Open Access Article
Open Access ArticleDeoxygenative phosphonation of ketones by titanium
Yuquan
Wang
a,
Kai
Yin
b,
Xiaobo
Pang
a and
Xing-Zhong
Shu
 *a
*a
aState Key Laboratory of Natural Product Chemistry (SKLAOC), College of Chemistry and Chemical Engineering, Lanzhou University, 222 South Tianshui Road, Lanzhou, 730000, China. E-mail: shuxingzh@lzu.edu.cn
bZhejiang Nanjiao Chemistry Co., Ltd., Shaoxing 312369, China. E-mail: ykgoals@163.com
First published on 9th February 2026
Abstract
Tertiary phosphine oxides are ubiquitous motifs with essential roles across synthetic chemistry and life sciences, yet C(sp3)–P coupling remains underdeveloped. Herein, we report an accessible di-oxo-titanium trichloride complex, operating in combination with Mn powder, that enables a deoxygenative C(sp3)–P coupling of ketones with secondary phosphine oxides. The transformation proceeds under mild conditions and shows good compatibility with carbonyl substrates, including cyclic and acyclic ketones, as well as aliphatic aldehydes. Functionalities such as carboxyl, acetal, alkyne, sulfonate, phenol, and alcohol are well tolerated.
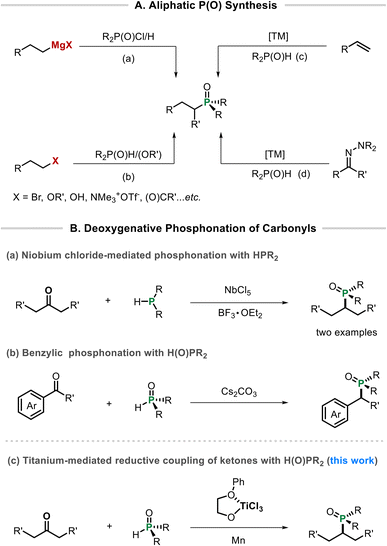
Tertiary phosphine oxides display diverse biological activities and serve as versatile ligands or ligand precursors.1 While C(sp2)–P bond formation is well established,2 strategies for constructing C(sp3)–P bonds remain underdeveloped. Traditional approaches typically involve substitution reactions of alkyl halides or alkyl metal reagents (Scheme 1A, a and b), such as Michaelis–Arbuzov rearrangements3 or H-phosphinate additions.4 Other methods include cross-coupling reactions,5 hydrophosphination of olefins (Scheme 1A, c),6 and carbene insertion into P(O)–H bonds (Scheme 1A, d).7 Despite these advances, further extension of phosphonation strategies to more readily available functional groups would enhance the practical utility of this chemistry.
Ketones are among the most widely available functionalities in organic synthesis, rendering them attractive precursors for accessing aliphatic tertiary phosphine oxides via direct phosphonation. In this context, the pioneering work by Suzuki and co-workers disclosed a niobium chloride–mediated deoxygenative phosphonation of aldehydes with P(III) reagents, in which only two ketone examples were demonstrated (Scheme 1B, a).8 Very recently, the use of bench-stable H(O)PR2 for phosphonation of benzylic ketones has been realized (Scheme 1B, b).9 Related progress has also been made in the deoxygenative phosphonation of esters and acids to install H(O)PR2 at benzylic positions.10 However, despite these advances, a broadly applicable and mild deoxygenative phosphonation of structurally diverse ketones remains underdeveloped.
Radical-based strategies offer new opportunities to expand the scope of coupling partners. For example, MacMillan and co-workers recently reported two isolated cases of radical phosphonation of NHC-activated primary alcohols with HP(O)Ar2 to afford alkyl–P(O)Ar2 products.11 However, progress in this area remains limited, in sharp contrast to the numerous reports on radical alkylation of P(III) reagents such as ClPPh2, Me3SnPPh2, P(OR)3, and P(SPh)3.12 Moreover, methods enabling the installation of phosphine oxides at stable and readily available functional positions would substantially broaden their practical utility.
Low-valent titanium complexes play a pivotal role in enabling radical transformations of C–O compounds,13,14 especially for carbonyl compounds.15 Since the discovery of the McMurry coupling,16 a wide range of reductive carbonyl transformations have been realized, including pinacol couplings via carbonyl–carbonyl dimerization,17 umpolung-type carbonyl–cyano couplings to form α-hydroxy ketones,18 as well as reductive cross-couplings and [3 + 2] cycloadditions with halides,19N,N′-dimethylformamide,20 and activated alkenes.21 These advances underscore the exceptional capacity of low-valent titanium species to mediate radical pathways and construct diverse C–C frameworks under mild conditions. However, their application in C–X bond-forming reactions remains largely unexplored.22
Motivated by these encouraging results and building upon our recent progress in the deoxygenation functionalization of C–O compounds,23 we introduce a titanium-mediated deoxygenative phosphonation reaction of ketones for constructing C(sp3)–P bonds (Scheme 1b). The method facilitates an intermolecular cross-coupling between ketones and diaryl phosphine oxides through a radical pathway, proceeds under mild conditions and shows good functional group compatibility. This transformation will provide a simplified and efficient alternative to the existing methods24 and expand the scope of radical transformations of carbonyl compounds by titanium.
We initiated our investigation by evaluating the reaction between ketone 1a and diphenylphosphine oxide 2a. Optimal efficiency was achieved using titanium complex 3m (2.2 equiv.), Mn powder (4.0 equiv.), and DMPU (0.2 M), affording the desired product in 80% isolated yields (Table 1, entry 1). Replacing DMPU with DMA led to a modest decrease in yield (entry 3), whereas THF, despite its widespread use in related titanocene systems, resulted in strong inhibition of product formation (entry 2). Lowering the reaction temperature to 60 °C (entry 4) or replacing Mn with Zn (entry 5), a reductant of weaker reducing strength, both resulted in substantial reductions in efficiency. Control experiments verified that both the Ti complex and Mn reductant are essential (entry 6), as omission of either component completely suppressed formation of the target product.
| Entry | Change from standard conditions | 4a (%) |
|---|---|---|
| a Reaction conditions: 1a (0.1 mmol) and 2a (0.15 mmol) were used; the yields were determined by 1H NMR analysis using CH2Br2 as an internal standard. b Isolated yield obtained from 1a (0.2 mmol) and 2a (0.3 mmol) is given. c Bench-top reaction. | ||
| 1 | None | 81 (80)b |
| 2 | THF | 18% |
| 3 | DMA | 69% |
| 4 | 60 °C | 71 |
| 5 | Zn instead of Mn | 11 |
| 6 | Without Ti or Mn | 0 |
|
||
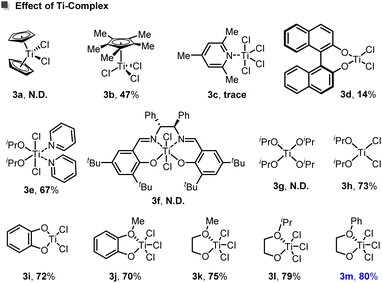
The ligand sphere around titanium exerts a decisive influence on the reaction outcome. Cp2TiCl2 (3a) predominantly promoted P-centered homocoupling of 2a and fails to deliver the desired product. In contrast, Cp*TiCl3 (3b) enabled formation of product 4a in moderate yield. Other classical titanium complexes, including TiCl4(collidine) (3c) and the alkoxide complex (iPrO)4Ti (3e), are ineffective, and unreacted substrates were recovered. Incorporation of less sterically encumbered di-oxo donors, as in complexes derived from isopropanol (3h) and catechol (3i), markedly improved coupling efficiency. Conversely, more rigid and sterically congested ligand frameworks, such as BINOL- or Salen-supported Ti complexes (3d and 3g), significantly reduce reactivity. Among all evaluated Ti precatalysts, the di-oxo titanium trichloride series incorporating simple 1,2-diol backbones (3j–3m) performed most effectively, especially the ethylene glycol-derived complexes (3k, 3l, and 3m). Within this group, the arylated complex 3m was identified as the optimal precatalyst for enabling the coupling.
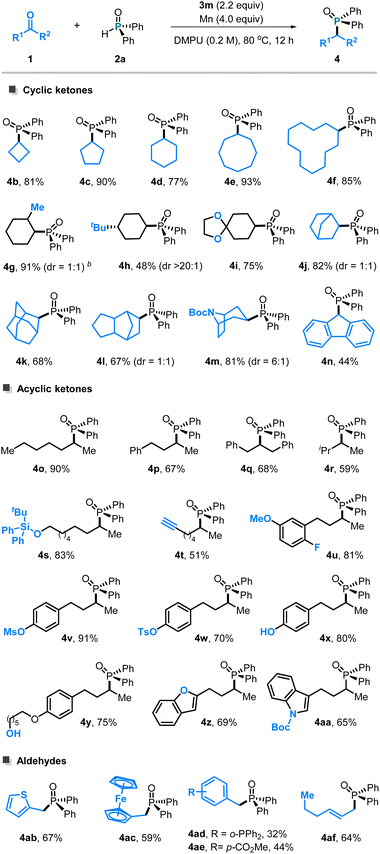
With the optimized conditions in hand, we evaluated the substrate scope of ketones with diphenylphosphine oxide 1b (Scheme 2). The reaction proved effective for cyclic ketones, accommodating ring sizes from 4 to 12 members (4b–4f) and delivering the corresponding phosphonated products in good yields. Structurally rigid polycyclic ketones also participated smoothly (4j–4f), demonstrating the capacity of this Ti platform to activate conformationally and sterically constrained carbonyl frameworks. Cyclic ketones bearing substituents at different ring positions (4g–4i), including those with steric crowding adjacent to the carbonyl site (4g), were equally compatible, indicating the steric tolerance of this titanium complex (Table 2).
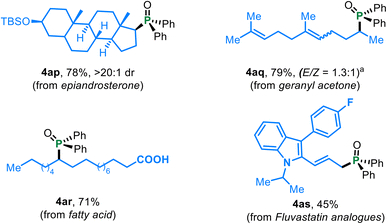 | ||
Scheme 2 Deoxygenative phosphonation of ketone-containing bioactive molecules. bKetone substrate 1ag (E/Z = 1.5![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) was used. 1) was used. | ||
The method is also effective for acyclic aliphatic ketones featuring varied chain lengths (4a, 4o–4p) or increased steric demand around the carbonyl unit (4q and 4r), furnishing products in 69–90% yields. Functionalities including silyl ethers (4s), alkynes (4r), ethers, fluorine (4u), and sulfonates (4v and 4w) are tolerated. Notably, ketones containing hydroxy-derived motifs (4v–4y), especially for the phenols (4x) and aliphatic alcohol-based frameworks (4y), which are typically ligating or deactivating in classical low-valent Ti systems, remained intact under the present conditions. This highlights the good stability and robustness of the titanium complex used.
This titanium system also enabled efficient deoxygenative phosphonation of aldehydes, demonstrating good tolerance toward diverse functional motifs. Aldehydes containing thiophene, ferrocenecarbonyl (4ac), phosphines (4ad), esters (4ae),10 and acrylaldehyde units (4af) all underwent smooth C–O cleavage and phosphonylation without compromised selectivity, further underscoring the versatility and practical utility of the method in complex settings.
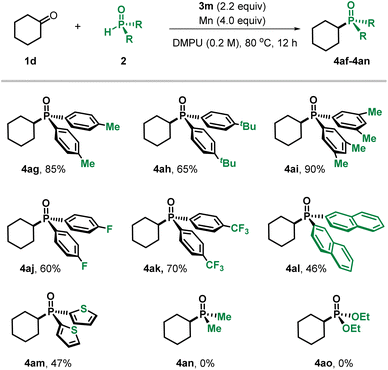
The substrate scope of phosphine oxides was then evaluated using cyclohexanone as the coupling partner (Table 3). Diverse diarylphosphine oxides participated in the transformation, including those decorated with electron-donating substituents (4ag–4ai) as well as electron-withdrawing groups (4aj–4ak), all undergoing smooth C–P bond formation. Phosphine oxides bearing conjugated naphthyl (4al) and heteroaryl (4am) variants were tolerated in the current titanium system. In contrast, phosphine oxides in which the aryl groups were replaced by alkyl (4an) and alkoxy (4ao) moieties did not furnish the desired products.
| a Cyclohexanone 1 (0.2 mmol) and phosphine oxides 2 (0.3 mmol) were used; isolated yields are given. |
|---|
|
The methodology further demonstrated competence in the late-stage functionalization of bioactive molecules. Ketone-containing compounds, including epiandrosterone (4ap), geranyl acetone (4aq), unprotected fatty acid (4ar), and fluvastatin analogues (4as), underwent deoxygenative phosphonation to furnish the corresponding C–P coupled products in synthetically useful yields.
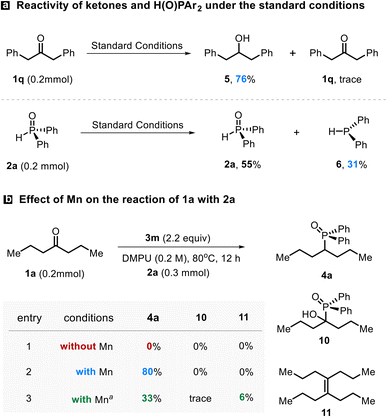
To gain mechanistic insight into the transformation, a series of control experiments were executed. Independent exposure of ketone 1q and phosphine oxide 2a to the standard manifold led to efficient reduction of 1q, affording the corresponding alcohol 5 in 76% yield,25 whereas phosphine oxide 2a exhibits substantially weaker interaction with the titanium species, with more than half recovered (Scheme 3a). These observations indicate that ketones possess higher reactivity over phosphine oxides toward the active Ti species.
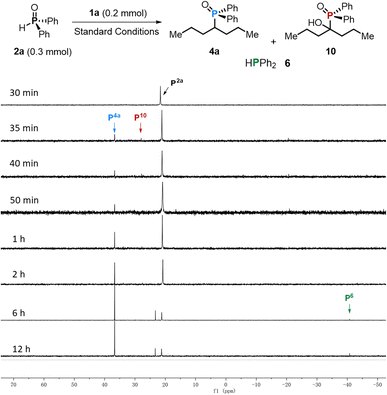
The role of Mn in Ti initiation was then examined. In the absence of Mn, no productive C–P coupling occurred, and both 1a and 2a were almost fully restored, consistent with a Mn-enabled Ti(IV) to low-valent Ti reduction even being required to access the catalytically competent state (Scheme 3b, entries 1 vs. 2). The filtrate obtained from preactivation of 3m with Mn dust (see the SI for details) promoted the coupling of 1a and 2a to afford 4a in 33% yield (Scheme 3b, entry 3), with 52% recovery of ketone 1a and minor formation of McMurry byproduct 11. These results indicate that in situ generated low-valent titanium species are required to initiate the transformation. 31P NMR analysis revealed a signal corresponding to α-hydroxyphosphonates (10) at 27.9 ppm,26 suggesting that it may be an intermediate in the transformation.
The real-time 31P NMR monitoring further validated this proposal. As shown in Fig. 1, product 4a appeared within 35 minutes, accompanied by a transient and low-intensity resonance for 10 at 27.9 ppm. The reaction reached completion within 2 hours without detection of other P-containing adducts derived from 2a. In contrast, the deoxygenated phosphine HPPh2 (6, −40.8 ppm) only emerged after extended reaction times (6 h) and accumulated gradually, indicating that 6 forms downstream and outside the catalytic C–P bond-forming process.
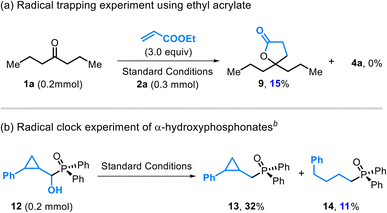
The intermediacy of radicals was supported by orthogonal experiments. Addition of Michael acceptors delivered a radical-trapped-esterified product 9 in 15% yield (Scheme 4a),27 indicating accessible carbon-centered radicals. A radical clock experiment employing cyclopropyl-containing α-hydroxyphosphonates 12 afforded the ring-opened product 14 (Scheme 4b), revealing radical-mediated C–O scission during deoxygenation. The initially formed alkenyl fragment from cyclopropane opening underwent subsequent reduction to 14 under the current Ti/Mn system.
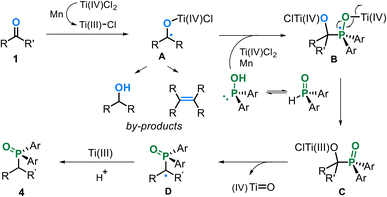
Based on the above results and literature reports,28 a plausible reaction mechanism is proposed (Scheme 5). The Lewis acidity of the Ti complex enables P(V) to P(III) isomerization,29 while carbonyl deoxygenation under the Ti(IV)/Mn manifold generates alkyl radical A, which is captured by P(III) species to form intermediate B.11,30 Subsequent β-scission restores the P(V) species and Ti(IV) reduction to afford low-valent complex C, followed by radical dehydroxylation and protonation to release the deoxygenatively phosphonated product.
Conclusions
In conclusion, we have developed a titanium-mediated deoxygenative C(sp3)–P coupling of ketones and aldehydes with secondary phosphine oxides, providing a practical route to diverse aliphatic organophosphorus compounds. The reaction proceeds via a distinct radical pathway, in which the combined Lewis acidity and single-electron reductive capacity of the titanium complex are essential for C–O bond radical activation and productive phosphonylation. The protocol operates under mild conditions and tolerates a wide range of functional groups, including carboxyl, phenolic OH, and alcohol groups that typically challenge classical polar phosphonation manifolds. Further deoxygenative functionalization of aldehydes/ketones is ongoing in our laboratory.Author contributions
X.-Z. S. conceived and supervised the project. X.-Z. S., X. P. and Y. W. designed the experiments. Y. W. performed the experiments and analysed the data. Y. K. assisted with data analysis and reaction design.Conflicts of interest
The authors declare no competing financial interests.Data availability
CCDC 2467845 contains the supplementary crystallographic data for this paper.31All experimental data, procedures for data analysis, and pertinent data sets are provided in the supplementary information (SI). Supplementary information is available. See DOI: https://doi.org/10.1039/d5sc09520d.
Acknowledgements
X.-Z. S. and X. P. thank the National Natural Science Foundation of China (22471104, 22271127, and 22401122), Science and Technology Program Projects of Gansu Province (25RCKA023), Science and Technology Major Program of Gansu Province of China (22ZD6FA006 and 23ZDFA015), and High-Level Talent Program of Lanzhou Municipality for their financial support.Notes and references
- (a) J. Sikorski and E. Logusch, in Aliphatic Carbon–Phosphorus Compounds as Herbicides, Marcel Dekker, 1992, pp. 737–806 Search PubMed; (b) M. Eto, in Handbook of Organophosphorus Chemistry, ed. R. Engel, Marcel Dekker, 1992, pp. 807–873 Search PubMed; (c) T. M. Shaikh, C.-M. Weng and F.-E. Hong, Coord. Chem. Rev., 2012, 256, 771–803 CrossRef CAS; (d) U. Pradere, E. C. Garnier-Amblard, S. J. Coats, F. Amblard and R. F. Schinazi, Chem. Rev., 2014, 114, 9154–9218 CrossRef CAS PubMed; (e) A. Gallen, A. Riera, X. Verdaguer and A. Grabulosa, Catal. Sci. Technol., 2019, 9, 5504–5521 RSC; (f) M. You, Z. Zhang, C. Chen, Z. Mei, X. Zhang, J. Bai, H. Huang and Q. Xiao, Green Chem., 2025, 27, 3743–3750 RSC; (g) C. Chen, Z. Mei, M. You, Z. Zhang, H. Huang and Q. Xiao, Org. Chem. Front., 2025, 12, 3681–3686 RSC.
- (a) T. Hirao, T. Masunaga, Y. Ohshiro and T. Agawa, Tetrahedron Lett., 1980, 21, 3595–3598 CrossRef CAS; (b) D. Gelman, L. Jiang and S. L. Buchwald, Org. Lett., 2003, 5, 2315–2318 CrossRef CAS PubMed; (c) L.-B. Han, C. Zhang, H. Yazawa and S. Shimada, J. Am. Chem. Soc., 2004, 126, 5080–5081 CrossRef CAS PubMed; (d) A. L. Schwan, Chem. Soc. Rev., 2004, 33, 218–224 Search PubMed; (e) Y. Gao, G. Wang, L. Chen, P. Xu, Y. Zhao, Y. Zhou and L.-B. Han, J. Am. Chem. Soc., 2009, 131, 7956–7957 CrossRef CAS PubMed; (f) G. Evano, K. Tadiparthi and F. Couty, Chem. Commun., 2011, 47, 179–181 RSC; (g) A. J. Bloomfield and S. B. Herzon, Org. Lett., 2012, 14, 4370–4373 CrossRef CAS PubMed; (h) C.-G. Feng, M. Ye, K.-J. Xiao, S. Li and J.-Q. Yu, J. Am. Chem. Soc., 2013, 135, 9322–9325 Search PubMed; (i) Y.-M. Li, M. Sun, H.-L. Wang, Q.-P. Tian and S.-D. Yang, Angew. Chem., Int. Ed., 2013, 52, 3972–3976 Search PubMed; (j) A.-X. Zhou, L.-L. Mao, G.-W. Wang and S.-D. Yang, Chem. Commun., 2014, 50, 8529–8532 RSC; (k) W. C. Fu, C. M. So and F. Y. Kwong, Org. Lett., 2015, 17, 5906–5909 CrossRef CAS PubMed; (l) J. Yang, J. Xiao, T. Chen, S.-F. Yin and L.-B. Han, Chem. Commun., 2016, 52, 12233–12236 Search PubMed; (m) J. Yang, T. Chen and L.-B. Han, J. Am. Chem. Soc., 2015, 137, 1782–1785 Search PubMed; (n) J. Yang, J. Xiao, T. Chen and L.-B. Han, J. Org. Chem., 2016, 81, 3911–3916 CrossRef CAS PubMed; (o) Y.-N. Ma and S.-D. Yang, Chem. Rec., 2016, 16, 977–986 CrossRef CAS PubMed; (p) L. Rout and T. Punniyamurthy, Coord. Chem. Rev., 2021, 431, 213675 CrossRef CAS; (q) X. Zhang, S.-Q. Wang and S.-D. Yang, Sci. China: Chem., 2023, 66, 2569–2575 CrossRef CAS; (r) M. Huang, H. Huang, M. You, X. Zhang, L. Sun, C. Chen, Z. Mei, R. Yang and Q. Xiao, Green Chem., 2024, 26, 295–299 RSC.
- (a) K. D. Berlin and G. B. Butler, Chem. Rev., 1960, 60, 243–260 CrossRef CAS; (b) A. K. Bhattacharya, Chem. Rev., 1981, 81, 415–430 Search PubMed; (c) P.-Y. Renard, P. Vayron, E. Leclerc, A. Valleix and C. Mioskowski, Angew. Chem., Int. Ed., 2003, 42, 2389–2392 CrossRef CAS PubMed; (d) P.-Y. Renard, P. Vayron and C. Mioskowski, Org. Lett., 2003, 5, 1661–1664 Search PubMed; (e) L. Longwitz and T. Werner, Angew. Chem., Int. Ed., 2020, 59, 2760–2763 CrossRef CAS PubMed.
- (a) E. K. Baylis, Tetrahedron Lett., 1995, 36, 9385–9388 CrossRef CAS; (b) J. Kehler, B. Ebert, O. Dahl and P. Krogsgaard-Larsen, Tetrahedron, 1999, 55, 771–784 CrossRef CAS; (c) Y. A. Rina and J. A. R. Schmidt, Organometallics, 2019, 38, 4261–4270 Search PubMed.
- (a) C.-T. Yang, J. Han, J. Liu, Y. Li, F. Zhang, M. Gu, S. Hu and X. Wang, RSC Adv., 2017, 7, 24652–24655 Search PubMed; (b) L. Chen, Y. Zhu, T. Chen, L. Liu, J.-S. Zhang and L.-B. Han, Org. Biomol. Chem., 2018, 16, 5090–5093 Search PubMed; (c) T. Chen, Q. Tan, X. Liu, L. Liu, T. Huang and L.-B. Han, Synthesis, 2021, 53, 95–106 Search PubMed; (d) Q. Li, C.-Q. Zhao, T. Chen and L.-B. Han, RSC Adv., 2022, 12, 18441–18444 RSC; (e) C.-K. Li, Z.-K. Tao, A. Shoberu, W. Zhang and J.-P. Zou, Org. Lett., 2022, 24, 6083–6087 CrossRef CAS PubMed; (f) R. Wang, J. Wang, Y. Zhang, B. Wang, Y. Xia, F. Xue, W. Jin and C. Liu, Adv. Synth. Catal., 2023, 365, 900–905 Search PubMed; (g) S.-Y. Zhao, Y.-Y. Huang, S.-H. Deng, Z.-P. Guan and Z.-B. Dong, Org. Chem. Front., 2024, 11, 4882–4894 RSC; (h) J.-L. Tu, Z. Shen and B. Huang, Tetrahedron, 2024, 168, 134352 CrossRef CAS; (i) D.-W. Shi, M. Li, R.-J. Yang, X.-Y. Zhao, T. Zhang, J. Huangfu, L. Xu, K. Wang, Y.-X. Ma and B. Yang, J. Org. Chem., 2025, 90, 5578–5585 CrossRef CAS PubMed.
- (a) T. Hirai and L.-B. Han, Org. Lett., 2007, 9, 53–56 CrossRef CAS PubMed; (b) B. Yang, H.-Y. Zhang and S.-D. Yang, Org. Biomol. Chem., 2015, 13, 3561–3565 RSC; (c) J. Liu, H.-Z. Xiao, Q. Fu and D.-G. Yu, Chem. Synth., 2021, 1, 9 CAS; (d) Q. Xu, C. Guo, B. Li, Z. Zhang, Y. Qiu, S. Tian, L. Zheng, L. Gu, W. Yan, D. Wang and J. Zhang, J. Am. Chem. Soc., 2022, 144, 4321–4326 CrossRef CAS PubMed; (e) G. Zhang, H. He, X. Chen, S.-F. Ni and R. Zeng, Org. Lett., 2023, 25, 5356–5360 CrossRef CAS PubMed; (f) M.-Q. Tang, Z.-J. Yang, A.-J. Han and Z.-T. He, Angew. Chem., Int. Ed., 2025, 64, e202413428 CrossRef CAS PubMed; (g) J.-T. Cheng, X. Yuan, Z. Yang, K. Liu and J. Wang, Angew. Chem., Int. Ed., 2025, 64, e202519578 CrossRef CAS PubMed; (h) W.-H. Wang, S.-Y. Zhang, Y.-X. Zhang, Y.-Q. Wang and Q.-W. Zhang, J. Am. Chem. Soc., 2025, 147, 14797–14805 CrossRef CAS PubMed; (i) C. Ma, X. Wang, T. Soós, J. Zhang and J. Yang, Nat. Commun., 2025, 16, 5436 CrossRef PubMed; (j) R. Lin, Y. Zhang, T. Xiong, H. Huang, P. Xie, Y. Zhang, N. Huang, W. Hu and J. Jiang, ACS Catal., 2025, 15, 16236–16246 CrossRef CAS; (k) M. Gong, C. Lin, X. Gu, N. Huang and J. Jiang, Org. Lett., 2025, 27, 10594–10599 CrossRef CAS PubMed.
- (a) L. Wu, X. Zhang, C.-Q. Chen and A.-K. Zhou, Org. Biomol. Chem., 2012, 10, 7859–7862 RSC; (b) Z.-S. Chen, Z.-Z. Zhou, H.-L. Hua, X.-H. Duan, J.-Y. Luo, J. Wang, P.-X. Zhou and Y.-M. Liang, Tetrahedron, 2013, 69, 1065–1068 CrossRef CAS; (c) S. Hyde, J. Veliks, B. Liégault, D. Grassi, M. Taillefer and V. Gouverneur, Angew. Chem., Int. Ed., 2016, 55, 3785–3789 CrossRef CAS PubMed; (d) R. Shen, C. Dong, J. Yang and L.-B. Han, Adv. Synth. Catal., 2018, 360, 4252–4258 CrossRef CAS; (e) L. Zhang, B. M. DeMuynck, A. N. Paneque, J. E. Rutherford and D. A. Nagib, Science, 2022, 377, 649–654 CrossRef CAS PubMed; (f) X. Mo, Y. Xie, L. Wei, X. Gu, M. Zhang, X. Zhang and J. Jiang, Org. Lett., 2023, 25, 2338–2343 CrossRef CAS PubMed.
- K. Suzuki, T. Hashimoto, H. Maeta and T. Matsumoto, Synlett, 1992, 1992, 125–128 CrossRef.
- F. Liu, J. Dong, R. Cheng, S.-F. Yin, L. Chen, L. Su, R. Qiu, Y. Zhou, L.-B. Han and C.-J. Li, Sci. Adv., 2025, 11, eads4626 CrossRef CAS PubMed.
- (a) M. B. Kurosawa, R. Isshiki, K. Muto and J. Yamaguchi, J. Am. Chem. Soc., 2020, 142, 7386–7392 CrossRef CAS PubMed; (b) L. Gan, T. Xu, Q. Tan, M. Cen, L. Wang, J. Zhao, K. Liu, L. Liu, W.-H. Chen, L.-B. Han, J. E. Nycz and T. Chen, Chem. Sci., 2023, 14, 5519–5526 RSC.
- N. B. Bissonnette, N. Bisballe, A. V. Tran, J. A. Rossi-Ashton and D. W. C. MacMillan, J. Am. Chem. Soc., 2024, 146, 7942–7949 CrossRef CAS PubMed.
- (a) D. H. R. Barton, D. Bridon and S. Z. Zard, Tetrahedron Lett., 1986, 27, 4309–4312 CrossRef CAS; (b) D. H. R. Barton and J. Zhu, J. Am. Chem. Soc., 1993, 115, 2071–2072 CrossRef CAS; (c) A. Sato, H. Yorimitsu and K. Oshima, J. Am. Chem. Soc., 2006, 128, 4240–4241 Search PubMed; (d) S. E. Vaillard, C. Muck-Lichtenfeld, S. Grimme and A. Studer, Angew. Chem., Int. Ed., 2007, 46, 6533–6536 Search PubMed; (e) D. T. Xie, H. L. Chen, D. Wei, B. Y. Wei, Z. H. Li, J. W. Zhang, W. Yu and B. Han, Angew. Chem., Int. Ed., 2022, 61, e202203398 CrossRef CAS PubMed; (f) S. K. Pagire, C. Shu, D. Reich, A. Noble and V. K. Aggarwal, J. Am. Chem. Soc., 2023, 145, 18649–18657 CrossRef CAS PubMed; (g) L. Chai, J. Wang, J. Yang, J. Yin, Z. Zhang, Y. Cheng, L. Zhu, X. S. Xue and C. Li, CCS Chem., 2024, 6, 1312–1323 CrossRef CAS; (h) J. Yin, X. Lin, L. Chai, C.-Y. Wang, L. Zhu and C. Li, Chem, 2023, 9, 1945–1954 CrossRef CAS.
- For selected examples of epoxides, see: (a) T. V. Rajanbabu and W. A. Nugent, J. Am. Chem. Soc., 1989, 111, 4525–4527 CrossRef CAS; (b) A. Gansäuer, H. Bluhm and M. Pierobon, J. Am. Chem. Soc., 1998, 120, 12849–12859 CrossRef; (c) Y. Zhao and D. J. Weix, J. Am. Chem. Soc., 2015, 137, 3237–3240 CrossRef CAS PubMed; (d) C. Yao, T. Dahmen, A. Gansäuer and J. Norton, Science, 2019, 364, 764–767 CrossRef CAS PubMed; (e) K. Y. Ye, T. McCallum and S. Lin, J. Am. Chem. Soc., 2019, 141, 9548–9554 Search PubMed; (f) A. Gansäuer, Synlett, 2020, 32, 447–456 Search PubMed; (g) L. Li, S. Yang, Z. Xu, S. Li, J. Jiang and Y.-Q. Zhang, J. Am. Chem. Soc., 2024, 146, 13546–13557 Search PubMed; (h) L. Zhang, W. Wang, C. Shen and K. Dong, ACS Catal., 2025, 15, 7578–7587 Search PubMed.
- For selected examples of alcohols, see: (a) E. E. van Tamelen and M. A. Schwartz, J. Am. Chem. Soc., 1965, 87, 3277–3278 CrossRef CAS; (b) J. E. McMurry, M. G. Silvestri, M. P. Fleming, T. Hoz and M. W. Grayston, J. Org. Chem., 1978, 43, 3249–3255 CrossRef CAS; (c) H. R. Diéguez, A. López, V. Domingo, J. F. Arteaga, J. A. Dobado, M. M. Herrador, J. F. Quílez del Moral and A. F. Barrero, J. Am. Chem. Soc., 2010, 132, 254–259 CrossRef PubMed; (d) T. Suga, S. Shimazu and Y. Ukaji, Org. Lett., 2018, 20, 5389–5392 CrossRef CAS PubMed; (e) T. Suga and Y. Ukaji, Org. Lett., 2018, 20, 7846–7850 CrossRef CAS PubMed; (f) T. Suga, T. Takahashi and Y. Ukaji, Adv. Synth. Catal., 2020, 362, 5622–5627 CrossRef CAS; (g) K. Sumiyama, N. Toriumi and N. Iwasawa, Eur. J. Org Chem., 2021, 2474–2478 Search PubMed; (h) L. Yao, J. Bao, Y. Wang and J. Gui, Org. Lett., 2024, 26, 1243–1248 CrossRef CAS PubMed; (i) X. Zheng, X.-J. Dai, H.-Q. Yuan, C.-X. Ye, J. Ma and P.-Q. Huang, Angew. Chem., Int. Ed., 2013, 52, 3578–3582 CrossRef; (j) T. Suga, Y. Takahashi, C. Miki and Y. Ukaji, Angew. Chem., Int. Ed., 2022, 61, e202112533 CrossRef CAS PubMed; (k) H. Tian, R. Zhang, L. Shi, C. Zhao and X. Wang, Chin. J. Chem., 2023, 41, 1783–1790 CrossRef CAS.
- For selected reviews, see: (a) B. Rossi, S. Prosperini, N. Pastori, A. Clerici and C. Punta, Molecules, 2012, 17, 14700–14732 CrossRef CAS PubMed; (b) J. Streuff and A. Gansäuer, Angew. Chem., Int. Ed., 2015, 54, 14232–14242 CrossRef CAS PubMed; (c) A. Rosales, I. Rodríguez-García, J. Muñoz-Bascón, E. Roldan-Molina, N. M. Padial, L. P. Morales, M. García-Ocaña and J. E. Oltra, Eur. J. Org Chem., 2015, 4567–4591 CrossRef CAS; (d) J. M. Botubol-Ares, M. J. Durán-Peña, J. R. Hanson, R. Hernández-Galán and I. G. Collado, Synthesis, 2018, 50, 2163–2180 CrossRef CAS; (e) J. M. Botubol-Ares, M. J. Durán-Peña, J. R. Hanson, R. Hernández-Galán and I. G. Collado, Synthesis, 2018, 50, 2163–2180 CrossRef CAS; (f) M. Manßen and L. L. Schafer, Chem. Soc. Rev., 2020, 49, 6947–6994 RSC; (g) X. Wu, Y. Chang and S. Lin, Chem, 2022, 8, 1805–1821 CrossRef CAS PubMed; (h) T. Hilche, S. L. Younas, A. Gansäuer and J. Streuff, ChemCatChem, 2022, 14, e202200530 CrossRef CAS; (i) J.-L. Tu and B. Huang, Org. Biomol. Chem., 2024, 22, 6650–6664 RSC.
- (a) J. E. McMurry and M. P. Fleming, J. Am. Chem. Soc., 1974, 96, 4708–4709 CrossRef CAS PubMed; (b) A. Fürstner and A. Hupperts, J. Am. Chem. Soc., 1995, 117, 4468–4475 CrossRef.
- (a) E. J. Corey, R. L. Danheiser and S. Chandrasekaran, J. Org. Chem., 1976, 41, 260–265 CrossRef CAS; (b) A. Gansäuer, Chem. Commun., 1997, 457–458 RSC; (c) M. S. Dunlap and K. M. Nicholas, Synth. Commun., 1999, 29, 1097–1106 CrossRef CAS; (d) F. Calogero, G. Magagnano, S. Potenti, F. Pasca, A. Fermi, A. Gualandi, P. Ceroni, G. Bergamini and P. G. Cozzi, Chem. Sci., 2022, 13, 5973–5981 RSC.
- (a) Y. Yamamoto, D. Matsumi and K. Itoh, Chem. Commun., 1998, 875–876 RSC; (b) J. Streuff, M. Feurer, P. Bichovski, G. Frey and U. Gellrich, Angew. Chem., Int. Ed., 2012, 51, 8661–8664 CrossRef CAS PubMed; (c) M. Feurer, G. Frey, H.-T. Luu, D. Kratzert and J. Streuff, Chem. Commun., 2014, 50, 5370–5372 RSC; (d) M. A. Unkrig-Bau, S. L. Leijendekker and J. Streuff, ChemCatChem, 2025, 17, e202401337 CrossRef CAS.
- (a) R. E. Estévez, J. Justicia, B. Bazdi, N. Fuentes, M. Paradas, D. Choquesillo-Lazarte, J. M. García-Ruiz, R. Robles, A. Gansäuer, J. M. Cuerva and J. E. Oltra, Chem.–Eur. J., 2009, 15, 2774–2791 CrossRef PubMed; (b) J. Justicia, I. Sancho-Sanz, E. Alvarez-Manzaneda, J. E. Oltra and J. M. Cuerva, Adv. Synth. Catal., 2009, 351, 2295–2300 CrossRef CAS; (c) F.-S. Li, Y.-Q. Chen, S.-J. Lin, C.-Z. Shi, X.-Y. Li, Y.-C. Sun, Z.-W. Guo and L. Shi, Org. Chem. Front., 2020, 7, 3434–3438 RSC; (d) A. Gualandi, F. Calogero, M. Mazzarini, S. Guazzi, A. Fermi, G. Bergamini and P. G. Cozzi, ACS Catal., 2020, 10, 3857–3863 CrossRef CAS.
- Y. Wang and X.-Z. Shu, Org. Lett., 2025, 27, 6059–6064 CrossRef PubMed.
- (a) R. E. Estévez, J. L. Oller-López, R. Robles, C. R. Melgarejo, A. Gansäuer, J. M. Cuerva and J. E. Oltra, Org. Lett., 2006, 8, 5433–5436 CrossRef PubMed; (b) J. Streuff, Chem.–Eur. J., 2011, 17, 5507–5510 CrossRef CAS PubMed; (c) P. Bichovski, T. M. Haas, D. Kratzert and J. Streuff, Chem.–Eur. J., 2015, 21, 2339–2342 CrossRef CAS PubMed; (d) P. Bichovski, T. M. Haas, M. Keller and J. Streuff, Org. Biomol. Chem., 2016, 14, 5673–5682 RSC; (e) W. Hao, X. Wu, J. Z. Sun, J. C. Siu, S. N. MacMillan and S. Lin, J. Am. Chem. Soc., 2017, 139, 12141–12144 CrossRef CAS PubMed.
- (a) A. Clerici and O. Porta, Tetrahedron Lett., 1990, 31, 2069–2072 CrossRef CAS; (b) R. Cannella, A. Clerici, N. Pastori, E. Regolini and O. Porta, Org. Lett., 2005, 7, 645–648 CrossRef CAS PubMed; (c) A. Clerici, R. Cannella, W. Panzeri, N. Pastori, E. Regolini and O. Porta, Tetrahedron Lett., 2005, 46, 8351–8354 CrossRef CAS; (d) A. Clerici, R. Cannella, N. Pastori, W. Panzeri and O. Porta, Tetrahedron, 2006, 62, 5986–5994 CrossRef CAS; (e) R. Cannella, A. Clerici, W. Panzeri, N. Pastori, C. Punta and O. Porta, J. Am. Chem. Soc., 2006, 128, 5358–5359 CrossRef CAS PubMed; (f) A. Clerici, A. Ghilardi, N. Pastori, C. Punta and O. Porta, Org. Lett., 2008, 10, 5063–5066 CrossRef CAS PubMed; (g) N. Pastori, C. Greco, A. Clerici, C. Punta and O. Porta, Org. Lett., 2010, 12, 3898–3901 CrossRef CAS PubMed; (h) B. Rossi, N. Pastori, A. Clerici and C. Punta, Tetrahedron, 2012, 68, 10151–10156 CrossRef CAS.
- (a) X. Pang, P.-F. Su and X.-Z. Shu, Acc. Chem. Res., 2022, 55, 2491–2509 CrossRef CAS PubMed; (b) X. Pang and X.-Z. Shu, Chin. J. Chem., 2023, 41, 1637–1652 CrossRef CAS; (c) H. Xie, J. Guo, Y.-Q. Wang, K. Wang, P. Guo, P.-F. Su, X. Wang and X.-Z. Shu, J. Am. Chem. Soc., 2020, 142, 16787–16794 CrossRef CAS PubMed; (d) H. Xie, S. Wang, Y. Wang, P. Guo and X.-Z. Shu, ACS Catal., 2022, 12, 1018–1023 CrossRef CAS; (e) P. Guo, K. Wang, W.-J. Jin, H. Xie, L. Qi, X.-Y. Liu and X.-Z. Shu, J. Am. Chem. Soc., 2021, 143, 513–523 CrossRef CAS PubMed; (f) W.-Y. Ma, G.-Y. Han, S. Kang, X. Pang, X.-Y. Liu and X.-Z. Shu, J. Am. Chem. Soc., 2021, 143, 15930–15935 CrossRef CAS PubMed; (g) H. Xie, S. Wang and X.-Z. Shu, J. Am. Chem. Soc., 2024, 146, 32269–32275 CrossRef CAS PubMed.
- (a) J. Wang, J. Xiao, Z.-L. Tang, D.-H. Lan and L.-B. Han, J. Org. Chem., 2024, 89, 5109–5117 CrossRef CAS PubMed; (b) X. Wu, S. Lu, W. Zhong and T. Xu, Nat. Commun., 2025, 16, 3628 CrossRef CAS PubMed; (c) X. Wu, A. Alimu, W. Zhong and T. Xu, Org. Lett., 2025, 27, 6025 CrossRef CAS PubMed.
- Isotope-labeling experiments indicate that the proton incorporated during the reduction originates from trace amounts of water present in the reaction system (see the SI for details).
- J. Qiao, Z. Liu, R.-N. Ci, Q.-G. Wang, B. Chen, C.-H. Tung and L.-Z. Wu, Quantum Dots Photocatalysis Enabled Direct Activation of Acetone for Radical-Radical Cross-Coupling, Adv. Funct. Mater., 2025, e02878 CrossRef.
- E. A. Boyd, C. Shin, D. J. Charboneau, J. C. Peters and S. E. Reisman, Science, 2024, 385, 847–853 CrossRef CAS PubMed.
- (a) W. G. Bentrude, Acc. Chem. Res., 1982, 15, 117–125 CrossRef CAS; (b) W. G. Bentrude, J. J. L. Fu and P. E. Rogers, J. Am. Chem. Soc., 1973, 95, 3625–3635 CrossRef.
- D. Zhao and R. Wang, Chem. Soc. Rev., 2012, 41, 2095–2108 Search PubMed.
- (a) J. A. Rossi-Ashton, A. K. Clarke, W. P. Unsworth and R. J. K. Taylor, ACS Catal., 2020, 10, 7250–7261 CrossRef CAS PubMed; (b) R. S. Shaikh, S. J. S. Düsel and B. König, ACS Catal., 2016, 6, 8410–8414 Search PubMed; (c) R. S. Shaikh, I. Ghosh and B. König, Chem.–Eur. J., 2017, 23, 12120–12124 CrossRef CAS PubMed; (d) Y. Jian, M. Chen, B. Huang, W. Jia, C. Yang and W. Xia, Org. Lett., 2018, 20, 5370–5374 CrossRef CAS PubMed; (e) W. Lecroq, P. Bazille, F. Morlet-Savary, M. Breugst, J. Lalevée, A. C. Gaumont and S. Lakhdar, Org. Lett., 2018, 20, 4164–4167 CrossRef CAS PubMed; (f) H. Zeng, Q. Dou and C. J. Li, Org. Lett., 2019, 21, 1301–1305 Search PubMed.
- CCDC 2467845: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2ntzxd.
| This journal is © The Royal Society of Chemistry 2026 |