Open Access Article
Open Access ArticleThiophenol-mediated metal-free chemoselective conjugate reduction strategy for 3-alkylidene oxindoles
Baliram R. Patilab,
Chandrakant B. Nichinde ab,
Amardipsing S. Girase
ab,
Amardipsing S. Girase ab,
Suryakant S. Chaudhariab and
Anil K. Kinage
ab,
Suryakant S. Chaudhariab and
Anil K. Kinage *ab
*ab
aChemical Engineering and Process Development Division, CSIR-National Chemical Laboratory, Pune, 410008, India. E-mail: ak.kinage.ncl@csir.res.in
bAcademy of Scientific and Innovative Research (AcSIR), Ghaziabad-201 002, India
First published on 23rd February 2026
Abstract
A mild and metal-free thiophenol-mediated chemoselective conjugate reduction of 3-alkylidene oxindoles via a thiol–ene-type Michael addition was effectively carried out. Thiophenol as an efficient reductant is used in DMSO, and the transformation proceeds without the need for metal catalyst, external hydrogen source, or harsh reagents. The reaction displays a broad substrate scope, accommodating both electron-donating and electron-withdrawing substituents, and delivers 3-alkyloxindoles in high yields with excellent β-selectivity. This sustainable and operationally simple method unveils a distinct thiol-mediated reduction mechanism, providing a green and versatile approach for accessing reduced oxindole derivatives and related Michael acceptors.
Introduction
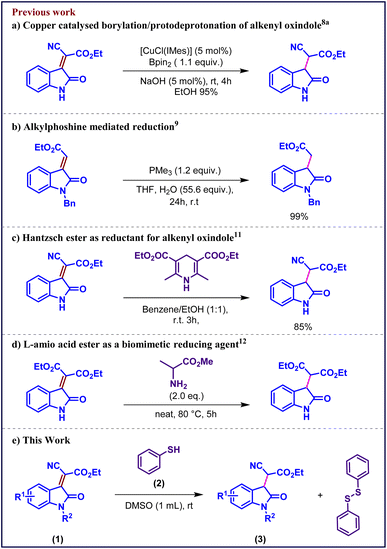
The oxindole framework represents a privileged structural motif that frequently occurs in a wide range of natural products and pharmacologically active molecules.1 Considerable attention has been directed toward oxindole derivatives owing to their diverse biological profiles, including antioxidant,2 neuroprotective,3 anticancer,4 and anti-HIV activities.5 Among these, 3-substituted oxindoles have emerged as key synthetic intermediates in the construction of spirocyclic and polycyclic heterocyclic architectures,6 which are prevalent in numerous bioactive natural products and therapeutic agents.7Despite their importance, conventional methods for accessing 3-substituted oxindoles often rely on transition-metal-catalyzed hydrogenation or stoichiometric reductants, which limit regioselectivity, functional-group tolerance, and sustainability. The predominant approach involves selective reduction of the conjugated C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C bond in 3-alkylidene oxindoles. Classical strategies include metal-catalyzed hydrogenation (Scheme 1a),8 alkylphosphane-mediated reductions (Scheme 1b)9 and biocatalytic processes utilizing baker's yeast or Pseudomonas strains.10 In recent years, biomimetic and photoredox systems have emerged as alternative methods, employing Hantzsch esters (Scheme 1c),11 L-amino acid esters (Scheme 1d),12 and visible-light-driven photoredox catalysis using formate donors or Eosin Y.13 However, these transformations often suffer from inherent limitations such as harsh reaction conditions, use of aggressive or expensive reagents, limited substrate scope, and challenging purification due to the generation of byproducts.
C bond in 3-alkylidene oxindoles. Classical strategies include metal-catalyzed hydrogenation (Scheme 1a),8 alkylphosphane-mediated reductions (Scheme 1b)9 and biocatalytic processes utilizing baker's yeast or Pseudomonas strains.10 In recent years, biomimetic and photoredox systems have emerged as alternative methods, employing Hantzsch esters (Scheme 1c),11 L-amino acid esters (Scheme 1d),12 and visible-light-driven photoredox catalysis using formate donors or Eosin Y.13 However, these transformations often suffer from inherent limitations such as harsh reaction conditions, use of aggressive or expensive reagents, limited substrate scope, and challenging purification due to the generation of byproducts.
Within this domain, the selective reduction of conjugated alkenes continues to be of substantial research interest. Despite notable advances in chemoselective reduction methodologies, the thiol-mediated reduction of conjugated alkenes in oxindole systems remains unexplored, presenting an opportunity to develop a mild, sustainable, and metal-free reductive strategy.
As we are working on the functionalization of oxindole moity14 extending that, herein we disclose a mild and chemoselective thiolphenol mediated reduction of oxindole-derived conjugated alkenes, providing straightforward access to 3-substituted oxindoles under metal-free conditions. This approach circumvents the disadvantages of metal-based systems and harsh reductants by taking advantage of the Michael addition of thiols to electron-deficient double bonds, providing a sustainable substitute for the production of medicinally significant oxindole derivatives.
Results and discussion
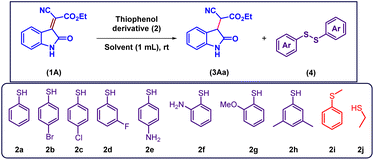
We initiate our study by examining the reaction of thiophenol with isatylidene cyanoester 1A in ethanol, anticipating the formation of a thia-Michael addition product. Remarkably, instead of the expected adduct, the reaction predominantly afforded the reduced product 3Aa via a selective conjugate C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C bond reduction pathway, corresponding product 3Aa is isolated as a mixture of diastereomers. The formation of 1,2-diphenyldisulfane 4 as a byproduct was also observed and confirmed by NMR analysis (Table 1, entry 1). Encouraged by this unexpected reactivity, we subsequently investigated the generality of the transformation using a series of isatin derived Michael acceptors.
C bond reduction pathway, corresponding product 3Aa is isolated as a mixture of diastereomers. The formation of 1,2-diphenyldisulfane 4 as a byproduct was also observed and confirmed by NMR analysis (Table 1, entry 1). Encouraged by this unexpected reactivity, we subsequently investigated the generality of the transformation using a series of isatin derived Michael acceptors.
| Entry | 2 (equiv.) | Solvent (1 mL) | Time (h) | Yieldb (%) |
|---|---|---|---|---|
| a Unless otherwise noted, a mixture of 1 (0.3 mmol, 1 equiv.) 2 (0.6 mmol, 2 equiv.) in DMSO (1 mL) stirred continuously till get formation of 3.b Isolated yield of two diastereomers.c 2a (0.9 mmol, 3 equiv.).d 2a (0.3 mmol, 1 equiv.); n. d. = not detected.e Only thio-Micheal addition product was observed. | ||||
| 1 | 2a (2.0) | EtOH | 5 | 35 |
| 2 | — | DMSO | 24 | n.d. |
| 3 | 2a (2.0) | DMF | 5 | 45 |
| 4 | 2a (2.0) | DCE | 3 | 62 |
| 5 | 2a (2.0) | ACN | 6 | Trace |
| 6 | 2a (2.0) | THF | 6 | Trace |
| 7 | 2a (2.0) | DCM | 3 | 60 |
| 8 | 2a (2.0) | DMSO | 1 | 92 |
| 9 | 2a (2.0) | H2O | 8 | n.d. |
| 10 | 2a (2.0) | Toluene | 3 | 55 |
| 11c | 2a (3) | DMSO | 0.5 | 92 |
| 12d | 2a (1.0) | DMSO | 3 | 30 |
| 13 | 2b (2.0) | DMSO | 1 | 92 |
| 14 | 2c (2.0) | DMSO | 1.5 | 88 |
| 15 | 2d (2.0) | DMSO | 1.5 | 85 |
| 16 | 2e (2.0) | DMSO | 2.5 | 63 |
| 17 | 2f (2.0) | DMSO | 2.5 | 72 |
| 18 | 2g (2.0) | DMSO | 5 | <20 |
| 19 | 2h (2.0) | DMSO | 5 | <20 |
| 20 | 2i (2.0) | DMSO | 12 | n.d. |
| 21 | 2j (2.0) | DMSO | 12 | n.d.e |
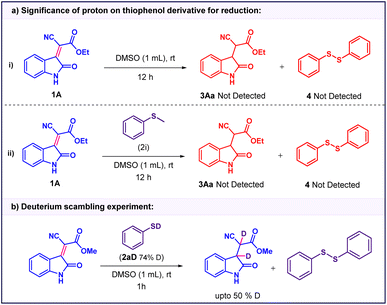
To verify the essential role of thiophenol, the reaction was performed in absence thiophenol no conversion of the starting material 1A was observed, and the substrate remained intact throughout the reaction course, indicating that thiophenol is crucial for initiating the reduction process (Table 1, entry 2). Subsequently, the influence of different solvents on the efficiency of the transformation was investigated. Among the solvents examined, DMSO provided the best yield of the desired reduced product with 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 dr ratio (Table 1, entries 3–10). The study on the effect of thiophenol loading revealed that the reaction exhibited equal efficiency with increase loading of thiophenol up to 3 equiv. and time of reaction also decrease effectively while reducing loading of thiophenol to 1 equiv. reduced yielding up to 30% and also increase reaction time (Table 1, entry 11 and 12). To assess the effect of substituents on thiophenol, a series of derivatives (2a–2i) were evaluated under the optimized conditions. Halogen-substituted thiophenols demonstrated enhanced reactivity, with 4-bromothiophenol (2b) providing the reduced product in excellent yield within 1 h. While thiophenols bearing electron-donating groups such as amino, methoxy, or methyl substituents exhibited significantly lower reactivity, affording the corresponding products in poor to moderate yields. No reaction occurred with thioanisole (2i), confirming that the S–H proton of thiophenol is essential for the conjugate reduction (Table 1, entry 13–20). We further evaluated the feasibility of the reaction with an aliphatic thiol; however, only the thio-Michael adduct was observed instead of the desired reduction (Table 1, entry 21).
1 dr ratio (Table 1, entries 3–10). The study on the effect of thiophenol loading revealed that the reaction exhibited equal efficiency with increase loading of thiophenol up to 3 equiv. and time of reaction also decrease effectively while reducing loading of thiophenol to 1 equiv. reduced yielding up to 30% and also increase reaction time (Table 1, entry 11 and 12). To assess the effect of substituents on thiophenol, a series of derivatives (2a–2i) were evaluated under the optimized conditions. Halogen-substituted thiophenols demonstrated enhanced reactivity, with 4-bromothiophenol (2b) providing the reduced product in excellent yield within 1 h. While thiophenols bearing electron-donating groups such as amino, methoxy, or methyl substituents exhibited significantly lower reactivity, affording the corresponding products in poor to moderate yields. No reaction occurred with thioanisole (2i), confirming that the S–H proton of thiophenol is essential for the conjugate reduction (Table 1, entry 13–20). We further evaluated the feasibility of the reaction with an aliphatic thiol; however, only the thio-Michael adduct was observed instead of the desired reduction (Table 1, entry 21).
With initial results in hand, we check efficiency of reaction with different alkylidene oxindole with substituents at the R3 and R4 positions summarizes in Table 2. Substrates 1A and 1B, containing strongly electron-withdrawing groups at these positions, displayed the highest reactivity, affording the corresponding products in comparable yields within 1 h (Table 2, entries 1–2). In contrast, substrate 1C, bearing a weak electron-withdrawing substituent, required a longer reaction time 3 h and furnished product 3C in lower yield compared to 3A and 3B. Replacement of the electron-withdrawing group at R3 with hydrogen (1D) led to a significant decrease in reaction rate 6 h and yield. Moreover, substrates bearing electron-donating substituents at R3 and R4 showed negligible conversion, and the reduced product 3E was not formed.
| Entry | Substrate | Product | R3 | R4 | Yieldb (%) |
|---|---|---|---|---|---|
a Unless otherwise noted, a mixture of 1 (0.3 mmol, 1 equiv.), thiophenol (0.6 mmol, 2.0 equiv.) in DMSO (1 mL) was stirred at rt up to the formation of products 3.b Isolated yield of products 3.c 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 Diasteriomeric mixture of product. N. R = No Result. 1 Diasteriomeric mixture of product. N. R = No Result. |
|||||
| 1 | 1A | 3Aa | CN | COOEt | 90c |
| 2 | 1B | 3B | CN | CN | 92 |
| 3 | 1C | 3C | COOEt | COOEt | 74 |
| 4 | 1D | 3D | H | COOEt | 56 |
| 5 | 1E | 3E | Me | Me | N. R |
Among all tested substrates (1A–1D), 1A proved to be the most reactive isomer under the optimized conditions, though without notable enhancement in yield or diastereomeric ratio (Table 2, entries 1–5).
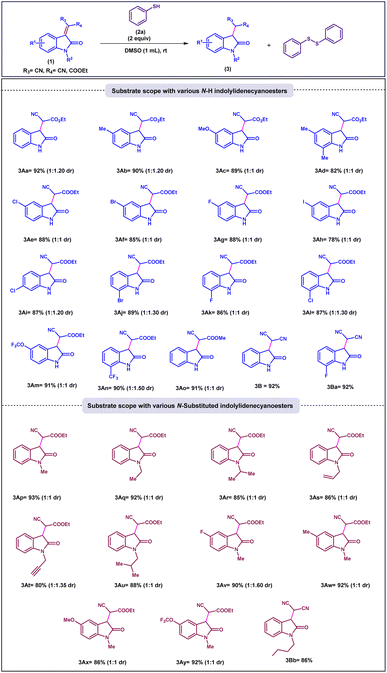
With the optimized reaction condition established, the substrate scope of indolylidene cyanoesters bearing various substituents was systematically investigated to assess the generality of the transformation. Initially, substrates featuring electron-donating groups such as 5-Me, 5-OMe, and 5,7-DiMe were examined. These substrates underwent smooth conversion to afford the corresponding reduced products (3Ab–3Ad) in good to excellent yields, although the reactions provided diastereoselectivity with an approximate dr of 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1. Encouraged by these results, the influence of halogen substituents at different positions of the indole ring (5-, 6-, and 7-) was explored. Substrates bearing halogens such as 5-Cl, 5-Br, and 5-F demonstrated enhanced reactivity, delivering products (3Ai–3Ak) in good to excellent yields. In contrast, the 5-I derivative (3Ah) displayed sluggish reactivity, affording a comparatively lower yield (78%), likely due to steric and electronic effects associated with the larger iodine atom. Further evaluation of the 6- and 7-halogen-substituted derivatives revealed excellent reactivity and yields (86–89%), with diastereomeric ratios reaching up to 1
1. Encouraged by these results, the influence of halogen substituents at different positions of the indole ring (5-, 6-, and 7-) was explored. Substrates bearing halogens such as 5-Cl, 5-Br, and 5-F demonstrated enhanced reactivity, delivering products (3Ai–3Ak) in good to excellent yields. In contrast, the 5-I derivative (3Ah) displayed sluggish reactivity, affording a comparatively lower yield (78%), likely due to steric and electronic effects associated with the larger iodine atom. Further evaluation of the 6- and 7-halogen-substituted derivatives revealed excellent reactivity and yields (86–89%), with diastereomeric ratios reaching up to 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1.3 (3Ai–3Al). Interestingly, indolylidene cyanoesters substituted with electron-withdrawing groups such as 5-OCF3 and 7-CF3 exhibited markedly enhanced reactivity, providing the desired products (3Am, 3An) in higher yields (90–91%) compared to their electron-donating counterparts (Scheme 2).
1.3 (3Ai–3Al). Interestingly, indolylidene cyanoesters substituted with electron-withdrawing groups such as 5-OCF3 and 7-CF3 exhibited markedly enhanced reactivity, providing the desired products (3Am, 3An) in higher yields (90–91%) compared to their electron-donating counterparts (Scheme 2).
Under the optimised reaction condition, the substrate scope was expanded to various N-substituted indolylidene cynoester derivatives, such as N-methyl, N-ethyl, N-isopropyl, N-propargyl, N-isobutyl and all these tolerated well and afforded the corresponding products with high to excellent yield and dr up to 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1.4 (3Ap–3Au). Additionally, the optimised reaction condition also work well for derivatives from 3Av to 3Ay which are substituted on both aromatic ring as well as N-substitution and also provided desired product in excellent yields (86–92%) and dr up to 1
1.4 (3Ap–3Au). Additionally, the optimised reaction condition also work well for derivatives from 3Av to 3Ay which are substituted on both aromatic ring as well as N-substitution and also provided desired product in excellent yields (86–92%) and dr up to 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1.6 (3Av–3Ay). Further, isatylidene malonitrile derivatives were employed under the optimised conditions the corresponding products were obtained with good yield (3B, 3Ba, 3Bb).
1.6 (3Av–3Ay). Further, isatylidene malonitrile derivatives were employed under the optimised conditions the corresponding products were obtained with good yield (3B, 3Ba, 3Bb).
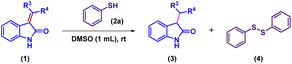
To identify the hydrogen source responsible for the reduction, a series of control experiments were performed. As indicated in the optimization studies, thiophenol is essential for the reaction, as no product formation was observed in its absence. To probe the role of the thiol proton, thioanisole (2i) in which the S–H proton of thiophenol is replaced by a methyl group (S–Me) was employed under identical conditions. In this case, the starting material remained unreacted, and no formation of the reduced product or 1,2-diphenyldisulfane was detected (Scheme 3a), confirming that the S–H bond of thiophenol is crucial for the reduction process. To gain deeper mechanistic insight, an isotopic scrambling experiment was subsequently conducted using deuterated thiophenol (2aD) (<75% D), enabling direct verification of the thiol proton's participation as the hydrogen donor in the transformation. Under the standard reaction conditions to afford the crude product 3Ao up to 91% yield with up to 50% D contents by 1H NMR (Scheme 3b). This isotope labelling experiments clearly indicate that the source of proton is only thiophenol (S–H) in reduction reaction.
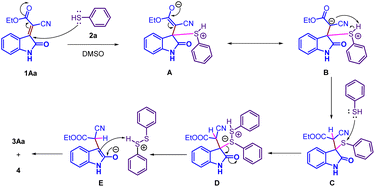
Based on previous literature precedents on thia-Michael reactions15 and our control experiments, we propose a plausible mechanism for the thiophenol-mediated, metal-free conjugate reduction of 3-alkylidene oxindoles. The transformation proceeds via a thiol–ene-type Michael addition–elimination sequence. Under the reaction conditions, thiophenol undergoes chemoselective nucleophilic attack at the C3-position of the 3-alkylidene oxindole, affording the thio-Michael adduct C via intermediate B. The pronounced polarization of the α, β-unsaturated system adjacent to the electron-withdrawing carbonyl group ensures high chemoselectivity in this conjugate addition step. Subsequent proton transfer furnishes the β-thiolated intermediate, which, in the presence of excess thiophenol, undergoes β-elimination or hydride transfer to expel the diphenyl disulfide (4) moiety and generate the reduced C–H bond (3Aa). Throughout the process, thiophenol (PhSH) serves a dual role as both the nucleophile and the hydrogen donor, thus enabling an efficient metal-free conjugate reduction pathway (Scheme 4).
Conclusions
In summary, we have developed an efficient thiophenol-mediated, metal-free, chemoselective conjugate reduction strategy for the transformation of 3-alkylidene oxindoles into their corresponding 3-alkyl oxindoles. This protocol operates under mild, transition-metal-free conditions and exhibits excellent chemoselectivity and functional group tolerance. Mechanistic investigations, supported by control experiments and literature precedents, suggest that the transformation proceeds via a thiol–ene-type Michael addition–elimination pathway, wherein thiophenol serves a dual role as both the nucleophile and the hydrogen donor. The pronounced polarization of the α, β-unsaturated oxindole framework ensures high chemoselectivity during the conjugate addition step, while the subsequent β-elimination or hydride transfer delivers the reduced product in a redox-neutral manner. Overall, this methodology provides a practical, sustainable, and mechanistically insightful approach to the selective reduction of C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C bond in electron-deficient isatylidene systems, enriching the synthetic utility of thiol-mediated transformations in modern organic synthesis.
C bond in electron-deficient isatylidene systems, enriching the synthetic utility of thiol-mediated transformations in modern organic synthesis.
Author contributions
B. R. P. and A. S. G. perform the experiment and developed the method. B. R. P. expanded the substrate scope. S. S. C. and C. B. N finalized the data, A. K. K. directed the project. B. R. P. and C.B.N. wrote main manuscript with the help of A. K. K and feedback from other authors.Conflicts of interest
No conflicts of interest to declare.Data availability
The data supporting this article have been included as part of the supplementary information (SI). Supplementary information is available. See DOI: https://doi.org/10.1039/d5ra09175f.Acknowledgements
B. R. P. and S. S. C. thank UGC for a fellowship. C. B. N. thanks CSIR-New Delhi, India, for the fellowship. We sincerely acknowledge the support from the central NMR, HRMS facility, and CSIR-NCL.Notes and references
-
(a) C. V. Galliford and K. A. Scheidt, Angew. Chem., Int. Ed., 2007, 46(46), 8748–8875 CrossRef CAS PubMed
; (b) S. Peddibhotla, Curr. Bioact. Compd., 2009, 5, 20–38 Search PubMed
; (c) J. J. Badillo, N. V. Hanhan and A. K. Franz, Curr. Opin. Drug Discovery Dev., 2010, 13, 758 CAS
; (d) S. Hibino and T. Choshi, Nat. Prod. Rep., 2002, 18, 66 Search PubMed
; (e) N. Ye, H. Chen, E. A. Wold, P. Y. Shi and J. Zhou, ACS Infect. Dis., 2016, 2, 382 Search PubMed
.
- B. Volk, J. Barkóczy, E. Hegedus, S. Udvari, I. Gacsályi, T. Mezei, K. Pallagi, H. Kompagne, G. Lévay, A. Egyed, L. G. J. Hársing, M. Spedding and G. Simig, J. Med. Chem., 2008, 51, 2522–2532 Search PubMed
.
- B. Volk, I. Gacasalyi, K. Pallagi, L. Posszvacz, I. Gynos and E. Szabo, J. Med. Chem., 2011, 54, 6657 Search PubMed
.
- B. Yu, D. Q. Yu and H. M. Liu, Eur. J. Med. Chem., 2015, 97, 673–698 CrossRef CAS PubMed
.
-
(a) T. Jiang, K. L. Kuhen, K. Wolff, H. Yin, K. Bieza, J. Caldwell, B. Bursulaya, T. Y.-H. Wu and Y. He, Bioorg. Med. Chem. Lett., 2006, 16(8), 2105–2108 CrossRef CAS PubMed
; (b) M. Eissenstat, T. Guerassina, S. Gulnik, E. Afonina, A. M. Silva, D. Ludtke, H. Yokoe, B. Yu and J. Erickson, Bioorg. Med. Chem. Lett., 2012, 22(15), 5078–5083 CrossRef CAS PubMed
.
-
(a) J.-Q. Zhao, Z.-J. Wu, M.-Q. Zhou, X.-Y. Xu, X.-M. Zhang and W.-C. Yuan, Org. Lett., 2015, 17, 5020–5023 CrossRef CAS PubMed
; (b) L. Chen, Y. You, M.-L. Zhang, J.-Q. Zhao, J. Zuo, X.-M. Zhang, W.-C. Yuan and X.-Y. Xu, Org. Biomol. Chem., 2015, 13, 4413–4417 RSC
; (c) A. Millemaggi and R. J. K. Taylor, Eur. J. Org Chem., 2010, 2010, 4527 CrossRef
; (d) F. Zhou, Y. L. Liu and J. Zhou, Adv. Synth. Catal., 2010, 352, 1381 CrossRef CAS
; (e) S. Pramanik, R. Karmakar and C. Mukhopadhyay, Tetrahedron, 2025, 184, 134757 CrossRef CAS
.
-
(a) C. Jaghta and M. Singh, Chemistry Select, 2025, 10, e00743 Search PubMed
; (b) Y. M. Khetmalis, M. Shivani, S. Murugesan and K. V. G. Chandra Sekhar, Biomed. Pharmacother., 2021, 141, 111842 CrossRef CAS PubMed
; (c) M. Kaur, M. Singh, N. Chadha and O. Silakari, Eur. J. Med. Chem., 2016, 123, 858–894 CrossRef CAS PubMed
.
-
(a) M. E. Contreira, D. S. Lüdtke and A. V. Moro, Tetrahedron Lett., 2018, 59, 2784–2787 CrossRef CAS
; (b) Y. Liu, Y. Mao, Y. Hu, J. Gui, L. Wang, W. Wang and S. Zhang, Adv. Synth. Catal., 2019, 361, 1554–1558 CrossRef CAS
.
- S.-H. Cao, X.-C. Zhang, Y. Wei and M. Shi, Eur. J. Org Chem., 2011, 2011(14), 2668–2672 CrossRef
.
-
(a) A. Rossetti, A. Sacchetti, M. Bonfanti, G. Roda, G. Rainoldi and A. Silvani, Tetrahedron, 2017, 73, 4584–4590 Search PubMed
; (b) J. Zhao, S. Guan, X. Zhou, W. Han, B. Cui and Y. Chen, Tetrahedron, 2016, 72, 3098–3104 Search PubMed
.
- S. J. Garden, C. R. W. Guimarães, M. B. Corréa, C. A. F. Oliveira, A. C. Pinto and R. Bicca de Alencastro, J. Org. Chem., 2003, 68, 8815–8822 CrossRef CAS PubMed
.
- J.-W. Ren, Q.-H. Zhang, C.-S. Han, H.-X. Zhang, Y.-B. Wang, H.-R. Shi, J.-H. Suna and Y.-F. Han, Org. Biomol. Chem., 2025, 23, 654–659 RSC
.
-
(a) C. U. Brzezinski, A. R. LeBlanc, M. G. Clerici and W. M. Wuest, Org. Lett., 2024, 26, 5534–5538 CrossRef CAS PubMed
; (b) A. Bhowmick, P. K. Warghude and R. G. Bhat, Asian J. Org. Chem., 2022, 11, e202200205 CrossRef CAS
.
-
(a) C. B. Nichinde, B. R. Patil, S. S. Chaudhari, B. P. Mali, R. G. Gonnade and A. K. Kinage, RSC Adv., 2023, 13, 13206–13212 RSC
; (b) B. R. Patil, C. B. Nichinde, S. S. Chaudhari, R. K. Gamidi and A. K. Kinage, RSC Adv., 2024, 14, 2873–2877 RSC
; (c) C. B. Nichinde, M. Bhati, A. S. Girase, B. R. Patil, S. S. Chaudhari, R. Krishna Gamidi, K. Joshi and A. K. Kinage, Eur. J. Org Chem., 2025, 28, e202401121 CrossRef CAS
; (d) B. R. Patil, C. B. Nichinde, S. S. Chaudhari and A. K. Kinage, Org. Biomol. Chem., 2025, 23, 6214–6219 RSC
; (e) S. S. Chaudhari, C. B. Nichinde, B. R. Patil, R. K. Gamidi and A. K. Kinage, Org. Biomol. Chem., 2024, 22, 1727–1732 Search PubMed
; (f) S. S. Chaudhari, C. B. Nichinde, B. R. Patil, A. S. Girase and A. K. Kinage, Select, 2025, 10, e202405996 Search PubMed
; (g) S. S. Chaudhari, C. B. Nichinde, B. R. Patil, A. S. Girase, S. H. Kaulage and A. K. Kinage, Org. Biomol. Chem., 2025, 23, 8687–8694 Search PubMed
.
-
(a) X. Huang, E. David, P. Jubault, T. Besset and S. Couve-Bonnaire, J. Org. Chem., 2020, 85, 14055–14067 Search PubMed
; (b) X. Ding, C. Tian, Y. Hu, L. Gong and E. Meggers, Eur. J. Org Chem., 2016, 2016, 887–890 Search PubMed
; (c) S. Shaw and J. D. White, Org. Lett., 2015, 17, 4564–4567 Search PubMed
; (d) P. Wadhwa, A. Kharbanda and A. Sharma, Asian J. Org. Chem., 2018, 7, 634–661 Search PubMed
; (e) P. B. Momo, E. F. Mizobuchi, R. Echemendía, I. Baddeley, M. N. Grayson and A. C. B. Burtoloso, J. Org. Chem., 2022, 87, 3482–3490 CrossRef CAS PubMed
.
| This journal is © The Royal Society of Chemistry 2026 |