Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Polymorphism-driven coordination geometry engineering for boosting nitrate electroreduction in Cu-pyrazolate chains
Zhanning
Liu†
 *,
Shanna
An†
,
Qingzhong
Xue
*,
Shanna
An†
,
Qingzhong
Xue
 * and
Jian
Tian
* and
Jian
Tian
 *
*
School of Materials Science and Engineering, Shandong Key Laboratory of Special Epoxy Resin, Shandong University of Science and Technology, Qingdao, 266590, China. E-mail: znliu@sdust.edu.cn; xueqz@upc.edu.cn; jiantian@sdust.edu.cn
First published on 9th March 2026
Abstract
Tailoring the coordination geometry of metal centers through polymorphism offers a powerful approach to isolating the structural origin of catalytic activity in metal–organic frameworks (MOFs). Herein, two copper-pyrazolate (Pz) polymorphs, α-Cu(Pz)2 and β-Cu(Pz)2 were synthesized, featuring identical chemical compositions and 1-periodic chain structures but distinct local coordination configurations. Remarkably, the β-Cu(Pz)2 exhibits a faradaic efficiency (FE) of 93.33% for the nitrate reduction reaction (NO3RR), significantly outperforming α-Cu(Pz)2 (53.10%). Comprehensive structural analyses, in situ spectroscopy, and density functional theory (DFT) calculations revealed that the coordination geometry governs the electronic structure of the Cu active centers. Specifically, the cis-configured β-Cu(Pz)2 enables more delocalized Cu 3d orbitals and stronger Cu–O (NO3−) electronic coupling, thereby promoting nitrate adsorption and activation. This work demonstrates that MOF polymorphism allows precise tuning of electronic structures, offering a fundamental design principle for the development of advanced electrocatalysts toward sustainable nitrogen-cycle chemistry.
Introduction
Ammonia (NH3) synthesis has been one of the largest chemical industries since the early twentieth century, owing to its crucial role in modern agriculture and its promising potential for energy storage.1–4 Among the various strategies for NH3 production, electrocatalytic synthesis via the nitrate reduction reaction (NO3RR) has recently attracted increasing attention as a promising alternative to the energy-intensive Haber–Bosch process.5–9 Moreover, by utilizing nitrate as the nitrogen feedstock, this approach integrates energy conversion with environmental remediation, providing a dual benefit for both ammonia synthesis and wastewater treatment.10,11 Nevertheless, the reduction of NO3− to NH3 is a complex process that involves the transfer of eight electrons and nine protons, accompanied by multiple hydrogenation and deoxygenation steps.12 This intricate reaction pathway often leads to the formation of various intermediates and by-products.13,14 Consequently, the rational design of efficient electrocatalysts and the elucidation of structural-performance relationships remain significant challenges.Metal–organic frameworks (MOFs), a class of highly crystalline organic–inorganic hybrid materials, have recently emerged as an ideal platform for designing electrocatalysts and conducting mechanistic studies.15–17 Their rich chemical diversity, facile functionalization, and modular architecture together provide a robust foundation for elucidating the molecular-level mechanisms underlying electrocatalytic processes. For instance, Cheng et al. demonstrated that introducing polar functional groups (–NH2 or –NO2) into ZIF-7 can enhance CO2 capture and activation during electrocatalytic CO2 reduction.18 Zuo and coworkers reported that a bimetallic Zn5-NiS4TP MOF shows superior electrocatalytic NO3RR performance,19 in which the Zn5 sites are responsible for reducing NO3− to NO2−, while [NiS4] sites within the ligand facilitate the subsequent reduction of NO2− to NH3. Our recent investigations have demonstrated that tuning the electron-withdrawing strength of halogen atoms (F, Cl, and Br) in the copper anilates can effectively modulate the electronic structure of Cu2+, thereby facilitating the deoxygenation of *NO intermediate.20,21 However, these strategies typically rely on introducing additional atoms or functional groups, which may complicate or obscure the precise elucidation of the intrinsic active sites.
Polymorphism, referring to the ability of a single compound to exist in two or more distinct crystalline forms,22–24 is widely observed in MOFs. Subtle variations in the arrangement or coordination geometry of the building blocks can induce pronounced changes in their physical and chemical properties, offering a powerful platform for unraveling structure-property relationships.25–27 For instance, the packing arrangement changes of ligand in cadmium squarates can induce dramatically different thermal expansion behaviors.25 In Al-based MOFs, polymorphism arising from cis-trans configurations of µ-OH-connected AlO4(OH)2 chains leads to markedly different water adsorption behaviors.27 However, its potential in electrocatalysis remains rarely explored. To this end, Cu2+ based MOFs provide an ideal model system, as Cu2+ centers not only exhibit a favorable alignment between the d-orbital energy levels and the LUMO π* of NO3−,28 but also display diverse coordination modes that can give rise to rich polymorphic behavior.29–31
Motivated by these points and as a proof of concept, we herein systematically investigated the electrocatalytic NO3RR performance of two 1-periodic copper-pyrazolate (Pz) polymorphs, α-Cu(Pz)2 and β-Cu(Pz)2. Remarkably, the β-Cu(Pz)2 exhibits a significantly enhanced NO3RR activity with the Faraday efficiency (FE) reaching 93.33%, while the α-Cu(Pz)2 only delivers a FE of 53.10%. Comprehensive analyses combining long-range and short-range structure investigations, in situ spectroscopic measurements, and density functional theory (DFT) calculations reveal that the coordination geometry of Cu centers plays a decisive role in modulating their electronic structures, thereby governing the adsorption and activation of nitrate species and ultimately determining the overall NO3RR performance.
Results and discussion
The α-Cu(Pz)2 and β-Cu(Pz)2 phases were synthesized via soft-chemistry methods, and the detailed procedures are given in the SI.32 Powder X-ray diffraction (PXRD) analyses, combined with Rietveld refinements (Fig. 1), confirmed that both products are phase pure and crystallize in distinct structures. Despite sharing the same chemical formula and both exhibiting infinite one-periodic (1p) chain motifs, their spatial arrangements are dramatically different. As shown in Fig. 1, the 1p chains in α-Cu(Pz)2 pack into an orthorhombic phase with the lattice parameters a = 7.922 Å, b = 11.504 Å, and c = 7.791 Å, whereas β-Cu(Pz)2 adopts a different unit cell with a = 16.970 Å, b = 6.236 Å, and c = 7.294 Å. The distinct packing modes stem from the local coordination differences within the 1p chain. In both samples, each Cu2+ is coordinated by four nitrogen atoms from four Pz ligands. However, in the α-Cu(Pz)2 phase, the Pz molecules adopt an alternating trans configuration, forming nearly linear 1p chains along the a-axis. In contrast, the β-Cu(Pz)2 phase displays a uniform cis configuration, in which the Pz molecules are oriented in the same direction, giving rise to a zig-zag chain conformation. Such distinct chain geometries induce different steric effects and packing behaviors, ultimately giving rise to the observed polymorphism of the Cu(Pz)2 system. The scanning electron microscope (SEM) images (Fig. S1) showed that both samples consist of nanosized particles with diameters in the range of 200–500 nm.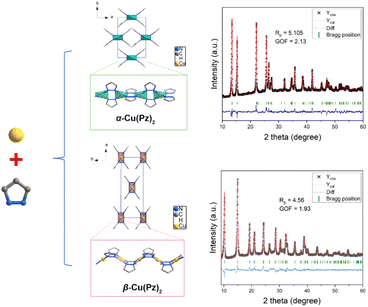 | ||
| Fig. 1 Illustration of crystal structures of α-Cu(Pz)2 and β-Cu(Pz)2 and the corresponding Rietveld refinement patterns. | ||
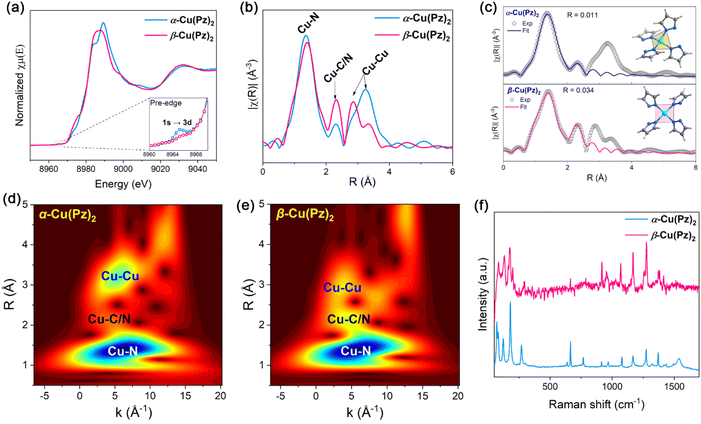
To gain atomic-level insight into the local coordination environments of α-Cu(Pz)2 and β-Cu(Pz)2 and to minimize potential effects from atomic occupancy disorder, Cu K-edge X-ray adsorption fine structure (XAFS) analyses were carried out. As shown in Fig. 2a, the X-ray absorption near-edge structure (XANES) spectra of the two compounds exhibit nearly identical adsorption edges, indicating that the Cu ions in both samples share similar oxidation states. This conclusion is further validated by the high-resolution X-ray photoelectron spectroscopy (XPS, Fig. S2), which confirms that the Cu ions in both samples are in the +2 oxidation state. Notably, the pre-edge peak at ∼8965 eV is more pronounced in α-Cu(Pz)2 than in β-Cu(Pz)2. Since this feature arises from the 1s → 3d electronic transition,33–35 which is typically intensified in non-centrosymmetric coordination environments, the result indicates that the Cu2+ site in α-Cu(Pz)2 possesses a less symmetric local coordination environment. This was further supported by the extended X-ray absorption fine structure (EXAFS) analyses. As shown in Fig. 2b, the first peak at ∼1.4 Å corresponds to the Cu–N bond, while the second coordination-shell peak at ∼2.3 Å is attributed to Cu–C/N correlations arising from the next-nearest-neighbor C/N atoms of the Pz ligand. It should be noted that the EXAFS data are not phase-corrected, resulting in an apparent radial shift of approximately 0.4 Å relative to the actual Cu–N distances.36 The EXAFS fitting results reveal that Cu2+ in both compounds is four-coordinate (Tables S1 and S2). However, the coordination geometries differ distinctly, being a distorted tetrahedron in α-Cu(Pz)2 and a square-planar configuration in β-Cu(Pz)2 (Fig. 2c). According to the Cambridge Crystallographic Data Centre (CCDC), most of the Cu(II)-N4 based complexes preferentially adopt a square-planar geometry. Thus, the distorted tetrahedral coordination environment in α-Cu(Pz)2 may render this phase metastable. Further DFT calculations showed that, the ground-state energy of β-Cu(Pz)2 is significantly lower than α-Cu(Pz)2 with ΔE0 = −0.12775 eV (Table S3), confirming the higher intrinsic stability of β-Cu(Pz)2. Fig. 2d and e display the wavelet transform (WT) contour plots of the two samples. The lobes observed at ∼5 Å−1 and ∼3 Å are assigned to the third coordination sphere, corresponding to Cu–Cu pairs. Obviously, the Cu–Cu distance in α-Cu(Pz)2 is longer than that in β-Cu(Pz)2, which is consistent with the quasi-linear and zig-zag configurations observed in their long-range structures. Moreover, the Raman spectra of the two compounds show similar characteristic bands (Fig. 2f), validating their identical building blocks.
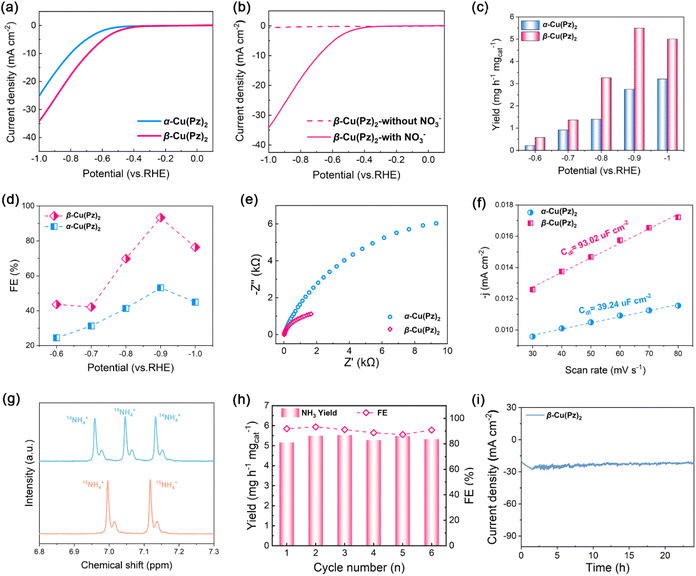
Electrocatalytic NO3RR performances of the two polymorphs were evaluated in a typical H-type electrolytic cell containing 0.1 M Na2SO4 and 0.1 M KNO3. The linear sweep voltammetry (LSV) curves of α-Cu(Pz)2 and β-Cu(Pz)2 are shown in Fig. 3a. The current density of β-Cu(Pz)2 is obviously higher than that of α-Cu(Pz)2 at the same potential, inferring its higher electrocatalytic activity. In addition, the pronounced increase in current density in the presence of NO3− confirms its electrocatalytic NO3RR performance (Fig. 3b). Based on the LSV results, a series of NO3RR measurements were conducted under different potentials ranging from −0.5–−1.0 V vs. RHE. The NH3 yield rates were quantified using a chronopotentiometry method (Fig. S3–S5). As shown in Fig. 3c and d, both NH3 yield rates and FEs display a volcanic trend with increasingly negative potentials. At all applied potentials, β-Cu(Pz)2 outperforms α-Cu(Pz)2. Remarkably, at −0.9 V vs. RHE, β-Cu(Pz)2 achieves an NH3 yield rate of 5.50 mg h−1 mgcat−1 with an FE of 93.33%, nearly twice that of α-Cu(Pz)2 (2.74 mg h−1 mgcat−1 and 53.10%) and outperforming most of the reported copper-based electrocatalysts (Table S3). These results indicate that changes in the local coordination geometry of the microenvironment exert a significant regulatory effect on the catalytic performance. To confirm the origin of ammonia production, isotope labeling experiments were performed. As shown in Fig. 3g, the 1H nuclear magnetic resonance (NMR) spectra using 15NO3− as the feeding nitrogen source display the characteristic doublet of 15NH4+, whereas 14NO3− produces 14NH4+ with a triplet signal. These results confirm that the NH4+ detected in the electrolyte does originate from nitrate reduction rather than other impurities. Besides, the electrochemical impedance spectroscopy (EIS) measurements were conducted to assess the conductivity of electrodes. As shown in Fig. 3e, the β-Cu(Pz)2 exhibits a significantly lower interfacial electron-transfer resistance. Meanwhile, the electrochemically active surface area (ECSA) measurements (Fig. 3f, S6) indicate that β-Cu(Pz)2 has a higher Cdl value of 93.02 µF cm−2, obviously exceeding that of α-Cu(Pz)2 (39.24 µF cm−2), suggesting a greater number of accessible active sites in β-Cu(Pz)2. The cycling performance of β-Cu(Pz)2 was evaluated at −0.9 V vs. RHE. Over six consecutive cycles, no significant decrease in NH3 yield rate or FE can be observed (Fig. 3h), and negligible metal ion leaching was detected. Moreover, the 24 h time-dependent current density profile shows negligible decay (Fig. 3i), further confirming its long-term stability and potential for practical applications.
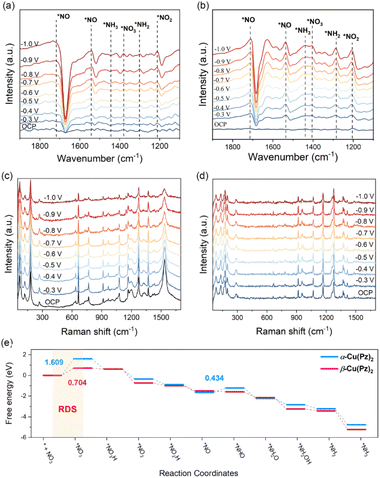
In situ Fourier transform infrared (FTIR) spectroscopy was employed to probe the possible reaction intermediates formed during the ammonia synthesis process over α-Cu(Pz)2 and β-Cu(Pz)2 catalysts. As shown in Fig. 4a and b, both catalysts exhibit similar intermediate-related signals as the applied potential shifts from open-circuit potential (OCP) to −1.0 V vs. RHE. Among them, the band at 1404 cm−1 is attributed to the adsorbed NO3− species, while the bands at 1205 cm−1, 1537 cm−1, and 1285 cm−1 correspond to the formed *NO2, *NO, and *NH2 intermediates,37,38 respectively. These characteristic peaks suggest that the electrocatalytic NO3RR proceeds through a successive deoxygenation and hydrogenation process. Notably, the intermediate-related IR bands of β-Cu(Pz)2 exhibit a markedly higher signal-to-noise ratio than those of α-Cu(Pz)2, implying enhanced catalytic performance. This observation is fully consistent with its superior NO3RR performance described above. Furthermore, in situ Raman spectra were used to detect the possible structural changes in the catalysts (Fig. 4c and d). Under the applied potentials, no additional Raman bands were observed, confirming the structural stability of the catalysts.
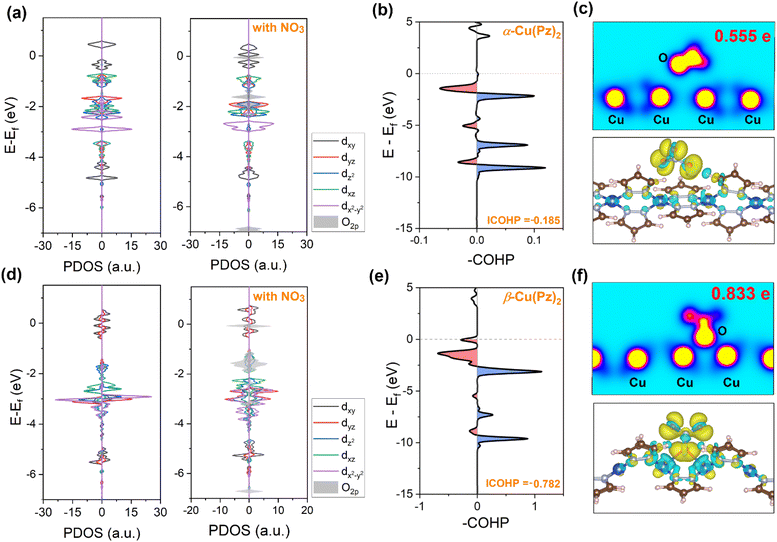
Subsequently, guided by the above in situ spectroscopic results, density functional theory (DFT) calculations were carried out to further elucidate the origin of their distinct catalytic activities (Fig. S7 and S8). The electrocatalytic NO3RR process primarily proceeds through the following pathway: (1) adsorption and activation of NO3− to form *NO3 radical; (2) successive deoxygenation to produce *NO and stepwise hydrogenation via proton-coupled electron transfer (PCET) to ultimately generate *NH3.39 As depicted in the free energy profiles (Fig. 4e), NO3− adsorption is the rate-determining step (RDS) throughout the overall reaction process for both catalysts. The corresponding energy barrier on β-Cu(Pz)2 (ΔG = 0.704 eV) is significantly lower than that on α-Cu(Pz)2 (ΔG = 1.609 eV), indicating a more favourable activation of the NO3− species. To gain deeper insight into this difference, the electronic structures and orbital interactions of the two catalysts were examined. The projected density of states (PDOS) of Cu 3d orbitals for pristine α-Cu(Pz)2 and β-Cu(Pz)2 are shown in Fig. 5a and d. In α-Cu(Pz)2, the Cu 3d states are relatively localized, with narrow distributions and weak overlap, indicating limited hybridization with the surrounding ligand orbitals. In contrast, β-Cu(Pz)2 exhibits broader and more continuous 3d states, implying enhanced delocalization and stronger Cu-ligand electronic coupling. Upon NO3− adsorption, β-Cu(Pz)2 displays a more pronounced overlap between the O 2p orbitals of NO3− and the Cu 3d orbitals, revealing stronger Cu–O electronic coupling and greater charge transfer capability. This conclusion is further supported by the integrated crystal orbital Hamilton population (ICOHP) analysis, where the Cu–O bonds in α-Cu(Pz)2 and β-Cu(Pz)2 with adsorbed NO3− are −0.185 eV and −0.782 eV (Fig. 5b and e), respectively. The more negative ICOHP value signifies a stronger chemical bonding interaction,40 corroborating that the *NO3 binds more tightly to β-Cu(Pz)2 than to α-Cu(Pz)2. The charge density difference maps (Fig. 5c and f) also demonstrate that the NO3− species gains approximately 0.555 e and 0.833 e from α-Cu(Pz)2 and β-Cu(Pz)2, respectively, confirming more significant charge transfer in the latter. Overall, these results reveal that the enhanced NO3RR performance of β-Cu(Pz)2 originates from its stronger Cu–O orbital hybridization and greater electron delocalization, which can effectively facilitate the NO3− activation and reduction.
Conclusions
In summary, two copper-pyrazolate polymorphs α-Cu(Pz)2 and β-Cu(Pz)2 were successfully synthesized. Both samples feature 1-periodic chain structures, while their local coordination geometry were dramatically different, with α-Cu(Pz)2 adopting a trans configuration and β-Cu(Pz)2 adopting a cis configuration. Electrocatalytic NO3RR measurements revealed that the β-Cu(Pz)2 demonstrates a markedly superior performance, achieving a FE of 93.33%, compared with 53.10% of α-Cu(Pz)2. This enhancement originates from their distinct electronic structures. In β-Cu(Pz)2, the Cu 3d orbitals are more delocalized, enabling greater electron donation to the adsorbed *NO3 species, thereby promoting the nitrate adsorption, activation, and subsequent reduction. These findings highlight the crucial role of coordination geometry engineering in tuning the electronic properties of active metal centers, providing valuable insights for the rational design of advanced electrocatalysts for sustainable ammonia synthesis and beyond.Author contributions
Conceptualization, Zhanning Liu; methodology and investigation, Zhanning Liu, and Shanna An; software, Zhanning Liu, Qingzhong Xue, and Jian Tian; funding acquisition, Zhanning Liu; resources, Qingzhong Xue and Jian Tian. All authors have read and agreed to the published version of the manuscript.Conflicts of interest
There are no conflicts to declare.Data availability
The data underlying this study are available in the published article and its supplementary information (SI). Supplementary information: experimetnal details, DFT calculations, SEM, XPS, electrochemical measurement results, and crystal structure models. See DOI: https://doi.org/10.1039/d5sc09385f.Acknowledgements
This work was supported by the National Natural Science Foundation of China (No. 22005340), Natural Science Foundation of Shandong Province (No. ZR2022JQ21), National Ten Thousand Talent Program for Young Top-notch Talents, Science and Technology Special Project of Qingdao City (No. 25-1-5-cspz-9-nsh), and Qingdao Natural Science Foundation (24-4-4-zrjj-194-jch).Notes and references
- D. R. MacFarlane, P. V. Cherepanov, J. Choi, B. H. Suryanto, R. Y. Hodgetts, J. M. Bakker, F. M. F. Vallana and A. N. Simonov, A roadmap to the ammonia economy, Joule, 2020, 4, 1186–1205 CrossRef CAS.
- K. Zhang, A. Cao, L. H. Wandall, J. Vernieres, J. Kibsgaard, J. K. Nørskov and I. Chorkendorff, Spin-mediated promotion of Co catalysts for ammonia synthesis, Science, 2024, 383, 1357–1363 CrossRef CAS.
- M. Wang, M. A. Khan, I. Mohsin, J. Wicks, A. H. Ip, K. Z. Sumon, C.-T. Dinh, E. H. Sargent, I. D. Gates and M. G. Kibria, Can sustainable ammonia synthesis pathways compete with fossil-fuel based Haber–Bosch processes?, Energy Environ. Sci., 2021, 14, 2535–2548 RSC.
- S. Wu, N. Salmon, M. M.-J. Li, R. Bañares-Alcántara and S. C. E. Tsang, Energy decarbonization via green H2 or NH3?, ACS Energy Lett., 2022, 7, 1021–1033 CrossRef CAS.
- S. Li, X. Fu, J. K. Nørskov and I. Chorkendorff, Towards sustainable metal-mediated ammonia electrosynthesis, Nat. Energy, 2024, 9, 1344–1349 CrossRef CAS.
- P. H. van Langevelde, I. Katsounaros and M. T. Koper, Electrocatalytic nitrate reduction for sustainable ammonia production, Joule, 2021, 5, 290–294 CrossRef.
- J. Liang, Z. Li, L. Zhang, X. He, Y. Luo, D. Zheng, Y. Wang, T. Li, H. Yan and B. Ying, Advances in ammonia electrosynthesis from ambient nitrate/nitrite reduction, Chem, 2023, 9, 1768–1827 CAS.
- M. A. Mushtaq, A. Kumar, W. Liu, Q. Ji, Y. Deng, G. Yasin, A. Saad, W. Raza, J. Zhao and S. Ajmal, A metal coordination number determined catalytic performance in manganese borides for ambient electrolysis of nitrogen to ammonia, Adv. Mater., 2024, 36, 2313086 CrossRef CAS.
- Y.-C. Liu, J.-R. Huang, H.-L. Zhu, X.-F. Qiu, C. Yu, X.-M. Chen and P.-Q. Liao, Electrosynthesis of pure urea from pretreated flue gas in a proton-limited environment established in a porous solid-state electrolyte electrolyser, Nat. Nanotechnol., 2025, 1–7 Search PubMed.
- Y. Du, H. Lu, J. Wu, Y. Zou, Z. F. Huang, J. J. Zou, T. Mu, J. Gao, X. D. Zhu and Y. C. Zhang, Selenium-Deficient FeSe2/Fe3O4 Electrocatalyst for Nitrate Reduction to Ammonia, Angew. Chem., Int. Ed., 2025, 64, e202420903 CrossRef CAS PubMed.
- D.-S. Huang, X.-F. Qiu, J.-R. Huang, M. Mao, L. Liu, Y. Han, Z.-H. Zhao, P.-Q. Liao and X.-M. Chen, Electrosynthesis of urea by using Fe2O3 nanoparticles encapsulated in a conductive metal–organic framework, Nat. Synth., 2024, 3, 1404–1413 CrossRef CAS.
- P. Muthukumar, Z. Ullah, X. Zhang, H. Ullah, Y. Liu, L. Li, S. Tian, X. Zhou, S. P. Anthony, Y. Zuo, C. Lv, X. Wang and C. Wang, Unlocking a Water Coordination Environment in Co-Based Metal–Organic Frameworks for Advanced Nitrate-to-Ammonia Electroreduction, J. Am. Chem. Soc., 2025, 147, 29949 CrossRef CAS.
- H. Xu, Y. Ma, J. Chen, W.-X. Zhang and J. Yang, Electrocatalytic reduction of nitrate–a step towards a sustainable nitrogen cycle, Chem. Soc. Rev., 2022, 51, 2710–2758 Search PubMed.
- G. Zhang, X. Li, K. Chen, Y. Guo, D. Ma and K. Chu, Tandem electrocatalytic nitrate reduction to ammonia on MBenes, Angew. Chem., Int. Ed., 2023, 62, e202300054 CrossRef CAS.
- Z. Yang, A. Li, H. Li, G. Lai, Y. Fu, Y. Zhang, K. Wang, S. Zeng, L. Xie and M. Li, Dimensionality Reduction of Metal–Organic Frameworks to Monolayers for Enhanced Electrocatalysis, Angew. Chem., Int. Ed., 2025, e202505399 CAS.
- N. Sun, S. S. A. Shah, Z. Lin, Y.-Z. Zheng, L. Jiao and H.-L. Jiang, MOF-based electrocatalysts: an overview from the perspective of structural design, Chem. Rev., 2025, 125, 2703–2792 CrossRef CAS PubMed.
- A. H. Mamaghani, J. Liu, Z. Zhang, R. Gao, Y. Wu, H. Li, M. Feng and Z. Chen, Promises of MOF-based and MOF-derived materials for electrocatalytic CO2 reduction, Adv. Energy Mater., 2024, 14, 2402278 CrossRef CAS.
- J. Chen, G. Wang, Y. Dong, J. Ji, L. Li, M. Xue, X. Zhang and H. M. Cheng, Controlling the Polarity of Metal–Organic Frameworks to Promote Electrochemical CO2 Reduction, Angew. Chem., Int. Ed., 2025, 64, e202416367 CrossRef CAS PubMed.
- Z. Zhang, Y. Lv, Y. Gu, X. Zhou, B. Tian, A. Zhang, Z. Yang, S. Chen, J. Ma and M. Ding, Dual Zn5− NiS4 Sites in a Redox-Active Metal–Organic Framework Enables Efficient Cascade Catalysis for Nitrate-to-Ammonia Conversion, Angew. Chem., Int. Ed., 2025, 64, e202418272 CrossRef CAS PubMed.
- C. Xing, J. Ren, L. Fan, J. Zhang, M. Ma, S. Wu, Z. Liu and J. Tian, π-d conjugated copper chloranilate with distorted Cu-O4 site for efficient electrocatalytic ammonia production, Adv. Funct. Mater., 2024, 34, 2409064 CrossRef CAS.
- Z. Liu, C. Xing, Y. Shan, M. Ma, S. Wu, R. Ge, Q. Xue and J. Tian, Molecular engineering of 1D conjugated copper anilate coordination polymers for boosting electrocatalytic nitrate reduction to ammonia, Chem. Sci., 2025, 16, 7010–7017 RSC.
- A. J. Cruz-Cabeza and J. Bernstein, Conformational polymorphism, Chem. Rev., 2014, 114, 2170–2191 CrossRef CAS.
- B. Karadeniz, D. Žilić, I. Huskić, L. S. Germann, A. M. Fidelli, S. Muratović, I. Lončarić, M. Etter, R. E. Dinnebier and D. Barišić, Controlling the polymorphism and topology transformation in porphyrinic zirconium metal–organic frameworks via mechanochemistry, J. Am. Chem. Soc., 2019, 141, 19214–19220 CrossRef CAS PubMed.
- T. J. Matemb Ma Ntep, M. Wahiduzzaman, E. Laurenz, I. Cornu, G. Mouchaham, I. Dovgaliuk, S. Nandi, K. Knop, C. Jansen and F. Nouar, When polymorphism in metal–organic frameworks enables water sorption profile tunability for enhancing heat allocation and water harvesting performance, Adv. Mater., 2024, 36, 2211302 CrossRef CAS.
- Z. Liu, R. Ma, J. Deng, J. Chen and X. Xing, Molecular packing-dependent thermal expansion behaviors in metal squarate frameworks, Chem. Mater., 2020, 32, 2893–2898 CrossRef CAS.
- Z. Chen, C.-H. Ho, X. Wang, S. M. Vornholt, T. M. Rayder, T. Islamoglu, O. K. Farha, F. Paesani and K. W. Chapman, Humidity-responsive polymorphism in CALF-20: a resilient MOF physisorbent for CO2 capture, ACS Mater. Lett., 2023, 5, 2942–2947 CrossRef CAS.
- T. J. Matemb Ma Ntep, M. Wahiduzzaman, E. Laurenz, I. Cornu, G. Mouchaham, I. Dovgaliuk, S. Nandi, K. Knop, C. Jansen, F. Nouar, P. Florian, G. FuIdner, G. Maruin, C. Janiak and C. Serre, When polymorphism in metal-organic frameworks enables water sorption profile tunability for enhancing heat allocation and water harvesting performance, Adv. Mater., 2024, 36, 2211302 CrossRef CAS.
- G.-F. Chen, Y. Yuan, H. Jiang, S.-Y. Ren, L.-X. Ding, L. Ma, T. Wu, J. Lu and H. Wang, Electrochemical reduction of nitrate to ammonia via direct eight-electron transfer using a copper–molecular solid catalyst, Nat. Energy, 2020, 5, 605–613 CrossRef CAS.
- Y. Yang, C. Zhang, C. Zhang, Y. Shi, J. Li, B. Johannessen, Y. Liang, S. Zhang, Q. Song and H. Zhang, Ligand-tuning copper in coordination polymers for efficient electrochemical C–C coupling, Nat. Commun., 2024, 15, 6316 CrossRef CAS.
- J. Jiao, X. Kang, J. Yang, S. Jia, Y. Peng, S. Liu, C. Chen, X. Xing, M. He and H. Wu, Steering the reaction pathway of CO2 electroreduction by tuning the coordination number of copper catalysts, J. Am. Chem. Soc., 2024, 146, 15917–15925 CrossRef CAS PubMed.
- Z.-H. Zhao, J.-R. Huang, P.-Q. Liao and X.-M. Chen, Highly efficient electroreduction of CO2 to ethanol via asymmetric C–C coupling by a metal–organic framework with heterodimetal dual sites, J. Am. Chem. Soc., 2023, 145, 26783–26790 CrossRef CAS PubMed.
- A. Cingoani, S. Galli, N. Mascicchi, L. Pandolfo, C. Pettinari and A. Sironi, Sorption-desorption behavior of bispyrazolato-copper(II) 1D coordination polymers, J. Am. Chem. Soc., 2005, 127, 6144–6145 CrossRef PubMed.
- B. L. Geoghegan, Y. Liu, S. Peredkov, S. Dechert, F. Meyer, S. DeBeer and G. E. Cutsail III, Combining valence-to-core x-ray emission and Cu k-edge x-ray absorption spectroscopies to experimentally assess oxidation state in organometallic Cu (I)/(II)/(III) complexes, J. Am. Chem. Soc., 2022, 144, 2520–2534 CrossRef CAS.
- L. León-Alcaide, C. Castillo-Blas, V. Martin-Diaconescu, I. Da Silva, D. A. Keen, T. D. Bennett and G. M. Espallargas, Solvent-free approach for the synthesis of heterometallic Fe–Zn-ZIF glass via a melt-quenched process, Chem. Sci., 2025, 16, 7946–7955 RSC.
- F. Farges, Ab initio and experimental pre-edge investigations of the Mn K-edge XANES in oxide-type materials, Phys. Rev. B, 2005, 71, 155109 CrossRef.
- V. L. Sushkevich, O. V. Safonova, D. Palagin, M. A. Newton and J. A. van Bokhoven, Structure of copper sites in zeolites examined by Fourier and wavelet transform analysis of EXAFS, Chem. Sci., 2020, 11, 5299–5312 RSC.
- X. Gu, J. Zhang, S. Guo, Y. Zhang, L. Xu, R. Jin and G. Li, Tiara Ni Clusters for Electrocatalytic Nitrate Reduction to Ammonia with 97% Faradaic Efficiency, J. Am. Chem. Soc., 2025, 14, 22785 CrossRef.
- L. Zhou, X. Chen, S. Zhu, K. You, Z. J. Wang, R. Fan, J. Li, Y. Yuan, X. Wang and J. Wang, Two-dimensional Cu Plates with Steady Fluid Fields for High-rate Nitrate Electroreduction to Ammonia and Efficient Zn-Nitrate Batteries, Angew. Chem., Int. Ed., 2024, 136, e202401924 CrossRef.
- D. R. Weinberg, C. J. Gagliardi, J. F. Hull, C. F. Murphy, C. A. Kent, B. C. Westlake, A. Paul, D. H. Ess, D. G. McCafferty and T. J. Meyer, Proton-coupled electron transfer, Chem. Rev., 2012, 112, 4016–4093 CrossRef CAS PubMed.
- S. Wang, Q. Jiang, S. Ju, C.-S. Hsu, H. M. Chen, D. Zhang and F. Song, Identifying the geometric catalytic active sites of crystalline cobalt oxyhydroxides for oxygen evolution reaction, Nat. Commun., 2022, 13, 6650 CrossRef CAS.
Footnote |
| † These authors contributed equally to this work. |
| This journal is © The Royal Society of Chemistry 2026 |