Open Access Article
Open Access ArticleUnraveling the positive effect of twisted helicene structure on narrowband electroluminescence
Cheng-Zhuo
Du†
a,
Minqiang
Mai†
b,
Pei-Han
Gao
a,
Yi-Chao
Zhao
a,
Xiang-Yu
Gao
a,
Dongdong
Zhang
*b,
Lian
Duan
 *b,
Chunming
Cui
*b,
Chunming
Cui
 a and
Xiao-Ye
Wang
a and
Xiao-Ye
Wang
 *ac
*ac
aState Key Laboratory of Elemento-Organic Chemistry, Frontiers Science Center for New Organic Matter, Haihe Laboratory of Sustainable Chemical Transformations, Academy for Advanced Interdisciplinary Studies, College of Chemistry, Nankai University, Tianjin, 300071, China. E-mail: xiaoye.wang@nankai.edu.cn; Web: http://wang.nankai.edu.cn
bKey Lab of Organic Optoelectronics and Molecular Engineering of Ministry of Education, Department of Chemistry, Tsinghua University, Beijing, 100084, China. E-mail: ddzhang@mail.tsinghua.edu.cn; duanl@mail.tsinghua.edu.cn
cBeijing National Laboratory for Molecular Sciences, Beijing, 100190, China
First published on 12th January 2026
Abstract
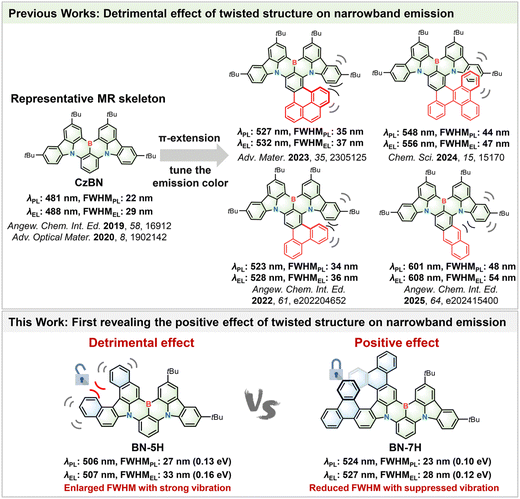
Multi-resonance (MR) materials based on 1,4-BN-heteroarenes have attracted extensive attention in recent years for their narrowband electroluminescence. Extending the π-conjugation of MR skeletons is a widely adopted strategy to regulate their emission colors, but it inevitably induces structural distortion and undesirable vibronic couplings, thus broadening the emission bandwidth. Herein, we design and synthesize new MR emitters via π-extension of a classic MR backbone (CzBN) and disclose how the twisted structure plays a positive role in reducing the emission bandwidth. Specifically, π-extension of CzBN to form a [5]helicene substructure (BN-5H) induces serious vibrations, while further extending the helicene moiety to build a [7]helicene substructure (BN-7H) suppresses undesirable vibrations by locking the conformation. As a consequence, BN-7H achieves a smaller full-width at half-maximum (FWHM) of 28 nm compared with BN-5H (33 nm) in organic light-emitting diodes and longer device lifetime. These results overturn the traditional cognition of the detrimental effect of highly twisted structures on narrowband emission and offer a new design concept for the future development of narrowband electroluminescence materials.
Introduction
Narrowband organic light-emitting diodes (OLEDs) are highly desirable for the future ultrahigh-definition displays with a wide color gamut.1 In 2016, Hatakeyama et al. developed a new type of multi-resonance (MR) materials by employing the opposite resonance effect of boron and nitrogen to separate the frontier molecular orbitals on different atoms,2 leading to narrowband emission. As a representative MR skeleton, CzBN has been widely studied due to its merits of easy functionalization and excellent device performance (Fig. 1).3–11 Extending the π-conjugation of CzBN by fusing benzene rings has been proven as a viable strategy to tune the emission color. Particularly, a number of green emitters for narrowband OLEDs have been developed in recent years based on this strategy.12–17 However, upon extension of the π-skeleton, the steric repulsion between the spatially close benzene rings inevitably intensifies structural distortion and leads to additional vibrations, thus broadening the emission spectra, with full-widths at half-maximum (FWHMs) mostly exceeding 30 nm. Therefore, it is generally believed that enhancing the planarity of the MR skeleton is beneficial to achieving narrowband emission.3,18–21Herein, we design and synthesize new MR emitters (BN-5H and BN-7H) via π-extension of CzBN and disclose the positive effect of the twisted structure on narrowband electroluminescence (Fig. 1). BN-5H, comprising a [5]helicene substructure, exhibits serious structural vibrations similar to those of previously reported CzBN derivatives.13,17 In contrast, BN-7H, with an elongated [7]helicene moiety, significantly suppresses the undesirable vibrations by locking the conformation. Moreover, the highly twisted structure of BN-7H also suppresses solid-state emission quenching and spectral broadening, which are common problems for planar MR emitters.22–24 Therefore, BN-7H achieves a smaller FWHM of only 28 nm (0.12 eV) compared with BN-5H (33 nm/0.16 eV) in OLEDs. The FWHM of BN-7H is the smallest among the reported green MR materials based on benzene-fused CzBN derivatives. Besides, a high maximum external quantum efficiency (EQEmax) of 26.2% and long operation lifetime of 560.1 h at 1000 cd m−2 are obtained for BN-7H-based devices. These results shed new light on the positive role of twisted helicene structures for narrowband electroluminescence materials.
Results and discussion
The synthetic routes to BN-5H and BN-7H are shown in Scheme 1. Although aza[5]helicene (5H) is commercially available, the higher homologue aza[7]helicene requires complicated synthesis in eight steps.25,26 We thus adopted dibenzoaza[7]helicene (7H), which can be easily synthesized in only two steps,27 as a building block to access the target compound BN-7H. First, precursors 5H-Br and 7H-Br were synthesized through aromatic nucleophilic substitution of compound 1 with 5H and 7H, respectively. Then, BN-5H and BN-7H were obtained via one-pot borylation in yields of 36% and 51%, respectively (for details, see the SI). Thermogravimetric analysis (TGA) indicates that BN-5H and BN-7H possess excellent thermal stability with high decomposition temperatures (Td) of 503 °C for BN-5H and 547 °C for BN-7H (Fig. S1), assuring the feasibility of thermal sublimation for further purification and OLED fabrications. According to the cyclic voltammetry (CV) characterizations (Fig. S2 and Table S1), the highest occupied molecular orbital (HOMO) and lowest unoccupied molecular orbital (LUMO) energy levels are calculated to be −5.42/–2.97 eV for BN-5H and −5.40/–3.01 eV for BN-7H, respectively. Therefore, the corresponding electrochemical energy gaps (Egap) are deduced as 2.45 eV and 2.39 eV for BN-5H and BN-7H, respectively.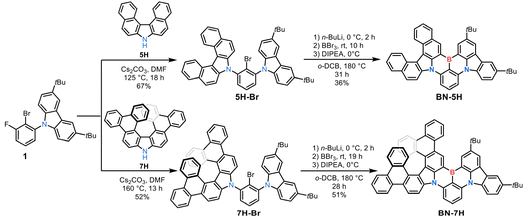 | ||
| Scheme 1 Synthetic routes to BN-5H and BN-7H. DMF: N,N-dimethylformamide; DIPEA: N,N-diisopropylethylamine; and o-DCB: 1,2-dichlorobenzene. | ||
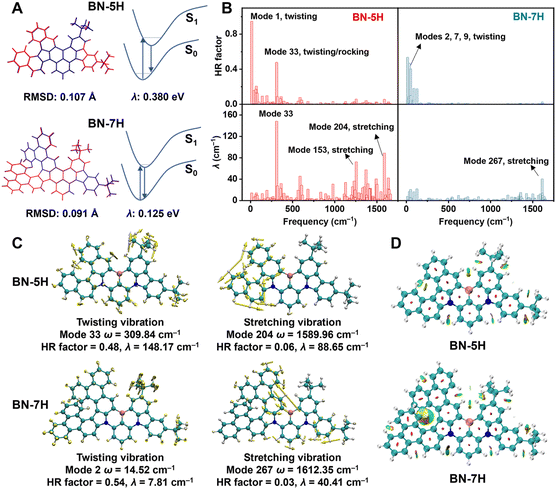
To further understand the geometric and electronic properties of BN-5H and BN-7H, time-dependent density functional theory (TD-DFT) calculations were performed at the B3LYP/6-311G(d,p) level. Compared with BN-5H, BN-7H features a more twisted geometry, with a dihedral angle of 44.98° between the terminal benzene rings (Fig. S13). According to the optimized geometries, the root-mean-square deviation (RMSD) values between S0 and S1 states were calculated to be 0.107 Å and 0.091 Å for BN-5H and BN-7H, respectively (Fig. 2A). Moreover, the total reorganization energy (λ) of BN-7H (0.125 eV) is much smaller than that of BN-5H (0.380 eV). These results indicate that the structural relaxation is effectively suppressed by further extending the helicene moiety.
To gain a deeper understanding of the vibronic coupling of BN-5H and BN-7H, the Huang-Rhys (HR) factors and λ at different vibrational modes were calculated using the Molecular Materials Property Prediction Package (MOMAP) software (Fig. 2B).28 For BN-5H, two large HR factors of 0.95 and 0.48 are observed in the low-frequency region, which are related to the out-of-plane twisting vibrations of the MR framework and the peripheral tert-butyl groups (Modes 1 and 33). Moreover, the high-frequency stretching vibrational modes 153 (1254.78 cm−1) and 204 (1589.96 cm−1) of BN-5H contribute significantly to λ (Fig. S14), which may induce obvious spectral broadening. In contrast, BN-7H displays significantly suppressed out-of-plane skeleton twisting vibrations (Modes 2, 7, 9) with smaller HR factors (0.54, 0.47, 0.40) in the region 14.52–50.94 cm−1. The suppressed vibrations of BN-7H can be attributed to the strong intramolecular noncovalent interactions from the terminal benzene rings, which can be visualized by the reduced density gradient (RDG) analysis (Fig. 2D).29 Besides, compared with BN-5H, the high-frequency stretching vibrations of BN-7H are almost negligible (Fig. S15). These results reveal the feasibility of introducing highly twisted structure to restrain common vibrations of MR emitters.
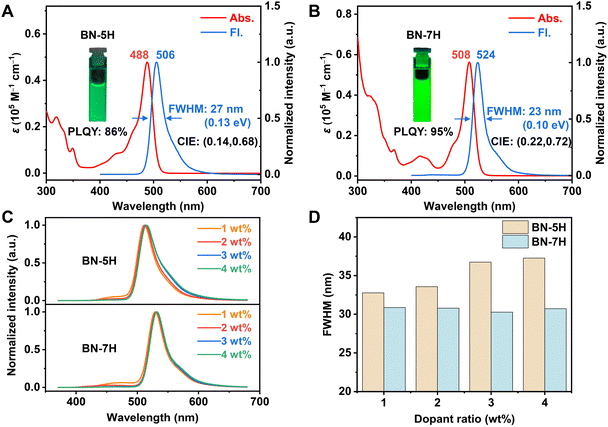
The photophysical properties of BN-5H and BN-7H were further studied in toluene solutions (1 × 10−5 M) (Fig. 3A and B), and the detailed data are summarized in Table 1. BN-5H and BN-7H exhibit strong absorption bands peaking at 488 nm and 508 nm, with molar extinction coefficients (ε) of 47![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 830 M−1 cm−1 and 56
830 M−1 cm−1 and 56![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 320 M−1 cm−1, respectively. The fluorescence maximum of BN-7H (524 nm) shows an obvious red-shift of 18 nm compared with that of BN-5H (506 nm) due to the π-extension. Benefiting from the suppressed vibrations by locking the conformation, BN-7H achieves a smaller FWHM value of only 23 nm (0.10 eV) in toluene than BN-5H (27 nm/0.13 eV). The Commission Internationale de L'Eclairage (CIE) coordinate of BN-7H is (0.22, 0.72), meeting the green color requirement of (0.21, 0.71) stipulated by the National Television System Committee (NTSC). Moreover, BN-7H exhibits a higher photoluminescence quantum yield (PLQY) of 95% than BN-5H (86%) due to the suppression of undesirable vibrations.
320 M−1 cm−1, respectively. The fluorescence maximum of BN-7H (524 nm) shows an obvious red-shift of 18 nm compared with that of BN-5H (506 nm) due to the π-extension. Benefiting from the suppressed vibrations by locking the conformation, BN-7H achieves a smaller FWHM value of only 23 nm (0.10 eV) in toluene than BN-5H (27 nm/0.13 eV). The Commission Internationale de L'Eclairage (CIE) coordinate of BN-7H is (0.22, 0.72), meeting the green color requirement of (0.21, 0.71) stipulated by the National Television System Committee (NTSC). Moreover, BN-7H exhibits a higher photoluminescence quantum yield (PLQY) of 95% than BN-5H (86%) due to the suppression of undesirable vibrations.
| Compound | λ abs (nm) | λ PL (nm) | FWHMb (nm/eV) | ΔESTc (eV) | HOMO/LUMOd (eV) | E g CV (eV) | PLQYe (%) | τ (ns) | k r (108 s−1) | k nr (107 s−1) | T d/Tgh (°C) | CIE (x, y)i |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| a Maximum wavelength of UV-vis absorption and fluorescence spectra in toluene solution (1 × 10−5 M) at 298 K. b FWHMs of the fluorescence spectra. c Obtained from the peak of the fluorescence and phosphorescence spectra in toluene solution at 77 K. d Obtained from the onset potentials of the first oxidative and reductive waves of CV curves, respectively. e Absolute photoluminescence quantum yield measured in oxygen-free toluene solution. f Fluorescence lifetime. g Radiative decay rate constant and non-radiative decay rate constant. h Decomposition temperature (Td) and glass transition temperature (Tg). i Commission Internationale de L'Eclairage (CIE) coordinates in the CIE 1931 chromaticity diagram. | ||||||||||||
| BN-5H | 488 | 506 | 27/0.13 | 0.50 | −5.42/–2.97 | 2.45 | 86 | 5.3 | 1.6 | 2.6 | 503/- | (0.14, 0.68) |
| BN-7H | 508 | 524 | 23/0.10 | 0.29 | −5.40/–3.01 | 2.39 | 95 | 4.0 | 2.4 | 1.3 | 547/258 | (0.22, 0.72) |
The transient photoluminescence spectra were next examined, and no obvious delayed components were observed in degassed solutions (Fig. S5), implying that these two emitters are fluorescence materials without thermally activated delayed fluorescence (TADF) characteristics, which may be associated with the large singlet-triplet energy gaps (ΔEST) of 0.50 eV for BN-5H and 0.29 eV for BN-7H (Fig. S3).30–33 The reduction of ΔEST of BN-7H is further investigated by the hole–electron analysis (Fig. S12). The fluorescence lifetimes (τ) were determined to be 5.3 ns and 4.0 ns for BN-5H and BN-7H, respectively, and the corresponding radiative decay rates (kr) and nonradiative decay rates (knr) of BN-5H and BN-7H were calculated and are summarized in Table 1. BN-7H displays a faster radiative decay process with kr of 2.4 × 108 s−1 and a significantly suppressed knr of only 1.3 × 107 s−1, while BN-5H suffers from a more serious energy loss with knr of 2.6 × 107 s−1. This difference indicates that the more twisted structure of BN-7H can lock the flexible conformation and suppress undesirable vibrations to reduce nonradiative energy dissipation.
The emission properties of the two compounds in solid state were then investigated (Fig. 3C), and 1,3-dihydro-1,1-dimethyl-3-(3-(4,6-diphenyl-1,3,5-triazin-2-yl)phenyl)indeno[2,1-b]carbazole (DMIC-TRZ) was chosen as the wide-energy-gap host.34 As the dopant concentration increases, the emission spectra of BN-5H doped films are significantly broadened with FWHM values increasing from 33 nm to 37 nm, while the FWHMs of BN-7H-doped films are basically unchanged (FWHM: 31 nm) (Fig. 3D). Moreover, the concentration-dependent emission quenching of BN-7H is also efficiently suppressed compared with BN-5H (Table S2). These results demonstrate the beneficial effect of highly twisted structures beyond vibrational suppression at the molecular level.
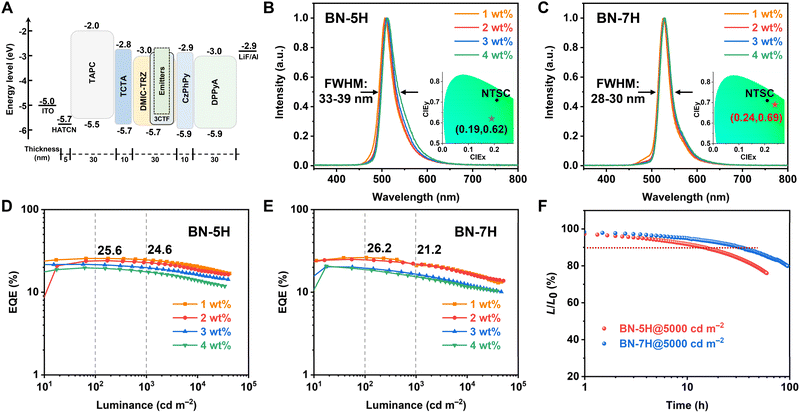
Considering that BN-5H and BN-7H are fluorescence emitters, TADF-sensitized OLEDs were fabricated to evaluate the electroluminescent (EL) properties. The corresponding energy level scheme and chemical structures of the materials used are depicted in Fig. 4A and S17. Particularly, 2,4,6-tris(2-(3,6-di-tert-butyl-9H-carbazol-9-yl)-5-(trifluoromethyl)phenyl)-1,3,5-triazine (3CTF) was chosen as the TADF sensitizer because of its small molecular dipole moment, substantial spectral overlap with these emitters, and high reverse intersystem crossing rate of 2.57 × 106 s−1 (Fig. S18).35 The EL characteristics are summarized in Table 2. Obviously, the devices based on BN-5H show lower tolerance to the dopant concentration, and the FWHM increases significantly with higher dopant concentrations (Fig. 4B). By contrast, the BN-7H-based devices display nearly unchanged emission peaks and FWHMs with increased dopant concentrations, and pure green emissions peaking at 527–529 nm with small FWHMs of only 28–30 nm (0.12–0.13 eV) were recorded (Fig. 4C). Such a result is one of the smallest FWHMs among the reported sensitized green OLEDs (Table S5).3,36–43 Besides, the CIE coordinates of BN-7H devices reach (0.24, 0.69), which is very close to the pure green requirement of the NTSC standard. Moreover, BN-7H shows outstanding device performance with EQEmax, current efficiency (CE), and power efficiency (PE) of 26.2%, 102.3 cd A−1, and 106.5 lm W−1, respectively (Fig. 4E). Meanwhile, the efficiency roll-off is especially low with the EQE remaining 21.2% at 1000 cd m−2.
| Emitter | Dopant concentration (wt%) | λ EL (nm) | FWHMb (nm/eV) | V on (V) | L max (cd m−2) | CEmaxe (cd A−1) | PEmaxf (lm W−1) | EQEg (%) | CIE (x, y)h |
|---|---|---|---|---|---|---|---|---|---|
| a The maximum EL wavelength. b FWHM of the EL spectrum. c Turn-on voltage at 1 cd m−2. d Maximum luminance. e Maximum CE. f Maximum PE. g Maximum EQE, and values at 100 cd m−2 and 1000 cd m−2. h CIE coordinates of the devices in the CIE 1931 chromaticity diagram. | |||||||||
| BN-5H | 1 | 507 | 34/0.16 | 2.6 | 42![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 450 450 |
77.1 | 78.2 | 25.6/25.6/24.6 | (0.19.0.62) |
| 2 | 509 | 33/0.16 | 2.6 | 40![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 090 090 |
76.6 | 74.8 | 24.1/24.0/22.9 | (0.19.0.65) | |
| 3 | 511 | 35/0.17 | 2.7 | 39![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 750 750 |
71.8 | 73.9 | 21.8/21.6/19.8 | (0.20.0.66) | |
| 4 | 513 | 39/0.18 | 2.7 | 34![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 360 360 |
67.7 | 67.0 | 19.7/19.3/17.6 | (0.23.0.67) | |
| BN-7H | 1 | 527 | 28/0.12 | 2.6 | 47![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 370 370 |
102.3 | 106.5 | 26.2/26.2/21.2 | (0.24.0.69) |
| 2 | 527 | 28/0.12 | 2.6 | 51![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 720 720 |
99.8 | 110.8 | 25.0/24.6/22.0 | (0.25.0.69) | |
| 3 | 527 | 29/0.13 | 2.6 | 46![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 890 890 |
81.0 | 90.9 | 20.3/19.7/16.6 | (0.26.0.69) | |
| 4 | 529 | 30/0.13 | 2.6 | 38![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 610 610 |
82.0 | 92.0 | 20.7/18.8/15.4 | (0.27.0.69) | |
We further evaluated the operational stabilities of these devices under an initial luminance (L0) of 5000 cd m−2 (Fig. 4F). The LT90 (lifetime to 90% of the initial luminance) of 14.5 and 33.5 h were recorded for BN-5H and BN-7H, respectively. By adopting a degradation acceleration factor (n) of 1.75,44 the LT90 at 1000 cd m−2 can be extrapolated as 242.4 h for BN-5H and 560.1 h for BN-7H, respectively. The device lifetime of BN-7H is more than twice that of BN-5H, indicating the advantages of the highly twisted structure in improving device stability.
Furthermore, it is noteworthy that BN-7H is chiral, with higher configurational stability than BN-5H due to the spatial overlapping between the terminal benzene rings (Fig. S26). Therefore, the P and M enantiomers of BN-7H were successfully separated by chiral high-performance liquid chromatography (HPLC) (Fig. S22). BN-7H exhibits circular dichroism (CD) responses with the largest absolute absorption dissymmetry factors (|gabs|) of 0.009 at 379 nm (Fig. S24), as well as circularly polarized luminescence (CPL) with the luminescence dissymmetry factors (|glum|) of 0.7 × 10−3 at 524 nm and CPL brightness (BCPL) of 19.0 M−1 cm−1 (Fig. S25), which are comparable to most of the reported circularly polarized MR materials (Table S6).45–49 However, due to the limited solubility hindering the large-scale chiral separation, the EL properties of BN-7H enantiomers were not studied. Further optimizing the molecular structures and chiral separation conditions can provide promising materials for circularly polarized OLEDs with narrowband emissions in the future.14,50–54
Conclusions
In summary, we have designed and synthesized two new MR emitters (BN-5H and BN-7H) via π-extension on one carbazole moiety of the classic MR backbone CzBN and disclosed the positive role of the twisted structure in achieving narrowband OLEDs. BN-5H with a [5]helicene substructure suffers from serious structural vibrations, which is similar to other reported twisted MR emitters. In sharp contrast, BN-7H, with a [7]helicene moiety, significantly suppresses the undesirable vibrations by locking the conformation. Furthermore, the twisted structure of BN-7H also more effectively suppresses solid-state spectral broadening and fluorescence quenching compared with BN-5H. As a result, BN-7H achieves a smaller FWHM of only 28 nm (0.12 eV) compared with BN-5H (33 nm/0.16 eV) in OLEDs. Additionally, the highly twisted skeleton endows BN-7H with a high EQEmax of 26.2% and long device lifetime of 560.1 h at 1000 cd m−2. This work has overturned the traditional cognition of the detrimental effect of twisted structure on narrowband emission and opened up a new avenue to the development of narrowband electroluminescence materials.Author contributions
Xiao-Ye Wang, Lian Duan, Dongdong Zhang and Chunming Cui conceived the project. Cheng-Zhuo Du, Pei-Han Gao, and Yi-Chao Zhao performed the synthesis and characterization of all materials. Minqiang Mai performed the photophysical measurements of the doped films and fabricated the devices. Xiang-Yu Gao characterized the chiroptical properties. Cheng-Zhuo Du performed the theoretical calculations. Xiao-Ye Wang and Cheng-Zhuo Du wrote the original draft. All authors discussed the progress of the research and commented on the manuscript.Conflicts of interest
There are no conflicts of interest to declare.Data availability
The data supporting this article have been included as part of the supplementary information (SI). Supplementary information: experimental details, synthetic procedures, characterization data, NMR spectra, theoretical calculations and electroluminescence data. See DOI: https://doi.org/10.1039/d5sc08405a.Acknowledgements
This work was financially supported by the National Key Research and Development Program of China (2022YFB4200600), the National Natural Science Foundation of China (22375106, 22221002, and 92256304), the Beijing National Laboratory for Molecular Sciences (BNLMS2023013), and the Haihe Laboratory of Sustainable Chemical Transformations. We gratefully acknowledge HZWTECH for providing computation facilities.Notes and references
- M. Sugawara, S. Y. Choi and D. Wood, IEEE Signal Process. Mag., 2014, 31, 170–174 Search PubMed
.
- T. Hatakeyama, K. Shiren, K. Nakajima, S. Nomura, S. Nakatsuka, K. Kinoshita, J. Ni, Y. Ono and T. Ikuta, Adv. Mater., 2016, 28, 2777–2781 Search PubMed
.
- J. Liu, Y. Zhu, T. Tsuboi, C. Deng, W. Lou, D. Wang, T. Liu and Q. Zhang, Nat. Commun., 2022, 13, 4876 CrossRef CAS PubMed
.
- L. Xing, J. Wang, W.-C. Chen, B. Liu, G. Chen, X. Wang, J.-H. Tan, S. S. Chen, J.-X. Chen, S. Ji, Z. Zhao, M.-C. Tang and Y. Huo, Nat. Commun., 2024, 15, 6175 CrossRef CAS PubMed
.
- Y. Pu, Q. Jin, Y. Zhang, C. Li, L. Duan and Y. Wang, Nat. Commun., 2025, 16, 332 CrossRef PubMed
.
- S. Madayanad Suresh, D. Hall, D. Beljonne, Y. Olivier and E. Zysman-Colman, Adv. Funct. Mater., 2020, 30, 1908677 CrossRef CAS
.
- Y. Xu, Q. Wang, X. Cai, C. Li, S. Jiang and Y. Wang, Angew. Chem., Int. Ed., 2023, 62, e202312451 CrossRef CAS PubMed
.
- M. Mamada, M. Hayakawa, J. Ochi and T. Hatakeyama, Chem. Soc. Rev., 2024, 53, 1624–1692 Search PubMed
.
- J. Kang, D. J. Shin and J. Y. Lee, Adv. Opt. Mater., 2025, 13, 2402653 CrossRef CAS
.
- Y. Zhang, C.-Z. Du, J.-K. Li and X.-Y. Wang, Chin. J. Org. Chem., 2023, 43, 1645–1690 Search PubMed
.
- Y.-H. He, J.-Y. Liu, Z. Zhang, G.-W. Chen, Y.-C. Wang, G. Yuan, F.-M. Xie, J.-X. Tang and Y.-Q. Li, Matter, 2025, 8, 102188 Search PubMed
.
- Y. Zhang, D. Zhang, J. Wei, X. Hong, Y. Lu, D. Hu, G. Li, Z. Liu, Y. Chen and L. Duan, Angew. Chem., Int. Ed., 2020, 59, 17499–17503 CrossRef CAS PubMed
.
- Y. Xu, Q. Wang, J. Wei, X. Peng, J. Xue, Z. Wang, S. J. Su and Y. Wang, Angew. Chem., Int. Ed., 2022, 61, e202204652 CrossRef CAS PubMed
.
- T. Huang, L. Yuan, X. Lu, Y. Qu, C. Qu, Y. Xu, Y.-X. Zheng and Y. Wang, Chem. Sci., 2024, 15, 15170–15177 RSC
.
- Q. Wang, L. Yuan, C. Qu, T. Huang, X. Song, Y. Xu, Y.-X. Zheng and Y. Wang, Adv. Mater., 2023, 35, 2305125 Search PubMed
.
- Q. Wang, Y. Xu, T. Huang, Y. Qu, J. Xue, B. Liang and Y. Wang, Angew. Chem., Int. Ed., 2023, 62, e202301930 CrossRef CAS PubMed
.
- H. Chen, M. Du, C. Qu, Q. Jin, Z. Tao, R. Ji, G. Zhao, T. Zhou, Y. Lou, Y. Sun, W. Jiang, L. Duan and Y. Zhang, Angew. Chem., Int. Ed., 2025, 64, e202415400 CrossRef CAS PubMed
.
- S. Jiang, Y. Yu, D. Li, Z. Chen, Y. He, M. Li, G. X. Yang, W. Qiu, Z. Yang, Y. Gan, J. Lin, Y. Ma and S. J. Su, Angew. Chem., Int. Ed., 2023, 62, e202218892 CrossRef CAS PubMed
.
- G. Meng, J. Zhou, T. Huang, H. Dai, X. Li, X. Jia, L. Wang, D. Zhang and L. Duan, Angew. Chem., Int. Ed., 2023, 62, e202309923 CrossRef CAS PubMed
.
- X. Wang, L. Wang, G. Meng, X. Zeng, D. Zhang and L. Duan, Sci. Adv., 2023, 9, eadh1434 CrossRef CAS PubMed
.
- X. Huang, J. Liu, Y. Xu, G. Chen, M. Huang, M. Yu, X. Lv, X. Yin, Y. Zou, J. Miao, X. Cao and C. Yang, Natl. Sci. Rev., 2024, 11, nwae115 Search PubMed
.
- P. Jiang, J. Miao, X. Cao, H. Xia, K. Pan, T. Hua, X. Lv, Z. Huang, Y. Zou and C. Yang, Adv. Mater., 2022, 34, 2106954 Search PubMed
.
- Y. K. Qu, D. Y. Zhou, F. C. Kong, Q. Zheng, X. Tang, Y. H. Zhu, C. C. Huang, Z. Q. Feng, J. Fan, C. Adachi, L. S. Liao and Z. Q. Jiang, Angew. Chem., Int. Ed., 2022, 61, e202201886 CrossRef CAS PubMed
.
- Y. Zhang, J. Wei, D. Zhang, C. Yin, G. Li, Z. Liu, X. Jia, J. Qiao and L. Duan, Angew. Chem., Int. Ed., 2022, 61, e202113206 CrossRef CAS PubMed
.
- K. Uematsu, K. Noguchi and K. Nakano, Phys. Chem. Chem. Phys., 2018, 20, 3286–3295 RSC
.
- B. M. Gross and M. Oestreich, Synthesis, 2021, 53, 2512–2516 CrossRef CAS
.
- C. Maeda, K. Nagahata, T. Shirakawa and T. Ema, Angew. Chem., Int. Ed., 2020, 59, 7813–7817 CrossRef CAS PubMed
.
- Y. Niu, W. Li, Q. Peng, H. Geng, Y. Yi, L. Wang, G. Nan, D. Wang and Z. Shuai, Mol. Phys., 2018, 116, 1078–1090 CrossRef CAS
.
- E. R. Johnson, S. Keinan, P. Mori-Sánchez, J. Contreras-García, A. J. Cohen and W. Yang, J. Am. Chem. Soc., 2010, 132, 6498–6506 CrossRef CAS PubMed
.
- Y. Hu, M. Huang, H. Liu, J. Miao and C. Yang, Angew. Chem., Int. Ed., 2023, 62, e202312666 CrossRef CAS PubMed
.
- J. Liu, X. Yin, M. Huang, J. Miao, N. Li, Z. Huang and C. Yang, Adv. Mater., 2025, 37, 2411610 CrossRef CAS PubMed
.
- C. Lee, Y. T. Lee, C. Y. Chan, C. W. Park, S. Lee, H. I. Kwon, D. Boo, Y. Tsuchiya, S. Y. Lee and C. Adachi, Adv. Opt. Mater., 2025, 13, 2402862 CrossRef CAS
.
- Z. Li, Z. Li, S. Zhang, M. Liu, G. Gao, J. You and Z. Bin, Sci. China Mater., 2024, 67, 1581–1587 CrossRef CAS
.
- D. Zhang, C. Zhao, Y. Zhang, X. Song, P. Wei, M. Cai and L. Duan, ACS Appl. Mater. Interfaces, 2017, 9, 4769–4777 CrossRef CAS PubMed
.
- T. Huang, Q. Wang, S. Xiao, D. Zhang, Y. Zhang, C. Yin, D. Yang, D. Ma, Z. Wang and L. Duan, Angew. Chem., Int. Ed., 2021, 60, 23771–23776 CrossRef CAS PubMed
.
- Y. Zhang, G. Li, L. Wang, T. Huang, J. Wei, G. Meng, X. Wang, X. Zeng, D. Zhang and L. Duan, Angew. Chem., Int. Ed., 2022, 61, e202202380 CrossRef CAS PubMed
.
- L. Wu, Z. Xin, D. Liu, D. Li, J. Zhang, Y. Zhou, S. Wu, T. Wang, S.-J. Su, W. Li and Z. Ge, Adv. Mater., 2025, 37, 2416224 Search PubMed
.
- M. Quan, Z.-L. Zhu, G. Chen, J. Miao and C. Yang, Angew. Chem., Int. Ed., 2025, 64, e202512162 CrossRef CAS PubMed
.
- J. Liu, X. Yin, M. Huang, J. Miao, N. Li, Z. Huang and C. Yang, Adv. Mater., 2025, 37, 2411610 CrossRef CAS PubMed
.
- Z.-G. Wu, Y. Xin, C. Lu, W. Huang, H. Xu, X. Liang, X. Cao, C. Li, D. Zhang, Y. Zhang and L. Duan, Angew. Chem., Int. Ed., 2024, 63, e202318742 CrossRef CAS PubMed
.
- Y.-T. Lee, C.-Y. Chan, N. Matsuno, S. Uemura, S. Oda, M. Kondo, R. W. Weerasinghe, Y. Hu, G. N. I. Lestanto, Y. Tsuchiya, Y. Li, T. Hatakeyama and C. Adachi, Nat. Commun., 2024, 15, 3174 Search PubMed
.
- J. Liu, J. Miao, J. Dong, Z. Chen, Z. Huang and C. Yang, Adv. Mater., 2025, e13987 Search PubMed
.
- T. Fan, S. Zhu, X. Cao, X. Liang, M. Du, Y. Zhang, R. Liu, D. Zhang and L. Duan, Angew. Chem., Int. Ed., 2023, 62, e202313254 Search PubMed
.
- G. Meng, H. Dai, Q. Wang, J. Zhou, T. Fan, X. Zeng, X. Wang, Y. Zhang, D. Yang, D. Ma, D. Zhang and L. Duan, Nat. Commun., 2023, 14, 2394 CrossRef CAS PubMed
.
- L. Yuan, Z.-L. Tu, J.-W. Xu, H.-X. Ni, Z.-P. Mao, W.-Y. Xu and Y.-X. Zheng, Sci. China:Chem., 2023, 66, 2612–2620 CrossRef CAS
.
- G. Meng, J. Zhou, X.-S. Han, W. Zhao, Y. Zhang, M. Li, C.-F. Chen, D. Zhang and L. Duan, Adv. Mater., 2024, 36, 2307420 CrossRef CAS PubMed
.
- L. Li, Y. Xu, Y. Sun, Y. Qu, W. Cui, L. Guo, P. Zheng, Y. Wang and C. Li, Adv. Mater., 2026, 38, e11560 CrossRef CAS PubMed
.
- X. Cai, J. Wei, Z. Li, Y. Pu, Y. Wu and Y. Wang, Chem. Sci., 2025, 16, 11539–11547 Search PubMed
.
- J. Wang, D. Chen, J. M. Moreno-Naranjo, F. Zinna, L. Frédéric, D. B. Cordes, A. P. McKay, M. J. Fuchter, X. Zhang and E. Zysman-Colman, Chem. Sci., 2024, 15, 16917–16927 RSC
.
- Q. Wang, L. Yuan, C. Qu, T. Huang, X. Song, Y. Xu, Y. X. Zheng and Y. Wang, Adv. Mater., 2023, 35, 2305125 CrossRef CAS PubMed
.
- Y. Yang, N. Li, J. Miao, X. Cao, A. Ying, K. Pan, X. Lv, F. Ni, Z. Huang, S. Gong and C. Yang, Angew. Chem., Int. Ed., 2022, 61, e202202227 CrossRef CAS PubMed
.
- X.-J. Liao, D. Pu, L. Yuan, J. Tong, S. Xing, Z.-L. Tu, J.-L. Zuo, W.-H. Zheng and Y.-X. Zheng, Angew. Chem., Int. Ed., 2023, 62, e202217045 CrossRef CAS PubMed
.
- F. Zhang, F. Rauch, A. Swain, T. B. Marder and P. Ravat, Angew. Chem., Int. Ed., 2023, 62, e202218965 CrossRef CAS PubMed
.
- H. Jiang, D. Ai, J. Jin, Z. Xie and W.-Y. Wong, Sci. China:Chem., 2025, 68, 2804–2819 CrossRef CAS
.
Footnote |
| † These authors contributed equally. |
| This journal is © The Royal Society of Chemistry 2026 |