Open Access Article
Open Access ArticleVisible-light-mediated synthesis of 2-oxetanes via Giese addition to α-oxy radicals
Matthew
Liu
 a,
Elvis C.
McFee
a,
Elvis C.
McFee
 a and
Corinna S.
Schindler
a and
Corinna S.
Schindler
 *abc
*abc
aDepartment of Chemistry, University of British Columbia, 2036 Main Mall, Vancouver, BC V6T 1Z1, Canada. E-mail: schindler@chem.ubc.ca
bDepartment of Biochemistry and Molecular Biology, University of British Columbia, 2350 Health Sciences Mall, Vancouver, BC V6T 1Z3, Canada
cBC Cancer Research Institute, 675 West 10th Ave, Vancouver, BC V5Z 1L3, Canada
First published on 6th February 2026
Abstract
Oxetanes have recently garnered large amounts of interest from synthetic chemists due to their unique structural and chemical properties. Despite substantial advances for the formation of 3,3-disubstituted oxetanes, its 2-functionalized counterparts remain a challenging synthetic motif. Furthermore, methods to form alkyl oxetanes remain underdeveloped. Herein, we disclose a novel protocol for the synthesis of various alkyl and aryl 2-oxetanes leveraging a Giese addition to α-oxy radicals, furnishing the desired products in up to 95% yield.
Introduction
Oxetanes are considered privileged motifs in modern drug discovery due to their unique ability to modulate key physicochemical properties, including lipophilicity, metabolic stability, and aqueous solubility.1–6 Their incorporation into bioactive molecules holds the potential to improve pharmacokinetic profiles, making them attractive scaffolds for medicinal chemists.1,5,7 As a result, there is growing interest in developing efficient strategies for the functionalization of oxetane scaffolds to enable structure–activity relationship (SAR) exploration and diversification of lead compounds.7 Among the potential sites for derivatization, the 3-position of aryl-substituted oxetanes has been most extensively targeted,8–17 in part due to the ability to generate stabilized benzylic radicals under photoredox-catalyzed conditions.18 For example, visible-light-promoted decarboxylative alkylation of 3-aryl-oxetanes (1) enables the generation of benzylic radicals that engage efficiently in C–C bond-forming reactions, providing streamlined access to 3-functionalized oxetanes (2, Fig. 1). This reactivity is facilitated by radical stabilization from the adjacent aromatic ring, and multiple methods rely on this strategy to allow for alkylation and arylation at the 3-position (3 to 4, Fig. 1).18 | ||
| Fig. 1 Oxetane decarboxylation and functionalization. Previous reports and this study. (A) Previous work highlighting methods to functionalize oxetanes at the 3- and 2- position.18,19 (B) Current work demonstrating the synthesis of various alkyl and aryl 2-oxetanes via a Giese addition to α-oxy radicals. | ||
In contrast, the functionalization of oxetanes at the 2-position remains a significant synthetic challenge. The lack of electronic stabilization at this site renders radical intermediates less accessible (6), and few methods have been developed to directly forge C–C bonds at the 2-position of oxetanes.20–26 Recently, progress has been made by harnessing decarboxylative activation of 2-aryl-substituted oxetane carboxylic acids (7) to generate α-oxy radicals.19 Under photoredox conditions, these intermediates can engage in bond-forming reactions, offering rare examples of direct substitution at the 2-position (8). However, such strategies remain limited in scope and are typically restricted to aryl-substituted systems.19 To address this limitation, the present work establishes a general approach for the synthesis of 2-substituted oxetanes (10) via Giese-type addition of α-oxy radicals to electron-deficient alkenes. Notably, Mykhailiuk and co-workers recently successfully developed a Giese-addition approach from azetidine-2-carboxylic acids to access functionalized azetidines.27 Using readily available oxetane carboxylic acids (9) to serve as radical precursors, and under visible-light irradiation, decarboxylation generates transient α-oxy radicals that can add to activated olefins to form C–C bonds at the 2-position (B. Fig. 1). This method proceeds under mild conditions, tolerates a variety of alkyl and aryl substituents, and provides a valuable platform for expanding the synthetic utility of oxetanes in the context of drug development.
Results and discussion
To begin our studies towards the development of a novel protocol for the visible-light-mediated synthesis of various alkyl and aryl 2-oxetanes via a Giese addition to α-oxy radicals, oxetane-2-carboxylic acid (9) and 4-vinylpyridine (10) were chosen as model substrates and optimal conditions were explored (Table 1). Upon irradiating 2.0 equivalents of alkene (10) with oxetane-2-carboxylic acid (9) in conjunction with an excess of base in 0.2 M DMF, the desired product can be observed in 70% yield (entry 1 and Table 1). Varying the base used (entry 2 and Table 1) (see SI for additional details) revealed productive reactivity, although LiOH·H2O was found to provide the best product yield. Importantly, evaluation of various photocatalysts revealed that both organic and metal-based photocatalysts were sufficient in providing reactivity, including Ir1, 4CzIPN, and MesAcr1 (entry 4, 5 and Table 1). Subsequent efforts focused on varying the reaction solvent identified DMF as being superior, providing the highest product yield compared to other solvents, although reactivity was still observed with ethyl acetate and THF (59% and 62% product yield, respectively) (see SI for additional details). Furthermore, when increasing alkene loading to 2.5 equivalents, a slight increase in product yield was observed. Finally, decreasing reaction concentration from 0.2 M to 0.1 M highlighted an increase in product yield from 73 to 77% (entry 6, 7 and Table 1). By performing the reaction open to air, a noticeable decrease in product yield was observed (11% reduction in product yield) (see SI for additional information). Optimal reaction conditions using 2.5 mol% of Ir1, 2.5 equivalents of alkene 10, 1.5 equivalents of LiOH·H2O, in 0.1 M DMF (degassed for 2 minutes) were found to furnish the desired product in up to 80% yield. Control experiments were also conducted, highlighting that both light and base were required to provide productive reactivity (entries 9, 10 and Table 1), with temperature playing no significant role in product yield with the absence of light (entry 11 and Table 1).| Entry | Photocat. (mol%) | Alkene (equiv.) | Base (equiv.) | Conc. (M) | Yield (%) |
|---|---|---|---|---|---|
| a Average yields over 3 runs. n.r = No Reactivity. | |||||
| 1 | Ir1 PF6 (2.5) | 2.0 | LiOH·H2O (1.1) | 0.2 | 70 |
| 2 | Ir1 PF6 (2.5) | 2.0 | Cs2CO3 (1.1) | 0.2 | 68 |
| 3 | Ir2 (2.5) | 2.0 | LiOH·H2O (1.1) | 0.2 | n.r |
| 4 | MesAcr1 (2.5) | 2.0 | LiOH·H2O (1.1) | 0.2 | 21 |
| 5 | 4CzIPN (2.5) | 2.0 | LiOH·H2O (1.5) | 0.2 | 72 |
| 6 | Ir1 PF6 (2.5) | 2.0 | LiOH·H2O (1.5) | 0.2 | 73 |
| 7 | Ir1 PF6 (2.5) | 2.0 | LiOH H2O (1.5) | 0.1 | 77 |
| 8 | Ir1 PF6 (2.5) | 2.5 | LiOH·H2O (1.5) | 0.1 | 80a |
| 9 | No light | n.r | |||
| 10 | No.base | n.r | |||
| 11 | No light 40 °C | n.r | |||

|
|||||
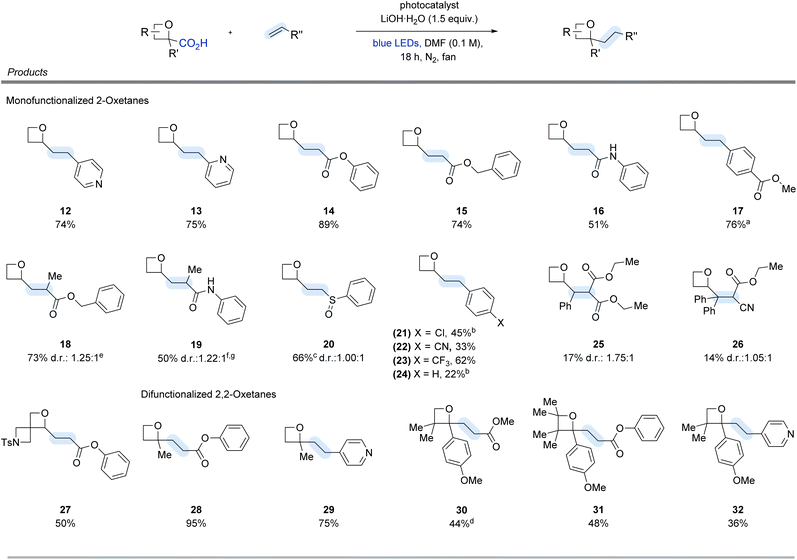
To demonstrate the utility of our protocol, we subjected various mono- and di-functionalized 2-oxetanes and alkene partners to optimized or modified conditions to assess their reactivity. For monofunctionalized 2-oxetanes (Fig. 2), acrylates such as phenyl- and benzyl acrylate gave the desired products 14 and 15 in in 89% and 74% yield, respectively. Styrene was also a viable radical acceptor, affording the desired oxetane 24, albeit with lower yields (22%). Various styrene derivatives, including methyl 4-vinylbenzoate and 4-chlorostyrene also furnished the corresponding oxetane products 17 and 21 in 76% and 45% yield, respectively. Furthermore, α-methyl-substituted alkenes such as benzyl methacrylate and N-phenylmethacrylamide afforded products 18 and 19 in 73% and 50% yield, respectively. Importantly, heteroaryl substrates such as 4-vinyl- (12) and 2-vinylpyridine (13) were well tolerated, underscoring the method's applicability to late-stage functionalization of medicinally relevant compounds. Importantly, alkenes bearing a tri- or tetra-substituted (25 and 26, respectively) pattern were also amenable to reactivity. Oxetanes bearing an azetidinyl substituents (27) also furnished the desired product in 50% yield, demonstrating the implementation of this protocol even in spirocyclic scaffolds. To further highlight the generality of our method, a range of difunctionalized 2,2-oxetane carboxylic acids were subjected to reactivity. Using 2-methyloxetane-2-carboxylic acid in conjunction with either phenyl acrylate or 4-vinylpyridine, the desired 2,2-difunctionalized oxetanes (28 and 29) can be obtained in 95% and 75% yield, respectively. Furthermore, leveraging recent results from our lab on the hydrodecarboxylation of 2-aryl oxetanes via the generation of an α-oxy radical,19 we envisioned an extension of this work in applying substrates towards this process, thereby accessing the corresponding Giese adducts (30–32). Di- and tetra-substituted 2-aryl oxetanes such as 2-(4-methoxyphenyl)-3,3,4,4-tetramethyloxetane-2-carboxylic acid and 2-(4-methoxyphenyl)-3,3-dimethyloxetane-2-carboxylic acid were observed to undergo productive reactivity with either 4-vinylpyridine (32, 36%), methyl acrylate (30, 44%), or phenyl acrylate (31, 48%).
Finally, the utility of our process was further exemplified with a 1.0 mmol scale up reaction (Fig. 3), which is shown to effectively produce the corresponding product 14 in 82% yield.
Given that photoredox decarboxylative Giese reactions typically proceed via a reductive quenching pathway, the following mechanism is proposed (A, Fig. 4).28–30 Following photocatalyst excitation of the ground state photocatalyst via visible-light, the corresponding photoexcited species (*Ir1) engages in an oxidative single electron transfer (SET) process with 31, made accessible by a base-mediated deprotonation of 30. Facile decarboxylation followed by the conjugate addition to a Michael-acceptor generates the carbon-centered radical intermediate 33. The reduced photocatalyst species (Ir1red) participates in a second single electron transfer with intermediate 33, reducing it to the corresponding carbanion 34, in addition to the regeneration of Ir1, completing the catalytic cycle.29 The carbanion intermediate 34 is subsequently protonated, resulting in the desired 2-functionalized oxetane product 35. We next sought out to investigate the impact of the α-oxy radical on radical philicity (B and C, Fig. 4). Recently, Nagib and coworkers present an experimentally validated database of radical polarities31,† Using this, alkenes with varying radical polarities values (ω) were investigated and the corresponding oxetane product yields show a correlation between product yields and increasing electrophilicity in the alkene (B and C, Fig. 4), suggesting that the α-oxy radical formed may be inherently nucleophilic.
Conclusions
We herein developed a novel method for the synthesis of various alkyl and aryl 2-oxetanes through a visible-light-mediated decarboxylation and subsequent Giese addition of the resulting α-oxy radicals. Our protocol provides a framework that is complementary to existing strategies for functionalization of 3-oxetanes.8–18 This method contributes to the synthetic toolbox for medicinal and organic chemists interested in incorporating oxetanes as bioisosteres1,4,32–38 for modulating the physicochemical activity of drugs or for generating building blocks for drug discovery.Author contributions
M. L and E. C. M designed and conducted experiments. All authors have given approval to the final version of the manuscript.Conflicts of interest
The authors declare no competing financial interest.Data availability
All data supporting the findings of this study are available within the article and its supplementary information (SI) file. Supplementary information: general information, experimental procedures, characterization data, and spectroscopic data. See DOI: https://doi.org/10.1039/d5sc07764h.Acknowledgements
This work was supported by the Natural Sciences and Engineering Research Council of Canada (NSERC). This research was performed, in part, with funding from the Canada Research Chairs Program. M. L thanks S. A. Chamness for helpful discussions. The authors thank J. Zhu for performing mass spectrometry and P. Xu for aid in setting up NMR spectroscopic parameters. The authors thank D. A. Nagib and K. Mikhaeel for their support with radical polarity calculations.Notes and references
- G. Wuitschik, M. Rogers-Evans, K. Müller, H. Fischer, B. Wagner, F. Schuler, L. Polonchuk and E. M. Carreira, Oxetanes as Promising Modules in Drug Discovery, Angew. Chem., Int. Ed., 2006, 45(46), 7736–7739, DOI:10.1002/anie.200602343.
- J. A. Burkhard, G. Wuitschik, M. Rogers-Evans, K. Müller and E. M. Carreira, Oxetanes as Versatile Elements in Drug Discovery and Synthesis, Angew. Chem., Int. Ed., 2010, 49(48), 9052–9067, DOI:10.1002/anie.200907155.
- A. F. Stepan, K. Karki, W. S. McDonald, P. H. Dorff, J. K. Dutra, K. J. DiRico, A. Won, C. Subramanyam, I. V. Efremov, C. J. O'Donnell, C. E. Nolan, S. L. Becker, L. R. Pustilnik, B. Sneed, H. Sun, Y. Lu, A. E. Robshaw, D. Riddell, T. J. O'Sullivan, E. Sibley, S. Capetta, K. Atchison, A. J. Hallgren, E. Miller, A. Wood and R. S. Obach, Metabolism-Directed Design of Oxetane-Containing Arylsulfonamide Derivatives as γ-Secretase Inhibitors, J. Med. Chem., 2011, 54(22), 7772–7783, DOI:10.1021/jm200893p.
- J. A. Burkhard, G. Wuitschik, J.-M. Plancher, M. Rogers-Evans and E. M. Carreira, Synthesis and Stability of Oxetane Analogs of Thalidomide and Lenalidomide, Org. Lett., 2013, 15(17), 4312–4315, DOI:10.1021/ol401705a.
- J. J. Rojas and J. A. Bull, Oxetanes in Drug Discovery Campaigns, J. Med. Chem., 2023, 66(18), 12697–12709, DOI:10.1021/acs.jmedchem.3c01101.
- J. A. Bull, R. A. Croft, O. A. Davis, R. Doran and K. F. Morgan, Oxetanes: Recent Advances in Synthesis, Reactivity, and Medicinal Chemistry, Chem. Rev., 2016, 116(19), 12150–12233, DOI:10.1021/acs.chemrev.6b00274.
- J. J. Rojas and J. A. Bull, Oxetanes in Drug Discovery Campaigns, J. Med. Chem., 2023, 66(18), 12697–12709, DOI:10.1021/acs.jmedchem.3c01101.
- S. A. Green, S. Vásquez-Céspedes and R. A. Shenvi, Iron–Nickel Dual-Catalysis: A New Engine for Olefin Functionalization and the Formation of Quaternary Centers, J. Am. Chem. Soc., 2018, 140(36), 11317–11324, DOI:10.1021/jacs.8b05868.
- Y. Zhang, Y. Zhang and X. Shen, Alkoxyl-Radical-Mediated Synthesis of Functionalized Allyl Tert-(Hetero)Cyclobutanols and Their Ring-Opening and Ring-Expansion Functionalizations, Chem Catal., 2021, 1(2), 423–436, DOI:10.1016/j.checat.2021.03.014.
- J. J. Rojas, R. A. Croft, A. J. Sterling, E. L. Briggs, D. Antermite, D. C. Schmitt, L. Blagojevic, P. Haycock, A. J. P. White, F. Duarte, C. Choi, J. J. Mousseau and J. A. Bull, Amino-Oxetanes as Amide Isosteres by an Alternative Defluorosulfonylative Coupling of Sulfonyl Fluorides, Nat. Chem., 2022, 14(2), 160–169, DOI:10.1038/s41557-021-00856-2.
- R. A. Croft, J. J. Mousseau, C. Choi and J. A. Bull, Structurally Divergent Lithium Catalyzed Friedel–Crafts Reactions on Oxetan-3-Ols: Synthesis of 3,3-Diaryloxetanes and 2,3-Dihydrobenzofurans, Chem.–Eur. J., 2016, 22(45), 16271–16276, DOI:10.1002/chem.201604031.
- R. A. Croft, J. J. Mousseau, C. Choi and J. A. Bull, Lithium-Catalyzed Thiol Alkylation with Tertiary and Secondary Alcohols: Synthesis of 3-Sulfanyl-Oxetanes as Bioisosteres, Chem.–Eur. J., 2018, 24(4), 818–821, DOI:10.1002/chem.201705576.
- R. A. Croft, J. J. Mousseau, C. Choi and J. A. Bull, Oxetane Ethers Are Formed Reversibly in the Lithium-Catalyzed Friedel–Crafts Alkylation of Phenols with Oxetanols: Synthesis of Dihydrobenzofurans, Diaryloxetanes, and Oxetane Ethers, Tetrahedron, 2018, 74(38), 5427–5435, DOI:10.1016/j.tet.2018.02.069.
- M. A. J. Dubois, M. A. Smith, A. J. P. White, A. Lee Wei Jie, J. J. Mousseau, C. Choi and J. A. Bull, Short Synthesis of Oxetane and Azetidine 3-Aryl-3-Carboxylic Acid Derivatives by Selective Furan Oxidative Cleavage, Org. Lett., 2020, 22(14), 5279–5283, DOI:10.1021/acs.orglett.0c01214.
- M. A. J. Dubois, A. R. Croft, Y. Ding, C. Choi, D. R. Owen, J. A. Bull and J. J. Mousseau, Investigating 3,3-Diaryloxetanes as Potential Bioisosteres through Matched Molecular Pair Analysis, RSC Med. Chem., 2021, 12(12), 2045–2052, 10.1039/D1MD00248A.
- P. J. Saejong, J. Rojas, C. P. Denis, A. J. White, A. Sophie Voisin-Chiret, C. Choi and A. J. Bull, Synthesis of Oxetane and Azetidine Ethers as Ester Isosteres by Brønsted Acid Catalysed Alkylation of Alcohols with 3-Aryl-Oxetanols and 3-Aryl-Azetidinols, Org. Biomol. Chem., 2023, 21(27), 5553–5559, 10.1039/D3OB00731F.
- J. J. Rojas and J. A. Bull, 4-Membered Ring Carbocations: A Positive Development in the Synthesis of 3,3-Disubstituted Oxetanes and Azetidines, Chimia, 2023, 77(4), 192–195, DOI:10.2533/chimia.2023.192.
- M. A. J. Dubois, J. J. Rojas, A. J. Sterling, H. C. Broderick, M. A. Smith, A. J. P. White, P. W. Miller, C. Choi, J. J. Mousseau, F. Duarte and J. A. Bull, Visible Light Photoredox-Catalyzed Decarboxylative Alkylation of 3-Aryl-Oxetanes and Azetidines via Benzylic Tertiary Radicals and Implications of Benzylic Radical Stability, J. Org. Chem., 2023, 88(10), 6476–6488, DOI:10.1021/acs.joc.3c00083.
- E. C. McFee, K. A. Rykaczewski and C. S. Schindler, Photoredox-Catalyzed Decarboxylation of Oxetane-2-Carboxylic Acids and Unique Mechanistic Insights, Angew. Chem., Int. Ed., 2025, 64(5), e202405125, DOI:10.1002/anie.202405125.
- J. L. Sloane, A. B. Santos, E. M. Simmons and T. C. Sherwood, Metallaphotoredox-Mediated Decarboxylative Cross-Coupling of α-Oxy Morpholine Carboxylic Acids and (Hetero)Aryl Halides, Org. Lett., 2023, 25(23), 4219–4224, DOI:10.1021/acs.orglett.3c01478.
- D. Ravelli, M. Zoccolillo, M. Mella and M. Fagnoni, Photocatalytic Synthesis of Oxetane Derivatives by Selective C–H Activation, Adv. Synth. Catal., 2014, 356(13), 2781–2786, DOI:10.1002/adsc.201400027.
- D. F. Fernández, M. González-Esguevillas, S. Keess, F. Schäfer, J. Mohr, A. Shavnya, T. Knauber, D. C. Blakemore and D. W. C. MacMillan, Redefining the Synthetic Logic of Medicinal Chemistry. Photoredox-Catalyzed Reactions as a General Tool for Aliphatic Core Functionalization, Org. Lett., 2024, 26(14), 2702–2707, DOI:10.1021/acs.orglett.3c00994.
- J. Jin and D. W. C. MacMillan, Direct α-Arylation of Ethers through the Combination of Photoredox-Mediated C–H Functionalization and the Minisci Reaction, Angew. Chem., Int. Ed., 2015, 54(5), 1565–1569, DOI:10.1002/anie.201410432.
- S. Paul, D. Filippini, F. Ficarra, H. Melnychenko, C. Janot and M. Silvi, Oxetane Synthesis via Alcohol C–H Functionalization, J. Am. Chem. Soc., 2023, 145(29), 15688–15694, DOI:10.1021/jacs.3c04891.
- K. F. Morgan, R. Doran, R. A. Croft, I. A. Hollingsworth and J. A. Bull, 2-Sulfinyl Oxetanes: Synthesis, Stability and Reactivity, Synlett, 2015, 27, 106–110, DOI:10.1055/s-0035-1560588.
- A. O. Davis, A. R. Croft and A. J. Bull, Synthesis of Diversely Functionalised 2,2-Disubstituted Oxetanes: Fragment Motifs in New Chemical Space, Chem. Commun., 2015, 51(84), 15446–15449, 10.1039/C5CC05740J.
- O. P. Datsenko, A. Baziievskyi, I. Sadkova, B. Campos, J. T. I. Brewster, J. Kowalski, R. J. Hinklin and P. K. Mykhailiuk, Alkyl Azetidines Via Batch and Flow Photochemistry, Org. Lett., 2025, 27(21), 5318–5323, DOI:10.1021/acs.orglett.5c00922.
- J. D. Griffin, M. A. Zeller and D. A. Nicewicz, Hydrodecarboxylation of Carboxylic and Malonic Acid Derivatives via Organic Photoredox Catalysis: Substrate Scope and Mechanistic Insight, J. Am. Chem. Soc., 2015, 137(35), 11340–11348, DOI:10.1021/jacs.5b07770.
- M. D. Kitcatt, S. Nicolle and A.-L. Lee, Direct Decarboxylative Giese Reactions, Chem. Soc. Rev., 2022, 51(4), 1415–1453, 10.1039/D1CS01168E.
- L. Chu, C. Ohta, Z. Zuo and D. W. C. MacMillan, Carboxylic Acids as A Traceless Activation Group for Conjugate Additions: A Three-Step Synthesis of (±)-Pregabalin, J. Am. Chem. Soc., 2014, 136(31), 10886–10889, DOI:10.1021/ja505964r.
- J. J. A. Garwood, A. D. Chen and D. A. Nagib, Radical Polarity, J. Am. Chem. Soc., 2024, 146(41), 28034–28059, DOI:10.1021/jacs.4c06774.
- G. Wuitschik, M. Rogers-Evans, A. Buckl, M. Bernasconi, M. Märki, T. Godel, H. Fischer, B. Wagner, I. Parrilla, F. Schuler, J. Schneider, A. Alker, W. B. Schweizer, K. Müller and E. M. Carreira, Spirocyclic Oxetanes: Synthesis and Properties, Angew. Chem., Int. Ed., 2008, 47(24), 4512–4515, DOI:10.1002/anie.200800450.
- G. Wuitschik, E. M. Carreira, B. Wagner, H. Fischer, I. Parrilla, F. Schuler, M. Rogers-Evans and K. Müller, Oxetanes in Drug Discovery: Structural and Synthetic Insights, J. Med. Chem., 2010, 53(8), 3227–3246, DOI:10.1021/jm9018788.
- J. S. Scott, A. M. Birch, K. J. Brocklehurst, H. S. Brown, K. Goldberg, S. D. Groombridge, J. A. Hudson, A. G. Leach, P. A. MacFaul, D. McKerrecher, R. Poultney, P. Schofield and P. H. Svensson, Optimisation of Aqueous Solubility in a Series of G Protein Coupled Receptor 119 (GPR119) Agonists, MedChemComm, 2012, 4(1), 95–100, 10.1039/C2MD20130E.
- M. McLaughlin, R. Yazaki, T. C. Fessard and E. M. Carreira, Oxetanyl Peptides: Novel Peptidomimetic Modules for Medicinal Chemistry, Org. Lett., 2014, 16(16), 4070–4073, DOI:10.1021/ol501590n.
- G. P. Möller, S. Müller, B. T. Wolfstädter, S. Wolfrum, D. Schepmann, B. Wünsch and E. M. Carreira, Oxetanyl Amino Acids for Peptidomimetics, Org. Lett., 2017, 19(10), 2510–2513, DOI:10.1021/acs.orglett.7b00745.
- P. Lassalas, K. Oukoloff, V. Makani, M. James, V. Tran, Y. Yao, L. Huang, K. Vijayendran, L. Monti, J. Q. Trojanowski, V. M.-Y. Lee, M. C. Kozlowski, A. B. Smith, K. R. Brunden and C. Ballatore, Evaluation of Oxetan-3-Ol, Thietan-3-Ol, and Derivatives Thereof as Bioisosteres of the Carboxylic Acid Functional Group, ACS Med. Chem. Lett., 2017, 8(8), 864–868, DOI:10.1021/acsmedchemlett.7b00212.
- P. Mukherjee, M. Pettersson, J. K. Dutra, L. Xie and C. W. am Ende, Trifluoromethyl Oxetanes: Synthesis and Evaluation as a Tert-Butyl Isostere, ChemMedChem, 2017, 12(19), 1574–1577, DOI:10.1002/cmdc.201700333.
Footnote |
| † Radical polarity values for 4-vinylpyridine were provided by D. A. Nagib and K. Mikhaeel by personal communication. |
| This journal is © The Royal Society of Chemistry 2026 |