 Open Access Article
Open Access ArticleEffects of activating agent and pyrolysis temperature on the pore structure and adsorption performance of sweet potato vine-based activated carbon
Wei Wei * and
Na Ren
* and
Na Ren
College of Biochemical Engineering, Beijing Union University, No. 18 Sanqu, Fatouxili, Chaoyang District, Beijing, 100023, China. E-mail: wlweiwei@buu.edu.cn; Fax: +86 10 52072157; Tel: +86 10 52072157
First published on 7th April 2026
Abstract
China produces a large amount of sweet potato vines annually that require processing. In this study, sweet potato vine was used as a precursor to prepare activated carbons with two chemical activating agents (ZnCl2 and K2CO3). The obtained activated carbons were characterized by N2 adsorption/desorption isotherms, scanning electron microscopy (SEM), and Fourier transform infrared (FTIR) spectroscopy. The synthesized activated carbons were subsequently applied for the adsorption of methylene blue (MB). The effects of the activating agent and pyrolysis temperature on the properties and adsorption capacity of the activated carbons were investigated. The activated carbon prepared with K2CO3 at 800 °C (CK800) exhibits the highest surface area (1523.9 m2 g−1) and total pore volume (0.7247 cm3 g−1), and is composed of micropores and mesopores. In contrast, the sample activated with ZnCl2 at 500 °C (CZn500) shows a surface area of 1347.2 m2 g−1 and is dominated by mesopores with a mesopore volume of 1.0953 cm3 g−1. Both activated carbons exhibited high adsorption capacities for methylene blue. Their adsorption isotherms followed the Langmuir model, with maximum adsorption capacities reaching 775.2 mg g−1 (CK800) and 400 mg g−1 (CZn500), respectively. The adsorption kinetics were consistent with the pseudo-second-order kinetic model. This is primarily attributed to their large BET surface area and high total pore volume. Simultaneously, mechanisms such as electrostatic attraction, π–π stacking, hydrophobic interaction, and hydrogen bonding also make important contributions to the materials' high adsorption performance. The prepared materials demonstrate that sweet potato vine can serve as a biomass feedstock for the production of activated carbon. Different preparation methods can impart distinct functionalities and physicochemical properties to the activated carbon. By selecting appropriate activating agents and pyrolysis temperatures, it is possible to successfully produce sweet potato vine-based activated carbon with high BET specific surface area, large pore volume, and excellent methylene blue adsorption performance.
1 Introduction
Dyes are extensively utilized across multiple industries including the textiles, leather, paper, and printing industries. However, their discharge poses significant environmental concerns, as dye contamination not only pollutes water bodies but also adversely affects aquatic flora and fauna, ultimately threatening human health through bioaccumulation in the food chain. The adsorption process serves as an effective method for treating dye-containing wastewater. Activated carbon has been widely employed as an adsorbent due to its excellent adsorption performance. However, its large-scale application remains limited by high production costs. Recently, researchers have shown growing interest in developing activated carbons from agricultural and industrial wastes, owing to their environmental sustainability, cost-effectiveness, and remarkable adsorption capacity. Various agricultural waste materials have been investigated as potential precursors, including sugarcane bagasse,1,2 spent coffee grounds,3 shells,4–8 bamboo waste,9 stones,10–12 seeds,13,14 peels,15–18 stalk19 and leaves.20,21 Despite the substantial annual production of vine biomass, its utilization as a precursor for activated carbon preparation has received minimal attention. Only a limited number of studies have reported on this.22China produces over 20 million tons of sweet potatoes annually, accounting for approximately 30% of the global yield. During harvesting, only a small portion of sweet potato vines (SPV) are utilized as animal feed, while the majority has traditionally been discarded through field burning – a practice now prohibited due to its environmental impact. SPV represent an attractive raw material due to their low cost, abundant availability, renewability, and environmental friendliness. Chemically, SPV primarily consist of cellulose, hemicellulose, lignin, and minor mineral components.23 These characteristics make SPV a promising precursor for activated carbon production. Nevertheless, limited studies have explored the utilization of SPV for this purpose. This study aimed to develop cost-effective activated carbon from SPV and evaluate its efficacy in removing textile dyes from aqueous solutions. The production of activated carbon from biomass typically involves two key steps: carbonization and activation. In this work, we employed a one-stage method using different chemical activating agents (ZnCl2 and K2CO3), combining pyrolysis and activation in a single step to optimize energy efficiency. The results demonstrate that SPV can be effectively converted into high-quality activated carbon with large BET surface area and pore volume when processed with suitable activating agents and optimal pyrolysis temperatures. The resulting activated carbons exhibited excellent adsorption performance for methylene blue (MB) removal from aqueous solutions. These findings collectively demonstrate that SPV-derived activated carbon represents a promising and cost-effective adsorbent for practical applications.
2 Materials and methods
2.1 Material and reagents
SPV were collected from a local farm in Beijing, China. The fresh vines were first air-dried at ambient temperature, followed by oven-drying until constant weight was achieved. The dried material was then ground into fine powder. MB was procured from Shanghai Chemical Reagent Factory (Shanghai, China). All other chemical reagents, obtained from Lanyi Chemical Reagent Company (Beijing, China), were of analytical grade purity.2.2 Preparation of activated carbons
The SPV powder was separately impregnated with ZnCl2 and K2CO3 solutions for 24 hours, using mass ratios of 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 (SPV
2 (SPV![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) ZnCl2) and 1
ZnCl2) and 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1.5 (SPV
1.5 (SPV![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) K2CO3), respectively. After impregnation, the samples were first air-dried at room temperature, followed by oven-drying at 100 °C for 12 hours. The dried mixtures then underwent simultaneous carbonization and activation under argon atmosphere at various predetermined temperatures. The resulting products were cooled, washed with deionized water, and finally oven-dried at 100 °C to constant weight. The obtained activated carbons were labeled as CZnx and CKx, where ‘x’ represents the carbonization/activation temperature, and the prefixes indicate the activating agent used (ZnCl2 or K2CO3, respectively).
K2CO3), respectively. After impregnation, the samples were first air-dried at room temperature, followed by oven-drying at 100 °C for 12 hours. The dried mixtures then underwent simultaneous carbonization and activation under argon atmosphere at various predetermined temperatures. The resulting products were cooled, washed with deionized water, and finally oven-dried at 100 °C to constant weight. The obtained activated carbons were labeled as CZnx and CKx, where ‘x’ represents the carbonization/activation temperature, and the prefixes indicate the activating agent used (ZnCl2 or K2CO3, respectively).
2.3 Characterization of activated carbons
The surface morphology of activated carbons was examined using field-emission scanning electron microscopy (Sigma 300, Zeiss, Germany). Surface functional groups were characterized by Fourier-transform infrared spectroscopy (FT-IR, PerkinElmer, USA). Pore structure analysis was performed through nitrogen physisorption measurements using a Micromeritics ASAP 2020 analyzer (USA). Elemental analysis was performed using an elemental analyzer (EA 2400 II, PerkinElmer, USA) operating in CHN mode. Samples were combusted at 950 °C in an oxygen-rich atmosphere, and the resulting gases (CO2, H2O, and N2) were separated by frontal chromatography and detected by a thermal conductivity detector (TCD). Acetanilide was used as a calibration standard.For pHPZC determination, selected activated carbons were added to 50 mL of 0.1 mol L−1 NaCl solution. The initial pH was adjusted to values ranging from 2 to 11 using 0.1 M HCl or NaOH solutions. The sealed flasks were then agitated in a thermostatic shaker for 48 hours. The pHPZC was identified as the point where the final pH curve intersected the initial pH line (ΔpH = 0).24
2.4 Adsorption experiments
For adsorption experiments, 50 mg of activated carbon samples were added to 250 mL glass flasks containing 100 mL of MB solutions at varying concentrations. The flasks were agitated in a thermostatic shaker until adsorption equilibrium was achieved. Sampling was performed at predetermined time intervals throughout the experiments. MB concentrations were quantified using a UV-vis spectrophotometer at 640 nm wavelength, with reference to a pre-established calibration curve. The equilibrium adsorption capacity, qe (mg g−1), was calculated using the following equation:
 | (1) |
The amount of adsorption at time t, qt (mg g−1) was calculated by the following equation:
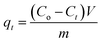 | (2) |
The pH effect studies were conducted by mixing 50 mg of selected activated carbons with 100 mL MB solution. The solution pH was systematically adjusted to values between 2 and 10 using 0.1 M HCl or NaOH solutions.
CK800 and CZn500 were regenerated by soaking in a mixture of 0.1 M HCl and ethanol (50% v/v) with ultrasonic treatment for 30 minutes, followed by washing with deionized water until neutral. After drying, the regenerated samples were reused for the adsorption of MB to evaluate their reusability.
3 Results and discussion
3.1 Characterization of activated carbons
| Samples | Elemental content (%) | SBET (m2 g−1) | Vtotal (cm3 g−1) | Vmicro (cm3 g−1) | Vmeso (cm3 g−1) | Vmi Vt−1 (%) | Vme Vt−1 (%) | Pore size (nm) | ||
|---|---|---|---|---|---|---|---|---|---|---|
| C | N | H | ||||||||
| CK500 | 68.17 | 2.10 | 1.89 | 532.9 | 0.2846 | 0.2001 | 0.0845 | 70.31 | 29.69 | 2.14 |
| CK600 | 72.67 | 1.92 | 1.67 | 684.6 | 0.3466 | 0.2722 | 0.0744 | 78.53 | 21.47 | 2.03 |
| CK700 | 75.39 | 0.80 | 1.53 | 1193.1 | 0.5857 | 0.4368 | 0.1489 | 74.58 | 25.42 | 1.96 |
| CK800 | 77.26 | 0.67 | 1.46 | 1523.9 | 0.7247 | 0.4266 | 0.2981 | 58.87 | 41.13 | 1.90 |
| CZn400 | 58.92 | 2.28 | 2.26 | 671.0 | 0.3499 | 0.1279 | 0.2220 | 36.55 | 63.45 | 2.09 |
| CZn500 | 60.70 | 2.10 | 1.54 | 1347.2 | 1.0953 | 0.000 | 1.0953 | 0 | 100 | 3.25 |
| CZn600 | 62.61 | 2.31 | 1.62 | 1186.3 | 0.8367 | 0.0262 | 0.8105 | 3.13 | 96.87 | 2.82 |
| CZn700 | 70.80 | 3.41 | 1.24 | 932.1 | 0.7658 | 0.0168 | 0.7490 | 2.19 | 97.81 | 3.28 |
Fig. 1 presents the N2 adsorption–desorption isotherms and corresponding pore size distributions of activated carbons prepared with different chemical activating agents and pyrolysis temperatures. As summarized in Table 1, these materials exhibit distinct BET surface areas, pore volumes, and average pore diameters. The results clearly demonstrate that the choice of chemical activator and thermal treatment conditions significantly influence the textural properties of the resulting activated carbons, producing materials with varying surface areas, pore size distributions, and pore volumes.
 | ||
| Fig. 1 N2 adsorption–desorption isotherms and pore size distributions of activated carbons prepared with K2CO3 (a and b) and ZnCl2 (c and d) at various carbonization/activation temperatures. | ||
The N2 adsorption–desorption isotherms of CK samples reveal a distinct evolution in pore structure with increasing carbonization temperature. CK500, CK600, and CK700 display typical Type-I isotherms, confirming their predominantly microporous nature, which is further supported by the pore size distribution and pore volume analyses. In contrast, the isotherms of CK800 exhibit hysteresis loops characteristic of Type-IV behavior, indicating the development of mesoporosity. CK800 features a mixed pore structure comprising both micropores and mesopores. This is likely attributable to the collapse of micropores or their blockage by volatiles under high temperatures. Both BET surface area and total pore volume demonstrate a positive correlation with pyrolysis temperature, increasing progressively from 500 to 800 °C. Remarkably, the sample CK800 achieved a maximum BET surface area of 1523.9 m2 g−1 accompanied by a pore volume of 0.7247 cm3 g−1, significantly surpassing commercial activated carbons.25,26 These findings underscore the critical role of pyrolysis temperature in determining pore characteristics. During thermal treatment, cellulose, hemicellulose, and lignin undergo degradation, dehydration, and carbonization, leading to carbon skeleton aromatization and pore development. The K2CO3 activator decomposes under argon atmosphere to form K, K2O, CO, and CO2. When the pyrolysis temperature exceeds the boiling point of potassium (759 °C), metallic potassium diffuses into the carbon matrix and reacts with the carbon, promoting the decomposition of volatile organic compounds and thereby forming a well-developed porous structure. Simultaneously, under high-temperature conditions, CO2 can undergo an oxidation reaction with the disordered carbon present in the already formed biochar (C + CO2 → 2CO), which selectively etches the carbon skeleton, gradually forming and expanding its microporous and mesoporous structures.
The N2 adsorption–desorption isotherms of CZn samples exhibit Type IV characteristics with distinct hysteresis loops in the relative pressure range of 0.55–0.99, confirming the presence of mesoporous structures. These observations are further supported by the corresponding pore size distribution and pore volume analyses. The textural properties show a temperature-dependent trend, where both BET surface area and total pore volume initially increase with carbonization temperature up to 500 °C before decreasing at higher temperatures. The maximum values achieved at 500 °C (1347.2 m2 g−1surface area and 1.0953 cm3 g−1 pore volume) are comparable to mesoporous carbons (e.g., 1267 m2 g−1 and 0.96 cm3 g−1, respectively) as reported in literature.27 The activation mechanism involves several key processes: (1) ZnCl2, as a hygroscopic salt, induces cellular dehydration through osmotic pressure, enhancing precursor porosity; (2) during pyrolysis, water reacts with zinc chloride to form zinc oxide hydrate, simultaneously generating HCl that contributes to pore enlargement; (3) elevated temperatures (400–500 °C) intensify these reactions, promoting volatile compound formation and consequent pore development. The observed surface area reduction above 500 °C likely results from structural collapse of the porous framework under excessive thermal treatment.
Table 2 presents a comparative analysis of BET surface areas and total pore volumes for various biomass-derived activated carbons reported in previous studies. The results demonstrate that the SPV-based activated carbons prepared using ZnCl2 and K2CO3 activation exhibit comparable textural properties to many other biomass-derived carbons, indicating their potential for adsorption applications.
| Material | Activation method | BET surface area (m2 g−1) | Total pore volume (cm3 g−1) | Average pore size (nm) | Reference |
|---|---|---|---|---|---|
| Rice husks | ZnCl2 | 749.51 | 0.9956 | 1.53 | 2 |
| Sugarcane bagasse | ZnCl2 | 1386.58 | 0.9947 | 1.55 | 2 |
| Loofah vine | KOH | 904.6 | 0.677 | 2.99 | 28 |
| Orange peel | H3PO4 | 79.73 | 0.03 | 20.1 | 29 |
| Tamarind seeds | KOH | 1172 | 0.618 | 2.1 | 13 |
| Spent coffee grounds | KOH | 827.91 | 2.7 | 3 | |
| Olive stone | H3PO4 | 710 | 1.25 | 11 | |
| Sugarcane bagasse | KOH | 234.58 | 1 | ||
| Bamboo biomass | Na2SiO | 381.6 | 0.52 | 5.43 | 9 |
| Groundnut shell | KOH | 412 | 0.322 | 7 | |
| Pine fruit shells | H3PO4 | 1022.13 | 0.566 | 2.215 | 6 |
| Coconut shells | NaOH | 345 | 0.2 | 1.92 | 8 |
| Palm leave | KOH | 540 | 0.261 | 5.13 | 21 |
| Khat leaves | KOH | 340 | 3.93 | 3.7 | 20 |
| Black garlic peels | 463.9 | 0.373 | 17 | ||
| Banana peels | H3PO4 | 867 | 0.032 | 16 | |
| Palm stones | CO2 | 19.53 | 0.007 | 1.43 | 12 |
| Coconut shells | Alkali from cocoa pod husks | 1250.20 | 0.61 | 2.754 | 30 |
| Plane tree seeds | NaOH | 1508.6 | 0.7063 | 2.0462 | 26 |
| Cellulose | H3PO4 | 561 | 31 | ||
| Commercial activated carbon | 1185 | 0.57 | 26 | ||
| Sweet potato vine | ZnCl2 | 1347.2 | 1.0953 | 3.25 | This work |
| Sweet potato vine | K2CO3 | 1523.9 | 0.7247 | 1.90 | This work |
![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O bonds); 1537 cm−1 (C
O bonds); 1537 cm−1 (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C aromatic ring vibrations); 1160 cm−1 (C–O stretching in ether/ester groups); 875 cm−1 (C–H bending vibrations). These spectral features confirm the presence of oxygen-containing functional groups and aromatic structures on the carbon surfaces. The temperature-dependent variations in peak intensities reflect the thermal stability differences between the two activation systems.
C aromatic ring vibrations); 1160 cm−1 (C–O stretching in ether/ester groups); 875 cm−1 (C–H bending vibrations). These spectral features confirm the presence of oxygen-containing functional groups and aromatic structures on the carbon surfaces. The temperature-dependent variations in peak intensities reflect the thermal stability differences between the two activation systems.
3.2 Adsorptive removal of MB using activated carbons
According to Polanyi's adsorption potential theory,32 the adsorption potential ε is inversely related to pore size (dp, nm). In microporous carbons, the characteristic adsorption energy E0 (kJ mol−1) follows the Dubinin–Stoeckli relationship:33,34
| E0 = 15.8/dp | (3) |
This relationship indicates that micropores (<2 nm) generate significantly higher adsorption potentials than mesopores (2–50 nm). For dye molecules such as MB (molecular dimensions ∼1.4 nm × 0.6 nm × 0.4 nm), the enhanced interaction arises from the superposition of Lennard-Jones potentials from opposing pore walls,35 leading to strong adsorption through micropore filling at low concentrations.36
Thus, the superior adsorption capacity of CK700 (478 mg g−1) compared to CZn600 (369 mg g−1) is related to its abundant micropore structure, which provides a higher adsorption potential and facilitates effective micropore filling. In contrast, the mesopore-dominated structure of CZn600 limits its adsorption primarily to surface coverage, resulting in a lower overall capacity despite its comparable specific surface area.
FTIR analysis was conducted to further elucidate the adsorption mechanism. After MB adsorption (Fig. 2), several new peaks characteristic of MB appeared at approximately 1500, 1417, 1395, 1330, and 880–800 cm−1, corresponding to aromatic C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C stretching, and C–N stretching/–CH3 bending.37,38 Notably, the peak at 1417 cm−1 shifted from its typical position (ca. 1395 cm−1), indicating electrostatic interactions between the cationic MB molecules and the carbon surface.39,40 Meanwhile, the aromatic C
C stretching, and C–N stretching/–CH3 bending.37,38 Notably, the peak at 1417 cm−1 shifted from its typical position (ca. 1395 cm−1), indicating electrostatic interactions between the cationic MB molecules and the carbon surface.39,40 Meanwhile, the aromatic C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C peak at ∼1600 cm−1 exhibited enhanced intensity and a slight shift, suggesting π–π stacking interactions between MB and the graphitic microcrystallites of the activated carbon.41,42 Raman spectra (Fig. S3) show that the activated carbon is composed mainly of amorphous carbon with some graphitic crystallites. The effect of solution pH on adsorption capacity (Fig. 6b) confirms that electrostatic attraction plays a significant role, particularly for CZn500, where the surface charge transitions from positive to negative around its pHPZC of 6.68. Furthermore, the hydrophobic nature of the carbon surface promotes hydrophobic interactions with the MB molecule. These results suggest that MB adsorption is governed by a combination of π–π stacking and electrostatic interactions, complementing the physical adsorption associated with the pore structure. Based on the characterization results and adsorption experiments, a multi-faceted adsorption mechanism is proposed, as illustrated in Fig. 4. The MB adsorption capacity of the prepared activated carbon indicates promising performance under the specific batch conditions used in this study.31,43–48
C peak at ∼1600 cm−1 exhibited enhanced intensity and a slight shift, suggesting π–π stacking interactions between MB and the graphitic microcrystallites of the activated carbon.41,42 Raman spectra (Fig. S3) show that the activated carbon is composed mainly of amorphous carbon with some graphitic crystallites. The effect of solution pH on adsorption capacity (Fig. 6b) confirms that electrostatic attraction plays a significant role, particularly for CZn500, where the surface charge transitions from positive to negative around its pHPZC of 6.68. Furthermore, the hydrophobic nature of the carbon surface promotes hydrophobic interactions with the MB molecule. These results suggest that MB adsorption is governed by a combination of π–π stacking and electrostatic interactions, complementing the physical adsorption associated with the pore structure. Based on the characterization results and adsorption experiments, a multi-faceted adsorption mechanism is proposed, as illustrated in Fig. 4. The MB adsorption capacity of the prepared activated carbon indicates promising performance under the specific batch conditions used in this study.31,43–48
The activated carbons CK800 and CZn500 were selected for adsorption studies. The adsorption capacity of CK800 and CZn500 were maintained about 75% (535.8 mg g−1) and 78% (304.2 mg g−1) respectively after four adsorption-regeneration cycles, showing good reusability. Fig. 5 presents the MB adsorption isotherms and kinetics for these two materials. The isotherm data were analyzed using the Langmuir, Freundlich and Temkin adsorption models to characterize the equilibrium distribution of adsorbed molecules between the liquid and solid phases. The calculated isotherm parameters and corresponding correlation coefficients (R2) for tested models are presented in Table 3. As summarized in Table 3, the Langmuir isotherm model provided the best fit to the experimental data, demonstrating excellent linearity (R2 = 0.999) across the entire concentration range studied. This strong correlation suggests that MB adsorption on both CK800 and CZn500 is reasonably well described by the Langmuir equation over the tested concentration range, although biochar and activated carbon often possess heterogeneous surfaces.
 | ||
| Fig. 5 Effects of MB concentration (A) and contact time (B) on the adsorption of onto activated carbons. | ||
| Samples | Langmuir | Freundlich | Temkin | ||||||
|---|---|---|---|---|---|---|---|---|---|
| qmax (mg g−1) | KL (L g−1) | R2 | KF ((mg g−1) (L mg−1)1/n) | n | R2 | KT (L g−1) | B (J mol L−1) | R2 | |
| CK800 | 775.2 | 0.364 | 0.999 | 734.689 | 29.507 | 0.987 | 5.19 × 1011 | 23.610 | 0.983 |
| CZn500 | 400.0 | 1.923 | 0.999 | 242.759 | 8.326 | 0.600 | 1.06 × 104 | 28.132 | 0.754 |
According to the Langmuir model calculations, the maximum adsorption capacities of CK800 and CZn500 are 775.2 mg g−1 and 400.0 mg g−1, respectively. To investigate the adsorption mechanism, two kinetic models – pseudo-first-order and pseudo-second-order – were employed to analyze the adsorption process. The results demonstrate that MB adsorption followed the pseudo-second-order kinetic model, with the corresponding parameters summarized in Table 4.
| Samples | Pseudo-first-order model | Pseudo-second-order model | ||||
|---|---|---|---|---|---|---|
| qe (mg g−1) | k1 (h−1) | R2 | qe (mg g−1) | k2 (g mg−1 h−1) | R2 | |
| CK800 | 7.258 | 0.635 | 0.339 | 438.6 | 0.1165 | 0.999 |
| CZn500 | 176 | 0.324 | 0.939 | 383.1 | 0.385 | 0.999 |
To further elucidate the rate-controlling steps, the kinetic data were fitted to the intraparticle diffusion model (Fig. S4 and Table 5) and Boyd model (Fig. S5). The adsorption process of CK-800 toward the target adsorbate exhibits a distinct three-stage characteristic, governed by a combined rate control of film diffusion and intraparticle diffusion. The fitting results of the intraparticle diffusion model show that in the first stage, the rate constant kid1 = 544.29 mg g−1 h−0.5 and the intercept C1 = 126.62 > 0, indicating that film diffusion (boundary layer effect) dominates the initial stage with a fast adsorption rate. In the second stage, the rate constant decreases to kid2 = 174.90 mg g−1 h−0.5 and the intercept C2 = 308.59 > 0, suggesting that intraparticle diffusion becomes the main rate-controlling step, while film diffusion still contributes. In the third stage, the rate constant approaches zero (kid3 = 1.29), indicating that adsorption reaches equilibrium. The progressive decrease in rate constants (kid1 > kid2 ≫ kid3) and the gradual increase in intercepts (C1 < C2 < C3) across the three stages are consistent with the typical characteristics of multi-step adsorption. Meanwhile, the Boyd plot is nonlinear and does not pass through the origin, further confirming that intraparticle diffusion is not the sole rate-controlling step, and that the microporous structure of CK-800 hinders pore diffusion, causing deviation from the ideal Fickian diffusion behavior. Integrating the results from both models, the adsorption process of CK-800 is governed by a combined control of film diffusion and intraparticle diffusion, with hindered diffusion within the microporous structure being the fundamental reason for the reduced intraparticle diffusion rate.
| Samples | First stage of intra-particle diffusion | Second stage of intra-particle diffusion | Third stage of intra-particle diffusion | ||||||
|---|---|---|---|---|---|---|---|---|---|
| Kid1 (mg g−1 h−0.5) | C1 | R2 | Kid2 (mg g−1 h−0.5) | C2 | R2 | Kid3 (mg g−1 h−0.5) | C3 | R2 | |
| CK800 | 544.29 | 126.62 | 0.964 | 174.90 | 308.59 | 0.932 | 1.29 | 433.82 | 0.284 |
| CZn500 | 207.93 | 0.5422 | 0.9903 | 85.84 | 168.85 | 0.9997 | 12.12 | 319.06 | 0.9531 |
For the mesoporous CZn-500, the adsorption process toward the target adsorbate is dominated by intraparticle diffusion. The fitting results of the intraparticle diffusion model show three distinct linear stages. In the first stage, the intercept C1 = 0.5422 is very close to zero, indicating that film diffusion (boundary layer effect) makes a negligible contribution in the initial stage. In the second stage, the rate constant decreases to kid2 = 85.84 mg g−1 h−0.5, and intraparticle diffusion becomes the main rate-controlling step. In the third stage, the rate constant kid3 = 12.12 mg g−1 h−0.5 remains relatively high compared to microporous materials, reflecting the favorable diffusion properties enabled by the mesoporous structure. The Boyd plot exhibits good linearity (R2 = 0.931) with an intercept of 0.278, further confirming that intraparticle diffusion is the predominant rate-controlling step, with only a minor contribution from boundary layer effects.
In comparison with the microporous material CK-800, which exhibits a combined control mechanism of film diffusion and intraparticle diffusion, the mesoporous structure of CZ500 effectively reduces pore diffusion resistance, leading to adsorption behavior that more closely approximates the ideal intraparticle diffusion-controlled model.
4 Conclusion
Activated carbons were prepared from SPV using ZnCl2 and KCO3 as activating agents, respectively. Their performance as adsorbents was then evaluated for the removal of MB from aqueous solutions. Experimental results demonstrate that the prepared activated carbons possess high surface areas and total pore volumes. The activated carbon synthesized with K2CO3 predominantly features micropores at lower temperatures, while exhibiting a micro-mesoporous composite structure at elevated temperatures. As the pyrolysis temperature increases, the specific surface area and total pore volume of activated carbons both show an increasing trend. The sample prepared at 800 °C achieved a surface area of 1523.9 m2 g−1 with a total pore volume of 0.7247 cm3 g−1. In contrast, the ZnCl2-activated carbon exhibits a mesopore-dominated porous structure, with the sample treated at 500 °C showing the highest specific surface area and mesopore volume, reaching 1347.2 m2 g−1 and 1.0953 cm3 g−1, respectively. Both the activated carbons exhibited exceptional adsorption performance for methylene blue. The adsorption process was influenced by initial dye concentration, contact time, and solution pH. The adsorption process was well described by the Langmuir isothermal model, suggesting a homogeneous monolayer adsorption. Kinetic studies indicated that the adsorption followed pseudo-second-order kinetics.This approach effectively addresses the challenges related to the disposal and management of agricultural waste. Furthermore, it contributes to lower production costs, diversifies the raw material sources for activated carbon, and may offer potential for supporting the treatment of dye-contaminated wastewater.
Author contributions
Wei Wei: supervision, funding acquisition, conceptualization, writing – original draft. Na Ren: data curation, formal analysis, writing – review & editing.Conflicts of interest
The authors declare no competing financial interest.Data availability
The data that support the findings of this study are available from the corresponding author upon reasonable request.Supplementary information (SI): XRD, Raman, adsorption isotherms, fitting curves of Webber–Morris intra-particle diffusion and Boyd model. See DOI: https://doi.org/10.1039/d6ra01057a.
Acknowledgements
This work was financially supported by the National Natural Science Foundation of China (No. 51772031).References
- D. Dermawan, S. I. C. Karno, T. A. Ramadani, D. R. Widiana, A. C. Ni’am, Y.-F. Wang and A. Setiawan, Miner. Eng., 2025, 232, 109575 Search PubMed.
- E. R. Raut, M. A. Bedmohata and A. R. Chaudhari, Mater. Today: Proc., 2022, 66, 1875–1884 CAS.
- R. Campbell, B. Xiao and C. Mangwandi, J. Environ. Manage., 2024, 366, 121682 Search PubMed.
- I. A. Olowonyo, K. K. Salam, M. O. Aremu and A. Lateef, Waste Manag. Bull., 2024, 1, 217–233 Search PubMed.
- R. S. Brishti, R. Kundu, M. A. Habib and M. H. Ara, Results Chem., 2023, 5, 100727 Search PubMed.
- I. Hamadneh, R. A. Abu-Zurayk, A. Aqel, A. Al-Mobydeen, L. Hamadneh, Y. Al-Dalahmeh, F. Hannoon, R. Albuqain, S. Alsotari and A. H. Al-Dujaili, Desalination Water Treat., 2022, 264, 293–306 Search PubMed.
- D. Mani, D. Elango, A. Priyadharsan, L. A. Al-Humaid, N. D. Al- Dahmash, S. Ragupathy, P. Jayanthi and Y.-H. Ahn, Environ. Res., 2023, 231, 116026 CrossRef CAS.
- J. Saleem, Z. K. B. Moghal, S. Pradhan and G. McKay, RSC Adv., 2024, 14, 33797–33808 RSC.
- Y. Guo and Q. Wang, Environ. Technol. Innovation, 2023, 32, 103318 CrossRef CAS.
- J. De Smedt, P. J. Arauzo and F. Ronsse, Bioresour. Technol., 2025, 419, 132040 CrossRef CAS PubMed.
- K. Haddad, M. Ghafer, H. Salman and H. El-Rakkad, Results Chem., 2025, 14, 102137 CrossRef CAS.
- A. A. Ali and H. M. Abdul-Hameed, Desalination Water Treat., 2025, 321, 100951 CrossRef CAS.
- P. Janthabut, P. Athikaphan, P. Jekruk, P. Kanchunsi, P. Kongkoed, S. Nijpanich, S. Theerakulpisut, A. Neramittagapong and S. Neramittagapong, Environ. Chall., 2025, 20, 101243 CrossRef CAS.
- P. M. Thabede and N. D. Shooto, S. Afr. J. Chem. Eng., 2025, 53, 256–266 Search PubMed.
- A. Martín-Lorenzo, M. Hoyos and A. Álvarez-Gómez, J. Anal. Appl. Pyrolysis, 2025, 192, 107280 CrossRef.
- S. Phal, C. Guigui, A. Didi, R. Tan and Y. Bessiere, J. Water Proc. Eng., 2025, 77, 108495 CrossRef.
- D. Duan, S. Xu, Y. Zhou, H. Mo, W. Yang, Y. Zhang and R. Ruan, J. Anal. Appl. Pyrolysis, 2025, 191, 107229 CrossRef CAS.
- A. S. Almaghrabi, E. M. Bakhsh, K. Akhtar, M. A. Alghamdi and S. B. Khan, Results Chem., 2025, 15, 102277 CrossRef CAS.
- B. Ren, X. Han, Y. Jin, Y. Zhang, X. Song, L. Zhao and R. Liu, J. Environ. Chem. Eng., 2025, 13, 118989 CrossRef CAS.
- A. A. Alswat, A. M. Ashmali, T. M. Alqasmi, H. R. Alhassani and F. T. Alshorifi, Heliyon, 2023, 9, e14301 CrossRef CAS PubMed.
- M. A. Islam, M. K. Nazal, A. A. Akinpelu, M. Sajid, N. A. Alhussain, R. E. K. Billah and L. Bahsis, Diamond Relat. Mater., 2024, 147, 111375 CrossRef CAS.
- G. Qin, C. Wang, Q. Yang and W. Wei, Desalination Water Treat., 2023, 283, 237–246 CrossRef CAS.
- T. Wang, X. Dong, Z. Jin, W. Su, X. Ye, C. Dong and Q. Lu, Bioresour. Technol., 2015, 192, 799–801 CrossRef CAS PubMed.
- F. Marrakchi, M. Bouaziz and B. H. Hameed, Colloids Surf., A, 2017, 535, 157–165 CrossRef CAS.
- K. Kuśmierek, L. Dąbek and A. Świątkowski, Desalination Water Treat., 2025, 321, 100996 CrossRef.
- V. Dodevski, M. Marinović-Cincović and B. Janković, Carbon Resour. Convers., 2025, 100377 Search PubMed.
- M. Marzuki, A. H. Jawad, N. I. I. M. Zazulir, R. R. R. Deris, R. Wu and Z. A. Alothman, Diamond Relat. Mater., 2025, 159, 112869 CrossRef CAS.
- C. Wang, Q. Yang, N. Ren, Z. Zhao, W. Wei and G. Qin, Chem. Eng. Commun., 2022, 209, 62–68 CrossRef CAS.
- A. I. Licona-Aguilar, A. M. Torres-Huerta, M. A. Domínguez-Crespo, M. L. X. Negrete-Rodríguez, E. Conde-Barajas, S. B. Brachetti-Sibaja and A. E. Rodríguez-Salazar, Chem. Eng. J., 2024, 492, 152102 CrossRef CAS.
- D. T. Ojo, O. S. Amuda, K. K. Salam, O. F. Oyediran and B. B. Yekini, Next Nanotechnol., 2025, 8, 100198 CrossRef CAS.
- N. Anter, A. Hannioui, A. Medaghri-Alaoui and M. Ghazoui, RSC Adv., 2025, 15, 49780–49795 RSC.
- M. Polanyi, Science, 1963, 141, 1010–1013 CrossRef CAS PubMed.
- M. M. Dubinin, Carbon, 1989, 27, 457–467 CrossRef CAS.
- D. E. Morel and F. Stoeckli, Chimia, 1980, 34, 503–504 Search PubMed.
- R. D. Kaminsky, W. C. Conner and E. Maglara, Langmuir, 1994, 10, 1556–1565 CrossRef CAS.
- C. H. Giles, T. H. MacEwan, S. N. Nakhwa and D. Smith, J. Chem. Soc., 1960, 3973–3993, 10.1039/JR9600003973.
- A. Kumar and K. P. Srivastava, Orient. J. Chem., 2002, 18, 2 Search PubMed.
- A. Javed, M. Islam, Y. O. Al-Ghamdi, M. Iqbal, M. Aljohani, S. Sohni, S. S. A. Shah and S. A. Khan, Int. J. Biol. Macromol., 2024, 256, 128363 CrossRef CAS PubMed.
- Y. Li, Q. Du, T. Liu, J. Sun, Y. Wang, S. Wu, Z. Wang, Y. Xia and L. Xia, Carbohydr. Polym., 2013, 95, 501–507 CrossRef CAS PubMed.
- A. Bhattacharyya, S. Ghorai, D. Rana, I. Roy, G. Sarkar, N. R. Saha, J. T. Orasugh, S. De, S. Sadhukhan and D. Chattopadhyay, Mater. Chem. Phys., 2021, 260, 124090 CrossRef CAS.
- C. E. Anson, C. S. Creaser and G. Richard Stephenson, Spectrochim. Acta, Part A, 1996, 52, 1183–1191 CrossRef.
- W. Xiao, X. Jiang, X. Liu, W. Zhou, Z. N. Garba, I. Lawan, L. Wang and Z. Yuan, J. Clean. Prod., 2021, 284, 124773 CrossRef CAS.
- D. D. Anuse, S. A. Patil, A. A. Chorumale, A. G. Kolekar, P. P. Bote, L. S. Walekar, M. G. Mali and S. P. Pawar, Clean. Water, 2025, 4, 100118 CrossRef.
- S. R. Mishra, T. Mandal, P. Kumar, V. Verma, R. N. Senapati, M. Kumar and V. Singh, Surf. Interfaces, 2025, 69, 106770 CrossRef CAS.
- Y. Liu, J. Wang, L. Xu, D. Qian and Y. Mu, J. Environ. Chem. Eng., 2025, 13, 118317 CrossRef CAS.
- A. A. R. Andrade, M. C. Silva, L. H. S. Crespo, T. L. Silva, A. L. Cazetta, L. Spessato and V. C. Almeida, J. Mol. Liq., 2025, 427, 127435 CrossRef CAS.
- X. Fan, B. Qiu, X. Tian and X. Yang, Microporous Mesoporous Mater., 2025, 394, 113625 CrossRef CAS.
- M. Usama, S. Atta, M. N. Qasim, A. Q. Alshammari, A. K. Alanazi, N. A. Khan, M. Mateen and M. N. Akhtar, RSC Adv., 2025, 15, 46553–46569 RSC.
| This journal is © The Royal Society of Chemistry 2026 |




