Open Access Article
Open Access ArticleEngineering porous organic polymers for enhanced CO2 capture: from synthesis to implementation
Mohammed G.
Kotp
 and
Shiao-Wei
Kuo
and
Shiao-Wei
Kuo
 *
*
Department of Materials and Optoelectronic Science, Center for Functional Polymers and Supramolecular Materials, National Sun Yat-sen University, Kaohsiung 804, Taiwan. E-mail: kuosw@faculty.nsysu.edu.tw
First published on 7th April 2026
Abstract
The escalating concentration of atmospheric carbon dioxide (CO2) necessitates the development of efficient and scalable carbon capture technologies. Porous organic polymers (POPs) have emerged as a leading class of solid-state adsorbents, offering an exceptional combination of high surface area, tuneable porosity, and robust chemical stability. This review provides a comprehensive analysis of the engineering of POPs for enhanced CO2 capture, traversing the journey from molecular design to implementation. We show the fundamental characteristics of POPs, including their classification and unique structure–property relationships that govern adsorption performance. The core of the review critically examines diverse synthetic strategies for creating POPs, with a focus on tailoring pore architecture and chemical functionality—such as amine incorporation and heteroatom doping—to optimize CO2 capacity, selectivity, and regeneration kinetics. We further assess the performance of POPs under realistic conditions, analysing the critical impact of humidity, co-adsorbates, and long-term cycling on their practical viability. The economic and environmental prospects of POP-based capture are evaluated through techno-economic assessments. Finally, we highlight emerging trends of multi-functional POPs, and outline a roadmap for future research. The review concludes that while challenges in scalability and cost remain, POPs hold immense promise as next-generation adsorbents, with the potential to play a pivotal role in achieving a sustainable and net-zero future.
1 Introduction
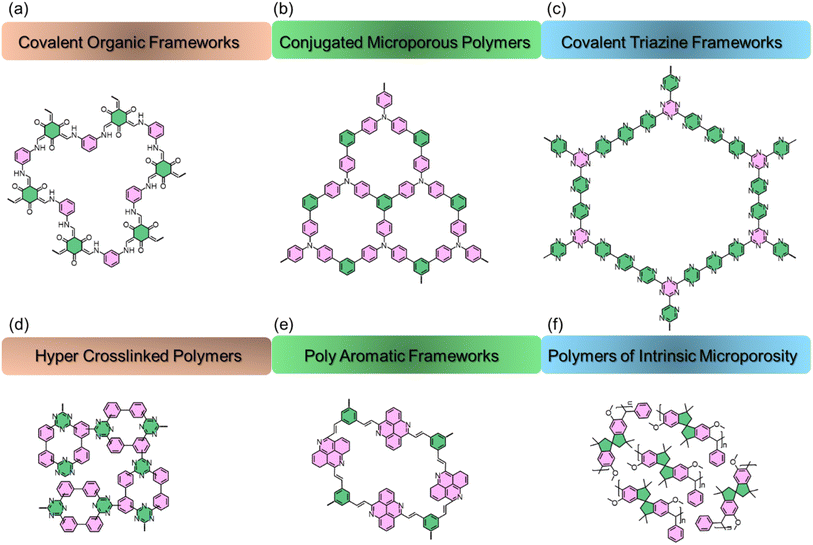
The relentless increase in atmospheric carbon dioxide (CO2) concentration, primarily driven by anthropogenic fossil fuel combustion, represents one of the most pressing challenges of our time.1–3 This accumulation of CO2 is the principal driver of global climate change, manifesting in rising global temperatures, ocean acidification, and an increased frequency of extreme weather events.4 The urgency of this crisis is underscored by international efforts, such as the Paris Agreement, which aim to limit global warming to well below 2 °C above pre-industrial levels.5–7 Achieving this ambitious goal necessitates a multi-pronged strategy that not only transitions to renewable energy sources but also actively deploys technologies for carbon capture, utilization, and storage (CCUS). Notably, beyond its role as a greenhouse gas, CO2 has emerged as a valuable C1 building block for polymerization and advanced materials development. Within this framework, developing efficient and scalable methods for capturing CO2 from point sources like flue gas, or directly from the atmosphere, is a critical scientific and engineering imperative to mitigate the most severe consequences of climate change and ensure a sustainable future.8 In response to this urgent need, a portfolio of carbon capture technologies has been developed, primarily categorized by their separation mechanism.9 The current benchmark, aqueous amine scrubbing, relies on chemisorption where amines react with CO2 to form carbamates.10 While effective, this process is energy-intensive, requiring high temperatures for sorbent regeneration, and suffers from solvent corrosion and degradation.11 Alternative approaches, such as cryogenic distillation and membrane separation, offer different trade-offs between selectivity and energy demand.12 This landscape has thus spurred intensive research into solid adsorbents as a promising alternative, with materials like zeolites, metal–organic frameworks (MOFs), and activated carbons being widely investigated.13 These materials primarily operate through physisorption, which generally offers lower regeneration energies. However, challenges remain in balancing capacity, selectivity, stability, and cost-effectiveness, driving the search for next-generation materials with tailored properties.Amidst the search for advanced solid adsorbents, porous organic polymers (POPs) have emerged as a particularly promising class of materials to address the limitations of existing technologies.14–28 POPs are a diverse family of covalently linked amorphous or semi-crystalline frameworks that boast an exceptional degree of synthetic tunability (Fig. 1) including covalent triazine frameworks (CTFs), conjugated microporous polymers (CMPs), hyper-crosslinked polymers (HCPs), covalent organic frameworks (COFs), polyaromatic frameworks (PAFs), and polymers of intrinsic microporosity (PIMs).
Most critically, this allows for the precise engineering of their pore architecture, surface area, and chemical functionality to enhance CO2 affinity and selectivity. Unlike many benchmark materials, POPs are typically composed of lightweight, robust elements (e.g., C, H, N, O, and B), granting them high physicochemical stability and low regeneration energy requirements. Their fully organic nature also often confers superior hydrothermal stability compared to MOFs, a crucial advantage for processing humid flue gases.29,30 Clearly, while MOFs offer high crystallinity and tunable coordination environments, their performance can be limited under humid conditions due to moisture sensitivity and, in some cases, potential metal leaching. Zeolites exhibit excellent thermal and mechanical stability; however, their rigid inorganic frameworks restrict structural tunability and functional diversity. Activated carbons are economically attractive but generally lack precise control over pore architecture and surface chemistry. In contrast, POPs combine high chemical and thermal stability with metal-free, purely covalent frameworks that allow extensive synthetic flexibility. Their modular design enables controlled pore engineering and heteroatom functionalization to enhance CO2 affinity while maintaining structural robustness. These advantages make POPs particularly promising candidates for adsorption-based CO2 capture under a wide range of operating conditions.
Capitalizing on the immense design versatility of POPs outlined above, this review provides a comprehensive and forward-looking analysis of the engineering evolution of POPs for efficient CO2 capture, bridging molecular-level design with practical implementation. Unlike previous reviews that primarily summarize material performance, this work integrates synthetic strategies, pore environment engineering, and binding-site modulation into a unified structure–property–application framework. We systematically elucidate the fundamental design principles and synthetic methodologies used to construct POPs with tailored porosity and enhanced CO2 affinity.
Beyond evaluating CO2 adsorption capacities across different POP families, we critically analyse structure–property relationships and adsorption mechanisms to extract generalizable design guidelines. Importantly, while several excellent reviews have recently examined POPs for CO2 capture—focusing on synthetic methodologies, individual POP subclasses, or performance metrics—a comprehensive framework that systematically connects molecular design principles with practical implementation challenges remains lacking. This review addresses this gap by uniquely integrating three critical dimensions: (i) the relationship between synthetic strategies and pore engineering outcomes across major POP families (CMPs, COFs, CTFs, and HCPs); (ii) the derivation of generalizable structure–function correlations that guide the rational design of high-performance adsorbents; and (iii) an in-depth analysis of real-world deployment factors—including stability under realistic conditions, shaping and pelletization, mechanical integrity, techno-economic feasibility, and Technology Readiness Level (TRL) positioning. By bridging fundamental materials chemistry with engineering considerations, this review provides a holistic roadmap for advancing POPs from laboratory discoveries toward scalable carbon capture technologies. Furthermore, we assess the suitability of POPs across various capture scenarios (post-combustion, pre-combustion, and direct air capture (DAC)) and identify the key scientific and engineering challenges that must be overcome to translate laboratory advances into real-world carbon capture technologies.
By combining molecular insight with application-oriented evaluation, this review aims to provide a strategic roadmap for the rational design and industrial advancement of next-generation POP-based CO2 adsorbents.
2 Implementation contexts for CO2 capture
POPs can be strategically engineered for CO2 capture implementation in various contexts including post-combustion, pre-combustion, and DAC, as shown in Fig. 2. In pre-combustion capture from high-pressure syngas (H2-rich streams), the focus shifts to POPs with high volumetric surface area and robust frameworks to maximize CO2 uptake through physisorption.31 For the challenging task of DAC, where CO2 concentrations are extremely dilute, POPs require ultra-strong chemisorption sites, such as alkylamines, to effectively bind CO2 molecules from the atmosphere, while maintaining stability for repeated capture–release cycles.32–34 Notably, for post-combustion capture from flue gas (N2-rich streams), POPs are designed with high CO2/N2 selectivity, often achieved by incorporating nitrogen-containing groups like amines for chemisorption or tuning pore sizes for enhanced physisorption.35,36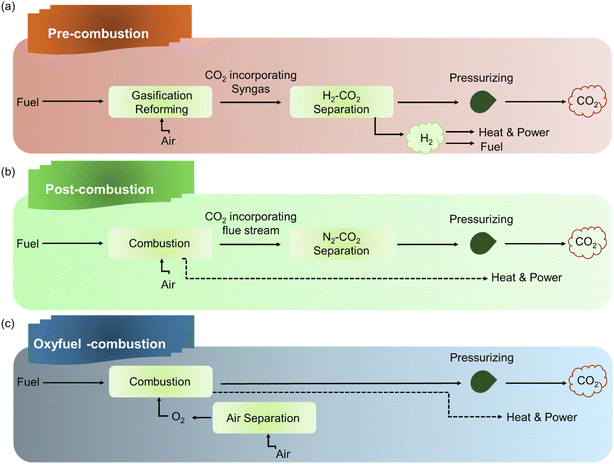 | ||
| Fig. 2 Different approaches used for CO2 capture including (a) pre-combustion capture, (b) post-combustion capture, and (c) oxy-fuel combustion. | ||
2.1 Pre-combustion capture
Pre-combustion capture occurs upstream of combustion, targeting a high-pressure synthesis gas (“syngas”) produced from the gasification of fossil fuels or biomass.37,38 This stream presents a distinct separation challenge, containing a high concentration of CO2 (typically 15–60%) mixed with a valuable H2 product, all under significantly elevated pressures (Fig. 2(a)).39,40 In the pre-combustion capture scenario, the role of POPs shifts towards maximizing performance under high-pressure conditions.41,42 The critical properties become a combination of high CO2/H2 selectivity and high working capacity. Selectivity is essential to ensure the purification of the valuable H2 fuel stream by efficiently separating out CO2. Simultaneously, the material must possess a high working capacity to make the process efficient and economically viable. The focus is on designing POPs that can capture a significant mass of CO2 directly from the high-pressure syngas mixture, leveraging the elevated partial pressure of CO2 to drive high uptake within the polymer's pores.Under the high-pressure conditions characteristic of pre-combustion streams, the adsorption mechanism in POPs relies predominantly on physisorption.43 The elevated CO2 partial pressure favours this type of interaction, as it drives a large amount of gas into the pores. Consequently, the design goal for POPs in this application is twofold: (i) to achieve a high volumetric surface area to create ample space for gas confinement and (ii) to incorporate a high density of CO2-philic sites (such as polar functional groups) within the pores. This synergistic design maximizes the working capacity by ensuring that a significant volume of CO2 can be captured per cycle through enhanced van der Waals and dipole–quadrupole interactions, while still allowing for relatively low-energy regeneration. A defining advantage of POPs in pre-combustion capture is their inherent robustness under harsh operational conditions.44 The syngas environment is often at high pressure and can contain significant moisture, which can degrade less stable materials.45 The extensive covalent bonds forming the backbone of POPs grant them exceptional thermal and mechanical stability, allowing them to withstand high-pressure cycling without structural collapse.46 Furthermore, many classes of POPs demonstrate remarkable hydrothermal stability, resisting degradation in the presence of water vapor.47,48 This durability ensures a long operational lifespan and consistent performance, which is a critical advantage over other adsorbents that may swell, decompose, or lose activity under similar demanding conditions.
2.2 Post-combustion capture
Post-combustion capture targets the removal of CO2 from flue gas, the exhaust produced by burning fossil fuels in power plants and industrial facilities.49,50 This gas stream is primarily composed of N2 and contains a relatively dilute concentration of CO2 at near-ambient pressure and temperature (Fig. 2(b)).51 The central role of POPs in post-combustion capture is to function as molecular sieves that preferentially adsorb CO2 over N2.52 Given the massive abundance of N2 in the flue gas, the most critical performance metric is high CO2/N2 selectivity. POPs are therefore strategically engineered to exhibit a strong thermodynamic affinity for CO2 molecules, ensuring selective capture from the mixed gas stream.53 Actually, this high CO2/N2 selectivity is achieved through two primary adsorption mechanisms, which can be engineered independently or synergistically within the POP framework.54,55 Physisorption relies on weak van der Waals forces. It is optimized by designing POPs with precise ultra-microporosity, where pore diameters are tuned to be less than 1 nm. At this scale, pores closely fit the kinetic diameter of the CO2 molecule, creating a confined environment that significantly enhances the physical interaction energy between the pore wall and CO2, leading to selective adsorption.56–58 On the other hand, chemisorption involves a stronger, more specific chemical reaction. It is facilitated by the strategic incorporation of nitrogen-rich functional groups such as amines, azo groups, and triazine rings into the polymer backbone or as pendant groups.59,60 These nitrogen sites act as Lewis bases, which directly and reversibly chemisorb the acidic CO2 molecule.61 This creates a much stronger binding affinity than physisorption, dramatically increasing selectivity, particularly under low-concentration conditions. A pivotal advantage of POPs in post-combustion capture lies in their robust material properties, which offer significant practical benefits over conventional liquid amine scrubbers.62 As solid adsorbents, POPs circumvent critical operational drawbacks associated with amine solutions, such as solvent evaporation, equipment corrosion, and solvent degradation.63 Furthermore, the covalent bonds within the porous polymer framework provide high thermal and chemical stability, allowing them to withstand the demanding conditions of capture and regeneration cycles.64,65 This intrinsic stability, combined with the typically lower binding energy of physisorptive POPs, often leads to a substantially lower energy for regeneration, directly addressing one of the primary economic and efficiency challenges in large-scale carbon capture.662.3 Oxy-fuel combustion
The oxy-fuel combustion protocol represents a distinct pre-combustion strategy where the primary separation involves oxygen rather than CO₂.67 In this process, fossil fuels are burned in a mixture of high-purity oxygen and recycled flue gas, rather than air. This eliminates the large volume of nitrogen from the system, resulting in a flue gas stream that is primarily composed of CO2 and water vapor, which are easily separated by condensation. The central challenge, therefore, shifts to the energy-intensive air separation step to produce the required pure oxygen (Fig. 2(c)). While the application of POPs in oxy-fuel combustion is less explored than in other capture scenarios, their potential role is significant. POPs can be engineered as high-performance O2/N2 adsorbents for oxygen production.68 The focus here is on designing frameworks with ultra-high O2/N2 selectivity, potentially leveraging the paramagnetic nature of O2 to enhance separation from diamagnetic N2. By enabling more efficient, lower-energy air separation units, POPs could directly address the major cost and efficiency bottleneck of the oxy-fuel process, facilitating the generation of the high-purity CO2 stream that is its ultimate goal.692.4 CH4 purification (natural gas/biogas upgrading)
Beyond flue gas treatments, POPs demonstrate significant utility in the critical energy-related process of methane (CH4) purification, often referred to as natural gas or biogas upgrading. Raw natural gas and biogas extracted from landfills or anaerobic digesters contain CO2 as a primary impurity, which reduces the fuel's energy density and promotes pipeline corrosion. The separation challenge here involves isolating CO2 from a mixture where CH4 is the valuable product, a task complicated by the similar kinetic diameters and non-polar nature of the two molecules. In this scenario, the role of POPs is to act as highly selective sieves for CO2/CH4 separation. The key properties are high CO2/CH4 selectivity and high CO2 working capacity, often at medium to high pressures. This selectivity is engineered by leveraging the differences in molecular properties; CO2 has a significant quadrupole moment and higher polarizability compared to the more inert CH4. POPs can be designed with ultra-microporous pores that preferentially interact with the CO2 molecule through enhanced van der Waals forces or by incorporating polar functional groups (e.g., nitrogen sites or oxygen groups) that engage in dipole–quadrupole interactions with CO2.56,70 By selectively removing CO2, POPs facilitate the production of high-purity, pipeline-quality methane, enhancing fuel efficiency and reducing greenhouse gas emissions from fuel sources.2.5 DAC
DAC represents the most thermodynamically challenging carbon capture methodology. It involves adsorbing CO2 directly from the ambient atmosphere, where it is present at an extremely dilute concentration of approximately 400 ppm. This ultra-dilute nature is the fundamental source of the challenge. This translates to a very low partial pressure of CO2 (around 0.04 kPa), which significantly reduces the driving force for adsorption compared to flue gas streams (where CO2 partial pressure can be 100–1000 times higher). Consequently, the primary hurdle in DAC is not just the capacity to hold CO2, but the affinity required to selectively bind these scarce molecules against a vast background of nitrogen, oxygen, and water vapor.71 This demands sorbents with exceptionally strong and selective binding sites that can effectively capture CO2 at this minimal concentration.72 Furthermore, the energy input required to release the captured CO2 and regenerate the sorbent is intrinsically tied to the strength of this initial binding, creating a critical trade-off between capture efficiency and regeneration cost that is far more acute in DAC than in any other capture technology. Actually, the paramount requirement for POPs in DAC is an ultra-high affinity for CO2. The exceptionally low partial pressure of CO2 in ambient air (∼0.04 kPa) provides a minimal driving force for adsorption, rendering conventional physisorption—which relies on weak van der Waals forces—largely ineffective for the initial capture step. At such extreme dilution, the interaction energy provided by physisorption is often insufficient to overcome the entropy penalty of confining a single CO2 molecule from a vast reservoir of air.73Therefore, POPs engineered for DAC must be functionalized to facilitate strong chemisorption. This is typically achieved by incorporating strong Lewis base sites, such as primary or secondary amines (–NH2 or –NH), into the porous framework.74 These amine groups react reversibly with CO2 to form stable carbamate or bicarbonate species, a chemical reaction that provides the substantial binding energy required to selectively “trap” CO2 molecules even when they are vastly outnumbered by other gases. This chemisorptive mechanism is non-negotiable for achieving meaningful CO2 uptake from the atmosphere, as it creates a sufficiently deep energy well to overcome the thermodynamic limitations of the ultra-dilute source. The central challenge in designing POPs for DAC lies in resolving a fundamental thermodynamic contradiction: the material must possess ultra-high affinity to effectively capture CO2 at trace concentrations, yet it must also allow for low-energy regeneration to release the purified CO2 and recycle the sorbent. This creates a critical trade-off, as the strong chemisorptive bonds (e.g., carbamate formation) necessary for efficient capture from air typically require significant energy input—often in the form of high-temperature steam or vacuum—to break, thereby increasing the overall cost and energy penalty of the process. Consequently, the focus of studies is on creating smart POPs that strike a delicate balance. The objective is to engineer binding sites that are strong enough to sequester CO2 from a 400 ppm source, but weak enough to facilitate release under relatively mild conditions. This involves sophisticated molecular design, such as tuning the alkalinity of amine groups, incorporating cooperative binding effects, or creating humidity-swing mechanisms that use moisture instead of heat to trigger CO2 release. Furthermore, exceptional hydrolytic stability is non-negotiable, as the POPs will be continuously exposed to ambient humidity, which can degrade many organic functional groups and lead to significant performance loss over time. The ideal DAC sorbent is thus a stable, precisely tuned platform where binding strength is optimized not for maximum capture alone, but for the optimal balance between capture efficiency and regeneration economics.
3 Fundamental concepts and metrics in carbon capture
To critically evaluate the performance of POPs for CO2 capture, it is essential to understand the key metrics and terminologies used in the field.3.1 Surface area, porosity, and CO2 capacity
3.2 Enthalpy of adsorption (ΔHads or Qst)
Q st is a critical thermodynamic parameter that describes the strength of the interaction between the CO2 molecule and the adsorbent surface. It is a measure of the heat released during adsorption. Lower ΔHads (typically 20–40 kJ mol−1) suggests physisorption, dominated by weak van der Waals forces. This is advantageous for low-energy regeneration but may result in low selectivity and capacity under dilute conditions.77 Higher ΔHads (typically 50–100 kJ mol−1) indicates chemisorption, involving strong covalent or ionic bonds (e.g., with amine groups). While this provides high selectivity and capacity at low CO2 concentrations, it necessitates higher energy input for sorbent regeneration.783.3 Selectivity
Selectivity quantifies the ability of an adsorbent to preferentially capture one gas (CO2) over another (e.g., N2 or CH4).3.4 Recyclability and stability
Mostly, for any CO2 adsorbent to be viable for industrial applications, it must demonstrate long-term recyclability and stability.81,82 This is evaluated through multiple adsorption–desorption cycles, where the material is exposed to repeated capture and regeneration conditions. First, recyclability is confirmed if the CO2 capacity remains high over numerous cycles (e.g., >10–20 cycles with minimal loss). Furthermore, the material must exhibit sufficient thermal stability to withstand regeneration temperatures, as well as chemical stability against moisture, acidic gases (SOx and NOx), and other flue gas contaminants. These properties are essential for maintaining performance over numerous adsorption–desorption cycles, rendering the material robust and economically viable.4 Evolution of POPs for carbon dioxide capture
The quest for efficient carbon capture has driven the exploration and development of a wide array of porous materials, each with distinct advantages and limitations. The journey often begins with traditional adsorbents, such as zeolites and activated carbons, which primarily rely on physisorption. Zeolites, with their highly ordered, ionic structures, offer high initial CO2 uptake and selectivity under dry conditions but suffer from significant sensitivity to moisture and high regeneration energy.9,83–85 Activated carbons, prized for their high surface area, low cost, and hydrophobicity, typically exhibit lower CO2 selectivity due to their broad pore size distribution.86–88 The landscape advanced with the rise of MOFs. These materials represented a paradigm shift due to their extraordinary surface areas, unparalleled tunability, and precise pore chemistry.89–91 MOFs demonstrated record-breaking CO2 capacities, particularly under high-pressure conditions, and their structures could be post-synthetically modified with amines to enhance selectivity.92,93 However, challenges related to their hydrothermal stability, the cost of metal precursors, and long-term degradation under real flue gas conditions have prompted the search for more robust alternatives.This pursuit has firmly established POPs as a leading-edge class of materials in the carbon capture field. POPs uniquely combine the high surface areas and tunability of MOFs with the robust stability of traditional carbon-based materials.94–96 Constructed entirely from strong covalent bonds between light organic elements, they exhibit exceptional thermal and chemical stability, resisting moisture and corrosive environments.97 Their purely organic nature allows for unparalleled synthetic design, enabling the precise incorporation of CO2-philic functional groups—such as amines, azo groups, or triazines—directly into the polymer backbone.98 This synergy of permanent porosity, structural resilience, and molecular-level design freedom positions POPs as a versatile and highly promising platform, capable of being engineered for the specific demands of post-combustion, pre-combustion, and DAC scenarios.
4.1 Fundamentals of POPs
Basically, POPs are a class of advanced materials characterized by rigid, covalently linked organic subunits that create a permanent and well-defined network of pores.75,99 Their defining feature is their composition, being constructed solely from light, non-metallic elements (e.g., C, H, O, N, and S), which results in high thermal and chemical stability.64,65,100–103 POPs are primarily classified based on the nature of their chemical bonds and synthesis strategies. The main categories include HCPs, which feature extensive covalent linkages; PIMs, which have rigid and contorted backbones that cannot pack efficiently; COFs, which are highly crystalline structures with ordered pores; and CMPs, which incorporate π-conjugated electronic structures.104 This diversity in bonding and structure allows for precise tuning of their physical and chemical properties for specific applications like CO2 capture.17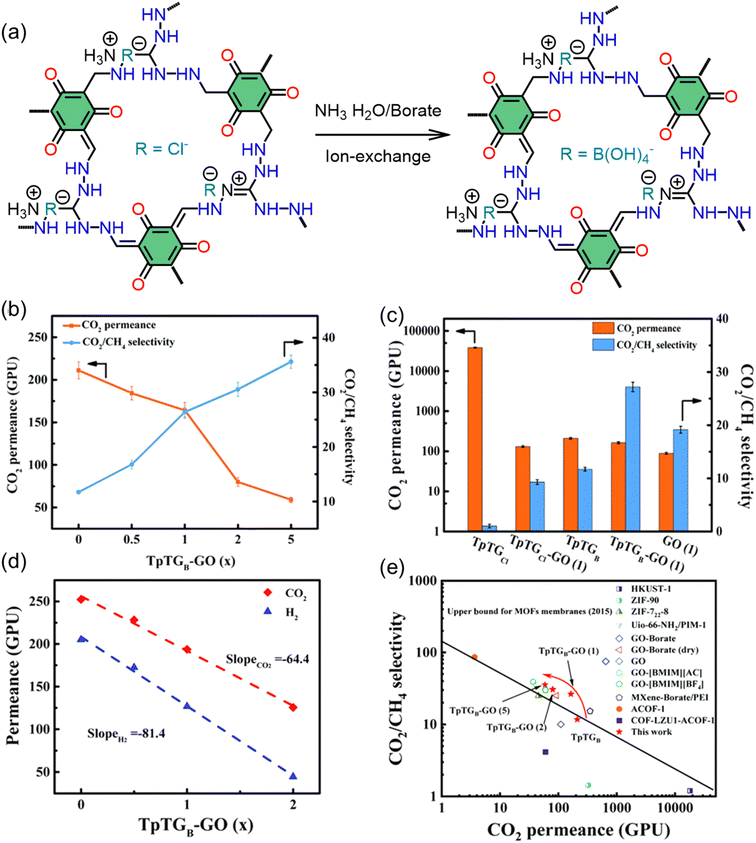 | ||
| Fig. 3 (a) Synthetic route for TpTGCl and its conversion to TpTGB via ion exchange, (b) gas separation records for TpTGB-GO membranes, (c) TpTGB-GO (1) versus control membranes, (d) linear fit showing the relationship between gas permeance and GO content, and (e) comparison of this work's membrane performance with notable 2D and framework membranes.111 Adapted with authorization of the Royal Society of Chemistry. | ||
Very recently, by introducing mesoporous silica templates, Kao et al.112 successfully produced COFs with markedly improved pore ordering, higher surface areas, and more defined channel architectures. These ordered pore systems facilitated more efficient molecular diffusion while enabling precise spatial arrangement of functional groups, ultimately resulting in higher CO2 uptake (3.46 mmol g−1 at 273 K) compared to non-templated analogues. This directly supports the idea that COFs with well-defined, crystalline pore channels provide a platform for atomic-level tenability, allowing targeted incorporation of binding sites that strengthen CO2 interactions and improve selectivity. The superior performance of the templated COFs in CO2 adsorption demonstrates how structural regularity and controllable pore environments amplify adsorption efficiency, aligning with the central claim of this review that ordered and tuneable COF architectures offer unique advantages for high-performance CO2 capture applications as displayed in Fig. 4.
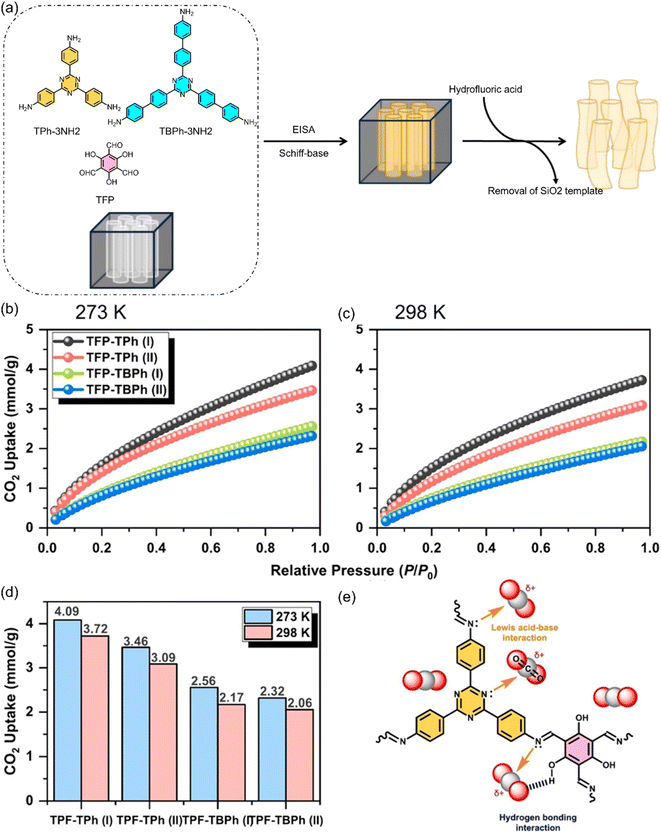 | ||
| Fig. 4 (a) Preparation of TFP-TPh COFs and TFP-TBPh COFs using silica as a template and CO2 adsorption curves of TFP COFs at (b) 273 K and (c) 298 K, with (d) a summary of their CO2 uptake capacities. (e) Suggested adsorption mechanism of CO2 in TFP-based COFs.112 Adapted with authorization of Elsevier. | ||
Furthermore, the combination of high surface area for substantial physisorption capacity and the ability to integrate Lewis basic sites (like nitrogen-rich triazine or imine linkages) directly into the scaffold walls makes COFs compelling candidates for creating highly efficient and selective adsorbents. Their robust covalent bonds, typically boroxine, imine, or triazine linkages, further ensure the thermal and chemical stability required for cyclic capture and regeneration processes. Yu and his group demonstrated that amine-functionalized TaTp-COF achieves exceptional CO2 capture performance by combining high surface area with uniformly distributed secondary amines formed via enol-to-ketoamine tautomerization. The material shows strong CO2 binding, high CO2/N2 selectivity, and excellent stability even under low-concentration flue-gas conditions, confirming the power of integrating Lewis basic sites directly into a porous COF framework. Their results support our claim that merging large physisorption capacity with built-in nitrogen-rich functionalities yields highly efficient and selective CO2 adsorbents (Fig. 5).113
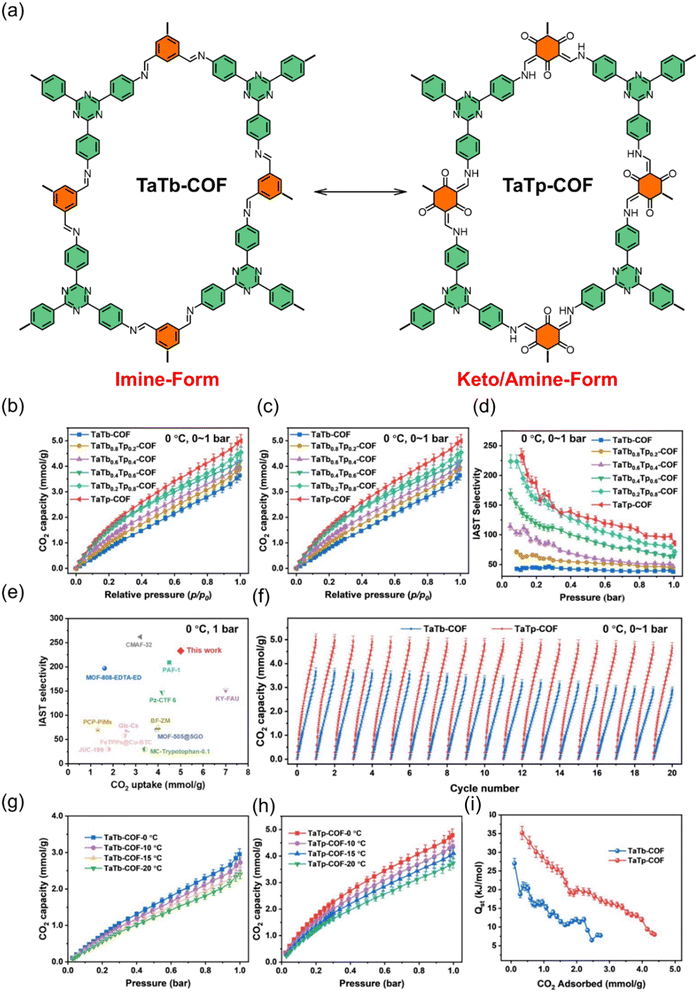 | ||
| Fig. 5 (a) Synthesis and structures of TaTb-COF and TaTp-COF, (b and c) N2 adsorption and CO2 adsorption performance, (d) IAST-calculated CO2/N2 selectivity at 273 K, (e) comparison of CO2 uptake and selectivity with benchmark materials, (f) CO2 cycling stability, (g and h) temperature-dependent CO2 adsorption, and (i) and isosteric heats of adsorption.113 Adapted with authorization of Elsevier. | ||
Our assertion that COFs provide a versatile platform for designing selective adsorbents is strongly supported by recent foundational work from the recent Nobel Prize winner Yaghi and colleagues, which also delineates a critical path for optimization.114 In a seminal study combining advanced simulations and experiments on amine-functionalized COF-999-NH2, they confirmed the platform's designability but uncovered a key challenge for DAC applications. While the modular structure allowed for the precise integration of amine sites (in this case, via reduction of a nitro-functionalized COF), the simulations revealed that water molecules are persistently retained in the pores, forming hydrogen-bonded networks with the nitrile and amine groups. This retained water initiates unwanted side reactions and, crucially, competes directly with CO2 for adsorption sites, undermining the theoretical uptake capacity. The profound insight from this work is that simply adding amine functionalities is insufficient for optimal DAC performance. Instead, Yaghi's group proposes a decisive design rule: to maximize CO2 capture efficiency from humid air, COFs must be engineered with inherently hydrophobic pore environments that actively exclude water while preserving the targeted CO2-binding sites. This work exemplifies the sophisticated structure–property understanding achievable with COFs and directly informs the next generation of designs, moving beyond mere functionalization to the holistic engineering of the pore atmosphere for unparalleled selectivity in the presence of humidity.
Furthermore, this inherent designability of COFs, combining high surface area with integrated functionality, is powerfully validated by recent pioneering work. Yaghi and colleagues addressed the stringent requirements of DAC by synthesizing COF-999, a crystalline framework constructed with robust olefin linkages to ensure stability.115 They then executed a precise post-synthetic modification, covalently grafting amine initiators to grow polyamine chains within its pores—directly integrating high-density Lewis basic sites deriving COF-999-NH2 (Fig. 6). This engineered material exhibits a substantial CO2 capacity from 400 ppm air (0.96–2.05 mmol g−1, humidity-enhanced), fast kinetics (18.8 min to reach half-capacity), and, critically, full regeneration at a low temperature of 60 °C. Most significantly, COF-999 retained 100% of its performance over more than 100 cycles using ambient outdoor air, conclusively demonstrating the thermal, chemical, and cycling stability afforded by its robust covalent architecture. This study stands as a paradigm, proving that the synergistic combination of a stable porous scaffold and precisely incorporated chemisorptive sites in COFs can yield highly efficient, durable, and practical adsorbents for the most challenging capture scenarios.
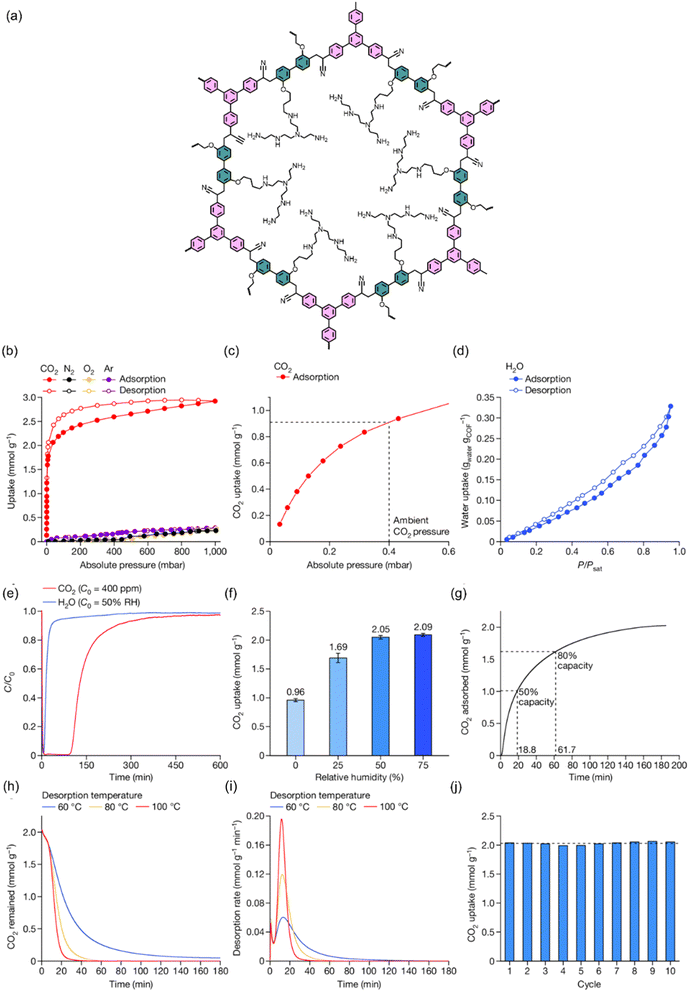 | ||
| Fig. 6 (a) Structure of COF-999-NH2, (b) single-component gas adsorption isotherms at 25 °C. (c) CO2 uptake at ambient pressure (0.4 mbar), (d) H2O vapor sorption isotherm at 25 °C, (e) CO2/H2O breakthrough curves under humid simulated air (400 ppm CO2, 50% RH), (f) CO2 uptake at different relative humidity levels, (g) CO2 adsorption kinetics under humid conditions, (h and i) CO2 desorption kinetics and rates at 60–100 °C, and (j) cycling performance under humid conditions showing an average working capacity of 2.03 mmol g−1.115 Adapted with authorization of Springer Nature. | ||
Earlier, our team reported CMPs built from triphenylamine- and thiadiazole-containing units, showing that tuning the electronic structure of the polymer backbone strongly influences CO2 uptake (Fig. 7). The TPPDA-TPA CMP, which contains more electron-rich and nucleophilic sites, exhibits nearly double the CO2 adsorption capacity of the less electron-rich TPPDA-ThZ CMP, confirming that enhancing pore polarity and electron density significantly improves CO2 affinity and CO2/N2 selectivity. This study directly supports our argument that electron-rich aromatic units and heteroatom incorporation allow CMPs to be engineered for superior CO2 capture performance.65
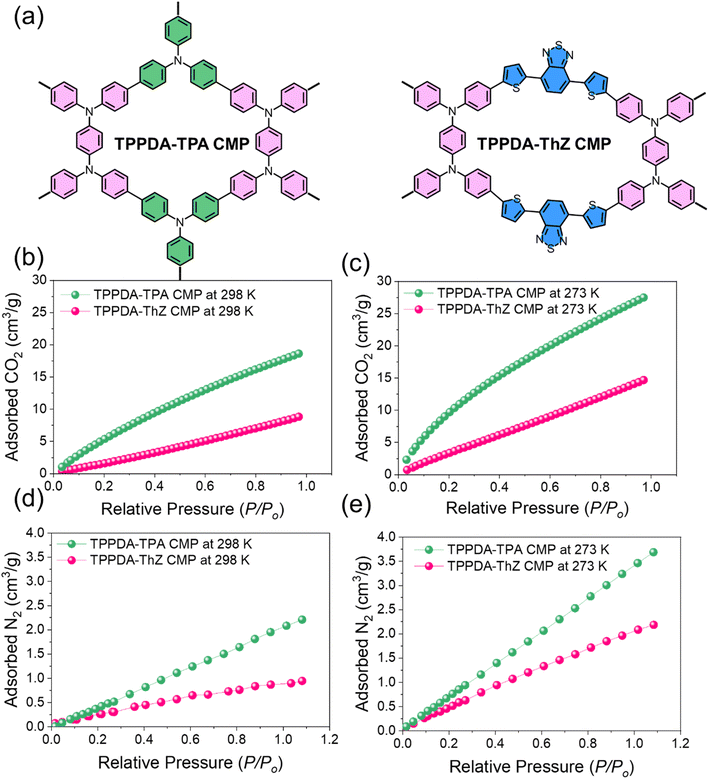 | ||
| Fig. 7 (a) Molecular scheme of TPPDA-TPA and TPPDA-ThZ CMPs; CO2 adsorption of TPPDA-TPA CMP and TPPDA-ThZ CMP at (b) 298 K and (c) 273 K, and their N2 adsorption at (d) 298 K and (e) 273 K.65 Adapted with authorization of the Royal Society of Chemistry. | ||
Furthermore, Mohamed et al.118 demonstrated that engineering CMPs with electron-rich units and abundant heteroatoms (N and O) significantly enhances their CO2 capture performance. Their Anthra-DHTP CMP, which combines a higher BET surface area with polar phenolic OH groups and nitrogen sites, shows substantially higher CO2 uptake and stronger CO2 binding (Qst = 29 kJ mol−1) than the less electron-dense TPE-DHTP CMP. This confirms our review argument that tuning the electronic structure and heteroatom content of CMPs creates a more polar pore environment, strengthening interactions with acidic CO2 and boosting both capacity and selectivity, as shown in Fig. 8.
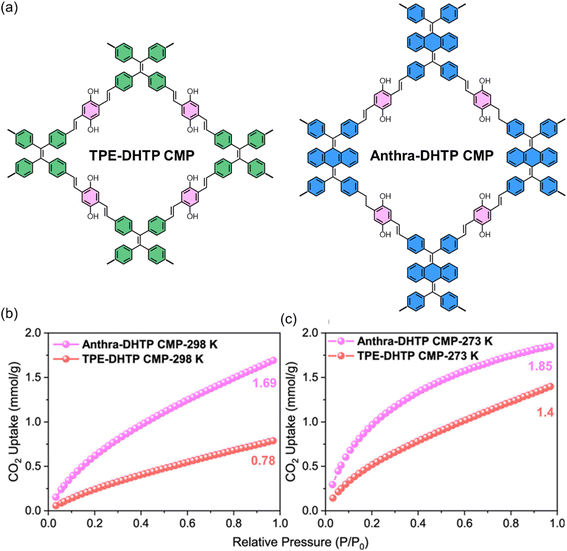 | ||
| Fig. 8 (a) Molecular structures of TPE-DHTP and Anthra-DHTP CMPs and CO2 adsorption performance of TPE-DHTP and Anthra-DHTP CMPs measured at (b) 298 K and (c) 273 K.118 Adapted with authorization of American Chemical Society. | ||
Jia et al. showed that carbazole-based CMPs, which contain electron-rich aromatic units, could be precisely engineered through controlled electropolymerization to adjust their micropore size and fractional free volume. By tuning the polymerization degree, the researchers created CMP membranes with optimized microporosity and enhanced CO2 transport, demonstrating how the electronic structure and packing of CMPs can be finely controlled to improve CO2 affinity and separation performance. Their results strongly support our review's argument that CMPs combine tunable electronic environments with customizable microporous structures, making them powerful platforms for selective and efficient CO2 capture.119 By integrating polar oxygen-containing acyl groups and constructing a 3D network of aligned hollow acyl-functionalized CMPs (AC-CMPs), the material achieves both high CO2 affinity through a polar pore environment and fast mass transfer through hierarchical porosity as displayed in Fig. 9. The resulting AC-CMPs show strong CO2/N2 selectivity, stable performance under humidity and heat, and efficient flue-gas filtration, clearly demonstrating how the chemical tunability and structural versatility of CMPs enable advanced, selective, and robust CO2 capture technologies.120
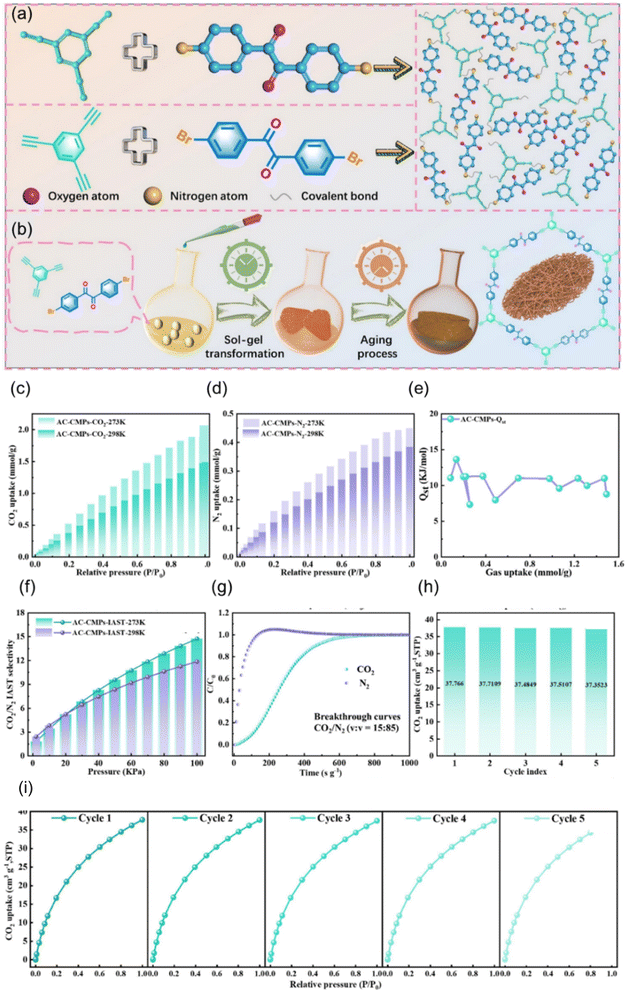 | ||
Fig. 9 (a) Formation of AC-CMP monoliths through self-assembly, (b) schematic of the AC-CMP synthesis process at various stages, (c and d) CO2 and N2 adsorption by AC-CMPs at 273 K and 298 K over 0–1.0 bar, (e) isosteric heat of CO2 adsorption calculated from 273 K isotherms using the Clausius–Clapeyron equation, (f) IAST-predicted CO2/N2 (15![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 85 v/v) selectivity at 273 K and 298 K, (g) CO2/N2 breakthrough curves at 298 K (flow: 10 mL min; CO2: 15%, and N2: 85%; 1 bar), (h) CO2 adsorption capacity over 5 cycles, and (i) cyclic CO2 adsorption performance.120 Adapted with authorization of Elsevier. 85 v/v) selectivity at 273 K and 298 K, (g) CO2/N2 breakthrough curves at 298 K (flow: 10 mL min; CO2: 15%, and N2: 85%; 1 bar), (h) CO2 adsorption capacity over 5 cycles, and (i) cyclic CO2 adsorption performance.120 Adapted with authorization of Elsevier. | ||
Maharana et al. developed nitrogen-rich CTFs (BMTz-CTFs) using a triazole-functionalized benzonitrile monomer, enabling a highly porous structure with abundant N sites. The optimized material (BMTz-CTF600) achieved a very high surface area (1557 m2 g−1), mixed micro/mesoporosity, and partially graphitic domains that together enhance CO2 affinity. As a result, it showed excellent CO2 uptake (5.77 mmol g−1 at 273 K, 1 bar) and high CO2/N2 selectivity (IAST = 82). These nitrogen functionalities also provided strong redox activity, endowing the material with exceptional supercapacitor performance and long-term stability (Fig. 10).126
 | ||
Fig. 10 (a) Synthesis route and proposed chemical structure of BMTz-CTFs, (b) N2 adsorption (filled) and desorption (open) isotherms of BMTz-CTFs at 77 K, (c) pore size distribution calculated from the 77 K N2 isotherm, (d) CO2 adsorption isotherms up to 1 bar, (e) N2 adsorption isotherms up to 1 bar, (f) IAST-predicted CO2/N2 (15![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 85) selectivity at 298 K and (g) isosteric heat of CO2 adsorption.126 Adapted with authorization of the Royal Society of Chemistry. 85) selectivity at 298 K and (g) isosteric heat of CO2 adsorption.126 Adapted with authorization of the Royal Society of Chemistry. | ||
Huang et al.127 developed the first photo-switchable CTFs for low-energy CO2 capture and release. Unlike MOF-based photoresponsive adsorbents—which often suffer from poor thermal and chemical stability—the metal-free CTFs combined high porosity, strong structural stability, and tunable azobenzene photochromic groups. Light irradiation (UV/visible) allowed reversible CO2 adsorption/desorption, and the switching efficiency was further improved by increasing azobenzene content or applying structural alleviation strategies. Actually, the Huang study demonstrates that CTFs can be engineered into highly stable, tuneable, and low-energy CO2-responsive materials, strongly supporting the idea of using CTFs for selective CO2 capture (Fig. 11).
 | ||
| Fig. 11 (a) Schematic representation of CO2 capture and release through chemical transformation (cis/trans), and CO2 adsorption isotherms of (b) PCTF0, (c) PCTF1, (d) mPCTF1, and (e) PCTF2 measured in the dark (black) and after 5 hours of UV irradiation at 273 K (purple).127 Adapted with authorization of John Wiley and Sons. | ||
Earlier, we synthesized nitrogen-rich covalent triazine frameworks (ANT-CTF-10-500 and ANT-CTF-20-500) using an ionothermal method with ZnCl2 at 500 °C.128 These CTFs showed high thermal stability (up to 81% char yield), moderate surface areas (106–170 m2 g−1), and strong CO2 affinity, achieving uptake values up to 2.14 mmol g−1. Their high nitrogen content and polar triazine rings created CO2-philic pore environments, confirming that CTFs intrinsically enhance CO2 adsorption without requiring post-functionalization (Fig. 12).
 | ||
| Fig. 12 (a) Synthesis protocol of ANTs-CTFs and CO2 capture capacity of ANT-CTF-10-500 and ANT-CTF-20-500 at (b) 298 K and (c) 273 K.129 Adapted with authorization of the Royal Society of Chemistry. | ||
Further, Zhao et al. used DFT simulations to investigate how covalent triazine frameworks (pym-CTF) capture CO2 under both dry and humid conditions. They found that CO2 interacts favourably inside the pores, while in humid environments a stable seven-membered ring forms through multiple hydrogen bonds with H2CO3, showing strong affinity under both conditions. Importantly, ammoniating the CTF greatly increased the adsorption energies for CO2 and H2CO3, demonstrating that chemical functionalization can significantly boost capture performance (Fig. 13).130
 | ||
| Fig. 13 Optimized CO2 adsorption configurations on pym-CTF at (a) site 1, (b) site 2, (c) site 3, and (d) site 4.130 Adapted with authorization of Elsevier. | ||
 | ||
| Fig. 14 (a) Mercapto functionalization of TPA-CH POP, (b and c) uptake records of CO2 and (d and e) N2 by the TPA-CH POP and the TPA-CH POP-SH nanocomposite, respectively, at two different temperatures of (b and d) 273 K and (c and e) 298 K.64 Adapted with authorization of MDPI. | ||
For CO2 capture, HCPs offer significant practical benefits. Their synthesis readily generates a high density of ultra-micropores (<1 nm), which are crucial for strong physisorption of CO2 at low pressures due to the overlapping potential from adjacent pore walls.133 While they are traditionally considered physisorbents, their chemical structure provides an excellent platform for post-synthetic modification.134 The aromatic backbone can be easily functionalized to incorporate chemisorptive sites, thereby enhancing CO2 affinity and selectivity.135,136 Combined with their high thermal stability and demonstrated recyclability, HCPs stand out as highly practical and economically viable candidates for large-scale post-combustion carbon capture applications. Alemin et al.136 reported that the combination of porosity engineering with post-synthetic chemical modification to create HCPs can lead to both high CO2 uptake and exceptional CO2/N2 selectivity (Fig. 15). By first tuning the pore structure using different cross-linkers and then introducing polar functional groups (NO2 and NH2), Alemin et al. showed that CO2 selectivity can be dramatically improved even when the surface area decreases. In particular, converting HCP-F-NO2 to HCP-F-NH2 generates strong CO2-binding sites that boost CO2/N2 selectivity to 100—far higher than that of the unmodified framework. Breakthrough and recyclability tests further confirm practical stability.
 | ||
Fig. 15 (a) Post-synthetic modification of dimethoxymethane (HCP-F), (b) CO2/N2 adsorption selectivity of HCP-F-NO2, (c) CO2/N2 adsorption selectivity of HCP-F-NH2, (d) CO2 heat of adsorption for both materials, and (e–g) selectivity was determined from the initial slope of Henry's law at 273.15 K (pressure < 0.3 bar). The heat of adsorption was calculated from CO2 isotherms at 273.15 and 298.15 K. Dynamic CO2/N2 (15![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 85 v/v) breakthrough curves at 298 K and 3 mL min−1 for HCP-F, HCP-F-NO2, and HCP-F-NH2, respectively.136 Adapted with authorization of John Wiley and Sons. 85 v/v) breakthrough curves at 298 K and 3 mL min−1 for HCP-F, HCP-F-NO2, and HCP-F-NH2, respectively.136 Adapted with authorization of John Wiley and Sons. | ||
Recently, Ma and his team et al.52 developed a dually functionalized hyper-cross-linked polymer (HCP-A-S) that combines ultramicropores (0.34 nm) with two strong CO2-binding groups (–NH2 and –SO3H). This design allows the material to precisely sieve CO2 over N2 and significantly enhance CO2 affinity through polar interactions (Fig. 16). As a result, the polymer shows high CO2 uptake, excellent CO2/N2 selectivity, and low-energy regeneration, maintaining full performance over repeated cycles.
 | ||
| Fig. 16 (a) Sulphonation of HCP-A,CO2/N2; (b) selectivity of HCP-A-S and HCP-A, dynamic CO2 capture at 298 K and 1 bar; (c) breakthrough curves for N2 (black), HCP-A (purple), and HCP-A-S (orange); (d) CO2 adsorption stability over six adsorption–regeneration cycles using helium.52 Adapted with authorization of Elsevier. | ||
Liu et al.137 demonstrated a pore-engineering strategy to enhance HCP performance by using Friedel–Crafts alkylation followed by HFA etching to create hydroxyl-functionalized HCPs (HCPs-X-HFA) (Fig. 17). This treatment greatly increases surface area, pore volume, and the number of –OH groups, resulting in high CO2 uptake (up to 3.41 mmol g−1 at 0 °C) and improved CO2/N2 selectivity. The introduced hydroxyl sites not only strengthen CO2 binding through hydrogen bonding but also make the materials effective recyclable catalysts for CO2 conversion into cyclic carbonates under mild conditions (60 °C, 1 bar). Liu's work demonstrates well how post-synthetic pore and functionality engineering can produce HCPs with dual CO2 adsorption and catalytic capabilities, supporting their versatility in carbon capture and utilization.
 | ||
Fig. 17 (a) Synthesis of HCPs-X-HFA (X = 1, 2, or 3) using OPh-POSS and DX in the following ratios: 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 6 (X = 1), 1 6 (X = 1), 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 9 (X = 2), and 1 9 (X = 2), and 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 12 (X = 3), (b) CO2 and (c) N2 adsorption isotherms of HCPs at 0 °C, (d) comparison of CO2 uptake at 0 °C and 1 bar with top-performing materials, (e) CO2 isosteric heat of adsorption (Qst) for HCPs, (f) IAST-predicted CO2/N2 (15 12 (X = 3), (b) CO2 and (c) N2 adsorption isotherms of HCPs at 0 °C, (d) comparison of CO2 uptake at 0 °C and 1 bar with top-performing materials, (e) CO2 isosteric heat of adsorption (Qst) for HCPs, (f) IAST-predicted CO2/N2 (15![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 85 v/v) selectivity at 0 °C, and (g) CO2 adsorption cycling performance of HCPs-2-HFA.137 Adapted with authorization of American Chemical Society. 85 v/v) selectivity at 0 °C, and (g) CO2 adsorption cycling performance of HCPs-2-HFA.137 Adapted with authorization of American Chemical Society. | ||
 | ||
| Fig. 18 (a) Schematic representation of CO2 capture and separation over PAF-45DPA, (b) N2 adsorption–desorption isotherms of PAF-45DPA at 77 K, (c) pore size distribution from QSDFT analysis, (d) N2 adsorption–desorption isotherms of PAF-45 and PDPA, (e) CO2 and N2 adsorption–desorption isotherms of PAF-45, PAF-45DPA, and PDPA at 273 K, and (f) IAST-predicted CO2/N2 selectivity at 273 K for PAF-45DPA, PDPA, and PAF-45.143 Adapted with authorization of Elsevier. | ||
 | ||
| Fig. 19 (a) Structures of PIM-1 and its functionalized derivatives with their infinite-dilution sorption coefficients, (b) Robeson plot comparing pure- and mixed-gas CO2/CH4 separation performance of aged PIMs, and (c) mixed-gas CO2 permeability and CO2/CH4 selectivity of PIM-NH2 as a function of CO2 fraction, with corresponding CH4 permeabilities.146 Adapted with authorization of the Royal Society of Chemistry. | ||
To provide clear guidance for the field, we present a quantitative benchmarking of representative POP subclasses (Table 1), comparing key metrics including CO2 capacity, selectivity, heat of adsorption, stability, and synthesis complexity. This analysis reveals distinct performance trade-offs that inform material selection for specific capture scenarios and highlights opportunities for future innovation.
| POP subclass | Representative material | CO2 (mmol g−1) at 1 bar | Q st (kJ mol−1) | Selectivity (CO2/N2, IAST) | Selectivity (CO2/CH4) | Stability (cycles) | Regeneration temperature (°C) | Synthesis complexity | Ref. |
|---|---|---|---|---|---|---|---|---|---|
| COFs | TaTp-COF | 5.0 at 273 K | 35 | 233 (15![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 85) 85) |
NA | Excellent (>10 cycles) | Not specified | Moderate | 113 |
| CMPs | AC-CMPs | 2.07 at 273 K | 13 | 15 (15![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 85) 85) |
NA | Excellent (>5 cycles) | 60 °C | Moderate | 120 |
| CTFs | BMTz-CTFs-600 | 5.77 at 273 K | 38 | 82 (50![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 50) 50) |
NA | Not specified | Not specified | High | 126 |
| HCPs | HCPs-3-HFA | 3.41 at 273 K | 29.2 | 65 (15![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 85) 85) |
NA | 75% (>7 cycles) | Not specified | Low | 137 |
| PAF | PAF-45DPA/PSF | 3.55 mmol g−1 | 32.7 | 63 (15![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 85) 85) |
N/A | 79 GPU 10-day period | Not specified | Simple | 143 |
| PIMs | PIM-1 | — | — | N/A | 13.5 ± 0.8 | N/A | N/A | Moderate | 146 |
4.2 Structure–property relationships in POPs
The performance of POPs in CO2 capture is not a product of chance but is directly governed by their molecular-level architecture.147 Understanding the structure–property relationship is fundamental to the rational design of next-generation adsorbents. These relationships can be analyzed in terms of the interplay among the building blocks, the resulting porous network, and the chemical functionality.4.2.2.1 Surface area. The high BET surface area provides a greater number of adsorption sites, generally correlating with higher gas uptake capacity, particularly for physisorption.151 Liao et al.152 developed hydroxyl-functionalized ionic hyper-cross-linked polymers using a one-step Friedel–Crafts method, achieving very high surface areas (560–1480 m2 g−1) and rich microporosity. These structural features, combined with ionic sites, resulted in exceptionally high and reversible CO2 uptake, reaching 157.5 mg g−1 at 273 K and 1 bar—surpassing many reported ionic polymers. The materials also served as efficient, recyclable catalysts for CO2 conversion due to synergistic effects from hydroxyl groups. This study clearly demonstrates that maximizing BET surface area significantly enhances available adsorption sites and directly boosts CO2 capture performance, reinforcing the importance of high-surface-area porous polymers in selective CO2 adsorption (Fig. 20).
 | ||
| Fig. 20 (a) Molecular representation of the rational design of ionic HCPs, (b–f) CO2 adsorption isotherms of HCPs measured from 273 to 338 K, and (g) comparison of CO2 uptake among the different HCPs.152 Adapted with authorization of Elsevier. | ||
4.2.2.2 Pore size. Actually, this is a critical parameter for selectivity as ultra-micropores (<0.8 nm) exert a strong van der Waals potential on confined CO2 molecules (kinetic diameter ∼0.33 nm), leading to high adsorption energy and excellent molecular sieving for separations such as CO2/N2. In this context, He et al.153 demonstrated that both intrinsic defects and precise pore size control are critical for maximizing CO2 adsorption in porous carbons. Their combined experimental and multiscale simulation study showed that defects distort graphene layers, creating more micropores and additional high-energy adsorption sites that strengthen CO2 binding. Through slit-pore simulations, they identified an optimal pore size of ∼7 Å, where CO2 forms a stable monolayer with the highest adsorption energy; larger pores lead to weaker bilayer adsorption. Defect-rich activated carbons (VAC700 and VAC900) also generated more ultramicropores, further enhancing CO2 uptake.
4.3 Chemical functionality and binding energy
The chemical composition of the pore walls dictates the strength of the CO2–framework interaction. | ||
| Fig. 21 (a) Tuneable amine functionalized POPs utilized for CO2 capture, dynamic CO2/N2 (15/85 v/v) breakthrough curves for amino-functionalized POPs at 298 K and 1.0 bar (flow rate: 0.5 mL min−1): (b) PDVB, (c) POP-1, (d) POP-2, (e) POP-3, (f) POP-3, (g) POP-4, (h) POP-5, and (i) POP-5, and (j) comparison of breakthrough performance for PDVB and all amino-functionalized POPs.154 Adapted with authorization of Elsevier. | ||
The most effective POPs for CO2 capture are designed by synergistically combining these elements. An ideal adsorbent might feature a rigid 3D scaffold to ensure stability, a high density of ultra-micropores for strong physisorption, and a strategic distribution of amine groups for enhanced selectivity. This allows for the optimization of the trade-off between high capture capacity (governed by porosity) and strong affinity (governed by chemistry), while maintaining the structural integrity required for long-term recyclability.
4.4 Computational insights and predictive modelling
Molecular dynamics (MD) simulations provide a powerful computational lens through which researchers can observe and understand the dynamic behaviour of CO2 molecules within the porous landscapes of POPs. Unlike static models, MD simulates the motions of atoms and molecules over time under specified conditions of temperature and pressure, governed by a force field that describes interatomic interactions. This approach offers unique, time-resolved insights that are often inaccessible through experimentation alone. The primary strength of MD simulations in CO2 capture research lies in elucidating transport phenomena and dynamic adsorption processes.Jeong et al.155 developed nitrogen-rich POPs and incorporated them into mixed-matrix membranes to enhance CO2/N2 separation. MD simulations, combined with experiments, showed that these POP fillers improve CO2 separation by increasing both CO2 solubility and diffusivity within the membrane—effects directly linked to the nitrogen-functionalized porous structure. MD played a key role in revealing how CO2 molecules move through the POP-filled membrane, identifying faster CO2 transport pathways and stronger CO2–polymer interactions compared with N2 (Fig. 22). Consequently, MD simulations bridge the gap between the static structure of a POP and its dynamic performance. By providing atomistic-level details on gas motion and host–guest interactions, they serve as an indispensable tool for interpreting experimental results, validating hypotheses, and guiding the design of next-generation POPs with optimized pore geometries and surface chemistries for fast and selective CO2 capture.
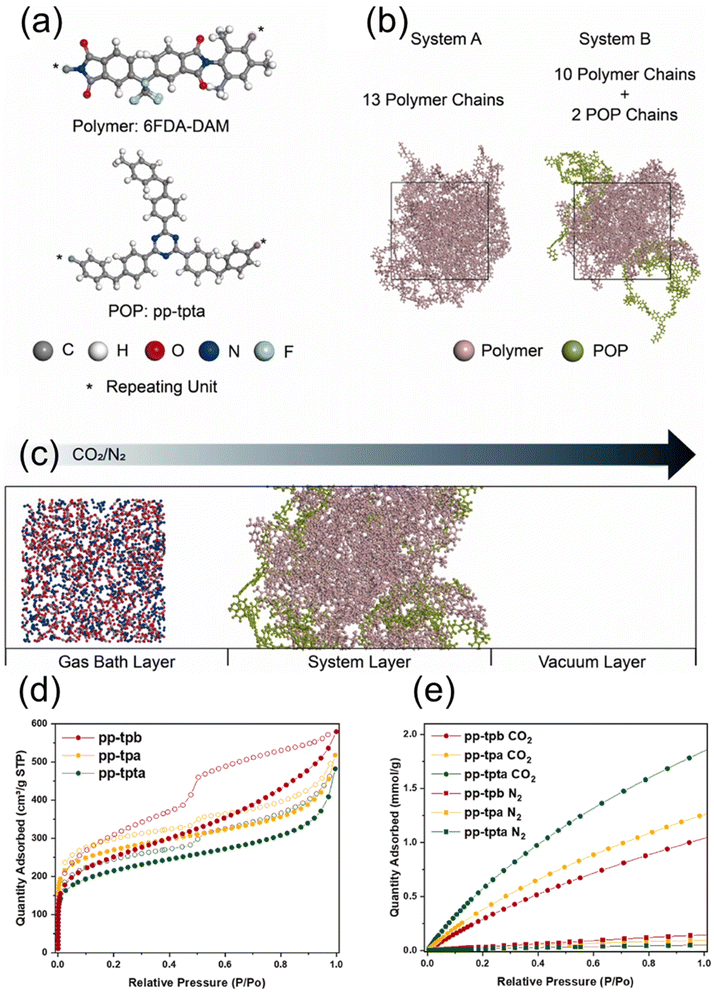 | ||
| Fig. 22 (a) Molecular structures of the polymer (6FDA-DAM) and the POP (POP, pp-tpta), (b) equilibrated models of system A (pure polymer) and system B (polymer/POP blend), including system details, (c) NEMD simulation setup showing the gas bath, membrane layer, and vacuum region, where a mixture of 200 CO2 and 800 N2 molecules diffuses along the z-axis, (d) N2 physisorption isotherms of POPs at 77 K, and (e) CO2 and N2 adsorption isotherms of POPs at 25 °C.155 Adapted with authorization of Elsevier. | ||
5 Performance under real-world conditions
5.1 Effect of humidity and water vapor
The performance of POPs under real-world flue gas or ambient air conditions is critically dependent on their interaction with water vapor, which is a major and ubiquitous component. The presence of humidity can have complex and often competing effects on CO2 capture performance, ranging from cooperative enhancement to detrimental competition, making it a paramount consideration for practical implementation.156 | ||
| Fig. 23 (a) CO2 capture over the hierarchically porous amine moiety, (b) effect of temperature and (c) effect of humidity on the CO2 adsorption capacity of polyHIPE-V1T2-HCP-NH.159 Adapted with authorization of Elsevier. | ||
5.2 Presence of co-adsorbates (SO2, NOx, and O2)
Real-world flue gas is a complex mixture containing not only CO2 and N2 but also trace acidic and oxidative impurities, primarily sulfur oxides (SOx), nitrogen oxides (NOx), and oxygen (O2). The presence of these co-adsorbates poses a significant challenge to the long-term performance and stability of POPs, as they can compete for adsorption sites, react irreversibly with the framework, and lead to permanent degradation.5.3 Temperature and pressure variations
The performance of POPs in CO2 capture is not static but is profoundly influenced by the operational temperature and pressure of the gas stream. Understanding these dependencies is crucial for selecting the appropriate adsorbent for a given capture scenario and for designing an efficient adsorption–desorption cycle. | ||
| Fig. 24 Molecular visualizations of (a) ITQ-29 and (b) zeolite 5A. Atom colors: Si (yellow), O (red), Al (pink), Na (purple), and Ca (green). Lattice parameters: ITQ-29, a = b = c = 23.70 Å; zeolite 5A, a = b = c = 24.84 Å. (c) van't Hoff plot of CO2 adsorption on zeolite 5A, with the negative slope indicating an exothermic process, and (d) isosteric heat of adsorption (Qst) versus CO2 uptake, showing a decrease with higher loading due to the gradual filling of lower-energy adsorption sites.168 Adapted with authorization of MDPI. | ||
5.3.2.1 Post-combustion capture (low pressure). At the near-ambient pressures of flue gas, the performance of a POP hinges on its uptake in the low-pressure region of the isotherm (0–1 bar). Materials must be engineered for strong affinity (e.g., ultra-micropores and amines) to achieve meaningful capacity under these dilute conditions.169
5.3.2.2 Pre-combustion capture (high pressure). In high-pressure syngas streams (tens of bars), the POP's performance is determined by its high-pressure capacity. Here, a high surface area and large pore volume are critical to maximize the amount of CO2 that can be stored via physisorption. The working capacity for a Pressure Swing Adsorption (PSA) cycle is the difference in uptake between the high adsorption pressure and the low desorption pressure.133
5.4 Long-term stability and degradation
The transition of POPs from laboratory marvels to industrially viable adsorbents is contingent upon their long-term stability under continuous operation. This encompasses the material's ability to retain its structural integrity, porosity, and chemical functionality over thousands of adsorption–desorption cycles in the presence of real flue gas constituents. Degradation, the irreversible loss of performance, can occur through several distinct but often interconnected mechanisms.5.4.1.1 Hydrolytic degradation. Sensitive chemical linkages, such as boronate esters, some imines, or esters, can be susceptible to cleavage in the presence of heat and moisture, leading to a collapse of the porous network.170
5.4.1.2 Oxidative degradation. Trace oxygen O2 in the flue gas, especially at elevated regeneration temperatures, can gradually oxidize the polymer. This is particularly detrimental to amine functionalities, which can be converted to nitro groups or other oxidized species, losing their ability to chemisorb CO2. The conjugated backbones of CMPs are also vulnerable.171,172
5.4.1.3 Acidic gas attack. The presence of SOx NOx can cause irreversible chemical poisoning. These strongly acidic gases form stable salts (sulfates and nitrates) with basic sites like amines, permanently removing them from the adsorption pool.173,174
5.5 From powder to product: shaping, cost, and technology readiness of POPs
For POPs to move from the research laboratory into real industrial applications, they must be transformed from fine powders into practical forms that can be used in large-scale equipment. The as-synthesized POPs are typically fine powders that would cause a high-pressure drop and handling difficulties if used directly in an industrial column, so they must be shaped into larger, robust forms such as pellets, beads, or monoliths. The most common method is binder-assisted granulation, where the POP powder is mixed with a binder material like clay or another polymer and then pressed or extruded into pellets. Choosing the right binder is a balancing act: while a small amount can significantly increase pellet strength, too much binder or the pressing process itself can block the POP's pores and reduce its CO2 capture capacity. A more advanced approach is monolith fabrication, where the material is shaped into a honeycomb-like structure with straight, open channels that allow gas to flow through with very little resistance, making them ideal for fast cycling processes. Once shaped, mechanical strength is essential for long-term use. Inside an adsorption column, pellets and monoliths must withstand constant gas pressure, temperature changes during regeneration, and the weight of the material itself without cracking or crumbling. Researchers have shown that optimized pellets can withstand pressures, while emerging monolithic POP structures demonstrate good resilience by bouncing back after compression. The cost of the final shaped product is another major factor in commercial viability. Key cost drivers include the price of starting materials, where using cheap, widely available chemicals like common diamines can potentially bring material costs down. The synthesis method itself matters too—traditional routes using toxic solvents and high temperatures add cost, while greener methods using benign solvents like ethanol at room temperature have already been scaled to produce sub-kilogram quantities. The shaping process also adds its own cost, which must be factored into the final price. To measure where this technology stands, researchers use the TRL scale from 1 (basic research) to 9 (proven in operation). Most POP research currently sits at TRL 2–3, where concepts are proven in the lab. Reaching the next levels will require scaling up synthesis to produce kilograms of material, proving that shaped pellets or monoliths can withstand thousands of capture-and-release cycles using real flue gas containing water and impurities and successfully testing the material in small pilot-scale reactor systems.1796 Emerging trends and future directions
6.1 Machine learning and AI-driven design
The field of porous material discovery is undergoing a paradigm shift with the integration of Machine Learning (ML) and AI, moving away from reliance on intuition towards a predictive, data-driven design cycle for POPs. This approach leverages computational power to uncover complex, non-linear relationships between a POP's chemical structure, its synthesis conditions, and its resulting properties, thereby dramatically accelerating the development of next-generation materials for CO2 capture.6.2 High-throughput screening approaches
Complementing AI-driven design, high-throughput screening (HTS) represents a powerful experimental paradigm for rapidly evaluating vast libraries of POPs. This approach adapts methodologies from pharmaceutical and materials science to systematically synthesize and test numerous POP variants in parallel, dramatically accelerating the empirical discovery and optimization process. By miniaturizing and automating synthesis and characterization, HTS enables a comprehensive exploration of the multi-dimensional parameter space governing POP performance.6.3 Multi-functional POPs
Moving beyond their role as passive adsorbents, a frontier in POPs research is the design of multi-functional materials that not only capture CO2 but also actively transform it or perform additional valuable tasks within a single integrated platform. This approach enhances the economic viability and utility of carbon capture by creating systems that do more than just sequester CO2, potentially enabling a circular carbon economy. The development of multi-functional POPs represents a shift from a singular focus on adsorption metrics towards a holistic design of “smart” materials that integrate capture, sensing, and conversion. This model not only addresses the challenge of CO2 emissions but also adds value to the capture process, creating a more compelling and sustainable pathway for its implementation.6.4 DAC applications
The application of POPs in DAC represents one of the most challenging yet potentially transformative directions for this polymer class. DAC requires the selective extraction of CO2 from the ultra-dilute atmosphere, a task with unique thermodynamic and kinetic demands that push the limits of adsorbent design. POPs, with their unparalleled synthetic tunability, are at the forefront of developing next-generation solid sorbents for this critical negative emissions technology.6.5 Next-generation synthesis methods
The pursuit of commercially viable POPs for CO2 capture is driving innovation not only in molecular design but also in every method used to create them. Next-generation synthesis aims to exceed the limitations of traditional solvothermal routes, focusing on principles of scalability, sustainability, and precision to enable the practical production of advanced POPs. These emerging methodologies seek to make the synthesis faster, greener, and more amenable to industrial manufacturing.The pursuit of sustainable POPs can be guided by recent advances in biomass-derived carbons. For example, phosphorus-doped carbons from lotus petiole waste using sodium phytate as a green activating agent achieve notable CO2 uptake (2.51 mmol g−1) through a one-step, environmentally friendly process.180 Similarly, the use of sodium metaborate tetrahydrate (NaBO2·4H2O) as a non-corrosive alternative to KOH or ZnCl2 for activating water chestnut shells represents a significant step towards greener activation chemistry.181,182 These examples demonstrate that high performance and sustainability can be synergistically achieved.
Importantly, the future of sustainable CO2 capture lies in the convergence of performance with green chemistry. For POPs, this translates directly into several of our actionable research directions, including prioritizing bio-based monomers (e.g., from lignin, vanillin, tannins, or other renewable resources) over petrochemical-derived building blocks; adopting solvent-free (mechanochemical) or aqueous-based methods and replacing toxic or precious-metal catalysts with earth-abundant or organocatalysts; and moving beyond performance metrics (capacity and selectivity) to systematically evaluate the environmental footprint of the entire material life cycle using Life Cycle Assessment (LCA) tools.
By embedding these principles into the design pipeline, the next generation of POPs can achieve not only high efficiency but also true environmental sustainability, ensuring that they contribute positively to the net-zero goal they are intended to serve.
7 Challenges and opportunities
7.1 Current limitations
Despite their considerable promise, the widespread deployment of POPs for CO2 capture faces several significant challenges that must be addressed to transition from laboratory excellence to industrial relevance.7.2 Scalability barriers
A primary obstacle is the difficulty in synthesizing many high-performance POPs on a kilogram-to-ton scale. Actually, the intricate, multi-step synthesis of organic building units is often cost-prohibitive and resource-intensive at large scales. Further, many protocols rely on extended reaction times (days), high temperatures and pressures, and air-sensitive catalysts, which pose significant engineering and safety challenges for scale-up. Furthermore, the use of large volumes of toxic, high-boiling-point solvents generates substantial waste, and the energy-intensive purification (e.g., Soxhlet extraction) becomes impractical commercially. Most importantly, processing the synthesized powder into mechanically robust, shaped bodies for use in industrial reactors without losing porosity or performance remains a non-trivial challenge.7.3 Stability issues
Long-term operational stability under real flue gas conditions is a critical concern. Firstly, polymer frameworks, particularly those with hydrolytically sensitive linkages (e.g., boronate esters and some imines), can decompose in the presence of heat and moisture. Furthermore, functional groups like amines are susceptible to oxidative degradation by O2 or irreversible poisoning by SOx and NOx. Secondly, over multiple adsorption–desorption cycles, some amorphous POPs can undergo physical aging and pore collapse, especially when exposed to steam during regeneration, leading to a permanent loss of surface area and capacity. Thirdly, in dynamic reactor systems, POP particles must resist abrasion and fragmentation to prevent powdering and increased pressure drop.7.4 Cost competitiveness
The overall cost of CO2 capture using POPs must be competitive with established technologies like amine scrubbing. Notably, the combined cost of complex monomers, precious metal catalysts, and solvent management often results in a prohibitively high price for the final adsorbent. Moreover, while – in many reported cases – some POPs offer lower regeneration energy than amines, this advantage must be substantial enough to offset their potentially higher initial cost. The energy required for compression and vacuum pumping also contributes to the operational expense. On the other hand, a short operational lifespan due to chemical or physical degradation necessitates frequent sorbent replacement, severely impacting the levelized cost of capture. A material with a high initial capacity but poor stability is ultimately not economically viable.7.5 Opportunities for innovation
The challenges and knowledge gaps facing POPs simultaneously delineate a rich landscape for transformative innovation. By addressing these hurdles through creative scientific and engineering approaches, POPs have the potential to redefine the economics and efficiency of carbon capture.8 Conclusions and outlook
This review has detailed the significant progress in engineering POPs for CO2 capture, establishing them as a formidable class of materials due to their exceptional tunability, high surface area, and robust frameworks. Our review demonstrates that the strategic design of pore architecture is paramount for achieving strong physisorption and high selectivity, while chemical functionalization, particularly amine grafting and heteroatom doping, dramatically enhances CO2 affinity and selectivity in low-pressure scenarios such as post-combustion capture and direct air capture. Furthermore, the stability of the covalent bonds within the framework is a critical differentiator, with materials featuring C–C, triazine, and other robust linkages demonstrating superior resistance to harsh operational conditions compared to more labile counterparts. While performance in idealized laboratory settings is promising, real-world viability is contingent upon performance under realistic conditions, including the presence of moisture, trace oxidants, and acidic gases, which can lead to competitive adsorption and degradation. To transition POPs from academic interest to industrial reality, future research must focus on several concrete and actionable directions. First, targeted pore engineering should move beyond surface area maximization to focus on the precise design of ultra-micropores (<0.8 nm) with narrow size distributions tailored for specific CO2 partial pressures, achieved through molecular building blocks with fixed geometries that lock in optimal pore dimensions, while hierarchical pore architectures integrating mesopores can enhance adsorption kinetics. Second, advanced heteroatom functionalization should explore cooperative binding sites—such as amine–hydroxyl pairs or amine–triazine combinations—that work synergistically to achieve an enthalpy of adsorption in the “Goldilocks zone” (50–70 kJ mol−1), balancing strong selectivity with low-temperature regeneration (≤80 °C), while systematically investigating doping density and spatial distribution to maximize site utilization without pore blocking. Third, scalable and green synthesis must prioritize solvent-free or aqueous-based routes using earth-abundant, low-cost monomers derived from biomass or commodity chemicals, replacing precious metal catalysts with organocatalysts or recyclable heterogeneous catalysts and transitioning from batch to continuous flow manufacturing for consistent, large-scale production with reduced environmental footprints. Fourth, the development of hybrid and composite materials—including POP-polymer mixed-matrix membranes, POP-metal nanoparticle composites for integrated capture and conversion, and POP-ionic liquid hybrids—offers untapped potential for multifunctional performance. Fifth, dedicated studies on long-term stability under realistic conditions are needed to elucidate molecular degradation mechanisms, guiding the design of self-healing or protective frameworks and the development of accelerated aging protocols that reliably predict multi-year performance. Sixth, data-driven discovery and validation require the creation of open-access, standardized databases to train robust machine learning models for high-throughput virtual screening and inverse design, predicting exact monomer combinations and topologies required to meet specific performance targets before experimental validation. Finally, application-specific design must tailor POPs for individual capture scenarios: ultra-microporous, hydrolytically stable frameworks with moderate amine density for post-combustion capture; ultra-high surface area materials with large pore volumes for pre-combustion capture; frameworks with hydrophobic pore environments that exclude water while hosting optimized amine sites for DAC; and size-selective pores for CO2/CH4 separation with minimal methane loss for natural gas upgrading. By systematically addressing these concrete research directions—moving from broad statements to targeted, actionable strategies—POPs are poised to transition from academic curiosity to a cornerstone technology in the global effort to achieve a net-zero future.Author contributions
Mohammed G. Kotp: writing – review & editing, writing – original draft, visualization, investigation, data curation, conceptualization. Shiao-Wei Kuo: review & editing, resources, project administration.Conflicts of interest
There are no conflicts to declare.Data availability
No primary research results, software or code have been included and no new data were generated or analysed as part of this review.Acknowledgements
The study was supported by the National Science and Technology Council, Taiwan (NSTC 114-2223-E-110-001).References
- Q. Ge, Y. Liu, W. You, Y. Li, W. Wang, L. Yang, L. Xie, K. Li, L. Wang and M. Ma, Sci. Adv., 2025, 11, eadx5714 CrossRef CAS PubMed.
- B. Shao, Y. Jiang, S. Li, Z. Xie, Z.-Q. Wang, S. Dai, H. Liu, F. Qian and J. Hu, ACS Catal., 2025, 15, 18315–18325 CrossRef CAS.
- Y.-S. Chang, M.-Y. Ho, C.-W. Wu and Y.-J. Chang, Process Saf. Environ. Prot., 2025, 200, 107419 CrossRef CAS.
- S. R. Pacella, C. A. Brown, R. G. Labiosa, B. Hales, T. C. Mochon Collura, W. Evans and G. G. Waldbusser, J. Geophys. Res., Oceans, 2024, 129, e2023JC020313 CrossRef CAS PubMed.
- A. P. Schurer, M. E. Mann, E. Hawkins, S. F. Tett and G. C. Hegerl, Nat. Clim. Change, 2017, 7, 563–567 CrossRef PubMed.
- S. I. Seneviratne, J. Rogelj, R. Séférian, R. Wartenburger, M. R. Allen, M. Cain, R. J. Millar, K. L. Ebi, N. Ellis and O. Hoegh-Guldberg, Nature, 2018, 558, 41–49 CrossRef CAS PubMed.
- M. Meinshausen, J. Lewis, C. McGlade, J. Gütschow, Z. Nicholls, R. Burdon, L. Cozzi and B. Hackmann, Nature, 2022, 604, 304–309 CrossRef CAS PubMed.
- Y. Kumar and J. Sangwai, Energy Fuels, 2025, 39, 5007–5033 CrossRef CAS.
- S. A. Ali, S. N. Shah, M. A. Karim, S. A. M. Hashmi, F. Ahmad, K. Habib, A. Sami and M. Abdullah, Energy Fuels, 2025, 39, 9285–9315 CrossRef CAS.
- P. Stiernet, A. Verdin, N. Johnsson, A. Jaworski, D. Prietzel, N. Hedin and J. Yuan, ACS Appl. Mater. Interfaces, 2025, 17, 60400–60410 CrossRef CAS PubMed.
- H. Qiu, Q. Long, Y. Bai, X. Fu, C. Liu, G. Jing, Z. Zhou and B. Lv, Chem. Eng. J., 2025, 520, 165388 CrossRef CAS.
- P.-Y. Liu, Y.-F. Lu, Y.-C. Kuo and Y.-J. Lin, Ind. Eng. Chem. Res., 2025, 64, 24779–24796 CrossRef CAS.
- Y. Song, J. Li, D. Chi, Z. Xu, J. Liu, M. Chen and Z. Wang, Chem. Commun., 2025, 61, 15972–16001 RSC.
- M. G. Mohamed, M. G. Kotp, A. Osama Mousa, Y.-S. Li and S.-W. Kuo, ACS Appl. Energy Mater., 2025, 4, 2389–2402 CrossRef.
- C.-W. Hsiao, A. M. Elewa, M. G. Mohamed, M. G. Kotp, M. M.-C. Chou and S.-W. Kuo, Colloids Surf., A, 2024, 699, 134658 CrossRef CAS.
- M. M. Ayad, W. A. Amer and M. G. Kotp, Mol. Catal., 2017, 439, 72–80 CAS.
- Y.-C. Kao, P.-H. Chen, C.-Y. Chen, H.-W. Chen, W.-C. Chen, M. Ejaz, M. G. Kotp, M. G. Mohamed, H. Karim and S.-W. Kuo, Polymer, 2025, 338, 129041 CrossRef CAS.
- M. G. Kotp, S.-W. Kuo and A. F. M. EL-Mahdy, Colloids Surf., A, 2024, 685, 133210 CrossRef CAS.
- M. Hussain, A. S. Ali, T. Kousar, F. Mahmood, A. Haruna, Z. U. Zango, H. Adamu, M. G. Kotp, I. A. Abdulganiyyu and B. E. Keshta, Sustain. Chem. One World, 2025, 5, 100047 CrossRef.
- M. G. Kotp, J. Lüder, S.-W. Kuo and A. F. M. EL-Mahdy, Mater. Adv., 2024, 5, 4142–4150 RSC.
- M. G. Kotp, A. F. M. EL-Mahdy, T.-L. Yang and S.-W. Kuo, Microporous Mesoporous Mater., 2022, 331, 111669 CrossRef CAS.
- M. G. Kotp, A. F. M. EL-Mahdy and S.-W. Kuo, Polym. Chem., 2025, 16, 422–432 RSC.
- M. M. Ayad, W. A. Amer, M. G. Kotp, I. M. Minisy, A. F. Rehab, D. Kopecký and P. Fitl, RSC Adv., 2017, 7, 18553–18560 RSC.
- M. G. Kotp, C.-L. Chang and A. F. M. EL-Mahdy, J. Water Process Eng., 2023, 53, 103675 CrossRef.
- M. G. Kotp, S. U. Sharma, J.-T. Lee, A. F. M. EL-Mahdy and S.-W. Kuo, J. Taiwan Inst. Chem. Eng., 2022, 134, 104310 CrossRef CAS.
- M. Ejaz, M. G. Mohamed, M. G. Kotp, A. M. Elewa and S.-W. Kuo, Colloids Surf., A, 2025, 722, 137239 CrossRef CAS.
- M. G. Kotp, N. L. Torad, H. Nara, W. Chaikittisilp, J. You, Y. Yamauchi, A. F. M. EL-Mahdy and S.-W. Kuo, J. Mater. Chem. A, 2023, 11, 15022–15032 RSC.
- M. G. Kotp, M. G. Mohamed, P.-T. Wang, A. E. Hassan, A. M. Elewa and S.-W. Kuo, ACS Polym. Au, 2025, 5, 379–393 CrossRef CAS PubMed.
- H. Kumar, O. A. Britto, M. Thamizharasan, G. Arthanareeswaran and M. R. Viswanathan, Mater. Chem. Front., 2026, 10, 184–205 RSC.
- X. Zhang, H. Zhang, S. Gu, J. Tang and G. Yu, ACS Appl. Mater. Interfaces, 2025, 17, 42530–42540 CrossRef CAS PubMed.
- C. F. Martin, E. Stöckel, R. Clowes, D. J. Adams, A. I. Cooper, J. J. Pis, F. Rubiera and C. Pevida, J. Mater. Chem., 2011, 21, 5475–5483 RSC.
- D. R. Kumar, C. Rosu, A. R. Sujan, M. A. Sakwa-Novak, E. W. Ping and C. W. Jones, ACS Sustain. Chem. Eng., 2020, 8, 10971–10982 CAS.
- G. T. Rochelle, Carbon Capture Sci. Technol., 2024, 11, 100192 CAS.
- M. H. Nawaz, S. Santhoshkumar, T. H. Ho, B. Arumugam, M. G. Mohamed, T. V. Vu, Y. M. Nabil, M. G. Kotp, L.-W. Tu and H.-D. Yang, Chem. Eng. J., 2025, 527, 171521 CrossRef.
- A. Aslani, H. Masoumi and A. Ghaemi, CO2 Adsorbents: Advances in Materials, Technologies, and Applications for a Sustainable Future, 2025 Search PubMed.
- M. G. Mohamed, T.-C. Chen and S.-W. Kuo, Macromolecules, 2021, 12, 5866–5877 CrossRef.
- I. Ul Hassan, S. A. Hussen, R. Sathyamurthy, U. Zahid, U. Ahmed, V. M. Reddy and A. G. Abdul Jameel, Energy Fuels, 2025, 39, 13931–13968 CrossRef CAS.
- M. Shen, W. Guo, L. Tong, L. Wang, P. K. Chu, S. Kawi and Y. Ding, Chem. Soc. Rev., 2025, 54, 2762–2831 RSC.
- K. Atsonios, K. Panopoulos, A. Doukelis, A. Koumanakos and E. Kakaras, Energy, 2013, 53, 106–113 CrossRef CAS.
- F. Raganati and P. Ammendola, Energy Fuels, 2024, 38, 13858–13905 CrossRef CAS.
- S. Nandi, J. Rother, D. Chakraborty, R. Maity, U. Werner-Zwanziger and R. Vaidhyanathan, J. Mater. Chem. A, 2017, 5, 8431–8439 RSC.
- Y. Wang, F. Gao, Y. Niu, J. Zhang, K. Chen, Y. Zhou, X. Tang, S. Zhao and H. Yi, J. Mater. Chem. A, 2025, 13, 23323–23353 RSC.
- M. Vorokhta, M. I. M. Kusdhany, M. Švábová, M. Nishihara, K. Sasaki and S. M. Lyth, Sep. Purif. Technol., 2025, 354, 129054 CrossRef CAS.
- D. Chakraborty, R. Chatterjee, S. Mondal, S. K. Das, V. Amoli, M. Cho and A. Bhaumik, ACS Appl. Mater. Interfaces, 2023, 15, 48326–48335 CrossRef CAS PubMed.
- Y. Wang, J. Guo, L. Qu, P. Webley, H. Ding and G. K. Li, Chem Catal., 2025, 5, 101254 CAS.
- R. Norouzbeigi, CO2 Adsorbents: Advances in Materials, Technologies, and Applications for a Sustainable Future, 2025, p. 125 Search PubMed.
- T.-L. Lee, A. M. Elewa, M. G. Kotp, H.-H. Chou and A. F. M. El-Mahdy, Chem. Commun., 2021, 57, 11968–11971 RSC.
- M. G. Kotp, M. G. Mohamed, A. O. Mousa and S.-W. Kuo, Eur. Polym. J., 2025, 227, 113786 CrossRef CAS.
- Y. Du, T. Gao, G. T. Rochelle and A. S. Bhown, Int. J. Greenhouse Gas Control, 2021, 111, 103473 CrossRef CAS.
- F. Xia, Z. Yang, A. Adeosun, A. Gopan, B. M. Kumfer and R. L. Axelbaum, Fuel, 2016, 181, 1170–1178 CrossRef CAS.
- M. De Joannon, A. Chinnici, P. Sabia and R. Ragucci, Chem. Eng. J., 2012, 211, 318–326 CrossRef.
- P. Ma, W. Zhou, C. Yu, Z. Liu, Q. Chen, S.-P. Sun and X. Wang, Sep. Purif. Technol., 2025, 380, 135242 CrossRef.
- B. Ma, N. Qin, Q.-Q. Yan, S. Zhang, X. Wang, L. Bao and X. Lu, Digital Discovery, 2026, 5, 523–547 RSC.
- H. Xu, Y. Chen and Z. He, Sep. Purif. Technol., 2025, 382, 136054 CrossRef.
- S. Pal, E. P. Roberts, M. Trifkovic and G. Natale, Commun. Mater., 2025, 6, 130 CrossRef CAS PubMed.
- P. D. Raju, A. R. Sujatha, S. Krishnan and C. V. Suneesh, Nanoscale, 2025, 17, 22100–22121 RSC.
- Y. Zhou, S. Li, Y. Bai, B. Ji, X. Kong, B. Hu and J. Zhang, Energy Fuels, 2024, 39, 626–637 CrossRef.
- D. Wu, M. Yang, J. Yu, M. Dyballa, P. Yang, M. Li, G. Hou, M. Hunger and W. Dai, Chem. Soc. Rev., 2025, 54, 9192–9244 RSC.
- L. Jiang, F. Bao, Y. Peng, Z. Zhao, Y. Wang, Y. Song, J. Li, K. Lv, J. Zhang and K. Yuan, Microporous Mesoporous Mater., 2025, 381, 113338 CrossRef CAS.
- S. Raza, A. Abid, I. Areej, S. Nazeer, A. K. Qureshi and B. Tan, ACS Appl. Polym. Mater., 2024, 6, 6843–6851 CrossRef CAS.
- H. Zhang, H. Wang, T. Gao, S. Pan, C. Liu, C. Li and X. Tao, Carbon, 2025, 234, 120004 CrossRef CAS.
- M. Abdalla, M. Essalhi, M. H. Elsayed, A. Sabbah, M. G. Mohammed, I. H. Aljundi and M. M. Abdelnaby, ACS Appl. Polym. Mater., 2025, 7, 8731–8742 CrossRef CAS.
- A. Mollahosseini, M. N. Dafchahi, S. K. Salestan, J. W. Chew, M. Mozafari, M. Soroush, S. Hrapovic, U. D. Hemraz, R. Giro and M. B. Steiner, Energy Environ. Sci., 2025, 18, 5025–5092 RSC.
- M. G. Kotp and S.-W. Kuo, Polymers, 2024, 13, 1759 CrossRef PubMed.
- M. G. Kotp, A. F. M. El-Mahdy, M. M. Chou and S.-W. Kuo, New J. Chem., 2024, 48, 14435–14443 RSC.
- P. N. Singh, M. G. Mohamed, M. G. Kotp, T. Mondal, S. V. Chaganti, M. Ibrahim, S. U. Sharma, Y. Ye and S.-W. Kuo, ACS Appl. Polym. Mater., 2025, 7, 3324–3336 CrossRef CAS.
- J. R. Serrano, P. Piqueras, E. J. Sanchis and F. J. García, Energy Convers. Manage., 2025, 342, 120034 CrossRef CAS.
- H. Pal, A. Karmakar, A. Sadhukhan, K. Koner, S. Karak, R. K. Sharma, M. Ghosh, K. K. Dey, B. Pathak and S. Kundu, Adv. Funct. Mater., 2024, 34, 2408255 CrossRef CAS.
- J. Zhou, M. Deissenroth-Uhrig and M. Gallei, Adv. Funct. Mater., 2025, e20959 Search PubMed.
- Y. Song and S. Ma, Chem. Sci., 2025, 16, 11740–11767 RSC.
- M. K. Wong, J. J. Foo, J. Y. Loh and W. J. Ong, Adv. Energy Mater., 2024, 14, 2303281 CrossRef CAS.
- C. Y. Chuah, Y. L. Ho, A. M. H. Syed, K. G. K. Thivyalakshmi, E. Yang, K. Johari, Y. Yang and W. C. Poon, Ind. Eng. Chem. Res., 2025, 64, 4117–4147 CrossRef CAS.
- X. Wang, Y. Chen, A. Lindbråthen, Z. Waris and L. Deng, Chem. Eng. J., 2025, 512, 162377 CrossRef CAS.
- S. Zhao, Y. Zhang, L. Li, J. Feng, W. Qiu, Y. Ning, Z. Huang and H. Lin, Sep. Purif. Technol., 2025, 354, 129586 CrossRef CAS.
- M. G. Kotp and S.-W. Kuo, Mater. Today Chem., 2024, 41, 102299 CrossRef.
- M. Joshi, X. Ren, T. Lin and R. Joshi, Small, 2025, 21, 2406706 CrossRef CAS PubMed.
- L. Joos, K. Lejaeghere, J. M. Huck, V. Van Speybroeck and B. Smit, Energy Environ. Sci., 2015, 8, 2480–2491 RSC.
- G. Cui, J. Wang and S. Zhang, Chem. Soc. Rev., 2016, 45, 4307–4339 RSC.
- A. Chamoun-Farah, L. M. Cañada, J. F. Brennecke and B. D. Freeman, J. Membr. Sci., 2025, 727, 124081 CrossRef CAS.
- R. Krishna and J. M. van Baten, Sep. Purif. Technol., 2025, 362, 131757 CrossRef CAS.
- S. A. Khan, S. Ahmed, S. Ali and F. Altaf, Carbon Neutrality, 2025, 4, e70063 CrossRef CAS.
- V. Ramar, X. Zhang, H. Zhang, H. Tan and Y. Zhao, Small Methods, 2025, 9, e00717 CrossRef CAS PubMed.
- E. Pérez-Botella, S. Valencia and F. Rey, Chem. Rev., 2022, 122, 17647–17695 CrossRef PubMed.
- M. Zhang, J. Huang, F. Meng, C. Zhang and Z. Zhang, Dalton Trans., 2025, 54, 17383–17399 RSC.
- M. Ejaz, M. G. Mohamed and S.-W. Kuo, Polym. Chem., 2023, 14, 2494–2509 RSC.
- A. Kumari, K. Kaushik, A. Shankar, R. Aneja, A. Chauhan and V. K. Saini, Fuel, 2025, 399, 135651 CrossRef CAS.
- G. Li, H. Yu, D. Ji, C. Zhu, K. Thu and T. Miyazaki, Energy, 2025, 328, 136506 CrossRef CAS.
- M. G. Mohamed, Y.-C. Kao, B.-X. Su, H. Karim and S.-W. Kuo, Sep. Purif. Technol., 2025, 387, 136692 CrossRef.
- C. A. Trickett, A. Helal, B. A. Al-Maythalony, Z. H. Yamani, K. E. Cordova and O. M. Yaghi, Nat. Rev. Mater., 2017, 2, 17045 CrossRef CAS.
- H. Lyu, O. I.-F. Chen, N. Hanikel, M. I. Hossain, R. W. Flaig, X. Pei, A. Amin, M. D. Doherty, R. K. Impastato and T. G. Glover, J. Am. Chem. Soc., 2022, 144, 2387–2396 CrossRef CAS PubMed.
- R. W. Flaig, T. M. Osborn Popp, A. M. Fracaroli, E. A. Kapustin, M. J. Kalmutzki, R. M. Altamimi, F. Fathieh, J. A. Reimer and O. M. Yaghi, J. Am. Chem. Soc., 2017, 139, 12125–12128 CrossRef CAS PubMed.
- G. Lee and S. H. Jhung, Fuel, 2025, 394, 135143 CrossRef CAS.
- K. Klemenčič, A. Krajnc, A. Puškarić, M. Huš, D. Marinič, B. Likozar, N. Z. Logar and M. Mazaj, Angew. Chem., 2025, 137, e202424747 CrossRef.
- W. Song, Y. Wen, Y. Cho, X. Zhang, D. Kang, E. Shin, D. G. Yu, G. Li, Y. Liao and I. D. Kim, Adv. Mater., 2025, e13138 Search PubMed.
- K. Patra, S. Dey, C. Solanki, A. Sengupta and V. K. Mittal, ACS Appl. Eng. Mater., 2025, 3, 1130–1165 CrossRef CAS.
- J. He, D. Wen, Y. Wang, Y. Qian, J. Guo, Z. Yun and W. Yang, Appl. Catal., A, 2025, 708, 120604 CrossRef CAS.
- Y. Wu, Y. Wu, Y. Sun, W. Zhao and L. Wang, Adv. Mater., 2024, 36, 2312460 CrossRef CAS PubMed.
- A. K. Sekizkardes, P. Wang, J. Hoffman, S. Budhathoki and D. Hopkinson, Mater. Adv., 2022, 3, 6668–6686 RSC.
- M. G. Kotp, M. G. Mohamed and S.-W. Kuo, Chem. Sci., 2025, 16, 20718–20754 RSC.
- M. G. Kotp and S.-W. Kuo, Electrochim. Acta, 2025, 531, 146440 CrossRef CAS.
- M. G. Kotp, I. M. Minisy, B. Al-Saida and S.-W. Kuo, Carbohydr. Polym., 2025, 356, 123399 CrossRef CAS PubMed.
- M. G. Kotp, N. L. Torad, J. Lüder, A. El-Amir, W. Chaikittisilp, Y. Yamauchi and A. F. M. EL-Mahdy, J. Mater. Chem. A, 2023, 11, 764–774 RSC.
- M. G. Mohamed, C.-C. Chen and S.-W. Kuo, React. Funct. Polym., 2025, 214, 106286 CrossRef CAS.
- M. G. Mohamed, A. F. M. EL-Mahdy, M. G. Kotp and S.-W. Kuo, Mater. Adv., 2022, 3, 707–733 RSC.
- A. F. M. El-Mahdy, C.-H. Kuo, A. Alshehri, C. Young, Y. Yamauchi, J. Kim and S.-W. Kuo, J. Mater. Chem. A, 2018, 6, 19532–19541 RSC.
- A. F. M. El-Mahdy, C. Young, J. Kim, J. You, Y. Yamauchi and S.-W. Kuo, ACS Appl. Mater. Interfaces, 2019, 11, 9343–9354 CrossRef CAS PubMed.
- M. G. Mohamed, E. C. Atayde Jr, B. M. Matsagar, J. Na, Y. Yamauchi, K. C.-W. Wu and S.-W. Kuo, J. Taiwan Inst. Chem. Eng., 2020, 112, 180–192 CrossRef CAS.
- A. F. EL-Mahdy, Y.-H. Hung, T. H. Mansoure, H.-H. Yu, Y.-S. Hsu, K. C. Wu and S.-W. Kuo, J. Taiwan Inst. Chem. Eng., 2019, 103, 199–208 CrossRef CAS.
- M. E. Tohidi and A. Amiri, Adv. Mater., 2025, 38, e11083 CrossRef PubMed.
- C. Qian and X. Zhao, Acc. Chem. Res., 2025, 58, 1192–1209 CrossRef CAS PubMed.
- L. Yang, H. Yang, H. Wu, L. Zhang, H. Ma, Y. Liu, Y. Wu, Y. Ren, X. Wu and Z. Jiang, J. Mater. Chem. A, 2021, 9, 12636–12643 RSC.
- Y.-C. Kao, K.-T. Yeh, M. G. Mohamed, H. Karim, W.-H. Su and S.-W. Kuo, Sep. Purif. Technol., 2025, 375, 133827 CrossRef CAS.
- F. Zhao, F. Xu, H. García and J. Yu, J. Colloid Interface Sci., 2025, 700, 138532 CrossRef CAS PubMed.
- H. Daglar, Z. Zhou, R. Zhu, P. Parihar, J. I. Siepmann, O. M. Yaghi and L. Gagliardi, J. Am. Chem. Soc., 2025, 1, 1614–1622 Search PubMed.
- Z. Zhou, T. Ma, H. Zhang, S. Chheda, H. Li, K. Wang, S. Ehrling, R. Giovine, C. Li, A. H. Alawadhi, M. M. Abduljawad, M. O. Alawad, L. Gagliardi, J. Sauer and O. M. Yaghi, Nature, 2024, 635, 96–101 CrossRef CAS PubMed.
- M. G. Kotp, A. M. Elewa, A. F. M. EL-Mahdy, H.-H. Chou and S.-W. Kuo, ACS Appl. Energy Mater., 2021, 4, 13140–13151 CrossRef CAS.
- Y. W. Ahmed, A. Loukanov and H. C. Tsai, Adv. Healthcare Mater., 2025, 14, 2403743 CrossRef CAS PubMed.
- M. G. Mohamed, C.-C. Chen, M. Ibrahim, A. Osama Mousa, M. H. Elsayed, Y. Ye and S.-W. Kuo, JACS Au, 2024, 4, 3593–3605 CrossRef CAS PubMed.
- Y. Jia, Y. Lu, H. Yang, Y. Chen, F. Hillman, K. Wang, C. Z. Liang and S. Zhang, Adv. Funct. Mater., 2024, 34, 2407499 CrossRef CAS.
- B. Chai, S. Wang, Z. Li, Y. Jiang, X. Liu, M. Cui, X. Yu, Y. Xu, Y. Lei and L. Zhao, Fuel, 2024, 369, 131776 CrossRef CAS.
- Y. Ren, S. Yang and Y. Xu, Acc. Chem. Res., 2025, 58, 474–487 CrossRef CAS PubMed.
- C. Lin, P. Feng, P. Geng, S. Zhang, Y. Chen, Y. Shen and Y. Zheng, Chem. Commun., 2025, 61, 16354–16371 RSC.
- Y. Chen, X. Hu, J. Guo, Z. Guo, H. Zhan and S. Du, Eur. Polym. J., 2022, 171, 111215 CrossRef CAS.
- T. Guo, J. Chen, Y. Zhang and A. H. Bedane, Sep. Purif. Technol., 2025, 359, 130794 CrossRef CAS.
- R. Kishan, P. Rani, S. Kumar and C. Nagaraja, Energy Fuels, 2025, 39, 18586–18596 CrossRef CAS.
- A. K. Maharana, S. K. Sarkar, S. Mukherjee, R. Sarkar, G. Rambabu, K. Sugamata and S. Das, J. Mater. Chem. A, 2025, 13, 11717–11731 RSC.
- Q. Huang, Z. Zhan, R. Sun, J. Mu, B. Tan and C. Wu, Angew. Chem., 2023, 135, e202305500 CrossRef.
- W. Song, Y. Wen, Z. Wang, H. Xu, Q. Liao, Y. Tang, D.-G. Yu and I. Kim, Langmuir, 2024, 40, 16670–16689 CrossRef CAS PubMed.
- M.-C. Lin, S.-W. Kuo and M. G. Mohamed, Mater. Adv., 2024, 5, 6222–6233 RSC.
- Y. Zhao, Y. Hu, Y. Ma, L. Ding, S. Zhang, Y. Fu and X. Wang, Appl. Surf. Sci., 2025, 685, 161963 CrossRef CAS.
- M. Ejaz, M. G. Mohamed, Y.-T. Chen, K. Zhang and S.-W. Kuo, J. Energy Storage, 2024, 78, 110166 CrossRef CAS.
- Y. Gu, S. U. Son, T. Li and B. Tan, Adv. Funct. Mater., 2021, 31, 2008265 CrossRef CAS.
- S. Ullah, J. Hu, S. Manzoor and B. Tan, J. Mater. Chem. A, 2025, 13, 35082–35112 RSC.
- S. Krishnan and C. V. Suneesh, Mater. Today Commun., 2021, 27, 102251 CrossRef CAS.
- H. Ouyang, K. Song, J. Du, Z. Zhan and B. Tan, Chem. Eng. J., 2022, 431, 134326 CrossRef CAS.
- Y. Alemin, J. Hu, P. Xie, X. Wang, H. Gao and B. Tan, Macromol. Rapid Commun., 2025, 46, 2500020 CrossRef CAS PubMed.
- P. Liu, F. Zhao, Z. Xu, Q. Liao, C. Yang, X. Hu, P. Chen and T. Zhao, ACS Sustain. Chem. Eng., 2025, 13, 9903–9912 CrossRef CAS.
- Y. Tian, F. Cui, Z. Bian, X. Tao, H. Wang, N. Zhang and G. Zhu, Acc. Chem. Res., 2024, 57, 2130–2143 CrossRef CAS PubMed.
- M. Li, H. Ren, F. Sun, Y. Tian, Y. Zhu, J. Li, X. Mu, J. Xu, F. Deng and G. Zhu, Adv. Mater., 2018, 30, 1804169 CrossRef PubMed.
- H. Ali, Y. Orooji, A. Y. A. Alzahrani, H. M. Hassan, Z. Ajmal, D. Yue and A. Hayat, ACS Nano, 2025, 19, 7482–7545 CrossRef CAS PubMed.
- T. Ben, Y. Li, L. Zhu, D. Zhang, D. Cao, Z. Xiang, X. Yao and S. Qiu, Energy Environ. Sci., 2012, 5, 8370–8376 RSC.
- T. Ben, C. Pei, D. Zhang, J. Xu, F. Deng, X. Jing and S. Qiu, Energy Environ. Sci., 2011, 4, 3991–3999 RSC.
- S. Zhang, J. Li, J. Liu, S. Jiang, X. Chen, H. Ren, T. X. Liu, X. Zou and G. Zhu, J. Membr. Sci., 2021, 632, 119372 CrossRef CAS.
- N. B. McKeown, Curr. Opin. Chem. Eng., 2022, 36, 100785 CrossRef.
- T. H. Lee, P. A. Dean, J. Y. Yeo and Z. P. Smith, Adv. Mater., 2025, e13892 Search PubMed.
- K. M. Rodriguez, F. M. Benedetti, N. Roy, A. X. Wu and Z. P. Smith, J. Mater. Chem. A, 2021, 9, 23631–23642 RSC.
- Z. Zhong, X. Wang and B. Tan, Chem.–Eur. J., 2025, 31, e202404089 CrossRef CAS PubMed.
- S. Fajal, S. Dutta and S. K. Ghosh, Mater. Horiz., 2023, 10, 4083–4138 RSC.
- D. Luo, M. Li, Q. Ma, G. Wen, H. Dou, B. Ren, Y. Liu, X. Wang, L. Shui and Z. Chen, Chem. Soc. Rev., 2022, 51, 2917–2938 RSC.
- V. Guillerm and M. Eddaoudi, Acc. Chem. Res., 2021, 54, 3298–3312 CrossRef CAS PubMed.
- Y. Zhi, J. Shao, C. Liu, Q. Xiao, M. Demir, M. K. Al Mesfer, M. Danish, L. Wang and X. Hu, Sep. Purif. Technol., 2025, 361, 131253 CrossRef CAS.
- Q. Liao, Y. Yuan and J. Cao, J. Colloid Interface Sci., 2024, 665, 958–968 CrossRef CAS PubMed.
- M. He, H. Zhao, J. Jia, W. Zhou, Z. Wang, K. An, Y. Jiao, X. Yang, X. Zhang and T. Fan, Environ. Res., 2025, 267, 120701 CrossRef CAS PubMed.
- X. Yang, N. Zhou, X. Xie, Z. Dai, B. T. Goh, Z. Chen and Y. Xiong, Sep. Purif. Technol., 2025, 361, 131257 CrossRef CAS.
- Y. Jeong, Y. Kim, W. Jeong, H. L. Choi, J. Kim and T.-H. Bae, J. Membr. Sci., 2025, 738, 124749 CrossRef.
- B. Ray, S. R. Churipard and S. C. Peter, J. Mater. Chem. A, 2021, 9, 26498–26527 RSC.
- L. Yang, Y. Sheng and D. Liu, Dalton Trans., 2026, 55, 1515–1547 RSC.
- C. Zhang, S. Sun, S. Xu and C. Wu, Biomass Bioenergy, 2022, 166, 106608 CrossRef CAS.
- S. Wang, X. Qiu, Y. Chen and S. Chen, Microporous Mesoporous Mater., 2022, 330, 111585 CrossRef CAS.
- A. Kuang, M. Kuang, H. Yuan, G. Wang, H. Chen and X. Yang, Appl. Surf. Sci., 2017, 410, 505–512 CrossRef CAS.
- N. MacDowell, N. Florin, A. Buchard, J. Hallett, A. Galindo, G. Jackson, C. S. Adjiman, C. K. Williams, N. Shah and P. Fennell, Energy Environ. Sci., 2010, 3, 1645–1669 RSC.
- P. Bollini, S. Choi, J. H. Drese and C. W. Jones, Energy Fuel., 2011, 25, 2416–2425 CrossRef CAS.
- J. S. Carneiro, G. Innocenti, H. J. Moon, Y. Guta, L. Proaño, C. Sievers, M. A. Sakwa-Novak, E. W. Ping and C. W. Jones, Angew. Chem., Int. Ed., 2023, 62, e202302887 CrossRef CAS PubMed.
- S. S. Hamid, L. Kerber and A. B. Clarke, Nat. Commun., 2025, 16, 8923 CrossRef CAS PubMed.
- M. Debruyne, V. Van Speybroeck, P. Van Der Voort and C. V. Stevens, Green Chem., 2021, 23, 7361–7434 RSC.
- W. Zhang, G. Li, H. Yin, K. Zhao, H. Zhao and T. An, Environ. Sci.: Nano, 2022, 9, 81–104 RSC.
- Y. Ran, K. Peng, Z. Li, X. Guo, H. Chen, S. Cai, L. Su, M. Niu, D. Lu and H. Wang, Chem. Eng. J., 2025, 514, 163149 CrossRef CAS.
- M. A. Elsayed, S. Zhou, X. Zhao, G. W. Manggada, Z. Chen, F. Wang and Z. Tang, Nanomaterials, 2025, 15, 1077 CrossRef CAS PubMed.
- S. Podder, H. Jungi and J. Mitra, Chem.–Eur. J., 2025, 31, e202500865 CrossRef CAS PubMed.
- Y.-C. Su, G. Chen, Y.-J. Lai, G.-Z. Song, T.-L. Wu and Y.-C. Yeh, Chem. Soc. Rev., 2026, 55, 819–868 RSC.
- S. A. Bedell, Energy Procedia, 2009, 1, 771–778 CrossRef CAS.
- A. P. Hallenbeck and J. R. Kitchin, Ind. Eng. Chem. Res., 2013, 52, 10788–10794 CrossRef CAS.
- S. I. Matsumoto, Y. Ikeda, H. Suzuki, M. Ogai and N. Miyoshi, Appl. Catal., B, 2000, 25, 115–124 CrossRef CAS.
- M. Gómez-García, V. Pitchon and A. Kiennemann, Environ. Int., 2005, 31, 445–467 CrossRef PubMed.
- Z.-X. Low, P. M. Budd, N. B. McKeown and D. A. Patterson, Chem. Rev., 2018, 118, 5871–5911 CrossRef CAS PubMed.
- D. Cangialosi, V. M. Boucher, A. Alegría and J. Colmenero, Soft Matter, 2013, 9, 8619–8630 RSC.
- M. Meier, E. John, D. Wieckhusen, W. Wirth and W. Peukert, Powder Technol., 2009, 188, 301–313 CrossRef CAS.
- X. Lin, S. Kim, M. Abdallah and R. J. Farrauto, Appl. Catal., A, 2025, 709, 120659 CrossRef.
- R. Bonné, Industrial Production Of Porous Materials, in Handbook of Porous Materials, World Scientific, 2020 Search PubMed.
- Y. Zhi, J. Shao, J. Wang, X. Liu, Q. Xiao, M. Demir, U. B. Simsek, L. Wang and X. Hu, Molecules, 2025, 30, 3990 CrossRef CAS PubMed.
- J. Shao, Y. Wang, M. Che, Q. Xiao, M. Demir, M. K. Al Mesfer, L. Wang, X. Hu and Y. Liu, J. Energy Inst., 2025, 102273 CrossRef CAS.
- J. Wang, Y. Wang, X. Liu, Q. Xiao, M. Demir, M. K. Almesfer, S. G. Colak, L. Wang, X. Hu and Y. Liu, Molecules, 2025, 30, 2564 CrossRef CAS PubMed.
| This journal is © The Royal Society of Chemistry 2026 |