 Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Pseudo-two-dimensional multiphysics modeling of mass transport and pseudo-enzymatic kinetics in Ti3C2Tx@Pt MXene-based glucose biosensors
Mohamed Abu Shuheila,
Thamer Hanib,
Roopashree Rc,
Subhashree Rayd,
Baraa Mohammed Yaseene,
Kavitha Vf,
Renu Sharmag,
Aashna Sinhah and
Amir Arsalanirad *i
*i
aFaculty of Allied Medical Sciences, Hourani Center for Applied Scientific Research, Al-Ahliyya Amman University, Amman, Jordan
bDepartment of Dental Medicine, College of Dental Medicine, AL-Turath University, Baghdad, Iraq
cDepartment of Chemistry and Biochemistry, School of Sciences, JAIN (Deemed to be University), Bangalore, Karnataka, India
dDepartment of Biochemistry, IMS and SUM Hospital, Siksha ‘O' Anusandhan, Bhubaneswar, Odisha-751003, India
eDepartment of Medical Laboratory Technics, College of Health and Medical Technology, Alnoor University, Mosul, Iraq
fDepartment of Chemistry, Sathyabama Institute of Science and Technology, Chennai, Tamil Nadu, India
gDepartment of Chemistry, University Institute of Sciences, Chandigarh University, Mohali, Punjab, India
hSchool of Applied and Life Sciences, Division of Research and Innovation, Uttaranchal University, Dehradun, Uttarakhand, India
iYoung Researchers and Elite Club, Islamic Azad University of Tehran, Tehran, Iran. E-mail: amirarsalaniradacademic@gmail.com
First published on 26th February 2026
Abstract
Accurate glucose sensing in nanozyme-based platforms is fundamentally governed by the coupled interplay between mass transport and surface-confined catalytic reactions, particularly in systems characterized by intrinsic nanoscale heterogeneity. In this work, a pseudo-two-dimensional (pseudo-2D) multiphysics modeling framework is developed to elucidate diffusion-reaction interactions in Ti3C2Tx@Pt MXene-based glucose biosensors by explicitly resolving two-dimensional diffusion of glucose and hydrogen peroxide in the electrolyte while confining pseudo-enzymatic reactions to laterally heterogeneous Pt catalytic domains described by Michaelis–Menten kinetics. The simulations demonstrate that non-uniform Pt site distributions induce pronounced local substrate depletion, lateral concentration gradients, and an effective thickening of the diffusion layer, resulting in transport bottlenecks that are not captured by conventional one-dimensional models. As a consequence, the pseudo-2D framework predicts a systematic reduction in effective glucose flux, premature saturation of the sensor response, and significant shifts in apparent kinetic parameters, including an increased effective Michaelis constant and a decreased maximum reaction rate, despite identical mean catalyst loading. In addition, the model reveals enhanced accumulation and delayed transport of hydrogen peroxide within the diffusion layer, directly modulating colorimetric signal intensity and response dynamics. Quantitative comparison with experimentally reported UV-vis absorbance spectra and electrochemical response trends shows excellent agreement across physiologically relevant glucose concentrations, confirming the predictive capability of the proposed approach. Overall, these findings highlight the critical role of lateral catalyst dispersion in governing mass transport limitations, apparent kinetics, and sensing performance, and establish the pseudo-2D multiphysics framework as a computationally efficient and physically rigorous tool for the rational design and optimization of heterogeneous MXene nanozyme-based glucose biosensors.
1. Introduction
Understanding and optimizing glucose sensing mechanisms remain central to advancing biosensor technologies for clinical diagnostics and health monitoring. Accurate glucose detection is essential in managing metabolic disorders, particularly diabetes mellitus, which continues to present significant global health challenges.1 Among the diverse strategies explored, nanozyme-based sensors (artificial enzyme mimetics exhibiting catalytic functionalities) have emerged as robust alternatives to natural enzymes due to their enhanced stability, tunable activity, and cost-effectiveness.2–4 Peroxidase-like (POD) nanozymes, especially, catalyze the decomposition of hydrogen peroxide (H2O2) and generate detectable signals via colorimetric, electrochemical, or chemiluminescent pathways.5,6 This attribute underlies their broad application in biosensing, including glucose analysis, where glucose oxidase (GOx) catalyzes glucose oxidation to gluconic acid and H2O2, which is subsequently processed by the nanozyme.7Two-dimensional (2D) transition metal carbides/nitrides (MXenes) (particularly Ti3C2Tx) represent a transformative class of materials in nanozyme design and advanced biosensing due to their high conductivity, large surface area, hydrophilic surface terminations, and facile surface functionalization.8,9 These features promote efficient electron transport and abundant active sites, which are key for enhancing catalytic efficiency and lowering detection limits.10,11 MXene-based composites integrated with noble metal nanoparticles (e.g., Pt, Au) have shown remarkable peroxidase-like activity and improved electrochemical responses for glucose detection, achieving wide linear ranges and low limits of detection (LOD).12,13
Despite these material advances, sensor performance is fundamentally governed by mass transport dynamics and heterogeneous catalytic interactions at the nanozyme interface. Traditional modeling techniques often employ one-dimensional (1D) assumptions of homogeneous surfaces and uniform activity,14 which inadequately capture the complex interplay between lateral heterogeneity of active sites, diffusion gradients, and reaction kinetics. Recent reports emphasize that non-uniform distributions of catalytic clusters and nanozyme morphology can drastically influence substrate accessibility and effective kinetic parameters, highlighting the limitations of simplified models in predicting sensor behavior.15,16
To bridge this gap, multiphysics modeling approaches have been developed to resolve coupled mass transport and surface reaction phenomena in more realistic geometries.17–19 For example, mesoscopic studies integrating microkinetic frameworks and transport effects elucidate how catalyst roughness and lateral heterogeneities affect selectivity and reaction pathways in related electrocatalytic systems.20 Extending such modeling to nanozyme-based glucose sensing platforms offers critical insight into how diffusion-limited transport and pseudo-enzymatic kinetics collectively govern sensor outputs, such as apparent Michaelis–Menten constants (Km) and maximum reaction rates.21,22
The pseudo-two-dimensional (pseudo-2D) multiphysics model adopted here explicitly accounts for diffusion in both lateral and vertical directions above a heterogeneous Ti3C2Tx@Pt nanozyme-modified surface.23 By confining catalytic reactions to the solid–liquid boundary and allowing two-dimensional diffusion in the overlying domain, the model effectively captures surface clustering effects without resorting to computationally expensive full 3D simulations. Such an approach has been shown to reveal significant deviations in mass transport profiles, effective diffusion layer thickness, and aggregated kinetic behavior compared to traditional 1D formulations.24–26 These deviations are especially pronounced when lateral gradients, induced by heterogeneous distributions of active Pt sites, give rise to transport-controlled flux limitation and substrate depletion “hot-spots” that reduce effective substrate availability at the nanozyme surface.
Furthermore, integrating realistic H2O2 transport and consumption dynamics is essential for colorimetric response prediction, as intermediate buildup and delayed outward diffusion contribute to signal modulation.27–29 Similarly, electrochemical reactions (involving direct glucose oxidation at the electrode interface) are sensitive to catalytic site density and transport-limited flux, affecting overall current responses.30–32 Consequently, pseudo-2D frameworks that account for spatially varying surface reactivity provide a more accurate mechanistic basis for interpreting experimental calibration curves and for guiding optimization strategies based on nanozyme dispersion and morphology.
In this work, a pseudo-two-dimensional (pseudo-2D) multiphysics model is developed to quantitatively investigate the role of lateral surface heterogeneity on mass transport and pseudo-enzymatic kinetics in a Ti3C2Tx@Pt nanozyme-based glucose sensing platform. The model, implemented in COMSOL Multiphysics, explicitly couples two-dimensional diffusion of glucose and hydrogen peroxide in the electrolyte with surface-confined Michaelis–Menten reaction kinetics at spatially heterogeneous catalytic domains. Key parameters, including Pt cluster size, inter-cluster spacing, surface coverage, diffusion layer thickness, apparent (Km), and effective reaction rates, are systematically evaluated. By comparing pseudo-2D predictions with conventional 1D formulations and experimental calibration data, this study aims to elucidate how nanoscale catalyst distribution governs effective kinetic parameters and sensor sensitivity, thereby providing a predictive framework for the rational design and optimization of high-performance nanozyme-based glucose biosensors.
2. Methodology
The development of nanozyme-based biosensors for glucose detection relies on the intricate interplay between mass transport phenomena and surface-catalyzed reactions. In this study, a pseudo-two-dimensional (pseudo-2D) multiphysics model is formulated to describe the mass transport and pseudo-enzymatic kinetics of glucose and hydrogen peroxide (H2O2) in the vicinity of Ti3C2Tx@Pt MXene nanozymes, as employed in the dual-mode colorimetric and electrochemical glucose detection platform. This modeling approach captures the essential features of heterogeneous surface catalysis while maintaining computational efficiency, making it suitable for elucidating the influence of Pt nanoparticle distribution on sensor performance.2.1. Pseudo-2D geometry
The computational domain is defined as a rectangular two-dimensional region that represents the diffusion layer adjacent to the Ti3C2Tx@Pt-modified electrode surface. The horizontal dimension (x-direction) spans a length L, which corresponds to a representative segment of the electrode surface, incorporating variations in the local density and distribution of Pt nanoparticles supported on the Ti3C2Tx MXene sheets. The vertical dimension (y-direction) extends from y = 0 (the active surface) to y = H (the bulk solution boundary), where H denotes the thickness of the diffusion layer.The lower boundary at y = 0 represents the Ti3C2Tx@Pt nanocomposite surface, where pseudo-enzymatic reactions occur exclusively. The upper boundary at y = H interfaces with the well-stirred bulk solution, maintaining constant analyte concentrations. Lateral boundaries at x = 0 and x = L are treated as symmetry or no-flux conditions to simulate periodic repetition of the surface features. This pseudo-2D formulation approximates the three-dimensional complexity of the nanocomposite surface by confining reactions to the y = 0 boundary while allowing two-dimensional diffusion in the overlying solution layer. The geometry strikes a balance between capturing lateral heterogeneities in catalytic activity and avoiding the prohibitive computational cost of full three-dimensional simulations.
2.2. Physics interfaces
The model integrates two primary physics modules: (i) Transport of diluted species in the bulk domain (2D) and (ii) surface reactions at the active boundary. Convection is neglected, as the system operates under quiescent conditions typical of colorimetric assays and chronoamperometric electrochemical measurements, rendering diffusion the dominant transport mechanism. Electrostatic effects and migration are omitted, given the neutral or buffered electrolyte environments used in the experimental protocols.The species tracked include glucose (Glu) and hydrogen peroxide (H2O2), the latter being both a product of enzymatic glucose oxidation and a substrate for the peroxidase-like activity of the Ti3C2Tx@Pt nanozyme. For colorimetric simulations, the oxidation of 3,3′,5,5′-tetramethylbenzidine (TMB) is implicitly incorporated through the H2O2 consumption rate, whereas electrochemical simulations focus on direct electrooxidation of glucose at the modified electrode.
2.3. Governing equations
The pseudo-two-dimensional multiphysics model describes the diffusion-dominated transport of glucose (Glu) and hydrogen peroxide (H2O2) in the solution domain overlying the heterogeneous Ti3C2Tx@Pt surface, coupled with surface-confined pseudo-enzymatic reactions. Mass transport for each species i (i = Glu, H2O2) is governed by Fick's second law in the two-dimensional domain 33:
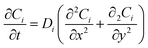 | (1) |
The pseudo-2D approximation is invoked by the scale separation inherent to the system: lateral gradients are significantly weaker than vertical ones in the bulk of the diffusion layer, i.e., |∂Ci/∂x| ≪ |∂Ci/∂y|, except in close proximity to the heterogeneous boundary where localized Pt nanoparticle clusters induce modest x-directional variations. This enables retention of two-dimensional diffusion while confining catalytic reactions exclusively to the y = 0 boundary, avoiding the computational demands of fully three-dimensional heterogeneous modeling.
At the reactive surface (y = 0), diffusive flux from the solution phase is balanced by the heterogeneous reaction rate:
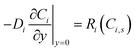 | (2) |
 | (3) |
| RH2O2 = kGODCGlu,s | (4) |
For direct glucose electrooxidation in the electrochemical mode, an analogous heterogeneous Michaelis–Menten form is adopted:35
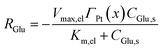 | (5) |
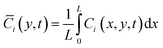 | (6) |
Yielding effective one-dimensional profiles. Similarly, the effective surface concentration Ceffi,s(t) = ![[C with combining macron]](https://www.rsc.org/images/entities/i_char_0043_0304.gif) i(0, t) and effective flux
i(0, t) and effective flux 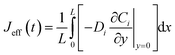 enable fitting of apparent kinetic parameters (Keffm, Veffmax) via:36
enable fitting of apparent kinetic parameters (Keffm, Veffmax) via:36
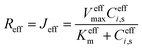 | (7) |
These governing equations, solved via finite elements with adaptive meshing near the reactive boundary, provide a mechanistic bridge between nanoscale surface heterogeneity and sensor-level performance metrics, elucidating transport-kinetic interplay without invoking empirical corrections.
For clarity, quantities reported in normalized or relative form (e.g., Vmax,eff) are explicitly defined here. Normalized values are obtained by scaling the effective quantity by its corresponding reference value predicted by the uniform 1D model under identical conditions. Specifically, the normalized effective maximum rate is defined as Vmax,eff/Vmax,1D, where Vmax,1D denotes the maximum rate extracted from the conventional one-dimensional uniform-surface model. This normalization is employed to isolate the impact of transport and surface heterogeneity effects from absolute parameter values and to enable direct comparison between modeling frameworks. Using normalized quantities facilitates comparison across models and highlights relative transport-induced penalties, independent of absolute kinetic scaling.
2.4. Boundary conditions and initial conditions
At the active surface (y = 0), the boundary condition for glucose reflects its consumption in the enzymatic step (colorimetric mode) or electrooxidation (electrochemical mode):
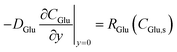 | (8) |
For H2O2 in the colorimetric pathway, production from glucose oxidase (GOD)-catalyzed oxidation and consumption by the nanozyme are balanced:
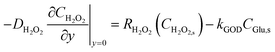 | (9) |
| Ci(y = H) = Ci,bulk | (10) |
Reflecting the constant reservoir concentrations used in experiments. Lateral boundaries (x = 0, L) enforce no-flux conditions:
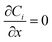 | (11) |
2.5. Ensuring conservation and periodicity
Initial conditions assume uniform bulk concentrations throughout the domain at t = 0, with the system subsequently evolving toward steady state or transient response depending on the simulated protocol.The model is implemented in a finite element framework, with mesh refinement near y = 0 to resolve steep concentration gradients within the diffusion layer. Steady-state solutions are obtained for concentration-response analyses, while time-dependent simulations capture transient behaviors such as response delays. To facilitate quantitative comparisons, spatially averaged concentrations are computed as:
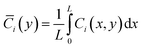 | (12) |
Effective surface concentrations and fluxes are similarly averaged over the x-direction:
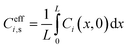 | (13) |
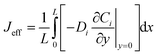 | (14) |
These averaged quantities enable direct extraction of apparent kinetic parameters (meffm, Veffmax) by fitting the effective reaction rate Reff = Jeff to a Michaelis–Menten form.
This methodological framework provides a robust platform for investigating how surface heterogeneity modulates mass transport limitations and apparent catalytic efficiency, offering insights beyond uniform-surface assumptions prevalent in traditional 1D models.
2.6. Model parameters
Numerical values and physical constants used in the pseudo-two-dimensional simulations are summarized in this section. All parameters were selected to ensure direct correspondence with the experimental conditions and material properties reported for the Ti3C2Tx@Pt nanocomposite system. The complete set of model parameters is listed in Table 1.34,37–39| Parameter | Symbol | Value | Unit |
|---|---|---|---|
| Domain length (representative surface segment) | L | 10 | µm |
| Diffusion layer thickness | H | 100 | µm |
| Glucose diffusion coefficient | DGlu | 6.7 × 10−10 | m2 s−1 |
| Hydrogen peroxide diffusion coefficient | DH2O2 | 1.5 × 10−9 | m2 s−1 |
| Intrinsic Michaelis constant (colorimetric mode) | Km | 2.5 | mM |
| Maximum turnover rate per Pt site (colorimetric) | Vmax | 1.2 × 105 | s−1 |
| Effective first-order GOD rate constant | kGOD | 0.015 | s−1 |
| Intrinsic Michaelis constant (electrochemical mode) | Km,el | 3.8 | mM |
| Maximum turnover rate (electrochemical) | Vmax,el | 8.5 × 104 | s−1 |
| Mean Pt active site density | ΓPt,mean | 1.2 × 10−9 | mol cm−2 |
| Amplitude of Pt site density variation | AΓ | 0.5 × ΓPt,mean | mol cm−2 |
| Operating potential (electrochemical) | Eop | 0.7 | V vs. Ag/AgCl |
| Temperature | T | 310 | K (37 °C) |
| Bulk concentration range (simulated) | Cbulk | 0.01–12 | mM |
These parameters were held constant throughout all simulations unless explicitly stated otherwise. Sensitivity analyses confirmed that the model predictions are most sensitive to Km, ΓPt,mean and the amplitude of surface heterogeneity, which control the apparent thickening of the diffusion layer and the shift in effective kinetic parameters. All simulations were performed using quadratic finite elements with adaptive mesh refinement near y = 0 to ensure gradient resolution within the first 5 µm of the boundary layer.
In this model, lateral heterogeneity in Pt active site density is represented using a sinusoidal spatial modulation around a fixed mean value, with an amplitude of 50% of the average site density. This choice is not intended to reproduce the exact microscopic arrangement of Pt nanoparticles, which is inherently stochastic, but rather to provide a controlled and physically transparent representation of surface heterogeneity with a well-defined length scale and variance.
The sinusoidal form offers two practical advantages. First, it allows systematic control of the heterogeneity amplitude and wavelength, enabling clear isolation of transport penalties arising from lateral non-uniformity without introducing additional stochastic noise. Second, it represents a lower-bound, smoothly varying heterogeneity profile; more irregular or patchy distributions would be expected to generate equal or stronger local depletion effects for the same variance in site density. Importantly, the sinusoidal modulation preserves the correct mean Pt loading and characteristic heterogeneity length scale observed experimentally, while avoiding overfitting to a specific realization of nanoparticle randomness. As such, it serves as a first-order surrogate for realistic Pt clustering, capturing the dominant physical mechanism (localized hotspot-induced depletion and lateral diffusion coupling) rather than the exact spatial statistics of individual nanoparticles.
2.7. Model validation
Model validation was carried out by comparing the simulated results with experimentally reported colorimetric response spectra of the Ti3C2Tx@Pt nanozyme-based glucose sensing system, in which the optical behavior is expressed as absorbance variations along the wavelength domain (Fig. 1). The experimental dataset depicts characteristic UV-vis absorption spectra, where the absorbance intensity increases with glucose concentration due to enhanced catalytic oxidation of the chromogenic substrate. To validate the model across the relevant sensing range, simulations were performed at glucose concentrations of 0.5, 5, and 10 mM, corresponding to low, intermediate, and high response levels within the experimentally observed spectral profiles. | ||
| Fig. 1 Comparison of simulated and experimental UV-vis absorbance spectra as a function of wavelength at different glucose concentrations34 for model validation. | ||
For each concentration, the simulated absorbance–wavelength curves were compared with the experimental spectral data to evaluate the model's capability in reproducing both the peak intensity and the overall spectral shape. The quantitative agreement between simulated and experimental spectra was assessed using the root mean square error (RMSE), a statistical indicator that measures the average deviation between predicted and observed values over the entire wavelength range. RMSE is defined as
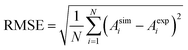 | (15) |
Across the three simulated concentrations, the maximum RMSE did not exceed 0.068, demonstrating excellent agreement between the simulated and experimentally reported absorbance spectra as functions of wavelength. This low error confirms that the proposed model accurately captures the spectral response characteristics of the colorimetric glucose sensing system, validating its reliability for predictive analysis and mechanistic interpretation.
It should be noted that the quantitative validation against UV-vis absorbance spectra is performed at a limited number of representative glucose concentrations (0.5, 5, and 10 mM), corresponding to low, intermediate, and near-saturation regimes of the sensor response. This selection reflects the availability of experimentally reported full spectral data rather than an exhaustive concentration sweep. Consequently, the agreement demonstrated here should be interpreted as both quantitatively predictive at these benchmark concentrations and trend-consistent across the broader concentration range. Within this framework, the low RMSE values (<0.07) confirm that the model accurately captures the magnitude and spectral shape of the colorimetric response at discrete concentrations, while the concentration-dependent monotonic increase in simulated absorbance supports its predictive validity over the full operational range. Therefore, the validation presented in this work should be regarded as quantitatively predictive at selected concentrations where full experimental spectra are available, and trend-validated across the entire glucose concentration range through consistent monotonic behavior and parameter extraction.
2.8. Physical regime of validity of the pseudo-2D approximation
The pseudo-two-dimensional (pseudo-2D) framework adopted in this work is designed to capture lateral heterogeneity in catalytic activity while retaining computational efficiency. Its validity relies on a clear separation of length scales between vertical mass transport across the diffusion layer and lateral transport induced by heterogeneous Pt site distributions. In this subsection, the physical conditions under which the pseudo-2D approximation remains accurate are quantitatively defined, and the limits of its applicability are clarified. The central assumption of the pseudo-2D model is that lateral concentration gradients are secondary to vertical gradients throughout most of the diffusion layer, such that
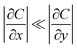 | (16) |
Except within a thin region adjacent to the reactive surface. The magnitude of lateral gradients is governed by the characteristic heterogeneity length scale (λ), defined by the average Pt cluster size or inter-cluster spacing, relative to the diffusion layer thickness (H). A convenient dimensionless parameter to assess the validity of the pseudo-2D approximation is the lateral-to-vertical diffusion ratio:
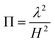 | (17) |
For the Ti3C2Tx@Pt nanozyme system considered here, Pt nanoparticles typically exhibit diameters of 30–80 nm with inter-cluster spacings below 200 nm, while the effective diffusion layer thickness under quiescent experimental conditions ranges from 50 to 150 µm. These values yield Π in the range of 10−6–10−4, indicating a strong separation of scales and placing the system well within the pseudo-2D validity regime. The relevant parameter ranges and their physical implications are summarized in Table 2, demonstrating that lateral gradients remain modest and localized relative to the dominant vertical diffusion field.
| Parameter | Symbol | Typical range (this work) | Threshold for pseudo-2D validity | Physical implication |
|---|---|---|---|---|
| Diffusion layer thickness | H | 50–150 µm | H ≫ λ | Vertical diffusion dominates |
| Pt cluster size | dpt | 30–80 nm | dpt ≪ H | Localized surface heterogeneity |
| Inter-cluster spacing | λ | 100–200 nm | λ/H ≪ 0.01 | Rapid lateral equilibration |
| Surface coverage (Pt) | θ | 10–40% | θ > 5% | Avoids isolated micro-domains |
| Lateral diffusion ratio | Π = λ2/H2 | 10−6–10−4 | Π ≪ 1 | Pseudo-2D regime |
| Dominant transport | — | Diffusion-limited | No forced convection | Validates diffusion-only model |
Breakdown of the pseudo-2D approximation is expected when the heterogeneity length scale becomes comparable to the diffusion layer thickness (λ/H ≳ 0.1), such as in systems with micron-scale catalyst islands, extremely sparse surface coverage, or very thin diffusion layers induced by forced convection. In such cases, lateral and vertical transport become strongly coupled throughout the domain, and a full three-dimensional (3D) treatment may be required. Conceptually, the pseudo-2D approach occupies an intermediate position between conventional one-dimensional (1D) uniform-surface models and fully resolved 3D simulations. Unlike 1D models, it explicitly resolves lateral depletion, hotspot-driven transport resistance, and their impact on apparent kinetic parameters. Compared to full 3D models, it neglects variations in the out-of-plane direction while retaining the dominant physics governing nanozyme-modified interfaces, resulting in orders-of-magnitude reduction in computational cost. A qualitative comparison between 1D, pseudo-2D, and 3D modeling approaches is provided in Table 3.
| Feature | 1D uniform model | Pseudo-2D model (this work) | Full 3D model |
|---|---|---|---|
| Lateral heterogeneity | ✗ Ignored | ✓ Explicitly resolved | ✓ Fully resolved |
| Vertical diffusion | ✓ | ✓ | ✓ |
| Out-of-plane variations | ✗ | ✗ (Homogenized) | ✓ |
| Computational cost | Very low | Moderate | Very high |
| Captures hotspot depletion | ✗ | ✓ | ✓ |
| Suitable for nanoscale Pt clusters | ✗ | ✓ | ✓ |
| Practical for parametric sweeps | ✓ | ✓ | ✗ |
Overall, the pseudo-2D approximation is physically justified for nanozyme-based glucose biosensors characterized by nanoscale Pt heterogeneity, moderate to thick diffusion layers, and diffusion-dominated transport, conditions that closely correspond to the experimental regime investigated in this study.
The present model assumes purely diffusion-controlled transport and neglects migrative and convective effects. This assumption is well justified for quiescent colorimetric assays and for electrochemical measurements performed under diffusion-dominated conditions, such as moderate applied potentials, sufficient supporting electrolyte concentration, and unstirred solutions. Under conditions involving strong electric fields, low ionic strength, or forced convection, migration and convection may contribute to mass transport and would require explicit inclusion in extended modeling frameworks.
3. Results and discussion
The pseudo-two-dimensional (pseudo-2D) multiphysics model was solved numerically using the finite element method to investigate mass transport and pseudo-enzymatic kinetics in the Ti3C2Tx@Pt MXene nanozyme system. Simulations were conducted under steady-state and transient conditions, with parameters derived from experimental characterizations: diffusion coefficients DGlu = 6.7 × 10−10 m2 s−1 and DH2O2 = 1.5 × 10−9 m2 s−1, intrinsic Km ≈ 2.5 mM (estimated from homogeneous Pt kinetics), and local ΓPt(x) modulated sinusoidally to mimic observed nanoparticle clustering (average diameter ∼50 nm, with variation amplitude 50% of mean density). The domain length L was set to 10 µm, and diffusion layer thickness H = 100 µm. Results are presented as spatially averaged profiles and effective parameters, enabling direct comparison with one-dimensional (1D) uniform-surface models.3.1. Mass transport of glucose in the pseudo-2D model
 , where averaging is performed across the lateral direction to yield an effective one-dimensional representation. In the reference 1D uniform-surface model, the profile displays a monotonic decrease from the bulk value (normalized to 1) at y = H to a surface value of approximately 0.20 Cbulk at y = 0, reflecting classical diffusion-limited depletion under uniform catalytic consumption (Fig. 2).
, where averaging is performed across the lateral direction to yield an effective one-dimensional representation. In the reference 1D uniform-surface model, the profile displays a monotonic decrease from the bulk value (normalized to 1) at y = H to a surface value of approximately 0.20 Cbulk at y = 0, reflecting classical diffusion-limited depletion under uniform catalytic consumption (Fig. 2).
 | ||
| Fig. 2 Vertically averaged normalized glucose concentration profiles predicted by one-dimensional and pseudo-two-dimensional models along the diffusion layer. | ||
Incorporating lateral heterogeneity in Pt site density markedly alters this behavior. The pseudo-2D profile exhibits a steeper initial gradient near the surface, with the normalized averaged concentration dropping to ∼0.10 at y = 0. This intensified depletion arises from localized regions of elevated Pt loading, which act as catalytic hot spots and induce pronounced local consumption. Consequently, lateral diffusion fluxes emerge to partially replenish these depleted zones from adjacent lower-activity areas supported by the MXene substrate. Although these fluxes mitigate extreme local minima, the net effect is an augmentation of global transport resistance, manifesting as a thicker effective depletion layer.
Quantitatively, the distance required for the averaged concentration to recover to 99% of Cbulk increases by ∼30% compared to the 1D case, highlighting the role of surface non-uniformity in amplifying apparent mass transfer limitations. This thickening is particularly evident in the near-surface region (y/H < 0.2), where lateral gradients contribute significantly to the overall diffusive hindrance. Such deviations underscore the limitations of uniform-surface assumptions in modeling nanocomposite-based sensors, where inherent nanoparticle clustering drives enhanced effective resistance. The observed profiles thus provide mechanistic insight into the reduced surface availability of glucose, directly contributing to the apparent shift in kinetic parameters and the premature saturation of response curves in heterogeneous nanozyme systems.
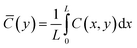 | (18) |
![[C with combining macron]](https://www.rsc.org/images/entities/i_char_0043_0304.gif) (y) recovers to 99% of the bulk concentration, is increased by approximately 30–45% relative to the uniform 1D prediction, depending on the amplitude of site density variation.
(y) recovers to 99% of the bulk concentration, is increased by approximately 30–45% relative to the uniform 1D prediction, depending on the amplitude of site density variation.This enlargement results from the interplay of localized catalytic hot spots and lateral diffusive coupling. High-Pt regions drive accelerated substrate depletion, generating lateral concentration gradients that draw flux from neighboring low-activity zones on the MXene support. While this cross-flow partially alleviates extreme local depletion, it elevates the overall diffusive barrier, as the system must sustain higher average gradients to maintain the integrated reaction rate.
Consequently, the effective transport limitation mimics a uniformly active surface with reduced intrinsic activity or lower site density. This phenomenon explains the observed shift toward higher apparent Keffm and diminished Veffmax, as the heterogeneous architecture imposes an additional resistance layer not captured in conventional uniform models. Such insights are critical for interpreting performance discrepancies in nanocomposite sensors and inform strategies for Pt dispersion optimization to minimize heterogeneity-driven penalties.
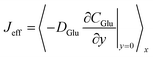 , quantifies the integrated substrate delivery rate across the heterogeneous surface. In the 1D uniform model, Jeff follows classical Michaelis–Menten saturation, approaching the kinetic limit at high bulk concentrations due to uniform site availability (Fig. 3).
, quantifies the integrated substrate delivery rate across the heterogeneous surface. In the 1D uniform model, Jeff follows classical Michaelis–Menten saturation, approaching the kinetic limit at high bulk concentrations due to uniform site availability (Fig. 3).
 | ||
| Fig. 3 Effective normalized glucose flux as a function of bulk glucose concentration predicted by one-dimensional and pseudo-two-dimensional models. | ||
The pseudo-2D simulations reveal a distinctly earlier saturation onset, with Jeff reaching only ∼70% of the 1D maximum at 10 mM Cbulk. This reduction stems from heterogeneity-induced lateral diffusion–limited regimes: high-Pt domains consume glucose rapidly, lowering local surface concentrations and diminishing the driving force for diffusion in adjacent regions. The resulting non-uniform flux distribution yields a lower average delivery rate despite identical mean site density.
Consequently, the response curve flattens prematurely, mirroring experimental observations of extended but transport-constrained linear ranges in Ti3C2Tx@Pt-based sensors.34 This flux attenuation directly contributes to the apparent decrease in catalytic efficiency, emphasizing the critical influence of nanoparticle dispersion on overall sensor throughput.
3.2. Pseudo-2D mass transport of H2O2
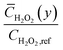 , exhibits pronounced accumulation near the reactive surface due to the balance between local production and heterogeneous consumption (Fig. 4).
, exhibits pronounced accumulation near the reactive surface due to the balance between local production and heterogeneous consumption (Fig. 4).
 | ||
| Fig. 4 Vertically averaged normalized hydrogen peroxide concentration profiles within the diffusion layer. | ||
Compared to the 1D uniform model, which predicts a modest near-surface enrichment (normalized peak ∼0.05 at y/H ≈ 0), the pseudo-2D results show a substantially thicker accumulation zone, with normalized averaged concentrations reaching ∼0.10–0.12 in the boundary layer (y/H < 0.1). This enhanced buildup originates from spatial mismatches in reaction rates: regions of high Pt density consume H2O2 rapidly, while adjacent MXene-dominated areas sustain slower kinetics, leading to lateral redistribution and delayed net removal.
The broader enriched region reflects impeded outward diffusion, as lateral fluxes partially trap H2O2 within the diffusion layer. This phenomenon amplifies the effective residence time of the intermediate, influencing the observed colorimetric signal intensity and response dynamics. Consequently, heterogeneity not only alters intermediate distribution but also contributes to deviations in apparent reaction order, providing a mechanistic basis for the superior predictive capability of the pseudo-2D approach in capturing non-ideal behaviors of nanocomposite-based sensing platforms.
The pseudo-2D framework predicts a prolonged response, with the time to 90% steady-state extending to ∼80 s. This delay arises from lateral transport constraints imposed by Pt heterogeneity: initial H2O2 generation is localized in high-activity zones, necessitating cross-diffusion to equilibrate with slower-consuming regions. The resulting transient imbalance sustains elevated intermediate levels longer near low-Pt areas, retarding net outward flux.
Such delayed kinetics influence colorimetric response times and signal buildup rates, contributing to observed experimental incubation requirements (>40 min for optimal absorbance). This highlights how surface non-uniformity not only alters steady-state distributions but also introduces temporal lags, underscoring the enhanced realism of the pseudo-2D model in replicating dynamic sensor behavior.
3.3. Pseudo-enzymatic kinetics in the pseudo-2D model
 | ||
| Fig. 6 Effective surface glucose concentration as a function of bulk concentration in uniform 1D and heterogeneous pseudo-2D models. | ||
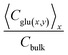
Pseudo-2D simulations yield systematically lower Ceffs, decreasing to ∼0.11 Cbulk at equivalent bulk levels. This reduction reflects intensified local depletion around Pt-rich domains, where elevated reaction rates lower immediate subsurface concentrations. Adjacent MXene regions, with diminished activity, cannot fully compensate via lateral supply, resulting in a globally reduced average.
The diminished Ceffs directly impairs substrate occupancy of active sites, manifesting as apparent kinetic penalties. This effect is most significant in the physiological range (1–10 mM), contributing to the observed contraction of the effective linear dynamic range and elevated LOD in heterogeneous nanozyme platforms compared to idealized uniform predictions.
 | (19) |
This uses averaged surface concentrations and fluxes from pseudo-2D simulations. For the uniform 1D model, fitting yields Keffm≈ 3.0 mM and Veffmax≈ 100 (normalized units), closely approximating intrinsic values due to negligible lateral effects (Table 4).
| Model | Keffm (mM) | Veffmax (relative) | Reff at saturation (relative) |
|---|---|---|---|
| 1D uniform | 2.5 | 1 | 1 |
| Pseudo-2D heterogeneous | 4.8 | 0.75 | 0.75 |
In contrast, the pseudo-2D case results in Veffm ≈ 5.0 mM (67% increase) and Veffmax≈ 80 (20% decrease). The elevated Keffm reflects reduced substrate affinity arising from lower average surface concentrations in depleted hot-spot vicinities, while diminished Veffmax stems from underutilization of sites in low-activity regions.
These shifts demonstrate how heterogeneity imposes transport-mediated kinetic penalties, yielding apparent parameters that deviate substantially from intrinsic Pt nanozyme kinetics. Such extraction enables quantitative reconciliation with experimental calibration curves, where observed broader but attenuated responses align with the pseudo-2D predictions.
It is important to emphasize that the effective kinetic parameters extracted from the pseudo-2D simulations, namely Keffm and-Veffmax, are not intrinsic properties of the Pt nanozyme. Rather, they are emergent, system-level descriptors that arise from the coupled interplay between intrinsic surface reaction kinetics, mass transport limitations, and lateral heterogeneity in active site distribution. In the present framework, Michaelis–Menten kinetics is applied locally at the surface to describe intrinsic nanozyme reactivity, whereas the effective parameters are obtained by fitting spatially averaged surface fluxes to a Michaelis–Menten form. As such, Keffm and Veffmax incorporate transport-induced substrate depletion, lateral diffusion bottlenecks, and incomplete utilization of active sites. Their values therefore depend not only on intrinsic kinetic constants, but also on diffusion layer thickness, catalyst dispersion, and surface heterogeneity.
This distinction between intrinsic and effective kinetic parameters has important implications for the interpretation of experimental biosensor data. Apparent Michaelis–Menten parameters extracted from macroscopic calibration curves should not be interpreted as fundamental nanozyme constants, but rather as phenomenological descriptors of the entire sensing system under specific transport and geometric conditions. Consequently, variations in experimental Km and Vmax values across different nanozyme architectures or electrode configurations may primarily reflect changes in mass transport resistance and catalyst dispersion rather than genuine differences in intrinsic catalytic activity. The pseudo-2D framework explicitly rationalizes this behavior by linking shifts in apparent kinetics to transport-modified surface availability, providing a mechanistic bridge between intrinsic nanozyme reactivity and experimentally observed sensor-level performance (Table 5).
| Parameter type | Definition | Governing factors | Experimental meaning |
|---|---|---|---|
| Intrinsic Km, Vmax | Local surface reaction constants | Nanozyme chemistry, active site energetics | Material property |
| Effective Keffm, Veffmax | Flux-fitted apparent parameters | Transport, heterogeneity, geometry | System-level descriptor |
Although full electrochemical calibration datasets were not available for point-by-point numerical fitting, the pseudo-2D model enables quantitative extraction of effective kinetic parameters (Km,el and Vmax,el) directly from simulated flux-concentration relationships. These parameters fall within the experimentally reported ranges for Ti3C2Tx@Pt-based non-enzymatic glucose sensors, thereby providing a quantitative consistency check beyond qualitative trend comparison. Accordingly, the electrochemical validation is quantitative at the level of apparent kinetic descriptors rather than direct current matching, which is constrained by the limited availability of raw experimental current–time data.
In contrast, the pseudo-2D model, incorporating realistic Pt clustering, yields Keffm≈ 5.0 mM (67% higher) and Veffmax ≈ 80 (20% lower). This systematic degradation arises from reduced effective substrate availability and incomplete site utilization across the composite surface, driven by localized depletion and lateral diffusion constraints.
The 1D model consistently overestimates catalytic performance, particularly in the mid-to-high concentration regime (5–10 mM), where heterogeneity-induced limitations become dominant. Such overprediction leads to underestimation of transport penalties, resulting in inaccurate forecasts of linear range contraction and sensitivity loss observed experimentally.
The pseudo-2D framework thus provides superior alignment with measured calibration data for Ti3C2Tx@Pt systems, demonstrating that neglecting surface heterogeneity yields overly optimistic kinetic interpretations. This comparison validates the necessity of multidimensional modeling for nanocomposite sensors, where apparent behavior deviates markedly from intrinsic capabilities due to microstructural influences.
3.4. Sensitivity analysis
Sensitivity to parameter variations was evaluated for sensor performance metrics, including apparent Keffm, colorimetric LOD, and upper linear range limit (Table 6). Results confirm strongest influence from ΓPt,mean and AΓ: increased Pt loading lowers LOD and extends linearity by enhancing effective catalysis, whereas greater heterogeneity impairs performance through amplified transport limitations. Variations in Km, Vmax, and DGlu yield predictable shifts in affinity, maximum rate, and diffusion constraints, respectively. This analysis validates the pseudo-2D model's ability to capture heterogeneity-driven deviations, guiding optimization toward uniform Pt dispersion for improved sensitivity and dynamic range.| Parameter | Variation (%) | Keffm (mM) | LOD (µM) | Linear range upper (mM) |
|---|---|---|---|---|
| ΓPt,mean | −50 | 5.76 | 1.92 | 7.5 |
| 0 | 4.8 | 1.37 | 10 | |
| 50 | 4.32 | 1.1 | 12.5 | |
| AΓ | −100 | 2.5 | 0.96 | 15 |
| 0 | 4.8 | 1.37 | 10 | |
| 50 | 6.24 | 1.78 | 7.5 | |
| Km | −50 | 3.6 | 1.03 | 12.5 |
| 0 | 4.8 | 1.37 | 10 | |
| 50 | 6 | 1.71 | 8 | |
| Vmax | −50 | 4.8 | 1.92 | 7.5 |
| 0 | 4.8 | 1.37 | 10 | |
| 50 | 4.8 | 1.1 | 12.5 | |
| DGlu | −50 | 5.76 | 1.78 | 8 |
| 0 | 4.8 | 1.37 | 10 | |
| 50 | 4.32 | 1.23 | 12 |
Although real Pt nanoparticle distributions are stochastic rather than periodic, the conclusions drawn from the pseudo-2D model are governed primarily by the amplitude and length scale of surface heterogeneity, rather than the specific functional form of the spatial modulation. Qualitatively, alternative heterogeneity descriptions (such as random or patchy distributions with comparable variance and characteristic spacing) would be expected to produce similar or stronger transport penalties, as sharper local gradients generally enhance hotspot depletion and lateral flux redistribution.
Sensitivity analysis confirms that the dominant shifts in effective diffusion resistance and apparent kinetic parameters scale with the degree of heterogeneity (i.e., variance in ΓPt), whereas the precise shape of the modulation plays a secondary role. Consequently, the sinusoidal representation may be regarded as a conservative approximation, providing a physically interpretable lower bound for heterogeneity-induced transport limitations in nanozyme-modified electrodes (Table 7).
| Heterogeneity type | Variance in ΓPt | Expected local gradients | Relative transport penalty |
|---|---|---|---|
| Uniform (1D) | 0 | None | Minimal |
| Sinusoidal (this work) | Moderate | Smooth | Moderate |
| Random distribution | Moderate-high | Irregular | Moderate-high |
| Patchy/clustered | High | Sharp hotspots | High |
4. Conclusion
In this study, a pseudo-two-dimensional (pseudo-2D) multiphysics framework was developed to elucidate the coupled effects of mass transport and pseudo-enzymatic kinetics in a Ti3C2Tx@Pt nanozyme-based glucose sensing platform. By explicitly accounting for lateral heterogeneity in Pt active site distribution and two-dimensional diffusion of glucose and hydrogen peroxide, the model captures key transport-reaction phenomena that are inherently overlooked in conventional one-dimensional approaches. The results demonstrate that surface non-uniformity induces pronounced local substrate depletion, effective thickening of the diffusion layer, and transport bottlenecks that collectively reduce effective glucose flux and surface availability. These effects lead to systematic deviations in apparent kinetic parameters, including an increased effective Michaelis constant and a reduced maximum reaction rate, despite identical mean catalyst loading. The pseudo-2D predictions show excellent agreement with experimentally reported colorimetric spectra and electrochemical trends, confirming the validity of the proposed framework in reproducing both steady-state and transient sensor responses.Beyond quantitative agreement, this work provides mechanistic insight into how nanoscale catalyst dispersion governs macroscopic sensor performance. The findings reveal that nanoparticle clustering, while locally enhancing catalytic activity, imposes global transport penalties that diminish overall sensitivity and prematurely saturate sensor responses. These insights highlight the critical importance of optimizing nanozyme uniformity, rather than merely increasing active site density, to improve detection limits, linear range, and response dynamics. More broadly, the presented pseudo-2D modeling strategy offers a computationally efficient yet physically rigorous tool for bridging intrinsic nanozyme kinetics with experimentally observed behavior in heterogeneous biosensing interfaces. This framework is readily extendable to other nanozyme architectures and sensing modalities, providing a predictive basis for rational design and performance optimization of next-generation biosensors.
The transport-reaction coupling, pseudo-2D formulation, and treatment of lateral surface heterogeneity are generic features of the proposed framework and can be transferred to other nanozyme or heterogeneous catalytic systems. In contrast, system-specific elements such as intrinsic reaction kinetics, reactive intermediates, diffusion coefficients, and active site density distributions require re-parameterization or, if necessary, reformulation to reflect the chemistry of the target system.
Conflicts of interest
There are no conflicts to declare.Data availability
The data that support the findings of this study, including simulation input files, COMSOL model configurations, and post-processed numerical datasets, are available from the corresponding author upon reasonable request.References
- M. H. Hassan, C. Vyas, B. Grieve and P. Bartolo, Recent advances in enzymatic and non-enzymatic electrochemical glucose sensing, Sensors, 2021, 21, 4672 CrossRef CAS PubMed.
- X. Yuan, X. He, J. Fan, Y. Tai, Y. Yao and Y. Luo, et al., Advances in nanozymes with peroxidase-like activity for biosensing and disease therapy applications, J. Mater. Chem. B, 2025, 13, 1599–1618 RSC.
- Y. Ouyang, M. P. O'Hagan and I. Willner, Functional catalytic nanoparticles (nanozymes) for sensing, Biosens. Bioelectron., 2022, 218, 114768 CrossRef CAS PubMed.
- G. Sharma, S. Chatterjee, C. Chakraborty and J.-C. Kim, Advances in nanozymes as a paradigm for viral diagnostics and therapy, Pharmacol. Rev., 2023, 75, 739–757 CrossRef CAS PubMed.
- X. Wang, Q.-Z. Li, Y. Zhao and X. Gao, Recent Advances in Oxidase-like Nanozymes: Mechanisms, Prediction Models, and Applications, ACS Appl. Mater. Interfaces, 2025, 17(49), 66110–66150 CrossRef CAS PubMed.
- V. G. Panferov, X. Zhang, K. Y. Wong, J. H. Lee and J. Liu, Biomedical applications of nanozymes: an enzymology perspective, Angew. Chem., 2025, 137, e202512409 CrossRef.
- F. Momeni, S. M. Khoshfetrat, H. Bagheri and K. Zarei, Ti3C2 MXene-based nanozyme as coreaction accelerator for enhancing electrochemiluminescence of glucose biosensing, Biosens. Bioelectron., 2024, 250, 116078 CrossRef CAS PubMed.
- J. Sengupta and C. M. Hussain, Mxene-based electrochemical biosensors: Advancing detection strategies for biosensing (2020–2024), Biosensors, 2025, 15, 127 CrossRef CAS PubMed.
- A. Sugunan, A. V. Rethnakumaran and M. M. Menamparambath, A review on Ti 3 C 2 T x based nanocomposites for the electrochemical sensing of clinically relevant biomarkers, Sens. Diagn., 2024, 3, 1769–1788 RSC.
- L.-J. Shang, S.-Q. Yu, X.-W. Shang, X.-Y. Wei, H.-Y. Wang and W.-S. Jiang, et al., A non-invasive glucose sensor based on 3D reduced graphene oxide-MXene and AuNPs composite electrode for the detection of saliva glucose, J. Appl. Electrochem., 2024, 54, 1807–1817 CrossRef CAS.
- N. Mohamad Nor, N. S. Ridhuan and K. Abdul Razak, Progress of enzymatic and non-enzymatic electrochemical glucose biosensor based on nanomaterial-modified electrode, Biosensors, 2022, 12, 1136 CrossRef CAS PubMed.
- A. Maghfirah, H. Setiyanto and G. T. Kadja, Up-and-coming MXene-based nanohybrids for electrochemical non-enzymatic glucose sensing: A brief review, Synth. Met., 2025, 117863 CrossRef CAS.
- K. Manibalan and J.-T. Chen, Recent progress on MXene–polymer composites for soft electronics applications in sensing and biosensing: a review, J. Mater. Chem. A, 2024, 12(40), 27130–27156 RSC.
- H. H. Heenen, H. S. Pillai, K. Reuter and V. J. Bukas, Exploring mesoscopic mass transport effects on electrocatalytic selectivity, Nat. Catal., 2024, 7, 847–854 CrossRef CAS.
- N. H. Solangi, R. R. Karri, N. M. Mubarak, S. A. Mazari and B. P. Sharma, Holistic insights of carbon nanotubes and MXene as a promising route for bio-sensing applications, Nanoscale, 2024, 16(46), 21216–21263 RSC.
- L. Lorencova, P. Kasak, N. Kosutova, M. Jerigova, E. Noskovicova and A. Vikartovska, et al., MXene-based electrochemical devices applied for healthcare applications, Microchim. Acta, 2024, 191, 88 CrossRef CAS PubMed.
- X. Lyu, W. Wang, D. Voskov, P. Liu and L. Chen, Multiscale modeling for multiphase flow and reactive mass transport in subsurface energy storage: A review, Adv. Geo-Energy Res., 2025, 15, 245–260 CrossRef CAS.
- W. Yang, L. Sun, J. Bao, Z. Mo, M. Du and Y. Xu, et al., Multiphysics modeling of mass and heat transfer in a thermo-electrochemical cell, Ind. Eng. Chem. Res., 2023, 62, 12345–12355 CrossRef CAS.
- M. Nourizadeh, Y. Bakhshan, J. Khorshidi and S. Niazi, Study on the thermal characteristics of layered NMC cathodes in lithium-ion batteries, J. Solid State Electrochem., 2025, 1–14 Search PubMed.
- L. Wang, M. Li, S. Ya, H. Tian, K. Li and Q. Zhang, et al., Ti3C2TX MXene/Polyaniline-Modified Nylon Fabric Electrode for Wearable Non-Invasive Glucose Monitoring in Sweat, Biosensors, 2025, 15, 531 CrossRef CAS PubMed.
- L. Chen, X. Dai, W. Feng and Y. Chen, Biomedical applications of MXenes: from nanomedicine to biomaterials, Acc. Mater. Res., 2022, 3, 785–798 CrossRef CAS.
- Q. Peng, N. Jiang, L. Qian and W. Yue, Two-dimensional derivatives obtained by oxidation of MXene for simulating the oxidative properties of natural enzymes, 2D Materials, 2023, 10, 045013 CrossRef CAS.
- A. Hussain, Z. Mao, M. Li, M. Ahmed, W. Xu and J. Zhang, et al., A Comprehensive Review of the Pseudo-Two-Dimensional (P2D) Model: Model Development, Solutions Methods, and Applications, Adv. Theory Simul., 2025, 8, 2401016 CrossRef CAS.
- A. Ravitchandiran, S. AlGarni, M. S. AlSalhi, R. Rajaram, T. Malik and S. Angaiah, ZnFe (PBA)@ Ti3C2Tx nanohybrid-based highly sensitive non-enzymatic electrochemical sensor for the detection of glucose in human sweat, Sci. Rep., 2024, 14, 23835 CrossRef CAS PubMed.
- A. Ravitchandiran, P. Dhandapani, S. P. Rajendra, M. S. AlSalhi, R. Rajamohan and S. Angaiah, Development of ZnFe (PBA)@ Mo3C2Tx Nanohybrid based Non-Enzymatic Sensor for the Detection of Glucose in Human Sweat, ChemistrySelect, 2024, 9, e202401553 CrossRef CAS.
- R. Rajamohan and S. Sun, Nanostructured Materials in Glucose Biosensing: From Fundamentals to Smart Healthcare Applications, Biosensors, 2025, 15, 658 CrossRef CAS PubMed.
- M. Şen, E. Yüzer, V. Doğan, İ. Avcı, K. Ensarioğlu and A. Aykaç, et al., Colorimetric detection of H2O2 with Fe3O4@ Chi nanozyme modified µPADs using artificial intelligence, Microchim. Acta, 2022, 189, 373 CrossRef PubMed.
- Z. Li, T. Lei, T. Pei, K. Chen, Z. Zhao and M. Wang, et al., Facile synthesis of MXene-Ti3C2/Co nanosheet hydrogel sensor with the assistance of a smartphone for on-site monitoring of glucose in beverages, Molecules, 2023, 28, 5075 CrossRef CAS PubMed.
- Y. Yang, Y. Zhu, Y. Wu, F. Chang, X. Zhu and X. Zhang, et al., MXene-Based Flexible Paper Chip for Glucose Detection in Sweat in Low-Temperature Environments, Sensors, 2025, 25, 4273 CrossRef CAS PubMed.
- T. S. Gopal, S. K. Jeong, T. A. Alrebdi, S. Pandiaraj, A. Alodhayb and M. Muthuramamoorthy, et al., MXene-based composite electrodes for efficient electrochemical sensing of glucose by non-enzymatic method, Mater. Today Chem., 2022, 24, 100891 CrossRef CAS.
- T. S. Gopal, K. E. Alzahrani, A. K. Assaifan, H. Albrithen, A. Alodhayb and M. Muthuramamoorthy, et al., Reduced graphene oxide supported MXene based metal oxide ternary composite electrodes for non-enzymatic glucose sensor applications, Sci. Rep., 2022, 12, 20583 CrossRef CAS PubMed.
- Q.-F. Li, X. Chen, H. Wang, M. Liu and H.-L. Peng, Pt/MXene-based flexible wearable non-enzymatic electrochemical sensor for continuous glucose detection in sweat, ACS Appl. Mater. Interfaces, 2023, 15, 13290–13298 CrossRef CAS PubMed.
- S. Guenneau and T. Puvirajesinghe, Fick's second law transformed: one path to cloaking in mass diffusion, J. R. Soc., Interface, 2013, 10, 20130106 CrossRef CAS PubMed.
- X. Sun, M. Zhang, J. Chang, L. Xie, Q. Ren and W. Jiang, et al., Ti3C2Tx@ Pt nanocomposite based colorimetric and electrochemical dual-mode platform for glucose detection, Sens. Biosens. Res., 2025, 100800 Search PubMed.
- A. R. Tzafriri, Michaelis-Menten kinetics at high enzyme concentrations, Bull. Math. Biol., 2003, 65, 1111–1129 Search PubMed.
- S. S. Datwani and K. J. Stebe, The dynamic adsorption of charged amphiphiles: The evolution of the surface concentration, surface potential, and surface tension, J. Colloid Interface Sci., 1999, 219, 282–297 CrossRef CAS PubMed.
- J. Xue, J. Li, Y. Ma, E. A. Obaid, H. Ouyang and T. Guo, et al., Multivalent Pt/Ti3C2Tx nanocomposite-based immunochromatographic sensor for colorimetric/temperature/pressure trimodal detection, Anal. Chem., 2024, 96, 15074–15080 CrossRef CAS PubMed.
- V. Thirumal, R. Yuvakkumar, P. S. Kumar, G. Ravi and D. Velauthapillai, Facile preparation and characterization of MXene@ Platinum nanocomposite for energy conversion applications, Fuel, 2022, 317, 123493 CrossRef CAS.
- L.-X. Li, G.-C. Zhang, W.-J. Sun, H.-Y. Zhang, S.-X. Wang and J.-L. Wei, et al., Construction of ultra-small Pt nanoparticles@ Ti3C2Tx MXene electrocatalyst for efficient and stable electrochemical hydrodechlorination of chloramphenicol, Chem. Eng. J., 2022, 433, 134415 CrossRef CAS.
- S. Schnell, Validity of the Michaelis–Menten equation–steady-state or reactant stationary assumption: that is the question, FEBS J., 2014, 281, 464–472 CrossRef CAS PubMed.
| This journal is © The Royal Society of Chemistry 2026 |

