Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
A general route to β,β-carbocyclic sidechains in peptides: an aqueous metallaphotoredox approach driven by green light
Samuel
Gary
,
Pei-Hsuan
Chen
,
Nin
Mai
and
Steven
Bloom
 *
*
Department of Medicinal Chemistry, Gray-Little Hall, Lawrence, KS 66045, USA. E-mail: spbloom@ku.edu
First published on 5th February 2026
Abstract
Amino acids with β,β-carbocyclic sidechains are valuable replacements for endogenous Val, Leu, and Ile, with therapeutic benefits. When placed into ordinary peptides, these annulated variants improve metabolic stability, cell permeability, and receptor affinity and selectivity. Yet, their appearance in modern peptide drugs is often limited to β,β-cyclopentyl- and β,β-cyclohexyl-rings, one reason being the limited availability of resin- and solution-compatible β,β-carbocyclic amino acids for direct coupling. More ‘exotic’ rings, i.e., those with different sizes, chemical compositions, and geometric preferences, could be superior, but finding and assessing their benefits calls for more general ways to incorporate and test them. Herein, we pioneer a modular route to convert a single unsaturated residue, known as β-sulfonyldehydroamino acid (ΔSulf), in a peptide into many unique β,β-carbocycles—cyclic, polycyclic, and heteroatom-containing—in two telescoped steps. First, an unprecedented photocatalyst, Pyronin Y, in an original combination with an organodiiodide, cobalt porphyrin catalyst, sacrificial amine, and green LEDs converts ΔSulf into a Δ-amino acid with a pendant iodide. Adding Zn/Cu couple then triggers an intramolecular and stereoselective Giese cyclization. We detail the mechanism of our procedure, highlighting the interplay between aqueous metallaphotoredox catalysis, halogen-atom abstraction, and ligand-controlled cyclization using spectroscopy, cyclic voltammetry, intermediate-trapping, and radical-clock experiments.
Introduction
Peptides are the third most abundant therapeutic modality—tailing behind biologics and small molecules—and have gained increasing traction in recent years.1 They are incredibly specific and highly potent, display broad chemical and biological diversity, are well-tolerated, and have outstanding therapeutic efficacy.2,3 Many of these salient features can be enhanced by introducing at least one Non-Proteinogenic Amino Acid (NPAA) into the peptide. These NPAAs can take many forms, including those that mirror endogenous residues (isosteric replacements), and those for which no biological counterpart exists. NPAAs that mimic endogenous residues often harbor additional atoms, groups, or stereocenters that promote non-covalent interactions—hydrogen-bonding, π-stacking, space-filling—to drive in vivo activity and therapeutic competence.4,5 One example is cyclic β,β-disubstituted (cββ) amino acids. These synthetic residues are analogous to native Val, Leu and Ile, but their cyclic three-dimensional structure increases their effective size, conformational diversity, and metabolic and plasma stability.6–9 Unsurprisingly, cββs have found their way into several FDA-approved peptide drugs, including epelsiban/retosiban,10,11 zilucoplan12 and telaprevir,13 as well as pre-clinical dipeptidyl peptidase inhibitors8 and anti-apoptotic agents,14Fig. 1. Some peptides even incorporate multiple cββs, such as SEN304, which mimics the hydrophobic core of the Aβ42 peptide (LVFFL) and has been investigated as a possible treatment for Alzheimer's disease.15 But these examples are predominately limited to commercial cββs with cyclopentyl- and cyclohexyl-rings. Other rings may be superior, but their lack of commercial availability prevents them from being sampled and, ultimately, adopted into peptide drugs. Standard approaches to insert cββs into peptides rely on Horner–Wadsworth–Emmons (HWE) reactions between phosphonoglycine (Schmidt's reagent) and designer ketones,16,17 electrophilic alkylation of glycine residues,18,19 or redox-mediated cross-dehydrogenative coupling on N-arylglycine residues.19–23 Although moderately successful, these methods have limitations including poor chemoselectivity when using peptides with acidic or oxidizable sidechains, risks of α-epimerization, and inability to be performed at interior positions in the peptide, Fig. 1. Therefore, most cββs are prepared in a separate chemical synthesis and then integrated into the nascent polypeptide using liquid-phase or solid-phase peptide synthesis. To accelerate cββ peptide synthesis and discovery, it would be better if a single progenitor residue could first be positioned in the peptide and then transformed, in parallel, into a bevy of new cββs, yielding a comprehensive inventory of peptides for biochemical testing. Such an approach would not only streamline hit-to-lead campaigns with cββs but allow medicinal chemists to quickly customize a diverse set of probe molecules to map protein binding sites and elucidate complex structure–activity relationships. Hereafter, we describe our approach to addressing this timely challenge.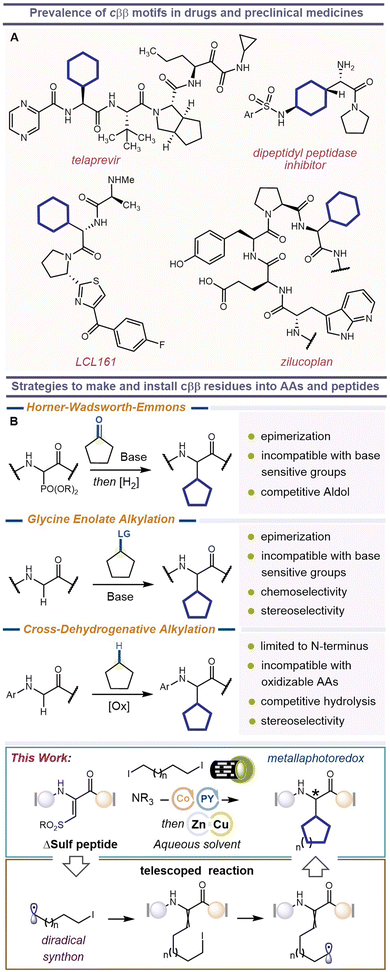 | ||
| Fig. 1 (A) Examples of β,β-cycloalkyl amino acids found in bioactive molecules. (B) Current methods used in the synthesis of β,β-cycloalkyl amino acid derivatives and this work. | ||
Experimental design plan
We envisioned a divergent approach to cββs using our recently reported β-sulfonyldehydroamino acid (ΔSulf).24 This new bioorthogonal reagent can undergo two successful C–C bond-forming events by intercepting two different C-centered radicals. The first C-radical displaces the sulfonyl group, while the second C-radical performs a Giese reaction to furnish the saturated β,β-disubstituted product. If we use a single reagent that can give rise to both radicals—a diradical synthon—then we could fashion an annulated product, i.e., the desired cββ, Fig. 1. It might also be possible to bias cyclization to deliver D-cββs and L-cββs in a stereoselective manner.Results and discussion
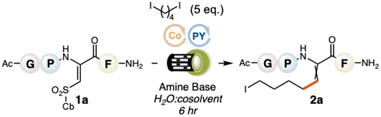
To explore our design plan, we selected a model tetrapeptide, AcNH-Gly-Pro-ΔSulfCb-Phe-CONH2 (1a), where the sulfonyl group of our dehydroamino acid is capped with a cyclobutyl (Cb) ring. We surveyed diiodobutane (diradical synthon) with twenty-eight photoredox catalysts, using 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 H2O
1 H2O![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) TFE as solvent and N,N-diisopropylethylamine (DIPEA) as a sacrificial amine. We also surveyed electrochemical conditions; see SI S11–S13. None of these afforded the 5-membered cββ product (3a). The only noteable product was the β-alkylated dehydroamino acid 2a, uniquely formed with a green-light (525 nm) activated Pyronin Y (PY+) photocatalyst in 15% conversion. 2a is produced when the first radical is formed and displaces the sulfonyl group, but the second radical that closes the ring through intramolecular conjugate addition is not generated. The formation of 2a could not be enhanced by substituting the organodiiodide with other diradical synthons, e.g., 1,4-Katritzky salts, -N-hydroxyphthalimide esters, and -dibromides; see SI S14. Because the second C–I bond remains intact, it is still possible to forge the annulated cββ product from 2a through conjugate addition of the newly tethered alkyliodide onto the adjoining π-system. We found that this could be done by adding Cu(acac)2 and Zn powder to the crude reaction mixture,25 delivering the desired cββ; a two-step approach done in a single reaction vessel without purification of the intermediate.
TFE as solvent and N,N-diisopropylethylamine (DIPEA) as a sacrificial amine. We also surveyed electrochemical conditions; see SI S11–S13. None of these afforded the 5-membered cββ product (3a). The only noteable product was the β-alkylated dehydroamino acid 2a, uniquely formed with a green-light (525 nm) activated Pyronin Y (PY+) photocatalyst in 15% conversion. 2a is produced when the first radical is formed and displaces the sulfonyl group, but the second radical that closes the ring through intramolecular conjugate addition is not generated. The formation of 2a could not be enhanced by substituting the organodiiodide with other diradical synthons, e.g., 1,4-Katritzky salts, -N-hydroxyphthalimide esters, and -dibromides; see SI S14. Because the second C–I bond remains intact, it is still possible to forge the annulated cββ product from 2a through conjugate addition of the newly tethered alkyliodide onto the adjoining π-system. We found that this could be done by adding Cu(acac)2 and Zn powder to the crude reaction mixture,25 delivering the desired cββ; a two-step approach done in a single reaction vessel without purification of the intermediate.
The combination of PY+ and DIPEA was further optimized for cββ formation under 525 nm irradiation. One immediate challenge was the low conversion to the intermediate β-iodoalkyl-dehydroamino acid (15% conversion). A possible reason for this poor conversion is that primary alkyl radicals—from our organodiiodide—are highly reactive and prone to side reactions, e.g., H-atom abstraction, intramolecular cyclization, and homodimerization.26–28 Adding a transition metal salt could sequester the alkyl radical and help guide the first addition step (sulfonyl displacement) through an organometallic mechanism. Including a cobalt porphyrin, CoII [tetra(p-methoxyphenyl)]porphyrin (CoIITMPP), nearly doubled our conversion (29%). This finding is significant not only for its ability to improve our yield, but that synergistic reactions involving metals and photocatalysts are incredibly rare in water, with only a handful of examples being reported.29–31 However, CoII also encouraged the conjugate addition of TFE to ΔSulfCb. Switching the cosolvent to MeCN eliminated this background reaction and provided a small boost in conversion (30%). Decreasing the loadings of PY+ and CoIITMPP to 7 mol% and 1 mol%, respectively, further increased the conversion to 41%. At this point, we gathered that a change in either CoII catalyst, amine base, or ΔSulf substrate would be needed to bolster conversions beyond 40%. A survey of electronically distinct porphyrin and phthalocyanine cobalt ligands revealed that our original CoIITMPP was optimal; see SI S16. Using tri-n-butylamine (NBu3) improved conversions to 53%. And finally, exchanging the cyclobutyl group of ΔSulfCb for an n-propyl group (ΔSulfnPr, 1b) gave 56% conversion. Refinements in concentration, i.e., moving from 2 mM to 4 mM and adding phosphate buffer (pH 7, 10 mM overall), provided an optimal and reproducible yield of 63% (4![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 Z
1 Z![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) E geometric isomers), respectively. Key optimizations are summarized in Table 1. Even with these optimized conditions, we still did not observe the formation of 3a. Moreover, isolating 2a and resubjecting it to our optimized protocol failed to produce 3a, suggesting that our metallaphotoredox system does not induce cyclization. The only observable byproducts in our reaction were reductive cleavage of the β-sulfonyl group from 1b, reduction of the resulting dehydroalanine residue to saturated alanine, and hydrodehalogenation of 2a. As a final point of interest, we checked the compatibility of our system with different amino acids, including aromatic (Phe), aliphatic (Leu), oxidizable (Trp, Tyr, Met), β-branched (Val), and basic (His, Lys) residues, using a series of dipeptides, i.e., AcNH-ΔSulfCy-AA-CO2Me, where AA = amino acid. All residues except Lys and Met (oxidized to sulfoxide) were tolerated; avg. 41% for alkylation (step 1) and avg. 30% for cyclization (step 2). See SI S17–S23 for dipeptide results.
E geometric isomers), respectively. Key optimizations are summarized in Table 1. Even with these optimized conditions, we still did not observe the formation of 3a. Moreover, isolating 2a and resubjecting it to our optimized protocol failed to produce 3a, suggesting that our metallaphotoredox system does not induce cyclization. The only observable byproducts in our reaction were reductive cleavage of the β-sulfonyl group from 1b, reduction of the resulting dehydroalanine residue to saturated alanine, and hydrodehalogenation of 2a. As a final point of interest, we checked the compatibility of our system with different amino acids, including aromatic (Phe), aliphatic (Leu), oxidizable (Trp, Tyr, Met), β-branched (Val), and basic (His, Lys) residues, using a series of dipeptides, i.e., AcNH-ΔSulfCy-AA-CO2Me, where AA = amino acid. All residues except Lys and Met (oxidized to sulfoxide) were tolerated; avg. 41% for alkylation (step 1) and avg. 30% for cyclization (step 2). See SI S17–S23 for dipeptide results.
| Entry | Photocatalyst | Metal catalyst | Amine | Solvent | Conversion |
|---|---|---|---|---|---|
| a Reactions performed with ΔSulfCb and 5 eq. of amine base at [2 mM] unless stated otherwise. b Reaction performed with ΔSulfnPr. c Reaction performed at [4 mM]. | |||||
| 1 | PY+ (0.1 eq.) | — | DIPEA | H2O:TFE | 15% |
| 2 | PY+ (0.1 eq.) | CoTMPP (0.1 eq.) | DIPEA | H2O:TFE | 29% |
| 3 | PY+ (0.1 eq.) | CoTMPP (0.1 eq.) | DIPEA | H2O:MeCN | 30% |
| 4 | PY+ (0.07 eq.) | CoTMPP (0.01 eq.) | DIPEA | H2O:MeCN | 41% |
| 5 | PY+ (0.07 eq.) | CoTMPP (0.01 eq.) | NBu3 | H2O:MeCN | 53% |
| 6b | PY+ (0.07 eq.) | CoTMPP (0.01 eq.) | NBu3 | H2O:MeCN | 56% |
| 7b,c | PY+ (0.07 eq.) | CoTMPP (0.01 eq.) | NBu3 | H2O:MeCN | 56% |
| 8 , | PY + (0.07 eq.) | CoTMPP (0.01 eq.) | NBu 3 | Kpi:MeCN | 63% |
| 9b,c | PY+ (0.01 eq.) | — | NBu3 | Kpi:MeCN | 32% |
| 10b,c | PY+ (0.07 eq.) | CoTMPP (0.01 eq.) | — | Kpi:MeCN | 0% |
| 11b,c | — | CoTMPP (0.01 eq.) | NBu3 | Kpi:MeCN | 0% |
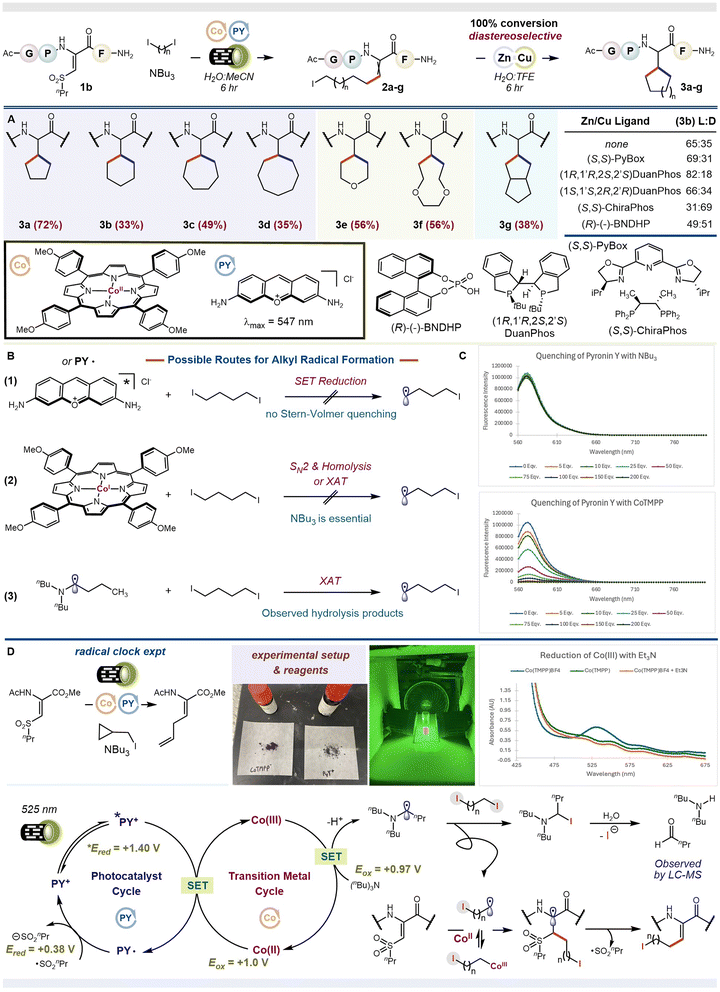
We examined the scope of our reaction, Fig. 2A. We found that ring sizes of 5-, 6-, 7-, and 8- (compounds 3a–d) could be fashioned from the corresponding diiodides using our standard peptide and optimized metallaphotoredox conditions, followed by addition of Cu(acac)2 and Zn powder to the crude reaction mixture. We found that including TFE could accelerate the ring-closing step, affording complete (100%) conversion in 6 h. We also found that one of the two possible diastereomers from ring closure is favored. By comparison to authentic samples, we deduced that our reaction favors the L-diastereomer (dr 65![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 35); See SI S75–S78. This innate bias could be enhanced when a chiral ligand, e.g., (1R,1′R,2S,2′S)DuanPhos, was coordinated to Cu(acac)2 in the second step on the crude reaction material; shown for compound 3b (dr 82
35); See SI S75–S78. This innate bias could be enhanced when a chiral ligand, e.g., (1R,1′R,2S,2′S)DuanPhos, was coordinated to Cu(acac)2 in the second step on the crude reaction material; shown for compound 3b (dr 82![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 18). However, using the opposing (1S,1′S,2R,2′R)DuanPhos enantiomer failed to produce the opposite peptide diastereomer, indicating a matched vs. mismatched case, respectively. Using a different ligand altogether, i.e., ChiraPhos, flipped the selectivity to favor the D-diastereomer (dr 31
18). However, using the opposing (1S,1′S,2R,2′R)DuanPhos enantiomer failed to produce the opposite peptide diastereomer, indicating a matched vs. mismatched case, respectively. Using a different ligand altogether, i.e., ChiraPhos, flipped the selectivity to favor the D-diastereomer (dr 31![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 69), overriding the innate preference of the peptide substrate.32–34 (comparable diastereoselectivities were obtained when using purified 2b in place of the crude mixture). Bidentate and tridentate amine-based ligands did not influence the stereochemical outcome of our reaction and chiral Brønsted acids tended to give racemic mixtures. A more comprehensive survey of chiral ligands is included in the SI, see S24–S31. We plan to examine other phosphine-based ligands in future reports, including chiral variants thereof. Diiodides that form heteroatom-containing cββs also worked (compounds 3e and 3f) as did ones that yield bicyclic cββs (compound 3g). In all cases, the peptide products could be purified by routine flash chromatography in quantities suitable for early-phase medicinal chemistry projects (>0.1 mg, with purities exceeding 65%), as previously shown by our lab.35 In fact, we tested a few of our products as inhibitors of Aβ42 fibril formation, but they were not effective.
69), overriding the innate preference of the peptide substrate.32–34 (comparable diastereoselectivities were obtained when using purified 2b in place of the crude mixture). Bidentate and tridentate amine-based ligands did not influence the stereochemical outcome of our reaction and chiral Brønsted acids tended to give racemic mixtures. A more comprehensive survey of chiral ligands is included in the SI, see S24–S31. We plan to examine other phosphine-based ligands in future reports, including chiral variants thereof. Diiodides that form heteroatom-containing cββs also worked (compounds 3e and 3f) as did ones that yield bicyclic cββs (compound 3g). In all cases, the peptide products could be purified by routine flash chromatography in quantities suitable for early-phase medicinal chemistry projects (>0.1 mg, with purities exceeding 65%), as previously shown by our lab.35 In fact, we tested a few of our products as inhibitors of Aβ42 fibril formation, but they were not effective.
Mechanistic studies
To unravel the synergistic effect between PY+ and CoIITMPP, a series of mechanistic experiments were performed. First, we conducted a radical clock experiment to probe the involvement of free radicals in our reaction. ΔSulfnPr was reacted with 1-iodomethylcyclopropane in the presence of PY+, CoIITMPP, and NBu3. Loss of an iodine atom generates a primary carbon-centered radical that unfurls the cyclopropane ring, forming an intermediate homoallylic radical (k ∼108)36 that can conjugate to ΔSulfnPr. Addition of a new alkene fragment to ΔSulfnPr can, therefore, be taken as evidence for the involvement of a transient alkyl radical. Indeed, we observed the ring-opened olefin product, Fig. 2D & SI S64. Including 3 equivalents of TEMPO, a reagent commonly used to trap free radicals, abolished all reactivity. Together, these initial results show that our metallaphotoredox conditions turn C–I bonds into free carbon-radicals that engage ΔSulfnPr through open–shell conjugate addition.How does the carbon-radical form from the diiodide? We envisioned three scenarios involving PY+ (λmax 547 nm) and CoIITMPP (Q1 band λmax 548 nm), both of which absorb the green light used in our standard reaction, Fig. 2B: (1) SET reduction of the organodiiodide by excited state *PY+ or ground state PY˙, the latter being formed via reductive quenching of *PY+ by sacrificial NBu3 or CoIITMPP, (2) SN2-like oxidative addition of CoITMPP into the C–I bond followed by photolytic cleavage. The requisite CoI species is formed from CoIIvia oxidative quenching of *PY+ or reduction by NBu3, and (3) iodine-atom abstraction by neutral Bu2NC·H(CH2)2CH3, formed from oxidation of NBu3 by *PY+ or *CoIITMPP. Stern–Volmer quenching is an excellent way to interrogate these three possibilities (Fig. 2C). For *PY+, no significant quenching was observed with NBu3, CH3(CH2)3I, or ΔSulfnPr, the last study being done with and without NBu3. Even though quenching of the excited state photocatalyst by NBu3 may appear insignificant, the fact that our reaction works when only NBu3 and PY+ are present, as shown in Table 1, suggests that this process, common to many photoredox reactions,37,38 can still operate, leading to intermediate dehydroamino acid 2a when cobalt is not present. Only CoIITMPP significantly quenched *PY+, appearing linear at low concentrations of CoIITMPP and exponential at higher concentrations of CoIITMPP (≥100 equivalents). This change indicates that both dynamic (diffusion-controlled SET) and static (EDA complexation or ground state electron transfer) mechanisms are operative at high CoIITMPP concentrations.39 Because our reaction is performed with sub-stoichiometric amounts of CoII, only linear quenching should be considered further. This notion is also supported by the fact that no new spectral peaks appear when reaction quantities of CoIITMPP and PY+ are combined. In other words, no new complexes or catalytic entities are formed in the ground state. The ability of CoIITMPP to quench *PY+ suggests that CoII can function beyond capturing and stabilizing a transient alkyl radical; it can function as a redox mediator. Interestingly, its fluorescence is also quenched by PY+, highlighting that an intimate redox pair exist between CoIITMPP and PY+ in the excited state. Note: the prior experiment required excitation of the CoIITMPP Soret band (λmax 433 nm), as no fluorescence was observed at λex > 500 nm. No other reagent combinations showed any significant quenching. To summarize, our Stern–Volmer studies indicate that electron and/or energy transfer occurs best between *CoIITMPP/PY+ and *PY+/CoIITMPP pairs; a result which means that scenario 1 is not the sole mechanism. The fact that the *CoII lifetime is very short (τ1/2 ∼ 12 ps)40 also makes chemistries that invoke it unlikely. Hence, the interaction between *PY+/CoIITMPP is the most probable reaction pathway leading to alkyl radical formation. See SI S48–S60 for complete spectroscopic data.
Which way do electrons or energy flow, i.e., from *PY+ + CoII → PY˙++ + CoI or *PY+ + CoII → PY˙ + CoIII? And, does the resulting intermediate drive carbon-radical formation? To answer this next question, we performed a series of control reactions, systematically removing each component from our reaction mixture. If an electron moves from *PY+→CoII, the resulting CoI species (scenario 2) should be able to complete our reaction in the absence of NBu3, forming the alkyl radical through iodine-atom abstraction or an SN2-like mechanism. The same is not true if an electron moves from CoII → *PY+, yielding CoIII. In this case, NBu3 would be critically essential, serving as both a sacrificial reductant and possible iodine-atom abstractor. We found that no reaction occurred in the absence of NBu3, consistent with *PY+ oxidation of CoII. Hence, scenario 2 is not viable. We also observed no reaction when PY+ was removed, suggesting that CoIITMPP does not independently drive productive chemistry, either by electron or energy transfer from its ground or excited states. To further validate the *PY+ + CoII → PY˙ + CoIII pathway, we determined the excited state redox potentials of *PY+ and the ground state redox potentials of CoIITMPP. PY+ shows irreversible ground state oxidation at EOx = 1.27 V vs. SCE and irreversible reduction at ERed = −0.79 V vs. SCE.41 Combined with absorbance and fluorescence measurements, the excited state redox potentials of *PY+ are estimated as *ERed = +1.40 V vs. SCE and *EOx = −0.92 V vs. SCE. Ground state CoII/CoIII oxidation occurs at a peak potential of EOx = 1.0 V vs. SCE.42 Thus, SET oxidation of CoIITMPP by *PY+ is thermodynamically feasible (∼−10 kcalmol−1). See SI S61, S62 and S65 for spectroscopic data. We are now left with scenario 3. Formation of Bu2NC·H(CH2)2CH3 abstractor could occur if the newly generated CoIIITMPP oxidizes NBu3 (EOx = 0.97 V vs. SCE) to +˙NBu3, which is subsequently deprotonated. To test the feasibility of this step, we prepared CoIIITMPP+BF4− according to methods by Jones.43 For this species, a new absorption peak was observed at 533 nm. Addition of NBu3 resulted in quantitative disappearance of this new spectral feature and the reemergence of bands consistent with CoIITMPP. We interpreted this as oxidation of NBu3 by CoIIITMPP to yield +˙NBu3 and CoII. No complex was evident between NBu3 and the resulting CoII, as later confirmed by separate UV-vis studies; see SI S49. The final step is to show that Bu2NC·H(CH2)2CH3 can abstract an iodine atom from our organodiiodide to yield an alkyl radical. Fortunately, this situation already has literature support, being studied by Leonori.44–46 Additionally, we performed two control reactions, substituting NBu3 for pyridine and DABCO. Neither of these bases can form the requisite α-amino radical needed for halide abstraction. Hence, neither base should work in our reaction. Indeed, this was the case. The resulting α-iodo amine or, more precisely, its iminium congener, is hydrolyzed to dibutylamine and butyraldehyde, with HNBu2 being detected by LC-MS of our crude reaction mixture, see SI S79. From the above experiments, we propose the following mechanism shown in Fig. 2D. Upon green light irradiation, *PY+ oxidizes CoIITMPP to CoIIITMPP, which oxidizes NBu3 to +˙NBu3, returning CoII. Deprotonation of +˙NBu3 (pKa ∼8)47 yields an α-amino radical that abstracts one of the two iodine atoms from our organodiiodide. Conjugate addition of the resulting carbon-centered radical to ΔSulfnPr yields a β-sulfonyl radical, which undergoes β-scission to release ˙SulfnPr and the alkylated dehydroamino acid with the second C–I bond still intact. (Note: the free carbon-radical could also be reversibly captured by CoII, stabilizing it for subsequent addition to ΔSulfnPr.48,49 It does not covalently modify PY+ or CoIITMPP according to UV-vis tracking of these two reaction components, which appear unaltered at 2 h into the reaction; see SI S49) Reduction of the sulfonyl radical (ERed = +0.38 V vs. SCE for ˙SO2Me)50 by reduced PY˙ delivers a sulfinate salt, closing the photoredox cycle. Intramolecular cyclization is then induced in a separate chemical step by adding Cu(acac)2 and Zn powder.
Conclusions
In conclusion, we developed a one-pot, two-step process to transform β-sulfonyldehydroamino acids in peptides into one of several cββs using organodiiodides as ‘ring-transfer reagents’. In the first step, we show that a potent halogen-atom abstractor, borne from the unique combination of an unprecedented photocatalyst, PY+, and CoIITMPP and green light, attaches the diiodide to the β-sulfonyldehydroamino acid, excising the sulfonyl group and one of the two iodine atoms. Adding Zn/Cu couple then induces ring closure in the same reaction vessel. This two-step process has the advantage of enabling different ring chemotypes to be successfully made and to control their stereochemistry through judicious choice of ligand in step 2. Overall, our procedure provides peptide medicinal chemists with a powerful new synthetic tool and offers key mechanistic insights for designing cooperative metallaphotoredox and halogen-atom transfer reactions in water and at longer wavelengths.Author contributions
S. G. and S. B. conceived the project. S. G. carried out the method development and experimental and mechanistic investigations, with assistance from P.-H. C. and N. M. S. B. supervised the project and secured funding. All authors contributed to data analysis and participated in writing and editing the final manuscript.Conflicts of interest
The authors declare no competing financial interest.Data availability
Supplementary information (SI): experimental optimizations, general considerations, experimental procedures, and characterization data for all compounds including NMR spectra and LC-MS spectra. See DOI: https://doi.org/10.1039/d5sc08845c.Acknowledgements
We thank Farjana Afroj and Guangyu Zhang of the University of Kansas for their help in reviewing this manuscript. Some of the material presented in this manuscript is also covered in the author's (S. Gary) graduate thesis: publication number 32041662 & submission ID 20080. The thesis has been embargoed until 2026. We have also filed a provisional patent (US Patent Appl. No. 63/858,734) discussing this work. This work was performed in the Department of Medicinal Chemistry at the University of Kansas and supported the National Institute of General Medical Sciences (NIGMS) of the National Institutes of Health under award number R35GM147169. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institutes of Health.Notes and references
- K. Sharma, K. K. Sharma, A. Sharma and R. Jain, Peptide-based drug discovery: Current status and recent advances, Drug Discovery Today, 2023, 28(2), 1–15, DOI:10.1016/j.drudis.2022.103464.
- J. Fetse, S. Kandel, U. F. Mamani and K. Cheng, Recent advances in the development of therapeutic peptides, Trends Pharmacol. Sci., 2023, 44(7), 425–441, DOI:10.1016/j.tips.2023.04.003.
- L. Wang, N. X. Wang, W. P. Zhang, X. R. Cheng, Z. B. Yan, G. Shao, X. Wang, R. Wang and C. Y. Fu, Therapeutic peptides: current applications and future directions, Signal Transduction Targeted Ther., 2022, 7, 1, DOI:10.1038/s41392-022-00904-4.
- Y. Ding, J. P. Ting, J. S. Liu, S. Al-Azzam, P. Pandya and S. Afshar, Impact of non-proteinogenic amino acids in the discovery and development of peptide therapeutics, Amino Acids, 2020, 52(9), 1207–1226, DOI:10.1007/s00726-020-02890-9.
- J. L. Hickey, D. Sindhikara, S. L. Zultanski and D. M. Schultz, Beyond 20 in the 21st Century: Prospects and Challenges of Non-canonical Amino Acids in Peptide Drug Discovery, ACS Med. Chem. Lett., 2023, 14(5), 557–565, DOI:10.1021/acsmedchemlett.3c00037.
- O. Nyeki, K. S. Szalay, L. Kisfaludy, E. Karpati, L. Szporny, G. B. Makara and B. Varga, Synthesis of Angiotensin-Ii Antagonists with Variations in Position 5, J. Med. Chem., 1987, 30(10), 1719–1724, DOI:10.1021/jm00393a006.
- H. Morii, H. Uedaira, K. Ogata, S. Ishii and A. Sarai, Shape and energetics of a cavity in c-Myb probed by natural and non-natural amino acid mutations, J. Mol. Biol., 1999, 292(4), 909–920, DOI:10.1006/jmbi.1999.3099.
- E. R. Parmee, J. F. He, A. Mastracchio, S. D. Edmondson, L. Colwell, G. Eiermann, W. P. Feeney, B. Habulihaz, H. B. He and R. Kilburn, et al., 4-Amino cyclohexylglycine analogues as potent dipeptidyl peptidase IV inhibitors, Bioorg. Med. Chem. Lett., 2004, 14(1), 43–46, DOI:10.1016/j.bmcl.2003.10.016.
- P. Udompholkul, A. Garza-Granados, G. Alboreggia, C. Baggio, J. McGuire, S. D. Pegan and M. Pellecchia, Characterization of a Potent and Orally Bioavailable Lys-Covalent Inhibitor of Apoptosis Protein (IAP) Antagonist, J. Med. Chem., 2023, 66(12), 8159–8169, DOI:10.1021/acs.jmedchem.3c00467.
- R. Shinghal, A. Barnes, K. M. Mahar, B. Stier, L. Giancaterino, L. D. Condreay, L. Black and S. W. McCallum, Safety and Efficacy of Epelsiban in the Treatment of Men with Premature Ejaculation: A Randomized, Double-Blind, Placebo-Controlled, Fixed-Dose Study, J. Sex. Med., 2013, 10(10), 2506–2517, DOI:10.1111/jsm.12272.
- S. Thornton, G. Valenzuela, C. Baidoo, M. J. Fossler, T. H. Montague, L. Clayton, M. Powell, J. Snidow, B. Stier and D. Soergel, Treatment of spontaneous preterm labour with retosiban: a phase II pilot dose-ranging study, Br. J. Clin. Pharmacol., 2017, 83(10), 2283–2291, DOI:10.1111/bcp.13336.
- M. Shirley, Zilucoplan: First Approval, Drugs, 2024, 84(1), 99–104, DOI:10.1007/s40265-023-01977-3.
- J. G. McHutchison, M. P. Manns, A. J. Muir, N. A. Terrault, I. M. Jacobson, N. H. Afdhal, E. J. Heathcote, S. Zeuzem, H. W. Reesink and J. Garg, et al., Telaprevir for Previously Treated Chronic HCV Infection, N. Engl. J. Med, 2010, 362(14), 1292–1303, DOI:10.1056/NEJMoa0908014.
- N. Pemmaraju, B. Z. Carter, P. Bose, N. Jain, T. M. Kadia, G. Garcia-Manero, C. E. Bueso-Ramos, C. D. DiNardo, S. Bledsoe and N. G. Daver, et al., Final results of a phase 2 clinical trial of LCL161, an oral SMAC mimetic for patients with myelofibrosis, Blood Adv., 2021, 5(16), 3163–3173, DOI:10.1182/bloodadvances.202000.
- K. Kokkoni, H. Scott, H. Amijee, J. M. Mason and A. J. Doig, N-Methylated Peptide Inhbitors of B-Amyloid Aggregation and Toxicity. Optimization of the Inhibitor Structure, Biochemistry, 2006, 45, 9906–9918, DOI:10.1021/BI060837S.
- F. Wakenhut, T. D. Tran, C. Pickford, S. Shaw, M. Westby, C. Smith-Burchnell, L. Watson, M. Paradowski, J. Milbank and D. Stonehouse, et al., The Discovery of Potent Nonstructural Protein 5A (NS5A) Inhibitors with a Unique Resistance Profile-Part 2, ChemMedChem, 2014, 9(7), 1387–1396, DOI:10.1002/cmdc.201400046.
- H. L. Shi, Y. Wan and Y. Q. Zhang, Synthesis of Diverse Oxetane Amino Acids via Visible-Light-Induced Photocatalytic Decarboxylative Giese-Type Reaction, Chin. J. Chem., 2024, 42(12), 1341–1346, DOI:10.1002/cjoc.202300750.
- P. Kong, Y. W. Ye, X. Zhang, X. Z. Bao and C. D. Huo, Alkylation of Glycine Derivatives through a Synergistic Single-Electron Transfer and Halogen-Atom Transfer Process, Org. Lett., 2024, 26(36), 7507–7513, DOI:10.1021/acs.orglett.4c02352.
- S. Yang, H. Hu, J. H. Li and M. Chen, Direct Alkylation of Glycine Derivativesvia Photoinduced Palladium Catalysis, Utilizing Intermolecular Hydrogen Atom Transfer Mediated by Alkyl Radicals, ACS Catal., 2023, 13(24), 15652–15662, DOI:10.1021/acscatal.3c04075.
- M. H. Babu and J. Sim, Radical-Mediated C-H Alkylation of Glycine Derivatives: A Straightforward Strategy for Diverse α-Unnatural Amino Acids, Eur. J. Org Chem., 2022, 2022, 45, DOI:10.1002/ejoc.202200859.
- M. S. Segundo, I. Guerrero and A. Correa, Co-Catalyzed C(sp3)–H Oxidative Coupling of Glycine and Peptide Derivatives, Org. Lett., 2017, 19(19), 5288–5291 CrossRef PubMed.
- G.-Z. Wang, M.-C. Fu, B. Zhao and R. Shang, Photocatalytic decarboxylative alkylations of C(sp3)-H and C(sp2)-H bonds enabled by ammonium iodide in amide solvent, Sci. China Chem., 2021, 64, 439–444 CrossRef CAS.
- P. Kong, P. Zhang, Y. Ye, C. Yang, Y. Yuan and C. Huo, Catalytic Decarboxylative Alkylation of Glycine Derivatives by Aliphatic Carboxylic Acids, Org. Lett., 2025, 27(30), 8200–8205 CrossRef CAS PubMed.
- P.-H. Chen and S. Bloom, Divergent Synthesis of ΔAA-Peptides using a Bioorthogonal pro-Amino Acid and Aqueous Flavin Photocatalyst: Green Light Enhances Catalyst Performance and Product Selectivity, Angew. Chem., Int. Ed., 2025, 64(39), e202511832, DOI:10.1002/anie.202511832R1.
- Our lab has previously shown that Zn/Cu couple mediates intramolecular cyclization between alkyliodides and dehydroamino acids in peptides, S. Gary and S. Bloom, Peptide Carbocycles: From -SS- to -CC- via a Late-Stage Snip- and-Stitch, ACS Cent. Sci., 2022, 8(11), 1537–1547, DOI:10.1021/acscentsci.2c00456.
- P. Poizot, V. Jouikov and J. Simonet, Glassy carbon modified by a silver-palladium alloy: cheap and convenient cathodes for the selctive reductive homocoupling of alkyl iodides, Tetrahedron Lett., 2009, 50, 822–824 CrossRef CAS.
- F. O. Ginah, T. A. Donovan Jr, S. D. Suchan, D. R. Pfennig and G. W. Ebert, Homocoupling of Alkyl Halides and Cyclization of a,w-Dihaloalkanes via Activated Copper, J. Org. Chem., 1990, 55, 584–589 CrossRef CAS.
- J. F. Garst and J. T. Barbas, On the mechanism of alkyl dimer formation in reactions of alkyl iodides with sodium naphthalene. Evidence from reactions of 1,4-diiodobutane, Tetrahedron Lett., 1969, 10, 3125–3128 CrossRef.
- M. Chilamari, J. R. Immel, P.-H. Chen, B. M. Alghafli and S. Bloom, Flavin Metallaphotoredox Catalysis: Synergistic Synthesis in Water, ACS Catal., 2022, 12(7), 4175–4181, DOI:10.1021/acscatal.2c00773.
- D. K. Kölmel, J. Meng, M. H. Tsai, J. M. Que, R. P. Loach, T. Knauber, J. Q. Wan and M. E. Flanagan, On-DNA Decarboxylative Arylation: Merging Photoredox with Nickel Catalysis in Water, ACS Comb. Sci., 2019, 21(8), 588–597, DOI:10.1021/acscombsci.9b00076.
- D. K. Kölmel, A. S. Ratnayake and M. E. Flanagan, Photoredox cross-electrophile coupling in DNA-encoded chemistry, Biochem. Biophys. Res. Commun., 2020, 533(2), 201–208, DOI:10.1016/j.bbrc.2020.04.028.
- H. M. Key and S. J. Miller, Site- and Stereoselective Chemical Editing of Thiostrepton by Rh-Catalyzed Conjugate Arylation: New Analogues and Collateral Enantioselective Synthesis of Amino Acids, J. Am. Chem. Soc., 2017, 139(43), 15460–15466, DOI:10.1021/jacs.7b08775.
- X. X. Qi, S. Jambu, Y. N. Ji, K. M. Belyk, N. R. Panigrahi, P. S. Arora, N. A. Strotman and T. N. Diao, Late-Stage Modification of Oligopeptides by Nickel-Catalyzed Stereoselective Radical Addition to Dehydroalanine, Angew. Chem., Int. Ed., 2022, 61(48), e202213315, DOI:10.1002/anie.202213315.
- C. J. Richards and R. A. Arthurs, Catalysit optimization for asymmetric synthesis by ligand chirality element addition: a perspective on stereochemical cooperativity, Chem.–Eur. J., 2017, 23(48), 11460–11478, DOI:10.1002/chem.201700926.
- J. R. Immel, M. Chilamari and S. Bloom, Combining flavin photocatalysis with parallel synthesis: a general platform to optimize peptides with non-proteinogenic amino acids, Chem. Sci., 2021, 12, 10083–10091 RSC.
- P. S. Engel, S. L. He, J. T. Banks, K. U. Ingold and J. Lusztyk, Clocking tertiary cyclopropylcarbinyl radical rearrangements, J. Org. Chem., 1997, 62(5), 1210–1214, DOI:10.1021/jo961685h.
- M. H. Shaw, J. Twilton and D. W. C. MacMillan, Photoredox Catalysis in Organic Chemistry, J. Org. Chem., 2016, 81(16), 6898–6926 Search PubMed.
- N. Romero and D. A. Nicewicz, Organic Photoredox Catalysis, Chem. Rev., 2016, 116(17), 10075–10166 CrossRef CAS PubMed.
- E. Ciotta, P. Prosposito and R. Pizzoferrato, Positive curvature in Stern-Volmer plot described by a generalized model for static quenching, J. Lumin., 2019, 206, 518–522, DOI:10.1016/j.jlumin.2018.10.106.
- G. R. Loppnow, D. Melamed, A. R. Leheny, A. D. Hamilton and T. G. Spiro, Excited-State Photophysics of Donor-AppendedCobalt(II) Porphyrins from Picosecond Transient Absorption-Spectroscopy, J. Phys. Chem., 1993, 97(35), 8969–8975, DOI:10.1021/j100137a022.
- L. L. Zhao, Y. Y. Zeng, S. D. Gong, C. G. Li, J. F. Xia, X. Zhou, F. F. Zhang and Z. H. Wang, Pyronin Derivatives as Efficient Electrochemiluminescence Emitters in Aqueous Solution, J. Electrochem. Soc., 2023, 170, 4, DOI:10.1149/1945-7111/acc979.
- M. Guergueb, S. Nasri, J. Brahmi, F. Loiseau, F. Molton, T. Roisnel, V. Guerineau, I. Turowska-Tyrk, K. Aouadi and H. Nasri, Effect of the coordination of π-acceptor 4-cyanopyridine ligand on the structural and electronic properties of -tetra(-methoxy) and -tetra(-chlorophenyl) porphyrin cobalt(II) coordination compound. Application in the catalytic degradation of methylene blue dye, RSC Adv., 2020, 10(12), 6900–6918, 10.1039/c9ra08504.
- K. Venkatasubbaiah, X. Zhu, E. Kays, K. I. Hardcastle and C. W. Jones, Co(III)-Porphyrin-Mediated Highly Regioselective Ring-Opening. of Terminal Epoxides with Alcohols and Phenols, ACS Catal., 2011, 1, 489–492 CrossRef CAS.
- T. Constantin, M. Zanini, A. Regni, N. S. Sheikh, F. Juliá and D. Leonori, Aminoalkyl radicals as halogen-atom transfer agents for activation of alkyl and aryl halides, Science, 2020, 367(6481), 1021–1026, DOI:10.1126/science.aba2419.
- X. Sun and K. Zheng, Electrochemical halogen-atom transfer alkylation via α-aminoalkyl radical activation of alkyl iodides, Nat. Commun., 2023, 14, 6825 CrossRef CAS.
- J. Wu, R. Purushothaman, F. Kallert, S. Homolle and L. Ackermann, Electrochemical Glycosylation via Halogen-Atom-Transfer for C-Glycoside Assembly, ACS Catal., 2024, 14(15), 11532–11544 Search PubMed.
- X. M. Zhang, S. R. Yeh, S. Hong, M. Freccero, A. Albini, D. E. Falvey and P. S. Mariano, Dynamics of Alpha-Ch Deprotonation and Alpha-Desilylation Reactions of Tertiary Amine Cation Radicals, J. Am. Chem. Soc., 1994, 116(10), 4211–4220, DOI:10.1021/ja00089a010.
- M. Kojima and S. Matsunaga, The Merger of Photoredox and Cobalt Catalysis, Trends Chem., 2020, 2(5), 410–426, DOI:10.1016/j.trechm.2020.01.004.
- A. Pai, S. De and A. Thakur, Cobalt-based Photocatalysis: From Fundamental Principles to Applications in the Generation of C–X (X=C, O, N, H, Si) Bond, Chem.–Eur. J., 2025, 31, e202403667 CrossRef PubMed.
- H. M. Guo, B. Q. He and X. S. Wu, Direct Photoexcitation of Xanthate Anions for Deoxygenative Alkenylation of Alcohols, Org. Lett., 2022, 24(17), 3199–3204, DOI:10.1021/acs.orglett.2c00889.
| This journal is © The Royal Society of Chemistry 2026 |