 Open Access Article
Open Access ArticleSequential determination of calcium chemical phases in gypsum-associated fluorite ores using ICP-OES
Xiao Wangab,
Na Guo *ab,
Liming Ganab,
Jiuffen Liuc,
Xin Weiab and
Tao Heab
*ab,
Liming Ganab,
Jiuffen Liuc,
Xin Weiab and
Tao Heab
aXi'an Mineral Resources Survey, China Geological Survey, Xi'an, China. E-mail: 380403034@qq.com
bTechnology Innovation Center for Gold Ore Exploration, China Geological Survey, Xi'an, China
cNatural Resources Survey, China Geological Survey, Beijing, China
First published on 20th January 2026
Abstract
Fluorite is a crucial non-metallic strategic mineral resource, commonly occurring in hydrothermal vein-type and sedimentary metamorphic fluorite deposits. Sedimentary metamorphic fluorite (CaF2) frequently coexists with associated minerals, such as gypsum (CaSO4·2H2O) and calcite (CaCO3). Accurately determining the contents of calcium sulphate, calcium carbonate, and calcium fluoride is essential for understanding mineralization mechanisms and for guiding the development of subsequent beneficiation and metallurgical processes. Existing analytical methods for fluorite are primarily developed for single-type deposits. They generally focus on the determination of calcium carbonate and calcium fluoride while not accounting for the calcium sulphate phase. Consequently, their applicability is somewhat constrained, and they may encounter challenges when applied to complex fluorite ore systems containing gypsum. In this study, sodium chloride was employed to separate calcium sulphate, hydrochloric acid was used to isolate calcium carbonate, and a boric acid–hydrochloric acid mixture was applied to dissolve calcium fluoride from the residue. An analytical method was developed for the sequential determination of calcium sulphate, calcium carbonate, and calcium fluoride in gypsum-associated fluorite ores by inductively coupled plasma optical emission spectrometry (ICP-OES). The separation and determination conditions for each phase were systematically investigated. Three gypsum-associated fluorite samples were analyzed in parallel according to the proposed method, yielding relative standard deviations (RSD, n = 7) of ≤4.84% for all three phases. The standard addition method was applied for recovery testing, with recovery rates ranging from 90.5% to 98.0%. This method enables the separation and continuous determination of calcium sulphate, calcium carbonate, and calcium fluoride phases. With a straightforward process and efficient operation, it saves time, labor, and costs, thereby significantly enhancing analytical efficiency. The technique is particularly applicable for phase analysis of calcium in gypsum-associated fluorite ores.
1. Introduction
Fluorite is a critical non-metallic strategic mineral resource, often referred to as the “second rare earth”. It is widely utilized in traditional industries such as fluorine chemistry, metallurgy, and construction materials, as well as in strategic emerging industries, including new energy and advanced material.1–4 Due to its significance, fluorite has been classified as a strategic resource by numerous countries, including the United States, China, the European Union, and Iran.5–7 Fluorite deposits are predominantly categorized into hydrothermal vein and sedimentary metamorphic types. Sedimentary metamorphic fluorite (CaF2) frequently occurs in association with minerals such as gypsum (CaSO4·2H2O) and calcite (CaCO3), resulting in a complex mineral composition and relatively low overall utilization efficiency.8–12 The primary calcium-bearing phases in gypsum-associated fluorite ore include calcium sulphate, calcium carbonate, and calcium fluoride. Calcium sulphate (CaSO4) is a typica deleterious gangue mineral in the beneficiation of fluorite (CaF2). Its primary effect is to impair the separation efficiency between fluorite and gangue minerals, thereby directly reducing the quality of fluorite concentrate and beneficiation recovery. Calcium carbonate (CaCO3) is also one of the most common harmful gangue minerals in fluorite (CaF2) beneficiation. The primary challenge lies in its highly similar floatability characteristics relative to fluorite, which hinders effective separation between the two minerals. This difficulty ultimately compromises the purity of the fluorite concentrate and diminishes the overall economic efficiency of the beneficiation process. The rapid and accurate determination of calcium's chemical phase composition is critical for both advancing theoretical studies on fluorite mineralization and optimizing beneficiation and metallurgical processes.13–17At present, the methodologies employed for chemical phase analysis encompass volumetric analysis,18,19 X-ray fluorescence spectrometry (XRF),20–24 X-ray diffraction (XRD),25,26 inductively coupled plasma optical emission spectrometry (ICP-OES),27,28 and gas chromatography,29,30 and others. For the chemical phase analysis of fluorite, XRD is a convenient method but suffers from low detection sensitivity and micro-absorption effects, and is limited by the sensitivity of crystallinity and preferred orientation-interfering with intensity calculation in quantitative analysis. The existing wet-chemical approaches are restricted to single-type fluorite analysis techniques, which only account for the phase states of calcium carbonate and calcium fluoride. The traditional method employs acetic acid to separate calcium carbonate. Subsequently, EDTA titration is used for the quantitative determination of calcium carbonate and calcium fluoride. However, this method has several limitations. These include a cumbersome operational procedure and difficulty in identifying the titration endpoint. Both of these factors may compromise the accuracy of the analytical results.31–33 The XRF method also uses acetic acid containing calcium to separate calcium carbonate. After that, the residue is fused and sampled to determine calcium fluoride. Although this method is relatively easy to perform, it is only suitable for the analysis of single-phase calcium fluoride.34 The ICP-OES method likewise uses acetic acid to separate calcium carbonate. The remaining residue after filtration is extracted with an aluminum trichloride solution. Then, the concentrations of calcium carbonate and calcium fluoride are determined from the resulting extract.35,36 Due to the coexistence of calcium sulphate in gypsum–fluorite ore, the core challenge in the chemical phase analysis of calcium is the effective separation of calcium sulphate, calcium carbonate, and calcium fluoride within the sample. Consequently, the aforementioned methods are not suitable, and there is an urgent need to establish a continuous and reliable analytical method for determining the chemical phases of calcium sulphate, calcium carbonate, and calcium fluoride in gypsum-associated fluorite ore.
Based on previous research, this study concentrated on the separation system of calcium sulphate, calcium carbonate, and calcium fluoride in gypsum-associated fluorite ore. Calcium sulphate was dissolved using sodium chloride and separated by filtration from the other calcium compounds. Subsequently, calcium carbonate was dissolved with hydrochloric acid in the presence of calcium ions and separated from calcium fluoride by filtration. Finally, the remaining calcium fluoride in the residue was dissolved using a mixture of boric acid and hydrochloric acid. By combining this separation procedure with ICP-OES analysis, a method was developed for determining the chemical phases of calcium in gypsum-associated fluorite ore. This approach achieved the complete separation and sequential determination of calcium sulphate, calcium carbonate, and calcium fluoride. The deviation between the calculated sum of calcium from individual phases and the measured total calcium content ranged from −3.17% to 0.50%, satisfying both the principle of mass balance and the requirements for analytical accuracy. The recovery rates for each phase demonstrated good accuracy. In comparison with the conventional approach involving separate isolation followed by EDTA volumetric determination of calcium, this procedure offers enhanced simplicity, yielding accurate and reliable analytical results. It is particularly suitable for the chemical phase analysis of calcium sulphate, calcium carbonate, and calcium fluoride in fluorite ores containing gypsum.
2. Material and methods
2.1. Instrument and reagents
The ICP-OES (iCAP-PRO, PerkinElmer, USA) was used under the following operating conditions: a power range of 600 to 1600 W, a power stability of 0.1%. Gas flow is precisely controlled using a high-precision mass flowmeter. The experimental parameters were optimized based on signal-to-background ratio comparisons, and the optimized operating conditions of the instrument are summarized in Table 1.| Item | Parameter |
|---|---|
| Power of RF | 1150 W |
| Speed of flush pump | 75 rpm |
| Gas flow of auxiliary | 0.5 L min−1 |
| Gas flow of atomizer | 0.60 L min−1 |
| Test number | 3 |
| Washing time | 25 s |
| Atomizer | Salt-tolerant |
| Determination wavelength of Ca | 315.886 nm |
The analytical balance (BS124S, Sartoris, Beijing) with a sensitivity of 0.01 mg was used. The ultrapure water system (MILLI-Q ADVANTRGE A10) with a resistivity of 18.25 MΩ cm was used.
Sodium chloride: analytical grade, used for dissolving calcium sulphate.
Hydrochloric acid: analytical grade, used for dissolving calcium carbonate.
Boric acid: analytical grade, used for dissolving calcium fluoride.
Calcium standard stock solution: 1000 mg L−1, used for the preparation of the standard curve.
Calcium sulphate standard working solution: pipette varying volumes of the calcium standard stock solution into 100 mL volumetric flasks. Add 4.0 mL of a 0.20 g per mL sodium chloride solution to each flask, and dilute to the mark with deionized water. This yields solutions with calcium mass concentrations of 0.00, 1.00, 5.00, 10.0, 20.0, 50.0, and 100.0 mg L−1.
Calcium carbonate standard working solution: pipette varying volumes of the calcium standard stock solution into 100 mL volumetric flasks. Add 2.0 mL of 10% (v/v) hydrochloric acid solution to each flask, and dilute to the mark with deionized water. This yields solutions with calcium mass concentrations of 0.00, 1.00, 5.00, 10.0, 20.0, 50.0, and 100.0 mg L−1.
Calcium fluoride standard working solution: pipette varying volumes of the calcium standard stock solution into 100 mL volumetric flasks. Add 1.0 mL of a 0.040 g per mL boric acid solution to each flask, and dilute to the mark with deionized water. This yields solutions with calcium mass concentrations of 0.00, 1.00, 5.00, 10.0, 20.0, 50.0, and 100.0 mg L−1.
2.2. Experimental principle
The samples were dissolved using sodium chloride to extract calcium sulphate, hydrochloric acid to dissolve calcium carbonate, and a boric acid–hydrochloric acid mixture to dissolve calcium fluoride. The three filtrate phases were analyzed for calcium content using ICP-OES. Based on the measured calcium concentrations in each phase, the corresponding concentrations of calcium sulphate, calcium carbonate, and calcium fluoride were calculated. A flow chart summarizing the methodology is shown in Fig. 1.2.3. Experimental methods
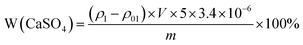 | (1) |
In the formula, W(CaSO4) denotes the mass percentage of calcium sulphate in the sample, ρ1 represents the measured Ca concentration in the sample (mg L−1), ρ01 is the blank measurement from the sodium chloride solution (mg L−1), V is the dilution volume (L), m is the sample mass (g), 5 is the dilution factor, and 3.4 is the conversion factor for converting Ca into CaSO4.
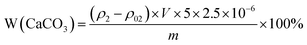 | (2) |
In the formula, W(CaCO3) denotes the mass percentage of calcium carbonate in the sample, ρ2 represents the measured Ca concentration in the sample (mg L−1), ρ02 is the blank measurement from the hydrochloric acid solution (mg L−1), V is the dilution volume (L), m is the sample mass (g), 5 is the dilution factor, and 2.5 is the conversion factor for converting Ca into CaCO3.
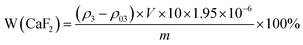 | (3) |
In the formula, W(CaF2) denotes the mass percentage of calcium fluoride in the sample, ρ3 represents the measured Ca concentration in the sample (mg L−1), ρ03 is the blank measurement from the boric acid and hydrochloric acid solution (mg L−1), V is the dilution volume (L), m is the sample mass (g), 10 is the dilution factor, and 1.95 is the conversion factor for converting Ca into CaF2.
3. Results and discussion
3.1. Selection of phase separation conditions for calcium sulphate
Sodium chloride is a symmetrical electrolyte, and a high concentration of sodium chloride solution can significantly increase the ionic strength of the solution. Due to the significantly higher solubility product constant (Ksp) of calcium sulphate (Ksp: 9.1 × 10−6) compared to calcium fluoride (Ksp: 2.7 × 10−11) and calcium carbonate (Ksp: 2.8 × 10−9), a concentrated sodium chloride solution can effectively promote the dissolution of calcium sulphate, whereas the dissolution of calcium carbonate and calcium fluoride remains limited. When dissolving calcium sulphate in NaCl solution, calcium sulphate dihydrate (CaSO4·2H2O) gradually transforms into anhydrous calcium sulphate (CaSO4) at temperatures exceeding 40 °C. The solubility of anhydrous CaSO4 decreases sharply with rising temperature. Meanwhile, at high NaCl concentrations, elevated temperature accelerates the formation of calcium sulphate ion pairs, enhancing short-range electrostatic interactions and further reducing solubility with increasing temperature. Thus, dissolving CaSO4 at room temperature is more effective.37,38 Experiments optimized NaCl concentration and dissolution time using pure mineral powders of gypsum (CaSO4·2H2O), calcite (CaCO3), and fluorite (CaF2). | ||
| Fig. 2 The dissolution rate of calcium sulphate, calcium carbonate, and calcium fluoride in varying concentrations of sodium chloride. | ||
Experimental results reveal that the dissolution rate of calcium sulphate exhibits a characteristic trend: it initially increases and subsequently decreases with the increasing concentration of sodium chloride. The maximum dissolution rate of calcium sulphate is observed at a sodium chloride concentration of 0.20 g mL−1, at which point calcium sulphate is nearly completely dissolved. These findings are consistent with the predictions of the Debye–Hückel theory. At low ionic concentrations, an increase in sodium chloride concentration leads to an increase in ionic strength and a decrease in the activity coefficient, thereby enhancing the dissolution rate of calcium sulphate. However, at higher salt concentrations, calcium sulphate tends to form ion pairs, and the enhanced short-range electrostatic interactions result in a reduction of its solubility. In contrast, the dissolution rates of calcium carbonate and calcium fluoride in sodium chloride solution are extremely low and can be considered negligible. Based on these observations, it is recommended to use a 20 mL volume of 0.20 g per mL sodium chloride solution for the effective dissolution of calcium sulphate.
The dissolution efficiency of calcium sulphate gradually increases with the extension of the dissolution time. When the dissolution duration exceeds 40 minutes, the substance nearly reaches complete dissolution. Consequently, 40 minutes was selected as the optimal dissolution duration.
3.2. Selection of phase separation conditions for calcium carbonate
Hydrochloric acid can effectively dissolve calcium carbonate in dolomite and calcite.34,39 Under heating, a 10% hydrochloric acid solution effectively dissolves the calcium carbonate phase, thereby enabling its separation from the calcium fluoride phase. The experimental parameters, such as hydrochloric acid concentration and dissolution time, were optimized accordingly.The results indicate that the dissolution rate of calcium carbonate gradually increases with rising hydrochloric acid concentration. When the concentration reaches or exceeds 10% (v/v), calcium carbonate is nearly completely dissolved. At this concentration, the dissolution rate of calcium fluoride remains very low and can be considered negligible. Therefore, 10 mL of 10% (v/v) hydrochloric acid is used to effectively dissolve calcium carbonate.
The results show that the measured calcium carbonate content gradually stabilizes with increasing dissolution time. When the dissolution time exceeds 30 minutes, calcium carbonate is nearly completely dissolved. Therefore, 30 minutes is selected as the optimal dissolution time.
3.3. Selection of phase separation conditions for calcium fluoride
Boric acid and hydrochloric acid can effectively dissolve calcium fluoride under heating conditions,40 converting it into calcium chloride, which has a higher solubility. The corresponding chemical reaction is shown in eqn (4):| 2CaF2 + 4HCl + H3BO3 → 2CaCl2 + HBO4 + 3H2O | (4) |
The experimental conditions, including boric acid concentration and dissolution time, were optimized.
 | ||
| Fig. 6 Dissolution rate of calcium carbonate and calcium fluoride in varying concentrations of boric acid. | ||
It can be observed from the results that the dissolution rate of calcium fluoride gradually increases with the increasing concentration of boric acid. When the concentration of boric acid reaches or exceeds 0.040 g mL−1, calcium fluoride is nearly completely dissolved. Therefore, a boric acid concentration of 0.040 g mL−1 is recommended for the effective dissolution of calcium fluoride.
The results indicate that the measured calcium fluoride content gradually stabilizes with increasing contact time with boric acid and hydrochloric acid. Nearly complete dissolution is achieved when the dissolution duration exceeds 30 minutes. Therefore, 30 minutes was selected as the optimal dissolution duration.
3.4. Calibration curve and determination of detection limit
In accordance with the “Conformity assessment-Guidance on validation and verification of chemical analytical methods” (GB/T 27417-2017), the method detection limit was determined. Eleven blank samples were prepared using a mixture of low-grade calcium sulphate, calcium carbonate, and calcium fluoride, as specified in Section 2.3 Experimental methods, and analyzed by ICP-OES. The standard deviations (S) of the blank measurements for calcium sulphate, calcium carbonate, and calcium fluoride were calculated. Limit of detection (LOD) is determined according to eqn (5).| LOD = t(n−1,0.99) × S | (5) |
The detection limits were determined by multiplying the standard deviations by a factor of 3. The lower limits of quantification were obtained by multiplying the detection limits by 4, thus establishing the quantification range for each compound. The resulting detection limits for calcium sulphate, calcium carbonate, and calcium fluoride are presented in Table 2. The obtained detection limits and quantification ranges satisfy the analytical requirements for the determination of chemical phases in gypsum-bearing fluorite ore.
| Elements | Phase state transformation | Detection limit (%) | Detection ranges (%) | Correlation coefficient (R) |
|---|---|---|---|---|
| Ca | CaSO4 | 0.0092 | 0.0367–85.00 | 0.9991 |
| CaCO3 | 0.0061 | 0.0245–62.50 | ||
| CaF2 | 0.0049 | 0.0198–97.50 |
3.5. Precision of the method
Three representative samples of gypsum-bearing fluorite ore were selected, with calcium sulphate content ranging from 8.52% to 29.79%, calcium carbonate from 1.76% to 10.00%, and calcium fluoride from 15.41% to 39.25%. According to the experimental procedure, the contents of calcium sulphate, calcium carbonate, and calcium fluoride were measured in parallel seven times. The results are presented in Table 3. The mean bias across all phase states does not exceed 1.83%. The relative standard deviations (RSDs) for all phases were ≤4.84%, demonstrating satisfactory precision.| Sample | Chemical phase state | Measured value (%) | Average value (%) | Mean bias (%) | RSD (%) |
|---|---|---|---|---|---|
| XS1 | Calcium sulphate phase | 8.43, 8.57, 8.47, 8.81, 8.26, 8.64, 8.47 | 8.52 | 0.90 | 2.02 |
| Calcium carbonate phase | 1.78, 1.80, 1.60, 1.83, 1.75, 1.70, 1.85 | 1.76 | 0.44 | 4.84 | |
| Calcium fluoride phase | 39.51, 38.96, 39.23, 39.64, 38.88, 39.43, 39.12 | 39.25 | 1.63 | 0.73 | |
| XS2 | Calcium sulphate phase | 30.23, 29.65, 30.02, 29.34, 29.95, 29.89, 29.48 | 29.79 | 1.83 | 1.06 |
| Calcium carbonate phase | 6.35, 6.05, 6.08, 6.28, 6.05, 6.40, 6.00 | 6.17 | 1.02 | 2.67 | |
| Calcium fluoride phase | 15.46, 15.52, 15.44, 15.37, 15.54, 15.23, 15.27 | 15.41 | 0.70 | 0.79 | |
| XS3 | Calcium sulphate phase | 16.73, 16.46, 16.76, 16.86, 16.39, 16.59, 16.69 | 16.64 | 0.97 | 1.03 |
| Calcium carbonate phase | 9.95, 10.13, 9.83, 10.03, 10.15, 10.03, 9.88 | 10.00 | 0.68 | 1.21 | |
| Calcium fluoride phase | 26.15, 26.23, 26.44, 26.15, 26.44, 26.33, 26.05 | 26.26 | 0.89 | 0.58 |
3.6. Accuracy of the method
Due to the lack of standard reference materials for gypsum-synthesized fluorite phase analysis, three representative samples were selected. Spiked recovery experiments were performed on calcium sulphate, calcium carbonate, and calcium fluoride to assess the analytical accuracy of the method.Two aliquots of each XS1–XS3 sample were accurately weighed and analyzed separately. Following the established experimental procedure, one aliquot was used for the determination of calcium sulphate, calcium carbonate, and calcium fluoride. The second aliquot was spiked with 0.02 g of pure gypsum (calcium sulphate), 0.01 g of calcite (calcium carbonate), and 0.04 g of fluorite (calcium fluoride). The concentrations of each phase in the spiked samples were subsequently determined, and the corresponding recovery rates were calculated. As shown in Table 4, the recovery rates ranged from 90.5% to 98.0%, indicating good analytical accuracy. Simultaneously, the total calcium content in the actual samples of XS1–XS3 was determined. The results were compared with the sum of the calcium contents obtained from the three sequential phases. The results are presented in Table 5. The deviation between the calculated sum of calcium from individual phases and the measured total calcium content ranged from −3.17% to 0.50%, satisfying both the principle of mass balance and the requirements for analytical accuracy.
| Sample | Chemical phase state | Content of fluorine (mg) | Recovery (%) | ||
|---|---|---|---|---|---|
| Measured | Added | Measured after added | |||
| XS1 | Calcium sulphate phase | 16.9 | 20 | 35.6 | 93.5 |
| Calcium carbonate phase | 3.56 | 10 | 12.9 | 93.4 | |
| Calcium fluoride phase | 78.1 | 40 | 115 | 92.3 | |
| XS2 | Calcium sulphate phase | 60.2 | 20 | 78.3 | 90.5 |
| Calcium carbonate phase | 11.8 | 10 | 21.6 | 98.0 | |
| Calcium fluoride phase | 31.2 | 40 | 70.3 | 97.8 | |
| XS3 | Calcium sulphate phase | 32.3 | 20 | 51.5 | 96.0 |
| Calcium carbonate phase | 20.8 | 10 | 29.9 | 91.0 | |
| Calcium fluoride phase | 52.4 | 40 | 91.6 | 91.0 | |
| Sample | Total calcium (%) | Calcium content in each chemical phase (%) | Deviation (%) | |||
|---|---|---|---|---|---|---|
| Calcium sulphate phase | Calcium carbonate phase | Calcium fluoride phase | Sum of calcium content in each phase state | |||
| XS1 | 23.88 | 2.51 | 0.704 | 20.14 | 23.35 | −2.22 |
| XS2 | 19.87 | 8.83 | 2.46 | 7.95 | 19.24 | −3.17 |
| XS3 | 22.18 | 4.90 | 3.96 | 13.43 | 22.29 | 0.50 |
4. Conclusions
In this study, a chemical separation technique was employed to systematically isolate calcium sulphate, calcium carbonate, and calcium fluoride phases present in gypsum-symbiotic fluorite ore. By combining this separation strategy with ICP-OES a method was developed for the sequential determination of calcium chemical phases in gypsum-associated fluorite ores. The results demonstrated good accuracy, reliability. The recovery rates for each phase ranged from 90.5% to 98.0%, demonstrating good reproducibility. This approach effectively addresses the lack of suitable methodologies for chemical phase analysis of gypsum-symbiotic fluorite ores. Compared to XRD, this method features a lower detection limit, delivers accurate quantitative results for each phase, and minimizes reliance on model-dependent assumptions. Compared to traditional EDTA titration, this method effectively integrates separation techniques with ICP-OES, leveraging its ease of operation, high sensitivity, and wide linear range. Moreover, it provides a valuable reference for ICP-OES application in the chemical phase analysis of other minerals. The primary limitation of this method is that the presence of apatite in associated fluorite deposits may lead to elevated measured values for calcium sulphate and calcium carbonate, resulting in an overestimation of their concentrations. This method is specifically applicable to the chemical phase analysis of calcium sulphate, calcium carbonate, and calcium fluoride in fluorite deposits associated with gypsum.Author contributions
Xiao Wang: conceptualization, formal analysis, writing-original draft. Liming Gan: conceptualization, methodology. Jiufeng Liu: validation, visualization, formal analysis. Xin Wei: validation, visualization. Tao He: investigation, formal analysis. Na Guo: conceptualization, methodology, writing-review & editing, supervision.Conflicts of interest
There are no conflicts to declare.Data availability
The data supporting the findings of this study are openly available in the manuscript and its supplementary materials. Raw ICP-OES analytical data are deposited in the Mendeley Data repository (https://doi.org/10.17632/4j8993h9pf.1). Supplementary information (SI) is available. See DOI: https://doi.org/10.1039/d5ra07208e.Acknowledgements
This work was supported by the China Geological Survey project (DD20251126 and DD20251103).Notes and references
- M. Cherai, L. Rddad, F. Talbi and E. J. M. Carranza, Model. Earth Syst. Env., 2023, 9, 3135–3150 CrossRef.
- W. Li, C. Long, A. Shang, C. Song, A.-H. I. MOurad and A. M. M. Ibrahim, Opt Laser. Technol., 2025, 189, 113123 Search PubMed.
- Е. А. Bogdanova, V. M. Skachkov and К. V. Nefedova, Phys. Chem. Aspects Study Clusters, Nanostruct. Nanomater., 2024, 16, 779–793 Search PubMed.
- Y. M. Fedorchuk, L. A. Leonova, E. V. Solodov and E. A. Guba, Izv. Akad. Nauk SSSR, Ser. Khim., 2024, 335, 244–252 Search PubMed.
- Z. Gao, C. Zhang and B. McFadzean, Minerals, 2025, 15, 679 CrossRef CAS.
- S. Masoudi, E. Ezzati, N. Rashidnejad-Omran and A. Moradzadeh, Resour. Policy, 2017, 52, 100–106 CrossRef.
- A. Wang and X. Yuan, Bull. Chin. Acad. Sci., 2022, 37, 1550–1559 Search PubMed.
- X. Xiong, Technical Requirements for Evaluating the Potential of Important Chemical and Mineral Resources, Geological Publishing House, Beijing, 2010 Search PubMed.
- D. Zhang, J. Kang, H. Huang, W. Zhu and R. Wang, Chin. J. Process Eng., 2023, 23, 1–14 CAS.
- M. Dahhou, A. El Hamidi, M. El Moussaouiti and M. A. Arshad, Materialia, 2021, 20, 101204 CrossRef CAS.
- Y. Li, H. Zou, H. Liu, X. Jiang and D. Li, Rock Miner. Anal., 2020, 39, 300–310 CAS.
- T. U. Schlegel, T. Wagner and T. Fusswinkel, Chem. Geol., 2020, 548, 119674 CrossRef CAS.
- L. Li, T. Liu, Z. Yuan and C. Zhang, Conserv. Util. Miner. Resour., 2015, 6, 46–53 Search PubMed.
- Z. Gao, C. Wang, W. Sun, Y. Gao and P. B. Kowalczuk, Adv. Colloid Interface Sci., 2021, 290, 102382 Search PubMed.
- Q. Zhao, L. Mo, L. Hu and M. Xu, J. Ind. Eng. Chem., 2025, 146, 378–390 Search PubMed.
- Y. Wang, X. Zhang, J. Zhang and W. Zhao, Min. Metall. Explor., 2022, 39, 855–862 Search PubMed.
- P. Liu, Y. Tian, C. Zhang, M. Tian and W. Sun, Processes, 2025, 13, 1609 CrossRef CAS.
- H. Lu, D. Xie, Q. CUI and X. Yang, Metall. Anal., 2023, 43, 86–93 CAS.
- B. Huang, Z. zhang, L. Yang, J. Gao, Y. Li and S. Chen, Chin. J. Inorg. Anal. Chem., 2011, 1, 6–12 CAS.
- M. Petric, R. Bohinc, K. Bučar, M. Žitnik, J. Szlachetko and M. Kavčič, Anal. Chem., 2015, 87, 5632–5639 CrossRef CAS PubMed.
- J. Kawai, X-Ray Spectroscopy for Chemical State Analysis, Springer, 2023 Search PubMed.
- J. Aosong, W. Longhua and L. Zhu, Rock Miner. Anal., 2024, 43, 659–675 Search PubMed.
- A. Ohbuchi, K. Otori, K. Hagiwara, Y. Koike and T. Nakamura, ISIJ Int., 2021, 61, 2249–2255 CrossRef CAS.
- J. Yuan, Rock Miner. Anal., 2007, 26, 419–420 CAS.
- M. Zhang, Y. Cai, L. Xiao and L. Wang, Rock Miner. Anal., 2023, 42, 513–522 CAS.
- Y. Li, F. Sun, Y. Zhou and L. Zeng, J. Spectrosc., 2015, 1, 859365 Search PubMed.
- X. Wang, C. Chen, L. Feng, T. Jin, N. Chen and W. Fang, Metall. Anal., 2024, 44, 89–96 CAS.
- Z. Feng, L. Kuai and H. Shao, Metall. Anal., 2024, 44, 59–64 CAS.
- Z. Fan, J. Zhao, X. Liu, B. Luo, L. Zhou, D. Nie, Y. Wu, Z. Kang and W. Tao, J. Chromatogr. A, 2022, 1683, 463547 CrossRef CAS PubMed.
- H. Zhu, H. Zheng, T. Zhang, Q. He, H. Li, T. Yang, B. Yue, Y. Luo, X. Wang and J. Xie, J. Chromatogr. A, 2025, 1741, 465616 CrossRef CAS PubMed.
- W. Chen and S. Zhan, Guangdong Chem. Ind., 2022, 37, 1550–1559 Search PubMed.
- SAC, Fluorspar—Determination of Calcium Fluoride Content—EDTA Titration Method and Distillation-Potentiometric Titration Method, Standards Press of China, Beijing, 2017 Search PubMed.
- C. Xia, X. Tian, Z. Wang, M. Chen, Z. Qiu and W. Zhao, Chin. J. Inorg. Anal. Chem., 2023, 13, 813–824 CAS.
- Y. Yuan, F. Yang, J. Wu, B. Li, G. Fang and M. Wen, Metall. Anal., 2023, 43, 34–39 CAS.
- L. A, P. Wu, M. Mao, P. He, L. Ma and Y. Sun, Metall. Anal., 2022, 42, 32–38 Search PubMed.
- Y. Zhang, X. Hao, S. Liu, Z. Tian, Y. Dong and Y. Jiao, Metall. Anal., 2023, 43, 83–89 Search PubMed.
- Q. Wang, L. Zhang, J. Hu and X. Shi, Chem. Geol., 2024, 650, 122001 CrossRef CAS.
- N. S. Kistanova, A. D. Chashchukhina, O. S. Kudryashova and E. A. Khayrulina, Environ. Earth Sci., 2023, 82, 565 CrossRef CAS.
- I. B. Ivanishin and H. A. Nasr-El-Din, J. Petrol. Sci. Eng., 2021, 202, 108463 CrossRef CAS.
- M. Tarnopolskaia, A. Y. Bychkov, Y. V. Shvarov and Y. A. Popova, Geochem. Int., 2017, 55, 355–359 CrossRef CAS.
| This journal is © The Royal Society of Chemistry 2026 |





