Open Access Article
Open Access ArticleEnhanced hydrophilic properties and performance evaluation of PVDF-TiO2-nZVI-SiO2 nanocomposite membranes for the remediation of heavy metal contaminated wastewater
Murtala Namakka *ab,
Md Rezaur Rahman
*ab,
Md Rezaur Rahman a,
Khairul Anwar Bin Mohamed Saida and
Bavya Devi Karuppasamyc
a,
Khairul Anwar Bin Mohamed Saida and
Bavya Devi Karuppasamyc
aDepartment of Chemical Engineering and Energy Sustainability, Universiti Malaysia Sarawak, Malaysia. E-mail: abuhuzaifah33@yahoo.com; rmrezaur@unimas.my
bDepartment of Chemical Engineering, Ahmadu Bello University Zaria-Nigeria, Nigeria
cKPR Institute of Engineering and Technology, India
First published on 21st January 2026
Abstract
Exposure to heavy metal contaminated water causes irreversible harm to humans and the environment. Despite the recent progress in nanofiltration for heavy metal remediation, advanced innovations are necessary to improve its effectiveness and membrane hydraulic performance. In this article, a hydrophilic multifunctional PVDF-TiO2-nZVI-SiO2 nanocomposite membrane material was developed using a phase inversion technique for the remediation of lead and hexavalent chromium contaminants. The synthesized TiO2-nZVI-SiO2 nanocomposite and PVDF-TiO2-nZVI-SiO2 nanocomposite membranes were characterized via various characterization techniques including FESEM, FTIR, EDX, XRD, XPS, TGA, water contact angle, and solvent content analysis. The novel TiO2-nZVI coated SiO2 nanocomposite membranes were fabricated by varying TiO2-nZVI-SiO2 nanocomposite loadings from 0.1 g (M1) to 0.3 g (M5) and the optimal modification was determined via detailed performance evaluation under varying concentrations viz. 20 ppm, 30 ppm, 40 ppm and 50 ppm. The removal efficiencies of the PVDF- TiO2-nZVI-SiO2 membranes were consistently stable despite increase in nanocomposite loadings with optimum membrane (M5) achieving 99.8% removal efficiency during the 50 minutes filtration operation. The TiO2-nZVI-SiO2 nanocomposite combines synergistic redox reaction and hydrophilic properties via TiO2-nZVI and stabilization against particle aggregation from the surface SiO2. Long-term stability study, the influence of contaminant type and loading on PVDF- TiO2-nZVI-SiO2 membrane and hydraulic performance corroborated sustained hydraulic properties and stable performance exceeding 90% across all contaminant loadings.
1 Introduction
Lack of safe drinking water due to contamination from harmful inorganic and organic chemicals has been declared a global concern by the United Nations since 2023, threatening 26% of the global population. Amid the current surge of global population and technological advancements, the demand for sustainable water and energy security is never more crucial than now. However, the global demand of clean water, free of harmful contaminants, for human consumption, food and energy security will likely be overstretched by the recent advancements in AI technologies and Data Centres which utilise large-scale computational models that require clean water for thermal management.1 The water used for cooling data centres to maintain optimum operations of these technologies, is often sourced from municipal water supplies adding to a compounding environmental concern.1,2 These make the development of effective materials for the treatment of industrial effluents and other wastewater sources highly critical. Efficient wastewater purification technologies capable of removing emerging contaminants such as dyes,3,4 emulsions5 and heavy metals6 are sustainable alternatives for the increasing water demands. Heavy metal pollution is a contemporary environmental challenge associated with the discharge of heavy metals into water systems from industrialisation and rapid urban developments. Recent studies have investigated the harmful effects of heavy metal contaminants such as arsenic,7,8 cadmium,9,10 chromium11,12 and lead.13,14 Despite the remediation intervention, heavy metal contaminations remain a significant concern. One of the most complex heavy metal contaminations is the hexavalent chromium, Cr(VI) contamination which often occur from soil or volcanic dust sources. Cr(VI) is highly toxic, carcinogenic, corrosion resistance with relatively high melting points compared to Cr(III) oxidation state which forms insoluble hydroxides when exposed to water.11,15 Lead Pb2+ contamination has been reported to inadequately affect ecosystem due to its enormous toxicity,16 with current available remediation techniques lacking sustainability.16,17 Therefore, new remediation materials and strategies for the removal of lead and hexavalent chromium(VI) are essential in mitigating potential health hazards and future environmental contaminations. Conventional methods such as chemical precipitation,18,19 ion exchange,20,21 electrochemical22,23 and adsorption methods are commonly employed with relative success.24,25 Although limitations including selectivity in ion exchange, efficacy of resin generation, production of passive non-conductive layer in electrochemical method are a major drawback. Consequently, advanced oxidation processes coupled with the utilization of biological wastewater treatment strategies have been explored to enhance target ion selectivity while maintaining stable adsorption capacity.22,23 Nevertheless, long operation time,26 excessive sludge formation,27 remains a challenge in adopting these strategies.28,29 Hence, adsorption method remains the most widely employed approach for both lead and Cr(VI) remediation compared to other remediation strategies.30,31 The efficiency of adsorption method significantly lies on the nature and catalytic activity of the adsorbent with iron and silica-based adsorbent recording the higher Cr(VI) adsorption capacity.32 However, the approach requires post treatment cost coupled with unstable adsorption efficiency due to passivation of iron active sites after the chemical reduction of Cr(VI) to Cr(III) over time.33–35To address this challenge, it is crucial to develop effective and sustainable remediation approach capable of preventing formation of passive layers and nanomaterial aggregation while efficiently combating the diverse range of toxic heavy metals from wastewater effluents.16 Zero valent iron (ZVI) is characterized by its exceptional reactivity, injective ability and large surface area electron donor that reduces emerging contaminants.36 However, individual nZVI can easily passivate or agglomerated thereby limiting overall performance. Researchers have reported enhanced stability of nZVI by incorporating it a support materials forming stable nanocomposite with less potential passivation properties shielding active nZVI surface particularly TiO2 which is also a studied adsorbent capable of mineralizing heavy metal contaminants37 with potential recombination. The coating of TiO2-nZVI was reported with efficient dispersibility, reducing capacity, adsorption properties and a significant photocatalytic.38
In this study, an efficient, advanced adsorbent incorporated PVDF membrane was developed for removal of emerging Pb2+ and Cr(VI) contaminants. The synergistic combination of nZVI,39 silica SiO2, and TiO2 have shown successful application in environmental remediation with recent application reporting 100% MB dye removal.40 Previous studies utilize direct chemical reaction in the synthesis of nZVI-TiO2-SiO2. In the current study a layer-by-layer patter was adopted to systematically developed three layers of the nanocomposite by coating the TiO2-nZVI with SiO2 using CTAB to enable hierarchy in the nanocomposite structural interface. Subsequently, PVDF-TiO2-nZVI-SiO2 nanocomposite membrane was fabricated via phase inversion by varying the nZVI-TiO2-SiO2 nanocomposite concentration from 0.1 g to 0.30 g for M1 to M5. The nZVI-TiO2-SiO2 and nanocomposite membrane were characterized via various techniques. Performance of the nanocomposite membrane was assessed via various techniques including solvent content, water contact angle, water flux and the remediation of Pb2+ and Cr(VI) contaminants.
2 Materials and methods
2.1 Materials
For the fabrication of TiO2-nZVI and SiO2 coated nanocomposite, All procured Chemicals including Sodium borohyas procured, FeSO4·7H2O, ethanol, CH3CH2OH, methanol CH3OH, sodium hydroxide, NaOH, hydrochloric acid HCl, titanium oxide TiO2 (CAS No: 13![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 463-67-7), Tetraethyl orthosilicate TEOS Si(OC2H5)4, NH3, 1-propanol C3H8O 99.5%, and Cetyltrimethylammonium Bromide CTAB, were used as procured from Sigma-Aldrich Sdn. Bhd. (Malaysia). Synthesized nZVI, and TiO2-nZVI were prepared. Chemicals for nanocomposite membrane fabrication includes Poly vinylidene fluoride, (PVDF) (CAS No: 24
463-67-7), Tetraethyl orthosilicate TEOS Si(OC2H5)4, NH3, 1-propanol C3H8O 99.5%, and Cetyltrimethylammonium Bromide CTAB, were used as procured from Sigma-Aldrich Sdn. Bhd. (Malaysia). Synthesized nZVI, and TiO2-nZVI were prepared. Chemicals for nanocomposite membrane fabrication includes Poly vinylidene fluoride, (PVDF) (CAS No: 24![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 937-79-9, Mm 534
937-79-9, Mm 534![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 g mol−1) as the primary polymer matrix for its intrinsic chemical resistivity and thermal stability, N-methyl-2-pyrrolidone (NMP) as a solvent to ensure a homogeneous dispersion of TiO2-nZVI-SiO2 nanoparticles in the casting solution. Throughout the experimental procedures, all aqueous reagent solutions were prepared with ultrapure water while industrial grade nitrogen, N2 utilized in this work, was provided by Universiti of Malysia Sarawak.
000 g mol−1) as the primary polymer matrix for its intrinsic chemical resistivity and thermal stability, N-methyl-2-pyrrolidone (NMP) as a solvent to ensure a homogeneous dispersion of TiO2-nZVI-SiO2 nanoparticles in the casting solution. Throughout the experimental procedures, all aqueous reagent solutions were prepared with ultrapure water while industrial grade nitrogen, N2 utilized in this work, was provided by Universiti of Malysia Sarawak.
2.2 Synthesis of TiO2-nZVI nanoparticles
The TiO2-nZVI nanocomposite was synthesized following the procedure described in ref. 41 and 37 with little modifications. Briefly, a precursor solution was prepared by dissolving 5.56 g of ferrous sulfate heptahydrate (FeSO4·7H2O) and a specific amount of pre-synthesized TiO2 nanoparticles in 200 ml of ultrapure water within a three-necked flask. The mixture was continuously purged with N2 gas under constant stirring for 30 minutes to achieve a homogeneous, oxygen-free TiO2–FeSO4 solution. Sodium hydroxide (NaOH) and hydrochloric acid (HCl) (0.01 mol L−1) were used to adjust the solution pH. Concurrently, sodium borohydride (NaBH4) reducing agent solution was formulated by dissolving 1.60 g of NaBH4 in 50 ml deionized water. The reduction of ferrous ions, Fe2+ to zero valent iron Fe0 on the TiO2 particles was subsequently initiated by the dropwise addition of the NaBH4 solution into the TiO2–FeSO4 solution (see eqn (1)–(3)).| FeSO4·7H2O + 2NaBH4 → Fe0 + NaSO4 + 7H2O + 2B(OH)3 | (1) |
| Fe2+ + 2BH4 − + 6H2O → Fe0 + 2B(OH)3 + 7H2↑ | (2) |
| Fe0 → Fe2+ + 2e− | (3) |
The reaction was allowed to proceed for 25 minutes; the cessation of gas evolution and complete formation of black colored particles indicates quantitative reduction of Fe2+ to Fe0. The solution was allowed to settle for few minutes, washed several times using 95% ethanol to remove excess reactants and uncoated TiO2 particles before centrifuging for 5 minutes at 20![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 rpm at room temperature. The binary composite was dried at 90 °C for 4 hours in a tube furnace under nitrogen condition. Dried TiO2-nZVI nanoparticles were stored for further characterizations.
000 rpm at room temperature. The binary composite was dried at 90 °C for 4 hours in a tube furnace under nitrogen condition. Dried TiO2-nZVI nanoparticles were stored for further characterizations.
2.3 Synthesis of TiO2-nZVI-SiO2 nanoparticles
The TiO2-nZVI-SiO2 nanocomposites were synthesized by modifying the procedure described in ref. 42. Briefly, specified amount of TiO2-nZVI- nanoparticles were prepared and ultrasonically dispersed in 75% ethanol solutions. The mixture was continuously sonicated before introducing 10 ml Ammonia and n-propanol with 2.1 g of cationic surfactant Cetyltrimethylammonium Bromide, CTAB solutions. Subsequently tetraethyl orthosilicate was added and the reaction was allowed to proceed at 85 °C for 6 hours. The silica coated TiO2-nZVI nanoparticles were collected, washed several times with 75% ethanol and centrifuged for 5 minutes at 20![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 rpm under room temperature conditions. Subsequently, the particles were dried at 95 °C in a tube furnace and stored for further characterization and membrane fabrications.
000 rpm under room temperature conditions. Subsequently, the particles were dried at 95 °C in a tube furnace and stored for further characterization and membrane fabrications.
2.4 Fabrication of TiO2-nZVI-SiO2 nanocomposite membranes
Polyvinylidene fluoride (PVDF) and NMP were used as the base polymer and PVDF solvent for the preparation of dope solutions, while synthesized TiO2-nZVI-SiO2 nanoparticles were incorporated into the membrane dope solution to enhance hydrophilicity, self-cleaning and overall performance of the nanocomposite membrane. The dope solutions were prepared following the procedure described in ref. 43 and 44 with the following modifications. Briefly, PVDF pellets (14% w v−1) were added to a predetermined volume of NMP solvent in a 250 ml conical flask. TiO2-nZVI-SiO2 nanoparticles were subsequently introduced at varying concentrations viz. 0.1 g, 0.15 g, 0.20 g, 0.25 g and 0.30 g. The solutions were covered using foil paper and subjected to 350 rpm magnetic stirring at 85 °C for 24 hours to ensure effective dissolution and homogenous dispersion of the TiO2-nZVI-SiO2 nanoparticles in the dope solutions. To eliminate possibly entrapped air bubbles introduced during stirring, which could potentially introduce defects in the cast membrane, the homogenously dispersed dope solutions were transferred to an oven and held at 50 °C for 1 hour, this procedure reduces the solution viscosity and allows microbubbles to coalesce. Subsequently, the bubble-free dope solution (approximately 10 ml) was cast onto a clean, dry glass substrate (21 cm × 30 cm) using a doctor blade film applicator and immediately immersed in a coagulation bath containing deionized water maintained at room temperature (approximately 25 °C). This step helps initiate the non-solvent induced phase separation process, resulting in the solidification of the polymer solution and the formation of a hydrophilic TiO2-nZVI-SiO2 nanocomposite membrane. Following phase inversion, the nanocomposite membranes were transferred to another coagulation bath containing fresh deionized water for 12 hours (overnight). The coagulation bath water was replaced with fresh distilled water to ensure the complete extraction of residual NMP solvent and any soluble components from the TiO2-nZVI-SiO2 nanocomposite membrane matrix. Finally, the nanocomposite membranes were stored in a fridge for further analysis. Table 1 shows the TiO2-nZVI-SiO2 nanocomposite membrane designated code and compositions.| Code | PVDF (wt%) | Concentration of TiO2-nZVI-SiO2 (grams) | NMP solvent (v%) |
|---|---|---|---|
| Mo | 14 | 0 | 86 |
| M1 | 14 | 0.10 | 86 |
| M2 | 14 | 0.15 | 86 |
| M3 | 14 | 0.20 | 86 |
| M4 | 14 | 0.25 | 86 |
| M5 | 14 | 0.30 | 86 |
2.5 Characterizations
Synthesized TiO2-nZVI, SiO2 coated TiO2-nZVI and fabricated nanocomposite membranes were characterized via XRD, FTIR, TGA, FESEM, EDX, and XPS analytical techniques to corroborate the physicochemical properties and chemical bonding of the synthesised nanocomposite material.Fourier-transform infrared (FTIR) spectroscopy was performed using a Shimadzu IRAffinity-1 spectrometer to characterize the molecular bond structures and functional groups of the nanoparticles and the fabricated membranes. Both analyses were conducted in attenuated total reflectance (ATR) mode across a wavenumber range of 4000 to 400 cm−1, with a spectral resolution of 4 cm−1 and an accumulation of 32 scans per spectrum. For measurement, a section of each sample was placed on the ATR crystal and secured with a swivel press to ensure optimal surface contact.
The surface morphology and elemental composition of the synthesized nanocomposites were characterized by using field emission scanning electron microscopy coupled with energy-dispersive X-ray spectroscopy (FESEM-EDS), which are well-established techniques for microstructural and elemental analysis.45,46 The FESEM images (×15![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 and ×50
000 and ×50![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 magnification) were acquired with a Hitachi S-4700 instrument, operating at an accelerating voltage of 20 kV, which illustrates the morphological structure of the TiO2-nZVI, silica coated TiO2-nZVI nanocomposites and modified PVDF- TiO2-nZVI-SiO2. Subsequently, the thermal stability of the TiO2-nZVI and the influence of SiO2 coating on the physicochemical properties of the synthesized TiO2-nZVI-SiO2 nanocomposite material was evaluated via TGA analysis under the following conditions: on set temperature of 323.69 °C, Inflect. Point temperature 372.92 °C, end set temperature 388.95 °C and midpoint 351.52 °C under constant N2 supply. The XPS analysis was conducted to investigate the chemical bonding and elemental interactions within the nanocomposite material. The analysis followed the procedure described in ref. 47 and 48 and the deconvolutions and peaks identification were guided by the standard procedure described in ThermoFisher scientific. Briefly. The analysis was carried out in Alpha-K XPS spectrometer using monochromatic aluminium radiation at 0.3 eV FWHM, 1.5% pass energy resolution and 11.75 eV to 23.5 eV measurement pass energy resolution as described in operation manual. The crystallinity and phase composition of the SiO2 coated TiO2-nZVI nanoparticles and the fabricated nanocomposite membranes were analysed using X-ray diffraction (XRD). Measurements were performed at ambient temperature on a Rigaku SmartLab Powder X-ray diffractometer. Data were collected over a 2θ angular range from 0° to 80°. The crystalline index (CrI), serving as a measure of the degree of crystallinity, was determined using the empirical equations described in ref. 49. The crystallite size was calculated using origin software (see S1CA 1–10), by employing Debye–Scherrer equation and the standard procedure of the ASTM F3419-22 (2022) standard.50
000 magnification) were acquired with a Hitachi S-4700 instrument, operating at an accelerating voltage of 20 kV, which illustrates the morphological structure of the TiO2-nZVI, silica coated TiO2-nZVI nanocomposites and modified PVDF- TiO2-nZVI-SiO2. Subsequently, the thermal stability of the TiO2-nZVI and the influence of SiO2 coating on the physicochemical properties of the synthesized TiO2-nZVI-SiO2 nanocomposite material was evaluated via TGA analysis under the following conditions: on set temperature of 323.69 °C, Inflect. Point temperature 372.92 °C, end set temperature 388.95 °C and midpoint 351.52 °C under constant N2 supply. The XPS analysis was conducted to investigate the chemical bonding and elemental interactions within the nanocomposite material. The analysis followed the procedure described in ref. 47 and 48 and the deconvolutions and peaks identification were guided by the standard procedure described in ThermoFisher scientific. Briefly. The analysis was carried out in Alpha-K XPS spectrometer using monochromatic aluminium radiation at 0.3 eV FWHM, 1.5% pass energy resolution and 11.75 eV to 23.5 eV measurement pass energy resolution as described in operation manual. The crystallinity and phase composition of the SiO2 coated TiO2-nZVI nanoparticles and the fabricated nanocomposite membranes were analysed using X-ray diffraction (XRD). Measurements were performed at ambient temperature on a Rigaku SmartLab Powder X-ray diffractometer. Data were collected over a 2θ angular range from 0° to 80°. The crystalline index (CrI), serving as a measure of the degree of crystallinity, was determined using the empirical equations described in ref. 49. The crystallite size was calculated using origin software (see S1CA 1–10), by employing Debye–Scherrer equation and the standard procedure of the ASTM F3419-22 (2022) standard.50
 | (4) |
| At = Ac + Am | (5) |
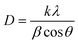 | (6) |
2.6 Nanocomposite membrane performance analysis
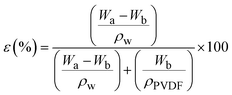 | (7) |
 | (8) |
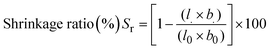 | (9) |
 | (10) |
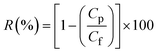 | (11) |
3 Results and discussion
3.1 FTIR of nZVI, TiO2-nZVI, and TiO2-nZVI-SiO2 nanocomposite
Fourier transform infrared spectroscopy with attenuated total reflectance (FTIR-ATR) was employed to characterize the functional groups present in the synthesized nZVI, nZVI-TiO2, and TiO2-nZVI-SiO2 nanoparticles (Fig. 1). The spectra for all three nanoparticles exhibited characteristic absorption peaks correlated with the vibrational modes of their constituent chemical bonds. The FTIR spectra of the nZVI (a) and nZVI coated TiO2 (b) nanoparticles were nearly identical, indicating that the composite is predominantly composed of the coated nZVI surface layer. In the FTIR spectrum of the TiO2-nZVI composite, a broad absorption band observed at 3400.23 cm−1 is attributed to O–H stretching vibrations, which is consistent with the typical range for this functional group.57 The peaks at 1590.78 cm−1 and 2104 cm−1 collectively suggest the presence of O–H bonds associated with the formation of α-FeOOH.58,59 Furthermore, a shift in the Ti–O–Ti stretching vibration absorption band from 720 cm−1 to a lower wavenumber of 516.09 cm−1 corroborate the successful compositional integration of nZVI in TiO2 surface layer.In FTIR analysis of TiO2-nZVI-SiO2 shown in (c), both peak at 1030, –OH tensile vibration peak (2104 cm−1), and –OH stretching (3400 cm−1), peaks became sharp with a clear C–H vibrations around 2800 cm−1, this can be associated with the surface coating by SiO2 nanoparticles on the TiO2-ZVI, thereby increasing the overall –OH functional group on the TiO2-nZVI-SiO2 nanocomposite material.
The effective encapsulation of TiO2-nZVI within a silica matrix is essential for maintaining stability and reactivity, coupled with reduced passivation potential of nZVI surfaces, particularly in aqueous environments. Similar findings were reported in ref. 60. Moreover, the silica coating has been shown to mitigate the oxidation of nZVI, a factor critical for preserving its reductive capabilities. The vibrational nodes for the metal–oxygen bonds (Ti–O–Ti and Fe–O) were identified in the 484–500 cm−1 range, which are critical for the structural support of nZVI by TiO2 40, amorphous SiO2 peak with Si–O bending vibrations was noted at 680 cm−1.61,62 At little higher wavenumbers, the absorption bands between 1000 and 1006 cm−1 were assigned to Si–O–Ti or Si–O–Si asymmetric stretching vibrations.63–65 The presence of surface hydroxyl groups, vital for the material's adsorptive and catalytic properties, was confirmed by multiple vibration intensities in both nanoparticle samples. These included Fe–O vibrations (500–742 cm−1), O–H bending at 1630 cm−1, more profound for TiO2-nZVI, and coated SiO2 sample, and O–H stretching nodes related to Fe–OH, Si–OH and Ti–OH species at 600 cm−1, and 3400 cm−1 respectively. A comparative spectral analysis of the nanoparticles showed consistent peaks profiles, suggesting a stable composite structure.
3.2 FESEM/EDS analysis of TiO2-nZVI-SiO2 nanocomposite
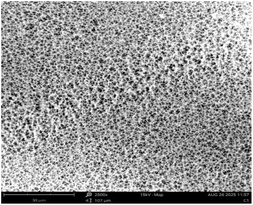
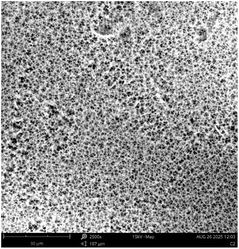
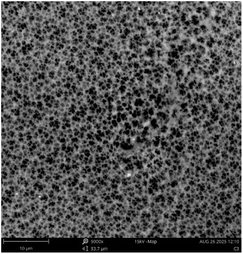
Fig. 2 depicts the FESEM images at ×15![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 and ×50
000 and ×50![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 magnification of the morphological structure of TiO2-nZVI and the silica coated TiO2-nZVI nanocomposites. As shown in Fig. 2(a). The morphological structure of the TiO2-nZVI nanoparticles, examined at a magnification of ×50
000 magnification of the morphological structure of TiO2-nZVI and the silica coated TiO2-nZVI nanocomposites. As shown in Fig. 2(a). The morphological structure of the TiO2-nZVI nanoparticles, examined at a magnification of ×50![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 magnification reveals a regular spherical shape with a moderately uniform distribution of particles. Similar characteristic morphology was reported by Huang et al. (2018). In Fig. 2(b) under magnifications of ×15
000 magnification reveals a regular spherical shape with a moderately uniform distribution of particles. Similar characteristic morphology was reported by Huang et al. (2018). In Fig. 2(b) under magnifications of ×15![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000, the silica coated TiO2-nZVI nanoparticle material, despite the homogeneity observed in TiO2-nZVI (Fig. 2(a)), exhibits few irregularly shaped particles with a non-uniform distribution, this characteristic morphological property was also observed in previous research findings41 indicating the presence of mesopores a morphological trait commonly associated with silica coated nZVI-based nanocomposites.66,67
000, the silica coated TiO2-nZVI nanoparticle material, despite the homogeneity observed in TiO2-nZVI (Fig. 2(a)), exhibits few irregularly shaped particles with a non-uniform distribution, this characteristic morphological property was also observed in previous research findings41 indicating the presence of mesopores a morphological trait commonly associated with silica coated nZVI-based nanocomposites.66,67
 | ||
Fig. 2 FESEM analysis of TiO2-nZVI ×50![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 magnification (a), silica coated TiO2-nZVI nanoparticles ×15 000 magnification (a), silica coated TiO2-nZVI nanoparticles ×15![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 magnification (b) and (c) selected area of silica coated TiO2-nZVI ×50 000 magnification (b) and (c) selected area of silica coated TiO2-nZVI ×50![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 magnification. 000 magnification. | ||
The ×50![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 magnified view in image (c) elucidates a detailed particle morphology. The relatively uniform surface morphology observed suggests an efficient silica coating, as supported by previous studies that highlight the benefits of silica for enhancing the dispersibility and stability of metal nanoparticles.60,63 Fig. 3 shows the elemental compositions of the synthesized nanocomposite material, from this figure, energy-dispersive X-ray spectroscopy (EDS) analysis corroborated the elemental composition and purity of the synthesized nZVI-TiO2-SiO2 nanocomposites. The EDS spectra reveal a progressive mass ratio of silicon, zero-valent iron (nZVI), and titanium atoms with relatively higher mass ratio of oxygen. Higher mass percentage of titanium with nearly equal mass percentage of silicon and zero valent iron (15.78% and 15.48% mass percent respectively) confirm the existence of the Fe0 and Si as a coating layer of approximately equal mass percents in the nanocomposite material. Ti mass percents 18.52% exist relatively higher after oxygen (50.22%) in the nanocomposite as the titania (TiO2) nanoparticles are the “core” material in the nanocomposite. The distribution and relative abundance of the incorporated nanoparticles are governed by interfacial interactions, redox reactions, and particle nucleation and growth dynamics. These fundamental mechanisms are themselves highly dependent on the reaction pH, as established in prior studies.40,68–70
000 magnified view in image (c) elucidates a detailed particle morphology. The relatively uniform surface morphology observed suggests an efficient silica coating, as supported by previous studies that highlight the benefits of silica for enhancing the dispersibility and stability of metal nanoparticles.60,63 Fig. 3 shows the elemental compositions of the synthesized nanocomposite material, from this figure, energy-dispersive X-ray spectroscopy (EDS) analysis corroborated the elemental composition and purity of the synthesized nZVI-TiO2-SiO2 nanocomposites. The EDS spectra reveal a progressive mass ratio of silicon, zero-valent iron (nZVI), and titanium atoms with relatively higher mass ratio of oxygen. Higher mass percentage of titanium with nearly equal mass percentage of silicon and zero valent iron (15.78% and 15.48% mass percent respectively) confirm the existence of the Fe0 and Si as a coating layer of approximately equal mass percents in the nanocomposite material. Ti mass percents 18.52% exist relatively higher after oxygen (50.22%) in the nanocomposite as the titania (TiO2) nanoparticles are the “core” material in the nanocomposite. The distribution and relative abundance of the incorporated nanoparticles are governed by interfacial interactions, redox reactions, and particle nucleation and growth dynamics. These fundamental mechanisms are themselves highly dependent on the reaction pH, as established in prior studies.40,68–70
The EDX analysis of the TiO2-nZVI-SiO2 nanoparticles confirms the results of the FTIR analysis and a successful synthesis of nZVI-TiO2-SiO2 nanocomposite material with the desired elemental composition. The results highlight the effective two-layer coating of nZVI and SiO2. The uniform distribution of nZVI and silicon highlights the effectiveness of the coating method, which is crucial for maximizing the material's reactivity and stability in environmental applications.
3.3 X-ray diffraction (XRD) analysis of nZVI, TiO2-nZVI and TiO2-nZVI-SiO2 nanocomposite
Fig. 4 shows the crystalline structure and phase composition of the synthesized nZVI, TiO2-nZVI, TiO2-nZVI-SiO2 nanoparticles analyzed using Rigaku SmartLab Powder X-ray Diffractometer. The resulting patterns Fig. 4(a and b) exhibits characteristic peaks of a binary crystalline material coated by an amorphous Silica matrix. The XRD peaks in Fig. 4(a) (see Table 2) corroborates the existence of dominant anatase titanium dioxide, TiO2 and a metallic zero valent iron, Fe0.71–74 The most prominent peak at 25.3° is indexed to the 101 plane75 of the anatase phase (JCPDS No. 21-1272). This confirmed the presence of the TiO2 photocatalytic component which forms the core of the synthesized TiO2-nZVI-SiO2 nanocomposite and reinforces its overall stability and chemical activity.76–78 The anatase 37.8° indexed 004 plane, 48.0° indexed 200 plane and a distinct peak corresponding to the 105 plane at 53.9° confirms the high purity of the anatase phase of the TiO2 component. In addition, there is nearly complete absence of the main rutile (110) peak at 27.4° (see Fig. 4(a)). | ||
| Fig. 4 X-ray diffraction pattern of nZVI, TiO2-nZVI and TiO2-nZVI-SiO2 nanoparticles (a) TiO2-ZVI-SiO2 (b) Comparison of diffraction patterns of ZVI, TiO2-nZVI and TiO2-nZVI-SiO2 nanoparticles. | ||
| Phase | 2θ (°) | Crystal plane (hkl) | JCPDS card | Ref. |
|---|---|---|---|---|
| TiO2 (anatase) | 25.3 | (101) | 21-1272 | 75 |
| TiO2 (anatase) | 37.8 | (004) | 21-1272 | 75 |
| TiO2 (anatase) | 48.0 | (200) | 21-1272 | 75 |
| TiO2 (anatase) | 53.9 | (105) | 21-1272 | 75 |
| Fe | 44.7 | (110) | 06-0696 | 79 |
| Fe | 65.1 | (200) | 06-0696 | 80 |
| Amorphous SiO2 | 15–30 (broad hump), 20.2 | (111) | 00-022-1536 | 81 |
A critical peak of zero-valent iron, magnetic and reductive component of the TiO2-nZVI-SiO2 nanocomposite, is observed at 44.7°, indexed (110) JCPDS 06-0696 which is the fingerprint of the nZVI materials.79 However, a small wide peak at 65.1° indexed to the iron (200) plane is associated with the Fe0. The peak reflection at 35.4° could be associated with the formation of iron oxide shells such as Fe3O4 and γ-Fe2O3 which form upon the exposure of Fe0 to air during XRD analysis. However, these narrow passive layers usually produced via surface oxidation often serve to stabilize the highly reactive nZVI core.82–84 The broad silica hump across approximately 15° to 30°, coupled with the absence of sharp, characteristic peaks of SiO2 in the form of quartz, revealed that the SiO2 component exists predominantly in an amorphous state.85,86 The amorphous SiO2 acts functionally as an inert and a stabilizer.63 Hence, it is highly essential for mitigating the intrinsic agglomeration potential of the nZVI and its possible influence on the synthesized TiO2-nZVI nanocomposite.58 In addition, amorphous SiO2 coating bolsters the available surface area of the coated TiO2-nZVI material for synergistic catalytic and reductive reactions.81
3.4 XPS analysis of TiO2-nZVI-SiO2 nanocomposite
Fig. 5 illustrates the X-ray photoelectron spectroscopy analysis of the nanocomposite material. The full survey spectrum in Fig. 5(a) confirmed the presence of titanium (Ti), iron (Fe), and silicon (Si) in the synthesized nanocomposite. Fig. 5(b) deconvoluted via Gaussian model provides a detailed survey of the chemical and electronic interaction of titanium spectrum (Ti 2p) in the nanocomposite material. The well-defined doublet of the Ti 2p3/2 and Ti 2p1/2 peaks were observed at binding energy, BE = 458.7 eV and BE = 464.4 eV respectively. The peaks binding energy separation were found to be BE = 5.7 eV conforming with the predominant +4 oxidation state, Ti4+. The absence of Ti3+ suggest a successful integration of TiO2 with the nZVI.87 In Fig. 5(c), on the other hand, the characteristic Fe 2p3/2 and Fe 2p1/2 spin orbit splitting peaks with two minor peaks located at BE = 705.6 eV and BE = 718.6 eV respectively which are consistent with the XPS findings in ref. 88 and 89. These minor peaks associated with lower intensities are characteristic property of nano zero valent iron, Fe0.90 Subsequently, the passivated iron layers Fe 2p3/2 and Fe 2p½ located at BE = 711 eV and BE = 724.5 eV are a specific characteristic of oxidized Fe0 iron species (Fe2+ and Fe3+) with a potential formation of Fe–OOH and Fe3O4 iron oxides,82,83 confirming the core shell structure of coated nZVI on TiO2 nanoparticles. Fig. 5(d) depicts the detailed survey region of Si 2p, the deconvolution shows two distinct peaks correlated to bulk SiO2 and silicon metal interface (Si–O–Metal) located at BE = 103.5 eV and BE = 102.5 eV respectively. The bulk SiO2 binding energy is consistent with tetrahedral network within the amorphous SiO2 matrix91–93 corroborating the XRD findings of the amorphous nature of silica layer (refer to Section 3.4), coating the nanocomposite material. The silicon metal interface will enable strong interfacial interaction of Si–O–x with Fe0,63 leading to the formation of Si–O–Fe,63 while making SiO2 a strong chemical interface supporting electron coupling in the TiO2-nZVI-SiO2 nanocomposite material. In the O 1s spectrum (Fig. 3(b)), a prominent peak at approximately BE = 529 eV corresponds to the lattice oxygen bond in metal oxides (M−O), while a smaller deconvoluted peak at BE = 532.5 eV is attributed to O–H bonds,94,95 confirming the presence of oxygen atoms associated with surface hydroxyl groups on SiO2,96 TiO2 and Fe–OOH or Fe2O3.4 The detailed region analysis of the C 1s XPS spectra was deconvoluted into four distinct peaks via similar Gaussian model (see Fig. 5(f)). The centered peak at BE = 284.8 eV is assigned to C–C and C–H bonds associated with adventitious hydrocarbon contaminants.97 the peak at BE = 286.0 eV to 287.0 eV and BE = 288.5 eV to 289.5 eV is attributed to oxidized C–O or C–OH and C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O or O–C
O or O–C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O functional groups.98–101 The presence of these oxidized byproducts could be associated with the exposure of adventitious carbon from residual organic precursors utilized in the nanoparticle synthesis to the atmospheric air. Recalling that the synthesized particles were washed several times with ethanol as described in Section 3. At low-binding energy BE = 283.0–284.0 eV signifies C–Metal bonds, particularly metal carbides such as Ti–C, Fe–C102 and Ti–Fe–C.103 However, there was no anatase peak indicating Ti–C bond which is commonly located near 281 eV, suggesting absence of carbon–oxygen replacement in the anatase TiO2 lattice.104,105 Consequently, the formation of these metal carbides at low BE signifies a successful chemical interaction between the nZVI with carbon species confirming electronically coupled and stable nanocomposite. These properties are crucial for overall material catalytic activity, functionality and structural integrity. The overall nanocomposite components selection was designed for synergistic activity, where the TiO2 shell can facilitate the photocatalytic activity and enhanced electron transfer from the underlying nZVI, a mechanism known to boost contaminant reduction. Furthermore, the SiO2 matrix not only stabilizes the composite against aggregation but also modulate interfacial electron dynamics as shown in the Si 2p spectral analysis.
O functional groups.98–101 The presence of these oxidized byproducts could be associated with the exposure of adventitious carbon from residual organic precursors utilized in the nanoparticle synthesis to the atmospheric air. Recalling that the synthesized particles were washed several times with ethanol as described in Section 3. At low-binding energy BE = 283.0–284.0 eV signifies C–Metal bonds, particularly metal carbides such as Ti–C, Fe–C102 and Ti–Fe–C.103 However, there was no anatase peak indicating Ti–C bond which is commonly located near 281 eV, suggesting absence of carbon–oxygen replacement in the anatase TiO2 lattice.104,105 Consequently, the formation of these metal carbides at low BE signifies a successful chemical interaction between the nZVI with carbon species confirming electronically coupled and stable nanocomposite. These properties are crucial for overall material catalytic activity, functionality and structural integrity. The overall nanocomposite components selection was designed for synergistic activity, where the TiO2 shell can facilitate the photocatalytic activity and enhanced electron transfer from the underlying nZVI, a mechanism known to boost contaminant reduction. Furthermore, the SiO2 matrix not only stabilizes the composite against aggregation but also modulate interfacial electron dynamics as shown in the Si 2p spectral analysis.
 | ||
| Fig. 5 TiO2-ZVI-SiO2 XPS analysis. Full survey response (a), and (b–f) detailed survey of regions Ti 2p, Fe 2p, Si 2p, O 1s, and C 1s. | ||
3.5 TGA analysis of TiO2-nZVI-SiO2 nanocomposite compared with TiO2-nZVI nanoparticles
Fig. 6 shows the thermal gravimetric analysis of TiO2-nZVI-SiO2 and compares the degradation properties with the precursor TiO2-nZVI composite to evaluate the influence of the SiO2 layer on the thermal behavior and structural stability. From Fig. 6(a–c), the TiO2-nZVI composite shows multi-stage thermal degradation profile (see S1 TGA 1&2), with a total weight loss of 38 w% across the TGA temperature range. The first degradation stage at 150 °C, characterized by 5 wt% to 8 wt% loss, is attributed to the removal of physiosorbed water and residual solvent molecules.41,106 Weight loss from 150 °C to 800 °C is associated with the decomposition of organic species, such as –O–H groups on the nanocomposite surface, and the oxidation of the nZVI core to iron oxides such as Fe3O4 or Fe2O3.107 The constant degradation of the composite material at this stage implies nearly complete thermal oxidation of the reactive nZVI core and the initial exposure of TiO2 shell. In (Fig. 7(c)) the TiO-nZVI-SiO2 composite revealed better thermal stability, with a total mass loss of 3% across the TGA temperature range. The flat TGA curve indicates high thermal stability of the SiO2-coated nanocomposite material, which is consistent with the findings in ref. 107 where SiO2 was reported to enhance the thermal stability of TiO2 in TiO2–SiO2 nanocomposite. The SiO2 layer functions as a protective barrier that restricts the diffusion of oxygen and moisture to the nZVI, slowing the thermal oxidation and overall aggregation of the SiO2-coated nanocomposite. | ||
| Fig. 6 TGA Analysis of TiO2-nZVI and TiO2-nZVI-SiO2 (a and c), weight 1st order differential of TiO2-Nzvi and TiO2-nZVI-SiO2 (b and d) nanocomposite. | ||
3.6 Membrane properties and performance evaluation
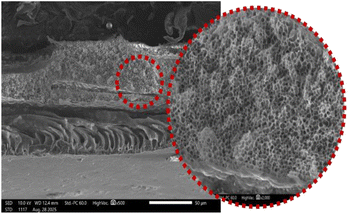
The results from the FESEM analyses (refer to Table 3) further corroborates the incorporation of TiO2-nZVI-SiO2 nanoparticles in the PVDF matrix, as observed by the induced concentration-dependent morphology associated with the presence of titania particles. Consequently, at lower TiO2-nZVI-SiO2 loadings 0.10 g to 0.20 g, the nanocomposite exhibited a refined surface pore structure with increased mean pore size while maintaining consistent membrane porosity.123,126,127 Results from this cross-sectional morphology corroborate the influence of nanocomposite loadings in the formation of extended macro-voids in the membrane structure.49,128,129 These morphological properties are associated to the TiO2-nZVI-SiO2 ability in enhancing the phase inversion process while promoting instantaneous de-mixing and solvent to nonsolvent exchange kinetics. Similarly, higher TiO2-nZVI-SiO2 loadings (0.25 g to 0.30 g) instigate a proportional morphological transition. The increased dope solution viscosity delays phase separation, leading to looser sub-layer, more prominent surface pores, and increased overall porosity to 87.53% (see Table 4). This shift, governed by thermodynamic characteristics, results in a structure characterized by higher porosity and a thinner selective layer, which in turn could enhance water permeability.130,131 However, it could also compromise rejection performance due to the less compact morphology. Table 4 shows characteristics of fabricated nanocomposite membrane.
| Modified membrane | Average membrane thickness (µm) | Membrane pore size × 10−3(µm) | Membrane porosity (%) | Crystallinity (%) |
|---|---|---|---|---|
| 0.10 g TiO2-nZVi-SiO2 | 49 | 11.874 | 83.81 | 34.86 |
| 0.15 g TiO2-nZVi-SiO2 | 53 | 11.836 | 85.30 | 38.11 |
| 0.20 g TiO2-nZVi-SiO2 | 55 | 11.747 | 85.90 | 39.42 |
| 0.25 g TiO2-nZVi-SiO2 | 56 | 11.770 | 85.92 | 45.17 |
| 0.30 g TiO2-nZVi-SiO2 | 58 | 11.746 | 87.53 | 58.39 |
Fig. 11 shows the results from solvent content analysis (11a) and the relationship between membrane porosity and membrane shrinkage ratio (11b). The solvent content of the nanocomposite membrane was evaluated using water, methanol and ethanol with the relationship described in eqn (8). The results from this analysis revealed a proportional relation of H2O content with membrane porosity and hydrophilicity conforming the findings in ref. 43. A significant decline in the trend, from 0.78 to 0.11, was observed across the modified nanocomposite membranes (M1 to M5). This decline corresponds to a decreased solvent polarity, as demonstrated by the ethanol and methanol solvents shown in Fig. 11a. To further evaluate this membrane characteristic property and its relationship with membrane porosity, membrane percent shrinkage ratio was computed following eqn (9) and compared with the membrane porosity,136 see Fig. 11b. Consistent with prior research, the findings establish an inverse correlation between membrane porosity and the shrinkage ratio.137 The modified membrane, M5 with the maximum porosity and nanocomposite loading showed minimum shrinkage ratio corroborating its suitability for purification technology.
 | ||
| Fig. 12 Pure water analysis of PVDF and TiO2-nZVI-SiO2 modified nanocomposite membranes (Mo = pristine PVDF; M1 = 0.1 g, M2 = 0.15 g, M3 = 0.20 g, M4 = 0.25 g and M5 = 0.3 g TiO2-ZVI-SiO2). | ||
To evaluate the overall performance of the membrane material and determine the optimum nanocomposite loading, an initial 10 ppm concentration of lead (Pb2+) solution was utilized. The results of filtration experiments obtained for the modified nanocomposite membranes are shown in Fig. 13(a and b). From Fig. 13(a) the concentration gradient shows a sharp decline across all the membranes. The initial rapid decline suggests the impact of combined mechanisms of surface adsorption and size exclusion following redox reaction during the filtration operation,145 a steady state Pb2+ removal rate was subsequently established, confirming the long-term stability of the nanocomposite membranes. The removal efficiencies shown in Fig. 13(b) were found to be proportional to the nanocomposite loading with optimum membrane (M5) achieving 99.8% removal efficiency during the 50 minutes filtration operations. This excellent performance can be associated with an increased number of reactive sites at higher loading of the nanocomposite materials as more of the nZVI and TiO2 nanoparticles are introduced into the membrane matrix, providing synergistic Pb2+ reduction and adsorption capabilities on the modified membrane surface. Namakka et al., (2024) reported similar synergistic behavior of nZVI, TiO2 and SiO2 recording 99.9% methylene blue dye removal.40 Similar mechanisms were reported for chromium(VI)123 and chlorophenol146 removal using nZVI-TiO2 nanocomposite membranes. The enhanced adsorption and reduction capabilities of the developed nZVI-TiO2 components facilitate the production of reduced lead Pb2+ species which are subsequently retained by the modified membrane matrix. While SiO2 coating, in addition to stabilizing nZVI-TiO2 and preventing aggregation, ensures effective dispersion and maximum available reactive sites. The optimum membrane (M5) was further utilized in assessing the membrane stability and to investigate the influence of heavy metal types and concentrations by varying concentration of Pb2+ and chromium(VI) solutions. The results are shown in Fig. 14(a–d).
The results in Fig. 14(a and b) show the performance of the M5 nanocomposite membrane under 70 minutes of filtration time at 1 bar pressure. The concentration of the tank solutions was varied across the heavy metal filtration solutions: 20 ppm, 30 ppm, and 40 ppm for both Pb2+ and Cr(VI). Subsequently, the concentration of Cr(VI) was increased to 50 ppm to further explore the membrane boundaries and evaluate overall membrane stability, see Fig. 14(c).
The results revealed maximum removal efficiencies at lower Pb2+ and Cr(VI) loadings, which decline as contaminants concentrations increase (refer to Fig. 14(a)). The maximum efficiencies 97.7%, 96.9%, and 96% for 20 ppm, 30 ppm, and 40 ppm Pb2+ respectively. Similar trends were observed in Cr(VI) removal experiment (refer to Fig. 14(b)), in which 94.3%, 92%, and ∼90% were obtained for 20 ppm, 30 ppm, and 40 ppm, respectively. The trends confirmed the concentration-dependent behaviour of the nanocomposite membrane, which conforms with the findings in ref. 147 and 148. Lower removal rate observed (refer to Fig. 14a and b) with increasing heavy metal loading suggests high concentration of Pb2+ and Cr(VI) ions on the reactive sites of the membrane matrix, limiting the kinetic rate of the reduction reaction.149,150 The plateau at low efficiencies corresponding to higher metal ion loadings (30 ppm and 40 ppm) can be associated with the possible active site's saturation causing overall mass transfer resistance through the nanocomposite membrane.149 Although the 40 ppm curves show relatively higher removal efficiency (96.9%) in the removal of Pb2+ in contrast to the 90% obtained for Cr(VI), the nanocomposite membrane proved to be highly efficient for both Pb2+ and Cr(VI) remediation applications. It is imperative to note that the mechanism involved in contaminants removal depends on the nature of the contaminants in addition to the catalytic activity of the nanocomposite membrane. For example, the disparity in the removal efficiencies observed in Pb2+ and Cr(VI) can be strongly influenced by the reaction mechanism involved. Since Pb2+ cations are easily reduced via chemical reduction into metallic lead or converted to Pb (OH)2 or oxyhydroxide, which can be easily retained by the nanocomposite membrane matrix. However, the Cr(VI) reduction mechanisms involve the initial generation of Cr3+ species by redox reaction with nZVI components of the nanocomposite membrane followed by subsequent precipitation or adsorption by the TiO2–SiO2 components. The synergistic mechanisms of every nanocomposite component allow for efficient removal of these contaminants (>90%).
To further confirm the adsorption behavior of these contaminants onto the PVDF-TiO2-nZVI-SiO2 nanocomposite membranes and their interaction with TiO2-nZVI-SiO2 particles active sites, equilibrium adsorption isotherm analysis was performed at 25 °C following the procedure described by ref. 114. M1 to M5 membrane data were analyzed using the pseudo first order (PFO), and pseudo second order (PSO) kinetic models. Subsequently the optimal membrane (M5) was subjected to adsorption isotherm analysis using Langmuir isotherms, and the parameters are detailed in S2. Both PSO, PFO and Langmuir model demonstrated a sufficient fit with a higher linear correlation coefficient R2 ranging from 0.96 to 0.99. The R2 values (refer to S2) for the PFO and PSO kinetic models corroborate a strong correlation between the experimental data for Pb2+ removal and the respective kinetic models, confirming how these models accurately described the adsorption rate.151 The higher R2 value of M5 observed (>0.99) for the PSO model suggests chemisorption as the rate limiting step of the adsorption process,152 in which Pb2+ ions are removed by chemical interactions, such as the sharing or exchange of electrons between the Pb2+ ions and the hydroxyl functional groups present on the surface of the optimal PVDF-TiO2-nZVI-SiO2 nanocomposite membrane, confirming the chemical nature of the binding mechanism, which is characteristic of highly efficient, functionalized adsorbent. Similar findings were reported in previous studies.114
Fig. 15(a) shows the Cr(IV) rejection rate at various loadings on M5 membrane. The figures compared overall performance stability with efficiency of the optimum membrane. The results indicate that the Cr(VI) rejection rate declines as the initial concentration of Cr(VI) ions increases, with an average overall rejection rate of 92% across the Cr(VI) contaminant loadings, suggesting a balanced and effective removal efficiency throughout the filtration period. The sustained performance despite higher contaminants loading demonstrates the ability of the SiO2 coating in preventing leaching and ensuring stabilization of nZVI-TiO2 against aggregation. Despite the conventional principle that higher concentration of the contaminant results to a greater mass transfer resistance and potential blockage of catalytic active sites,149 the rejection stability obtained in this study (>90% at higher Cr(IV) and Pb2+ concentrations) confirmed a continual availability of active sites from the nanocomposite membrane for the mineralization Pb2+ and chemical reduction of Cr(VI) to Cr3+.
 | ||
| Fig. 15 Comparison of (a) Cr(VI) rejection rate (%) at 20 pm, 30 ppm, 40 ppm and 50 ppm of the optimal membrane (M5) with (b) the overall membrane efficiency over the filtration time. | ||
In Fig. 15(b) the rejection rate of Cr(VI) and Pb2+ at 20 ppm, 30 ppm, 40 ppm, and 50 ppm were compared with the filtration time to evaluate the influence of normalized filtration time, flux stability and membrane performance.
The long-term stability and reusability of the developed PVDF-TiO2-nZVI-SiO2 optimal nanocomposite membrane was investigated through 5 ppm Pb2+ solution over 7 cycles of filtration operation as shown in Fig. 16. A slow flux decrease was observed after 4.7 hours of filtration operation, attributable to the accumulation of surface foulants. However, the membrane flux was regenerated following hydraulic cleaning treatment. Furthermore, the separation efficiency remained above 98% confirming the stability and structural integrity of the PVDF-TiO2-nZVI-SiO2 nanocomposite membrane. FTIR analysis of the optimal membrane (refer to Fig. 17) was further conducted and compared with spectral peaks before reusability analysis. The results from this analysis corroborate a consistent hydraulic and anti-fouling performance of the optimum nanocomposite membrane associated with influence of enhanced hydrophilic properties,43,52 observed in the water contact angle analysis discussed in the previous section. The hydrophilic properties and flux stability of the developed nanocomposite membrane is a crucial finding of this study that contribute to the operational challenges in practical membrane applications particularly where there is an intermittent flux decline or a trade-off between membrane hydraulic properties and membrane purification performance.
 | ||
| Fig. 16 Long-term stability studies of the optimal PVDF-TiO2-nZVI-SiO2 nanocomposite membrane using 5 ppm Pb2+ solution. | ||
 | ||
| Fig. 17 FTIR spectra comparing the optimal PVDF-TiO2-nZVI-SiO2 nanocomposite membrane before and after Pb2+ treatment. | ||
The FTIR spectra comparing the PVDF-TiO2-nZVI-SiO2 nanocomposite membrane M5 before and after Pb2+ treatment is shown in Fig. 17. The results revealed changes in peak intensities and provided molecular insight on the Pb2+ chemisorption removal process with the functional groups on the membrane surface. The change in peaks intensities and shift of the O–H stretching vibrations peaks observed around 3000 to 3500 cm−1 regions indicate active participation of these functional groups in binding Pb2+ ions. Furthermore, there is an observed changes in the shape and positions of Si–O, Fe–O peaks at 1000–1200 cm−1, these spectral changes corroborate the formation of a chemical bond or chelation complex between the Pb2+ ions and the oxygen-containing functional groups of the PVDF-TiO2-nZVI-SiO2 nanocomposite which could be basis for the high R2 value observed in the PSO studies (refer to S2).
4 Conclusion
In conclusion, this study provided a comprehensive investigation of the synthesis, characterization, and performance evaluation of a novel PVDF-TiO2-nZVI-SiO2 nanocomposite membrane for the removal of Cr(VI) and Pb2+ from aqueous solutions. The physicochemical properties of the synthesized TiO2-nZVI-SiO2 nanocomposite were investigated via various techniques with XPS and TGA analysis confirming the thermal stability and successful coating of the SiO2 on the TiO2-nZVI surface. The developed PVDF-TiO2-nZVI-SiO2 nanocomposite membrane showed stable hydraulic properties, enhanced hydrophilicity and long-term stability. The optimum nanocomposite loading (M5) was determined and the Pb2+ removal was found to be higher, 99.8% compared to Cr(VI) 94.3% which required the formation of Cr3+ prior to co-precipitation and membrane matrix retention. The removal efficiencies exceed 90% across all nanocomposite membranes and contaminants loadings.Author contributions
All authors contributed to the study conception and design. Material preparation, experiments, analysis and writing the first draft were performed by Murtala Namakka. And all authors commented on previous versions of the manuscript. All authors read and approved of the final manuscript.Conflicts of interest
The authors have no relevant financial or non-financial interests to disclose.Data availability
Data described in this research are provided in the supplementary information (SI) file. Supplementary information is available. See DOI: https://doi.org/10.1039/d5ra08723f.Acknowledgements
This work was funded by Universiti Malaysia Sarawak under the Vice Chancellor Higher Impact Research Scheme, Grant Number (UNI/FO2/VC-HIRG/85508/P10-03).References
- M. Herrera, X. Xie, A. Menapace, A. Zanfei and B. M. Brentan, Sustainable AI infrastructure: A scenario-based forecast of water footprint under uncertainty, J. Clean. Prod., 2025, 526(August), 146528, DOI:10.1016/j.jclepro.2025.146528.
- J. Cowls, A. Tsamados, M. Taddeo and L. Floridi, The AI gambit: leveraging artificial intelligence to combat climate change—opportunities, challenges, and recommendations, AI Soc., 2023, 38(1), 283–307, DOI:10.1007/s00146-021-01294-x.
- Y. Zhao, W. Yu, R. Li, Y. Xu, Y. Liu and T. Sun, et al., Electric field endowing the conductive polyvinylidene fluoride (PVDF)-graphene oxide (GO)-nickel (Ni) membrane with high-efficient performance for dye wastewater treatment, Appl. Surf. Sci., 2019, 483(January), 1006–1016, DOI:10.1016/j.apsusc.2019.04.054.
- S. M. Botsa, Reduced graphene oxide – assisted TiO2 – Fe2O3 ternary nanocomposite for efficient visible – light driven photocatalysis of nitrobenzene and dye pollutants, Adv. Compos. Hybrid Mater., 2025, 8, 384, DOI:10.1007/s42114-025-01431-w.
- G. Jingling, Y. S. Bin Xiang and J. Li, Janus smart materials with asymmetrical wettability for on-demand oil/water separation: a comprehensive review, J. Mater. Chem. A, 2023, 11(46), 25093–25114 RSC . https://pubs.rsc.org/en/content/articlelanding/2023/ta/d3ta04160c.
- H. An, T. Liu, X. Xiao, M. Liu, Y. Hu and P. Wei, et al., Magnetic biochar-supported nanoscale zero-valent iron for remediation of arsenic and cadmium-contaminated soils: The role of free radicals, Environ. Res., 2025, 276, 121484, DOI:10.1016/j.envres.2025.121484.
- C. Zhang, D. Shi, C. Wang, G. Sun, H. Li and Y. Hu, et al., Pristine/magnesium-loaded biochar and ZVI affect rice grain arsenic speciation and cadmium accumulation through different pathways in an alkaline paddy soil, J. Environ. Sci., 2024, 147, 630–641 CrossRef PubMed.
- H. Alijani and Z. Shariatinia, Effective aqueous arsenic removal using zero valent iron doped MWCNT synthesized by in situ CVD method using natural α-Fe2O3as a precursor, Chemosphere, 2017, 171, 502–511, DOI:10.1016/j.chemosphere.2016.12.106.
- A. Khan, S. Ahmad, M. Nafees, Z. Mahmood, S. Ali and W. Du, et al., Chemosphere Alleviated cadmium toxicity in wheat ( Triticum aestivum L.) by the coactive role of zinc oxide nanoparticles and plant growth promoting rhizobacteria on TaEIL1 gene expression , biochemical and physiological changes, Chemosphere, 2024, 364, 143113, DOI:10.1016/j.chemosphere.2024.143113.
- V. Khum-in, J. Suk-in, P. In-ai, K. Piaowan, Y. Praimeesub and K. Rintachai, et al., Combining magnet-assisted soil washing and soil amendment with zero-valent iron to restore safe rice cultivation in real cadmium-contaminated paddy fields, Chemosphere, 2023, 340, 139816, DOI:10.1016/j.chemosphere.2023.139816.
- S. Xie, Water contamination due to hexavalent chromium and its health impacts: exploring green technology for Cr (VI) remediation, Green Chem. Lett. Rev., 2024, 17(1), 1–19, DOI:10.1080/17518253.2024.2356614.
- Z. Ren, H. Tang, H. Li and Q. Jing, Journal of Water Process Engineering Column experimental study on the removal of hexavalent chromium from water by modified cellulose filter paper loaded with nano zero-valent iron, J. Water Proc. Eng., 2024, 59, 104920, DOI:10.1016/j.jwpe.2024.104920.
- L. Zhang, Y. Zhu, H. Gu, S. S. Lam, X. Chen and C. Sonne, et al., A review of phytoremediation of environmental lead (pb) contamination, Chemosphere, 2024, 362, 142691, DOI:10.1016/j.chemosphere.2024.142691.
- W. Zhan, Y. Yuan, X. Zhang, Y. Liang, S. Song and M. de Jesús Martínez-López, et al., Efficient and selective Lead(II) removal within a wide concentration range through chemisorption and electrosorption coupling process via defective MoS2 electrode, Sep. Purif. Technol., 2024, 329, 125183, DOI:10.1016/j.seppur.2023.125183.
- M. A. Irshad, S. Sattar, R. Nawaz, S. A. Al-Hussain, M. Rizwan and A. Bukhari, et al., Enhancing chromium removal and recovery from industrial wastewater using sustainable and efficient nanomaterial: A review, Ecotoxicol. Environ. Saf., 2023, 263, 115231, DOI:10.1016/j.ecoenv.2023.115231.
- I. R. Chowdhury, S. Chowdhury, M. A. J. Mazumder and A. Al-Ahmed, Removal of lead ions (Pb2+) from water and wastewater: a review on the low-cost adsorbents, Appl. Water Sci., 2022, 12, 1–33, DOI:10.1007/s13201-022-01703-6.
- V. Kumar, S. K. Dwivedi and S. Oh, A critical review on lead removal from industrial wastewater: Recent advances and future outlook, J. Water Proc. Eng., 2022, 45(2021), 102518, DOI:10.1016/j.jwpe.2021.102518.
- Q. Chen, Z. Luo, C. Hills, G. Xue and M. Tyrer, Precipitation of heavy metals from wastewater using simulated flue gas: Sequent additions of fly ash, lime and carbon dioxide, Water Res., 2009, 43(10), 2605–2614, DOI:10.1016/j.watres.2009.03.007.
- M. C. Benalia, L. Youcef, M. G. Bouaziz, S. Achour and H. Menasra, Removal of Heavy Metals from Industrial Wastewater by Chemical Precipitation: Mechanisms and Sludge Characterization, Arabian J. Sci. Eng., 2022, 47(5), 5587–5599, DOI:10.1007/s13369-021-05525-7.
- E. Sgreccia, C. Rogalska, F. S. Gallardo Gonzalez, P. Prosposito, L. Burratti and P. Knauth, et al., Heavy metal decontamination by ion exchange polymers for water purification: counterintuitive cation removal by an anion exchange polymer, J. Mater. Sci., 2024, 59(7), 2776–2787, DOI:10.1007/s10853-024-09356-3.
- A. Q. Jasim and S. K. Ajjam, Removal of heavy metal ions from wastewater using ion exchange resin in a batch process with kinetic isotherm, S. Afr. J. Chem. Eng., 2024, 49, 43–54, DOI:10.1016/j.sajce.2024.04.002.
- Z. Wang, Z. Tan, H. Li, S. Yuan, Y. Zhang and Y. Dong, Direct current electrochemical method for removal and recovery of heavy metals from water using straw biochar electrode, J. Clean. Prod., 2022, 339, 130746, DOI:10.1016/j.jclepro.2022.130746.
- E. C. Okpara, O. E. Fayemi, O. B. Wojuola, D. C. Onwudiwe and E. E. Ebenso, Electrochemical detection of selected heavy metals in water: a case study of African experiences, RSC Adv., 2022, 12(40), 26319–26361 RSC.
- E. H. Khader, S. A. Muslim, N. M. C. Saady, N. S. Ali, I. K. Salih and T. J. Mohammed, et al., Recent advances in photocatalytic advanced oxidation processes for organic compound degradation: A review, Desalination Water Treat., 2024, 318, 100384, DOI:10.1016/j.dwt.2024.100384.
- B. Yan, Y. Dai, L. Xin, M. Li, H. Zhang and H. Long, et al., Research progress in the degradation of printing and dyeing wastewater using chitosan based composite photocatalytic materials, Int. J. Biol. Macromol., 2024, 263, 130082, DOI:10.1016/j.ijbiomac.2024.130082.
- T. Munawar, F. Mukhtar, K. M. Batoo, A. Mazhar, M. S. Nadeem and S. Hussain, et al., Sunlight-activated Mo-doped La2CuO4/rGO perovskite oxide nanocomposite for photocatalytic treatment of diverse dyes pollutant, Mater. Sci. Eng. B, 2024, 304, 117355, DOI:10.1016/j.mseb.2024.117355.
- D. Fatta-Kassinos, M. I. Vasquez and K. Kümmerer, Transformation products of pharmaceuticals in surface waters and wastewater formed during photolysis and advanced oxidation processes - Degradation, elucidation of byproducts and assessment of their biological potency, Chemosphere, 2011, 85(5), 693–709, DOI:10.1016/j.chemosphere.2011.06.082.
- L. Rizzo, Bioassays as a tool for evaluating advanced oxidation processes in water and wastewater treatment, Water Res., 2011, 45(15), 4311–4340, DOI:10.1016/j.watres.2011.05.035.
- C. R. Holkar, A. J. Jadhav, D. V. Pinjari, N. M. Mahamuni and A. B. Pandit, A critical review on textile wastewater treatments: Possible approaches, J. Environ. Manage., 2016, 182, 351–366, DOI:10.1016/j.jenvman.2016.07.090.
- W. S. Chai, J. Y. Cheun, P. S. Kumar, M. Mubashir, Z. Majeed and F. Banat, et al., A review on conventional and novel materials towards heavy metal adsorption in wastewater treatment application, J. Clean. Prod., 2021, 296, 126589, DOI:10.1016/j.jclepro.2021.126589.
- B. He, W. Zhang, Y. Diao, S. Sun, Y. Zhang and W. Zhao, et al., Mechanistic study of the adsorption capabilities of heavy metals on the surface of ferrihydrite: batch sorption, modeling, and density functional theory, RSC Adv., 2025, 15(2), 1072–1080 Search PubMed.
- L. Mohapatra, Y. F. Tsang, X. Dou and K. Baek, MnFe-layered double hydroxides grown on spherical SiO2 to construct core-shell heterostructures for enhanced simultaneous photocatalytic redox Reactions of Cr(VI) and As(III), J. Water Proc. Eng., 2022, 50, 103236, DOI:10.1016/j.jwpe.2022.103236.
- Q. Jing, W. You, S. Qiao, Y. Ma and Z. Ren, Comprehensive understanding of adsorption and reduction on 2,4-DCP and Cr(VI) removal process by NZVI-rGO: Performance and mechanism, J. Water Proc. Eng., 2023, 51, 103413, DOI:10.1016/j.jwpe.2022.103413.
- R. Fu, Y. Yang, Z. Xu, X. Zhang, X. Guo and D. Bi, The removal of chromium (VI) and lead (II) from groundwater using sepiolite-supported nanoscale zero-valent iron (S-NZVI), Chemosphere, 2015, 138, 726–734, DOI:10.1016/j.chemosphere.2015.07.051.
- M. Kazemi, M. Peyravi and M. Jahanshahi, Multilayer UF membrane assisted by photocatalytic NZVI@TiO2 nanoparticle for removal and reduction of hexavalent chromium, J. Water Proc. Eng., 2020, 37, 101183, DOI:10.1016/j.jwpe.2020.101183.
- A. M. Zafar, A. Naeem, M. A. Minhas, M. J. Hasan, S. Rafique and A. Ikhlaq, Removal of reactive dyes from textile industrial effluent using electrocoagulation in different parametric conditions of aluminum electrodes, Total Environ. Adv., 2024, 9, 200087, DOI:10.1016/j.teadva.2023.200087.
- Z. Hejri, M. Hejri, M. Omidvar and S. Morshedi, Synthesis of TiO2/nZVI nanocomposite for nitrate removal from aqueous solution, Int. J. Ind. Chem., 2019, 10(3), 225–236, DOI:10.1007/s40090-019-0186-3.
- M. Zhao, Z. Niu, Y. Xu, F. Fan, W. Wu and D. Pan, One-step electrospinning preparation of magnetic NZVI@TiO2 nanofibers for enhanced immobilization of U(VI) from aqueous solution, J. Radioanal. Nucl. Chem., 2023, 332(4), 1083–1091, DOI:10.1007/s10967-022-08696-1.
- M. Namakka and R. Rahman, RSC Advances Insights into micro-and nano-zero valent iron materials : synthesis methods and multifaceted applications, RSC Adv., 2024, 14, 30411–30439, 10.1039/D4RA03507K.
- M. Namakka, M. R. Rahman, K. A. Mohamad Bin Said, K. K. Kuok, F. A. Md Yusof and M. S. M. Al-Saleem, et al., Unveiling the synergistic effect of an nZVI–SiO 2 –TiO 2 nanocomposite for the remediation of dye contaminated wastewater, Mater. Adv., 2024, 9292–9313, 10.1039/d4ma00853g.
- M. Namakka, M. R. Rahman, K. A. Mohamad Bin Said, K. K. Kuok, F. A. Md Yusof and M. S. M. Al-Saleem, et al., Unveiling the synergistic effect of an nZVI–SiO 2 –TiO 2 nanocomposite for the remediation of dye contaminated wastewater, Mater. Adv., 2024, 9292–9313, 10.1039/d4ma00853g.
- S. S. Koçak, O. S. Özdemir, M. Asiltürk and İ. Atmaca, Thermal and Optical Characteristics of TiO2@SiO2, Fe3O4@SiO2, and ZnO@SiO2 Core–Shell Nanoparticles and Their Water-Based Nanofluids, Int. J. Thermophys., 2025, 46, 3–38, DOI:10.1007/s10765-025-03558-w.
- H. Nawaz, M. Umar, A. Ullah, H. Razzaq, K. M. Zia and X. Liu, Polyvinylidene fluoride nanocomposite super hydrophilic membrane integrated with Polyaniline-Graphene oxide nano fillers for treatment of textile effluents, J. Hazard. Mater., 2021, 403, 123587, DOI:10.1016/j.jhazmat.2020.123587.
- M. R. Rahman, A. James, M. Said, M. Namakka, M. U. Khandaker, W. H. Jiunn, J. Y. Al-Humaidi, R. H. Althomali and M. M. Rahman, A TiO2 grafted bamboo derivative nanocellulose polyvinylidene fluoride (PVDF) nanocomposite membrane for wastewater treatment by a photocatalytic process, Mater. Adv., 2024, 7617–7636, 10.1039/d4ma00716f.
- D. Sharma, S. Kanchi and K. Bisetty, Biogenic synthesis of nanoparticles: A review., Arab. J. Chem., 2019, 12, 3576–3600 Search PubMed.
- S. Aziz and A. Abdel-Karim, Dual-functional ultrafiltration biocatalytic membrane containing laccase/nanoparticle for removal of pollutants: A review, Environ. Nanotechnol. Monit. Manag., 2023, 20, 100852, DOI:10.1016/j.enmm.2023.100852.
- G. H. Major, V. Fernandez, N. Fairley, E. F. Smith and M. R. Linford, Guide to XPS data analysis: Applying appropriate constraints to synthetic peaks in XPS peak fitting, J. Vac. Sci. Technol. A, 2022, 40(6), 063201, DOI:10.1116/6.0001975.
- A. C. Baily, T. Nunney, Thermo Fisher Scientific, East Grinstead, West Sussex, UK, XPS Analysis of a Hard Disk Platter by Rapid Depth Profiling, 2019 Search PubMed.
- A. James, M. Rezaur Rahman, K. Anwar Mohamed Said, M. Namakka, K. Kuok Kuok and M. Uddin Khandaker, et al., Lithium Chloride-Mediated enhancement of dye removal capacity in Borneo bamboo derived nanocellulose-based nanocomposite membranes (NCMs), J. Mol. Liq., 2024, 413, 125973, DOI:10.1016/j.molliq.2024.125973.
- ASTM F3419-22. ASTM International, Standard Test Method for Mineral Characterization of Equine Surface Materials by X-Ray Diffraction (XRD) Techniques, West Conshohocken, PA, USA, 2022 Search PubMed.
- M. R. Rahman, A. Z. Sueraya, K. A. B. M. Said, M. Namakka, A. James and I. M. M. Rahman, et al., Impact of Graphene/Nanocellulose on Nanocomposite Membrane for Methylene Blue Dye Removal and Antifouling Performance, J. Appl. Polym. Sci., 2025, 1–21 Search PubMed.
- G. Hurwitz, G. R. Guillen and E. M. V. Hoek, Probing polyamide membrane surface charge, zeta potential, wettability, and hydrophilicity with contact angle measurements, J. Membr. Sci., 2010, 349(1–2), 349–357 CrossRef CAS.
- T. Iqbal, R. Sahrash, A. Siddiqa, S. Afsheen, M. B. Tahir and M. I. Khan, et al., Preparation and characterization of polyvinylidene fluoride/1-butyl-3-methylimidazolium bromide-based ionogel membranes for desalination applications, Int. J. Environ. Sci. Technol., 2019, 16(11), 7081–7092, DOI:10.1007/s13762-019-02207-8.
- C. S. Ong, W. J. Lau, P. S. Goh, B. C. Ng, T. Matsuura and A. F. Ismail, Effect of PVP Molecular Weights on the Properties of PVDF-TiO2 Composite Membrane for Oily Wastewater Treatment Process, Separ. Sci. Technol., 2014, 49(15), 2303–2314 CrossRef CAS.
- P. L. N. Khui, M. R. Rahman, K. A. bin M. Said, M. Namakka, M. Shahabuddin and M. S. M. Al-Saleem, et al., Enhanced flux and surface modification of PVDF hollow fiber membranes using bamboo cellulose/PVA composite coatings, Polym. Bull., 2025,(0123456789), 12909–12927, DOI:10.1007/s00289-025-06023-8.
- R. Razavi, A. Shakeri and H. Salehi, A novel thin film nanocomposite forward osmosis membrane modified with zwitterionic GO-g-PSBMA with high fouling resistance and heavy metal ions rejection, J. Environ. Chem. Eng., 2025, 13(1), 115308, DOI:10.1016/j.jece.2025.115308.
- M. R. Rahman, A. Z. Sueraya, K. A. B. M. Said, M. Namakka, A. James and I. M. M. Rahman, et al., Impact of Graphene/Nanocellulose on Nanocomposite Membrane for Methylene Blue Dye Removal and Antifouling Performance, J. Appl. Polym. Sci., 2025, 1–21 Search PubMed.
- M. Namakka, M. R. Rahman, K. A. Bin Mohamad Said and A. Muhammad, Insights into micro-and nano-zero valent iron materials: synthesis methods and multifaceted applications, RSC Adv., 2024, 14(41), 30411–30439 Search PubMed.
- R. Zhang, R. Napolano, B. Xi, A. M. Salazar, Q. Shi and Y. Zhao, et al., Mechanistic insights into Cr(VI) removal by a combination of zero-valent iron and pyrite, Chemosphere, 2023, 330, 138693, DOI:10.1016/j.chemosphere.2023.138693.
- Q. B. Nguyen, C. Kim and I. Hwang, Roles of silica coating on nanosized zero-valent iron in sequential reduction–oxidation process in a system containing persulfate, J. Hazard. Mater., 2024, 480, 135946, DOI:10.1016/j.jhazmat.2024.135946.
- P. Y. Jia, X. M. Liu, G. Z. Li, M. Yu, J. Fang and J. Lin, Sol-gel synthesis and characterization of SiO2@CaWO 4,SiO2@CaWO4:Eu3+/Tb3+ core-shell structured spherical particles, Nanotechnology, 2006, 17(3), 734–742 CrossRef CAS.
- S. Saravanan, R. S. Dubey, S. Saravanan and R. S. Dubey, Synthesis of SiO2 Nanoparticles by Sol-Gel Method and Their Optical and Structural Properties Thin films for Opto-Electronic Applications View project Nanomaterials View project Synthesis of SiO2 Nanoparticles by Sol-Gel Method and Their Optical and Structural Properties, Rom. J. Inf. Sci. Technol., 2020, 23, 1 Search PubMed . https://www.researchgate.net/publication/338828540.
- C. Ratanaphain, D. Viboonratanasri, P. Prompinit, S. Krajangpan, E. Khan and P. Punyapalakul, Mechanistic study of iodoacetamide degradation by functionalized SiO2-coated nZVI: The role of surface functional groups on adsorption, dehalogenation and hydrolysis, J. Water Proc. Eng., 2024, 63, 105485, DOI:10.1016/j.jwpe.2024.105485.
- A. Rosales and K. Esquivel, SiO2@TiO2 composite synthesis and its hydrophobic applications: A review, Catalysts, 2020, 10(2), 1–17, DOI:10.3390/catal10020171.
- Z. Liu, G. Chen, F. Hu and X. Li, Synthesis of mesoporous magnetic MnFe2O4@CS-SiO2 microsphere and its adsorption performance of Zn2+ and MB studies, J. Environ. Manage., 2020, 263, 110377, DOI:10.1016/j.jenvman.2020.110377.
- Q. Li, H. Wang, Z. Chen, X. He, Y. Liu and M. Qiu, et al., Adsorption-reduction strategy of U(VI) on NZVI-supported zeolite composites via batch, visual and XPS techniques, J. Mol. Liq., 2021, 339, 116719, DOI:10.1016/j.molliq.2021.116719.
- M. Zhao, Z. Niu, Y. Xu, F. Fan, W. Wu and D. Pan, One-step electrospinning preparation of magnetic NZVI@TiO2 nanofibers for enhanced immobilization of U(VI) from aqueous solution, J. Radioanal. Nucl. Chem., 2023, 332(4), 1083–1091 CrossRef CAS.
- H. Hu, D. Zhao, C. Wu and R. Xie, Sulfidized Nanoscale Zerovalent Iron Supported by Oyster Powder for Efficient Removal of Cr (VI): Characterization, Performance, and Mechanisms, Materials, 2022, 15(11), 1–16, DOI:10.3390/ma15113898.
- S. Xie, J. Su, J. Zhao, H. Yang and H. Qian, An amorphous zero-valent iron decorated by Fe3O4 significantly improves the Fenton-like reaction, J. Alloys Compd., 2022, 929, 167306, DOI:10.1016/j.jallcom.2022.167306.
- M. Namakka, M. R. Rahman, K. A. M. B. Said, M. Abdul Mannan and A. M. Patwary, A review of nanoparticle synthesis methods, classifications, applications, and characterization, Environ. Nanotechnol. Monit. Manag., 2023, 20, 100900, DOI:10.1016/j.enmm.2023.100900.
- L. Liu, J. Zhao, X. Liu, S. Bai, H. Lin and D. Wang, Reduction and removal of As(V) in aqueous solution by biochar derived from nano zero-valent-iron (nZVI) and sewage sludge, Chemosphere, 2021, 277, 130273, DOI:10.1016/j.chemosphere.2021.130273.
- J. Hou, Y. Li, H. Ci, L. Miao, G. You and J. Wu, et al., Influence of aggregation and sedimentation behavior of bare and modified zero-valent-iron nanoparticles on the Cr(VI) removal under various groundwater chemistry conditions, Chemosphere, 2022, 296, 133905, DOI:10.1016/j.chemosphere.2022.133905.
- Y. P. Sun, X. Q. Li, J. Cao, W. X. Zhang and H. P. Wang, Characterization of zero-valent iron nanoparticles, Adv. Colloid Interface Sci., 2006, 120(1–3), 47–56 CrossRef CAS PubMed.
- M. M. El-shafei, A. Hamdy and M. M. Hefny, Zero-valent iron nanostructures: synthesis, characterization and application, J. Environ. Biotechnol. Res., 2018, 7, 1–10 Search PubMed.
- W. Li, R. Liang, A. Hu, Z. Huang and Y. N. Zhou, Generation of oxygen vacancies in visible light activated one-dimensional iodine TiO2 photocatalysts, RSC Adv., 2014, 4(70), 36959–36966, 10.1039/C4RA04768K.
- N. Prabhakar, A. M. Isloor, M. Padaki and A. Fauzi Ismail, Fabrication of TiO2@ZIF-67 metal organic framework composite incorporated PVDF membranes for the removal of hazardous reactive black 5 and Congo red dyes from contaminated water, Chem. Eng. J., 2024, 498, 155270, DOI:10.1016/j.cej.2024.155270.
- S. Kalidhasan and H. Y. Lee, Preparation of TiO2-deposited silica-based catalysts for photocatalytic decomposition of chloro-pesticide to environmentally less toxic species, Chemosphere, 2022, 290, 133300, DOI:10.1016/j.chemosphere.2021.133300.
- W. Biao, N. Awanis Hashim, M. F. B. Rabuni, O. Lide and A. Ullah, An innovative strategy for polyester microplastic fiber elimination from laundry wastewater via coupled separation and degradation using TiO2-based photocatalytic membrane reactor, Sep. Purif. Technol., 2025, 356, 129929, DOI:10.1016/j.seppur.2024.129929.
- C. G. Soria-Hernández, L. M. Palacios-Pineda, A. Elías-Zúñiga, I. A. Perales-Martínez and O. Martínez-Romero, Investigation of the effect of carbonyl iron micro-Particles on the mechanical and rheological properties of isotropic and anisotropic MREs: Constitutive magneto-mechanical material model, Polymers, 2019, 11(10), 2–22, DOI:10.3390/polym11101705.
- Y. Chen, X. F. Zhang, A. J. Wang, Q. L. Zhang, H. Huang and J. J. Feng, Ultrafine Fe3C nanoparticles embedded in N-doped graphitic carbon sheets for simultaneous determination of ascorbic acid, dopamine, uric acid and xanthine, Microchim. Acta, 2019, 186(9), 660, DOI:10.1007/s00604-019-3769-y.
- S. Mallakpour and M. Naghdi, Application of SiO2 nanoparticles with double layer coverage consist of citric acid and l(+)-ascorbic acid for the production of poly(vinyl chloride)/SiO2 nanocomposite films with enhanced optical and thermal properties, Polym. Bull., 2016, 73(6), 1701–1717 Search PubMed.
- S. H. Ammar, A. Ibrahim Elaibi and Sh. Mohammed I, Core/shell Fe3O4@Al2O3-PMo magnetic nanocatalyst for photocatalytic degradation of organic pollutants in an internal loop airlift reactor, J. Water Proc. Eng., 2020, 37, 101240, DOI:10.1016/j.jwpe.2020.101240.
- A. S. Astuti, M. Muldarisnur and U. S. R. A. Zulhadjri, Enhancement in photoluminescence performance of carbon-based Fe3O4@ZnO–C nanocomposites, Vacuum, 2023, 211, 111935, DOI:10.1016/j.vacuum.2023.111935.
- M. Yaghoobi, F. Asjadi and M. Sanikhani, A facile one-step green hydrothermal synthesis of paramagnetic Fe3O4 nanoparticles with highly efficient dye removal, J. Taiwan Inst. Chem. Eng., 2023, 144 Search PubMed.
- N. Lertthanaphol, P. Sereerattanakorn, S. Tulaphol, T. Maihom, T. K. Phung and G. Garbarino, et al., Selective conversion of aqueous sorbitol to sorbitan by amorphous Silica-Alumina catalysts, Chem. Eng. J., 2025, 511, 161918, DOI:10.1016/j.cej.2025.161918.
- A. Perzon and P. Ulvskov, Journal Pre of, Solid State Sci., 2019, 108117, DOI:10.1016/j.solidstatesciences.2025.108117.
- S. Madan, R. Shaw, S. Tiwari and S. K. Tiwari, Enhancing corrosion stability and shelf life of nanoscale zero-valent iron via encapsulation in porous Ze-TiO2 matrix: An interface for simultaneous oxidation and adsorption of As(III), Colloids Surf., A, 2020, 607, 125381, DOI:10.1016/j.colsurfa.2020.125381.
- Q. Li, H. Wang, Z. Chen, X. He, Y. Liu and M. Qiu, et al., Adsorption-reduction strategy of U(VI) on NZVI-supported zeolite composites via batch, visual and XPS techniques, J. Mol. Liq., 2021, 339, 116719, DOI:10.1016/j.molliq.2021.116719.
- X. Li, M. Gao, Y. Huo, H. Liu, J. Li and T. Huang, et al., Impacts of shell structure on nitrate-reduction activity and air stability of nanoscale zero-valent iron, Environ. Sci. Pollut. Res., 2022, 29(53), 80683–80692, DOI:10.1007/s11356-022-21460-y.
- L. Wang, C. Song, X. Jiang, T. Li and H. Wang, Mechanochemical synthesis of redox-active Fe-based nanocomposites for efficient Cr(VI) remediation in water and soil, Sep. Purif. Technol., 2025, 377, 134212, DOI:10.1016/j.seppur.2025.134212.
- L. Guo, Synthesis of Amorphous-Coated and Amorphous-Doped Nanomaterials, in Amorphous Nanomaterials, ed. L. Guo, 2021, DOI:10.1002/9783527826360.ch8.
- B. Alessi, M. Macias-Montero, C. Maddi, P. Maguire, V. Svrcek and D. Mariotti, Bridging energy bands to the crystalline and amorphous states of Si QDs, Faraday Discuss., 2020, 222, 390–404 Search PubMed.
- M. Kitabata, T. Taddese and S. Okazaki, Molecular Dynamics Study on Wettability of Poly(vinylidene fluoride) Crystalline and Amorphous Surfaces, Langmuir, 2018, 34(40), 12214–12223 CrossRef CAS PubMed.
- P. J. Tapia, J. Gallego and J. F. Espinal, Calcination Temperature Effect in Catalyst Reactivity for the CO SELOX Reaction Using Perovskite-like LaBO3 (B: Mn, Fe, Co, Ni) Oxides, Catal. Lett., 2021, 151(12), 3690–3703, DOI:10.1007/s10562-021-03601-z.
- S. Jain, J. Shah, N. S. Negi, C. Sharma and R. K. Kotnala, Significance of interface barrier at electrode of hematite hydroelectric cell for generating ecopower by water splitting, Int. J. Energy Res., 2019, 43(9), 4743–4755 Search PubMed.
- H. Wu, L. Zheng, J. Zhan, N. Du, W. Liu and J. Ma, et al., Recycling silicon-based industrial waste as sustainable sources of Si/SiO2 composites for high-performance Li-ion battery anodes, J. Power Sources, 2020, 449, 227513, DOI:10.1016/j.jpowsour.2019.227513.
- G. Greczynski and L. Hultman, C 1s Peak of Adventitious Carbon Aligns to the Vacuum Level: Dire Consequences for Material's Bonding Assignment by Photoelectron Spectroscopy, ChemPhysChem, 2017, 18(12), 1507–1512 Search PubMed.
- X-ray TTSK alpha. Investigating the oxidation of a cobalt-based catalyst using X-ray photoelectron spectroscopy.
- A. Bushell, P. Mack, E. Grinstead and W. Sussex, Identifying Structures on a Plasma-Modified Polymer Surface Authors, Thermo Fisher Scientific Inc., 2019, AN52333_E:3 Search PubMed.
- T. T. Scientific, Depth Profiling of an Organic FET with XPS and Argon Cluster Ions, Application Note, 2019 Search PubMed.
- Y. C. G. Kwan, G. M. Ng and C. H. A. Huan, Identification of functional groups and determination of carboxyl formation temperature in graphene oxide using the XPS O 1s spectrum, Thin Solid Films, 2015, 590, 40–48, DOI:10.1016/j.tsf.2015.07.051.
- G. Greczynski, D. Primetzhofer and L. Hultman, Reference binding energies of transition metal carbides by core-level x-ray photoelectron spectroscopy free from Ar + etching artefacts, Appl. Surf. Sci., 2018, 436, 102–110, DOI:10.1016/j.apsusc.2017.11.264.
- O. Wilhelmsson, S. Bijelovic, M. Lindquist, B. André, U. Wiklund and P. Svedlindh, et al., Deposition and characterization of magnetic Ti-Fe-C nanocomposite thin films, Thin Solid Films, 2010, 518(10), 2607–2616, DOI:10.1016/j.tsf.2009.07.195.
- X. Yu, L. Jiajun, Y. Xianglin, Z. Feng, L. Yongjun and L. Junbo, Preparation of graphdiyne-doped TiO2/SiO2 composite for enhanced photocatalytic activity, J. Nanopart. Res., 2020, 22(12), 365, DOI:10.1007/s11051-020-05097-x.
- S. Reghunath, D. Pinheiro and S. D. KR, A review of hierarchical nanostructures of TiO2: Advances and applications, Appl. Surf. Sci. Adv., 2021, 3, 100063, DOI:10.1016/j.apsadv.2021.100063.
- P. Yu, H. Yu, Q. Sun and B. Ma, Filter paper supported nZVI for continuous treatment of simulated dyeing wastewater, Sci. Rep., 2019, 9(1), 1–8 Search PubMed.
- S. S. Koçak, O. S. Özdemir, M. Asiltürk and İ. Atmaca, Thermal and Optical Characteristics of TiO2@SiO2, Fe3O4@SiO2, and ZnO@SiO2 Core–Shell Nanoparticles and Their Water-Based Nanofluids, Int. J. Thermophys., 2025, 46, 1–31, DOI:10.1007/s10765-025-03558-w.
- A. Anand, D. Meena, K. K. Dey and M. C. Bhatnagar, Enhanced piezoelectricity properties of reduced graphene oxide (RGO) loaded polyvinylidene fluoride (PVDF) nanocomposite films for nanogenerator application, J. Polym. Res., 2020, 27(12), 1–11 Search PubMed . https://link.springer.com/article/10.1007/s10965-020-02323-x.
- H. Zhang, Y. Zhu and L. Li, Fabrication of PVDF/graphene composites with enhanced β phase via conventional melt processing assisted by solid state shear milling technology, RSC Adv., 2020, 10(6), 3391–3401 RSC . https://pubs.rsc.org/en/content/articlehtml/2020/ra/c9ra09459h.
- A. Zaienah and S. Rezaur, Impact of titanium dioxide/graphene in polyvinylidene fluoride nanocomposite membrane to intensify methylene blue dye removal, Antifouling Performance , and Reusability, 2024, pp. 1–14 Search PubMed.
- N. Ismail, M. Essalhi, M. Rahmati, Z. Cui, M. Khayet and N. Tavajohi, Experimental and theoretical studies on the formation of pure β-phase polymorphs during fabrication of polyvinylidene fluoride membranes by cyclic carbonate solvents, Green Chem., 2021, 23(5), 2130–2147 Search PubMed.
- S. Acarer-Arat, M. Tüfekci, İ. Pir and N. Tüfekci, Nanocellulose in polyvinylidene fluoride (PVDF) membranes: assessing reinforcement impact and modelling techniques, J. Environ. Chem. Eng., 2024, 12(6), 114749 CrossRef CAS.
- W. Zhang, G. Wu, H. Zeng, Z. Li, W. Wu and H. Jiang, et al., The preparation, structural design and application of electroactive poly (vinylidene fluoride) - based materials for wearable sensors and human energy harvesters, Polymers, 2023, 15(13), 2766 Search PubMed.
- A. James, M. R. Rahman, S. K. A. B. Mohamad, M. Namakka, M. Shahabuddin and M. S. M. Al-Saleem, et al., Multifunctional PVDF membranes incorporating graphene, TiO2, and nanocellulose: synergistic effects on filtration and antifouling performance, RSC Adv., 2025, 15(38), 31471–31497 RSC.
- A. James, M. Rezaur Rahman, K. Anwar Mohamed Said, M. Namakka, K. Kuok Kuok and M. Uddin Khandaker, et al., Lithium Chloride-Mediated enhancement of dye removal capacity in Borneo bamboo derived nanocellulose-based nanocomposite membranes (NCMs), J. Mol. Liq., 2024, 413, 125973, DOI:10.1016/j.molliq.2024.125973.
- A. Ramazani, Z. Shaghaghi, H. Aghahosseini, P. A. Asiabi and S. W. Joo, Silica nanoparticles as a highly efficient catalyst for the onepot synthesis of sterically congested 2-(dibenzylamino)-2-Aryl acetamide derivatives from by phthaldehyde isomers, isocyanides and dibenzylamine, Bull. Chem. Soc. Ethiop., 2016, 30(3), 413–420 CrossRef CAS.
- M. Pyo, S. Jeong, J. H. Kim, M. J. Jeon and E. J. Lee, Hydrophobicity and membrane distillation performance of glass fiber membranes modified by dip coating of pure PDMS, J. Environ. Chem. Eng., 2024, 12(3), 112534, DOI:10.1016/j.jece.2024.112534.
- K. M. Jeong, Y. Li, D. G. Yoo, N. K. Lee, H. G. Lee and S. Ando, et al., Effects of crosslinking agents on the physical properties of polyimide/amino-functionalized graphene oxide hybrid films, Polym. Int., 2018, 67(5), 588–597 CrossRef CAS.
- J. Sun, H. Bi, S. Su, H. Jia, X. Xie and L. Sun, One-step preparation of GO/SiO2 membrane for highly efficient separation of oil-in-water emulsion, J. Membr. Sci., 2018, 553, 131–138 Search PubMed.
- H. A. Budiarti, R. N. Puspitasari, A. M. Hatta and R. D. D. Sekartedjo, Synthesis and Characterization of TiO2@SiO2 and SiO2@TiO2 Core-Shell Structure Using Lapindo Mud Extract via Sol-Gel Method, Procedia Eng., 2017, 170, 65–71 CrossRef CAS.
- R. Liu, H. Jiang, P. Li, J. Yin, Z. Xu and X. Chen, et al., Degradation mechanism of antibiotics by flower-like Bi2S3 sensitized anatase/rutile TiO2 coupled with SiO2 photonic crystals, J. Water Proc. Eng., 2024, 58, 104827, DOI:10.1016/j.jwpe.2024.104827.
- N. S. Fuzil, N. H. Othman, N. H. Alias, F. Marpani, M. S. Mat Shayuti and M. Z. Shahruddin, et al., MoS2-TiO2 coated PVDF-based hollow fiber membranes for permeate flux enhancement in membrane distillation, J. Environ. Chem. Eng., 2023, 11(3), 109866, DOI:10.1016/j.jece.2023.109866.
- M. Kazemi, M. Peyravi and M. Jahanshahi, Multilayer UF membrane assisted by photocatalytic NZVI@TiO2 nanoparticle for removal and reduction of hexavalent chromium, J. Water Proc. Eng., 2020, 37, 101183, DOI:10.1016/j.jwpe.2020.101183.
- G. C. Bleier, J. Watt, C. K. Simocko, J. M. Lavin and D. L. Huber, Reversible Magnetic Agglomeration – A Mechanism for Thermodynamic Control over Nanoparticle Size, Angew. Chem., Int. Ed., 2018, 57, 7678 CrossRef CAS PubMed.
- M. A. Ashraf, W. Peng and Y. Zare, Effects of Size and Aggregation/Agglomeration of Nanoparticles on the Interfacial/Interphase Properties and Tensile Strength of Polymer Nanocomposites, Nanoscale Res. Lett., 2018, 13, 214, DOI:10.1186/s11671-018-2624-0.
- B. Sutariya and S. Karan, A realistic approach for determining the pore size distribution of nanofiltration membranes, Sep. Purif. Technol., 2022, 293, 121096, DOI:10.1016/j.seppur.2022.121096.
- D. Wibowo, F. Mustapa, S. Selviantori, M. Idris, A. Mahmud and M. Maulidiyah, et al., CA/PEG/chitosan membrane incorporated with TiO2 nanoparticles for strengthening and permselectivity membrane for reverse osmosis desalination, Environ. Nanotechnol. Monit. Manag., 2023, 20, 100848, DOI:10.1016/j.enmm.2023.100848.
- T. M. H. Le, R. Wang and S. Sairiam, Self-protecting PVDF-PDA-TiO2 membranes towards highly efficient and prolonged dye wastewater treatment by photocatalytic membranes, J. Membr. Sci., 2023, 683, 121789, DOI:10.1016/j.memsci.2023.121789.
- M. R. Rahman, A. James, K. A. Mohamed Said, M. Namakka, M. U. Khandaker and W. H. Jiunn, et al., A TiO 2 grafted bamboo derivative nanocellulose polyvinylidene fluoride (PVDF) nanocomposite membrane for wastewater treatment by a photocatalytic process, Mater. Adv., 2024, 7617–7636 Search PubMed.
- F. Yan, H. Chen, Y. Lü, Z. Lü, S. Yu and M. Liu, et al., Improving the water permeability and antifouling property of thin-film composite polyamide nanofiltration membrane by modifying the active layer with triethanolamine, J. Membr. Sci., 2016, 513, 108–116 Search PubMed.
- P. G. Ingole, R. R. Pawar, M. I. Baig, J. D. Jeon and H. K. Lee, Thin film nanocomposite (TFN) hollow fiber membranes incorporated with functionalized acid-activated bentonite (ABn-NH) clay: Towards enhancement of water vapor permeance and selectivity, J. Mater. Chem. A, 2017, 5(39), 20947–20958 RSC.
- M. Shokouhian and S. Solouki, p-Phenylenediamine-grafted multi-walled carbon nanotubes as a hydrophilic modifier in thin-film nanocomposite membrane, Polym. Bull., 2020, 77(7), 3485–3498, DOI:10.1007/s00289-019-02899-5.
- L. L. S. Silva, J. A. Caldara, A. M. Rocco, C. P. Borges and F. V. Fonseca, Evaluation of nano zero-valent iron (nZVI) Activity in solution and immobilized in hydrophilic PVDF membrane for drimaren red X-6BN and bisphenol-a removal in water, Processes, 2019, 7(12), 904, DOI:10.3390/pr7120904.
- B. Ozbey-Unal, C. Balcik and B. Van der Bruggen, Development of a novel hydrophilic SiO2/PVDF Janus membrane via different modification methods for robust antiwetting and antifouling membrane distillation, J. Water Proc. Eng., 2023, 54, 104021, DOI:10.1016/j.jwpe.2023.104021.
- Q. Zhao, H. Yang, L. Tong and R. Jin, Pollution Characteristics of Pyrite Surface in Cyanide Tailings by PCA-Assisted ToF-SIMS and Their Correlation with the Contact Angle, Jom, 2023, 76(1), 547–557, DOI:10.1007/s11837-023-06143-4.
- M. R. Bilad, E. Guillen-Burrieza, M. O. Mavukkandy, F. A. Al Marzooqi and H. A. Arafat, Shrinkage, defect and membrane distillation performance of composite PVDF membranes, Desalination, 2015, 376, 62–72, DOI:10.1016/j.desal.2015.08.015.
- R. Ambedkar, G. Shanker and S. Singh, Investigation of photo-Fenton assisted antibiotic degradation using nZVI/Cu bimetallic heterostructures embedded PVDF film, Surf. Interfaces, 2025, 72, 107122, DOI:10.1016/j.surfin.2025.107122.
- J. Mun, H. M. Park, E. Koh and Y. T. Lee, Enhancement of the crystallinity and surface hydrophilicity of a PVDF hollow fiber membrane on simultaneous stretching and coating method, J. Ind. Eng. Chem., 2018, 65, 112–119, DOI:10.1016/j.jiec.2018.04.019.
- A. Siddiqa, H. Razzaq, S. Qaisar, S. Liaqat, M. Arshad and R. Gill, PVDF-Nanodiamonds Composite Membranes: Fabrication, Characterization and Water Treatment Applications, Der Pharma Chemica, 2017, 9(15), 32–40 Search PubMed.
- B. Hao, W. Jarman, H. Peng, K. Li, A. M. Khan and F. Russo, et al., High performance crosslinked polyvinylidene fluoride (PVDF) membranes for solvent permeation, Membranes, 2025, 209, 106174, DOI:10.1016/j.reactfunctpolym.2025.106174.
- M. Namakka, MdR. Rahman, M. Sa’don N bt, M. K. B. Bakri and A. Z. Sueraya, Chemisorption and physisorption of polymer nanocomposite membranes, Polymer Nanocomposite Membranes in Water Treatment and Desalination, 2025, pp. 423–446, https://www.sciencedirect.com/science/article/abs/pii/B9780443238796000117 Search PubMed.
- A. Z. Sueraya, MdR. Rahman, N. Sa’don M. bt, A. Anak James, M. K. B. Bakri and M. Namakka, Polymer nanocomposite membranes for dye removal, Polymer Nanocomposite Membranes in Water Treatment and Desalination, 2025, pp. 187–207, https://www.sciencedirect.com/science/article/abs/pii/B9780443238796000063 Search PubMed.
- N. Hamzah, C. P. Leo and B. S. Ooi, Superhydrophobic PVDF/TiO2-SiO2 Membrane with Hierarchical Roughness in Membrane Distillation for Water Recovery from Phenolic Rich Solution Containing Surfactant, Chin. J. Polym. Sci., 2019, 37(6), 609–616 CrossRef CAS.
- J. Gong, B. Xiang, R. Jin, Z. Li, W. Liu and J. Li, Robust peony-like Cu3(PO4)2/UiO-66-NH2/PVA membranes with exceptional anti-fouling performance for high flux emulsion separation, Sep. Purif. Technol., 2025, 379, 134915, DOI:10.1016/j.seppur.2025.134915.
- K. Xiao, J. Sun, Y. Mo, Z. Fang, P. Liang and X. Huang, et al., Effect of membrane pore morphology on microfiltration organic fouling: PTFE/PVDF blend membranes compared with PVDF membranes, Desalination, 2014, 343, 217–225, DOI:10.1016/j.desal.2013.09.026.
- Q. Ma, H. Zhang, X. Deng, Y. Cui, X. Cheng and X. Li, et al., Electrochemical fabrication of NZVI/TiO2 nano-tube arrays photoelectrode and its enhanced visible light photocatalytic performance and mechanism for degradation of 4-chlorphenol, Sep. Purif. Technol., 2017, 182, 144–150 Search PubMed.
- M. Namakka, MdR. Rahman, N. Sa’don M. bt, M. K. B. Bakri and A. Z. Sueraya, Polymer Nanocomposite Membranes in Water Treatment and Desalination, Recent Developments, Future Opportunities, and Sustainable Applications, Woodhead Publishing in Materials, 2025, https://www.sciencedirect.com/science/article/abs/pii/B9780443238796000117 Search PubMed.
- A. Z. Sueraya, M. R. Rahman, K. A. B. M. Said, M. Namakka, D. Kanakaraju and J. Y. Al-Humaidi, et al., Impact of titanium dioxide/graphene in polyvinylidene fluoride nanocomposite membrane to intensify methylene blue dye removal, antifouling performance, and reusability, J. Appl. Polym. Sci., 2024, 1–14 Search PubMed.
- F. G. Ávila, J. Cabrera-Sumba, S. Valdez-Pilataxi, J. Villalta-Chungata, L. Valdiviezo-Gonzales and C. Alegria-Arnedo, Removal of heavy metals in industrial wastewater using adsorption technology: Efficiency and influencing factors, Clean Eng. Technol., 2025, 24, 100879, DOI:10.1016/j.clet.2025.100879.
- A. Kagalkar, S. Dharaskar, N. Chaudhari, V. Vakharia and R. R. Karri, Enhanced metal ion adsorption using ZnO-MXene nanocomposites with machine learning-based performance prediction, Sci. Rep., 2025, 15(1), 1–26 Search PubMed.
- M. Alimohammady, M. Jahangiri, M. Salavati-Niasari and A. M. Aljeboree, Heavy metal adsorption by graphene oxide modified with 5-amino-3(2-thienyl)pyrazole using central composite design/response surface methodology (CCD/RSM), RSC Adv., 2025, 15(44), 36837–36860 RSC.
- N. S. Mohamed, M. Abbas and M. Trari, Understanding the rate-limiting step adsorption kinetics onto biomaterials for mechanism adsorption control, Prog. React. Kinet. Mech., 2024, 49, 1–26 Search PubMed.
| This journal is © The Royal Society of Chemistry 2026 |