 Open Access Article
Open Access ArticleNatural-based catalysts for 5-hydroxymethylfurfural production from cane syrup for pharmaceutical applications
Nattee Akkarawatkhoosith a,
Rathakron Suknasaenga,
Yada Chaiyasita,
Attasak Jaree
a,
Rathakron Suknasaenga,
Yada Chaiyasita,
Attasak Jaree b and
Tiprawee Tongtummachat*a
b and
Tiprawee Tongtummachat*a
aBio-Based Chemical and Biofuel Engineering Laboratory, Department of Chemical Engineering, Faculty of Engineering, Mahidol University, Phuttamonthon 4 Road, Nakhon Pathom, 73170, Thailand. E-mail: tiprawee.ton@mahidol.ac.th
bCenter for High-Value Products from Bioresources (HVPB), Department of Chemical Engineering, Faculty of Engineering, Kasetsart University, Bangkok, 10900, Thailand
First published on 20th January 2026
Abstract
In this study, naturally derived carboxylic acids, including citric, malic, acetic, and lactic acids, were investigated as novel non-toxic catalysts for producing pharmaceutical-grade 5-HMF from cane syrup, thereby demonstrating a safer and more sustainable alternative to conventional synthesis routes. A continuous catalytic biphasic microreactor system was employed with dimethyl carbonate as an environmentally benign extraction solvent. Among the acids tested, citric acid exhibited the highest catalytic performance and was selected for further systematic optimization. Response surface methodology was utilized to identify and optimize key reaction parameters, including reaction temperature, residence time, organic-to-aqueous (O![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) A) volumetric ratio, initial syrup concentration, and catalyst loading. Optimal conditions were established as a reaction temperature of 180 °C, a residence time of 60 min, an O
A) volumetric ratio, initial syrup concentration, and catalyst loading. Optimal conditions were established as a reaction temperature of 180 °C, a residence time of 60 min, an O![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) A ratio of 0.5
A ratio of 0.5![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, an initial syrup concentration of 6.25 g L−1, and a catalyst loading of 30 wt%, yielding a 5-HMF yield of 76.9% with a selectivity of 89.6%. Following purification, the obtained 5-HMF exhibited a purity of 99%. These outcomes exceed previously reported yields for comparable renewable feedstocks, highlighting this approach as a promising pathway to producing high-purity 5-HMF for food and pharmaceutical applications.
1, an initial syrup concentration of 6.25 g L−1, and a catalyst loading of 30 wt%, yielding a 5-HMF yield of 76.9% with a selectivity of 89.6%. Following purification, the obtained 5-HMF exhibited a purity of 99%. These outcomes exceed previously reported yields for comparable renewable feedstocks, highlighting this approach as a promising pathway to producing high-purity 5-HMF for food and pharmaceutical applications.
1 Introduction
5-Hydroxymethylfurfural (5-HMF) has emerged as one of the “top-value” bio-based platform chemicals due to its wide range of applications as a raw material, including polymer precursors, biofuels, and active pharmaceutical ingredients (APIs).1 Among these applications, 5-HMF has gained rapid attention as an API.2 Pharmacological studies report anti-sickling,3 antioxidant, and cytoprotective properties,4 positioning highly pure 5-HMF as a promising candidate for the treatment of sickle-cell disease,5 ischemia-reperfusion injury, and other oxidative stress disorders.6 Consequently, there is a growing global demand for pharmaceutical-grade 5-HMF, necessitating production methods with high selectivity, minimal contaminants, and sustainable practices consistent with Good Manufacturing Practice (GMP) and FDA standards. However, traditional syntheses, which focus on bio-based polymer applications, have relied on strong mineral acids or metal-chloride acids. Although effective, these catalysts are corrosive, generate salt waste, and raise toxicological concerns that complicate downstream purification, making them unsuitable for pharmaceutical applications.7 Therefore, alternative safer production pathways are required to overcome these challenges.Switching from highly toxic catalysts, such as mineral acids, to less hazardous organic acids presents a promising alternative. Sajid et al. (2020)8 investigated a range of organic acids, including p-toluenesulfonic acid (pTSA), oxalic acid, maleic acid, malonic acid, and succinic acid, for the conversion of fructose to 5-HMF in batch systems. Among these, pTSA demonstrated the highest 5-HMF yield (up to 90.2%) at 120 °C within 30 min using dimethyl sulfoxide (DMSO) as the solvent. Despite its high efficiency, pTSA is not suitable for food or pharmaceutical applications because it is toxic and not naturally derived. Therefore, identifying food-grade and naturally occurring organic acids represents a viable approach to addressing these limitations. Furthermore, the use of DMSO poses additional challenges due to its high boiling point, which complicates solvent recovery.9
The choice of solvent is also crucial for the effective production of biphasic 5-HMF. Common solvents, such as methyl isobutyl ketone (MIBK) and DMSO, are preferred due to their good partitioning properties. However, the residual levels of solvents in pharmaceutical products must comply with strict FDA guidelines, which limit MIBK concentration to approximately 4500 ppm. Identifying safer and more easily removable solvents is therefore essential. Alternative solvents such as ethanol and acetone have been explored. For instance, Zhu et al. (2022)10 synthesized 5-HMF from glucose in an acetone-water system using AlCl3 and HCl catalysts, achieving a yield of 64.5% and a purity of 85%. Nevertheless, the toxicity of these catalysts still limits their pharmaceutical applications, highlighting the necessity for safer and greener alternatives.
The selection of raw materials also significantly impacts production costs, feasibility, and overall efficiency. Fructose, a monosaccharide sugar, is usually preferred due to its high conversion efficiency. However, its limited availability and higher cost reduce its commercial viability. Alternatively, syrup, a widely available and cost-effective sugar source used extensively in food and pharmaceutical industries,11 is an attractive choice for sustainable and economically viable 5-HMF production.12,13
Our previous research14 demonstrated the continuous, catalyst-free biphasic production of 5-HMF from syrup using dimethyl carbonate (DMC), a green solvent. This process achieved a 69.5% yield and 77.9% selectivity at 190 °C, with a residence time of 134 min and an organic-to-aqueous volume ratio of 0.5![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1. Although the results are promising, the yield remains lower than that obtained with conventional methods employing mineral acid catalysts. This highlights the need for further process improvements, potentially by adding safer, greener catalytic alternatives.
1. Although the results are promising, the yield remains lower than that obtained with conventional methods employing mineral acid catalysts. This highlights the need for further process improvements, potentially by adding safer, greener catalytic alternatives.
In this study, we explored renewable, low-toxicity natural organic acids—citric acid, malic acid, acetic acid, and lactic acid—as catalysts to enhance our previously developed continuous biphasic 5-HMF production method in a microreactor using DMC as the solvent. These catalysts, approved for food and pharmaceutical applications, have not yet been examined for this particular purpose. DMC was selected for its favorable safety profile, lower boiling point, and reduced environmental impact compared with conventional solvents such as methyl isobutyl ketone (MIBK).15 We systematically evaluated and optimized key operational variables influencing 5-HMF yield/selectivity using response surface methodology and compared our results with existing literature. The outcomes of this research will provide valuable insights into sustainable, economically feasible, and pharmaceutical-grade production of 5-HMF.
2 Materials and methods
2.1 Materials
Commercial food-grade natural cane syrup (Mitr Phol Co., Ltd) obtained from a local market was employed as the raw material. The syrup comprised 31% fructose, 30% glucose, and 39% sucrose.14 It was utilized as received without any additional pretreatment. Methanol (≥99%), deionized (DI) water, methyl isobutyl ketone (≥99%), and acetonitrile (≥99%) were obtained from RCI Labscan. AR-grade citric acid (≥99%) and lactic acid (≥85%) were obtained from KemAus. Malic acid (≥99%) was purchased from HiMedia, and acetic acid (≥99%) was supplied by Merck. All organic acids were used as received, without further purification. Dimethyl carbonate (≥98.5%), and analytical standards (5-hydroxymethylfurfural (5-HMF, ≥98.0%), levulinic acid (≥97.5%), and formic acid (≥97.5%)) were purchased from Sigma-Aldrich. Sucrose (≥99.5%), fructose (≥99.0%), and glucose (≥99.5%) were obtained from Merck.2.2 5-HMF production
The continuous production of 5-HMF was carried out using a continuous-flow microreactor (volume: 4 mL; OD: 1/16″). The homogeneous aqueous feed solution (aqueous phase), consisting of syrup (6.25–25 g L−1) and a selected catalyst (1–30 wt%), along with DMC as the organic solvent (organic phase), were separately delivered into a micromixer using two HPLC pumps. The volumetric flow rates of the feed streams were adjusted to achieve the desired residence times (30–80 min), corresponding to total flow rates of 0.133–0.050 mL min−1 and the organic-to-aqueous phase volume ratio (O![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) A ratio; 0.5
A ratio; 0.5![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 to 5
1 to 5![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1). Upon mixing, a biphasic stream was formed and introduced into the microreactor, which was placed inside an oven set to the desired reaction temperature (140–190 °C). To prevent solvent vaporization, a back-pressure regulator was installed at the reactor outlet to maintain a constant pressure of 1000 psi. The effluent was subsequently separated into organic and aqueous phases. Both phases were analyzed to determine the concentrations of 5-HMF, unreacted sugars, and by-products. These data were used to calculate key reaction performance metrics, including syrup conversion, 5-HMF selectivity, and 5-HMF yield, as defined by eqn (1)–(3). The experimental setup is illustrated in Fig. 1.
1). Upon mixing, a biphasic stream was formed and introduced into the microreactor, which was placed inside an oven set to the desired reaction temperature (140–190 °C). To prevent solvent vaporization, a back-pressure regulator was installed at the reactor outlet to maintain a constant pressure of 1000 psi. The effluent was subsequently separated into organic and aqueous phases. Both phases were analyzed to determine the concentrations of 5-HMF, unreacted sugars, and by-products. These data were used to calculate key reaction performance metrics, including syrup conversion, 5-HMF selectivity, and 5-HMF yield, as defined by eqn (1)–(3). The experimental setup is illustrated in Fig. 1.
 | (1) |
 | (2) |
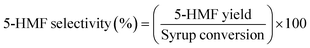 | (3) |
2.3 Product analysis
The analysis of sugar compounds, specifically glucose, fructose, and sucrose, was conducted using high-performance liquid chromatography (HPLC) equipped with a refractive index detector (RI). An ACE Excel 5 NH2 column (250 mm × 4.6 mm, 5 µm particle size) was employed for chromatographic separation, with the column temperature maintained at a constant 40 °C. The mobile phase consisted of a mixture of acetonitrile and deionized water in a volumetric ratio of 82![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 18 (v/v), delivered at a flow rate of 1 mL min−1. Each sample was introduced into the system via a 10 µL injection, and the concentrations of the individual sugars (g L−1) were quantified using external standard calibration curves prepared from pure reference compounds.
18 (v/v), delivered at a flow rate of 1 mL min−1. Each sample was introduced into the system via a 10 µL injection, and the concentrations of the individual sugars (g L−1) were quantified using external standard calibration curves prepared from pure reference compounds.
For the identification and quantification of dehydration products and by-products, including 5-HMF, levulinic acid, and formic acid, an HPLC system equipped with a photodiode array (PDA) detector was employed. Chromatographic separation was achieved using an ACE Excel C18 column (250 mm × 4.6 mm, 5 µm particle size), also operated at 40 °C. The mobile phase was composed of deionized water and acetonitrile in a 90![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 10 (v/v) ratio, with a flow rate of 1 mL min−1 maintained. Samples were injected at a fixed volume of 10 µL, and the resulting chromatograms were analyzed to determine compound concentrations by referencing standard calibration curves.
10 (v/v) ratio, with a flow rate of 1 mL min−1 maintained. Samples were injected at a fixed volume of 10 µL, and the resulting chromatograms were analyzed to determine compound concentrations by referencing standard calibration curves.
2.4 Post-treatment purification process
The 5-HMF product obtained from the continuous-flow catalytic process was purified following the procedure described in our previous work.16 Briefly, the organic and aqueous product phases were first separated using an in-line membrane separator. The organic 5-HMF phase was subsequently purified by adsorption onto Amberlyst 21, followed by vacuum evaporation, yielding 5-HMF in 99% purity. The aqueous phase was extracted with MIBK, and the resulting organic extract was collected after the membrane separator was purified using the same adsorption and evaporation steps as applied to the organic phase.3 Results and discussion
3.1 Catalyst-free 5-HMF production
Continuous catalyst-free production of 5-HMF from syrup was initially conducted to confirm the necessity of a catalyst. The experiments were performed based on previous results14 at a syrup concentration of 25 g L−1, an O![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) A ratio of 1
A ratio of 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, and residence times of 40–80 min. Preliminary experiments were conducted at a reaction temperature of 150 °C to enable clearer evaluation of the intrinsic catalytic effects by minimizing thermal degradation and secondary reactions. As shown in Fig. 2, the results indicated low 5-HMF yields (ranging from 2.8% to 4.2%) in the absence of a catalyst. Extending the residence time up to 80 min did not significantly improve the yield. This was due to the absence of acid sites, which leaves the hexose-dehydration step kinetically hindered. Note that, however, increasing both reaction temperature (≥180 °C) and residence time (≥200 min) might provide a positive effect on the 5-HMF yield, the stronger degradation of 5-HMF via secondary reactions (humins and organic acids (LA and FA)) is encountered, resulting in low selectivity and difficulties in downstream processing.17 For example, Asghari and Yoshida (2006)18 investigated the synthesis of 5-HMF from fructose under subcritical water conditions. They reported a yield of approximately 20% at 240 °C. However, when the residence time was extended by about fivefold, the 5-HMF yield rapidly declined to around 5%. It should be noted that achieving high 5-HMF yields (>50%) is difficult under non-catalytic conditions,19 as glucose is scarcely converted to 5-HMF via direct dehydration via the acyclic pathway.20 These findings confirm that a catalyst is essential for enhancing 5-HMF production performance (yield/selectivity) at shorter residence time through both cyclic and acyclic pathways. Given the goal of developing a 5-HMF production system for pharmaceutical applications, the selected catalysts must be safe, naturally derived, and broadly utilized in the food and pharmaceutical industries. Therefore, four natural catalysts were chosen for investigation: (1) acetic acid, (2) citric acid, (3) lactic acid, and (4) malic acid. These substances are widely used and play a significant role in both the food and pharmaceutical sectors.
1, and residence times of 40–80 min. Preliminary experiments were conducted at a reaction temperature of 150 °C to enable clearer evaluation of the intrinsic catalytic effects by minimizing thermal degradation and secondary reactions. As shown in Fig. 2, the results indicated low 5-HMF yields (ranging from 2.8% to 4.2%) in the absence of a catalyst. Extending the residence time up to 80 min did not significantly improve the yield. This was due to the absence of acid sites, which leaves the hexose-dehydration step kinetically hindered. Note that, however, increasing both reaction temperature (≥180 °C) and residence time (≥200 min) might provide a positive effect on the 5-HMF yield, the stronger degradation of 5-HMF via secondary reactions (humins and organic acids (LA and FA)) is encountered, resulting in low selectivity and difficulties in downstream processing.17 For example, Asghari and Yoshida (2006)18 investigated the synthesis of 5-HMF from fructose under subcritical water conditions. They reported a yield of approximately 20% at 240 °C. However, when the residence time was extended by about fivefold, the 5-HMF yield rapidly declined to around 5%. It should be noted that achieving high 5-HMF yields (>50%) is difficult under non-catalytic conditions,19 as glucose is scarcely converted to 5-HMF via direct dehydration via the acyclic pathway.20 These findings confirm that a catalyst is essential for enhancing 5-HMF production performance (yield/selectivity) at shorter residence time through both cyclic and acyclic pathways. Given the goal of developing a 5-HMF production system for pharmaceutical applications, the selected catalysts must be safe, naturally derived, and broadly utilized in the food and pharmaceutical industries. Therefore, four natural catalysts were chosen for investigation: (1) acetic acid, (2) citric acid, (3) lactic acid, and (4) malic acid. These substances are widely used and play a significant role in both the food and pharmaceutical sectors.
 | ||
Fig. 2 5-HMF yield under catalyst-free conditions at different residence times; (150 °C, 25 g L−1, 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, and 40–80 min). 1, and 40–80 min). | ||
3.2 Catalytic 5-HMF production
Continuous catalytic 5-HMF production from syrup was performed under conditions similar to those for catalyst-free 5-HMF production. Four natural catalysts were preliminarily tested, including (1) acetic acid, (2) citric acid, (3) lactic acid, and (4) malic acid. The catalyst concentration was 10 wt%. The results, as shown in Fig. 3, indicate that the highest 5-HMF yield of 24.5% was achieved with citric acid, followed by malic acid (22.0%), lactic acid (20.0%), and acetic acid (16.4%). The superior catalytic efficiency of citric acid can be attributed to its unique chemical structure and strong acidity (low pKa).8,18 Citric acid possesses three carboxylic acid groups (–COOH) and a pKa value of 3.13, which allows it to donate protons readily. This facilitates the selective dehydration of fructose to 5-HMF via a cyclic pathway.21 In contrast, malic acid (pKa 3.40) and lactic acid (pKa 3.86), though capable of catalyzing the reaction, exhibit relatively lower activity. This can be attributed to their lower acidity or fewer available carboxyl groups, which limit their ability to stabilize intermediate species and effectively drive the reaction forward. Acetic acid (pKa 4.76), possessing only a single carboxyl group and the weakest acidity among the tested acids, exhibited the poorest catalytic dehydration performance, resulting in the lowest 5-HMF yield. Its limited proton-donating ability hinders efficient dehydration of fructose intermediates and fails to suppress competing side reactions, thereby reducing selectivity toward 5-HMF formation.22 The weak acid strength leads to incomplete catalytic dehydration, while allowing parallel degradation pathways—such as thermal decomposition and self-condensation—to dominate. This explains why, despite showing the highest syrup conversion, acetic acid produced the lowest 5-HMF yield. However, the catalytic production of 5-HMF with these catalysts significantly increased the 5-HMF yield compared to the catalyst-free system (4.2%) (see Fig. 2), confirming the catalyst requirement for this process. | ||
Fig. 3 Comparison of 5-HMF yield and selectivity over various organic acid catalysts; (150 °C, 25 g L−1, 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, 80 min, and 10 wt%). 1, 80 min, and 10 wt%). | ||
Further investigation of each candidate was still required to select the best catalyst prior to the optimization section. To identify the best candidate, the full factorial design (3 levels 2 factors) was employed under the conditions as follows: catalyst concentrations of 1 to 10 wt%, residence times of 40 to 80 min, while the other parameters were kept constant at the syrup concentration of 25 g L−1, O![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) A ratio of 1
A ratio of 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, and reaction temperature of 150 °C. The 5-HMF yield was selected as the primary indicator for evaluating catalyst performance, as it directly reflects the efficiency of syrup conversion into the desired 5-HMF product and encompasses both the activity and selectivity of the catalyst. The results are discussed in the following sections.
1, and reaction temperature of 150 °C. The 5-HMF yield was selected as the primary indicator for evaluating catalyst performance, as it directly reflects the efficiency of syrup conversion into the desired 5-HMF product and encompasses both the activity and selectivity of the catalyst. The results are discussed in the following sections.
3.3 Catalytic 5-HMF production with various organic acids
The production of 5-HMF from syrup using acetic acid as a catalyst was first studied under the following conditions: 25 g L−1 syrup concentration, 150 °C reaction temperature, a 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 organic-to-aqueous phase ratio, and residence times ranging from 40 to 80 min. Catalyst loadings varied from 1 to 10 wt%. As shown in Fig. 4(a), the highest 5-HMF yield of 16.4% was achieved at 80 min and 10 wt% catalyst. The results indicate that increasing both residence time and catalyst loading enhances the yield of 5-HMF. However, prolonging the residence time alone, especially at low catalyst concentrations (<5 wt%), led to only modest improvements. A similar trend was observed in the work of Souza et al.,23 for instance, at 5 wt%, loading and extending the residence time from 40 to 80 min increased the yield from 1.01% to 4.7%. In contrast, increasing the catalyst loading from 5 to 10 wt% at a fixed residence time of 40 min improved the yield to 5.26%. These findings suggest that catalyst concentration has a more pronounced impact than residence time, likely due to increased proton (H+) availability, which facilitates sugar dehydration reactions. However, excessive acid concentrations may also promote 5-HMF degradation to side products such as levulinic and formic acids. Thus, acetic acid was found to have limited catalytic efficiency for continuous 5-HMF production due to the interaction effects of time and catalyst concentration.
1 organic-to-aqueous phase ratio, and residence times ranging from 40 to 80 min. Catalyst loadings varied from 1 to 10 wt%. As shown in Fig. 4(a), the highest 5-HMF yield of 16.4% was achieved at 80 min and 10 wt% catalyst. The results indicate that increasing both residence time and catalyst loading enhances the yield of 5-HMF. However, prolonging the residence time alone, especially at low catalyst concentrations (<5 wt%), led to only modest improvements. A similar trend was observed in the work of Souza et al.,23 for instance, at 5 wt%, loading and extending the residence time from 40 to 80 min increased the yield from 1.01% to 4.7%. In contrast, increasing the catalyst loading from 5 to 10 wt% at a fixed residence time of 40 min improved the yield to 5.26%. These findings suggest that catalyst concentration has a more pronounced impact than residence time, likely due to increased proton (H+) availability, which facilitates sugar dehydration reactions. However, excessive acid concentrations may also promote 5-HMF degradation to side products such as levulinic and formic acids. Thus, acetic acid was found to have limited catalytic efficiency for continuous 5-HMF production due to the interaction effects of time and catalyst concentration.
Lactic acid was then evaluated under the same conditions used for acetic acid. The results (see Fig. 4(b)) showed that both catalyst loading and residence time had a stronger effect on 5-HMF yield than observed with acetic acid. At 5 wt% after 40 min of catalyst loading, the 5-HMF yield reached 8.28%, increasing to 14.5% at 80 min. Alternatively, using 10 wt% lactic acid at 40 min gave an 11% yield. The improved performance can be attributed to lactic acid's stronger acidity (pKa 3.86 vs. 4.76 for acetic acid), which enhances protonation and facilitates the dehydration of sugars. The maximum 5-HMF yield of 20% was achieved at 80 min and 10 wt% catalyst. Notably, a yield of 18.8% was already attained at 60 min, suggesting lactic acid enables efficient conversion under moderate conditions.
Malic acid was also tested under identical reaction conditions and consistently delivered higher 5-HMF yields (see Fig. 4(c)) compared to acetic and lactic acids. This enhanced performance is due to its lower pKa and dicarboxylic structure, which allows greater proton donation.24 For example, malic acid contains two carboxyl groups, unlike acetic and lactic acids, which contain only one. At 80 min and 10 wt% loading, the highest 5-HMF yield reached 22.0%. This stronger acidity makes malic acid a more effective catalyst under the studied conditions.
Citric acid was evaluated using the same setup and demonstrated the highest catalytic performance among all organic acids studied (see Fig. 4(d)). As a tricarboxylic acid, citric acid provides three carboxyl groups, enhancing proton availability far beyond that of acetic (1), lactic (1), or malic acid (2). This increased acidity promotes faster and more efficient dehydration of sugar to 5-HMF. Moreover, citric acid's excellent water solubility (≈59% w/w at 20 °C) ensures efficient dispersion in the aqueous phase, enhancing contact with dissolved sugars. However, the high proton concentration supplied by citric acid's tricarboxylic framework also accelerates secondary reactions of 5-HMF; consequently, prolonged residence times promote rapid re-hydration and humin formation.25,26 Adjusting the O![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) A ratio counteracts this effect by extracting 5-HMF as soon as it forms, thereby suppressing the undesired pathways and preserving yield and selectivity (as detailed in Section 3.4.3). Under optimal conditions (80 min, 10 wt% catalyst), citric acid yielded a maximum of 26.9%. Given its superior catalytic activity and stabilizing properties, citric acid was selected for further studies to optimize 5-HMF production from syrup.
A ratio counteracts this effect by extracting 5-HMF as soon as it forms, thereby suppressing the undesired pathways and preserving yield and selectivity (as detailed in Section 3.4.3). Under optimal conditions (80 min, 10 wt% catalyst), citric acid yielded a maximum of 26.9%. Given its superior catalytic activity and stabilizing properties, citric acid was selected for further studies to optimize 5-HMF production from syrup.
3.4 Influence of process variables on 5-HMF performance
The investigation of key parameters influencing 5-HMF production from syrup using citric acid as the selected catalyst was conducted via a one-factor-at-a-time (OFAT) approach. The parameters examined were catalyst loading, reaction temperature, organic-to-aqueous phase volume ratio, and initial syrup concentration. This systematic approach enabled the thorough evaluation of individual effects on 5-HMF yield, selectivity, and conversion prior to optimization using Design of Experiments (DOE).![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 organic-to-aqueous volume ratio, 150 °C reaction temperature). The experimental results, illustrated in Fig. 5, clearly demonstrate the significant influence of catalyst concentration on both 5-HMF yield and syrup conversion.
1 organic-to-aqueous volume ratio, 150 °C reaction temperature). The experimental results, illustrated in Fig. 5, clearly demonstrate the significant influence of catalyst concentration on both 5-HMF yield and syrup conversion.
 | ||
Fig. 5 Effect of catalyst loading on 5-HMF yield and syrup conversion; (80 min, 25 g L−1, 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, 150 °C, and 1–30 wt%). 1, 150 °C, and 1–30 wt%). | ||
At the lowest citric acid concentration (1 wt%), the yield of 5-HMF was only 12.49%, accompanied by a relatively low syrup conversion of 30.0%, reflecting insufficient catalytic activity at low acid loadings. As the concentration of citric acid increased, both the yield and syrup conversion notably improved. At 5 wt%, the yield slightly improved to 13.46%, while conversion increased more substantially to 40.18%. When the citric acid concentration was further increased to 10 and 20 wt%, yields progressively rose to 24.54% and 27.90%, respectively, with corresponding syrup conversions of 38.52% and 44.80%. These results indicate that higher catalyst loadings enhance the dehydration of sugars to 5-HMF by increasing the proton availability within the reaction medium.27 However, when the catalyst loading reached 30 wt%, the syrup conversion significantly increased to 58.0%, but the corresponding 5-HMF yield was only 31.66%. This marked increase in conversion, without a proportionate enhancement in yield, strongly suggests an escalation in undesirable side reactions at excessively high catalyst concentrations. These findings align with prior research indicating that overly acidic conditions can induce side reactions, such as the rehydration of 5-HMF into levulinic and formic acids or the formation of humin through polymerization.28
In summary, while increasing citric acid concentration positively influences syrup conversion and overall reaction rate, excessively high acid loading reduces selectivity due to competing side reactions. Therefore, 20 wt% citric acid was selected as the optimal concentration. This catalyst loading was consequently chosen as a reference condition for subsequent investigations into other process parameters.
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1. As shown in Fig. 6, increasing the reaction temperature slightly improved the 5-HMF yield but significantly promoted side reactions.
1. As shown in Fig. 6, increasing the reaction temperature slightly improved the 5-HMF yield but significantly promoted side reactions.
 | ||
Fig. 6 Effect of reaction temperature on 5-HMF yield and syrup conversion; (80 min, 25 g L−1, 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, 20 wt%, and 150–190 °C). 1, 20 wt%, and 150–190 °C). | ||
At 150 °C, the 5-HMF yield was 27.9%, with a relatively high selectivity of 62.3%. When the temperature was increased to 190 °C, the yield rose modestly to 30.61%; however, the selectivity dropped sharply to 31.7%. These results indicate that while higher temperatures can enhance the reaction kinetics of sugar dehydration to 5-HMF, they also intensify secondary reactions that degrade 5-HMF into by-products such as levulinic acid, formic acid, and polymeric humins. This degradation pathway is well-documented in previous studies, which have shown that 5-HMF is thermally unstable under acidic conditions, especially at elevated temperatures.29
Moreover, high temperatures may promote not only the rehydration of 5-HMF but also condensation reactions with other reactive intermediates, leading to the formation of insoluble humins. These side reactions reduce the effective yield and increase purification costs, which are critical considerations for industrial applications. The observed sharp drop in selectivity at 190 °C highlights the need to moderate the temperature in acid-catalyzed systems to preserve product integrity.
In contrast, operating at 170 °C provided a better balance between yield and selectivity. At this temperature, the 5-HMF yield was higher than that at 150 °C, while selectivity remained within an acceptable range. This suggests that 170 °C offers sufficient thermal energy to drive the dehydration reaction efficiently without severely accelerating 5-HMF degradation pathways. Similar optimal temperature ranges have been reported in related systems using organic or heterogeneous acid catalysts.30
Therefore, 170 °C was selected as the working temperature for subsequent experiments on other variables. It represents a practical compromise between product yield, selectivity, and reaction severity. However, it should be noted that the precise optimal temperature will be re-evaluated in the next phase of this study using a statistically designed experimental approach (Design of Experiments, DOE), which will account for interaction effects and allow for more accurate process optimization.
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/b_char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/b_char_2009.gif) A ratio. The following parameter investigated was the volume ratio of the organic to aqueous phase, an important factor in biphasic reaction systems for 5-HMF production. The experiments were performed under controlled conditions: a reaction temperature of 170 °C, the citric acid concentration of 20 wt%, residence time of 80 min, and initial syrup concentration of 25 g L−1. The results, presented in Fig. 7, demonstrate that increasing the volume ratio of the organic phase relative to the aqueous phase has an adverse effect on the 5-HMF yield.
A ratio. The following parameter investigated was the volume ratio of the organic to aqueous phase, an important factor in biphasic reaction systems for 5-HMF production. The experiments were performed under controlled conditions: a reaction temperature of 170 °C, the citric acid concentration of 20 wt%, residence time of 80 min, and initial syrup concentration of 25 g L−1. The results, presented in Fig. 7, demonstrate that increasing the volume ratio of the organic phase relative to the aqueous phase has an adverse effect on the 5-HMF yield.
 | ||
Fig. 7 Effect of O![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) A ratio on 5-HMF yield and syrup conversion; (80 min, 25 g L−1, 20 wt%, 170 °C, and 0.5:1–5:1). A ratio on 5-HMF yield and syrup conversion; (80 min, 25 g L−1, 20 wt%, 170 °C, and 0.5:1–5:1). | ||
In biphasic systems, the organic phase (solvent phase) facilitates the in situ extraction of 5-HMF from the aqueous phase, thereby protecting it from secondary degradation reactions such as rehydration to levulinic acid and formic acid or condensation into humins. Several studies have highlighted the advantage of using an organic overlay to enhance 5-HMF selectivity and yield in acid-catalyzed systems.31 However, in this study, when the volume of the organic phase exceeded a certain threshold, a counterproductive dilution effect was observed. In addition, the flow regime shifted to a longer slug-length pattern, reducing the interfacial area between the organic and aqueous phases and consequently lowering the overall mass-transfer efficiency.
Specifically, at elevated organic-to-aqueous ratios, the 5-HMF concentration in the organic phase decreased markedly, reflecting limited mass transfer across the interface and diminished extraction efficiency. Excess organic solvent volume can modify interfacial dynamics, reduce interfacial contact, and disrupt mixing behavior, collectively hindering mass transfer and extraction kinetics.32
The highest 5-HMF yield of 32.0% was achieved at a volume ratio of 0.5![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, indicating an optimal balance between effective extraction and minimal dilution. This finding is consistent with previous reports emphasizing the need for carefully optimized phase ratios to maximize extraction performance and minimize unnecessary solvent use.33 Thus, the organic-to-aqueous phase volume ratio plays a critical role in determining not only the yield and selectivity of 5-HMF but also the process's sustainability and cost-effectiveness.34 This parameter must be finely tuned to maximize product recovery while minimizing solvent usage and downstream separation costs.
1, indicating an optimal balance between effective extraction and minimal dilution. This finding is consistent with previous reports emphasizing the need for carefully optimized phase ratios to maximize extraction performance and minimize unnecessary solvent use.33 Thus, the organic-to-aqueous phase volume ratio plays a critical role in determining not only the yield and selectivity of 5-HMF but also the process's sustainability and cost-effectiveness.34 This parameter must be finely tuned to maximize product recovery while minimizing solvent usage and downstream separation costs.
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1. The experimental results, shown in Fig. 8, indicate that lowering the initial syrup concentration significantly reduced the occurrence of side reactions and enhanced 5-HMF production efficiency.
1. The experimental results, shown in Fig. 8, indicate that lowering the initial syrup concentration significantly reduced the occurrence of side reactions and enhanced 5-HMF production efficiency.
 | ||
Fig. 8 Effect of initial syrup concentration on 5-HMF yield and syrup conversion; (80 min, 20 wt%, 170 °C, 0.5![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, and 6.25–25 g L−1). 1, and 6.25–25 g L−1). | ||
At the lowest concentration of 6.25 g L−1, the highest 5-HMF yield of 57.69% was achieved, along with a syrup conversion of 87.57%. These results suggest that dilute conditions limit the accumulation of reactive intermediates, thereby suppressing secondary reactions such as humin formation and rehydration of 5-HMF. This phenomenon aligns with prior studies indicating that lower substrate concentrations reduce the probability of intermolecular condensation and polymerization side reactions.35 Moreover, under dilute conditions, the dehydration of monosaccharides to 5-HMF can proceed more selectively, as heat- and mass-transfer limitations are minimized and local acid concentrations remain effective throughout the reaction medium.36 Therefore, an initial syrup concentration of 6.25 g L−1 was selected as the most suitable condition for further optimization.
A shorter residence time (60 min) was further evaluated at an initial syrup concentration of 6.25 g L−1 to suppress side reactions occurring at prolonged residence times and to enhance process productivity. The results showed that the 5-HMF yield of 56.50% was obtained at 60 min, which slightly decreased at 80 min (57.69%). Despite the marginal increase in yield at longer residence times, selectivity significantly reduced from 70.0% at 60 min to 65.8% at 80 min, indicating an increased contribution of side reactions at prolonged residence times. Consequently, a residence time of ≤60 min was considered more suitable for subsequent optimization studies, balancing yield, selectivity, and process efficiency.
In summary, the results of the OFAT investigation highlight that residence time, temperature, and citric acid concentration are the key factors influencing 5-HMF production from syrup. These variables govern both the extent of sugar dehydration and the formation of unwanted by-products. Consequently, these parameters were selected for further study in the optimization phase using a design-of-experiments approach, specifically response surface methodology (RSM) coupled with a Box–Behnken design (BBD). The defined operating ranges for these parameters are summarized in Table 1.
| Variables | Symbol | Levels | Constraints | ||
|---|---|---|---|---|---|
| −1 | 0 | 1 | |||
| Independent variables | |||||
| Reaction temperature (°C) | X1 | 140 | 160 | 180 | In the range |
| Residence time (min) | X2 | 30 | 45 | 60 | In the range |
| Catalyst amount (g L−1) | X3 | 10 | 20 | 30 | In the range |
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) |
|||||
| Dependent variable | |||||
| 5-HMF yield (%) | Y1 | Optimize | |||
| 5-HMF selectivity (%) | Y2 | Optimize | |||
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) |
|||||
| Control variables | |||||
| Feed concentration (g L−1) | 6.25 | ||||
O![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) A ratio (v/v) A ratio (v/v) |
0.5![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1 |
||||
3.5 Optimization process
The experimental results summarized in Table 1 were used to establish the relationships between the independent variables (reaction temperature, residence time, and citric acid concentration) and the response variables (5-HMF yield and selectivity) via the BBD-RSM technique, yielding eqn (4) and (5). The validity and accuracy of the predictive models were rigorously evaluated using statistical analysis. Specifically, the lack-of-fit test for eqn (4) yielded a p-value of 0.35, indicating no significant lack of fit and thus affirming the model's suitability for describing the experimental data. The coefficient of determination (R2) was notably high at 99.1%, further underscoring the model's precision and reliability in predicting experimental outcomes. Similarly, eqn (5) demonstrated comparable statistical robustness, reinforcing the model's predictive credibility (p-value for lack-of-fit of 0.56 and R2 of 98.7%).Using these statistically validated models, optimal conditions for maximizing 5-HMF production were identified: a reaction temperature of 180 °C, a residence time of 60 min, and a citric acid concentration of 30 wt%. Under these conditions, the model predicted a 5-HMF yield of 75% with a selectivity of 93%.
To experimentally validate the model predictions, verification experiments were performed under the identified optimal conditions. The experimental results revealed a 5-HMF yield of 76.9% and a selectivity of 89.6%, with minor percentage deviations of 2.5% and 3.8%, respectively, from the predicted values. This close agreement between predicted and observed outcomes confirms that the developed regression models are reliable and can be confidently used to guide process optimization and scaling up production. Crude 5-HMF produced under the optimized conditions was purified using the procedure described in our earlier work,16 affording a product with 99% purity.
| Y1 = 997.46–10.63X1 − 8.48X2 + 2.35X3 + 0.03X12 + 0.022X22 − 0.062X32 + 0.041X1X2 + 0.001X1X3 + 0.001X2X3 | (4) |
| Y2 = 553.83–7.19X1 − 1.58X2 − 3.467X3 + 0.024X12 + 0.004X22 − 0.026X32 + 0.007X1X2 + 0.026X1X3 − 0.023X2X3 | (5) |
3.6 Interaction effect of process variables
The interaction between reaction temperature and residence time was thoroughly evaluated, as illustrated in Fig. 9(a). A pronounced positive interaction effect was observed for 5-HMF yield across the studied range. Specifically, at shorter residence times (e.g., 30 min), increasing the reaction temperature from 140 °C to 180 °C significantly boosted the 5-HMF yield, attributed to the accelerated kinetics of sugar dehydration. This beneficial interaction was even more pronounced at extended residence times: at 180 °C, the increase in yield from 40 to 60 min was roughly three times greater than that at 140 °C. These observations underline the amplifying effect of high temperatures on residence time, making both parameters critical for optimizing yield. However, regarding 5-HMF selectivity, the interaction becomes more nuanced. High temperatures combined with extended residence times reduce selectivity due to intensified side reactions such as rehydration to levulinic and formic acids, and the formation of humins.37 Thus, careful management of temperature and residence time is necessary to maximize both yield and selectivity.Fig. 9(c) demonstrates the interaction between reaction temperature and catalyst loading, highlighting a pronounced synergistic effect at elevated temperatures. For example, increasing catalyst loading from 10 to 20 wt% at 180 °C resulted in an approximately ten-fold greater improvement in 5-HMF yield compared to lower temperatures. This indicates that catalyst effectiveness significantly depends on sufficient thermal activation. Additionally, the positive effect on selectivity indicates optimal catalyst loading and temperature conditions preferentially drive sugar dehydration towards 5-HMF rather than unwanted side products. However, excessive catalyst loadings at high temperatures risk promoting undesired reactions. Thus, simultaneous optimization of both temperature and catalyst loading is critical for achieving high yields and selectivity.
The interaction effect of residence time and catalyst loading, illustrated in Fig. 9(b), exhibited variable outcomes dependent on catalyst loading. At high catalyst loading (30 wt%), the 5-HMF yield initially increased with residence time but eventually decreased due to enhanced side reactions under prolonged acidic conditions.38 Conversely, at low catalyst loading (10 wt%), 5-HMF yield exhibited only slight improvements before plateauing, demonstrating catalyst-limited reaction kinetics, where further extension of residence time provided negligible gains.39 A similar trend was observed for selectivity (see Fig. 10); elevated catalyst loadings, combined with extended residence times, significantly reduced selectivity by increasing the degradation of 5-HMF into undesirable by-products. At low catalyst loading, selectivity remained relatively stable, though yields were comparatively modest. These findings highlight the importance of carefully balancing catalyst amounts and residence times to avoid substantial yield and selectivity losses due to side reactions.
3.7 Process comparison
The 5-HMF production system developed in this study was compared with other production methods in terms of production conditions, 5-HMF yield and selectivity, and the types of solvents and catalysts employed. These comparisons are summarized in Table 2. The comparison considers three distinct categories: (1) catalyst-free systems, (2) hazardous catalyst systems, which utilize catalysts unsuitable for food and pharmaceutical applications due to their toxicity, and (3) green catalyst systems, employing safe catalysts and solvents compatible with food and pharmaceutical industries. However, a direct comparison proved challenging since the system presented in this work is among the first to integrate both safe solvents and catalysts specifically for 5-HMF production.| No | Raw material | Organic solvent | Catalyst (amount) | Promoter (amount) | Temp (°C) | Time (min) | Ratio (v/v) | Yield (%) | Selectivity (%) | Ref. |
|---|---|---|---|---|---|---|---|---|---|---|
a Fructose![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) glucose glucose![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) sucrose fraction (0.31 sucrose fraction (0.31![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0.30 0.30![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0.39).b Fructose 0.39).b Fructose![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) glucose glucose![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) sucrose fraction (0.55 sucrose fraction (0.55![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0.41 0.41![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0.04). 0.04). |
||||||||||
| Catalyst free system | ||||||||||
| 1 | Syrupa | MIBK | None | None | 190 | 134 | 0.5![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1 |
69.5 | 77.9 | 17 |
| 2 | Syrupa | DMC | None | None | 190 | 80 | 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1 |
66.8 | NA | 14 |
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) |
||||||||||
| Hazardous catalyst system | ||||||||||
| 2 | Glucose | MIBK | AlCl3/HCl (40 mM/40 mM) | NaCl (20 wt%) | 160 | 16 | 4![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1 |
66.2 | 79.5 | 40 |
| 3 | Glucose | MIBK | HCl (150 mM) | None | 180 | 3 | 0.5![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1 |
81.7 | 89.8 | 37 |
| 4 | Syrupb | MIBK | HCl (50 mM) | None | 155 | 16 | 4![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1 |
81 | NA | 41 |
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) |
||||||||||
| Green catalyst system | ||||||||||
| 5 | Syrupa | DMC | Citric acid (30%) | None | 180 | 60 | 0.5![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1 |
76.9 | 89.6 | This work |
As indicated in Table 2, catalyst-free systems (e.g., No. 1 in Table 2) achieved a maximum 5-HMF yield of approximately 69.5%, with a selectivity of 77.9%. These relatively modest outcomes were attributed to the absence of catalysts, requiring considerably longer residence times to achieve acceptable conversions. In contrast, systems employing hazardous catalysts such as HCl or combined HCl/AlCl3 (No. 2–4, Table 2) provided significant improvements in both yield (typically increased by more than 15%) and reaction rates (reduced residence time by more than 800%) compared to catalyst-free conditions. For instance, study No. 4 used syrup as the feedstock with HCl as the catalyst, yielding 5-HMF in 81% yield. Nonetheless, these hazardous catalyst systems are unsuitable for applications in the food and pharmaceutical sectors due to their inherent toxicity, environmental risks, and potential regulatory issues.
In the current research, the selected approach prioritized safety and sustainability by employing DMC as the solvent and citric acid as the catalyst—both of which are classified as safe and environmentally friendly for food and pharmaceutical production. Under these conditions, a 5-HMF yield of 76.9% with a high selectivity of 89.6% was achieved. Although slightly lower in yield than hazardous catalyst-based systems, this result is significantly superior to catalyst-free approaches, highlighting a favorable balance between production efficiency and safety considerations.
Thus, the developed process offers a promising alternative that aligns with green chemistry and sustainability principles, making it suitable for industrial-scale production of 5-HMF for food and pharmaceutical applications.
4 Conclusion
The primary objective of this study was to evaluate natural catalysts suitable for use in a continuous biphasic microreactor system that utilized DMC as a safe extraction solvent. Four natural catalysts—citric, malic, acetic, and lactic acids—were systematically investigated under various experimental conditions. Among them, citric acid was identified as the most effective catalyst and selected for further optimization studies. RSM was employed to examine and optimize key operating parameters, including reaction temperature, residence time, catalyst concentration, initial syrup concentration, and O![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) A ratio. Both main and interaction effects of these parameters on 5-HMF yield and selectivity were clearly demonstrated through RSM. Optimal conditions were determined to be a reaction temperature of 180 °C, a residence time of 60 min, a catalyst loading of 30 wt%, and an O
A ratio. Both main and interaction effects of these parameters on 5-HMF yield and selectivity were clearly demonstrated through RSM. Optimal conditions were determined to be a reaction temperature of 180 °C, a residence time of 60 min, a catalyst loading of 30 wt%, and an O![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) A ratio of 0.5
A ratio of 0.5![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, achieving a maximum 5-HMF yield of 76.9% with a selectivity of 89.6%. Following purification, the final product exhibited a high purity of 99%. These results surpass previously reported yields for similar renewable feedstocks, highlighting the effectiveness and practical potential of the developed low-toxicity process for sustainable 5-HMF production.
1, achieving a maximum 5-HMF yield of 76.9% with a selectivity of 89.6%. Following purification, the final product exhibited a high purity of 99%. These results surpass previously reported yields for similar renewable feedstocks, highlighting the effectiveness and practical potential of the developed low-toxicity process for sustainable 5-HMF production.
Author contributions
Nattee Akkarawatkhoosith: resources, conceptualization, methodology, investigation, formal analysis, validation, writing – original draft, writing – review & editing. Rathakron Suknasaeng: formal analysis, investigation. Yada Chaiyasit: formal analysis, investigation. Attasak Jaree: resources, supervision, writing – review & editing. Tiprawee Tongtummachat: resources, conceptualization, methodology, investigation, formal analysis, validation, funding acquisition, resources, project administration, writing – original draft, writing – review & editing.Conflicts of interest
The authors declare no conflicts of interest.Data availability
All data supporting the findings of this study are available within the article. Additional raw data or materials used in this work are available from the corresponding author upon request.Acknowledgements
This research project is supported by Mahidol University (Strategic Research Fund): 2024. During the preparation of this work, the author(s) used ChatGPT in order to improve language and readability. After using this tool/service, the author(s) reviewed and edited the content as needed and take(s) full responsibility for the content of the publication.References
- Q. S. Kong, X. L. Li, H. J. Xu and Y. Fu, Conversion of 5-hydroxymethylfurfural to chemicals: A review of catalytic routes and product applications, Fuel Process. Technol., 2020, 209, 106528, DOI:10.1016/j.fuproc.2020.106528.
- P. P. Pagare, M. McGinn, M. S. Ghatge, V. Shekhar, R. T. Alhashimi, B. D. Pierce, O. Abdulmalik, Y. Zhang and M. K. Safo, The antisickling agent, 5-hydroxymethyl-2-furfural: Other potential pharmacological applications, Med. Res. Rev., 2024, 44, 2707–2729, DOI:10.1002/med.22062.
- G. J. Kato, M. P. Lawrence and L. G. Mendelsohn, et al., Phase 1 clinical trial of the candidate anti-sickling agent Aes-103 in adults with sickle cell anemia, Blood, 2013, 122, 1009, DOI:10.1182/blood.V122.21.1009.1009.
- W. Li, X. N. Qu, Y. Han, S. W. Zheng, J. Wang and Y. P. Wang, Ameliorative effects of 5-hydroxymethyl-2-furfural (5-HMF) from Schisandra chinensis on alcoholic liver oxidative injury in mice, Int. J. Mol. Sci., 2015, 16, 2446–2457, DOI:10.3390/ijms16022446.
- O. Abdulmalik, M. K. Safo and Q. Chen, et al., 5-Hydroxymethyl-2-furfural modifies intracellular sickle haemoglobin and inhibits sickling of red blood cells, Br. J. Haematol., 2005, 128, 552–561, DOI:10.1111/j.1365-2141.2004.05332.x.
- B. Ya, L. Zhang, L. Zhang, Y. Li and L. Li, 5-Hydroxymethyl-2-furfural prolongs survival and inhibits oxidative stress in a mouse model of forebrain ischemia, Neural Regen. Res., 2012, 7, 1722–1728, DOI:10.3969/j.issn.1673-5374.2012.22.008.
- L. Hu, Y. Jiang, Z. Wu, X. Wang, A. He, J. Xu and J. Xu, State-of-the-art advances and perspectives in the separation of biomass-derived 5-hydroxymethylfurfural, J. Clean. Prod., 2020, 276, 124219, DOI:10.1016/j.jclepro.2020.124219.
- M. Sajid, Y. Bai, D. Liu and X. Zhao, Organic acid catalyzed production of platform chemical 5-hydroxymethylfurfural from fructose: Process comparison and evaluation based on kinetic modeling, Arab. J. Chem., 2020, 13, 7430–7444, DOI:10.1016/j.arabjc.2020.08.019.
- M. Sayed, N. Warlin, C. Hulteberg, I. Munslow, S. Lundmark, O. Pajalic, P. Tunå, B. Zhang, S. H. Pyo and R. Hatti-Kaul, 5-Hydroxymethylfurfural from fructose: An efficient continuous process in a water–dimethyl carbonate biphasic system with high yield product recovery, Green Chem., 2020, 22, 5402–5413, 10.1039/D0GC01422B.
- H. Zhu, X. Guo, Y. Si, Q. Du, Y. Cheng, L. Wang and X. Li, Insights into pathways and solvent effects of fructose dehydration to 5-hydroxymethylfurfural in acetone–water solvent, Chem. Eng. Sci., 2023, 267, 118352, DOI:10.1016/j.ces.2022.118352.
- C. Lin, H. Wu, J. Wang, J. Huang, F. Cao, W. Zhuang, Y. Lu, J. Chen, H. Jia and P. Ouyang, Preparation of 5-hydroxymethylfurfural from high fructose corn syrup using organic weak acid in situ as catalyst, Ind. Eng. Chem. Res., 2020, 59, 4358–4366, DOI:10.1021/acs.iecr.9b06602.
- A. L. M. Kognou, S. Shrestha, Z. Jiang, C. Xu, F. Sun and W. Qin, High-fructose corn syrup production and its new applications for 5-hydroxymethylfurfural and value-added furan derivatives: Promises and challenges, J. Bioresour. Bioprod., 2022, 7, 148–160, DOI:10.1016/j.jobab.2022.03.004.
- S. Souzanchi, L. Nazari, K. T. V. Rao, Z. Yuan, Z. Tan and C. Xu, 5-HMF production from industrial grade sugar syrups derived from corn and wood using niobium phosphate catalyst in a biphasic continuous-flow tubular reactor, Catal. Today, 2023, 407, 274–280, DOI:10.1016/j.cattod.2021.07.032.
- T. Tongtummachat, A. Jaree, K. Thongkan, W. Chuphueak and N. Akkarawatkhoosith, Continuous production of 5-hydroxymethylfurfural: A catalyst-free approach using non-toxic solvents for pharmaceutical applications, Chem. Eng. J. Adv., 2024, 18, 100597, DOI:10.1016/j.ceja.2024.100597.
- T. Shen, L. Hou, J. Gosset, H. Wang, S. Leng, Y. Boumghar, S. Barghi and C. Xu, Recent advances in processes and technologies for production of 5-hydroxymethylfurfural and 2,5-furandicarboxylic acid from carbohydrates, Chem. Eng. J., 2024, 500, 156470, DOI:10.1016/j.cej.2024.156470.
- T. Tongtummachat, K. Thongkan, W. Chuphueak, A. Jaree and N. Akkarawatkhoosith, Efficient continuous-flow separation and purification processes of 5-hydroxymethylfurfural, Chem. Eng. J. Adv., 2024, 20, 100641, DOI:10.1016/j.ceja.2024.100641.
- T. Tongtummachat, A. Jaree and N. Akkarawatkhoosith, Green synthesis of 5-hydroxymethylfurfural through non-catalytic conversion of glucose in a microreactor, Energy Convers. Manag. X, 2021, 12, 100141, DOI:10.1016/j.ecmx.2021.100141.
- F. S. Asghari and H. Yoshida, Acid-catalyzed production of 5-hydroxymethylfurfural from D-fructose in subcritical water, Ind. Eng. Chem. Res., 2006, 45, 2163–2173, DOI:10.1021/ie051025+.
- B. Song, Y. Yu and H. Wu, Tuning glucose decomposition in hot-compressed γ-valerolactone/water mixtures: From isomerization to dehydration reactions, Fuel, 2019, 238, 225–231, DOI:10.1016/j.fuel.2018.10.163.
- H. Kimura, M. Nakahara and N. Matubayasi, Solvent effect on pathways and mechanisms for D-fructose conversion to 5-hydroxymethyl-2-furaldehyde: In situ 13C NMR study, J. Phys. Chem. A, 2013, 117, 2102–2113, DOI:10.1021/jp312002h.
- T. Istasse and A. Richel, Mechanistic aspects of saccharide dehydration to furan derivatives for reaction media design, RSC Adv., 2020, 10, 23720–23742, 10.1039/D0RA03892J.
- G. Yang, E. A. Pidko and E. J. M. Hensen, Mechanism of Brønsted acid-catalyzed conversion of carbohydrates, J. Catal., 2012, 295, 122–132, DOI:10.1016/j.jcat.2012.08.002.
- R. L. De Souza, H. Yu, F. Rataboul and N. Essayem, 5-Hydroxymethylfurfural (5-HMF) production from hexoses: Limits of heterogeneous catalysis in hydrothermal conditions and potential of concentrated aqueous organic acids as reactive solvent system, Challenges, 2012, 3, 212–232, DOI:10.3390/challe3020212.
- Y. Li, M. Ma, R. Liu, Y. Liu and Z. Cao, Biocatalytic valorization of biobased 5-hydroxymethylfurfural to 5-hydroxymethyl-2-furfurylamine in a three-constituent deep eutectic solvent–water system, ACS Sustain. Chem. Eng., 2022, 10, 8452–8463, DOI:10.1021/acssuschemeng.2c01481.
- E. S. Morais, M. G. Freire, C. S. R. Freire and A. J. D. Silvestre, Improved production of 5-hydroxymethylfurfural in acidic deep eutectic solvents using microwave-assisted reactions, Int. J. Mol. Sci., 2022, 23, 1959, DOI:10.3390/ijms23041959.
- H. Matsumiya and T. Hara, Conversion of glucose into 5-hydroxymethylfurfural with boric acid in molten mixtures of choline salts and carboxylic acids, Biomass Bioenergy, 2015, 72, 227–232, DOI:10.1016/j.biombioe.2014.11.001.
- Z. Wu, Y. Yu and H. Wu, Hydrothermal reactions of biomass-derived platform molecules: Mechanistic insights into 5-hydroxymethylfurfural (5-HMF) formation during glucose and fructose decomposition, Energy Fuels, 2023, 37, 2115–2126, DOI:10.1021/acs.energyfuels.2c03462.
- H. Matsumiya and T. Hara, Conversion of glucose into 5-hydroxymethylfurfural with boric acid in molten mixtures of choline salts and carboxylic acids, Biomass Bioenergy, 2015, 72, 227–232, DOI:10.1016/j.biombioe.2014.11.001.
- C. Antonetti, D. Licursi, E. Fulignati, G. Valentini and R. Galletti, New frontiers in the catalytic synthesis of levulinic acid: From sugars to raw and waste biomass as starting feedstock, Catalysts, 2016, 6, 196, DOI:10.3390/catal6120196.
- A. A. Rosatella, S. P. Simeonov, R. F. M. Frade and C. A. M. Afonso, 5-Hydroxymethylfurfural (HMF) as a building-block platform: Biological properties, synthesis and synthetic applications, Green Chem., 2011, 13, 754–793, 10.1039/C0GC00401D.
- M. Sayed, N. Warlin, C. Hulteberg, I. Munslow, S. Lundmark, O. Pajalic, P. Tunå, B. Zhang, S. H. Pyo and R. Hatti-Kaul, 5-Hydroxymethylfurfural from fructose: An efficient continuous process in a water–dimethyl carbonate biphasic system with high yield product recovery, Green Chem., 2020, 22, 5402–5413, 10.1039/D0GC01422B.
- E. C. Sindermann, A. Holbach, A. D. Haan and N. Kockmann, Single stage and countercurrent extraction of 5-hydroxymethylfurfural from aqueous phase systems, Chem. Eng. J., 2016, 283, 251–259, DOI:10.1016/j.cej.2015.07.029.
- L. Hu, Y. Jiang, Z. Wu, X. Wang, A. He, J. Xu and J. Xu, State-of-the-art advances and perspectives in the separation of biomass-derived 5-hydroxymethylfurfural, J. Clean. Prod., 2020, 276, 124219, DOI:10.1016/j.jclepro.2020.124219.
- D. W. Rackemann and W. O. S. Doherty, The conversion of lignocellulosics to levulinic acid, Biofuels Bioprod. Bioref., 2011, 5, 198–214, DOI:10.1002/bbb.267.
- R. J. Van Putten, J. C. van der Waal, E. de Jong, C. B. Rasrendra, H. J. Heeres and J. G. de Vries, Hydroxymethylfurfural, a versatile platform chemical made from renewable resources, Chem. Rev., 2013, 113, 1499–1597, DOI:10.1021/cr300182k.
- F. Constant, S. Jourdain, C. Esposito, J. Degrève and B. Delmon, The origin, composition, and applications of industrial humins – a review, Green Chem., 2025, 27, 3136–3166, 10.1039/D4GC06244B.
- T. Tongtummachat, A. Jaree and N. Akkarawatkhoosith, Conversion of glucose to 5-hydroxymethylfurfural in a microreactor, Front. Chem., 2020, 7, 951, DOI:10.3389/fchem.2019.00951.
- A. Mukherjee, M. J. Dumont and V. Raghavan, Review on the production of 5-hydroxymethylfurfural (HMF) from biomass, Carbohydr. Res., 2015, 402, 23–37, DOI:10.1016/j.carres.2014.09.008.
- X. Qi, M. Watanabe, T. M. Aida and R. L. Smith Jr, Catalytic conversion of carbohydrates into 5-hydroxymethylfurfural in high-temperature water, Green Chem., 2008, 10, 799–805, 10.1039/B804703K.
- W. Guo, H. J. Heeres and J. Yue, Continuous synthesis of 5-hydroxymethylfurfural from glucose using a combination of AlCl3 and HCl as catalyst in a biphasic slug flow capillary microreactor, Chem. Eng. J., 2020, 381, 122754, DOI:10.1016/j.cej.2019.122754.
- W. Guo, Z. Zhang, J. Hacking, H. J. Heeres and J. Yue, Selective fructose dehydration to 5-hydroxymethylfurfural from a fructose–glucose mixture over a sulfuric acid catalyst in a biphasic system: Experimental study and kinetic modelling, Chem. Eng. J., 2021, 409, 128182, DOI:10.1016/j.cej.2020.128182.
| This journal is © The Royal Society of Chemistry 2026 |




