Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Investigation of the dynamic behavior of metallopolymers by combined experimental and theoretical methods
Milena
Jäger
 ab,
Michael Freduah
Agyemang
cde,
Wanja T.
Schulze
ab,
Michael Freduah
Agyemang
cde,
Wanja T.
Schulze
 c,
Julian
Kimmig
c,
Julian
Kimmig
 ab,
Thomas
Bätz
ab,
Chiara
Wondraczek
c,
Stefan
Zechel
abgh,
Alexander
Croy
ab,
Thomas
Bätz
ab,
Chiara
Wondraczek
c,
Stefan
Zechel
abgh,
Alexander
Croy
 c,
Michael
Schmitt
c,
Michael
Schmitt
 cd,
Jürgen
Popp
cd,
Jürgen
Popp
 cde,
Stefanie
Gräfe
cde,
Stefanie
Gräfe
 cdf,
Martin D.
Hager
cdf,
Martin D.
Hager
 *abgh and
Ulrich S.
Schubert
*abgh and
Ulrich S.
Schubert
 *abdgh
*abdgh
aLaboratory of Organic and Macromolecular Chemistry (IOMC), Friedrich Schiller University Jena, Humboldtstr. 10, 07743 Jena, Germany. E-mail: ulrich.schubert@uni-jena.de; martin.hager@uni-jena.de
bJena Center for Soft Matter (JCSM), Friedrich Schiller University Jena, Philosophenweg 7, 07743 Jena, Germany
cInstitute of Physical Chemistry (IPC), Friedrich Schiller University Jena, Helmholzweg 4, 07743 Jena, Germany
dAbbe Center of Photonics (ACP), Friedrich Schiller University Jena, Albert-Einstein-Straße 6, 07745 Jena, Germany
eLeibniz Institute of Photonic Technology, Albert-Einstein-Straße 9, 07745 Jena, Germany
fFraunhofer Institute for Applied Optics and Precision Engineering, Albert-Einstein-Straße 7, 07745 Jena, Germany
gHelmholtz-Institute for Polymers in Energy Applications Jena (HIPOLE Jena), Lessingstr. 12-14, 07443 Jena, Germany
hHelmholtz Zentrum Berlin für Materialien und Energie GmbH (HZB), Hahn-Meitner-Platz 1, 14109 Berlin, Germany
First published on 13th March 2026
Abstract
In this study, we investigate the structural changes in dynamic metallopolymers during stimulus application, i.e. thermal treatment. For this purpose, we focused on the synthesis of polymers containing terpyridine moieties as ligands in the side chains that were complexed with either iron(II) or zinc(II) salts. The resulting crosslinked metallopolymers were characterized using differential scanning calorimetry (DSC), thermogravimetric analysis (TGA) and elemental analysis (EA). Rheology experiments, including dynamic mechanical thermal analysis (DMTA), frequency sweeps, stress relaxation and time–temperature superposition were conducted to study the stimuli-responsive mechanical properties. Hereby, the activation energy determined by stress relaxation as a combination of the metal complex and the polymer matrix could be determined. Additionally, computational master curves were obtained and the resulting relaxation spectra were analyzed. Beside the macroscopic material properties, temperature-dependent Raman spectroscopy and density functional theory (DFT) calculations were utilized to gain information on the changes on the molecular level. In this context, morphological changes in the polymer matrix were observed, which might be correlated to the presence of supramolecular aggregates. The changes on the molecular level could be linked to the macroscopic properties.
Introduction
In nature, metal complexes play an integral part in biological processes. For example, in hemoglobin or chlorophyll, the incorporation of a metal ion in the organic framework leads to unique functions like oxygen transport or photosynthesis.1–3 Inspired by these examples, synthetic polymers have become a widely researched field. Today, polymers are applied in various areas of the society, such as in medicine,4 in packaging5 or as electronic devices.6 However, traditional polymers are characterized by static and largely unalterable properties. Hence, the field of polymer research has advanced from static structures to dynamic materials.7 These kinds of polymers belong to an interesting class of materials that are capable of undergoing reversible and controlled changes in their properties due to different internal or external stimuli.8–10 In general, dynamic polymers can be divided into two groups, the dynamic covalent11 and the dynamic supramolecular polymers.12 Covalent polymers are considered dynamic when it is possible to break and to reform the bonds reversibly.13 This can be achieved by a variety of literature known reactions, such as the thermally reversible Diels–Alder reaction,14–16 the transesterification17,18 or the hydrolysis of boronic esters,19,20 the thermal structural reorganization of alkoxyamines,21,22 the degenerative imine bond exchange,23,24 the disulfide metathesis25,26 or the reorganization of hindered urea bonds.27,28 In contrast, dynamic supramolecular polymers can be based on hydrogen bonds,29,30 host–guest interactions,31,32 π–π stacking33,34 or metallo-supramolecular interactions.35,36 The usage of hydrogen bonds or π–π stacking in the design of supramolecular polymers leads to a rapid and reversible assembly, but the resulting materials are often sensitive to temperature or solvents, which limits the material's mechanical strength.37,38 Host–guest interactions exhibit high selectivity and self-organization but are also sensitive to temperatures.39 In contrast to these supramolecular binding motifs, metallopolymers are tunable and can be more resistant to moisture or higher temperatures, which leads to more robust materials and broader design possibilities.40–44Furthermore, metallopolymers combine classical organic polymers with metal ions or complexes to benefit from each other's properties.45 The advantages of the polymers like lightweight,46 tunable properties47 and cost-effectiveness48 are merged with the properties of metal complexes like catalytic activity,49 bioactivity,50 conductivity51 and others making these polymers an unique material class.52–54 This combination of benefits leads to many different potential applications of metallopolymers, for example as self-healing55,56 or shape-memory materials.42,57,58
There are different ways to incorporate a metal ion into the polymer structure. Ciardelli et al.59 postulated three different types of metal-containing polymers. First, that metal ions can be bound to a chain of the polymer by electrostatic attraction, by coordination or by linking the metal complex as a pendant or an end group to the polymer. Second, the metal complexes can be incorporated into the polymer as part of the chain by covalent linkage or by metal ligand coordination in the main chain. Lastly, the metal ions or the complex can physically interact with the polymer chain.59,60
In literature, ligands are typically chosen as the building blocks to incorporate the metal ions into the polymer.43 Therefore, bidirectional, ditopic ligands with various interactions, such as ionic bonding,61 metal-arene complexation62 and coordinative bonding56 are applied. The coordinative binding of the metal ion to the ligand is the most prominent way to design metallopolymers shown in literature.42,56,63–65 Hereby a variety of different ligands containing different heteroatoms can be applied. A highly dynamic coordination environment can be achieved by the usage of imidazole41 or histidine-inspired ligands,66 which leads to reversible networks.42 Other ligands like porphyrins67 or salens68 can integrate new functionalities into the polymer like electronic69,70 or optical features.67 Furthermore, different pyridine-based ligands can be utilized, therefore mono-,71 bi-58 or tridentate systems64 are applied. Here, the binding constant increases from pyridine to terpyridines, showcasing the chelating effect of those ligands.72
In addition to the choice of ligand, it is also crucial to select the right metal salt because it influences the properties and the dynamics of the resulting metallopolymer. Each metal ion differs in geometry, ligand field stabilization and binding strength.73 These factors determine the dynamic nature of the resulting metallopolymer networks and affect the macroscopic behavior of the materials. Therefore, different metal ions from the p-block like Al(III)74 or Bi(III),75 the d-block and the f-block are typically chosen. The d-block transition metals like Fe(II/III)76 or Ru(II/III)77 are more commonly used than the p-block metal ions because they offer a wide range of redox states and coordination geometries,76,77 while the f-block lanthanide ions like Eu(III)78 or Tb(III)79 exhibit unique optical properties that can be used for, e.g., stimuli-responsive photoluminescence.80
Overall, by selecting specific ligand/metal combinations, the properties can be adjusted from highly reversible, i.e. low binding constant of the supramolecular unit, to nearly irreversible based on a high association constant.42,72 Applying this principle enables a tailor-made design of the materials thereof with tunable and on-demand properties.63
Terpyridine ligands have gained significant attention due to their coordination to transition metals featuring a high degree of versatility and an adjustable binding constant by choosing the respective metal ion.64,65,76,81 The resulting metallopolymers can be addressed by different stimuli, e.g., temperature,57 and can be utilized for different applications as smart materials. In this context, self-healing56,63,64 and shape-memory polymers42,57,58 could be obtained by the utilization of this ligand.
However, the focus of previous works was on synthesizing and characterizing those materials, while there are still unanswered questions about the structural dynamics, in particular during stimulus application. Rheology experiments were also conducted before but mostly to confirm the results from self-healing63 or shape-memory investigations.57 In a previous study from our group, Bode et al.63 confirmed the correlation between the self-healing ability and the supramolecular bond lifetime linked to the crossover points in frequency sweep measurements, while Meurer et al.57 confirmed the shape-memory ability of the used metallopolymers via frequency sweeps and thermomechanical analysis. However, a detailed investigation of the macroscopic and molecular processes of those metallopolymers during thermal treatment is still missing preventing further optimization of such systems.
In this study, we aim to provide more in-depth detail of the structural and molecular dynamics of terpyridine-based metallopolymers using Fe(II) and Zn(II) metal ions. For this purpose, a combination of rheological investigations and Raman spectroscopy are applied. While rheology provides insights into the viscoelastic behavior and structural changes,82 Raman spectroscopy offers a perspective on the bond interactions and coordination changes.83 To complement these results theoretical calculations enable predictions of the molecular behavior and the structural changes.84 With this approach, we aim to build a more complete picture of the material's response and behavior that could lead to the development of new smart materials with better and on-demand functionalities.
Results and discussion
Synthesis of the metallopolymers
In order to investigate the reversible moieties in metallopolymers, first, linear polymers featuring ligands in the side-chain were prepared. As the ligand, 6-(2,2′:6′,2″-terpyridin-4′-yloxy)-hexyl-methacrylate (Tpy-MA) was selected due to its well-established coordination properties, making it particularly suitable for the metallopolymer synthesis.42,57,58,85 The ligand was synthesized following a previously reported procedure.86 The linear polymers were synthesized via reversible addition–fragmentation chain-transfer (RAFT) polymerization. Methyl methacrylate (MMA, P1), butyl methacrylate (BMA, P2), or 2-ethylhexyl methacrylate (2-EHMA, P3) were chosen as comonomers and were polymerized together with Tpy-MA. Hereby, a terpyridine-content of ca. 10% was aimed to introduce a significant amount of coordination functionalities, to have a controlled, but not too dense crosslinking and to compare the resulting systems to literature known metallopolymers.86 The resulting polymers were characterized using elemental analysis (EA), differential scanning calorimetry (DSC), thermogravimetric analysis (TGA), and size exclusion chromatography (SEC) (all results are depicted in the SI; see Table 1 for a summary of these). 1H NMR spectroscopy confirmed a consistent incorporation of approximately 9% Tpy-MA across all polymers. The SEC results revealed narrow dispersity (<1.25) and similar molar masses ranging from 12![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 500 g mol−1 for P1 to 23
500 g mol−1 for P1 to 23![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 500 g mol−1 for P3, as expected for the RAFT polymerization process. The glass transition temperatures (Tg) obtained from DSC measurements are similar to former results from our group.56,85 Since similar Tpy-MA content, comparable molar masses and comparable degrees of polymerization were obtained, the polymers could be utilized to prepare metallopolymer networks, which are highly comparable. Afterwards, the metallopolymers were synthesized by converting the linear polymers with either FeSO4 × 7 H2O (P1-Fe, P2-Fe, P3-Fe) or Zn(TFMS)2 (P1-Zn, P2-Zn, P3-Zn). These metal salts were dissolved in methanol and added to chloroform solutions of the polymers (see Scheme 1), exploiting the high binding affinity of the terpyridine ligands for metal ions.87,88 All metallopolymers were characterized by EA. The determined compositions are in the expected range (see SI Table S6). DSC, and TGA (see Table 1 and SI Table S6) provided insights into the thermal properties of the metallopolymers. Unfortunately, it was not possible to determine Tg from the DSC measurements for all metallopolymers (see SI Fig. S10–S12). Hence, dynamic mechanical thermal analysis was utilized for the determination, which will be described in a following section. For the degradation temperatures, all metallopolymers featured a lower decomposition temperature (Td) compared to the linear polymers. For example, P2 shows degradation at 238 °C while P2-Fe and P2-Zn exhibit a Td of 210 °C and 215 °C, respectively. Since all metallopolymers have a first degradation between 150 °C and 270 °C they are stable enough for the switching and the following analysis. To provide additional context and to improve the understanding of the metallopolymer properties, polymer networks without ligands or metal salts were synthesized for comparison. MMA (P1-cc), BMA (P2-cc) and 2-EHMA (P3-cc) were crosslinked with 1,6-hexanediol dimethacrylate (HDMA) as the covalent crosslinker and the networks were obtained via free radical polymerization (FRP) leading to a 5% content of HDMA within the polymer, which results in a similar crosslinking density compared to the metallopolymers. These polymer networks were subsequently characterized using EA, DSC, and TGA (see Table S8) to analyze their composition, thermal behavior and stability. The determined compositions via EA were in the expected range. The DSC measurements revealed comparable glass transition temperatures to the linear polymers, but slightly lower values compared to the metallopolymers, while the degradation temperatures lie in the same range, which makes these networks highly useful for comparison. Furthermore, model complexes [(Tpy)2Fe]2+, [(Tpy)2Zn]2+ and poly(methyl methacrylate) (PMMA) were synthesized as well (see SI Tables S1 and S4).
500 g mol−1 for P3, as expected for the RAFT polymerization process. The glass transition temperatures (Tg) obtained from DSC measurements are similar to former results from our group.56,85 Since similar Tpy-MA content, comparable molar masses and comparable degrees of polymerization were obtained, the polymers could be utilized to prepare metallopolymer networks, which are highly comparable. Afterwards, the metallopolymers were synthesized by converting the linear polymers with either FeSO4 × 7 H2O (P1-Fe, P2-Fe, P3-Fe) or Zn(TFMS)2 (P1-Zn, P2-Zn, P3-Zn). These metal salts were dissolved in methanol and added to chloroform solutions of the polymers (see Scheme 1), exploiting the high binding affinity of the terpyridine ligands for metal ions.87,88 All metallopolymers were characterized by EA. The determined compositions are in the expected range (see SI Table S6). DSC, and TGA (see Table 1 and SI Table S6) provided insights into the thermal properties of the metallopolymers. Unfortunately, it was not possible to determine Tg from the DSC measurements for all metallopolymers (see SI Fig. S10–S12). Hence, dynamic mechanical thermal analysis was utilized for the determination, which will be described in a following section. For the degradation temperatures, all metallopolymers featured a lower decomposition temperature (Td) compared to the linear polymers. For example, P2 shows degradation at 238 °C while P2-Fe and P2-Zn exhibit a Td of 210 °C and 215 °C, respectively. Since all metallopolymers have a first degradation between 150 °C and 270 °C they are stable enough for the switching and the following analysis. To provide additional context and to improve the understanding of the metallopolymer properties, polymer networks without ligands or metal salts were synthesized for comparison. MMA (P1-cc), BMA (P2-cc) and 2-EHMA (P3-cc) were crosslinked with 1,6-hexanediol dimethacrylate (HDMA) as the covalent crosslinker and the networks were obtained via free radical polymerization (FRP) leading to a 5% content of HDMA within the polymer, which results in a similar crosslinking density compared to the metallopolymers. These polymer networks were subsequently characterized using EA, DSC, and TGA (see Table S8) to analyze their composition, thermal behavior and stability. The determined compositions via EA were in the expected range. The DSC measurements revealed comparable glass transition temperatures to the linear polymers, but slightly lower values compared to the metallopolymers, while the degradation temperatures lie in the same range, which makes these networks highly useful for comparison. Furthermore, model complexes [(Tpy)2Fe]2+, [(Tpy)2Zn]2+ and poly(methyl methacrylate) (PMMA) were synthesized as well (see SI Tables S1 and S4).
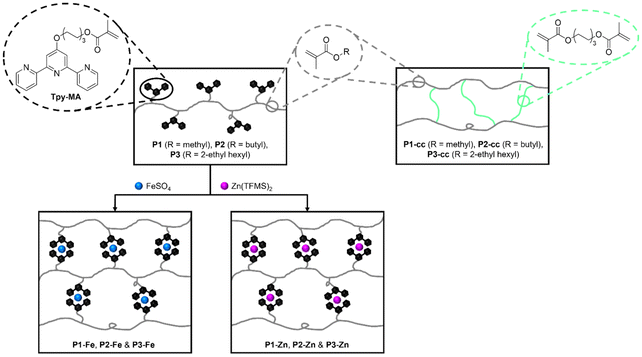 | ||
| Scheme 1 Schematic representation of the synthesis of all metallopolymers P1-Fe to P3-Zn and of the polymer networks P1-cc to P3-cc. | ||
| Polymer | Tpy content [%]a | SECb | T d [°C] | T g [°C] | MP | T d /°C | T g /°C | ||
|---|---|---|---|---|---|---|---|---|---|
| M n/g mol−1 | M w/g mol−1 | Đ | |||||||
| a Determined via NMR spectroscopy (in CD2Cl2 at 300 MHz). b Eluent: CHCl3/NEt3/i-PrOH (94/4/2), PMMA standards. c Degradation temperatures determined via TGA measurements: 25 to 600 °C, 20 K min−1, nitrogen atmosphere. d Glass transition temperatures determined via DSC measurements: −110 to 150 °C, 3rd heating run, 10 K min−1, nitrogen atmosphere. e Glass transition temperatures determined via DMTA: 25 to 100, 130 or 150 °C, 2 K min−1, 1 Hz. | |||||||||
| P1 | 9.3 | 12![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 500 500 |
15![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 100 100 |
1.21 | 167 | 76 | P1-Fe | 148 | 108 |
| P1-Zn | 162 | 121 | |||||||
| P2 | 9.1 | 13![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 300 300 |
15![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 300 300 |
1.15 | 238 | 25 | P2-Fe | 210 | 50 |
| P2-Zn | 215 | 89 | |||||||
| P3 | 9.4 | 23![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 500 500 |
26![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 300 300 |
1.12 | 298 | 3 | P3-Fe | 223 | 50 |
| P3-Zn | 268 | 71 | |||||||
Complexation behavior in solution
Metallopolymers and metal complexes can undergo dynamic and reversible transitions, in particular in solution.89 Understanding the reversibility in solution is also crucial for understanding the reversible behavior in the solid state. Therefore, we firstly investigated the metal complexes, which were utilized for the metallopolymer synthesis, in solution. For this purpose, isothermal titration calorimetry (ITC) was applied, since it provides valuable insights into the binding constants of the metal complexes as it has been demonstrated in other studies.42,57 The results of these already published ITC measurements for Zn(TFMS)2 and FeSO4 with the terpyridine ligand are summarized in Table 2.42,57 Within these measurements, a single binding event with a stoichiometry of 2 was found for the terpyridine ligand and the used metal salts, indicating that two terpyridine units binds simultaneously to one metal ion.42,57 Furthermore, it should be noted that the counter anions can play a significant role in the binding affinities of the complexes, which was previously discussed in literature.63 The choice of counter anion was based on the non-binding nature of the anions to the metal ions, in contrast to, e.g., chloride which acts as a bridging ligand in Mn(II).63 In addition, FeSO4 and Zn(TFMS)2 are both air-stable. Furthermore, the iron complex exhibits a logarithmic association constant of 23 (corresponding to the maximum measurable value with our instrument),57 significantly higher than the 15 observed for the zinc complex.42 Consequently, the bis-terpyridine iron(II) complex is much more stable compared to its zinc analogue. To validate these results, density functional theory (DFT) calculations were performed. The complexation of the two metal ions has been modeled with an isodesmic reaction, using zinc(II) and iron(II) ions that are explicitly coordinated by methanol molecules, which was part of the solvent mixture utilized in the ITC measurements.57 The resulting free Gibbs energy (Gcalc) and the association constant ln![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Kcalc are summarized in Table 2. Here, the logarithmic association constant is again higher for the bis-terpyridine iron complex with 130 compared to the zinc complex with 72, which aligns well with the ITC results. Furthermore, the free Gibbs energy was determined via the DFT calculations. These complex formation energies are higher for the [(Tpy)2Fe]2+ complex with −58 kJ mol−1 from experimental results and −328 kJ mol−1 from theoretical calculations compared to the [(Tpy)2Zn]2+ complex. One should mention that absolute association constants are notoriously difficult to predict, with deviations of the logarithmic constant by a factor of 2 to 10 being very common.90–92 One cause of the reported deviations could be that the calculated values assume pure methanol as the solvent, deviating from the experiment. Additionally, it can be shown that the anion's electrostatic effect plays an important role. Table S13 displays the values for a repeated calculation with additional chloride ions to mimic the effect of the anions, showing a drastic reduction of the calculated association constants and improved alignment with the experiments. If the values for both salts are put into proportion, the result for the relative free Gibbs energies are similar for the experimental and theoretical approach. These results make the two chosen salts very suitable candidates for the formation and investigation of structural changes in metallopolymers, since one more stable and one less stable complex are analyzed.
Kcalc are summarized in Table 2. Here, the logarithmic association constant is again higher for the bis-terpyridine iron complex with 130 compared to the zinc complex with 72, which aligns well with the ITC results. Furthermore, the free Gibbs energy was determined via the DFT calculations. These complex formation energies are higher for the [(Tpy)2Fe]2+ complex with −58 kJ mol−1 from experimental results and −328 kJ mol−1 from theoretical calculations compared to the [(Tpy)2Zn]2+ complex. One should mention that absolute association constants are notoriously difficult to predict, with deviations of the logarithmic constant by a factor of 2 to 10 being very common.90–92 One cause of the reported deviations could be that the calculated values assume pure methanol as the solvent, deviating from the experiment. Additionally, it can be shown that the anion's electrostatic effect plays an important role. Table S13 displays the values for a repeated calculation with additional chloride ions to mimic the effect of the anions, showing a drastic reduction of the calculated association constants and improved alignment with the experiments. If the values for both salts are put into proportion, the result for the relative free Gibbs energies are similar for the experimental and theoretical approach. These results make the two chosen salts very suitable candidates for the formation and investigation of structural changes in metallopolymers, since one more stable and one less stable complex are analyzed.
| Complex | DFT | ITC | |||||
|---|---|---|---|---|---|---|---|
| G calc/kJ mol−1 | G calc(Fe)/Gcalc(Zn) | ln![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Kcalc Kcalc |
G exp/kJ mol−1 | G exp(Fe)/Gexp(Zn) | ln![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Kexp Kexp![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 42,57 42,57 |
n | |
| [(Tpy)2Fe]2+ | −328.277 | 1.81 | 130.306 | −58.009 | 1.54 | 23.026 | 1.91 |
| [(Tpy)2Zn]2+ | −181.657 | 72.107 | −37.767 | 14.991 | 1.90 | ||
Rheological investigation
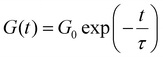 | (1) |
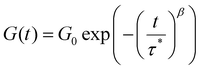 | (2) |
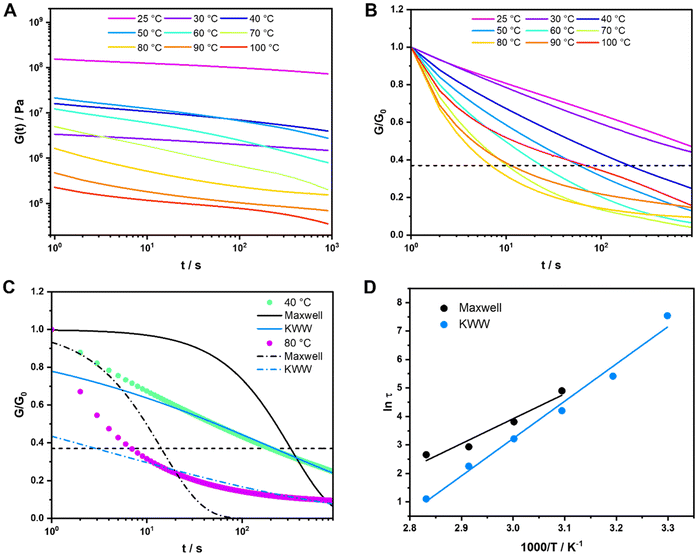
Fig. 2C demonstrates that the Maxwell model does not fit the measured data very well; on the other hand, the stretched exponential function from the KWW model fits the data much better, indicating that the measured samples are not ideal and simple. This is also the case for all other metallopolymers and crosslinked polymers (see SI). Since both models do not fit the measurements perfectly, it would in principle be possible to fit the stress relaxation data using a modified KWW function using a fast and a slow relaxation as shown in literature.112 However, in the following section we report that the presented metallopolymers exhibit multiple, i.e. five to seven, relaxation modes. Therefore, it is not possible to only use a single or a two-term fitting for these plots. Hence, the Maxwell model (eqn (1)) and the KWW model (eqn (2)) are depicted, even though we exhibit more than one relaxation. To compare both shown approaches the following calculations were performed for both models. Using τ from the Maxwell model and τ* from the KWW model, it is possible to calculate the activation energies (EA). Therefore, the Arrhenius correlation of EA was utilized (see eqn (3)).113
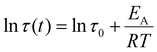 | (3) |
The resulting Arrhenius plots of P2-Zn in a temperature range from 50 °C to 80 °C for the Maxwell model and from 30 °C to 80 °C for the KWW model are presented in Fig. 2D and for the additional metallopolymers and polymer networks in the SI, Fig. S53–S58. Furthermore, the calculated activation energies are summarized in Table 3. The found EA values range from 132.0 kJ mol−1 (P1-Fe) to 41.0 kJ mol−1 (P3-Zn) for the metallopolymers. These values are in the range of activation energies found for other vitrimers114 and are comparable to the free Gibbs energies determined via ITC measurements (see Table 2). However, the values are significantly smaller compared to those of the DFT-modeled Gibbs free energies (Table 2). For P3-Fe it was not possible to determine EA because the relaxation did not follow a vitrimeric behavior. As shown in Table 3 the EA values differ a lot comparing the results from Maxwell and KWW fits. In general, all values for KWW are higher compared to Maxwell. The values for the metallopolymers containing MMA (P1-Fe and P1-Zn) are higher than the ones containing BMA (P2-Fe and P2-Zn) or 2-EHMA (P3-Zn). Only for the MMA-containing metallopolymers, the one containing iron exhibits a higher activation energy compared to its counterpart metallopolymers containing zinc. This result has already been described by the theoretical calculations of the free Gibbs energy (see Table 2) and is also in line with the complexation behavior in solution. However, this is not the case for the other metallopolymers. Another observation is that the EA values determined by the Maxwell and KWW approach of P1-cc and P2-cc lie in the same range as their corresponding metallopolymers P1-Fe, P1-Zn and P2-Fe, P2-Zn, respectively. The comparison to the covalently crosslinked networks reveal that the polymer matrix has a significant influence on the activation energy determined by the stress relaxation measurements. These results lead to the assumption that the resulting EA values are a combination of the activation of the metal–ligand complex and the Tg of the polymer matrix. Another approach to analyze the stress relaxation measurements is to construct a master curve using the time–temperature superposition principle.115 Hereby, the resulting curve was fitted using the KWW equation (eqn (2)) and the resulting shift factors aT could be correlated to the τ* values to calculate the activation energy. The resulting plots are depicted in the SI (Fig. S59–S69) and the EA values are summarized in Table S9. However, not all master curves follow the KWW equation very well, which leads to unrealistic EA values, e.g., for P3-Zn, where an activation energy of 228 kJ mol−1 was obtained while only a value of 42 kJ mol−1 could be determined using the previously described KWW approach. Furthermore, these findings suggest that stress relaxation measurements are not the best method to clarify the structural properties of metallopolymers, i.e. the supramolecular binding motifs of the used bis-terpyridine metal complexes.
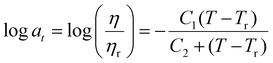 | (4) |
| Sample | WLF C1 | WLF C2/°C | |||||||
|---|---|---|---|---|---|---|---|---|---|
| P1-Fe | T/°C | 100 | 110 |
![[1 with combining low line]](https://www.rsc.org/images/entities/b_char_0031_0332.gif) ![[2 with combining low line]](https://www.rsc.org/images/entities/b_char_0032_0332.gif) ![[0 with combining low line]](https://www.rsc.org/images/entities/b_char_0030_0332.gif)
|
130 | 140 | 28.71 | 174.91 | |
| a T | 3000 | 59 | 1 | 0.027 | 0.0015 | ||||
| b T | 0.85 | 1 | 1 | 1 | 1 | ||||
| P1-Zn | T/°C | 100 | 110 |
![[1 with combining low line]](https://www.rsc.org/images/entities/b_char_0031_0332.gif) ![[2 with combining low line]](https://www.rsc.org/images/entities/b_char_0032_0332.gif) ![[0 with combining low line]](https://www.rsc.org/images/entities/b_char_0030_0332.gif)
|
130 | 140 | 9.84 | 68.38 | |
| a T | 3000 | 55 | 1 | 0.05 | 0.01 | ||||
| b t | 0.9 | 1 | 1 | 1 | 1 | ||||
| P2-Fe | T/°C | 40 | 50 | 60 |
![[7 with combining low line]](https://www.rsc.org/images/entities/b_char_0037_0332.gif) ![[0 with combining low line]](https://www.rsc.org/images/entities/b_char_0030_0332.gif)
|
80 | 90 | 13.99 | 180.26 |
| a T | 800 | 56 | 6.5 | 1 | 0.18 | 0.043 | |||
| P2-Zn | T/°C | 80 | 90 | 100 |
![[1 with combining low line]](https://www.rsc.org/images/entities/b_char_0031_0332.gif) ![[1 with combining low line]](https://www.rsc.org/images/entities/b_char_0031_0332.gif) ![[0 with combining low line]](https://www.rsc.org/images/entities/b_char_0030_0332.gif)
|
120 | 130 | 8.15 | 109.85 |
| a T | 490 | 55 | 7 | 1 | 0.21 | 0.06 | |||
| P3-Fe | T/°C | 40 | 50 | 60 |
![[7 with combining low line]](https://www.rsc.org/images/entities/b_char_0037_0332.gif) ![[0 with combining low line]](https://www.rsc.org/images/entities/b_char_0030_0332.gif)
|
80 | 90 | 10.20 | 141.44 |
| a T | 380 | 45 | 6.1 | 1 | 0.21 | 0.06 | |||
| P3-Zn | T/°C | 70 | 80 | 90 |
![[1 with combining low line]](https://www.rsc.org/images/entities/b_char_0031_0332.gif) ![[0 with combining low line]](https://www.rsc.org/images/entities/b_char_0030_0332.gif) ![[0 with combining low line]](https://www.rsc.org/images/entities/b_char_0030_0332.gif)
|
110 | 120 | 31.75 | 619.72 |
| a T | 150 | 15 | 3 | 1 | 0.3 | 0.1 | |||
| P1-cc | T/°C | 90 | 100 | 110 |
![[1 with combining low line]](https://www.rsc.org/images/entities/b_char_0031_0332.gif) ![[2 with combining low line]](https://www.rsc.org/images/entities/b_char_0032_0332.gif) ![[0 with combining low line]](https://www.rsc.org/images/entities/b_char_0030_0332.gif)
|
130 | 140 | 4.56 | 63.90 |
| a T | 4500 | 120 | 7 | 1 | 0.23 | 0.1 | |||
| b T | 0.85 | 0.9 | 0.95 | 1 | 1.05 | 1.1 | |||
In this equation two material constants C1 and C2, often described as the WLF parameters, are utilized, while Tr is the reference temperature for generating the master curve.
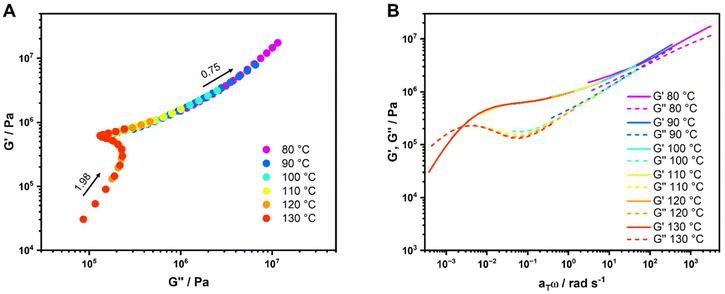
For this study, the measurements were conducted around Tg so the WLF-equation is applicable. The WLF parameters C1 and C2 were determined via linear plotting (see SI, Fig. S95–S103). The results of this analysis are presented in Table 4. The WLF parameters vary significantly and there is no clear trend in comparison between the metal ions used. Overall, the TTS principle is a good way to analyze the metallopolymers, but no other structural changes than in the frequency sweeps (i.e. activation of the complexes at higher temperatures) could be determined.
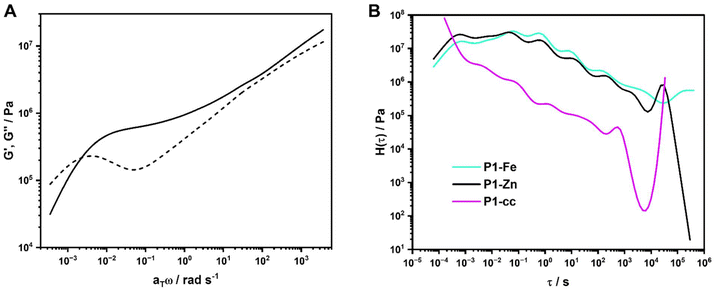 | ||
| Fig. 4 (A) Computational master curve of P2-Zn at Tref = 120 °C, (B) computational relaxation spectrum of P1-Fe, P1-Zn and P1-cc. | ||
For better understanding of the master curves, we decided to model relaxation spectra H(τ)134,138 using the RheoSpec program. These plots reveal the underlying relaxation modes that cannot be seen in any other measurements. Since the derivation of H(τ) from the frequency sweep measurements of the storage and loss modulus over a limited frequency range is hard, it is only possible to calculate an approximate function, because small changes in G′ and G″ from the experimental data lead to variations in the relaxation spectrum.137
There are two different methods for the use of relaxation spectra, the discrete139 and the continuous mode.140 We used the continuous mode, since it is a more compact method to study the underlying relaxation responses.135,137 The resulting spectra of the samples can be found in SI (Fig. S113–S121). For a better understanding of this data, a comparison between the samples with the same comonomer and the samples containing the same metal salt were carried out (see Fig. 4B and Fig. S122–S125). In Fig. 4B all samples containing MMA as the comonomer are summarized, here all three relaxation curves look very similar. P1-Fe and P1-Zn look almost identical, the sample containing Zn(II) is just shifted slightly to longer times. Since the frequency sweep measurements were performed up to 140 °C, the Tg of PMMA is quite close, this leads to a better resolution in the relaxation spectra. For the samples containing BMA and 2-EHMA as a comonomer (see SI, Fig. S122 and S123) the Tg is lower, and the conducted temperature range in the frequency sweep measurements are much higher compared to the glass transition temperature. As for those samples, there is no clear difference between the relaxation curves. Furthermore, when comparing all samples containing Fe(II) (see Fig. S124) or Zn(II) (see Fig. 4B) no clear distinction between the BMA- and 2-EHMA-containing metallopolymers is possible.
There are about seven different peaks in the H(τ) spectrum for the MMA-containing samples and around five to six peaks for the BMA- and EHMA-containing samples. This leads to the conclusion that the metallopolymers are very complex materials and there are multiple relaxations visible for the metal–ligand complex itself and the polymer backbone. In literature, there are often only two distinctive peaks shown in the H(τ) spectra that could be assigned to distinctive relaxation processes.141–143 However, there are also examples of polymers featuring more complex behavior, like polyimides134 or polyurethanes.144
Raman spectroscopy and DFT modelling
Raman spectroscopy is a labelfree analytical technique that provides highly specific molecular information. It has extensively been applied to study ligand binding, complexation, and structural changes in various systems.57,85,145 Building on its well-established molecular specificity, Raman spectroscopy is used in this study to validate the formation of metal complexes within the polymeric matrix and to investigate their behavior at different temperatures, thereby providing deeper insight into their molecular-level structural dynamics. To achieve this, the free terpyridine monomer (Tpy-MA), as well as the zinc and iron model complexes, [(Tpy)2Zn]2+ and [(Tpy)2Fe]2+ were first investigated to identify characteristic vibrational bands that could serve as marker bands to confirm whether Zn(TFMS)2 or FeSO4 metal salts successfully complex with the terpyridine ligand. Upon complexation, significant Raman spectral changes were observed, particularly in the zinc complex [(Tpy)2Zn]2+, where the most pronounced changes occurred (Fig. S126). For instance, the Raman band at 339 cm−1 in free Tpy-MA spectrum is shifted to 349 cm−1 in the zinc complex, [(Tpy)2Zn]2+, accompanied by the appearance of a new band at 313 cm−1, forming a doublet attributed to the N–Zn stretching vibration.146 A higher intensity and slight shifting of the above-mentioned band can also be seen in the DFT modeled Raman spectra (see SI, Fig. S147 and S148) supporting the experimental results. Here the band at approximately 324 cm−1 for the free ligand is shifted to 326 cm−1 with a much higher intensity for the bis-terpyridine zinc complex. In contrast, iron complexation results in a broad band at 310 cm−1 (see Fig. S126) corresponding to N–Fe stretching vibrations. These shifts occur due to coordination of the terpyridine nitrogen to the metal center, where the metal accepts additional electrons density from the terpyridine ligand. This electron transfer also quenches the intense δ(C–C) band at 997 cm−1 observed in the free Tpy-MA spectrum, resulting in a significant reduction in intensity for [(Tpy)2Fe]2+ and almost complete disappearance in [(Tpy)2Zn]2+. This phenomenon can also be seen in Fig. S147 and S148 of the modeled spectra where the mentioned band for the free terpyridine ligand disappears for both complexes. The weak doublet at 457 cm−1 and 469 cm−1 in the free Tpy-MA spectrum merges into a broader band at about 461 cm−1 in [(Tpy)2Fe]2+, reflecting the distinct electronic and geometric environment introduced by iron complexation. This band is assigned to the N–Fe stretching vibration.146,147 Moving to the higher wavenumbers, the Raman band at 644 cm−1 intensifies and blue-shifts by approximately 6 cm−1 for both zinc and iron complexes. Meanwhile, the band at 623 cm−1 shifts to 639 cm−1 in the zinc complex and appears as a shoulder in the iron complex. In the breathing modes spectral range, the δ(C–C) and δ(C–H) bands at 1030 cm−1 and 1041 cm−1 shifts and intensify to 1020 cm−1 and 1030 cm−1 for the zinc complex and 1023 cm−1 and 1039 cm−1 for the iron complex. The higher wavenumber shift and the intensification of the 1023 cm−1 band in the iron complex, suggests a stronger coordination of the terpyridine ligand to Fe2+ than to Zn2+, which is in line with the ITC results mentioned above.147 The described shifts are also visible in the modeled spectra, where at approximately 1020 cm−1 a significant increase in intensity for both complexes is visible. At even higher wavenumbers, the νas(C–C) band at 1201 cm−1 shifts and merges with another νas(C–C) band at 1251 cm−1 to form a single band at 1264 cm−1 in the zinc complex. This merging is likely due to mode coupling during zinc coordination. In contrast, a new band emerges at 1289 cm−1 in the iron complex, indicating a unique vibrational mode that arises exclusively from the iron coordination corresponding to a newly formed band for the C−C bond.148 The ν(N–C) broad band at about 1400 cm−1 also undergoes significant changes, broadening into two bands centered at 1435 cm−1. The symmetric doublet ν(CNC, C–C) at 1446 cm−1 and 1568 cm−1 shifts to 1474 cm−1 and 1499 cm−1 in the zinc complex and to 1469 cm−1 in the iron complex. Additionally, the ν(CC, CN) band at 1356 cm−1 shifts by +12 cm−1 in the zinc complex and by +6 cm−1 in the iron complex. These shifts arise due to geometrical changes and mode coupling effects during complexation.146 Furthermore, complex changes in the ν(CN, CC) region between 1550 and 1650 cm−1 were observed, which might be attributed to cis-to-trans transformations during complex formation.145,146,149 A comparison of the determined band positions of the metal complexes by Raman and by DFT calculations can be found in the SI Table S14. To ensure that these identified characteristic bands can be used for validation, the ligand-containing polymers (P1, P2, and P3) without the metal complexes were analyzed to check for similarities. The analysis revealed that most of the identified bands can reliably confirm the presence of the complexes. However, the bands at 639 cm−1, 700 cm−1, 1030 cm−1 (zinc complex) and 1039 cm−1 (iron complex), and the region 1550 cm−1 to 1650 cm−1 overlaps with the polymer matrix, making them unsuitable for validation. The metallopolymers were subsequently measured and analyzed to validate the formation of the metal complexes. As shown in Fig. 5A and B for the iron and zinc-based metallopolymers, respectively, the identified characteristic Raman bands indicative for complexation, as described above, were consistently present across all metallopolymers. Some bands exhibited shifts of up to +5 cm−1, while others remain unchanged (localized and unaffected by the polymeric matrix). Additionally, broadening of certain vibrational modes, such as the band at 1264 cm−1, the band at 754 cm−1, and even the N–Zn band at 313 cm−1, was observed. This broadening and shifting are attributed to the influence of the polymeric environment on the vibrational modes. Furthermore, it was observed with the P1-Zn metallopolymer that new bands emerged outside the previously identified characteristic bands following complexation. These new bands appear at approximately 263 cm−1, 367 cm−1, and 667 cm−1 and were also observed in the crosslinked polymer P1-cc, although their signal intensities were weak, with the exception of the 367 cm−1 band (see SI Fig. S140). The bands at 263 cm−1 and 367 cm−1 corresponds to C–C/C–O out of plane bending, while the band at 667 cm−1 is attributed to the wagging vibration of the C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O in the ester moiety of the methacrylate.150–152 Upon complexation, these signals become significantly more intense, displaying almost equal intensities showing an influence of the metal complex spectrum by the presence of the surrounding polymer. Afterwards, temperature-dependent Raman measurements were conducted with a focus of the complexed metallopolymers P1-Fe to P3-Zn. Interestingly, during these measurements, the intensities of the complexation sensitive Raman bands decreased as the temperature increased (see SI, Fig. S141). This can be attributed to the fact that an increase in temperature disrupts the influence of the polymer environment on the coordination of the metal complex. However, the band positions remained relatively unchanged, except for the 667 cm−1 band, which shifted slightly to 665 cm−1 at 130 °C and further to 663 cm−1 at 140 °C and 150 °C. This might suggest a subtle change in the coordination environment and its geometry and also corresponds to the changes observed in the rheology at those temperatures. Thus, these changes in the molecular pattern fits to the observed reversibility in the frequency sweeps (and in the TTS) for the zinc complexes. The characteristic ν(Zn–N) bands at 312 cm−1 and 349 cm−1 showed minimal shifts in peak position for increasing temperature, highlighting the stability of the metal–ligand complex even at elevated temperatures. This finding corresponds perfectly to the DMTA measurements, in which G′ is also higher than G″. Specifically, at 140 °C, the band at 312 cm−1 slightly shifts to 310 cm−1 before returning to 312 cm−1 at 150 °C. The 349 cm−1 band also exhibited minor back-and-forth shifts of 2 cm−1 starting from 70 °C. It should be noted that these fluctuations do not follow any observable trend, suggesting that the complex remains stable and localized, with no significant structural change in the metal complex. Similar characteristics were observed for the Zn–N vibration bands in the other metallopolymers, P2-Zn and P3-Zn (see SI, Fig. S142, S143 and Table S12 for the Raman band positions with respect to temperature.) The temperature-dependent Raman analysis shows that the observed Raman spectral changes arise primarily from thermally induced morphological changes in the polymer matrix and localized vibrational changes in the coordination environment of the Zn2+ ions, while the terpyridine-zinc complex itself remains intact. This suggests a molecular mechanism that enables the reversible behavior of the metallopolymers. The finding is well in line with the DMTA measurements, which suggest a polymer network even at higher temperatures and no opening of the bonds (which would correspond to a dissociative mechanism). However, the dynamic behavior in the frequency-dependent measurements might be explainable with these structural changes observed in the temperature-dependent Raman measurements, since the reversibility by exchange reaction will presumably lead to a change in the molecular environment of the metal complexes. Alternatively, it could also be possible that certain aggregates, e.g., ionic clusters are formed and the thermal treatment would lead to an exchange/reversibility of these. Such aggregates are known for supramolecular polymers containing polar groups in a non-polar polymer environment.55,86,97,153,154 Fig. S144 through S146 illustrates the temperature-dependent Raman spectra of the P1-Fe, P2-Fe, and P3-Fe metallopolymers, respectively. Unfortunately, a significant loss in spectra intensity is observed for P1-Fe and P2-Fe at temperatures above 80 °C, while P3-Fe exhibits a similar decline beginning at around 100 °C. Here, the decline in intensity could be correlated to a temperature-dependent process that leads to environmental changes in the polymer network without significantly influencing the chemical structure. Sample degradation could be excluded, which was proven by the TGA and rheology measurements indicating stability of the metallopolymers at these high temperatures. Thus, these polymers could not be investigated in detail by temperature-dependent Raman measurements.
O in the ester moiety of the methacrylate.150–152 Upon complexation, these signals become significantly more intense, displaying almost equal intensities showing an influence of the metal complex spectrum by the presence of the surrounding polymer. Afterwards, temperature-dependent Raman measurements were conducted with a focus of the complexed metallopolymers P1-Fe to P3-Zn. Interestingly, during these measurements, the intensities of the complexation sensitive Raman bands decreased as the temperature increased (see SI, Fig. S141). This can be attributed to the fact that an increase in temperature disrupts the influence of the polymer environment on the coordination of the metal complex. However, the band positions remained relatively unchanged, except for the 667 cm−1 band, which shifted slightly to 665 cm−1 at 130 °C and further to 663 cm−1 at 140 °C and 150 °C. This might suggest a subtle change in the coordination environment and its geometry and also corresponds to the changes observed in the rheology at those temperatures. Thus, these changes in the molecular pattern fits to the observed reversibility in the frequency sweeps (and in the TTS) for the zinc complexes. The characteristic ν(Zn–N) bands at 312 cm−1 and 349 cm−1 showed minimal shifts in peak position for increasing temperature, highlighting the stability of the metal–ligand complex even at elevated temperatures. This finding corresponds perfectly to the DMTA measurements, in which G′ is also higher than G″. Specifically, at 140 °C, the band at 312 cm−1 slightly shifts to 310 cm−1 before returning to 312 cm−1 at 150 °C. The 349 cm−1 band also exhibited minor back-and-forth shifts of 2 cm−1 starting from 70 °C. It should be noted that these fluctuations do not follow any observable trend, suggesting that the complex remains stable and localized, with no significant structural change in the metal complex. Similar characteristics were observed for the Zn–N vibration bands in the other metallopolymers, P2-Zn and P3-Zn (see SI, Fig. S142, S143 and Table S12 for the Raman band positions with respect to temperature.) The temperature-dependent Raman analysis shows that the observed Raman spectral changes arise primarily from thermally induced morphological changes in the polymer matrix and localized vibrational changes in the coordination environment of the Zn2+ ions, while the terpyridine-zinc complex itself remains intact. This suggests a molecular mechanism that enables the reversible behavior of the metallopolymers. The finding is well in line with the DMTA measurements, which suggest a polymer network even at higher temperatures and no opening of the bonds (which would correspond to a dissociative mechanism). However, the dynamic behavior in the frequency-dependent measurements might be explainable with these structural changes observed in the temperature-dependent Raman measurements, since the reversibility by exchange reaction will presumably lead to a change in the molecular environment of the metal complexes. Alternatively, it could also be possible that certain aggregates, e.g., ionic clusters are formed and the thermal treatment would lead to an exchange/reversibility of these. Such aggregates are known for supramolecular polymers containing polar groups in a non-polar polymer environment.55,86,97,153,154 Fig. S144 through S146 illustrates the temperature-dependent Raman spectra of the P1-Fe, P2-Fe, and P3-Fe metallopolymers, respectively. Unfortunately, a significant loss in spectra intensity is observed for P1-Fe and P2-Fe at temperatures above 80 °C, while P3-Fe exhibits a similar decline beginning at around 100 °C. Here, the decline in intensity could be correlated to a temperature-dependent process that leads to environmental changes in the polymer network without significantly influencing the chemical structure. Sample degradation could be excluded, which was proven by the TGA and rheology measurements indicating stability of the metallopolymers at these high temperatures. Thus, these polymers could not be investigated in detail by temperature-dependent Raman measurements.
Conclusions
In the current study, our aim was to understand and describe the structural changes happening in metallopolymers. We therefore prepared metallopolymers containing terpyridine moieties as ligands in the side chains that form homoleptic complexes with Fe(II) or Zn(II) ions. The metallopolymers were characterized using DSC, TGA and elemental analysis. Rheological investigations were conducted, focusing on DMTA, stress relaxation, frequency sweeps and the TTS principle. Herein we concluded that frequency sweeps are the best method to characterize structural changes in such metallopolymers. Here it was possible to observe crossovers in the plots for the zinc containing bis-terpyridine complexes, which is well aligned with the higher reversibility of this bond. Stress relaxation measurements revealed activation energies of the overall relaxation process (i.e. polymer matrix and metal complexes). Furthermore, computational rheology was applied to construct master curves and relaxation spectra. Here it was possible to see different relaxation modes for the Fe(II) and Zn(II) containing metallopolymers clearly indicating a reversibility/relaxation caused by the metal complexes. For further investigation, Raman spectroscopy and DFT calculations were conducted. The formation of the complexes could be demonstrated with both methods, and temperature-dependent Raman spectroscopy revealed no significant structural changes of the metal complexes but rather a thermal-induced morphological change of the polymer matrix. These findings align with the DMTA measurements, where no dissociation of the bonds of the polymer network could be observed. The frequency-dependent dynamics may stem from reversible structural changes, which are also observable in the temperature-dependent Raman spectra, likely due to exchange reactions of metal complexes or exchanges within certain aggregates.Overall, this study demonstrates that dynamic metallopolymers can be comprehensively characterized using a combination of complementary analytical techniques. In particular, DMTA, frequency-dependent measurements and TTS are very useful rheological approaches, whereas Raman spectroscopy in combination with DFT calculations provides valuable insights into the molecular structure.
In future studies, other ligands and metal salts should be studied to investigate their structural behavior and see if those changes occur only within the polymer matrix or within metal complexes inside the metallopolymers.
Author contributions
Synthesis of monomer, polymers and metallopolymers: M. J.; SEC, DSC, TGA: M. J.; rheology measurements: M. J.; Raman measurements: M. F. A.; DFT calculations: W. T. S., C. W.; RheoSpec: J. K.; ITC-measurements: T. B.; writing of the manuscript: M. J., M. F. A., S. Z.; interpretation of data: M. J., M. F. A., S. Z., M. S.; supervision: S. Z., A. C., M. S., J. P., S. G., M. D. H., U. S. S.; concept of the study: S. Z., M. D. H., U. S. S.; correction of the manuscript: S. Z., A. C., M. S., J. P., S. G., M. D. H., U. S. S.; funding: J. P., S. G., M. D. H., U. S. S.Conflicts of interest
There are no conflicts to declare.Data availability
The data supporting this article is included in the supplementary information (SI). Supplementary information: information about the used chemicals and instruments, the synthesis of the polymers, metallopolymers and polymer networks as well as detailed information about the characterization of those, e.g., NMR, SEC, DSC, TGA and elemental analysis data and curves. Additionally, the plots of the DMTA, frequency sweeps, stress relaxation, Arrhenius and TTS; the measured Raman spectra as well as the DFT modeled Raman spectra. See DOI: https://doi.org/10.1039/d6py00053c.All primary data can be found under the following https://doi.org/10.5281/zenodo.18182255.
Acknowledgements
The authors would like to thank the Deutsche Forschungsgemeinschaft (research unit FuncHeal, FOR 5301, projects P1, P5 and P6, project number: 455748945) for financial support.References
- D. Hao, G. R. Asrar, Y. Zeng, X. Yang, X. Li, J. Xiao, K. Guan, J. Wen, Q. Xiao, J. A. Berry and M. Chen, Global Change Biol., 2021, 27, 2144–2158 CrossRef CAS PubMed.
- C. Ciaccio, A. Coletta and M. Coletta, Mol. Aspects Med., 2022, 84, 101022 CrossRef CAS PubMed.
- R. M. Winslow, Respir. Physiol. Neurobiol., 2007, 158, 121–127 CrossRef CAS PubMed.
- M. B. Oliveira and J. F. Mano, Biotechnol. Prog., 2011, 27, 897–912 CrossRef CAS PubMed.
- A. Göthlich, S. Koltzenburg and G. Schornick, Chem. Unserer Zeit, 2005, 39, 262–273 CrossRef.
- K. Lee, J. Y. Kim, S. H. Park, S. H. Kim, S. Cho and A. J. Heeger, Adv. Mater., 2007, 19, 2445–2449 CrossRef CAS.
- N. Roy, B. Bruchmann and J.-M. Lehn, Chem. Soc. Rev., 2015, 44, 3786–3807 RSC.
- F. Liu and M. W. Urban, Prog. Polym. Sci., 2010, 35, 3–23 CrossRef CAS.
- M. Wei, Y. Gao, X. Li and M. J. Serpe, Polym. Chem., 2017, 8, 127–143 RSC.
- A. Bratek-Skicki, Appl. Surf. Sci. Adv., 2021, 4, 100068 CrossRef.
- F. García and M. M. J. Smulders, J. Polym. Sci., Part A:Polym. Chem., 2016, 54, 3551–3577 CrossRef PubMed.
- Y. Yang and M. W. Urban, Adv. Mater. Interfaces, 2018, 5, 1800384 CrossRef.
- S. J. Rowan, S. J. Cantrill, G. R. L. Cousins, J. K. M. Sanders and J. F. Stoddart, Angew. Chem., Int. Ed., 2002, 41, 898–952 CrossRef PubMed.
- Y. Chujo, K. Sada and T. Saegusa, Macromolecules, 1990, 23, 2636–2641 CrossRef CAS.
- S. Terryn, J. Brancart, E. Roels, R. Verhelle, A. Safaei, A. Cuvellier, B. Vanderborght and G. Van Assche, Macromolecules, 2022, 55, 5497–5513 CrossRef CAS.
- W. T. Schulze, S. Schwalbe, K. Trepte, A. Croy, J. Kortus and S. Gräfe, J. Chem. Phys., 2023, 158, 164102 CrossRef CAS PubMed.
- B. Zhou, T. Deng, C. Yang, M. Wang, H. Yan, Z. Yang, Z. Wang and Z. Xue, Adv. Funct. Mater., 2023, 33, 2212005 CrossRef CAS.
- O. R. Cromwell, J. Chung and Z. Guan, J. Am. Chem. Soc., 2015, 137, 6492–6495 CrossRef CAS PubMed.
- C. Kim, H. Ejima and N. Yoshie, J. Mater. Chem. A, 2018, 6, 19643–19652 RSC.
- J. J. Cash, T. Kubo, A. P. Bapat and B. S. Sumerlin, Macromolecules, 2015, 48, 2098–2106 CrossRef CAS.
- H. Otsuka, K. Aotani, Y. Higaki, Y. Amamoto and A. Takahara, Macromolecules, 2007, 40, 1429–1434 CrossRef CAS.
- M. Aiba, T. Higashihara, M. Ashizawa, H. Otsuka and H. Matsumoto, Macromolecules, 2016, 49, 2153–2161 CrossRef CAS.
- A. Chao, I. Negulescu and D. Zhang, Macromolecules, 2016, 49, 6277–6284 CrossRef CAS.
- G. P. Carden, M. L. Martins, G. Toleutay, S. Ge, B. Li, S. Zhao and A. P. Sokolov, Macromolecules, 2024, 57, 8621–8631 CrossRef CAS.
- H. Otsuka, S. Nagano, Y. Kobashi, T. Maeda and A. Takahara, Chem. Commun., 2010, 46, 1150–1152 RSC.
- Q. Zhang, D.-H. Qu, B. L. Feringa and H. Tian, J. Am. Chem. Soc., 2022, 144, 2022–2033 CrossRef CAS PubMed.
- Y. Li, Y. Wang, S. Wang, Z. Ye, C. Bian, X. Xing, T. Hong and X. Jing, Macromolecules, 2022, 55, 9091–9102 CrossRef CAS.
- M. A. Bin Rusayyis and J. M. Torkelson, ACS Macro Lett., 2022, 11, 568–574 CrossRef CAS PubMed.
- C. Du, X. N. Zhang, T. L. Sun, M. Du, Q. Zheng and Z. L. Wu, Macromolecules, 2021, 54, 4313–4325 CrossRef CAS.
- Z. Zhou, S. Chen, X. Xu, Y. Chen, L. Xu, Y. Zeng and F. Zhang, Prog. Org. Coat., 2021, 154, 106213 CrossRef CAS.
- R. Bai, H. Zhang, X. Yang, J. Zhao, Y. Wang, Z. Zhang and X. Yan, Polym. Chem., 2022, 13, 1253–1259 RSC.
- Y. Liu, J. Wan, X. Zhao, J. Zhao, Y. Guo, R. Bai, Z. Zhang, W. Yu, H. W. Gibson and X. Yan, Angew. Chem., Int. Ed., 2023, 62, e202302370 CrossRef CAS PubMed.
- J. Dai, Z. Wang, Z. Wu, Z. Fang, S. Heliu, W. Yang, Y. Bai and X. Zhang, ACS Appl. Polym. Mater., 2023, 5, 2575–2582 CrossRef CAS.
- X. Yuan, X. Lin, F. Dong, X. Huang, H. Liu and X. Xu, ACS Appl. Polym. Mater., 2025, 7, 1328–1337 CrossRef CAS.
- D. Zhao, J. Yang, X. Tian, J. Wei, Q. Li and Y. Wang, Chem. Eng. J., 2022, 434, 134806 CrossRef CAS.
- G. Chen, B. Jin, Q. Zhao and T. Xie, J. Mater. Chem. A, 2021, 9, 6827–6830 RSC.
- A. W. Bosman, R. P. Sijbesma and E. W. Meijer, Mater. Today, 2004, 7, 34–39 CrossRef CAS.
- P. Ximenis, D. Martínez, L. Rubert and B. Soberats, Chem. Soc. Rev., 2025, 54, 11659–11698 RSC.
- H.-J. Schneider and A. K. Yatsimirsky, Chem. Soc. Rev., 2008, 37, 263–277 RSC.
- Y. Zhu, W. Zheng, W. Wang and H.-B. Yang, Chem. Soc. Rev., 2021, 50, 7395–7417 RSC.
- D. Mozhdehi, J. A. Neal, S. C. Grindy, Y. Cordeau, S. Ayala, N. Holten-Andersen and Z. Guan, Macromolecules, 2016, 49, 6310–6321 CrossRef CAS.
- J. Meurer, J. Hniopek, T. Bätz, S. Zechel, M. Enke, J. Vitz, M. Schmitt, J. Popp, M. D. Hager and U. S. Schubert, Adv. Mater., 2021, 33, 2006655 CrossRef CAS PubMed.
- A. C. Jackson, F. L. Beyer, S. C. Price, B. C. Rinderspacher and R. H. Lambeth, Macromolecules, 2013, 46, 5416–5422 CrossRef CAS.
- R. D. Mukhopadhyay and A. Ajayaghosh, Chem. Soc. Rev., 2023, 52, 8635–8650 RSC.
- S. Götz, S. Zechel, M. D. Hager, G. R. Newkome and U. S. Schubert, Prog. Polym. Sci., 2021, 119, 101428 CrossRef.
- Y. Yao, S. Jin, H. Zou, L. Li, X. Ma, G. Lv, F. Gao, X. Lv and Q. Shu, J. Mater. Sci., 2021, 56, 6549–6580 CrossRef CAS.
- K. P. Nair, V. Breedveld and M. Weck, Macromolecules, 2008, 41, 3429–3438 CrossRef CAS.
- C. Sun, F. Pan, H. Bin, J. Zhang, L. Xue, B. Qiu, Z. Wei, Z.-G. Zhang and Y. Li, Nat. Commun., 2018, 9, 743 Search PubMed.
- K. C. Gupta and A. K. Sutar, Coord. Chem. Rev., 2008, 252, 1420–1450 CrossRef CAS.
- D.-L. Ma, H.-Z. He, K.-H. Leung, D. S.-H. Chan and C.-H. Leung, Angew. Chem., Int. Ed., 2013, 52, 7666–7682 CrossRef CAS PubMed.
- A. R. Yaul, V. V. Dhande, G. B. Pethe and A. S. Aswar, Bull. Chem. Soc. Ethiop., 2014, 28, 255–264 CrossRef.
- Y. Liu, S.-C. Yiu, C.-L. Ho and W.-Y. Wong, Coord. Chem. Rev., 2018, 375, 514–557 Search PubMed.
- Y. Sekine, R. Akiyoshi and S. Hayami, Coord. Chem. Rev., 2022, 469, 214663 CrossRef CAS.
- S. D. Bella, Chem. Soc. Rev., 2001, 30, 355–366 RSC.
- B. Sandmann, S. Bode, M. D. Hager and U. S. Schubert, in Adv. Polym. Sci, ed. V. Percec, Springer International Publishing, Cham, 2013, pp. 239–257 Search PubMed.
- S. Bode, R. K. Bose, S. Matthes, M. Ehrhardt, A. Seifert, F. H. Schacher, R. M. Paulus, S. Stumpf, B. Sandmann, J. Vitz, A. Winter, S. Hoeppener, S. J. Garcia, S. Spange, S. van der Zwaag, M. D. Hager and U. S. Schubert, Polym. Chem., 2013, 4, 4966–4973 RSC.
- J. Meurer, T. Bätz, J. Hniopek, S. Zechel, M. Schmitt, J. Popp, M. D. Hager and U. S. Schubert, J. Mater. Chem. A, 2021, 9, 15051–15058 RSC.
- J. Meurer, T. Bätz, J. Hniopek, M. Jäger, S. Zechel, M. Schmitt, J. Popp, M. D. Hager and U. S. Schubert, Polymers, 2022, 14, 1833 CrossRef CAS PubMed.
- F. Ciardelli, E. Tsuchida and D. Wöhrle, in Macromolecule-Metal Complexes, Springer, Berlin, Heidelberg, 1996 Search PubMed.
- A. Wild, A. Winter, F. Schlütter and U. S. Schubert, Chem. Soc. Rev., 2011, 40, 1459–1511 RSC.
- W.-Y. Wong, S.-M. Chan, K.-H. Choi, K.-W. Cheah and W.-K. Chan, Macromol. Rapid Commun., 2000, 21, 453–457 CrossRef CAS.
- P. Yang, P. Pageni, M. P. Kabir, T. Zhu and C. Tang, ACS Macro Lett., 2016, 5, 1293–1300 CrossRef CAS PubMed.
- S. Bode, M. Enke, R. K. Bose, F. H. Schacher, S. J. Garcia, S. van der Zwaag, M. D. Hager and U. S. Schubert, J. Mater. Chem. A, 2015, 3, 22145–22153 RSC.
- S. Götz, M. Abend, S. Zechel, M. D. Hager and U. S. Schubert, J. Appl. Polym. Sci., 2019, 136, 47064 CrossRef.
- T. Bätz, H. H. Haeri, S. Langstein, S. Zechel, M. D. Hager, D. Hinderberger and U. S. Schubert, Polymer, 2024, 307, 127258 CrossRef.
- M. Enke, S. Bode, J. Vitz, F. H. Schacher, M. J. Harrington, M. D. Hager and U. S. Schubert, Polymer, 2015, 69, 274–282 CrossRef CAS.
- W. Bian, H. Lian, Y. Zhang, F. Tai, H. Wang, Q. Dong, B. Yu, X. Wei and Q. Zhao, J. Organomet. Chem., 2017, 835, 25–30 CrossRef CAS.
- C. F. Pereira, A. Olean-Oliveira, D. N. David-Parra and M. F. S. Teixeira, Talanta, 2018, 190, 119–125 CrossRef CAS PubMed.
- J. Polozhentseva, M. Novozhilova and M. Karushev, Int. J. Mol. Sci., 2022, 23, 1795 CrossRef CAS PubMed.
- H. Zhan, S. Lamare, A. Ng, T. Kenny, H. Guernon, W.-K. Chan, A. B. Djurišić, P. D. Harvey and W.-Y. Wong, Macromolecules, 2011, 44, 5155–5167 CrossRef CAS.
- M. Ahmadi, C. Sprenger, G. Pareras, A. Poater and S. Seiffert, Soft Matter, 2023, 19, 8112–8123 RSC.
- R. Dobrawa and F. Würthner, J. Polym. Sci., Part A:Polym. Chem., 2005, 43, 4981–4995 CrossRef CAS.
- K. N. Aziz, K. M. Ahmed, R. A. Omer, A. F. Qader and E. I. Abdulkareem, Rev. Inorg. Chem., 2025, 45, 1–19 CrossRef CAS.
- A. Kumar, S. Bawa, J. Bera, U. Shankar, S. Sahu and A. Bandyopadhyay, J. Appl. Polym. Sci., 2023, 140, e53242 CrossRef CAS.
- J. Fritsch, D. Mansfeld, M. Mehring, R. Wursche, J. Grothe and S. Kaskel, Polymer, 2011, 52, 3263–3268 CrossRef CAS.
- Y. Liang, D. Strohecker, V. Lynch, B. J. Holliday and R. A. Jones, ACS Appl. Mater. Interfaces, 2016, 8, 34568–34580 CrossRef CAS PubMed.
- J. J. Walsh, Q. Zeng, R. J. Forster and T. E. Keyes, Photochem. Photobiol. Sci., 2012, 11, 1547–1557 CrossRef CAS PubMed.
- Z. Zhang, Y.-N. He, L. Liu, X.-Q. Lü, X.-J. Zhu, W.-K. Wong, M. Pan and C.-Y. Su, Chem. Commun., 2016, 52, 3713–3716 RSC.
- W.-X. Feng, S.-Y. Yin, M. Pan, H.-P. Wang, Y.-N. Fan, X.-Q. Lü and C.-Y. Su, J. Mater. Chem. C, 2017, 5, 1742–1750 RSC.
- D. Zhao, L. Guo, Q. Li, C. Yue, B. Han, K. Liu and H. Li, Adv. Mater., 2024, 36, 2405164 CrossRef CAS PubMed.
- N. Hannewald, M. Enke, I. Nischang, S. Zechel, M. D. Hager and U. S. Schubert, J. Inorg. Organomet. Polym. Mater., 2020, 30, 230–242 CrossRef CAS.
- H. A. Barnes, Rheol. Rev., 2003, 1–36 Search PubMed.
- T. Okada, K. Komatsu, T. Kawamoto, T. Yamanaka and H. Kagi, Spectrochim. Acta, Part A, 2005, 61, 2423–2427 CrossRef PubMed.
- O. C. Adekoya, G. J. Adekoya, E. R. Sadiku, Y. Hamam and S. S. Ray, Pharmaceutics, 2022, 14, 1972 CrossRef CAS PubMed.
- J. Meurer, T. Bätz, J. Hniopek, C. Bernt, S. Zechel, M. Schmitt, J. Popp, M. D. Hager and U. S. Schubert, J. Mater. Chem. A, 2022, 10, 25106–25117 RSC.
- S. Bode, L. Zedler, F. H. Schacher, B. Dietzek, M. Schmitt, J. Popp, M. D. Hager and U. S. Schubert, Adv. Mater., 2013, 25, 1634–1638 CrossRef CAS PubMed.
- C. Wei, Y. He, X. Shi and Z. Song, Coord. Chem. Rev., 2019, 385, 1–19 CrossRef CAS PubMed.
- U. S. Schubert and C. Eschbaumer, Angew. Chem., Int. Ed., 2002, 41, 2892–2926 CrossRef CAS PubMed.
- L. Leibler, M. Rubinstein and R. H. Colby, Macromolecules, 1991, 24, 4701–4707 CrossRef CAS.
- H. Chen, R. Shi and H. Ow, ACS Omega, 2019, 4, 20665–20671 CrossRef CAS PubMed.
- O. Gutten, I. Beššeová and L. Rulíšek, J. Phys. Chem. A, 2011, 115, 11394–11402 CrossRef CAS PubMed.
- O. Gutten and L. Rulíšek, Inorg. Chem., 2013, 52, 10347–10355 CrossRef CAS PubMed.
- D. M. Parragh, C. Scheuerlein, N. Martin, R. Piccin, F. Ravotti, G. Pezzullo, T. Koettig and D. Lellinger, Polymers, 2024, 16, 407 CrossRef CAS PubMed.
- T. Mezger, in Angewandte Rheologie: Mit Joe Flow auf der Rheologie-Straße, Anton Paar GmbH, Graz, 2017, pp. 9833–9841 Search PubMed.
- H. Zhang, D. Wang, N. Wu, C. Li, C. Zhu, N. Zhao and J. Xu, ACS Appl. Mater. Interfaces, 2020, 12, 9833–9841 CrossRef CAS PubMed.
- R. K. Bose, N. Hohlbein, S. J. Garcia, A. M. Schmidt and S. van der Zwaag, Phys. Chem. Chem. Phys., 2014, 17, 1697–1704 RSC.
- S. Chen and W. H. Binder, Acc. Chem. Res., 2016, 49, 1409–1420 CrossRef CAS PubMed.
- S. Ge, Y.-H. Tsao and C. M. Evans, Nat. Commun., 2023, 14, 7244 CrossRef CAS PubMed.
- J. Zhao, Z. Zhang, C. Wang and X. Yan, CCS Chem., 2023, 6, 41–56 CrossRef.
- V. Zhang, B. Kang, J. V. Accardo and J. A. Kalow, J. Am. Chem. Soc., 2022, 144, 22358–22377 CrossRef CAS PubMed.
- C. Taplan, M. Guerre and F. E. Du Prez, J. Am. Chem. Soc., 2021, 143, 9140–9150 CrossRef CAS PubMed.
- X. Kuang, G. Liu, X. Dong and D. Wang, Mater. Chem. Front., 2016, 1, 111–118 RSC.
- S. Xu, D. Sheng, Y. Zhou, H. Wu, H. Xie, X. Tian, Y. Sun, X. Liu and Y. Yang, New J. Chem., 2020, 44, 7395–7400 RSC.
- Y. Vidavsky, M. R. Buche, Z. M. Sparrow, X. Zhang, S. J. Yang, R. A. DiStasio Jr. and M. N. Silberstein, Macromolecules, 2020, 53, 2021–2030 CrossRef CAS.
- R. D. Sudduth, J. Appl. Polym. Sci., 2001, 82, 527–540 CrossRef CAS.
- D. Berne, S. Laviéville, E. Leclerc, S. Caillol, V. Ladmiral and C. Bakkali-Hassani, ACS Polym. Au, 2025, 5, 214–240 CrossRef CAS.
- Q. Peng, Z. Zhu, C. Jiang and H. Jiang, Adv. Ind. Eng. Polym. Res., 2019, 2, 61–68 Search PubMed.
- W. Denissen, J. M. Winne and F. E. D. Prez, Chem. Sci., 2015, 7, 30–38 RSC.
- F. Meng, M. O. Saed and E. M. Terentjev, Macromolecules, 2019, 52, 7423–7429 CrossRef CAS.
- L. E. Porath and C. M. Evans, Macromolecules, 2021, 54, 4782–4791 CrossRef CAS.
- N. Rabiei, S. H. Amirshahi and M. Haghighat Kish, Phys. Rev. E, 2019, 99, 032502 CrossRef CAS.
- N. Nishiie, R. Kawatani, S. Tezuka, M. Mizuma, M. Hayashi and Y. Kohsaka, Nat. Commun., 2024, 15, 8657 CrossRef CAS PubMed.
- M. Capelot, M. M. Unterlass, F. Tournilhac and L. Leibler, ACS Macro Lett., 2012, 1, 789–792 CrossRef CAS PubMed.
- M. Guerre, C. Taplan, J. M. Winne and F. E. Du Prez, Chem. Sci., 2020, 11, 4855–4870 RSC.
- M. L. Martins, X. Zhao, Z. Demchuk, J. Luo, G. P. Carden, G. Toleutay and A. P. Sokolov, Macromolecules, 2023, 56, 8688–8696 CrossRef CAS.
- J. Dealy and D. Plazek, Rheol. Bull., 2009, 78, 16–31 Search PubMed.
- R. G. Ricarte and S. Shanbhag, Polym. Chem., 2024, 15, 815–846 RSC.
- L.-I. Palade, V. Verney and P. Attané, Rheol. Acta, 1996, 35, 265–273 CrossRef CAS.
- L. I. Palade, V. Verney and P. Attane, Macromolecules, 1995, 28, 7051–7057 CrossRef CAS.
- F. Zhuge, J. Brassinne, C.-A. Fustin, E. van Ruymbeke and J.-F. Gohy, Macromolecules, 2017, 50, 5165–5175 CrossRef CAS.
- R. J. Hickey, T. M. Gillard, M. T. Irwin, T. P. Lodge and F. S. Bates, Soft Matter, 2016, 12, 53–66 RSC.
- R. J. Sheridan and C. N. Bowman, Macromolecules, 2012, 45, 7634–7641 CrossRef CAS.
- R. K. Bose, M. Enke, A. M. Grande, S. Zechel, F. H. Schacher, M. D. Hager, S. J. Garcia, U. S. Schubert and S. van der Zwaag, Eur. Polym. J., 2017, 93, 417–427 CrossRef CAS.
- R. Kampes, J. Meurer, J. Hniopek, C. Bernt, S. Zechel, M. Schmitt, J. Popp, M. D. Hager and U. S. Schubert, Front. Soft Matter, 2022, 2, 14 Search PubMed.
- J.-E. Potaufeux, R. Tavernier, J. Odent and J.-M. Raquez, Macromolecules, 2024, 57, 7884–7892 CAS.
- H. Fang, X. Gao, F. Zhang, W. Zhou, G. Qi, K. Song, S. Cheng, Y. Ding and H. H. Winter, Macromolecules, 2022, 55, 10900–10911 CrossRef CAS.
- C. D. Han and M. S. Jhon, J. Appl. Polym. Sci., 1986, 32, 3809–3840 CrossRef.
- D. G. Fesko and N. W. Tschoegl, J. Polym. Sci., Part C, 1971, 35, 51–69 CrossRef.
- J. Ahmed, R. Auras, T. Kijchavengkul and S. K. Varshney, J. Food Eng., 2012, 111, 580–589 CrossRef CAS.
- C. Smithson, M. Stamenović, M. Nujkić and S. Putić, Zast. Mater., 2014, 55, 395–400 Search PubMed.
- P. A. O'Connell and G. B. McKenna, J. Chem. Phys., 1999, 110, 11054–11060 CrossRef.
- J. Rault, J. Non-Cryst. Solids, 2000, 271, 177–217 CrossRef CAS.
- M. L. Williams, R. F. Landel and J. D. Ferry, J. Am. Chem. Soc., 1955, 77, 3701–3707 CrossRef CAS.
- V. Montano, S. J. Picken, S. Van Der Zwaag and S. J. Garcia, Phys. Chem. Chem. Phys., 2019, 21, 10171–10184 RSC.
- J. Honerkamp and J. Weese, Rheol. Acta, 1993, 32, 65–73 CrossRef CAS.
- A. Stankiewicz, Materials, 2024, 17, 1527 CrossRef CAS PubMed.
- J. Song, N. Holten-Andersen and G. H. McKinley, Soft Matter, 2023, 19, 7885–7906 RSC.
- L. Xi, R. Luo, Q. Ma, C. Tu and Y. I. Shah, Constr. Build. Mater., 2022, 354, 129182 CrossRef.
- I. Emri and N. W. Tschoegl, Rheol. Acta, 1993, 32, 311–322 CrossRef CAS.
- M. Baumgaertel and H. H. Winter, Rheol. Acta, 1989, 28, 511–519 CrossRef CAS.
- C. S. Y. Tan, G. Agmon, J. Liu, D. Hoogland, E.-R. Janeček, E. A. Appel and O. A. Scherman, Polym. Chem., 2017, 8, 5336–5343 RSC.
- P. Edera, S. Chappuis, M. Cloitre and F. Tournilhac, Polymer, 2024, 299, 126916 CrossRef CAS.
- S. Ge, Y.-H. Tsao and C. M. Evans, Nat. Commun., 2023, 14, 7244 CrossRef CAS PubMed.
- V. Montano, M. Senardi, S. van der Zwaag and S. J. Garcia, Phys. Chem. Chem. Phys., 2020, 22, 21750–21760 RSC.
- T. Bätz, M. F. Agyemang, J. Meurer, J. Hniopek, S. Zechel, M. Schmitt, J. Popp, M. D. Hager and U. S. Schubert, Polym. Chem., 2025, 16, 492–502 RSC.
- M. Presselt, B. Dietzek, M. Schmitt, J. Popp, A. Winter, M. Chiper, C. Friebe and U. S. Schubert, J. Phys. Chem. C, 2008, 112, 18651–18660 CrossRef CAS.
- I. Šloufová, B. Vlčková, P. Mojzeš, I. Matulková, I. Císařová, M. Procházka and J. Vohlídal, J. Phys. Chem. C, 2018, 122, 6066–6077 CrossRef.
- I. Šloufová, B. Vlčková, M. Procházka, J. Svoboda and J. Vohlídal, J. Raman Spectrosc., 2014, 45, 338–348 CrossRef.
- J. Kožíšek, J. Svoboda, J. Zedník, B. Vlčková and I. Šloufová, J. Phys. Chem. B, 2021, 125, 12847–12858 CrossRef.
- G. Socrates, in Infrared and Raman characteristic group frequencies: tables and charts, Wiley, Chichester Weinheim, 3rd edn, 2001 Search PubMed.
- C. Apip, A. Martínez, M. Meléndrez, M. Domínguez, T. Marzialetti, R. Báez, G. Sánchez-Sanhueza, A. Jaramillo and A. Catalán, Saudi Dent. J., 2021, 33, 944–953 CrossRef PubMed.
- M. Rosemal, H. Haris, K. Sathasivam and S. Mohan, Pharm. Chem., 2010, 2, 316–323 Search PubMed.
- A. Eisenberg, Macromolecules, 1970, 3, 147–154 CrossRef CAS.
- F. Herbst, K. Schröter, I. Gunkel, S. Gröger, T. Thurn-Albrecht, J. Balbach and W. H. Binder, Macromolecules, 2010, 43, 10006–10016 CrossRef CAS.
| This journal is © The Royal Society of Chemistry 2026 |