Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Smart nanosystems for disease-resistant potatoes: a new Frontier in nanobiotechnology
Bo Chen†
a,
Mohammad Umair Rafiq†b,
Muhammad Usmanc,
Sarmad Frogh Arshadb,
Akhtar Hameedb and
Manzar Abbas *de
*de
aSchool of Agriculture, Forestry and Food Engineering, Yibin University, Yibin 644000, China
bInstitute of Plant Protection, Muhammad Nawaz Shareef University of Agriculture, Multan 66000, Pakistan
cDepartment of Biochemistry and Biotechnology, Muhammad Nawaz Shareef University of Agriculture, Multan 66000, Pakistan
dInner Mongolia Saikexing Institute of Breeding and Reproductive Biotechnology in Domestic Animal, Hohhot, China. E-mail: abbas2472@hotmail.com
eNational Center of Technology Innovation for Dairy, Hohhot 010020, China
First published on 17th March 2026
Abstract
Microbial diseases in potato crops pose significant threats to production quality and crop protection. Yield losses due to these diseases are unsustainable for a world that is increasingly reliant on potato-based diets. Conventional management strategies, including chemical and biological controls, have been employed, but they often disrupt biodiversity conservation. The resulting ecological degradation has driven researchers to seek sustainable alternatives. Emerging technologies now address phytopathogens—viruses, fungi, and bacteria—while minimizing the adverse effects of traditional methods. Among these, nanotechnology, utilizing materials with at least one dimension between 1–100 nm, has revolutionized plant health through nanopesticides and targeted pesticide delivery systems. These innovations exhibit minimal ecological impact while demonstrating potent antimicrobial activity against key potato diseases such as early blight, late blight, common scab, soft rot, and blackleg. Nanoparticles (NPs) generate reactive oxygen species (ROS), which lyse microbial cells while simultaneously activating defense-related signaling pathways (salicylic acid and jasmonic acid pathways). These pathways upregulate pathogenesis-related (PR) genes, enhancing PR protein synthesis to combat microbial invasion. Nanotechnology has enabled the design of advanced nano-biosensors for disease detection. By leveraging nanoparticle properties such as a high surface-area-to-volume ratio, photoluminescence, electrical conductivity, and biomolecular interaction, these sensors precisely identify microbial biomarkers. Additionally, the small size, surface charge, controlled release, and tunable surface chemistry of nanoparticles help in optimizing targeted gene and drug deliveries. Nanotechnology further enhances genome-editing tools, like CRISPR/Cas9 and RNA interference (RNAi), facilitating the development of disease-resistant transgenic potato varieties. It also induces the production of antioxidant enzymes, osmolytes, stress-responsive genes, and structural barriers to mitigate abiotic stresses. In summary, nanotechnology offers a multidisciplinary approach for combating phytopathogens, ensuring sustainable potato cultivation with minimal ecological disruption.
1 Introduction
Potato belongs to the Solanaceae family of plants with the scientific name Solanum tuberosum L.; it bears tubers in the underground part of the soil and has been recognized as one of the most produced and consumed crops globally after wheat, rice and maize.1 Some European countries, like Ireland, have adopted potato as a staple food because of its reasonably high yields and rich nutrient profile. The presence of nutritional components such as potassium, vitamin C, vitamin B6, proteins, thiamine and niacin in potatoes increase their essentiality as these components are important for maintaining human health. The climate and soil resilience of the potato crop ensures its availability across the globe.2 Nowadays, the potato crop has valorized the global food industry due to its nutritive and economic value. The food processing industries of potatoes have been transformed extensively into production units of value-added products. By-products like potato starch are used in soups, sauces, gravies and puddings and as binding agents and texture enhancers in meats and baked items. The production of alcoholic beverages and fermentation into spirits due to their high starch content has also supported the alcohol industry. Potatoes have recently emerged as a viable source of bioplastics as they are developed into biodegradable packaging materials that replace traditional plastic packaging, and this is a broader trend towards sustainability and for decreasing the environmental impacts of plastic trash.1.1 Potato productivity under siege
The production of potatoes was estimated to be approximately 375 million tonnes, while its global consumption was approximately 236 million metric tonnes based on the latest statistics of FAO in 2023. China is the largest producer of potatoes, followed by India, Germany, France, the Netherlands, the UK, Belgium, Russia, Ukraine, the USA, Canada, Argentina and Australia. Asia has been a major contributor to the production of staple food crops like wheat, rice and potatoes. Potato is the second most targeted crop in Asia after wheat, rice and maize. In Asia in 2022, potatoes were harvested in an area of approximately 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 336
336![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 800 hectares with a production of approximately 203
800 hectares with a production of approximately 203![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 338
338![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 tons. China, the world's potato superpower, accounts for more than 20% of the global potato-growing area and harvest. China produces approximately 92 million metric tons of potatoes every year, ∼66 kg per capita. Based on its contribution to world potato production, we can say that if there is a drought in China that affects potato output, this might have consequences for the entire world food supply. India is the next largest potato producing country, with a production of approximately 50 million metric tons every year. Several other Asian countries including Pakistan, Bangladesh, Iran, Japan and Turkey are among the top 20 producers of potatoes. Most of the world would go hungry without potatoes from Asia.
000 tons. China, the world's potato superpower, accounts for more than 20% of the global potato-growing area and harvest. China produces approximately 92 million metric tons of potatoes every year, ∼66 kg per capita. Based on its contribution to world potato production, we can say that if there is a drought in China that affects potato output, this might have consequences for the entire world food supply. India is the next largest potato producing country, with a production of approximately 50 million metric tons every year. Several other Asian countries including Pakistan, Bangladesh, Iran, Japan and Turkey are among the top 20 producers of potatoes. Most of the world would go hungry without potatoes from Asia.
As per the Trade Development Authority of Pakistan (TDAP), Pakistan contributes around 4% to world potato production, while according to the Pakistan Economic Survey 2022-23, the total potato cultivating area was around 341![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 hectares, producing 8
000 hectares, producing 8![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 320
320![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 tons in the country. In 2023-24, potato production was estimated at 8
000 tons in the country. In 2023-24, potato production was estimated at 8![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 441
441![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 tons using the cultivated area of 339
000 tons using the cultivated area of 339![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 hectares, an increase in production by 1.5%. Potato production in Pakistan is mainly focused in Punjab and then in KPK, Balochistan and Sindh. Two major regions of Pakistan renowned for potato cultivation are Punjab and Gilgit Baltistan. In Punjab, potato production was estimated to be over 8.1 million tons, which was harvested from an area of around 808
000 hectares, an increase in production by 1.5%. Potato production in Pakistan is mainly focused in Punjab and then in KPK, Balochistan and Sindh. Two major regions of Pakistan renowned for potato cultivation are Punjab and Gilgit Baltistan. In Punjab, potato production was estimated to be over 8.1 million tons, which was harvested from an area of around 808![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 hectares.
000 hectares.
Considering the production figures of potatoes, the lion's share of this production is lost at the pre- and post-harvest stages due to diseases or other factors. Potato diseases have remained a major concern for improving potato production in Pakistan. Thus, for better production, protection is necessary. Potatoes are not only consumed directly by consumers, but different value-added products of this crop have increased its importance.3 As the world relies on this crop, it is necessary to grow safe and healthy crops with high production yields which are often disturbed by different biological problems such as attacks by pathogens.4
1.2 The invisible yield thief
Potato has remained below the threshold of different vulnerable pathogens including viruses, bacteria, fungi and nematodes. These pathogens not only affect the production of the crop but also deteriorate the quality of tubers. Considering the importance of this crop, it is necessary to protect it from destructive microbes. Blackleg, ring rot, soft rot, zebra chip, pink eye, bacterial wilt and common scab are important bacterial diseases of potatoes that cause huge losses of the crop. Fungal diseases like early blight, late blight, brown leaf spot, black scurf, silver scurf, stem canker, black dot, skin spot, gangrene, verticillium wilt, wart disease, violet root rot, and watery wound rot have negative impacts on potato quality and quantity. Potato Virus Y, Potato Virus X, Potato leaf roll virus, Potato mop top virus, Tobacco necrotic virus, Tobacco rattle virus, and latent and mild mosaic viruses damage the crop aggressively. Root knot nematodes, potato cyst nematodes, and sting nematodes affect the potato crop vigorously. These diseases not only destroy its quality and yield but also affect its market value indirectly. All major diseases (bacterial, viral, fungal and nematode) of potato are mentioned in Table 1.| Diseases | Pathogens | Symptoms | Treatment | Ref. |
|---|---|---|---|---|
| Blackleg of potato | Pectobacterium atrosepticum | Blackening and rotting of the stem starts from the base upward; leaves become yellow, wilt and show premature senescence | Mercuric chloride, Agristep- or Semesan bel copper-based fungicides like Mancozeb | 13–15 |
| Ring rot of potato | Clavibacter michiganensis | A soft cheese-like rotting of the vascular ring is the earliest sign, hence the term “ring” rot. In severe cases, it completely rots and cracks the tuber's skin | Roccal, zephiran, hydrogen peroxide (5–20%) | 16, 17 and 18 |
| Soft rot of potato | Dickeya solani | Soft rot symptoms often include soft and moist tissues of tubers that are cream-colored | 5-Nitro-8-hydroxyquinoline, chlorine dioxide | 19 and 20 |
| Zebra chip disease | Candidatus Liberibacter | Chlorosis and purpling of foliage, upward rolling and burning of leaves, stunting and the development of aerial tubers | Thiamethoxam, fludioxonil, difenoconazole | 21, 22 and 23 |
| Pink eye disease | Pseudomonas fluorescens | Pink but slightly elevated regions on damp. The afflicted areas are typically located around the eyes and at the tuber stem end. Often present on freshly dug tubers but harder to detect on dried ones | Chlorothalonil, copper-based fungicides | 24, 25 an 26 |
| Bacterial wilt of potato | Ralstonia solanacearum | Wilting and yellowing of leaves and stunted plants. Vascular browning is ubiquitous, with bacterial ooze splashing from the incision | Methyl bromide, 1,3-dichloropropene, metam sodium with chloropicrin | 27, 28 and 29 |
| Common scab of potato | Streptomyces scabiei | Slight protrusions with sunken centers may occur and be covered with tiny amounts of corky tissue. Scab layers are transformed into lesions | Kasugamycin, copper-based fungicides | 30, 31 and 32 |
| Brown rot of potato | Ralstonia solanacearum | Symptoms emerge with brown staining of vascular rings that leads to rotting. Plant wilts with immediate effects | Copper nitrate, sodium hydroxide, magnesium sulphate polyvinylpyrrolidone | 33, 34 and 35 |
| Aerial stem rot of potato | Pectobacterium spp. | Black to brown slimy decay on potato stems | Copper hydroxide famoxadone | 36, 37 and 38 |
| Early blight | Alternaria solani | Lesions appear on leaves with a targeted spot having concentric rings. These frequently occur few weeks after emergence as extremely black or brown patches on lower leaves that eventually coalesce | Azoxystrobin, difenoconazole | 39, 40 and 41 |
| Late blight | Phytophthora infestans | Tuber skin becomes darker brown, while the internal rot becomes reddish brown. Granular rot may remain at the surface or spread to the center of the tuber | Zorvec endavia, ranman, revus, mancozeb | 42, 43 and 44 |
| Brown leaf spot | Alternaria alternata | On leaves, it produces little dark brown necrotic tissue patches with dark brown edges. Starting as tiny lesions, the spots might merge to cover a significant portion of the leaf or petiole surface | Fossil (azoxystrobin + difenoconazole), topsin (thiophanate methyl) | 45, 46 and 47 |
| Black scurf | Rhizoctonia solani | Affected tubers may exhibit growth abnormalities that resemble glyphosate damage and exhibit slowed growth in conjunction with stronger brown skin patches known as netting or elephant hide | Pencycuron, flutolanil, iprodione | 48, 49 and 50 |
| Silver scurf | Helminthosporium solani | Small silvery gray specks expand into circles with a slightly darker boundary. These circles expand and join together. Unwashed tubers with silver scurfs often have a sooty look due to spore formation | Pentachloronitro-benzene | 51, 52 and 53 |
| Stem canker | Rhizoctonia solani | Plants remain stunted and have folding of upper leaves. Brown, slightly recessed lesions with definite edges develop at the stem base and stolons. If the lesions are severe, they may unite and girdle the stem | Tolclofos-methyl + thiram, fosetyl-aluminium | 54, 55 and 56 |
| Black dot | Colletotrichum coccodes | Vascular disease may trigger stem wrapping, leading to foliar wilt. The affected skin looks light brown to undamaged but with spots. Later, boring dark brown spots may form | Fluazinam, azoxystrobin | 57, 58 and 59 |
| Skin spot | Polyscytalum pustulans | Dark grey and slightly elevated spots on tubers. A mature fungal lesion typically creates an empty ring around a rising center. Lesions may appear singly or in bunches, and they can be randomly scattered or localized around the eyes, stolons, and injured skin | 60, 61 and 62 | |
| Gangrene | Phoma exigua var. Foveata | The potato has discolored, dark sunken and irregularly shaped patches. Gangrene also produces interior decay and cavities filled with sparse gray or yellow mould | Fluazinam | 63, 64 and 65 |
| Verticillium wilt | Verticillium dahliae | The disease is frequently recognized as scattered patches in a field and can cause stunting, premature plant senescence and diminished output. Plants lose turgor and wilt on hot sunny days | Chlorothalonil, propiconazole, thiophanate-methyl, azoxystrobin | 66, 67 and 68 |
| Wart disease | Synchytrium endobioticum | Galls (wart-like growths) start out green, change brown and eventually black. Potato wart is seen at the time of harvest. Tubers have abnormal wart-like growth, ranging in size from a pin to a fist | Famoxadone + cymoxanil, tolclofos methyl + thiram, dimethomorph + copperoxychloride, mandipropamid + mancozeb, zoxamide + mancozeb | 69, 70 and 71 |
| Violet root rot | Rhizoctonia crocorum | Infected roots and tubers develop distinctive red-violet mycelium, which is occasionally accompanied by little black sclerotia. The mycelium then penetrates the tube | Flutolanil, fludioxonil | 72 and 73 |
| Watery wound rot | Pythium ultimum | Affected flesh may be grey to brown in color with a dark border. It is damp and soon liquefies | Metalaxyl, mancozeb | 74, 75 and 76 |
| Potato stem end rot | Fusarium oxysporum | A slightly sunken circular lesion appears with corky rot on the potato surface at the end of the stem | Mefenoxam | 77, 78 and 79 |
| Potato virus Y | Aphid | Symptoms include leaf deformation, mottling and crinkling, chlorotic mosaics in haulm, vein necrosis, reduced leaflet size and overall reduction in vigour | Thiamethoxam, (25 WG @ 150 g ha−1 | 80, 81 and 82 |
| Potato virus X | Mechanical transmission | Mosaic, chlorosis, necrotic lesions and decreased leaf size are morphological symptoms | Ribavirin | 83, 84 and 85 |
| Potato leafroll virus | Green Peach Aphid | In the upper leaves, a slight rolling and red/orange tinge may occur. The bottom leaves may roll and become dry and brittle, having a papery feel. The upper leaves upturn and show yellowing. Plants remain stunted | Pyrethroids, carbamate, organophosphates, neonicotinoids | 86, 87 and 88 |
| Potato mop top virus | Spongospora subterranea | Dark arcs and rings on the tuber skin are visible. The lower leaves have yellow spots, and the upper leaves are pale green. Stunted plants may bunch upper leaves with wavy/rolled margins (hence mop top). Tubers may be distorted, russetted, and cracked and may have net-like surface patches | Fluazinam, cyazofamid | 89, 90 and 91 |
| Tobacco necrotic virus | Olpidium brassicae | Dark brown and increased patches on leaves later to dark sunken lesions in ring/horseshoe form | Cross protection | 92, 93 and 94 |
| Tobacco rattle virus | Trichodorus spp. | The infection starts with brown flecking and arcs that are concentric. Stems become stunted with mottled leaves | Abamectin, azoxystrobin | 95, 96 and 97 |
| Mosaic virus | Aphids | Poor growth, yellowing, stunting, mottling or mosaic patterns and plant deformities are the symptoms | Thiamethoxam (25 WG @ 150 g ha−1 | 98, 99 and 100 |
| Root knot nematode | Meloidogyne incognita | Form swelling on roots known as galls, which are small in size, while egg masses appear on feeder roots. Wilting appears on above ground parts | Cadusafos | 101, 102 and 103 |
| Potato cyst nematode | Globodera rostochiensis | Form cysts on roots, while above-ground symptoms include plant stunting, flower delaying, leaf yellowing and ultimate wilting | Fluazaindolizine | 104, 105 and 106 |
| Sting nematode | Belonolaimus spp. | They are linked with severely misshapen, abnormally russetted and scruffy tubers. Wilting and stunting of plants are the ultimate effects | Fluensulfone, 1,3-dichloropropene | 106, 107 and 108 |
Different management strategies, like cultural, physical, biological and chemical, have been applied against such microbial load over time, but these strategies do not eliminate the risk of pathogens. Chemical control measures have detrimental effects on both plant and consumer health. On the contrary, the continuous application of chemical pesticides leads to the development of microbial resistance. Such detrimental effects of pesticides have led researchers to develop resistant potato cultivars with enhanced yield traits using genetic engineering techniques to mitigate the effects of pathogens (Fig. 1).
1.3 Recognition gap: undetected pathogen effector and its toll on plant defense
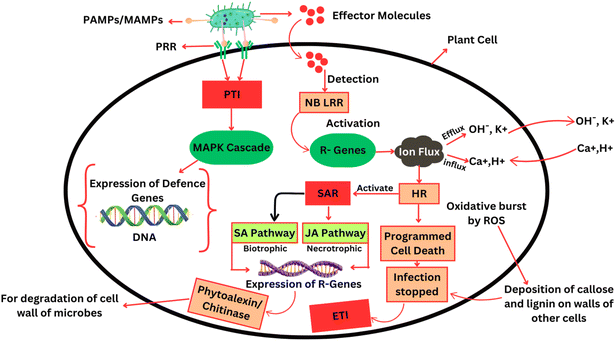
Micro-organisms present on or within plants produce various signals based on volatile chemical compounds, hormones, hormone mimics, proteins and carbohydrates.5 Microbial life depends on signals derived from proteins and carbohydrates that are often described as Microbe or Pathogen-Associated Molecular Patterns (MAMPs or PAMPs).6 Plants produce distinct Pattern Recognition Receptors (PRRs) localized in the plasma membrane that bind MAMPs/PAMPs and regulate plant immunological responses. PAMP/Pattern Triggered Immunity (PTI), also known as basal resistance, is the first line of defense in which most plant species limit pathogen infection in response to MAMPs/PAMPs.7 In the meantime, microbes release effector molecules during invasion, which are essential components of pathogenesis. Plants produce a series of immune reactions known as Effector Triggered Immunity (ETI) that recognize effector molecules.8 Plants that do not identify these effector molecules become susceptible and cannot activate their defense mechanisms.9,10 In the case of the detection of effector molecules, plants activate their defense systems, like the expression of R genes, production of antimicrobial proteins like phytoalexins, closure of stomatal pathways, over-production of reactive oxygen species (ROS) and expression of hypersensitive response (HR), followed by programmed cell death.11 With the expression of HR, plants may undergo systemic acquired resistance (SAR) through signaling pathways (jasmonic acid (JA) and salicylic acid (SA) pathways). Through these signals, plants activate R-genes, which encode pathogenesis-related (PR) proteins, to combat pathogens.12,141.4 Rising threat: agrochemical-resistant pathogens in global potato security
Microbial resistance refers to the ability of bacteria, fungi and viruses to endure agents, like antibiotics or pesticides, that normally kill them. Microbial resistance develops through various mechanisms initiated by pathogens.109 Pathogens continue to develop infections leading to severe diseases like late blight or black leg, which reduce crop yields. Therefore, high doses of chemicals or additional applications are applied to control resistant strains that increase production costs and environmental damage. For sustainable potato production and food security, the fight against microbial resistance is necessary.110 The resistance of potato pathogens against synthetic pesticides is caused by the presence of different mechanisms by which pathogens survive and thrive in the presence of chemical treatments. To be able to develop appropriate management strategies, it is important to understand these mechanisms in depth.111 Here are some key mechanisms of microbial resistance:2 Nanobiotechnology: a next-generation shield against crop pathogens
The pursuit of sustainable development is vital for safeguarding the availability of essential resources and meeting the basic needs of both present and future populations.9 However, microbial resistance to chemical control in food crops has posed serious concerns regarding food safety. The anticipation of biological problems related to food safety has become paramount to feeding the increasing population of the world. A variety of modern cultural, physical, biological and chemical approaches have been developed to ensure the protection of these food crops against microbial resistance. These technologies have revolutionized the traditional world by diverting it into a global village. Nanobiotechnology, the rising technology, consists of two major domains: nanotechnology and biotechnology.117 Nanobiotechnology has transformed into a paradigm-shifting technology that has the potential to completely change the 21st century by building a foundation for sustainable and high-quality civilizations.118 Nanotechnology may be defined as the use of material particles with at least one dimension of 1-100 nm.119 This concept was first introduced in 1959 by an American physicist, Richard Feynman, a Nobel Prize winner, during a meeting of the American Physical Society.120 A Japanese scientist, Norio Taniguch, in 1974 proposed the term “nanotechnology” with its proper definition and application, which was given as “nanotechnology is primarily concerned with the separation, consolidation and deformation of materials by a single atom or molecules”.121 The other domain, “biotechnology”, refers to the technology used to implement the combination of natural and engineering sciences for the welfare of organisms.122 Moreover, nanobiotechnology is referred to as designing and organizing materials on the nanoscale to develop smart devices with extraordinary physicochemical and biological properties and their applications in various fields of life such as agriculture and medicine.123The term nanomaterials (NMs) is an umbrella term that describes materials with a minimum dimension of the nanoscale (1–100 nm), making it a term used to describe nanoparticles, nanocomposites and functionalized nanostructures. In particular, nanoparticles (NPs) refer to discrete nanoscale particles (e.g. Ag, CuO, or ZnO particles) that can possess inherent physicochemical or antimicrobial behavior but do not contain engineered complexity. Conversely, nanosystems are engineered nano-architectures where nanoparticles are engineered with other functionalities, like encapsulation, surface engineering, controlled release or targeting. In this review, the term smart nanosystems is used solely to refer to novel nanosystems with advanced or responsive or programmable behavior such as stimulus release (e.g., pH, enzymes, and ROS), stimulus-responsive activation, or delivery to a particular plant tissue or cellular compartment. Based on these, simple metallic nanoparticles lacking these functional characteristics are not considered smart systems but conventional antimicrobial nanoparticles.124
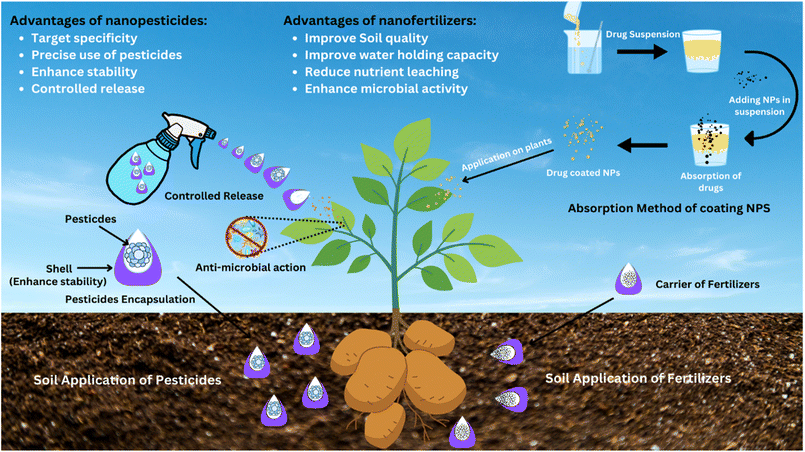
2.1 Role of nanobiotechnology in agriculture
Nowadays, the commercial use of nanotechniques has increased by merging nanotechnology and biotechnology to make them more functional in industries.125 Along with being introduced on an industrial level, nano-biotechnology has had a major impact in agriculture, related to the production and protection of crops. In this way, nanobiotechnology has solved problems such as food availability, for an increasing population.126 Products like nano-pesticides, nano-biosensors and nano-fertilizers have been developed to enhance crop production and reduce crop loss caused by microbial attack.127 These innovative products ensure sustainable agriculture because they replace synthetic agrochemicals that deteriorate biodiversity. The repetitive use of synthetic fertilizers disturbs the soil structure, texture, fertility and soil microbiota,128 while the extensive use of pesticides develops microbial resistance against them. Microbes with developed resistance are more harmful and even lead to epidemic outbreaks. Nano-biosensors are designed to mitigate the vandalism of pesticides by detecting their toxicities.129 Microbial resistance is mitigated by delivering anti-microbial agents with the use of nanocarriers. These nanocarriers deliver anti-microbials by releasing them at specific target sites.130 Thus, nanobiotechnology greatly enhances crop yield in terms of quality and quantity.The interaction of antimicrobial nanoparticles with phytopathogens occurs directly and suppresses them without loading the active ingredients. Some commonly used antimicrobial nanoparticles include silver (Ag), copper (Cu/CuO), zinc oxide (ZnO), magnesium oxide (MgO), and titanium dioxide (TiO2). They cause various effects: membrane disruption, ion release, enzyme inhibition and ROS generation, giving them a broad spectrum of action and limiting the resistance chances, but phytotoxicity and environmental persistence must be optimized by dose. The biological behavior of plants exposed to metallic nanoparticles is strongly dependent on the concentration, and it depends on the size of the particles, surface chemistry, plant species, and the stage of development. Although low or optimized doses of Ag-, Cu-, and Zn-based nanoparticles can lead to pathogen suppression and host defense reactions, phytotoxic processes, including oxidative stress disequilibrium, chlorophyll deterioration, root development, membrane harm, and photosynthetic activity, have been linked to excessive concentrations.136 Notably, the effective concentration region of pathogens can overlap with concentrations that have adverse biological impacts on plants, meaning that a specific set of metallic nanoparticles has a tight safety margin. Thus, the establishment of crop-specific dose–response relationships and the determination of biologically safe concentration ranges are important to reduce the risk of phytotoxicity and to ensure the viability of the use of nano-enabled disease management tools. The main mechanism of immune-activating nanoparticles is associated with priming or stimulation of the innate defense system of the plant instead of attacking pathogens.137 Silica nanoparticles, carbon-based nanomaterials (carbon nanotubes, graphene derivatives), salicylic acid-loaded nanoparticles, and biopolymer nanosystems are the most common immune-activating nanoparticles capable of inducing salicylic acid (SA) and jasmonic acid (JA) signaling, PR gene expression, and systemic acquired resistance (SAR). This immune modulation provides a non-toxic alternative to disease management that is highly chemical-intensive, results in the enhancement of host resistance, and decreases the pressure of a particular pathogen with time. These types of nanoparticles offer a multi-layered disease management solution: pathogen suppression, effective delivery, and immune activation. Future studies must aim at refining the combinations of these nanosystems to be applicable in the fields of long-term safety and regulation acceptance.138
The various nanoparticle systems for potato disease management, as summarized in Table 2, are discussed below to showcase their relative antimicrobial performance and functional application (Fig. 2).
| Nanosystems | Carrier molecule | Physical properties | Size (nm) | Conc | Diseases | Ref. |
|---|---|---|---|---|---|---|
| CuO/MgO NPs | None/Stabilized via green synthesis | High surface area, porous, mesoporous structure | 6 | 3 mg mL−1 | Potato Brown rot | 139 |
| Chitosan NPs | Chitosan polymer | Biodegradable, mucoadhesive, film-forming, hydrophilic | 93.76 | 200 µg mL−1 | Bacterial wilt | 140 |
| ZnO NPs | Stabilized with plant extracts/starch | Photocatalytic, UV-absorbing, piezoelectric, semiconducting | 11.5 | 30 mg L−1 | Late blight | 141 |
| TiO2 NPs | Surfactants or polymer matrices | Photocatalytic, UV-active, high chemical stability | 20–200 | 50 µg mL−1 | Soft rot | 142 |
| Ag NPs | Stabilized with plant extract or PEG | High conductivity, plasmonic, antimicrobial | 12.7 | 25 ppm | Early blight | 143 |
| MgO NPs | Green synthesis or surfactant-coated | Alkaline, thermally stable, low solubility | 52.5–57.3 | 200 mg L−1 | Black scurf | 144 |
| Fe NMs | Oleic acid or dextran | Magnetic, redox-active, biocompatible | 5–100 | 300 µg mL−1 | Blackleg | 145 |
| rGO-CuO NPs | Reduced graphene oxide (rGO) | High surface area, conductive, hybrid (CuO + graphene) | 5–50 | 1 mg L−1 | Root rot | 146 |
| Cur NPs | Curcumin encapsulated in a polymer matrix | Hydrophobic, antioxidant, fluorescent | 45 | 10 mg mL−1 | Potato virus YNTN | 147 |
| Salicylic acid NPs | Biopolymer (e.g., chitosan) or lipid vesicles | Bioactive, hormone-like, systemic inducer | 20–26.6 | 2.5 mM | Potato leaf roll virus | 148 |
| Ag NMs | PEG or polyvinylpyrrolidone (PVP) | Antiviral, surface-active, crystalline | 12.6 ± 5 | 200 ppm | Tomato spotted wilt virus | 149 |
| Ag NMs | Biopolymer (starch/chitosan) | Stable, antimicrobial, optical-active | 55 | 100 µg mL−1 | Root Knot Nematode | 150 |
The main limitation in the comparison of the antimicrobial efficacy of the nanoparticle-based interventions summarized in Table 2 is that these studies do not always report standardized concentrations. The doses of nanoparticles are quantified in various measures such as mg L−1, ppm, µg mL−1, and mM, which vary by experimental design, the route of application (foliar spray, soil amendment or hydroponic exposure), and the biological scale. Although aqueous systems can be normalized to a limited degree (e.g., mg L−1 = ppm), dosing measures specific to plants cannot be compared quantitatively. Thus, the performance of various nanosystems is viewed qualitatively, focusing on relative performance trends, dose–response behaviour, and the mechanistic mode of action as opposed to absolute concentration values.
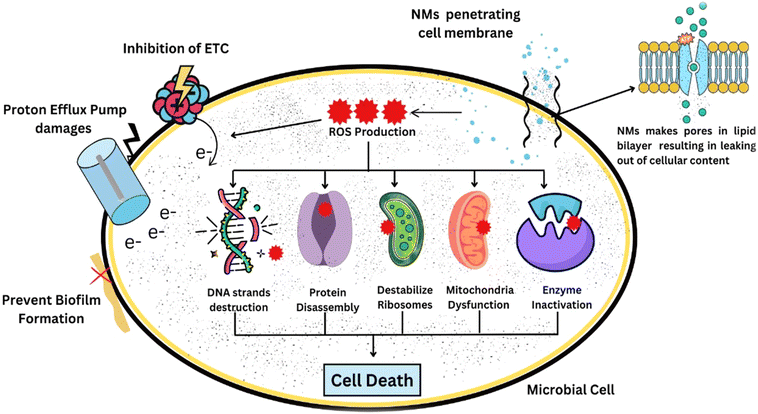
Metallic and metal oxide nanoparticles, like Ag, CuO, ZnO and MgO, can be highly effective as antimicrobials with a broad spectrum of activity, mainly via membrane disruption, release of ions and generation of reactive oxygen species (ROS). These include silver nanoparticles that are highly effective at relatively low doses against bacterial and fungal pathogens such as early blight and bacterial wilt, but the issue of phytotoxicity requires optimization of the dose. The nanoparticles contain zinc oxide and magnesium oxide, which offer a compromise between antimicrobial activities and environmental friendliness and hence can be used repeatedly in agriculture. The polymeric nanoparticles, especially the chitosan-based systems, have moderate antimicrobial activity but have better performance as delivery systems because they are biodegradable and adhesive to plants and have the capacity to stimulate host defense. Chitosan nanoparticles have particular applications in controlling bacterial wilt and viral diseases when combined with bioactive compounds or genetic materials.151 Carbon-based and hybrid nanosystems, including reduced graphene oxide-metal composites, have greater effectiveness due to synergistic effects, which involve physical damage to membranes and the induction of oxidative stress. Altogether, metal-based nanoparticles could be useful for attaining rapid pathogen exclusion, but polymeric and hybrid nanosystems could have better safety, release regulation, and immune-priming properties. These relative distinctions highlight that nanoparticles need to be disease-specific and chosen based on efficacy, levels of phytotoxicity and environmental sustainability as opposed to antimicrobial capabilities alone.152
2.2 Plant protection: enhancing biosecurity through smart nanosystems
Silver nanoparticles exhibit strong antimicrobial behaviour because they can be used to control bacterial and fungal pathogens, causing common potato diseases such as Ralstonia solanacearum (bacterial wilt) or Phytophthora infestans (late blight). However, these nanoparticles can destroy microbial cell walls and prevent resistant strains from developing.140 For example, ZnO nanoparticles have anti-fungal and antibacterial properties and can be used to control potato diseases. These nanoparticles are also being explored as a way to influence plant immunity and warfare against microbial resistance (Fig. 3).162
By acting as carriers, they increase the life of biopesticides because some are destroyed when exposed to UV light such as avermectin.171 Avermectin is loaded into hollow mesoporous silica nanoparticles, which provide a protective shell and release it at a specific site.172 Coating NMs with a drug is another method of drug delivery through NPs. In the absorption method of coating NMs, a drug suspension is prepared, and NMs are mixed in the suspension. They absorb the drugs, and when these drug-coated NMs are applied on infected plants, the drugs reach the target sites with ease. Both the drug and NMs activate antagonistic mechanisms against pathogens. The drug coating completely depends on its solubility in liquid dispersion agents, and solubility depends on the presence of an end functional group either in the drugs or the dispersion agent. In addition, NMs deliver fertilizers and nutrients to plants easily.173
3 Plant disease detection with advanced nanosystems
3.1 Nano-biosensors for disease diagnosis
The early detection of plant pathogens effectively helps monitor crop health and develop an integrated disease management strategy. The real-time detection of microbes has become possible, significantly enhancing disease detection due to the intervention of nano-biosensors.174 Nano-biosensors are synthesized using nanotechnological methods and are composed of nanomaterials such as nano-probes, carbon quantum dots, carbon nanotubes, nanowires and magnetic nanoparticles. Additionally, inorganic nanoparticles, such as Ag, Au and metal oxide nanomaterials, are generally utilized in their fabrication. Previous studies175,176 detected the genomic DNA of Ralstonia solanacearum, the causative agent of bacterial wilt in potato, by functionalizing gold nanoparticles (AuNPs) with single-stranded oligonucleotides. These nanobiosensors facilitate the real-time monitoring of plant health and monitor soil environmental conditions, like pH, temperature, humidity and nutrients. They also detect and monitor the pesticide residues, fertilizer concentrations and other agrochemicals in the crop production system.177 The advancement of nanobiosensors integrates nano-science, computer science, electronics and biology to make them highly sensitive diagnostic tools. Nanobiosensors function by converting chemical signals into digital data that can be measured and monitored by applying electronic devices.178 The fundamental components of nanobiosensors include bio-receptors, transducers and detectors. In the context of plant disease detection, bio-receptors identify microbial signs and symptoms and relay chemical signals to a transducer that converts them into measurable electrical signals. These detectors analyze and amplify these signals into digital data that allow for the precise diagnosis of pathogens.179Historically, the widespread applications of pesticides were employed not only for the management of plant diseases but also inadvertently imposed stress on plant health. However, advancements in plant pathogen detection techniques have enabled significantly precise disease management strategies. Recently, site and target-specific pesticide applications have minimized the adverse effects on plant physiology and overall ecosystem health. The judicious and controlled use of pesticides contributes to biodiversity conservation by minimizing non-target impacts.180 Agriculture has significantly proven to be the most affected sector due to the extensive application of agrochemicals, including pesticides, fungicides and insecticides, which often contain heavy metal ions.181 These toxic metal ions are introduced to crops and soil, subsequently leaching into water bodies. Upon the consumption of food crops, heavy metal ions accumulate in the human body, posing serious threats due to their carcinogenic nature.182 Advanced nanotechnological approaches such as nanobiosensors, have been employed to detect carcinogenic metal ions. In recent advancements, smart nano-biosensors have replaced conventional nano-biosensors for plant pathogen detection and chemical diagnostics. These wearable sensors designed for direct implantation on plant structures such as stems and leaves, exhibit superior flexibility, biocompatibility, cost-effectiveness and lightweight properties, thereby garnering significant interest among phytopathological researchers.183
4 First line of defense: nano-sensors for early disease detection
Early disease detection avoids the spread of pathogens targeting potato crops.184 Biosensors with nanotechnology capabilities identify the presence of a pathogen at extremely low concentrations before the disease spreads, which may require early intervention to prevent loss, thereby reducing the chances of resistance development.185 Optical and electrochemical sensors identify some plant stress responses or microbial markers that could be evolved to treat the disease more precisely without the requirement of broad-spectrum antibiotics and postpone drug resistance onset.1864.1 Nanoimaging
Nanoimaging has emerged as a revolutionary research technique in plant pathology that enables the identification and examination of plant diseases with nanometer-scale precision. Nanoimaging combines nanotechnology with sophisticated imaging modalities to visualize pathogens, plant tissue responses, and disease development at the cellular and subcellular levels. The ultra-high resolution offered by nanoimaging technologies such as Transmission Electron Microscopy (TEM), Scanning Electron Microscopy (SEM), and Quantum Dot (QD)-mediated fluorescent imaging, enables the early and highly specific detection of plant pathogens weeks before the appearance of overt symptoms.187 Underlying nanoimaging is the use of engineered nanomaterials, including metallic nanoparticles (e.g., gold and silver) and semiconductor quantum dots, as biosensing tags or contrast agents. Nanoparticles are typically bound to antibodies, nucleic acids, or ligands with high binding affinity to pathogen-specific markers. Upon injection into infected plant tissues, these nanoprobes bind specifically to targeted structures and generate high-contrast signals in electron- or fluorescence-based imaging systems, thereby enabling direct visualization of pathogenic invasions at ultra-high resolutions.188One of the greatest advantages of nanoimaging for plant disease diagnosis is its ability to detect pathogens during incubation before symptoms appear. This is significant in precision agriculture because early treatment can prevent yield loss and limit the spread of pathogens. For instance, quantum dot-labeled antibodies can efficiently track specific viral or bacterial pathogens in plant tissue at nanometer resolution, which is more sensitive and specific than conventional serological or molecular assays.189 Furthermore, nanoimaging methods are crucial for describing structural and functional changes in plant tissue under pathogenic stress. TEM and SEM are among the methods used to provide comprehensive evaluations of cell wall integrity modifications, intracellular transport mechanisms, and organelle morphology in response to pathogen invasion. TEM provides high spatial resolution although it is limited in intact plant samples. Sample preparation includes fixation, dehydration, sectioning, and staining, which can disrupt the original cellular structure and nanoparticle distribution within cells. TEM also provides a two-dimensional ultra-structure from ultra-thin sections, which is not suitable for 3D localization and long-distance transport of nanoparticles in complex plant tissues. Hence, additional imaging techniques such as confocal laser scanning microscopy or X-ray techniques, are required to better understand nanoparticle distribution in plants. Such specificity is crucial for understanding host–pathogen interactions and the generation of disease-resistant crop varieties.190
Apart from diagnostics, nanoimaging is also crucial in establishing the administration and efficacy of nanotherapeutics. Following the administration of nanoparticles for the targeted delivery of agrochemicals or immunomodulators into plant cells, tracking can be performed using nanoimaging techniques. Tracking thus far enables precise localization, release kinetics, and interaction with pathogens, ultimately enhancing the efficacy of treatment and minimizing environmental residues.191 The integration of nanoimaging methods with computerized analytical systems has enormously amplified their diagnostic capabilities. High-resolution images acquired using transmission electron microscopy (TEM) or fluorescence microscopy can be processed by applying computational algorithms and machine learning schemes, enabling the automation of pathogen detection, measurement of infection intensity, and prediction of disease progression. This fusion of nanotechnology, imaging methods, and artificial intelligence is ready to result in revolutionary changes in plant health monitoring systems.192
Although it has many advantages, nanoimaging in plant pathology is not without limitations. Its high cost of equipment, requirement of advanced laboratory facilities, and requirement of highly skilled personnel may limit its application in common farming routines, especially in the developing world. Moreover, the toxicity and environmental hazards of nanoparticles necessitate rigorous risk assessments and the development of biodegradable and biocompatible nanomaterials. To overcome these limitations, recent advances have emphasized the field applicability and miniaturization of nanoimaging platforms. Continuous work on portable imaging systems, lab-on-chip systems, and smartphone-based diagnostic platforms is aimed at enabling the on-site, real-time diagnosis of plant diseases. These technological innovations seek to democratize the accessibility of nanodiagnostics as accessible tools in both cutting-edge research environments and field-level agricultural operations. In short, nanoimaging is an effective and precise tool for plant disease detection, observation, and interpretation.193 By providing early, sensitive, and specific imaging of pathogen activities and host responses, nanoimaging significantly enhances the field of plant diagnostics and has great potential for sustainable and intelligent agricultural practices.
5 Nanobiotechnology in disease resistance
5.1 Genetic approaches in nanobiotechnology
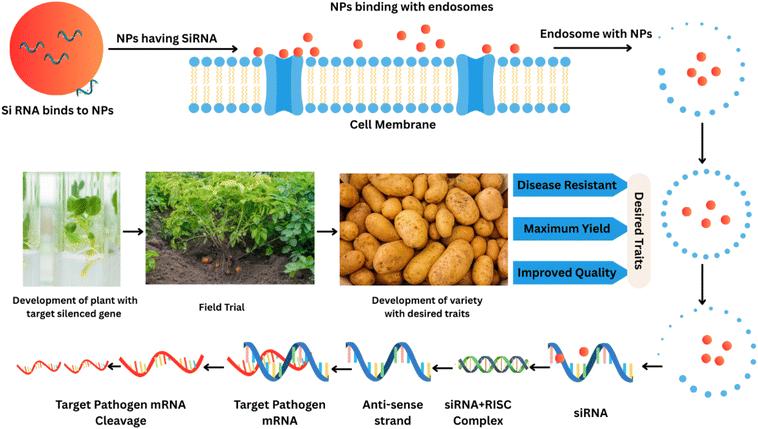
Genetic techniques through nanobiotechnology are being revolutionized to modify plant resistance, especially against microbial diseases. Among the most exciting uses is the use of NPs to create gene delivery systems. Encapsulating these genes allows these NPs to introduce the genes that give plants resistance to different diseases into plant cells more effectively. Nanoparticles can, in turn, be engineered to speed up transformation rates by shielding genetic material from degradation and allowing for absorption in plant tissue. This technique can directly insert beneficial genes into potato plants to make them 10 times more resistant to bacterial, fungal and viral infections. Recently, another significant development in genetic engineering has been the application of CRISPR-nano systems. This method utilizes CRISPR/Cas9 gene editing technology to exactly alter the DNA in the potato plant, which is delivered by nanoparticles.194 Scientists can deliver CRISPR components of particular genes to the target cells and modify them to alter their susceptibility to microbial infection. The targeted change is accomplished through the induction of advantageous alleles, or through the knockout of deleterious regulatory genes, to improve plant immunity. The combination of CRISPR technology and nanoparticles simultaneously increases gene editing efficiency while decreasing off-target effects, making the procedure safer and more accurate. However, given all these, there seems to be a lot of opportunity for utilizing nanobiotechnology-based genetic strategies to develop potato cultivars that are more resistant to microbial challenges. CRISPR gene editing combined with nanoparticle-based gene delivery offers a promising strategy for developing resilient crops, thereby reducing the need for chemical treatments and supporting sustainable agriculture. Nanoparticles can deliver RNAi molecules to improve a plant's defense mechanism or silence genes that promote pathogen virulence. This technique reduces the development of microbial resistance by focusing on the infection's use of specific molecular pathways.5.2 Genetic warfare: nanoparticles disrupting microbial resistance
| Nanosystems | Genome editing tools | Target pathogens | Target gene | Mode of action | Application method | Focus area | Ref. |
|---|---|---|---|---|---|---|---|
| Carbon quantum dots | CRISPR-Cas9 | Ralstonia solanacearum | StSR4 (resistance gene) | Disruption of a susceptible gene | Nanoparticle-mediated foliar spray | Bacterial wilt resistance | 198 |
| Chitosan nanoparticles | CRISPR-Cas9 | Potato virus Y (PVY) | Viral coat protein gene | Virus genome silencing | In planta DNA-free delivery | Virus resistance | 199 |
| Gold nanoparticles | CRISPR, RNAi | Fungal/bacterial pathogens | miRNA targets | RNA silencing + gene knockout | Infiltration into potato leaves | Dual protection strategy | 200 |
| Silica-based nano-DNA complexes | CRISPR/Cas9 | Pseudomonas syringae | S gene (susceptibility) | Targeted gene disruption | Nanocarrier foliar coating | Bacterial speck resistance | 201 |
| Lipid-based nanocarriers | CRISPR-Cas | Soil-borne microbes | R genes and effector-binding genes | Priming of immunity | Soil-root zone nanoinjection | Root disease prevention | 202 |
| Layered double hydroxide (LDH) nanosheets | Talen, CRISPR | Mixed bacterial load | PPO, StNPR1 | Transcription regulation | PEG-mediated protoplast transfection | Stress-resilient potatoes | 203 |
| Functionalized nanoparticles | ZFN, CRISPR | General pathogens | GBSSI (starch modulation gene) | Knockout for structural resistance | Somatic cell transformation | Resistance + yield traits | 204 |
| Chitosan-CaNPs | CRISPR-Cas9 | Potato virus Y | PVY coat protein | RNA interference | Direct leaf infiltration | Broad-spectrum antiviral defense | 205 |
| Polymeric nanoparticles | CRISPR | Phytophthora infestans (late blight) | Rpi-vnt1 | Resistance gene introgression | Agrobacterium + nanoparticles | Blight suppression | 206 |
| Chitosan nanoparticles (CS-NPs) | CRISPR-Cas9 | Phytophthora infestans | StRLK, Rpi-blb2, StWRKY8 | Knockout of susceptibility genes; enhanced PR protein expression | Foliar spray/root absorption | Disease resistance engineering | 207 |
| Lipid-based nanoparticles (LNPs) | CRISPR-Cas12a | Pseudomonas syringae | StDMR6-1 | Gene silencing via HDR-mediated knock-in | Vacuum infiltration | Bacterial resistance | 208 |
| Silica mesoporous nanoparticles (MSNs) | TALEN | Ralstonia solanacearum (bacterial wilt) | StSWEET10 (sugar transporter) | Disrupt pathogen nutrient uptake | Soil drench/hydroponics | Metabolic engineering | 209 |
| Silica nanocarriers | SHREDER | Leptinotarsa decemlineata (beetle) | Male-specific gene disruption | X-shredder gene drive reduces the beetle population, affecting the disease vector | Microinjection into eggs | Pest-based disease vector control | 210 |
| Polymeric nanoassemblies | CRISPR/dMac3-Cas9 | Fungal pathogens (mixed) | Starch metabolism genes | Multiplex editing to alter tuber chemistry, favoring resistance | Tuber-specific editing | Dual focus: Quality & microbial defense | 211 |
| Mesoporous silica nanoparticles | ZFN | Mixed microbial pathogens | StNPR1, StR3a | Activates defense-related transcriptional cascades | Soil root zone inoculation | General resistance boost | 212 |
| Nano-clay-based carriers | TALEN + CRISPR mix | Fusarium spp. | Defense enzymes (PAL, PR-1) | Enhances pathogen recognition and anti-fungal response | Leaf and tuber treatment | Fungal immunity | 213 |
| Gold nanoparticles (AuNPs) | CRISPR/Cas13a | Potato Virus Y (PVY) | Viral RNA (PVY) | RNA targeting system blocks viral replication | Foliar nanoparticle delivery | RNA virus interference | 214 |
Virus-specific nano-strategies can be tremendously useful in the treatment of potato viral diseases, especially PVY and PLRV. RNAi based on nanoparticles is carried out by utilizing siRNAs or dsRNAs that are viral in origin and are used to silence important viral genes, including those involving the coat protein and replication proteins. Polymeric, lipid, or inorganic nanoparticles are employed as nanoparticle carriers of RNA, resulting in greater antiviral efficacy and prolongation of antiviral duration. Nanoparticles may also be utilized to conjugate coat proteins of viruses, disrupting the assembly of the virus, its stability, or motility. Nano-based antivirals are more specific, less toxic to off-target and less prone to the development of resistance, making them the most suitable in the control of persistent and continually evolving potato viruses compared to traditional chemical-based antivirals.219
The targeted administration reduces detrimental effects on the environment through the efficient use of synthetic pesticides.215,220 Additionally, by employing nanocarriers, the stability and bioavailability of these medicines can be increased, guaranteeing prolonged release and better pathogen engagement. This is because the targeted mode of action can avoid conventional resistance routes. This strategy interferes with the life cycle of phytopathogens and reduces the chances of resistance emergence.221 Delivery methods mediated by nanoparticles are used to lower infection rates in potato crops. Furthermore, potato cultivars with improved resistance profiles can be developed by combining nanotechnology with conventional breeding and genetic engineering techniques.147 To achieve these advantages completely and handle any possible hazards related to the use of nanoparticles in agricultural systems, more in-depth research is vital.215
Although nanotechnology has a promising future in sustainable potato disease management, there are some limitations and environmental hazards that should be considered to ensure its better application in the field. The frequent application of engineered nanomaterials can result in their accumulation in agricultural soils, which can potentially affect soil physicochemical characteristics and positive microbial communities. Moreover, nanoparticles that are absorbed by the roots or deposited on foliage can be translocated into edible tubers, raising concerns about trophic transfer and long-term food safety. Some nanomaterials can also cause unexpected ecotoxicological impacts on non-target organisms in high concentrations due to oxidative stress or membrane damage. Hence, future research must focus on dose optimization, biodegradable or stimulus-responsive nanosystems, long-term field validation, and standardized regulatory motions so that the safety of the environment and acceptance by people are achieved.228
Another factor is whether long-term or repeated exposure to nanomaterials may lead to the development of resistance to plant pathogens. Compared with traditional agrochemicals, which often target sites, many nanomaterials have multi-modal antimicrobial effects such as physical disruption of the membranes, ion release and the production of reactive oxygen species, which decrease the chances of developing stable resistance. Nevertheless, these adaptive responses such as improvements in antioxidant defenses, biofilm formation or changed cell surface properties, cannot be eliminated entirely during long-term selective pressure. Consequently, nano-enabled strategies must be applied responsibly because dose optimization, rotation or combination with biological or conventional controls and avoiding chronic sub-lethal exposure may reduce the chances of resistance formation.229
6 Plant defense system: modulating innate immunity through nanomaterials
6.1 Activation of the defense system against microbiota
NPs enhance the immunological response of potato plants, resulting in the activation of the defense system. Roots or leaves of plants are immersed in NPs, like silica-, metal oxide (zinc oxide and copper)- and carbon-based nanomaterials.230 These NPs induce defense mechanisms through ROS production and serve as signaling molecules. ROS stimulate the plant's immune system depending on the defense-related genes that are activated.226,231 This improved immune response increases the production of defensive enzymes such as peroxidase, polyphenol oxidase and phytoalexins, which are naturally occurring antimicrobial compounds. Nanoparticles may also activate plant defense signaling pathways, including jasmonic acid and salicylic acid (SA), which induce PR genes to encode pathogenesis-related (PR) proteins that help protect plants against pathogen attack. This type of resistance is known as systemic acquired resistance (SAR).232 The SAR is the plant's natural defense system against bacteria, viruses and fungi, which is activated upon the attack of pathogens with no need for pesticide application.233 Besides ROS and hormone stimulation, nanoparticles trigger resistance via SA and JA signaling interactions rather than just their stimulation. Biotic resistance is typically associated with SA, and necrotrophic with JA, but the proportion of the two can be modulated by nanoparticle treatment based on the nanoparticle type, concentration, and mode of administration. Silica and metal oxide NPs can trigger SA signaling priming and tune genes sensitive to JA without antagonism and in concerted immunity.Functional genetic analysis and gene expression confirm the immunity caused by nanoparticles. PR genes (PR1, PR2, and PR5) are consistently up-regulated in response to NP treatment; their functionality validates their contribution to the resistance caused by nanoparticles to differentiate between antimicrobial and immune responses in plants.234 The introduction of nanoparticles in agriculture has reduced the overuse of conventional pesticides, leading to a reduction in microbial resistance due to the repeated use of chemicals.226 In addition to activating the defense system, nanoparticles are used to deliver small amounts of fertilizer, insecticide or antimicrobial agents under a time-released system to ensure the death of the targeted pathogen with minimal input of chemicals. This reduces environmental pollution, lowers the chance of developing pest resistance and develops sustainable agriculture. Silica and carbon-based nanoparticles play a role in boosting the natural defense systems of plants. For example, silica nanoparticles (SiO2-NPs) may help potato plants by developing systemic acquired resistance (SAR) towards disease.235 For example, carbon nanotubes (CNTs) have been beneficially affecting plant growth and thence immunological response without heavy chemical treatment, aiding in the resistance of potatoes to microbial diseases.236
6.2 Plant defense systems against abiotic stresses
Nanosystems have the potential to improve the resistance of potato plants against three primary abiotic stresses: temperature (heat and cold), humidity (drought and waterlogging), and UV radiation. Despite abiotic stress tolerance seeming marginal to disease resistance, growing evidence suggests that abiotic stresses (drought, temperature extremes, and waterlogging) have a significant and detrimental effect on plant disease resistance through the disruption of physiological barriers and immune signaling pathways. Stress-induced disruption of redox homeostasis, membrane integrity and the hormonal signaling systems in potato has the potential to impair defense mechanisms, thus reducing the disease severity. Based on this, nano-mediated mitigation of abiotic stress can indirectly increase disease resistance by maintaining immune competence, cellular homeostasis and plant resilience under related stress conditions. Hence, the adaptation of abiotic stress control is directly connected to the development of disease-resistant potato systems with robustness.237 We describe the mechanisms behind the protection by nanoparticles and cite recent studies with an emphasis on sustainable potato production applications.Reciprocal grafting proved that potato tubers synthesize anthocyanins in situ, not from leaves. UV-B irradiation of higher intensity (2.5 kJ m−2 days−1) significantly enhanced total anthocyanin contents and some anthocyanidins (pelargonidin and peonidin) in red-fleshed potatoes. Transcriptome analysis indicated upregulation of anthocyanin-related enzymatic genes (PAL, C4H, 4CL, CHS, CHI, F3H, F3′5′H, ANS, UFGTs, and GSTs) with UV-B treatment. Transcription factors, like StbHLH1 and several MYB factors, were induced at increased expression under UV-B, controlling anthocyanin biosynthesis. Low-dose UV-B treatment (5 kJ m−2) promoted the expression of anthocyanin structural genes without altering agronomic traits, while higher doses (10 kJ m−2) damaged plants and reduced yield. This emphasizes the need to optimize nanoparticle and UV exposure for desirable effects without stress damage.
6.3 Nanosystems trigger general defense mechanisms
Nanoparticles induce potato plant defense in various ways. They enhance enzymatic antioxidant defense (SOD, CAT, APX, and GPX) and boost non-enzymatic antioxidants (ascorbate, glutathione, phenols, and flavonoids). This enhances the suppression of ROS from abiotic stress. Nanoparticles trigger osmolyte production such as proline, glycine betaine, and sugars, which maintain turgor and protect macromolecules.236 They affect stress hormone signaling (ABA, ethylene, and jasmonic acid) and trigger calcium signaling for stress. Nanoparticle treatment can precondition defenses for quicker responses to future stresses. They maintain chlorophyll retention and photosystem II activity, protecting thylakoid membranes and retaining photosynthesis for recovery.2426.4 Omics
Recent technological advancements in omics technologies have improved the understanding of nanoparticle-induced stress tolerance. Nanoparticle treatment controls gene expression related to perception, signaling, and response to stress. Important pathways controlled are MAPK signaling, phenylpropanoid biosynthesis, and hormone metabolism. NMs control stress-related proteins (HSPs, dehydrins, and ROS-scavenging enzymes) and protein phosphorylation, influencing functionality. NMs also reorganize metabolite profiles, sustaining stress tolerance and protective compounds such as anthocyanins, flavonoids, and phenolic acids. Nanoparticles can induce DNA methylation changes in gene expression, while histone modifications can determine stress-responsive chromatin states. These omics strategies reveal the intricate control of nanoparticles on plant stress responses, moving away from phenomenology to mechanistic understanding.2417 Conclusion
The extensive use of pesticides to control diseases plays a role in the development of pathogen resistance. Nanobiotechnology has played a significant role in conferring microbial resistance and enhancing disease resistance in potato plants through the manipulation of different nanomaterials at the nanoscale. As carriers, NMs organize the controlled release of drug delivery against the attack of pathogens in order to mitigate the effects of synthetic chemicals, which deteriorate the environment. NMs have proven themselves to be shields against pathogens and their chemical-based management strategies in order to conserve biodiversity. In addition to disease protection in the potato crop, they have offered extensive roles to be used as carriers of RNAi in order to silence the virulence genes of microbes. Despite playing an adventitious role in modern plant disease management, the field still needs a lot of improvement. The delivery and penetration of NMs in both plant and microbial cells need to be approved because both depend on the size and the charge of NMs. There is a lot of research gap existing on defining the cost of synthesis and applications, their uptake by plants, their stability and solubility, shelf life and other properties of NMs that should be the focus of researchers and nano-scientists. Environmental safety, regulatory frameworks and phytotoxicity concerns still need discussions to improve them. All these improvements will be very important in describing the ability of nanobiotechnology to revolutionize agriculture, assure global food safety and develop sustainability.8 Future directions and challenges
The promising future of nanobiotechnology in plant disease management with several directions for research and development includes the development of multifunctional NMs like targeted delivery as a carrier for disease detection, disease treatment, nutrient supplementation, and as an immunity booster simultaneously in plants to improve the plant health status. The development of nanobiosensors for disease detection and the reduction of chemical usage and dosage for disease treatments provide enough areas for future directions. Environmental protection, dose-dependent phytotoxicity, and regulatory provisions need further research before the application of nanoparticle-based formulations in agriculture on a large scale. Future studies need to focus on long-term plant safety research, setting of crop-specific exposure limits and alignment with new regulatory standards to enable nano-enabled plant protection technologies to be both effective and environmentally friendly. All these future directions lead nanobiotechnology to greater precision, stability and effectiveness for plant disease management strategies. The difficulty in the handling and preparation of NMs on a large scale, their side effects when applied on plants and the challenges in the evaluation of risk assessments for their use are the major obstacles that hinder the adoption of nanobiotechnology as a plant disease management strategy. Future studies must focus on the mechanistic degradation of immune responses induced by nanoparticles using functional genomic methods. Studies with hormone signaling mutants, PR gene knockouts, and CRISPR/Cas editing of key defense regulators will be crucial to understand SA–JA interactions and establish causal defense pathways. The integration of transcriptomics, proteomics, and metabolomics analyses with gene perturbation provides a system-level understanding of disease resistance triggered by nanoparticles, allowing for the sensible design of new-generation nano-enabled crop protection methods. To further enhance translational potential, future studies must focus on designing biodegradable and stimulus-responsive nanomaterials that can degrade or activate in response to particular biological or environmental signals, thus minimizing environmental persistence. Moreover, the integration of nanosensor platforms with AI and machine learning (ML) models may enable real-time disease surveillance, predictive disease forecasting, and precise nano-enabled intervention deployment. These data-driven and adaptive plant protection systems represent an essential next step towards sustainable and intelligent disease-resistant potato production.Conflicts of interest
The authors declare no conflicts of interest.Abbreviations
| NMs | Nanomaterials |
| NPs | Nanoparticles |
| CNTs | Carbon nanotubes |
| QD | Quantum dot |
| CS-NPs | Chitosan nanoparticles |
| LNPs | Lipid-based nanoparticles |
| AuNPs | Gold nanoparticles |
| SiO2-NPs | Silica nanoparticles |
| MSNs | Mesoporous silica nanoparticles |
| LDH | Layered double hydroxide |
| ROS | Reactive oxygen species |
| PR | Pathogenesis-related |
| RNAi | RNA interference |
| siRNAs | small interfering RNAs |
| CRISPR | Clustered regularly interspaced short palindromic repeats |
| Cas9 | CRISPR-associated protein 9 |
| ZFNs | Zinc finger nucleases |
| TALENs | Transcription activator-like effector nucleases |
| SHREDDER | Short hairpin RNA enhancing degradation of disease-associated exons via RNA interference |
| FAO | Food and agriculture organization |
| KPK | Khyber Pakhtunkhwa |
| TDAP | Trade development authority of Pakistan |
| TEM | Transmission electron microscopy |
| SEM | Scanning electron microscopy |
| SOD | Superoxide dismutase |
| CAT | Catalase |
| POD | Peroxidase |
| APX | Ascorbate peroxidase |
| GPX | Glutathione peroxidase |
| MAPK signalling | Mitogen-activated protein kinase |
| HSFs | Heat shock factors |
| HSPs | Heat shock proteins |
| MAMPs | Microbe-associated molecular patterns |
| PAMPs | Pathogen-associated molecular patterns |
| PRRs | Pattern recognition receptors |
| PTI | Pattern triggered immunity |
| ETI | Effector triggered immunity |
| HR | Hypersensitive response |
| SAR | Systemic acquired resistance |
| PEI | Polyethyleneimine |
| NHEJ | Non-homologous end joining |
| HDR | Homology-directed repair |
| DMR | Differentially methylated region |
| NLRs | Nucleotide-binding domain and leucine-rich repeat-containing receptors |
Data availability
No primary research results, software or code have been included and no new data were generated or analyzed as part of this review.Acknowledgements
This work was supported by the Yibin Science and Technology Plan (2022NY011), Research on the Bio-synthesis of a Long-acting, Highly Active Follicle-Stimulating Hormone (FSH) for Enhanced Ovulation in Dairy Cows, funded by National Center of Technology Innovation for Dairy (2025-QNJJ-010), Science and Technology Project of Inner Mongolia Autonomous Region (2025YFHH0267).References
- H. De Jong, Am. J. Potato Res., 2016, 93, 415–429 CrossRef.
- M. E. Calıskan, A. Bakhsh and K. Jabran, Potato Production Worldwide, Academic Press, 2022 Search PubMed.
- D. Islamova, U. Kholiyorov, K. Dustmukhammad, F. Shafkarov and K. Numonov, E3S Web of Conferences, 215, p. , p. 02001 Search PubMed.
- S. K. Gupta and T. S. Thind, Disease Problems in Vegetable Production, Scientific Publishers, 2nd edn, 2018, p. 616 Search PubMed.
- N. W. A. Hamid and K. Nadarajah, Int. J. Mol. Sci., 2022, 23, 8998 CrossRef PubMed.
- C. J. Chiquito-Contreras, T. Meza-Menchaca, O. Guzmán-López, E. C. Vásquez, J. Ricaño-Rodríguez, C. J. Chiquito-Contreras, T. Meza-Menchaca, O. Guzmán-López, E. C. Vásquez and J. Ricaño-Rodríguez, Front. Biosci., 2024, 16, 9 CrossRef PubMed.
- X. Q. Yu, H. Q. Niu, C. Liu, H. L. Wang, W. Yin and X. Xia, Plant Biotechnol. J., 2024, 22, 2113–2128 CrossRef.
- B. C. Remick, M. M. Gaidt and R. E. Vance, Annu. Rev. Immunol., 2023, 41, 453–481 CrossRef.
- J. Mensah, Cogent Soc. Sci., 2019, 5, 1653531 Search PubMed.
- S. Zhang, C. Li, J. Si, Z. Han and D. Chen, Int. J. Mol. Sci., 2022, 23, 6758 CrossRef PubMed.
- I. P. de León and M. Montesano, Int. J. Mol. Sci., 2013, 14, 3178–3200 CrossRef.
- B. Verma, N. Kumar and G. Pratap, Dr A. B. Jadhao, Research Trends in Agricultural Sciences, 2023, vol. 2, p. 87 Search PubMed.
- A. Hameed, M. Zeeshan, R. Binyamin, M. W. Alam, S. Ali, M. S. Zaheer, H. Ali, M. W. Riaz, H. H. Ali and M. S. Elshikh, PeerJ, 2024, 12, e17518 CrossRef.
- J. Wu, J. Liu, S. Liu, U. Handique, Q. Sun, Z. Feng and R. Zhang, Eur. J. Plant Pathol., 2024, 169, 121–130 CrossRef CAS.
- E. Selem, A. Gouda, W. A. Hassanin and F. M. Reda, Bulletin of Faculty of Science, Zagazig University, 2024, vol. 2024, pp. 126–131 Search PubMed.
- E. Osdaghi, J. M. van der Wolf, H. Abachi, X. Li, S. H. De Boer and C. A. Ishimaru, Mol. Plant Pathol., 2022, 23, 911–932 CrossRef CAS PubMed.
- S. A. Khmeleva, L. K. Kurbatov, K. G. Ptitsyn, O. S. Timoshenko, D. D. Morozova, E. V. Suprun, S. P. Radko and A. V. Lisitsa, Int. J. Mol. Sci., 2024, 25, 12218 CrossRef CAS.
- E. S. Hajiyev, A. D. Mammadova, A. M. Karimova, S. V. Hajiyeva and R. T. Aliyev, Recommended for publication by the Scientific and Technical Council of the Mayak Research Station of the Institute of Vegetable and Melon Growing of the NAAS, Protocol No. 1 dated February 29, 2024. Responsible for the publication: Oleksandr POZNYAK, 2024, p. 74 Search PubMed.
- R. Czajkowski, M. C. M. Pérombelon, J. A. van Veen and J. M. van der Wolf, Plant Pathol., 2011, 60, 999–1013 CrossRef.
- Y. Degefu, J. Phytopathol., 2024, 172, e13282 CrossRef CAS.
- E. J. Wenninger and A. Rashed, Annu. Rev. Entomol., 2024, 69, 139–157 CrossRef CAS.
- C. S. Padilla, S. C. Irigoyen, M. Ramasamy, M. B. Damaj, M. M. Dominguez, D. Rossi, R. H. Bedre, W. O. Dawson, C. El-Mohtar, M. S. Irey and K. K. Mandadi, Plant Biotechnol. J., 2025, 23, 1876–1878 CrossRef CAS.
- R. Martinez-Gutierrez, M. E. M. Herrera, C. Trejo, I. H. Almeyda-Leon and H. A. Lopez-Delgado, Trop. Subtrop. Agroecosyst., 2025, 28 DOI:10.56369/tsaes.5303.
- E. Schlachter, S. Ruth, M. Satoh-Cruz and J. F. Willbur, Plant Health Prog., 2024, 25, 305–315 CrossRef.
- S. Banerjee, In Molecular and Biotechnological Tools for Plant Disease Management, J. T. Chen, M. Khan, A. Parveen and J. K. Patra, Springer Nature Singapore, 2024, pp. 119–158 Search PubMed.
- L. Nateghi, E. Hosseini and F. Mirmohammadmakki, Iran. J. Microbiol., 2024, 16, 62 Search PubMed.
- M. A. A. Seleim, M. F. F. Bereika, O. H. M. Ibrahim, A. I. Alqubaie and K. A. M. Abo-Elyousr, J. Plant Dis. Prot., 2023, 131, 65–75 CrossRef.
- G. Ababa, Cogent Food Agric., 2024, 10, 2407953 CrossRef.
- L. A. Okiro, R. M. Mulwa, M. E. Oyoo and S. Nyalala, CBI Rev., 2024, 19, 001 Search PubMed.
- S. Kumar, S. K. Biswas, A. Kumar, V. D. Rajput, R. Kumar, S. Kumar, K. H. Thanuja and R. Kumar, Potato Res., 2023, 67, 271–292 CrossRef.
- N. Uysal, A. Bozkurt and E. Elçi, Potato Res., 2025, 68, 2749–2775 CrossRef CAS.
- L. Giroux, I. Isayenka, S. Lerat, N. Beaudoin and C. Beaulieu, Front. Microbiol., 2024, 15, 1466927 Search PubMed.
- M. J. Ryley, Hist. Record. Aust. Sci., 2023, 35, 142–150 CrossRef.
- A. Rabea, E. Naeem, N. M. Balabel and G. E. Daigham, Bot. Stud., 2023, 64, 20 Search PubMed.
- M. M. Haque, G. C. Das, Md. M. Faysal, M. A. Hossain, M. Haque, S. Miah, J. Farthouse, M. Rahman and Md. N. H. Mehedi, Int. J. Radiat. Biol., 2025, 101, 382–390 Search PubMed.
- J. Wu, R. Zhang, Q. Sun, Z. Feng and U. Handique, Plant Dis., 2024, 108, 2976–2982 CrossRef CAS PubMed.
- D. A. Johnson, J. K. S. Dung, T. F. Cummings and B. K. Schroeder, Plant Dis., 2011, 95, 285–291 CrossRef.
- J. L. Valdez-López, E. Peréz-López, C. I. Mora-Zamudio, J. Méndez-Lozano, E. A. Rodríguez-Negrete, M. E. Santos-Cervantes and N. E. Leyva-López, Plant Dis., 2025, 109, 1581 CrossRef.
- A. Jain, S. Sarsaiya, Q. Wu, Y. Lu and J. Shi, Bioengineered, 2019, 10, 409–424 CrossRef CAS.
- E. M. Moradzade and M. Hosseini, J. Appl. Res. Plant Protect., 2024, 13, 87–95 Search PubMed.
- S. Sharma, R. K. Tiwari, V. Sagar and C. Maharana, in Approaches for Potato Crop Improvement and Stress Management, eds. S. M. P. Khurana, J. E. Bradshaw and V. Bhardwaj, Springer Nature Singapore, 2024, pp. 179–231 Search PubMed.
- A. Kaur, V. Sharma and A. Kumar, Potato Res., 2022, 65, 863–879 CrossRef CAS.
- Y. B. Naim and Y. Cohen, J. Fungi, 2023, 9, 1046 CrossRef.
- S. Upadhyay, J. Jain and R. Prasad, Int. J. Exp. Res. Rev., 2024, 38, 15–25 CrossRef.
- M. Atiq, M. F. Ullah, N. A. Rajput, A. Hameed, S. Ahmad, M. Usman, A. Hasnain, A. Nawaz, S. Iqbal and H. Ahmad, Arch. Phytopathol. Plant Protect., 2024, 57, 387–404 CrossRef CAS.
- T. Schmey, C. S. Tominello-Ramirez, C. Brune and R. Stam, Mol. Plant Pathol., 2024, 25, e13435 CrossRef.
- J. Park, S. Kim, M. Jo, S. An, Y. Kim, J. Yoon, M.-H. Jeong, E. Y. Kim, J. Choi, Y. Kim and S.-Y. Park, J. Fungi, 2024, 10, 53 CrossRef CAS.
- S. Jain, R. Rani, P. Salaria and S. Kaur, Am. J. Potato Res., 2024, 101, 306–311 CrossRef.
- S. Chaudhary, M. Lal, S. Sagar, S. Sharma and M. Kumar, Trop. Plant Pathol., 2023, 49, 169–192 CrossRef.
- R. Kumar, P. Mishra, P. Kumar, P. K. Verma, D. Singh and P. Ahlawat, Ecol. Environ., 2023, 41, 1002–1009 Search PubMed.
- S. I. Macchiavelli-Girón, S. A. Jordan, R. E. Macchiavelli, J. Spychalla, E. Monette, J. Crane, Y. Chen and A. J. Gevens, Plant Dis., 2025, 0363 Search PubMed.
- G. Irshad and M. F. A. F. Naz, Mycopath, 2014, 11 Search PubMed.
- M. Sanzo-Miró, A. Medina, L. A. Terry and M. C. Alamar, Int. J. Food Microbiol., 2024, 423, 110843 CrossRef.
- S. Chaudhary, M. Lal, S. Sagar, S. Sharma and M. Kumar, Trop. Plant Pathol., 2023, 49, 169–192 Search PubMed.
- S. Abo-Ake, W. Naffaa and M. J. Mando, Jordan J. Biol. Sci., 2024, 17, 21–27 Search PubMed.
- S. A. H. Naqvi, A. Abbas, M. Farhan, R. Kiran, Z. Hassan, Y. Mehmood, A. Ali, N. Ahmed, M. Z. Hassan, A. F. Alrefaei, F. Ölmez, S.-H. Yang and F. S. Baloch, Plants, 2024, 13, 715 Search PubMed.
- M. A. Kuznetsova, N. V. Statsyuk, V. N. Demidova, I. N. Semeniuk, T. I. Smetanina, A. Y. Ukolova and A. A. Vyatchinov, Agronomy, 2024, 14, 1373 Search PubMed.
- M. Sanzo-Miró, D. M. Simms, F. I. Rezwan, L. A. Terry and M. C. Alamar, Am. J. Potato Res., 2023, 100, 362–370 Search PubMed.
- Y. Zhao, D. Wang, C. Zhang, J. Zhang, L. Xu, Y. Cheng, X. He and H. Zhou, J. Phytopathol., 2024, 172, e13310 Search PubMed.
- P. C. Bethke, Am. J. Potato Res., 2023, 100, 253–264 Search PubMed.
- P. Wang, L. Cheng, J. Pan, L. Ma, X. Hu, Z. Zhang, D. Li, Y. Zhu, S. Chang, P. Yuan, P. Kear, L. Lassois, G. Zhu, S. Huang, H. Du and C. Zhang, aBIOTECH, 2025, 6, 22–32 Search PubMed.
- I. M, K. Shehu, T. U and I. TAFINTA, Ann. Biol. Sci., 2014, 2, 1–4 Search PubMed.
- A. Arshaghi, M. Ashourian and L. Ghabeli, Multimed. Tool. Appl., 2022, 82, 5725–5742 CrossRef.
- S. R. Mohammed, I. D. Eskov and E. M. Zeitar, Open Agric. J., 2020, 14, 339 CrossRef CAS.
- D. Semyalo, Y. Kim, E. Omia, M. A. A. Arief, H. Kim, E.-Y. Sim, M. S. Kim, I. Baek and B.-K. Cho, Agriculture, 2024, 14, 2014 CrossRef.
- S. Kc, A. Poudel, D. Oli, S. Ghimire and P. B. Angon, Int. J. Phytopathol., 2023, 12, 207–219 Search PubMed.
- D. Wang, H. Weng, Y. Zhao, H. Zhou, H. Guo, H. Cheng, J. Shen, M. Yin, S. Yan and X. Su, J. Agric. Food Chem., 2025, 73, 7121–7130 CAS.
- M. Li, J. Yang, H. Li, Y. Wang, X. Cheng, G. Han, T. Bisseling and J. Zhao, Front. Microbiol., 2024, 15, 1396044 Search PubMed.
- B. T. L. H. van de Vossenberg, C. Prodhomme, J. H. Vossen and T. A. J. van der Lee, Mol. Plant Pathol., 2022, 23, 461–474 Search PubMed.
- E. Çakir and F. Demirci, Interdepartmental Thematic Scientific Collection of Phytosanitary Safety, 2024, pp. 301–313 Search PubMed.
- R. Asakaviciute, A. Zelya, A. Kacergius, T. Andriychuk, G. Zelya, A. Skoreyko and A. Razukas, Sci. Rep., 2025, 15, 5915 CrossRef CAS.
- K. Ogero and R. van der Vlugt, in Handbook of Vegetable and Herb Diseases, Springer International Publishing, Cham, 2023, pp. 1–59 Search PubMed.
- J. R. A. Reiabi and A. Salariyan, J. Appl. Plant Biol., 2024, 2, 1–10 Search PubMed.
- S. Dangi, Oomycete Potato Pathogens: Characterization, Diagnostics, and Disease Management, Doctoral Dissertation, University of Idaho, 2022 Search PubMed.
- J. R. Henrie, Investigating Secondary Metabolites of Streptomyces and Bacillus Bacteria that Inhibit Colony Growth of Pathogenic Potato Fungi (Colletotrichum coccodes, Pythium ultimum), Master’s thesis, Brigham Young University, 2024.
- M. Beltramino, S. Landoni, M. Grbich, E. Vuletic, M. Bressano and A. Albarracín Orio, Eur. J. Plant Pathol., 2024, 171, 111–117 CrossRef.
- Md. Aktaruzzaman, S. J. Xu, J. Y. Kim, J. H. Woo, Y. I. Hahm and B. S. Kim, Mycobiology, 2014, 42, 206–209 CrossRef PubMed.
- A. Bibi, F. Mubeen, A. Rizwan, I. Ullah, M. Hammad, M. A. B. Waqas, A. Ikram, Z. Abbas, D. Halterman and N. A. Saeed, J. Fungi, 2024, 10, 701 Search PubMed.
- S. Sharma, R. K. Tiwari, V. Sagar and C. Maharana, in Approaches for Potato Crop Improvement and Stress Management, Springer Nature Singapore, Singapore, 2024, pp. 179–231 Search PubMed.
- J. Quenouille, N. Vassilakos and B. Moury, Mol. Plant Pathol., 2013, 14, 439–452 CrossRef CAS PubMed.
- A. Kajla, A. Kumar and S. Kumar, Biochem. Cell. Arch., 2023, 24, 1059–1065 Search PubMed.
- M. Mostafa and Md. M. Hossain, Online J. Biol. Sci., 2024, 24, 858–876 Search PubMed.
- J. Verchot, Mol. Plant Pathol., 2022, 23, 315–320 CrossRef CAS.
- A. Saidi, Z. Hajibarat, A. M. Gorji and R. Ahmadvand, Nucleus, 2024, 68, 227–241 CrossRef.
- M. E. M. Tibambre, J. D. S. Correa and F. A. A. Gutierrez, J. Exp. Mol. Biol., 2024, 25, 267–276 CrossRef CAS.
- R. R. Kumar, M. Ansar, K. Rajani, J. Kumar and T. Ranjan, BMC Res. Notes, 2020, 13, 523 CrossRef.
- J. Ali, A. Bayram, M. Mukarram, F. Zhou, M. F. Karim, M. M. A. Hafez, M. Mahamood, A. A. Yusuf, P. J. H. King, M. F. Adil, Z. Ma and I. H. Shamsi, Sustainability, 2023, 15, 11150 CrossRef CAS.
- M. Kholmatova, V. Fayziev, B. Akhmadaliev, T. Absamatov, A. Temirov, T. Tangriev, D. Jovlieva and M. Askarova, J. wildl. biodivers, 2024, 8, 149–161 Search PubMed.
- R. Falloon, I. Kirkwood, C. Delmiglio, C. Bleach, J. Monk and S. Clelland, N. Z. Plant Protect, 2024, 77, 25–35 Search PubMed.
- L. Mao, H. Shi, M. U. Sial, L. Zhang, L. Zhu, C. Wu and X. Liu, Crop Prot., 2024, 184, 106877 CrossRef CAS.
- H. Kamal, K. V. Kotapati, K. Tanaka and H. R. Pappu, Int. J. Mol. Sci., 2024, 25, 6990 CrossRef CAS.
- S. G. Bobev, K. De Jonghe and M. Maes, Plant Dis., 2018, 102, 829 CrossRef.
- G. Parrella and B. Moury, PLoS One, 2024, 19, 0302692 CrossRef PubMed.
- J. Kwon, K. Mori, T. Maoka, T. Sano and K. S. Nakahara, Virus Res., 2024, 348, 199436 CrossRef CAS PubMed.
- B. R. Shaver, P. Agudelo and S. B. Martin, Crop Sci., 2016, 56, 1330–1336 CrossRef CAS.
- N. L. Anglin, S. K. R. Yellarreddygari, N. C. Gudmestad, V. Sathuvalli, C. R. Brown, M. Feldman, W. S. De Jong, D. S. Douches, R. G. Novy and J. J. Coombs, Am. J. Potato Res., 2023, 101, 1–16 Search PubMed.
- V. Rajesh, V. Rakesh, A. Jeevalatha and A. Ghosh, in Approaches for Potato Crop Improvement and Stress Management, Springer Nature Singapore, Singapore, 2024, pp. 289–323 Search PubMed.
- T. Jiang and T. Zhou, Plants, 2023, 12, 2830 CrossRef CAS.
- A. Kajla, A. Kumar and S. Kumar, Biochem. Cell. Arch., 2024, 24, 1059–1065 Search PubMed.
- R. Muzerengwa, N. Muzhinji, D. Garwe, T. J. Chisango, M. Nyoni and F. Magama, Reverse Transcriptase-PCR-based techniques for the detection and identification of potato virus Y and tobacco mosaic virus in tobacco, Preprint, Res. Sq., 2025, 5932739 Search PubMed.
- N. Vovlas, D. Mifsud, B. B. Landa and P. Castillo, Plant Pathol., 2005, 54, 657–664 CrossRef.
- I. O. Giannakou, D. G. Karpouzas, I. Anastasiades, N. G. Tsiropoulos and A. Georgiadou, Pest Manage. Sci., 2005, 61, 961–972 CrossRef CAS PubMed.
- J. B. Pinheiro, G. O. da Silva, T. J. Pinto, D. F. Cunha, F. S. Rafael, L. A. Santos, M. L. Florentino, C. F. Ragassi, A. D. de Carvalho and A. da S. Pereira, Hortic. Bras., 2024, 42, e2535 CrossRef.
- B. A. Williamson-Benavides and A. Dhingra, Horticulturae, 2021, 7, 33 CrossRef.
- R. K. Walia and M. R. Khan, in Nematode Problems in Crops and Their Management in South Asia, Cambridge Scholars Publishing, Cambridge, UK, 2024, pp. 369–379 Search PubMed.
- P. Spychalla and W. S. De Jong, Crop Sci., 2024, 64, 1167–1182 CrossRef CAS.
- M. F. Narzikulova, O. M. Mamarakhimov, A. E. Mamatov, K. O. Ziyadullayev and F. T. Akhmedova, in BIO Web of Conferences, EDP Sciences, 2024, vol. 141, p. 01017 Search PubMed.
- P. Opdensteinen, R. Charudattan, J. C. Hong, E. N. Rosskopf and N. F. Steinmetz, Plant Biotechnol. J., 2024, 22, 2444–2460 CrossRef CAS.
- M. Stachelek, M. Zalewska, E. Kawecka-Grochocka, T. Sakowski and E. Bagnicka, Ann. Anim. Sci., 2021, 21, 63–87 CrossRef CAS.
- H. Shi, W. Li, Y. Zhou, J. Wang and S. Shen, Heliyon, 2023, 9, e22390 CrossRef PubMed.
- R. W. Mwangi, M. Mustafa, K. Charles, I. W. Wagara and N. Kappel, J. Agric. Food Res., 2023, 14, 100827 Search PubMed.
- T. Islam, Danishuddin, N. T. Tamanna, M. N. Matin, H. R. Barai and M. A. Haque, Plants, 2024, 13, 2737 CrossRef CAS.
- A. Gaurav, P. Bakht, M. Saini, S. Pandey and R. Pathania, Microbiology, 2023, 169, 001333 CrossRef CAS PubMed.
- R. Mirghani, T. Saba, H. Khaliq, J. Mitchell, L. Do, L. Chambi, K. Diaz, T. Kennedy, K. Alkassab, T. Huynh, M. Elmi, J. Martinez, S. Sawan and G. Rijal, AIMS Microbiol., 2022, 8, 239–277 Search PubMed.
- J. Yang, W. Li, T. B. Ng, X. Deng, J. Lin and X. Ye, Front. Microbiol., 2017, 8, 832 CrossRef.
- Q. Wang, S. Wei, A. F. Silva and J. S. Madsen, ISME J., 2023, 17, 846–854 CrossRef CAS.
- S. Malik, K. Muhammad and Y. Waheed, Molecules, 2023, 28, 661 CrossRef CAS.
- P. V. Tawade and K. L. Wasewar, in Environmental Applications of Microbial Nanotechnology, Elsevier, 2023, pp. 43–64 Search PubMed.
- P. Szczyglewska, A. Feliczak-Guzik and I. Nowak, Molecules, 2023, 28, 4932 CrossRef CAS PubMed.
- S. A. Ahire, A. A. Bachhav, T. B. Pawar, B. S. Jagdale, A. V. Patil and P. B. Koli, Results Chem., 2022, 4, 100633 CrossRef CAS.
- S. Bayda, M. Adeel, T. Tuccinardi, M. Cordani and F. Rizzolio, Molecules, 2019, 25, 112 CrossRef.
- V. Gupta, M. Sengupta, J. Prakash and B. C. Tripathy, in Basic and Applied Aspects of Biotechnology, 2016, pp. 1–21 Search PubMed.
- M. Rai and G. Avila-Quezada, Nanotechnology in Plant Health, CRC Press Boca Raton, Florida, USA, 2024 Search PubMed.
- E. B. Hussein, F. A. Rasheed, A. S. Mohammed and K. F. Kayani, RSC Adv., 2025, 15, 41061–41107 RSC.
- J. Saxena, A. Singh and A. Jyoti, Nanobiotechnology: Principles and Applications, Bentham Science Publishers, 2023, p.205 Search PubMed.
- S. Yadav, N. Sawarni, T. Dahiya, J. S. Rana, M. Sharma and B. Batra, in Agricultural and Environmental Nanotechnology. Interdisciplinary Biotechnological Advances, ed. F. Fernandez-Luqueno and J. K. Patra, Springer, Singapore, 2023 Search PubMed.
- A. Y. Ghidan, T. M. A. Antary, A. Y. Ghidan and T. M. A. Antary, in Applications of Nanobiotechnology, IntechOpen, 2019, pp. 1–14 Search PubMed.
- O. I. Ogidi and U. M. Akpan, in One Health Implications of Agrochemicals and Their Sustainable Alternatives, eds M. C. Ogwu and S. Chibueze Izah, Springer Nature, Singapore, 2023, pp. 765–792 Search PubMed.
- F. C. Christopher, P. S. Kumar, F. J. Christopher, G. J. Joshiba and P. Madhesh, J. Clean. Prod., 2020, 269, 122356 CrossRef CAS.
- H. F. Hetta, Y. N. Ramadan, A. I. Al-Harbi, E. A. Ahmed, B. Battah, N. H. A. Ellah, S. Zanetti and M. G. Donadu, Biomedicines, 2023, 11, 413 CrossRef CAS.
- A. Rana, K. Thakur and M. Thakur, in Innovative Methods in Horticultural Crop Improvement: Biosensors and Nanosensors, Springer Nature Switzerland, Cham, 2024, pp. 237–259 Search PubMed.
- S. Machado, R. Pereira and R. M. O. Sousa, Sci. Total Environ., 2023, 896, 166401 CrossRef CAS.
- R. Verma and S. Saroop, in Pesticides in the Environment, Elsevier, 2024, pp. 251–292 Search PubMed.
- N. Vovlas, D. Mifsud, B. B. Landa and P. Castillo, Plant Pathol., 2005, 54, 657–664 CrossRef.
- R. Fatima, P. Katiyar and K. Kushwaha, Front. Nanotechnol., 2025, 7, 1564188 CrossRef.
- D. Thapliyal, G. D. Verros and R. K. Arya, Polymers, 2025, 17, 247 CrossRef CAS PubMed.
- M. Rai and G. Avila-Quezada, Nanotechnology in Plant Health, CRC Press Boca Raton, Florida, USA, 2024 Search PubMed.
- S. M. Banihashemi Jozdani, Z. Hashemian, S. Ebrahim Damavandi, Z. Elyasigorji and M. Vosough, Nano Biomed. Eng., 2024, 16, 357–369 CrossRef.
- A. Rabea, E. Naeem, N. M. Balabel and G. E. Daigham, Bot. Stud., 2023, 64, 20 CrossRef CAS PubMed.
- A. M. Khairy, M. R. Tohamy, M. A. Zayed, S. F. Mahmoud, A. M. El-Tahan, M. T. El-Saadony and P. K. Mesiha, Saudi J. Biol. Sci., 2022, 29, 2199–2209 CrossRef CAS PubMed.
- A. A. AlHarethi, Q. Y. Abdullah, H. J. AlJobory, A. M. Anam, R. A. Arafa and K. Y. Farroh, Discover Nano, 2024, 19, 105 CrossRef CAS PubMed.
- A. Hossain, Y. Abdallah, M. A. Ali, M. M. I. Masum, B. Li, G. Sun, Y. Meng, Y. Wang and Q. An, Biomolecules, 2019, 9, 863 CrossRef CAS PubMed.
- A.-W. Ismail, N. Sidkey, R. Arafa, R. Fathy and A. El-Batal, Br. Biotechnol. J., 2016, 12, 1–11 CrossRef.
- A. M. Ismail, Egypt. J. Phytopathol., 2024, 49, 116–130 CrossRef.
- A. A. Shoeib, N. A. Ashmawy, A. Kamal and S. A. E. F. Zaki, Microb. Cell Factories, 2022, 22, 101 CrossRef.
- S. E. El-Abeid, Y. Ahmed, J.-A. Daròs and M. A. Mohamed, Nanomaterials, 2020, 10, 1001 CrossRef CAS.
- N. Helmy, S. Afifi, B. Othman, K. El-Dougdoug and A. Faiesal, Acta Fytotech. Zootech., 2023, 26 Search PubMed.
- T. Shoala, A. A. Al-Karmalawy, M. O. Germoush, S. M. ALshamrani, M. A. Abdein and N. S. Awad, Horticulturae, 2021, 7, 402 CrossRef.
- M. M. Elsharkawy, in Nanoparticles in Plant Biotic Stress Management, eds M. Khan and J. T. Chen, Springer, Singapore, 2024, pp. 181–206 Search PubMed.
- V. Arumugam, A. H. Bhat, I. K. S. Savarirayan, F. S. Ataya and D. Fouad, J. Nanopart. Res., 2024, 26, 249 CrossRef CAS.
- I. Jahan Lithi, K. I. A. Nakib, A. M. Sarwaruddin Chowdhury and M. S. Hossain, Nanoscale Adv., 2025, 7, 2446–2473 RSC.
- R. Ren, C. Lim, S. Li, Y. Wang, J. Song, T.-W. Lin, B. W. Muir, H.-Y. Hsu and H.-H. Shen, Nanomaterials, 2022, 12, 3855 CrossRef CAS PubMed.
- V. N. Zeyruk, S. V. Vasilieva, G. L. Belov, M. K. Derevyagina, O. A. Bogoslovskaya, I. P. Olkhovskaya, A. G. Yablokov and N. N. Glushchenko, Potato Res., 2021, 65, 273–288 CrossRef.
- G. R. Rudramurthy, M. K. Swamy, U. R. Sinniah and A. Ghasemzadeh, Molecules, 2016, 21, 836 CrossRef.
- L. Wang, C. Hu and L. Shao, Int. J. Nanomed., 2017, 12, 1227–1249 CrossRef CAS PubMed.
- K. Skłodowski, S. J. Chmielewska-Deptuła, E. Piktel, P. Wolak, T. Wollny and R. Bucki, Int. J. Mol. Sci., 2023, 24, 2104 CrossRef.
- K. A. Abd-Elsalam, Nanofungicides: Novel Applications in Plant Disease Control, Elsevier, 2023, p. 400 Search PubMed.
- P. P. Fu, Q. Xia, H.-M. Hwang, P. C. Ray and H. Yu, J. Food Drug Anal., 2014, 22, 64–75 CrossRef CAS.
- Z. Yu, Q. Li, J. Wang, Y. Yu, Y. Wang, Q. Zhou and P. Li, Nanoscale Res. Lett., 2020, 15, 115 CrossRef CAS.
- Y. Sun, Y. Jiang, Y. Li, Q. Wang, G. Zhu, T. Yi, Q. Wang, Y. Wang, O. Parkash Dhankher, Z. Tan, I. Lynch, J. C. White, Y. Rui and P. Zhang, Chem. Sci., 2024, 15, 4709–4722 RSC.
- P. R. More, S. Pandit, A. D. Filippis, G. Franci, I. Mijakovic and M. Galdiero, Microorganisms, 2023, 11, 369 CrossRef CAS.
- J. Ali, D. Alam, R. Noori, S. Faridi and M. Sardar, in Advances in Antifungal Drug Development: Natural Products with Antifungal Potential, Springer Nature Singapore, Singapore, 2024, pp. 411–437 Search PubMed.
- G. Singh, K. Ramadass, P. Sooriyakumar, O. Hettithanthri, M. Vithange, N. Bolan, E. Tavakkoli, L. Van Zwieten and A. Vinu, J. Controlled Release, 2022, 343, 187–206 CrossRef CAS.
- M. Jabran, M. A. Ali, S. Muzammil, A. Zahoor, F. Ali, S. Hussain, G. Muhae-Ud-Din, M. Ijaz and L. Gao, Chem. Biol. Technol. Agric., 2024, 11, 75 CrossRef.
- G. M. Daraban, R.-M. Hlihor and D. Suteu, Toxics, 2023, 11, 983 CrossRef CAS.
- P. Dutta, A. Kumari, M. Mahanta, K. K. Biswas, A. Dudkiewicz, D. Thakuria, A. S. Abdelrhim, S. B. Singh, G. Muthukrishnan, K. G. Sabarinathan, M. K. Mandal and N. Mazumdar, Front. Microbiol., 2022, 13, 935193 CrossRef.
- C. O. Egwu, C. Aloke, K. T. Onwe, C. I. Umoke, J. Nwafor, R. A. Eyo, J. A. Chukwu, G. O. Ufebe, J. Ladokun, D. T. Audu, A. O. Agwu, D. C. Obasi and C. O. Okoro, Molecules, 2024, 29, 2584 CrossRef CAS PubMed.
- Y. Zhou, J. Wang, C. Liang and F. Zheng, in Nanotechnology in Societal Development, Springer Nature Singapore, 2024, pp. 375–394 Search PubMed.
- V. K. Upadhayay, M. K. Chitara, D. Mishra, M. N. Jha, A. Jaiswal, G. Kumari, S. Ghosh, V. K. Patel, M. G. Naitam, A. K. Singh, N. Pareek, G. Taj, D. Maithani, A. Kumar, H. Dasila and A. Sharma, Front. Microbiol., 2023, 14, 1133968 CrossRef PubMed.
- S. Yousefi, W. Weisany, S. E. Hosseini and M. Ghasemlou, Food Hydrocolloids, 2024, 150, 109655 CrossRef CAS.
- Y. Pang, Z. Qin, S. Wang, C. Yi, M. Zhou, H. Lou and X. Qiu, Colloid Polym. Sci., 2020, 298, 1001–1012 CrossRef CAS.
- A. E. Kaziem, Y. Gao, Y. Zhang, X. Qin, Y. Xiao, Y. Zhang, H. You, J. Li and S. He, J. Hazard. Mater., 2018, 359, 213–221 CrossRef CAS PubMed.
- R. Singh and J. W. Lillard, Exp. Mol. Pathol., 2009, 86, 215–223 CrossRef CAS PubMed.
- M. K. I. Rahmani, H. M. A. Ghanimi, S. F. Jilani, M. Aslam, M. Alharbi, R. Alroobaea and S. Sengan, Big Data Res., 2023, 34, 100412 CrossRef.
- M. Ramesh, R. Janani, C. Deepa and L. Rajeshkumar, Biosensors, 2022, 13, 40 CrossRef.
- S. Khaledian, M. Nikkhah, M. Shams-bakhsh and S. Hoseinzadeh, J. Gen. Plant Pathol., 2017, 83, 231–239 CrossRef CAS.
- A. Bharti, U. Jain and N. Chauhan, Plant Nano Biol., 2024, 9, 100079 CrossRef CAS.
- V. Naresh and N. Lee, Sensors, 2021, 21, 1109 CrossRef CAS PubMed.
- R. Mondal, P. Dam, J. Chakraborty, M. L. Paret, A. Katı, S. Altuntas, R. Sarkar, S. Ghorai, D. Gangopadhyay, A. K. Mandal and A. Husen, Heliyon, 2022, 8, e12207 CrossRef CAS.
- M. Tudi, H. D. Ruan, L. Wang, J. Lyu, R. Sadler, D. Connell, C. Chu and D. T. Phung, Int. J. Environ. Res. Publ. Health, 2021, 18, 1112 CrossRef CAS.
- A. Alengebawy, S. T. Abdelkhalek, S. R. Qureshi and M.-Q. Wang, Toxics, 2021, 9, 42 CrossRef CAS PubMed.
- A. Rashid, B. J. Schutte, A. Ulery, M. K. Deyholos, S. Sanogo, E. A. Lehnhoff and L. Beck, Agronomy, 2023, 13, 1521 CrossRef CAS.
- S. Chandna, K. Gogde, R. Kaur, V. Sagar and J. Bhaumik, in The Role of Nanoparticles in Plant Nutrition under Soil Pollution: Nanoscience in Nutrient Use Efficiency, Springer International Publishing, Cham, 2022, pp. 199–221 Search PubMed.
- G. Al-Kateb, M. M. Mijwil, M. Aljanabi, M. Abotaleb, S. R. K. Priya and P. Mishra, Potato Res., 2024, 68, 449–463 Search PubMed.
- H. M. Valenzuela-Amaro, A. Aguayo-Acosta, E. R. Meléndez-Sánchez, O. de la Rosa, P. G. Vázquez-Ortega, M. A. Oyervides-Muñoz, J. E. Sosa-Hernández and R. Parra-Saldívar, Biosensors, 2023, 13, 922 CrossRef CAS PubMed.
- M. M. Hassan, X. Yi, M. Zareef, H. Li and Q. Chen, Trends Food Sci. Technol., 2023, 142, 104230 CrossRef CAS.
- M. A. Khiyami, H. Almoammar, Y. M. Awad, M. A. Alghuthaymi and K. A. Abd-Elsalam, Biotechnol. Biotechnol. Equip., 2014, 28, 775–785 CrossRef.
- P. I. Ishaque, A. Aslam, M. R. Ali, M. A. Z. Khan, A. Shakeel, S. F. Ghani, A. Suleman, A. Asif, K. Amjad and S. Rafique, Scholars Acad. J. Pharm., 2025, 14, 128–162 CrossRef.
- A. Chaturvedi, D. Tripathi and R. Ranjan, Front. Nanotechnol., 2025, 7, 1545792 CrossRef.
- R. Kajla, R. Leija-Cardenas, M. M. Shivalingaiah, M. W. Shabbir and Z. Ou, Nanomaterials, 2025, 15, 1752 CrossRef CAS.
- A. Rathore, W. Hasan, R. C, K. Pujar, R. Singh, N. Panotra and S. N. Satapathy, Uttar Pradesh J. Zool., 2024, 45, 46–71 CrossRef.
- B. Lee, S. Yoon, J. W. Lee, Y. Kim, J. Chang, J. Yun, J. C. Ro, J.-S. Lee and J. H. Lee, ACS Nano, 2020, 14, 17125–17133 CrossRef CAS.
- A. Bharti, U. Jain and N. Chauhan, Plant Nano Biol., 2024, 9, 100079 CrossRef CAS.
- S. Ahmar, T. Mahmood, S. Fiaz, F. Mora-Poblete, M. S. Shafique, M. S. Chattha and K. H. Jung, Front. Plant Sci., 2021, 12, 663849 CrossRef PubMed.
- T. Wang, J. Li, R. Hu, X. Shentu, Z. Ye, X. Yu and K. Sun, Plants, 2025, 14(23), 3649 CrossRef CAS.
- G. A. Khan and M. Mathesh, Plant Commun., 2025, 6, 101422 CrossRef CAS PubMed.
- Z. Sarfraz, Y. Zarlashat, A. Ambreen, M. Mujahid and M. S. Iqbal, Funct. Plant Biol., 2025, 52, 24357 CrossRef PubMed.
- F. Chen, L. Chen, Z. Yan, J. Xu, L. Feng, N. He, M. Guo, J. Zhao, Z. Chen, H. Chen, G. Yao and C. Liu, Front. Plant Sci., 2024, 15, 1478398 CrossRef CAS PubMed.
- J. K. Tiwari, J. A, N. Tuteja and S. M. P. Khurana, Mol. Biol. Rep., 2022, 49, 12109–12119 CrossRef CAS PubMed.
- M. Mujtaba, D. Wang, L. B. Carvalho, J. L. Oliveira, A. do Espirito Santo Pereira, R. Sharif, S. Jogaiah, M. K. Paidi, L. Wang, Q. Ali and L. F. Fraceto, ACS Agric. Sci. Technol., 2021, 1, 417–435 CrossRef CAS.
- H. Akhtar, M. Usman, R. Binyamin, A. Hameed, S. F. Arshad, H. M. U. Aslam, I. A. Khan, M. Abbas, H. E. M. Zaki, G. Ondrasek and M. S. Shahid, Agronomy, 2024, 14, 2175 CrossRef CAS.
- N. Salaria, K. Thakur, B. Singh, S. Kukreja and U. Goutam, in Harnessing Crop Biofortification for Sustainable Agriculture, Springer Nature Singapore, 2024, pp. 179–192 Search PubMed.
- P. Yuan, M. Usman, W. Liu, A. Adhikari, C. Zhang, V. Njiti and Y. Xia, Biotechnol. J., 2024, 19, e202400457 CrossRef CAS PubMed.
- D. Beniwal, S. Chauhan and H. K. Dhillon, Genet. Modif. Org., 2024, 63, 63–77 Search PubMed.
- K. Aaliya, S. Iftikhar, M. N. Sattar, F. Saeed, F. K. Ahmed, S. Faiz, A. A. Rezk and K. A. Abd-Elsalam, in CRISPR-based Gene Editing, Apple Academic Press, 2025, pp. 199–238 Search PubMed.
- S. Das Dangol, A. Barakate, J. Stephens, M. E. Çalıskan and A. Bakhsh, Plant Cell, Tissue Organ Cult., 2019, 139, 403–416 CrossRef.
- M. Sutula, D. Tussipkan, B. Kali and S. Manabayeva, Plants, 2025, 14, 1983 CrossRef CAS PubMed.
- M. Karlsson, N. P. Kieu, M. Lenman, S. Marttila, S. Resjö, M. A. Zahid and E. Andreasson, Hortic. Res., 2024, 11, 130 CrossRef.
- V. Toppo, A. Shaikh, W. N. Sudheer, C. P, J. M. Al-Khayri and P. Nagella, in Custom-Designed Crop Breeding, John Wiley & Sons, Ltd, 2025, pp. 653–688 Search PubMed.
- A. F. Thabet, H. A. Boraei, O. A. Galal, M. F. M. El-Samahy, K. M. Mousa, Y. Z. Zhang, M. Tuda, E. A. Helmy, J. Wen and T. Nozaki, Sci. Rep., 2021, 11, 14484 CrossRef CAS.
- N. Kumari, A. Kumar, S. Sharma, P. Thakur, S. Chadha and A. Dhiman, J. Plant Biochem. Biotechnol., 2024, 33, 108–128 CrossRef.
- M. A. Arshad, B. A. Akbar, N. Shehzadi, N. Iqbal, M. Z. Mushtaq, S. Rouf and A. Jawad, J. Plant Sci., 2025, 13, 38–58 Search PubMed.
- O. A. Fabiyi, in Nanophytopathology, CRC Press, 2023, pp. 23–38 Search PubMed.
- B. Poudel, CRISPR/Cas9 Based Gene Editing in Wheat and Potato to Enhance Multiple Disease Resistance, PhD thesis, McGill University, 2024.
- M. K. Ray, A. K. Mishra, Y. K. Mohanta, S. Mahanta, I. Chakrabartty, N. A. Kungwani, S. K. Avula, J. Panda and R. N. Pudake, Agriculture, 2023, 13, 1856 CrossRef CAS.
- M. M. Nizamani, A. C. Hughes, H.-L. Zhang and Y. Wang, Sci. Total Environ., 2024, 928, 172473 CrossRef CAS.
- N. S. M. Jalaluddin, M. Asem, J. A. Harikrishna and A. A. H. A. Fuaad, Molecules, 2023, 28, 2700 CrossRef PubMed.
- A. Chen, L. Halilovic, J.-H. Shay, A. Koch, N. Mitter and H. Jin, Curr. Opin. Plant Biol., 2023, 76, 102441 CrossRef CAS.
- U. Vasquez-Gutierrez, G. A. Frias-Treviño, L. A. Aguirre-Uribe, S. N. Ramírez-Barrón, J. Mendez-Lozano, A. Hernández-Juárez and H. García-Ruíz, Plants, 2025, 14, 3118 CrossRef CAS PubMed.
- S. Ghosh, S. Patra and S. Ray, ACS Omega, 2023, 8, 22345–22351 CrossRef CAS.
- H. Le, C. Karakasyan, T. Jouenne, D. L. Cerf and E. Dé, Appl. Sci., 2021, 11, 10695 CrossRef CAS.
- P. A. Nazarov, D. N. Baleev, M. I. Ivanova, L. M. Sokolova and M. V. Karakozova, Acta Nat., 2020, 12, 46–59 CrossRef CAS.
- Y. Gai and H. Wang, Agronomy, 2024, 14, 1615 CrossRef.
- J. González-Jiménez, B. Andersson, L. Wiik and J. Zhan, Field Crops Res., 2023, 299, 108977 CrossRef.
- R. Ridhi, G. S. S. Saini and S. K. Tripathi, Mater. Sci. Eng. B, 2024, 304, 117383 CrossRef CAS.
- A. Wahab, M. Muhammad, S. Ullah, G. Abdi, G. M. Shah, W. Zaman and A. Ayaz, Sci. Total Environ., 2024, 926, 171862 CrossRef CAS PubMed.
- D. L. Wasule, P. R. Shingote and S. Saxena, Discover Nano, 2024, 19, 118 CrossRef CAS PubMed.
- A. Yadav and K. Yadav, J. Environ. Nanotechnol., 2024, 13, 48–62 CrossRef CAS.
- M. Gul, R. S. Khan, Z. U. Islam, S. Khan, A. Shumaila, S. Umar, S. Khan, Brekhna, M. Zahoor and A. Ditta, Mol. Biol. Rep., 2024, 51, 92 CrossRef CAS.
- L. M. Carrillo-Lopez, C. Villanueva-Verduzco, E. Villanueva-Sánchez, M. L. Fajardo-Franco, M. Aguilar-Tlatelpa, R. I. Ventura-Aguilar and R. M. Soto-Hernández, Plants, 2024, 13, 2634 CrossRef CAS PubMed.
- D. V. Francis, A. Asif and Z. F. R. Ahmed, in Nanoparticles in Plant Biotic Stress Management, ed. M. Khan and J. T. Chen, Springer, Singapore, 2024. pp. 451–491 Search PubMed.
- R. Singh, P. Choudhary, S. Kumar and H. K. Daima, Environ. Sci.: Nano, 2024, 11, 2324–2351 RSC.
- J. M. Rajwade, R. G. Chikte and K. M. Paknikar, Appl. Microbiol. Biotechnol., 2020, 104, 1437–1461 CrossRef CAS.
- M. Jabran, M. S. Shafique, A. Abbas, W. Han, M. A. Ali and L. Gao, BMC Plant Biol., 2025, 25, 1517 CrossRef CAS PubMed.
- S. Sujata, K. R. Aarti and A. Kumar, Biochem. Cell. Arch., 2024, 24, 1029 CrossRef.
- X. Lv, H. Sha, Z. Ye, Y. Wang and B. Mao, Environ. Sci.: Nano, 2023, 10(12), 3232–3252 RSC.
- A. Y. Jassim, A. Karnwal, G. Pant, A. Gaur, M. Selvaraj, J. Dutta, R. S. K. Sachan, M. Kumar and N. Nesterova, J. Plant Interact., 2025, 20, 2586338 CrossRef.
- A. R. Sallam, A. A. Mahdi and K. Y. Farroh, Plant Cell Biotechnol. Mol. Biol., 2022, 23, 1–16 Search PubMed.
- I. Ullah, M. D. Toor, A. Basit, H. I. Mohamed, M. Gamal, N. A. Tanveer and S. T. Shah, Water, Air, Soil Pollut., 2023, 234, 666 CrossRef CAS.
- W. A. Al-Selwey, A. A. Alsadon, M. M. Alenazi, M. Tarroum, A. A. Ibrahim, A. Ahmad, M. Osman and M. F. Seleiman, Horticulturae, 2023, 9, 883 CrossRef.
- A. Maholiya, P. Ranjan, R. Khan, S. Murali, R. C. Nainwal, P. S. Chauhan, N. Sathish, J. P. Chaurasia and A. K. Srivastava, Environ. Sci.: Nano, 2023, 10, 959–995 RSC.
- C. Mony, P. Kaur, J. E. Rookes, D. L. Callahan, S. V. Eswaran, W. Yang and P. K. Manna, Environ. Sci.: Nano, 2022, 9, 3659–3683 RSC.
Footnote |
| † These authors have contributed equally to this work and share first authorship. |
| This journal is © The Royal Society of Chemistry 2026 |