD patterns of the np phases (Fig. S1).3DED data were collected from four individual submicron-sized single crystals of both ZIF-7 and ZIF-9, with rotational coverage of each dataset ranging from 75.2° to 94.7° for ZIF-7 and 82.2° to 94.5° for ZIF-9 (Fig. 2a, b and Table S2, see SI for experimental details). Indexing of the datasets provided triclinic unit cells (space group P![[1 with combining macron]](https://www.rsc.org/images/entities/char_0031_0304.gif) ) with very similar unit cell parameters for both materials (Table S2), and data completeness values of 88% for ZIF-7 and 82% for ZIF-9. These results confirm the close structural analogy between the two frameworks. Moreover, the unit cell parameters and the atomic coordinates align well with the ZIF-7-activated structure previously determined from neutron diffraction data (Table 1), thereby validating that model and clearly distinguishing it from the earlier ZIF-7-II model.
) with very similar unit cell parameters for both materials (Table S2), and data completeness values of 88% for ZIF-7 and 82% for ZIF-9. These results confirm the close structural analogy between the two frameworks. Moreover, the unit cell parameters and the atomic coordinates align well with the ZIF-7-activated structure previously determined from neutron diffraction data (Table 1), thereby validating that model and clearly distinguishing it from the earlier ZIF-7-II model.
The data quality of the 3DED data was sufficient to locate all hydrogen atoms from the Fourier difference maps35 and to refine all non-hydrogen atoms with atomic ADPs that reached physically sound components; a level of structural detail not achievable in the previous neutron diffraction study (Fig. 3). Notably, while the lp phases of both ZIFs feature only one crystallographically unique M2+ ion and two distinct bim− linkers per asymmetric unit, the np phases exhibit three unique M2+ ions and six distinct bim− linkers, reflecting their lower symmetry and higher structural complexity.
Compared to the high-symmetry solvated lp phases, the np phases adopt collapsed, highly distorted conformations, resulting in a dense crystal packing and loss of void volume. The internal connectivity of the building units in the np phase is still characteristic of the sod topology, indicating that the phase transition maintains chemical bonding within the framework. To visualise these structural differences between the lp and np phases, we constructed a graphical representation based on a single sodalite-type cage (a distorted truncated octahedron), which serves as the repeating unit of the network structure (the network is based on a body-centred packing of these sodalite-type cages). Since the structural differences between both phases of ZIF-7 and ZIF-9 are minimal, only ZIF-9 structures are presented here for clarity; the corresponding ZIF-7 structures are provided in the SI (Fig. S5). The vertex representations of the sodalite cages, as well as atomistic representations of the individual ring configurations of the lp and np phases, are set side by side to better visualise the structural differences between the two phases (Fig. 4). In the lp phase, the sodalite network exhibits a rhombohedral distortion (space group R![[3 with combining macron]](https://www.rsc.org/images/entities/char_0033_0304.gif) ) from the ideal cubic Im
) from the ideal cubic Im![[3 with combining macron]](https://www.rsc.org/images/entities/char_0033_0304.gif) m symmetry due to the bulk of the bim− linkers and their arrangement in the network. Conversely, the lp phase features two distinct types of 6-membered rings (1 and 2), occurring in a 1
m symmetry due to the bulk of the bim− linkers and their arrangement in the network. Conversely, the lp phase features two distinct types of 6-membered rings (1 and 2), occurring in a 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3 ratio, and one type of 4-membered ring (3), all arranged around a
3 ratio, and one type of 4-membered ring (3), all arranged around a ![[3 with combining macron]](https://www.rsc.org/images/entities/char_0033_0304.gif) axis, generating the rhombohedrally distorted sodalite cage. Upon transition to the np phase (space group P
axis, generating the rhombohedrally distorted sodalite cage. Upon transition to the np phase (space group P![[1 with combining macron]](https://www.rsc.org/images/entities/char_0031_0304.gif) ), the point symmetry of the sodalite cage is reduced to
), the point symmetry of the sodalite cage is reduced to ![[1 with combining macron]](https://www.rsc.org/images/entities/char_0031_0304.gif) , resulting in four unique 6-membered rings (1′, 2′, 3′, 4′) and three distinct 4-membered rings (5′, 6′, 7′). In contrast, the previous study reporting the ZIF-7-activated phase erroneously proposed that the np structure of ZIF-7 contains only three unique 6-membered rings and a single 4-membered ring, overlooking three additional distinct rings that emerge upon symmetry reduction.21
, resulting in four unique 6-membered rings (1′, 2′, 3′, 4′) and three distinct 4-membered rings (5′, 6′, 7′). In contrast, the previous study reporting the ZIF-7-activated phase erroneously proposed that the np structure of ZIF-7 contains only three unique 6-membered rings and a single 4-membered ring, overlooking three additional distinct rings that emerge upon symmetry reduction.21
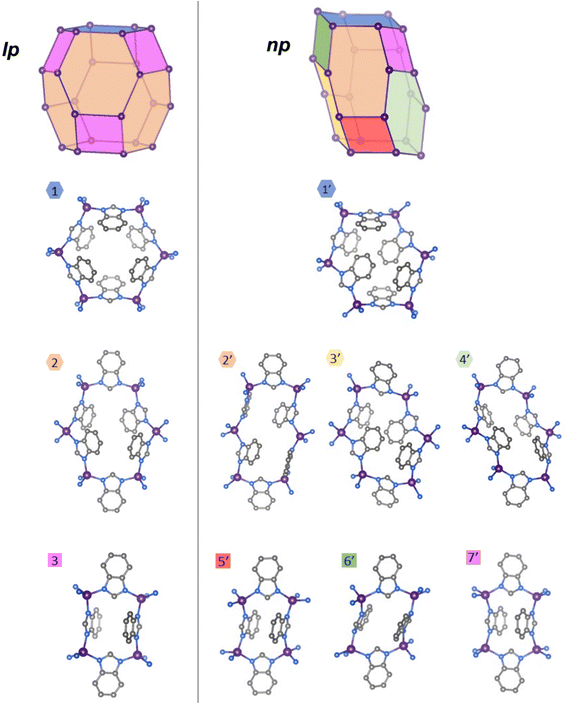 | ||
| Fig. 4 Structural representations of ZIF-9 np (right) sodalite-like cage and the unique four-membered rings and six-membered rings in comparison to the lp phase1 (left, CCDC VEJZEQ). Co, purple; N, blue; C, grey; hydrogen atoms are omitted for clarity. | ||
Comparison of the ring conformations in the lp and np phases reveals pronounced distortions in the np structures, particularly among the 6-membered rings. These undergo significant asymmetric elongation and compression, accompanied by substantial rotations of some bim− linkers about the M–bim–M connection axes, as well as out-of-plane twisting relative to the M–bim–M planes. These cooperative deformations result in a denser packing of the framework building units. Some of the 4-membered rings also exhibit marked deviations from their original geometries. For instance, ring 6′ adopts an oblique rhombic configuration in contrast to the nearly square-shaped ring 3 in the lp phase, reflecting the overall compression of the sodalite cage. Such structural distortions enhance van der Waals contacts between adjacent linkers, thereby contributing to the enthalpic stabilisation of the guest-free np phase.
The structural models derived from 3DED were verified by Rietveld refinement of the models against high-resolution PXRD data recorded at room temperature (Fig. 2c). Due to the low symmetry, severe peak overlap, and peak broadening, the Rietveld model used rigid bodies for the bim− linkers and applied distance and angle restraints for the [MN4] tetrahedra. H atoms were omitted from the Rietveld fit, due to their minimal contribution to the PXRD patterns. Notably, the lattice parameters obtained from the Rietveld fits of the PXRD data (Table S3) are 0.5% to 4.3% smaller than those derived from 3DED (Table S2), resulting in significantly smaller unit cell volumes from PXRD data. Specifically, the unit cell volume of ZIF-7 from PXRD is 8.8% smaller, and that of ZIF-9 is 5.2% smaller compared to 3DED. This difference cannot be attributed to the measurement temperatures (98 K for 3DED vs. 25 °C for PXRD), as lower temperatures would be expected to yield smaller unit cell volumes (see discussion about the thermal expansion behaviour of ZIF-9 and ZIF-9 below). We therefore attribute the discrepancy to the lower accuracy and precision of the unit cell parameters obtained from 3DED due to various experimental challenges associated with the instrument.43 Hence, we use the crystallographic parameters from the PXRD data refinements for the subsequent discussion of the variations in framework volumes and pore volumes. The volumes per M(bim)2 formula unit extracted from PXRD data change from 406.12(3) Å and 406.05(2) Å for the lp phases of ZIF-7 and ZIF-9 to 339.9(5) Å and 332.99(5) Å for the np phases, reflecting a compression by 16% (ZIF-7) and 18% (ZIF-9), respectively. Using the void calculation routine of the software Mercury (probe radius 1.2 Å and grid spacing 0.2 Å), the np phases feature only 3.5% void fraction for ZIF-7 and 2.6% for ZIF-9 (Fig. 5). This is in stark contrast to the reported lp phases, which possess a void fraction of 26.1% for ZIF-7 and 25.2% for ZIF-9 (neglecting solvent guests in the calculation). Remarkably, the obtained crystallographic densities equal 1.41 g cm−3 for ZIF-7 and 1.42 g cm−3 for ZIF-9, essentially identical to the density estimated from molecular dynamics simulations,19 reporting a value of 1.42 g cm−3.
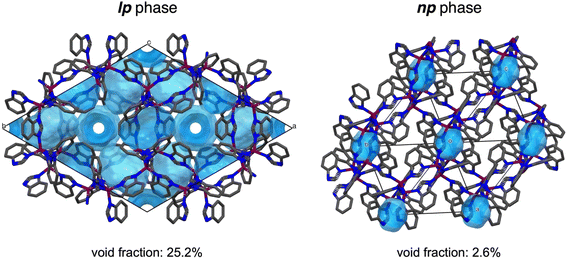 | ||
| Fig. 5 Representation of the void space in the lp phase (CCDC VEJZEQ)1 and np phase of ZIF-9 (CCDC deposition code: 2493471). The voids are calculated with a probe radius of 1.2 Å and a grid spacing of 0.2 Å. A similar viewing direction of the frameworks was attempted (along [001] for the lp phase and along [111] for the np phase). For the lp phase, atoms from solvent guests have been removed. For the np phase, the structure from the Rietveld refinement is taken with H atoms added at idealised positions for the void space calculation. Co, purple; N, blue; C, grey; hydrogen atoms are omitted for clarity. The void space is highlighted in light blue. Unit cell boundaries are shown as thin black lines. | ||
The isolated, tiny pores of the np phases are further in agreement with the well-known CO2 adsorption behaviour of ZIF-7 and ZIF-9, which exhibit extremely low gas uptake prior to the np–lp phase transformation.28 Moreover, the structural validity of the np phases is verified by dispersion corrected periodic DFT calculations using the QUICKSTEP/CP2K code44 at a level of theory established for such systems45 (for details see SI). Zero Kelvin optimizations of guest-free ZIF-7, starting from structural models of the lp and the np phase, resulted in local minima, with the compact np phase energetically favoured by about 12 kJ mol−1 per Zn(bim)2 formula unit. Moreover, a geometry optimisation starting from the previously reported ZIF-7-II phase does not stabilize to a nearby local minimum, but instead converges to the geometry found for the lp phase, highlighting that the proposed ZIF-7-II phase is not a local minimum at this level of theory.
To rationalise the energetic preference for the np phase, we performed a Hirshfeld surface analysis based on the DFT-optimised lp and np structures of ZIF-7 (Fig. S6). The dnorm-mapped Hirshfeld surfaces (where dnorm is the normalised contact distance relative to the sum of van der Waals radii, highlighting contacts close to the van der Waals separation) and the corresponding fingerprint plots (Fig. S7 and Table S4) show a clear shift towards shorter inter-linker (non-bonded) contacts in the np phase, consistent with a markedly tighter packing of the framework. In particular, the np structure exhibits substantially shorter H⋯H and C⋯H contacts between neighbouring linkers, indicative of enhanced dispersion-dominated stabilisation and more pronounced C–H⋯π interactions compared to the lp phase.
Thermal phase behaviour
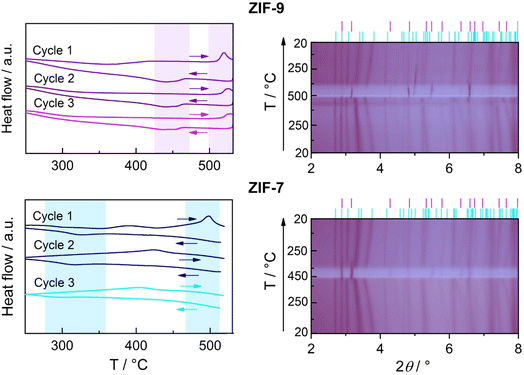
Previous work has shown that guest-free ZIF-7 can undergo a thermally-induced, entropy-driven transition from the np to the lp phase between 500 and 700 °C, depending on the atmosphere (CO2, N2, or vacuum).19 The high-temperature lp phase closely resembles the low-temperature, guest-occupied lp phase, but has a higher enthalpy than the np phase. Motivated by the close structural analogy between ZIF-7 and ZIF-9, we investigated whether ZIF-9 exhibits a similar thermoresponsive behaviour and compared its response to that of ZIF-7.For ZIF-9, DSC measurements on activated samples over three heating/cooling cycles revealed a distinct endothermic peak in the first upscan, with an onset at ∼512 °C (Fig. 6). TGA traces show negligible mass loss up to this temperature, indicating that the signal arises from a thermally-driven np–lp transition. Above ∼535 °C, pronounced mass loss indicates the onset of decomposition,42 prompting us to set 535 °C as the upper temperature limit for cycling experiments. The np–lp transition enthalpy (ΔHnp–lp) from the first upscan is ∼1.6 kJ mol−1 (Table S5). Upon subsequent cooling from 535 °C back to 200 °C, the reverse exothermic lp–np transition is observed around 465 °C, giving a lower enthalpy (ΔHlp–np ∼ −1.3 kJ mol−1), likely due to signal broadening and/or partial decomposition of the material at high temperatures. The pronounced shift of the transition temperature for the reverse transition is characteristic of a first-order phase transition and also in line with the pressure hysteresis observed in gas sorption isotherms involving the related, guest-induced np–lp phase transition.17,46 In subsequent cycles, the onset temperature of the np–lp transition signal increases slightly, so that incomplete baseline recovery up to the maximum temperature of 535 °C prevented reliable determination of ΔHnp–lp by peak integration. Cooling cycles 2 and 3 yielded ΔHlp–np values 17% and 33% lower in magnitude than in cycle 1, and displayed slightly reduced onset temperatures, consistent with progressive framework degradation during thermal cycling.
Variable temperature (VT)-PXRD measurements on ZIF-9 corroborate the DSC results. The np–lp phase transition is clearly visible around 480 °C (Fig. 6 and S17) and confirmed by profile fitting (Fig. S9 and Table S6). The transition temperature is notably reduced compared to the DSC experiment, which can be attributed to the much slower effective heating rate of the VT-PXRD experiment (4–5 min for X-ray exposure and detector readout per pattern) relative to DSC (10 °C min−1).47 Progressive shifts of the np phase reflections prior to the phase transition indicate strong thermal expansion of this phase. Pawley fitting48 of the VT-PXRD patterns (Fig. S9 and Table S6) and subsequent analysis of the derived temperature-dependent lattice parameters with the PAScal software package49 reveal that the framework contracts along the long axis of the stretched sodalite cages as the temperature increases (the coefficient of thermal expansion, CTE, is −22 MK−1), while expanding in the perpendicular directions (CTEs are +22 M K−1 and +138 M K−1; Tables S7 and S8, Fig. S10 and S11). The negative thermal expansion (NTE) behaviour of the np phase is characteristic of its structural evolution towards the rhombohedral lp phase, which features a less stretched sodalite cage (Fig. 4). Upon cooling, the reverse lp–np transition starts at ∼415 °C (Fig. S18) and is completed by ∼350 °C, again showing hysteresis relative to heating (Fig. 6).
Complementary DSC and VT-PXRD analyses of ZIF-7 confirm analogous behaviour, but at lower transition temperatures. DSC shows a rather broad endotherm with an onset at 471 °C (ΔHnp–lp = 1.6 kJ mol−1), and VT-PXRD reveals the np–lp transition at ∼450 °C (Fig. 6, S12 and S19). The lower transition temperature of ZIF-7 compared to ZIF-9 aligns with the lower pressures required for the CO2-induced np–lp transition in ZIF-7, reflecting the higher relative stability of the np phase in ZIF-9.28,50 Notably, a broad exotherm at ∼380 °C precedes the np–lp transition in DSC and coincides with a small weight-loss in TGA (Fig. S8), suggesting this feature may be ascribed to partial decomposition. The thermal expansion behaviour of the np phase of ZIF-7 prior to the phase transition is likewise anisotropic (Tables S10 and S11, Fig. S13 and S14), featuring NTE analogous to ZIF-9 (CTEs are −16 M K−1, +10 M K−1 and +178 M K−1). On cooling, the exothermic lp–np transition is observed with an onset at 384 °C in DSC and ∼380 °C in VT-PXRD (Fig. 6 and S20). The broadened exothermic lp–np peak in DSC precluded reliable enthalpy determination. This may either reflect sluggish phase transition kinetics in ZIF-7 or progressive decomposition during thermal cycling. The latter is consistent with diminished and shifted phase transition signatures in subsequent DSC cycles (Table S5); however, VT-PXRD shows no indication of ZIF-7 decomposition after one single heating–cooling cycle.
Conclusions
Combining 3DED and PXRD with DFT calculations enabled the structural resolution of the elusive guest-free phases of ZIF-7 and ZIF-9. In the absence of guest molecules, both frameworks adopt dense, collapsed np structures that crystallise in the triclinic P![[1 with combining macron]](https://www.rsc.org/images/entities/char_0031_0304.gif) space group. These phases exhibit pronounced structural distortions compared to the higher symmetry lp forms, with strongly deformed sodalite cages, minimised void fractions, and increased framework densities. Such distortions account for the sigmoidal gas sorption behaviour widely reported for these materials.
space group. These phases exhibit pronounced structural distortions compared to the higher symmetry lp forms, with strongly deformed sodalite cages, minimised void fractions, and increased framework densities. Such distortions account for the sigmoidal gas sorption behaviour widely reported for these materials.
The comprehensive structural analysis corrects earlier misassignments of the np phase of ZIF-7 and demonstrates that the dense forms are stabilised by cooperative framework deformations and enhanced van der Waals contacts between linkers. Comparison of ZIF-7 and ZIF-9 reveals their close structural analogy, while VT-PXRD and DSC highlight strong similarities in their thermal expansion behaviour and subtle differences in the thermally induced, entropy-driven np–lp phase transitions, signifying variations in the relative stabilities of their np and lp phases.
Altogether, this study provides a coherent structural picture of the guest-free np phases of ZIF-7 and ZIF-9, resolves longstanding ambiguities in their description, and provides direct insight into their temperature-induced responsiveness. More broadly, it showcases the power of 3DED to unravel complex MOF structures that remain difficult to access by conventional X-ray diffraction techniques, and underscores the value of combining calorimetric methods with in situ diffraction studies to understand transformation mechanisms of flexible MOFs.
Author contributions
AK: formal analysis, investigation, validation, visualization, writing – original draft, writing – review & editing. ESG: formal analysis, investigation, writing – review & editing. RP: formal analysis, investigation, writing – review & editing. JK: formal analysis, investigation. RS: validation, resources, supervision, writing – review & editing. AKI: validation, resources, supervision, writing – review & editing. SH: conceptualization, funding acquisition, project administration, validation, resources, supervision, writing – review & editing.Conflicts of interest
There are no conflicts to declare.Data availability
CCDC 2493469–2493472 contain the supplementary crystallographic data for this paper.51a–dThe data supporting this article have been included as part of the supplementary information (SI). Supplementary information: include experimental and methodogical details, synthesis protocols, additional PXRD, 1H NMR, DFT, TGA and DSC data, and Hirshfeld surface analyses. See DOI: https://doi.org/10.1039/d5sc08614k.
Acknowledgements
The Deutsche Forschungsgemeinschaft (project 447344931) and the VolkswagenStiftung (programme “Experiment!”, grant number A131005) are acknowledged for funding. The authors thank DELTA Dortmund for allocation of beamtime at beamline BL9 and Dr Christian Sternemann and Dr Michael Paulus for their help with the VT-PXRD experiments. Dr Mirko Eckardt (formerly University of Bayreuth) and Prof. Mirijam Zobel (RWTH Aachen) are acknowledged for recording the PXRD pattern of ZIF-7 used for the Rietveld refinement.References
- K. S. Park, Z. Ni, A. P. Côté, J. Y. Choi, R. Huang, F. J. Uribe-Romo, H. K. Chae, M. O'Keeffe and O. M. Yaghi, Proc. Natl. Acad. Sci. U. S. A., 2006, 103, 10186–10191 CrossRef CAS PubMed.
- A. Phan, C. J. Doonan, F. J. Uribe-Romo, C. B. Knobler, M. O'Keeffe and O. M. Yaghi, Acc. Chem. Res., 2010, 43, 58–67 CrossRef CAS PubMed.
- X. Huang, J. Zhang and X. Chen, Chin. Sci. Bull., 2003, 48, 1531–1534 Search PubMed.
- J. B. James and Y. S. Lin, J. Phys. Chem. C, 2016, 120, 14015–14026 CrossRef CAS.
- M. Gao, R.-K. Huang, B. Zheng, P. Wang, Q. Shi, W.-X. Zhang and J. Dong, Nat. Commun., 2022, 13, 4569 CrossRef CAS PubMed.
- P. Iacomi and G. Maurin, ACS Appl. Mater. Interfaces, 2021, 13, 50602–50642 CrossRef CAS PubMed.
- A. Schneemann, V. Bon, I. Schwedler, I. Senkovska, S. Kaskel and R. A. Fischer, Chem. Soc. Rev., 2014, 43, 6062–6096 RSC.
- S. Krause, N. Hosono and S. Kitagawa, Angew. Chem., Int. Ed., 2020, 59, 15325–15341 CrossRef CAS PubMed.
- J. Zhang, Y. Tan and W.-J. Song, Microchim. Acta, 2020, 187, 234 CrossRef CAS PubMed.
- B. Chen, Z. Yang, Y. Zhu and Y. Xia, J. Mater. Chem. A, 2014, 2, 16811–16831 RSC.
- E. Sadeghi Pouya, A. Arami-Niya, K. Jeong, X. Yang, G. Xiao and E. F. May, Chem. Eng. J., 2024, 501, 157567 CrossRef CAS.
- X. Yang, A. Arami-Niya, G. Xiao and E. F. May, Langmuir, 2020, 36, 14967–14977 CrossRef CAS PubMed.
- R. Pallach, J. Keupp, K. Terlinden, L. Frentzel-Beyme, M. Kloß, A. Machalica, J. Kotschy, S. K. Vasa, P. A. Chater, C. Sternemann, M. T. Wharmby, R. Linser, R. Schmid and S. Henke, Nat. Commun., 2021, 12, 4097 CrossRef CAS PubMed.
- J. Wieme, K. Lejaeghere, G. Kresse and V. Van Speybroeck, Nat. Commun., 2018, 9, 4899 CrossRef CAS PubMed.
- L. Kollias, D. C. Cantu, V.-A. Glezakou, R. Rousseau and M. Salvalaglio, Adv. Theory Simul., 2020, 3, 2000092 CrossRef CAS.
- S. Aguado, G. Bergeret, M. P. Titus, V. Moizan, C. Nieto-Draghi, N. Bats and D. Farrusseng, New J. Chem., 2011, 35, 546–550 RSC.
- A. Noguera-Díaz, J. Villarroel-Rocha, V. P. Ting, N. Bimbo, K. Sapag and T. J. Mays, J. Chem. Technol. Biotechnol., 2019, 94, 3787–3792 CrossRef.
- J. van den Bergh, C. Gücüyener, E. A. Pidko, E. J. M. Hensen, J. Gascon and F. Kapteijn, Chem.–Eur. J., 2011, 17, 8832–8840 CrossRef CAS PubMed.
- Y. Du, B. Wooler, M. Nines, P. Kortunov, C. S. Paur, J. Zengel, S. C. Weston and P. I. Ravikovitch, J. Am. Chem. Soc., 2015, 137, 13603–13611 Search PubMed.
- Y. K. Jo, S.-Y. Jeong, Y. K. Moon, Y.-M. Jo, J.-W. Yoon and J.-H. Lee, Nat. Commun., 2021, 12, 4955 CrossRef CAS PubMed.
- R. A. Klein, S. Shulda, P. A. Parilla, P. L. Magueres, R. K. Richardson, W. Morris, C. M. Brown and C. M. McGuirk, Chem. Sci., 2021, 12, 15620–15631 RSC.
- L. W. Bingel, J. D. Evans, T. Kim, J. K. Scott and K. S. Walton, Chem. Mater., 2024, 36, 11756–11769 Search PubMed.
- X. Huang, A. Martín-Calvo, M. J. J. Mulder, S. C. J. van Acht, J. J. Gutiérrez-Sevillano, J. C. García-Navarro and S. Calero, ACS Sustain. Chem. Eng., 2023, 11, 8020–8034 Search PubMed.
- K. Roztocki, F. Formalik, V. Bon, A. Krawczuk, P. Goszczycki, B. Kuchta, S. Kaskel and D. Matoga, Chem. Mater., 2022, 34, 3430–3439 Search PubMed.
- M. K. Taylor, T. Runčevski, J. Oktawiec, M. I. Gonzalez, R. L. Siegelman, J. A. Mason, J. Ye, C. M. Brown and J. R. Long, J. Am. Chem. Soc., 2016, 138, 15019–15026 CrossRef CAS PubMed.
- J. M. Falkowski, P. I. Ravikovitch, M. S. Abdulkarim, G. M. Muraro, S. F. Liu, C. Paur, E. A. Strohmaier and S. C. Weston, J. Mater. Chem. A, 2022, 10, 1425–1432 RSC.
- K. A. Forrest, A. Halder, C. M. McGuirk and B. Space, J. Am. Chem. Soc., 2025, 147, 22141–22160 CrossRef CAS PubMed.
- C. M. McGuirk, T. Runčevski, J. Oktawiec, A. Turkiewicz, M. K. Taylor and J. R. Long, J. Am. Chem. Soc., 2018, 140, 15924–15933 CrossRef CAS PubMed.
- P. Zhao, G. I. Lampronti, G. O. Lloyd, M. T. Wharmby, S. Facq, A. K. Cheetham and S. A. T. Redfern, Chem. Mater., 2014, 26, 1767–1769 CrossRef CAS PubMed.
- P. Zhao, H. Fang, S. Mukhopadhyay, A. Li, S. Rudić, I. J. McPherson, C. C. Tang, D. Fairen-Jimenez, S. C. E. Tsang and S. A. T. Redfern, Nat. Commun., 2019, 10, 999 CrossRef CAS PubMed.
- Z. Meng, C. G. Jones, S. Farid, I. U. Khan, H. M. Nelson and K. A. Mirica, Angew. Chem., Int. Ed., 2022, 61, e202113569 CrossRef CAS PubMed.
- J.-H. Dou, M. Q. Arguilla, Y. Luo, J. Li, W. Zhang, L. Sun, J. L. Mancuso, L. Yang, T. Chen, L. R. Parent, G. Skorupskii, N. J. Libretto, C. Sun, M. C. Yang, P. V. Dip, E. J. Brignole, J. T. Miller, J. Kong, C. H. Hendon, J. Sun and M. Dincă, Nat. Mater., 2021, 20, 222–228 CrossRef CAS PubMed.
- D. Lenzen, J. Zhao, S.-J. Ernst, M. Wahiduzzaman, A. Ken Inge, D. Fröhlich, H. Xu, H.-J. Bart, C. Janiak, S. Henninger, G. Maurin, X. Zou and N. Stock, Nat. Commun., 2019, 10, 3025 CrossRef PubMed.
- Z. Huang, M. Ge, F. Carraro, C. Doonan, P. Falcaro and X. Zou, Faraday Discuss., 2021, 225, 118–132 RSC.
- T. Gruene, J. J. Holstein, G. H. Clever and B. Keppler, Nat. Rev. Chem., 2021, 5, 660–668 CrossRef CAS PubMed.
- M. Ge, Y. Wang, F. Carraro, W. Liang, M. Roostaeinia, S. Siahrostami, D. M. Proserpio, C. Doonan, P. Falcaro, H. Zheng, X. Zou and Z. Huang, Angew. Chem., Int. Ed., 2021, 60, 11391–11397 CrossRef CAS PubMed.
- Z. Huang, T. Willhammar and X. Zou, Chem. Sci., 2021, 12, 1206–1219 RSC.
- M. Gemmi, M. G. I. La Placa, A. S. Galanis, E. F. Rauch and S. Nicolopoulos, J. Appl. Crystallogr., 2015, 48, 718–727 CrossRef CAS.
- M. Gemmi and A. E. Lanza, Acta Crystallogr., Sect. B:Struct. Sci., Cryst. Eng. Mater., 2019, 75, 495–504 CrossRef CAS PubMed.
- M. Gemmi, E. Mugnaioli, T. E. Gorelik, U. Kolb, L. Palatinus, P. Boullay, S. Hovmöller and J. P. Abrahams, ACS Cent. Sci., 2019, 5, 1315–1329 CrossRef CAS PubMed.
- A. Sala, M. D. Faye Diouf, D. Marchetti, L. Pasquale and M. Gemmi, Cryst. Growth Des., 2024, 24, 3246–3255 Search PubMed.
- L. T. L. Nguyen, K. K. A. Le, H. X. Truong and N. T. S. Phan, Catal. Sci. Technol., 2012, 2, 521–528 RSC.
- P. Brázda, M. Klementová, Y. Krysiak and L. Palatinus, IUCrJ, 2022, 9, 735–755 CrossRef PubMed.
- T. D. Kühne, M. Iannuzzi, M. Del Ben, V. V. Rybkin, P. Seewald, F. Stein, T. Laino, R. Z. Khaliullin, O. Schütt, F. Schiffmann, D. Golze, J. Wilhelm, S. Chulkov, M. H. Bani-Hashemian, V. Weber, U. Borštnik, M. Taillefumier, A. S. Jakobovits, A. Lazzaro, H. Pabst, T. Müller, R. Schade, M. Guidon, S. Andermatt, N. Holmberg, G. K. Schenter, A. Hehn, A. Bussy, F. Belleflamme, G. Tabacchi, A. Glöß, M. Lass, I. Bethune, C. J. Mundy, C. Plessl, M. Watkins, J. VandeVondele, M. Krack and J. Hutter, J. Chem. Phys., 2020, 152, 194103 CrossRef PubMed.
- V. Haigis, Y. Belkhodja, F.-X. Coudert, R. Vuilleumier and A. Boutin, J. Chem. Phys., 2014, 141, 064703 Search PubMed.
- S. Bendt, M. Hovestadt, U. Böhme, C. Paula, M. Döpken, M. Hartmann and F. J. Keil, Eur. J. Inorg. Chem., 2016, 2016, 4440–4449 CrossRef CAS.
- H. E. Kissinger, J. Res. Natl. Bur. Stand., 1956, 57, 217 CrossRef CAS.
- G. S. Pawley, J. Appl. Crystallogr., 1981, 14, 357–361 CrossRef CAS.
- M. J. Cliffe and A. L. Goodwin, J. Appl. Crystallogr., 2012, 45, 1321–1329 CrossRef CAS.
- S. Henke, M. T. Wharmby, G. Kieslich, I. Hante, A. Schneemann, Y. Wu, D. Daisenberger and A. K. Cheetham, Chem. Sci., 2018, 9, 1654–1660 RSC.
- (a) CCDC 2493469: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2ppnhk; (b) CCDC 2493470: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2ppnjl; (c) CCDC 2493471: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2ppnkm; (d) CCDC 2493472: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2ppnln.
Footnote |
| † Current affiliation: Department of Chemistry, Ångström Laboratory, Uppsala University, 75120 Uppsala, Sweden. |
| This journal is © The Royal Society of Chemistry 2026 |