Open Access Article
Open Access ArticleSolvent-controlled, chemodivergent oxidative anionic Fries rearrangement of O-aryl carbamates under aerobic conditions
Riccardo
Gnavi
 ab,
Federica
De Nardi
ab,
Federica
De Nardi
 b,
Carolina
Meazzo
b,
Carolina
Meazzo
 b,
Simone
Ghinato
b,
Simone
Ghinato
 b,
Ettore
Grimaldi
b,
Ettore
Grimaldi
 b,
Andrea
Maranzana
b,
Andrea
Maranzana
 b,
Cristina
Prandi
b,
Cristina
Prandi
 b and
Marco
Blangetti
b and
Marco
Blangetti
 *b
*b
aScuola Universitaria Superiore IUSS Pavia, Piazza della Vittoria 15, I-27100 Pavia, Italy
bDipartimento di Chimica, Università di Torino, Via P. Giuria 7, I-10125 Torino, Italy. E-mail: marco.blangetti@unito.it
First published on 13th January 2026
Abstract
We disclose herein a mild and efficient organolithium-mediated protocol which enables the chemodivergent transformation of ortho-cresol-derived O-aryl carbamates into diverse molecular structures by simply changing the nature of the reaction medium, working under air and at room temperature. The use of the biobased 2-MeTHF as solvent allows the chemoselective preparation of α-hydroxy arylacetamides in a single synthetic operation with a remarkable functional group tolerance. Our strategy, which exploits the presence of molecular oxygen arising from the use of bench-type aerobic conditions, relies on a one-pot anionic homo-Fries rearrangement/amide enolate autoxidation sequence with two consecutive C–C/C–O bond formation events occurring at the same carbon atom. Furthermore, we also describe the successful use of protic and bioinspired Deep Eutectic Solvents (DESs) as an effective tool to tune the chemoselectivity of the proposed transformation. The fast internal protonolysis of the anion solution, operated by the protic reaction medium, results in an interrupted metalation/rearrangement sequence, enabling the chemoselective preparation of arylacetamides under bench-type aerobic conditions owing to the efficient suppression of the oxidation step.
Introduction
The α-hydroxy amide function is of utmost importance in several areas of organic chemistry owing to its widespread occurrence in agrochemicals and pharmaceuticals with diversified biological targets.1 In this framework, a recurrent motif in bioactive molecules is the α-hydroxy arylacetamide skeleton, the amide derivative of the naturally occurring mandelic acid, which represents a key building block in both the development of novel drug candidates and the production of fine chemicals (Fig. 1A).2 Hence, the development of new bench-stable reagents and effective methodologies for the chemoselective preparation of α-hydroxy arylacetamide derivatives using cheap and readily available synthons is of remarkable synthetic value. Besides the traditional approaches based on the amidation of α-hydroxy acids3 or the reduction of α-keto amides,4 several de novo synthetic methods relying on the assembly of novel C–C bonds have been developed (Fig. 1B), including the nucleophilic homologation of aldehydes by carbamoyl anions,5 multicomponent approaches with isocyanides6 or difluorocarbenes,7 and the base-promoted anionic [1,2]-rearrangement of O-benzyl carbamates.8 A more powerful approach towards mandelic acid amides relies on the α-hydroxylation of arylacetamides by means of a selective C(sp3)–O bond formation. Excellent indirect strategies have been developed, including the chemoselective α-oxyamination of electrophilically activated arylacetamides promoted by TEMPO9 and the anionic [2,3]-rearrangement of O-functionalized hydroxamic acids.10 Direct methods based on the aerobic oxidation of C(sp3)–H bonds are of particular interest, as molecular oxygen is an inexpensive and ubiquitous reagent, and its exploitation in synthetic protocols (rather than its depletion) is of high value also from a sustainability point of view.11 In this context, the direct aerobic α-hydroxylation of arylacetamides (or α-aryllactams) has been accomplished by means of transition metal-promoted,12 organocatalytic,13 and base-promoted14 approaches (Fig. 1B). However, oxygen-mediated processes frequently suffer from both low selectivity and undesired over-oxidation or degradation pathways.15 In this context, opportunities for the preparation of α-hydroxy arylacetamides might also arise from the oxidation of nucleophilic polar s-block organometallic reagents, a well-established reaction typically promoted by metal oxenoids (hydroperoxide metal salts)16 or occurring by autoxidation of the organometallic species in the presence of triplet molecular oxygen17 (Fig. 1C). Despite the enormous progress in this area, especially in the oxidation of aryllithium compounds18 and enolates,19 the use of molecular oxygen for the α-hydroxylation of alkali-metal amide enolates has been barely investigated.20 Recent advancements in the chemistry of s-block organometallic compounds have clearly established that transformations involving organolithium, Grignard, and organosodium reagents could be efficiently performed under “bench-type” conditions, working at room temperature in the presence of air, using non-dried sustainable reaction media.21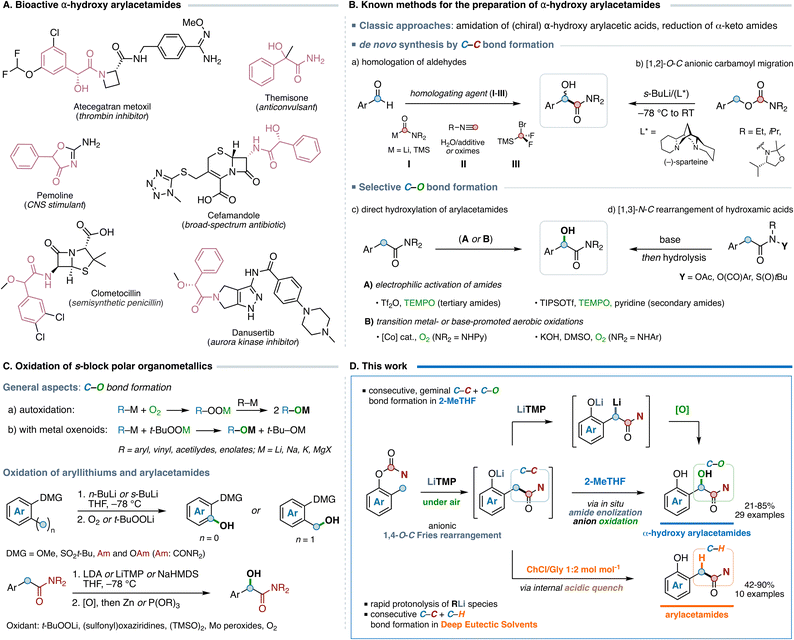 | ||
| Fig. 1 (A and B) State of the art in the synthesis of α-hydroxy arylacetamides. (C) General overview of the oxidation of organometallic reagents and (D) aim of this work. | ||
Supported by our recent findings on the reactivity of polar organometallic reagents under non-conventional conditions,22 we envisaged the possibility of designing a streamlined organolithium-mediated strategy to access α-hydroxy arylacetamides which exploits the presence of molecular oxygen in the reaction mixture, arising from the use of bench-type aerobic conditions. We thus herein report a systematic study on the synergistic combination of lithium amides and molecular oxygen to promote the transformation of ortho-cresol derived O-aryl carbamates into α-hydroxy arylacetamides using a one-pot, tandem anionic 1,4-O–C Fries (homo-Fries)23 rearrangement/oxidation sequence which involves two bond-making events (one C–C and one C–O bond) occurring at the same carbon atom, working under air and at room temperature, using 2-MeTHF as solvent (Fig. 1D). Mechanistic insights revealed that the oxidation of the intermediate lithium amide enolate triggered by molecular oxygen follows a classic autoxidation pathway with a SN2-type cleavage process. Furthermore, we also describe the successful use of bioinspired Deep Eutectic Solvents (DESs) as an effective tool to tune the chemoselectivity of the proposed transformation. The use of a protic heterogeneous solvent mixture allows the suppression of the oxidation step owing to the fast protonolysis of the organolithium species (Fig. 1D), enabling the chemoselective preparation of arylacetamides24 under bench-type aerobic conditions.
Results and discussion
Reaction development
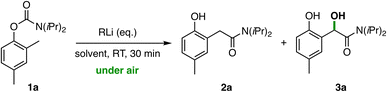
We started our investigation on the anionic homo-Fries rearrangement/oxidation sequence of O-2-tolyl carbamates under bench-type aerobic conditions using the sterically hindered N,N-diisopropyl carbamate 1a as a model substrate (Table 1). In a preliminary experiment, a solution of compound 1a (0.2 mmol, 0.2 M) in cyclopentyl methyl ether (CPME) was reacted with a freshly prepared solution of LiTMP (1 M in CPME, 2.0 equiv.) in the absence of an external electrophile, working at room temperature and under air (Table 1, entry 1).22d Aqueous quench of the anion solution after 30 min afforded a mixture of unreacted carbamate 1a (64%) and the arylacetamide 3a hydroxylated at the benzylic position in 34% yield. The structure was confirmed by NMR spectroscopy and X-ray analysis of 3a, which crystallised by slow, room temperature evaporation of a chloroform solution into the monoclinic P21/n space group.25 Performing the metalation of 1a using 2-MeTHF as reaction medium under the same reaction conditions led to a significant increase of the reaction yield of 3a (54%), alongside the formation of unidentified byproducts (entry 2). Pleasingly, increasing the amount of metalating agent (3 equiv.) resulted in the chemoselective formation of the desired α-hydroxy arylacetamide 3a in a satisfactory 70% yield (entry 3). The use of higher amounts of LiTMP or, alternatively, longer reaction times had a detrimental effect on the reaction yield due to the partial over-oxidation of 3a to the corresponding α-ketoamide byproduct (see the SI for details). Similar results were obtained when the reaction was performed using either the emerging eco-friendly alternative 4-MeTHP (entry 4) or other ethereal solvents with high coordinating ability for organolithiums, such as 1,2-dimethoxyethane (DME, entry 5) and tetrahydrofuran (THF, entry 6). While the use of less coordinating solvents had no effect on the anionic rearrangement, a significant impact on the oxidation step was observed as demonstrated by the considerable amount of unoxidized 2a (17–35%) detected in the reaction mixture (entries 7–9). By contrast, treatment of carbamate 1a with the less basic and sterically hindered lithium amide LDA (entry 10) resulted in a significant decrease of the reaction yield (34%). Analogously, the use of common alkyllithium compounds was less effective, resulting in a loss of selectivity due to the formation of ortho-Fries (s-BuLi)22d or several unidentified byproducts (t-BuLi) in non-negligible amounts (entries 11 and 12). Noteworthy, the stability of the organolithium species in 2-MeTHF is remarkable under these bench-type aerobic conditions. Performing the reaction on 1a in 2-MeTHF either under a dry Ar/O2 atmosphere or in the presence of humidified air (82% RH) provided comparable yields of 3a (71% and 69%, respectively) to those obtained with a standard bench-type (open air) experimental setup (39% RH, see the SI for details). These results clearly indicate that almost no protonolysis of the organolithium species occurs under air over the reaction time, owing to the beneficial effect of the highly hydrophobic 2-MeTHF on the stability of lithium amides.22b,26 Consequently, the reaction could be performed even at high moisture levels without significantly affecting the reaction yield.| Entry | RLi (eq.) | Solvent | 2a (%) | 3a (%) |
|---|---|---|---|---|
| a Reaction conditions: 1a (0.2 mmol), solvent (1 mL), RLi, room temperature (RT), under air (relative humidity = 39%). CPME = cyclopentyl methyl ether, 2-MeTHF = 2-methyltetrahydrofuran, 4-MeTHP = 4-methyltetrahydropyran, DME = 1,2-dimethoxyethane, MTBE = methyl t-butyl ether). LiTMP (1 M in 2-MeTHF), LDA (1 M in 2-MeTHF), t-BuLi (1.7 M in pentane), s-BuLi (1.4 M in cyclohexane). b Determined by 1H NMR using n-heptane as the internal standard. c Unreacted 1a (64%) was recovered. d Yield of isolated 3a = 68%. e The corresponding N,N-diisopropyl-2-oxoacetamide was also detected in the reaction mixture. See the SI for details. f The ortho-rearranged salicylamide (45%) was observed in the crude reaction mixture. See ref. 22d. | ||||
| 1 | LiTMP (2) | CPME | 0 | 34c |
| 2 | LiTMP (2) | 2-MeTHF | 0 | 54 |
| 3 | LiTMP (3) | 2-MeTHF | 0 | 70d |
| 4 | LiTMP (3) | 4-MeTHP | 0 | 53e |
| 5 | LiTMP (3) | DME | 0 | 62e |
| 6 | LiTMP (3) | THF | 0 | 54e |
| 7 | LiTMP (3) | Et2O | 35 | 50 |
| 8 | LiTMP (3) | MTBE | 17 | 48 |
| 9 | LiTMP (3) | n-hexane | 32 | 57 |
| 10 | LDA (3) | 2-MeTHF | 0 | 34 |
| 11 | t-BuLi (3) | 2-MeTHF | 0 | 32 |
| 12 | s-BuLi (3) | 2-MeTHF | 10 | 36e,f |
Scope of the reaction
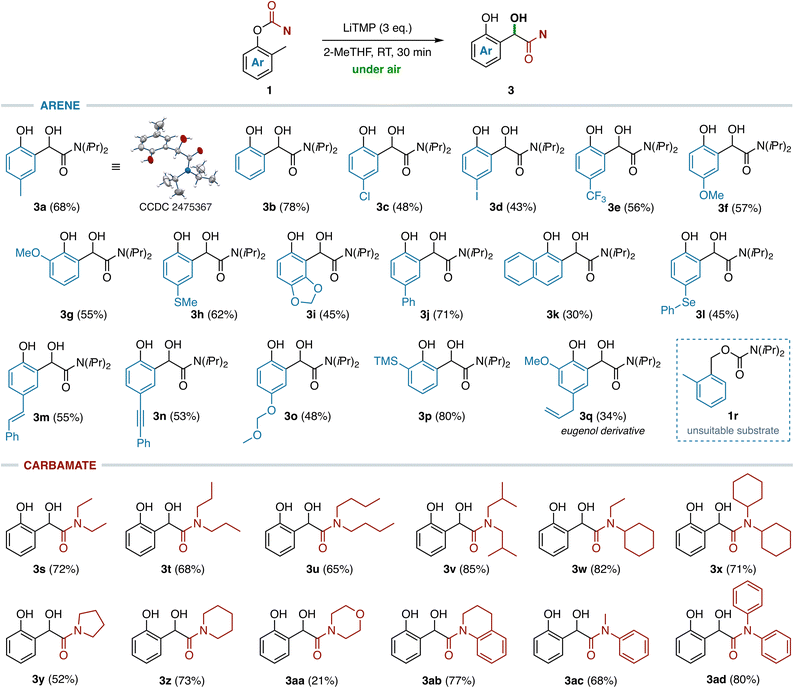
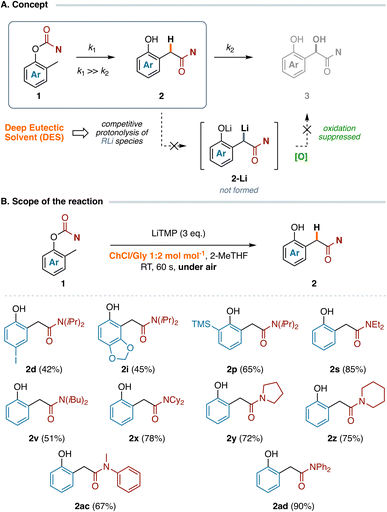
With optimized reaction conditions in hand (Table 1, entry 3), the scope and limitations of this transformation were evaluated for a series of functionalized O-aryl carbamates 1 bearing different substituents at both the O-aryl ring and the amine moiety, using the commodity metalating agent LiTMP in 2-MeTHF under air. The anionic Fries rearrangement of O-aryl N,N-diisopropylcarbamates 1a–q proceeded smoothly en route to a variety of substituted α-hydroxy arylacetamides bearing neutral (3a–b), electron-donating (3f–i and 3o), halogenated (3c and 3e) groups at the aromatic moiety, and polyaromatic rings (3j–k) without formation of byproducts arising from competitive directed ortho-, remote27 or peri-28 metalation processes (Scheme 1). Remarkably, the synthesis of compound 3a has been easily scaled up to 4.0 mmol of carbamate 1a (1 g) with comparable efficiency in terms of yield and selectivity (66% versus 68% on a small scale, see the SI). Pleasingly, our methodology also tolerates the presence of common organolithium-sensitive functional groups, allowing the chemoselective preparation of several polyfunctionalized α-hydroxy arylacetamides without competitive pathways, such as X/Li exchange (3d), transmetalation (3l), carbolithiation (3m–n) or α-lithiation (3h and 3p) reactions. Treatment of carbamate 1q, prepared from the ortho-methyl derivative of eugenol, with LiTMP gave access to the corresponding polysubstituted α-hydroxy arylacetamide 3q, albeit in a modest 34% yield. Other sterically hindered O-tolyl carbamates derived from secondary (cyclo)alkyl (1s–1aa) and aryl (1ab–ac) amines performed as well, thereby delivering a series of α-hydroxylated tertiary acetamides 3s–ad in good yields (21–85%). By contrast, when the reaction was performed on carbamate 1r the sole [1,2]-carbamoyl migration product, arising from the competitive α-lithiation at the O-benzylic position,8b was recovered in 88% yield (see the SI). Similarly, attempts to extend our methodology to 2-ethylphenol derivatives were unsuccessful. Treatment of the N,N-diisopropyl carbamate of 2-ethylphenol under optimized reaction conditions resulted in a partial anionic homo-Fries rearrangement29 which afforded the corresponding non-oxidized arylacetamide in 36% yield (alongside 52% of unreacted starting material), whereas no oxidation products were detected in the reaction mixture (see the SI).Mechanistic investigations
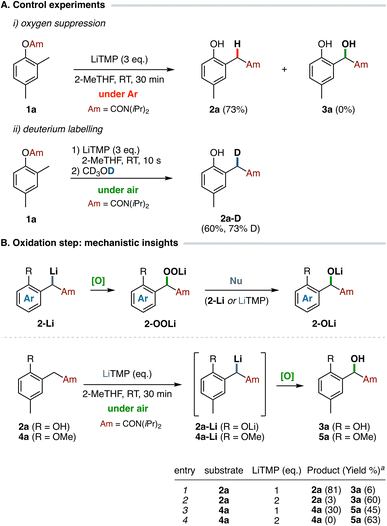
To gain more mechanistic insights into the anionic homo-Fries rearrangement/oxidation sequence, additional control experiments were performed (Scheme 2A). Carbamate 1a was treated with LiTMP (3 equiv.) under an inert atmosphere (Ar), using dry and thoroughly degassed 2-MeTHF as solvent. Under these conditions, the sole arylacetamide 2a arising from the benzylic metalation/anionic Fries rearrangement was isolated in 73% yield, confirming the effective role of molecular oxygen as the oxidant in the reaction sequence.30 Additionally, electrophilic quenching with deuterium of the anion solution generated by metalation of 1a with LiTMP under air yielded compound 2a-D with good deuterium incorporation (73% D) at the benzylic position after only 10 s of metalation, resulting from a fast enolization process occurring after the carbamoyl migration step. Taken together, these results confirmed that our developed protocol, which allows the chemoselective conversion of O-aryl carbamates to α-hydroxy arylacetamides in a single synthetic operation, exploits both a cascade of LiTMP-mediated transformations and the presence of molecular oxygen arising from the use of bench-type conditions.Further mechanistic investigations were subsequently devoted to the oxidation step. The nature of the species involved in the aerobic oxidation of organolithium compounds by molecular oxygen has been clearly established, and oxenoid-type compounds were often proposed as intermediates. The cleavage of the intermediate lithium peroxide is often rationalized by the fast interaction of the electrophilic lithium peroxide with a second molecule of non-oxidized organolithium to form the corresponding lithium alkoxide (autoxidation process).31 In our proposed anionic homo-Fries rearrangement/oxidation sequence, we speculated that the cleavage of the intermediate lithium peroxide 2-OOLi could be performed either by the non-oxidized amide enolate 2-Li (autoxidation) or, alternatively, by the excess of lithium amide LiTMP (Scheme 2B). The latter should account for the required three equivalents of metalating agent (one for each metalation step and an additional equivalent for the cleavage of 2-OOLi species) to achieve a satisfactory yield of hydroxylated acetamide 3, as emerged from the optimization studies (see Table 1, entry 3). Hence, additional experiments were performed on arylacetamide 2a to elucidate the nature of the species involved in the nucleophilic cleavage of the intermediate peroxide 2-OOLi generated during the oxidation step (Scheme 2B). As expected, treatment of 2a with a stoichiometric amount of LiTMP (1 equiv.) was ineffective in promoting the oxidation due to the protonolysis of the metalating agent by the more acidic phenolic hydroxy group, leading to the exclusive formation of the corresponding lithium phenoxide and the consequent recovery of unreacted 2a upon acidic quench (Scheme 2B, entry 1). By contrast, the use of a two-fold amount of LiTMP resulted in the complete α-hydroxylation of 2a owing the effective aerobic oxidation of amide enolate 2a-Li, generated in turn by the additional equivalent of metalating agent (entry 2). These findings strongly suggest a classic autoxidation pathway promoted by the non-oxidized 2a-Li, rather than a LiTMP-induced cleavage of the intermediate oxenoid-type species which should theoretically afford an equimolar mixture of 2a:3a. Interestingly, a similar result was observed upon metalation of the anisole-derived acetamide 4a with a stoichiometric amount of LiTMP (1 equiv.), which afforded an almost 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 mixture of unreacted 4a and α-hydroxy arylacetamide 5a (entry 3). By contrast, complete conversion of 4a was solely observed in the presence of a two-fold excess of metalating agent (entry 4), suggesting in this case a competitive behaviour of LiTMP as nucleophile versus4a-Li in the cleavage of the intermediate lithium peroxide.
1 mixture of unreacted 4a and α-hydroxy arylacetamide 5a (entry 3). By contrast, complete conversion of 4a was solely observed in the presence of a two-fold excess of metalating agent (entry 4), suggesting in this case a competitive behaviour of LiTMP as nucleophile versus4a-Li in the cleavage of the intermediate lithium peroxide.
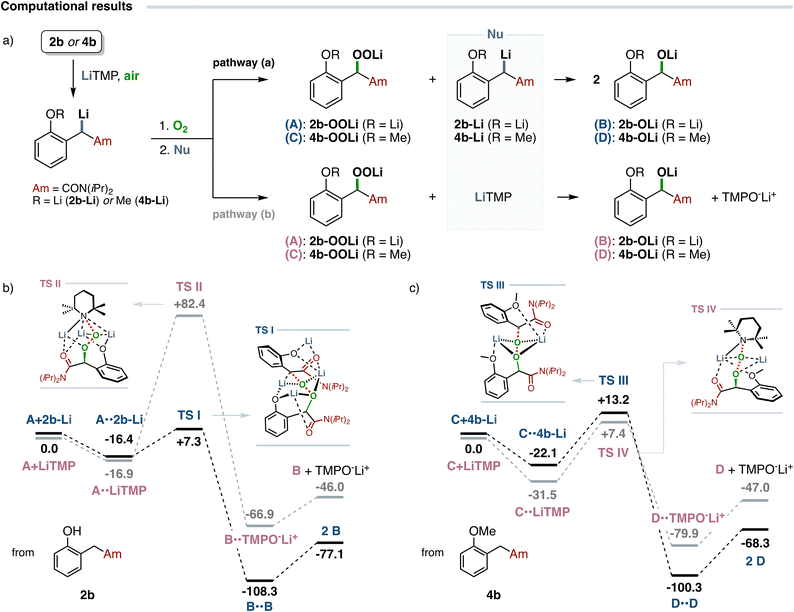
This experimental evidence has been further investigated by DFT calculations. The reaction free energies involved in the nucleophilic cleavage of model oxenoid-type intermediates, bearing a lithium alkoxide (2b-OOLi, A) or a methoxy- (4b-OOLi, C) substituent at the aromatic ring, operated by either the parent non-oxidized organolithiums 2b-Li/4b-Li (pathway a) or LiTMP (pathway b) have been estimated (Scheme 3a). Computational results disclosed that the cleavage of hydroperoxide salt 2b-OOLi (A, Scheme 3b) by the non-oxidized amide enolate 2b-Li,32 according to an established autoxidation pathway which generates two molecules of lithium alkoxide 2b-OLi (B) as products, proceeds through a transition state (TS I) in which the key structural element is a six-membered cyclic (–Li–O–)3 core organized into a chair-like conformation. The significant elongation of the O–O bond (from 1.43 Å in 2b-OOLi to 1.75 Å in TS I), and the almost linear alignment of the former 2b-Li nucleophilic carbon with the electrophilic oxygen in TS I (CLi–O–O angle = 16.3°), strongly suggest a SN2-type process involved in the cleavage of hydroperoxide salt 2b-OOLi.33 The relatively low cleavage barrier of the O–O bond (estimated ΔG‡ = 23.7 kcal mol−1), and the significant thermodynamic stability of the resulting lithium alkoxide with respect to the starting reactants, further corroborate the hypothesis that a classic autoxidation process of amide enolate 2b-Li occurs in the presence of molecular oxygen. Conversely, the interaction of 2b-OOLi with the lithium amide LiTMP (Scheme 3b), which should produce the lithium alkoxide 2b-OLi (B) and the N-hydroxy TMP lithium salt (TMPO−Li+) species, was significantly disfavoured due to the high free energy barrier required for the heterolytic O–O bond cleavage (estimated ΔG‡ = 99.3 kcal mol−1), arising from the formation of a more sterically crowded and less organized transition state (TS II). As emerged from the calculations, the computed geometry of TS II clearly discourages a nucleophilic cleavage pathway on 2b-OOLi operated by LiTMP (O–O = 1.40 Å, NLi–O–O angle = 101.0°).
A different scenario emerged from computational investigations on the oxidation of the metalated anisole-derived arylacetamide 4b (Scheme 3c). In close analogy to the phenoxide-derived intermediate 2b-OOLi, the nucleophilic cleavage of the hydroperoxide salt 4b-OOLi (C) operated by the unoxidized amide enolate 4b-Li proceeds through the formation of an organized heterodimeric transition state (TS III) containing a bent four-membered (–Li–O–)2 cyclic core (estimated ΔG‡ = 35.3 kcal mol−1). Interestingly, in this case the exergonic O–O bond cleavage of the electrophilic 4b-OOLi by LiTMP proceeds via the formation of a relatively more stable transition state TS IV (−5.8 kcal mol−1versusTS III) with a comparable free energy barrier (estimated ΔG‡ = 38.9 kcal mol−1). The geometries of both TSs, showing comparable O–O bond distances (1.73 Å and 1.74 Å for TS III and TS IV, respectively) and an almost linear orientation of the nucleophile with the peroxide moiety (CLi–O–O = 19.2°, TS III and NLi–O–O = 10.5°, TS IV), were in good accordance with those involved in a SN2 cleavage process. Overall, these simulations disclosed that in the presence of a lithium alkoxide group on the aromatic ring (arising from the preliminary anionic Fries rearrangement step) the oxidation of the lithium amide enolate follows a classic autoxidation pathway, where the intermediate hydroperoxide salt interacts with the non-oxidized enolate to yield two molecules of the corresponding lithium alkoxide.34 In contrast, in the absence of a complementary anionic site at the aromatic ring (e.g. in the case of arylacetamides 4a–b) the analysis of the free energy barriers indicates that the lithium amide strongly competes against the amide enolate (or is privileged) in the nucleophilic cleavage of the electrophilic peroxide. These findings are in good agreement with the experimental results (see Scheme 2B), where the complete oxidation of arylacetamides 2a (R = OLi) and 4a (R = OMe) has been achieved in the presence of one (2a, entry 2, autoxidation pathway) or two (4a, entry 4) equivalents of metalating agent, respectively.
Hence, we are inclined to propose the initial benzylic metalation of carbamate 1, followed by a fast anionic 1,4-O-C Fries rearrangement, to form the corresponding intermediate arylacetamide 2, which is promptly enolized by the excess of metalating agent (Scheme 4).35 The resulting 2-Li species then undergoes a spontaneous autoxidation process to deliver the α-hydroxy arylacetamide 3 upon protonolysis of the corresponding alkoxide 2-OLi, generated in situ from the nucleophilic cleavage of the intermediate oxenoid species 2-OOLi by the unoxidized amide enolate 2-Li. Altogether, four synthetic steps are involved in the aerobic tandem protocol developed herein: the regioselective lateral (benzylic) lithiation, an anionic carbamoyl migration, a chemoselective lithiation α to amide carbonyl, and the autoxidation of the resulting amide enolate promoted by oxygen. This allows, overall, the consecutive formation of a C–C and a C–O bond at the same carbon atom in a single synthetic operation.
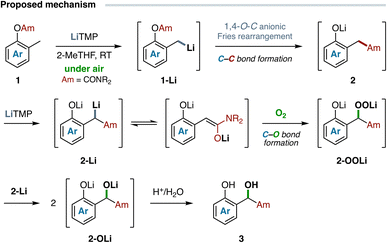 | ||
| Scheme 4 Proposed reaction mechanism based on experimental data and estimated reaction free energies. | ||
Interrupted anionic Fries rearrangement in deep eutectic solvents
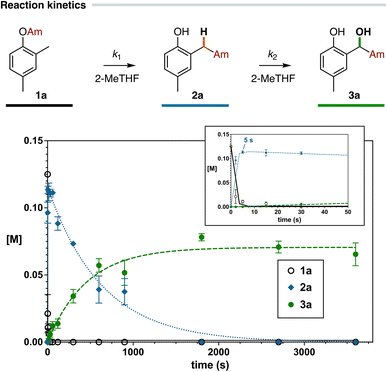
Whereas we have shown that the presence of atmospheric molecular oxygen in the reaction mixture, a commonly detrimental species for organolithium-promoted transformations, could be efficiently exploited to assemble hydroxylated acetamide-based structural motifs, final considerations have been devoted to the possibility of suppressing the anion oxidation step while maintaining the use of bench-type aerobic conditions.Deuterium labelling experiments performed on carbamate 1a (see Scheme 2A) suggest that the first steps of the reaction sequence (anionic Fries rearrangement and enolization) are faster than the oxidation process, as demonstrated by the relatively high deuterium incorporation in the arylacetamide 2a observed after only 10 s. Kinetic studies of the reaction, using the model carbamate 1a (0.2 mmol) as a substrate in 2-MeTHF (1 mL), revealed the classic profile of a sequential transformation (Fig. 2). During the initial stage of the reaction the oxidation product 3a was not formed. Subsequently, a slow consumption of 2a occurred with the concomitant steady production of 3a over 30 min. A comparison of the observed rate constants (k1 and k2)36 confirmed that the oxidation of the amide enolate is the rate determining step of the consecutive process.
Consequently, the different kinetics of these two steps could be exploited to perform a rapid quench of the reaction mixture before the oxidation occurs. The rapid protonolysis of the anion solution (e.g. by performing an external acidic quench) represents the most intuitive and immediate route to avoid the oxidation of the enolized arylacetamide; however this approach could lack reproducibility due to the short reaction times involved. An alternative and more intriguing possibility is offered by the use of protic Deep Eutectic Solvents (DESs), mixtures of specific hydrogen bond donors (HBDs) and acceptors (HBAs) combined in eutectic molar ratios with peculiar physical properties, high biodegradability and biocompatibility.37 Several studies have disclosed the ability of protic DESs to promote the kinetic activation of highly polar s-block organometallic reagents and, consequently, to improve both the reaction time and the chemoselectivity of several organometallic-mediated transformations without the need for external additives.38 Overall, the activating effect of DESs facilitates the reactivity of air-sensitive RLi and RMgX species in a protic and aerobic environment over the competitive protonolysis process, which usually becomes predominant after short reaction times.39 Hence, we envisaged that a combination of the intrinsic properties of protic DESs (acidity and activating effect on organolithiums) with the fast kinetics of the anionic Fries rearrangement step at room temperature could be efficiently exploited to perform in situ a rapid quench of the organolithium species in solution generated upon the metalation/carbamoyl migration process, namely the excess of LiTMP and the amide enolates 2-Li. In principle, this would suppress the formation of the corresponding α-hydroxylated compounds 3, allowing the preparation of arylacetamides 2 in a chemoselective fashion working under bench-type aerobic conditions (Scheme 5A). A series of carbamates 1 (0.2 mmol, 1 M in 2-MeTHF), differently substituted at both the aromatic ring and the amide nitrogen, were thus treated with LiTMP (3 equiv.) using a choline chloride (ChCl)/glycerol (Gly) 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 mol mol−1 deep eutectic mixture as the reaction medium (1 g), under air at RT and with vigorous stirring (Scheme 5B).40
2 mol mol−1 deep eutectic mixture as the reaction medium (1 g), under air at RT and with vigorous stirring (Scheme 5B).40
Pleasingly, under heterogeneous conditions the rearrangement products 2 were exclusively formed within only 60 s in good yields (42–90%), and the corresponding oxidation compounds 3 have never been observed even after a longer reaction time (0.5 h) owing to the efficient protonolysis of the organolithium species by the protic deep eutectic mixture. As expected, when carbamate 1a (0.2 mmol, 1 M in 2-MeTHF) was treated with LiTMP (0.6 mmol) under heterogeneous aerobic conditions, using the ChCl/Gly 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 mol mol−1 deep eutectic mixture as solvent, the kinetic profile of the reaction lacked the formation of the oxidation product, and a fast conversion of 1a into the rearranged arylacetamide 2a was observed (k1 = 3.0 ± 0.8 s−1, see the SI).
2 mol mol−1 deep eutectic mixture as solvent, the kinetic profile of the reaction lacked the formation of the oxidation product, and a fast conversion of 1a into the rearranged arylacetamide 2a was observed (k1 = 3.0 ± 0.8 s−1, see the SI).
Conclusions
In summary, we have developed a general and efficient organolithium-mediated protocol which exploits the presence of molecular oxygen, a commonly detrimental species in the chemistry of polar s-block organometallic reagents, to build poly-substituted scaffolds of remarkable synthetic value in a single synthetic operation. Our methodology enables the transformation of O-aryl carbamates into α-hydroxy arylacetamides by resorting to a one-pot, tandem sequence of C–C and C–O bond formation events occurring at the same carbon atom, working under air and at room temperature, using the biobased 2-MeTHF as solvent. Our strategy allows a significant increase in the structural complexity of a simple molecular skeleton with remarkable functional group tolerance while suppressing the formation of overoxidation products without the need for external additives. Furthermore, we have shown that the use of a protic heterogeneous solvent mixture represents an effective tool to tune the chemoselectivity of the proposed transformation. The fast internal protonolysis of the anion solution operated by the protic reaction medium results in the efficient suppression of the oxidation step, enabling the chemoselective preparation of arylacetamides in an aerobic environment as a result of an interrupted metalation/rearrangement sequence. Overall, our strategy allows for the generation of diverse molecular structures in a chemodivergent fashion by simply changing the nature of the reaction medium, thus enlarging the portfolio of organolithium-mediated transformations under non-conventional conditions. The development of other consecutive anionic migration strategies under bench-type aerobic conditions are under investigation and will be reported in due course.Author contributions
Riccardo Gnavi (methodology, investigation, visualization, writing review and editing); Federica De Nardi and Carolina Meazzo (methodology, investigation, writing review and editing); Simone Ghinato (methodology, investigation); Ettore Grimaldi (investigation); Andrea Maranzana (investigation, software, formal analysis, writing review and editing); Cristina Prandi (supervision, funding acquisition, writing review and editing); and Marco Blangetti (conceptualization, funding acquisition, project administration, supervision, visualization, formal analysis, writing – original draft, writing – review and editing). All authors have given approval to the final version of the manuscript.Conflicts of interest
There are no conflicts to declare.Data availability
CCDC deposition number 2475367 contains the supplementary crystallographic data for this paper.25The data underlying this study are available in the published article and its supplementary information (SI). Supplementary information: general procedures, experimental details, characterization data for both new and known compounds, copies of 1H, 13C, 19F and 2H NMR spectra, and computational details. See DOI: https://doi.org/10.1039/d5sc09227b.
Acknowledgements
We warmly thank Dr Rosangela Santalucia and Dr Emanuele Priola for HRMS and X-ray analyses, respectively. This publication was produced while Mr Riccardo Gnavi was attending the PhD programme in Sustainable Development And Climate Change at the University School for Advanced Studies IUSS Pavia, Cycle XXXIX, with the support of a scholarship financed by the Ministerial Decree no. 118 of 2nd March 2023, based on the NRRP – funded by the European Union-Next Generation EU – Mission 4 “Education and Research”, Component 1 “Enhancement of the offer of educational services: from nurseries to universities” – Investment 3.4 “Advanced teaching and university skills”. This work was also carried out under the framework of the National PRIN 2022 project “Unlocking Greener Metal-assisted Synthetic Tactics by Sustainable Solvents and Technologies” (SUSMET) (Project no. 20228W9TBL) funded by “Unione Europea–Next Generation EU, Missione 4 Componente 2 (CUP: D53D23010260006)”. The computational study was funded by “Unione Europea–Next Generation EU, Missione 4 Componente 1 CUP D13C22001340001 – CN00000013”, Spoke 7- Materials and Molecular Sciences. This research was also supported by EU funding within the MUR PNRR Extended Partnership initiative on Emerging Infectious Diseases (Project no. PE00000007, INF-ACT). The authors acknowledge support from the Project CH4.0 under the MUR program “Dipartimenti di Eccellenza 2023–2027” (CUP: D13C22003520001).Notes and references
- (a) F. Depeint, W. R. Bruce, N. Shangari, R. Mehta and P. J. O'Brien, Chem. Biol. Interact., 2006, 163, 94–112 CrossRef CAS PubMed; (b) M. Sani, D. Belotti, R. Giavazzi, W. Panzeri, A. Volonterio and M. Zanda, Tetrahedron Lett., 2004, 45, 1611–1615 CrossRef CAS; (c) M. Bassetto, S. Ferla, F. Pertusati, S. Kandil, A. D. Westwell, A. Brancale and C. McGuigan, Eur. J. Med. Chem., 2016, 118, 230–243 CrossRef CAS PubMed; (d) S.-L. Zhang, W. Zhang, Z. Yang, X. Hu and K. Y. Tam, Eur. J. Pharm. Sci., 2017, 110, 87–92 CrossRef CAS PubMed; (e) M. R. Wood, K. M. Schirripa, J. J. Kim, S. D. Kuduk, R. K. Chang, C. N. Di Marco, R. M. DiPardo, B.-L. Wan, K. L. Murphy, R. W. Ransom, R. S. L. Chang, M. A. Holahan, J. J. Cook, W. Lemaire, S. D. Mosser, R. A. Bednar, C. Tang, T. Prueksaritanont, A. A. Wallace, Q. Mei, J. Yu, D. L. Bohn, F. C. Clayton, E. D. Adarayn, G. R. Sitko, Y. M. Leonard, R. M. Freidinger, D. J. Pettibone and M. G. Bock, Bioorg. Med. Chem. Lett., 2008, 18, 716–720 CrossRef CAS PubMed.
- (a) T. R. Herrin, J. M. Pauvlik, E. V. Schuber and A. O. Geiszler, J. Med. Chem., 1975, 18, 1216–1223 CrossRef CAS PubMed; (b) Y. Xie, X.-H. Ruan, H.-Y. Gong, Y.-H. Wang, X.-B. Wang, J.-P. Zhang, Q. Li and W. Xue, J. Heterocycl. Chem., 2017, 54, 2644–2649 CrossRef CAS; (c) J. P. Ley and H.-J. Bertram, Tetrahedron, 2001, 57, 1277–1282 CrossRef CAS; (d) H. A. Schenck, P. W. Lenkowski, I. Choudhury-Mukherjee, S.-H. Ko, J. P. Stables, M. K. Patel and M. L. Brown, Biorg. Med. Chem., 2004, 12, 979–993 CrossRef CAS PubMed; (e) G. V. Kaiser, M. Gorman and J. A. Webber, J. Infect. Dis., 1978, 137, S10–S16 CrossRef PubMed; (f) J. Verhaegen and L. Verbist, J. Antimicrob. Chemother., 1998, 41, 381–385 CrossRef CAS PubMed; (g) H. J. Meulenbeld, R. H. J. Mathijssen, J. Verweij, R. de Wit and M. J. A. de Jonge, Expert Opin. Investig. Drugs, 2012, 21, 383–393 CrossRef CAS PubMed.
- (a) A. R. Katritzky, H.-Y. He and K. Suzuki, J. Org. Chem., 2000, 65, 8210–8213 CrossRef CAS PubMed; (b) S. E. Kelly and T. G. LaCour, Synth. Commun., 1992, 22, 859–869 CrossRef CAS.
- (a) J.-J. Zhang, J.-H. Wang, Y.-H. Sang, H.-J. Qin, C. Chen and W. Liu, Org. Biomol. Chem., 2025, 23, 4922–4926 RSC; (b) G. N. Rao and G. Sekar, J. Org. Chem., 2023, 88, 4008–4016 CrossRef CAS PubMed; (c) K. Soai and H. Hasegawa, J. Chem. Soc., Perkin Trans., 1985, 1, 769–772 RSC; (d) N. C. Mamillapalli and G. Sekar, Chem. Commun., 2014, 50, 7881–7884 RSC; (e) S. Golla and H. P. Kokatla, J. Org. Chem., 2022, 87, 9915–9925 CrossRef CAS PubMed; (f) N. C. Mamillapalli and G. Sekar, RSC Adv., 2014, 4, 61077–61085 RSC.
- (a) M. A. Ganiek, M. R. Becker, G. Berionni, H. Zipse and P. Knochel, Chem. Eur. J., 2017, 23, 10280–10284 CrossRef CAS PubMed; (b) A. Nagaki, Y. Takahashi and J.-i. Yoshida, Angew. Chem., Int. Ed., 2016, 55, 5327–5331 CrossRef CAS PubMed; (c) R. F. Cunico, Tetrahedron Lett., 2002, 43, 355–358 CrossRef CAS; (d) Y. Yao, W. Tong and J. Chen, Mendeleev Commun., 2014, 24, 176–177 CrossRef CAS; (e) U. Schöllkopf and H. Beckhaus, Angew. Chem., Int. Ed., 1976, 15, 293 CrossRef.
- (a) T. Yamada, T. Hirose, S. Ōmura and T. Sunazuka, Eur. J. Org Chem., 2015, 2015, 296–301 CrossRef CAS; (b) L. A. Martinho, T. P. F. Rosalba and C. K. Z. Andrade, Eur. J. Org Chem., 2022, 2022, e202201199 CrossRef CAS; (c) M. Serafini, A. Griglio, E. Oberto, T. Pirali and G. C. Tron, Tetrahedron Lett., 2017, 58, 4786–4789 CrossRef CAS; (d) R. Frey, S. G. Galbraith, S. Guelfi, C. Lamberth and M. Zeller, Synlett, 2003, 2003, 1536–1538 Search PubMed; (e) N. Yu, J.-F. Lv, S.-M. He, K.-C. He, W.-H. Zheng, Y.-Q. Zhou, K. Jiang, X.-C. Pan and Y. Wei, Org. Lett., 2024, 26, 1358–1363 CrossRef CAS PubMed.
- A. Liu, S. Sun, Q. Xie, R. Huang, T. Kong, C. Ni and J. Hu, Org. Chem. Front., 2023, 10, 5343–5351 RSC.
- (a) K. Tomooka, H. Shimizu, T. Inoue, H. Shibata and T. Nakai, Chem. Lett., 1999, 28, 759–760 CrossRef; (b) P. Zhang and R. E. Gawley, J. Org. Chem., 1993, 58, 3223–3224 CrossRef CAS; (c) A. K. Ghosh, A. J. Basu, C.-S. Hsu and M. Yadav, Chem.–Eur. J., 2022, 28, e202200941 CrossRef CAS PubMed.
- (a) A. de la Torre, D. Kaiser and N. Maulide, J. Am. Chem. Soc., 2017, 139, 6578–6581 CrossRef CAS PubMed; (b) X. Li, F. Lin, K. Huang, J. Wei, X. Li, X. Wang, X. Geng and N. Jiao, Angew. Chem., Int. Ed., 2017, 56, 12307–12311 CrossRef CAS PubMed.
- (a) R. V. Hoffman, N. K. Nayyar and W. Chen, J. Org. Chem., 1992, 57, 5700–5707 CrossRef CAS; (b) A. J. Clark, Y. S. S. Al-Faiyz, M. J. Broadhurst, D. Patel and J. L. Peacock, J. Chem. Soc., Perkin Trans., 2000, 1, 1117–1127 RSC.
- C. Tang, X. Qiu, Z. Cheng and N. Jiao, Chem. Soc. Rev., 2021, 50, 8067–8101 RSC.
- T. Inukai, T. Kano and K. Maruoka, Org. Lett., 2021, 23, 792–796 CrossRef CAS PubMed.
- R. D. Shinde, A. R. Paraskar, J. Kumar, E. Ghosh, T. K. Paine and S. Bhadra, J. Org. Chem., 2024, 89, 9666–9671 CrossRef CAS PubMed.
- (a) G. Y. Zhang Xinming, T. Guide and Z. Wu, Chin. J. Org. Chem., 2017, 37, 2993–2999 CrossRef; (b) X. Zhang, Y. Yu, W. Li, L. Shi and H. Li, J. Org. Chem., 2022, 87, 16263–16275 CrossRef CAS PubMed.
- (a) R. Stuhr, P. Bayer and A. J. von Wangelin, ChemSusChem, 2022, 15, e202201323 Search PubMed; (b) C. Xu, X. Li and L. Bai, J. Org. Chem., 2022, 87, 4298–4304 CrossRef CAS PubMed; (c) B. Song, S. Wang, C. Sun, H. Deng and B. Xu, Tetrahedron Lett., 2007, 48, 8982–8986 CrossRef CAS.
- (a) Y. Minko and I. Marek, Org. Biomol. Chem., 2014, 12, 1535–1546 RSC; (b) M. Julia, V. Pfeuty-Saint Jalmes, K. Plé, J.-N. Verpeaux and G. Hollingworth, Bull. Soc. Chim. Fr., 1996, 133, 15–24 CAS.
- J. L. Wardell, in Comprehensive Organometallic Chemistry, ed. G. Wilkinson, F. G. A. Stone and E. W. Abel, Pergamon, Oxford, 1982, vol. 1, pp. 46–47 Search PubMed.
- (a) K. A. Parker and K. A. Koziski, J. Org. Chem., 1987, 52, 674–676 Search PubMed; (b) M. Julia, V. P. Saint-Jalmes and J.-N. Verpeaux, Synlett, 1993, 1993, 233–234 Search PubMed; (c) G. Boche and J. C. Lohrenz, Chem. Rev., 2001, 101, 697–756 Search PubMed.
- (a) F. A. Davis and B. C. Chen, Chem. Rev., 1992, 92, 919–934 CrossRef CAS; (b) B.-C. Chen, P. Zhou, F. A. Davis and E. Ciganek, Org. React., 2004, 1–356 Search PubMed.
- (a) M. Ohtawa, M. J. Krambis, R. Cerne, J. M. Schkeryantz, J. M. Witkin and R. A. Shenvi, J. Am. Chem. Soc., 2017, 139, 9637–9644 Search PubMed; (b) M. Y. Kim, J. E. Starrett Jr. and S. M. Weinreb, J. Org. Chem., 1981, 46, 5383–5389 CrossRef CAS; (c) T. L. LaPorte, M. Hamedi, J. S. DePue, L. Shen, D. Watson and D. Hsieh, Org. Process Res. Dev., 2008, 12, 956–966 Search PubMed; (d) S. Hanessian and G. McNaughton-Smith, Bioorg. Med. Chem. Lett., 1996, 6, 1567–1572 CrossRef CAS; (e) H. H. Wasserman and B. H. Lipshutz, Tetrahedron Lett., 1975, 16, 1731–1734 CrossRef; (f) M. Velasco, U. Hernández, J. L. Terán, D. Gnecco, M. L. Orea, D. M. Aparicio, V. Gómez-Calvario, S. Bernès and J. R. Juárez, Tetrahedron Lett., 2019, 60, 820–824 CrossRef CAS; (g) J. C. Estévez, R. J. Estévez and L. Castedo, Tetrahedron, 1995, 51, 10801–10810 CrossRef; (h) H. Lubin, A. Tessier, G. Chaume, J. Pytkowicz and T. Brigaud, Org. Lett., 2010, 12, 1496–1499 CrossRef CAS PubMed.
- M. J. Rodríguez-Álvarez, N. Ríos-Lombardía, S. E. García-Garrido, C. Concellón, V. del Amo, V. Capriati and J. García-Álvarez, Molecules, 2024, 29 CrossRef PubMed.
- (a) F. Marra, F. Morelli, F. De Nardi, E. Priola, I. Coldham and M. Blangetti, Org. Biomol. Chem., 2025, 23, 8029–8038 RSC; (b) F. Marra, F. De Nardi, F. Rossi, E. Priola, C. Prandi and M. Blangetti, Eur. J. Org Chem., 2024, 27, e202400313 CrossRef CAS; (c) S. Ghinato, C. Meazzo, F. De Nardi, A. Maranzana, M. Blangetti and C. Prandi, Org. Lett., 2023, 25, 3904–3909 CrossRef CAS PubMed; (d) S. Ghinato, F. De Nardi, P. Bolzoni, A. Antenucci, M. Blangetti and C. Prandi, Chem. Eur. J., 2022, 28, e202201154 CrossRef CAS PubMed; (e) S. Ghinato, D. Territo, A. Maranzana, V. Capriati, M. Blangetti and C. Prandi, Chem. Eur. J., 2021, 27, 2868–2874 Search PubMed.
- M. Korb and H. Lang, Chem. Soc. Rev., 2019, 48, 2829–2882 RSC.
- P. M. P. Ferreira, K. D. C. Machado, S. N. Lavorato, F. C. E. Oliveira, J. D. N. Silva, A. A. C. D. Almeida, L. D. S. Santos, V. R. Silva, D. P. Bezerra, M. B. P. Soares, C. Pessoa, M. O. D. Moraes Filho, J. R. D. O. Ferreira, J. M. Sousa, V. G. Maltarollo and R. J. Alves, Toxicol. Appl. Pharmacol., 2019, 380, 114692 CrossRef CAS PubMed.
- CCDC 2475367 (compound 3a) Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2p2tk5.
- F. F. Mulks, L. J. Bole, L. Davin, A. Hernán-Gómez, A. Kennedy, J. García-Álvarez and E. Hevia, Angew. Chem., Int. Ed., 2020, 59, 19021–19026 CrossRef CAS PubMed.
- D. Tilly, J. Magolan and J. Mortier, Chem.–Eur. J., 2012, 18, 3804–3820 Search PubMed.
- J. Clayden, C. S. Frampton, C. McCarthy and N. Westlund, Tetrahedron, 1999, 55, 14161–14184 CrossRef CAS.
- (a) A. V. Kalinin, M. A. J. Miah, S. Chattopadhyay, M. Tsukazaki, M. Wicki, T. Nguen, A. L. Coelho, M. Kerr and V. Snieckus, Synlett, 1997, 1997, 839–841 CrossRef; (b) J. Fässler, J. A. McCubbin, A. Roglans, T. Kimachi, J. W. Hollett, R. W. Kunz, M. Tinkl, Y. Zhang, R. Wang, M. Campbell and V. Snieckus, J. Org. Chem., 2015, 80, 3368–3386 CrossRef PubMed.
- The hydroxylated compound 3a was not observed under these conditions. When the metalation was performed under Ar for 5 min, and the reaction was then stirred under air for 30 min, compound 3a was produced in 70% yield after 30 min.
- G. Sosnovsky and J. H. Brown, Chem. Rev., 1966, 66, 529–566 Search PubMed.
- Calculated CC-Li–CC
![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O bond length: 1.39 Å.
O bond length: 1.39 Å. - G. Boche, K. Möbus, K. Harms, J. C. W. Lohrenz and M. Marsch, Chem. Eur. J., 1996, 2, 604–607 CrossRef CAS PubMed.
- Treatment of 2a-Li species with 1 eq. of t-BuOOLi (0.5 M in 2-MeTHF) afforded compound 3a in 43% yield after workup. See SI for details.
- S. R. Wang, S. An, Y.-X. Xin, Y.-Y. Jiang and W. H. Liu, Estimated ΔG of enolization for compound 2b = –13.1 kcal mol−1 at 298 K (see SI for details). Data are in good accordance with previously reported results, JACS Au, 2024, 4, 4435–4444 CrossRef PubMed.
- Estimated by nonlinear least-squares fit of the experimental kinetic data using the integrated rate laws for a consecutive transformation.
- B. B. Hansen, S. Spittle, B. Chen, D. Poe, Y. Zhang, J. M. Klein, A. Horton, L. Adhikari, T. Zelovich, B. W. Doherty, B. Gurkan, E. J. Maginn, A. Ragauskas, M. Dadmun, T. A. Zawodzinski, G. A. Baker, M. E. Tuckerman, R. F. Savinell and J. R. Sangoro, Chem. Rev., 2021, 121, 1232–1285 Search PubMed.
- (a) L. Cicco, A. Fombona-Pascual, A. Sánchez-Condado, G. A. Carriedo, F. M. Perna, V. Capriati, A. Presa Soto and J. García-Álvarez, ChemSusChem, 2020, 13, 4967–4973 Search PubMed; (b) A. W. J. Platten, I. Manasi, M. Campana, K. J. Edler and E. Hevia, ChemSusChem, 2025, 18, e202402083 CrossRef CAS PubMed; (c) C. Vidal, J. García-Álvarez, A. Hernán-Gómez, A. R. Kennedy and E. Hevia, Angew. Chem., Int. Ed., 2014, 53, 5969–5973 CrossRef CAS PubMed; (d) G. Dilauro, M. Dell'Aera, P. Vitale, V. Capriati and F. M. Perna, Angew. Chem., Int. Ed., 2017, 56, 10200–10203 CrossRef CAS PubMed; (e) A. Sánchez-Condado, G. A. Carriedo, A. Presa Soto, M. J. Rodríguez-Álvarez, J. García-Álvarez and E. Hevia, ChemSusChem, 2019, 12, 3134–3143 Search PubMed; (f) G. Dilauro, C. Luccarelli, A. F. Quivelli, P. Vitale, F. M. Perna and V. Capriati, Angew. Chem., Int. Ed., 2023, 62, e202304720 Search PubMed; (g) S. Ghinato, G. Dilauro, F. M. Perna, V. Capriati, M. Blangetti and C. Prandi, Chem. Commun., 2019, 55, 7741–7744 RSC; (h) D. Arnodo, S. Ghinato, S. Nejrotti, M. Blangetti and C. Prandi, Chem. Commun., 2020, 56, 2391–2394 Search PubMed.
- Half-life times of aryllithiums (6.26 s) and benzyllithiums (6.57 s) in the choline chloride (ChCl)/glycerol (Gly) 1
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 mol mol−1 deep eutectic mixture have been previously estimated. See ref. 38g and 38h.
2 mol mol−1 deep eutectic mixture have been previously estimated. See ref. 38g and 38h. - In the absence of 2-MeTHF as an additive no lithiation occurred and the unreacted starting materials 1 were quantitatively recovered after workup.
| This journal is © The Royal Society of Chemistry 2026 |