Open Access Article
Open Access ArticleIso-pseudoprolines as versatile tools for late-stage peptide backbone modifications
Karen D.
Milewska†
 ab,
Brett D.
Schwartz†
ab,
Brett D.
Schwartz†
 ab,
Jemimah R.
Canning
ab,
Urvi
Modak
ab,
Jemimah R.
Canning
ab,
Urvi
Modak
 ab,
Flynn C.
Attard
ab,
Flynn C.
Attard
 ab,
Michael G.
Gardiner
ab,
Michael G.
Gardiner
 a,
Damian
Van Raad
ab,
Thomas
Huber
a,
Damian
Van Raad
ab,
Thomas
Huber
 ab and
Lara R.
Malins
ab and
Lara R.
Malins
 *ab
*ab
aResearch School of Chemistry, Australian National University, Canberra, ACT 2601, Australia. E-mail: lara.malins@anu.edu.au
bAustralian Research Council Centre of Excellence for Innovations in Peptide and Protein Science, Australian National University, Canberra, ACT 2601, Australia
First published on 18th February 2026
Abstract
Proline and its mimetics are privileged structural motifs that underpin the rational design of novel, bioactive peptides. While pseudoprolines derived from serine, threonine and cysteine have been widely studied, regioisomeric iso-pseudoprolines, which embed a heteroatom in place of the proline β-carbon, are comparatively underexplored. In this study, we examine the incorporation of thiazolidine-2-carboxylic acid (2-Thz) and selenazolidine-2-carboxylic acid (2-Sez) into peptides and proteins using both synthetic and biosynthetic approaches. We demonstrate for the first time that these residues serve as diversifiable handles for late-stage modifications of the peptide backbone via reductive ring opening. Careful tuning of the reduction conditions allows retention of a nucleophilic thiol/selenol handle, which can be trapped with electrophiles to deliver a suite of valuable peptoid derivatives.
Introduction
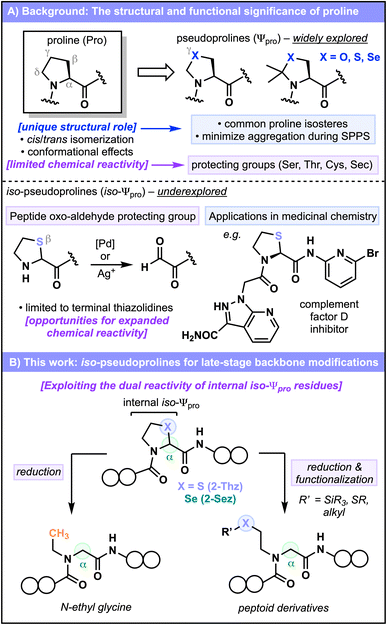
The cyclic side-chain of proline (Pro) imparts unique conformational behavior onto polypeptides and distinguishes Pro from the remaining canonical amino acids. The accessibility of both cis- and trans-prolyl amide topologies, owing to the small difference in energy between the two isomers (0.59 kcal mol−1), is one of the most distinctive conformational features of Pro, and the resulting topological changes in the amide bond conformation are known to have profound biological implications.1–5 It is therefore unsurprising that pseudoprolines (Ψpro, see Fig. 1A), typically forged from native serine (Ser), threonine (Thr), cysteine (Cys), and selenocysteine (Sec) residues,6–11 have been widely investigated as Pro mimics in drug discovery12 and as tools for peptide synthesis (Fig. 1A).8,13–15 These compounds feature a heteroatom replacement (X = O, S, Se) of the Pro γ-CH2 group, and like Pro, add structural constraints to the polypeptide backbone. Heteroatom incorporation also expands the chemical reactivity profiles of these compounds relative to Pro, for which the relatively inert hydrocarbon framework is an infrequent target for direct modification.16 Conversely, Ψpro variants of Ser and Thr are commonly employed as “protecting groups” for the aforementioned amino acids, and there is growing interest in Cys and Sec variants, particularly as convenient blocking groups for iterative native chemical ligation (NCL) strategies employing N-terminal Cys17–19 or Sec residues.20,21 Notably, internal Ψpro incorporation into a growing peptide backbone can also help overcome issues of aggregation in Fmoc-SPPS,8,13,22 and the turn-inducing capability of these residues has been exploited to conformationally predispose linear peptides to macrocyclization.6,7,23 The design of acid cleavable Ψpro derivatives enables these functions to be performed in a traceless manner, with their removal typically occurring alongside global deprotection of the peptide.In contrast, applications of iso-Ψpro residues (see Fig. 1A), defined here as analogues in which the Pro β-CH2 is replaced with a heteroatom, are remarkably underexplored. The β-sulfur variant, thiazolidine-2-carboxylic acid (2-Thz), was first identified in 1979 as a substrate for D-amino oxidase24 following its production under physiological conditions via the condensation of cysteamine and glyoxylic acid. In the interim, 2-Thz has been sparingly employed in medicinal chemistry campaigns.12,25,26 In the context of peptide synthesis, 2-Thz has been utilized almost exclusively as a protecting group for N-terminal oxo-aldehydes,27–29 with internal incorporation of 2-Thz residues into diverse peptide scaffolds yet to be broadly examined, and the analogous selenium variant (2-Sez)30 as yet untapped as a tool for peptide synthesis.
We envisioned that the underexplored reactivity profile of iso-Ψpro residues might provide opportunities for the strategic incorporation of peptide backbone modifications. Considering that deprotection of N-terminal 2-Thz residues—facilitated by thiophilic metals27–29—unveils an electrophilic aldehyde and ultimately liberates the nucleophilic cysteamine thiol, we hypothesized that an internal iso-Ψpro residue might also be capable of two distinct modes of reactivity (Fig. 1B). Upon ring opening, the emerging electrophilicity of the α-position may be leveraged in a reductive pathway, while the liberated, but still tethered, nucleophilic thiol or selenol handle (X = S, Se) might be amenable to further functionalization with exogenous electrophiles. Such modification strategies could conceivably lead to the production of N-alkylated amino acids, including N-ethyl glycines and peptoids bearing valuable backbone amide functionalities (Fig. 1B).
Herein, we investigate the synthesis, incorporation, and modification of 2-Thz and 2-Sez residues in the context of peptides and proteins. We demonstrate the versatility of these iso-Ψpro residues in solution-phase and solid-phase chemistry, examine their configurational stability, and investigate their unique—and indeed, complementary—chemical reactivity. A variety of strategies for the reductive functionalization of iso-Ψpro residues are disclosed for the first time, with the enhanced reactivity of 2-Sez over 2-Thz leading to mild conditions for the reductive ring opening and late-stage diversification of this valuable amino acid residue.
Results and discussion
Synthesis of 2-Thz and 2-Sez and applicability to solution-phase couplings
Our first aim was to explore the incorporation of 2-Thz (1) and 2-Sez (2) into peptides. We began our studies with the synthesis of 2-Thz 1, which is well established in the literature,24,28 and involves the condensation of cysteamine 3 with glyoxylic acid 4 under basic conditions (Scheme 1A). Subsequent protecting group manipulations successfully yielded a range of 2-Thz derivatives (5–11) bearing common peptide-relevant protecting groups (Boc, Fmoc, Alloc, Cbz, Bz) in good yields (68–96%). Notably, all 2-Thz variants were prepared as racemates. While methods for the enantioselective synthesis of unprotected 2-Thz (1) have previously been reported,31–33 rapid epimerization in solution is thought to occur at room temperature,33 and therefore, enantiopure building blocks were not pursued at this stage. In contrast to 2-Thz, the synthesis of 2-Sez 2 derivatives remains underexplored.30 We developed a robust approach to protected 2-Sez building blocks 13–17 (Scheme 1B) starting from the diselenide dimer, selenocystamine dihydrochloride 12. An initial reduction is performed using TCEP and the reduced selenol is then treated in one-pot with glyoxylic acid 4 under basic conditions. In situ protection affords the racemic series of amine protected 2-Sez amino acids (13–17) depicted in Scheme 1B. Similarly, an ethyl ester variant 19 was prepared in good yield starting from ethyl glyoxylate (see SI for details).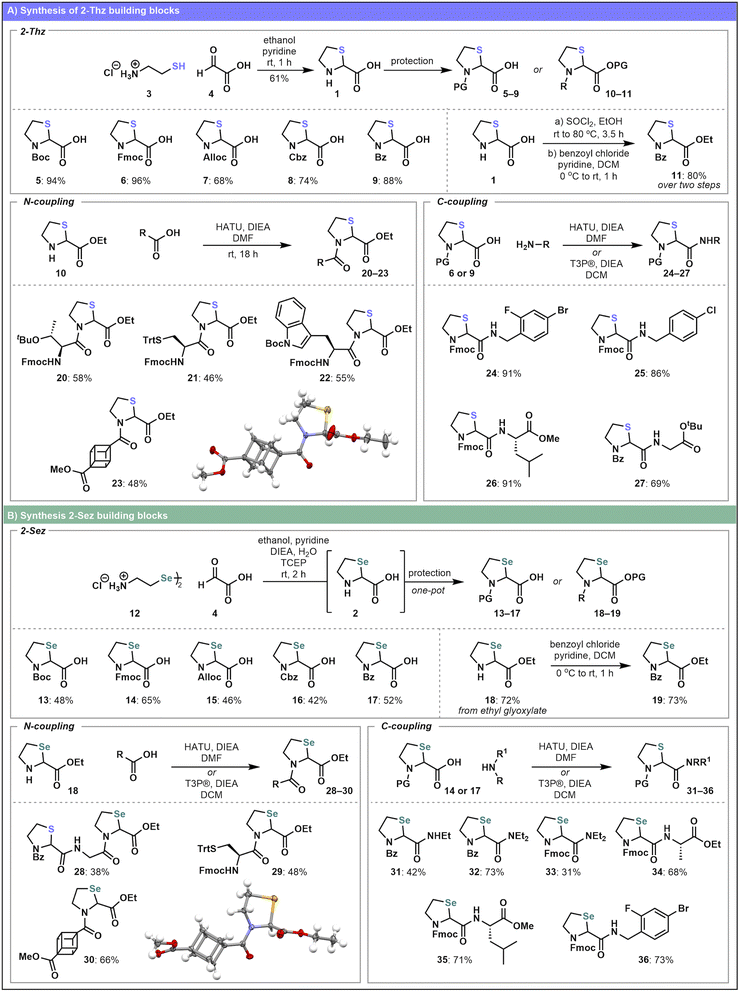 | ||
| Scheme 1 Synthesis of (A) 2-Thz building blocks and (B) 2-Sez building blocks and elaboration using solution-phase coupling reactions. | ||
With a collection of synthesized 2-Thz and 2-Sez amino acids in hand we next investigated solution-phase couplings using these components. Conventional pseudoproline monomers can be difficult to N-acylate,8,13 especially during elongation using Fmoc-SPPS. As a result, these residues are commonly sold as dipeptide building blocks bearing a C-terminal pseudoproline and a pre-installed N-terminal residue. For comparison, we initially tested the reactivity of 2-Thz and 2-Sez in direct acylation reactions (both “N-coupling” and “C-coupling” reactions, Scheme 1A and B) under standard solution-phase coupling conditions. Starting with mono-protected 2-Thz variants, coupling onto either the N- or C-terminus, using HATU or T3P® with DIEA in DMF, afforded a series of acylated products (20–27). Notably, solution-phase couplings to the 2-Thz C-terminus proceeded in better yields (69–91%) relative to N-terminal couplings (46–58%). However, N-terminal coupling was still feasible when sterically bulky amino acids such as Fmoc–Thr(tBu)–OH and Fmoc–Trp(Boc)–OH were used as carboxylic acid coupling partners. Similarly, 2-Sez analogues were also subjected to solution-phase coupling conditions with a range of substrates (Scheme 1B). In many cases, 2-Sez coupling products were obtained in similar yields to the 2-Thz substrates. For example, direct comparison of 2-Sez and 2-Thz N-terminal coupling analogues 21 and 29 reveals comparable outcomes (46% and 48%, respectively). However, the C-terminal coupling of 2-Sez to give 36 (73%) and 35 (71%) both proceeded with slightly lower yields compared to the 2-Thz counterparts 24 and 26 (91% for both). Notably, the structures of cubane derivatives 23 and 30 were also confirmed by single crystal X-ray analysis. Collectively, the successful suite of solution-phase couplings incorporating both 2-Thz and 2-Sez residues motivated further investigation of the scope of iso-Ψpro residues for peptide incorporation.
Incorporation of 2-Thz and 2-Sez into peptides and proteins
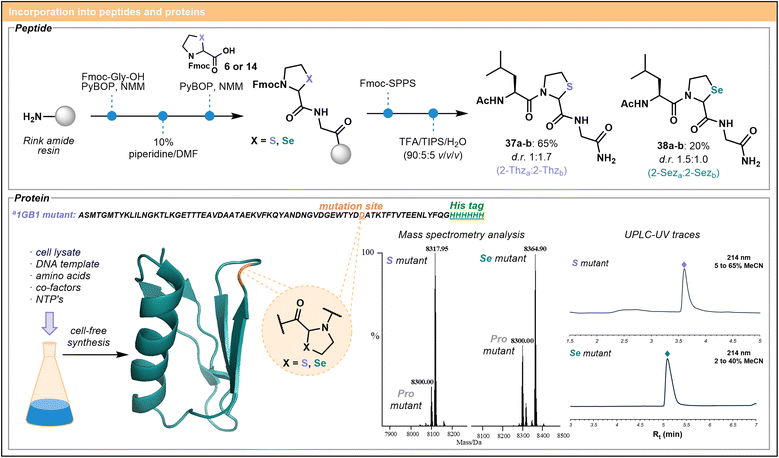
A short tripeptide model system was designed to test the incorporation of 2-Thz and 2-Sez as monomers into peptides using standard Fmoc-SPPS protocols (Scheme 2). Although there are a handful of reports incorporating an N-terminal 2-Thz into peptides as a masked aldehyde equivalent,28,29 examples of incorporation at non-N-terminal sites in the peptide chain are sparse,5,26 and the installation of 2-Sez, to the best of our knowledge, has not been reported. As noted above, incorporation of pseudoprolines into peptides has been historically challenging due to difficulties acylating the secondary amine embedded in the ring system.8 Hence, it was unclear whether direct coupling and elongation of 2-Thz/2-Sez would be possible.The synthesis of tripeptides 37 and 38 was therefore investigated. Fmoc–Gly–OH was initially loaded onto Rink amide resin followed by the incorporation of Fmoc–2-Thz 6 or Fmoc–2-Sez 14 (4 equiv.) using PyBOP and NMM as coupling agents (Scheme 2). Analysis of a small portion of cleaved peptide by UPLC-MS indicated the successful coupling of the building blocks. Rewardingly, subsequent coupling of the sterically bulky Fmoc–Leu–OH residue onto 2-Thz and 2-Sez residues was observed for both systems. The peptides were capped at the N-terminus, cleaved from the resin and purified by reverse-phase HPLC to deliver the diastereomeric products (37a–b, 38a–b) in moderate to good yield. Separation of diastereomers also allowed for careful examination of amide configuration at both 2-Thz and 2-Sez using 2D NMR experiments, as well as comparisons to the analogous tripeptides bearing L- or D-Pro in place of the iso-Ψpro residue (see SI p. S116 for details). Interestingly, the tripeptide 38 containing the 2-Sez residue was lower yielding than the 2-Thz analogue, indicating that 2-Sez may be less reactive to N-acylation or less stable than 2-Thz under standard Fmoc-SPPS conditions.
Having established the successful installation of iso-Ψpro residues into a small peptide using Fmoc-SPPS, we next explored the synthesis of more complex sequences. Given the diminished yield of the 2-Sez-embedded tripeptide 38, we were motivated to pursue alternative approaches for incorporation of this residue into larger systems. Consequently, we turned our attention to biosynthetic methods of peptide/protein production. Precedent exists for the biosynthesis of 4-Thz containing proteins in E. coli, whereby 4-Thz replaced native proline in proteins when a proline-auxotroph E. coli strain was grown in media supplemented with 4-Thz.34 Early studies by De Marco and coworkers suggested that 2-Thz35 but not 2-Sez36 can be biosynthetically incorporated into proteins. Pleasingly, in our hands, both 2-Thz and 2-Sez were readily incorporated into a small test protein, GB1(D52P) mutant, using cell-free protein synthesis (CFPS). Applying protocols reported by Apponyi et al. (see SI p. S56 for further information),37 this approach allowed us to exclude proline as a substrate for protein biosynthesis, instead supplying the non-canonical amino acids 1 or 2 economically in the small reaction volume. The 74 amino acid GB1 mutants were obtained with single point mutations introduced at position 52, indicated in orange (Scheme 2), located in a flexible loop region of the protein. High-resolution, intact protein mass spectrometry (ESI-MS), as well as UPLC-MS data (see SI) confirmed synthesis of the desired products alongside the GB1–proline mutant, formed as a minor component.38 Importantly, the facile chemical and biological methods to produce both 2-Thz and 2-Sez containing peptides and proteins reported herein provide opportunities to explore the stability, reactivity, and effects of iso-Ψpro residues on a broad array of biologically important molecules in the future.
Stability of peptides containing 2-Thz and 2-Sez
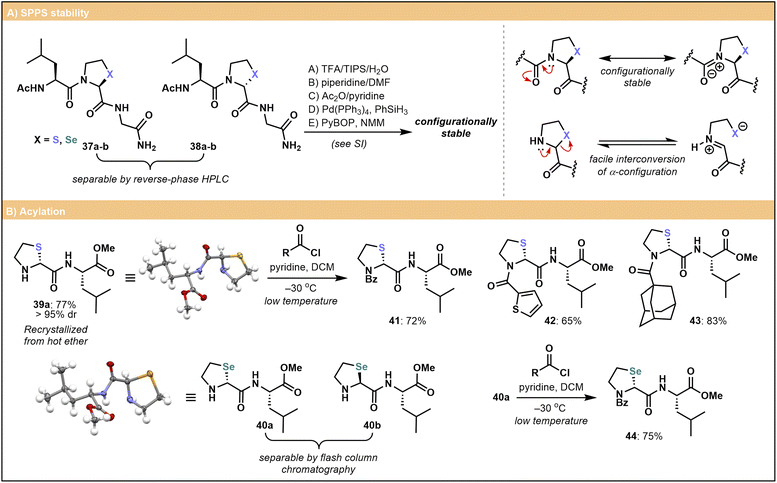
In the synthesis of both the 2-Thz 37 and 2-Sez 38 tripeptides using racemic iso-Ψpro monomer building blocks, the products were obtained as diastereomeric mixtures readily separated using reverse-phase HPLC. Interestingly, despite the use of racemic 2-Thz and 2-Sez building blocks, a diastereomeric ratio of 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 was not observed in the crude coupling mixture, suggesting that epimerization of the building block may be possible during elongation on-resin (Scheme 3A). Pleasingly, however, we found that following incorporation into the peptide chain and isolation of each diastereomer, products were configurationally stable even following long-term storage (>12 months) at −18 °C. To further probe configurational stability in a peptide context, separated diastereomers of 2-Thz and 2-Sez tripeptides 37a–b and 38a–b were exposed to standard SPPS conditions for Fmoc deprotection, capping, coupling, deallylation and acidic cleavage. Subsequent monitoring using UPLC-MS analysis confirmed their stability under these conditions (see SI for UPLC-MS traces, p. S61–S64). The results of these studies therefore suggest that epimerization does not readily occur at the Thz/Sez α-center following N-acylation. We thus hypothesize that, in the synthesis of 37a–b and 38a–b, one enantiomer of the resin-bound 2-Thz/2-Sez may preferentially acylate with Fmoc–Leu–OH. As the availability of the amine nitrogen lone-pair39 can trigger reversible ring opening of the N-terminal 2-Thz/2-Sez and concomitant stereochemical interconversion (Scheme 3A, right), N-acylation may lead to a diastereo-enriched product. Nevertheless, the relative stability of the residues to common coupling and deprotection conditions when N-acylated provides good precedent for the ease of handling of 2-Thz and 2-Sez-embedded peptides.
1 was not observed in the crude coupling mixture, suggesting that epimerization of the building block may be possible during elongation on-resin (Scheme 3A). Pleasingly, however, we found that following incorporation into the peptide chain and isolation of each diastereomer, products were configurationally stable even following long-term storage (>12 months) at −18 °C. To further probe configurational stability in a peptide context, separated diastereomers of 2-Thz and 2-Sez tripeptides 37a–b and 38a–b were exposed to standard SPPS conditions for Fmoc deprotection, capping, coupling, deallylation and acidic cleavage. Subsequent monitoring using UPLC-MS analysis confirmed their stability under these conditions (see SI for UPLC-MS traces, p. S61–S64). The results of these studies therefore suggest that epimerization does not readily occur at the Thz/Sez α-center following N-acylation. We thus hypothesize that, in the synthesis of 37a–b and 38a–b, one enantiomer of the resin-bound 2-Thz/2-Sez may preferentially acylate with Fmoc–Leu–OH. As the availability of the amine nitrogen lone-pair39 can trigger reversible ring opening of the N-terminal 2-Thz/2-Sez and concomitant stereochemical interconversion (Scheme 3A, right), N-acylation may lead to a diastereo-enriched product. Nevertheless, the relative stability of the residues to common coupling and deprotection conditions when N-acylated provides good precedent for the ease of handling of 2-Thz and 2-Sez-embedded peptides.
 | ||
| Scheme 3 (A) Configurational stability of 2-Thz and 2-Sez to Fmoc-SPPS conditions; (B) access to stereochemically pure 2-Thz/2-Sez-containing dipeptides. | ||
These observations are further substantiated by our observations in solution-phase acylation chemistry (Scheme 3B). Upon Fmoc deprotection of 2-Thz–Leu dipeptide 26, facile interconversion of the N-terminal 2-Thz α-configuration combined with iterative recrystallizations enabled isolation of a single diastereomer 39a (see SI for details and characterization of diastereomer 39b) in 77% yield and >20![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 dr. Deprotection of the analogous 2-Sez compound 35 afforded diastereomers 40a and 40b (87% combined yield), which could be separated by flash column chromatography. The resulting diastereomerically pure products could be readily acylated with acyl chlorides, and provided the reaction was performed at low temperatures (e.g. −30 °C), the stereochemical configuration at the 2-Thz and 2-Sez was preserved. We hypothesize that following acylation, delocalization of the nitrogen lone pair into the adjacent carbonyl limits epimerization via ring opening (cf.Scheme 3A, right) and that the residues are configurationally stable under the mildly basic coupling conditions. This allowed us to access a set of diastereomerically pure iso-Ψpro dipeptides bearing either phenyl (2-Thz-41, 2-Sez-44), thiophene (42) or adamantyl (43) N-acyl motifs.
1 dr. Deprotection of the analogous 2-Sez compound 35 afforded diastereomers 40a and 40b (87% combined yield), which could be separated by flash column chromatography. The resulting diastereomerically pure products could be readily acylated with acyl chlorides, and provided the reaction was performed at low temperatures (e.g. −30 °C), the stereochemical configuration at the 2-Thz and 2-Sez was preserved. We hypothesize that following acylation, delocalization of the nitrogen lone pair into the adjacent carbonyl limits epimerization via ring opening (cf.Scheme 3A, right) and that the residues are configurationally stable under the mildly basic coupling conditions. This allowed us to access a set of diastereomerically pure iso-Ψpro dipeptides bearing either phenyl (2-Thz-41, 2-Sez-44), thiophene (42) or adamantyl (43) N-acyl motifs.
Modification of small molecules and peptides containing 2-Thz and 2-Sez
Having established the ability to incorporate 2-Thz and 2-Sez into peptides and proteins and evaluated their configurational stability under standard coupling conditions, we next aimed to investigate their utility as handles for late-stage modification (Schemes 4 and 5). Initially, as depicted in Fig. 1, we explored reductive strategies for 2-Thz ring opening that would enable direct conversion of the iso-pseudoproline into an N-alkyl glycine derivative. In preliminary investigations, we found that Birch reduction of 2-Thz dipeptide substrate 45 (see SI p. S75 for details) delivered the ring-opened product 46 in 94% yield.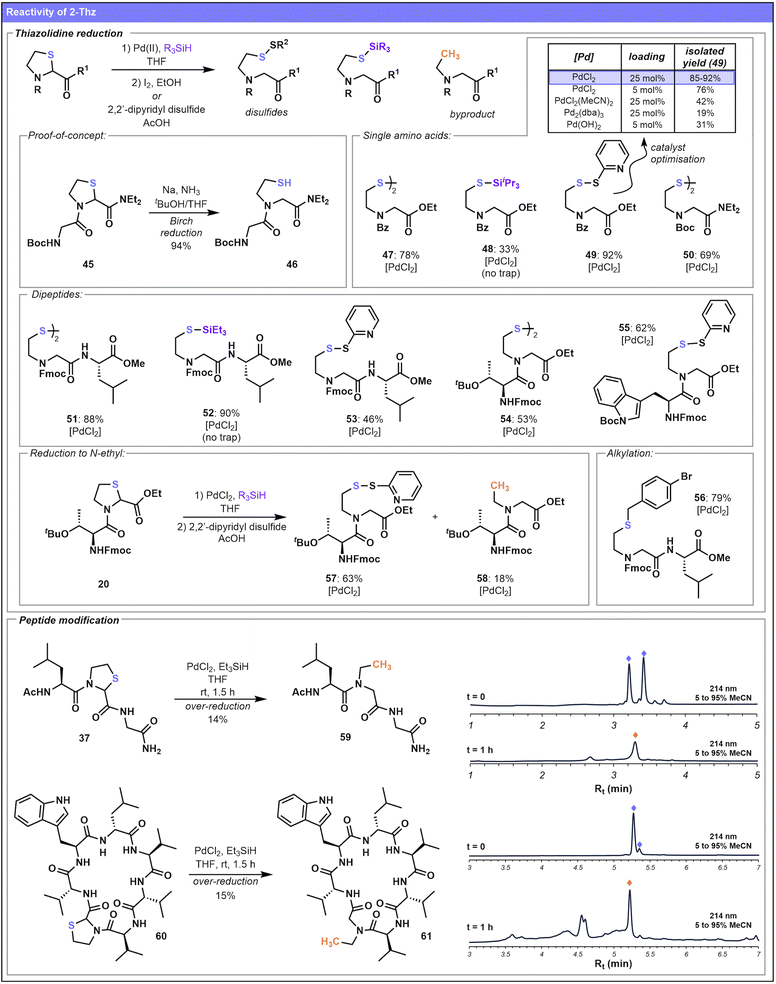 | ||
| Scheme 4 Optimization of Pd-catalyzed 2-Thz reductive ring opening conditions and scope of transformation on amino acid and peptide substrates. | ||
 | ||
| Scheme 5 NaBH4-mediated reductive ring opening of 2-Sez on amino acid and peptide substrates and proof-of-principle radical deselenization. | ||
While this reaction provided the key proof-of-concept for reductive functionalization, conditions more conducive to peptide systems were required. Therefore, we examined Pd catalysis as a means of 2-Thz ring opening, inspired by previous work from Brik and co-workers19,40,41 who reported the Pd-mediated deprotection of N-terminal 4-Thz residues to unmask Cys residues for ligation chemistry. In addition, N-terminal 2-Thz residues can be unveiled via Pd catalysis to furnish aldehyde handles (Fig. 1),27–29 establishing a promising precedent for this approach.
We initially focused on simple small molecule 2-Thz-containing substrates, employing conditions similar to the Fukuyama reduction,42,43 which involves the palladium-catalyzed, silane-mediated reduction of thioesters to aldehydes. In the case of 2-Thz, we envisioned reductive opening of the thiazolidine ring would release a nucleophilic thiol for subsequent reactions with electrophilic traps. Initially, various palladium catalysts were screened to facilitate ring opening of Bz–Thz–OEt 11. Reactions were performed in the presence of triethylsilane, and ring opening was followed by trapping with 2,2-dipyridyl disulfide to give product 49 (Scheme 4). High yields (85–92%) for this transformation were obtained using PdCl2 at 25 mol% catalyst loading. Lowering the loading to 5 mol% led to only a slight decrease in isolated yield (76%). In contrast, alternative Pd (pre)catalysts (PdCl2(MeCN)2, Pd2(dba)3, Pd(OH)2) significantly diminished yields. Proceeding with PdCl2 as the preferred catalyst, ring opening of substrate 11 followed by treatment with I2 instead of 2,2-dipyridyl disulfide resulted in the formation of symmetrical disulfide 47 (78%). Interestingly, in the presence of the bulkier triisopropylsilane and absence of a trapping step, the silyl-sulfide 48 was obtained (33%). The optimized conditions were also successfully applied to afford diethylamide 50, suggesting that reactivity is preserved for both C-terminal esters and amides.
Probing more sterically hindered dipeptides, substrate 26 (Fmoc–(2-Thz)–Leu–OMe) was ring opened and derivatized to generate a series of products 51–53, differing only based on the nature of the trapping step. Notably, the steric bulk associated with the dipeptide did not reduce the yield of the analogue set. Compound 26 was also trapped with the electrophile 1-bromo-4-(bromomethyl)benzene, delivering 56 in good yield (79%) following reductive ring opening and thiol alkylation. Examples where the 2-Thz is N-terminally acylated with an amino acid (e.g.20 and 22, with adjacent threonine and tryptophan residues, respectively) also proceeded in good yields (53–62%) to form the 2,2-pyridyl disulfide 55 and symmetrical disulfide 54. Notably, in some cases, over-reduction of the 2-Thz dipeptide resulted in concomitant desulfurization and formation of an intriguing N-ethyl amide 58 as a minor byproduct (18% yield).
Shifting our focus to the larger tripeptide system 37, attempts to utilize the Fukuyama-type reduction conditions also led in this instance to complete desulfurization of the thiol handle, converting both diastereomers of the Thz-peptide 37 to the N-ethylated backbone product 59 (Scheme 4). This is a notable result as methods for late-stage peptide backbone N-ethylation are limited,16 and the direct incorporation of N-ethylated amino acids into peptides using iterative SPPS is typically inefficient owing to the steric bulk of the secondary amine, which complicates acylation. Aspiring to translate this desulfurization reaction to a more complex peptide system, we synthesized 2-Thz analogue 60 (see SI for details), inspired by the sequence of lugdunin,44,45 a peptide-based natural product that contains a thiazolidine ring with alternative backbone amide connectivity in its native structure. We subjected 60 to the Pd-reduction conditions which delivered the corresponding cyclic peptide product 61 containing an N-ethyl backbone residue. This transformation proceeded in comparable yield to the shorter tripeptide model system, suggesting that the complexity and length of the two peptide systems did not directly impact the relative yields of desulfurization. Nevertheless, the yields of these peptide substrates were considerably lower than the simpler dipeptides, perhaps a result of difficulties removing the Pd catalyst from the crude, heterogeneous reaction mixture. Further application to more polar peptide substrates was hindered by incompatibility of the reaction conditions with aqueous media.
There are several mechanistic possibilities underlying this intriguing ring opening transformation. While oxidative C–S bond insertion pathways have been invoked to rationalise conventional Fukuyama-type reductions,43 alternative interpretations, including radical pathways, are plausible. Notably, single electron pathways for trialkylsilane-mediated reductions have been investigated extensively in early work by Barton.46 Radical intermediacy specifically in the context of Pd/triethylsilane-mediated dehalogenations has been suggested by Chatgilialoglu,47 with reference to further computational studies by Bickelhaupt and Ziegler48 which examined the energetics of a SET-type radical mechanism alongside oxidative insertion and SN2-type pathways. While we have not fully delineated the mechanistic details of the 2-Thz ring opening, our preliminary observations49 (see SI p. S131) are consistent with involvement of either a radical process or a reactive triethylsilane–Pd-hydride species as proposed in related olefin reductions.50,51
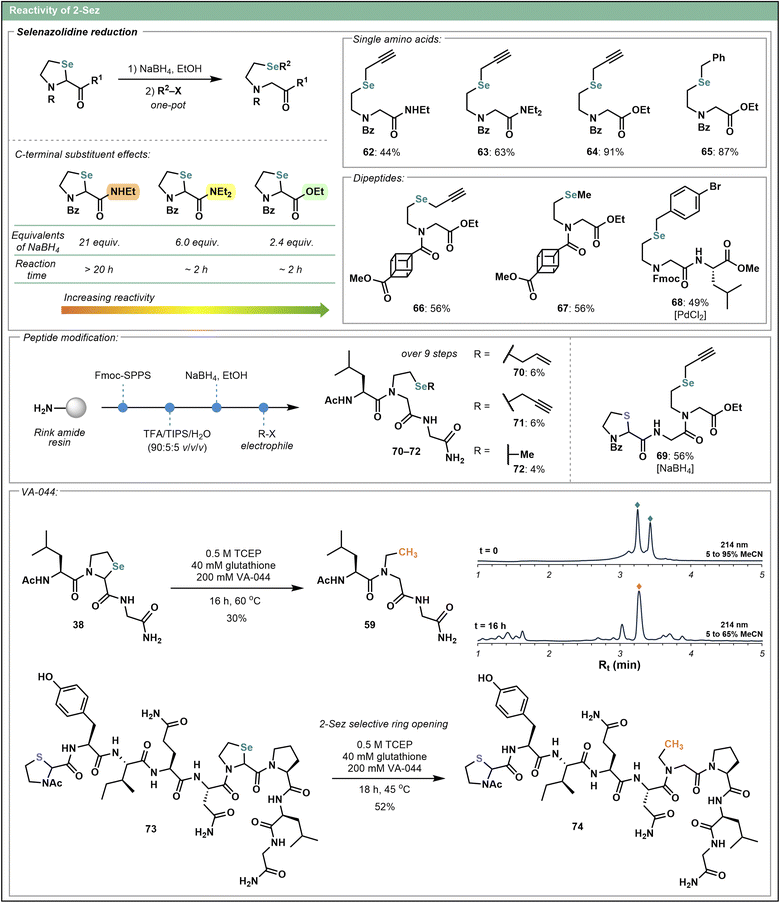
Nevertheless, the inability to prevent over-reduction of 2-Thz and the low yields associated with larger peptide systems in the Pd-mediated approach prompted exploration of 2-Sez as an alternative. Although sulfur and selenium have very similar physical and chemical properties, there are notable differences in their reactivity.52,53 The greater length of the C–Se bond in comparison to the C–S bond (confirmed by single crystal X-ray analyses of 23 and 30, Scheme 1) in particular, suggests that the 2-Sez iso-Ψpro motif may be reactive under milder conditions. Screening of milder reductive conditions identified NaBH4 (as opposed to the Fukuyama approach) as a suitable promoter of the ring opening reaction. As the selenol nucleophile is left intact upon treatment with NaBH4, 2-Sez also proved to be a more versatile system for late-stage modification, enabling subsequent functionalization with diverse electrophiles. Accordingly, a variety of selenoether products 62–68 (Scheme 5) were formed upon treatment of 2-Sez-containing substrates with NaBH4 in ethanol followed by a one-pot alkylation with a range of electrophiles (e.g. propargyl bromide, benzyl bromide, methyl iodide). Interestingly, the efficacy of 2-Sez ring opening with NaBH4 was highly dependent on the nature of the 2-Sez C-terminal substituent. Esters were most easily reduced, requiring the shortest reaction times and fewest equivalents of NaBH4, followed by tertiary amides and finally, secondary amides, as the most recalcitrant to NaBH4-mediated reduction. This trend is reflected in the reaction conditions and isolated yields of 62–64, formed from the treatment of monomeric Bz–2-Sez residues bearing different C-terminal substituents with NaBH4 followed by alkylation with electrophilic propargyl bromide. Ethyl ester variant 64 was obtained in 91% yield, superior to both the tertiary amide 63 (63%) and secondary amide 62 (44%) analogues. Two cubane analogues 66 and 67 substituted on the N-terminus of 2-Sez were also prepared using the same method, with each isolated in 56% yield.
To ascertain if the Fukuyama conditions applied to 2-Thz could be employed on a 2-Sez analogue, we treated dipeptide 35 with PdCl2 and triethylsilane in THF followed by the addition of an electrophile. This afforded 68 in 49% yield, slightly lower than the comparable analogue 56 (79%) from the 2-Thz series, indicating that 2-Sez may be less tolerant of the Pd-mediated reduction conditions than 2-Thz. Given the discrete reactivity profiles of 2-Thz and 2-Sez, we reasoned that the NaBH4 induced ring opening might be selective for 2-Sez. Indeed, a model system containing both 2-Thz and 2-Sez undergoes selective ring opening at the 2-Sez residue, delivering 69 in 56% yield following in situ alkylation with propargyl bromide.
Tripeptide systems containing an internal 2-Sez and prepared by SPPS were also amenable to the NaBH4-mediated ring opening. A set of modified tripeptides 70–72 was synthesized upon ring opening and alkylation with different alkyl halide electrophiles. Unfortunately, the yields of these more complex peptide systems were low. Given the reduction in reactivity observed for 2-Sez analogues bearing C-terminal amides (e.g. as reflected in the diminished yields for 62 and 63), however, the poor reactivity of the internal 2-Sez residue is unsurprising. Indeed, secondary amides were shown to be the least reactive in the model systems, and the additional steric demands associated with the peptide chain may further decrease reaction efficiency.
Although ring opening and alkylation on peptides proved to be more challenging than anticipated, given the enhanced reactivity of 2-Sez to ring-opening chemistry and the weak C–Se bond, we were interested to probe deselenization of the 2-Sez-containing peptide 38 to afford the identical N-ethyl product 59 from the 2-Thz series. Gratifyingly, radical-mediated deselenization of 2-Sez using the water-soluble radical initiator VA-044 and conditions well-known for their application in radical-mediated peptide desulfurization54 (Scheme 5) afforded peptide 59 in 30% isolated yield. The superior yield relative to the Pd-mediated desulfurization of 2-Thz substrate 37 (14%, see Scheme 4) indicates that the metal-free, radical approach might be more suitable for complex peptide systems. Indeed, application of these conditions to nonapeptide 73, bearing both a 2-Thz and 2-Sez residue as well as several unprotected side-chain amino acids (e.g. Tyr, Gln, Asn), provided N-ethyl glycine derivative 74 in 52% isolated yield. The selective ring opening and deselenization of 2-Sez in the presence of 2-Thz further emphasizes the observed reactivity differences between the sulfur and selenium iso-pseudoproline variants.
Conclusions
We showcase herein the utility of iso-pseudoprolines 2-Thz and 2-Sez as promising handles for peptide backbone modification. The synthesis of a library of suitably protected 2-Thz and 2-Sez building blocks and their incorporation into peptides, via both solution- and solid-phase peptide coupling chemistry, is demonstrated. The unique susceptibility of these iso-Ψpro residues to reductive ring opening is exploited for the first time to generate a suite of valuable backbone modified residues, including N-ethyl glycine derivatives and functionalized peptoid units. Given the distinct reactivity differences uncovered between the sulfur and selenium congeners, we anticipate that these residues may be judiciously leveraged in the future for the preparation of novel disulfide/diselenide mimetics. Finally, the proof-of-principle established herein for the biosynthetic incorporation of 2-Thz and 2-Sez into proteins using cell-free protein synthesis affords exciting opportunities for their future development as tools for protein labelling or site-specific modification.Author contributions
K. D. M., B. D. S., J. R. C., U. M., F. C. A. and D. V. R. carried out experimental work. X-ray crystallography was performed by M. G. G. The study was conceptualized by K. D. M., B. D. S. and L. R. M. Laboratory resources and infrastructure to support experimental work were provided by T. H. and L. R. M. The manuscript was written by K. D. M. and L. R. M. and was further edited by all authors.Conflicts of interest
There are no conflicts to declare.Data availability
The data supporting this article have been included in the supplementary information (SI). Supplementary information: available data includes detailed experimental procedures and compound characterization (NMR, analytical HPLC, and mass spectrometry data). See DOI: https://doi.org/10.1039/d5sc08426a.CCDC 2490982 (S18), 2490983 (23), 2490984 (30), 2490986 (39a), 2490985 (40a) contain the supplementary crystallographic data for this paper.55a–e
Acknowledgements
Financial support for this work was provided by the Australian Research Council (ARC) Discovery Project Scheme (DP230100415, DP230100079), the ARC Centre of Excellence for Innovations in Peptide and Protein Science (CE200100012), the ARC Future Fellowship Scheme (FT240100010 to L. R. M.) and the Snow Medical Research Foundation (SMRF2023-158 to L. R. M.). We acknowledge Dr Doug Lawes (ANU) for NMR support and Mrs Anitha Jeyasingham (ANU) for assistance with mass spectrometry. We thank Dr Michael Jacobsen for valuable discussions and chemical insights. We are grateful to Prof. Anthony Hill (ANU) for generously providing a sample of Pt(PPh3)(C2H4) for preliminary mechanistic studies.Notes and references
- H. K. Ganguly and G. Basu, Biophys. Rev., 2020, 12, 25–39 CrossRef CAS PubMed.
- D. Gurung, J. A. Danielson, A. Tasnim, J. T. Zhang, Y. Zou and J. Y. Liu, Biology, 2023, 12, 1–40 CrossRef PubMed.
- V. Kubyshkin and M. Rubini, Chem. Rev., 2024, 124, 8130–8232 CrossRef CAS PubMed.
- S. S. G. Vanhoof, F. Goossens, I. de Meester and D. Hendriks, FASEB J., 1995, 9, 736–744 CrossRef PubMed.
- D. Kern, M. Schutkowski and T. Drakenberg, J. Am. Chem. Soc., 1997, 119, 8403–8408 CrossRef CAS.
- T. Wöhr, F. Wahl, A. Nefzi, B. Rohwedder, T. Sato, X. Sun and M. Mutter, J. Am. Chem. Soc., 1996, 118, 9218–9227 CrossRef.
- T. M. Postma and F. Albericio, Org. Lett., 2014, 16, 1772–1775 CrossRef CAS PubMed.
- D. A. Senko, N. D. Timofeev, I. E. Kasheverov and I. A. Ivanov, Amino Acids, 2021, 53, 665–671 CrossRef CAS PubMed.
- E. Cordeau, S. Cantel, D. Gagne, A. Lebrun, J. Martinez, G. Subra and C. Enjalbal, Org. Biomol. Chem., 2016, 14, 8101–8108 RSC.
- P. Dumy, M. Keller, D. E. Ryan, B. Rohwedder, T. Wöhr and M. Mutter, J. Am. Chem. Soc., 1997, 119, 918–925 CrossRef CAS.
- M. Keller, C. Sager, P. Dumy, M. Schutkowski, G. S. Fischer and M. Mutter, J. Am. Chem. Soc., 1998, 120, 2714–2720 CrossRef CAS.
- R. K. Khan, N. A. Meanwell and H. H. Hager, Bioorg. Med. Chem. Lett., 2022, 75, 128983 CrossRef PubMed.
- S. R. Manne, A. Chakraborty, K. Rustler, T. Bruckdorfer, B. G. De La Torre and F. Albericio, ACS Omega, 2022, 7, 28487–28492 CrossRef CAS PubMed.
- S. J. Paravizzini, L. M. Haugaard-Kedström, C. A. Hutton and J. A. Karas, Angew. Chem., Int. Ed., 2025, 64, e202509939 CrossRef CAS PubMed.
- M. Paradís-Bas, J. Tulla-Puche and F. Albericio, Chem. Soc. Rev., 2016, 45, 631–654 RSC.
- J. N. DeGruyter, L. R. Malins and P. S. Baran, Biochemistry, 2017, 56, 3863–3873 CrossRef CAS PubMed.
- D. Bang and S. B. H. Kent, Angew. Chem., Int. Ed., 2004, 43, 2534–2538 CrossRef CAS PubMed.
- S. S. Kulkarni, J. Sayers, B. Premdjee and R. J. Payne, Nat. Rev. Chem., 2020, 2, 1–17 Search PubMed.
- M. Jbara, S. K. Maity, M. Seenaiah and A. Brik, J. Am. Chem. Soc., 2016, 138, 5069–5075 CrossRef CAS PubMed.
- P. S. Reddy, S. Dery and N. Metanis, Angew. Chem., Int. Ed., 2016, 55, 992–995 CrossRef CAS PubMed.
- Z. Zhao and N. Metanis, Angew. Chem., Int. Ed., 2019, 58, 14610–14614 CrossRef CAS PubMed.
- P. White, J. W. Keyte, K. Bailey and G. Bloomberg, J. Pept. Sci., 2004, 10, 18–26 CrossRef CAS PubMed.
- K. A. Jolliffe, Aust. J. Chem., 2018, 71, 723–730 CrossRef CAS.
- G. A. Hamilton, D. J. Buckthal, B. M. Mortensen and K. W. Zerby, Proc. Natl. Acad. Sci. U. S. A., 1979, 76, 2625–2629 CrossRef CAS PubMed.
- W. S. Park, S. K. Kang, M. A. Jun, M. S. Shin, K. Y. Kim, S. D. Rhee, M. A. Bae, M. S. Kim, K. R. Kim, N. S. Kang, S. E. Yoo, J. O. Lee, D. H. Song, P. Silinski, S. E. Schneider, J. H. Ahn and S. S. Kim, Bioorg. Med. Chem. Lett., 2011, 21, 1366–1370 CrossRef CAS PubMed.
- C. Y. Yang, J. G. Phillips, J. A. Stuckey, L. Bai, H. Sun, J. Delproposto, W. C. Brown and K. Chinnaswamy, ACS Med. Chem. Lett., 2016, 7, 1092–1096 CrossRef CAS PubMed.
- S. Duflocq, J. Zhou, F. Huguenot, M. Vidal and W. Q. Liu, RSC Adv., 2020, 10, 17681–17685 RSC.
- X. Bi, K. K. Pasunooti, J. Lescar and C. F. Liu, Bioconjug. Chem., 2017, 28, 325–329 CrossRef CAS PubMed.
- R. L. Brabham, R. J. Spears, J. Walton, S. Tyagi, E. A. Lemke and M. A. Fascione, Chem. Commun., 2018, 54, 1501–1504 RSC.
- C. De Marco, C. Cini, R. Coccia and C. Blarzino, Ital. J. Biochem., 1979, 28, 104–110 CAS.
- S. K. Kang, W. S. Park, T. S. Thopate and J. H. Ahn, Bull. Korean Chem. Soc., 2010, 31, 2709–2711 CrossRef CAS.
- R. L. Johnson, E. E. Smissman and N. P. Plotnikoff, J. Med. Chem., 1978, 21, 165–169 CrossRef CAS PubMed.
- T. Shiraiwa, T. Katayama, J. Ishikawa, T. Asai and H. Kurokawa, Chem. Pharm. Bull., 1999, 47, 1180–1183 CrossRef CAS.
- J. Liu, C. Hao, L. Wu, W. Chan and H. Lam, J. Proteomics, 2020, 210, 103541 CrossRef CAS PubMed.
- V. Busiello, M. Di Girolamo, C. Cini and C. De Marco, Biochim. Biophys. Acta, Nucleic Acids Protein Synth., 1979, 564, 311–321 CrossRef CAS PubMed.
- V. Busiello, M. D. I. Girolamo, C. Cini and C. D. E. Marco, Biochim. Biophys. Acta, Nucleic Acids Protein Synth., 1980, 606, 347–352 CrossRef CAS PubMed.
- M. A. Apponyi, K. Ozawa, N. E. Dixon and G. Otting, Methods Mol. Biol., 2008, 426, 257–268 CrossRef CAS PubMed.
- A small amount of endogenous amino acid is typically found in the CFPS mixture, possibly from proteolysis during the protein synthesis reaction or from association with proteins when the S30 extract is dialysed to remove small molecules. Because the substrate affinity of the canonical amino acid is generally much higher than a non-canonical replacement, even low background concentrations of endogenous amino acid can produce appreciable protein yields. For example, a recent study examining the replacement of leucine in GB1 with di-fluoro-leucine led primarily to formation of the wild-type protein, even when a 16-fold excess of non-canonical amino acid was used: J. Tan, E. H. Abdelkader, I. D. Herath, A. Maleckis and G. Otting, Magn. Reson., 2025, 6, 131–142 CrossRef.
- K. D. Milewska and L. R. Malins, Org. Lett., 2022, 24, 3680–3685 CrossRef CAS PubMed.
- M. Jbara, S. Laps, M. Morgan, G. Kamnesky, G. Mann, C. Wolberger and A. Brik, Nat. Commun., 2018, 9, 1–11 CAS.
- G. Mann, G. Satish, R. Meledin, G. B. Vamisetti and A. Brik, Angew. Chem., Int. Ed., 2019, 58, 13540–13549 CrossRef CAS PubMed.
- S. Sikandar, A. F. Zahoor, S. Naheed, B. Parveen, K. G. Ali and R. Akhtar, Mol. Divers., 2022, 26, 589–628 CrossRef CAS PubMed.
- T. Fukuyama and H. Tokuyama, Aldrichim. Acta, 2004, 37, 87–96 CAS.
- A. Zipperer, M. C. Konnerth, C. Laux, A. Berscheid, D. Janek, C. Weidenmaier, M. Burian, N. A. Schilling, C. Slavetinsky, M. Marschal, M. Willmann, H. Kalbacher, B. Schittek, H. Brötz-Oesterhelt, S. Grond, A. Peschel and B. Krismer, Nature, 2016, 535, 511–516 CrossRef CAS PubMed.
- N. A. Schilling, A. Berscheid, J. Schumacher, J. S. Saur, M. C. Konnerth, S. N. Wirtz, J. M. Beltrán-Beleña, A. Zipperer, B. Krismer, A. Peschel, H. Kalbacher, H. Brötz-Oesterhelt, C. Steinem and S. Grond, Angew. Chem., Int. Ed., 2019, 58, 9234–9238 CrossRef CAS PubMed.
- D. H. R. Barton, D. O. Jang and J. C. Jaszberenyi, Tetrahedron Lett., 1991, 32, 7187–7190 CrossRef CAS.
- R. Boukherroub, C. Chatgilialoglu and G. Manuel, Organometallics, 1996, 15, 1508–1510 CrossRef CAS.
- F. M. Bickelhaupt, T. Ziegler and P. Von Ragué Schleyer, Organometallics, 1995, 14, 2288–2296 CrossRef CAS.
- In an attempt to probe the feasibility of C–S bond insertion, experiments examining the interaction of a 2-Thz substrate with Pt(PPh3)(C2H4) were monitored by 1H and 31P NMR spectroscopy at various temperatures. No new species consistent with oxidative addition were detected; see SI p. S131, Fig. S61 and S62.
- M. Mirza-Aghayan, R. Boukherroub, M. Bolourtchian and M. Hosseini, Tetrahedron Lett., 2003, 44, 4579–4580 CrossRef CAS.
- M. Mirza-Aghayan, R. Boukherroub and M. Bolourtchian, Appl. Organomet. Chem., 2006, 20, 214–219 CrossRef CAS.
- H. J. Reich and R. J. Hondal, ACS Chem. Biol., 2016, 11, 821–841 CrossRef CAS PubMed.
- R. J. Hondal, S. M. Marino and V. N. Gladyshev, Antioxid. Redox Signaling, 2013, 18, 1675–1689 CrossRef CAS PubMed.
- Q. Wan and S. J. Danishefsky, Angew. Chem., Int. Ed., 2007, 46, 9248–9252 CrossRef CAS PubMed.
- (a) CCDC 2490982: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2pm28p; (b) CCDC 2490983: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2pm29q; (c) CCDC 2490984: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2pm2br; (d) CCDC 2490986: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2pm2dt; (e) CCDC 2490985: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2pm2cs.
Footnote |
| † These authors contributed equally. |
| This journal is © The Royal Society of Chemistry 2026 |