Open Access Article
Open Access Article[1H]/[2H] discriminated bianthryl atropisotopomers: enantiospecific syntheses from BINOL and direct multi-spectroscopic analyses of their isotopic chirality
Florian
Rigoulet
 a,
Guillaume
Dauvergne
a,
Jean-Valère
Naubron
a,
Guillaume
Dauvergne
a,
Jean-Valère
Naubron
 *bd,
Philippe
Lesot
*bd,
Philippe
Lesot
 *cd,
Christie
Aroulanda
*cd,
Christie
Aroulanda
 c and
Yoann
Coquerel
c and
Yoann
Coquerel
 *ad
*ad
aAix Marseille Univ, CNRS, Centrale Med, ISM2, 13397 Marseille, France. E-mail: yoann.coquerel@univ-amu.fr
bAix Marseille Univ, CNRS, Centrale Med, FSCM, 13397 Marseille, France. E-mail: jean-valere.naubron@univ-amu.fr
cUniversité Paris-Saclay, CNRS UMR 8182, ICMMO, RMN en Milieu Orienté, Bât. 670, 17 Avenue des Sciences, 91400, Orsay, France. E-mail: philippe.lesot@universite-paris-saclay.fr
dCentre National de la Recherche Scientifique, CNRS, 3 Rue Michel-Ange, 75016 Paris, France
First published on 26th January 2026
Abstract
The synthesis of two [1H]/[2H] discriminated bianthryl atropisotopomers (i.e., isotopically chiral atropisomers, vide infra) with ≥97% enantiomeric excess (ee) from the atropisomers of commercial (aS)- and (aR)-1,1′-bi-2-naphthol (BINOL, >99% ee) is described. The approach is based on enantiospecific [4 + 2] cycloaddition reactions of aryne atropisomer intermediates with [2H4]-furan. Overall, the six-step (from enantiopure BINOL) reaction sequence to the bianthryl atropisotopomers described herein occurs without significant erosion of the molecular stereogenic information. This is substantiated experimentally by three direct spectroscopic methods that do not necessitate chemical derivatization: (i) mirrored specific optical rotations as a proof of optical activity as well as of their equivalent ee, (ii) mirrored vibrational circular dichroism (VCD) spectra as a proof of their equivalent ee and for the confirmation of their absolute configurations (AC), and (iii) for the first time, anisotropic 2H/13C NMR in lyotropic chiral liquid crystals for the determination of the ee of one of them. In a broader and foreseeable perspective, the enantiospecific synthetic approach considered in this work to access bianthryl atropisotopomers from BINOL is simple and scalable, which should enable the production of a diverse range of atropisotopomers in the near future, possibly utilizing never-examined isotopes (e.g., [3H]). The availability of this class of isotopically chiral compounds in high isotopic and enantiomeric purity is a real asset for the development of precision chiral spectroscopies.
Introduction
The presence of isotopes in molecules (both chiral and non-chiral), whether of natural abundance or not, is of immense value in science and technology. Isotopes are ubiquitous in areas ranging from fundamental and applied science in physics, biology, and chemistry to major societal applications such as dating, fraud detection, health and energy production.1 A well-known illustration of this is the so-called “deuterium switch” in drug discovery, in which all-[1H] molecules are replaced by their [2H]-containing isotopologues, which has major impacts on drug efficacy and safety beyond simple pharmacokinetic improvements.2 However, this approach is limited by the “synthetic and analytical challenges in controlling site and extent of deuterium enrichment,” as Pirali and her co-workers so aptly put it.2 Another illustration of this is the ability of the [1H]/[2H] isotopes in molecules to control the selectivity of some chemical reactions toward one product or another through quantum mechanical tunneling, which is the third paradigm in chemical reactivity, alongside kinetic and thermodynamic control.3 This approach is also limited by synthetic and analytical challenges. The natural terrestrial abundance of the [2H] isotope of hydrogen in ocean water (V-SMOW value) is equal to 155.76(5) ppm (≈0.0156%, about 1 in 6420 atoms).4 Thus, it is clear that [2H]-glycine (1, Fig. 1a) with both a [1H] atom and a [2H] atom at its methylene C atom exists naturally on planet Earth in tiny amounts. The [1H] and [2H] atoms differ in the composition of their nuclei: the [1H] atom nucleus contains a single particle (a proton), while the [2H] atom nucleus contains two, namely, one proton and one neutron. Hence, the methylene C atom in 1 is stereogenic, and compound 1 is a chiral molecule by virtue of the [1H]/[2H] isotopic substitution.5 The chirality in 1, which arises solely from the different composition of the nuclei of the two hydrogen isotopes at its methylene C atom, is referred to as isotopic chirality, and the enantiomers of 1 are referred to as enantioisotopomers. Enantioenriched isotopically chiral molecules are able to trigger asymmetric autocatalysis with enantiomeric excess (ee) amplification in some reactions.6 Consequently, naturally occurring isotopically chiral molecules, possibly existing locally in not strictly racemic amounts, have been considered as a reasonable hypothesis to address the question of the origin of homochirality in terrestrial living organisms, which is a fundamental issue of considerable importance to humanity. If present, any (extremely tiny) ee in terrestrial naturally occurring isotopically chiral molecules would be impossible to detect for long. Thus, research on isotopically chiral molecules has relied on the designed and controlled incorporation of isotopes into molecules at spatially defined positions via precision chemical synthesis to obtain enantioenriched samples. However, this is an incredibly difficult task for which no perfect example exists, i.e., an example in which a chemical purity of >97%, isotopic purity of >97% and an ee of >97% could be achieved and verified experimentally. Prototypical examples of synthetic enantioisotopomers based on [1H]/[2H] discrimination are [2H]-glycine (1, 77–82% ee),7 [2H]-ethyl tosylate (2, 90% ee),8 and [2H]-ethyl benzene (3, 97% ee).9,10 A spectacular complex example is [2H6]-neopentane (4, 90–96% ee).11 The isotopic chirality in compounds 1–4 derives from a single stereogenic carbon atom (central isotopic chirality). With the recent progress in stereoselective synthetic organic chemistry, [2H]-enriched enantioisotopomers with central isotopic chirality have changed status from curiosities in academic laboratories decades ago to incredibly enabling tools in contemporary science. Compounds 1–4 are illustrative of this, and more examples exist.12 While atropisomerism can also lead to enantioisotopomers (based on axial isotopic chirality), this class of compounds has been much less examined. We propose the name “atropisotopomers” for atropisomerism-based enantioisotopomers. The first reported non-racemic [1H]/[2H] discriminated atropisotopomers were the biphenyl-based molecules 5![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 13 and 6,14 which were synthesized more than 50 years ago with modest ee. More recently, a promising new platform to synthesize non-racemic [1H]/[2H] discriminated atropisotopomers around a stereogenic C(sp2)–N(sp2) bond was developed, which led to compounds 7a,b (98% ee).15 To complete the list of existing non-racemic atropisotopomers based on [1H]/[2H] discrimination, the beautiful atropisotopomeric macrocyclic cyclotriveratrylene derivatives 8a,b
13 and 6,14 which were synthesized more than 50 years ago with modest ee. More recently, a promising new platform to synthesize non-racemic [1H]/[2H] discriminated atropisotopomers around a stereogenic C(sp2)–N(sp2) bond was developed, which led to compounds 7a,b (98% ee).15 To complete the list of existing non-racemic atropisotopomers based on [1H]/[2H] discrimination, the beautiful atropisotopomeric macrocyclic cyclotriveratrylene derivatives 8a,b![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 16 and the superb atropisotopomeric cryptophane 8c
16 and the superb atropisotopomeric cryptophane 8c![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 17 should also be mentioned. Some isolated examples of enantioisotopomers based on planar chirality18 and inherent chirality19 have also been reported.
17 should also be mentioned. Some isolated examples of enantioisotopomers based on planar chirality18 and inherent chirality19 have also been reported.
Except for compound 3 (and two of its analogues), for which an enantioselective copper-catalyzed synthesis can be achieved based on the hydrodeuteration of styrene,9,20 isotopically chiral molecules are typically obtained either by diastereoselective approaches using a chiral auxiliary (covalently bonded to the substrate or the reagent) assuming no racemization in the subsequent steps,7,8,11 or by enantiospecific syntheses, i.e., from standard chiral enantioenriched precursors of known absolute configurations (AC) and verified ee to which the heavy isotope atoms are added in late or final synthetic step(s) via functional group interconversion (for instance, Br → [2H], Br → C[2H]3, OH → OC[2H]3), presumably without racemization.13–17
One challenge is the analysis of chirality in isotopically chiral molecules, i.e., the experimental determination of their AC [with either (R)- or (S)-configured stereogenic atoms in the cases of central isotopic chirality, or either (aR)- or (aS)-configured stereogenic bonds in the case of axial isotopic chirality] and of their ee. Indeed, the chirality in these molecules derives exclusively from an asymmetric distribution of the masses of their nuclei, while they have virtually the same electron distribution as their all-[1H]-isotopologues. To a first approximation, isotopically chiral compounds are electronically achiral. Thus, analytical methods based on electronic molecular properties that are commonly employed to discriminate enantiomers are a priori not suitable to discriminate enantioisotopomers. This is the case for optical rotation, electronic circular dichroism (ECD) spectroscopy and single-crystal anomalous X-ray diffraction analysis, which are classically employed to determine the AC, and for chiral chromatography (gas phase or liquid phase), which is generally used to measure the ee. Enantioisotopomers are “minimal” test cases for evaluating how sensitive analytical tools are to molecular chirality.
In practice, tiny specific optical rotations ([α]25D) have been recorded for some enantioisotopomers since as early as 1949,21 and more recently for the enantioisotopomers of atropisotopomers 7a,b and 8c. Additionally, weak but detectable ECD signals were recorded for the enantioisotopomers of atropisotopomers 8a–c![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 17,22 and some isotopically chiral [2.2]paracyclophane.18 The AC of isotopically chiral molecules is generally assumed from the AC of their synthetic precursors. However, it can be experimentally determined/verified using vibrational circular dichroism (VCD) and/or Raman optical activity (ROA) spectroscopies, as the mass ratio of the [2H] and [1H] nuclei is 1.998 ≈ 2, thus producing experimentally detectable and computable differences in the vibration frequencies of their [12C]–[2H] and [12C]–[1H] bonds.23 Among other available techniques, gas-phase Coulomb explosion imaging enabled the determination of the AC of [1H]/[2H] discriminated enantioisotopomers,24 and notably, the AC of 3 was recently deduced using a new tool in the box, chiral tag molecular rotational resonance (CTMRR) spectroscopy in the gas phase.9,25
17,22 and some isotopically chiral [2.2]paracyclophane.18 The AC of isotopically chiral molecules is generally assumed from the AC of their synthetic precursors. However, it can be experimentally determined/verified using vibrational circular dichroism (VCD) and/or Raman optical activity (ROA) spectroscopies, as the mass ratio of the [2H] and [1H] nuclei is 1.998 ≈ 2, thus producing experimentally detectable and computable differences in the vibration frequencies of their [12C]–[2H] and [12C]–[1H] bonds.23 Among other available techniques, gas-phase Coulomb explosion imaging enabled the determination of the AC of [1H]/[2H] discriminated enantioisotopomers,24 and notably, the AC of 3 was recently deduced using a new tool in the box, chiral tag molecular rotational resonance (CTMRR) spectroscopy in the gas phase.9,25
The ee of isotopically chiral molecules based on [1H]/[2H] discrimination is also generally assumed to be identical to the ee of their synthetic precursors. In the cases in which the ee has been verified experimentally, this has most often been performed by indirect methods, i.e., using chemical derivatization to generate pairs of diastereomers (mixing standard and isotopic stereogenicity) and assuming no enantiomerization in the process.10,15 Subsequently, the measurement of the diastereomeric ratio by spectroscopic methods, typically using quantitative isotropic NMR based on chemical shift differences (Δδiso), can be observed for some nuclei. The real challenge is the direct experimental determination of the ee of enantioenriched isotopically chiral samples, which has rarely been achieved to date. Chiral chromatography is impracticable in these cases, with only a few cases of partial resolution having been achieved.26 For enantioisotopomers containing a remote Lewis-base coordinating site, the use of lanthanide chiral shift reagents (LCSR) has proved possible in some isolated examples having a stereogenic [1H]/[2H] methylene C atom.27 In practice, direct ee measurements of enantioisotopomers can only be performed by two means: (i) using CTMRR spectroscopy in the gas phase, e.g., for 3,9,28 and (ii) by anisotropic NMR spectroscopy in a condensed phase, i.e., NMR in enantiopure, chiral liquid crystals (CLC), e.g., for 2.8,29
In this article, we describe the syntheses of the two π-extended atropisotopomers 9 and 10, together with the extensive analysis of their chirality via polarimetry, using VCD for the determination of their AC and anisotropic NMR for the determination of their ee. Remarkably, the described enantiospecific synthetic approach to the atropisotopomers 9 and 10 is not based on a late-stage functional group interconversion as in previous approaches. In the described approach, the extended π-conjugated chiral carbon backbone is constructed enantiospecifically through a bidirectional Diels–Alder/deoxygenation two-step synthetic sequence involving arynes. Concerning the determination/confirmation of the AC of [1H]/[2H] discriminated atropisotopomers, we show that this can be confidently achieved using solid state VCD spectroscopy. Regarding the direct measurement of the ee of the enantioisotopomers of atropisotopomers, this study establishes for the first time that it can be realized by anisotropic NMR methods using both the 2H and 13C nuclei as magnetically active probes; however, while this was a successful approach for the enantioisotopomers of atropisotopomer 10 with C1-symmetry, the enantioisotopomers of atropisotopomer 9 with C2-symmetry resisted this analysis.
Results and discussion
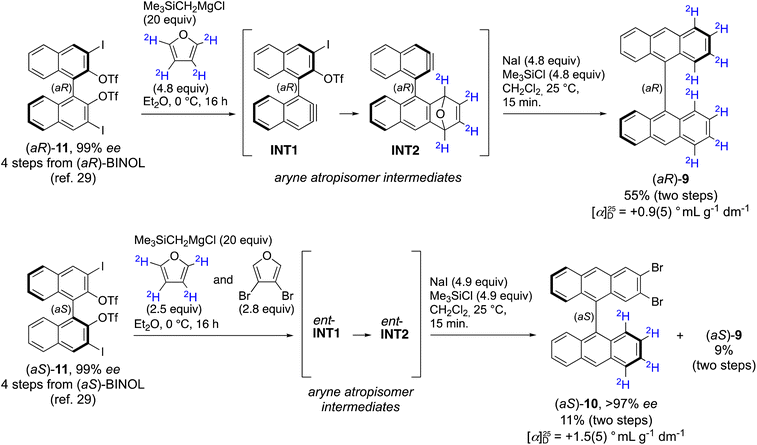
Using ortho-iodoaryl triflate precursors and trimethylsilylmethyl magnesium chloride, the Coquerel group demonstrated that aryne atropisomers (e.g., INT1 and INT2, Scheme 1) can be enantiospecifically generated and reacted with full retention of the chiral information of their precursors.30 In fact, the Diels–Alder reaction of aryne atropisomers with furan occurs with a half-reaction time <150 ns at temperatures higher than −20 °C, which is much faster than the rotation around their stereogenic C(sp2)–C(sp2) axis, thus ensuring enantiospecificity.31 Using both enantiomers of the enantiopure (99% ee) BINOL derivative 11 (derived in four steps and 77% yield from 99% ee BINOL) as the precursors of bis(aryne) atropisomers, fully enantiospecific bidirectional reactions were realized, enabling the spatially controlled syntheses of some congested atropisomers in high enantiomeric purity (>98% ee).32 Using [2H4]-furan (≥98% [2H] atoms) as the arynophile in a bidirectional Diels–Alder/deoxygenation two-step reaction sequence with both enantiomers of the bis(aryne) atropisomer precursor 11 (99% ee), the two enantioisotopomers of atropisotopomer 9 were prepared in an enantiospecific manner: (aR)-9 derived from (aR)-11 and (aS)-9 derived from (aS)-11, with 55% and 53% yield over two steps, respectively (Scheme 1). In another reaction using both [2H4]-furan and 3,4-dibromofuran as arynophiles in a three-component transformation, the two enantioisotopomers of atropisotopomer 10 were obtained (in 11% and 10% yield for (aS)-10 and (aR)-10, respectively), together with the two enantioisotopomers of atropisotopomer 9 (9% yield). In contrast to existing approaches to atropisotopomers, the extended π-conjugated carbon backbone of both 9 and 10 is constructed in an enantiospecific manner, not simply by functional group interconversion as in previously reported cases.13–17It was previously demonstrated that the bidirectional reactions of the bis(aryne) atropisomer precursor 11 occur with full retention of the chiral information, i.e., with the products having unchanged AC and identical (or nearly) ee to precursor 11 (enantiospecific reactions).32 Assuming that the rotation around the C(sp2)–C(sp2) in 9,9′-bianthryl is blocked at temperatures below 140 °C (with 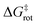 ≈ 185 kJ mol−1 as evaluated by DFT methods, see Section S5 in the SI), and despite the fact that the 12C–2H bond is 0.005 Å shorter than the 12C–1H bond,33 both atropisotopomers 9 and 10 are configurationally stable compounds with locked AC. No racemization of atropisotopomers 9 and 10 is possible at a significant rate under the examined experimental conditions. Thus, it can be reasonably hypothesized that the enantioisotopomers of atropisotopomers 9 and 10 were obtained with ee ≥98%, and with retention of the AC of their stereogenic axes.
≈ 185 kJ mol−1 as evaluated by DFT methods, see Section S5 in the SI), and despite the fact that the 12C–2H bond is 0.005 Å shorter than the 12C–1H bond,33 both atropisotopomers 9 and 10 are configurationally stable compounds with locked AC. No racemization of atropisotopomers 9 and 10 is possible at a significant rate under the examined experimental conditions. Thus, it can be reasonably hypothesized that the enantioisotopomers of atropisotopomers 9 and 10 were obtained with ee ≥98%, and with retention of the AC of their stereogenic axes.
The first evidence of the non-racemic nature of samples of 9 and 10 arose from measurement of their specific optical rotations: the [α]25D of (aR)-9 is +0.9(5)° mL g−1 dm−1 (c = 1.0, CHCl3) and the [α]25D of (aS)-9 is −0.9(5)° mL g−1 dm−1 (c = 1.0, CHCl3); similarly, the [α]25D of (aS)-10 is +1.5(5)° mL g−1 dm−1 (c = 0.42, CHCl3) and the [α]25D of (aR)-10 is −1.5(5)° mL g−1 dm−1 (c = 0.50, CHCl3).
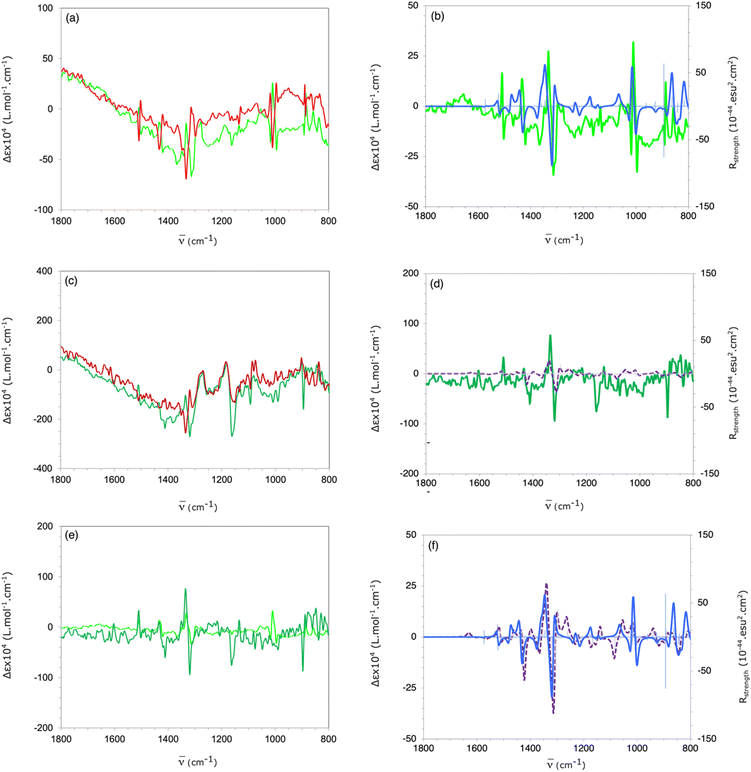
The AC of the enantioisotopomers of atropisotopomers 9 and 10 were confirmed by solid-state VCD spectroscopy (Fig. 2). For 9, IR measurements in CD2Cl2 at 0.1 M gave a low absorbance (below 0.1 AU), which was insufficient for reliable VCD analysis (recommended minimum ≈ 0.2 AU). Increasing the concentration to achieve the optimal ∼0.8 AU was precluded by solubility and material constraints. Consequently, the VCD spectra of 9 and 10 were recorded in the solid-state using ca. 5.5 mg of material in KBr pellets. The pellets were carefully prepared and measured on a rotating holder to minimize circular and linear birefringence and linear dichroism artefacts. The spectra of (aS)-9 and (aR)-9 are mirror images (Fig. 2a), ruling out major artefacts, and the experimental spectrum of 9 derived from (aR)-11 agrees well with the simulated spectrum of (aR)-9 computed at the B3LYP-D3(BJ)/6-311G(df,pd) DFT level of theory (Fig. 2b), confirming the enantiomeric relationship with their precursors (aR)-11 and (aS)-11, respectively. The VCD spectra of (aS)-10 and (aR)-10, although showing some significant artefacts around 1160 and 900 cm−1, are mirror images for the most diagnostically relevant bands, notably at 1315 cm−1 (Fig. 2c). They closely resemble those of the corresponding enantiomers of 9, especially in the 1250–1600 cm−1 region (Fig. 2e), in which the most intense bands arise from CAr–H and CAr–CAr deformation modes delocalized over both anthracene units; these features are accurately reproduced by the simulated spectra (Fig. 2f). The AC assignment of the enantioisotopomers of 10, based on the comparison of their measured VCD spectra with those of 9, was further supported by the calculated VCD spectrum of (aR)-10, which matches the experimental spectrum of the sample derived from (aR)-11 (Fig. 2d). Comparison of the IR spectra of 9 and 10 (Fig. S7 in the SI) reveals two bands at 1411 and 1091 cm−1, which can be assigned to vibrational modes involving the two bromine atoms. The calculated rotational strengths for these modes are extremely small, accounting for the absence of detectable VCD signals. Similarly, attempts to measure the VCD bands corresponding to the CAr–[2H] stretching modes around 2300 cm−1 were unsuccessful, as the VCD response was too weak to be distinguished from baseline noise.
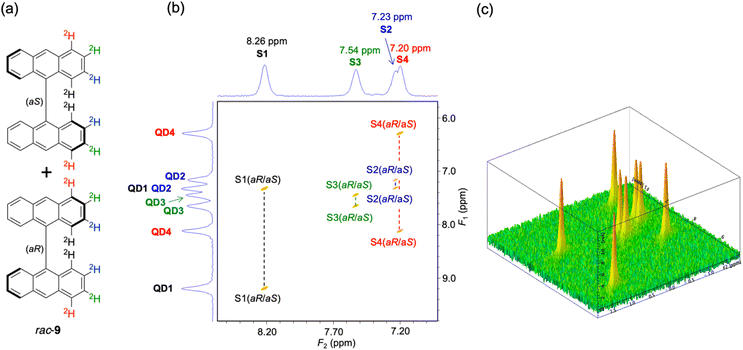
The question of the direct experimental measurement of the ee of the samples of the enantioisotopomers of atropisotopomers 9 and 10 is the most arduous one. Among the two methods available to do so, CTMRR spectroscopy in the gas phase is a priori not suitable because both atropisotopomers 9 and 10 would be difficult to vaporize [for (aS)-9 and (aR)-9: mp > 360 °C; for (aS)-10 and (aR)-10: mp = 278 °C, dec.], which leaves anisotropic NMR spectroscopy as the only viable option. To evaluate whether anisotropic NMR spectroscopy would be able to discriminate the enantioisotopomers of atropisotopomer 9, i.e., (aS)-9 and (aR)-9, a pseudo-racemic sample of rac-9 was prepared by mixing equal amounts (5.0 mg) of (aS)-9 and (aR)-9 obtained as described in Scheme 1. A sample composed of 3.0 mg of rac-9, 101 mg of the commercially available chiral homopolymer PBLG (poly-γ-benzyl-L-glutamate, DP = 768) and 502 mg of chloroform (stabilized with ethanol <1%) was analyzed via anisotropic 2H–{1H} 2D NMR with a 14.1 T spectrometer equipped with a selective 2H cryogenic probe (92.1 MHz) (see Fig. 3 and details in Section S4 of the SI).34 Conceptually, this analysis is based on the ability of the PBLG polymer to spatially orient, on average, each enantiomer of atropisotopomer 9 (and 10) along distinct magnetically detectable directions. When the difference in molecular orientation is significant in the CLC mesophase, two spectral signatures (one for each enantiomer) based on the difference in 2H residual quadrupolar couplings (2H-RQC) are then observed. In practice, the use of QUadrupole Order SpectroscopY (QUOSY) nD experiments such as phased 2H–{1H} Q-COSY Fz (spectrum of 9 in Fig. 3) or a phased 2H–{1H} Q-resolved 2D sequence (spectra of 10 in Fig. 4) instead of basic single-pulse 2H–{1H} 1D-NMR experiments is analytically advantageous. These reduce peak overlap and enable deuterium quadrupolar doublets (2H-DQ) to be identified based on their respective δ(2H). This considerably simplifies the analysis of complex anisotropic 2H–{1H} spectra. In both experiments, the 2H chemical shifts and 2H quadrupolar doublets are separated in the F2 and F1 dimensions, respectively.35 In the initial 2H–{1H} Q-COSY Fz 2D NMR experiments recorded at 9.4 T using a QXO probe (not shown), only four quadrupolar doublets were observed for rac-9, which indicated that no spectral enantiodiscrimination occurred under these conditions. The applicability of a higher-field 14.1 T spectrometer equipped with a selective 2H cryogenic probe was then examined. As shown in Fig. 3, only four quadrupolar doublets were observed for rac-9 in this case at 310 K; the same result was observed at 277 K and 325 K. This indicates that the inability of the PBLG chiral matrix to spectrally discriminate the enantioisotopomers of 9 is not a matter of spectroscopic resolution or sensitivity. The origin of this lies elsewhere (vide infra). Thus, for now, the ee of the samples of (aS)-9 and (aR)-9 synthesized as described above remain undetermined by a direct experimental method.
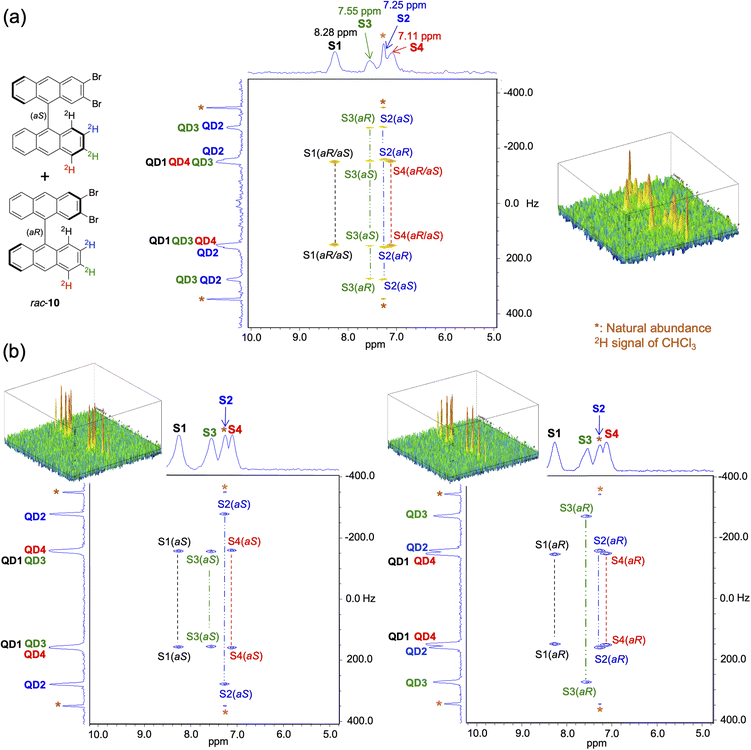
Using the same approach, the ability of the enantioisotopomers of atropisotopomer 10, i.e., (aS)-10 and (aR)-10, to be discriminated by anisotropic NMR was evaluated. A sample composed of 3.0 mg of rac-10, 110 mg of the chiral polymer PBLG (DP = 768) and 570 mg of chloroform was analyzed using anisotropic NMR spectroscopy with a 9.4 T spectrometer (QXO probe). In the corresponding 2H–{1H} Q-resolved 2D NMR spectrum (Fig. 4a), six quadrupolar doublets are observed, which indicates that spectral enantiodiscrimination occurs for two out of the four [2H] atoms in rac-10 under these conditions. More precisely, enantiodiscrimination is observed for the [2H] atoms at the 2 (blue) and 3 (green) positions of the bianthryl carbon backbone (Fig. 4a). Thus, the ee of both samples of enantioisotopomers (aS)-10 and (aR)-10 synthesized as described above can be directly determined by chiral anisotropic NMR. Accordingly, these samples were analyzed under similar experimental conditions. In the corresponding phased 2H–{1H} Q-resolved 2D NMR spectra of enantioisotopomers (aS)-10 and (aR)-10 (Fig. 4b), only four quadrupolar doublets are visible, meaning that these samples contain a single enantioisotopomer within the detection limit of the NMR spectrometer (see details in Section S4 of the SI). From these analyses, the ee of both enantioisotopomers of atropisotopomer 10, (aS)-10 and (aR)-10 can be reliably evaluated as ≥97%.
The naturally occurring isotope 13C (1.1%) is also an interesting magnetically active species for the spectral discrimination of enantioisotopomers, although this method is less sensitive in terms of enantiodiscrimination than 2H NMR for [2H]-enriched enantioisotopomers.36 In the case of rac-10, spectral enantiodiscrimination is also observed via anisotropic 13C–{1H} NMR through the difference in the 13C residual chemical shift anisotropy (13C-RCSA) between the enantioisotopomers,29 leading to a doubling of the number of signals for each discriminated 13C site compared to the 13C–{1H} spectrum recorded in isotropic (achiral) solvents. For instance, it can be observed for rac-10 that four 13C atoms show a doubling of their 13C resonances in the anisotropic 13C–{1H} NMR spectrum of rac-10 (Fig. 5, middle). In contrast, the anisotropic 13C–{1H} NMR spectra of (aR)-10 and (aS)-10 show a single resonance for the same 13C atoms, indicating that the minor enantioisotopomer is not detected (or does not emerge from the noise) in these cases, confirming that its ee is likely >95% (Fig. 5, top and bottom). These complementary anisotropic NMR investigations using 13C nuclei probes conclusively confirm the ee of both enantioisotopomers of atropisotopomer 10 as having been accurately determined from the 2H nuclei analyses. Overall, the anisotropic NMR analyses discussed herein are the first direct experimental determination of the ee for enantioisotopomers of an atropisotopomer, and also the highest ee (≥97%) determined by this technique for any synthetic enantioisotopomer.
 | ||
| Fig. 5 Anisotropic 13C–{1H} 1D NMR spectra (100.4 MHz) and 13C peak assignment of rac-10 (middle), (aR)-10 (top) and (aS)-10 (bottom) recorded in the chiral mesophase PBLG/CHCl3 at 305 K. | ||
At this early stage of our work, it is not clear why the enantioisotopomers of atropisotopomer 10 could be spectrally discriminated while the enantioisotopomers of atropisotopomer 9 could not be under comparable anisotropic NMR conditions. However, it can reasonably be hypothesized that the virtually null dipole moment of atropisotopomer 9, a compound with C2-symmetry, translates into a poor difference in the preferred diastereotopic spatial orientation of its enantioisotopomers in the PBLG chiral matrix, thus making the enantiodiscrimination very difficult to detect spectrally through anisotropic NMR experiments. This is extremely instructive for the development of the technique, which is on-going in our laboratories.
Conclusions
The bidirectional reactions of the enantiopure (or nearly, ee = 99%) bis(aryne) atropisomers (aS)-11 and (aR)-11, which were efficiently derived from both enantiomers of BINOL (ee ≥ 99%), with [2H4]-furan as either the sole arynophile or in combination with 3,4-dibromofuran enabled the expeditious syntheses of the isotopically chiral enantioisotopomers of the [1H]/[2H] discriminated atropisotopomers (aR)-9/(aS)-9 and (aR)-10/(aS)-10 (ee ≥ 97% for 10, ee not yet experimentally verified for 9). The originality and the relevance of this enantiospecific synthetic approach to atropisotopomers lies in its efficiency: it simultaneously forges the extended π-conjugated carbon backbone (the 9,9′-bianthryl backbone) and the isotopically stereogenic axis.Atropisotopomers 9 and 10 exhibit the largest π-conjugated electronic systems among atropisotopomers, with two separated 14-electron aromatic circuits. This is a feature of these molecules that we have not yet examined, especially in terms of circularly polarized UV-vis light–matter interactions (absorption and emission) among other electronic properties. Mirror specific optical rotations with a magnitude of [α]25D ≈ 1 were recorded for the enantioisotopomers of the atropisotopomers 9 and 10. Specific optical rotations of comparable intensity have been reported for the few examples of highly enantio-enriched isotopically chiral molecules reported in the past decades. Using VCD in the solid state, it was possible to record sufficiently intense mirror signals for both enantioisotopomers of the atropisotopomers 9 and 10, which confirmed their absolute configurations (AC) with the greatest confidence. Comparable VCD analyses of the few highly enantioenriched [1H]/[2H] discriminated enantioisotopomers that have been successfully analyzed to date also allowed their AC to confidently be determined. In terms of the analysis of isotopic chirality in molecules, the greatest innovation in the work described herein is the first direct determination of the ee of both enantioisotopomers of an atropisotopomer using anisotropic NMR, which is the only available method to do so in condensed phases. The few previous successful applications of this analytical technique for the determination of the ee of isotopically chiral molecules have been limited to examples with central chirality.
Remarkably, it has been demonstrated to be possible to use both the 2H and the 13C nuclei as magnetically active probes to determine directly the ee of the samples of the enantioisotopomers of atropisotopomer 10, (aR)-10 and (aS)-10. In this case, the 2H-based NMR experiments provided more accurate information for the ee determination than the 13C-based NMR experiments, reflecting the relative magnetic sensitivity of both nuclei (ca. 100× sensitivity for [2H] vs. [13C]). Although ee determination by anisotropic NMR was only possible for atropisotopomer 10 under the examined conditions (using PBLG as the lyotropic CLC), the ee of ≥97% for both samples of (aR)-10 and (aS)-10 is notably the highest ee determined by this technique for any enantioisotopomers. This represents a major step forward in the analysis of isotopic chirality, which calls for further development (e.g., using other chiral lyotropic chiral phases).
The π-extended 9,9′-bianthryl platform used to forge atropisotopomers 9 and 10 is configurationally stable and cannot racemize at a significant rate at temperatures below 140 °C for centuries. Considering the availability of the enantiopure precursors (aR)-11 and (aS)-11 from BINOL (99% ee, 4 steps, 77%), the versatility of the synthetic options enabled by aryne chemistry,37 and the equally versatile synthetic options for derivatization offered by the C(sp2)–Br bond, we can hypothesize that the synthetic chemistry described herein for the prototypical atropisotopomers 9 and 10 is generalizable and extendable to any designed [1H]/[2H] discriminated atropisotopomers with a 9-anthryl or a 9,9′-bianthryl carbon backbone, as well as beyond, e.g., to those based on other isotopes such as [1H]/[3H], [2H]/[3H] and [12C]/[13C]38 discrimination. This offers simple, practical, and cheap synthetic access to π-extended atropisotopomer molecules to enable the development of precision analytical methods for the analysis of isotopic chirality using polarimetry, VCD, anisotropic NMR and more. The work described herein was conceived with the objective of propelling research in the domain of molecular chirality, from precision synthesis to precision analysis, and further development is on-going in our laboratories.
Author contributions
F. R. and G. D. performed the syntheses. J.-V. N. realized the VCD studies. P. L. and C. A. carried out the anisotropic NMR studies. Y. C. conceived the study, secured the funding, designed the synthesis experiments, and drafted the manuscript with revisions from all authors. All authors contributed to the discussion.Conflicts of interest
The authors declare no conflicts of interest.Data availability
The data supporting this article have been included as part of the supplementary information (SI). Supplementary information: detailed synthetic procedures, details of the polarimetry and spectroscopic analyses, and the theoretical determination of the rotation barrier in 9,9′-bianthryl. See DOI: https://doi.org/10.1039/d5sc09200k.Acknowledgements
This work was funded in part by the French Agence Nationale de la Recherche—ANR (ANR-19-CE07-0041). G. D. thanks the French MESR for a PhD fellowship. This work was also supported by the computing facilities of the Centre Régional de Compétences en Modélisation Moléculaire de Marseille—CRCMM. Institutional financial support from Aix Marseille University, Université Paris-Saclay, the Centre National de la Recherche Scientifique—CNRS, and Centrale Méditerranée is gratefully acknowledged. P. L. would like to thank the Île de France Region for partially funding the “Cryomorphose” project (grant EX069247).References
- (a) E. Benfante, Isotopes for the Nation's Future: Research Opportunities and Plans, Nova Science Publishers Inc, Hauppauge, 2012 Search PubMed; (b) Organisation for Economic Co-Operation and Development - Nuclear Energy Agency's, Beneficial Uses and Production of Isotopes, OECD Publishing, Paris, 2001 Search PubMed.
- R. M. Concetta Di Martino, B. D. Maxwell and T. Pirali, Deuterium in drug discovery: progress, opportunities and challenges, Nat. Rev. Drug Discovery, 2023, 22, 562 CrossRef PubMed.
- (a) P. R. Schreiner, Quantum Mechanical Tunneling Is Essential to Understanding Chemical Reactivity, Trends Chem., 2020, 2, 980 Search PubMed; (b) A. Danho, B. Bernhardt, D. Gerbig, M. Alešković and P. R. Schreiner, Cage Alkyl Carbenes Provide Experimental Evidence for Isotope-Controlled Selectivity in Competing Tunneling Reactions, J. Am. Chem. Soc., 2025, 147, 16717 CrossRef CAS PubMed.
- R. Gonfiantini, Standards for stable isotope measurements in natural compounds, Nature, 1978, 271, 534 Search PubMed.
- (a) E. L. Eliel and S. H. Wilen, Stereochemistry of Organic Compounds, Wiley, New York, 1994 Search PubMed; (b) A. Meddour, I. Canet, A. Loewenstein, J. M. Péchiné and J. Courtieu, Observation of Enantiomers, Chiral by Virtue of Isotopic Substitution, through Deuterium NMR in a Polypeptide Liquid Crystal, J. Am. Chem. Soc., 1994, 116, 9652 Search PubMed.
- (a) T. Kawasaki, Y. Matsumura, T. Tsutsumi, K. Suzuki, M. Ito and K. Soai, Asymmetric Autocatalysis Triggered by Carbon Isotope (13C/12C) Chirality, Science, 2009, 324, 492 Search PubMed; (b) T. Kawasaki, M. Shimizu, D. Nishiyama, M. Ito, H. Ozawa and K. Soai, Asymmetric autocatalysis induced by meteoritic amino acids with hydrogen isotope chirality, Chem. Commun., 2009, 2009, 4396 RSC.
- R. M. Williams, D. Zhai and P. J. Sinclair, Asymmetric synthesis of (R)- and (S)-[2-2H1]glycine, J. Org. Chem., 1986, 51, 5021 Search PubMed.
- T. Naret, P. Lesot, A. R. Puente, P. L. Polavarapu, D.-A. Buisson, J. Crassous, G. Pieters and S. Feuillastre, Chemical Synthesis of [2H]-Ethyl Tosylate and Exploration of Its Crypto-optically Active Character Combining Complementary Spectroscopic Tools, Org. Lett., 2020, 22, 8846 Search PubMed.
- M. D. Mills, R. E. Sonstrom, Z. P. Vang, J. L. Neill, H. N. Scolati, C. T. West, B. H. Pate and J. R. Clark, Enantioselective Synthesis of Enantioisotopomers with Quantitative Chiral Analysis by Chiral Tag Rotational Spectroscopy, Angew. Chem., Int. Ed., 2022, 61, e202207275 CrossRef CAS PubMed.
- J. Küppers, R. Rabus, H. Wilkes and J. Christoffers, Optically Active 1-Deuterio-1-phenylethane – Preparation and Proof of Enantiopurity, Eur. J. Org Chem., 2019, 2629 CrossRef.
- (a) J. Haesler, I. Schindelholz, E. Riguet, C. G. Bochet and W. Hug, Absolute configuration of chirally deuterated neopentane, Nature, 2007, 446, 526 Search PubMed; (b) A. Masarwa, D. Gerbig, L. Oskar, A. Loewenstein, H. P. reisenauer, P. Lesot, P. R. Schreiner and I. Marek, Synthesis and Stereochemical Assignment of Crypto-Optically Active 2H6-Neopentane, Angew. Chem., Int. Ed., 2015, 54, 13106 CrossRef CAS PubMed.
- (a) J. A. Berson and L. D. Pedersen, Thermal stereomutation of optically active trans-cyclopropane-1,2-d2, J. Am. Chem. Soc., 1975, 97, 238 CrossRef CAS; (b) S. J. Cianciosi, K. M. Spencer, T. B. Freedman, L. A. Nafie and J. E. Baldwin, Synthesis and gas-phase vibrational circular dichroism of (+)-(S,S)-cyclopropane-1,2-2H2, J. Am. Chem. Soc., 1989, 111, 1913 Search PubMed; (c) J. S. Chickos, M. Bausch and R. Alul, Stereospecific synthesis of optically active succinic-d2 acid, J. Org. Chem., 1981, 46, 3559 Search PubMed; (d) D. Cavagnat, L. Lespade and T. Buffeteau, Vibrational Absorption and Circular Dichroism Studies of trans-(3S,4S)-d6-Cyclopentene in the Gas Phase, J. Phys. Chem. A, 2007, 111, 7014 Search PubMed; (e) J. Drabowicz, A. Zajac, P. Lyzwa, P. J. Stephens, J.-J. Pan and F. J. Devlin, Determination of the absolute configurations of isotopically chiral molecules using vibrational circular dichroism (VCD) spectroscopy: the isotopically chiral sulfoxide, perdeuteriophenyl-phenyl-sulfoxide, Tetrahedron: Asymmetry, 2008, 19, 288 Search PubMed; (f) F. Saito, D. Gerbig, J. Becker and P. R. Schreiner, Breaking the Symmetry of a Meso Compound by Isotopic Substitution: Synthesis and Stereochemical Assignment of Monodeuterated cis-Perhydroazulene, Org. Lett., 2021, 23, 113 CrossRef CAS PubMed.
- K. Mislow, R. Graeve, A. J. Gordon and G. H. Wahl, Conformational Kinetic Isotope Effects in the Racemization of 9,10-Dihydro-4,5-dimethylphenanthrene, J. Am. Chem. Soc., 1964, 86, 1733 CrossRef CAS.
- M. Bloch, N. Lau, H. Musso and U.-I. Záhorszky, (S)-[3,3’-2H2]Bimesityl, Chem. Ber., 1972, 105, 1790 CrossRef CAS.
- (a) S. Miwa, R. Senda, K. Saito, A. Sato, Y. Nakamura and O. Kitagawa, Asymmetric Synthesis of Isotopic Atropisomers based on ortho-CH3/CD3 Discrimination and Their Structural Properties, J. Org. Chem., 2022, 87, 13501 Search PubMed; (b) Y. Watanabe, U. Take, R. Senda, A. Sato and O. Kitagawa, Synthesis of Atropisomeric Quinazolin-4-one Derivatives Based on Remote H/D and 12C/13C Discrimination, J. Org. Chem., 2025, 90, 784 Search PubMed.
- (a) A. Collet and J. Gabard, Optically Active (C3)-CycIotriveratrylene-d9. Energy Barrier for the “Crown to Crown” Conformational Interconversion of Its Nine-Membered-Ring System, J. Org. Chem., 1980, 45, 5400 CrossRef CAS; (b) J. Caneceill and A. Collet, Optical Activity due to Isotopic Substitution. Synthesis and Absolute Configuration of (+)- and (-)-(C3)-[2H3]Cyclotribenzylene ([2H3][1.1.1]Orthocyclophane), J. Chem. Soc., Chem. Commun., 1983, 1983, 1145 RSC; (c) O. Lafon, P. Lesot, H. Zimmermann, R. Poupko and Z. Luz, Chiral Discrimination in the 13C and 2H NMR of the Crown and Saddle Isomers of Nonamethoxy-Cyclotriveratrylene in Chiral Liquid-Crystalline Solutions, J. Phys. Chem. B, 2007, 111, 9453 Search PubMed; (d) P. Lesot, O. Lafon, H. Zimmermann and Z. Luz, Enantiodiscrimination in Deuterium NMR Spectra of Flexible Chiral Molecules with Average Axial Symmetry Dissolved in Chiral Liquid Crystals: The Case of Tridioxyethylenetriphenylene, J. Am. Chem. Soc., 2008, 130, 8754 Search PubMed.
- T. Brotin, N. Daugey, J. Kapitan, N. Vanthyune, M. Jean, E. Jeanneau and T. Buffeteau, Synthesis and Chiroptical Properties of a Chiral Isotopologue of syn-Cryptophane-B, J. Org. Chem., 2023, 88, 4829 CrossRef CAS PubMed.
- P. H. Hoffman, E. C. Ong, O. E. Weigang Jr. and M. J. Nugent, [2.2]Paracyclophane system optical activity. IV. Circular dichroism of (-)-(S)-4-deuterio[2.2]paracyclophane, J. Am. Chem. Soc., 1974, 96, 2620 CrossRef CAS.
- T. Miura, T. Nakamuro, S. G. Stewart, Y. Nagata and M. Murakami, Synthesis of Enantiopure C3-Symmetric Triangular Molecules, Angew. Chem., Int. Ed., 2017, 56, 3334 Search PubMed.
- An enantioselective synthesis of an enantioisotopomer would have been achieved recently, however without experimental verification of the AC and the ee of the product, which calls for caution in interpretation, see: P. Ren, Q. Zhao, Y. R. Chi and T. Zhu, Carbene-catalyzed enantioselective construction of a quasi-symmetrical spirocyclic hydroquinone with a minor chiral distinction, Chem. Sci., 2025, 16, 8940 Search PubMed.
- (a) E. R. Alexander and A. G. Pinkus, Optical Activity in Compounds Containing Deuterium. I. 2,3-Dideutero-trans-Menthane, J. Am. Chem. Soc., 1949, 71, 1786 CrossRef CAS; (b) E. L. Eliel, The Reduction of Optically Active Phenylmethylcarbinyl Chloride with Lithium Aluminum Deuteride, J. Am. Chem. Soc., 1949, 71, 3970 Search PubMed.
- (a) A. Collet and G. Gottarelli, Circular Dichroism of (C3)-Cyclotriveratrylene-d9: An Example of Exciton Optical Activity due to Isotopic Substitution, J. Am. Chem. Soc., 1981, 103, 5912 CrossRef CAS; (b) J. Canceill, A. Collet and G. Gottarelli, Optical activity due to isotopic substitution. Synthesis, stereochemistry, and circular dichroism of (+)- and (-)-[2,7,12-2H3]cyclotribenzylene, J. Am. Chem. Soc., 1984, 106, 5997 Search PubMed.
- L. A. Nafie, Vibrational Optical Activity: Principles and Applications, Wiley, Hoboken, 2011 Search PubMed.
- (a) P. Herwig, K. Zawatzky, M. Grieser, O. Heber, B. Jordon-Thaden, C. Krantz, O. Novotný, R. Repnow, V. Schurig, D. Schwalm, Z. Vager, A. Wolf, O. Trapp and H. Kreckel, Imaging the Absolute Configuration of a Chiral Epoxide in the Gas Phase, Science, 2013, 342, 1084 Search PubMed; (b) K. Zawatzky, P. Herwig, M. Grieser, O. Heber, B. Jordon-Thaden, C. Krantz, O. Novotný, R. Repnow, V. Schurig, D. Schwalm, Z. Vager, A. Wolf, H. Kreckel and O. Trapp, Coulomb Explosion Imaged Cryptochiral (R,R)-2,3-Dideuterooxirane: Unambiguous Access to the Absolute Configuration of (+)-Glyceraldehyde, Chem. – Eur. J., 2014, 20, 5555 Search PubMed.
- Z. P. Vang, R. E. Sonstrom, H. N. Scolati, J. R. Clark and B. H. Pate, Assignment of the absolute configuration of molecules that are chiral by virtue of deuterium substitution using chiral tag molecular rotational resonance spectroscopy, Chirality, 2023, 35, 856 Search PubMed.
- (a) K. Kimata, M. Kobayashi, K. Hosoya, T. Araki and N. Tanaka, Chromatographic Separation Based on Isotopic Chirality, J. Am. Chem. Soc., 1996, 118, 759 Search PubMed; (b) K. Kimata, K. Hosoya, T. Araki and N. Tanaka, Direct Chromatographic Separation of Racemates on the Basis of Isotopic Chirality, Anal. Chem., 1997, 69, 2610 CrossRef CAS PubMed; (c) W. H. Pirkle and K. Z. Gan, A direct chromatographic separation of enantiomers chiral by virtue of isotopic substitution, Tetrahedron: Asymmetry, 1997, 8, 811 Search PubMed.
- H. L. Holcomb, S. Nakanishi and T. C. Flood, Stereochemistry at Carbon of the Cyclometalation of 8-(a-Deuterioethyl)quinoline by Palladium(II) Salts, Organometallics, 1996, 15, 4228 CrossRef CAS.
- R. E. Sonstrom, Z. P. Vang, H. N. Scolati, J. L. Neill, B. H. Pate and J. R. Clark, Rapid Enantiomeric Excess Measurements of Enantioisotopomers by Molecular Rotational Resonance Spectroscopy, Org. Process Res. Dev., 2023, 27, 1185 Search PubMed.
- (a) P. Lesot, C. Aroulanda, P. Berdagué, A. Meddour, D. Merlet, J. Farjon, N. Giraud and O. Lafon, Multinuclear NMR in polypeptide liquid crystals: Three fertile decades of methodological developments and analytical challenges, Prog. Nucl. Magn. Reson. Spectrosc., 2020, 116, 85 Search PubMed; (b) C. Aroulanda and P. Lesot, Molecular enantiodiscrimination by NMR spectroscopy in chiral oriented systems: Concept, tools, and applications, Chirality, 2022, 34, 182 CrossRef CAS PubMed.
- (a) Y.-L. Wei, G. Dauvergne, J. Rodriguez and Y. Coquerel, Enantiospecic Generation and Trapping Reactions of Aryne Atropisomers, J. Am. Chem. Soc., 2020, 142, 16921 Search PubMed; (b) Y. Coquerel, Aryne Atropisomers: Chiral Arynes for the Enantiospecific Synthesis of Atropisomers and Nanographene Atropisomers, Acc. Chem. Res., 2023, 56, 86 CrossRef CAS PubMed.
- G. Dauvergne, N. Vanthuyne, M. Giorgi, J. Rodriguez, Y. Carissan and Y. Coquerel, Determination of the Rate Constant of the [4 + 2] Cycloaddition Between an Aryne Atropisomer and Furan in Solution, J. Org. Chem., 2022, 87, 11141 Search PubMed.
- G. Dauvergne, J.-V. Naubron, M. Giorgi, X. Bugaut, J. Rodriguez, Y. Carissan and Y. Coquerel, Enantiospecific Syntheses of Congested Atropisomers through Chiral Bis(aryne) Synthetic Equivalents, Chem. – Eur. J., 2022, e202202473 CrossRef CAS PubMed.
- J. D. Dunitz and R. M. Ibberson, Is Deuterium Always Smaller than Protium?, Angew. Chem., Int. Ed., 2008, 47, 4208 CrossRef CAS PubMed.
- H. Kovacs, D. Moskau and M. Spraul, Cryogenically cooled probes—a leap in NMR technology, Prog. Nucl. Magn. Reson. Spectrosc., 2005, 46, 131 Search PubMed.
- (a) D. Merlet, B. Ancian, J. Courtieu and P. Lesot, Two-dimensional Deuterium NMR Spectroscopy of Chiral Molecules Oriented in a Polypeptide Liquid Crystal: Application for the Enantiomeric Analysis through Natural Abundance Deuterium NMR, J. Am. Chem. Soc., 1999, 121, 5249 Search PubMed; (b) O. Lafon, P. Lesot, D. Merlet and J. Courtieu, Modified z-gradient filtering as a mean to obtain phased deuterium autocorrelation 2D NMR spectra in oriented solvents, J. Magn. Reson., 2004, 171, 135 Search PubMed.
- P. Lesot, O. Lafon, J. Courtieu and P. Berdagué, Analysis of the 13C NMR Spectra of Molecules, Chiral by Isotopic Substitution, Dissolved in a Chiral Oriented Environment: Towards the Absolute Assignment of the pro-R/pro-S Character of Enantiotopic Ligands in Prochiral Molecules, Chem. – Eur. J., 2004, 10, 3741 CrossRef CAS PubMed.
- J. Shi, L. Li and Y. Li, o-Silylaryl Trifates: A Journey of Kobayashi Aryne Precursors, Chem. Rev., 2021, 121, 3892 Search PubMed.
- The first [12C]/[13C] discriminated atropisotopomers (around a C–N stereogenic axis) were reported only recently: R. Senda, Y. Watanabe, S. Miwa, A. Sato and O. Kitagawa, Synthesis of Isotopic Atropisomers Based on 12C/13C Discrimination, J. Org. Chem., 2023, 88, 9579 CrossRef CAS PubMed.
| This journal is © The Royal Society of Chemistry 2026 |