 Open Access Article
Open Access ArticleA review of lead leakage monitoring in perovskite solar cells: emerging detection technologies, just transition to clean energy and perspectives
Benjamin K. Korira,
Samantha Ndlovub,
Vincent O. Nyamori b and
Joshua K. Kibet
b and
Joshua K. Kibet *a
*a
aDepartment of Chemistry, Egerton University, PO. Box 536-20115, Njoro, Kenya. E-mail: jkibet@egerton.ac.ke
bSchool of Agriculture and Science, University of KwaZulu-Natal, Westville Campus, Private Bag X54001, Durban 4000, South Africa
First published on 27th February 2026
Abstract
Lead-based perovskite solar cells (LPSCs) offer promise for low-cost, highly efficient alternatives to current solar energy technologies, with the potential for scale-up production and high-throughput coating. However, lead leakage, which is considered a health hazard, instability due to moisture intrusion into the perovskite core, and degradation challenges remain unresolved. Without reliable monitoring and mitigation strategies, the scalability of LPSCs raises regulatory and environmental problems. Currently, systematic detection and quantification of lead (Pb) leakage are scarcely developed. Therefore, there is urgent need for real-time monitoring systems for better Pb leakage detection and monitoring to ensure public and environmental health protection. Accordingly, leakage of Pb into the environment has far-reaching etiological risks to animal and plant health. For instance, the bioaccumulation of Pb in the human body can exacerbate the development of cardiovascular diseases and damage to the central nervous system. This study provides a detailed overview of the approaches used to monitor leakage in perovskite solar cells (PSCs), closing the loop in Pb recycling, and the outlook for perovskite recycling. Here, we analyse Pb leakage monitoring and prevention approaches undertaken in recent years by reviewing scholarly databases. This study also explores the latest and emerging detection technologies, such as spectroscopic methods, electrochemical sensors, fluorescence assays, and in situ monitoring techniques. Just transition toward green energy access together with environmental sustainability has been explored in the event that LPSCs are ultimately deployed and commercialised. Further, the potential of artificial intelligence (AI) systems has been reviewed to understand leakage events associated with the degradation of LPSCs. To this end, this review not only provides up-to-date state-of-the-art research and developments in perovskite research but also provides insights into suitable Pb leakage monitoring and prevention strategies toward mechanically stable and environmentally sustainable LPSCs.
1. Introduction
Increasing public awareness and justification for the adoption of solar energy by energy consumers is the primary factor in revitalising the acceptance of the photovoltaic technology.1,2 Concurrently, as perovskite solar cell (PSC) technology is projected to grow, environmental safety is a major concern that must be balanced with affordability and efficiency. While solar energy offers significant reductions in environmental pollution, energy justice has emerged as an important concept that highlights equity, inclusivity and fairness in the transition to clean energy. The anticipated widespread deployment of photovoltaics raises concerns about environmental implications related to resource extraction, hazardous materials, and end-of-life management, highlighting the need to address these issues through the lens of just transitions (JT) and equitable distributions of benefits and burdens.3 Accordingly, the growing demand for renewable energy alternatives, particularly driven by their favourable ratio of efficiency and cost, has gained significant momentum in the 21st century.4 However, the degradation of lead-based perovskite solar cells (LPSCs), such as methylammonium lead triiodide (MAPbI3), poses a significant challenge to solar cell stability and has unprecedented consequences for environmental and public health.5 Whereas perovskite solar deployments have achieved rapid advancements in terms of efficiencies, there are issues that need to be addressed like the adoption of efficient Pb leakage monitoring strategies and methods for mitigating Pb pollution from LPSCs. Further, its commercial deployment has been hindered by its susceptibility to temperature and moisture, and more significantly, the toxicity of lead (Pb).6 Currently, there is a lack of an appropriate treatment of MAPbI3 to mitigate Pb pollution and to determine how this hazardous material can be recovered and recycled. Different methods, including the most versatile inductive coupled plasma hyphenated with mass spectrometry (ICP-MS) or inductive coupled plasma atomic emission spectroscopy (ICP-AES), have been employed to quantify hazardous elements in environmental matrices,7 including analytical techniques such as potentiometric and atomic adsorption spectroscopy.8 However, ICP-MS and ICP-AES require highly trained experts and well-equipped laboratories.9 Trace elements in the environment and food have also been determined using electrochemical methods, particularly voltammetry techniques.The United Nations Sustainable Development Goal (SDG #7) highlights the global recognition and support for secure, affordable, and readily accessible renewable energy, climate action (SDG #13), and reduced inequalities (SDG #10). As the world strives to accomplish the challenge of “net zero” carbon in the context of SDGs, solar cell technologies have undergone the fastest rate of growth.10 The International Renewable Energy Agency (IRENA) has predicted the dominance of photovoltaics in the coming years due to low costs and potentially high efficiencies.11 Remarkably, organic–inorganic hybrid halide (CH3NH3PbX3, X = Cl, Br, I) PSCs offer promise for the advancement of photovoltaic technologies.12 Perovskites exhibit solution-processability and a unique combination of optoelectronic properties that have enabled these devices to achieve efficiencies exceeding 27%.13 Different from lightweight properties, perovskites are semi-transparent and/or flexible by means of appropriate designs.
The recyclability and life cycle assessment (LCA) of perovskite solar technologies are gaining momentum through sustainable technologies.14 LPSCs have emerged as leading candidates in the renewable energy landscape because of their low cost, simplicity in production, and high performance. Nonetheless, the scalability of modules, operational instability, and materials toxicity, particularly Pb, pose serious challenges that directly relate to the ecological impact of long-term applications – these challenges inhibit deployment and commercial feasibility.15 It has been widely reported previously that high-performing PSCs contain Pb, which is known to be toxic to plants and other living organisms.16 PSC materials can be easily degraded under certain conditions and can dissolve in water, releasing toxic Pb ions (Pb2+).17 For practical integration of PSCs, addressing Pb toxicity, stability, and high efficiency is a key priority for the safe deployment of this promising technology, as summarized in Fig. 1. Accordingly, the use of efficient and stable Pb encapsulants is envisaged to control Pb leakage in perovskites through their lifespans. In this regard, industrial pioneers and researchers have explored Pb recycling and immobilisation strategies, as replacing Pb with other halides may require significant research time and extensive effort.
 | ||
| Fig. 1 Key factors influencing the commercialisation of lead-based PSCs.18 | ||
The need to improve the understanding of end-of-life (EoL) management, particularly on discarded or damaged modules that can leach into water and soil systems, and potentially threaten human health and ecosystems, is necessary.19 The development of robust encapsulation and recycling procedures is crucial, given the adverse effects of Pb leakage from PSCs, even at low doses. It is important to understand perovskite degradation pathways since even low doses of Pb contamination exceed the regulatory threshold of safe exposure under acidic or humid conditions.20 Degradation of weathered or damaged perovskite modules is especially rapid in coastal or tropical conditions, or even in high-energy conditions.21 Critical analysis of reported incidents helps identify critical design flaws and inform the design and fabrication of safer device architectures, the use of Pb-absorbing interlayers, and multilayer encapsulation strategies. Concurrently, fundamental research is ongoing to understand the electronic, chemical, and physical properties of halide perovskites, and will continue to drive the future of device scalability and performance.18
Studies have shown that MAPbI3-based devices are susceptible to degradation under tropical-humidity conditions, prompting the need for robust Pb containment protocols.14 Besides thermal-based extraction methods, closed-loop recycling systems that can extract Pb through solvent-based methods in damaged Pb-based solar modules have been considered to enhance Pb recovery with minimal by-products. In the context of earlier investigations, recycling and immobilizing Pb within the perovskite structure reduces environmental risk and enables this new-generation technology to be scaled. The widespread adoption of PSCs focuses on addressing Pb leakage, its detrimental effects, and the strategies for Pb recovery and immobilization.22,23 Different from earlier studies that have examined the toxicity risks of Pb leaching or degradation of PSCs through a material science lens in isolation, this study situates Pb leakage monitoring and links it to JT to clean energy. This review discusses ICP-MS as an indispensable gold standard versus a comparative critique of field deployable techniques, such as in situ spectroscopic techniques, electrochemical sensors, and fluorescence tests from descriptive analysis and practical applicability. Also, the novelty of this work lies in the emphasis on how real-time monitoring can help monitor Pb2+ leaching into water systems, linking demonstrated AI applications to conceptual approaches to Pb leakage monitoring, and EoL management of PSCs, which are still far less explored in the literature. The objectives of this review articles, thus, are four-fold (1) critical analysis of Pb leakage monitoring strategies in PSCs, (2) risk assessment of the hazardous Pb in PSCs and its relationship with monitories strategies to inform the ecological and public health protection policies, (3) nuanced overview of predictive modelling and AI in device design, degradation and recycling, and (4) situating the current Pb2+ leakage monitoring strategies and JT in the paradigm of clean energy.
2. Methodology
This review paper systematically and thematically summarizes existing evidence regarding the environmental, just transitions and technological issues of Pb-based halide PSCs. The approach involved critical examination of the state-of-the-art in terms of Pb leakage detection, degradation pathways, recycling methods, and the emergence of artificial intelligence (AI) in predictive diagnostics. Accordingly, this study integrates scholarly publications and industry developments, with a particular focus on LCA and environmental sustainability. A set of scholarly data databases (Web of Science, Scopus, IEEE Xplore, ScienceDirect, and Google Scholar) was examined to ensure sufficient coverage. Keywords used in the search strategy include, but are not limited to, “halide perovskites”, “leakage of lead”, “bioaccumulation”, “encapsulation technologies”, “life cycle assessment”, “recovery of lead”, “artificial intelligence in photovoltaics”, “end-of-life management”, and just transitions. Peer-reviewed articles, patents, and industrial reports, which commented directly on the environmental attributes and technological developments of PSCs, were eligible. Articles published between 2010 and 2025 were included in the study. Consequently, this article assesses Pb leaching mechanisms and environmental effects, bioaccumulation of Pb in water bodies and soil, and its impact on human health. This is informed by studies in toxicology and environmental evaluation, with an intention to highlight the urgency of combating Pb contamination from decommissioned or damaged LPSCs. Secondly, current encapsulation engineering and containment technologies like polymeric barrier films and ion-exchange layers are assessed, focusing on minimising Pb migration under both working and degradation conditions. Also, this study investigates Pb recovery and recycling techniques, which are differentiated by thermal, solvent-based, and closed-loop systems. LCA models indirectly measure the impact on the environment of these practices, particularly recovery performance and secondary waste minimisation optimization. The possibility of predicting leakage or degradation patterns using AI-powered systems is discussed. This overview presents sensor integration strategies and machine learning models for real-time monitoring and predictive maintenance of perovskite solar modules. Technologies and strategies are synthetically compared, performance, cost, environmental safety, and commercial viability analysed in relation to data advanced in literature. Future outlooks are identified by mapping emerging trends in Pb-free alternatives, just transitions and AI applications, and critically evaluated the weaknesses of the current approaches to be used in developing sustainable perovskite technologies.3. Overview on the toxicity of lead
Unlike the immediate threat of degradation and leaching, a systemic risk of long-term environmental Pb build-up exists in perovskite modules. Studies have shown that even low concentrations of Pb2+ released into soil and groundwater can be bio-accumulated in plants and aquatic organisms, which subsequently enter the food chain.18,24 This raises concerns about chronic exposure in agricultural systems25 where solar farms may compromise crop safety and livestock health. The persistence of Pb in the environment makes its elimination prohibitively costly and technologically challenging, highlighting the significance of novel Pb immobilisation strategies.15 Even under minimal exposure, Pb is a critical concern for PSCs. The face density of Pb in a typical PSC with a 0.5 µm can be determined using eqn (1).26
 | (1) |
 | ||
| Fig. 2 Potential absorption and distribution Pb in human bodies as a result of degradation of Pb-containing perovskite that is dispersed in air and dissolved in water.30 | ||
The toxic nature of Pb in PSCs underscores the importance of strict occupational safety measures during module fabrication, installation, and recycling to guarantee the safety of both workers and the communities.22 The EoL management of perovskite modules is another pressing concern.31,32 Without proper recycling facilities, discarded modules can be left in landfills, where the leaching of Pb2+ ions into groundwater becomes inevitable.33 Case studies have demonstrated that populations around landfills can be exposed to Pb-containing leachates that exceed safe drinking water levels.34 To quantify the risks, research has shown that simulation methods such as hail impact tests have been used to determine Pb leakage rate as expressed in eqn (2).35
 | (2) |
4. Detection of lead leakage in PSCs
Addressing Pb pollution from damaged perovskites requires sensitive detection methods and effective Pb recovery strategies.38 By integrating kinetic and thermodynamic considerations, spectroelectrochemical methods such as cyclic voltammetry have been employed to understand the operational conditions of LPSCs.39 Cyclic voltammetry has proven effective for probing Pb ion behaviour in monitoring and detection. This technique provides information on Pb2+ adsorption, electrode kinetics, and redox processes. The reduction of Pb2+ to metallic Pb, followed by oxidation to Pb2+, yields characteristic peaks that can be used for Pb quantification. Cyclic voltammetry allows for real-time monitoring of Pb leakage due to its high sensitivity to trace Pb2+ quantities and the ability to distinguish complexed species from free ions.40,41 A recent study by Joji et al.42 proposed the use of biomimetic receptors immobilized on electrode surfaces to concentrate Pb2+ at the electrode interface. The study suggested an effective way of improving detection limits and Pb recovery efficiency by using biomimetic ligands to immobilize Pb2+. Cycling potentials allow Pb2+ to be removed from solution by reducing it at the electrode surface. Other studies have reported recovery efficiencies exceeding 95% in Pb recovery using cyclic voltammetry, ensuring that the hazardous Pb is not only detected but also reclaimed for reuse.41,43Remarkably, cyclic voltammetry has been used to detect Pb2+ at micromolar concentrations, making it suitable for detecting Pb2+ in water contaminated by decommissioned or damaged perovskite solar modules. It also ensured controlled Pb2+ release by electrochemically reducing and depositing Pb on electrode surfaces. The efficacy of cyclic voltammetry for Pb detection and recovery has been enhanced by combining it with chelation strategies that bind Pb2+ ions, such as gallic acid derivatives, phosphate-based polymers, ethylenediaminetetraacetic acid (EDTA)-like ligands, and crown esters. For instance, Li et al.44 introduced dispersible gallic acid chelators into PSCs and found that they reduced Pb immobilization and leakage with improved defect passivation and perovskite grain growth. Thin polymer coatings have also been used to form highly stable complexes with Pb2+, making them suitable for in situ Pb immobilization.15 Other studies have also shown success in “on-device Pb sequestration” by incorporating chelating agents such as ion-exchange layers and polymeric matrices.45 In addition, EDTA solutions have also been used to sequester Pb ions before electrochemical reduction. Biomimetic peptides that mimic natural proteins and crown esters that provide cavity-like binding sites when incorporated in perovskite solar modules with enhanced sensitivity and selectivity in Pb detection.42
Toward this end, the use of electrochemical sensors in aqueous leachates from PSCs, water systems of soil matrices are prone to surface fouling due to deposition of degradation products, salts, or organic matter.46 This deposition increases charge-transfer resistance and alters the capacitance of the double-layered electrode, consequently leading to reduced selectivity and loss of sensitivity. The inorganic precipitates and macromolecules distort the voltametric signals by blocking the active sites. Prior reports have shown that surface fouling can be mitigated by the use of antifouling polymers, hydrophilic coatings, and nanostructured carbon coatings.47 Further to this, in situ restoration has also been achieved by the use of catalytic surfaces or electrochemical pulsing. These antifouling strategies are known to extend sensor lifetimes. These sensors also suffer from instability of the reference electrode (typically Ag|AgCl) due to poisoning and electrode degradation.46 There have been attempts to resolve this instability issue, such as the use of solid-state composite reference electrodes and sulphide-resistant Ag|AgCl electrodes. Other reports have also shown that the use of fibrous solid-state Ag|AgCl minimizes instabilities and enhance mechanical robustness of the sensor.48 In addition, to quantify Pb2+ in complex systems, the electrochemical sensors should have stable calibration curves. However, due to repeated recycling, the changes at the electrode surfaces and environmental fluctuations lead to calibration drift. This implies that these sensors require drift correction methods and adaptive calibration protocols.49
Prior studies on Pb quantification under regulatory or environmental conditions have underscored the importance of physical and chemical encapsulants and the dependence of leaching on test conditions. These scientific reports provide a basis for estimating the total toxic Pb that could be leached from perovskite solar modules; specific mechanisms and contributing factors to ion transport remain insufficiently understood. Various methods have been used to quantify and recover Pb from PSCs. For instance, ICP-MS has been used to give precise Pb2+ concentration in aqueous leaching tests.50 Additionally, closed-loop recycling with cation-exchange resins is a method employed to capture Pb from waste organic solvents, appropriate for the circular economy of PSC materials. This technique offers high binding capacity and high selectivity to Pb2+. Aqueous tests simulate practical degradation factors under moisture conditions and quantify the extent of Pb release into water systems, thereby informing relevant regulatory standards. Solvent-based methods have also been used in conjunction with aqueous methods, whereby the PSC is dissolved in a controlled medium to enable precise measurement of Pb leaching. Additionally, kinetic modelling of Pb leaching quantifies release rates under different simulated stress conditions, allowing for extrapolation of data to device lifetimes. The overall Pb recovery from various recycling and/or recovery strategies is determined using eqn (3).51
 | (3) |
Detection limits (DL) of the different detection and monitoring approaches reveal their specific analytical abilities.52 DL demonstrates the lowest level of the analyte distinguishable from the background noise under optimal conditions. To ensure precision, statistical accuracy is achieved by determining DL values multiple times. It is computed using the raw intensity data from the standard and the blank, considering eqn (4).52
 | (4) |
| Method | Principle of detection | Strengths | Limitations | Detection limit | Response time | Ref. |
|---|---|---|---|---|---|---|
| Fluorescent sensors | Enhanced Pb2+ binding or fluorescent quenching | Portable, highly sensitive, and allows real-time monitoring | May suffer from interference from other ions in the matrix and requires calibration | ∼0.1–1 µg L−1 | Seconds–minutes | 54 |
| Electrochemical sensors | Pb2+ ions detected via changes in current/voltage at modified electrodes | It can be integrated in encapsulation layers, portable, and fast response | Unstable under long-term operation and lower sensitivity to ICP-MS | ∼0–10 µg L−1 | Seconds | 55 |
| ICP-MS | Appropriate for environmental monitoring studies. Ionization of sample and mass ratio detection | Ultra-sensitive up to ppt levels, widely validated, and has high security | High cost, not appropriate for in-field monitoring, and requires destructive sampling | ∼0.001–0.01 µg L−1 | Minutes–hours | 52 and 56 |
| Colorimetric assay | Visible color change upon Pb2+ binding with chromogenic reagents | Low cost, does not need advanced equipment, and easy visual detection | Has limited sensitivity, semi-quantitative, and accuracy is dependent on environmental conditions | ∼50–100 µg L−1 | Minutes | 8 and 57 |
| Encapsulation/barrier monitoring | Monitor Pb2+ leakage after leakage suppression via engineered encapsulation | Enhances device stability, allows for self-healing, and prevents leakage rather than detecting | Requires validation for long-term performance and allows for indirect detection | Not applicable | Long-term (months–years) | 58 |
| Chemical adsorption/scavenger layers | Functional layers such as sulphides and phosphates capture Pb2+ | Can be integrated in solar cell configurations and mitigates environmental contamination | Its effectiveness is dependent on material compatibility and is not a direct detection method | Not applicable | Continuous | 2 and 59 |
The presence of Pb in the matrix of PSCs and Pb by-products resulting from material degradation currently impedes the large-scale deployment of MAPbI3-based cells.30 Various techniques have been employed to quantify Pb leakage in PSCs. For instance, Pancini et al.50 designed a fluorescent organic sensor (FS) to detect Pb leakage as MAPbI3 perovskites degrade to PbI2. Pb-selective ligand binding was developed to enable measurement of the fluorescence of Pb2+ ions. Simulated rainwater was exposed to the perovskite module, and sensor performance was evaluated at varying Pb2+ concentrations. The researchers noted that even low levels of damage to MAPbI3 solar cells could release quantifiable amounts of Pb2+ into water systems. These findings suggest that damaged MAPbI3 solar cells may pose severe environmental risks. Furthermore, Pancini and his co-authors introduced the potential of fluorescent organic sensors as a field-deployable technique that may be integrated into solar farm monitoring packages.50
Fluorescence signals are sensitive to humidity, ambient light, and temperature and are optimum when the signal-to-noise ratio is high.60 Also, due to the presence of organic matter, interference from solar radiation, and scattering from dust, the signals can be distorted. Consequently, these sensors have been fitted with optical shielding and selective filters to minimize background interference.61 Quality assurance protocols and United States Geological Survey (USGS) guidelines have also compelled researchers to ensure that the application of such systems in monitoring water systems is reliable and reproducible. Moreover, these sensors are also susceptible to reduced emission intensity and photobleaching due to prolonged UV exposure. To enhance the photostability of these sensors, the application of inorganic or organic shells has been suggested to co-encapsulate the fluorophores.62 The environmental factors can also interfere with the quantitative accuracy of the fluorescence sensors. Nonetheless, the adaptive calibration schemes have been suggested to make these sensors appropriate for field applications in detecting heavy metals like Pb2+ in aqueous matrices.62
Lam et al.63 performed a multi-technique study of Pb leakage pathways in MAPbI3 perovskite films. The research team utilised time-resolved photoluminescence (TRPL) to measure non-radiative recombination centres and carrier dynamics, low-dose in situ electron microscopy to visualise structural transformations at the nanoscale, and solid-state magic-angle spinning nuclear magnetic resonance (MAS NMR) to track ion motion and identify chemical changes in the perovskite. The research group used material simulations and modelling to validate their experimental findings. Remarkably, previous reports have shown that the electric field increases the rates of defect formation and ion migration. Vacancy formation and iodide migration have been shown to trigger the decomposition of MAPbI3 into the toxic PbI2.63 The acceleration of Pb2+ migration, a serious ecological threat, has been reported in the presence of electric fields. The authors have also identified the importance of crystalline and morphology as crucial to the durability of the device. Their work also highlighted the complex interrelationship between material stability and electric fields in defect accumulations and ion migration.
Another promising approach to monitoring Pb leakage is the use of electrochemical sensors.64 They are selective electrode-based Pb2+ sensors that detect changes in ionic concentration in water. Recent findings have confirmed that electrochemical detection is sensitive and has high response times, making it a suitable technique for tracking the degradation of perovskite modules in real-time under outdoor conditions.65 Unlike fluorescence-based methods, electrochemical sensors can be integrated into low-cost monitoring systems, providing a scalable approach to large solar farms.66 In addition to laboratory detection, field-deployable kits are being developed to monitor the presence of Pb on the ground. Portable colorimetric assays entail chromogenic reagents that change colour when Pb2+ ions are present.67 These kits allow non-experts to quickly determine the contamination levels in soil and water surrounding perovskite installations. These are inexpensive and less sensitive than ICP-MS or TRPL, but they can be used in preliminary screening and community-wide surveillance, especially in areas with limited well-developed laboratory infrastructure.
ICP-MS is an indispensable technique when ultra-trace level and high-accuracy quantification is required. It can measure Pb2+ levels to parts-per-trillion (ppt) and parts-per-million (ppm) with exceptional precision.68 This method allows assessments of environmental risks and compliance with strict standards, including the US Environmental Protection Agency (EPA) and the EU Regulation on Registration, Evaluation, Authorization, and Restriction of Chemicals (REACH), where electrochemical or colorimetric techniques lack sensitivity.50 It is also robust for handling complex systems, particularly when Pb2+ leaches into biological, water, or soil samples with minimal interference. ICP-MS has been applied to quantify Pb2+ leakage with high precision over time, to examine degradation pathways of MAPbI3-based PSCs, and to correlate Pb mobilization with degradation factors, encapsulation strategies, and PSCs architectures.17 Also, it provides isotopic resolution, allowing differentiation between Pb2+ from damaged or degraded PSCs and background contamination for regulatory reporting and gaining mechanistic insights. It is well-established that complex matrices distort signals and induce interference in simple assays; thus, ICMP-MS with mass filtering and plasma ionization is required to meet strict standards and conduct environmental risk assessments.50,69 Simple techniques may not be suitable because internationally accepted accuracy is needed in litigation and certification. Thus, the advanced monitoring of complex environmental contaminants and toxic metals, including Pb, cannot be conducted without high-resolution analytical techniques, including ICP-MS.69 The detection limit of Pb in water can be modelled using eqn (5).70
 | (5) |
In Pb quantification, however, simple methods and low-cost detection, such as portable electrochemical probes, colorimetric analysis, and fluorescent sensors, are employed in routine screening and monitoring.71 Remarkably, these techniques can be adequate in quality control in device fabrication and preliminary screening. Moreover, these simple, portable, and inexpensive methods also work well in on-site testing and community-level monitoring. For instance, fluorescent sensors have been utilized to monitor Pb2+ leaching without requiring complex instrumentation.72 This democratization of Pb2+ detection is consistent with the JT to clean energy, ensuring that PSCs can be fabricated and the leaching of the hazardous Pb can be monitored even in resource-limited settings without prohibitive costs.73 Accordingly, simple methods used in Pb2+ detection serve as complementary techniques rather than exclusive, since they provide benchmark precision and broad monitoring capability. Simple techniques such as electrochemical sensors are field deployable and affordable, thus significant in the detection of emerging pollutants within water systems.73,74
Advanced spectroscopic methods have also been used to investigate Pb leakage pathways. X-ray photoelectron spectroscopy (XPS) and X-ray absorption near-edge structure (XANES) have been employed to determine the chemical state of Pb in degraded perovskite films.75 These methods provide insight into the oxidation and conversion of PbI2 to other Pb oxides or salts, which dictate mobility and toxicity. Such approaches are not field-deployable but are necessary to understand the fundamental chemistry of degradation and to develop more resilient perovskite formulations. Recent ICP-MS and accelerated ageing experiments have shown that multilayer polymer encapsulants and glass–glass encapsulation can significantly reduce Pb2+ release under simulated rainfall and humidity.76 However, as encapsulation layers are degraded by mechanical stress or weathering, the leakage increases exponentially. This highlights the importance of combining detection methods with durability tests to predict long-term risks. Combined monitoring systems that connect multiple detection methods to form a cumulative risk evaluation are also critical in analysing perovskite degradation. Fluorescence sensors, for example, may be combined with ICP-MS validation and electrochemical probes to provide high-precision laboratory validation and rapid field measurements.50 Such hybrid solutions can ensure that both short-term and long-term monitoring needs are met, allowing regulatory authorities and industry leaders to implement safety standards in the scale-up of PSCs. These frameworks represent a significant step toward achieving a balance between the extreme efficiency of perovskites and the need to ensure environmental safety.
The Pb2+ monitoring and detection applications are constrained by several factors. Since PSC modules are expected to reduce costs per watt due to scaling, Pb2+ leaching monitoring infrastructure continues to be a hidden cost. For instance, ICP-MS is an expensive instrument that demands skilled operators to calibrate the instrument and analyse the spectra.72 Colorimetric, electrochemical, and fluorescent methods have the advantage of being portable, but their accuracy under real-world conditions (UV-radiation, dust, and humidity) remains uncertain.77 In addition, scavenger/encapsulation layers to immobilize Pb need precise deposition, increasing the costs for scale fabrication of PSCs. Generally, the degradation pathways tend to be non-linear and often environmentally dependent, making the release of PbI2 and Pb2+ difficult to detect, thus compromising the efficacy of Pb detection and monitoring techniques.78 Finally, the implementation of these monitoring methods faces impediments related to strict policy frameworks, which may delay the commercialization and certification of laboratory breakthroughs.
5. Mechanisms of MAPbI3 decomposition and lead leakage
PSCs are semiconductor materials that have chemical components that exhibit weak bonding through ionic interactions, hydrogen bonding, and van der Waals forces. PSCs containing Pb can be hazardous to the environment and public health since degradation ions such as Pb2+ can readily migrate to various layers when exposed to degradation environments (Fig. 3). Unlike the widely deployed silicon solar cells, perovskites designed for outdoor applications are susceptible to damage due to natural factors such as wind stress, snow, hailstorms, or fires during operation.18 Improved understanding of possible health and environmental hazards and strategies for management will make LPSCs commercially viable and sustainable. The degradation mechanisms of perovskites are associated with intrinsic and extrinsic factors arising from the crystallographic or the molecular structure of the perovskite and inherent practical application conditions. | ||
| Fig. 3 Overview of Pb leakage in the environment from perovskite solar modules.79 | ||
5.1. Intrinsic degradation factors
Previous studies have discussed hysteresis, crystal defects, ion migration, optical absorption limitation, and thermal instability as common factors that impede the intrinsic stability of perovskites.80,81 The open circuit voltage (Voc) of perovskites is correlated with electron and hole separation quasi-Fermi levels within the perovskite layer during illumination.82 Therefore, the presence of defect-induced non-radiative recombination processes reduces the quasi-Fermi level splitting, thereby lowering the Voc.81 Besides, fill factor (FF) is also dependent on the Voc, and thus, these defects that arise from antistites substitutions, interstitials, and atomic vacancies also decrease the FF. The loss of volatile MAPbI3 results in the creation of iodide vacancies and, consequently, trap states in the device.81 During degradation, Pb0 also forms, increasing non-radiative recombination and inhibiting efficient charge transfer. In addition, the presence of defect density at the grain boundary also affects perovskite performance by lowering Voc and Jsc. The presence of Frenkel defects also contributes to hysteresis due to repetitive charging and discharging cycles.83 Under thermal drift or bias voltage, the cations and anions in the perovskite migrate within the perovskite, causing device instability. Ion migration has been reported to alter the crystal structure and relocate traps, leading to lattice collapse in perovskites and ultimately influencing device performance. The migration ions may also move to other functional layers of the perovskite, including the electron transport layer (ETL), hole transport layer (HTL), and electrodes, potentially corroding these layers, as shown in Fig. 4. The corrosion of these functional layers significantly reduces interlayer conductivity and forms a stable silver iodide (AgI) compound that is detrimental to the device's overall performance.81 | ||
| Fig. 4 Ion migration and re-orientation in the perovskite under forward and reverse scans.80 | ||
Previous studies have shown that hysteresis due to defect accumulation at the transport layer/perovskite interface reduces device stability. While the origin of these defects is still under serious investigation, it is crucial to estimate their concentration and location using impedance spectroscopy. Typically, impedance spectra show a high-frequency response associated with dipole depolarisation, ionic diffusion, chemical capacitance, or geometric capacitance within the active layer.80 It also gives the low-frequency responses associated with factors such as ionic diffusion, device degradation, trap states, electron accumulation at interfaces, and dielectric effects.80 Notably, impedance spectra reveal the origins of ion defects, but conflicting interpretations hinder improved understanding of device hysteresis.
5.2. Extrinsic degradation factors
During solar cell operation, halide PSCs interact with external factors, including electric bias, oxygen, moisture, light, heat, or external agents. Light induces the redistribution of metal and halide ions in the MAPbI3 film. This results in increased photoluminescence due to iodide migration. Light-induced ion migration causes film segregation, resulting in halide-deficient and halide-rich regions.84 The interaction of oxygen with the perovskite layer results in the formation of highly reactive superoxide species, generated by the photolysis of water. This light-induced degradation induces vacancy migration in the perovskite film, leading to a decline in device performance. In addition, perovskites are known to be stable within particular temperature ranges. Under heat, MAPbI3 may decompose into methyl iodide, ammonia, and lead iodide (PbI2) at temperatures exceeding 85 °C in an inert atmosphere.84 Perovskites predominantly use organic ETLs and HTLs, which can degrade at elevated temperatures. When HTLs are exposed to oxygen, the hole-transport barrier increases, reducing hole extraction at the HTL/perovskite interface. Moreover, the perovskite reacts with oxygen in the presence of water, which is an irreversible chemical process. Light-induced degradation has been mitigated through advanced device encapsulation strategies. A good encapsulant ensures Pb immobilization by capturing ions if the device is broken or damaged.85 Table 2 summarises studies that have been conducted to enhance device stability and ensure Pb immobilization, with promising results. Formamidinium iodide (FAPbI3) and MAPbI3 are highly hygroscopic and readily decompose into their respective precursor ions.86 Therefore, when exposed to humidity, water molecules form weak hydrogen bonds with organic molecules within the device structure. Without physical encapsulants, perovskite films cannot withstand continuous exposure to humidity and fail the damp-heat test, which involves 85% relative humidity.85 Accordingly, both extrinsic (environmental) and intrinsic (chemical) factors need to be considered before the deployment and commercialization of PSCs.81| Encapsulation material | Initial PCE | Durability of the standard test | Conditions | Retention of peak PCE | Standardized tests performed | Ref. |
|---|---|---|---|---|---|---|
| Thiol-functionalized perfluoroalkyl | 21.79% | 500 h | 85 °C, MPP at N2-filled chamber | 90.1% | No | 87 |
| 500 h | Room temperature 85% RH | 95% | ||||
| Polyacrylic acid (PAA) ionogel | 22.87% | 1000 h | 85 °C, 85% relative humidity | 95.2% | Damp heat, thermal cycling | 88 |
| 200 cycles | −40 to 80 °C cycling | 96.1% | ||||
| DMDP (front), EDTMP-PEO (back) | 20.27% | 500 h | MPP at room temperature | 80% | No | 45,89 |
| Multilayer Al2O3 and pV3D3 | 20.1% | 300 h | 50 °C, 50% relative humidity, dark | 98.9% | No | 90 |
| Alucone and Al2O3 bilayer stack | 17.01% | 2100 h | 30 °C, 80% relative humidity, dark | 96% | No | 91 |
| Al2O3 and 1H, 1H, 2H, 2H-perfluoro decyl-trichlorosilane stack | 19.7% | 500 h | 85 °C, 85% relative humidity | 78% | Damp heat not long enough | |
| PIB wide blanket | 19% | 1800 h | 85 °C, 85% relative humidity | 95% | Damp heat and humidity freeze | 92 |
| 30 cycles | −40 to 80 °C cycling | 95% | ||||
| Resin UVR-C (front), C100 (back) | 25.5% | 500 h | Ambient conditions | 97% | No | 93 |
| Sulfonic acid-based resin | 20.1% | 500 h | 45 °C, MPP, ambient RH | 83% | No | 94 |
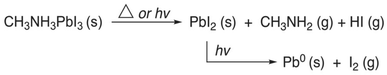
Pb-related environmental and public health impacts are measured as a function of exposure risk. Exposure probability depends on mobility, concentration, and the presence of toxic constituents in the environment. MAPbI3 is known to undergo various degradation pathways and seep into soil and water systems, circulating in the atmosphere and natural ecosystems. Under humid environments, MAPbI3 (CH3NH3PbI3) decomposes into CH3NH3I and PbI2 according to Rxn (a). Subsequently, CH3NH3I undergoes deprotonation by water molecules, producing CH3NH2 and HI. Further, HI decomposes into water and I2 in the presence of oxygen. Under elevated temperature, MAPbI3 degrades to produce PbI2 and CH3NH3I, which then decomposes into HI gas and CH3NH2, as shown in Rxn (b). Similarly, FAPbI3-based perovskite degrades when exposed to temperatures above 230 °C, as depicted in Rxn (c). Boyen et al.95 conducted a simulated fire experiment showing that burning PSCs release PbI2 into the smoke. These findings suggested that Pb compounds disperse in the air, polluting the environment.18 Therefore, the degradation factors of perovskite materials induced by heat and, to a greater extent, ultraviolet radiation cannot be underestimated. It is well-established that ultraviolet radiation degrades organic cations in the perovskite material, leading to chemical decomposition and phase segregation, as shown in Rxn. (b).
 | (a) |
 | (b) |
 | (c) |
Under degradation considerations, MAPbI3 decomposes to form PbI2, a significant environmental concern. PbI2 is insoluble in water, with a solubility constant (Ksp) of 8.0 × 10−9 to 1.84 × 10−8.96 This compound cannot be dissolved in water; however, PbO powder can be absorbed through human skin, posing serious health risks. Severe Pb exposures can be classified as accidental, disposal-related, or due to harsh weather conditions. The processing of perovskites may involve toxic solvents that can Pb to accidents by inhalation, ingestion, and even contact with the skin.97 Further, there are risks associated with module-induced breakage of Pb due to the density of the material and combustion during waste disposal of Pb-based solar modules.34 Other potential sources of Pb exposure related to perovskite solar technologies include severe weather events (such as hurricanes, tornadoes, and hailstones) and transport-related risks (such as vehicle collisions). Risk exposure pathways, including landfill leaching and groundwater and soil contamination, should be well understood to mitigate Pb leakage. Although the efficiencies are impressive, MAPbI3 formulations have been found to degrade in the presence of water, heat, and light, releasing Pb-based by-products such as lead carbonate, lead phosphate, lead iodide, and lead hydroxide.18 The routes of exposure and the risks that MAPbI3 may pose are multi-pathways, varying in risk levels based on pre-existing contamination levels, concentration, and source,98 as summarised in Table 3.
| Value chain stage | Causation | Health and safety risk type |
|---|---|---|
| PSC design | Identification of Pb vs. non-Pb containing perovskites | N/A |
| Device fabrication | Manufacturing process | Contaminated effluence |
| Solvent selection | Skin contact | |
| Machining process | Inhalation | |
| Transportation and staging | Improper transportation or packaging | Skin contact |
| Accident during transportation | ||
| Installation | Breakage/damage during installation | Skin contact |
| Operation | Aging | Water and soil system contamination |
| Weather | Skin contact | |
| Fire | Inhalation | |
| End-of-life management | Landfill or recycling process | Water and soil system contamination |
| Disassembly, staging, and transportation | Skin contact | |
| Inhalation |
6. Recent advances in the prevention of lead leakage in LPSCs
Physical encapsulation (Fig. 5) is the most widely used method to ensure that PSCs remain Pb-leakage-free.18,99 Encapsulation refers to the use of protective layers, such as glass–glass lamination, polymer films, or multilayer composites, that block water, oxygen, and mechanical forces.18 Glass–glass encapsulation, in particular, has been shown to reduce Pb2+ leakage by more than 90% under simulated rainfall conditions, and this effectiveness is also observed in the real world.100 Recent advancements use internal encapsulation, which involves using materials that interact with Pb to prevent its migration or capture it. In contrast, external encapsulants serve as physical barriers to prevent degradation due to harsh conditions and minimise Pb leakage. Fig. 5 depicts the external and internal encapsulation of PSCs. Nevertheless, encapsulation is not without challenges: microcracks, delamination, and thermal expansion mismatches can degrade barrier integrity over time, especially under harsh weather conditions. Thus, while encapsulation provides a physical barrier, its ultimate success depends on structural design and material reliability.76 Chemical absorption strategies complement physical barriers, actively immobilising Pb2+ upon release from degraded perovskite films.15 Pb2+ also interacts with absorbent compounds (phosphates, sulfates, and carbonates) to form insoluble salts such as Pb3(PO4)2 or PbSO4, which are more stable and less mobile in the environment. An example is the use of phosphate-based additives incorporated into encapsulation layers, which are reported to reduce Pb leakage by up to 96% compared to unencapsulated devices.58 Functionalized polymers and nanomaterials that incorporate chelating functional groups (e.g., thiols, amines) have also been designed to selectively trap Pb2+, thereby capturing them within the device infrastructure. The techniques not only reduce environmental risks but also enhance device stability by reducing ion migration routes.101 | ||
| Fig. 5 Physical encapsulation of perovskites to capture and prevent lead migration: (a) external encapsulation and (b) internal encapsulation.18 | ||
Hybrid designs that utilise the integrated strength of physical encapsulation and chemical absorption are gaining popularity as efficient methods for preventing leakage.18 By incorporating Pb-absorbing agents to encapsulation layers, researchers have developed dual-protection systems that should not only prevent external stressors but also repair any Pb ions that escape.15 For instance, the encapsulation of phosphate-based scavengers into polymer encapsulants has been more effective in hail, fire, and heavy rain environments and has released significantly less Pb than when encapsulated.58 Moreover, self-healing encapsulation materials provide an additional layer of resilience. These polymers can self-heal microcracks caused by mechanical stress, thereby preserving both the physical barrier and chemical scavenging properties over extended working lifetimes.102 These hybrid systems represent a promising avenue for realising commercial-scale deployment of perovskites. Even though encapsulation and absorption strategies are effective when functioning, Pb leakage cannot be avoided unless a lifecycle view is applied that covers fabrication, usage, and EoL handling.103 Recycling protocols that integrate encapsulation removal and Pb recovery are crucial in preventing landfill contamination. Closed-loop recycling, which involves recycling recovered Pb and incorporating it into new perovskite devices, not only reduces environmental risks but also makes PSC technology consistent with the principles of a circular economy.76 Life cycle analysis studies have shown that recycling and recovery can reduce the toxicity footprint of perovskites compared to silicon photovoltaics.104 Thus, leakage prevention must be incorporated throughout the entire value chain, including material design and disposal, to ensure that PSCs are not only high-performing but also environmentally sustainable.
6.1. Sustainable decommissioning of perovskite modules
Although perovskite commercialisation is still in its infancy, the issue of recycling has been widely debated in the context of sustainable technologies.105 Successful EoL management of LPSCs relies on an enhanced understanding of the device life cycle, which is crucial (Fig. 6).106 The first step in decommissioning LPSCs is to identify its critical components. This intricate journey begins with sourcing raw materials, including the absorber layer, and sustainable fabrication for creating small-area perovskites to minimise efficiency losses.106 The material components can be categorised by their cost and sustainability, and the urgency for recycling can be determined accordingly. | ||
| Fig. 6 Lifecycle of perovskite solar technology.106 | ||
Directive 2008/98/EC (Fig. 7(a)) outlines the waste hierarchy pyramid, which can be used to inform the sustainable decommissioning of PSC modules.105 At the top is prevention, which involves embedding eco-design principles into solar cell design, such as the use of physical encapsulants and the incorporation of durable and stable materials. In addition, perovskite modules contain valuable materials, including transparent conductive oxides, halides, Pb, and tin, which can be reused or recycled via in situ regeneration, solvent extraction, and chemical precipitation. Notably, recycling these materials supports the circular economy and simultaneously prevents the leaching of toxic contaminants from hazardous waste into the environment. In the lower tiers of the hierarchy, (energy recovery and disposal), requires that wastes can be incinerated or disposed of on land fields, but should remain as the last pathway for waste management.105 Encapsulate, capture, and recycle (ECR) are emerging material chemistry approaches that have been investigated to potentially mitigate the health and environmental hazards associated with LPSC devices,51 as illustrated in Fig. 7(b).
 | ||
| Fig. 7 Waste hierarchy pyramid as outlined in the directive 2008/98/EC of the European Parliament and of the Council of 19 November 2008 on wastes and repealing certain directives105 (a) and (b) emerging approaches for waste management of perovskite solar modules.103 | ||
In situ recycling of the perovskite layer is an emerging technique that mitigates Pb leakage, a common issue during perovskite recycling. Unlike the energy-intensive recycling strategies such as chemical precipitation, thermal treatment, and solvent extraction, in situ recycling minimizes these processing steps and directly regenerates PbI2. Xu et al.107 demonstrated that PbI2 can be recovered and reintroduced into devices, resulting in robust performance comparable to that of devices fabricated with fresh precursors. Remarkably, differences in environmental benefits, including in situ recycling, support the circular economy in perovskite device fabrication, thereby reducing overall material costs. This paradigm shift aligns with the European Union directive 2008/98/EC on waste management, which prioritises recycling, reuse, and prevention.108
6.2. Recovery of material components from LPSCs
Few studies have been conducted beyond the recovery and reuse of perovskite material components, specifically focusing on Pb2+.109 The sustainability and feasibility of recycling perovskite modules are crucial in the design of future perovskite devices. Common LPSC materials, along with their advantages, disadvantages, and recycling implications, are summarised in Table 4. Transparent conductive oxide (TCO) substrates and gold contacts are the most environmentally impactful and expensive materials used in halide perovskites. Research has shown that gold can contribute up to 65% of the carbon footprint and constitute up to 90% of the embedded material cost, due to the energy requirements during its deposition and the material value.110 The process of recovering perovskites enables the reuse of the recovered foil, but this remains a scientific challenge. Before reuse, these foils should be melted into suitable gold wires/pellets, which is an energy-intensive process, and may not be economically viable for large-scale commercialisation.110 However, due to its material benefits, such as high conductivity and stability, gold remains preferred in device configurations that support circular economy.| Material layer | Material options | Advantages | Limitations | Recycling implications |
|---|---|---|---|---|
| Substrate | Glass | Excellent thermal stability | Rigid | Highly compatible with mechanical recycling |
| Strong barrier against moisture and chemicals | Relatively expensive | Can be integrated into existing silicon PV recycling streams | ||
| Polymers (PET, PEN) | Flexible and lightweight | Poor heat resistance | Recycling is challenging under harsh thermal/chemical treatments | |
| Lower cost | Brittle under stress | |||
| Transparent conducting electrode | FTO (fluorine-doped tin oxide) | Good thermal stability | Higher surface roughness | Durable and less prone to degradation during recycling |
| Cost-effective | ||||
| ITO (indium-doped tin oxide) | Smooth surface | Lower thermal stability | Recycling is critical to recover indium, a scarce raw material | |
| High optical transparency | Expensive | |||
| ETL (electron transport layer) | Inorganic (TiO2, SnO2, ZnO) | Stable | Strong adhesion to substrates complicates separation | Difficult to recycle due to tight bonding with substrates |
| Low-cost | ||||
| Easy fabrication | ||||
| Organic (PCBM) | Low processing temperature | Poor stability | Degrades under heat/solvent exposure | |
| Low electron mobility | Removal requires toxic solvents (e.g., chlorobenzene) | |||
| HTL (hole transport layer) | Inorganic (NiOx, CuSCN) | Chemically and thermally stable | Possible interfacial reactions | Strong adhesion reduces recyclability |
| Cost-effective | ||||
| Organic (PEDOT: PSS, spiro-OMeTAD, PTAA) | Solution-processable | High cost | Dopants complicate chemical separation during recycling | |
| Enhanced charge transport | Complex synthesis, poor long-term stability | |||
| Perovskite layer | Pb-based | High efficiency | Toxicity concerns | Pb recovery is essential to prevent environmental contamination |
| Tin-based | Non-toxic | Easily oxidized | Oxidation complicates recovery and reuse | |
| Air instability | ||||
| Bismuth-based | Stable | Low efficiency | Recycling pathways are not yet established | |
| Low toxicity | Early-stage technology | |||
| Back electrode | Gold (Au) | Excellent conductivity | Very expensive. Requires vacuum deposition | Can be selectively etched and recovered |
| High work function | ||||
| Silver (Ag) | Lower cost than gold | Diffusion into perovskite | Recoverable, but diffusion complicates separation | |
| Requires vacuum deposition | ||||
| Copper (Cu) | Low cost | Prone to oxidation | Residual halides contaminate copper during recycling | |
| Good conductivity | ||||
| Carbon (C) | Cheap | Low work function | Difficult to isolate during recycling | |
| Chemically inert |
Before recovering the device components, the perovskite to be recycled is first peeled off to expose the film. The perovskite is then dissolved in organic solvents such as DMF, dimethyl sulfoxide (DMSO), and nontoxic butylamine (BA).111 Previous reports on the use of polar solvents yielded a solution containing Pb2+, followed by separation and extraction of PbI2 using precipitants or adsorbents for reuse in new devices.112 DMF has been successfully used as a solvent to dissolve EoL perovskites to obtain Pb2+, followed by the addition of Fe-decorated hydroxyapatite hollow composite with a negative surface charge to adsorb Pb2+. Subsequently, water can be added to the resulting solution to dissolve it, and KI is then added to obtain pure PbI2. This method has been successfully used to obtain a recycling yield of 99.97%.112 Similarly, Pb2+ can be adsorbed by the cation exchange resin (CER) method with a remarkable recycling rate of 99.99%.51 PbI2 is then separated and extracted by reacting with the solution containing NaI and HNO3, as shown in Fig. 8(a). Other studies have also suggested using the eutectic solvent ethylene glycol and choline chloride to dissolve the perovskite and separate Pb2+ from the solvent by electrodeposition, achieving a Pb recycling rate of 99.8%.102 In addition, NH3·H2O can also be used to achieve similar extraction results.113 Fundamental studies have also explored in situ recycling of MAPbI3 by dissolving the HTL in chlorobenzene and using a tape to strip the Ag electrode.107 Subsequently, PbI2 is then obtained by the thermal degradation of MAPbI3. Afterwards, a re-spin coating of MAI is performed on the PbI2 layer to form a new perovskite film (Fig. 8(b)). The performance of the re-prepared devices (14.84%) was higher than that of the control devices (14.35%).107 Performances of re-prepared devices can be better enhanced by the use of hydroxypropyl-β-cyclodextrin crosslinked with 1,2,3,4-Butanetetracarboxylic acid (HPβCD-BTCA), which can chelate Pb2+ as a Pb2+ adsorbent and as a passivator of the perovskite.114 The resulting composite of HPβCD-BTCA@PbI2 can be used, and the PCE of the as-prepared devices reached 20% higher than that of the control devices (19.63%).114 Besides, the PbI2 recovered from HPβCD-BTCA@PbI2 composite material showed a purity of 98.9%.114
 | ||
| Fig. 8 Lead recovery from perovskites: (a) use of cation-exchange resin (CER) to recycle solar modules51 and (b) illustration of in situ recycling of PbI2 from PSC and re-preparation of new solar cells.107 | ||
Ion migration may also lead to the degradation of PSC materials, resulting in irreversible changes to the material properties of HTL.110 Furthermore, the use of encapsulants such as ethylene-vinyl acetate (EVA) complicates the recycling process due to their chemical stability and strong adhesion. Innovative approaches introduced to overcome this challenge include the use of New Industrial Encapsulation (NICE).79 Moreover, the use of elastomers and thermoplastics has been proposed as an alternative to offer superior delamination properties compared to EVA.116–118 The EoL recycling of perovskite modules may also be hindered by the use of highly toxic antisolvents, such as chlorobenzene and toluene, as well as by solvents such as dimethylformamide. The perovskite precursors are also required for the selective recovery of materials from weathered or damaged perovskites, posing significant health and environmental risks. This underscores the need for sustainable and eco-friendly fabrication techniques, such as the use of green solvents (e.g., water, ethanol, and methanol) and green antisolvents (e.g., 2-methyltetrahydrofuran and anisole). Apart from the use of green solvents and antisolvents, it is also necessary to design HTLs that are highly soluble in green solvents.118
Recycling can be challenging due to conflicts or overlaps of the material's thermal and chemical properties. Previous studies proposed integrating AI algorithms to develop a more comprehensive understanding of the optimal recycling strategy, which could help recover the most valuable, costly, or hazardous materials.119 During recycling, it is also scientifically feasible to recover highly pure materials that deliver efficiencies comparable to those of pristine photovoltaics. Recycling involves mechanical, thermal, or chemical treatment of materials, which can increase the propensity for material contamination or damage, potentially introducing unintentional dopants that affect material performance.14 Therefore, recycling strategies may not be relied upon due to reproducibility issues and inconsistent performance. Furthermore, thermal and solvent-based recycling methods pose environmental challenges. Pb-containing compounds may volatilize into the environment, posing significant risks of Pb contamination.109 Aprotic solvents such as DMF may lead to the generation of toxic solvent waste, increasing health and environmental risks.79 PSCs are fabricated from nanoscale materials, and the recovered materials from recycling are often minimal; the cost of layer-by-layer purification may outweigh the device's market value.120 Recycling is also a high-energy-intensive process, particularly during the thermal recovery processes.121 According to previous studies, the LCA of recovering valuable materials from waste perovskites, such as aluminium, glass, Pb, nickel, gold, and silver, indicates that the total cost calculated for recovery ranges from the most effective method to the least ($10.70–$32.45).122 A summary of challenges in Pb recycling is presented in Fig. 9.
 | ||
| Fig. 9 Challenges in recycling of perovskite solar modules.79 | ||
6.3. Closed-loop recycling of lead
Although Pb remains a hazardous element, innovative approaches are being developed to mitigate the toxicity of LPSCs. Researchers are also addressing recycling and recovery methodologies to mitigate the environmental risks associated with Pb leaks to enable the re-use of useful materials. For instance, in situ regeneration in real-time has been proposed to restore Pb directly within the device, rather than precipitating or dissolving the device. Real-time monitoring implies tracking the changes in MAPbI3-PSCs “as they happen,” generally with second-to-minute resolution. Unlike in ex situ monitoring, the device is being monitored in real operating conditions (presence of environmental stress, bias, and illumination) as the data is being collected without disrupting module operation.123 Real-time monitoring of MAPbI3 modules has been attained through the use of electrochemical sensors, optical spectroscopy, and impedance analysis without interruptions. For instance, Suchan et al.124 employed optical in situ monitoring to study how perovskite film formation operates under stress conditions, and reported how optoelectronic characteristics of the perovskite change with time. Similarly, Yuan et al.125 and colleagues applied short-wavelength infra-red holography to understand how stress-induced defects affect perovskite performance in real-time. Moreover, scientific evidence has demonstrated how real-time monitoring of PV modules can be used to monitor moisture ingression, phase segregation, and ion migration continuously during device operation.126,127 These studies demonstrate success in situ monitoring in real-time when tailoring electronic properties during film growth, tracking how stress defects form, and the time dependence of PV degradation. Real-time monitoring in situ may also be extended to various growth recipes and synthesis environments.Another frequently studied approach to Pb recovery is chemical precipitation, where degraded perovskite modules are dissolved in aqueous solutions and reacted with precipitating agents, such as sulfates, phosphates, or carbonates.15,23 These reagents reduce the soluble Pb2+ ions to the insoluble PbSO4 or Pb3(PO4)2 forms, which can be filtered and recovered.23 This method not only prevents Pb from leaching into the environment but also allows the recovered compounds to be reused in industrial processes. Another promising avenue is the use of solvent extraction and ion-exchange approaches.128 In this approach, organic solvents or functionalized resins selectively trap Pb2+ from perovskite waste streams. The resulting bound Pb is strippable and can be recaptured in concentrated form. This method has shown significant recovery efficiencies and minimised the generation of secondary waste.128,129 Moreover, ion-exchange resins are reusable and reproducible, making them economically more viable when recycled at scale.22,110
Electrochemical recovery is also gaining interest in closing the loop in Pb recovery. With a favourable electrochemical potential, Pb2+ ions in solution can be reduced and plated out as metallic Pb on the electrode surfaces.115 This technique not only recovers Pb in a useful metallic form but can also be incorporated into closed-loop recycling networks. Electrochemical recovery is particularly attractive because it can be powered by renewable energy sources, thereby reducing the environmental footprint of perovskite recycling even further.130 Further to direct recovery, scientists are exploring encapsulation-based recycling systems. Such systems include protective encapsulation layers and built-in sorbents or scavengers that capture Pb2+ ions released during degradation. At the end of a module's lifetime, the immobilized Pb encapsulation material can be reclaimed and recycled.18 This proactive approach reduces the risk of environmental pollution during the solar cell's operational phase.23 A schematic illustration of strategies for Pb recycling from PSCs is presented in Fig. 10.
 | ||
| Fig. 10 Lead recovery strategies from lead-based perovskites: (a) in situ regeneration, (b) solvent extraction, (c) electrochemical deposition, and (d) adsorption–desorption.131 | ||
Perovskites degrade into volatile organic compounds (VOCs) and non-volatile lead iodide (PbI2). The VOCs escape into the atmosphere, while PbI2 can be recycled by supplementing organic perovskite to restore the perovskite film (Fig. 10(a)). However, this method has limited applicability because it depends on the morphology and crystallinity of the residual PbI2. Thus, the success of this method depends on the initial chemical composition and preparation methods of the perovskites.131 In solvent extraction, the degraded perovskite is immersed in polar protic solvents, such as 2-propanol, deionised water, ethanol, or methanol, to extract the highly soluble MAI and the low-solubility PbI2. Subsequently, aprotic solvents such as DMF or green solvents, including dialkylamines, butylamine, or methylamine, are added to dissolve the Pb compounds. The use of green solvents is preferred due to DMF's toxicity. While some studies have reported the success of solvent extraction for Pb recovery, this method requires large volumes of solvent and has limited selectivity. Electrochemical deposition has been adopted for all Pb-based devices because it allows recovery of up to 99.8% of Pb when properly dissolved.110,115 Table 5 presents studies that have proposed recovery methods to mitigate the risk of Pb pollution and promote the recycling of key material components for future perovskites.
| Method | Lead source | Pb retrieval (%) | Pb recovery (%) | Device efficiency (fresh PbI2) | Device efficiency (recycled PbI2) | Ref. |
|---|---|---|---|---|---|---|
| Electrochemical | Devices in molten LiCl–KCl perovskite films dissolved in a deep eutectic solvent | >99.8 | 98 | — | — | 43 |
| 98.7–99.8 | 99.8 | — | — | 115 | ||
| Ion exchange | Planar TiO2 devices in DMF | 99.99 | 99.97 | 16.7 | 16.5 | 112 |
| Pb (NO3) 2 aqueous solution | — | — | 19.3 | 19.0 | 132 | |
| PbI2 (aq) from degraded devices | 88 | 96.5 | 21.0 | 21.0 | 133 | |
| PbI2 from mini modules in DMF | 99.6 | 99.2 | 21.0 | 20.5 | 51 | |
| Solvent extraction/recrystallisation | PbI2 from carbon-based devices in DM | 99.9 | 95.7 | 12.2 | 11.4 | 113 |
| Inverted devices in butylamine | — | 98.9 | 17.0 | 17.0 | 134 | |
| TiO2 devices in DMF | — | — | — | 13.5 | 135 | |
| Synthetic solution (TiO2, spiro-OMeTAD, PbI2, MAI in water) | 91.3 | — | — | — | 136 | |
| Full device in bleaching solution | 99.9 | 100.0 | 21.0 | — | 137 |
Previous studies have developed a closed-loop cycle to recycle lead iodide (PbI2) harvested from waste organic solvents used in the manufacture of PSCs.138 This process isolates and purifies the precursor, which is then reused, rather than discarding solvent waste streams that are contaminated with dissolved PbI2. This approach not only reduces the amount of toxic Pb entering the environment but also minimizes the need for virgin Pb resources, providing a clear solution to one of the most pressing sustainability challenges associated with scaling up perovskite technology.138 Among their findings is the conclusion that the reused PbI2 can be used in fresh perovskite films without significant deterioration in device performance. This repurposed material produced perovskite layers with crystallinity, morphology, and optoelectronic properties comparable to those produced with commercial PbI2. A closed-loop system also has economic and environmental advantages. The reuse of PbI2 allows manufacturers to not only lower the price of raw materials but also the cost of hazardous waste management.136,139 The technique also minimises the environmental impact of perovskite production, thereby addressing one of the primary concerns associated with the use of toxic Pb. Accordingly, recycling reduces the risk of Pb leaching into soils and water systems, which is among the greatest challenges to the spread of perovskites in the renewable energy market.18
7. Applications of AI-powered algorithms in lead leakage monitoring
Machine learning (ML) algorithms (a branch of AI) have transformed the material science landscape, offering the capability to identify degradation pathways, predict material properties, or even propose optimal processing and synthesis conditions.140 Different from giving data on PV characteristics of PSCs, ML models have been trained on labelled data to elucidate degradation behaviour. Moreover, explainable AI (XAI) is essential in validating recycling processes by determining which variables have the most significant impact on recovery success.141 This transparency aids regulatory compliance and industry scalability of LPSCs. AI-assisted approaches facilitate the design of a circular manufacturing model in which recovered Pb is recycled and used to fabricate new PSCs, thereby minimizing environmental footprint and material waste.142 PSCs are sensitive to environmental factors, including humidity, UV radiation, and thermal cycling, and such measures are indicators of one of the major obstacles to long-term stability of PSCs.81,143 AI methods can mitigate these problems through prediction and diagnosis.144,145 The neural networks are especially helpful in modelling the complex kinetics of degradation, which allows a scientist not only to estimate how long these devices will last, but also to predict the type of failures that might also occur.146 This is supplemented by support vector machines, which classify degradation patterns using sensor data, differentiating between reversible and irreversible damage.147 Visual inspection with convolutional neural networks (CNNs) could identify morphological defects in perovskite films, including pin holes, cracking, and phase segregation.148 These insights play a key role in quality control and early intervention. This is achievable by learning reinforcement-based dynamic encapsulation systems that adaptively modify barrier properties in dynamically changing environments to enhance device longevity.148AI strategies could reduce development time and costs, paving the way for commercially viable, environmentally responsible PV technologies. PSC technology requires EoL control to mitigate environmental contamination due to Pb and allow sustainable use. AI practice in the field of LPSCs supports predictive maintenance, automated disassembly, and smart recycling decisions, toward circular economy.149 Support vector machine and machine learning schemes are capable of predicting the performance collapse of a machine based on the data of its operation, enabling timely intervention and Pb recovery.150 Explainable AI can be applied to LCA, quantifying the environmental impact of various EoL alternatives to support policy-making and stakeholder engagement.151 In addition, convolutional neural networks can simplify this step by detecting damaged or contaminated modules via visual inspection, thereby improving the efficiency of the recycling pipeline.152 Through such AI-driven applications, ML algorithms will contribute constructively to the development of sustainable and scalable LPSCs.151 A summary of various AI techniques that may be applicable to PSC research is presented in Table 6.
| AI technique | Application area | Advantages | Limitations | Ref. |
|---|---|---|---|---|
| Machine learning | Material screening and composition prediction | Rapid identification of stable, high-efficiency materials reduces experimental workload | Requires large, high-quality datasets; may over fit to known materials | 153 |
| Neural networks | Modelling degradation kinetics | Model nonlinear relationships between fabrication parameters and PCE; adaptable to new data | Often lacks interpretability; sensitive to noise in training data | 154 |
| Explainable AI | Manufacturing optimization, material recovery, and decision support | Provides insights into model decisions; improves trust and transparency | Still emerging in materials science; limited toolkits | 155 |
| Support vector machines | Classification of degradation patterns and failure modes | Effective for small datasets; robust to overfitting | Less effective for large, noisy datasets; limited scalability | 156 |
| Convolutional neural networks | Image-based defect detection in perovskite films | High accuracy in visual inspection enables real-time quality control | Requires labeled image datasets; computationally demanding | 148 |
Researchers at the University of Washington developed physicochemical ML models to predict the lifetime of MAPbI3-based solar cells.146 The research team further utilised various regression algorithms to predict device stability, employing Lasso regressions. Notably, the findings of this research group emphasised the importance of AI in quantifying the parameters that improve operational stability.146 AI has also been applied to accelerated ageing experiments, where perovskite modules were exposed to simulated stress conditions, such as high humidity, UV radiation, and high temperatures.157 Trained ML models based on these datasets can extrapolate long-term degradation trends to short-term experiments, reducing the time and cost of stability tests by many orders of magnitude. This technique can be particularly beneficial for estimating the extent of Pb leakage under extreme weather conditions that are difficult to replicate in a laboratory yet vital to practical applications.58 More so, AI can help with automated defect detection in perovskite films.158 Computer vision algorithms trained on microscopy images can detect microcracks, pinholes, and phase-segregation patterns that often precede Pb leakage. When installed on manufacturing lines, these systems can provide real-time quality control, enabling defective modules to be identified and corrected before being fabricated. This not only improves the reliability of the devices but also reduces the risk of faulty products polluting the environment.33
Another prospective application of AI in this field is integrating predictive modelling with real-time sensor data. By combining information from fluorescence sensors, electrochemical measurements, and ICP-MS, it is possible to identify early indicators of Pb leakage before it becomes harmful. Such predictive frameworks enable operators to implement proactive measures, such as module replacement or encapsulation strengthening, which reduce environmental contamination risks.50,159 Joji et al.42 utilised a biomimetic sensing moiety to detect Pb ions and amino acids in water leaked from LPSCs. They demonstrated the use of pulse voltammetry, utilising a zeolitic imidazolate framework-nitrogen-doped (ZIF-N) as the sensing element. These studies have demonstrated the potential of integrating AI predictive modelling with real-time sensor data in controlling Pb leakage from PSCs. Another notable contribution of AI is in the extraction of characteristics from complex degradation datasets. The mechanisms that result in the degradation of PSCs are multifaceted and encompass multiple processes that interact with one another, including ion transportation, water ingress, and thermal stress.104,160 Traditional statistical methods often overlook these nonlinear interactions. Nevertheless, deep learning models can process large multidimensional datasets to reveal latent relationships between environmental stressors and Pb release. This enables researchers to determine the most feeble degradation triggers and develop more robust device architectures.50
There are existing studies have successfully applied AI algorithms like ML to forecast new solar cell designs with strong performance and mechanical stability. ML-assisted designs have been employed to optimize defect tolerance and spectral response of perovskite photodetectors.161 This highlights the huge prospects of AI in analysing complex PSCs behaviour relevant to the MAPbI3 degradation pathways. Electrochemical techniques have also been combined with AI algorithms to create AI-assisted sensors, which have enhanced the selectivity and sensitivity of Pb2+ in soil and water systems. Design and development of PSCs have also utilized AI to accelerate the discovery and validation of experimental and theoretical data.162 However, some approaches are still speculative. For instance, whereas there exist proposals to use AI algorithms in predicting degradation due to mechanical stress, UV exposure, and humidity, there exist no industrially validated datasets to train robust AI models. The idea of using AI-based Internet of Things (IoT) sensor networks to monitor real-time Pb degradation is still at infancy since there exist no prototypes of portable sensors that have been integrated in autonomous self-learning networks. Secondly, no empirical data exists to support the use of AI in governance integration and policy frameworks, particularly in alignment with JT frameworks.
8. Life cycle assessment of lead-based perovskite solar cells
LCA of PSCs has been employed to examine the material composition of solar cell devices, particularly the amount of Pb in commonly used halide perovskites. Currently, the densities and molecular weights of formamidinium lead iodide (FAPbI3, 4.10g cm−3, 636 g mol−1), methylammonium lead bromide (MAPbBr3, 3.83 g cm−3, 482 g mol−1), and (MAPbI3, 4.09 g cm−3, 619.98 g mol−1) are typically employed to assess Pb concentration.163 These values act as a baseline against which the potential environmental risks of Pb leakage can be evaluated when producing, using, and disposing of perovskite-based devices.31 Among the most problematic aspects of the life cycle of PSCs is that they are prone to degradation under real-world operating conditions.18 The breakdown of perovskite films is also susceptible to degradation from various factors, including exposure to oxygen, moisture, heat, light, and even electric fields, which can reduce their stability and lifespan.18 These stressors are largely unavoidable in operation and therefore constitute a significant barrier to commercialising perovskite photovoltaics. Not only does degradation impact efficiency, but it also raises serious concerns about the release of toxic substances, such as Pb, into the environment.103The purpose of LCA is to conduct a systematic evaluation of the environmental impact of PSCs within a lifecycle framework, encompassing the mining of raw materials, device manufacturing, operation, and disposal.164 This holistic approach enables researchers to identify the stages at which environmental burdens are most critical and recommend effective mitigation measures. LPSCs, despite their high-power conversion efficiencies, must be evaluated against conventional silicon solar cells, which are more stable though require energy-intensive manufacturing.164 Comparative LCA studies have shown that PSCs generally have shorter energy payback times and lower greenhouse gas emissions than silicon-based technologies.97 However, the presence of Pb is a major drawback, where improper excavation or gradual wear can result in soil and water contamination. Researchers are therefore exploring Pb-free alternatives, such as tin-based perovskites, albeit with lower efficiency and stability challenges.104 The sustainability analysis of perovskite photovoltaics remains centred on the performance-environmental safety trade-off.31 The introduction of LCA into the perovskite field provides a framework that guides material development, device design, and recycling decisions. By quantifying the environmental impact at each stage of the product life cycle, stakeholders can make knowledge-based decisions to increase production without ecological degradation.103 The future will require improvements in encapsulation measures, recycling techniques, and the development of less toxic materials to ensure that PSCs can make a positive contribution to the global transition to renewable energy.14,18,165
Among the documented comparative LCA of PSCs, scientists at Chalmers University of Technology compared the environmental performance of perovskite modules with that of traditional silicon solar cells.166 The study found that perovskites also had a significantly shorter energy payback time because they were prepared as low-temperature solutions. However, their findings also highlighted that the instability of perovskites and the potential for releasing Pb during degradation posed environmental hazards and, therefore, are not environmentally sustainable.166 An extensive case study by Khan167 at the German Aerospace Centre (DLR) explored the LCA of future perovskite/silicon tandem solar cells. The study noted that tandem designs could reduce the overall carbon footprint of photovoltaics by improving efficiency and reducing the material volume per unit of electricity generated. The experiment, nonetheless, noted that environmental benefits were highly dependent on the stability of the perovskite layer because frequent replacement or degradation of the layer would offset the gains.167
9. Just transition to clean energy
Just transitions (JT) reflect the recognition that the transition to renewable energy is to mitigate greenhouse gas emissions and prioritize equity, justice, and community welfare. Energy justice is multidimensional, encompassing environmental health, community participation, equitable access, and labour rights.168 LPSCs containing toxic Pb raise serious ethical concerns about the trade-off between technological promise and etiological risks, owing to their strong potential to deliver high efficiency and stability.38 They offer a pathway to the democratization of solar energy, but they raise significant concerns regarding toxicity, environmental equity, and the uneven distribution of burdens and benefits. The risk of Pb leakage during the manufacturing, use, or disposal of perovskites remains unresolved despite ongoing research. Whereas efficiency, stability, and scale improvements dominate emerging scientific reports, and toxicity is occasionally addressed as a secondary concern through encapsulation or recycling.169 This underscores the tendency of industrial pioneers to prioritize performance metrics over social and environmental problems. Communities around manufacturing and waste-dumping sites also experience unprecedented consequences of new technologies.170 In cases of LPSCs, such communities can be disproportionately exposed to dangerous materials and the far-reaching implications of industrial effluents.171 Without proactive measures such as recycling mandates, the deployment of LPSCs risks reproducing the injustices of past energy systems, where the rich minority would hold the gains and the burdens would fall on vulnerable communities.Theoretically, low-cost PSCs could democratize solar adoption by reducing barriers to entry.172 However, with rapid advancement of LPSCs is envisaged to accelerate decarbonisation, critical issues, such as the use of toxic materials, remain uncertain. The rush to deploy LPSCs without appropriate safeguards creates new injustices to communities exposed to Pb pollution. To ensure equitable outcomes, participatory governance should involve communities in discussions about siting, production, and waste management.173 This would ensure that the benefits of LPSC deployment, such as improved access to clean, secure, and affordable energy, are distributed equally. There should also be transparent monitoring of emissions and strict recycling protocols to mitigate Pb leaching, with these protocols implemented in ways that are accountable and accessible to communities.
Marginalized groups such as rural communities, the indigenous population, and low-income households should be prioritized in expanding access to solar energy to mitigate existing inequalities.174 Previous studies have also shown that policies and regulations can serve as mediators in the development of risk management actions of LPSCs guided by justice principles. Strong governance frameworks, including safe material management, recycling mandates, and international standards, are essential for addressing inequitable harm.175 There is still a need for interdisciplinary research that bridges the gap between social justice and materials science, ensuring that innovations in PSCs are driven by equity and justice considerations. JT is not only about decarbonisation but also about ensuring that societies are more inclusive, equitable, resilient, and secure while embracing green energy transition.170
9.1. Relevant laws and legislation
Research laboratories and several companies are advancing LPSCs toward commercialisation; now, relevant laws and regulations need to be implemented to reduce and restrict Pb pollution. The highest-performing and most promising perovskite solar panels to be commercialised contain a highly toxic Pb, which can pose a potential risk to human health and the environment, as Pb2+ are soluble in water and can be absorbed into biological systems.34 Once a Pb particle is inside the body, it remains in soft tissues, and with continued exposure, it can be absorbed into the teeth and bones, potentially reducing its half-life to 20–30 years.176 The Restriction of Hazardous Substances (RoHS) and the United States Resource Conservation and Recovery Act (RCRA) both aim to reduce the risks associated with the use of hazardous materials and with disposal by regulating the transportation, generation, treatment, and disposal of hazardous waste.177 According to the EU, the RoHS directive primarily prevents the distribution of potentially hazardous commercial electrical and electronic (EE) products by restricting the use of hazardous substances and evaluating them on a per-weight basis. Here, the tolerated weight concentration limit for Pb in PSCs is 1000 mg kg−1 for the EU market.177 Moody et al.177 reported that the leaching of perovskite PVs poses a higher risk to environmental health than the Pb content itself, potentially endangering human life. The National Ambient Air Quality Standards (NAAQS) regulate the air concentrations of ozone, nitrogen dioxide, sulfur dioxide, carbon(IV) oxide, and Pb. The maximum safe level of Pb in ambient air is 0.15 µg m−3, based on a three-month average.178Accordingly, the legal framework for human health integrates specific global standards. For instance, the European Union (EU) has set standards and restrictions on the use of sludge containing metals, including Pb. EU limits Pb concentration in “homogenous materials” to 0.1% by weight.179 With respect to this, solar modules, “intended to be used in a system that is designed, assembled and installed by professionals for permanent use at a defined location”, are excepted from this restriction. Nevertheless, this legislation does not apply to devices embedded in portable systems or consumer electronics; specific legislation regulating immobilisation will be needed in the future. According to the World Health Organisation (WHO), the total Pb content in drinking water should not exceed 10 µg L−1. In China, the upper limit of Pb content in agricultural soils is 250 mg kg−1 to limit human exposure. Additionally, the United Nations (UN) Food and Agriculture Organisation (FAO) has established maximum Pb concentrations in canned food and beverages at 250 mg kg−1 and 150 mg kg−1, respectively.180 Elsewhere, the United States has set the safe-to-use Pb level at 5 µg L−1, with the goal of ensuring zero Pb content in drinking water.180 Also, the Centres for Disease Control (CDC) has set the limit blood Pb concentration of 3.5 µg dL−1.181
The Waste Electrical and Electronic Equipment (WEEE) directive involves large quantities of metals that can be isolated, recovered, and recycled, thereby providing substantial environmental benefits by decongesting landfills of potentially hazardous materials and reducing the risk of neurological disorders and cancer.182 Photovoltaics that can be reused, repaired, upgraded, remanufactured, disassembled, and efficiently recycled would have lower EoL costs, leading to a competitive Levelized Cost of Energy (LCOE) advantage.183 On the other hand, the RoHS directive limits Pb content to ≤0.1 wt% in homogeneous materials (i.e., materials with uniform composition throughout).183 Prior studies have shown that mass roll-to-roll (R2R) manufacturing could substantially reduce the environmental footprint per watt of electricity production.167 Research has also highlighted that without efficient recycling strategies, the toxicity of Pb-containing waste may pose a severe ecological hazard in the long term.167 Remarkably, a comparative LCA of Pb-based and Pb-free perovskites revealed that whereas Sn-based perovskites may reduce toxicity concerns, they do not compete with Pb-based perovskites in terms of efficiency and stability.169 However, the net environmental impact of tin-based devices may be higher due to shorter life and lower energy efficiency. Thus, improved encapsulation and recycling of LPSCs could be a more sustainable approach than switching to less efficient Pb-free perovskites.166
10. Future perspectives
Integration of Pb detection and monitoring techniques with JT principles into the deployment of PSCs remains a fundamental gap that demands attention for future research. Although detection and monitoring techniques have made remarkable advancements, they are restricted to laboratory-based demonstrations, which do not represent real-world conditions.184 In practice, PSCs are subjected to mechanical stress, temperature variations, and humidity.38 Therefore, without rigorous field validation, Pb leaching may continue until public trust and environmental safety are undermined. Moreover, the available ML algorithms data are fragmented, and there are limited generalizability and reproducibility of predictive models towards PSCs. This is a technological hurdle that hinders the development of globally acceptable monitoring systems. Therefore, additional research must focus on developing open-access datasets, which will help benchmark and expedite the development of universally acceptable monitoring systems. Accordingly, the shared resources will enable model frequency, support disassembly and recycling strategies.Fundamental studies on encapsulation engineering have underscored them as primary mitigation strategies to address Pb leakage. However, this approach fails to resolve issues such as EoL management, circular economy strategies, policy integration, recycling, safe disposal, and socio-technical considerations.38 Future investigations should focus on bridging the gap of justice concerns and technological detection methods. Encapsulation mechanisms that adapt to the environment should be developed. Additionally, participatory monitoring frameworks should involve affected communities in decision-making about waste management, production and siting to ensure adherence to policies like WHO drinking water limit and CDC lead threshold of the blood. This will make sure that technical innovation connects to equitable governance. Sustainability and performance principles should also guide the selection of materials used in PSCs.185
Whereas transition to lead-free PSC has been reported in prior studies as a solution to minimize the Pb leaching of PSCs, Pb-free alternatives have other analytic issues that need considerations.185 Sn-based devices, including Sn-based devices, are prone to instabilities and lower efficiency since the oxidation of Sn2+ to Sn4+ is rapid. In view of this, the focus shifts to monitoring Pb2+ mobilization to oxidation–state transitions in Sn. This implies that instead of using the already optimized techniques such as ICP-MS, in situ methods such as X-ray absorption and spectrochemical probes of redox systems must be improved. Additionally-Bi-based PSCs, can leach various species into the environment due to environmental degradation factors. While Pb-free alternatives are considered less toxic, they pose introduce scientific difficulties in terms of detection and quantification of Sb3+, Bi3+, and other leached, soluble PSCs species. Therefore, the challenge of Pb toxicity evolves to multi-element quantification and stability assessment. Furthermore, the transition to lead-free PSCs presents a paradigm shift regarding the application of the monitoring strategies.104 In the PSC settings, the ICP-MS is considered a gold standard, yet the focus will be on monitoring and quantification methods that prioritize field-applications and accessibility. This means that simple electrochemical instruments such as spectroscopic probes and electrochemical sensors that measure oxidation dynamics in real time will become prominent. The monitoring will also shift to less toxicology-based and more durability-based, and scientists will anticipate for materials that do not introduce unforeseen risks.104 Eventually, the shift to Pb-free will involve adaptive, cost-efficient, and inclusive technologies that can track the environmental interactions and degradation pathways of the modules.
11. Conclusions
In this review, we have provided a concise discussion on the prospects of perovskites, degradation pathways of MAPbI3-based devices, the environmental hazards associated with Pb leakage, Pb leakage detection strategies, including potential integration of AI to monitor Pb leakage, strategies for Pb immobilisation, recycling methods, and end-of-life management of decommissioned modules. Also, the balance between JT in embracing clean energy and the deployment of LPSCs among communities has been extensively discussed. Currently, perovskite solar technology is a leading candidate in the solar energy market; however, industrial advancement remains hindered by challenges in upscaling and stability. Encapsulation engineering strategies have been tested to address concerns about oxygen- and water-related degradation; nonetheless, inherent challenges related to light and thermal effects remain unresolved. To be able to compete with the widely deployed and most mature solar technology, industrial pioneers of perovskites must strive to narrow the efficiency gap between laboratory- and commercial-scale devices. By adopting strategies such as green solvents, device recycling, and encapsulation, the environmental impact of perovskites can be significantly reduced. Accordingly, AI offers a transformative pathway that not only accelerates predictive modelling and design strategies but also enables more effective monitoring of perovskite degradation. Ultimately, AI will transform the perovskite technology landscape, as AI-guided perovskite technologies are key to transitioning to sustainable solar power. As perovskite technology nears commercialisation, its successful deployment hinges on the use of green precursors to enable safer, more sustainable fabrication and recycling strategies. While Pb-free perovskite could offer new end-of-life management platforms, these devices are plagued by low efficiencies. Recent advances in Pb sequestration, particularly in external and internal encapsulation strategies for Pb capture and improved mechanical stability, offer promise for deployment and just transition protocols. However, recycling perovskites poses enormous challenges related to device stability and recycled device efficiency. Serious research is underway to develop a potential paradigm shift toward Pb-free perovskites to reduce health and environmental risks associated with LPSCs. Consequently, Pb-free perovskites will have different material composition, including the active layer, ETLs, and HTLs, offering the scientific community opportunities for recycling and environmental sustainability. The future outlook for perovskites is that additional research should focus on green, sustainable recycling protocols. Bearing this in mind, research on perovskites should not be restricted to one family (Pb-based devices), but also include new materials such as non-perovskites and chalcogenide-based perovskites with novel structures that offer bankability in terms of stability and optoelectronic properties. Future research should also focus on other applications of perovskite, such as light-emitting diodes (LEDs), detectors, lasers, and memory devices.Conflicts of interest
The authors declare no conflicts of interest.Consent for publication
This article has the consent of all the authors.Data availability
No new data were generated, as this is a review paper. Secondary research data has been used.Acknowledgements
The authors are grateful for support from the UK government's Ayrton Challenge Programme via the International Science Partnerships Fund (ISPF) as part of the REACH-PSM project. This work received funding from UK Research and Innovation (UKRI) through APP48267) REACH-PSM.References
- W. P. Wall, B. Khalid, M. Urbański and M. Kot, Factors influencing consumer's adoption of renewable energy, Energies, 2021, 14(17), 5420 CrossRef CAS.
- J. Singh and V. Mishra, Development of sustainable and ecofriendly metal ion scavenger for adsorbing Cu2+, Ni2+ and Zn2+ ions from the aqueous phase, Sep. Sci. Technol., 2022, 57(3), 354–371, DOI:10.1080/01496395.2021.1913421.
- N. S. Dutta, E. Gill, B. K. Arkhurst, M. Hallisey, K. Fu and K. Anderson, JUST-R metrics for considering energy justice in early-stage energy research, Joule, 2023, 7(3), 431–437 CrossRef.
- H. M. Saleh and A. I. Hassan, The challenges of sustainable energy transition: A focus on renewable energy, Appl. Chem. Eng., 2024, 7(2), 2084 CrossRef.
- A. D. Khan, M. Mustajab, S. Moeen, M. Imran, M. Ikram, Q. Khan and M. Khan, Advancements in the stability, protection and lead-free strategies of perovskite solar cells: a critical review, Environ. Sci.:Adv., 2024, 3(7), 1004–1029 CAS.
- S. Ali, S. Naseer, M. Rehman and Z. Wei, Recent trends and sources of lead toxicity: a review of state-of-the-art nano-remediation strategies, J. Nanopart. Res., 2024, 26(7), 168 CrossRef CAS.
- S. Lata and N. G. Ansari, Analytical Techniques for Heavy Metal Analysis, in Heavy Metal Contamination in the Environment, pp. 180–202. CRC Press, 2024) Search PubMed.
- S. B. Ruvubu and I. Roy, Advances in Heavy Metal Sensing: Utilizing Immobilized Chromogenic Reagents, Nanomaterials Perovskite and Nanonzymes, Crit. Rev. Anal. Chem., 2024, 1–28 Search PubMed.
- B. H. Durmishi, A. Durmishi and A. Shabani, The role of instrumental methods in chemical analysis for environmental protection, Int. J. Chem. Mater. Sci., 2025, 10(1), 1–22 Search PubMed.
- M. Bera, S. Das, S. Garai, S. Dutta, M. R. Choudhury, S. Tripathi and G. Chatterjee, Advancing energy efficiency: innovative technologies and strategic measures for achieving net zero emissions, Carbon Footprints, 2025, 4(1), 3 CrossRef.
- A. Ashraf and M. Sagheer, Renewable energy capacity and technological innovations: A review of global trends and future directions, Environ. Prog. Sustain. Energy, 2025, e70071 CrossRef CAS.
- L. Liu, Y. Tang, Y. Ma and B. Hu, Spin-Orbital Ordering Effects of Light Emission in Organic–Inorganic Hybrid Metal Halide Perovskites, Adv. Mater., 2025, 37(25), 2411913 CrossRef CAS PubMed.
- L. Zhang, M. Zhang, H. Wang, Z. Li, Z. Zhang, Y. Song, X. Song, S. Wang, J. Chen and W. Li, Diverse perovskite solar cells: progress, challenges, and perspectives, Adv. Mater., 2025, e12221 Search PubMed.
- A. Urbina, The balance between efficiency, stability and environmental impacts in perovskite solar cells: a review, J. Phys. Energy, 2020, 2(2), 022001 CrossRef CAS.
- H. Zhang, J.-W. Lee, G. Nasti, R. Handy, A. Abate, M. Grätzel and N.-G. Park, Lead immobilization for environmentally sustainable perovskite solar cells, Nature, 2023, 617(7962), 687–695 CrossRef CAS PubMed.
- L. M. Cavinato, E. Fresta, S. Ferrara and R. D. Costa, Merging biology and photovoltaics: How nature helps Sun-Catching, Adv. Energy Mater., 2021, 11(43), 2100520 CrossRef CAS.
- H. Luo, P. Li, J. Ma, L. Han, Y. Zhang and Y. Song, Sustainable Pb management in perovskite solar cells toward eco-friendly development, Adv. Energy Mater., 2022, 12(30), 2201242 CrossRef CAS.
- J. Suo, H. Pettersson and B. Yang, Sustainable approaches to address lead toxicity in halide perovskite solar cells: A review of lead encapsulation and recycling solutions, EcoMat, 2025, 7(1), e12511 CrossRef CAS.
- F. Znidi, M. Morsy and M. N. Uddin, Navigating challenges and solutions for metal-halide and carbon-based electrodes in perovskite solar cells (NCS-MCEPSC): An environmental approach, Heliyon, 2024, 10(12), e32843 CrossRef CAS PubMed.
- C. H. Chen, S. N. Cheng, L. Cheng, Z. K. Wang and L. S. Liao, Toxicity, leakage, and recycling of lead in perovskite photovoltaics, Adv. Energy Mater., 2023, 13(14), 2204144 CrossRef CAS.
- F. Li, Y. Yang, D. Kadyrma, A. Dosmukhambetova, Z. Liu, Z. Kalkozova, R. Chen and H. Wang, Beyond the Horizon: Exploration of Perovskite Solar Cells in Extreme Environments, ChemPhotoChem, 2025, 9(4), e202400337 CrossRef CAS.
- R. G. Charles, A. Doolin, R. García-Rodríguez, K. V. Villalobos and M. L. Davies, Circular economy for perovskite solar cells–drivers, progress and challenges, Energy Environ. Sci., 2023, 16(9), 3711–3733 RSC.
- W. Li, T. Mi, T. Tian, M. Yang and H. Pang, Mitigating Lead Toxicity in Halide Perovskite Solar Cells: Strategies for Sustainable Development, Inorganics, 2025, 13(4), 123 CrossRef CAS.
- P. Wu and F. Zhang, Recent advances in lead chemisorption for perovskite solar cells, Trans. Tianjin Univ., 2022, 28(5), 341–357 CrossRef CAS.
- G. G. Njema, N. Rono, B. C. Mosonik and J. K. Kibet, A review on advances towards achieving net-zero carbon footprint through sustainable agrivoltaic technology, Bull. Natl. Res. Cent., 2025, 49(1), 38, DOI:10.1186/s42269-025-01331-5.
- A. Sidler, F. Schmidt, B. Vallat, F. Grifoni, S. N. Habisreutinger, R. Suhonen, H. J. Snaith, A. Schäffer and M. Lenz, Assessment of soil impacts from lead release by lead-halide perovskite solar cells based on outdoor leaching tests, EES Solar, 2026, 2, 118–126 RSC.
- J. Dou, Y. Bai and Q. Chen, Challenges of lead leakage in perovskite solar cells, Mater. Chem. Front., 2022, 6(19), 2779–2789, 10.1039/D2QM00632D.
- T. Debnath, E. K. Kim, K. G. Lee and N. C. D. Nath, Halide perovskite solar cells with biocompatibility, Adv. Energy Sust. Res., 2020, 1(1), 2000028 CrossRef.
- J. Casanova-Chafer, R. Garcia-Aboal, P. Atienzar and E. Llobet, Unraveling the gas-sensing mechanisms of lead-free perovskites supported on graphene, Acs Sens., 2022, 7(12), 3753–3763 CrossRef CAS PubMed.
- I. Maietta, C. Otero-Martínez, S. Fernández, L. Sánchez, Á. González-Fernández, L. Polavarapu and R. Simón-Vázquez, The toxicity of lead and lead-free perovskite precursors and nanocrystals to human cells and aquatic organisms, Advanced Science, 2025, 12(13), 2415574 CrossRef CAS PubMed.
- W. Akram, X. Li, S. Ahmed, Z. Ouyang and G. Li, A review of life cycle assessment and sustainability analysis of perovskite/Si tandem solar cells, RSC Sustain., 2025, 3(1), 21–36 RSC.
- P. Zhu, C. Chen, J. Dai, Y. Zhang, R. Mao, S. Chen, J. Huang and J. Zhu, Toward the commercialization of perovskite solar modules, Adv. Mater., 2024, 36(15), 2307357 CrossRef CAS PubMed.
- Z. Zhang, P. Wu and F. Zhang, Lead Management In Perovskite Solar Cells, in Handbook of Perovskite Solar Cells, CRC Press, 2024, vol. 3, pp. 1–35 Search PubMed.
- C. E. Torrence, C. S. Libby, W. Nie and J. S. Stein, Environmental and health risks of perovskite solar modules: Case for better test standards and risk mitigation solutions, Iscience, 2023, 26(1), 105763 CrossRef PubMed.
- M. Tafurt-Cardona, C. E. Eismann, C. A. Suárez, A. A. Menegário, K. S. Luko and É. S. Junior, In situ selective determination of methylmercury in river water by diffusive gradient in thin films technique (DGT) using baker's yeast (Saccharomyces cerevisiae) immobilized in agarose gel as binding phase, Anal. Chim. Acta, 2015, 887, 38–44 CrossRef CAS PubMed.
- P. Yun, S. Devahastin and N. Chiewchan, Microstructures of encapsulates and their relations with encapsulation efficiency and controlled release of bioactive constituents: A review, Compr. Rev. Food Sci. Food Saf., 2021, 20(2), 1768–1799 CrossRef CAS PubMed.
- P. Baraneedharan, S. Sekar, S. Murugesan, D. Ahamada, S. A. B. Mohamed, Y. Lee and S. Lee, Recent advances and remaining challenges in perovskite solar cell components for innovative photovoltaics, Nanomaterials, 2024, 14(23), 1867 CrossRef CAS PubMed.
- G. G. Njema, S. N. Lemomo, P. K. Tanui and J. K. Kibet, A Review of the Strategies Used in the Recovery and Passivation of Lead in Lead-Based Perovskite Solar Cells: Balancing Device Efficiency, Toxicity, and Environmental Sustainability, Nat. Sci., 2025, e70041 Search PubMed.
- S. Min, M. Jeon, J. Cho, J. H. Bang and P. V. Kamat, Spectroelectrochemical insights into the intrinsic nature of lead halide perovskites, Nano Converg., 2024, 11(1), 49 CrossRef CAS PubMed.
- V. V. Halali, C. Sanjayan, V. Suvina, M. Sakar and R. G. Balakrishna, Perovskite nanomaterials as optical and electrochemical sensors, Inorg. Chem. Front., 2020, 7(14), 2702–2725 RSC.
- A. D. Ballantyne, J. P. Hallett, D. J. Riley, N. Shah and D. J. Payne, Lead acid battery recycling for the twenty-first century, R. Soc. Open Sci., 2018, 5(5), 171368 CrossRef PubMed.
- J. Joji, T. S. K. Naik and P. C. Ramamurthy, Biomimetic Approaches for Detecting Lead in Water Contaminated by Perovskite Solar Cells, Langmuir, 2025, 41(5), 3236–3248 CrossRef CAS PubMed.
- H. Wang, X. Chen, X. Li, J. Qu, H. Xie, S. Gao, D. Wang and H. Yin, Recovery of lead and iodine from spent perovskite solar cells in molten salt, Chem. Eng. J., 2022, 447, 137498 CrossRef CAS.
- J. Sun, H. Fu, H. Jing, X. Hu, D. Chen, F. Li, Y. Liu, X. Qin and W. Huang, Synergistic Integration of Halide Perovskite and Rare-Earth Ions toward Photonics, Adv. Mater., 2025, 37(12), 2417397 CrossRef CAS PubMed.
- X. Li, F. Zhang, H. He, J. J. Berry, K. Zhu and T. Xu, On-device lead sequestration for perovskite solar cells, Nature, 2020, 578(7796), 555–558 CrossRef CAS PubMed.
- H. Lv, G. Zhang, W. Yang, X. Dai, Y. Huang, J. Ni and Q. Wang, Portable anti-fouling electrochemical sensor for soil heavy metal ions detection based on the screen-printed carbon electrode modified with silica isoporous membrane, J. Electroanal. Chem., 2023, 930, 117141 CrossRef CAS.
- Z. He, W. Lian, Y. Lv, Z. Duan and Z. Fan, Review of the Mitigation Scale Performance of Anti-Fouling Coatings Surface Characteristics on Industrial Heat Exchange Surfaces, Coatings, 2025, 16(1), 40 CrossRef.
- Y. Tian, D. Lei, P. Wang, J. Bao, Y. Wang, T. Zhao and W. Guo, Research Progress on the Stability and Durability of Ag/AgCl Prepared by Anodic Chlorination Method for Chloride Ion Sensors in Cement-Based Materials, Buildings, 2025, 15(13), 2290 CrossRef.
- X. Dong and S. Han, Real-Time Correction and Long-Term Drift Compensation in MOS Gas Sensor Arrays Using Iterative Random Forests and Incremental Domain-Adversarial Networks, Micromachines, 2025, 16(9), 991 CrossRef PubMed.
- L. Pancini, R. Montecucco, V. Larini, A. Benassi, D. Mirani, G. Pica, M. De Bastiani, F. Doria and G. Grancini, A fluorescent sensor to detect lead leakage from perovskite solar cells, Mater. Adv., 2023, 4(11), 2410–2417 RSC.
- B. Chen, C. Fei, S. Chen, H. Gu, X. Xiao and J. Huang, Recycling lead and transparent conductors from perovskite solar modules, Nat. Commun., 2021, 12(1), 5859 CrossRef CAS PubMed.
- V. Tomoko, Thermo Scientific iCAP RQ ICP-MS: Typical Limits of Detection. A Technical Note. TN43427-EN 117, 2017 Search PubMed.
- D. Liu, T. Zhu, G. Li, Z. Song, Y. Yan and C. Li, Inhibition of Lead Toxicity of Perovskite Solar Cells, Adv. Energy Mater., 2026, e05840 CrossRef.
- W.-Q. Lai, Y.-F. Chang, F.-N. Chou and D.-M. Yang, Portable FRET-based biosensor device for on-site lead detection, Biosensors, 2022, 12(3), 157 CrossRef CAS PubMed.
- M. Sabah and A. Fethi, Electrochemical detection of lead in real samples using carbon nanostructure and inactivated E. Coli as low-cost sensitive biosensor with high electrocatalytic performance, Electrocatalysis, 2022, 13(6), 773–783 CrossRef CAS.
- R. D. Ross, Y. Han, H.-Y. Jeong, J. M. Ynzunza, R. H. Lavroff, A. Banerjee, A. Prajapati, C. G. Morales-Guio, J. M. Velázquez and A. N. Alexandrova, Solution and Active Site Speciation Drive Selectivity for Electrocatalytic Reactive Carbon Capture in Diethanolamine over Ni–N–C Catalysts, J. Am. Chem. Soc., 2026, 148(3), 3962–3975 CrossRef CAS PubMed.
- A. Netzahual-Lopantzi, L. Juárez-Santacruz and E. García-Nieto, A rapid colorimetric method for the determination of lead (II) at low concentrations in aqueous solution, Int. J. Environ. Sci. Technol., 2023, 20(12), 13191–13198 CrossRef CAS.
- K. Ma, X. Li, F. Yang and H. Liu, Lead leakage of Pb-based perovskite solar cells, Coatings, 2023, 13(6), 1009 CrossRef CAS.
- H. Teymourinia, C. Gonzales, J. J. Gallardo, M. Salavati-Niasari, J. Bisquert, J. Navas and A. Guerrero, Interfacial passivation of perovskite solar cells by reactive ion scavengers, ACS Appl. Energy Mater., 2021, 4(2), 1078–1084 CrossRef CAS.
- S. I. Mohammad, H. S. Jabbar, A. Vasudevan, I. Sapaev, M. Rekha, S. Gayathri, H. Zabebah, R. Sharma, P. Samantsinghar and S. Mahmoodi, Comprehensive advances in CsPbBr 3 perovskite quantum dots for ultrasensitive fluorescent nanosensors in food safety monitoring, Nanoscale Adv., 2026, 8, 422–457 RSC.
- A. Bukhamsin, J. Kosel, I. Blilou and K. N. Salama, Accelerating adoption of species-agnostic plant sensors for precision farming, Nat. Rev. Electr. Eng., 2025, 2(1), 58–70 CrossRef.
- S. Paul, P. Barman, N. Dey and M. Watkinson, Recent developments in pyrene-based fluorescence recognition and imaging of Ag+ and Pb 2+ ions: Synthesis, applications and challenges, Sens. diagn., 2024, 3(6), 946–967 RSC.
- Y. Lam, L. L. Nguyen, Q. Zhang, D. Bradley, T. Salim, P. Li, P. Mishra, A. Mueller, S. Mondal and K. S. Chong, Multi-technique Detection of Lead Iodide Hybrid Perovskite Degradation Pathways under Varying Electric Fields, 2023 Search PubMed.
- M. Shellaiah, K. W. Sun, N. Thirumalaivasan, M. Bhushan and A. Murugan, Sensing utilities of cesium lead halide perovskites and composites: A comprehensive review, Sensors, 2024, 24(8), 2504 CrossRef CAS PubMed.
- M. Dehghanipour, A. Kumar, M. Al-Hedrewy, P. Kanjariya, A. Rajiv, A. Shankhyan, K. Jayabalan and S. K. Samal, Emerging perovskite-based electrochemical biosensors for different biomarkers detection: innovations, applications, and future prospects, Microchim. Acta, 2025, 192(8), 474 CrossRef CAS PubMed.
- P. Nath, K. R. Mahtaba and A. Ray, Fluorescence-based portable assays for detection of biological and chemical analytes, Sensors, 2023, 23(11), 5053 CrossRef CAS PubMed.
- H. Xu, W. Qin, J. Zhu and Y.-G. Liu, A smartphone-assisted chromogenic and fluorogenic dual-channel sensor for specific determination of toxic Pb2+ ions with multifaceted applications, Tetrahedron, 2024, 168, 134316 CrossRef CAS.
- K. E. Adesina, C. J. Burgos, T. R. Grier, A. S. Sayam and A. J. Specht, Ways to measure metals: From ICP-MS to XRF, Curr. Environ. Health Rep., 2025, 12(1), 7 CrossRef PubMed.
- K. Zhu, H. Jia, S. Zhao, T. Xia, X. Guo, T. Wang and L. Zhu, Formation of environmentally persistent free radicals on microplastics under light irradiation, Environ. Sci. Technol., 2019, 53(14), 8177–8186 CrossRef CAS.
- A. Shrivastava and V. B. Gupta, Methods for the determination of limit of detection and limit of quantitation of the analytical methods, Chron. Young Sci., 2011, 2(1), 21–25 CrossRef.
- T. Hu, Q. Lai, W. Fan, Y. Zhang and Z. Liu, Advances in portable heavy metal ion sensors, Sensors, 2023, 23(8), 4125 CrossRef CAS PubMed.
- P. K. Mehta, J. Lee, E.-T. Oh, H. J. Park and K.-H. Lee, Ratiometric fluorescence sensing system for lead ions based on self-assembly of bioprobes triggered by specific Pb2+–peptide interactions, ACS Appl. Mater. Interfaces, 2023, 15(11), 14131–14145 CAS.
- Y. Chen, H. Xu, M. S. Khan, S. Han and S. Zhu, Recent advances in layered double hydroxides for pharmaceutical wastewater treatment: A critical review, Crit. Rev. Environ. Sci. Technol., 2025, 1–27 Search PubMed.
- L. Wang, H. Liu, L. Li, R. Guo, X. Yang, X. Chen, Y. Wen, M. Cao, J. Yan and G. Liu, High-selectivity NIR Biosensor and in vivo imaging for Hg2+ detection based on a DNA-wrapped Single-Chirality Carbon Nanotube, J. Hazard. Mater., 2025, 138979 CrossRef CAS PubMed.
- A. V. Spyrou, K. Zodhiates and Y. Deligiannakis, Comparison of Single Atoms vs. Sub-Nanoclusters as Co-Catalysts in Perovskites and Metal Oxides for Photocatalytic Technologies, Nanomaterials, 2025, 15(3), 226 CrossRef CAS PubMed.
- E. Leccisi and V. Fthenakis, Life-cycle human-and eco-toxicity assessment of emerging lead-based perovskite compared to conventional photovoltaic panels, MRS Bull., 2024, 49(12), 1240–1250 CrossRef CAS.
- A. Ko and C. Liao, based colorimetric sensors for point-of-care testing, Anal. Methods, 2023, 15(35), 4377–4404 RSC.
- Y. Wang, Y. Ma, Y. Zhang, R. Liu, L. Li, N. Zhang, P. Wang and L. Zhang, Suppressing the Residual PbI2 and Releasing Residual Stress to Regulate Crystallization of Perovskite Film in MAPbI3 PSCs via Doping Functional L-AA-rGO, Small Met., 2026, 10(3), e02000 CrossRef CAS PubMed.
- V. Selvanathan, N. H. Suhaimi, A. W. M. Zuhdi, Y. B. Kar, P. Chelvanathan, M. Akhtaruzzaman and T. S. Kiong, Recycling Strategies for Lead Halide Perovskite Solar Cells: Current Approaches, Challenges, and Future Directions, J. Sci. Adv. Mater. Devices, 2025, 100969 CrossRef CAS.
- M. B. Rahman, M. H. Miah, M. U. Khandaker and M. A. Islam, Selection of a compatible electron transport layer and hole transport layer for the mixed perovskite FA 0.85 Cs 0.15 Pb (I 0.85 Br 0.15) 3, towards achieving novel structure and high-efficiency perovskite solar cells: a detailed numerical study by SCAPS-1D, RSC Adv., 2023, 13(25), 17130–17142 RSC.
- M. H. Miah, M. B. Rahman, M. Nur-E-Alam, M. A. Islam, M. Shahinuzzaman, M. R. Rahman, M. H. Ullah and M. U. Khandaker, Key degradation mechanisms of perovskite solar cells and strategies for enhanced stability: issues and prospects, RSC Adv., 2025, 15(1), 628–654 RSC.
- P. Caprioglio, M. Stolterfoht, C. M. Wolff, T. Unold, B. Rech, S. Albrecht and D. Neher, On the relation between the open-circuit voltage and quasi-fermi level splitting in efficient perovskite solar cells, Adv. Energy Mater., 2019, 9(33), 1901631 CrossRef.
- F. P. Sabino, G. M. Dalpian and A. Zunger, Light-Induced Frenkel Defect Pair Formation Can Lead to Phase-Segregation of Otherwise Miscible Halide Perovskite Alloys, Adv. Energy Mater., 2023, 13(44), 2301539, DOI:10.1002/aenm.202301539.
- E. Ramírez, R. Betancur, J. F. Montoya, E. Velilla, D. Ramírez and F. Jaramillo, Encapsulation against extrinsic degradation factors and stability testing of perovskite solar cells, Recent Advances in Multifunctional Perovskite Materials, 2022 Search PubMed.
- S. S. Dipta, M. A. Rahim and A. Uddin, Encapsulating perovskite solar cells for long-term stability and prevention of lead toxicity, Appl. Phys. Rev., 2024, 11(2), 021301 CAS.
- A. H. Howlader and A. Uddin, Progress and challenges of chloride–iodide perovskite solar cells: a critical review, Nanomanufacturing, 2023, 3(2), 177–216 CrossRef.
- H. Zhang, K. Li, M. Sun, F. Wang, H. Wang and A. K. Y. Jen, Design of superhydrophobic surfaces for stable perovskite solar cells with reducing lead leakage, Adv. Energy Mater., 2021, 11(41), 2102281 CrossRef CAS.
- X. Xiao, M. Wang, S. Chen, Y. Zhang, H. Gu, Y. Deng, G. Yang, C. Fei, B. Chen and Y. Lin, Lead-adsorbing ionogel-based encapsulation for impact-resistant, stable, and lead-safe perovskite modules, Sci. Adv., 2021, 7(44), eabi8249 CrossRef CAS PubMed.
- X. Li, F. Zhang, J. Wang, J. Tong, T. Xu and K. Zhu, On-device lead-absorbing tapes for sustainable perovskite solar cells, Nat Sustain., 2021, 4(12), 1038–1041 CrossRef.
- Y. I. Lee, N. J. Jeon, B. J. Kim, H. Shim, T. Y. Yang, S. I. Seok, J. Seo and S. G. Im, A low-temperature thin-film encapsulation for enhanced stability of a highly efficient perovskite solar cell, Adv. Energy Mater., 2018, 8(9), 1701928 CrossRef.
- H. Wang, Y. Zhao, Z. Wang, Y. Liu, Z. Zhao, G. Xu, T.-H. Han, J.-W. Lee, C. Chen and D. Bao, Hermetic seal for perovskite solar cells: An improved plasma enhanced atomic layer deposition encapsulation, Nano Energy, 2020, 69, 104375 CrossRef CAS.
- Y. Lv, H. Zhang, R. Liu, Y. Sun and W. Huang, Composite encapsulation enabled superior comprehensive stability of perovskite solar cells, ACS Appl. Mater. Interfaces, 2020, 12(24), 27277–27285 CrossRef CAS PubMed.
- R. Ullah, N. Khan, R. Khattak, M. Khan, M. S. Khan and O. M. Ali, Preparation of Electrochemical Supercapacitor Based on Polypyrrole/Gum Arabic Composites, Polymers, 2022, 14(2), 242 CrossRef CAS PubMed.
- S. Chen, Y. Deng, X. Xiao, S. Xu, P. N. Rudd and J. Huang, Preventing lead leakage with built-in resin layers for sustainable perovskite solar cells, Nat Sustain., 2021, 4(7), 636–643 CrossRef.
- B. Conings, A. Babayigit and H.-G. Boyen, Fire safety of lead halide perovskite photovoltaics, ACS Energy Lett., 2019, 4(4), 873–878 CrossRef CAS.
- F. Schmidt, M. Amrein, S. Hedwig, M. Kober-Czerny, A. Paracchino, V. Holappa, R. Suhonen, A. Schäffer, E. C. Constable, H. J. Snaith and M. Lenz, Organic solvent free PbI2 recycling from perovskite solar cells using hot water, J. Hazard. Mater., 2023, 447, 130829, DOI:10.1016/j.jhazmat.2023.130829.
- R. Vidal, J.-A. Alberola-Borràs, S. N. Habisreutinger, J.-L. Gimeno-Molina, D. T. Moore, T. H. Schloemer, I. Mora-Seró, J. J. Berry and J. M. Luther, Assessing health and environmental impacts of solvents for producing perovskite solar cells, Nat Sustain., 2021, 4(3), 277–285 CrossRef.
- M. L. Rencheck, C. Libby, A. Montgomery and J. S. Stein, Managing potential environmental and human health risks of lead halide perovskite photovoltaic modules, Sol. Energy, 2024, 269, 112337 CrossRef CAS.
- Q.-Q. Chu, Z. Sun, D. Wang, B. Cheng, H. Wang, C.-P. Wong and B. Fang, Encapsulation: The path to commercialization of stable perovskite solar cells, Matter, 2023, 6(11), 3838–3863 CrossRef CAS.
- W. Yang, Y. Zhang, C. Xiao, J. Yang and T. Shi, A review of encapsulation methods and geometric improvements of perovskite solar cells and modules for mass production and commercialization, Nano Mater. Sci., 2025, 7(6), 790–809 CrossRef CAS.
- S. Kim, S. H. Cho, K. Jeong, J. Lee, Y. Jung, M. Choi and Y. S. Lee, Intensifying Chelation of Pb-Related Defects for Enhancing Stability in Halide Perovskite Thin-Film Solar Cells, Sol. RRL, 2025, 202500212 Search PubMed.
- P. Xu, J. Liu, S. Wang, J. Chen, B. Han, Y. Meng, S. Yang, L. Xie, M. Yang and R. Jia, Dynamic covalent polymer engineering for stable and self-healing perovskite solar cells, Mater. Horiz., 2023, 10(11), 5223–5234 RSC.
- V. K. Ravi, B. Mondal, V. V. Nawale and A. Nag, Don't let the lead out: new material chemistry approaches for sustainable lead halide perovskite solar cells, ACS Omega, 2020, 5(46), 29631–29641 CrossRef CAS PubMed.
- M. H. Miah, M. U. Khandaker, M. J. Hossen, I. Jahan, M. Shahinuzzaman, M. Nur-E-Alam, M. Y. Hanfi, M. H. Ullah and M. A. Islam, Lead-free alternatives and toxicity mitigation strategies for sustainable perovskite solar cells: a critical review, Mater. Adv., 2025, 6(9), 2718–2752 RSC.
- V. Larini, M. Degani, S. Cavalli and G. Grancini, Sustainable decommissioning of perovskite solar cells: from waste to resources, Chem. Soc. Rev., 2025, 54(15), 7252–7270 RSC.
- C. Yang, W. Hu, J. Liu, C. Han, Q. Gao, A. Mei, Y. Zhou, F. Guo and H. Han, Achievements, challenges, and future prospects for industrialization of perovskite solar cells, Light: Sci. Appl., 2024, 13(1), 227 CrossRef CAS PubMed.
- J. Xu, Z. Hu, L. Huang, X. Huang, X. Jia, J. Zhang, J. Zhang and Y. Zhu, In situ recycle of PbI2 as a step towards sustainable perovskite solar cells, Prog. Photovolt., 2017, 25(12), 1022–1033 CrossRef CAS.
- B. Bokor, Legal analysis of the EU regulatory framework on circular economy and sustainability principles in plastic food packaging, Clean. Waste Syst., 2025, 12, 100412, DOI:10.1016/j.clwas.2025.100412.
- A. Dhingra, O. Thakur and R. Pandey, Evaluation of Toxicity of Lead in Perovskite Solar Cells: Current Status and Future Challenges, in Perovskite Solar Cells: Reshaping the Future Energy Landscape, Springer, 2025, pp. 279–298 Search PubMed.
- K. Valadez-Villalobos and M. L. Davies, Remanufacturing of perovskite solar cells, RSC Sustain., 2024, 2(8), 2057–2068 RSC.
- X. Wu, D. Zhang, X. Wang, X. Jiang, B. Liu, B. Li, Z. Li, D. Gao, C. Zhang and Y. Wang, Eco-friendly perovskite solar cells: From materials design to device processing and recycling, EcoMat, 2023, 5(7), e12352 CrossRef CAS.
- S. Y. Park, J.-S. Park, B. J. Kim, H. Lee, A. Walsh, K. Zhu, D. H. Kim and H. S. Jung, Sustainable lead management in halide perovskite solar cells, Nat Sustainability, 2020, 3(12), 1044–1051 CrossRef.
- S. Zhang, L. Shen, M. Huang, Y. Yu, L. Lei, J. Shao, Q. Zhao, Z. Wu, J. Wang and S. Yang, Cyclic utilization of lead in carbon-based perovskite solar cells, ACS Sustain. Chem. Eng., 2018, 6(6), 7558–7564 CrossRef CAS.
- M. Yang, T. Tian, Y. Fang, W.-G. Li, G. Liu, W. Feng, M. Xu and W.-Q. Wu, Reducing lead toxicity of perovskite solar cells with a built-in supramolecular complex, Nat Sustain., 2023, 6(11), 1455–1464 CrossRef.
- C. G. Poll, G. W. Nelson, D. M. Pickup, A. V. Chadwick, D. J. Riley and D. J. Payne, Electrochemical recycling of lead from hybrid organic–inorganic perovskites using deep eutectic solvents, Green Chem., 2016, 18(10), 2946–2955 RSC.
- J. Li, R. Xia, W. Qi, X. Zhou, J. Cheng, Y. Chen, G. Hou, Y. Ding, Y. Li and Y. Zhao, Encapsulation of perovskite solar cells for enhanced stability: Structures, materials and characterization, J. Power Sources, 2021, 485, 229313 CrossRef CAS.
- S. M. Sivasankar, C. D. O. Amorim and A. F. D. Cunha, Progress in thin-film photovoltaics: A review of key strategies to enhance the efficiency of CIGS, CdTe, and CZTSSe solar cells, J. Compos. Sci., 2025, 9(3), 143 CrossRef CAS.
- G. Wei, Y. Zhou, Z. Hou, Y. Li, Q. Liu, J. Chen and D. He, Review of C-Si PV Module Recycling and Industrial Feasibility, EES Solar, 2025 Search PubMed.
- C. I. Sprague, V. De La Asunción-Nadal and A. García-Fernández, Implementing AI in Advanced Recycling of Perovskite Solar Cells, in 2024 9th International Conference on Smart and Sustainable Technologies (SpliTech), IEEE, 2024, pp. 1–4 Search PubMed.
- J. Príncipe, L. Andrade, T. M. Mata and A. A. Martins, Comparative Life Cycle Assessment of Perovskite Solar Cell Production: Mesoporous n-i-p Versus Inverted p-i-n Architectures, Adv. Energy Sust. Res., 2025, 6(6), 2400368 CrossRef.
- A. Babayigit, H.-G. Boyen and B. Conings, Environment versus sustainable energy: The case of lead halide perovskite-based solar cells, MRS Energy Sustain., 2018, 5, E1 CrossRef.
- E. McCalmont, A. Ravilla, T. O'Hara, B. Carlson, J. Kellar and I. Celik, Life cycle cost assessment of material recovery from perovskite solar cells, MRS Adv., 2023, 8(6), 317–322 CrossRef CAS.
- R. Szostak, A. de Souza Gonçalves, J. N. de Freitas, P. E. Marchezi, F. L. de Araújo, H. C. N. Tolentino, M. F. Toney, F. das Chagas Marques and A. F. Nogueira, In situ and operando characterizations of metal halide perovskite and solar cells: insights from lab-sized devices to upscaling processes, Chem. Rev., 2023, 123(6), 3160–3236 CrossRef CAS PubMed.
- K. Suchan, J. Just, P. Becker, E. L. Unger and T. Unold, Optical in situ monitoring during the synthesis of halide perovskite solar cells reveals formation kinetics and evolution of optoelectronic properties, J. Mater. Chem. A, 2020, 8(20), 10439–10449 RSC.
- E. Yuan, H. Huang, D. Zhang, Z. Zheng, D. Sun, J. Dong and Y. Qiu, Real-time three-dimensional monitoring and analysis of perovskite solar cell using short wavelength infrared digital holography, Appl. Phys. Lett., 2024, 125(23), 234102 CrossRef CAS.
- B. Xia, J. Jin, N. Wang and Q. Tai, Degradation Evolution of Perovskite Solar Cells via In Situ Real-Time Optical Observation, Adv. Funct. Mater., 2024, 34(6), 2308581 CrossRef CAS.
- G. R. Han, M. N. An, H. Jang, N. S. Han, J. Kim, K. S. Jeong, T. H. Yoon and M. Cho, In situ and real-time ultrafast spectroscopy of photoinduced reactions in perovskite nanomaterials, Nat. Commun., 2025, 16(1), 4956 CrossRef CAS PubMed.
- Q. Zou, Z. Lin, Z. Li, G. Hu, S. Zhou, Y. Zheng, L. Huang, S. Liang, H. Duan and S. Yuan, Closed-loop recycling of lead iodide precursor from waste organic solvents in the production of perovskite solar cells: Toward sustainable photovoltaics, Waste Manage., 2025, 206, 115049 CrossRef CAS.
- Z. Wu, M. Sytnyk, J. Zhang, G. Babayeva, C. Kupfer, J. Hu, S. Arnold, J. Hauch, C. Brabec and I. M. Peters, Closing the loop: recycling of MAPbI 3 perovskite solar cells, Energy Environ. Sci., 2024, 17(12), 4248–4262 RSC.
- D. Kumar, Y. N. Guguloth, J. K. Rath and T. Singh, Comparative analysis of recycling strategies for high-yield fabrication of PSCs: Resolving residual impurity challenges via targeted post-treatment, EES Solar, 2025, 1(6), 1074–1092 RSC.
- H. Zhang and N.-G. Park, Towards sustainability with self-healing and recyclable perovskite solar cells, EScience, 2022, 2(6), 567–572 CrossRef CAS.
- J. S. Hong, H. J. Kim, C. H. Sohn, O. Y. Gong, J. H. Choi, K. H. Cho, G. S. Han, K. T. Nam and H. S. Jung, High-throughput Pb recycling for perovskite solar cells using biomimetic whitlockite, Energy Environ. Mater., 2023, 6(3), e12374 CrossRef CAS.
- M. Ren, Y. Miao, T. Zhang, Z. Qin, Y. Chen, N. Wei, X. Qian, T. Wang and Y. Zhao, Lead stabilization and iodine recycling of lead halide perovskite solar cells, ACS Sustain. Chem. Eng., 2021, 9(48), 16519–16525 CrossRef CAS.
- X. Feng, Q. Guo, J. Xiu, Z. Ying, K. W. Ng, L. Huang, S. Wang, H. Pan, Z. Tang and Z. He, Close-loop recycling of perovskite solar cells through dissolution-recrystallization of perovskite by butylamine, Cell Rep. Phys. Sci., 2021, 2(2), 100341 CrossRef CAS.
- A. Binek, M. L. Petrus, N. Huber, H. Bristow, Y. Hu, T. Bein and P. Docampo, Recycling perovskite solar cells to avoid lead waste, ACS Appl. Mater. Interfaces, 2016, 8(20), 12881–12886 CrossRef CAS.
- F. Schmidt, M. Amrein, S. Hedwig, M. Kober-Czerny, A. Paracchino, V. Holappa, R. Suhonen, A. Schäffer, E. C. Constable and H. J. Snaith, Organic solvent free PbI2 recycling from perovskite solar cells using hot water, J. Hazard. Mater., 2023, 447, 130829 CrossRef CAS PubMed.
- K. Wang, T. Ye, X. Huang, Y. Hou, J. Yoon, D. Yang, X. Hu, X. Jiang, C. Wu and G. Zhou, “One-key-reset” recycling of whole perovskite solar cell, Matter, 2021, 4(7), 2522–2541 CrossRef CAS.
- T. O'Hara, A. Ravilla, E. McCalmont, B. Carlson, J. Kellar, Z. Song and I. Celik, Novel method of recycling perovskite solar cells using iodide solutions, MRS Adv., 2023, 8(6), 296–301 CrossRef.
- F. Barayeva, A. Beisenbayev, B. Zhussipbay, Y. Olzhabay, D. Aidarkhanov and A. Ng, Feasible Strategies for Lead Recovery and Recycling from End-of-Life Perovskite Solar Cells Towards Environmentally Sustainable Technologies, Eurasian Chem.-Technol. J., 2025, 27(3), 199–208 CrossRef.
- V. de la Asunción-Nadal, C. I. Sprague, B. Guijarro-Berdiñas, U. B. Cappel and A. García-Fernández, Machine learning for perovskite solar cells: a comprehensive review on opportunities and challenges for materials scientists, EES Solar, 2025, 1(6), 927–957 RSC.
- L. Klein, S. Ziegler, F. Laufer, C. Debus, M. Götz, K. Maier-Hein, U. W. Paetzold, F. Isensee and P. F. Jäger, Discovering process dynamics for scalable perovskite solar cell manufacturing with explainable AI, Adv. Mater., 2024, 36(7), 2307160 CrossRef CAS.
- S. Antony Jose, C. A. D. Cook, J. Palacios, H. Seo, C. E. Torres Ramirez, J. Wu and P. L. Menezes, Recent Advancements in Artificial Intelligence in Battery Recycling, Batteries, 2024, 10(12), 440 CrossRef CAS.
- F. Song, D. Zheng, J. Feng, J. Liu, T. Ye, Z. Li, K. Wang, S. Liu and D. Yang, Mechanical durability and flexibility in perovskite photovoltaics: advancements and applications, Adv. Mater., 2024, 36(18), 2312041 CrossRef CAS PubMed.
- S. H. Godasiaei, Optimizing the performance of vapor-deposited perovskite solar cells through advanced predictive modeling, J. Mater. Sci., 2024, 59(21), 9398–9422 CrossRef CAS.
- S. Subba, P. Rai and S. Chatterjee, Machine Learning Approaches in Advancing Perovskite Solar Cells Research, Adv. Theory Simul., 2025, 8(3), 2400652 CrossRef CAS.
- W. A. Dunlap-Shohl, Y. Meng, P. P. Sunkari, D. A. Beck, M. Meilă and H. W. Hillhouse, Physiochemical machine learning models predict operational lifetimes of CH 3 NH 3 PbI 3 perovskite solar cells, J. Mater. Chem. A, 2024, 12(16), 9730–9746 RSC.
- R. Bhaduri and S. Manasa, Perovskite Solar Cell Stability Analysis Using Entropy-Based Support Vector Machines Learning, Prog. Photovolt., 2025, 33(9), 962–979 CrossRef CAS.
- R. Khanam, M. Hussain, R. Hill and P. Allen, A comprehensive review of convolutional neural networks for defect detection in industrial applications, IEEE Access, 2024, 12, 94250–94295 Search PubMed.
- W. Shafik, An Overview of Artificial Intelligence Solutions for the Maintenance and Evaluation of Photovoltaic Systems, Energy Conversion Systems-Based Artificial Intelligence: Applications and Tools, 2025, pp. 23–53 Search PubMed.
- M. Shahin, F. F. Chen, A. Hosseinzadeh and N. Zand, Using machine learning and deep learning algorithms for downtime minimization in manufacturing systems: An early failure detection diagnostic service, J. Adv. Manuf. Technol., 2023, 128(9), 3857–3883 CrossRef.
- A. R. Hering, C. M. Sutter-Fella and M. S. Leite, An AI-accelerated pathway for reproducible and stable halide perovskites, Chem. Soc. Rev., 2025, 54(22), 10397–10426 RSC.
- A. Glaws, J. W. Schall, A. Ballen, A. Louks, K. O. Davis, A. F. Palmstrom, J. Ugirumurera and D. B. Kern, Explainable artificial intelligence relates perovskite luminescence images to current-voltage metrics, Energy, 2025, 100640 Search PubMed.
- C. H. Chan, M. Sun and B. Huang, Application of machine learning for advanced material prediction and design, EcoMat, 2022, 4(4), e12194 CrossRef CAS.
- P. Raut, D. K. Panda and A. K. Goyal, A Comprehensive Review on Next-Generation Modelling and Optimization for Semiconductor Devices, IEEE Access, 2025, 13, 123724–123742 Search PubMed.
- A. Puthanveettil Madathil, X. Luo, Q. Liu, C. Walker, R. Madarkar and Y. Qin, A review of explainable artificial intelligence in smart manufacturing, Int. J. Prod. Res., 2025, 1–44 Search PubMed.
- S. K. Rath, M. Sahu, S. P. Das, S. K. Bisoy and M. Sain, A comparative analysis of SVM and ELM classification on software reliability prediction model, Electronics, 2022, 11(17), 2707 CrossRef.
- K. Hasan, S. B. Yousuf, M. S. H. K. Tushar, B. K. Das, P. Das and M. S. Islam, Effects of different environmental and operational factors on the PV performance: A comprehensive review, Energy Sci. Eng., 2022, 10(2), 656–675 CrossRef.
- J. Zhang, B. Liu, Z. Liu, J. Wu, S. Arnold, H. Shi, T. Osterrieder, J. A. Hauch, Z. Wu and J. Luo, Optimizing perovskite thin-film parameter spaces with machine learning-guided robotic platform for high-performance perovskite solar cells, Adv. Energy Mater., 2023, 13(48), 2302594 CrossRef CAS.
- T. D. Doan, T. H. Y. Pham, D. D. Luong, N. H. Thi, H. T. Oanh, T. T. Le, H. T. Nguyen, T. K. D. Hoang and M. H. Hoang, A highly sensitive electrochemical sensor for the detection of lead (II) ions utilizing rice-shaped bimetallic MOFs incorporated reduced graphene oxide, RSC Adv., 2025, 15(7), 5356–5368 RSC.
- S. Baumann, G. E. Eperon, A. Virtuani, Q. Jeangros, D. B. Kern, D. Barrit, J. Schall, W. Nie, G. Oreski and M. Khenkin, Stability and reliability of perovskite containing solar cells and modules: degradation mechanisms and mitigation strategies, Energy Environ. Sci., 2024, 17(20), 7566–7599 RSC.
- Y. Wang, D. Sun, B. Zhao, T. Zhu, C. Liu, Z. Xu, T. Zhou and C. Xu, Data-Driven Perovskite Design via High-Throughput Simulation and Machine Learning, Processes, 2025, 13(10), 3049 CrossRef CAS.
- A. Li and C. Yi, From Data-Driven to Intelligence-Driven: Innovating Research Paradigms of Perovskite Solar Cells, Sol. RRL, 2025, 9(21), e202500681 CrossRef.
- O. Grånäs, D. Vinichenko and E. Kaxiras, Establishing the limits of efficiency of perovskite solar cells from first principles modeling, Sci. Rep., 2016, 6(1), 36108, DOI:10.1038/srep36108.
- Y. Rong, L. Liu, A. Mei, X. Li and H. Han, Beyond efficiency: the challenge of stability in mesoscopic perovskite solar cells, Adv. Energy Mater., 2015, 5(20), 1501066 CrossRef.
- S. Ma, G. Yuan, Y. Zhang, N. Yang, Y. Li and Q. Chen, Development of encapsulation strategies towards the commercialization of perovskite solar cells, Energy Environ. Sci., 2022, 15(1), 13–55 RSC.
- J. Stasiulionis, Life Cycle Assessment of Perovskite Solar Cells and Comparison to Silicon Solar Cells, 2015 Search PubMed.
- A. A. Khan, Life Cycle Assessment (LCA) of Future Perovskite Tandem Solar Cells, PhD thesis, Uni. Freiburg., 2020, https://elib.dlr.de/138075/1/Abeer%20Ali%20Khan_Life%20Cycle%20Assessment%20%28LCA%29%20of%20Future%20Perovskite%20Tandem%20Solar%20Cells_Final%20Version.pdf.
- S. Sareen and S. Shokrgozar, Desert geographies: solar energy governance for just transitions, Globalizations, 2022, 1–17 Search PubMed.
- G. Schileo and G. Grancini, Lead or no lead? Availability, toxicity, sustainability and environmental impact of lead-free perovskite solar cells, J. Mater. Chem., 2021, 9(1), 67–76 CAS.
- P. García-García, Ó. Carpintero and L. Buendía, Just transitions to renewables in mining areas: Local system dynamics, Renew. Sustain. Energy Rev., 2024, 189, 113934 CrossRef.
- J. Li, H.-L. Cao, W.-B. Jiao, Q. Wang, M. Wei, I. Cantone, J. Lü and A. Abate, Biological impact of lead from halide perovskites reveals the risk of introducing a safe threshold, Nat. Commun., 2020, 11(1), 310 CrossRef CAS PubMed.
- A. Sengupta, M. A. Afroz, B. Sharma, S. Choudhary, N. Pant, Y. Gulia, N. P. K. R. Pai, D. Angmo and S. Satapathi, Commercialization of perovskite solar cells: opportunities and challenges, Sustain. Energy Fuels, 2025, 9(15), 3999–4022 RSC.
- B. K. Sovacool, M. L. Barnacle, A. Smith and M. C. Brisbois, Towards improved solar energy justice: Exploring the complex inequities of household adoption of photovoltaic panels, Energy Policy, 2022, 164, 112868 CrossRef.
- B. Roose, E. M. Tennyson, G. Meheretu, A. Kassaw, S. A. Tilahun, L. Allen and S. D. Stranks, Local manufacturing of perovskite solar cells, a game-changer for low-and lower-middle income countries?, Energy Environ. Sci., 2022, 15(9), 3571–3582 RSC.
- K. Ukoba, K. O. Yoro, O. Eterigho-Ikelegbe, C. Ibegbulam and T.-C. Jen, Adaptation of solar energy in the Global South: Prospects, challenges and opportunities, Heliyon, 2024, 10(7), e28009 Search PubMed.
- A. Babayigit, A. Ethirajan, M. Muller and B. Conings, Toxicity of organometal halide perovskite solar cells, Nat. Mater., 2016, 15(3), 247–251, DOI:10.1038/nmat4572.
- N. Moody, S. Sesena, D. W. deQuilettes, B. D. Dou, R. Swartwout, J. T. Buchman, A. Johnson, U. Eze, R. Brenes and M. Johnston, Assessing the regulatory requirements of lead-based perovskite photovoltaics, Joule, 2020, 4(5), 970–974 CrossRef.
- U. E. P. Agency, in National Ambient Air Quality Standards (NAAQS), Environmental Protection Agency, Washington DC, 2012 Search PubMed.
- H. Hudcová, J. Vymazal and M. Rozkošný, Present restrictions of sewage sludge application in agriculture within the European Union, Soil Water Res., 2019, 14(2), 104–120 CrossRef.
- Z. Li, X. Wu, B. Li, S. Zhang, D. Gao, Y. Liu, X. Li, N. Zhang, X. Hu and C. Zhi, Sulfonated graphene aerogels enable safe-to-use flexible perovskite solar modules, Adv. Energy Mater., 2022, 12(5), 2103236 Search PubMed.
- C. L. P. Prevention, Recommended actions based on blood lead level. National Center for Environmental Health, Division of Emergency and Environmental Health Services, 2018 Search PubMed.
- V. Stratiotou Efstratiadis and N. Michailidis, Sustainable recovery, recycle of critical metals and rare earth elements from waste electric and electronic equipment (circuits, solar, wind) and their reusability in additive manufacturing applications: a review, Metals, 2022, 12(5), 794 Search PubMed.
- J. M. Kadro and A. Hagfeldt, The end-of-life of perovskite PV, Joule, 2017, 1(1), 29–46 CrossRef.
- A. K. M. Yahia and M. Shahjalal, Recent developments and challenges in fracture mechanics–based fatigue life prediction, ASRC Procedia: Global Perspectives in Science and Scholarship, 2025, vol. 1, pp. 1202–1237 Search PubMed.
- J. Zhang, N. Chang, C. Fagerholm, M. Qiu, L. Shuai, R. Egan and C. Yuan, Techno-economic and environmental sustainability of industrial-scale productions of perovskite solar cells, Renew. Sustain. Energy Rev., 2022, 158, 112146 CrossRef CAS.
| This journal is © The Royal Society of Chemistry 2026 |
