DOI:
10.1039/D5RA09289B
(Review Article)
RSC Adv., 2026,
16, 14688-14727
Smart adsorbent frameworks enabling high-efficiency pharmaceutical degradation via adsorption
Received
1st December 2025
, Accepted 25th January 2026
First published on 17th March 2026
Abstract
Pharmaceutical residues are increasingly detected in aquatic systems and present ecological and human health concerns due to their biological activity, structural complexity, and limited biodegradability. Poor removal via conventional wastewater treatment processes drives the development of smart adsorbents that offer tailored chemistry and stimuli-responsive behavior to selectively capture and degrade pharmaceuticals. This review summarizes recent advances in functionalized carbons, stimuli-responsive polymers, metal–organic frameworks, magnetic composites, and hybrid nanozymes that interact with pharmaceuticals through π–π stacking, electrostatic attraction, hydrogen bonding, and metal–ligand coordination. Special attention is paid to how surface functionalities, pore architectures, and pH-dependent speciation govern adsorption kinetics, isotherms, and selectivity. Coupling adsorption with catalytic degradation processes is highlighted as a synergistic strategy that can enable in situ transformation of adsorbed pharmaceuticals into less toxic products, overcoming drawbacks associated with adsorption-only systems, such as Fenton, photo-Fenton, or peroxymonosulphate activation. Key structure–property relationships, performance descriptors, and recyclability considerations are discussed in establishing a unified smart adsorbent design framework. Finally, critical knowledge gaps and future opportunities are identified, including scalable synthesis, selectivity tuning, regeneration, and integration into continuous treatment systems. This review offers guidelines for the rational development of next-generation smart adsorbents for efficient and sustainable removal of pharmaceutical pollutants.
Introduction
Pharmaceutical effluents (PEs) are wastewaters generated at different stages of pharmaceutical production, including the processes encompassing active pharmaceutical ingredient (API) synthesis, drug product formulation, and antibiotic production. Pharmaceutical effluents are categorized based on their origin or production methods. Among these categories are (A) chemical waste, (B) analysis residues and buffers, and (C) laboratory and surface cleaning waste.1 Fig. 1(a) pharmaceutical pollution pathways.2 Currently, the observed aqueous concentrations of different pharmaceutical compounds vary from nanograms to micrograms per liter.3 The presence of heavy metals in drinking water is hazardous to consumers. Metal ions in water can originate from the leaching of ore waste and from anthropogenic resources, which mainly include industrial wastewater and solid waste deposits. Currently, with the rapid development of industrial areas and the associated increase in heavy metal concentration in water systems, water pollution has substantially increased. Treatment of metal ions such as Pb, Cu, Hg, Cd, Cr, and Zn attracts particular attention because of their higher toxicity even at trace concentrations.4 Fig. 1(b) hydroxyl radical generation.5 Once administered, pharmaceuticals and personal care products (PCs) are incompletely metabolized by the body. Both original compounds and metabolites are subsequently released into the aquatic environment.6 They pose a threat to human health due to their persistence in the ecosystem, bioaccumulation, and biomagnification in the food webs. These also lead to adverse effects on human health, which is why PCs are a major cause for concern globally.7 Fig. 1(c) sources of pharmaceutical contamination.8 Consequently, they are now categorized as emerging pollutants.9 Fig. 1(d) general pathway of degradation. A huge quantity of waste is being produced worldwide, both solid and liquid. It has been estimated that the yearly production of waste will increase from 2.01 to 3.40 billion tonnes worldwide by 2050. However, a very small amount of waste is being recycled. The remaining waste is either disposed of improperly or left untreated, resulting in various forms of pollution, including land, water, and air pollution, as well as thermal and visual pollution, ultimately causing serious harm to both human health and the environment. Table 1 shows the pharmaceutical categories most often found in water.10
 |
| | Fig. 1 (a) Pharmaceutical pollution pathways2 (Adapted with permission from Elsevier (Tiwari, S. B., Ouarda, Drogui, Tyagi and Buelna, Bioresour. Technol., 2017), copyright 2017). (b) Hydroxyl radical generation.5. (Adapted with permission from Elsevier (Wang and Zhuan, Sci. Total Environ., 2020), copyright 2020). (c) Sources of pharmaceutical contamination8 (Adapted under a Creative Commons CC BY Attribution 4.0 International License, Mansouri, F., C. K., Roche, N., Ksibi, M., removal of pharmaceuticals from water by adsorption and advanced oxidation processes: state of the art and trends, Applied Sciences, 2021, 11(14), 6659). (d) General pathway of degradation. | |
Table 1 Pharmaceutical categories most frequently found in water, including the compounds discussed, fall into several key therapeutic categories11 (Adapted with permission from Elsevier (Rivera-Utrilla et al., Chemosphere, 2013), copyright 2013)
| Drug class |
Examples |
| Anti-inflammatories and analgesics (pain and inflammation relief) |
Acetylsalicylic acid (aspirin), diclofenac, ibuprofen, paracetamol |
| Antiepileptics (seizure control) |
Carbamazepine |
| Antidepressants |
Benzodiazepines |
| β-Blockers (cardiovascular agents) |
Metoprolol, atenolol, and propranolol |
| Lipid-lowering drugs (cholesterol management) |
Fibrates |
| Antibiotics (a broad group for bacterial infection treatment) |
Macrolides, penicillins, β-lactams, quinolones, tetracyclines and sulphonamides |
| Antiulcer drugs and antihistamines (acid reduction) |
Ranitidine and famotidine |
The efficiency of removal of pollutants, the low cost of adsorbents, and the possibility of regenerating or recycling the adsorbents are considered crucial. Various types of adsorbents, such as activated carbon, agricultural waste, and natural and synthetic polymers, and their composites, have been used to remove pollutants from water as well as wastewater.12 Fig. 2(a) conceptual overview of smart adsorbents in pharmaceutical wastewater treatment. The application of metal–organic framework materials as catalysts for decomposing organic pollutants has provided viable solutions for various environmental issues. Recent developments on the use of metal–organic frameworks for the removal and degradation of organic pollutants are discussed. These are classified according to the various applications that the metal–organic frameworks have been employed in, such as adsorbent materials, immobilized enzymes, nanozymes, photocatalysts, and, finally, as components in Fenton and sulphate radical technologies.13 Fig. 2(b) from pollutant transfer to pollutant destruction: adsorption and catalytic degradation. The smart adsorbent framework for pharmaceutical drug removal operates through a synergistic mechanism combining adsorption and Fenton-like catalytic degradation. Initially, the pharmaceutical molecules are selectively captured by the hydrochar-based smart adsorbent via hydrogen bonding, π–π interactions, and electrostatic attractions between the drug functional groups and surface hydroxyl, carboxyl, and carbonyl groups of the adsorbent. This targeted adsorption enriches the pollutants in the vicinity of the bimetallic Fe/Cu active centers, enabling efficient generation of hydroxyl radicals (˙OH) through the Fenton-like reaction, where Fe2+/Cu+ reacts with hydrogen peroxide. The adsorbed drugs are subsequently degraded by these radicals into smaller intermediates, eventually leading to complete mineralization to CO2 and H2O. Spectroscopic analyses, such as FTIR and XPS, demonstrate modifications in functional group signals, confirming strong adsorption, while EPR studies detect the presence of ˙OH radicals, evidencing catalytic activity. Complementary density functional theory (DFT) calculations further elucidate the adsorption energies and electron–transfer processes at the Fe Cu sites, providing a molecular-level understanding of how the smart adsorbent framework enhances pharmaceutical degradation efficiency through the combined adsorption Fenton process. Fig. 2(c) smart adsorbent mechanism for pharmaceutical degradation via adsorption–Fenton process. Fig. 2(d) smart adsorbent–Fenton degradation flowchart.
 |
| | Fig. 2 (a) Conceptual overview of smart adsorbents in pharmaceutical wastewater treatment. (b) From pollutant transfer to pollutant destruction: adsorption and catalytic degradation. (c) Smart adsorbent mechanism for pharmaceutical degradation via the adsorption–Fenton process. (d) Smart adsorbent–Fenton degradation flowchart. | |
Traditionally, antibiotics are chemical substances designed to suppress or eliminate microbial proliferation. Substances are derived from microbes, and some are semi-synthetic or entirely man-made.14 The following key aspects should be considered: (1) the presence of antibiotics in various environmental locations, specifically surface water, soil and wastewater; (2) antibiotics exerting toxic effects on non-target lifeforms, affecting both aquatic and terrestrial species; and (3) current treatment methods for degrading and removing antibiotics, such as hydrolysis, oxidation, biodegradation and adsorption.15 Fig. 3(a) Specific pathway of degradation through adsorption. Anaerobic bioprocessing is emerging as a potential opportunity to combat the issues associated with antibiotics-loaded pharmaceutical wastewater. The treatment of antibiotic wastewater is vital, and the operation of anaerobic treatment systems must be optimized.16 Fig. 3(b) general mechanism for degraded products. Three structurally different families of antibiotics were screened for their efficiencies of degradation: Amoxicillin (beta-lactam), Ciprofloxacin (fluoroquinolone), and Streptomycin (aminoglycoside).17 The direct dangers posed to human health by pharmaceutical residues present in untreated wastewater include their contamination of plants through irrigation and fish that are consumed by people.8 Fig. 3(c) conversion process of degraded techniques. Most pharmaceuticals are only partially removed during standard wastewater treatment. After their entry into the environment, they also do not easily biodegrade.18 Fig. 3(d). Ozonation, biodegradation, photocatalysis, and the Fenton process are the most explored methods for the elimination of pharmaceuticals from polluted water. Advanced oxidation processes (AOPs) are effective for degrading synthetic pollutants such as pharmaceuticals; however, they can sometimes result in the formation of more harmful intermediate compounds. Besides, AOPs are costly and challenging to operate, particularly when aiming for the complete degradation of persistent compounds. The nature of the selected adsorbent, however, plays a pivotal role in adsorption success.19 Fig. 4(a) study of the elimination of key pharmaceutical contaminants (carbamazepine, caffeine, ibuprofen and diclofenac) utilizing Th-CDP and the P-POP adsorbents.20 Fig. 4(b) successful removal techniques for different antibiotics. Advanced treatment technologies include a range of processes such as membrane-based filtration, granular activated carbon systems, biologically active porous media filters, ozonation, and advanced oxidation processes (AOPs), as summarized in Table 2 ref. 21.
 |
| | Fig. 3 (a) Specific pathway of degradation through adsorption. (b) General mechanism for degraded products. (c) Conversion processes in degradation techniques. (d) 3-D schematic of drug degradation reactions. | |
 |
| | Fig. 4 (a) Removal of pharmaceuticals (caffeine, diclofenac, carbamazepine, and ibuprofen) with the help of the P-POP and Th-CDP adsorbent20 (Adapted with permission from Elsevier (Song et al., J. Hazard. Mater., 2024), copyright 2023). (b) Successful removal techniques for different antibiotics. Advanced treatment technologies include a range of processes such as membrane-based filtration, granular activated carbon systems, biologically active porous media filters, ozonation, and advanced oxidation processes (AOPs), as summarized in Table 2.21 (Adapted with permission from Elsevier (Jumah et al., J. Environ. Chem. Eng., 2024), copyright 2022). (c) Key pharmaceutical adsorption mechanism using wood-based adsorbents22 (Adapted under a Creative Commons CC BY Attribution 4.0 International License, Godiya, C. B., Leiviskä, T., wood-derived adsorbents for the removal of pharmaceutical contamination from wastewater: a review, Environmental Chemistry Letters, 2025, 1–31). (d) Graphical representation showing water contamination and its treatment by a nanotechnology-assisted approach to obtain treated water23 (Adapted under a Creative Commons CC BY Attribution 4.0 International License, Saroa, A., et al., Nanotechnology-assisted treatment of pharmaceuticals contaminated water, Bioengineered, 2023, 14(1), 2260919). | |
Table 2 Advantages, drawbacks and removal efficiencies of different advanced wastewater treatment processes for pharmaceutical removal24 (Adapted with permission from Elsevier (Stadlmair et al., Chemosphere, 2018), copyright 2018
| Advanced treatment method |
Typical removal efficiency |
Main advantages |
Main drawbacks |
Typical pharmaceutical removal efficiency |
References |
| Physical |
Conventional activated sludge (CAS) |
Minor to intermediate |
No hazardous byproducts are produced |
Competition reduces the overall adsorption capacity from dissolved organic carbon (DOC) and the issue of safe waste disposal |
20–60% |
25 |
| Activated carbon in powdered or granular form |
Moderate to high |
26 |
| High-pressure membrane filtration (nanofiltration, reverse osmosis) |
Medium to high |
No toxic byproducts |
High energy intensity (energy-intensive operation), the issue of membrane fouling and the challenge of concentrate stream disposal |
85–99% |
27 |
| Biological |
Membrane bioreactor (MBR) |
Limited to average |
Absence of harmful byproducts |
Membrane fouling and diminished adsorption capacity due to the aging of the MBR sludge |
60–95% |
28 |
| Biofiltration |
Intermediate |
Minimal energy usage and zero waste production |
Large spatial requirements and performance variability |
40–85% |
26 |
| Enzymatic processes |
Under review |
No biological waste is generated |
The system is characterized by high selectivity and durability, but suffers from unproven technical feasibility |
50–95% |
26 |
| Chemical |
AOP: different combinations of hydrogen peroxide, ozone and UV respectively |
Moderate to high |
Can oxidize a wide range of pharmaceuticals (i.e., it is non-selective) |
High power consumption and the possibility of toxic by-product formation |
60–99% |
25 and 29 |
In 1903, Tswett's discovery of adsorption chromatography was developed as a novel analytical technique following the realization that adsorption processes exhibit selectivity. Tswett advocated for its use in separating various mixtures.30 Adsorption is a process in which pollutants, called adsorbates, transfer from a fluid (liquid or gas) and then interact or react on a solid surface, known as an adsorbent.31 Fig. 4(c) promising innovations set to transform adsorption technology.32 Adsorption is a well-established surface-based water-treatment technique, the effectiveness of which is determined by the available active sites, pore structure, surface area and various complex interactions.33 Fig. 4(d) graphical representation showing water contamination and its treatment by nanotechnology assisted approach to obtain treated water.23 Among water–treatment processes, adsorption presents the simplest design, with a small initial capital outlay for cost and space. Advantages include effectiveness at low adsorbate concentrations, low capital investment, the ability to reuse and regenerate adsorbents, and adaptability to both batch and continuous processes. Adsorption is an energy-saving technique that can achieve up to 90% removal under mild operating conditions. Photocatalysis generates acidic conditions, around pH 3.0, requiring a neutralizing stage after treatment, with considerable sludge production.34 Fig. 5(a) this figure presents a schematic of pharmaceutical entry routes into surface water and the compositional distribution of these drugs found in U.S. surface water.35 Regarding the removal of antibiotics from aquatic systems, adsorption processes generally use either batch or continuous methods. A wide variety of adsorbents may be employed, such as biochar, activated carbons (ACs), ion exchange resins, zeolites, clay minerals, and carbon nanotubes (CNTs). Hybrid systems using a variety of membrane technologies in conjunction with traditional methods of filtration, sedimentation, ozonolysis, chlorination, and photocatalysis have been developed; these do not come without their own set of problems.21 Fig. 5(b) exposure of pharmaceuticals in the environment.36 The overall expense of an adsorption process is generally governed by three factors: capital investment, energy use, and chemical costs. One of the most promising methods to decrease such costs is the utilization of inexpensive adsorbents synthesized from waste.31 Adsorption is considered a prime method for the eradication of antibiotics from contaminated water, as it has 90–100% efficiency for concentrations in the mg L−1 range.37 Adsorptive separation and purification processes can occur through both steric (based on size and shape exclusion) and kinetic (based on differences in adsorption rates) mechanisms.30 Fig. 5(d) competitive interaction of H2O and CO2 on active sites. Table 3 shows a roadmap of adsorbent research for pharmaceutics removal from water. Table 4 summarises basic types of adsorbents that can be used.38
 |
| | Fig. 5 (a) Schematic showing various pathways by which pharmaceuticals enter surface water, and distribution of the types of pharmaceuticals found in surface water of the USA. WW (wastewater); DW (drinking water); CAFO (concentrated animal feeding operation); WWTP (wastewater treatment plant); and DWTP (drinking water treatment plant)35 (Adapted under a Creative Commons CC BY Attribution 4.0 International License, RP, D., Pharmaceuticals in the Surface Water of the USA: A Review, Current Environmental Health Reports, 2014, 1(2), 113–122). (b) Release of pharmaceuticals in the environment36 (Adapted with permission from Elsevier (Kayode-Afolayan, Ahuekwe and Nwinyi, Sci. Afr., 2022), copyright 2022). (c) Competitive interactions of H2O and CO2 on active sites. Table 3 shows a roadmap of adsorbent research for pharmaceutics removal from water. Table 4 summarises basic types of adsorbents that can be used.38 (Adapted under a Creative Commons CC BY Attribution 4.0 International License, Zhao, J., et al., Understanding the effect of H2O on CO2 adsorption capture: mechanism explanation, quantitative approach and application, Sustainable Energy & Fuels, 2020, 4(12), 5970–5986). | |
Table 3 Roadmap of adsorbent research for pharmaceutical removal
| Time period |
Adsorbent type |
Advances |
Pharmaceutical removal relevance |
Limitations |
References |
| 1970s–1990s |
Conventional materials: clays, zeolites |
Basis of adsorption technology |
Efficient for general organics used as baseline adsorbent |
Low selectivity |
39 |
| 1990s–2005 |
Modified carbons and natural sorbents |
Surface functionalization |
Enhanced electrostatic interaction |
Limited target specificity |
40 |
| 2005–2015 |
Nanoadsorbents: CNTs, metal oxides |
Increased surface reactivity, faster kinetics |
Nanomaterials show high capacity toward drugs |
Aggregation cost |
39 |
| 2015–2020 |
Functional MOFs and porous frameworks |
Tunable porosity, functional design for selectivity |
MOFs show high pharma uptake |
Stability and regeneration still challenge |
41 |
| 2020–2025 |
Hybrid and bio-derived: biochar, hydrochar |
Combining adsorbent types: magnetic recovery |
Good removal of antibiotics |
Regeneration complexity |
42 |
| 2023–present |
Advanced nanostructure and specific adsorbents |
Highest capacities and partition coefficients for drugs |
Record adsorption capacity materials |
Scalability |
43 |
| Future directions |
Smart/selective and multifunctional systems |
Stimuli-responsive, integrated adsorbent/catalyst |
Toward simulation removal + degradation |
Requires validation in real wastewater |
44 |
Table 4 Basic types of adsorbents30 (Adapted with permission from Elsevier (Dąbrowski, Adv. Colloid Interface Sci., 2001), copyright 2001)
| Carbon adsorbents |
Mineral adsorbents |
Other adsorbents |
| Activated carbons |
Silica gels |
Synthetic polymers |
| Activated carbon fibres |
Activated alumina |
Composite adsorbents |
| Carbonaceous |
Pillared clays |
|
| Fullerenes |
Zeolites |
Mixed sorbents |
| Heterofullerenes |
Clay minerals |
|
| Mesocarbon microbeads |
Hydroxides of metals |
X-elutrilithe, X = Zn, Ca |
| Molecular carbon sieves |
Oxides of metals |
Complex mineral carbons |
| Nanomaterials |
Porous clay heterostructures (PCHs) |
|
The adsorption process falls into two distinct forms. Physical adsorption is influenced by van der Waals forces between the adsorbent and adsorbate molecules. Chemical adsorption involves reactions occurring directly at a molecular level between a substance (adsorbate) that is being adsorbed and the surface of the material doing the adsorbing (adsorbent).45 Fig. 6(a) bio-regeneration of saturated adsorbents.46 Membrane-based adsorption techniques allow greatly improved water purification by combining the best of both worlds: the ability of materials to adsorb pollutants and the selective separation of membranes.32 Fig. 6(b) chemical modifications to biochar improve the removal of pollutants and its reuse potential.47
 |
| | Fig. 6 (a) Bio-regeneration of saturated adsorbents46 (Adapted under a Creative Commons CC BY Attribution 4.0 International License, Yadav, S., et al., Cutting-edge regeneration technologies for saturated adsorbents: a systematic review on pathways to circular wastewater treatment system, Environmental Monitoring and Assessment, 2025, 197(2), 215). (b) Chemical modifications of biochar improve the removal of pollutants and its reuse potential47 (Adapted with permission from Elsevier (Alshehri and Pugazhendhi, J. Hazard. Mater. Adv., 2024), copyright 2024). (c) Main methods used for adsorbent modifications48 (Adapted under a Creative Commons CC BY Attribution 4.0 International License, Dehghani, M. H., et al., Recent advances on sustainable adsorbents for the remediation of noxious pollutants from water and wastewater: A critical review, Arabian Journal of Chemistry, 2023, 16(12), 105303). (d) Pharmaceutical adsorption processes using nanotubes obtained by green routes49 (Adapted with permission from Elsevier (Alshehri and Pugazhendhi, J. Hazard. Mater. Adv., 2024), copyright 2024). | |
Adsorption processes range from the nanometer scale of pores, through micrometer-sized crystals and millimeter-sized adsorbent particles.50 Fig. 6(c) the main methods used for the adsorbent modification.48 The adsorbent's cost makes up about 70% of the total expense, and developing adsorbents using inexpensive materials derived from waste and biomass provides a sustainable and economical solution. This is crucial because actual wastewater and effluents almost always contain multiple components that can compete for adsorption sites, which, in turn, lowers the ideal loading capacity for a pure substance in the adsorption system.51 Fig. 6(d) indicate adsorptive removal of pharmaceutical pollutants using green-synthesized nanotube.49 Smart adsorbents offer a novel technology for the removal of pharmaceutical pollutants, which not only provide high adsorption capacity but also promote the catalytic degradation of organic molecules. Current trends in research and development are oriented towards the use of functionalized carbons, metal-based hybrid adsorbents, and metal oxide-functionalized adsorbents, which provide multiple avenues for improvement in efficiency.
Fundamentals of adsorption in pharmaceutical degradation
Pharmaceuticals that are widely used in animal welfare (veterinary medicine) can enter the environment through various pathways. After being consumed, drugs such as antimicrobials are constantly released into natural ecosystems via excreta (urine and feces). Veterinary medicines encompass a wide spectrum of pharmacological classes. The extensive use of these compounds in livestock farming has led to growing concerns, as agricultural practices increasingly serve as a significant pathway for pharmaceutical contaminants to enter the environment as shown in Table 5 ref. 52.
Table 5 Categories of veterinary pharmaceuticals: these pharmaceuticals fall into a range of pharmacological categories52 (Adapted under a Creative Commons CC BY Attribution 4.0 International License, Wychodnik, K., et al., Poultry farms as a potential source of environmental pollution by pharmaceuticals, Molecules, 2020, 25(5), 1031)
| Major drug class |
Specific sub-categories/types |
Examples of agents |
References |
| Antiparasitics |
Endectocides, ectoparasiticides, endoparasiticides |
Anthelmintics and antiprotozoals |
53 |
| Anti-infective agents |
Antifungals, antimicrobials |
General antibiotics, antifungals |
54 |
| Hormones |
Various regulatory compounds |
Estrogens, progestins, etc. |
55 |
| Anti-inflammatory drugs |
Steroidal and non-steroidal types |
Corticosteroids, NSAIDs |
56 |
| Central nervous system agents |
Anesthetics, tranquilizers, sedatives |
Agents affecting mood and consciousness |
57 |
| Gastrointestinal and other agents |
Bronchodilators, antacids, diuretics, emetics |
Drugs for respiratory, digestive, and fluid balance |
58 |
Several pharmaceuticals, including diclofenac, ibuprofen, phenazone, naproxen, ketoprofen, indomethacin, and acetylsalicylic acid, have been detected in overland water. This involves two aspects: the disposal of unwanted chemicals and metabolic excretion.59 Pharmaceuticals enter the environment from two main types of sources. Point source pollution originates from distinct, identifiable points and, therefore, can be quantifiable for mathematical modeling. Prime examples are industrial and healthcare facility effluents, along with traditional wastewater treatment systems such as sewage treatment plants and septic tanks, all of which directly release pharmaceuticals into soil resources. Diffuse pollution is a lot more challenging to trace to a single source because it occurs across large areas.60 Fig. 7(a) origin and routes of pharmaceutical products.61 Pharmaceutical compounds in wastewater are effectively removable through various processes, including sorption, biodegradation, and abiotic transformation, such as photolytic degradation. Biodegradation may be complete (a process termed mineralization), yielding carbon dioxide (CO2) and water (H2O), or incomplete/partial, where the pharmaceutical may be conjugated or degraded into a metabolite, somewhat similar to processes in the human body.62 Antibiotics are also commonly detected in water matrices. Due to their antibacterial properties, it is not common that they are efficiently eliminated by wastewater treatment plants. Anti-inflammatory pharmaceuticals, such as diclofenac, naproxen, aspirin and ibuprofen, are among the dominant PPs detected in the environment, representing about 15% of all the PPs detected.63 Fig. 7(b) routes of pharmaceutical contamination in aquatic environments and corresponding biological treatment solutions (bioremediation).64 Humans can be exposed to non-therapeutic or recreational drugs by two broad routes. (1) Environmental recycling: ingestion of minute amounts of drugs present in the surroundings may take place through contaminated drinking water and food. These are mainly residues coming out of drugs released into the environment and going into the food and water intake. (2) Drug waste: exposure may also take place from the intake of drug wastes, either accidently (taking leftover drug wastes, for example) or intentionally (consuming leftover drug wastes).65 Fig. 7(c) publication history of research on pharmaceutical pollutants between 2010 and 2021.63 Fig. 7(d) main therapeutic categories.66 Pharmaceuticals present in the environment can affect human health, mainly through our drinking water and food. Both surface and groundwater, which we rely on for our water supply, can become contaminated with these drugs. Despite the fact that pharmaceutical concentrations in drinking water and food are typically low, the possibility of prolonged, continuous exposure is a significant concern.67 Among the many types of drug residues found, certain pharmaceuticals are frequently detected. These commonly reported categories include: antibiotics, lipid-regulating agents, e.g. statins, anti-inflammatory drugs, beta-blockers, contrast media, and contraceptives.68 Antibiotics have been widely used for infectious disease therapy since the 1940s. In addition to human medicine, they are also used to enhance growth in fish farms and livestock. A major source of environmental contamination by antibiotics is hospitals.69 Fig. 7(e) mechanisms of various kinds of adsorptions: (A) physisorption and (B) chemisorption.70 Essentially, the removal of pharmaceutical compounds is achieved through three main routes involving physical, biological, and chemical methods. Most WWTPs apply a primary system, which relies on physical and chemical methods of treatment. In this manner, some pharmaceuticals could be adsorbed and thus removed, but many still remain in the water. This is because most pharmaceutical compounds cannot be easily metabolized as a carbon source by the microorganisms in the activated sludge. Furthermore, these compounds can sometimes suppress the effectiveness of these beneficial microorganisms or potentially build up (bioaccumulate) within the food chain. Tertiary water treatment methods offer advanced solutions for further purification. These can include, biological systems for removing nitrogen compounds, ion exchange processes to eliminate various ions, chemical precipitation to specifically target and remove phosphorus, distillation to remediate volatile organic compounds, liquid–liquid extraction for separating pollutants, adsorption onto activated carbon to effectively eliminate both organic and inorganic contaminants, and advanced oxidation processes (AOPs), which are highly efficient in decomposing toxic and bio refractory persistent organic contaminants.71 Fig. 8(a) application of nanomaterials for water treatment.70 The key step during the degradation of pharmaceuticals, as it enriches pollutants on the adsorbent surface and creates efficient interaction with reactive sites. Adsorption behavior has recently been determined by surface functionality, porosity, and surface charge to have a direct impact on degradation kinetics. Therefore, a clear understanding of adsorption fundamentals provides a key basis for designing high-performance smart adsorbents for pharmaceutical removal.
 |
| | Fig. 7 (a) Origin and routes of pharmaceutical products61 (Adapted under a Creative Commons CC BY Attribution 4.0 International License, Gupta, N., et al., Pollution by pharma industries: an overview, Environmental Remediation Technologies, Regulations and Safety, 2023, 55). (b) Routes to pharmaceutical contamination in aquatic environments and corresponding biological treatment solutions (bioremediation)64 (Adapted under a Creative Commons CC BY Attribution 4.0 International License, Ortúzar, M., et al., Pharmaceutical pollution in aquatic environments: a concise review of environmental impacts and bioremediation systems, Frontiers in Microbiology, 2022, 13, 869332). (c) Publication trends in the period of 2010–2021 of pharmaceutical pollutants63 (Adapted under a Creative Commons CC BY Attribution 4.0 International License, Bustos Bustos, E., et al., Detection and treatment of persistent pollutants in water: general review of pharmaceutical products, ChemElectroChem, 2022, 9(12), e202200188). (d) Main therapeutic categories66 (Adapted under a Creative Commons CC BY Attribution 4.0 International License, Rodríguez-Serin, H., et al., Literature review: Evaluation of drug removal techniques in municipal and hospital wastewater, International Journal of Environmental Research and Public Health, 2022, 19(20), 13105). (e) Mechanisms of various kinds of adsorptions: (A) physisorption and (B) chemisorption70 (Adapted under a Creative Commons CC BY Attribution 4.0 International License, AlAqad, K. M., et al., Adsorbent materials for water treatment: a review of current trends and future challenges, Environmental Pollution and Management, 2025, 2, 1–3). | |
 |
| | Fig. 8 (a) Nanomaterials used in water treatment applications70 (Adapted under a Creative Commons CC BY Attribution 4.0 International License, AlAqad, K. M., et al., Adsorbent materials for water treatment: a review of current trends and future challenges, Environmental Pollution and Management, 2025, 2, 1–3). (b) Drug (Carbamazepine) adsorption mechanisms onto activated carbon and biochar72 (Adapted under a Creative Commons CC BY Attribution 4.0 International License, Décima, M. A., et al., A review on the removal of carbamazepine from aqueous solution by using activated carbon and biochar, Sustainability, 2021, 13(21), 11760). (c) Mechanism of adsorption of heavy metals on carbon-based adsorbents73 (Adapted under a Creative Commons CC BY Attribution 4.0 International License, Sheraz, N., et al., Comprehensive assessment of carbon-, biomaterial- and inorganic-based adsorbents for the removal of the most hazardous heavy metal ions from wastewater, RSC Advances, 2024, 14(16), 11284–11310). (d) Evaluation of adsorption kinetics74 (Adapted with permission from Elsevier (Simonin, J.-P., Chem. Eng. J., 2016), copyright 2016). | |
Smart adsorbent frameworks
Adsorption is the reversible process in which fluid molecules stick to a solid surface, forming a thin layer. The subsequent detachment of these molecules from the surface is desorption.75 Fig. 8(b) drug (Carbamazepine) adsorption mechanisms onto biochar and activated carbon.72 This is driven by the same weak intermolecular forces that cause vapors to condense. The energy involved is rather low, less than 40 kJ mol−1. Because the forces are weak in physisorption, it proceeds under mild thermal conditions, demanding little energy activation. It occurs naturally at low temperatures. Unlike chemical adsorption (chemisorption), physisorption can create more than a monolayer of adsorbate on the surface. Chemical adsorption (or chemisorption) is a form of adsorption where a reaction mechanism takes place between the adsorbent (surface) and the adsorbate (substance being adsorbed). This process requires a high activation energy and occurs at high temperatures, often with an energy change greater than 40 kJ mol−1.76 Fig. 8(c) mechanism of adsorption of heavy metals on carbon-based adsorbents.73 Adsorption isotherm models, which consider both equilibrium data and adsorbent properties, describe the fundamental interaction mechanisms that occur between pollutants and adsorbent materials. This system is useful for defining the fundamental behavior of the adsorption process.77 Fig. 8(d) evaluation of adsorption kinetics.74 Rationally designed frameworks incorporate surface functionalization, metal active sites, and hybrid architecture to promote synergistic adsorption–degradation pathways. In this way, smart adsorbents represent a promising and sustainable platform for next-generation pharmaceutical wastewater treatments. A comprehensive overview of theoretical isotherm and kinetic models employed to interpret adsorption behavior in biochar-assisted removal of pharmaceutical contaminants from aqueous systems as represented in Table 6 ref. 78.
Table 6 Summary of different theoretical models applied for predicting isotherm and kinetic data of biochar-mediated adsorption of PCs78 (Adapted under a Creative Commons CC BY Attribution 4.0 International License, Chauhan, S., et al., Biochar-mediated removal of pharmaceutical compounds from aqueous matrices via adsorption, Waste Disposal & Sustainable Energy, 2023, 5(1), 37–62)
| Models |
Equations |
Model parameters |
Biochar precursor |
Pharmaceutical compounds |
References |
| Langmuir |
 |
qe (mg g−1) = equilibrium adsorption capacity |
Banana stem fibers |
Carbamazepine (CBZ) |
79 |
| qL (mg g−1) = maximum adsorption capacity |
Date stone seeds |
Ibuprofen (IBF) |
| KL (L mg−1) = Langmuir isotherm constant |
Pomelo peel |
Amoxicillin (AMX) |
| Ce (mg L−1) = equilibrium adsorbate concentration in solution |
| Freundlich |
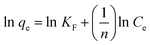 |
KF [(mg g−1) (L mg−1)] = Freundlich isotherm constant |
Municipal solid waste (MSW) |
Tetracycline (TC) |
80 |
| n = heterogeneity factor |
Gliricidia sepium |
Caffeine (CAF) |
| Gooseberry seed shells |
Naproxen (NPX) |
| Sips |
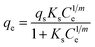 |
qs (mg g−1) = maximum uptake of adsorbate per unit mass of adsorbent |
Giant reed |
Amoxicillin (AMX) |
81 |
| Ks (L mg−1)1/m) = Sips constant related to energy of adsorption |
MSW |
| m = Sips parameter characterizing the system heterogeneity |
Ciprofloxacin (CFX) |
| Redlich–Peterson (R–P) |
 |
KR (L g−1) Redlich–Peterson isotherm constant |
Bovine bone |
Sulfamethoxazole (SMX) |
82 |
| αR and β are constants used for fitting the model |
Pure glucose |
Paracetamol (PRC) |
| Pine sawdust |
Caffeine (CAF) |
| Dubinin–Radushkevich (D–R) |
ln![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) qe = ln qe = ln![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) qD − Bε2 qD − Bε2 |
qD (mg g−1) = theoretical isotherm saturation capacity |
Cherry stalk |
Ciprofloxacin (CFX) |
83 |
| B (mol2 kJ−2) = Dubinin–Radushkevich isotherm constant |
Pomelo peel |
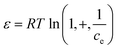 |
ε (kJ mol−1) = adsorption potential derived from Polanyi adsorption potential theory |
Pomelo peel waste |
Carbamazepine (CBZ) |
| R = universal gas constant (8.314 J mol−1 K−1) |
Pure glucose |
| T (Kelvin) = temperature |
Paracetamol (PRC) |
| Temkin |
 |
KT (L g−1) = equilibrium binding constant corresponding to the maximum binding energy |
Corn husk |
Caffeine (CAF) |
84 |
| Gliricidia sepium |
Tetracycline (TC) |
| b = Temkin isotherm constant |
Cherry stalk |
Ciprofloxacin (CFX) |
| Tetracycline (TC) |
| Pseudo-first-order (PFO) |
ln(qe − qt) = ln![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) qe − k1t qe − k1t |
qt (mg g−1) = amount of adsorbate adsorbed at any time t (min) |
Pure glucose |
Amoxicillin (AXM) |
85 |
| k1 (min−1) = PFO rate constant |
Giant reed |
Paracetamol (PRC) |
| Pseudo-second-order (PSO) |
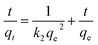 |
k2 (g mg−1 min−1) = PSO rate constant |
Gooseberry seed shells |
Amoxicillin (AXM) |
81 |
| Giant reed |
Paracetamol (PRC) |
| Intra-particle diffusion (IPD) |
qt = kt t1/2 |
kt (mg g−1 min−1/2) = IPD rate constant |
Cherry stalk |
Ciprofloxacin (CFX) |
86 |
| Textile effluent sludge |
Ofloxacin (OFL) |
| Corn husk |
Tetracycline (TC) |
| Levofloxacin (LEV) |
| Boyd's film diffusion |
 |
F(t) = ratio of qt to qe corresponding to fractional achievement of equilibrium Bt = mathematical function of F |
Bagasse biomass |
Tetracycline (TC) |
87 |
| Bt = −0.4977 − ln (1, −, F, (t), 0.86 <F(t)> 1 |
Cherry stalk |
Ciprofloxacin (CFX) |
| Elovich |
 |
α (mg g−1 min−1) = rate of adsorption during the initial phase |
Raw bamboo |
Sulfamethoxazole (SMX) |
88 |
| β (g mg−1) = desorption constant |
Pure glucose |
Paracetamol (PRC) |
Engineering high-efficiency adsorption systems
(1) Langmuir isotherm: the Langmuir theory, one of the earliest explanations for adsorption, is based on a kinetic approach and the following core assumptions: (i) the adsorption process occurs with uniform, equivalent sites. (ii) Every site can only hold a single adsorbate molecule (monolayer adsorption). (iii) The bonds are strong enough to keep the adsorbate attached, and there are no interactions (attraction or repulsion) between them. A significant limitation of the Langmuir isotherm is its unrealistic premise that the heat of adsorption remains constant and does not depend on how much of the surface is covered by the adsorbate. (2) Freundlich isotherm: this isotherm is a popular experimental framework frequently employed to characterize adsorption processes occurring on heterogeneous surfaces. The model contains two parameters, KF and n. Parameter n is a measure of surface heterogeneity, meaning it tells how far the surface binding locations are from having the same adsorption energy. The linear form is:89| |
 | (1) |
(3) Langmuir–Freundlich isotherm: the Langmuir–Freundlich isotherm represents a highly effective and functional model for adsorption equilibrium studies, as it incorporates some aspects of both Langmuir and Freundlich models. Though the original model was developed for gas adsorption on solids, a similar equation is presently used extensively in adsorption from liquid phases.89
| |
 | (2) |
The rate of adsorption is crucial because it directly influences how efficient the process is.90 The thermodynamic parameters change with the temperature variation in an adsorption process, as it is sensitive to temperature. (4) Gibbs free energy of change (ΔG°): ΔG° demonstrates whether an adsorption process will occur and if it is viable. A negative value of ΔG° represents that the process is spontaneous, whereas a positive ΔG° reveals the opposite: one that will not occur spontaneously.91
| |
ΔG° = −RT![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) ln ln![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) KC KC
| (3) |
where
R is the universal gas constant (J mol
−1 Kg
−1 K
−1),
T is the temperature (K), and
KC is the equilibrium constant. For example, a comprehensive thermodynamic investigation revealed that the adsorption entropies and enthalpies for CO
2 and H
2O are temperature dependent. This dependence causes a switch in the relative Gibbs free adsorption energy order among the two molecules.
38 Fig. 9(a) studies on dye removal utilizing a range of synthesized adsorbents.
92 The adsorption of pharmaceuticals occurs because of particular interaction mechanisms. Aromatic pharmaceuticals have a high interaction with π-rich materials, such as graphene oxide and biochar. The interaction occurs because of the π–π stacking interaction. Pharmaceutical polar groups such as the carboxylate ion and primary or secondary amines can form hydrogen bonds with the oxygen- or amine-functionalized surfaces. In cases where metal ions are present in adsorbent material, such as those with Fe- or Cu-based adsorbent surfaces, the carboxylate and heterocyclic groups can form inner–sphere complexes with the metal ions. Ionic strength and pH conditions can modulate the adsorption. However, the interaction mechanisms at the molecular scale are often inadequately described in the context of adsorption. The knowledge gap in this aspect is critical. Fundamental molecular interactions between drug molecules and the surface of adsorbent media (
Table 7) and, four adsorption isotherm models were employed, and their corresponding parameters were evaluated as shown in
Table 8.
93 A comparative evaluation of adsorption kinetics and isotherm models for pharmaceutical contaminants is shown in
Table 9. The major types of adsorbents used in industrial applications are presented in
Table 10.
94
 |
| | Fig. 9 (a) Dye adsorption over different kinds of designed materials92 (Adapted under a Creative Commons CC BY Attribution 4.0 International License, Haleem, A., et al., A comprehensive review on adsorption, photocatalytic and chemical degradation of dyes and nitro-compounds over different kinds of porous and composite materials, Molecules, 2023, 28(3), 1081). (b) Pharmaceutical waste: sources and consequences43 (Adapted under a Creative Commons CC BY Attribution 4.0 International License, Aziz, K., et al., Recent advances in nanomaterial-based adsorbents for removal of pharmaceutical pollutants from wastewater, Materials Horizons, 2025). (c) Preparation process involving solidification methods for composite adsorbents95 (Adapted under a Creative Commons CC BY Attribution 4.0 International License, Zhang, X., et al., Influential factors and optimization analysis of adsorption refrigeration system performance, AIP Advances, 2020, 10(10)). | |
Table 7 Molecular-level interactions between pharmaceuticals and adsorbent surfaces
| Drug functional group |
Representative drugs |
Adsorbent surface site |
Dominant interaction type |
pH/ionic strength influence |
References |
| Aromatic rings |
Diclofenac, ibuprofen, ciprofloxacin |
Graphene-basal planes, biochar |
π–π stacking |
Weak pH dependence, minimal ionic strength |
96 |
| Carboxylate (–COO−) |
Diclofenac, ibuprofen, naproxen |
–OH, –COOH, metal sites |
Electrostatic interaction |
Strongly pH-dependent |
97 |
| Amine |
Ciprofloxacin, amoxicillin |
–COOH, NH2 groups |
Hydrogen bonding |
Enhanced low pH |
98 |
| Heterocyclic |
Sulfonamides, fluoroquinolones |
Fe/MOF metal sites |
Metal–ligand coordination |
Favored around neutral pH |
99 |
| Hydroxyl |
Tetracycline |
–OH, –COOH |
Hydrogen bonding |
Best near neutral pH |
100 |
| Multiple functional group |
Ciprofloxacin, tetracycline |
GO-chitosan, Fe–Cu hybrids |
Synergistic electrostatic coordination |
Narrow optimal pH |
97 |
Table 8 Four isotherm models adopted in this work and their parameters93 (Adapted with permission from Elsevier (Özkaya, J. Hazard. Mater., 2006), copyright 2005)
| Isotherm |
Model |
| Freundlich |
qe = kf·Ce1/n |
| Langmuir |
 |
| Redlich–Peterson |
 |
| Toth |
 |
Table 9 Comparative overview of kinetic and isotherm models for pharmaceutical adsorption
| Adsorbent |
Pharmaceutical compounds |
Kinetic model |
Rate constant |
Isotherm model |
qmax (mg g−1) |
References |
| Activated carbon (AC) |
Tetracycline |
PSO |
k2 ∼ 0.001–0.01 g mg−1 |
Langmuir |
200–450 |
101 |
| Metal–organic framework (MOF-235) |
Ciprofloxacin |
PSO |
k2 ∼ 0.005–0.02 g mg−1 |
Langmuir |
300–600 |
102 |
| Graphene oxide (GO) |
Amoxicillin |
PSO |
k2 ∼ 0.002–0.015 g mg−1 |
Langmuir |
50–200 |
103 |
| Biochar |
Sulfamethoxazole |
PSO |
k2 ∼ 0.001–0.015 g mg−1 |
Langmuir |
150–200 |
103 |
| Magnetic nanocomposite |
Diclofenac |
PSO |
k2 ∼ 0.003–0.012 g mg−1 |
Langmuir |
180–400 |
103 |
| Zeolite |
Ibuprofen |
PFO/PSO |
k2 ∼ 0.01–0.05 min−1 |
Langmuir |
40–150 |
104 |
Table 10 Basic types of industrial adsorbents94 (Adapted under a Creative Commons CC BY Attribution 4.0 International License, Crini, G., et al., Conventional and non-conventional adsorbents for wastewater treatment, Environmental Chemistry Letters, 2019, 17(1), 195–213)
| Carbon adsorbents |
Mineral adsorbents |
Other adsorbents |
Target pharmaceuticals |
Adsorption capacity (mg g−1) |
References |
| Activated carbon |
Silica gels |
Synthetic polymers |
Diclofenac, ibuprofen |
120–350 |
105 |
| Activated carbon fiber |
Activated alumina |
Composite adsorbents (mineral carbons) |
Mixed pharmaceuticals |
150–400 |
106 |
| Carbonaceous materials |
Zeolites, clay minerals, pillared clays and inorganic nanomaterials |
|
Antibiotics, NSAIDs, ciprofloxacin |
30–210 |
107 |
| Quinolones |
50–300 |
| Fullerenes |
Metal hydroxides |
Mixed adsorbents |
NSAIDs, antibiotics |
100–260 |
107 |
| Molecular carbon sieves |
Metal oxides |
|
Tetracycline, SMX |
60–240 |
108 |
Activated carbon (AC) possesses high porosity and an extensive surface area; thus, the material is very effective at adsorption. Due to its unique properties, it can function as a catalyst or serve as a substrate for other catalysts. These characteristics, such as high porosity and catalytic and adsorptive capabilities, result directly from the activation it undergoes. Activated carbon is the most popular material used to remove dyes from wastewater. Dyes are commonly used in the textile industry, and colored wastewater is a significant problem. One of the characteristic features of activated carbon is its adsorption capacity, which refers to the amount of dye that it can remove.109 Renewable, biodegradable and nontoxic precursors are mainly used to develop bio-derived carbon nanostructures. Some examples of bio-derived carbon nanostructures are cellulose, graphene obtained from lignin, fullerene, and carbon nanodots derived from plant extracts.110 Fig. 9(b)) Pharmaceutical waste: sources and consequences.43 Biochar is produced via the fast pyrolysis of biomass, which can be sourced from various quarters, including forest materials such as sawdust, bark, wood shavings, and agricultural waste, such as bagasse and wheat straw. It is a solid biofuel that possesses a high heat value and serves as fuel in kilns and boilers.111 Fig. 9(c) preparation process of solidification methods for composite adsorbents.95 There are several carbonization processes by which biochar may be prepared. Among them, well-known techniques involve pyrolysis, pyro-gasification, and gasification. Various techniques used to synthesize the material include hydrothermal carbonization, torrefaction, and microwave pyrolysis. The material structure, surface chemistry, and operational conditions are optimized to maximize pharmaceutical uptake and stability. Thus, rational system engineering is of critical importance in translating advanced adsorbent materials into effective pharmaceutical treatment technologies in a scalable way.
Adsorption kinetic models
Adsorption kinetics are crucial because they determine how fast adsorption proceeds and help identify the main factors controlling the rate. (a) Pseudo-first-order: the PFO model, developed by Lagergren, is a non-reversible kinetic model. This model assumes a direct proportionality between the rate of adsorption and the number of unoccupied active sites on the adsorbent. The model is represented by the following non-linear equation:72where qt is the amount of adsorbate adsorbed at time t, qe is the amount of adsorbate adsorbed at equilibrium, and k1 is the pseudo-first-order rate constant.| | |
ln(qe − qt) = qe − k1t
| (5) |
where k1 is the apparent rate constant (L min−1), qt and qe (mg g−1) are the adsorption capacity at time t and at equilibrium. Its linearized form is also presented below: 72
(b) Pseudo-second-order: the PSO model of Ho and McKay is based on the premise that chemisorption, a chemical mechanism involving the sharing or exchanging of electrons, is the rate-determining step in adsorption. The PSO model equation can be expressed in eqn (6).72
| |
 | (6) |
The pseudo-second order and intraparticle diffusion models are the most commonly applied because they reflect the combined effects of surface reactions and mass transfer. In this respect, appropriate kinetic modeling is an important step, allowing a valid comparison between different adsorbents and enabling the selection and design of an efficient adsorption system. Due to outstanding physicochemical properties, MOFs and COFs have received multidisciplinary research interests. The excellent properties of COFs and MOFs and their derivatives are represented in CO2 reduction reactions and environmental pollutant treatments through strategies on sorption, photocatalysis, and electrocatalysis.112 Metal organic frameworks, often referred to as porous coordination polymers (PCPs), are a novel class of crystalline porous materials. They can be formed by the linkage of metal ions with organic molecules (ligands) to give a unique, highly structured framework.113 Fig. 10(a) Properties and applications of multifunctional MOFs.114 Of all the applications of MOFs, catalysis is one of the most extraordinary and fastest-growing areas. This is mainly due to several advantages of MOFs as catalysts. It is possible to design the framework to contain various types of active sites, metal centers, and functional groups on the organic ligands. The active sites are densely and homogeneously distributed within the whole material, which significantly enhances the activity and lifetime of the catalyst. The extensive network of pores and open channels allows reactants to reach every single active site, increasing efficiency. It is easy to control how the reactant molecules interact with the MOF. Having the form of a solid porous material, MOFs could easily be recovered and reutilized, making them an excellent choice for use as a recyclable catalyst.115 Fig. 10(b) various strategies for the synthesis of MOFs/Polymer composites.116 Several approaches are adopted to synthesize MFCs from the Fe3O4 magnetic nanoparticles and metal–organic frameworks, resulting in materials with unique design principles. Composites created from various combinations of nanomaterials and MOFs offer a huge range of functions and characteristics. This is the capability that makes MOF-based composites of interest for a variety of purposes. The large surface area makes MOF-based composites ideal for catalysis and adsorption processes. Porous MOF-based composites are excellent for gas storage and separation. MOF-based composites are often very chemically stable and can be effectively used in harsh conditions. The properties and performance of MOF-based composites are directly defined by the MOFs from which they were made.117 MOFs are crystalline, three-dimensional structures composed of organic ligands and metal ions. Because of the unique hybrid structure comprising inorganic and organic components, MOFs have become highly tunable. For this reason, they find applications in catalysis, water purification, and the removal of gases and toxic chemicals. Because of the crystalline, porous, and fragile nature of MOFs, their properties are very different from those of polymers, which are pliable, malleable solids that are processed by processing routes. Recently, crystalline MOFs have been hybridized with flexible polymers to create new materials with combined features of both.116 Covalent organic frameworks (COFs) are crystals made from light elements (such as carbon, hydrogen, oxygen, nitrogen, and sulfur) and are characterized by their long-range ordered structure, controlled pore sizes, high degree of crystallinity, good thermal stability, large specific surface area, and low density, and they have received considerable attention for applications in various areas. However, their application and large-scale synthesis are hindered because the powdered materials are not recyclable.118 The biocomposites are considered efficient adsorbents to remove contaminants from wastewater. Bentonite showed good removal behavior for most common industrial pollutants due to its surface being negatively charged. It is generally functionalized with other materials so that the drawbacks can be overcome and its adsorption efficiency performance can be enhanced.119 Fig. 10(c) biopolymer-based composite from agricultural waste biomass.120 Molecular-level strategies for elucidating the mechanisms of pharmaceutical pollutant removal are summarized in Table 11. The benefits and drawbacks of commonly used materials for removing pharmaceutical contaminants are presented in Table 12. Additionally, Table 13 outlines various agricultural wastes utilized for biochar production and their effectiveness in adsorbing antibiotics from water, as well as approaches for the removal of pharmaceuticals from surface water.121
 |
| | Fig. 10 (a) Properties and applications of multifunctional MOFs114 (Adapted under a Creative Commons CC BY Attribution 4.0 International License, Prajapati, M., et al., Recent advancement in metal–organic frameworks and composites for high-performance supercapacitors, Renewable and Sustainable Energy Reviews, 2023, 183, 113509). (b) Various strategies for the synthesis of MOFs/Polymer composites116 (Adapted with permission from Elsevier (Deeraj, J. S. J., Raman, Asok, Paul, Saritha, and Joseph, Surfaces Interfaces, 2023), copyright 2023). (c) Biopolymer-based composite from agricultural waste biomass.120 Molecular-level strategies for elucidating the mechanisms of pharmaceutical pollutant removal are summarized in Table 11. The benefits and drawbacks of commonly used materials for removing pharmaceutical contaminants are presented in Table 12. Additionally, Table 13 outlines various agricultural wastes utilized for biochar production and their effectiveness in adsorbing antibiotics from water, as well as approaches for the removal of pharmaceuticals from surface water.121. (Adapted under a Creative Commons CC BY Attribution 4.0 International License, TG, Y. G., et al., Biopolymer-based composites: an eco-friendly alternative from agricultural waste biomass, Journal of Composites Science, 2023, 7(6), 242). (d) Nanoparticles, nanofibers and nanoclays122 (Adapted under a Creative Commons CC BY Attribution 4.0 International License, Omanović-Mikličanin, E., et al., Nanocomposites: a brief review, Health and Technology, 2020, 10(1), 51–59). | |
Table 11 Molecular-level approaches for understanding pharmaceutical pollutant removal mechanisms
| Approach |
Techniques |
Insights provided |
References |
| Molecular-level interaction mechanism |
Molecular interaction |
Explains pollutant-adsorbent catalyst interactions in detail |
123 |
| Spectroscopic characterization |
FTIR, XPS, NMR, UV-Vis |
Functional group changes, surface interactions, oxidation states |
124 |
| Theoretical calculations |
DFT simulations |
Adsorption sites, binding energies, and electronic interactions |
125 |
| Combined approach |
Experimental + computational |
Comprehensive understanding of interaction mechanisms |
126 |
Table 12 Advantages and limitations of common materials for pharmaceutical pollutant removal
| Material type |
Key advantages |
Key limitations |
| Activated carbon (AC) |
High surface area, strong adsorption, widely available |
Costly regeneration, nonselective, potential secondary pollution |
| Biochar/hydrochar |
Renewable, low-cost, tunable surface chemistry |
Variable quality, lower adsorption than AC |
| Metal oxides (Fe, Cu, TiO2, etc.) |
Effective catalytic degradation, reusable, stable |
Agglomeration, limited selectivity, possible metal leaching |
| Bimetallic composites (FE-CU, FE-NI, and ETC) |
Synergistic effects, enhanced catalytic activity, fast removal |
Complex synthesis, higher cost, metal leaching risk |
| Polymeric adsorbents |
Selective adsorption, functional group tunability |
Limited thermal/chemical stability, expensive, slower kinetics |
| Magnetic nanomaterials |
Easy magnetic separation, high surface area |
Synthesis cost, aggregation, potential toxicity |
Table 13 Agricultural wastes used for biochar production and the adsorption of antibiotics from water121 (Adapted under a Creative Commons CC BY Attribution 4.0 International License, Onyekachukwu, E., et al., Low-Cost Adsorbents for the Removal of Pharmaceuticals from Surface Waters, Water, 2025, 17(17), 2619)
| Biochar feedstock |
Antibiotic |
Modification agent |
Specific surface area (m2 g−1) |
pH |
Adsorption capacity (mg g−1) |
References |
| Banana |
Furazolidone |
FeCl2·4H2O |
116.97 |
7.5 |
37.45 |
127 |
| Bamboo |
Sulfamethoxazole |
Fe2O3 |
61.48 |
6 |
212.8 |
128 |
| Corn stalk |
Enrofloxacin |
KOH |
22.69 |
3 |
58.29 |
129 |
| Pine wood |
Daptomycin |
Fe3O4 |
634.218 |
4 |
212.77 |
130 |
| Poplar |
Tetracycline |
KOH |
1.61 |
5 |
21.17 |
131 |
| Reed stalk |
Florfenicol |
FeSO4·7H2O |
254.6 |
6 |
9.29 |
132 |
Some of the vital parameters that need to be tested are as follows: (1) batch-to-batch consistency. (2) Testing for hazards and risks. (3) Testing the robustness of the process under variable conditions. (4) Yield or the extent of the reaction regarding the conversion of reactants to products.133 The formed composite material has been successfully used in drug delivery and demonstrated to be biocompatible.133 Fig. 10(d) nanoparticles, nanofibers and nanoclays.122
Nearly half of the studies (45%) focused on using natural clay minerals as adsorbents. The other 55% were dedicated to creating new, enhanced adsorbents by modifying or combining clay minerals using different techniques. This included: biopolymers (16%), metal pillared-clay minerals (11%), and organocations (17%).134 Fig. 11(a) sources and chemical structures of natural biopolymers used in bionanocomposite fabrication.135 In a water purification system, clay can remove about 70% of the waste. Removal of the final 30% can be achieved by utilizing activated carbon.136 The mechanisms of adsorption of the clay minerals through liquid and gaseous phases are: (i) physical and nonionic adsorption: weak physical forces are involved, such as van der Waals interactions. (ii) Ion exchange: an electrostatic process in which ions from the clay structure are exchanged with ions of the substance. (iii) Zeolite action: the ability of the porous clay to selectively trap and release molecules, similar to the actions of a zeolite.137 Such modifications render them very effective for the adsorption of organic molecules. Because of this, they are used to clean up organic-contaminated soils and water.138 Soil clay minerals act as a natural filter, cleaning water by removing pollutants through ion exchange and adsorption. This makes them effective and tough enough for use in a variety of environmental conditions.139 By changing the reaction conditions, many types of organoclays have been prepared.140 Pillared clays are another type of heterogeneous catalyst prepared from clays. Their wide applications in various organic reactions are due to their enhanced reactivity, porosity, thermal stability, reactivity and selectivity.141
 |
| | Fig. 11 (a) The molecular structures and sources of naturally derived biopolymer components for bionanocomposites135 (Adapted with permission from Elsevier (Xiong et al., Mater. Sci. Eng., R, 2018), copyright 2018). (b) Broad biopolymer classifications142 (Adapted with permission from Elsevier (Sharma, Malik and Jain, Mater. Today Commun., 2018), copyright 2018). (c) Synthesis of metal–organic frameworks143 (Adapted under a Creative Commons CC BY Attribution 4.0 International License, Aghayi-Anaraki, M., Safarifard, V., Fe3O4@ MOF magnetic nanocomposites: Synthesis and applications, European Journal of Inorganic Chemistry, 2020, 2020(20), 1916–1937). (d) Schematic of the synthesis and application of a MOF-inorganic material composite framework144 (Adapted with permission from Elsevier (Rani, Kasneryk and Opanasenko, Appl. Mater. Today, 2022), copyright 2022). | |
Limitations of surfactant-modified clay
Despite these advantages, surfactant modification of clay has certain disadvantages, including contaminant specificity, concentration dependency, and an unclear impact on surface area.145 Any polymer that is synthesized biologically or produced from biological materials is a biopolymer. Such polymers are biodegradable and often biocompatible. Microorganisms break these materials down completely into harmless natural elements, such as biomass, water and carbon dioxide, without producing any harmful byproducts during the process. Biopolymers may be divided into three types: (1) polysaccharide-based: examples include starch, chitosan and cellulose. (2) Protein-based: examples include whey protein, gelatin, and soy protein. (3) Microbial-derived: examples include polylactic acid and polyhydroxyalkanoates.146 Fig. 11(b) depicts the general categories of biopolymers.142 Agricultural byproducts and non-food waste materials form substantial feedstock for producing bioplastics. Manufacturing bioplastics from waste will increasingly enhance the sustainability of the agricultural economy, thus providing additional economic, social, and environmental benefits.147 Fig. 11(c) synthesis of metal–organic frameworks.143 Biocomposites derived from biopolymers are perfect for the creation of non-structural interior auto parts. In general, this creates the possibility to solve some of the most pressing environmental and economic problems.148 Fig. 11(d) schematic summary of the MOF inorganic material composite framework synthesis and application.144 Reinforcement is critical because these bio-based polymers naturally have high rates of water permeability and are biodegradable, which can limit their durability in construction applications. More than 100 types of agricultural waste are capable of generating a broad spectrum of biopolymers.149 Fig. 12(a) synthesis and types of chemically altered clay minerals used for the adsorption and removal of pollutants.145
 |
| | Fig. 12 (a) Modification types and synthesis steps of chemically modified clay minerals and adsorption of contaminants145 (Adapted under a Creative Commons CC BY Attribution 4.0 International License, Alomari, A. D. A., Chemically modified clay for adsorption of contaminants: trends, advantages and limitations—a concise review, International Journal of Environmental Analytical Chemistry, 2025, 105(10), 2302–2325). (b) Impact of heavy metals on the human body150 (Adapted under a Creative Commons CC BY Attribution 4.0 International License, Maftouh, A., et al., Comparative review of different adsorption techniques used in heavy metals removal in water, Biointerface Research and Applied Chemistry, 2023, 13(4)). (c) Schematic of the adsorption pathway and techniques150 (Adapted under a Creative Commons CC BY Attribution 4.0 International License, Maftouh, A., et al., Comparative review of different adsorption techniques used in heavy metals removal in water, Biointerface Research and Applied Chemistry, 2023, 13(4)). | |
The application of innovative technologies must be stimulated for processing the bio-based raw materials (agricultural waste) into valuable biofertilizers.151 Fig. 12(b) Impact of heavy metals on the human body.150 Sustainable agriculture in the 21st century will have to depend on research and development of new, renewable feedstocks for biopolymers in farming situations.152 Owing to their outstanding characteristics, bio-based polymers are widely utilized in various aspects. In medicine, they are commonly applied in regenerative medicine, bone implants, drug delivery, tissue engineering and regenerative medicine.153 The development of regenerative and clean sources of energy is an imperative approach to satisfy the increasingly growing demand for energy worldwide and to address the ecological concerns arising from the overexploitation of different resources.154 Fig. 12(c) illustrative flowchart of adsorption pathways and techniques.150 Surfactant-modified clays have enhanced affinity toward pharmaceutical pollutants; however, limited stability and risk of surfactant leaching reduce their practical use, and a decrease in performance is observed when the water matrices become more complex. Recent investigations have demonstrated that these constraints can adversely affect reusability and long-term operational efficiency. Thus, overcoming these limitations is still integral to developing surfactant-modified clays into effective pharmaceutical wastewater remediators.
Factors affecting adsorption efficiency
The effectiveness of liquid phase adsorption and, thus, the best way to run a water treatment process, is affected by various key factors. Adsorption efficiency is governed by several physicochemical parameters, including the surface chemistry and pore structure of the adsorbent, the nature of adsorbent–adsorbate interactions, solution pH, temperature, contact time, pressure, particle size, and the presence of competing ions or other co-existing species.155 Fig. 13(a) graphical representation of factors affecting adsorption efficiency.
 |
| | Fig. 13 (a) Graphical representation of factors affecting the adsorption efficiency. (b) Factors affecting the adsorption process. (c) Flowchart of the nature of factors affecting adsorption. (d) Mechanism of adsorption. | |
(1) Effect of solution pH: the pH is a crucial factor for effective adsorption because it influences how the adsorbent (the material doing the adsorbing) and the adsorbate interact. The optimal pH is necessary to achieve maximum extraction percentage for a given antibiotic. (2) Effect of contact time between adsorbate and adsorbent: With respect to this parameter, despite the goal of reducing operational time, a sufficiently long optimum contact time is necessary to attain maximum removal efficiency. This ensures that a sufficient amount of the substance, such as antibiotics, is removed. (3) Effect of the initial concentrations of adsorbate and adsorbent: the initial amounts of both the adsorbate (the substance being removed) and the adsorbent (the material doing the removal) determine when the system becomes saturated, a point known as breakthrough. Once breakthrough occurs, the adsorbate starts to pass through without being treated. Conversely, increasing the adsorbent dosage results in better removal, although this will also raise the operational cost.156 Fig. 13(b) factors affecting on adsorption process. (4) Surface chemistry: a key factor is surface chemistry, specifically the types and number of functional groups present. Surface functional groups form when unsaturated carbon atoms on a material's surface bond with atoms such as sulfur, hydrogen, oxygen, halogens and nitrogen. This process takes place during carbonization. (5) Effect of temperature: upon elevating the temperature within the adsorbent bed, the adsorption capacity decreases. This happens because the adsorbed molecules acquire more energy, allowing some to overcome the weak van der Waals forces. To reduce the risk of fire, continuous monitoring is required for carbon monoxide concentrations and temperature, as well as any operating parameters that directly impact thermal conditions.157 Fig. 13(c) flowchart of nature of factors affecting on adsorption. Fig. 13(d) mechanism of adsorption. (6) Effect of functional groups: a larger charge transfer shows a stronger interaction, corresponding to a higher adsorption energy. Despite the fact that no atomic rearrangement or destruction was observed, some models still showed adsorption energies higher than −50 kJ mol−1. When two functional groups are located next to each other on a surface, the combined effect generally results in a higher adsorption energy than that of a single, isolated functional group.158 Fig. 14(a) various forms of adsorbent–adsorbate interactions.159 (7) Effect of dosage of adsorbent: adsorption efficiency was measured at set time intervals for each dose, while all other conditions remained constant. The adsorption efficiency was evaluated as a function of time by systematically varying the adsorbent dosage.160 Fig. 14(b) analysis of experimental variables affecting adsorption capacity: this includes (a) pH, (b) adsorbent dose (at 50 mg L−1, 30 min, and 25 °C), (c) contact time (at 20 mg dose, pH 9, and 25 °C), and (d) initial CV concentration (at 20 mg dose, pH 9, and 30 min).161 Fig. 14(c) results of adsorption kinetics model fitting compared with the experimental kinetic data for (a) benzene and (b) toluene on activated carbon (AC). (Experimental conditions: 10–70 min adsorption time, 100 mL min−1 gas flow rate, 0.07 g adsorbent amount, and optimized initial concentration and temperature).162 Adsorption efficiency in pharmaceutical removal can be affected by the properties of the material itself, such as surface area, functional groups, and charges on the surface, and by the operational conditions, which include pH, contact time, temperature, and concentration of the substance to be removed. It has been revealed that significant roles are played by these factors in determining the efficiency of the adsorption process. Table 14 show the adsorption capacities and kinetic parameters and Table 15 represented recyclability and reusability of adsorbents.163
 |
| | Fig. 14 (A) Various forms of adsorbent–adsorbate interactions159 (Adapted with permission from Elsevier (Natarajan et al., Chemosphere, 2022), copyright 2022). (B) Analysis of experimental variables affecting adsorption capacity: effects of pH (a), adsorbent dose (Co–50 mg L; time–30 min; and T–25 °C) (b), contact time (dose–20 mg; pH–9; and T–25 °C) (c) and initial CV dye concentration (dose – 20 mg; pH–9; and time–30 min) (d) on adsorption capacity161 (Adapted under a Creative Commons CC BY Attribution 4.0 International License, Al-Shehri, H. S., et al., Effective adsorption of crystal violet from aqueous solutions with effective adsorbent: equilibrium, mechanism studies and modeling analysis, Environmental Pollutants and Bioavailability, 2021, 33(1), 214–226). (C) Adsorption kinetics model fitting results and experimental kinetics of benzene162 (Adapted under a Creative Commons CC BY Attribution 4.0 International License, Baytar, O., et al., High-performance gas-phase adsorption of benzene and toluene on activated carbon: response surface optimization, reusability, equilibrium, kinetic, and competitive adsorption studies, Environmental Science and Pollution Research, 2020, 27(21), 26191–26210). | |
Table 14 Adsorption capacities and kinetics
| Adsorbent type |
Pharmaceutical targets |
Max adsorption capacity (mg g−1) |
Kinetic model |
References |
| Wood-derived engineered sponge |
Tetracycline |
∼863.8 |
Pseudo second order |
22 |
| Wood-derived biochar |
Diclofenac |
∼350.0 |
Pseudo second order |
22 |
| Activated biochar (ASBS) |
Tetracycline |
∼402.7 |
Pseudo second order |
164 |
| Nitrogen-doped graphene |
Diclofenac |
∼278 |
Pseudo second order |
103 |
| Graphene oxide (GO) |
Trimethoprim |
∼218 |
Pseudo second order |
103 |
| Activated carbon |
Tetracycline |
∼909 |
Pseudo first order |
103 |
| Activated carbon (walnut) |
Diclofenac and others |
∼2.5–3.0 |
Pseudo first order |
103 |
| Chitosan-GO hydrogel |
Diclofenac |
∼129–131 |
Pseudo second order |
165 |
| Magnetic chitosan composites |
Carbamazepine |
∼95% removal in 25 min |
Pseudo second order |
165 |
| MOF-derived adsorbents |
Gemifloxacin |
∼876 |
Pseudo second order |
103 |
Table 15 Regeneration and reuse performance163 (Adapted under a Creative Commons CC BY Attribution 4.0 International License, Siyal, A. A., et al., A review of the developments in adsorbents for the efficient adsorption of ibuprofen from wastewater, RSC Advances, 2025, 15(23), 17843–17861)
| Adsorbent |
Regeneration method |
Number of cycles tested |
Retention |
References |
| Graphene oxide nanoparticles (GONPs) |
Methanol |
10 |
∼95.9% |
166 |
| Carbon nanospheres (CNs) |
DW wash |
6 |
∼16.9% |
167 |
| Pepper stem biochar |
NaOH |
4 |
∼22% |
168 |
| PMLE-E adsorbent |
Methanol |
8 |
∼22% |
169 |
Target pollutants and adsorption behavior
The widespread use of pharmaceuticals and dyes poses a global environmental risk with respect to their discharge. Annually, approximately 7 million tons of dyes and 100![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 to 200
000 to 200![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 tons of antibiotics are used worldwide.170 The primary routes of pharmaceutical contamination into water bodies are discharges from drug production plants and municipal sewage systems, and from hospital effluents. These compounds are considered hazardous. Advanced oxidation processes (AOPs), which include methods such as ozonation, the Fenton process, and photocatalysis, have been shown to effectively degrade synthetic contaminants.171 This has led to an increased focus on finding adsorbents that are highly efficient, safe, and affordable.172 Antibiotics are a type of pharmaceutical that often ends up in the environment, contaminating water and soil. Bacterial resistance occurs when bacteria that were once vulnerable to an antibiotic become resistant to it. This can be caused by the overuse of antibiotics and improper treatment of wastewater.173 As a result, the contaminated adsorbents then become a disposal concern, meaning that adsorption alone is not a complete solution for the issue of antibiotic residue.174 Continuous processes are typically more cost-effective than batch processes because batch processes have higher energy consumption due to start-up times and increased raw material costs, since materials are recycled less frequently. However, a key advantage of batch processing is the ability to combine multiple tasks into a single, larger unit.175 Continuous technology is considered advantageous for commercial production because it allows for flexible changes in production quantities and is easy to scale up. The use of continuous technology is expanding to include the production of active pharmaceutical ingredients (APIs) for both small molecules and biopharmaceuticals.176 The product is continuously harvested by removing the old medium and replacing it with fresh medium.177 Continuous production dramatically enhances the efficiency of the primary capture resin, resulting in a substantial 68% decrease in consumable costs.178 One advantage of batch processing is its flexibility. Conversely, continuous processes must operate year-round at a large scale to be profitable.179 Fig. 15(a) comparative schematics of monoclonal antibody production methods: illustrating the (A) traditional fed-batch process versus the (B) continuous manufacturing process.177 Used adsorbents are often treated as waste, but their disposal can be harmful to the environment because they may contain toxic, absorbed compounds. To address this, an environmentally friendly alternative is to desorb the pollutants and regenerate the adsorbents for reuse.180 Saturated adsorbents can be regenerated, and pollutants recovered using several methods, including chemical (acids, alkalis, other chemicals) regeneration. The maximum desorption capacities for these techniques were found to be 99.5%, 92.6%, 284%, 150%, and 66.61%, respectively.181 Adsorption using activated carbon is considered a superior way to treat both water and wastewater. It stands out from other chemical and physical techniques because of its highly efficient adsorption of diverse pollutants, its rapid adsorption rate, and its simple design.182 Adsorption techniques are particularly advantageous for environmental cleanup and sustainability because they utilize common and effective materials such as activated carbon, clay minerals, silica gel, zeolites, and polymers.183 In this regard, graphene materials are also very promising as next-generation conventional adsorbent replacements for the control of environmental pollution, representing very important scientific values. They feature strong adsorption capability and excellent recyclability.184 Regeneration processes allow used adsorbents to be retrieved and reused multiple times with no significant drop in performance. Reuse and regeneration, often achieved through an inexpensive process such as desorption, is crucial when adsorbents are used for wastewater treatment outside of their original location. Reusing adsorbents is beneficial because it reduces costs and the need for material disposal.185 Fig. 15(b) Possible mechanism of photocatalytic degradation.186,187 The repeatability and reusability tests demonstrate that the photocatalyst exhibits high structural and catalytic stability.188–190 Regeneration is a crucial part of the adsorption process from both economic and environmental perspectives. There are several regeneration methods that have yielded mixed results, including solvent washing, and thermal, chemical, and electrochemical regeneration. To make adsorption a more economical, valuable, and important method for treating wastewater, it is crucial to focus on regenerating and reusing the adsorbent, as well as recovering the solute.191 Percentage desorption was calculated as:192
000 tons of antibiotics are used worldwide.170 The primary routes of pharmaceutical contamination into water bodies are discharges from drug production plants and municipal sewage systems, and from hospital effluents. These compounds are considered hazardous. Advanced oxidation processes (AOPs), which include methods such as ozonation, the Fenton process, and photocatalysis, have been shown to effectively degrade synthetic contaminants.171 This has led to an increased focus on finding adsorbents that are highly efficient, safe, and affordable.172 Antibiotics are a type of pharmaceutical that often ends up in the environment, contaminating water and soil. Bacterial resistance occurs when bacteria that were once vulnerable to an antibiotic become resistant to it. This can be caused by the overuse of antibiotics and improper treatment of wastewater.173 As a result, the contaminated adsorbents then become a disposal concern, meaning that adsorption alone is not a complete solution for the issue of antibiotic residue.174 Continuous processes are typically more cost-effective than batch processes because batch processes have higher energy consumption due to start-up times and increased raw material costs, since materials are recycled less frequently. However, a key advantage of batch processing is the ability to combine multiple tasks into a single, larger unit.175 Continuous technology is considered advantageous for commercial production because it allows for flexible changes in production quantities and is easy to scale up. The use of continuous technology is expanding to include the production of active pharmaceutical ingredients (APIs) for both small molecules and biopharmaceuticals.176 The product is continuously harvested by removing the old medium and replacing it with fresh medium.177 Continuous production dramatically enhances the efficiency of the primary capture resin, resulting in a substantial 68% decrease in consumable costs.178 One advantage of batch processing is its flexibility. Conversely, continuous processes must operate year-round at a large scale to be profitable.179 Fig. 15(a) comparative schematics of monoclonal antibody production methods: illustrating the (A) traditional fed-batch process versus the (B) continuous manufacturing process.177 Used adsorbents are often treated as waste, but their disposal can be harmful to the environment because they may contain toxic, absorbed compounds. To address this, an environmentally friendly alternative is to desorb the pollutants and regenerate the adsorbents for reuse.180 Saturated adsorbents can be regenerated, and pollutants recovered using several methods, including chemical (acids, alkalis, other chemicals) regeneration. The maximum desorption capacities for these techniques were found to be 99.5%, 92.6%, 284%, 150%, and 66.61%, respectively.181 Adsorption using activated carbon is considered a superior way to treat both water and wastewater. It stands out from other chemical and physical techniques because of its highly efficient adsorption of diverse pollutants, its rapid adsorption rate, and its simple design.182 Adsorption techniques are particularly advantageous for environmental cleanup and sustainability because they utilize common and effective materials such as activated carbon, clay minerals, silica gel, zeolites, and polymers.183 In this regard, graphene materials are also very promising as next-generation conventional adsorbent replacements for the control of environmental pollution, representing very important scientific values. They feature strong adsorption capability and excellent recyclability.184 Regeneration processes allow used adsorbents to be retrieved and reused multiple times with no significant drop in performance. Reuse and regeneration, often achieved through an inexpensive process such as desorption, is crucial when adsorbents are used for wastewater treatment outside of their original location. Reusing adsorbents is beneficial because it reduces costs and the need for material disposal.185 Fig. 15(b) Possible mechanism of photocatalytic degradation.186,187 The repeatability and reusability tests demonstrate that the photocatalyst exhibits high structural and catalytic stability.188–190 Regeneration is a crucial part of the adsorption process from both economic and environmental perspectives. There are several regeneration methods that have yielded mixed results, including solvent washing, and thermal, chemical, and electrochemical regeneration. To make adsorption a more economical, valuable, and important method for treating wastewater, it is crucial to focus on regenerating and reusing the adsorbent, as well as recovering the solute.191 Percentage desorption was calculated as:192| |
 | (7) |
where Cad denotes the amount of metal ions adsorbed, and Cde is the concentration of metal ions desorbed.192 Fig. 15(c) desorption and regeneration process.193 Table 16 presents fundamental mechanisms involved in the adsorption of heavy metals onto engineered adsorbents.194
 |
| | Fig. 15 (a) Schematics of monoclonal antibody manufacturing processes: (A) traditional fed batch process, and (B) continuous manufacturing process177 (Adapted with permission from Elsevier (Madabhushi, Pinto and Lin, New Biotechnol., 2022), copyright 2022). (b) Possible mechanism of photocatalytic degradation186 (Adapted under a Creative Commons CC BY Attribution 4.0 International License, Titus, D., Samuel, E. J. J., Photocatalytic degradation of azo dye using biogenic SnO2 nanoparticles with antifungal property: RSM optimization and kinetic study, Journal of Cluster Science, 2019, 30(5), 1335–1345). (c) Desorption and regeneration processes.193 Table 16 presents fundamental mechanisms involved in the adsorption of heavy metals onto engineered adsorbents.194 (Adapted with permission from Elsevier (Bayuo et al., Adv. Colloid Interface Sci., 2024), copyright 2024). | |
Table 16 Adsorption mechanisms of heavy metals onto adsorbents194 (Adapted under a Creative Commons CC BY Attribution 4.0 International License, Wang, J., Guo, X., Adsorption kinetics and isotherm models of heavy metals by various adsorbents: An overview, Critical Reviews in Environmental Science and Technology, 2023, 53(21), 1837–1865)
| Adsorption mechanism |
Adsorption process |
References |
| Ion exchange |
Specific examples include the adsorption of modified biochar onto nanostructured zeolites |
194 |
| Electrostatic interaction |
Specific examples of adsorption applications include the removal of Cr onto protonated Fe2O3 surfaces, Cd onto MoS2 and metals onto specialized chelating resins and magnetic chitosan composites |
195 |
| Van der Waals interaction |
Specific adsorption applications include the removal of Cd onto MoS2 composites |
195 |
| Hydrogen bonding |
Specific applications demonstrated the adsorption of microplastics onto a chitosan magnetic composite |
196 |
| Establishment of strong bonds |
Specific applications demonstrated the adsorption of brown algae and fungi onto graphene oxide |
197 |
Photocatalytic degradation (PCD) is an increasingly important method for treating wastewater, particularly for water containing small amounts of stubborn organic pollutants. It offers several key advantages over other processes. Complete mineralization: it can break down pollutants entirely. No waste: it does not create a secondary waste disposal problem. Low cost: it is an economical option. Mild conditions: it operates effectively under mild temperatures and pressure. A good photocatalyst needs to have several key qualities: it must be photoactive, meaning it can be activated by light (either visible or near UV light), and it should be cheap and nontoxic.198 Fig. 16(a) basic process flow for common biochar modification techniques.199 Photocatalysis proven to be an effective method for the reduction of high-valent metal ions to their low-valent states as well as the degradation of organic pollutants under irradiation. Besides the selective interaction between target molecules and porous materials, other important parameters that can enhance the efficiency of photocatalysis include strong light absorption ability and efficient separation of electron–hole pairs.200 Photocatalytic reactions initiate a reaction when the catalyst absorbs a photon that has an energy higher than or equal to the band-gap energy (Ebg) of the catalyst.201 When a photocatalyst is exposed to UV light, an electron in the valence band gets a boost of energy and jumps to the conduction band, leaving behind a positively charged “hole” in the valence band. This process creates an electron–hole pair and is the initial step in the photocatalytic discoloration of a dye.202
| | |
Catalyst + hν → ecb− + hvb+
| (8) |
Here, e
cb− = electrons in the conduction band and h
vb+ = electron vacancy in the valence band.
202,203 AOPs developed for treating drinking water in the 1980s are today applied to a broad range of wastewaters. In AOP treatment, hydroxyl radicals (OH˙) or sulfate radicals (SO
4˙−) are generated. These powerful radicals can remove recalcitrant organic pollutants, trace contaminants, and even some inorganic substances.
204 These two main steps are common to all advanced oxidation processes, namely,
in situ production and their subsequent reaction with target pollutants.
205 Various techniques have been developed within this area of AOP, enabling the most appropriate options to be selected for particular treatment needs. Currently available cost analyses indicate that AOP systems are no more expensive than conventional pollutant removal technologies.
206 Fig. 16(b) mechanisms of biochar adsorption of pollutants in wastewater. (a) Mechanisms of biochar adsorption of heavy metals. (b) Mechanisms of inorganic salt adsorption by biochar. (c) Mechanisms of tetracycline adsorption by biochar.
199 Application development has been driven by a combination of the following: stricter regulations, pollution of water resources from agricultural and industrial activities, and the need for industries to meet effluent discharge standards.
207 Advanced oxidation processes (AOPs) encompass a range of techniques, including nonthermal plasmas, UV/O
3, photo-Fenton, UV/H
2O
2, supercritical water oxidation, radiolysis, sonolysis, Fenton and photocatalysis. In contrast, photochemical methods, such as photo-Fenton processes, generally require a precursor (catalyst) to generate these radicals.
208 Fig. 16(c) relationships between the three components of an adsorption system.
94 The ultimate goal is to fully mineralize the substances, yielding nontoxic final products such as carbon dioxide (CO
2), water (H
2O), and mineral acids (in cases where halogens are part of the pollutant).
209 Many municipalities employ AOPs as a supplementary step to eliminate persistent organic and inorganic contaminants. This helps traditional wastewater treatment plants comply with increasingly stringent environmental regulations.
210 Fig. 16(d) Management of spent adsorbents involves either regeneration to restore adsorption capacity or complete replacement of the material once adsorption sites are irreversibly exhausted.
94 Table 17 summarises representative chemical reactions involving advanced oxidation processes (AOPs).
210
 |
| | Fig. 16 (A) Simplified process scheme for common modified biochar preparation199 (Adapted under a Creative Commons CC BY Attribution 4.0 International License, Wang, Y., et al., Research status, trends, and mechanisms of biochar adsorption for wastewater treatment: a scientometric review, Environmental Sciences Europe, 2024, 36(1), 25). (B) Mechanisms of biochar adsorption of pollutants in wastewater. (a) Mechanisms of biochar adsorption of heavy metals. (b) Mechanisms of inorganic salt adsorption by biochar. (c) Mechanisms of tetracycline adsorption by biochar199 (Adapted under a Creative Commons CC BY Attribution 4.0 International License, Wang, Y., et al., Research status, trends, and mechanisms of biochar adsorption for wastewater treatment: a scientometric review, Environmental Sciences Europe, 2024, 36(1), 25). (C) Relationships between the three components of an adsorption system94 (Adapted under a Creative Commons CC BY Attribution 4.0 International License, Crini, G., et al., Conventional and non-conventional adsorbents for wastewater treatment, Environmental Chemistry Letters, 2019, 17(1), 195–213). (D) The two main strategies, regeneration step and replacement, that can be used to treat spent adsorbent after its usage94 (Adapted under a Creative Commons CC BY Attribution 4.0 International License, Crini, G., et al., Conventional and non-conventional adsorbents for wastewater treatment, Environmental Chemistry Letters, 2019, 17(1), 195–213). (E) Classifications of EPs that influence soil, atmosphere, aquatic life, animals, microbes, and people211 (Adapted under a Creative Commons CC BY Attribution 4.0 International License, Hashmi, Z., et al., Comparative analysis of conventional to biomass-derived adsorbent for wastewater treatment: a review, Biomass Conversion and Biorefinery, 2024, 14(1), 45–76). | |
Table 17 Representative reactions involving advanced oxidation process (AOP) mechanisms210 (Adapted under a Creative Commons CC BY Attribution 4.0 International License, Silva, J. A., Advanced Oxidation Process in the Sustainable Treatment of Refractory Wastewater: A Systematic Literature Review, Sustainability, 2025, 17(8))
| AOP method |
Representative reaction |
References |
| Ozonation |
O3 + H2O → 2OH + O2 |
212 |
| Fenton reaction |
Fe2+ + H2O2 → Fe3+ + ˙OH + OH− |
213 |
| Photo Fenton reaction |
Fe3+ + H2O + hν → Fe2+ + ˙OH + H+ |
214 |
| UV/H2O2 process |
H2O2 + hν → 2OH |
215 |
| Photocatalysis (TiO2-based) |
TiO2 + hν → e− + h+ + H2O → ˙OH e− + O2 → ˙O2˙– |
216 |
The two primary natural processes for eliminating hazardous compounds from water are photodegradation and biodegradation. Specifically, biodegradation involves the breakdown of pollutants, predominantly using bacteria and fungi naturally present in aquatic and soil environments.217 Biological treatment, utilizing microorganisms rather than chemicals, is more eco-friendly. However, it can only be used to treat particular types of organic pollutants effectively. Because of this limitation, combining different treatment methods is frequently essential to attain the required level of water purity. Bioremediation, a common biological wastewater treatment method, uses organisms, such as plants, fungi, and algae, to remove emerging contaminants.218 This metabolic activity enables the effective removal of a broad spectrum of contaminants.219 Fig. 16(e) classifications of EPs that influence soil, atmosphere, aquatic life, animals, microbes, and people.211
The Fenton process is recommended only for highly concentrated dimethyl sulphoxide (DMSO) pre-treatment or for the final polishing after biological treatment, especially when excess hydrogen peroxide is present.220 Fig. 17(a) the categorization of advanced oxidation processes is based on their primary components. Key abbreviations used in this classification are: ozonation, hydrogen peroxide, ultraviolet radiation, ultrasound energy and ferrous ion.221 Biological methods are the most effective for treating this type of wastewater. The use of a combination of anaerobic digestion followed by an aerobic system improves treatment efficiency while reducing both energy consumption and sludge production.222 Fig. 17(b) physical parameters affecting biological treatment efficiency.218 Fig. 17(c) applicability of water treatment technologies based on the amount of organic load.223 Adsorption behavior strongly depends on the physicochemical properties of the target pharmaceutical pollutants, including molecular structure, charge, and hydrophobicity. These properties govern interaction mechanisms such as electrostatic attraction, π–π interactions, and hydrogen bonding with adsorbent surfaces. Therefore, understanding pollutant-specific adsorption behavior is crucial for designing selective and high-performance adsorption systems. Table 18 presents a comprehensive summary of various pharmaceutical compounds, their applications, and their environmental impacts in aquatic systems.78
 |
| | Fig. 17 (a) Advanced oxidation processes (AOPs) classification. Abbreviations used: O3 ozonation; H2O2 hydrogen peroxide; UV ultraviolet radiation; US ultrasound energy; Fe2+ ferrous ion221 (Adapted under a Creative Commons CC BY Attribution 4.0 International License, Poyatos, J. M., et al., Advanced oxidation processes for wastewater treatment: state of the art, Water, Air, and Soil Pollution, 2010, 205(1), 187–204). (b) Physical parameters affecting biological treatment efficiency218 (Adapted with permission from Elsevier (Singh et al., J. Cleaner Prod., 2024), copyright 2024). (c) Applicability of water treatment technologies based on the amount of organic load223 (Adapted under a Creative Commons CC BY Attribution 4.0 International License, Sirés, I., et al., Electrochemical advanced oxidation processes: today and tomorrow. A review, Environmental Science and Pollution Research, 2014, 21(14), 8336–8367). (d) Improvements in biological treatment of flowback and produced water will require a thorough understanding of contaminants and microbial processes involved224 (Adapted under a Creative Commons CC BY Attribution 4.0 International License, Acharya, S. M., Chakraborty, R., Tringe, S. G., Emerging trends in biological treatment of wastewater from unconventional oil and gas extraction, Frontiers in Microbiology, 2020, 11, 569019). | |
Table 18 Summary of the wide array of pharmaceutical compounds (PCs), their use, and environmental effects in aqueous matrices78 (Adapted under a Creative Commons CC BY Attribution 4.0 International License, Chauhan, S., et al., Biochar-mediated removal of pharmaceutical compounds from aqueous matrices via adsorption, Waste Disposal & Sustainable Energy, 2023, 5(1), 37–62)
| Pharmaceutical class |
PCs |
Therapeutic use |
Adverse effects when left untreated in the environment |
References |
| Analgesic |
Ibuprofen (IBP) |
Analgesic drug and NSAID |
Endocrine-disrupting effects and neurological system damage in organisms, along with degradation of freshwater and groundwater quality |
225 and 226 |
| Ketoprofen (KETO) |
Analgesic drug and NSAID |
Aggregation in soil is a concern. Even more critically, aquatic organisms suffer long-term detrimental effects from continuous exposure to concentrations as low as nanograms to micrograms |
|
| Naproxen (NPX) |
| Diclofenac (DFC) |
NSAID |
Poisonous or damaging effects on a wide range of organisms |
| Acetaminophen (ACE) |
Antipyretic analgesic |
Due to the shedding of diverse metabolites, aquatic ecosystems face acute toxicity. A further significant consequence is the promotion of antibiotic resistance in bacterial populations |
| Antibiotic |
Ciprofloxacin (CFX) |
Synthetic antibiotic |
Adversely impacts surface quality |
227 and 228 |
| Sulfamethoxazole (SMX) |
Veterinary antibiotic and human |
Bioaccumulation in aquatic life drives the emergence of antibiotic-resistant genes throughout the ecosystem |
|
| Chlortetracycline (CTC) |
Veterinary antibiotic |
The effects include histological alterations in the gills of fish, often resulting from pro-oxidative activity (a stress response). Additionally, there is concurrent growth |
| Levofloxacin (LEV) |
Bacterial antibiotic |
The rise of antibiotic-resistant pathogens in humans and animals |
| Antimicrobial |
Triclosan (TCS) |
Antimicrobial ingredient |
Human impacts involve disorders of the endocrine system, the development of cancerous growths, and disturbances in thyroid hormone regulatory pathways |
229 and 230 |
| Anticonvulsant |
Carbamazepine (CBZ) |
Antiepileptic |
Its minimal degradation capacity results in its bioaccumulation and, consequently, its toxic effects |
231 and 232 |
| Stimulant drug |
Caffeine (CAF) |
Stimulant drug |
The substance negatively affects diverse marine and aquatic species, including sponges, bivalves, microalgae and corals |
233 and 234 |
| Antihypertensive |
Propranolol (PRO) |
Beta-blockers |
Due to the challenges associated with limiting its mobility in natural soil and sediment environments |
235 |
| Atenolol (ATO) |
The compound bioaccumulates in aquatic organisms and induces alterations in testosterone levels in male individuals |
| Metoprolol (MTP) |
Environmental and economic sustainability
The conversion of agricultural and biomass waste into valuable products is an important step within a circular bioeconomy for the sustainable use of resources. There is a need to evaluate these waste products to quantify their environmental impact and benefits.236 Fig. 17(d) effective advancement in the biological treatment of flowback and water necessitates a comprehensive understanding of the pollutants and the associated microbial processes.224 A life cycle assessment has been systematically used. This LCA was specifically designed to compare the environmental outcomes associated with producing adsorbents from different types of algae for producing specific adsorbents.237 The main aim was to find which life stages or processes for each adsorbent contributed the most to negative environmental impacts. The Life Cycle Assessment (LCA) in this study is based on specific assumptions regarding packaging, energy losses, transportation, bone sourcing, and alum waste management.238 In an LCA, the conclusions from the life cycle inventory (LCI) are organized into impact categories to assess environmental concerns: (1) climate change: this is a common impact category, especially in this case, because the production of both nanoadsorbents requires significant amounts of electricity. (2) Water use: this category accounts for total water required across the life cycle of the adsorbents, encompassing both primary production/application and all upstream activities (indirect processes). (3) Cumulative energy demand: the total energy required for both the foreground and background systems can change significantly as a product matures.239 Fig. 18(a) interpretation of life-cycle assessment.240 Scalability is a system's ability to grow in scale, magnitude, extent, and dimensions. The ability of a solution to be successfully duplicated elsewhere or later is known as replicability. Therefore, a scalability and replicability analysis for a smart grid solution is a predictive tool for forecasting implementation outcomes when the solution is applied to a larger scale (scalability) or transferred to a new location or time period (replicability). SRA analysis in smart grid projects highlights how the specific context of a solution influences its outcomes. SRA is a crucial tool for understanding how a smart grid solution's specific environment affects its outcomes. A full SRA involves: (i) identifying key contextual factors, and (ii) investigating the implications of their variation. Fig. 18(b) defining the scope of the current life cycle assessment model (cradle-to-gate).241 A number of dimensions can be considered in SRA analysis, including scalability in density, intranational replicability, scalability in size, and international replicability.242 With increased scalability, a system can exhibit improved stability and performance, coupled with reduced operational costs.243 Several metrics have been developed for distributed and parallel computing to quantify scalability.244 Based on the life cycle approach, the framework is supported by environmental indicators commonly adopted within a life cycle assessment study. Human toxicity potential measures toxicity to organisms on land.245 The interaction between humans and the environment is mainly symbiotic, and this stability has to be maintained. The relation among the parameters used for treatment showed a high significance level.246 Substantial work has been carried out to understand the specific mechanisms by which antibiotics attach to natural soils.247 Fig. 18(c) source of biosorbents.248 Surfactant-modified clays and smart adsorbents have high potential regarding their environmental and economic stability and could serve, therefore, as sustainable alternatives for the removal of pharmaceutical pollutants.
 |
| | Fig. 18 (a) Interpretation of life-cycle assessment240 (Adapted under a Creative Commons CC BY Attribution 4.0 International License, Pereira, P. H. F., et al., Prospective life cycle assessment (LCA) of activated carbon production, derived from banana peel waste for methylene blue removal, Adsorption, 2024, 30(6), 1081–1101). (b) System boundary for the cradle-to-gate LCA modelling of the present study241 (Adapted with permission from Elsevier (Nandikes et al., J. Ind. Eng. Chem., 2025), copyright 2025). (c) Source of biosorbents248 (Adapted under a Creative Commons CC BY Attribution 4.0 International License, Almeida-Naranjo, C. E., et al., Transforming waste into solutions: Raw and modified bioadsorbents for emerging contaminant removal, Journal of Environmental Chemical Engineering, 2025, 13, 116720). | |
Current challenges and research groups
In real-world applications, competitive adsorption is a common occurrence that significantly impacts an adsorbent's overall effectiveness. Even with competitive adsorption, the total amount of each metal ion adsorbed decreases, but the order of adsorption capacity remains the same as in single-metal systems. It is also considered that each metal species exhibits a different degree of penetrability into the porous structure of the adsorbent.249 Due to the challenges of experimentally measuring simultaneous adsorption of several substances, the ideal adsorbed solution theory (IAST) was developed.250 The major perspectives are: (i) characterize raw feedstock and CBB (biochar-based material). (ii) Contrast single and ternary metal adsorption patterns and characteristics. (iii) Evaluate the heavy metal adsorption capacities using both Freundlich and Langmuir models on data from single- and ternary-metal adsorption isotherms. (iv) Select the most accurate adsorption model to predict CBB's heavy metal retention capacity. (v) Create three-dimensional simulation graphics based on the experimental data to visualize heavy metal adsorption behaviors under different conditions.251 MOFs have the ability to selectively adsorb certain kinds of molecules because of their central metal ions and organic ligands of different types and structures. This means we can control the way substrate molecules bind to the MOF by controlling these components.252 Poor isolation and removal of target ions are mainly due to competitive adsorption in such systems where the concentration of coexisting salt cations is high.249 Different methods have been used to enhance wastewater's biodegradability and decrease its organic load through a combination of oxidation and coagulation before subsequent biological treatment. The combined Fenton and biological treatment process showed no toxicity to the microbes that were used.253 The growing occurrence of pharmaceuticals in wastewater is a concern because they are difficult to biodegrade. These compounds are considered emerging contaminants (ECs) because conclusive evidence linking them to long-term health effects in wildlife and humans is still lacking.254 The mechanical properties of ice are evaluated using standardized testing procedures. It is important to note that these recommendations are subject to change as new ice-testing methods are developed.255 Although standardizing antibiotic susceptibility testing is challenging due to several factors, an ideal testing medium has yet to be established. There is ongoing debate regarding the optimal methods for determining inoculum size, selecting disk content, controlling incubation conditions, and accurately interpreting results for both laboratory and clinical applications.256 When a test is given, it involves two steps: measurement and classification. Measurement is simply the process of giving numerical values to something. The quality of a measurement tool is determined by the reliability and validity of its scores. Reliability is about consistency: it is how consistently a tool measures a specific idea or concept. Validity is about accuracy: it is how well the tool actually measures what it is supposed to measure.257 Fig. 19: challenges and research group about adsorption. Despite the remarkable advancements achieved, areas that still need improvement include the recyclability of the adsorbent materials to some extent, the efficiency of the adsorption process to remove the pollutants, and scalability.
 |
| | Fig. 19 Challenges and research focus of the group on adsorption. | |
Future perspectives
Among the available techniques, adsorption is widely used and considered an effective removal method of contaminants from wastewater due to several advantages: it is easy to use, highly efficient, and often involves the use of low-cost materials. Furthermore, many adsorbents can be renewed and recycled. Commonly used adsorbents include activated carbon, agricultural residues, and a variety of both natural and man-made polymers and their composites. While current adsorbents can be highly effective at removing pollutants, most of them have limitations such as low selectivity and low adsorption capacity, which restrict their real-world application. Because these traditional adsorbents do not consistently provide the necessary pollutant removal effectiveness, there is a need to develop smart adsorbents that can efficiently capture pollutants even at trace concentrations.12 Fig. 20(a) schematic comparison of adsorbent types: (A) display high selectivity but poor desorption efficiency, (B) offer efficient desorption but compromise adsorption selectivity, (C) smart adsorbents with molecular gates: achieve the dual benefits of selective adsorption, (D) gate operation: an illustration of the molecular gates reversibly opening and closing via the photoisomerization of the AB component.258 Fig. 20(b) Remediation through smart adsorbent.12 Fig. 20(c) process oriented smart adsorbents.259 Adsorptive separation is a key process in many areas, including the chemical, food, and environmental sectors. Adsorbents are essential to this process because they perform separation tasks by undergoing adsorption (taking up substances) and desorption (releasing them). Process-oriented smart adsorbents (POSAs) are designed and developed based on the specific needs of adsorption and desorption processes. The goal is to develop highly efficient adsorbents and promote their use in practical industrial separation processes.259 Conventional adsorbents featuring static pore structures cannot achieve the two critical challenges in adsorptive separation, namely energy-efficient regeneration and selective adsorption. Smart adsorbents, unlike conventional adsorbents with fixed pores, can tune their pore properties in response to a particular condition of interest. This enables them to achieve the two critical objectives in adsorptive separation; that is, selective adsorption and energy-saving desorption, which is not possible with conventional materials.260 In water treatment, a hybrid method combining adsorption and chemical precipitation has been proposed. Although adsorption is an effective technique on its own, its extraction capability of heavy metals is restricted based on the surface area provided for adsorption. This new hybrid approach has successfully delivered excellent performance. In this hybrid approach, the shape of the adsorbent material is critical for the separation of heavy metals. The key to this hybrid approach is that heavy metal ions form crystals on the adsorbent, which significantly increases the removal capacity. However, a major limitation is that this crystal growth can block the pores of the adsorbent, preventing heavy metal ions from entering and ultimately reducing the overall removal effectiveness.261 Fig. 20(d) integration with hybrid technologies.261 Fig. 20(e) future aspects Fig. 20(f) adsorbent influence.
 |
| | Fig. 20 (a) (A) Schematic of traditional microporous adsorbents possessing excellent performance in selective adsorption but inefficient during desorption; (B) schematic of traditional mesoporous adsorbents possessing excellent performance in desorption but at a cost in adsorption selectivity; (C) after anchoring photoregulated molecular gates, smart adsorbents with the benefit of selective adsorption and efficient regeneration; and (D) schematic of molecular gates that can reversibly close/open through photoisomerization of AB258 (Adapted under a Creative Commons CC BY Attribution 4.0 International License, Cheng, L., Yan, N., Shan, S. F., Liu, X. Q., Sun, L. B., Smart adsorbents with photoregulated molecular gates for both selective adsorption and efficient regeneration, ACS Applied Materials & Interfaces, 2016, 8(35), 23404–23411). (b) Remediation through smart adsorbents12 (Adapted under a Creative Commons CC BY Attribution 4.0 International License, Nazarzadeh Zare, E., et al., Smart adsorbents for aquatic environmental remediation, Small, 2021, 17(34), 2007840). (c) Process-oriented smart adsorbents259 (Adapted under a Creative Commons CC BY Attribution 4.0 International License, Jiang, Y., T. P., Liu, X. Q., Sun, L. B., Process-oriented smart adsorbents: tailoring the properties dynamically as demanded by adsorption/desorption, Accounts of Chemical Research, 2021, 55(1), 75–86). (d) Integration with hybrid technologies261 (Adapted with permission from Elsevier (YG and K., Chemosphere, 2024), copyright 2024). (e) Future aspects. (f) Adsorbent influence. | |
Hybrid adsorption cooling systems are more effective than standalone systems. A hybrid system combines different cooling techniques, such as an adsorption cycle with a compression cycle, to improve overall efficiency.262 A hybrid approach that includes adsorption and membranes could have complementary effects that solve problems associated with individual processes, such as the inability to remove anions and dissolved organic matter (DOM) from water, due to the advantages of the hybrid method regarding its smaller footprint and better performance towards water and wastewater treatment. The most favored hybrid systems are membrane adsorbents and membrane-adsorption bioreactors due to their unique operational benefits.263 Despite all this research, these new materials rarely result in commercial products. In this respect, the opportunities and challenges of commercializing adsorbent materials were assessed following an analysis of the current status of companies that were interviewed.264 The primary commercialization barriers for adsorbents include (1) material properties: quality, availability, lifetime, and adaptability; (2) economic considerations: costs of regeneration, disposal, and transportation are unacceptably high; and (3) the costs of regeneration, disposal, and transportation are unacceptably high, legal and testing requirements: difficulties around environmental legislation, testing, and validation.265 Factors influencing adsorption capacity and their impacts are presented in Table 19.266
Table 19 Factors affecting adsorption capacity and their effects266 (Adapted under a Creative Commons CC BY Attribution 4.0 International License, Satyam, S., P.S., Innovations and challenges in adsorption-based wastewater remediation: A comprehensive review, Heliyon, 2024, 10(9))
| Factor |
Effect |
References |
| Surface area |
Increases the adsorption capacity by raising the number of available active sites |
267 |
| Porosity |
Increases the capacity by providing more physical space for the molecules |
268 |
| Functional groups |
Capacity is enhanced via specific interactions with the intended target molecules |
269 |
| Molecular size |
Adsorption is favored for smaller molecules because they have better access to the adsorption sites |
270 |
| Polarity |
Adsorption capacity is higher for polar molecules when the adsorbent is also polar |
271 |
| Concentration |
Capacity increases directly with higher concentration until saturation is reached |
272 |
| Temperature |
Although elevated temperatures may boost capacity, the actual effects are highly variable |
273 |
| pH |
Impacts capacity by modifying the charge and the ionization ratio of the target molecule |
274 |
Effective regulatory policies regarding adsorbent safety are important for a number of reasons: they instill public confidence through transparency in communications to stakeholders and the public, they protect public health and ecological systems, they allow innovation to occur since the rules are responsive to new technologies, and they help to certify the responsible and eco-friendly usage of technology in society. These policies also promote safe innovation and responsible development. They ensure that the benefits of new technologies are maximized while their potential risks are minimized.275 Fig. 21: advancements in sustainable technologies in smart adsorbent for environment and pharmaceutical remediation. The following are directions for advancing smart adsorbent designs by including hybrid degradation strategies and optimizing cost-effective and environmentally sustainable approaches to removing pharmaceutical pollutants in future research.
 |
| | Fig. 21 Advancements in sustainable technologies in smart adsorbents for environmental and pharmaceutical remediation. | |
Conclusion
The application of smart adsorbents for pharmaceutical removal and degradation has enormous potential but still faces issues such as scaling up, fouling by complex water sources, regeneration costs, and proper disposal. The focus for future research work could include data-driven design and fabrication, the development and application of adsorption-coupled advanced oxidation processes, and the utilization of biodegradable and waste materials as raw materials. Validating their application and designing regeneration processes that are less energy-intensive will be imperative for practical applications. Application and implementation of smart adsorbents within existing treatment plants through tertiary units or membrane-based adsorption systems can be one practical method to fill the widespread application gaps for pharmaceutical wastewater treatment. By doing so, smart adsorbents can lead to efficient and economically feasible pharmaceutical waste treatment.
Conflicts of interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Data availability
Since this manuscript is a review paper, no new experimental data were generated or analyzed in the course of this work. All data supporting the findings of this study are available within the cited references, which have been duly acknowledged in the manuscript.
Acknowledgements
The authors extend their appreciation to the Deanship of Scientific Research at Northern Border University, Arar, KSA, for funding this research work through project number “NBU-FFR-2026-1243-02”.
References
- A. S. Mramba, P. P. Ndibewu, L. L. Sibali and K. Makgopa, A review on electrochemical degradation and biopolymer adsorption treatments for toxic compounds in pharmaceutical effluents, Electroanalysis, 2020, 32(12), 2615–2634 CrossRef CAS.
- B. Tiwari, S. Balasubramanian, Y. Ouarda, P. Drogui, R. D. Tyagi and G. Buelna, Review on fate and mechanism of removal of pharmaceutical pollutants from wastewater using biological approach, Bioresour. Technol., 2017, 224, 1–2 CrossRef CAS PubMed.
- P. Bottoni, S. Caroli and A. B. Caracciolo, Pharmaceuticals as priority water contaminants, Toxicol. Environ. Chem., 2010, 92(3), 549–565 CrossRef CAS.
- S. Barkade, et al., Removal of lead and copper from wastewater using Bael fruit shell as an adsorbent, Mater. Today: Proc., 2022, 53, 65–70 CAS.
- J. Wang and R. Zhuan, Degradation of antibiotics by advanced oxidation processes: An overview, Sci. Total Environ., 2020, 701, 135023 CrossRef CAS PubMed.
- M. S. U. Rehman, et al., Global risk of pharmaceutical contamination from highly populated developing countries, Chemosphere, 2015, 138, 1045–1055 CrossRef CAS PubMed.
- S. Mahire, et al., Accumulation and effects of persistent organic pollutants and biogeographical solutions: appraisal of global environment, Arabian J. Geosci., 2023, 16(10), 570 CrossRef CAS.
- F. Mansouri, et al., Removal of pharmaceuticals from water by adsorption and advanced oxidation processes: state of the art and trends, Appl. Sci., 2021, 11(14), 6659 CrossRef.
- R. B. González-González, et al., Persistence, environmental hazards, and mitigation of pharmaceutically active residual contaminants from water matrices, Sci. Total Environ., 2022, 821, 153329 CrossRef PubMed.
- J. Narsude, et al., Decontamination of Sewage Wastewater by an Aeration Method Utilizing Water Hardness-Reducing Spirulina platensis, Curr. Microbiol., 2025, 82(5), 196 CrossRef CAS.
- J. Rivera-Utrilla, et al., Pharmaceuticals as emerging contaminants and their removal from water. A review, Chemosphere, 2013, 93(7), 1268–1287 CrossRef CAS.
- E. Nazarzadeh Zare, et al., Smart adsorbents for aquatic environmental remediation, Small, 2021, 17(34), 2007840 CrossRef CAS.
- J. Wei, et al., Recent advances in metal organic frameworks for the catalytic degradation of organic pollutants, Collagen Leather, 2023, 5(1), 33 CrossRef CAS.
- V. Homem and L. Santos, Degradation and removal methods of antibiotics from aqueous matrices–a review, J. Environ. Manag., 2011, 92(10), 2304–2347 CrossRef CAS.
- Q. Yang, et al., Antibiotics: An overview on the environmental occurrence, toxicity, degradation, and removal methods, Bioengineered, 2021, 12(1), 7376–7416 CrossRef.
- Y. Song, et al., Enhancement of anaerobic treatment of antibiotic pharmaceutical wastewater through the development of iron-based and carbon-based materials: A critical review, J. Hazard. Mater., 2024, 135514 CrossRef CAS.
- R. Jumah, et al., Chemical structure dependent electrochemical degradation of antibiotics using boron-doped diamond electrodes, J. Environ. Chem. Eng., 2024, 12(6), 114115 CrossRef CAS.
- P. N. Karungamye, Methods used for removal of pharmaceuticals from wastewater: A review, Appl. J. Environ. Eng. Sci., 2020, 6(4), 412–428 Search PubMed.
- G. Z. Kyzas, F. Jie, N. K. Lazaridis, D. N. Bikiaris and K. A. Matis, New approaches on the removal of pharmaceuticals from wastewaters with adsorbent materials, J. Mol. Liq., 2015, 209, 87–93 CrossRef CAS.
- A. Intisar, R. Arooj, S. Hafeez, N. Hussain, M. Irfan, N. Shakeel, K. A. Gill, A. Iqbal, M. Janczarek and T. Jesionowski, Adsorptive and photocatalytic degradation potential of porous polymeric materials for removal of pesticides, pharmaceuticals, and dyes-based emerging contaminants from water, Chemosphere, 2023, 336, 139203 CrossRef CAS PubMed.
- S. A. Mangla D and S. Ikram, Critical review on adsorptive removal of antibiotics: Present situation, challenges and future perspective, J. Hazard. Mater., 2022, 425, 127946 CrossRef.
- C. B. Godiya and T. Leiviskä, Wood-derived adsorbents for the removal of pharmaceutical contamination from wastewater: a review, Environ. Chem. Lett., 2025, 1–31 Search PubMed.
- A. Saroa, et al., Nanotechnology-assisted treatment of pharmaceuticals contaminated water, Bioengineered, 2023, 14(1), 2260919 CrossRef PubMed.
- L. F. Stadlmair, L. Thomas, J. E. Drewes and J. Grassmann, Enzymes in removal of pharmaceuticals from wastewater: A critical review of challenges, Chemoshpere, 2018, 205, 649–661 CrossRef CAS.
- I. Michael, et al., Urban wastewater treatment plants as hotspots for the release of antibiotics in the environment: a review, Water Res., 2013, 47(3), 957–995 CrossRef CAS.
- D. Holder, et al., A dynamic and complex monochloramine stress response in Escherichia coli revealed by transcriptome analysis, Water Res., 2013, 47(14), 4978–4985 CrossRef CAS.
- C. Bellona, et al., Factors affecting the rejection of organic solutes during NF/RO treatment—a literature review, Water Res., 2004, 38(12), 2795–2809 CrossRef CAS PubMed.
- N. Tadkaew, et al., Removal of trace organics by MBR treatment: the role of molecular properties, Water Res., 2011, 45(8), 2439–2451 CrossRef CAS.
- L. Shearer, S. Pap and S. W. Gibb, Removal of pharmaceuticals from wastewater: A review of adsorptive approaches, modelling and mechanisms for metformin and macrolides, J. Environ. Chem. Eng., 2022, 10(4), 108106 CrossRef CAS.
- A. Dąbrowski, Adsorption—from theory to practice, Adv. Colloid Interface Sci., 2001, 93(1–3), 135–224 CrossRef.
- S. Radovic, P. Sabolc, L. Niemi, J. Prodanović and M. T. Sekulic, A review on sustainable technologies for pharmaceutical elimination in wastewaters—A ubiquitous problem of modern society, J. Mol. Liq., 2023, 383, 122121 CrossRef CAS.
- S. Satyam and S. Patra, Innovations and challenges in adsorption-based wastewater remediation: a comprehensive review, Heliyon, 2024, e29573 CrossRef CAS.
- M. Jain, M. Ackmez, D. L. Ramasamy, M. Najafi, M. Usman, R. Zhu, G. Kumar, S. Shobana, V. K. Garg and M. Sillanpää, Orption, degradation, and mineralization of emerging pollutants (pharmaceuticals and agrochemicals) by nanostructures: a comprehensive review, Environ. Sci. Pollut. Res., 2020, 27, 34862–34905 CrossRef CAS PubMed.
- F. Mansouri, C. Khawla, N. Roche and M. Ksibi, Removal of pharmaceuticals from water by adsorption and advanced oxidation processes: state of the art and trends, Appl. Sci., 2021, 11(14), 6659 CrossRef CAS.
- P. D. Randhir, Pharmaceuticals in the Surface Water of the USA: A Review, Curr. Environ. Health Rep., 2014, 1(2), 113–122 CrossRef.
- S. D. Kayode-Afolayan, E. F. Ahuekwe and O. C. Nwinyi, Impacts of pharmaceutical effluents on aquatic ecosystems, Sci. Afr., 2022, 17, e01288 CAS.
- M. B. Ahmed, L. Z. John, H. H. Ngo and W. Guo, Adsorptive removal of antibiotics from water and wastewater: progress and challenges, Sci. Total Environ., 2015, 532, 112–126 CrossRef CAS.
- J. Zhao, et al., Understanding the effect of H 2 O on CO 2 adsorption capture: mechanism explanation, quantitative approach and application, Sustain. Energy Fuels, 2020, 4(12), 5970–5986 RSC.
- V. I. Isaeva, et al., Modern carbon–based materials for adsorptive removal of organic and inorganic pollutants from water and wastewater, Molecules, 2021, 26(21), 6628 CrossRef CAS.
- N. L. Nxumalo and P. N. Mahlambi, Molecularly imprinted polymer-based adsorbents for the selective removal of pharmaceuticals from wastewater: Adsorption kinetics, isotherms, and thermodynamics studies, Ind. Eng. Chem. Res., 2023, 62(41), 16525–16544 CrossRef CAS.
- L. Huang, R. Shen and Q. Shuai, Adsorptive removal of pharmaceuticals from water using metal-organic frameworks: A review, J. Environ. Manage., 2021, 277, 111389 CrossRef CAS PubMed.
- A. H. Nordin, et al., Recent advances in using adsorbent derived from agricultural waste for antibiotics and non-steroidal anti-inflammatory wastewater treatment: A review, Separations, 2023, 10(5), 300 CrossRef CAS.
- K. Aziz, et al., Recent advances in nanomaterial-based adsorbents for removal of pharmaceutical pollutants from wastewater, Mater. Horiz., 2025, 6043–6068 RSC.
- T. Luukkonen and J. Teeriniemi, Is there progress in the adsorbent development for water treatment?, iScience, 2025, 28(8), 112993 CrossRef CAS PubMed.
- L. W. Wang, R. Z. Wang, and R. G. Oliveira, A review on adsorption working pairs for refrigeration, Renew. Sustain. Energy Rev., 2009, 13(3), 518–534 CrossRef CAS.
- S. Yadav, et al., Cutting-edge regeneration technologies for saturated adsorbents: a systematic review on pathways to circular wastewater treatment system, Environ. Monit. Assess., 2025, 197(2), 215 CrossRef PubMed.
- M. A. Alshehri and A. Pugazhendhi, Biochar for wastewater treatment: Addressing contaminants and enhancing sustainability: Challenges and solutions, J. Hazard. Mater. Adv., 2024, 16, 100504 Search PubMed.
- M. H. Dehghani, et al., Recent advances on sustainable adsorbents for the remediation of noxious pollutants from water and wastewater: A critical review, Arab. J. Chem., 2023, 16(12), 105303 CrossRef CAS.
- E. D. Duarte, et al., Adsorption of pharmaceutical products from aqueous solutions on functionalized carbon nanotubes by conventional and green methods: A critical review, J. Clean. Prod., 2022, 372, 133743 CrossRef CAS.
- E. R. Alírio, Chemical engineering and environmental challenges. Cyclic adsorption/reaction technologies: Materials and process together!, J. Environ. Chem. Eng., 2020, 8(4), 103926 CrossRef PubMed.
- G. L. Dotto and M. Gordon, Current scenario and challenges in adsorption for water treatment, J. Environ. Chem. Eng., 2020, 8(4), 103988 CrossRef CAS.
- K. Wychodnik, et al., Poultry farms as a potential source of environmental pollution by pharmaceuticals, Molecules, 2020, 25(5), 1031 CrossRef CAS.
- E. Syranidou, et al., Bisphenol-A removal by the halophyte Juncus acutus in a phytoremediation pilot: characterization and potential role of the endophytic community, J. Hazard Mater., 2017, 323, 350–358 CrossRef CAS.
- L. J. Carter, et al., Uptake of pharmaceuticals influences plant development and affects nutrient and hormone homeostases, Environ. Sci. Technol., 2015, 49(20), 12509–12518 CrossRef CAS.
- A. Chakraborty, et al., Pharmaceuticals and personal care products as emerging environmental contaminants: prevalence, toxicity, and remedial approaches, ACS Chem. Health Saf., 2023, 30(6), 362–388 CrossRef CAS.
- T. P. T. Pham, C.-W. Cho and Y.-S. Yun, Environmental fate and toxicity of ionic liquids: a review, Water Res., 2010, 44(2), 352–372 CrossRef CAS.
- Q. Yu, S. Deng and G. Yu, Selective removal of perfluorooctane sulfonate from aqueous solution using chitosan-based molecularly imprinted polymer adsorbents, Water Res., 2008, 42(12), 3089–3097 CrossRef CAS PubMed.
- E. Nilsen, et al., Contaminants of legacy and emerging concern in largescale suckers (Catostomus macrocheilus) and the foodweb in the lower Columbia River, Oregon and Washington, USA, Sci. Total Environ., 2014, 484, 344–352 CrossRef CAS.
- K. Shalini, et al., A review on pharma pollution, Int. J. PharmTech Res., 2010, 2(4), 2265–2270 Search PubMed.
- W. C. Li, Occurrence, sources, and fate of pharmaceuticals in aquatic environment and soil, Environ. Pollut., 2014, 187, 193–201 CrossRef CAS.
- N. Gupta and et al., Pollution by pharma industries: an overview, Environmental Remediation Technologies, Regulations and Safety, 2023, p. 55 Search PubMed.
- V. Chander, et al., Pharmaceutical compounds in drinking water, J. Xenobiot., 2016, 6(1), 5774 Search PubMed.
- E. Bustos Bustos, G. Antonia Sandoval and C. Martínez-Sánchez, Detection and treatment of persistent pollutants in water: general review of pharmaceutical products, ChemElectroChem, 2022, 9(12), e202200188 CrossRef CAS.
- M. Ortúzar, et al., Pharmaceutical pollution in aquatic environments: a concise review of environmental impacts and bioremediation systems, Front. Microbiol., 2022, 13, 869332 CrossRef.
- C. G. Daughton, Pharmaceuticals as environmental pollutants: the ramifications for human exposure, International encyclopedia of public health, 2008, 5, 66–102 Search PubMed.
- H. Rodríguez-Serin, et al., Literature review: Evaluation of drug removal techniques in municipal and hospital wastewater, Int. J. Environ. Res. Public Health, 2022, 19(20), 13105 CrossRef.
- B. I. Ashiwaju, C. G. Uzougbo and O. F. Orikpete, Environmental impact of pharmaceuticals: A comprehensive review, Matrix Sci. Pharma, 2023, 7(3), 85–94 CrossRef.
- M. D. Hernando, et al., Environmental risk assessment of pharmaceutical residues in wastewater effluents, surface waters and sediments, Talanta, 2006, 69(2), 334–342 CrossRef CAS PubMed.
- S. Aydin, et al., Antibiotics in hospital effluents: occurrence, contribution to urban wastewater, removal in a wastewater treatment plant, and environmental risk assessment, Environ. Sci. Pollut. Res., 2019, 26(1), 544–558 CrossRef CAS.
- K. M. AlAqad, M. M. Abdelnaby, A. Tanimu, I. Abdulazeez and A. M. Elsharif, Adsorbent materials for water treatment: a review of current trends and future challenges, Environ. Pollut Manag., 2025, 2, 1–3 CrossRef.
- B. Feier, et al., Electrochemical detection and removal of pharmaceuticals in waste waters, Curr. Opin. Electrochem., 2018, 11, 1–11 CrossRef CAS.
- M. A. Décima, et al., A review on the removal of carbamazepine from aqueous solution by using activated carbon and biochar, Sustainability, 2021, 13(21), 11760 CrossRef.
- N. Sheraz, et al., Comprehensive assessment of carbon-, biomaterial-and inorganic-based adsorbents for the removal of the most hazardous heavy metal ions from wastewater, RSC Adv., 2024, 14(16), 11284–11310 RSC.
- J.-P. Simonin, On the comparison of pseudo-first order and pseudo-second order rate laws in the modeling of adsorption kinetics, Chem. Eng. J., 2016, 300, 254–263 CrossRef CAS.
- M. Alaqarbeh, Adsorption phenomena: definition, mechanisms, and adsorption types: short review, Rhazes: Green Appl. Chem., 2021, 13, 43–51 Search PubMed.
- N. M. Aljamali, R. Khdur and I. O. Alfatlawi, Physical and chemical adsorption and its applications, Int. j. Thermodyn. Chem. Kinet., 2021, 7(2), 1–8 Search PubMed.
- S. E. Saied Azizian, Chapter 6 - Adsorption isotherms and kinetics, Interface Science and Technology, M. Ghaedi, Elsevier, 2021, vol. 33, pp. 445–509 Search PubMed.
- S. Chauhan, et al., Biochar-mediated removal of pharmaceutical compounds from aqueous matrices via adsorption, Waste Dispos. Sustain. Energy, 2023, 5(1), 37–62 CrossRef PubMed.
- D. Chen, et al., Activated biochar derived from pomelo peel as a high-capacity sorbent for removal of carbamazepine from aqueous solution, RSC Adv., 2017, 7(87), 54969–54979 RSC.
- S. Keerthanan, et al., Caffeine removal by Gliricidia sepium biochar: Influence of pyrolysis temperature and physicochemical properties, Environ. Res., 2020, 189, 109865 CrossRef CAS.
- P. V. dos Santos Lins, et al., Evaluation of caffeine adsorption by MgAl-LDH/biochar composite, Environ. Sci. Pollut. Res., 2019, 26(31), 31804–31811 CrossRef CAS PubMed.
- F. Sayin, S. T. Akar and T. Akar, From green biowaste to water treatment applications: Utilization of modified new biochar for the efficient removal of ciprofloxacin, Sustain. Chem. Pharm., 2021, 24, 100522 CrossRef CAS.
- X. Tan, et al., One-pot synthesis of carbon supported calcined-Mg/Al layered double hydroxides for antibiotic removal by slow pyrolysis of biomass waste, Sci. Rep., 2016, 6(1), 39691 CrossRef CAS PubMed.
- A. Ashiq, et al., Municipal solid waste biochar-bentonite composite for the removal of antibiotic ciprofloxacin from aqueous media, J. Environ. Manag., 2019, 236, 428–435 CrossRef CAS PubMed.
- H. N. Tran, et al., Innovative spherical biochar for pharmaceutical removal from water: Insight into adsorption mechanism, J. Hazard Mater., 2020, 394, 122255 CrossRef CAS PubMed.
- D. Zhang, et al., Contribution of different sulfamethoxazole species to their overall adsorption on functionalized carbon nanotubes, Environ. Sci. Technol., 2010, 44(10), 3806–3811 CrossRef CAS.
- H. Swenson and N. P. Stadie, Langmuir's theory of adsorption: A centennial review, Langmuir, 2019, 35(16), 5409–5426 CrossRef CAS PubMed.
- J. Ouyang, et al., Biomass-derived activated carbons for the removal of pharmaceutical mircopollutants from wastewater: A review, Sep. Purif. Technol., 2020, 253, 117536 CrossRef CAS.
- S. Azizian and S. Eris, Adsorption isotherms and kinetics, in Interface Science and Technology, Elsevier, 2021, p. 445–509 Search PubMed.
- G. S. ElShafei, et al., Kinetics and thermodynamics of adsorption of cadusafos on soils, J. Hazard. Mater., 2009, 172(2–3), 1608–1616 CrossRef CAS.
- A. N. Ebelegi, N. Ayawei and D. Wankasi, Interpretation of adsorption thermodynamics and kinetics, Open J. Phys. Chem., 2020, 10(3), 166–182 CrossRef CAS.
- A. Haleem, et al., A comprehensive review on adsorption, photocatalytic and chemical degradation of dyes and nitro-compounds over different kinds of porous and composite materials, Molecules, 2023, 28(3), 1081 CrossRef CAS PubMed.
- B. Özkaya, Adsorption and desorption of phenol on activated carbon and a comparison of isotherm models, J. Hazard Mater., 2006, 129(1–3), 158–163 CrossRef.
- G. Crini, et al., Conventional and non-conventional adsorbents for wastewater treatment, Environ. Chem. Lett., 2019, 17(1), 195–213 CrossRef CAS.
- X. Zhang, et al., Influential factors and optimization analysis of adsorption refrigeration system performance, AIP Adv., 2020, 10(10), 105315 CrossRef.
- S.-W. Nam, et al., Adsorption characteristics of diclofenac and sulfamethoxazole to graphene oxide in aqueous solution, Chemosphere, 2015, 136, 20–26 CrossRef CAS PubMed.
- H. Mahmoodi, M. Fattahi and M. Motevassel, Graphene oxide–chitosan hydrogel for adsorptive removal of diclofenac from aqueous solution: preparation, characterization, kinetic and thermodynamic modelling, RSC Adv., 2021, 11(57), 36289–36304 RSC.
- S. Mirizadeh, A. Converti and A. A. Casazza, Chitosan-based adsorbents for antibiotic removal with insights from batch and dynamic studies: A review, Int. J. Biol. Macromol., 2025, 148399 CrossRef CAS.
- A. Verma, et al., Graphene oxide/chitosan hydrogels for removal of antibiotics, Environ. Technol., 2025, 1–31 Search PubMed.
- D. Veclani, M. Tolazzi and A. Melchior, Molecular interpretation of pharmaceuticals' adsorption on carbon nanomaterials: theory meets experiments, Processes, 2020, 8(6), 642 CrossRef CAS.
- R. Li, et al., Adsorption of toxic tetracycline, thiamphenicol and sulfamethoxazole by a granular activated carbon (GAC) under different conditions, Molecules, 2022, 27(22), 7980 CrossRef CAS.
- X. Huang, et al., Adsorption characteristics of metal–organic framework MIL-101 (Cr) towards sulfamethoxazole and its persulfate oxidation regeneration, RSC Adv., 2018, 8(49), 27623–27630 RSC.
- T. O. Ajiboye, P. O. Oladoye and E. O. Omotola, Adsorptive reclamation of pharmaceuticals from wastewater using carbon-based materials: A review, Kuwait J. Sci. Eng., 2024, 51(3), 100225 CrossRef.
- M. Gutiérrez, P. Verlicchi and D. Mutavdžić Pavlović, Study of the influence of the wastewater matrix in the adsorption of three pharmaceuticals by powdered activated carbon, Molecules, 2023, 28(5), 2098 CrossRef.
- R. Pinto, et al., Raman spectroscopy applied to diatoms (microalgae, Bacillariophyta): Prospective use in the environmental diagnosis of freshwater ecosystems, Water Res., 2021, 198, 117102 CrossRef CAS PubMed.
- Y. Tang, et al., Significance of manganese resistant Bacillus cereus strain WSE01 as a bioinoculant for promotion of plant growth and manganese accumulation in Myriophyllum verticillatum, Sci. Total Environ., 2020, 707, 135867 CrossRef CAS.
- S. Chen, et al., Investigation of highly efficient adsorbent based on Ni-MOF-74 in the separation of CO2 from natural gas, Chem. Eng. J., 2021, 419, 129653 CrossRef CAS.
- W. Sun, et al., Enhanced gaseous acetone adsorption on montmorillonite by ball milling generated Si–OH and interlayer under synergistic modification with H2O2 and tetramethylammonium bromide, Chemosphere, 2023, 321, 138114 CrossRef CAS.
- D. Angın, E. Altintig and T. E. Köse, Influence of process parameters on the surface and chemical properties of activated carbon obtained from biochar by chemical activation, Bioresour. Technol., 2013, 148, 542–549 CrossRef PubMed.
- A. K. Singh, V. U. Pandit, and S. L. Sonawane, Bio-based raw materials for preparation of carbon nanostructures, in Bio-derived Carbon Nanostructures, Elsevier, 2024, pp. 25–63 Search PubMed.
- R. Azargohar and A. Dalai, Biochar as a precursor of activated carbon, Appl. Biochem. Biotechnol., 2006, 131(1), 762–773 CAS.
- H. Lu, et al., Metal–Organic Frameworks/Covalent–Organic Frameworks-Based Materials in Organic/inorganic Pollutant Elimination and CO2 Reduction Applications, ChemNanoMat, 2025, 11(9), e202500244 CrossRef CAS.
- Q.-L. Zhu and Q. Xu, Metal–organic framework composites, Chem. Soc. Rev., 2014, 43(16), 5468–5512 RSC.
- M. Prajapati, et al., Recent advancement in metal-organic frameworks and composites for high-performance supercapatteries, Renew. Sustain. Energy Rev., 2023, 183, 113509 CrossRef CAS.
- B. Li, J. G. Ma and P. Cheng, Integration of metal nanoparticles into metal–organic frameworks for composite catalysts: design and synthetic strategy, Small, 2019, 15(32), 1804849 CrossRef.
- B. D. S Deeraj, J. S. Jayan, Akhila Raman, A. Asok, R. Paul, A. Saritha and K. Joseph, A comprehensive review of recent developments in metal-organic framework/polymer composites and their applications, Surf. Interfaces, 2023, 43, 103574 CrossRef.
- H. Meskher, A critical review about metal organic framework-based composites: potential applications and future perspective, J. Compos. Compd., 2023, 5(14), 25–37 CrossRef.
- M. Xiao, et al., Recent Progress in Covalent Organic Framework-Based Membranes: Design, Synthesis, and Applications in the Fields of Energy and the Environment, ACS Macro Lett., 2025, 14(8), 1201–1220 CrossRef CAS.
- A. Grouli, et al., Removal of Pollutants from Wastewater Using Fe-Doped Hydroxyapatite Encapsulated with Alginate, Biointerface Res. Appl. Chem., 2023, 13(5), 438 CAS.
- Y. T. G. Gowda, et al., Biopolymer-based composites: an eco-friendly alternative from agricultural waste biomass, J. Compos. Sci., 2023, 7(6), 242 CrossRef.
- E. Onyekachukwu, et al., Low-Cost Adsorbents for the Removal of Pharmaceuticals from Surface Waters, Water, 2025, 17(17), 2619 CrossRef CAS.
- E. Omanović-Mikličanin, et al., Nanocomposites: a brief review, Health Technol., 2020, 10(1), 51–59 CrossRef.
- G. Murtaza, et al., A review of mechanism and adsorption capacities of biochar-based engineered composites for removing aquatic pollutants from contaminated water, Front. Environ. Sci., 2022, 10, 1035865 CrossRef.
- C. X. Yang, et al., Preparation and characterization of phosphoric acid-modified biochar nanomaterials with highly efficient adsorption and photodegradation ability, Langmuir, 2021, 37(30), 9253–9263 CrossRef.
- M. Kebir, et al., Pharmaceutical pollutants adsorption onto activated carbon: isotherm, kinetic investigations and DFT modeling approaches, C. R. Chim., 2022, 25(S2), 9–25 CrossRef.
- Q. Yang, et al., The adsorption behavior and mechanism of Cu (II), Fe (II) and Co (II) on straw biochar and their Fenton-like performance for ciprofloxacin decontamination, J. Environ. Manage., 2025, 373, 123962 CrossRef CAS.
- W. Wang, et al., Adsorption of enrofloxacin on acid/alkali-modified corn stalk biochar, Spectrosc. Lett., 2019, 52(7), 367–375 CrossRef CAS.
- J. Heo, et al., Enhanced adsorption of bisphenol A and sulfamethoxazole by a novel magnetic CuZnFe2O4–biochar composite, Bioresour. Technol., 2019, 281, 179–187 CrossRef CAS.
- R. Gurav, et al., Treatment of furazolidone contaminated water using banana pseudostem biochar engineered with facile synthesized magnetic nanocomposites, Bioresour. Technol., 2020, 297, 122472 CrossRef CAS.
- T. Ai, et al., Daptomycin adsorption on magnetic ultra-fine wood-based biochars from water: kinetics, isotherms, and mechanism studies, Bioresour. Technol., 2019, 273, 8–15 CrossRef CAS PubMed.
- L. Huang, et al., Reaction mechanism of zero-valent iron coupling with microbe to degrade tetracycline in permeable reactive barrier (PRB), Chem. Eng. J., 2017, 316, 525–533 CrossRef CAS.
- H. Zhao and Y. Lang, Adsorption behaviors and mechanisms of florfenicol by magnetic functionalized biochar and reed biochar, J. Taiwan Inst. Chem. Eng., 2018, 88, 152–160 CrossRef CAS.
- J. Y. Lim, et al., Biomedically-relevant metal organic framework-hydrogel composites, Biomater. Sci., 2023, 11(8), 2661–2677 RSC.
- G. G. Hacıosmanoğlu, et al., Antibiotic adsorption by natural and modified clay minerals as designer adsorbents for wastewater treatment: A comprehensive review, J. Environ. Manage., 2022, 317, 115397 CrossRef PubMed.
- R. Xiong, et al., Naturally-derived biopolymer nanocomposites: Interfacial design, properties and emerging applications, Mater. Sci. Eng. R Rep., 2018, 125, 1–41 CrossRef.
- A. A. Adeyemo, I. O. Adeoye and O. S. Bello, Adsorption of dyes using different types of clay: a review, Appl. Water Sci., 2017, 7(2), 543–568 CrossRef CAS.
- S. Barakan and V. Aghazadeh, The advantages of clay mineral modification methods for enhancing adsorption efficiency in wastewater treatment: a review, Environ. Sci. Pollut. Res., 2021, 28(3), 2572–2599 CrossRef CAS PubMed.
- M. Cruz-Guzmán, et al., Heavy metal adsorption by montmorillonites modified with natural organic cations, Soil Sci. Soc. Am. J., 2006, 70(1), 215–221 CrossRef.
- G. K. Sarma, S. SenGupta and K. G. Bhattacharyya, Methylene blue adsorption on natural and modified clays, Sep. Sci. Technol., 2011, 46(10), 1602–1614 CrossRef CAS.
- A. A. El-Zahhar and G. A. Al-Hazmi, Organically modified clay for adsorption of petroleum hydrocarbon, Eur. Chem. Bull., 2015, 4(2), 87–91 Search PubMed.
- A. Moronta, Catalytic and adsorption properties of modified clay surfaces, in Interface Science and Technology, Elsevier, 2004, pp. 321–344 Search PubMed.
- B. Sharma, P. Malik and P. Jain, Biopolymer reinforced nanocomposites: A comprehensive review, Mater. Today Commun., 2018, 16, 353–363 CrossRef CAS.
- M. Aghayi-Anaraki and V. Safarifard, Fe3O4@ MOF magnetic nanocomposites: Synthesis and applications, Eur. J. Inorg. Chem., 2020, 2020(20), 1916–1937 CrossRef CAS.
- P. Rani, V. Kasneryk and M. Opanasenko, MOF-inorganic nanocomposites: Bridging a gap with inorganic materials, Appl. Mater. Today, 2022, 26, 101283 CrossRef.
- A. D. A. Alomari, Chemically modified clay for adsorption of contaminants: trends, advantages and limitations-a concise review, Int. J. Environ. Anal. Chem., 2025, 105(10), 2302–2325 CrossRef CAS.
- T. Selvam, et al., Agricultural Waste-Derived Biopolymers for Sustainable Food Packaging: Challenges and Future Prospects, Polymers, 2025, 17(14), 1897 CrossRef CAS PubMed.
- N. Patel, M. Feofilovs and D. Blumberga, Agro biopolymer: A sustainable future of agriculture–state of art review, Rigas Teh. Univ. Zinat. Raksti, 2022, 26(1), 499–511 CAS.
- R. Phiri, et al., Development of sustainable biopolymer-based composites for lightweight applications from agricultural waste biomass: A review, Adv. Ind. Eng. Polym. Res., 2023, 6(4), 436–450 CAS.
- C. Maraveas, Production of sustainable and biodegradable polymers from agricultural waste, Polymers, 2020, 12(5), 1127 CrossRef CAS PubMed.
- A. Maftouh, et al., Comparative review of different adsorption techniques used in heavy metals removal in water, Biointerface Res. Appl. Chem., 2023, 13(4), 397 CAS.
- D. Puglia, et al., The opportunity of valorizing agricultural waste, through its conversion into biostimulants, biofertilizers, and biopolymers, Sustainability, 2021, 13(5), 2710 CrossRef CAS.
- C. Valle, et al., Harnessing agri-food waste as a source of biopolymers for agriculture, Appl. Sci., 2024, 14(10), 4089 CrossRef CAS.
- A. Vinayak, S. Sharma, and G. B. Singh, Biopolymers from industrial waste, in Biopolymers: Recent Updates, Challenges and Opportunities, Springer, 2022, pp. 129–149 Search PubMed.
- X. Pan, F. Kong and M. Xing, Spatial separation of photo-generated carriers in g-C3N4/MnO2/Pt with enhanced H2 evolution and organic pollutant control, Res. Chem. Intermed., 2022, 48, 2837–2855 CrossRef CAS.
- E. Rápó and S. Tonk, Factors affecting synthetic dye adsorption; desorption studies: a review of results from the last five years (2017–2021), Molecules, 2021, 26(17), 5419 CrossRef PubMed.
- M. B. Ahmed, et al., Adsorptive removal of antibiotics from water and wastewater: progress and challenges, Sci. Total Environ., 2015, 532, 112–126 CrossRef CAS.
- N. Le-Minh, et al., Factors affecting the adsorption of gaseous environmental odors by activated carbon: A critical review, Crit. Rev. Environ. Sci. Technol., 2018, 48(4), 341–375 CrossRef CAS.
- Q. Yin, et al., DFT study on the effect of functional groups of carbonaceous surface on ammonium adsorption from water, Chemosphere, 2022, 287, 132294 CrossRef CAS.
- R. Natarajan, et al., Understanding the factors affecting adsorption of pharmaceuticals on different adsorbents–A critical literature update, Chemosphere, 2022, 287, 131958 CrossRef CAS.
- S. Tandon and N. Nandini, Adsorption efficiency of carbon from treated sugarcane bagasse in removing chromium (vi) from aqueous solutions by optimization of adsorption parameters, J. Appl. Nat. Sci., 2009, 1(2), 155 Search PubMed.
- H. S. Al-Shehri, et al., Effective adsorption of crystal violet from aqueous solutions with effective adsorbent: equilibrium, mechanism studies and modeling analysis, Environ. Pollut. Bioavailab., 2021, 33(1), 214–226 CrossRef CAS.
- O. Baytar, et al., High-performance gas-phase adsorption of benzene and toluene on activated carbon: response surface optimization, reusability, equilibrium, kinetic, and competitive adsorption studies, Environ. Sci. Pollut. Res., 2020, 27(21), 26191–26210 CrossRef CAS.
- A. A. Siyal, et al., A review of the developments in adsorbents for the efficient adsorption of ibuprofen from
wastewater, RSC Adv., 2025, 15(23), 17843–17861 RSC.
- Z. Shirani, V. Carrasco-Navarro and J. Sorvari, Effective Adsorption of Pharmaceuticals by Plant Based-Activated Biochar, Water, Air, Soil Pollut., 2025, 236(13), 872 CrossRef CAS.
- D. K. Kowanga and W. N. Nyairo, A review on decontamination of aqueous systems using chitosan-derived adsorbents, Discover Environ., 2025, 3(1), 237 CrossRef CAS.
- P. Banerjee, et al., Application of graphene oxide nanoplatelets for adsorption of ibuprofen from aqueous solutions: evaluation of process kinetics and thermodynamics, Process Saf. Environ. Prot., 2016, 101, 45–53 CrossRef CAS.
- A. A. Alluhaybi, et al., Synthesis and characterization of carbon nanospheres for adsorption of ibuprofen from aqueous solution: Optimization by Box–Behnken design, J. Mol. Liq., 2023, 383, 122059 CrossRef CAS.
- A. Naima, et al., Development of a novel and efficient biochar produced from pepper stem for effective ibuprofen removal, Bioresour. Technol., 2022, 347, 126685 CrossRef CAS PubMed.
- M. Sözbir, et al., Renewable terpene-based highly porous polymer monoliths for the effective removal of persistent pharmaceuticals of tetracycline and ibuprofen, Microporous Mesoporous Mater., 2023, 354, 112509 CrossRef.
- J. Yang, S. Shojaei and S. Shojaei, Removal of drug and dye from aqueous solutions by graphene oxide: adsorption studies and chemometrics methods, npj Clean Water, 2022, 5(1), 5 CrossRef CAS.
- G. Z. Kyzas, et al., New approaches on the removal of pharmaceuticals from wastewaters with adsorbent materials, J. Mol. Liq., 2015, 209, 87–93 CrossRef CAS.
- D. França, et al., The versatility of montmorillonite in water remediation using adsorption: Current studies and challenges in drug removal, J. Environ. Chem. Eng., 2022, 10(2), 107341 CrossRef.
- S. S. Priya and K. Radha, A review on the adsorption studies of tetracycline onto various types of adsorbents, Chem. Eng. Commun., 2017, 204(8), 821–839 CrossRef CAS.
- X. Zheng, et al., Enhanced degradation of ciprofloxacin by graphitized mesoporous carbon (GMC)-TiO2 nanocomposite: Strong synergy of adsorption-photocatalysis and antibiotics degradation mechanism, J. Colloid Interface Sci., 2018, 527, 202–213 CrossRef CAS.
- A. Goršek and P. Glavič, Design of Batch Versus Continuous Processes: Part III: Extended Analysis of Cost Parameters, Chem. Eng. Res. Des., 2000, 78(2), 231–244 CrossRef.
- K. Matsunami, et al., A large-scale experimental comparison of batch and continuous technologies in pharmaceutical tablet manufacturing using ethenzamide, Int. J. Pharm., 2019, 559, 210–219 CrossRef CAS PubMed.
- S. R. Madabhushi, N. D. Pinto and H. Lin, Comparison of process mass intensity (PMI) of continuous and batch manufacturing processes for biologics, New Biotechnol., 2022, 72, 122–127 CrossRef CAS.
- O. Yang, S. Prabhu and M. Ierapetritou, Comparison between batch and continuous monoclonal antibody production and economic analysis, Ind. Eng. Chem. Res., 2019, 58(15), 5851–5863 CrossRef CAS.
- P. T. Benavides, J. Salazar and U. Diwekar, Economic comparison of continuous and batch production of biodiesel using soybean oil, Environ. Prog. Sustain. Energy, 2013, 32(1), 11–24 CrossRef CAS.
- N. El Messaoudi, et al., Regeneration and reusability of non-conventional low-cost adsorbents to remove dyes from wastewaters in multiple consecutive adsorption–desorption cycles: a review, Biomass Convers. Biorefinery, 2024, 14(11), 11739–11756 CrossRef CAS.
- T. Sithole, A review on regeneration of adsorbent and recovery of metals: adsorbent disposal and regeneration mechanism, S. Afr. J. Chem. Eng., 2024, 50(1), 39–50 Search PubMed.
- S. Lata, P. Singh and S. Samadder, Regeneration of adsorbents and recovery of heavy metals: a review, Int. J. Environ. Sci. Technol., 2015, 12(4), 1461–1478 CrossRef CAS.
- K. Obodo and U. O. Aigbe, The types, characteristics, and management options (reusability/recyclability/final disposal) of commonly used adsorbents in environmental sustainability, in Adsorption Applications for Environmental Sustainability, IOP Publishing, Bristol, UK, 2023, p. 25 Search PubMed.
- X. Pan, et al., Research progress of graphene-based nanomaterials for the environmental remediation, Chin. Chem. Lett., 2020, 31(6), 1462–1473 CrossRef CAS.
- Y. Liu, et al., Green adsorbents for environmental remediation: synthesis methods, ecotoxicity, and reusability prospects, Processes, 2024, 12(6), 1195 CrossRef CAS.
- D. Titus and E. J. J. Samuel, Photocatalytic degradation of azo dye using biogenic SnO2 nanoparticles with antifungal property: RSM optimization and kinetic study, J. Cluster Sci., 2019, 30(5), 1335–1345 CrossRef CAS.
- V. Pandit, et al., In situ preparation of a novel organo-inorganic 6, 13-pentacenequinone–TiO 2 coupled semiconductor nanosystem: a new visible light active photocatalyst for hydrogen generation, J. Mater. Chem. A, 2015, 3(8), 4338–4344 RSC.
- V. U. Pandit, et al., Novel functionality of organic 6, 13-pentacenequinone as a photocatalyst for hydrogen production under solar light, Environ. Sci. Technol., 2014, 48(7), 4178–4183 CrossRef CAS PubMed.
- V. U. Pandit, et al., Solar light driven dye degradation using novel organo–inorganic (6, 13-pentacenequinone/TiO 2) nanocomposite, RSC Adv., 2015, 5(14), 10326–10331 RSC.
- K. Li, et al., Raney Ni as Recyclable and Selective Catalyst for the Reduction of α-Pinene to cis-Pinane with NaBH4 at Room Temperature, ChemistrySelect, 2019, 4(35), 10506–10509 CrossRef CAS.
- S. Kulkarni and J. Kaware, Regeneration and recovery in adsorption-a review, Int. J. Innov. Sci. Eng. Technol., 2014, 1(8), 61–64 Search PubMed.
- J. Bayuo, M. A. Abukari and K. B. Pelig-Ba, Desorption of chromium (VI) and lead (II) ions and regeneration of the exhausted adsorbent, Appl. Water Sci., 2020, 10(7), 171 CrossRef CAS.
- J. Bayuo, et al., Adsorption and desorption processes of toxic heavy metals, regeneration and reusability of spent adsorbents: economic and environmental sustainability approach, Adv. Colloid Interface Sci., 2024, 329, 103196 CrossRef CAS.
- J. Wang and X. Guo, Adsorption kinetics and isotherm models of heavy metals by various adsorbents: An overview, Crit. Rev. Environ. Sci. Technol., 2023, 53(21), 1837–1865 CrossRef CAS.
- S. Rehman, et al., Decoding the complex relation of financial development and carbon emission using bibliometric analysis, Cogent Bus. Manag., 2024, 11(1), 2294524 CrossRef.
- W. Sun, et al., Bibliometric analysis of assessment and evaluation in higher education: 2012–2023, Assess Eval. High Educ., 2024, 49(8), 1121–1135 CrossRef.
- J. Wang and G. Xuan, Adsorption kinetics and isotherm models of heavy metals by various adsorbents: An overview, Crit. Rev. Environ. Sci. Technol., 2023, 53(21), 1837–1865 CrossRef CAS.
- D. S. Bhatkhande, V. G. Pangarkar and A. A. C. M. Beenackers, Photocatalytic degradation for environmental applications–a review, J. Chem. Technol. Biotechnol. Int. Res. Process Environ. Clean Technol., 2002, 77(1), 102–116 CAS.
- Y. Wang, et al., Research status, trends, and mechanisms of biochar adsorption for wastewater treatment: a scientometric review, Environ. Sci. Eur., 2024, 36(1), 25 CrossRef.
- X. Liu, S. Wang and X. Wang, Challenges of porous nanomaterials in highly efficient elimination of pollutants from aqueous solution, Chin. Chem. Lett., 2024, 110679 Search PubMed.
- U. G. Akpan and B. H. Hameed, Parameters affecting the photocatalytic degradation of dyes using TiO2-based photocatalysts: a review, J. Hazard Mater., 2009, 170(2–3), 520–529 CrossRef CAS PubMed.
- M. Rauf and S. S. Ashraf, Fundamental principles and application of heterogeneous photocatalytic degradation of dyes in solution, Chem. Eng. J., 2009, 151(1–3), 10–18 CrossRef CAS.
- X. Pan, F. Kong and M. Xing, Spatial separation of photo-generated carriers in gC 3 N 4/MnO 2/Pt with enhanced H 2 evolution and organic pollutant control, Res. Chem. Intermed., 2022, 48(7), 2837–2855 CrossRef CAS.
- Y. Deng and R. Zhao, Advanced oxidation processes (AOPs) in wastewater treatment, Curr. Pollut. Rep., 2015, 1(3), 167–176 CrossRef CAS.
- D. B. Miklos, et al., Evaluation of advanced oxidation processes for water and wastewater treatment–A critical review, Water Res., 2018, 139, 118–131 CrossRef CAS PubMed.
- R. Andreozzi, et al., Advanced oxidation processes (AOP) for water purification and recovery, Catal. Today, 1999, 53(1), 51–59 CrossRef CAS.
- H. Suty, C. De Traversay and M. Cost, Applications of advanced oxidation processes: present and future, Water Sci. Technol., 2004, 49(4), 227–233 CrossRef CAS PubMed.
- K. E. O'Shea and D. D. Dionysiou, Advanced Oxidation Processes for Water Treatment, ACS Publications, 2012, p. 2112–2113 Search PubMed.
- C. G. Joseph, et al., Sonophotocatalysis in advanced oxidation process: a short review, Ultrason. Sonochem., 2009, 16(5), 583–589 CrossRef CAS.
- J. A. Silva, Advanced Oxidation Process in the Sustainable Treatment of Refractory Wastewater: A Systematic Literature Review, Sustainability, 2025, 17(8), 3439 CrossRef CAS.
- Z. Hashmi, et al., Comparative analysis of conventional to biomass-derived adsorbent for wastewater treatment: a review, Biomass Convers. Biorefinery, 2024, 14(1), 45–76 CrossRef CAS.
- U. v. Gunten, Ozonation of drinking water: Part I. Oxidation kinetics and product formation, Water Res., 2003, 37(7), 1443–1467 CrossRef.
- J. J. Pignatello, O. Esther and A. MacKay, Advanced oxidation processes for organic contaminant destruction based on the Fenton reaction and related chemistry, Crit. Rev. Environ. Sci. Technol., 2006, 36(1), 1–84 CrossRef CAS.
- V. A. Sadykov, S. F. Tikhov, N. N. Bulgakov and A. P. Gerasev, Catalytic oxidation of CO on CuOx revisited: Impact of the surface state on the apparent kinetic parameters, Catal. Today, 2009, 144(3–4), 324–333 CrossRef CAS.
- J. R. Bolton, K. G. Bircher, W. Tumas and C. A. Tolman, Figures-of-merit for the technical development and application of advanced oxidation technologies for both electric-and solar-driven systems (IUPAC Technical Report), Pure Appl. Chem., 2001, 73(4), 627–637 CrossRef CAS.
- A. Fujishima, Z. Xintong and D. A. Tryk, TiO2 photocatalysis and related surface phenomena, Surf. Sci. Rep., 2008, 63(12), 515–582 CrossRef CAS.
- I. Oller, S. Malato and J. A. Sánchez-Pérez, Combination of advanced oxidation processes and biological treatments for wastewater decontamination—a review, Sci. Total Environ., 2011, 409(20), 4141–4166 CrossRef CAS PubMed.
- D. Singh, et al., Strategies for biological treatment of waste water: A critical review, J. Clean. Prod., 2024, 454, 142266 CrossRef CAS.
- T. E. Schultz, Biological wastewater treatment, Chem. Eng., 2005, 1, 44 Search PubMed.
- S.-J. Park, et al., Biological treatment of wastewater containing dimethyl sulphoxide from the semi-conductor industry, Process Biochem., 2001, 36(6), 579–589 CrossRef CAS.
- J. M. Poyatos, et al., Advanced oxidation processes for wastewater treatment: state of the art, Water, Air, Soil Pollut., 2010, 205(1), 187–204 CrossRef CAS.
- P. Chowdhury, T. Viraraghavan and A. Srinivasan, Biological treatment processes for fish processing wastewater–A review, Bioresour. Technol., 2010, 101(2), 439–449 CrossRef CAS.
- I. Sirés, et al., Electrochemical advanced oxidation processes: today and tomorrow. A review, Environ. Sci. Pollut. Res., 2014, 21(14), 8336–8367 CrossRef.
- S. M. Acharya, R. Chakraborty and S. G. Tringe, Emerging trends in biological treatment of wastewater from unconventional oil and gas extraction, Front. Microbiol., 2020, 11, 569019 CrossRef PubMed.
- P. Chakraborty, S. Show, W. U. Rahman and G. Halder, Linearity and non-linearity analysis of isotherms and kinetics for ibuprofen remotion using superheated steam and acid modified biochar, Process Safety and Environmental Protection, 2019, 126, 193–204 CrossRef CAS.
- R. Luo, L. Xiaohui, H. Xu, Y. Sun and J. Wu, Effects of temperature, solution pH, and ball-milling modification on the adsorption of non-steroidal anti-inflammatory drugs onto biochar, Bull. Environ. Contam. Toxicol., 2020, 105(3), 422–427 CrossRef CAS PubMed.
- A. Ashiq, S. Binoy, N. Adassooriya, J. Walpita, A. U. Rajapaksha, Y. S. Ok and M. Vithanage, Sorption process of municipal solid waste biochar-montmorillonite composite for ciprofloxacin removal in aqueous media, Chemosphere, 2019, 236, 124384 CrossRef CAS PubMed.
- J. Li, G. Yu, L. Pan, C. Li, F. You, S. Xie, Y. Wang, J. Ma and X. Shang, Study of ciprofloxacin removal by biochar obtained from used tea leaves, J. Environ. Sci., 2018, 73, 20–30 CrossRef CAS PubMed.
- B. S. Delgado-Moreno L, G. Gasco, A. Méndez, M. El Azzouzi and E. Romero, New insights into the efficient removal of emerging contaminants by biochars and hydrochars derived from olive oil wastes, Sci. Total Environ., 2021, 752, 141838 CrossRef.
- G. S. Dhillon, K. Surinder, R. Pulicharla, S. K. Brar, M. Cledón, M. Verma and R. Y. Surampalli, Triclosan: current status, occurrence, environmental risks and bioaccumulation potential, Int. J. Environ. Res. Public Health, 2015, 12(5), 5657–5684 CrossRef CAS.
- M. Naghdi, T. Mehrdad, R. Pulicharla, T. Rouissi, S. K. Brar, M. Verma and R. Y. Surampalli, Pine-wood derived nanobiochar for removal of carbamazepine from aqueous media: Adsorption behavior and influential parameters, Arab. J. Chem., 2019, 12(8), 5292–5301 CrossRef CAS.
- G. Vernouillet, E. Philippe, A. Lajeunesse, C. Blaise, F. Gagné and P. Juneau, Toxic effects and bioaccumulation of carbamazepine evaluated by biomarkers measured in organisms of different trophic levels, Chemosphere, 2010, 80(9), 1062–1068 CrossRef CAS PubMed.
- S. Keerthanan, B. Amit, K. Mahatantila, C. Jayasinghe, Y. S. Ok and M. Vithanage, Engineered tea-waste biochar for the removal of caffeine, a model compound in pharmaceuticals and personal care products (PPCPs), from aqueous media, Environ. Technol. Innov., 2020, 19, 100847 CrossRef.
- S. Álvarez-Torrellas, R. Araceli, G. Ovejero, J. M. Gómez and J. García, Removal of caffeine from pharmaceutical wastewater by adsorption: influence of NOM, textural and chemical properties of the adsorbent, Environ. Technol., 2016, 37(13), 1618–1630 CrossRef.
- A. A. Godoy, K. Fábio and P. A. Pamplin, Occurrence, ecotoxicological effects and risk assessment of antihypertensive pharmaceutical residues in the aquatic environment-a review, Chemosphere, 2015, 138, 281–291 CrossRef CAS PubMed.
- R. C. Pundlik, et al., Life-cycle assessment of agricultural waste-based and biomass-based adsorbents, Biomass, Biofuels, Biochem., 2021, 669–695 CAS.
- S. Wang, et al., Life cycle assessment of carbon-based adsorbent preparation from algal biomass, J. Clean. Prod., 2023, 427, 139269 CrossRef CAS.
- T. L. Yami, et al., Life cycle assessment of adsorbents for fluoride removal from drinking water in East Africa, Int. J. Life Cycle Assess., 2015, 20(9), 1277–1286 CrossRef CAS.
- B. N. Kazemi A, A. Heydari and S. I. Olsen, Life cycle assessment of nanoadsorbents at early stage technological development, J. Clean. Prod., 2018, 174, 527–537 CrossRef.
- P. H. F. Pereira, et al., Prospective life cycle assessment prospective (LCA) of activated carbon production, derived from banana peel waste for methylene blue removal, Adsorption, 2024, 30(6), 1081–1101 CrossRef CAS.
- N. A. Nandikes G, S. I. Siddiqui and S. Oh, Sustainable water treatment using agricultural residue Adsorbents: Evaluating Efficacy and life cycle impacts, J. Ind. Eng. Chem., 2025, 889–900 CrossRef.
- A. Rodriguez-Calvo, R. Cossent and P. Frías, Scalability and replicability analysis of large-scale smart grid implementations: Approaches and proposals in Europe, Renew. Sustain. Energy Rev., 2018, 93, 1–15 CrossRef.
- D. Moldovan, H.-L. Truong, and S. Dustdar. Cost-aware scalability of applications in public clouds. in 2016 IEEE International Conference on Cloud Engineering (IC2E), IEEE, 2016 Search PubMed.
- H. Xiong and et al., Scalable architectures for platform-as-a-service clouds: Performance and cost analysis. in European Conference on Software Architecture, Springer, 2014 Search PubMed.
- E. Santoyo-Castelazo and A. Azapagic, Sustainability assessment of energy systems: integrating environmental, economic and social aspects, J. Clean. Prod., 2014, 80, 119–138 CrossRef.
- M. Aleem, et al., Coagulation-and adsorption-based environmental impact assessment and textile effluent treatment, Water, Air, Soil Pollut., 2020, 231(2), 45 CrossRef CAS.
- S. Wang and H. Wang, Adsorption behavior of antibiotic in soil environment: a critical review, Front. Environ. Sci. Eng., 2015, 9(4), 565–574 CrossRef CAS.
- C. E. Almeida-Naranjo, et al., Transforming waste into solutions: Raw and modified bioadsorbents
for emerging contaminant removal, J. Environ. Chem. Eng., 2025, 13, 116720 CrossRef CAS.
- B. K. Xiao and K. M. Thomas, Competitive adsorption of aqueous metal ions on an oxidized nanoporous activated carbon, Langmuir, 2004, 20(11), 4566–4578 CrossRef CAS PubMed.
- Z. Zhao, et al., Competitive adsorption and selectivity of benzene and water vapor on the microporous metal organic frameworks (HKUST-1), Chem. Eng. J., 2015, 259, 79–89 CrossRef CAS.
- J.-H. Park, et al., Competitive adsorption and selectivity sequence of heavy metals by chicken bone-derived biochar: batch and column experiment, J. Environ. Sci. Health Part A, 2015, 50(11), 1194–1204 CrossRef CAS.
- Z.-H. Zhang, et al., Selective and competitive adsorption of azo dyes on the metal–organic framework ZIF-67, Water, Air, Soil Pollut., 2016, 227(12), 471 CrossRef.
- R. Changotra, R. Himadri and A. Dhir, Treatment of real pharmaceutical wastewater using combined approach of Fenton applications and aerobic biological treatment, J. Photochem. Photobiol., A, 2019, 376, 175–184 CrossRef CAS.
- S. Wong, N. Norzita, I. M. Inuwa and O. Hassan, Recent advances in applications of activated carbon from biowaste for wastewater treatment: a short review, J. Clean. Prod., 2018, 175, 361–375 CrossRef CAS.
- J. Schwarz, et al., Standardized testing methods for measuring mechanical properties of ice, Cold Reg. Sci. Technol., 1981, 4(3), 245–253 CrossRef.
- I. Phillips and A King, Standardization of susceptibility testing methods, J. Chemother., 1997, 9(1), 13–18 CrossRef CAS PubMed.
- I. Himelfarb, A primer on standardized testing: History, measurement, classical test theory, item response theory, and equating, J. Chiropr. Educ., 2019, 33(2), 151–163 CrossRef.
- L. Cheng, J. Yao, N. Yan, S. F. Shan, X. Q. Liu and L. B. Sun, Smart adsorbents with photoregulated molecular gates for both selective adsorption and efficient regeneration, ACS Appl. Mater. Interfaces, 2016, 8(35), 23404–23411 CrossRef CAS PubMed.
- Y. Jiang, T. Peng, X. Q. Liu and L. B. Sun, Process-oriented smart adsorbents: tailoring the properties dynamically as demanded by adsorption/desorption, Acc. Chem. Res., 2021, 55(1), 75–86 CrossRef.
- J. J. Ding, Z. Jing, Y. X. Li, X. Q. Liu and L. B. Sun, Smart adsorbents functionalized with thermoresponsive polymers for selective adsorption and energy-saving regeneration, Ind. Eng. Chem. Res., 2017, 56(15), 4341–4349 CrossRef CAS.
- Y. G. Ko, Hybrid method integrating adsorption and chemical precipitation of heavy metal ions on polymeric fiber surfaces for highly efficient water purification, Chemosphere, 2024, 363, 142909 CrossRef CAS PubMed.
- A. A. Askalany, B. B. Saha, K. Kariya, I. M. Ismail, M. Salem, A. H. Ali and M. G. Morsy, Hybrid adsorption cooling systems–An overview, Renew. Sustain. Energy Rev., 2012, 16(8), 5787–5801 CrossRef CAS.
- S. S. Madaeni and S. Ehsan, Membrane-adsorption integrated systems/processes, Integr. Membr. Syst. Processes, 2016, 4, 343–373 Search PubMed.
- P. Kunwar, Commercialization challenges of adsorbent materials for water and wastewater treatment, Master’s thesis, 2022.
- P. J. Kunwar, L. Tero, H. Haapasalo and J. Majava, Addressing adsorbent materials commercialization challenges for water treatment in European markets through productization, Cogent Eng., 2024, 11(1), 2320952 CrossRef.
- S. Satyam and P. Sanjukta, Innovations and challenges in adsorption-based wastewater remediation: A comprehensive review, Heliyon, 2024, 10(9), 2024 CrossRef PubMed.
- M. Thommes, K. Katsumi, A. V. Neimark, J. P. Olivier, F. Rodriguez-Reinoso, J. Rouquerol and K. S. Sing, Physisorption of gases, with special reference to the evaluation of surface area and pore size distribution (IUPAC Technical Report), Pure Appl. Chem., 2015, 87(9–10), 1051–1069 CrossRef CAS.
- W. S. Kenneth Stafford, Reporting physisorption data for gas/solid systems with special reference to the determination of surface area and porosity (Recommendations 1984), Pure Appl. Chem., 1985, 57(4), 603–619 CrossRef.
- H. B. Foo KY, Insights into the modeling of adsorption isotherm systems, Chem. Eng. J., 2010, 156(1), 2–10 CrossRef.
- C. Pelekani and V. L. Snoeyink, Competitive adsorption between atrazine and methylene blue on activated carbon: the importance of pore size distribution, Carbon, 2000, 38(10), 1423–1436 CrossRef CAS.
- R. C. Bansal and G.M.A.c.a.C.p.M., Activated Carbon Adsorption, CRC press, 2005 Search PubMed.
- Y. S. Ho and G. McKay, Pseudo-second order model for sorption processes, Process Biochem., 1999, 34(5), 451–465 CrossRef CAS.
- X. Vecino, R. Devesa-Rey, D. M. de Lima Stebbins, A. B. Moldes, J. M. Cruz and N. A. Alcantar, Evaluation of a cactus mucilage biocomposite to remove total arsenic from water, Environ. Technol. Innov., 2016, 6, 69–79 CrossRef.
- G. Crini and E. Lichtfouse, Advantages and disadvantages of techniques used for wastewater treatment, Environ. Chem. Lett., 2019, 17(1), 145–155 CrossRef CAS.
- P. O. Isibor, Regulations and policy considerations for nanoparticle safety, in Environmental Nanotoxicology: Combatting the Minute Contaminants, Springer Nature Switzerland, Cham, 2024, vol. 20, pp. 295–316 Search PubMed.
|
| This journal is © The Royal Society of Chemistry 2026 |
Click here to see how this site uses Cookies. View our privacy policy here.  Open Access Article
Open Access Article *a,
Amna Islama,
Syed Khasim
*a,
Amna Islama,
Syed Khasim b,
Farooq Ahmadc,
Naveed Ahmadc and
Ibrahim A. Alsayer
b,
Farooq Ahmadc,
Naveed Ahmadc and
Ibrahim A. Alsayer c
c







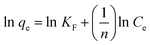
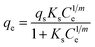

![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) qe = ln
qe = ln![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) qD − Bε2
qD − Bε2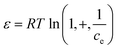

![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) qe − k1t
qe − k1t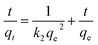




![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) ln
ln![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) KC
KC






![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 to 200
000 to 200![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 tons of antibiotics are used worldwide.170 The primary routes of pharmaceutical contamination into water bodies are discharges from drug production plants and municipal sewage systems, and from hospital effluents. These compounds are considered hazardous. Advanced oxidation processes (AOPs), which include methods such as ozonation, the Fenton process, and photocatalysis, have been shown to effectively degrade synthetic contaminants.171 This has led to an increased focus on finding adsorbents that are highly efficient, safe, and affordable.172 Antibiotics are a type of pharmaceutical that often ends up in the environment, contaminating water and soil. Bacterial resistance occurs when bacteria that were once vulnerable to an antibiotic become resistant to it. This can be caused by the overuse of antibiotics and improper treatment of wastewater.173 As a result, the contaminated adsorbents then become a disposal concern, meaning that adsorption alone is not a complete solution for the issue of antibiotic residue.174 Continuous processes are typically more cost-effective than batch processes because batch processes have higher energy consumption due to start-up times and increased raw material costs, since materials are recycled less frequently. However, a key advantage of batch processing is the ability to combine multiple tasks into a single, larger unit.175 Continuous technology is considered advantageous for commercial production because it allows for flexible changes in production quantities and is easy to scale up. The use of continuous technology is expanding to include the production of active pharmaceutical ingredients (APIs) for both small molecules and biopharmaceuticals.176 The product is continuously harvested by removing the old medium and replacing it with fresh medium.177 Continuous production dramatically enhances the efficiency of the primary capture resin, resulting in a substantial 68% decrease in consumable costs.178 One advantage of batch processing is its flexibility. Conversely, continuous processes must operate year-round at a large scale to be profitable.179 Fig. 15(a) comparative schematics of monoclonal antibody production methods: illustrating the (A) traditional fed-batch process versus the (B) continuous manufacturing process.177 Used adsorbents are often treated as waste, but their disposal can be harmful to the environment because they may contain toxic, absorbed compounds. To address this, an environmentally friendly alternative is to desorb the pollutants and regenerate the adsorbents for reuse.180 Saturated adsorbents can be regenerated, and pollutants recovered using several methods, including chemical (acids, alkalis, other chemicals) regeneration. The maximum desorption capacities for these techniques were found to be 99.5%, 92.6%, 284%, 150%, and 66.61%, respectively.181 Adsorption using activated carbon is considered a superior way to treat both water and wastewater. It stands out from other chemical and physical techniques because of its highly efficient adsorption of diverse pollutants, its rapid adsorption rate, and its simple design.182 Adsorption techniques are particularly advantageous for environmental cleanup and sustainability because they utilize common and effective materials such as activated carbon, clay minerals, silica gel, zeolites, and polymers.183 In this regard, graphene materials are also very promising as next-generation conventional adsorbent replacements for the control of environmental pollution, representing very important scientific values. They feature strong adsorption capability and excellent recyclability.184 Regeneration processes allow used adsorbents to be retrieved and reused multiple times with no significant drop in performance. Reuse and regeneration, often achieved through an inexpensive process such as desorption, is crucial when adsorbents are used for wastewater treatment outside of their original location. Reusing adsorbents is beneficial because it reduces costs and the need for material disposal.185 Fig. 15(b) Possible mechanism of photocatalytic degradation.186,187 The repeatability and reusability tests demonstrate that the photocatalyst exhibits high structural and catalytic stability.188–190 Regeneration is a crucial part of the adsorption process from both economic and environmental perspectives. There are several regeneration methods that have yielded mixed results, including solvent washing, and thermal, chemical, and electrochemical regeneration. To make adsorption a more economical, valuable, and important method for treating wastewater, it is crucial to focus on regenerating and reusing the adsorbent, as well as recovering the solute.191 Percentage desorption was calculated as:192
000 tons of antibiotics are used worldwide.170 The primary routes of pharmaceutical contamination into water bodies are discharges from drug production plants and municipal sewage systems, and from hospital effluents. These compounds are considered hazardous. Advanced oxidation processes (AOPs), which include methods such as ozonation, the Fenton process, and photocatalysis, have been shown to effectively degrade synthetic contaminants.171 This has led to an increased focus on finding adsorbents that are highly efficient, safe, and affordable.172 Antibiotics are a type of pharmaceutical that often ends up in the environment, contaminating water and soil. Bacterial resistance occurs when bacteria that were once vulnerable to an antibiotic become resistant to it. This can be caused by the overuse of antibiotics and improper treatment of wastewater.173 As a result, the contaminated adsorbents then become a disposal concern, meaning that adsorption alone is not a complete solution for the issue of antibiotic residue.174 Continuous processes are typically more cost-effective than batch processes because batch processes have higher energy consumption due to start-up times and increased raw material costs, since materials are recycled less frequently. However, a key advantage of batch processing is the ability to combine multiple tasks into a single, larger unit.175 Continuous technology is considered advantageous for commercial production because it allows for flexible changes in production quantities and is easy to scale up. The use of continuous technology is expanding to include the production of active pharmaceutical ingredients (APIs) for both small molecules and biopharmaceuticals.176 The product is continuously harvested by removing the old medium and replacing it with fresh medium.177 Continuous production dramatically enhances the efficiency of the primary capture resin, resulting in a substantial 68% decrease in consumable costs.178 One advantage of batch processing is its flexibility. Conversely, continuous processes must operate year-round at a large scale to be profitable.179 Fig. 15(a) comparative schematics of monoclonal antibody production methods: illustrating the (A) traditional fed-batch process versus the (B) continuous manufacturing process.177 Used adsorbents are often treated as waste, but their disposal can be harmful to the environment because they may contain toxic, absorbed compounds. To address this, an environmentally friendly alternative is to desorb the pollutants and regenerate the adsorbents for reuse.180 Saturated adsorbents can be regenerated, and pollutants recovered using several methods, including chemical (acids, alkalis, other chemicals) regeneration. The maximum desorption capacities for these techniques were found to be 99.5%, 92.6%, 284%, 150%, and 66.61%, respectively.181 Adsorption using activated carbon is considered a superior way to treat both water and wastewater. It stands out from other chemical and physical techniques because of its highly efficient adsorption of diverse pollutants, its rapid adsorption rate, and its simple design.182 Adsorption techniques are particularly advantageous for environmental cleanup and sustainability because they utilize common and effective materials such as activated carbon, clay minerals, silica gel, zeolites, and polymers.183 In this regard, graphene materials are also very promising as next-generation conventional adsorbent replacements for the control of environmental pollution, representing very important scientific values. They feature strong adsorption capability and excellent recyclability.184 Regeneration processes allow used adsorbents to be retrieved and reused multiple times with no significant drop in performance. Reuse and regeneration, often achieved through an inexpensive process such as desorption, is crucial when adsorbents are used for wastewater treatment outside of their original location. Reusing adsorbents is beneficial because it reduces costs and the need for material disposal.185 Fig. 15(b) Possible mechanism of photocatalytic degradation.186,187 The repeatability and reusability tests demonstrate that the photocatalyst exhibits high structural and catalytic stability.188–190 Regeneration is a crucial part of the adsorption process from both economic and environmental perspectives. There are several regeneration methods that have yielded mixed results, including solvent washing, and thermal, chemical, and electrochemical regeneration. To make adsorption a more economical, valuable, and important method for treating wastewater, it is crucial to focus on regenerating and reusing the adsorbent, as well as recovering the solute.191 Percentage desorption was calculated as:192













