 Open Access Article
Open Access ArticleOrganic supramolecular assemblage-confined photoluminescence
Hengzhi
Zhang
and
Yu
Liu
 *
*
College of Chemistry, State Key Laboratory of Elemento-Organic Chemistry, Nankai University, Tianjin 300071, P. R. China. E-mail: yuliu@nankai.edu.cn
First published on 1st April 2026
Abstract
Organic supramolecular hosts can selectively bond to small functional molecules with chromophores, inhibiting molecular motion and isolating quenching factors through spatial confinement, thereby enhancing fluorescence and phosphorescence emission and expanding their applications in chemistry, biology, and materials science. This review mainly focuses on supramolecular luminescent systems constructed from cucurbit[n]urils, cyclodextrins, and other macromolecules. It is very important that cucurbit[n]urils and cyclodextrins both possess rigid hydrophobic cavities, in which cucurbit[n]urils can bond to positively charged guest molecules due to the high negative potential of the carbonyl group at the portal, while cyclodextrin tends to bond to negatively charged guest molecules and has abundant hydroxyl groups, providing numerous modification sites, and both can be further assembled with biomacromolecules through bonding or modification. Biomacromolecules such as hyaluronic acid and chitosan can multivalently bind to guest molecules through electrostatic interactions and hydrogen bonds. Supramolecular luminescent systems formed by these organic macrocyclic hosts or macromolecules and guest molecules have been widely used to construct intelligent supramolecular assemblies and have been successfully applied to near-infrared cell imaging, in situ photodynamic therapy, anti-counterfeiting, information encryption, logic gates, and other fields. With the continuous emergence of novel luminescent groups and new macrocycles, photoluminescence confined within the assembly of organic supramolecular systems will undoubtedly boost their further development in fields such as constructing chiral transfer amplification systems, novel organic light-emitting diodes, and in vivo imaging diagnostics.
1 Introduction
Supramolecular luminescent materials achieved by assembly confinement and cascade confinement strategies have attracted widespread interest in recent years due to their ability to effectively suppress nonradiative transitions and isolate quenching factors, thereby enhancing the emission intensity of luminescent groups.1–3 Organic supramolecular self-assembly units primarily comprise macrocycles, such as cucurbit[n]uril (CB[n]) and cyclodextrin, as well as macromolecules like hyaluronic acid and chitosan. Cucurbit[n]uril is a type of cyclic molecule obtained by the condensation of glycoluril and formaldehyde.4 It usually has 6–8 repeating units. The high negative potentials imparted by the carbonyl groups on the glycoluril units confer a high bonding constant for positively charged guest molecules.5 Generally, CB[6], which has a smaller cavity, tends to bind with aliphatic ammonium salts;6 CB[7] (medium cavity) shows high affinity for phenylammonium salts or pyridinium salts;7,8 while CB[8] (large cavity) can accommodate adamantane, ferrocene derivatives, or two guest molecules, forming a host–guest complex with a stoichiometric ratio of 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2.9–12 Unlike cucurbit[n]urils, cyclodextrins are cyclic molecules formed by α-D-glucanopyranose linked end-to-end by 1,4-glycosidic bonds.13 The ends are hydrophilic while the inner walls are hydrophobic, giving them greater structural flexibility compared to cucurbit[n]urils. The abundance of hydroxyl groups also contributes to their good water solubility and modifiability.14–16 Especially, the 6-OH group of cyclodextrins is relatively reactive, making it easy to modify with different functional groups or macromolecules. Different from molecular cages, which possess encapsulation and isolation properties,17,18 supramolecular macrocycles tend to recognize guest molecules, and their bonding is reversible. Therefore, they can be used to construct supramolecular assemblies with dynamically tunable stimulus responses.19,20 Hyaluronic acid, chitosan, and other natural polysaccharide macromolecules possess negative and positive charges due to their carboxyl and amino groups, respectively, allowing them to bond with guest molecules through multivalent electrostatic interactions.21–25 Furthermore, the polysaccharide structure can form abundant hydrogen bonds, further immobilizing the luminescent group (Table 1). However, despite numerous reviews on supramolecular photoluminescence in recent years, organic supramolecular-confined luminescence has been rarely summarized in detail to the best of our knowledge.2,4 In this review, we summarize recently reported organic supramolecular-confined luminescence systems, mainly including (1) supramolecular luminescence systems constructed from cucurbit[n]urils (CB[n]s), (2) supramolecular luminescence systems constructed from cyclodextrins, and (3) supramolecular luminescence systems constructed from macromolecules (Scheme 1). We hope this review will promote the application of novel supramolecular luminescent materials in chemistry, materials science, biology, and other fields.
2.9–12 Unlike cucurbit[n]urils, cyclodextrins are cyclic molecules formed by α-D-glucanopyranose linked end-to-end by 1,4-glycosidic bonds.13 The ends are hydrophilic while the inner walls are hydrophobic, giving them greater structural flexibility compared to cucurbit[n]urils. The abundance of hydroxyl groups also contributes to their good water solubility and modifiability.14–16 Especially, the 6-OH group of cyclodextrins is relatively reactive, making it easy to modify with different functional groups or macromolecules. Different from molecular cages, which possess encapsulation and isolation properties,17,18 supramolecular macrocycles tend to recognize guest molecules, and their bonding is reversible. Therefore, they can be used to construct supramolecular assemblies with dynamically tunable stimulus responses.19,20 Hyaluronic acid, chitosan, and other natural polysaccharide macromolecules possess negative and positive charges due to their carboxyl and amino groups, respectively, allowing them to bond with guest molecules through multivalent electrostatic interactions.21–25 Furthermore, the polysaccharide structure can form abundant hydrogen bonds, further immobilizing the luminescent group (Table 1). However, despite numerous reviews on supramolecular photoluminescence in recent years, organic supramolecular-confined luminescence has been rarely summarized in detail to the best of our knowledge.2,4 In this review, we summarize recently reported organic supramolecular-confined luminescence systems, mainly including (1) supramolecular luminescence systems constructed from cucurbit[n]urils (CB[n]s), (2) supramolecular luminescence systems constructed from cyclodextrins, and (3) supramolecular luminescence systems constructed from macromolecules (Scheme 1). We hope this review will promote the application of novel supramolecular luminescent materials in chemistry, materials science, biology, and other fields.
| Supramolecular hosts | Advantages | Shortcomings |
|---|---|---|
| Cyclodextrins (CD) | Low prices, good water solubilities, easy modification | Lower binding constant and confinement ability |
| Curcubit[n]urils (CB[n]) | Rigid cavities to provide more efficient confinement, high affinities towards positively charged guests | Expensive, lower solubility, hard to modify |
| Linear macromolecules (e.g., hyaluronic acid, chitosan) | Naturally acquired, good biocompatibility, easy to construct larger-scale assemblies (∼100–500 nm) | Causes the solution to become excessively viscous at high concentrations |
 | ||
| Scheme 1 Schematic illustration of three types of organic supramolecular assemblage-confined photoluminescence. | ||
2 Supramolecular luminescent systems constructed from cucurbit[n]urils
Cucurbit[n]urils have a high negative potential portal carbonyl group, thus they have a strong bonding ability for positively charged guest molecules.26–28 For example, bromobenzoimide, a phosphorescent group with an aliphatic ammonium structure, does not emit phosphorescence itself (Fig. 1a)29 when it forms an assembly with CB[7] in a 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 stoichiometric ratio, where CB[7] can thread through the alkyl chain to form a pseudorotaxane structure. At the same time, its rigid cavity inhibits the vibration of the phosphorescent group, thereby inducing its room-temperature phosphorescence at 505 nm. Further assembly with amphiphilic calixarene can greatly improve the intensity and lifetime of phosphorescence emission, so as to construct a phosphorescence resonance energy transfer system and use it for cell imaging. Coincidentally, CB[7] can also bind with bromophenylpyridinium bearing a polyacrylamide side chain (Fig. 1b).30 The assembled polymer exhibits dual emission of fluorescence at around 380 nm and phosphorescence at around 500 nm due to the combined effect of macrocyclic confinement and the rigid matrix of acrylamide. The phosphorescence lifetime can reach up to 2.2 s, which makes it possible to use it for phosphorescence resonance energy transfer to obtain long-lifetime multicolor delayed fluorescence, which can be used for time-resolved information encryption. Similarly, copolymerizing pyridium-modified [2.2]paracyclophane with acrylamide produces only weak phosphorescence.31 However, when co-assembled with CB[8], the phosphorescence intensity is markedly enhanced, and it exhibits a large structure-dependent spectral shift, making it suitable for use in phosphorescence-based pure organic optical thermometers.
1 stoichiometric ratio, where CB[7] can thread through the alkyl chain to form a pseudorotaxane structure. At the same time, its rigid cavity inhibits the vibration of the phosphorescent group, thereby inducing its room-temperature phosphorescence at 505 nm. Further assembly with amphiphilic calixarene can greatly improve the intensity and lifetime of phosphorescence emission, so as to construct a phosphorescence resonance energy transfer system and use it for cell imaging. Coincidentally, CB[7] can also bind with bromophenylpyridinium bearing a polyacrylamide side chain (Fig. 1b).30 The assembled polymer exhibits dual emission of fluorescence at around 380 nm and phosphorescence at around 500 nm due to the combined effect of macrocyclic confinement and the rigid matrix of acrylamide. The phosphorescence lifetime can reach up to 2.2 s, which makes it possible to use it for phosphorescence resonance energy transfer to obtain long-lifetime multicolor delayed fluorescence, which can be used for time-resolved information encryption. Similarly, copolymerizing pyridium-modified [2.2]paracyclophane with acrylamide produces only weak phosphorescence.31 However, when co-assembled with CB[8], the phosphorescence intensity is markedly enhanced, and it exhibits a large structure-dependent spectral shift, making it suitable for use in phosphorescence-based pure organic optical thermometers.
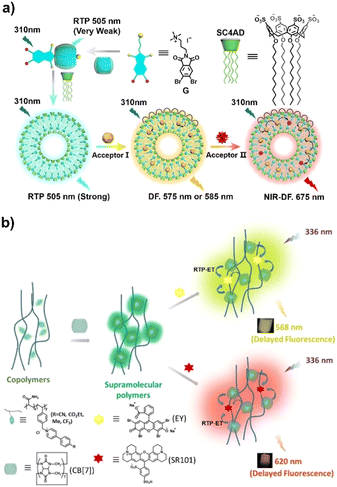 | ||
| Fig. 1 (a) Schematic illustration of the cascade phosphorescence resonance energy transfer system constructed from bromobenzoimide derivatives, CB[7], and SC4AD. This figure has been reproduced from ref. 29 with permission from Wiley-VCH GmbH, copyright 2021. (b) Schematic illustration of the long-lifetime room-temperature phosphorescent macromolecule constructed from acrylamide–phenylpyridium copolymers and CB[7]. This figure has been reproduced from ref. 30 with permission from Wiley-VCH GmbH, copyright 2022. | ||
Interestingly, photoisomerization of spiropyran can change the electronic properties of the molecule, thereby altering its assembly mode with cucurbit[n]urils. For example, spiropyran modified with cyanostyl styrene derivatives is a dicationic salt in the open-ring state (MC) and a monocationic salt in the closed-ring state (SP) (Fig. 2a).32 Utilizing the high affinity of CB[8] for cations, a light-controlled supramolecular shuttle can be assembled: in the closed-ring state, only cyanostyl styrene exhibits blue fluorescence under 365 nm excitation. When the assembly is placed in darkness, spiropyran gradually changes from SP to MC, followed by intramolecular fluorescence resonance energy transfer from cyanostyl styrene to MC, causing the fluorescence to gradually change from blue to green, and finally to orange. Irradiating the assembly with visible light can reverse this process. Similarly, when sulfonic acid-modified spiropyran is added to the linear supramolecular polymer composed of CB[8] and pyridinium-modified diarylethene, its closed-ring SP is negatively charged. Thus, it can undergo cascade assembly with positively charged supramolecular polymers to form nanorods or nanospheres. In contrast, the open-ring MC is neutral and does not undergo further assembly with supramolecular polymers33. Furthermore, the emission wavelength of this assembly can be orthogonally modulated by three wavelengths, thus enabling its use in multi-dimensional information encryption. Spiropyran derivatives modified with styrene derivatives can assemble with CB[7] and CB[8].34 After assembly with CB[7], the emission of the assembly gradually changes from blue to orange when placed in the dark. When assembled with CB[8], because CB[8] can bond to styrene units in a 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 stoichiometric ratio, forming a π–π stacked structure, the emission of the assembly redshifts. When placed in the dark, the emission gradually changes from yellow to red. This bidirectional molecular shuttle can be used for information encryption and cell imaging.
2 stoichiometric ratio, forming a π–π stacked structure, the emission of the assembly redshifts. When placed in the dark, the emission gradually changes from yellow to red. This bidirectional molecular shuttle can be used for information encryption and cell imaging.
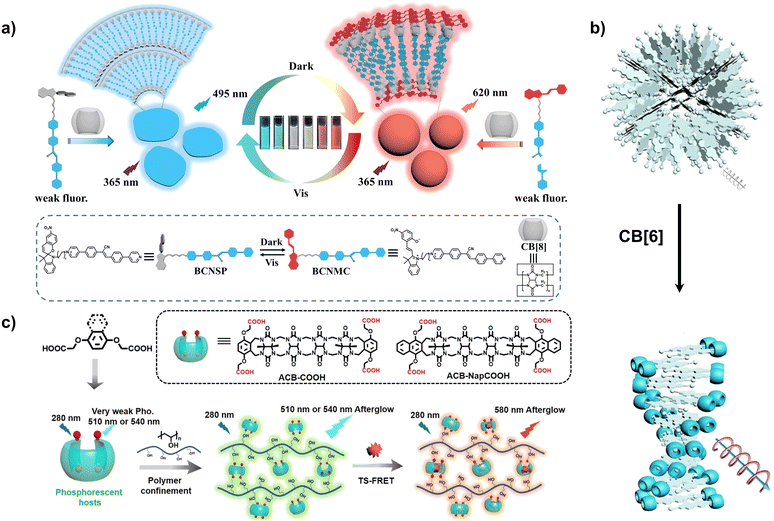 | ||
| Fig. 2 (a) Schematic illustration of the supramolecular shuttle constructed from spiropyran derivatives and CB[8]. This figure has been reproduced from ref. 32 with permission from Wiley-VCH GmbH, copyright 2023. (b) Schematic illustration of the CB[6]-induced chiral transfer amplification. This figure has been reproduced from ref. 35 with permission from Wiley-VCH GmbH, copyright 2024. (c) Schematic illustration of the phosphorescence of acyclic cucurbituril (ACB) induced by a rigid matrix. This figure has been reproduced from ref. 38 with permission from the authors, copyright 2024. | ||
Furthermore, by utilizing the molecular recognition between CB[6] with smaller cavities and chiral ammonium-modified luminescent group guests, supramolecular assemblies with chiral stacking patterns can be constructed, inducing strong CD signals and enhancing the luminescence intensity of the guest molecules (Fig. 2b).35 While CB[7] with larger cavities can also form supramolecular assemblies, it cannot induce chiral stacking patterns.
CB[8] with larger cavities can bind two guest molecules in a 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 stoichiometric ratio. For example, tetracation-modified phenothiazine derivatives can form supramolecular organic frameworks (SOFs) with CB[8] and emit red light at 710 nm.36 Adding a D/L phenylalanine tripeptide to the system as a chiral source allows it to bind CB[8] at the framework edge, where non-covalent crosslinking nodes have not formed, inducing circularly polarized luminescence throughout the framework. It is noteworthy that heating causes the assembly to disintegrate, thus losing its inducible chirality, while cooling restores the chiral luminescence. CB[8] can also be assembled with bromophenylpyridinium modified with aniline derivatives. When the aniline derivative is N,N-dimethylaniline with less steric hindrance, it can bond with CB[8] in a 2
2 stoichiometric ratio. For example, tetracation-modified phenothiazine derivatives can form supramolecular organic frameworks (SOFs) with CB[8] and emit red light at 710 nm.36 Adding a D/L phenylalanine tripeptide to the system as a chiral source allows it to bind CB[8] at the framework edge, where non-covalent crosslinking nodes have not formed, inducing circularly polarized luminescence throughout the framework. It is noteworthy that heating causes the assembly to disintegrate, thus losing its inducible chirality, while cooling restores the chiral luminescence. CB[8] can also be assembled with bromophenylpyridinium modified with aniline derivatives. When the aniline derivative is N,N-dimethylaniline with less steric hindrance, it can bond with CB[8] in a 2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 stoichiometric ratio, avoiding intramolecular twisted charge transfer (TICT) and promoting space charge transfer (TSCT), thereby obtaining enhanced near-infrared delayed fluorescence/phosphorescence dual emission.37 However, N,N-diphenylaniline with larger steric hindrance can only form an assembly with CB[8] in a 2
2 stoichiometric ratio, avoiding intramolecular twisted charge transfer (TICT) and promoting space charge transfer (TSCT), thereby obtaining enhanced near-infrared delayed fluorescence/phosphorescence dual emission.37 However, N,N-diphenylaniline with larger steric hindrance can only form an assembly with CB[8] in a 2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 stoichiometric ratio, and there is no phosphorescence emission due to TICT.
1 stoichiometric ratio, and there is no phosphorescence emission due to TICT.
Interestingly, acyclic cucurbituril (ACB) itself can also act as a luminescent group. For example, ACB with port-modified phenylcarboxylic acid derivatives exhibits long-lifetime room-temperature phosphorescence emission in polyvinyl alcohol films (Fig. 2c).38 The rigid coiled configuration of the cucurbituril tetramer restricts the movement of phenylcarboxylic acid, while the abundant hydrogen bonds in the PVA film provide further confinement, ultimately resulting in long-lifetime phosphorescence with a lifetime of 2.12 s. Furthermore, the cavity of ACB can bind various dyes and drugs, making it suitable for drug detection and phosphorescence resonance energy transfer.
3 Supramolecular luminescent systems constructed from cyclodextrins
Cyclodextrins are a class of cyclic polysaccharide molecules with hydrophobic cavities, exhibiting strong affinity for negatively charged or neutral guest molecules.39,40 For example, in the rigid matrix of polyvinyl alcohol (PVA), γ-cyclodextrin can bond to phenylboronic acid derivatives grafted onto PVA, effectively inhibiting the aggregation of luminescent groups and significantly enhancing the phosphorescence lifetime (Fig. 3a).41 Further doping with dyes such as rhodamine and SR101 allows them to be encapsulated within the cavities of γ-cyclodextrin, achieving phosphorescence resonance energy transfer to obtain multicolor afterglow longer than 50 s. In aqueous solution, β-cyclodextrin can still bind 4-phenylphenylboronic acid, inducing room-temperature phosphorescence with a lifetime of up to 1.03 s through the synergistic effect of macrocyclic confinement and intermolecular hydrogen bonding.42 Notably, only after ultrasonic treatment to encapsulate 4-phenylphenylboronic acid with β-cyclodextrin can room-temperature phosphorescence emission be induced. In the PVA matrix, β-cyclodextrin can bind terephthalic acid, inducing blue phosphorescence emission at 410 nm through spatial confinement and the rigid matrix (Fig. 3b).19 Interestingly, as the temperature increases, the binding effect of β-cyclodextrin with terephthalic acid weakens, causing terephthalic acid to leave the cyclodextrin cavity and aggregate, resulting in a redshift of the phosphorescence emission to 500 nm. Furthermore, the phosphorescence emission after heating shows good overlap with the excitation of rhodamine and SR101, while the phosphorescence emission before heating shows poor overlap with the excitation of the two dyes. Therefore, a thermally activated phosphorescence resonance energy transfer system can be constructed for multi-level anti-counterfeiting and information encryption.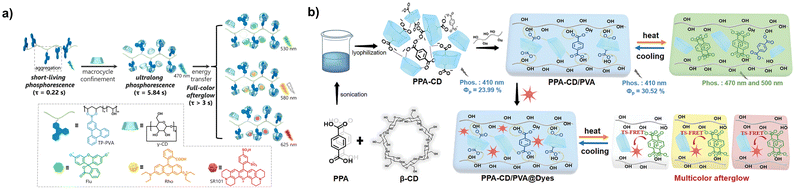 | ||
| Fig. 3 (a) Schematic illustration of the ultra-long-lifetime phosphorescence in PVA film achieved by phenylboronic acid derivatives and γ-CD. This figure has been reproduced from ref. 41 with permission from the authors, copyright 2023. (b) Schematic illustration of the thermal-activated reversible phosphorescence. This figure has been reproduced from ref. 19 with permission from Wiley-VCH GmbH, copyright 2024. | ||
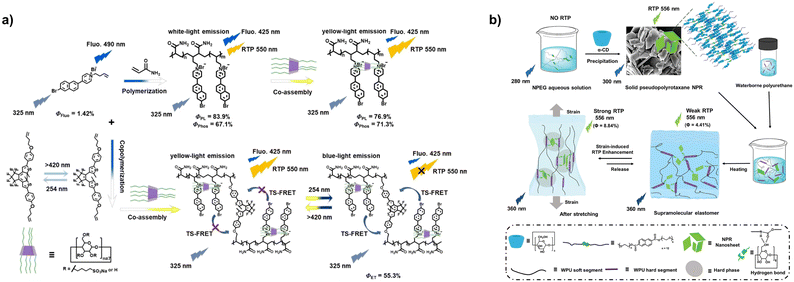
Multicharged cyclodextrins prepared by modifying cyclodextrins with different functional groups can bind guest molecules via electrostatic interactions.43,44 For example, a copolymer of bromonaphthylpyridinium with dual phosphorescence and fluorescence emission with acrylamide can be assembled with sulfobutylether-β-cyclodextrin (SBE-β-CD), enhancing the polymer's yellow room-temperature phosphorescence at 550 nm through spatial confinement (Fig. 4a),45 which mixes with the blue fluorescence emission at 425 nm to achieve strong white light emission. By copolymerizing the photo-switching molecule diarylethylene with monomers and controlling the ratio of yellow phosphorescence to blue fluorescence, white light emission can be obtained, which can be used for data encryption and white LEDs. Similarly, a copolymer of cyano-substituted 4-phenylpyridinium derivatives with acrylamide can also be assembled with SBE-β-CD.46 SBE-β-CD acts as a non-covalent crosslinking node and effectively improves the polymer's phosphorescence lifetime. This phosphorescence exhibits reversible responsiveness to light and heat; the phosphorescence intensity and lifetime decrease sharply after irradiation with a 275 nm UV lamp, while heating reverses this process, thus enabling its application in intelligent UV detection. SBE-β-CD can also form a sheet-like nanostructure via electrostatic interaction with the primary assembly of a tetraphenylethylene derivative and CB[8], significantly enhancing the yellow emission of the assembly at 600 nm.47 However, when assembled with a single tetraphenylethylene derivative, a spherical nanostructure is formed, but the lack of macrocyclic confinement results in insignificant luminescence enhancement. After adding the near-infrared dye aluminum phthalocyanine sulfonate to the sheet-like secondary assembly, fluorescence resonance energy transfer occurs, and near-infrared fluorescence emission is obtained at 691 nm with an efficiency of 75%. In addition, sulfonic acid-modified β-cyclodextrin can be assembled with the pH probe DPy-6C by electrostatic interaction.48 Under acidic conditions, the assembly exhibits weak red fluorescence, while under alkaline conditions, the stacking mode of the assembly is disrupted, resulting in a slight blue shift and significant enhancement of the luminescence, which has been successfully used for the quantitative detection of volatile organic amines in liquid and gas phases.
 | ||
| Fig. 4 (a) Schematic illustration of the SBE-β-CD-enhanced phosphorescence. This figure has been reproduced from ref. 45 with permission from Wiley-VCH GmbH, copyright 2022. (b) Schematic illustration of the elastomer with stress-responsive luminescence constructed from polyurethane and α-CD polypseudorotaxane. This figure has been reproduced from ref. 53 with permission from the authors, copyright 2024. | ||
Utilizing the binding ability of cyclodextrin cavities toward negatively charged dyes can enhance the efficiency of phosphorescence resonance energy transfer (PRET). For example, copolymerizing double-bond modified β-cyclodextrin and carbazole with acrylamide allows the rigid polyacrylamide to induce room-temperature phosphorescence emission of carbazole.49 Compared to copolymers without β-cyclodextrin, the terpolymer better enriches the dye, increasing the PRET efficiency from 27.3% to 97.4%.
The cavity size of α-cyclodextrin matches that of polyethylene glycol (PEG) chains, making it easy to thread through the PEG chains to form pseudorotaxane structures.50–52 Adding α-cyclodextrin to PEG-modified naphthalene esters results in pseudorotaxanes that produce green room-temperature phosphorescence (Fig. 4b).53 Further incorporating these pseudorotaxanes into polyurethane elastomers results in stress-enhanced phosphorescence emission because the hard segments of the polyurethane exert a stronger confinement effect on the pseudorotaxanes under stress.
Interestingly, supramolecular luminescent systems can also be constructed using cyclodextrins in some novel solid materials. For example, adding aromatic boric acid to a MOF formed by γ-CD and potassium ions allows γ-cyclodextrin to effectively bond with the aromatic boric acid, resulting in blue room-temperature phosphorescence emission from the MOF crystals.54 Adding different dyes can effectively generate phosphorescence resonance energy transfer, producing multicolor afterglow. Randomly methylated β-cyclodextrin and p-hydroxybenzoic acid can be hot-pressed to construct transparent supramolecular glasses with good transmittance and high fracture stress.55 Adding tetraphenylethylene derivatives and luminescent groups such as BODIPY to this glass can prepare supramolecular transparent luminescent materials. Furthermore, grafting monomers containing adamantyl groups onto the surface of a hydrogel, when treated with a solution containing naphthalimide-modified cyclodextrin, causes the naphthalimide units originally self-encapsulated within the cyclodextrin cavity to leave due to the competitive bonding of adamantane, promoting π–π stacking and producing green fluorescence emission.56 This can be used for the detection of aromatic hydrocarbon derivatives and multi-level information encryption.
4 Supramolecular luminescent systems constructed from linear macromolecules and other macrocycles
Biomacromolecules, such as hyaluronic acid (HA) and chitosan (CS), can also be used to construct supramolecular-confined luminescence systems. Among them, hyaluronic acid is a natural polysaccharide with a negatively charged carboxyl group on its surface, so it can bind guest molecules through electrostatic interactions.25 At the same time, it can be easily modified with various functional groups.57,58 For example, using hyaluronic acid (HACD) with a side chain modified with cyclodextrin as the host for secondary assembly to bind the primary assembly of dodecyl-bridged 6-bromoisoquinoline and CB[7], the cyclodextrin cavity can effectively encapsulate the primary assembly, and the positively charged primary assembly and the negatively charged HACD can also be assembled through electrostatic interactions.59 Secondary assembly increases the luminescence intensity by eight times and the phosphorescence lifetime from 59 µs to 581 µs. After incorporating near-infrared dyes such as Nile Blue or sulfonated porphyrin, near-infrared delayed fluorescence can be obtained through phosphorescence resonance energy transfer. In addition, due to the targeting of hyaluronic acid to tumor cells, the assembly has been successfully applied to targeted near-infrared imaging of live tumor cells. HACD can also effectively bond to the assembly of tetracation macrocycle and doxorubicin, promoting intermolecular photoelectron transfer (Fig. 5a).60 Under light irradiation, the assembly can generate singlet oxygen species with a high quantum yield of 185%, which enables it to perform efficient photodynamic therapy. The assembly exhibits good tumor cell targeting in vitro and in vivo, and shows excellent antitumor activity. HACD can also bond to the xanthracene part of the primary assembly of indole-bridged xanthracene derivative and CB[8], thereby providing further confinement, which increases the quantum yield of the assembly by 22 times (Fig. 5b).61 The cavity of β-cyclodextrin can also bind to the inclusion complex of the phosphorescence donor sulfonic acid-modified bromophenylpyridinium and CB[8] to achieve phosphorescence resonance energy transfer with an efficiency of 84.49%. The assembly also exhibits upconversion properties and can achieve three-photon excitation at 940 nm, thereby achieving targeted imaging that penetrates deep tissues. HACD can undergo secondary assembly with a ternary supramolecular complex formed by bromophenylpyridinium-modified tetraphenylethylene and CB[7]/CB[8], which possesses a donor–acceptor structure. This assembly simultaneously activates the intramolecular PRET from bromophenylpyridinium to tetraphenylethylene, resulting in near-infrared delayed fluorescence at 700 nm, which can be used for mitochondrial-targeted imaging of tumor cells.62 In addition, HACD can further confine the two-dimensional SOF network formed by CB[8] and bis(triphenylamine) derivatives to enhance its red fluorescence at 810 nm.63 Adding the bromophenylpyridinium derivative and CB[8] to this assembly as a phosphorescence donor gives near-infrared delayed fluorescence, which can be used for targeted near-infrared cell imaging and information encryption.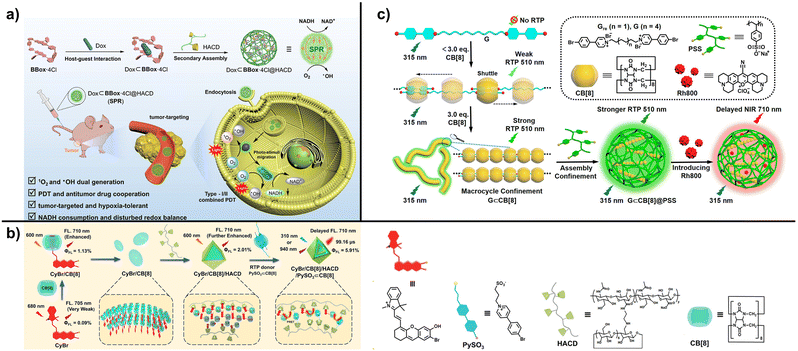 | ||
| Fig. 5 (a) Schematic illustration of the HACD co-assembled photodynamic therapy system. This figure has been reproduced from ref. 60 with permission from the American Chemical Society, copyright 2025. (b) Schematic illustration of the HACD co-assembled upconversion near-infrared emission. This figure has been reproduced from ref. 61 with permission from Wiley-VCH GmbH, copyright 2025. (c) Schematic illustration of the PSS co-assembled supramolecular phosphorescence resonance energy transfer system. This figure has been reproduced from ref. 68 with permission from the authors, copyright 2024. | ||
HA can also be modified by guest molecules containing other functional groups. For example, HA modified with mitochondrial-targeting peptides and bromophenylpyridinium salts can wrap around mitochondria.64 When CB[8] is added, it can bond bromophenylpyridinium in a 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 stoichiometric ratio, driving mitochondrial aggregation and emitting strong room-temperature phosphorescence at 500 nm. This mitochondrial aggregation can effectively enhance the antitumor efficiency of cisplatin. HA modified with bromophenylpyridinium alone can also bind CB[8] in a 2
2 stoichiometric ratio, driving mitochondrial aggregation and emitting strong room-temperature phosphorescence at 500 nm. This mitochondrial aggregation can effectively enhance the antitumor efficiency of cisplatin. HA modified with bromophenylpyridinium alone can also bind CB[8] in a 2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 stoichiometric ratio, producing green room-temperature phosphorescence emission and constructing a PRET system.65
1 stoichiometric ratio, producing green room-temperature phosphorescence emission and constructing a PRET system.65
Unlike HA, CS has numerous amino groups on its polysaccharide chain. These amino groups can be protonated, rendering CS positively charged and enabling it to assemble with negatively charged guest molecules. In addition, amino groups can coordinate with metal ions or undergo Schiff base reactions.66 For example, β-cyclodextrin derivatives containing aldehyde groups can form hydrogels with chitosan through Schiff base reactions, producing fluorescence emission at 315 nm due to aggregation.67 The gel has good injectability and can achieve multicolor luminescence based on fluorescence resonance energy transfer by doping with triphenylamine derivatives.
Interestingly, artificially synthesized macromolecules can also serve as the host for secondary assembly to further enhance confinement. For example, sodium poly(p-phenylenesulfonate) (PSS) can be further assembled with bromophenylpyridinium and CB[8] via electrostatic interactions to form uniform spherical nanoparticles, thereby further enhancing the phosphorescence lifetime (Fig. 5c).68 Adding rhodamine 800 to the assembly can yield near-infrared delayed fluorescence of 710 nm through phosphorescence resonance energy transfer. This can be used for cell imaging.
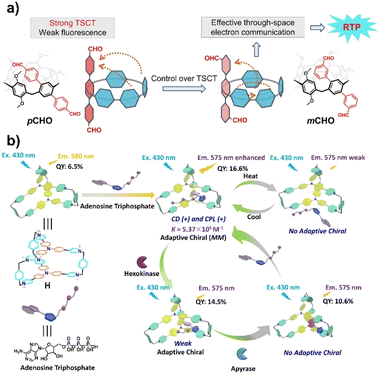
In addition, novel macrocyclic compounds such as pillararenes can also serve as the host for supramolecular-confined luminescence.69,70 For example, m-formylphenyl-substituted pillar[5]arenes can produce millisecond-level room-temperature phosphorescence emission through effective space charge transfer (TSCT), while formylphenyl-substituted columnar[5]arenes cannot produce long-lifetime phosphorescence emission due to excessively strong TSCT (Fig. 6a).71 Binding of guests with heavy atoms (e.g., bromoethane) to this pillar[5]arene derivative enhances quantum yield by promoting ISC and shortens phosphorescence lifetime, enabling the detection of volatile organic compounds. The hexacation cage constructed with triphenylamine has a rotatable rigid framework that can effectively bond nucleotides (Fig. 6b).72 Upon binding with adenosine triphosphate (ATP), the fluorescence intensity and quantum yield are significantly enhanced, and circularly polarized luminescence is caused by chiral transfer. This supramolecular system exhibits thermal responsiveness, and the circularly polarized luminescence disappears under the stepwise hydrolysis of ATP by hexokinase and adenosine triphosphate diphosphorylase. This chiral supramolecular transfer container has been successfully used for chiral logic gates and multi-level information encryption. Furthermore, the π-extended triphenylamine tetracation macrocycle enables photodynamic therapy with a high singlet oxygen quantum yield, and its cavity effectively binds the antitumor drug pemetrexed.73 This synergistic therapy achieved a good antitumor efficacy of 93.4% in a mouse model.
 | ||
| Fig. 6 (a) Schematic illustration of the room-temperature phosphorescence of pillar[5]arene derivatives induced by through-space electron communication. This figure has been reproduced from ref. 71 with permission from the American Chemical Society, copyright 2023. (b) Schematic illustration of the ATP-driven step-chiral transfer system. This figure has been reproduced from ref. 72 with permission from Wiley-VCH GmbH, copyright 2024. | ||
5 Conclusions
In this review, we mainly summarized the applications of supramolecular assembly-confined luminescence strategies based on cucurbit[n]urils, cyclodextrins, and other macromolecules in enhancing luminescence and extending phosphorescence lifetime, as well as cell imaging, photodynamic therapy, and anti-counterfeiting. Although cucurbit[n]urils and cyclodextrins both have rigid cavities, cucurbit[n]urils can bind to various positively charged molecules due to the negative potential on their edge, while cyclodextrins with different functional groups can bind guest molecules with positive or negative charge, both can effectively restricts molecular motion and isolates it from external quenching factors, thereby enhancing the phosphorescence or fluorescence emission of the luminescent group. Meanwhile, hyaluronic acid, a negatively charged linear polysaccharide, can assemble with not only guest molecules but also host–guest complexes through electrostatic interactions, enhancing its luminescence through multi-level confinement. Chitosan, also a linear polysaccharide, readily protonates due to its amino groups, resulting in a positive charge. This allows it to assemble with negatively charged guest molecules. Furthermore, the amino groups can condense with aldehyde groups to form dynamic chemical bonds. Artificially synthesized multicharged polymers, such as PSS, can also enhance the luminescence of guests through electrostatic interactions. Novel hosts, such as pillararenes and triphenylamine-based polycationic macrocycles, can bind different guests to achieve applications in bioimaging, synergistic therapy, and volatile organic compound detection. Despite the remarkable progress made in the supramolecular assembly-confined luminescence systems described above, further applications in the biomedical field still face challenges in terms of cost, in vivo stability, controllable release, and clinical trial data, as well as face the drawbacks of fatigue resistance and environmental tolerance in fields such as anti-counterfeiting and organic light-emitting diodes. We believe this review will promote the application of emerging macrocycles and novel luminescent groups in luminescent systems constructed through supramolecular confinement in in vivo imaging, synergistic therapy, smart materials, multi-level anti-counterfeiting, and encryption, and will also advance the development of supramolecular chemistry.Author contributions
H. Z. searched the literature and wrote the manuscript. Y. L. edited and modified the manuscript.Conflicts of interest
There are no conflicts to declare.Data availability
Data availability is not applicable to this article as no new data was created or analyzed in this studyAcknowledgements
We thank the National Natural Science Foundation of China (NNSFC, Grant No. 22131008), China Fundamental Research Funds for the Central Universities, and the Haihe Laboratory of Sustainable Chemical Transformations for financial support.Notes and references
- W.-L. Zhou, W. Lin, Y. Chen and Y. Liu, Chem. Sci., 2022, 13, 7976–7989 RSC.
- X. Zhou, H. Zhang and Y. Liu, Chem. Sci., 2024, 15, 18259–18271 RSC.
- J. Li, J. Wang, H. Li, N. Song, D. Wang and B. Z. Tang, Chem. Soc. Rev., 2020, 49, 1144–1172 RSC.
- H. Nie, Z. Wei, X.-L. Ni and Y. Liu, Chem. Rev., 2022, 122, 9032–9077 CrossRef CAS PubMed.
- C. Márquez, R. R. Hudgins and W. M. Nau, J. Am. Chem. Soc., 2004, 126, 5806–5816 CrossRef.
- Z.-Y. Zhang, Y. Chen and Y. Liu, Angew. Chem., Int. Ed., 2019, 58, 6028–6032 CrossRef CAS PubMed.
- M. Tang, Z. Yang, X. Tang, H. Ma, B. Xie, J.-F. Xu, C. Gao, D. Bardelang and R. Wang, J. Am. Chem. Soc., 2025, 147, 3488–3499 CrossRef CAS PubMed.
- J. Kong, S.-Q. Song, M. Huo, C. Zhang, C.-H. Liu and Y. Liu, Nano Today, 2025, 65, 102861 CrossRef CAS.
- J. Niu, J. Yu, X. Wu, Y.-M. Zhang, Y. Chen, Z. Yu and Y. Liu, Chem. Sci., 2024, 15, 13779–13787 RSC.
- J. Niu, X. Wu, J. Yu, Z. Lei, Y.-M. Zhang, L.-H. Wang, J. Wu, J. Han and Y. Liu, Chem. Sci., 2025, 16, 19244–19252 RSC.
- G. Liu, C. Tian, X. Fan, X. Xue, L. Feng, C. Wang and Y. Liu, JACS Au, 2023, 3, 2550–2556 CrossRef CAS.
- H. Wu, Y. Wang, L. O. Jones, W. Liu, B. Song, Y. Cui, K. Cai, L. Zhang, D. Shen, X.-Y. Chen, Y. Jiao, C. L. Stern, X. Li, G. C. Schatz and J. F. Stoddart, J. Am. Chem. Soc., 2020, 142, 16849–16860 CrossRef CAS PubMed.
- V. T. D'Souza and K. B. Lipkowitz, Chem. Rev., 1998, 98, 1741–1742 CrossRef PubMed.
- E. Delgado-Pinar, G. Utzeri, A. J. M. Valente, J. S. Seixas de Melo and A. A. A. C. Pais, Carbohydr. Polym., 2025, 356, 123361 CrossRef CAS PubMed.
- J.-H. Huang, Y.-J. Liu, Y. Si, Y. Cui, X.-Y. Dong and S.-Q. Zang, J. Am. Chem. Soc., 2024, 146, 16729–16736 CrossRef CAS PubMed.
- J. Li, Z. Wang, R. Luo, X. Quan, H. U. Fong, Q. Cheng, J. Wei, L. Wang, Y. Zhao and R. Wang, Adv. Sci., 2025, 12, 2414879 CrossRef CAS.
- Q.-Q. Wang, Acc. Chem. Res., 2024, 57, 3227–3240 CrossRef CAS PubMed.
- W. He, Y. Yu, K. Iizuka, H. Takezawa and M. Fujita, Nat. Chem., 2025, 17, 653–662 CrossRef CAS.
- X. Zhou, X. Zhao, X. Bai, Q. Cheng and Y. Liu, Adv. Funct. Mater., 2024, 34, 2400898 CrossRef CAS.
- J. Yang, H.-J. Ye, H.-M. Xiang, X. Zhou, P.-Y. Wang, S.-S. Liu, B.-X. Yang, H.-B. Yang, L.-W. Liu and S. Yang, Adv. Funct. Mater., 2023, 33, 2303206 CrossRef CAS.
- Y. Shan, H. Chu, S. Ye, G. Ji, J. Zhao, X. Si, Y. Zhong, Y. Tao, J. Shi and X. Fang, Adv. Sci., 2025, 12, e09309 CrossRef CAS PubMed.
- J. Tang, C. Li, W. Ma, Z. Ba, Z. Hu, I. Willner and C. Wang, Angew. Chem., Int. Ed., 2025, 64, e202503485 CrossRef CAS PubMed.
- Y. An, Z. Shen, F. Zhang, Q. Yang, Z. Han, M. Wang, H. Ma, L. Yu, W. Yuan and K. Sui, Adv. Sci., 2025, 12, 2415260 CrossRef CAS.
- G. Lim, H. R. Kim, C. Yoo, Y. K. Oh, Y. S. Park, S. Park, S. Jang, L. Priscilla and D. Y. Lee, ACS Nano, 2025, 19, 31169–31182 CrossRef CAS PubMed.
- Z. Liu, W. Lin and Y. Liu, Acc. Chem. Res., 2022, 55, 3417–3429 CrossRef CAS PubMed.
- D. Kolarski, M. L. Bossi, R. Lincoln, J. C. Fuentes-Monteverde, V. N. Belov and S. W. Hell, J. Am. Chem. Soc., 2025, 147, 28893–28902 CrossRef CAS PubMed.
- D. Kim, M. L. Bossi, V. N. Belov and S. W. Hell, Angew. Chem., Int. Ed., 2024, 63, e202410217 CrossRef CAS PubMed.
- M. Du and C. Li, Adv. Mater., 2024, 36, 2408484 CrossRef CAS.
- M. Huo, X.-Y. Dai and Y. Liu, Angew. Chem., Int. Ed., 2021, 60, 27171–27177 CrossRef CAS PubMed.
- W.-W. Xu, Y. Chen, Y.-L. Lu, Y.-X. Qin, H. Zhang, X. Xu and Y. Liu, Angew. Chem., Int. Ed., 2022, 61, e202115265 CrossRef CAS PubMed.
- X. Qiu, T. Zheng, M. Runowski, P. Woźny, I. R. Martín, K. Soler-Carracedo, C. E. Piñero, S. Lebedkin, O. Fuhr and S. Bräse, Adv. Funct. Mater., 2024, 34, 2313517 CrossRef CAS.
- R. Zhang, Y. Chen and Y. Liu, Angew. Chem., Int. Ed., 2023, 62, e202315749 CrossRef CAS PubMed.
- F. Zhu, X.-M. Chen, Y. Ge, M. Li, N. Li, J. Zhou and W. Chen, Adv. Funct. Mater., 2024, 34, 2312155 CrossRef CAS.
- R. Zhang, Z. Lei, Z. Yu, Y. Chen and Y. Liu, Adv. Sci., 2025, 12, e07090 CrossRef CAS.
- Y. Xu, A. Hao and P. Xing, Angew. Chem., Int. Ed., 2024, 63, e202409624 CAS.
- J. Yu, J. Niu, X. Xu and Y. Liu, Adv. Sci., 2024, 11, 2408107 CrossRef CAS PubMed.
- C. Yin, Z. Wu, Z.-A. Yan, P. Jiang, Y. Ding, T. Li, Y. Chen, H. Tian and X. Ma, J. Am. Chem. Soc., 2025, 147, 43286–43294 CrossRef CAS PubMed.
- M. Huo, S.-Q. Song, X.-Y. Dai, F.-F. Li, Y.-Y. Hu and Y. Liu, Chem. Sci., 2024, 15, 5163–5173 RSC.
- X. Dai, M. Huo, B. Zhang, Z. Liu and Y. Liu, Biomacromolecules, 2022, 23, 3549–3559 CrossRef CAS PubMed.
- H.-J. Yu, H. Wang, F.-F. Shen, F.-Q. Li, Y.-M. Zhang, X. Xu and Y. Liu, Small, 2022, 18, 2201737 CrossRef CAS PubMed.
- X.-K. Ma, Q. Cheng, X. Zhou and Y. Liu, JACS Au, 2023, 3, 2036–2043 CrossRef CAS PubMed.
- D. Li, Z. Liu, M. Fang, J. Yang, B. Z. Tang and Z. Li, ACS Nano, 2023, 17, 12895–12902 CrossRef CAS PubMed.
- Z. Liu and Y. Liu, Chem. Soc. Rev., 2022, 51, 4786–4827 RSC.
- P. Liu, Y. Chen, D. Yao, J. Jia, Y. Meng, P. Zhou, X. Gao, Y. Xie, L. Yao, S. Li, L. Wang, G. C. Bazan, S. Xu and C. Zhou, Angew. Chem., Int. Ed., 2025, 64, e202504581 CrossRef CAS PubMed.
- Y.-Y. Hu, X.-Y. Dai, X. Dong, M. Huo and Y. Liu, Angew. Chem., Int. Ed., 2022, 61, e202213097 CrossRef CAS PubMed.
- W.-W. Xu, Y. Chen, X. Xu and Y. Liu, Small, 2024, 20, 2311087 CrossRef CAS PubMed.
- M. Tian, Z. Wang, X. Yuan, H. Zhang, Z. Liu and Y. Liu, Adv. Funct. Mater., 2023, 33, 2300779 CrossRef CAS.
- Z. Liu, H. Chen, M. Tian, X. Sun, Y.-X. Li, J. Wu, R. Wang, B. Li, C. Li and Y. Liu, Aggregate, 2024, 5, e627 CrossRef CAS.
- Q. Cheng, X.-K. Ma, X. Zhou, Y.-M. Zhang and Y. Liu, Small, 2024, 20, 2309732 CrossRef CAS.
- K. Hashimoto, T. Shiwaku, H. Aoki, H. Yokoyama, K. Mayumi and K. Ito, Sci. Adv., 2023, 9, eadi8505 CrossRef CAS PubMed.
- L. Chen, X. Sheng, G. Li and F. Huang, Chem. Soc. Rev., 2022, 51, 7046–7065 RSC.
- Y. Jiang, Z. Zhang, Y.-X. Wang, D. Li, C.-T. Coen, E. Hwaun, G. Chen, H.-C. Wu, D. Zhong, S. Niu, W. Wang, A. Saberi, J.-C. Lai, Y. Wu, Y. Wang, A. A. Trotsyuk, K. Y. Loh, C.-C. Shih, W. Xu, K. Liang, K. Zhang, Y. Bai, G. Gurusankar, W. Hu, W. Jia, Z. Cheng, R. H. Dauskardt, G. C. Gurtner, J. B. H. Tok, K. Deisseroth, I. Soltesz and Z. Bao, Science, 2022, 375, 1411–1417 CrossRef CAS PubMed.
- Y. Zhang, Y. Chen, J.-Q. Li, S.-E. Liu and Y. Liu, Adv. Sci., 2024, 11, 2307777 CrossRef CAS.
- J. Zhang, J. Tang, Y. Zhang, Y. Chen and J. Gong, Green Chem., 2025, 27, 8875–8882 RSC.
- C. Cai, S. Wu, Y. Zhang, F. Li, Z. Tan and S. Dong, Nat. Commun., 2024, 15, 3929 CrossRef CAS PubMed.
- H. Yang, S. Li, J. Zheng, G. Chen, W. Wang, Y. Miao, N. Zhu, Y. Cong and J. Fu, Adv. Mater., 2023, 35, 2301300 CrossRef CAS.
- M. Tang, J. Qiu, Y. Lu, Z. Liu, Y. Liu, C. Luo, C. Fan and R. Wang, Angew. Chem., Int. Ed., 2025, 64, e202514530 CrossRef CAS PubMed.
- Y.-H. Song, Y.-J. Gu, Z. Lei, N.-K. Li, Y.-M. Zhang, Q. Yu and Y. Liu, Nano Lett., 2025, 25, 4476–4484 CrossRef PubMed.
- X.-Y. Dai, M. Huo, X. Dong, Y.-Y. Hu and Y. Liu, Adv. Mater., 2022, 34, 2203534 CrossRef CAS.
- Z. Lei, Y.-H. Song, Y.-L. Leng, Y.-J. Gu, M. Yu, Y. Chen, Q. Yu and Y. Liu, J. Med. Chem., 2025, 68, 5891–5906 CrossRef CAS PubMed.
- S. Song, X. Zhao, H. Zhang, L. Wang and Y. Liu, Small, 2025, e07202 CrossRef CAS.
- X. Zhou, X. Bai, F. Shang, H.-Y. Zhang, L.-H. Wang, X. Xu and Y. Liu, Nat. Commun., 2024, 15, 4787 CrossRef CAS PubMed.
- J. Yu, J. Niu, J. Yue, L.-H. Wang and Y. Liu, ACS Nano, 2023, 17, 19349–19358 CrossRef CAS.
- X.-Y. Dai, B. Zhang, Q. Yu and Y. Liu, J. Med. Chem., 2022, 65, 7363–7370 CrossRef CAS PubMed.
- J. Yu, H. Yu, J. Niu, Z. Lei and Y. Liu, Nano Lett., 2024, 24, 16124–16131 CrossRef CAS PubMed.
- R. Parhi, Environ. Chem. Lett., 2020, 18, 577–594 CrossRef CAS.
- J. Yu, H. Wang, X.-Y. Dai, J. Niu and Y. Liu, Chem. Commun., 2023, 59, 1457–1460 RSC.
- X.-Y. Dai, Q. Song, W.-L. Zhou and Y. Liu, JACS Au, 2024, 4, 216–227 CrossRef CAS PubMed.
- X. Qu, Z. Liu, Z. Wang, D. Yu, X. Huang, J. Yang, J. Ji, X. Wei and C. Yang, Chin. Chem. Lett., 2025, 36, 111024 CrossRef CAS.
- J.-L. Song, C. Chen, X. Li, Y. Jiang, Z. Peng, X.-Q. Wang, H.-B. Yang and W. Wang, Nat. Commun., 2024, 15, 10531 CrossRef CAS PubMed.
- H. Zhu, J. Liu, Y. Wu, L. Wang, H. Zhang, Q. Li, H. Wang, H. Xing, J. L. Sessler and F. Huang, J. Am. Chem. Soc., 2023, 145, 11130–11139 CrossRef CAS PubMed.
- J. Yu, H. Yu, Y. Qiu, H.-Y. Zhang, X. Xu and Y. Liu, Angew. Chem., Int. Ed., 2025, 64, e202418938 CrossRef CAS PubMed.
- Q. Liu, Z. Lei, F. Shang, X. Dong, E. Yang, Y.-M. Zhang, Q. Yu, X. Xu and Y. Liu, CCS Chem., 2025, 0, 1–18 Search PubMed.
| This journal is © The Royal Society of Chemistry 2026 |
