 Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
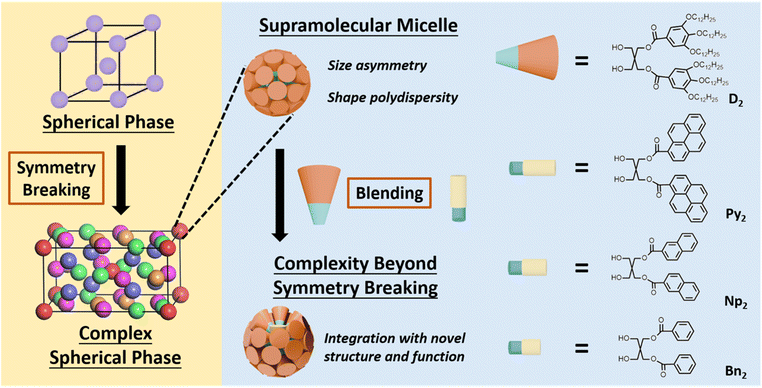
Toward functional and structurally complex Frank–Kasper phases via creating concavities on supramolecular micelles
Yong-Rui
Wang
a,
Jui-Heng
Weng
a,
Shing-Jong
Huang
a,
Chun-Jen
Su
 b,
U-Ser
Jeng
b,
U-Ser
Jeng
 b,
Po-Ya
Chang
b,
Wei-Tsung
Chuang
b,
Po-Ya
Chang
b,
Wei-Tsung
Chuang
 *b and
Chien-Lung
Wang
*b and
Chien-Lung
Wang
 *a
*a
aDepartment of Chemistry, National Taiwan University, No. 1, Sec. 4, Roosevelt Rd, Taipei 10617, Taiwan. E-mail: kclwang@ntu.edu.tw
bNational Synchrotron Radiation Research Center, 101 Hsin-Ann Road, Hsinchu 30092, Taiwan. E-mail: weitsung@nsrrc.org.tw
First published on 22nd January 2026
Abstract
In supramolecular chemistry, higher structural complexity enables emergent functions in ordered soft matter. To construct structurally complex and functional Frank–Kasper (FK) phases, heterogeneity in supramolecular micelles is introduced by blending rigid aromatic dendrons (Ar2) with flexible aliphatic dendrons (D2). This approach creates micelles with surface concavities while preserving long-range periodicity of the FK σ lattice. The concave domains serve as enzyme-like pockets that accommodate guest molecules and facilitate photodimerization reactions. Structural analyses confirm that these features enhance complexity in hierarchical architecture and enable catalytic performance. This work presents a versatile strategy for designing FK phases that integrate molecular recognition, supramolecular precision, and catalytic function within an ordered framework.
Introduction
Nature creates self-assembly systems that are precise, complex and dynamic to enable physiological functions that are essential for life.1 In comparison to their natural counterparts, artificial supramolecular systems still exhibit relatively limited structural complexity and functionality. To address this gap, researchers often tune two key parameters: multiplicity, which introduces chemical diversity in molecular composition and function, and interconnections, which define the bonding modes and dynamic interactions between components.2,3 Modulating these parameters can influence how individual components integrate into higher-order architectures. As this hierarchical integration becomes more sophisticated, the overall organization of the system may change, giving rise to emergent collective properties.2,3One strategy to enhance complexity is the incorporation of aperiodicity within periodic lattices. In block copolymers, factors such as conformational asymmetry,4–7 volume asymmetry,8–10 compositional asymmetry,11–15etc. have been applied to create symmetry breaking that leads to structural complexity in the spherical phases.16 Blending, in particular, provides a straightforward means of controlling both formation and stability of Frank–Kasper (FK) phases, where uneven interfacial curvature promotes non-spherical and multi-sized micellar motifs.11,12,17–23 However, blending-based polydispersity mainly alters micelle size and shape, and therefore offers only limited ways to introduce new structural features. Creating additional levels of complexity—such as spatially defined chemical environments, anisotropic interfaces, or directional binding sites—without disrupting the integration of the original FK lattice remains challenging. For example, although blending block copolymers can effectively shift the phase behavior of mixed systems, the resulting structures often retain a single pre-existing ordered phase, without generating finer structural features or new functional elements within that lattice.12 This highlights the difficulty of enhancing structural complexity while maintaining long-range order.
Dendrons and dendrimers offer a distinctive pathway to such complexity and structural features.24,25 Their generation-dependent shape evolution,26–28 precise molecular design,26,29–32 and versatile end-group modifications27,30,33,34 enable quasi-equivalent packing that relieves frustration and supports tetrahedrally close-packed FK lattices.35 Consequently, dendrons assemble into a remarkable diversity of structures—including cubic,27,29,30,35,36 tetragonal,28,31,35,37,38 quasicrystalline,28,37–39 hollow,33,34,37 and even chiral micelles34,39,40—surpassing what is typically achievable with block copolymers.31,37 In contrast, natural blended systems routinely exploit heterogeneity to generate structural multiplicity and dynamic functionality. Biological membranes, for instance, form compositionally heterogeneous microdomains such as lipid rafts, stabilized by cholesterol–sphingolipid interactions that enhance membrane responsiveness.41–44 Likewise, protein binding pockets provide concave hydrophobic cavities that couple structure to function, enabling selective guest recognition and catalysis by pre-organizing substrates and stabilizing transition states.45–48 These examples illustrate how blending-derived heterogeneity can simultaneously increase the complexity and functional adaptability of an assembly.
Motivated by these natural design principles—and by the ability of dendrons to introduce controlled heterogeneity—we sought to install designed concavities onto the surface of FK-phase supramolecular micelles. Our goal was to elevate the structural sophistication of the spherical assemblies while preserving the symmetry breaking and long-range integration of the FK lattice. As illustrated in Scheme 1, the diaryl amphiphiles (Ar2) are used as molecular rafts that create heterogeneity in supramolecular micelles formed by the amphiphilic di-dendron (D2). D2 molecule has been synthesized as the intermediate product of the Janus dendrimers for many research.49–59 In our previous study, D2 has demonstrated the ability to form FK σ phase with symmetry breaking.59 Since the Ar2 molecules are smaller and more rigid than D2, blending the Ar2 and D2 in the supramolecular micelles could create both heterogeneity and concavities in the supramolecular micelles of the FK phase. Catalytic functions mimic the natural hydrophobic pockets may also be added to the complex FK phase as the concavities on top of the Ar2 motifs could encapsulate guest molecules. Our characterization data indicated that on top of the symmetry breaking, the presence of the raft-inspired building blocks Ar2 in the supramolecular micelles of D2 created additional structural features and function to the FK σ phase. The overall integration of the synthetic self-assembly system is thus upgraded as the presence of the Ar2 advances both hierarchical complexity and functional adaptability of the spherical phase. This innovation aligns with Lehn's vision of progressive complexity,3 bridging the gap between structural sophistication and dynamic functionality in artificial self-assembly systems.
Results and discussion
Create concavities onto the FK σ phase
Schemes S1–S3 illustrate the synthetic routes for D2 and Ar2. The synthesis routes of these di-dendron molecules are referring to the published literature of dendron self-assembly.59 The molecular structures of these compounds were confirmed by 1H NMR, 13C NMR, and mass spectrometry, as presented in Fig. S2–S15. To incorporate the Ar2 into D2, both Ar2 and D2 were co-dissolved in CH2Cl2. After solvent removal, Ar2/D2 mixtures with varying molar fractions of Ar2 were obtained. The ordered phases of the mixtures were then prepared by heating to 150 °C—above the isotropization temperatures (Ti) of both D2 and Ar2 but below their decomposition thresholds (Fig. S16)—to ensure homogeneous mixing in the molten state. The resulting melts were cooled and annealed at 40 °C for 12 hours to facilitate self-assembly.
were obtained. The ordered phases of the mixtures were then prepared by heating to 150 °C—above the isotropization temperatures (Ti) of both D2 and Ar2 but below their decomposition thresholds (Fig. S16)—to ensure homogeneous mixing in the molten state. The resulting melts were cooled and annealed at 40 °C for 12 hours to facilitate self-assembly.
To confirm the uniform incorporation of molecular rafts into the ordered phase of D2, optical microscopy (OM) was employed for direct observation. As shown in Fig. 1a, macroscopic spherulitic structures were evident in the drop-cast samples of the Py2/D2 mixtures. With increasing Py2 content, the spherulitic morphology became increasingly disrupted. At  the emergence of dark aggregated domains suggests phase segregation of excess Py2. This conclusion is further supported by wide-angle X-ray scattering (WAXS) data (Fig. 1b), where distinct crystalline diffraction peaks attributed to Py2 are observed in the
the emergence of dark aggregated domains suggests phase segregation of excess Py2. This conclusion is further supported by wide-angle X-ray scattering (WAXS) data (Fig. 1b), where distinct crystalline diffraction peaks attributed to Py2 are observed in the  sample. In contrast, WAXS profiles of samples with
sample. In contrast, WAXS profiles of samples with  display no sharp crystalline peaks, but instead feature a broad amorphous halo. This indicates that the spherulitic structures observed via OM do not originate from angstrom-scale crystalline ordering. The scattering profiles of varies of Ar2 are shown in Fig. S17.
display no sharp crystalline peaks, but instead feature a broad amorphous halo. This indicates that the spherulitic structures observed via OM do not originate from angstrom-scale crystalline ordering. The scattering profiles of varies of Ar2 are shown in Fig. S17.
To probe the internal aggregate structures within these spherulites, small-angle X-ray scattering (SAXS) analysis was conducted (Fig. 1c). The SAXS profiles revealed that self-assemble micellization of the Py2/D2 mixtures crystalizes into the FK σ phase. To further clarify how the SAXS data support the micellar assignment and FK σ phase formation, the primary and higher-order peaks were analyzed. The peak ratios (q/q0) are consistent with the reflection pattern of the FK σ lattice,60 and the comprehensive analysis of the σ-phase structure and its indexed diffraction peaks is provided in Table S1. The characteristic peaks q410, q330, and q002 were used to calculate the corresponding lattice parameters (a, b, c) by the standard geometric relations of tetragonal phase, shown in Table S2.38 Based on these lattice parameters, the diameter of the supramolecular micelles within the FK σ lattice (Dsphere) was estimated, also performed in Table S2. These results confirm that the incorporation of Py2 does not inhibit the ability of D2 to act as a cone-shaped motif and self-assemble into the FK σ phase. However, the presence of Py2 shifts the diffraction peaks to higher q values and broadens them, indicating that the inclusion of rigid, small Py2 molecules introduces heterogeneity within the micelles and compresses the σ-phase lattice.
The formation of this FK σ phase from cone-shaped motifs represents a thermodynamically stable self-assembly, further validated by differential scanning calorimetry (DSC). As shown in Fig. 1d, the Ti of the mixtures systematically decreases with increasing Py2 content. This phase behavior is consistent with the principle of colligative properties, where the incorporation of solute molecules (Py2) lowers the Ti of the host component (D2).61 This behavior also confirms that Py2 is well-dispersed within the D2-rich ordered domains for compositions with  At
At  however, the Ti does not decrease further, indicating that the excess Py2 exceeds the micelle's incorporation capacity and instead undergoes phase separation to form Py2-rich domains, as corroborated by both Fig. 1a and b. Additionally, the broadening of all (hkl) diffraction peaks in the SAXS profiles supports the DSC results, reinforcing the conclusion that Py2 molecules are randomly and uniformly distributed within the supramolecular micelles of D2, resulting in both a reduction in Ti and broadened diffraction features.
however, the Ti does not decrease further, indicating that the excess Py2 exceeds the micelle's incorporation capacity and instead undergoes phase separation to form Py2-rich domains, as corroborated by both Fig. 1a and b. Additionally, the broadening of all (hkl) diffraction peaks in the SAXS profiles supports the DSC results, reinforcing the conclusion that Py2 molecules are randomly and uniformly distributed within the supramolecular micelles of D2, resulting in both a reduction in Ti and broadened diffraction features.
The miscibility of other Ar2 variants—Np2 and Bn2—with D2 was also assessed using the same protocol. The results, summarized in Fig. S18 and Tables S4–S9, confirm that both Np2 and Bn2 exhibit good miscibility with D2 up to  Moreover, increasing size mismatch between Ar2 and D2 leads to a greater suppression of Ti and more significant lattice contraction in the σ phase. This trend suggests that smaller molecular rafts introduce greater disruption to the micellar packing order, highlighting the influence of geometric compatibility on structural organization within FK σ phases.
Moreover, increasing size mismatch between Ar2 and D2 leads to a greater suppression of Ti and more significant lattice contraction in the σ phase. This trend suggests that smaller molecular rafts introduce greater disruption to the micellar packing order, highlighting the influence of geometric compatibility on structural organization within FK σ phases.
The heterogeneity of the Py2/D2 micelles and the dynamic behavior of Py2 within the supramolecular micelles were further investigated by solid-state 13C–1H heteronuclear correlation (HETCOR) nuclear magnetic resonance (NMR) spectroscopy. For pure D2 in the FK σ phase, cross-polarization (CP) is relatively inefficient due to the high segmental mobility; therefore, the HETCOR spectrum was acquired using a prolonged CP contact time (tc) of 2 ms. In contrast, the HETCOR spectrum of Py2 was recorded with a short CP contact time of 200 µs to suppress undesired long-range polarization transfer.
As shown in Fig. 2a and b, the characteristic spectral features of D2 and Py2 are clearly distinguishable. For D2, only cross-peaks originating from the dodecyl chains are observed, appearing in the ranges of δC = 10–50 ppm and δH = 0–3 ppm. In contrast, the expected resonances from the O–CH2 units (δC ≈ 65–75 ppm),62 the pentaerythritol core (δC = 50–65 ppm and δH = 2–5 ppm), and the aromatic moieties (δC = 100–120 ppm and δH = 5–8 ppm) are absent. This behavior can be attributed to their significantly higher mobility, which leads to inefficient 1H–13C dipolar recoupling under the applied CP conditions, as discussed in detail in Fig. S19. For Py2, in addition to the signals originating from the pentaerythritol core, an additional broad resonance at approximately δC ≈ 120 ppm was observed, which can be attributed to the Py segments. The aromatic 1H signals of Py2 are widely distributed and predominantly broadened due to strong residual 1H–1H dipolar couplings that persist even under MAS conditions, as well as the heterogeneous local environments experienced by the Py2 segments within the micelles.63–65 For the Py2/D2 mixtures, the cross-peaks associated with Py2 are clearly observed in the HETCOR spectra for samples with  in the HETCOR spectra, acquired with a prolonged contact time of 5 ms, as shown in Fig. 2c and d. In contrast, the Py2 signals in the
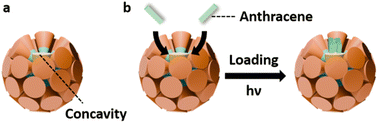
in the HETCOR spectra, acquired with a prolonged contact time of 5 ms, as shown in Fig. 2c and d. In contrast, the Py2 signals in the  mixture are too weak to be detected (Fig. S20) owing to the low Py2 concentration. To gain deeper insight into the location and orientation of the Py segments within the micelles, a 1H spin-diffusion (PSD) block was incorporated into the HETCOR pulse sequence, rather than further extending the CP contact time, in order to probe long-range intermolecular spatial correlations. This strategy was adopted for three main reasons. First, magnetization decay in the present approach is governed by T1 relaxation, whereas an extended CP contact time is limited by T1ρ relaxation, and T1 is typically much longer than T1ρ. Second, 1H–1H dipolar interactions play a crucial role in long-range intermolecular magnetization transfer; however, the spin-locking RF field ramped linearly from 40 to 50 kHz during CP may partially suppress these dipolar interactions and thus hinder spin diffusion. Third, excessively long CP contact times (>50 ms) may lead to power-handling issues in commercial MAS probeheads. As shown in Fig. 2e and f, at a spin-diffusion time (tSD) of 250 ms, magnetization from the 1H nuclei of the dodecyl chains is efficiently transferred to the Py2 segments. This effective magnetization transfers between the dodecyl chains and the Py segments indicates that the Py segments of the molecular rafts are primarily located within the hydrophobic shell of the supramolecular micelles, as schematically illustrated in Scheme 2a.
mixture are too weak to be detected (Fig. S20) owing to the low Py2 concentration. To gain deeper insight into the location and orientation of the Py segments within the micelles, a 1H spin-diffusion (PSD) block was incorporated into the HETCOR pulse sequence, rather than further extending the CP contact time, in order to probe long-range intermolecular spatial correlations. This strategy was adopted for three main reasons. First, magnetization decay in the present approach is governed by T1 relaxation, whereas an extended CP contact time is limited by T1ρ relaxation, and T1 is typically much longer than T1ρ. Second, 1H–1H dipolar interactions play a crucial role in long-range intermolecular magnetization transfer; however, the spin-locking RF field ramped linearly from 40 to 50 kHz during CP may partially suppress these dipolar interactions and thus hinder spin diffusion. Third, excessively long CP contact times (>50 ms) may lead to power-handling issues in commercial MAS probeheads. As shown in Fig. 2e and f, at a spin-diffusion time (tSD) of 250 ms, magnetization from the 1H nuclei of the dodecyl chains is efficiently transferred to the Py2 segments. This effective magnetization transfers between the dodecyl chains and the Py segments indicates that the Py segments of the molecular rafts are primarily located within the hydrophobic shell of the supramolecular micelles, as schematically illustrated in Scheme 2a.
According to the previous work in our group,59 in the σ phase of D2, each supramolecular micelle is constructed with ca. 19 cone-like D2 molecules. Therefore, at  each supramolecular micelle of Py2/D2 mixtures has to accommodate ca. 4 and 6 Py2 molecules, respectively. Combining the ssNMR results in Fig. 2 and the SAXS, WAXS and OM data in Fig. 1, it was found that the presence of Py2 creates structural heterogeneity to the supramolecular micelles of D2, but the loading capacity of the micelles is limited, as 6 Py2 molecules in each micelle is effective enough to create crowded environment that slows down the dynamics of the molecular rafts. The maximum loading capacity of each micelle in the σ phase is about 6 Py2 molecules, as the higher molar ratio of Py2 in the mixture led to the phase-separated Py2-rich domains.
each supramolecular micelle of Py2/D2 mixtures has to accommodate ca. 4 and 6 Py2 molecules, respectively. Combining the ssNMR results in Fig. 2 and the SAXS, WAXS and OM data in Fig. 1, it was found that the presence of Py2 creates structural heterogeneity to the supramolecular micelles of D2, but the loading capacity of the micelles is limited, as 6 Py2 molecules in each micelle is effective enough to create crowded environment that slows down the dynamics of the molecular rafts. The maximum loading capacity of each micelle in the σ phase is about 6 Py2 molecules, as the higher molar ratio of Py2 in the mixture led to the phase-separated Py2-rich domains.
A comparable diffusion-based experiment was carried out for the Np2/D2 system at a molar fraction of 20%, following the same experimental strategy as used for the Py2/D2 mixtures. As shown in Fig. S21, distinct intermolecular magnetization transfer between the aliphatic region of D2 and the aromatic resonances of Np2 was observed, indicating efficient spin diffusion between these components. This result demonstrates that replacing Py2 with a different aromatic molecular raft (Ar2) leads to a similar spatial proximity between the aromatic segments and the dodecyl-chain-rich hydrophobic domains of the D2 supramolecular micelles. The Np2/D2 diffusion data therefore support the generality of aromatic raft incorporation within concaved σ-phase micelles, rather than a behavior specific to the Py2 system.
Guest encapsulation and catalytic properties of the σ(Ar2)
As D2 and Ar2 are mismatch in size, incorporating molecular rafts Ar2 units into the supramolecular micelles of D2 create novel FK σ phases, denoted σ(Ar2), which contain concave domains as illustrated in Scheme 2a. The indentations on the supramolecular micelles may provide confined environments for guest encapsulation and potential catalytic activation resembling hydrophobic enzyme pockets. To probe this, anthracene (An)—a planar polycyclic aromatic hydrocarbon structurally similar to the Ar segments—was selected as the guest molecule. Meanwhile, the [4 + 4] photodimerization of An molecules under UV light66 was applied as the probe for both binding and reactivity within σ(Ar2) with the molar ratio as illustrated in Scheme 2b.
as illustrated in Scheme 2b.
To differentiate the dispersion environments, the An-loaded assemblies are denoted as An@σ for the assemble using simply D2 as the host (conventional micelles), and An@σ(Ar2) for those using Ar2/D2 mixtures as the hosts (concaved micelles).
SAXS profiles in Fig. 3a shows that guest loading perturbs the lattice of conventional micelles, causing more significant lattice contraction and peak broadening of the σ phase of D2, as shown in Table S11 and Table S12. The insignificant changes of the FK σ framework of σ(Ar2) indicate that the pre-organized concavities localize guest molecules and reduce the extent of global lattice distortion. Due to the size differences, the incorporation of Py2, Np2 and Bn2 results in concavities with difference depths in the micelles. Among the variants, σ(Np2) provides the optimal balance of geometric complementarity and interaction strength, stably encapsulating 2 – 3 eq. of An without phase-separation (Fig. S22 and S23). Collectively, these results establish that only concavities of suitable depth enable stable guest encapsulation, with σ(Np2) standing out by accommodating two An per concavity—precisely the stoichiometry for photodimerization—highlighting the unique role of geometry-tuned concavities in stabilizing and pre-organizing guests.
Since σ(Np2) exhibited the most favorable guest encapsulation, we next evaluated its impact on photoreactivity. As shown in Fig. 3b, crystalline An dimerized very slowly under UV irradiation (λ = 365 nm) due to the rigid packing of the lattice. In contrast, An dispersed in conventional micelles (An@σ) reached about 45% conversion within 2 h, consistent with the requirement for suitable intermolecular orientation;67 the modestly improved conversion can be attributed to the greater molecular mobility in the fluidic micellar environment. Remarkably, in σ(Np2) assemblies nearly complete photodimerization occurred within 1 h, demonstrating that the concavities not only stabilize guest encapsulation but also promote effective preorganization of the reactants. Collectively, these results show that concavity-engineered micelles enhance photoreactivity through the combined effects of confinement and spatial preorganization, thereby closely emulating the functional characteristics of enzymatic active sites.
Conclusions
To exceed complexity of the FK phase and equip functions to the bulk phase supramolecular micelles, in this study, aromatic (Ar2) and aliphatic (D2) dendrons are blended in controlled ratios to introduce heterogeneity and create surface concavities within the micellar domains of FK σ phase. These concavities increase architectural hierarchy through localized asymmetry while preserving the long-range periodicity of the FK σ lattice, as evidenced by SAXS and WAXS results. Solid-state NMR reveals that aromatic segments of the Ar2 reside in the hydrophobic micellar shell, confirming the formation of chemically distinct, heterogeneous domains. These features generate concavities that act as functional pockets for guest encapsulation without disrupting the FK σ lattice. SAXS and WAXS analyses confirm the incorporation of An into the ordered σ(Np2) framework, revealing concavities with optimal depth and packing to encapsulate the guest molecules. Building on this structural evidence, time-dependent ex situ1H NMR demonstrates that such concavities substantially accelerate An photodimerization, as reflected in the C/C0 profiles.Together, these findings establish σ(Np2) as the most effective architecture for promoting photoreactivity. More broadly, they highlight a generalizable strategy in which concavity confinement, molecular recognition, and dynamic guest exchange are embedded within a FK σ lattice, thereby functionally mimicking enzymatic active sites and advancing Lehn's vision of progressively complex soft materials.
Author contributions
Conceptualization, data curation, formal analysis: Y. R. W, J. H. W, W. T. C., and C. L. W. Funding acquisition: C. L. W. Investigation: Y. R. W, J. H. W, and S. J. H. Methodology: Y. R. W., S. J. H., and C. L. W. Project administration: Y. R. W., and C. L. W. Resources, software: S. J. H, C. J. S, U. S. J., P. Y. C., W. T. C., and C. L. W. Supervision, validation: W. T. C., and C. L. W. Visualization, writing – original draft: Y. R. W, and C. L. W. Writing – review & editing: Y. R. W, S. J. H., W. T. C and C. L. W.Conflicts of interest
There are no conflicts to declare.Data availability
All data supporting the findings of this study, including time-dependent 1H NMR spectra, SAXS/WAXS scattering profiles, ssNMR spectra, TGA and DSC thermograms, are provided in the supplementary information (SI). Additional raw data generated and analyzed during the current study are available from the corresponding author upon reasonable request. Supplementary information: the synthetic procedures, 1H and 13C NMR and MASS spectrum and TGA and DSC thermograms of the D2 and Ar2, Miller indices, lattice parameters, and S/WAXS patterns of FK σ phase of the Ar2/D2 mixtures, instrumentation of the analytical experiments, ssNMR results and its pulse sequences, characterization and catalytic analysis of An, An@σ, and An@σ(Ar2). See DOI: https://doi.org/10.1039/d5sc07961f.Acknowledgements
We gratefully acknowledge the TPS 13A beamline at the National Synchrotron Radiation Research Center (NSRRC), Taiwan, for providing access to the S/WAXS measurements. Thermogravimetric analysis (TGA) was performed using the Thermal Analysis System at the Instrumentation Center, National Taiwan University (NTU). Mass spectrometry services were provided by the Consortia of Key Technologies, NTU. We also thank Dr Hsi-Ching Tseng at the NTU Instrumentation Center for the kind assistance with NMR measurements. This work was supported by the National Science and Technology Council, Taiwan (NSTC 113-2123-M-002-008).Notes and references
- R. Montis, L. Fusaro, A. Falqui, M. B. Hursthouse, N. Tumanov, S. J. Coles, T. L. Threlfall, P. N. Horton, R. Sougrat and A. Lafontaine, Nature, 2021, 590, 275–278 CrossRef CAS PubMed.
- J.-M. Lehn, Science, 2002, 295, 2400–2403 CrossRef CAS PubMed.
- J. M. Lehn, Angew. Chem., Int. Ed., 2013, 52, 2836–2850 CrossRef CAS PubMed.
- Z. Gan, Z. Xu, K. Tian, D. Zhou, L. Li, Z. Ma, R. Tan, W. Li and X.-H. Dong, Nat. Commun., 2024, 15, 6581 CrossRef CAS PubMed.
- K. K. Lachmayr, C. M. Wentz and L. R. Sita, Angew. Chem., Int. Ed., 2020, 59, 1521–1526 CrossRef CAS PubMed.
- M. W. Bates, J. Lequieu, S. M. Barbon, R. M. Lewis III, K. T. Delaney, A. Anastasaki, C. J. Hawker, G. H. Fredrickson and C. M. Bates, Proc. Natl. Acad. Sci. U. S. A., 2019, 116, 13194–13199 CrossRef CAS PubMed.
- A. Reddy, M. S. Dimitriyev and G. M. Grason, Nat. Commun., 2022, 13, 2629 CrossRef CAS PubMed.
- A. Reddy, M. B. Buckley, A. Arora, F. S. Bates, K. D. Dorfman and G. M. Grason, Proc. Natl. Acad. Sci. U. S. A., 2018, 115, 10233–10238 CrossRef CAS PubMed.
- Y. Wang, J. Huang, X. Y. Yan, H. Lei, X. Y. Liu, Q. Y. Guo, Y. Liu, T. Liu, M. Huang and F. Bian, Angew. Chem., 2022, 134, e202200637 CrossRef.
- A. Reddy and G. M. Grason, Nat. Chem., 2019, 11, 865–867 CrossRef CAS PubMed.
- M. Liu, Y. Qiang, W. Li, F. Qiu and A.-C. Shi, ACS Macro Lett., 2016, 5, 1167–1171 CrossRef CAS PubMed.
- A. P. Lindsay, R. M. Lewis III, B. Lee, A. J. Peterson, T. P. Lodge and F. S. Bates, ACS Macro Lett., 2020, 9, 197–203 CrossRef CAS PubMed.
- K. K. Lachmayr and L. R. Sita, Angew. Chem., Int. Ed., 2020, 59, 3563–3567 CrossRef CAS PubMed.
- H. Takagi and K. Yamamoto, Macromolecules, 2019, 52, 2007–2014 CrossRef CAS.
- A. B. Chang and F. S. Bates, ACS Nano, 2020, 14, 11463–11472 CrossRef CAS PubMed.
- S. Lee, C. Leighton and F. S. Bates, Proc. Natl. Acad. Sci. U. S. A., 2014, 111, 17723–17731 CrossRef CAS PubMed.
- Z. Xu and W. Li, Chin. J. Chem., 2022, 40, 1083–1090 CrossRef CAS.
- G. S. Doerk, A. Stein, S. Bae, M. M. Noack, M. Fukuto and K. G. Yager, Sci. Adv., 2023, 9, eadd3687 CrossRef PubMed.
- J. Xie and A.-C. Shi, Langmuir, 2023, 39, 11491–11509 CrossRef CAS PubMed.
- H. Lei, X.-Y. Liu, Y. Wang, X.-H. Li, X.-Y. Yan, T. Liu, J. Huang, W. Li, L. Wang, X. Kuang, X. Miao, F. Bian, M. Huang, Y. Liu and S. Z. Cheng, J. Am. Chem. Soc., 2024, 146, 33403–33412 CrossRef CAS PubMed.
- X.-Y. Liu, X.-Y. Yan, Y. Liu, H. Qu, Y. Wang, J. Wang, Q.-Y. Guo, H. Lei, X.-H. Li, F. Bian, X.-Y. Cao, R. Zhang, Y. Wang, M. Huang, Z. Lin, E. W. Meijer, T. Aida, X. Kong and S. Z. Cheng, Nat. Mater., 2024, 23, 570–576 CrossRef CAS PubMed.
- X.-Y. Yan, Q.-Y. Guo, X.-Y. Liu, Y. Wang, J. Wang, Z. Su, J. Huang, F. Bian, H. Lin, M. Huang, Z. Lin, T. Liu, Y. Liu and S. Z. Cheng, J. Am. Chem. Soc., 2021, 143, 21613–21621 CrossRef CAS PubMed.
- Y. Liu, T. Liu, X.-Y. Yan, Q.-Y. Guo, H. Lei, Z. Huang, R. Zhang, Y. Wang, J. Wang, F. Liu, F.-G. Bian, E. W. Meijer, T. Aida, M. Huang and S. Z. Cheng, Proc. Natl. Acad. Sci. U. S. A., 2022, 119, e2115304119 CrossRef CAS PubMed.
- B. M. Rosen, C. J. Wilson, D. A. Wilson, M. Peterca, M. R. Imam and V. Percec, Chem. Rev., 2009, 109, 6275–6540 CrossRef CAS PubMed.
- V. Percec, S. Wang, N. Huang, B. E. Partridge, X. Wang, D. Sahoo, D. J. Hoffman, J. Malineni, M. Peterca and R. L. Jezorek, J. Am. Chem. Soc., 2021, 143, 17724–17743 CrossRef CAS PubMed.
- V. Percec, W.-D. Cho, P. Mosier, G. Ungar and D. Yeardley, J. Am. Chem. Soc., 1998, 120, 11061–11070 CrossRef CAS.
- V. Percec, W.-D. Cho and G. Ungar, J. Am. Chem. Soc., 2000, 122, 10273–10281 CrossRef CAS.
- V. Percec, C. M. Mitchell, W.-D. Cho, S. Uchida, M. Glodde, G. Ungar, X. Zeng, Y. Liu, V. S. Balagurusamy and P. A. Heiney, J. Am. Chem. Soc., 2004, 126, 6078–6094 CrossRef CAS PubMed.
- V. Percec, C.-H. Ahn, G. Ungar, D. Yeardley, M. Möller and S. Sheiko, Nature, 1998, 391, 161–164 CrossRef CAS.
- V. Percec, W.-D. Cho, G. Ungar and D. J. Yeardley, J. Am. Chem. Soc., 2001, 123, 1302–1315 CrossRef CAS.
- G. Ungar, Y. Liu, X. Zeng, V. Percec and W.-D. Cho, Science, 2003, 299, 1208–1211 CrossRef CAS PubMed.
- S. Hudson, H.-T. Jung, V. Percec, W.-D. Cho, G. Johansson, G. Ungar and V. Balagurusamy, Science, 1997, 278, 449–452 CrossRef CAS.
- V. Percec, B. C. Won, M. Peterca and P. A. Heiney, J. Am. Chem. Soc., 2007, 129, 11265–11278 CrossRef CAS PubMed.
- V. Percec, M. Peterca, A. E. Dulcey, M. R. Imam, S. D. Hudson, S. Nummelin, P. Adelman and P. A. Heiney, J. Am. Chem. Soc., 2008, 130, 13079–13094 CrossRef CAS PubMed.
- X. Zeng, G. Ungar, Y. Liu, V. Percec, A. E. Dulcey and J. K. Hobbs, Nature, 2004, 428, 157–160 CrossRef CAS PubMed.
- V. Percec, W.-D. Cho, M. Möller, S. A. Prokhorova, G. Ungar and D. J. Yeardley, J. Am. Chem. Soc., 2000, 122, 4249–4250 CrossRef CAS.
- B. M. Rosen, D. A. Wilson, C. J. Wilson, M. Peterca, B. C. Won, C. Huang, L. R. Lipski, X. Zeng, G. Ungar and P. A. Heiney, J. Am. Chem. Soc., 2009, 131, 17500–17521 CrossRef CAS PubMed.
- M. N. Holerca, D. Sahoo, B. E. Partridge, M. Peterca, X. Zeng, G. Ungar and V. Percec, J. Am. Chem. Soc., 2018, 140, 16941–16947 CrossRef CAS PubMed.
- V. Percec, M. R. Imam, M. Peterca, D. A. Wilson, R. Graf, H. W. Spiess, V. S. Balagurusamy and P. A. Heiney, J. Am. Chem. Soc., 2009, 131, 7662–7677 CrossRef CAS PubMed.
- V. Percec, M. R. Imam, M. Peterca, D. A. Wilson and P. A. Heiney, J. Am. Chem. Soc., 2009, 131, 1294–1304 CrossRef CAS PubMed.
- D. Lingwood and K. Simons, Science, 2010, 327, 46–50 CrossRef CAS.
- A. J. Sodt, M. L. Sandar, K. Gawrisch, R. W. Pastor and E. Lyman, J. Am. Chem. Soc., 2014, 136, 725–732 Search PubMed.
- K. Simons and D. Toomre, Nat. Rev. Mol. Cell Biol., 2000, 1, 31–39 CrossRef CAS PubMed.
- L. Rajendran and K. Simons, J. Cell Sci., 2005, 118, 1099–1102 CrossRef CAS PubMed.
- P. Setny and M. Geller, J. Chem. Phys., 2006, 125 Search PubMed.
- A. K. Shrive, G. M. Gheetham, D. Holden, D. A. Myles, W. G. Turnell, J. E. Volanakis, M. B. Pepys, A. C. Bloomer and T. J. Greenhough, Nat. Struct. Mol. Biol., 1996, 3, 346–354 Search PubMed.
- J. W. Barnett, M. R. Sullivan, J. A. Long, D. Tang, T. Nguyen, D. Ben-Amotz, B. C. Gibb and H. S. Ashbaugh, Nat. Chem., 2020, 12, 589–594 CrossRef CAS PubMed.
- M. Yoshizawa, T. Kusukawa, M. Kawano, T. Ohhara, I. Tanaka, K. Kurihara, N. Niimura and M. Fujita, J. Am. Chem. Soc., 2005, 127, 2798–2799 CrossRef CAS PubMed.
- Q. Xiao, M. Delbianco, S. E. Sherman, A. M. Reveron Perez, P. Bharate, A. Pardo-Vargas, C. Rodriguez-Emmenegger, N. Y. Kostina, K. Rahimi and D. Söder, Proc. Natl. Acad. Sci. U. S. A., 2020, 117, 11931–11939 CrossRef CAS PubMed.
- V. Percec, P. Leowanawat, H.-J. Sun, O. Kulikov, C. D. Nusbaum, T. M. Tran, A. Bertin, D. A. Wilson, M. Peterca and S. Zhang, J. Am. Chem. Soc., 2013, 135, 9055–9077 CrossRef CAS PubMed.
- D. Zhang, E. N. Atochina-Vasserman, D. S. Maurya, N. Huang, Q. Xiao, N. Ona, M. Liu, H. Shahnawaz, H. Ni and K. Kim, J. Am. Chem. Soc., 2021, 143, 12315–12327 CrossRef CAS PubMed.
- C. Rodriguez-Emmenegger, Q. Xiao, N. Y. Kostina, S. E. Sherman, K. Rahimi, B. E. Partridge, S. Li, D. Sahoo, A. M. Reveron Perez and I. Buzzacchera, Proc. Natl. Acad. Sci. U. S. A., 2019, 116, 5376–5382 CrossRef CAS.
- N. Yu. Kostina, D. Söder, T. Haraszti, Q. Xiao, K. Rahimi, B. E. Partridge, M. L. Klein, V. Percec and C. Rodriguez-Emmenegger, Angew. Chem., Int. Ed., 2021, 60, 8352–8360 CrossRef PubMed.
- S. Zhang, R. O. Moussodia, H. J. Sun, P. Leowanawat, A. Muncan, C. D. Nusbaum, K. M. Chelling, P. A. Heiney, M. L. Klein and S. André, Angew. Chem., 2014, 126, 11079–11083 CrossRef.
- S. E. Sherman, Q. Xiao and V. Percec, Chem. Rev., 2017, 117, 6538–6631 CrossRef CAS PubMed.
- N. Huang, Q. Xiao, M. Peterca, X. Zeng and V. Percec, Mol. Phys., 2021, 119, e1902586 CrossRef.
- V. Percec, D. A. Wilson, P. Leowanawat, C. J. Wilson, A. D. Hughes, M. S. Kaucher, D. A. Hammer, D. H. Levine, A. J. Kim and F. S. Bates, Science, 2010, 328, 1009–1014 CrossRef CAS PubMed.
- L.-C. Lee and Y. Zhao, J. Am. Chem. Soc., 2014, 136, 5579–5582 CrossRef CAS PubMed.
- C.-L. Wang, W.-T. Chuang, M.-T. Lee, Y.-R. Wang, S.-Y. Chen, H.-J. Huang, S.-Y. Liu, J.-M. Lin, C.-Y. Chen and Y.-C. Lee, ACS Appl. Mater. Interfaces, 2025, 17, 31403–31410 CrossRef CAS PubMed.
- T. Jun, H. Park, S. Jeon, S. Jo, H. Ahn, W.-D. Jang, B. Lee and D. Y. Ryu, J. Am. Chem. Soc., 2021, 143, 17548–17556 CrossRef CAS PubMed.
- H.-Y. Lin, B.-J. Zhong, H.-J. Liu, Y.-K. Wu, C.-H. Peng and C.-L. Wang, Cryst. Growth Des., 2024, 24, 2833–2840 CrossRef CAS.
- M. Seo, G. Seo and Y. K. Sang, Angew. Chem., Int. Ed., 2006, 45, 6306–6310 CrossRef CAS PubMed.
- C. S. Yannoni, Acc. Chem. Res., 1982, 15, 201–208 CrossRef CAS.
- L.-Q. Wang, J. Liu, G. J. Exarhos, K. Y. Flanigan and R. Bordia, J. Phys. Chem. B, 2000, 104, 2810–2816 CrossRef CAS.
- F. G. Vogt, J. S. Clawson, M. Strohmeier, A. J. Edwards, T. N. Pham and S. A. Watson, Cryst. Growth Des., 2009, 9, 921–937 CrossRef CAS.
- H. Bouas-Laurent, A. Castellan, J.-P. Desvergne and R. Lapouyade, Chem. Soc. Rev., 2000, 29, 43–55 RSC.
- S. L. Anderson, P. G. Boyd, A. Gładysiak, T. N. Nguyen, R. G. Palgrave, D. Kubicki, L. Emsley, D. Bradshaw, M. J. Rosseinsky and B. Smit, Nat. Commun., 2019, 10, 1612 CrossRef PubMed.
| This journal is © The Royal Society of Chemistry 2026 |









![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
: