 Open Access Article
Open Access ArticleNitric oxide-releasing hyaluronic acid as an antibacterial and immunomodulatory acne therapeutic
Samantha L. Picciottia,
Torrance M. Jenkinsa,
Tsian D. Ramrattana and
Mark H. Schoenfisch *ab
*ab
aDepartment of Chemistry, University of North Carolina at Chapel Hill, Chapel Hill, North Carolina 27599, USA. E-mail: schoenfisch@unc.edu
bEshelman School of Pharmacy, University of North Carolina at Chapel Hill, Chapel Hill, North Carolina 27599, USA
First published on 6th February 2026
Abstract
Acne vulgaris (acne) is a common skin disorder associated with significant psychosocial impact. Current clinical therapies include topical and systemic antibiotics, benzoyl peroxide, and retinoids. While moderately effective, these clinical therapies fail to target all four major pathogenic causes of acne and are associated with painful side effects. Nitric oxide (NO), an endogenous signaling molecule, represents a promising alternative to conventional acne treatments due to its innate antibacterial and immunomodulatory functions. As NO is highly reactive, macromolecular NO donors are required for its controlled, solution-phase delivery. Prior work has utilized silica nanoparticle scaffolds to store and deliver NO, with the silica scaffold being considered inert. Herein, NO-releasing hyaluronic acid (HA), an endogenously produced biopolymer, was modified with NO donors to enable a dual-action therapeutic capable of addressing the pathogenic factors responsible for acne development. The molecular weight of these HA derivatives proved important with respect to bactericidal activity against Cutibacterium acnes and ability to modulate keratinocyte proliferation, sebum production, and inflammation.
1. Introduction
Acne vulgaris (acne) is a chronic inflammatory disorder of the skin that affects more than 85% of adolescents and young adults, making it the most common skin disorder in the United States.1–3 While acne is not life-threatening, it has direct association with profound psychosocial effects.4 Those that experience severe acne often suffer from low self-esteem, anxiety, and depression, all of which heavily impact quality of life.4–6 In addition, acne poses a significant financial burden on both patients and healthcare systems, with annual costs incurring over $3 billion in the United States.6,7Characteristic acne lesions (e.g., open and closed comedones, papules, cysts) result from obstruction and inflammation within the pilosebaceous units (i.e., hair follicles and their accompanying sebaceous glands).2,3,8 Pilosebaceous units are primarily concentrated on the face, chest, and back, and as such, these areas are the most prone to acne development.2,3,8 Acne pathophysiology is multifactorial and involves the interplay of four key factors: (1) dysbiosis involving Cutibacterium acnes (C. acnes), (2) excessive keratinocyte proliferation within the pilosebaceous unit, (3) excessive production and altered composition of sebum, and (4) inflammation.3,5,9–13 Hormonal fluctuations resulting from several factors, including puberty, stress, diet, and genetics, stimulate excessive sebum production and contribute to hyperkeratinization.9–11,14 Sebum accumulation and irregular keratinocyte desquamation obstruct the pilosebaceous unit, thereby creating a low-oxygen, nutrient-rich environment ideal for C. acnes proliferation.10,14–16 Cutibacterium acnes is a major commensal bacterium of healthy skin; however, C. acnes evolves into an opportunistic pathogen upon homeostatic imbalance.13–15 The loss of diversity of individual C. acnes strains and alteration of the cutaneous microbiome are highly correlated with the pathogenesis of acne.13–15,17,18 Further, an inflammatory reaction fundamental to acne development is initiated from the interaction of C. acnes with sebum and skin cells within obstructed pilosebaceous units.3,5,15 Current conventional acne treatments (e.g., topical and systemic antibiotics, benzoyl peroxide, retinoids) primarily influence the skin microbiome, keratinocyte proliferation, and/or inflammation to modulate the visible symptoms of acne and are often prescribed in combination.3,5,19 Isotretinoin, a systemic retinoid, is the only approved treatment that targets all four pathogenic factors of acne.3,5,19 Unfortunately, these treatments suffer from low patient compliance due to perceptions of worsening acne, painful or severe side effects, and lengthy treatment regimens.3,5,6,19,20 Increasing antimicrobial resistance further limits the use of topical and oral antibiotics.21–24 Given the high prevalence of acne, the need for novel therapies that address the multiple pathogenic factors of acne while limiting off-target side effects is great.
Nitric oxide (NO), an endogenous signaling molecule, represents an attractive alternative to conventional acne treatments due to its innate antibacterial and immunomodulatory activities.25–28 The physiological roles of NO are concentration dependent, with high concentrations (i.e., nM–µM) eliciting broad-spectrum antibacterial activity via multiple mechanisms of action, including nitrosative and oxidative stresses induced by reactive byproducts of NO (e.g., peroxynitrite, dinitrogen trioxide) that elicit thiol nitrosation, DNA deamination, and bacterial membrane destruction through lipid peroxidation.25,29,30 Of importance, bacterial resistance to NO has not been observed in vitro,31,32 likely due to its multiple mechanisms of bactericidal action, small size, and/or high reactivity. At low concentrations (i.e., pM–nM), NO acts as an anti-inflammatory agent that can modulate immune cell function and inflammatory pathway activation to alter the release of inflammatory mediators.26,33,34 Low levels of NO are also believed to modulate sebum production by altering the proliferation and differentiation of sebocytes (i.e., sebaceous gland cells), as well as lipogenesis.35,36 Therefore, the native roles of endogenous NO in skin physiology have motivated the study of exogenous NO therapy for acne treatment.35–37
The high reactivity and short half-life of NO in biological milieu requires the use of chemical NO donors to modulate acne's pathogenic factors with exogenous NO.38–40 Chemical NO donors are attractive for exogenous delivery as they allow for controlled, solution-phase delivery of NO under physiological conditions.39,40 To date, chemical NO donors comprise both low molecular weight (i.e., small molecule) and macromolecular systems such as silica, dendrimers, and biopolymers.41–47 Often, macromolecular systems aid in mitigating toxicity.48 Silica-based nanoparticles that release NO have demonstrated promise as potential acne therapeutics, exhibiting the ability to modulate sebum production and C. acnes-induced inflammation by decreasing lipogenesis, clearing infection, and inhibiting innate immune response stimulation.35,36 While promising, the finite release of NO from such scaffolds leaves behind carriers that are considered inert and lack beneficial therapeutic attributes. The development of bioactive macromolecular NO-delivery scaffolds that can potentially further modulate the pathogenic factors of acne, even after NO release has ceased, is of great interest. Hyaluronic acid (HA), an endogenous biopolymer, represents an attractive NO-delivery scaffold as it is involved in key physiological processes, including cell signaling, wound healing, and tissue regeneration.49–53 As a drug delivery vehicle in the pharmaceutical and cosmetic industries, HA is favorable due to its physicochemical properties, including water solubility, biocompatibility, and biodegradability.49,52,54 Hyaluronic acid also promotes skin hydration, supports skin barrier function, and improves the appearance of scars, all of which may aid in acne management.52,54–57
Recently, we reported on NO-releasing HA biopolymers as unique NO-delivery vehicles for wound healing applications.46,47,58 Briefly, infected wounds treated with NO-releasing HA exhibited accelerated wound closure and decreased bacterial burden in a simple murine wound model compared to those treated with the vehicle or non-NO-releasing controls.46,47 Nitric oxide-releasing HA was also found to influence lipopolysaccharide (LPS)-stimulated macrophage phenotype and effector functions in vitro, highlighting its immunomodulatory activity.58 Given the promising antibacterial and immunomodulatory properties of NO-releasing HA as a wound healing agent, we sought to evaluate its potential as a multi-action acne therapeutic capable of addressing the pathogenic factors of acne. A series of HA molecular weights (i.e., 6 kDa, 90 kDa, 1000 kDa) were modified with secondary amine-containing alkylamine ligands and then reacted with NO gas at high pressures to form N-diazeniumdiolate NO donors. The resulting NO-releasing HA was utilized to elucidate the influence of both HA molecular weight and NO-release kinetics on modulation of bacterial viability, keratinocyte and sebocyte proliferation, sebum production, and toll-like receptor (TLR)-mediated inflammation.
2. Results and discussion
The biological functions of HA are molecular weight dependent, with high molecular weight HA (≥1 MDa) expressed in healthy tissue eliciting immunosuppressive and anti-inflammatory properties.59–61 In contrast, low molecular weight HA (1–800 kDa) is generally expressed in inflamed tissue and signals for immunostimulatory and pro-inflammatory action.59–61 In acne, HA has been shown to play a significant role in decreasing sebum production by regulating sebaceous gland size and lipid synthesis.62 However, the role of HA molecular weight in acne has not yet been explored. Due to the molecular weight-dependent properties of HA, a range of molecular weights were utilized for NO delivery.2.1 Synthesis and characterization of NO-releasing HA
Three molecular weights of HA, HA6 (9.0 ± 2.3 kDa, Đ = 1.7), HA90 (89.3 ± 8.0 kDa, Đ = 1.8), and HA1000 (1150.8 ± 161.0 kDa, Đ = 1.2) were modified with diethylenetriamine (DETA) or bis(3-aminopropyl)amine (DPTA) via carbodiimide chemistry, which resulted in both covalent and ionic modification. The amine-modified HA derivatives were then reacted with high pressures of NO gas under basic conditions to facilitate NO storage and tunable (via alkylamine structure) NO-release capabilities (Scheme S1).58 Following alkylamine modification, the molecular weights remained comparable to those of native HA (Table S1). Scanning electron microscopy (SEM) was used to evaluate the morphology of the unmodified and DETA-modified HA derivatives after hydration and subsequent lyophilization (Fig. S1). The representative SEM images of the unmodified HA6 and HA90 derivatives revealed porous structures, with porosity appearing to decrease after modification with DETA. Conversely, unmodified HA1000 was not porous, and modification with DETA had no influence on its morphology.Successful incorporation of the alkylamines was monitored via FTIR spectroscopy, 1H NMR spectroscopy, and CHN elemental analysis. The representative FTIR spectra revealed a shift in peak shape toward the carbonyl region (1650 cm−1) for all molecular weights, indicative of increased amide content (Fig. S2). The representative 1H NMR spectra revealed proton environments indicative of covalent modification around 3.39 and 2.40 ppm for the DETA- and DPTA-modified derivatives (Fig. S3–S11). The presence of unreacted alkylamine was also observed in each spectrum, supporting the hypothesis that the HA is both covalently and ionically modified with the alkylamines. Amine modification was further confirmed via CHN elemental analysis, which showed an increase in total nitrogen content from ∼3 wt% for unmodified HA to ∼8–10 wt% for the amine-modified HA derivatives (Table S2). The covalent modification efficiency and total amine content (i.e., covalently conjugated and ionically incorporated) were determined for the amine-modified HA derivatives using the integration ratios from the 1H NMR spectra and CHN elemental analysis, respectively.58 Covalent modification efficiency was estimated to be ∼20% for the HA6 derivatives, ∼11–14% for the HA90 derivatives, and ∼7–14% for the HA1000 derivatives, with total amine content for all derivatives between ∼50–70% (Table S3).
The molecular weight of the unmodified and DETA-modified HA derivatives was also studied over time to determine the short-term stability of the derivatives. After the initial molecular weight was determined, the HA derivatives were incubated at 37 °C to mimic physiological conditions, with analysis every 24 h. All unmodified and DETA-modified derivatives remained stable over a 72-h period, with no differences statistically at any time point from the initial starting molecular weight, highlighting the short-term stability of these materials (Fig. S12).
The release of NO was characterized in real time using a chemiluminescent nitric oxide analyzer (NOA; Fig. S13), with the NO-releasing HA derivatives dissolved in pH 7.4 phosphate-buffered saline (PBS) at 37 °C to mimic physiological conditions. The resulting NO payloads ranged from 0.30–0.60 µmol mg−1, corresponding to ∼15–30% conversion of secondary amines to N-diazeniumdiolate NO donors, with differences in functionalization efficiencies, NO totals, and NO-release kinetics dependent on the molecular weight of HA and alkylamine modification (Tables 1 and S4). Low molecular weight HA (i.e., HA6) exhibited significantly larger NO payloads than the higher molecular weight derivatives (i.e., HA90, HA1000) for both DETA and DPTA modifications. While not significant, NO payloads appeared to diminish as the molecular weight of HA was increased from 90 to 1000 kDa. During NO donor formation, NO gas may experience decreased accessibility to the HA scaffold for larger molecular weights, likely due to increasing chain entanglement and viscosity, with concomitant lower NO payloads.46 The DETA and DPTA alkylamines, differing only in alkyl chain length, were used to achieve tunable NO-release kinetics. The NO-releasing DETA-modified derivatives exhibited more sustained NO-release half-lives and durations than the NO-releasing DPTA-modified derivatives, attributable to enhanced N-diazeniumdiolate stabilization from the ethyl spacing of DETA versus the propyl spacing of DPTA.63 The molecular weight of HA also influenced the release durations, with the NO-releasing HA6 derivatives having the longest release durations, followed by the NO-releasing HA90 and HA1000 derivatives, respectively. Overall, the HA derivatives demonstrated tunable NO-release properties dependent on HA molecular weight and alkylamine modification.
| Material | [NO]tb (µmol mg−1) | t1/2c (min) | tdd (h) |
|---|---|---|---|
| a Error represents the standard deviation from n ≥ 3 separate syntheses.b Total NO released over full release duration.c Half-life of NO release.d Duration of NO release. *p < 0.05 for HA6 compared to HA90. ^p < 0.05 for HA6 compared to HA1000. †p < 0.05 for DETA modifications compared to DPTA modifications. | |||
| HA6-DETA/NO | 0.46 ± 0.07*,^,† | 41 ± 11† | 14.1 ± 2.2^ |
| HA90-DETA/NO | 0.32 ± 0.09 | 40 ± 9† | 10.9 ± 3.1† |
| HA1000-DETA/NO | 0.28 ± 0.05 | 30 ± 4 | 7.7 ± 1.1 |
| HA6-DPTA/NO | 0.63 ± 0.11*,^ | 24 ± 4 | 11.9 ± 1.4*,^ |
| HA90-DPTA/NO | 0.40 ± 0.05 | 22 ± 4 | 7.6 ± 1.5 |
| HA1000-DPTA/NO | 0.28 ± 0.09 | 24 ± 5 | 5.6 ± 1.4 |
2.2 In vitro antibacterial activity against C. acnes and S. aureus
Proliferation of C. acnes triggers the activation of the innate immune system and cutaneous inflammation. In addition, cutaneous microbiome dysbiosis is implicated in the pathophysiology of inflammatory acne and may lead to the proliferation of pathogenic species (e.g., Staphylococcus aureus; S. aureus) that further exacerbate inflammation.13,14,64 We thus sought to assess the antibacterial activity of NO-releasing HA against C. acnes and a methicillin-resistant S. aureus (MRSA) strain.The two Gram-positive bacterial species evaluated were treated with concentrations of unmodified, amine-modified, or NO-releasing HA ranging from 0.125–16 mg mL−1 for HA6 and HA90 derivatives and 0.125–8 mg mL−1 for HA1000 derivatives. The minimum bactericidal concentrations (MBCs), defined as the concentration of material required to induce a 3-log (i.e., 99.9%) reduction in viable bacterial colonies, were determined after 4 and 24 h of exposure. Of note, the treatment concentration for all HA1000 derivatives was limited by solution viscosity and solubility, with 8 mg mL−1 being the greatest concentration achievable for consistent HA delivery. As expected, control (i.e., both unmodified and amine-modified) HA derivatives did not elicit antibacterial activity over 24 h of treatment for either bacterial species, confirming that bactericidal activity from the NO-releasing HA was from the NO release rather than the HA scaffold (Table S5). The NO-releasing HA derivatives elicited antibacterial activity regardless of the NO donor modification, albeit to varying degrees (Table 2). Negligible antibacterial activity was observed from the NO-releasing derivatives after 4 h of treatment, as demonstrated by greater MBC4h values between 4 to greater than 16 mg mL−1 for both C. acnes and S. aureus. The observed antibacterial activity significantly increased between 4- and 24-h exposure, likely due to more complete release from the remaining NO donor after the 4-h time point given that both NO-releasing HA systems had release durations (i.e., td) greater than 4 h. For both strains, the MBC24h values ranged from 0.5–4 mg mL−1, whereby the NO-releasing HA6 derivatives required lower treatment concentrations to eradicate the bacteria compared to the NO-releasing HA90 and HA1000 derivatives. However, these molecular weight-dependent antibacterial properties were hypothesized to result from differences in NO loading, as the lower molecular weights achieved higher NO payloads (Table 1).
| Material | C. acnesb | S. aureusc | ||
|---|---|---|---|---|
| MBC4h (mg mL−1) | MBC24h (mg mL−1) | MBC4h (mg mL−1) | MBC24h (mg mL−1) | |
| a MBCs determined from n ≥ 3 separate experiments.b C. acnes treated in 2% v/v WCB in pH 7.4 PBS.c S. aureus treated in 2% v/v MHB in pH 7.4 PBS. | ||||
| HA6-DETA/NO | 4 | 0.5 | 8 | 1 |
| HA90-DETA/NO | 16 | 2 | >16 | 4 |
| HA1000-DETA/NO | 8 | 2 | >8 | 4 |
| HA6-DPTA/NO | 4 | 0.5 | 4 | 1 |
| HA90-DPTA/NO | 16 | 2 | 8 | 2 |
| HA1000-DPTA/NO | >8 | 4 | >8 | 4 |
To account for the variability in NO payloads and allow for comparisons between molecular weight and alkylamine modification, the MBC24h values were normalized to NO dose for both bacterial species. When comparing NO doses required for eradication, lower NO doses were needed for C. acnes versus S. aureus for all NO-releasing HA derivatives, highlighting the increased susceptibility of C. acnes to NO (Table S6). It is hypothesized that the increased susceptibility is due to the anaerobic nature of C. acnes. The NO dose was also evaluated as a function of molecular weight, with the NO-releasing HA6 derivatives requiring the lowest NO doses to achieve a 3-log reduction for both C. acnes and S. aureus. In contrast, the NO-releasing HA90 and HA1000 derivatives required higher NO doses that were comparable to each other. The lower molecular weight of HA6 may provide beneficial properties, including faster diffusion to the bacteria and/or increased bacterial localization for more efficient NO delivery.46,47 The identity of the alkylamine modification and resulting NO-release kinetics only influenced antibacterial activity toward C. acnes, with the DETA-modified derivatives requiring lower NO doses for eradication regardless of the HA molecular weight. This is likely a result of the slower NO-release kinetics associated with the DETA modification, suggesting that sustained release of NO is more effective against slower growing bacteria, such as C. acnes. This trend was not observed for S. aureus, indicating that differences in NO-release kinetics do not influence antibacterial activity against faster growing bacteria.
The antibacterial activity of NO-releasing HA was also assessed as a function of time via a time-kill assay to evaluate the rate of bactericidal action. For this assay, a greater starting concentration of bacteria was utilized to allow for colony counting. As such, a set dose of 8 mg mL−1 was used for all NO-releasing derivatives to ensure bacterial eradication. For both species, the NO-releasing HA6 derivatives reached bactericidal levels the fastest, followed by the NO-releasing HA90 and HA1000 derivatives, respectively, with slight differences in exposure time required based on alkylamine modification and bacterial strain (Fig. 1). Alkylamine modification did not strongly influence the rate of C. acnes killing (Fig. 1A and B). In contrast, derivatives modified with DPTA achieved a 3-log reduction faster than those modified with DETA against S. aureus, within 4 and 8 h for NO-releasing HA6 and HA90/HA1000, respectively (Fig. 1C and D). Both NO-releasing HA6 derivatives elicited complete eradication within 2 h against C. acnes compared to 4–8 h against S. aureus, corroborating the increased susceptibility of C. acnes to NO demonstrated in the MBC24h results. Together, the MBC and time-kill assays highlight the antibacterial properties of NO and suggest the promise of NO-releasing HA6 as a fast-acting antibacterial agent capable of modulating C. acnes and S. aureus proliferation.
2.3 In vitro cytotoxicity against human epidermal and sebaceous gland cells
The primary cells involved in the development of acne are keratinocytes and sebocytes, which are also the main cell types that comprise the epidermis and sebaceous glands, respectively.3,65 Keratinocytes play a crucial role in the development of acne, as their proliferation and abnormal desquamation leads to the formation of microcomedones, or clogged pores.12,65 Similarly, excessive proliferation and differentiation of sebocytes contributes to pore clogging through the overproduction of sebum.3,10 Both keratinocytes and sebocytes are immune-active cells involved in the inflammatory response of acne.65,66 As such, keratinocytes and sebocytes were used to evaluate the effects of HA and NO on cell proliferation and sebum production.While various clinical studies have demonstrated the safety of both HA and NO for dermatological applications separately, it is important to evaluate the cytotoxicity of the NO-releasing HA derivatives.67–70 The cytotoxicity of the unmodified, amine-modified, and NO-releasing HA derivatives was first evaluated against keratinocytes and sebocytes, which allowed for the determination of appropriate treatment concentrations for subsequent proliferation, sebum modulation, and inflammation assays. Keratinocytes and sebocytes were exposed to the DETA- and DPTA-modified HA derivatives of varying molecular weight for 24 h. After treatment, dose–response curves were generated to determine the concentration of material that reduces cellular metabolic activity by 50% (i.e., IC50) for each material (Fig. S14 and S15). While HA is considered a safe and biocompatible scaffold, the addition of amine-containing modifications and/or NO release has previously been shown to alter the native biocompatibility of the biopolymer.46,47,60,61 As expected, treatment with unmodified HA6, HA90, and HA1000 did not impact keratinocyte or sebocyte viability, with cell viability remaining greater than 80% up to the highest treatment concentrations tested and IC50 values of >16 mg mL−1 for HA6/HA90 and >8 mg mL−1 for HA1000 (Fig. 2).
Broadly, chemical modification of HA with DETA or DPTA did not impact keratinocyte or sebocyte viability, with corresponding IC50 values >16 mg mL−1 and >8 mg mL−1 for most of the amine-modified HA6/HA90 and HA1000 derivatives, respectively (Fig. 2). However, modification of HA6 with DETA and HA90 with both DETA or DPTA did elicit slight toxicity toward keratinocytes, as noted by IC50 values of 13.7 ± 0.7 mg mL−1 for HA6-DETA, 10.6 ± 0.3 mg mL−1 for HA90-DETA, and 12.6 ± 0.6 mg mL−1 for HA90-DPTA (Fig. 2A). The slight toxicity elicited upon alkylamine modification of HA6 and HA90 was not observed for the sebocytes, suggesting that the keratinocytes are more susceptible to these materials than the sebocytes (Fig. 2B). When loaded with NO, all HA derivatives exhibited a decrease in cell viability, with IC50 values for keratinocytes and sebocytes dropping to 1.5–5 mg mL−1 and 3.5–7 mg mL−1, respectively, highlighting that NO payloads are strongly influential to HA's toxicity profile (Fig. 2). Again, the NO-releasing HA derivatives elicited greater toxicity against keratinocytes, as observed through lower IC50 values compared to those of the sebocytes, further suggesting the increased susceptibility of keratinocytes to these materials. While there were overall trends of increasing IC50 values corresponding with increasing HA molecular weight, these trends are likely attributed to the decrease in NO payload as HA molecular weight is increased.
As the HA derivatives have variable NO payloads, the IC50 values were normalized to NO dose to allow for comparison between molecular weight and alkylamine modification for each cell line (Fig. S16). When comparing NO doses, slightly lower NO doses elicited greater toxicity toward the keratinocytes versus the sebocytes, again suggesting the increased susceptibility of the keratinocytes to NO. The molecular weight of HA had no influence on the cytotoxicity of the NO doses, as the NO doses were similar for all molecular weights. The identity of the alkylamine modification and resulting NO-release kinetics only influenced toxicity toward the sebocytes, with the DETA-modified derivatives eliciting toxicity at lower NO doses than the DPTA-modified derivatives, suggesting that sebocytes are more susceptible to the sustained release of NO. This trend was not observed for the keratinocytes, indicating that differences in NO-release kinetics have less of an effect on keratinocyte viability. Together, the resulting IC50 values informed the upper limit of treatment concentration for evaluation in the subsequent in vitro assays. Specifically, the proliferation, sebum modulation, and inflammation assays were performed using treatment concentrations with an upper limit of 1000 µg mL−1 to avoid significant cellular toxicity.
2.4 In vitro assessment of human epidermal and sebaceous gland cell proliferation
As stated previously, keratinocytes and sebocytes are heavily involved in the pathogenesis of acne, as their hyperproliferation contributes to microcomedone formation through abnormal desquamation and excess sebum production.3,10,12,65 As such, it is important to design therapeutics that can suppress aberrant cell proliferation, thereby reducing hyperkeratinization and sebum overproduction. To evaluate the effect of unmodified, amine-modified, and NO-releasing HA on keratinocyte and sebocyte proliferation, cells were treated once a day for 3 d using concentrations of material ranging from 1–1000 µg mL−1, as determined from the cytotoxicity assays. A lower cell seeding density and a 72-h incubation time were utilized to allow the cells sufficient space and time to proliferate. Cell proliferation relative to untreated control cells was determined after 72 h.Treatment with the unmodified, amine-modified, and NO-releasing HA derivatives at 1, 10, and 100 µg mL−1 did not significantly enhance or suppress keratinocyte proliferation, with relative proliferations ranging from ∼90–100% (Fig. 3A, C and E). However, treatment at 1000 µg mL−1 suppressed keratinocyte proliferation to varying degrees depending on HA molecular weight, alkylamine modification, and NO payload. Unmodified HA6 did not significantly suppress keratinocyte proliferation at 1000 µg mL−1, with the relative proliferation remaining at ∼90% (Fig. 3A). Modification of HA6 with DETA or DPTA significantly suppressed keratinocyte proliferation compared to unmodified HA6, with relative proliferations of ∼75%. The addition of NO release further suppressed keratinocyte proliferation compared to unmodified and amine-modified HA6, with the relative proliferation after treatment with HA6-DETA/NO and HA6-DPTA/NO at ∼45% and ∼0.5%, respectively. Similar trends were observed for keratinocytes treated with the HA90 and HA1000 derivatives at 1000 µg mL−1. The unmodified and amine-modified HA90 derivatives elicited significant suppression of keratinocyte proliferation, with proliferations relative to untreated cells at ∼70% (Fig. 3C). Again, the addition of NO release further suppressed keratinocyte proliferation compared to unmodified and amine-modified HA90, with the relative proliferation after treatment with HA90-DETA/NO and HA90-DPTA/NO at ∼60% and ∼40%, respectively. Of note, all unmodified, amine-modified, and NO-releasing HA1000 derivatives exhibited the same suppression of keratinocyte proliferation at 1000 µg mL−1, with relative proliferations of ∼50–60% (Fig. 3E). Sebocytes treated with the unmodified, amine-modified, and NO-releasing HA derivatives revealed the same concentration-, molecular weight-, alkylamine modification-, and NO release-dependent trends in proliferation as the keratinocytes (Fig. 3B, D and F). For both cell types, NO release played a significant role in the suppression of proliferation for the lower HA molecular weights (i.e., HA6, HA90), whereas at the higher HA molecular weight (i.e., HA1000), neither alkylamine modification nor NO release significantly altered the suppression of proliferation that unmodified HA elicited. This trend is likely due to the size of the biopolymer, as high molecular weight HA has immunosuppressive properties.52,60 Together, these results suggest that treatment with the NO-releasing HA derivatives does not promote excessive keratinocyte or sebocyte proliferation and even inhibits their proliferation at concentrations above 100 µg mL−1. Thus, NO-releasing HA represents a therapeutic capable of addressing the hyperkeratinization and excessive sebum production associated with acne.
2.5 In vitro modulation of sebum production
Increased sebum production is one of the most significant causes of acne development, as sebum provides an ideal anaerobic and nutrient-rich environment for C. acnes proliferation and subsequent innate immune stimulation.3 As sebocytes differentiate, they accumulate intracellular lipid droplets that are eventually released as sebum through holocrine secretion.10 To study the influence of HA and NO on sebocyte differentiation and lipid production, sebocytes were simultaneously stimulated with 1 µg mL−1 of insulin to induce differentiation and treated with 1–1000 µg mL−1 of the unmodified, amine-modified, or NO-releasing HA derivatives. The sebocytes were exposed to the insulin and test materials for 24 h prior to lipid quantification with Oil Red O, which is a dye for neutral, intracellular lipids. Cells stained with Oil Red O were imaged and the amount of Oil Red O per image, correlating to the amount of lipid accumulated, was quantified using ImageJ. Representative images of sebocytes stained with Oil Red O following treatment with 1000 µg mL−1 of the HA derivatives are shown in Fig. S17.At all concentrations tested, treatment with the unmodified, amine-modified, and NO-releasing HA derivatives modulated sebocyte differentiation and resulting lipid production, as all materials elicited a significant decrease in the amount of Oil Red O quantified compared to untreated, differentiated cells (Fig. 4 and S18). In general, the unmodified HA derivatives were most effective in modulating lipid production, with these derivatives exhibiting significantly less Oil Red O stain than some of their corresponding amine-modified counterparts and all of their corresponding NO-releasing counterparts, regardless of HA molecular weight or treatment concentration. It was expected that treatment with unmodified HA at these concentrations would attenuate sebum production, as Jung et al. previously demonstrated the functional role of high molecular weight HA in human sebaceous gland biology, specifically sebum production, in vitro and in vivo.62 Our results corroborate these previous findings and also confirm the role of low molecular weight HA in modulating sebum production. The addition of amine functionality did not strongly affect the native properties of unmodified HA, with all derivatives significantly decreasing lipid production for both DETA and DPTA modifications. Of interest, the amine-modified HA6 derivatives were significantly more effective than the HA90 and HA1000 derivatives at 1000 µg mL−1, while the amine-modified HA1000 derivatives were significantly more effective at 1 µg mL−1. Finally, NO release from the HA derivatives did significantly increase lipid production compared to the unmodified and amine-modified derivatives even though lipid production was modulated compared to the untreated control cells, suggesting that the HA backbone is most likely the major driver of the observed sebum-modulating properties. These results, together with the proliferation results, indicate that NO-releasing HA has the potential to modulate the aberrant sebocyte proliferation and differentiation characteristic of acne.
2.6 In vitro modulation of TLR-mediated inflammation
The proliferation of C. acnes produces a multitude of biologically active molecules (e.g., enzymes, short-chain fatty acids) that stimulate immune cells and skin cells within the pilosebaceous unit to secrete pro-inflammatory cytokines, initiating a strong inflammatory response.65 These biologically active molecules are identified by toll-like receptors (TLR) 2 and 4, which are overly expressed on the surface of keratinocytes, sebocytes, and tissue macrophages in acne lesions.5,65 The interaction of C. acnes with TLRs stimulates nuclear factor kappa B (NF-κB), a family of transcription factor proteins that activate the genes that produce pro-inflammatory cytokines and other signaling molecules, thereby triggering inflammation.65,66,71 Indeed, the NF-κB pathway serves as a key modulator of inflammation.Two NF-κB-induced secreted embryonic alkaline phosphatase (SEAP) reporter cell lines were utilized to evaluate the ability of unmodified, amine-modified, and NO-releasing HA derivatives to modulate TLR-mediated NF-κB inflammatory pathway activation. The two types of human embryonic kidney (HEK) 293 cells, which have NF-κB-induced SEAP expression and only express either TLR2 or TLR4, were treated with 1–1000 µg mL−1 of the HA derivatives for 24 h prior to quantifying the amount of SEAP expressed. The amount of SEAP activity is thus directly proportional to the strength of NF-κB activation. To stimulate the TLRs and induce NF-κB activation, 100 ng mL−1 of lipopolysaccharides (LPS) was added concurrently with the HA treatment.
The unmodified and amine-modified HA derivatives were first evaluated to understand the contributions of HA on modulation of TLR-mediated NF-κB activation. In general, the unmodified and amine-modified HA derivatives did not exhibit the ability to modulate TLR2- or TLR4-mediated inflammatory responses activated by LPS stimulation at any concentration, as revealed by SEAP expressions comparable to that of the LPS-stimulated control cells (Fig. S19 and S20). Similarly, the NO-releasing HA derivatives also did not influence NF-κB activation for the TLR2 or TLR4 cells at 1, 10, or 100 µg mL−1 treatment concentrations (Fig. S21). However, increasing the concentration of NO-releasing HA to 1000 µg mL−1 significantly decreased activation of the NF-κB inflammatory pathway for some derivatives (Fig. 5). Specifically, HA90-DPTA/NO and HA1000-DPTA/NO exhibited a significant decrease in SEAP concentration for the TLR2 cells, correlating with a significant decrease in TLR2-mediated NF-κB activation compared to the LPS-stimulated control (Fig. 5A). Of interest, HA6-DETA/NO, HA6-DPTA/NO, HA90-DETA/NO, and HA90-DPTA/NO all exhibited a significant decrease in SEAP concentration for the TLR4 cells, suggesting that NO released from lower molecular weights of HA (i.e., HA6, HA90) is more influential in modulating NF-κB activation through TLR4 than TLR2 (Fig. 5B). Differences in the ability of NO-releasing HA to modulate inflammation through interactions with TLR2 and TLR4 were expected, as HA has a stronger binding affinity for TLR4 compared to TLR2.72 Together, these results highlight NO's anti-inflammatory properties and potential to regulate TLR-mediated inflammation in acne.
2.7 Limitations
While the in vitro experiments presented herein demonstrate the promise of NO-releasing low molecular weight HA as an alternative acne therapeutic, in vivo validation is an essential next step to further confirm its translational potential. In vivo acne models are valuable for understanding acne pathogenesis and evaluating the efficacy of anti-acne therapies under physiological complexity. Current in vivo murine models emulate human acne via multiple pathogenic mechanisms, inducing inflammatory acne-like lesions through the intradermal injection of C. acnes and subsequent topical application of synthetic sebum.73,74 However, skin complexity (i.e., pH, enzymes) may alter the function of NO-releasing HA due to increased HA degradation and premature NO release. As such, the development of a topical formulation is also an essential next step to optimize the stability and bioavailability of NO-releasing HA for in vivo validation. For future in vivo studies, mice will be intradermally injected with 1 × 107 CFU of C. acnes prior to the application of freshly made synthetic sebum. Once acne lesions form, the mice will be treated with NO-releasing low molecular weight HA in a topical formulation once and twice daily for 14 d, with the non-NO-releasing HA and vehicle formulation as controls. Acne lesions will be visually monitored each day and scored based on skin pathology to evaluate effectiveness and irritation. At the end of the study, tissue from the acne lesions will be harvested and used for cytokine, bacterial burden, and histological analysis. Cytokine profiles and bacterial burdens will be analyzed via Luminex and quantitative polymerase chain reaction (qPCR) analysis, respectively, to evaluate the anti-inflammatory and antibacterial properties of NO-releasing HA in vivo.47,75 Hematoxylin and eosin (H&E) staining will also be performed on the tissue sections to evaluate changes in inflammation and inflammatory cell recruitment, epidermal/follicular wall thickness, and cystic structures with keratinized plugs. Together, these future in vivo studies will provide a more complete understanding of the influence of NO-releasing HA on acne, increasing its translational potential.3. Conclusion
Hyaluronic acid was modified with N-diazeniumdiolate NO donors to evaluate its potential as a multi-action acne therapeutic capable of addressing bacterial infection, keratinocyte proliferation, sebum production, and inflammation. While all NO-releasing HA derivatives demonstrated antibacterial activity against C. acnes and S. aureus, the NO-releasing HA6 derivatives demonstrated superior antibacterial activity compared to the NO-releasing HA90 and HA1000 derivatives, with bactericidal concentrations as low as 0.5 mg mL−1. The biocompatibility of native HA was not altered after alkylamine modification, as keratinocyte and sebocyte viability remained high. Of importance, antibacterial doses of NO led to only minimal cytotoxicity to keratinocytes and sebocytes. Cell proliferation results revealed that treatment at antibacterial concentrations suppressed keratinocyte and sebocyte proliferation, suggesting that these materials may modulate hyperkeratinization and sebum production. Both control (i.e., unmodified or amine-modified) and NO-releasing HA derivatives elicited a decrease in lipid production within sebocytes. However, the non-NO-releasing derivatives proved to be the most effective, highlighting the role of the HA scaffold in sebum modulation. Additionally, the NO-releasing HA derivatives exhibited anti-inflammatory properties through interactions with TLR2 and TLR4, suppressing activation of the NF-κB pathway.Together, these in vitro results demonstrate the promise of NO-releasing low molecular weight HA (i.e., HA6) as an alternative acne therapeutic. The dual activity of NO and HA provided the ability to target all four major pathogenic causes of acne with a single therapeutic. To further evaluate the therapeutic potential of low molecular weight NO-releasing HA, future studies should utilize an in vivo acne model, as physiological complexity may alter the function of NO-releasing HA.
4. Experimental section/methods
4.1 Materials
Ultra-low (<6 kDa; HA6), extra-low (80–110 kDa; HA90), and high (1.0–1.5 MDa; HA1000) molecular weight (MW) HA was purchased from Lotioncrafter (Eastsound, WA). Diethylenetriamine (DETA), bis(3-aminopropyl)amine (DPTA), N-hydroxysuccinimide (NHS), deuterium oxide (D2O), 2-propanol, lipopolysaccharides (LPS) from Escherichia coli O127:B8, adenine hydrochloride hydrate, hydrocortisone, insulin from bovine pancreas, cholera toxin, bovine serum albumin (BSA), Wilkins-Chalgren anaerobic agar (WCA), Oil Red O (ORO), and resazurin sodium salt were purchased from MilliporeSigma (St. Louis, MO). Hydrochloric acid (HCl), sodium methoxide (NaOMe; 5.4 M in methanol), fetal bovine serum (FBS), antibiotic–antimycotic (100×), penicillin–streptomycin (PS), human recombinant epidermal growth factor (EGF), high glucose Dulbecco's Modified Eagle Medium (DMEM) without pyruvate, low glucose DMEM with pyruvate, Ham's F-12 nutrient mix, 0.05% trypsin, keratinocyte serum-free medium, Wilkins-Chalgren anaerobic broth (WCB), 10% buffered formalin, and common laboratory salts and solvents were purchased from Thermo Fisher Scientific (Waltham, MA). Poly-L-lysine was purchased from R&D Systems (Minneapolis, MN). Normocin, HEK-Blue selective antibiotics, HEK-Blue-hTLR2 cells, HEK-Blue-hTLR4 cells, and QUANTI-Blue solution were purchased from InvivoGen (San Diego, CA). 1-Ethyl-3-(3-dimethylaminopropyl)carbodiimide hydrochloride (EDC) was purchased from TCI America (Portland, OR). Cellulose ester dialysis membranes, tryptic soy broth (TSB), tryptic soy agar (TSA), and cation-adjusted Mueller Hinton II broth (MHB) were purchased from VWR (Radnor, PA). Human epidermal keratinocytes (HEKs; HEK001) were purchased from the UNC Tissue Culture Facility. Immortalized human sebaceous gland cells (sebocytes; SEB-1) were donated by Professor Amanda Nelson from the Department of Dermatology at Pennsylvania State University College of Medicine (Hershey, PA). The Cutibacterium acnes (C. acnes) laboratory reference strain ATCC 11828 (type II phylotype, ribotype 2), isolated from an inflammatory subcutaneous abscess, was purchased from the American Type Culture Collection (ATCC; Manassas, VA). Staphylococcus aureus (S. aureus; LAC) was donated by Professor Tony Richardson from the Department of Microbiology and Molecular Genetics at the University of Pittsburgh (Pittsburgh, PA). Argon (Ar), carbon dioxide (CO2), nitrogen (N2), oxygen (O2), anaerobic medical mix (5% CO2, 10% hydrogen, balance N2), NO calibration (25.87 ppm balance N2), and pure NO (99.5%) gas cylinders were purchased from Airgas National Welders (Raleigh, NC). Unless otherwise specified, reagents were used as received without further purification. Phosphate-buffered saline (PBS) was used at 10 mM, pH 7.4, and 37 °C. Distilled water was purified to a resistivity of 18.2 MΩ cm and a total organic content of <6 ppb using a Millipore Milli-Q UV Gradient A10 system (Bedford, MA).4.2 Synthesis of amine-modified and NO-releasing HA
Modification of HA (HA6, HA90, and HA1000) with either DETA or DPTA was achieved following previously published protocols.46,47,58 Briefly, HA (1 g) was dissolved in 40, 100, or 330 mL of distilled water for HA6, HA90, and HA1000, respectively. After, EDC (2024 mg) and NHS (1215 mg) were added in a 4× molar excess, with respect to the monomer carboxylic acid groups, followed by 1 mL of 0.5 M HCl. The reaction solution was stirred for 20 min at room temperature to allow for carboxylic acid activation before a 4× molar excess of DETA (1141 µL) or DPTA (1475 µL) was added. The reaction solution was then stirred for an additional 72 h at room temperature. Amine-modified HA6 and HA90 were collected via precipitation in ethanol and centrifugation, washed with ethanol, and dried under vacuum overnight. Amine-modified HA1000 was purified via dialysis against distilled water for 48 h, with 3× daily water changes, frozen at −80 °C, and collected via lyophilization.N-Diazeniumdiolate NO donors were formed on the secondary amines of amine-modified HA following previously published protocols.46,47,58 Briefly, HA6 and HA90 derivatives (150 mg) were dissolved in an 8![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 or 7
2 or 7![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3 methanol/water solution (10 mL), respectively. The amine-modified HA1000 derivatives (100 mg) were dissolved in a 1
3 methanol/water solution (10 mL), respectively. The amine-modified HA1000 derivatives (100 mg) were dissolved in a 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 methanol/water solution (10 mL). An 8× molar excess of NaOMe (500 µL for HA6/HA90, 333 µL for HA1000) was added to all solutions before they were transferred to the stainless-steel reaction vessels of an HEL ChemScan II High Pressure Parallel Reactor (Herts, England). The reaction vessels were purged with argon for three short (10 s) and three long (10 min) purges at 100 psi to remove oxygen. The reaction vessels were then pressurized to 290 psi with NO gas under continuous magnetic stirring for 72 h for N-diazeniumdiolate formation. After 72 h, unreacted NO was removed from the reaction vessels following the previous argon purging protocol. The NO-releasing HA derivatives (HAMW-amine/NO) were precipitated in ethanol, collected via centrifugation, dried overnight under vacuum, and stored in vacuum-sealed bags at −20 °C prior to use.
1 methanol/water solution (10 mL). An 8× molar excess of NaOMe (500 µL for HA6/HA90, 333 µL for HA1000) was added to all solutions before they were transferred to the stainless-steel reaction vessels of an HEL ChemScan II High Pressure Parallel Reactor (Herts, England). The reaction vessels were purged with argon for three short (10 s) and three long (10 min) purges at 100 psi to remove oxygen. The reaction vessels were then pressurized to 290 psi with NO gas under continuous magnetic stirring for 72 h for N-diazeniumdiolate formation. After 72 h, unreacted NO was removed from the reaction vessels following the previous argon purging protocol. The NO-releasing HA derivatives (HAMW-amine/NO) were precipitated in ethanol, collected via centrifugation, dried overnight under vacuum, and stored in vacuum-sealed bags at −20 °C prior to use.
4.3 Characterization of amine-modified HA
The molecular weight determination of unmodified and amine-modified HA derivatives was performed using an aqueous gel permeation chromatography (GPC) system equipped with dual Shodex OHpak LB-806M polyhydroxy-methacrylate columns (New York, NY), a Waters 2414 refractive index detector (Milford, MA), and a Wyatt miniDawn TREOS multi-angle light scattering detector (Santa Barbara, CA). The 0.1 M phosphate buffer (PB) mobile phase was adjusted to pH 7.0 and supplemented with 0.1 M sodium nitrate and 0.02 wt% sodium azide. The HA samples were dissolved in the mobile phase at 1 mg mL−1 and filtered with 0.22 µm hydrophilic polyvinylidene fluoride (PVDF) filters prior to analysis. For short-term molecular weight stability, molecular weight was monitored every 24 h for 72 h, with incubation at 37 °C in between time points to mimic physiological conditions. The morphology of unmodified and amine-modified HA was evaluated via scanning electron microscopy (SEM). The HA derivatives were dissolved in distilled water at 20 mg mL−1, frozen at −80 °C, and lyophilized prior to imaging on a Hitachi S-4700 Cold Cathode Field Emission Scanning Electron Microscope (Tokyo, Japan) at 2 kV. Attenuated total reflectance Fourier transform infrared spectroscopy (FTIR) was performed using a Thermo Fisher Scientific Nicolet iS50 FTIR spectrometer (Waltham, MA). Proton nuclear magnetic resonance (1H NMR) spectra were recorded on a Bruker Avance NEO 400 MHz spectrometer (Billerica, MA) with a relaxation delay of 5 s. Chemical shifts are reported in parts per million (ppm) using the resonance of the deuterated solvent as an internal standard. Elemental (carbon, nitrogen, hydrogen; CHN) analysis was performed on a PerkinElmer 2400 Series II CHNS/O Analyzer (Waltham, MA). The unmodified and amine-modified HA derivatives (∼1 mg) were encapsulated in tin capsules prior to analysis. Covalently bound modification efficiency was determined from the integration ratios of the 1H NMR spectra using eqn (1), where Imod, Iref, and Nref represent the integration of the alkylamine-modified proton signal (i.e., ∼2.40 ppm), the integration of the unchanged HA reference signal, and the number of protons represented by the unchanged reference peak, respectively. The value 6 represents the theoretical number of protons in the alkylamine-modified signal.
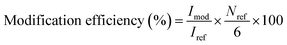 | (1) |
4.4 Characterization of NO-release properties
The real-time release of NO from HA derivatives was quantified using a Sievers 280i nitric oxide analyzer (NOA; Boulder, CO). The NOA was calibrated with air passed through a zero NO filter (0 ppm of NO) and 25.87 ppm of NO calibration gas (balance N2) prior to analysis. The NO-releasing HA derivatives (∼1 mg) were dissolved in 30 mL of deoxygenated PBS at 37 °C, and the liberated NO was carried to the NOA at 200 mL min−1 via nitrogen gas. Analysis was ended when NO release fell below the limit of quantification of the instrument (i.e. 10 ppb).4.5 Planktonic bactericidal assays
Cutibacterium acnes bacterial cultures were grown from frozen stocks (−80 °C) in WCB (5 mL) at 37 °C under anaerobic conditions for 48 h. A 1 mL aliquot of the bacterial solution was resuspended in 8 mL of fresh WCB and incubated under anaerobic conditions until reaching 1 × 108 CFU mL−1. The bacterial solution was subsequently diluted to 5 × 106 CFU mL−1 in 2% v/v WCB in pH 7.4 PBS (2 mL) prior to use. Staphylococcus aureus bacterial cultures were grown from frozen stocks (−80 °C) on TSA plates. Isolated colonies were grown overnight under vigorous shaking (250 rpm) in TSB (5 mL) at 37 °C. Overnight cultures were diluted 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 50 in fresh TSB (5 mL), grown to a concentration of 1 × 108 CFU mL−1, and subsequently diluted to 5 × 106 CFU mL−1 in 2% v/v MHB in pH 7.4 PBS (2 mL). Unmodified, amine-modified, and NO-releasing HA derivatives were dissolved in either 2% v/v WCB (C. acnes) or 2% v/v MHB (S. aureus) and pH adjusted to 7.4 with 5 M HCl. The treatments were added to the wells of 96-well plates and serially diluted 1
50 in fresh TSB (5 mL), grown to a concentration of 1 × 108 CFU mL−1, and subsequently diluted to 5 × 106 CFU mL−1 in 2% v/v MHB in pH 7.4 PBS (2 mL). Unmodified, amine-modified, and NO-releasing HA derivatives were dissolved in either 2% v/v WCB (C. acnes) or 2% v/v MHB (S. aureus) and pH adjusted to 7.4 with 5 M HCl. The treatments were added to the wells of 96-well plates and serially diluted 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2, resulting in wells containing 100 µL of 0.125–16 mg mL−1 of treatment for HA6 and HA90 derivatives and 0.125–8 mg mL−1 of treatment for HA1000 derivatives. After, 10 µL of the 5 × 106 CFU mL−1 bacterial solution (C. acnes or S. aureus) was added to each well. The 96-well plates were incubated anaerobically or aerobically for C. acnes and S. aureus, respectively, at 37 °C for 24 h. Untreated bacterial solutions were included for each plate to ensure bacterial viability over the 24-h duration. After 4 and 24 h, 10 µL aliquots from each well were plated on WCA (C. acnes) or TSA (S. aureus) plates and incubated for 72 h for C. acnes or overnight for S. aureus at 37 °C. The minimum bactericidal concentration after 4- and 24-h exposure (MBC4h, MBC24h) was defined as the minimum concentration in which no bacterial growth was present on the agar.
2, resulting in wells containing 100 µL of 0.125–16 mg mL−1 of treatment for HA6 and HA90 derivatives and 0.125–8 mg mL−1 of treatment for HA1000 derivatives. After, 10 µL of the 5 × 106 CFU mL−1 bacterial solution (C. acnes or S. aureus) was added to each well. The 96-well plates were incubated anaerobically or aerobically for C. acnes and S. aureus, respectively, at 37 °C for 24 h. Untreated bacterial solutions were included for each plate to ensure bacterial viability over the 24-h duration. After 4 and 24 h, 10 µL aliquots from each well were plated on WCA (C. acnes) or TSA (S. aureus) plates and incubated for 72 h for C. acnes or overnight for S. aureus at 37 °C. The minimum bactericidal concentration after 4- and 24-h exposure (MBC4h, MBC24h) was defined as the minimum concentration in which no bacterial growth was present on the agar.
4.6 Time-based planktonic bactericidal assays
Bacterial solutions containing 1 × 108 CFU mL−1 of C. acnes or S. aureus were prepared as described above and diluted to 1 × 107 CFU mL−1 in 2% v/v WCB or 2% v/v MHB, respectively (5 mL). The NO-releasing HA derivatives were dissolved in 2% v/v WCB or 2% v/v MHB at 16 mg mL−1 and pH adjusted to 7.4 with 5 M HCl. Aliquots (500 µL) were added to the wells of 24-well plates, followed by an equivalent volume of the 1 × 107 CFU mL−1 bacterial solution to each well (1 mL total volume) to bring the final concentration of NO-releasing HA derivatives to 8 mg mL−1. The 24-well plates were incubated anaerobically or aerobically for C. acnes and S. aureus, respectively, at 37 °C for 12 h. Untreated bacterial solutions were included to ensure bacterial viability over the 12-h duration. At discrete time points (i.e., 0, 2, 4, 8, and 12 h), aliquots (25 µL) were removed from the wells, serially diluted 10 to 100![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000-fold, plated on either WCA (C. acnes) or TSA (S. aureus) plates, and incubated at 37 °C for 72 or 24 h for C. acnes or S. aureus, respectively. Bacterial viability was determined via colony counting.
000-fold, plated on either WCA (C. acnes) or TSA (S. aureus) plates, and incubated at 37 °C for 72 or 24 h for C. acnes or S. aureus, respectively. Bacterial viability was determined via colony counting.
4.7 In vitro cytotoxicity assays
Human epidermal keratinocytes were grown in keratinocyte serum-free medium supplemented with 1 wt% PS and 5 ng mL–1 human recombinant EGF. Human sebaceous gland cells were grown in a 3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 low glucose DMEM to Ham's F-12 mixture supplemented with 2.5% v/v FBS, 24 µg mL–1 adenine hydrochloride hydrate, 45.2 ng mL–1 hydrocortisone, 10 ng mL–1 insulin, 3 ng mL–1 human recombinant EGF, 1.2 × 10−10 M cholera toxin, and 1 wt% antibiotic–antimycotic (penicillin/streptomycin/amphotericin B). All cells were incubated in 5% v/v CO2 at 37 °C until 80% confluence was reached. For both HEKs and SEB-1s, cell suspensions of 5 × 105 cells mL–1 were prepared, and aliquots (100 µL) were added to the wells of 96-well plates. The HEK and SEB-1 plates were then incubated for 24 h to allow the cells to adhere before the supernatant was removed and replaced with either unmodified, amine-modified, or NO-releasing HA derivatives (100 µL) in fresh media (pH adjusted to 7.4 with 1 M HCl). Test media consisting of low glucose DMEM and 1 wt% antibiotic–antimycotic was used for SEB-1 treatment. Treatment concentrations ranged from 0.03–16 mg mL−1 for HA6 and HA90 derivatives and 0.03–8 mg mL−1 for HA1000 derivatives. After 24-h exposure to the treatments, 0.1 mg mL–1 resazurin sodium salt solution in sterile distilled water (20 µL) was added to each well and incubated for 4 h before the fluorescence (Fl) of the solution in each well was measured at 544 nm excitation and 590 nm emission wavelengths using a Molecular Devices SpectraMax M2 spectrophotometer (San Jose, CA). The cell viability for each sample was calculated using eqn (2), where media/resazurin and untreated cells/resazurin served as the blank and control, respectively.
1 low glucose DMEM to Ham's F-12 mixture supplemented with 2.5% v/v FBS, 24 µg mL–1 adenine hydrochloride hydrate, 45.2 ng mL–1 hydrocortisone, 10 ng mL–1 insulin, 3 ng mL–1 human recombinant EGF, 1.2 × 10−10 M cholera toxin, and 1 wt% antibiotic–antimycotic (penicillin/streptomycin/amphotericin B). All cells were incubated in 5% v/v CO2 at 37 °C until 80% confluence was reached. For both HEKs and SEB-1s, cell suspensions of 5 × 105 cells mL–1 were prepared, and aliquots (100 µL) were added to the wells of 96-well plates. The HEK and SEB-1 plates were then incubated for 24 h to allow the cells to adhere before the supernatant was removed and replaced with either unmodified, amine-modified, or NO-releasing HA derivatives (100 µL) in fresh media (pH adjusted to 7.4 with 1 M HCl). Test media consisting of low glucose DMEM and 1 wt% antibiotic–antimycotic was used for SEB-1 treatment. Treatment concentrations ranged from 0.03–16 mg mL−1 for HA6 and HA90 derivatives and 0.03–8 mg mL−1 for HA1000 derivatives. After 24-h exposure to the treatments, 0.1 mg mL–1 resazurin sodium salt solution in sterile distilled water (20 µL) was added to each well and incubated for 4 h before the fluorescence (Fl) of the solution in each well was measured at 544 nm excitation and 590 nm emission wavelengths using a Molecular Devices SpectraMax M2 spectrophotometer (San Jose, CA). The cell viability for each sample was calculated using eqn (2), where media/resazurin and untreated cells/resazurin served as the blank and control, respectively.
 | (2) |
4.8 In vitro cell proliferation assays
The HEKs and SEB-1s were grown under the previously described conditions until 80% confluence was reached. A cell suspension of 2.5 × 105 or 1 × 105 cells mL–1 was prepared for HEKs and SEB-1s, respectively, and aliquots (100 µL) were added to the wells of 96-well plates. After the plates were incubated for 24 h, the supernatant was removed and replaced with unmodified, amine-modified, or NO-releasing HA derivatives in fresh media (180 µL) at 1, 10, 100, and 1000 µg mL−1. The plates were incubated for 72 h, with treatment repeated at 24 and 48 h to achieve a multiday treatment. Following 72-h exposure, 0.1 mg mL–1 resazurin sodium salt solution in sterile distilled water (40 µL) was added to each well and incubated for 6 h before the fluorescence of the solution in each well was measured as previously described. The relative cell proliferation was calculated for each sample using eqn (3), where media/resazurin and untreated cells/resazurin served as the blank and control, respectively.
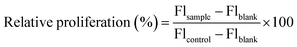 | (3) |
4.9 In vitro sebum modulation assays
Human sebocytes were grown under the previously described conditions until 80% confluence was reached. A cell suspension of 2.5 × 105 cells mL–1 was prepared, and aliquots (100 µL) were added to the wells of 96-well plates. The plates were incubated until cells reached 100% confluence within the wells. After, the supernatant was removed and replaced with 1, 10, 100, or 1000 µg mL−1 unmodified, amine-modified, or NO-releasing HA derivatives in SEB-1 test media containing 1 µg mL−1 of insulin to stimulate the cells (200 µL). Untreated cells with insulin stimulation served as controls. Following 24 h of concurrent insulin stimulation and treatment, the supernatant was removed and the cells were fixed for 30 min with 10% formalin (100 µL) in preparation for ORO staining. The cells were stained for 15 min with ORO (100 µL) before washing with 60% isopropanol (100 µL) and cold distilled water (100 µL) to remove excess stain prior to imaging. Wells were imaged with an Invitrogen EVOS XL Core Imaging System (Fair Lawn, NJ) using the 20× objective lens, with 5 images acquired per well. All materials were tested in triplicate, for a total of 15 images per treatment condition. The amount of stain per image was quantified using ImageJ software (Bethesda, MD).4.10 In vitro inflammation assays
HEK-Blue-hTLR2 and HEK-Blue-hTLR4 cells were grown in high glucose DMEM without pyruvate supplemented with 10% v/v FBS, 1 wt% PS, and 100 µg mL–1 Normocin antimicrobial reagent. The cells were incubated in 5% v/v CO2 at 37 °C until 80% confluence was reached. Cell suspensions of 4 × 105 cells mL–1 were prepared, and aliquots (100 µL) were added to the wells of 96-well plates and allowed to adhere overnight in a total of 200 µL of media. After, the supernatant was removed and replaced with 1, 10, 100, or 1000 µg mL−1 of unmodified, amine-modified, or NO-releasing HA derivatives in fresh media containing 100 ng mL−1 of LPS to stimulate the cells (200 µL). Cells without treatment or LPS stimulation and cells without treatment but with LPS stimulation served as negative and positive controls, respectively. After 24 h of concurrent HA treatment and LPS stimulation, cell supernatant (20 µL) from each well was removed and transferred to wells containing QUANTI-Blue solution (180 µL) to quantify secreted embryonic alkaline phosphatase (SEAP) activity. The plates were incubated for 1 h before the absorbance of the QUANTI-Blue solution was measured at 620 nm using a Molecular Devices SpectraMax M2 spectrophotometer (San Jose, CA).4.11 Statistical analysis
Weight-average molecular weight and dispersity, NO-release measurements, and NO donor functionalization are reported as the average ± standard deviation from n ≥ 3 separate syntheses. CHN elemental analysis and total amine content are reported as the average ± standard deviation from n = 3 separate syntheses. Bacterial time-kill results are presented as the average ± standard deviation from n ≥ 3 separate experiments. Cell viability results are presented as the average ± standard error of the mean from n ≥ 3 separate experiments with mammalian cells grown on different days. Normalized IC50 results are presented as the average ± standard deviation from n ≥ 3 separate experiments. Cell proliferation results are presented as the average ± standard deviation from n ≥ 3 separate experiments with mammalian cells grown on different days. Sebum modulation results are presented as the average ± standard deviation from n ≥ 15 images. The TLR activation results are presented as the average ± standard deviation from n = 3 separate samples. All statistical analyses were completed using GraphPad Prism 10 (San Diego, CA). Nonlinear regression (normalized response with variable slope) analysis was performed to determine IC50 values. Significance testing for NO totals, release durations, half-lives, IC50 values, NO dose values, and sebum production comparing HA molecular weight, and cell proliferation and sebum production comparing treatment concentrations, was performed via an ordinary one-way ANOVA with Tukey's correction for multiple comparisons. Significance testing for NO totals, release durations, half-lives, IC50 values, NO dose values, and sebum production comparing alkylamine modification, and cell proliferation and sebum production comparing unmodified, amine-modified, and NO-releasing derivatives, was performed via an ordinary one-way ANOVA with Šídák's correction for multiple comparisons. Significance testing for short-term molecular weight stability comparing molecular weight at all time points to initial molecular weight, sebum production comparing HA derivatives to insulin-stimulated cells, and TLR inflammation comparing HA derivatives to LPS-stimulated cells were performed via an ordinary one-way ANOVA with Dunnett's correction for multiple comparisons.Conflicts of interest
The corresponding author declares competing financial interest. M. H. Schoenfisch is a co-founder, is a member of the board of directors, and maintains a financial interest in KnowBIO, LLC. KnowBIO commercializes macromolecular nitric oxide storage and release scaffolds for the treatment of multiple diseases.Data availability
All data supporting this study are available within the article and supplementary information (SI). Supplementary information: HA modification schematic; weight-average molecular weight and dispersity; representative SEM images; FTIR spectra; 1H NMR spectra; elemental analysis; modification efficiency; short-term stability; real-time NO-release profiles; control MBC values; normalized NO dose values; dose-response curves for keratinocytes and sebocytes; representative stained sebocyte images; sebocyte lipid production; and TLR2/TLR4-mediated NF-κB activation. See DOI: https://doi.org/10.1039/d5ra08320f.Acknowledgements
This research was supported by the National Institutes of Health (DK132778). We thank Amanda Nelson, PhD for providing protocols and advice for the immortalized human sebaceous gland cell line.References
- S. Titus and J. Hodge, Diagnosis and Treatment of Acne, Am. Fam. Physician, 2012, 86, 734–740 Search PubMed.
- T. R. Walsh, J. Efthimiou and B. Dréno, Systematic Review of Antibiotic Resistance in Acne: An Increasing Topical and Oral Threat, Lancet Infect. Dis., 2016, 16, e22–e32 Search PubMed.
- M. Vasam, S. Korutla and R. A. Bohara, Acne Vulgaris: A Review of the Pathophysiology, Treatment, and Recent Nanotechnology Based Advances, Biochem. Biophys. Rep., 2023, 36, 101578 Search PubMed.
- M. Szepietowska, B. Bień, P. K. Krajewski, A. A. Stefaniak and Ł. Matusiak, Prevalence, Intensity and Psychosocial Burden of Acne Itch: Two Different Cohorts Study, J. Clin. Med., 2023, 12, 3997 CrossRef PubMed.
- S. Cruz, N. Vecerek and N. Elbuluk, Targeting Inflammation in Acne: Current Treatments and Future Prospects, Am. J. Clin. Dermatol., 2023, 24, 681–694 Search PubMed.
- K. A. Habeshian and B. A. Cohen, Current Issues in the Treatment of Acne Vulgaris, Pediatrics, 2020, 145, S225–S230 Search PubMed.
- M. Tassavor and M. J. Payette, Estimated Cost Efficacy of U.S. Food and Drug Administration-Approved Treatments for Acne, Dermatol. Ther., 2019, 32, e12765 Search PubMed.
- R. Nguyen and J. Su, Treatment of Acne Vulgaris, Paediatr. Child Health, 2011, 21, 119–125 CrossRef.
- Y. J. Bhat, I. Latief and I. Hassan, Update on Etiopathogenesis and Treatment of Acne, Indian J. Dermatol. Venereol. Leprol., 2017, 83, 298–306 CrossRef PubMed.
- J. Q. Del Rosso and L. Kircik, The Primary Role of Sebum in the Pathophysiology of Acne Vulgaris and Its Therapeutic Relevance in Acne Management, J. Dermatol. Treat., 2024, 35, 2296855 CrossRef PubMed.
- C. C. Zouboulis, Acne and Sebaceous Gland Function, Clin. Dermatol., 2004, 22, 360–366 CrossRef PubMed.
- D. M. Thiboutot, The Role of Follicular Hyperkeratinization in Acne, J. Dermatol. Treat., 2000, 11, 5–8 Search PubMed.
- B. Dréno, M. A. Dagnelie, A. Khammari and S. Corvec, The Skin Microbiome: A New Actor in Inflammatory Acne, Am. J. Clin. Dermatol., 2020, 21, S18–S24 CrossRef PubMed.
- C. Mayslich, P. A. Grange and N. Dupin, Cutibacterium Acnes as an Opportunistic Pathogen: An Update of Its Virulence-Associated Factors, Microorganisms, 2021, 9, 303 CrossRef CAS PubMed.
- T. X. Cong, D. Hao, X. Wen, X. H. Li, G. He and X. Jiang, From Pathogenesis of Acne Vulgaris to Anti-Acne Agents, Arch. Dermatol. Res., 2019, 311, 337–349 CrossRef CAS PubMed.
- M. J. Elston, J. P. Dupaix, M. I. Opanova and R. E. Atkinson, Cutibacterium acnes (Formerly Proprionibacterium acnes) and Shoulder Surgery, Hawai'i Journal of Health & Social Welfare, 2019, 78, 3–5 Search PubMed.
- B. Dréno, I. Dekio, H. Baldwin, A. L. Demessant, M. A. Dagnelie, A. Khammari and S. Corvec, Acne Microbiome: From Phyla to Phylotypes, J. Eur. Acad. Dermatol. Venereol., 2023, 38, 657–664 CrossRef PubMed.
- A. M. Schneider, Z. T. Nolan, K. Banerjee, A. R. Paine, Z. Cong, S. L. Gettle, A. L. Longenecker, X. Zhan, G. W. Agak and A. M. Nelson, Evolution of the Facial Skin Microbiome during Puberty in Normal and Acne Skin, J. Eur. Acad. Dermatol. Venereol., 2022, 37, 166–175 CrossRef PubMed.
- R. V. Reynolds, H. Yeung, C. E. Cheng, F. Cook-Bolden, S. R. Desai, K. M. Druby, E. E. Freeman, J. E. Keri, L. F. Stein Gold, J. K. L. Tan, M. M. Tollefson, J. S. Weiss, P. A. Wu, A. L. Zaenglein, J. M. Han and J. S. Barbieri, Guidelines of Care for the Management of Acne Vulgaris, J. Am. Acad. Dermatol., 2024, 90, 1006.e1–1006.e30 CrossRef PubMed.
- A. Tobiasz, D. Nowicka and J. C. Szepietowski, Acne Vulgaris—Novel Treatment Options and Factors Affecting Therapy Adherence: A Narrative Review, J. Clin. Med., 2022, 11, 7535 CrossRef CAS PubMed.
- B. L. Adler, H. Kornmehl and A. W. Armstrong, Antibiotic Resistance in Acne Treatment, JAMA Dermatol., 2017, 153, 810–811 CrossRef PubMed.
- H. Baldwin, Oral Antibiotic Treatment Options for Acne Vulgaris, Journal of Clinical and Aesthetic Dermatology, 2020, 13, 26–32 Search PubMed.
- S. Y. Chon, H. Q. Doan, R. M. Mays, S. M. Singh, R. A. Gordon and S. K. Tyring, Antibiotic Overuse and Resistance in Dermatology, Dermatol. Ther., 2012, 25, 55–69 CrossRef PubMed.
- A. S. Karadag, M. A. Kayıran, C. Y. Wu, W. Chen and L. C. Parish, Antibiotic Resistance in Acne: Changes, Consequences and Concerns, J. Eur. Acad. Dermatol. Venereol., 2021, 35, 73–78 Search PubMed.
- F. C. Fang, Perspectives Series: Host/Pathogen Interactions. Mechanisms of Nitric Oxide-Related Antimicrobial Activity, J. Clin. Invest., 1997, 99, 2818–2825 Search PubMed.
- G. Cirino, E. Distrutti and J. L. Wallace, Nitric Oxide and Inflammation, Inflammation Allergy:Drug Targets, 2006, 5, 115–119 Search PubMed.
- S. M. Andrabi, N. S. Sharma, A. Karan, S. M. S. Shahriar, B. Cordon, B. Ma and J. Xie, Nitric Oxide: Physiological Functions, Delivery, and Biomedical Applications, Adv. Sci., 2023, 10, e2303259 Search PubMed.
- S. H. Snyder and D. S. Bredt, Biological Roles of Nitric Oxide, Sci. Am., 1992, 266, 68–77 Search PubMed.
- A. W. Carpenter and M. H. Schoenfisch, Nitric Oxide Release: Part II. Therapeutic Applications, Chem. Soc. Rev., 2012, 41, 3742–3752 Search PubMed.
- E. M. Hetrick, J. H. Shin, N. A. Stasko, C. B. Johnson, D. A. Wespe, E. Holmuhamedov and M. H. Schoenfisch, Bactericidal Efficacy of Nitric Oxide-Releasing Silica Nanoparticles, ACS Nano, 2008, 2, 235–246 Search PubMed.
- R. A. McDonald, S. G. Nagy, M. Chambers, C. A. Broberg, M. J. R. Ahonen and M. H. Schoenfisch, Nitric Oxide-Releasing Prodrug for the Treatment of Complex Mycobacterium abscessus Infections, Antimicrob. Agents Chemother., 2024, 68, e0132723 Search PubMed.
- B. J. Privett, A. D. Broadnax, S. J. Bauman, D. A. Riccio and M. H. Schoenfisch, Examination of Bacterial Resistance to Exogenous Nitric Oxide, Nitric Oxide, 2012, 26, 169–173 CrossRef CAS PubMed.
- P. Tripathi, P. Tripathi, L. Kashyap and V. Singh, The Role of Nitric Oxide in Inflammatory Reactions, FEMS Immunol. Med. Microbiol., 2007, 51, 443–452 CrossRef CAS PubMed.
- F. S. Laroux, D. J. Lefer, S. Kawachi, R. Scalia, A. S. Cockrell, L. Gray, H. Van der Heyde, J. M. Hoffman and M. B. Grisham, Role of Nitric Oxide in the Regulation of Acute and Chronic Inflammation, Antioxid. Redox Signaling, 2000, 2, 391–396 CrossRef CAS PubMed.
- S. Bauman and N. Stasko, Methods of Decreasing Sebum Production in the Skin, US Pat., US8591876B2, 2013 Search PubMed.
- M. Qin, A. Landriscina, J. M. Rosen, G. Wei, S. Kao, W. Olcott, G. W. Agak, K. B. Paz, J. Bonventre, A. Clendaniel, S. Harper, B. L. Adler, A. E. Krausz, J. M. Friedman, J. D. Nosanchuk, J. Kim and A. J. Friedman, Nitric Oxide-Releasing Nanoparticles Prevent Propionibacterium acnes-Induced Inflammation by Both Clearing the Organism and Inhibiting Microbial Stimulation of the Innate Immune Response, J. Invest. Dermatol., 2015, 135, 2723–2731 CrossRef CAS PubMed.
- S. Settelmeier, T. Rassaf, U. B. Hendgen-Cotta and I. Stoffels, Nitric Oxide Generating Formulation as an Innovative Approach to Topical Skin Care: An Open-Label Pilot Study, Cosmetics, 2021, 8, 16 CrossRef CAS.
- T. S. Hakim, K. Sugimori, E. M. Camporesi and G. Anderson, Half-Life of Nitric Oxide in Aqueous Solutions with and without Haemoglobin, Physiol. Meas., 1996, 17, 267–277 CrossRef CAS PubMed.
- L. K. Keefer, Fifty Years of Diazeniumdiolate Research. From Laboratory Curiosity to Broad-Spectrum Biomedical Advances, ACS Chem. Biol., 2011, 6, 1147–1155 CrossRef CAS PubMed.
- K. M. Davies, D. A. Wink, J. E. Saavedra and L. K. Keefer, Chemistry of the Diazeniumdiolates. 2. Kinetics and Mechanism of Dissociation to Nitric Oxide in Aqueous Solution, J. Am. Chem. Soc., 2001, 123, 5473–5481 CrossRef CAS PubMed.
- J. H. Shin, S. K. Metzger and M. H. Schoenfisch, Synthesis of Nitric Oxide-Releasing Silica Nanoparticles, J. Am. Chem. Soc., 2007, 129, 4612–4619 Search PubMed.
- R. J. Soto, L. Yang and M. H. Schoenfisch, Functionalized Mesoporous Silica via an Aminosilane Surfactant Ion Exchange Reaction: Controlled Scaffold Design and Nitric Oxide Release, ACS Appl. Mater. Interfaces, 2016, 8, 2220–2231 Search PubMed.
- N. A. Stasko and M. H. Schoenfisch, Dendrimers as a Scaffold for Nitric Oxide Release, J. Am. Chem. Soc., 2006, 128, 8265–8271 Search PubMed.
- M. J. R. Ahonen, D. J. Suchyta, H. Zhu and M. H. Schoenfisch, Nitric Oxide-Releasing Alginates, Biomacromolecules, 2018, 19, 1189–1197 Search PubMed.
- Y. Lu, D. L. Slomberg and M. H. Schoenfisch, Nitric Oxide-Releasing Chitosan Oligosaccharides as Antibacterial Agents, Biomaterials, 2014, 35, 1716–1724 CrossRef CAS PubMed.
- S. E. Maloney, C. A. Broberg, Q. E. Grayton, S. L. Picciotti, H. R. Hall, S. M. Wallet, R. Maile and M. H. Schoenfisch, Role of Nitric Oxide-Releasing Glycosaminoglycans in Wound Healing, ACS Biomater. Sci. Eng., 2022, 8, 2537–2552 CrossRef CAS PubMed.
- S. E. Maloney, K. V. McGrath, M. J. R. Ahonen, D. S. Soliman, E. S. Feura, H. R. Hall, S. M. Wallet, R. Maile and M. H. Schoenfisch, Nitric Oxide-Releasing Hyaluronic Acid as an Antibacterial Agent for Wound Therapy, Biomacromolecules, 2021, 22, 867–879 CrossRef CAS PubMed.
- L. Yang, E. S. Feura, M. J. R. Ahonen and M. H. Schoenfisch, Nitric Oxide-Releasing Macromolecular Scaffolds for Antibacterial Applications, Adv. Healthcare Mater., 2018, 7, e1800155 Search PubMed.
- I. S. Bayer, Hyaluronic Acid and Controlled Release: A Review, Molecules, 2020, 25, 2649 Search PubMed.
- H. Yang, L. Song, Y. Zou, D. Sun, L. Wang, Z. Yu and J. Guo, Role of Hyaluronic Acids and Potential as Regenerative Biomaterials in Wound Healing, ACS Appl. Bio Mater., 2021, 4, 311–324 CrossRef CAS PubMed.
- M. G. Neuman, R. M. Nanau, L. Oruña-Sanchez and G. Coto, Hyaluronic Acid and Wound Healing, J. Pharm. Pharm. Sci., 2015, 18, 53–60 CAS.
- G. Abatangelo, V. Vindigni, G. Avruscio, L. Pandis and P. Brun, Hyaluronic Acid: Redefining Its Role, Cells, 2020, 9, 1743 CrossRef CAS PubMed.
- M. Dovedytis, Z. J. Liu and S. Bartlett, Hyaluronic Acid and Its Biomedical Applications: A Review, Eng. Regener., 2020, 1, 102–113 Search PubMed.
- A. Fallacara, E. Baldini, S. Manfredini and S. Vertuani, Hyaluronic Acid in the Third Millennium, Polymers, 2018, 10, 701 CrossRef PubMed.
- E. Papakonstantinou, M. Roth and G. Karakiulakis, Hyaluronic Acid: A Key Molecule in Skin Aging, Dermatoendocrinol, 2012, 4, 253–258 CrossRef CAS PubMed.
- S. Halachmi, D. Ben Amitai and M. Lapidoth, Treatment of Acne Scars With Hyaluronic Acid: An Improved Approach, J. Drugs Dermatol., 2013, 12, e121–e123 Search PubMed.
- C. Dierickx, M. K. Larsson and S. Blomster, Effectiveness and Safety of Acne Scar Treatment With Nonanimal Stabilized Hyaluronic Acid Gel, Dermatol. Surg., 2018, 44, S10–S18 CrossRef CAS PubMed.
- S. L. Picciotti, H. El-Ahmad, M. F. Warchol, M. P. Bucci, A. L. Lynch, T. M. Jenkins, M. E. Purvis, S. M. Wallet and M. H. Schoenfisch, Influence of Nitric Oxide-Releasing Hyaluronic Acid Form on Immune Modulation, Biomacromolecules, 2025, 26, 5753–5766 CrossRef CAS PubMed.
- J. S. Frenkel, The Role of Hyaluronan in Wound Healing, Int. Wound J., 2012, 11, 159–163 CrossRef PubMed.
- R. C. Gupta, R. Lall, A. Srivastava and A. Sinha, Hyaluronic Acid: Molecular Mechanisms and Therapeutic Trajectory, Front. Vet. Sci., 2019, 6, 192 Search PubMed.
- M. Antoszewska, E. M. Sokolewicz and W. Barańska-Rybak, Wide Use of Hyaluronic Acid in the Process of Wound Healing—A Rapid Review, Sci. Pharm., 2024, 92, 23 CrossRef CAS.
- Y. R. Jung, C. Hwang, J. M. Ha, D. K. Choi, K. C. Sohn, Y. Lee, Y. J. Seo, Y. H. Lee, C. D. Kim, J. H. Lee and M. Im, Hyaluronic Acid Decreases Lipid Synthesis in Sebaceous Glands, J. Invest. Dermatol., 2017, 137, 1215–1222 Search PubMed.
- J. A. Hrabie, J. R. Klose, D. A. Wink and L. K. Keefer, New Nitric Oxide-Releasing Zwitterions Derived from Polyamines, J. Org. Chem., 1993, 58, 1472–1476 CrossRef CAS.
- A. Niedźwiedzka, M. P. Micallef, M. Biazzo and C. Podrini, The Role of the Skin Microbiome in Acne: Challenges and Future Therapeutic Opportunities, Int. J. Mol. Sci., 2024, 25, 11422 CrossRef PubMed.
- E. Firlej, W. Kowalska, K. Szymaszek, J. Roliński and J. Bartosińska, The Role of Skin Immune System in Acne, J. Clin. Med., 2022, 11, 1579 CrossRef CAS PubMed.
- L. Huang, S. Yang, X. Yu, F. Fang, L. Zhu, L. Wang, X. Zhang, C. Yang, Q. Qian and T. Zhu, Association of Different Cell Types and Inflammation in Early Acne Vulgaris, Front. Immunol., 2024, 15, 1275269 CrossRef CAS PubMed.
- C. Zanchetta, A. Scandolera and R. Reynaud, Hyaluronic Acid in Topical Applications: The Various Forms and Biological Effects of a Hero Molecule in the Cosmetics Industry, Biomolecules, 2025, 15, 1656 Search PubMed.
- H. Baldwin, D. Blanco, C. McKeever, N. Paz, Y. N. Vasques, J. Quiring, C. Enloe, E. de León and N. Stasko, Results of a Phase 2 Efficacy and Safety Study with SB204, an Investigational Topical Nitric Oxide-Releasing Drug for the Treatment of Acne Vulgaris, Journal Of Clinical And Aesthetic Dermatology, 2016, 9, 12–18 Search PubMed.
- J. C. Browning, C. Enloe, M. Cartwright, A. Hebert, A. S. Paller, D. Hebert, E. K. Kowalewski and T. Maeda-Chubachi, Efficacy and Safety of Topical Nitric Oxide− Releasing Berdazimer Gel in Patients With Molluscum Contagiosum A Phase 3 Randomized Clinical Trial Supplemental Content, JAMA Dermatol., 2022, 158, 871–878 Search PubMed.
- T. Yazdanparast, A. Ayatollahi, A. Samadi, A. Sabzvari, H. Kafi and A. Firooz, Safety and Efficacy of a “High and Low Molecular Weight Hyaluronic Acid Hybrid Complex” Injection for Face Rejuvenation, J. Cosmet. Dermatol., 2025, 24, e70117 CrossRef PubMed.
- B. Zhang, Y. M. Choi, J. Lee, I. S. An, L. Li, C. He, Y. Dong, S. Bae and H. Meng, Toll-like Receptor 2 Plays a Critical Role in Pathogenesis of Acne Vulgaris, Biomedical Dermatology, 2019, 3, 4 CrossRef.
- R. Ebid, J. Lichtnekert and H. J. Anders, Hyaluronan Is Not a Ligand but a Regulator of Toll-Like Receptor Signaling in Mesangial Cells: Role of Extracellular Matrix in Innate Immunity, ISRN Nephrol., 2014, 2014, 714081 Search PubMed.
- G. Ladopoulos, C. Barda, K. Tsami, A. Vitsos, N. A. Didaras, D. Mossialos, H. Skaltsa, I. Sfiniadakis, G. T. Papaioannou and M. C. Rallis, Cutibacterium acnes Induces Acne-like Lesions in Hairless Mice Models–A Comparative Study, J. Pharmacol. Toxicol. Methods, 2024, 128, 107539 CrossRef CAS PubMed.
- Y. H. Jang, K. C. Lee, S. J. Lee, D. W. Kim and W. J. Lee, HR-1 Mice: A New Inflammatory Acne Mouse Model, Ann. Dermatol., 2015, 27, 257–264 CrossRef PubMed.
- S. L. Picciotti, H. El-Ahmad, M. P. Bucci, Q. E. Grayton, S. M. Wallet and M. H. Schoenfisch, Delivery of Nitric Oxide by Chondroitin Sulfate C Increases the Rate of Wound Healing through Immune Modulation, ACS Appl. Bio Mater., 2024, 7, 6152–6161 CrossRef CAS PubMed.
| This journal is © The Royal Society of Chemistry 2026 |





