Open Access Article
Open Access ArticleFluoroolefin-vinyl ether copolymer ionic fluorogels for PFAS remediation from water
Irene M.
Harmody
 a,
Haley P.
Macdonald
a,
Haley P.
Macdonald
 b,
Orlando
Coronell
b,
Orlando
Coronell
 b and
Frank A.
Leibfarth
b and
Frank A.
Leibfarth
 *a
*a
aDepartment of Chemistry, The University of North Carolina at Chapel Hill, Chapel Hill, North Carolina 27599, USA. E-mail: FrankL@email.unc.edu
bDepartment of Environmental Sciences & Engineering, The University of North Carolina at Chapel Hill, Chapel Hill, North Carolina 27599, USA
First published on 26th February 2026
Abstract
Per- and polyfluoroalkyl substances (PFAS) are a class of toxic chemicals that are ubiquitous in the environment and have contaminated water sources globally. As regulations limiting PFAS concentrations in drinking water are being established by regulatory agencies globally, there is an urgent demand for robust granular sorbents that remove PFAS from water selectively and are easily regenerated. Here we report a new class of Ionic Fluorogels (IF) that leverage fluoroolefin-vinyl ether (FVE) perfectly alternating copolymers as a polymer matrix for the development of ion exchange resins. The use of 2-chloroethyl vinyl ether as a comonomer provided a partially fluorinated polymer with electrophilic functionality, which was reacted with multivalent amines to install covalent crosslinks and cationic character in a single synthetic step. Systematic variation in the identity of fluoroolefin comonomer and multivalent amine resulted in a library of materials for structure–property evaluation. We found that the synergistic combination of a fluorophilic matrix and cationic charge leads to high PFAS sorption, and that the ratio of quaternary ammonium to tertiary amine is an important design criteria. Performance evaluation showed that the FVE-based IFs have high binding capacity for GenX, up to 770 milligrams GenX per gram IF, facile regeneration with no lapse in performance across five cycles, and higher PFAS selectivity than commercial ion exchange resin in both simulated and natural waters.
Introduction
Per- and polyfluorinated alkyl substances (PFAS) are a class of fluorinated synthetic chemicals1 which are used commercially in fluoropolymer manufacturing, waterproof coatings, food packaging, and aqueous fire-fighting foams (AFFF), among others.2 PFAS have been associated with numerous claims of endocrine, reproductive, immunotoxic, and other health effects.3,4 In part due to their toxicity, “legacy” PFAS, such as perfluorooctanoic acid (PFOA) and perfluorooctane sulfonic acid (PFOS), were phased out by 20065,6 and replaced with “emerging” PFAS including perfluoroalkyl ether acids (PFECAs).7,8 Although understanding of the toxicity and bioaccumulation of emerging PFECAs like GenX is still nascent, a growing body of evidence indicates that PFECAs are concerns for human and animal health.9In the United States, the U.S. Environmental Protection Agency (EPA) has investigated PFAS and determined that they are harmful to human health. In 2024, the U.S. EPA established legally enforceable Maximum Contaminant Limits (MCLs) in drinking water for six PFAS: PFOA, PFOS, PFHxS, PFNA, GenX, and PFBS. The limits are 4 parts per trillion (ppt) for PFOA and PFOS, 10 ppt for PFHxS, PFNA, and GenX, and a hazard index of less than one for a mixture containing two or more of PFHxS, PFNA, GenX, and PFBS.10 While subsequently the U.S. EPA announced its intention to rescind the limits and reconsider the regulatory determinations for PFHxS, PFNA, GenX, and PFBS, the limits for PFOA and PFOS remain in place.11 Public water utilities have until 2031 to implement PFAS remediation strategies and comply with the MCL. An estimated 7000 water utilities in the U.S. will have to install new water remediation systems or modify existing systems to meet the proposed MCLs, and the annual cost is estimated to be upwards of $2.7 billion.12
Implementation of efficient, cost-effective PFAS remediation technologies is critical. Reverse Osmosis (RO) and nanofiltration efficiently remove PFAS from water, however, these processes are difficult to implement on large scales, as they create large amounts of PFAS-laden residuals, are energy intensive, and may not be cost-effective for impacted communities.13 Granular sorbents, such as granular activated carbon (GAC) and ion exchange resins (IX), have shown promise in pilot studies; however, they often demonstrate poor performance for short-chain PFAS such as GenX and other PFECAs.14,15 In one practical example, the Sweeney Water Treatment Plant in the impacted community of Wilmington, NC, installed a $43 million system that removes PFECAs to low-ppt levels using 3 million pounds of GAC which is replaced every 270 days.16 The annual operating costs for this water treatment system are estimated at approximately $5 million.17
The expense of PFAS remediation by traditional granular sorbents and the impending scope of remediation motivates the development of materials that are selective for fluorinated chemicals. An effective PFAS sorbent would have high PFAS selectivity over natural organic matter and other co-constituents in water, resulting in higher-capacity sorbent beds, long operational lifetimes, and less material usage. Regenerability constitutes an additional attractive feature of an effective PFAS remediation sorbent that yields a longer usage life cycle and a concentrated PFAS waste that can be destroyed using emerging destruction technologies.18 However, development of practical removal technologies relies on fundamental understanding of PFAS sorption processes. Taking inspiration from GAC, many materials leverage hydrophobic interactions to selectively partition PFAS from solution. Furthermore, molecular recognition19–23 and supramolecular host–guest complexes24–26 implemented via cationic crosslinked resins27–30 and porous polymer sorbents31–36 have demonstrated preliminary promise to remove PFAS from water. These materials often lack selectivity for a wide range of PFAS at environmentally relevant concentrations such as those in natural waters that serve as drinking water sources.37
To address this lack of selectivity in natural waters, we were inspired by the use of fluorophilicity to selectively partition PFAS from aqueous solution.29,38–44 Previously, we reported Ionic Fluorogels (IFs), a selective class of PFAS remediation sorbents which leverage perfluoropolyethers (PFPEs) as fluorophilic matrices for PFAS removal. In a flow-through packed bed geometry, the IFs efficiently removed PFAS from natural waters spiked with part-per-trillion level PFAS, supporting our hypothesis that a synergistic combination of fluorous and electrostatic components enables high-performance PFAS remediation from natural waters (Fig. 1A).39 However, PFPEs present some possible environmental hazards,45 are expensive on scale, and suffer from hydrolytic degradation without chemical modification. Installation of aryl-ether linkages as PFPE end groups limited hydrolytic degradation (Fig. 1B),46 but concerns regarding costs and potential environmental hazard remain. Given the need for a scalable and chemically-stable PFAS remediation technology, we hypothesized that a complementary class of partially fluorinated copolymers, fluoroolefin-vinyl ether copolymers (FVEs), could serve as low-cost, hydrolytically stable, high-performance fluorophilic matrices for PFAS removal (Fig. 1C).
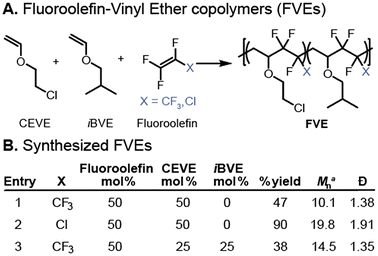
FVEs are tunable, partially fluorinated polymers synthesized through the perfectly alternating radical copolymerization of a fluoroolefin monomer and one or more vinyl ether monomers (Fig. 2A).47–49 The alternating character arises due to the difference in radical polarity50,51 between the fluoroolefin (e.g. tetrafluoroethylene, hexafluoropropylene, chlorotrifluoroethylene) and the vinyl ether (e.g. isobutyl vinyl ether, chloroethyl vinyl ether) during propagation.48,52,53 Vinyl ethers are readily available, electron-rich monomers that do not homopolymerize through radical polymerization.54,55 Fluoroolefins are electron-deficient monomers that are polymerized at an industrial scale using radical polymerization. The matching of radical polarity leads to a lower kinetic barrier (ΔG‡) toward crosspropogation, which results in an alternating polymer microstructure.53,56–71 Incorporation of a functional comonomer such as chloroethylvinyl ether (CEVE) enables post-polymerization modification to form graft copolymers62,72 or ionomers,49,61 further diversifying the structural manifold provided by the FVE copolymer platform.
We leveraged functional CEVE to provide a platform for the generation of a library of polymer networks to evaluate for PFAS remediation (Fig. 2B). Nucleophilic substitution of the alkyl chloride with tertiary amines results in formation of quaternary ammonium groups, installing both a covalent crosslink and permanent cationic character in a single synthetic step. We hypothesized that this new class of IFs would be efficient PFAS sorbents, as they would leverage both a fluorinated matrix and electrostatic interactions to remove PFAS from water.
Herein, we report FVE copolymer-based Ionic Fluorogels (FVE-IFs) as a library of granular resins for PFAS remediation. These modular materials demonstrate improved sorption of PFAS from simulated natural water relative to commercial ion exchange resins, high PFAS capacity, and facile regeneration. Competitive sorption experiments indicate that FVE-IFs show superior selectivity for PFAS over organic matter compared to a state-of-the-art ion exchange resin. The tunability and performance of FVE-IFs make them a promising platform for translationally relevant PFAS remediation technology.
Results and discussion
A significant advantage of the FVE-IF materials platform is the tunability of the FVE copolymer structure to access varied degrees of ionic content and fluorophilicty. To understand the effect of fluoroolefin identity and functional comonomer density, we first synthesized a representative suite of FVE copolymers by radical copolymerization (Fig. 2A). Hexafluoropropylene (HFP) and chlorotrifluoroethylene (CTFE) were identified as fluoroolefin monomers to modulate fluorophilicity by changing the backbone fluorination of the polymer. CEVE was chosen as the functional comonomer through which post-polymerization nucleophilic substitution would occur.49 Two FVEs with 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 feed ratios of fluoroolefin and CEVE were synthesized with different fluoroolefins to yield poly(HFP-alt-CEVE) and poly(CTFE-alt-CEVE) (Fig. 2B, entries 1 and 2). To investigate the role of functional comonomer loading, a terpolymer with a 2
1 feed ratios of fluoroolefin and CEVE were synthesized with different fluoroolefins to yield poly(HFP-alt-CEVE) and poly(CTFE-alt-CEVE) (Fig. 2B, entries 1 and 2). To investigate the role of functional comonomer loading, a terpolymer with a 2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1
1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 incorporation of HFP, CEVE, and isobutylvinyl ether (iBVE) (poly[(HFP-alt-CEVE)-co-(HFP-alt-iBVE)]) was also synthesized (Fig. 2B, entry 3). At a multi-gram scale, all three copolymers were isolated in yields consistent with literature precedent49 and at appropriate number average molar masses (Mn) for subsequent crosslinking reactions (Mn = 10–20 kg mol−1).
1 incorporation of HFP, CEVE, and isobutylvinyl ether (iBVE) (poly[(HFP-alt-CEVE)-co-(HFP-alt-iBVE)]) was also synthesized (Fig. 2B, entry 3). At a multi-gram scale, all three copolymers were isolated in yields consistent with literature precedent49 and at appropriate number average molar masses (Mn) for subsequent crosslinking reactions (Mn = 10–20 kg mol−1).
The resulting FVE copolymers were crosslinked via substitution with multi-functional nucleophiles (Fig. 3A and Table 1), leveraging the CEVE comonomer as a functional handle. The parent FVE copolymers are soluble in common organic solvents, therefore the reaction proceeded readily in acetone following an adapted literature procedure (see SI for a detailed procedure).49 Upon network formation, the reaction solution formed an insoluble gel. After reaction completion, the material was ground into a powder and purified via Soxhlet extraction in ethanol, followed by drying in vacuo at 50 °C. The products were obtained in quantitative yield. The FVE-IFs were characterized via scanning electron microscopy (SEM), thermogravimetric analysis (TGA), and differential scanning calorimetry (DSC) (Fig. S2–S4). SEM revealed the expected morphology of granular resins; the particles were irregularly shaped with significant crevices and other surface features but no observable long-range order or porosity in the micrometer range (Fig. 3B).
 | ||
| Fig. 3 Synthesis of FVE-IFs. (A) Synthesis of FVE-IFs via nucleophilic substitution crosslinking. (B) SEM images of ground FVE-IF-2. | ||
| Ref. | FVE | Multiamine | mol% FTEG | |
|---|---|---|---|---|
| Identity | mol% | |||
| FVE-IF-1 | Poly(HFP-alt-CEVE) | TMEDA | 10 | 0 |
| FVE-IF-2 | Poly(HFP-alt-CEVE) | TMEDA | 40 | 0 |
| FVE-IF-3 | CTFE-alt-CEVE | TMEDA | 40 | 0 |
| FVE-IF-4 | Poly(HFP-alt-CEVE) | TMEDA | 40 | 20 |
| FVE-IF-5 | Poly(CTFE-alt-CEVE) | TMEDA | 40 | 20 |
| FVE-IF-6 | Poly[(HFP-alt-CEVE)-co-(HFP-alt-iBVE)] | TMEDA | 40 | 0 |
| FVE-IF-7 | Poly(HFP-alt-CEVE) | TMEDA | 94 | 0 |
| FVE-IF-8 | Poly(HFP-alt-CEVE) | PMDTA | 37 | 0 |
| FVE-IF-9 | Poly(HFP-alt-CEVE) | HMTETA | 74 | 0 |
| FVE-IF-10 | Poly(HFP-alt-CEVE) | Me6TREN | 20 | 0 |
| FVE-IF-11 | Poly(HFP-alt-CEVE) | Me6TREN | 40 | 0 |
The library of FVE-IFs synthesized is shown in Table 1. The nucleophilic crosslinker loading is described by mole percent (mol%) incorporation relative to fluoroolefin repeat unit. Poly(HFP-alt-CEVE) was used as the primary FVE for FVE-IF synthesis due to safety concerns regarding transport and storage of CTFE.73 However, representative experiments using poly(CTFE-alt-CEVE) to form FVE-IF-3 and FVE-IF-5 were successful, demonstrating the ability to synthesize FVE-IFs with diverse fluoroolefin monomers. Using N,N,N′,N′-tetramethylethylenediamine (TMEDA) as a model nucleophile resulted in quantitative conversion of the soluble polymer to a crosslinked network, even when only 10 mol% of TMEDA was used (FVE-IF-1), or when CEVE incorporation was decreased (FVE-IF-6). Because a tertiary amine-based crosslinker was used, the nucleophilic substitution resulted in quaternary ammonium ion formation, such that network formation and permanent charge installation are achieved in one step. This represents an advance over existing technologies, which typically require separate reactions for network formation and quaternary ammonium group formation.
To investigate the effect of multiamine molecular weight and architecture, 1,1,4,7,7,-pentamethyldiethylenetriamine (PMDTA), 1,1,4,7,10,10-hexamethyltriethylenetetramine (HMTETA), and tris[2-(dimethylamino)ethyl]amine (Me6TREN) were each used as multiamines for FVE-IF synthesis (FVE-IF-8, FVE-IF-9, FVE-IF-10, FVE-IF-11). Incorporation of fluorinated tetraethylene glycol (FTEG) as a secondary nucleophile also resulted in network formation (FVE-IF-4, FVE-IF-5). FTEG incorporation introduced an additional fluorous component to the FVE-IFs, which we hypothesized would further improve PFAS sorption.
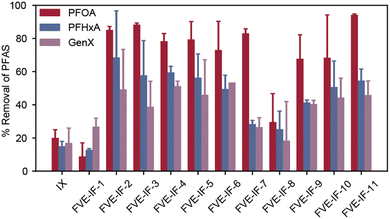
The resulting FVE-IFs were first subjected to PFAS remediation batch experiments under equilibrium conditions in simulated natural water. The parameters of this experiment are described in the SI and our previous work.39 Briefly, simulated natural water was prepared by dissolving 200 mg L−1 NaCl and 20 mg L−1 humic acid in deionized water. The salt content mimicked the salt content of natural water; however, the humic acid loading was one to two orders of magnitude higher than what would be expected in a practical remediation scenario, making this a challenging experiment for non-selective sorbents. Next, the sorbent (10 mg L−1) was suspended in the solution, then stirred 3 hours to hydrate. Upon hydration, the particles swelled marginally but retained the shape and structural integrity. For example, FVE-IF-2 swelled ∼50 wt% compared to its dry weight, which indicates that it is a loosely crosslinked polymer network (Table S2 and Fig. S5). Attempts to analyze their surface area through nitrogen adsorption were not successful, presumably because FVE-IFs behave like “gel” ion exchange resins with no permanent porosity. Finally, the solution was spiked with three representative PFAS at 1 μg L−1 each, resulting in a 20![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 times excess of humic acid relative to each PFAS. After 21 hours of stirring, the solution was analyzed via Liquid Chromatography-Mass Spectrometry (LC-MS) to determine how much of each PFAS was removed by the sorbent. Control experiments without sorbent or with a commercial ion exchange resin were also performed. The figure of merit for this experiment is percent removal of each PFAS relative to a control with no sorbent present, where higher percent removals correlate to better sorbent performance (Fig. 4).
000 times excess of humic acid relative to each PFAS. After 21 hours of stirring, the solution was analyzed via Liquid Chromatography-Mass Spectrometry (LC-MS) to determine how much of each PFAS was removed by the sorbent. Control experiments without sorbent or with a commercial ion exchange resin were also performed. The figure of merit for this experiment is percent removal of each PFAS relative to a control with no sorbent present, where higher percent removals correlate to better sorbent performance (Fig. 4).
Batch equilibrium sorption results for the FVE-IFs revealed informative structure–property–performance relationships (Fig. 4). FVE-IF-1, with 10 mol% incorporation of TMEDA relative to repeat unit (equating to a maximum of 20 mol% incorporation of quaternary ammonium groups given TMEDA contains two tertiary amines), demonstrated poor PFAS removal. However, FVE-IF-2, with 40 mol% incorporation of TMEDA, demonstrated over 85% PFOA removal, 68% PFHxA removal, and 49% GenX removal. FVE-IF-3, synthesized using poly(CTFE-co-CEVE), demonstrated similar performance to FVE-IF-2, synthesized using poly(HFP-alt-CEVE), indicating there may be little to no effect on performance when replacing the HFP trifluoromethyl moiety with chloride. FVE-IF-4 and FVE-IF-5 performed similarly to FVE-IF-2 and FVE-IF-3 despite incorporation of FTEG. We reason that this similarity across four materials of the library indicates that small differences in fluorophilicity do not affect overall PFAS sorption under these challenging water conditions. FVE-IF-6 also performed similarly to FVE-IFs 2–5, indicating that the incorporation of alkyl vinyl ethers does not reduce PFAS sorption. These initial results are encouraging, especially when compared to the high-performing commercial resin PFA694E (labeled IX in Fig. 4).
The use of FVEs as a platform enabled the incorporation of both quaternary ammonium and tertiary amine functionlity into a single sorbent. In one approach, the incorporation of iBVE lowered the density of electrophilic sites (i.e. CEVE repeat units), which results in a lower number of reactive sites for nucleophilic substitution by tertiary amines (FVE-IF-6). The use of an FVE with 50 mol% CEVE relative to HFP repeat unit and the use of 40 mol% TMEDA results in an FVE-IF with 63% quaternary ammonium and 37% tertiary amine, assuming quantitative conversion of chloroalkyl group to ammonium. While FVE-IF-6 performed similarly to resins with a high proportion of permanent positive change, further reductions in quaternary ammonium density to 53% had adverse effects on short chain PFAS removal (FVE-IF-7). We infer from the contrasting results between FVE-IF-6 and FVE-IF-7 that there is some ratio of strong base to weak base ion exchange sites below which short-chain PFAS sorption is reduced. However, since PFOA sorption by FVE-IF-6 and FVE-IF-7 were similar, we hypothesize that long-chain PFAS sorption is driven by fluorophilicity, which increases with increasing perfluoroalkyl chain length.74–76 These trends are consistent with previous investigations with PFPE IFs.39
Investigating other multiamine crosslinkers elucidated further structure–property–performance relationship trends. FVE-IF-8, which was crosslinked with 37 mol% PMDTA, demonstrated poor PFAS removal relative to its analog FVE-IF-2 crosslinked with 40 mol% TMEDA. PMDTA has an additional tertiary amine, which results in a higher crosslink density and higher density of quaternary ammonium groups. Finally, Me6TREN, a tripodal multiamine crosslinker, was used in FVE-IF-10 and FVE-IF-11. These two FVE-IFs had attractive sorption performance similar to to FVE-IFs 2–5. FVE-IF-10, with 20 mol% Me6TREN incorporation, 67% quaternary ammonium groups and 33% tertiary amines (presumingly only the sterically accessible distal amines react), demonstrates efficient sorption of both long- and short-chain PFAS. FVE-IF-11, which has 63% quaternary ammoniums and 37% tertiary amines, also demonstrated efficient sorption. To understand how fluorine content influences adsorption, Table S2 correlates wt% fluorine in the FVE-IFs to equilibrium sorption. Taken together, these initial batch equilibrium sorption results demonstrate that both TMEDA and Me6TREN serve as effective amines for high performance FVE-IF sorbents for PFAS removal, and that the ratio of quaternary ammonium to tertiary amine is an important design criteria to consider.
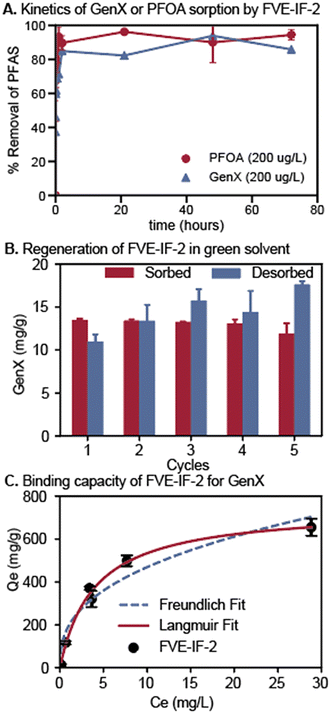
FVE-IF performance was investigated more thoroughly using FVE-IF-2 as a model sorbent and GenX as a model PFAS. First, the kinetics of PFOA and GenX sorption by FVE-IF-2 were investigated independently in pure water. At high concentration (200 μg L−1 PFAS, 10 mg L−1 sorbent), up to 93% GenX removal and 94% PFOA removal were observed (Fig. 5A). At low concentrations (1 μg L−1), PFAS sorption was near-quantitative after 21 hours (Fig. S6), with 95% PFOA and 97% GenX removal observed. No significant PFAS desorption was observed throughout the course of this experiment. Although the kinetics of PFPE IF PFAS sorption are faster,39 the performance of FVE-IF-2 is promising as a proof-of-concept.
The ability to regenerate and reuse PFAS remediation sorbents is crucial for both fundamental insights, as well as practical performance. Fundamentally, observing the binding affinity of PFAS to a sorbent under different conditions elucidates structure–property–performance relationships. Practically, sorbent regeneration via an environmentally conscious solvent mixture enables desorption of PFAS into a concentrated solution which may be further analyzed or destroyed. Additionally, regeneration of the sorbent extends its lifetime, significantly decreasing technology ownership cost and improving overall performance. Therefore, we conducted repetitive sorption and desorption experiments using FVE-IF-2 and GenX to probe the regenerability of the FVE-IFs. FVE-IF-2 was suspended in a solution of GenX in nanopure water (target dosing was 10 mg GenX per gram of FVE-IF-2), stirred 1 hour, and centrifuged. The supernatant was analyzed via LC-MS to determine how much GenX was sorbed. The FVE-IF-2 pellet was then suspended in a solution of 400 mM ammonium acetate in 50% aqueous ethanol, stirred 1 hour, and centrifuged. The supernatant was then analyzed to determine the extent of GenX desorption. The desorption brine solution was identified as a green alternative to harsher methanol-based regenerants used previously.39 This process was repeated over five cycles in triplicate (Fig. 5B). The results show that FVE-IF-2 is regenerable over at least five cycles with no decrease in sorbent performance.
The GenX binding capacity of FVE-IF-2 was measured by subjecting FVE-IF-2 to batch equilibrium sorption experiments in pure water spiked with a range of GenX concentrations ([GenX]0 = 0.2–50 mg L−1). Binding capacity, typically measured in milligrams of analyte per gram of sorbent (mg g−1), is a valuable metric for PFAS sorbents that indicates the maximum amount of analyte which can be sorbed to the resin at equilibrium. The amount of GenX sorbed to FVE-IF-2 at equilibrium was plotted against the concentration of GenX remaining in solution for each experiment, then the data were fit to both the Langmuir and Freundlich isotherms (Fig. 5C). The calculated capacity for FVE-IF-2 was 770 ± 50 mg g−1 according to the Langmuir isotherm (r2 = 0.99) (Table S2). This capacity is over 2.75 times higher than the calculated binding capacity for PFPE IFs (280 mg g−1).39
Despite only removing 50% of GenX in the batch sorption experiment in simulated natural water, FVE-IF-2 demonstrates outstanding GenX binding capacity in nanopure water. We hypothesize this disparity stems from some non-selective sorption of non-fluorinated organic matter by the FVE-IF. However, relative to commercial anion exchange resins, we hypothesized that the fluorinated matrix of the FVE-IF would result in improved selectivity for PFAS. To probe this difference in selectivity, we designed a proof-of-concept experiment which leveraged 1H and 19F NMR to measure fluorinated and non-fluorinated organic matter binding. Solutions of about 3 mM of PFOA, GenX, octanoic acid (OA), hexanoic acid (HxA), PFOA and OA, and GenX and HxA were prepared. To 1 mL of each solution, 20 mg of either FVE-IF-2 or Purolite PFA694E was added. The resulting suspensions were stirred 1 hour, then transferred to 1.5 mL tubes and centrifuged. Next, 450 μL supernatant was spiked with 40 μL D2O and 10 μL trifluoroethanol (as an internal standard), then the percent removal of each analyte was determined via19F NMR (PFOA, GenX) and 1H NMR (OA, HxA) relative to control reactions without sorbent. The results are summarized in Table S7 and Fig. 6. In experiments with only one analyte, FVE-IF-2 demonstrated near-quantitative removal of PFOA and GenX, as measured by 19F NMR, and no removal of OA or HxA, demonstrating significant selectivity for fluorinated analytes. Meanwhile, the IX removed all PFOA and GenX, as well as 43% of OA and 36% of HxA. In a mixed solution of PFOA and OA, FVE-IF-2 removed all detectable PFOA and 31% of OA, while IX removed 100% of both PFOA and OA. Likewise, in a mixed solution of GenX and HxA, FVE-IF-2 removed 98% of GenX and 31% of HxA, while IX removed 100% of GenX and 46% of HxA. Therefore, in a mixed solution of fluorinated and non-fluorinated organic matter, both FVE-IF-2 and IX are more selective for fluorinated molecules. However, FVE-IF-2 demonstrated significantly higher PFAS selectivity than IX, as evidenced by the results with mixed PFAS and non-fluorinated organic matter, in particular the PFOA and OA solution, and as supported by the superior FVE-IF-2 performance in the batch tests with simulated natural water depicted in Fig. 4. Coupled with the high GenX binding capacity, the selectivity of FVE-IFs for PFAS makes them a promising class of materials for translational application to water remediation systems.
Conclusion
FVE copolymers are an underexplored class of functional materials. Here, we demonstrated a new class of Ionic Fluorogels that synergistically combine an FVE copolymer fluorinated matrix with ion exchange via cationic quaternary ammonium groups for selective PFAS remediation from simulated natural water. The resulting polymer networks are high-performing sorbents with excellent binding capacity for GenX as well as facile regenerability in a green solvent. Finally, when compared to commercial ion exchange resins under identical conditions, FVE-IFs outperform IX under both simulated natural conditions, and in high-concentration NMR studies. These FVE-IFs represent a significant advance in the development of translationally relevant PFAS sorbents, as the principles of Ionic Fluorogels have been demonstrated to be general across multiple classes of fluorinated polymers.Conflicts of interest
Two international patent applications have been filed by the University of North Carolina at Chapel Hill on technology related to ionic fluorogels, including PCT/US2020/047365 and PCT/US2022/013249. F. A. L., O. C. and I. M. H. have a financial interest in Sorbenta Inc.Data availability
The data that supports the findings of this study, including chemical and materials characterization, methods, and procedures, can be found in the supplementary information (SI). See DOI: https://doi.org/10.1039/d5py00795j.Acknowledgements
This work was supported by the Strategic Environmental Research and Development Program of the U.S. Department of Defense (ER20-1211, F. A. L.), the NC Collaboratory (F. A. L. and O. C.), the National Institute of Environmental Health Sciences (P42ES031007, O. C.), and the U.S. National Science Foundation Graduate Research Fellowship Program (DGE-2040435, H. P. M.). The UNC Department of Chemistry's NMR Core Laboratory provided expertise and instrumentation that enabled this study with support from the U.S. National Science Foundation (CHE-1828183 and CHE-0922858).References
- Perfluoroalkyl and Polyfluoroalkyl Substances (PFAS), National Institute of Environmental Health Sciences, https://www.niehs.nih.gov/health/topics/agents/pfc (accessed 2025-07-26).
- M. G. Evich, M. J. B. Davis, J. P. McCord, B. Acrey, J. A. Awkerman, D. R. U. Knappe, A. B. Lindstrom, T. F. Speth, C. Tebes-Stevens, M. J. Strynar, Z. Wang, E. J. Weber, W. M. Henderson and J. W. Washington, Per- and Polyfluoroalkyl Substances in the Environment, Science, 2022, 375(6580) DOI:10.1126/science.abg9065.
- J. C. Dewitt, M. M. Peden-Adams, J. M. Keller and D. R. Germolec, Immunotoxicity of Perfluorinated Compounds: Recent Developments, Toxicol. Pathol., 2012, 40(2), 300–311, DOI:10.1177/0192623311428473.
- S. E. Fenton, A. Ducatman, A. Boobis, J. C. DeWitt, C. Lau, C. Ng, J. S. Smith and S. M. Roberts, Per- and Polyfluoroalkyl Substance Toxicity and Human Health Review: Current State of Knowledge and Strategies for Informing Future Research, Environ. Toxicol. Chem., 2021, 606–630, DOI:10.1002/etc.4890.
- US EPA's Federal Facilities Restoration and Reuse Office (FFRRO), Technical Fact Sheet – Perfluorooctane Sulfonate (PFOS) and Perfluorooctanoic Acid (PFOA), 2017. https://19january2021snapshot.epa.gov/sites/static/files/2017-12/documents/ffrrofactsheet_contaminants_pfos_pfoa_11-20-17_508_0.pdf (accessed 2025-01-28) Search PubMed.
- U.S. Environmental Protection Agency Designation of Perfluorooctanoic acid (PFOA) and perfluorooctanesulfonic acid (PFOS) as CERCLA hazardous substances (Final Rule, 89 FR 39124), Federal Register. U.S. Environmental Protection Agency. (2024, May 8). https://www.federalregister.gov/d/2024-08547 Search PubMed.
- M. Strynar, S. Dagnino, R. McMahen, S. Liang, A. Lindstrom, E. Andersen, L. McMillan, M. Thurman, I. Ferrer and C. Ball, Identification of Novel Perfluoroalkyl Ether Carboxylic Acids (PFECAs) and Sulfonic Acids (PFESAs) in Natural Waters Using Accurate Mass Time-of-Flight Mass Spectrometry (TOFMS, Environ. Sci. Technol., 2015, 49(19), 11622–11630, DOI:10.1021/acs.est.5b01215.
- F. Heydebreck, J. Tang, Z. Xie and R. Ebinghaus, Alternative and Legacy Perfluoroalkyl Substances: Differences between European and Chinese River/Estuary Systems, Environ. Sci. Technol., 2015, 49(14), 8386–8395 CrossRef CAS PubMed.
- T. Woodlief, S. Vance, Q. Hu and J. DeWitt, Immunotoxicity of Per- and Polyfluoroalkyl Substances: Insights into Short-Chain PFAS Exposure, Toxics, 2021, 9(5), 100, DOI:10.3390/toxics9050100.
- United States Environmental Protection Agency. Per- and Polyfluoroalkyl Substances (PFAS) Final PFAS National Primary Drinking Water Regulation, United States Environmental Protection Agency (accessed 2025-07-26) Search PubMed.
- EPA Press Office, EPA Announces It Will Keep Maximum Contaminant Levels for PFOA, PFOS, 2025. https://www.epa.gov/newsreleases/epa-announces-it-will-keep-maximum-contaminant-levels-pfoa-pfos Search PubMed.
- Black & Veatch and Corona Environmental Consulting, Estimating the National Cost to Remove PFAS from Drinking Water Using UCMR 5 Data, 2024. https://www.awwa.org/wp-content/uploads/Final-Technical-Memorandum-Updating-National-Cost-Estimate-for-PFAS-Standards-using-UCMR-5.pdf Search PubMed.
- A. B. Schantz, B. Xiong, E. Dees, D. R. Moore, X. Yang and M. Kumar, Emerging Investigators Series: Prospects and Challenges for High-Pressure Reverse Osmosis in Minimizing Concentrated Waste Streams, Environ. Sci., 2018, 4(7), 894–908, 10.1039/c8ew00137e.
- R. Medina, M. W. Pannu, S. A. Grieco, M. Hwang, C. Pham and M. H. Plumlee, Pilot-Scale Comparison of Granular Activated Carbons, Ion Exchange, and Alternative Adsorbents for per- and Polyfluoroalkyl Substances Removal, AWWA Water Sci., 2022, 4(5) DOI:10.1002/aws2.1308.
- C. Kassar, C. Graham and T. H. Boyer, Removal of Perfluoroalkyl Acids and Common Drinking Water Contaminants by Weak-Base Anion Exchange Resins: Impacts of Solution PH and Resin Properties, Water Res., 2022, 17, 100159, DOI:10.1016/j.wroa.2022.100159.
- C. Mojica, NCDEQ and EPA Representatives Tour Sweeney Plant Treatment Plant near Wilmington, WHQR Public Media, 2022 Search PubMed.
- Z. C. F. P. U. A. Solon, No, PFAS Found in Water Treated by GAC Filters, WECT News 6, Wilmington, 2022.
- M. J. Krause, E. Thoma, E. Sahle-Damesessie, B. Crone, A. Whitehill, E. Shields and B. Gullet, Supercritical Water Oxidation as an Innovative Technology for PFAS Destruction, J. Environ. Eng., 2022, 148(2), 5021006, DOI:10.1061/(ASCE)EE.1943-7870.0001957.
- R. B. Clark and J. E. Dick, Electrochemical Sensing of Perfluorooctanesulfonate (PFOS) Using Ambient Oxygen in River Water, ACS Sens., 2020, 5(11) DOI:10.1021/acssensors.0c01894.
- F. Cao, L. Wang, X. Ren and H. Sun, Synthesis of a Perfluorooctanoic Acid Molecularly Imprinted Polymer for the Selective Removal of Perfluorooctanoic Acid in an Aqueous Environment, J. Appl. Polym. Sci., 2016, 133(15), 1–10, DOI:10.1002/app.43192.
- A. H. Karoyo and L. D. Wilson, Tunable Macromolecular-Based Materials for the Adsorption of Perfluorooctanoic and Octanoic Acid Anions, J. Colloid Interface Sci., 2013, 402, 196–203, DOI:10.1016/j.jcis.2013.03.065.
- L. Du, Y. Wu, X. Zhang, F. Zhang, X. Chen, Z. Cheng, F. Wu and K. Tan, Preparation of Magnetic Molecularly Imprinted Polymers for the Rapid and Selective Separation and Enrichment of Perfluorooctane Sulfonate, J. Sep. Sci., 2017, 40(13), 2819–2826, DOI:10.1002/jssc.201700157.
- Q. Yu, S. Deng and G. Yu, Selective Removal of Perfluorooctane Sulfonate from Aqueous Solution Using Chitosan-Based Molecularly Imprinted Polymer Adsorbents, Water Res., 2008, 42(12), 3089–3097, DOI:10.1016/j.watres.2008.02.024.
- D. Shetty, I. Jahović, T. Skorjanc, T. S. Erkal, L. Ali, J. Raya, Z. Asfari, M. A. Olson, S. Kirmizialtin, A. O. Yazaydin and A. Trabolsi, Rapid and Efficient Removal of Perfluorooctanoic Acid from Water with Fluorine-Rich Calixarene-Based Porous Polymers, ACS Appl. Mater. Interfaces, 2020, 12(38), 43160–43166, DOI:10.1021/acsami.0c13400.
- W. Guo, B. M. Fung and S. D. Christian, NMR Study of Cyclodextrin Inclusion of Fluorocarbon Surfactants in Solution, Langmuir, 1992, 8(2), 446–451, DOI:10.1021/la00038a022.
- A. H. Karoyo, A. S. Borisov, L. D. Wilson and P. Hazendonk, Formation of Host-Guest Complexes of β-Cyclodextrin and Perfluorooctanoic Acid, J. Phys. Chem. B, 2011, 115(31), 9511–9527, DOI:10.1021/jp110806k.
- M. Ateia, M. F. Attia, A. Maroli, N. Tharayil, F. Alexis, D. C. Whitehead and T. Karanfil, Rapid Removal of Poly- and Perfluorinated Alkyl Substances by Poly(Ethylenimine)-Functionalized Cellulose Microcrystals at Environmentally Relevant Conditions, Environ. Sci. Technol. Lett., 2018, 5(12), 764–769, DOI:10.1021/acs.estlett.8b00556.
- Y. Koda, T. Terashima and M. Sawamoto, Fluorinated Microgels in Star Polymers: From In-Core Dynamics to Fluorous Encapsulation, Macromolecules, 2015, 48, 2901–2908, DOI:10.1021/acs.macromol.5b00166.
- Y. Koda, T. Terashima and M. Sawamoto, Fluorous Microgel Star Polymers: Selective Recognition and Separation of Polyfluorinated Surfactants and Compounds in Water, J. Am. Chem. Soc., 2014, 136(44), 15742–15748, DOI:10.1021/ja508818j.
- T. Terashima and M. Sawamoto, Microgel-Core Star Polymers as Functional Compartments for Catalysis and Molecular Recognition, in Progress in Controlled Radical Polymerization: Materials and Applications, ACS Symposium Series, American Chemical Society, 2012, vol. 1101, pp. 5–65. DOI:10.1021/bk-2012-1101.ch005.
- A. Alsbaiee, B. J. Smith, L. Xiao, Y. Ling, D. E. Helbling and W. R. Dichtel, Rapid Removal of Organic Micropollutants from Water by a Porous β-Cyclodextrin Polymer, Nature, 2016, 529(7585), 190–194, DOI:10.1038/nature16185.
- L. Xiao, Y. Ling, A. Alsbaiee, C. Li, D. E. Helbling and W. R. Dichtel, β-Cyclodextrin Polymer Network Sequesters Perfluorooctanoic Acid at Environmentally Relevant Concentrations, J. Am. Chem. Soc., 2017, 139(23), 7689–7692, DOI:10.1021/jacs.7b02381.
- W. Ji, L. Xiao, Y. Ling, C. Ching, M. Matsumoto, R. P. Bisbey, D. E. Helbling and W. R. Dichtel, Removal of GenX and Perfluorinated Alkyl Substances from Water by Amine-Functionalized Covalent Organic Frameworks, J. Am. Chem. Soc., 2018, 140(40), 12677–12681, DOI:10.1021/jacs.8b06958.
- L. Xiao, C. Ching, Y. Ling, M. Nasiri, M. J. Klemes, T. M. Reineke, D. E. Helbling and W. R. Dichtel, Cross-Linker Chemistry Determines the Uptake Potential of Perfluorinated Alkyl Substances by β-Cyclodextrin Polymers, Macromolecules, 2019, 52, 3747–3752, DOI:10.1021/acs.macromol.9b00417.
- A. M. J. Klemes, Y. Ling, C. Ching, V. Wu, E. Helbling and W. R. Dichtel, Reduction of a Tetrafluoroterephthalonitrile-β-Cyclodextrin Polymer to Remove Anionic Micropollutants and Perfluorinated Alkyl Substances from Water, Angew. Chem., Int. Ed., 2019, 58, 12049–12053, DOI:10.1002/anie.201905142.
- C. Wu, M. J. Klemes, B. Trang, W. R. Dichtel and D. E. Helbling, Exploring the Factors That Influence the Adsorption of Anionic PFAS on Conventional and Emerging Adsorbents in Aquatic Matrices, Water Res., 2020, 182, 115950, DOI:10.1016/j.watres.2020.115950.
- United States Environmental Protection Agency, Fifth Unregulated Contaminant Monitoring Rule Data Finder, United States Environmental Protection Agency, https://www.epa.gov/dwucmr/fifth-unregulated-contaminant-25 (accessed 2025-07-26) Search PubMed.
- Y. Koda, T. Terashima, A. Nomura, M. Ouchi and M. Sawamoto, Fluorinated Microgel-Core Star Polymers as Fluorous Compartments for Molecular Recognition, 2011, pp. 4574–4578. DOI:10.1021/ma201076y.
- E. Kumarasamy, I. M. Manning, L. B. Collins, O. Coronell and F. A. Leibfarth, Ionic Fluorogels for Remediation of Per- and Polyfluorinated Alkyl Substances from Water, ACS Cent. Sci., 2020, 6(4), 487–792, DOI:10.1021/acscentsci.9b01224.
- I. M. Manning, N. G. P. Chew, H. P. Macdonald, K. E. Miller, M. J. Strynar, O. Coronell and F. Leibfarth, Hydrolytically Stable Ionic Fluorogels for High-Performance Remediation of Per- and Polyfluoroalkyl Substances (PFAS) from Natural Water, Angew. Chem., Int. Ed., 2022, 61(41), e202208150, DOI:10.1002/anie.202208150.
- Y. Koda, T. Terashima and M. Sawamoto, Fluorinated Microgel Star Polymers as Fluorous Nanocapsules for the Encapsulation and Release of Perfluorinated Compounds, Polym. Chem., 2015, 6(31), 5663–5674, 10.1039/c5py00113g.
- X. Tan, J. Zhong, C. Fu, H. Dang, Y. Han, P. Král, J. Guo, Z. Yuan, H. Peng, C. Zhang and A. K. Whittaker, Amphiphilic Perfluoropolyether Copolymers for the Effective Removal of Polyfluoroalkyl Substances from Aqueous Environments, Macromolecules, 2021, 54(7), 3447–3457, DOI:10.1021/acs.macromol.1c00096.
- X. Tan, M. Sawczyk, Y. Chang, Y. Wang, A. Usman, C. Fu, P. Král, H. Peng, C. Zhang and A. K. Whittaker, Revealing the Molecular-Level Interactions between Cationic Fluorinated Polymer Sorbents and the Major PFAS Pollutant PFOA, Macromolecules, 2022, 55(3), 1077–1087, DOI:10.1021/acs.macromol.1c02435.
- Q. Quan, H. Wen, S. Han, Z. Wang, Z. Shao and M. Chen, Fluorous-Core Nanoparticle-Embedded Hydrogel Synthesized via Tandem Photo-Controlled Radical Polymerization: Facilitating the Separation of Perfluorinated Alkyl Substances from Water, ACS Appl. Mater. Interfaces, 2020, 12(21), 24319–24327, DOI:10.1021/acsami.0c04646.
- J. McCord, M. Strynar, J. Washington, M. Davis and E. G. S. Bergman, Emerging Chlorinated Polyfluorinated Ether Compounds Impacting the Waters of New Jersey Identified by Use of Nontargeted Analysis, 2020. DOI:10.1021/acs.estlett.0c00640.
- I. M. Manning, N. G. P. Chew, H. P. Macdonald, K. E. Miller, M. J. Strynar, O. Coronell and F. Leibfarth, Hydrolytically Stable Ionic Fluorogels for High-Performance Remediation of Per- and Polyfluoroalkyl Substances, 2022. DOI:10.1002/anie.202208150.
- Y. Tabata and T. A. D. U. Plessis, Radiation-Induced-Copolymerization Chlorotrifluoroethylene with Ethyl Vinyl Ether, J. Polym. Sci., Part A: Polym. Chem., 1972, 9, 3425–3435 CrossRef.
- C. Walling and F. R. Mayo, A Note on the “Alternating Effect” in Copolymerization, J. Polym. Sci., 1948, 3(6), 895–897, DOI:10.1002/pol.1948.120030610.
- D. Valade, F. Boschet and B. Améduri, Synthesis and Modification of Alternating Copolymers Based on Vinyl Ethers, Chlorotrifluoroethylene, and Hexafluoropropylene, Macromolecules, 2009, 42(20), 7689–7700, DOI:10.1021/ma900860u.
- J. J. A. Garwood, A. D. Chen and D. A. Nagib, Radical Polarity, J. Am. Chem. Soc., 2024, 146(41), 28034–28059, DOI:10.1021/jacs.4c06774.
- F. Parsaee, M. C. Senarathna, P. B. Kannangara, S. N. Alexander, P. D. E. Arche and E. R. Welin, Radical Philicity and Its Role in Selective Organic Transformations, Nat. Rev. Chem., 2021, 5(7), 486–499, DOI:10.1038/s41570-021-00284-3.
- B. Boutevin, F. Cersosimo and B. Youssef, Studies of the Alternating Copolymerization of Vinyl Ethers with Chlorotrifluoroethylene, Macromolecules, 1992, 25(11), 2842–2846, DOI:10.1021/ma00037a009.
- M. Gaboyard, Y. Hervaud and B. Boutevin, Photoinitiated Alternating Copolymerization of Vinyl Ethers with Chlorotrifluoroethylene, Polym. Int., 2002, 51(7), 577–584, DOI:10.1002/pi.902.
- D. Braun, H. Elsässer and F. Hu, Free Radical Terpolymerization of Three Non-Homopolymerizable Monomers. Part III, Eur. Polym. J., 2001, 37(9), 1779–1784, DOI:10.1016/S0014-3057(01)00050-7.
- W. G. S. Reyntjens and E. J. Goethals, New Materials from Poly (Vinyl Ethers), Polym. Adv. Technol., 2001, 12(1–2), 107–122 CrossRef CAS.
- T. Takakura, CTFE/Vinyl Ether Copolymers, in Modern Fluoropolymers, Wiley Interscience, New York, 1997, pp. 557–568 Search PubMed.
- A. Asakawa, M. Unoki, T. Hirono and T. Takayanagi, Waterborne Fluoropolymers for Paint Use, J. Fluorine Chem., 2000, 104(1), 47–51, DOI:10.1016/S0022-1139(00)00226-8.
- R. Hamiye, A. Alaaeddine, M. Awada, B. Campagne, S. Caillol, S. M. Guillaume, B. Ameduri and J.-F. Carpentier, From Glycidyl Carbonate to Hydroxyurethane Side-Groups in Alternating Fluorinated Copolymers, Polym. Chem., 2014, 5(17), 5089–5099, 10.1039/C4PY00547C.
- A. Alaaeddine, F. Boschet, B. Ameduri and B. Boutevin, Synthesis and Characterization of Original Alternated Fluorinated Copolymers Bearing Glycidyl Carbonate Side Groups, J. Polym. Sci., Part A: Polym. Chem., 2012, 50(16), 3303–3312, DOI:10.1002/pola.26112.
- G. Couture, B. Campagne, A. Alaaeddine and B. Améduri, Synthesis and Characterizations of Alternating Co- and Terpolymers Based on Vinyl Ethers and Chlorotrifluoroethylene, Polym. Chem., 2013, 4(6), 1960–1968, 10.1039/C2PY20977B.
- G. Couture, V. Ladmiral and B. Améduri, Methods to Prepare Quaternary Ammonium Groups-Containing Alternating Poly(Chlorotrifluoroethylene-Alt-Vinyl Ether) Copolymers, RSC Adv., 2015, 5(14), 10243–10253, 10.1039/C4RA09238D.
- D. Valade, F. Boschet and B. Ameduri, Grafting Polymerization of Styrene onto Alternating Terpolymers Based on Chlorotrifluoroethylene, Hexafluoropropylene, and Vinyl Ethers, and Their Modification into Ionomers Bearing Ammonium Side-Groups, J. Polym. Sci., Part A: Polym. Chem., 2010, 48(24), 5801–5811, DOI:10.1002/pola.24388.
- K. S. Santhosh Kumar and B. Ameduri, Synthesis and Characterization of Epoxy Functionalized Cooligomers Based on Chlorotrifluoroethylene and Allyl Glycidyl Ether, J. Polym. Sci., Part A: Polym. Chem., 2010, 48(16), 3587–3595, DOI:10.1002/pola.24138.
- M.-L. Tran-Do, J.-P. Habas and B. Ameduri, Oxygen-Tolerant Alternating Copolymerization of Fluorinated Monomers and Vinyl Ethers at Mild Temperature, ACS Appl. Polym. Mater., 2022, 4(2), 1401–1410, DOI:10.1021/acsapm.1c01780.
- P. Wang, J. Dai, L. Liu, Q. Dong, B. Jin and R. Bai, Xanthate-Mediated Living/Controlled Radical Copolymerization of Hexafluoropropylene and Butyl Vinyl Ether under 60Co γ-Ray Irradiation and Preparation of Fluorinated Polymers End-Capped with a Fluoroalkyl Sulfonic Acid Group, Polym. Chem., 2013, 4(6), 1760–1764, 10.1039/C3PY21158D.
- L. Liu, D. Lu, H. Wang, Q. Dong, P. Wang and R. Bai, Living/Controlled Free Radical Copolymerization of Chlorotrifluoroethene and Butyl Vinyl Ether under 60Co γ-Ray Irradiation in the Presence of S-Benzyl O-Ethyl Dithiocarbonate, Chem. Commun., 2011, 47(27), 7839–7841, 10.1039/C1CC11749A.
- J. Lee, G. Lopez, B. Améduri and M. Seo, Synthesis of Heterograft Copolymers with a Semifluorinated Backbone by Combination of Grafting-through and Grafting-from Polymerizations, Macromolecules, 2020, 53(8), 2811–2821, DOI:10.1021/acs.macromol.9b02493.
- F. Boschet and B. Ameduri, Copolymers of Chlorotrifluoroethylene: Synthesis, Properties, and Applications, Chem. Rev., 2014, 114(2), 927–980, DOI:10.1021/cr2002933.
- R. Tayouo, G. David, B. Améduri, J. Rozière and S. Roualdès, New Fluorinated Polymers Bearing Pendant Phosphonic Acid Groups. Proton Conducting Membranes for Fuel Cell, Macromolecules, 2010, 43(12), 5269–5276, DOI:10.1021/ma100703k.
- G. Puts, G. Lopez, T. Ono, P. Crouse and B. Ameduri, Radical Copolymerisation of Chlorotrifluoroethylene with Isobutyl Vinyl Ether Initiated by the Persistent Perfluoro-3-Ethyl-2,4-Dimethyl-3-Pentyl Radical, RSC Adv., 2015, 5(52), 41544–41554, 10.1039/C5RA05066A.
- A. Alaaeddine, F. Boschet and B. Ameduri, Synthesis of Methallylic Monomers Bearing Ammonium Side-Groups and Their Radical Copolymerization with Chlorotrifluoroethylene, J. Polym. Sci., Part A: Polym. Chem., 2014, 52(12), 1721–1729, DOI:10.1002/pola.27173.
- D. J. Siegwart, J. K. Oh and K. Matyjaszewski, ATRP in the Design of Functional Materials for Biomedical Applications, Prog. Polym. Sci., 2012, 37(1), 18–37, DOI:10.1016/j.progpolymsci.2011.08.001.
- Safety Data Sheet, Chlorotrifluoroethylene, Saint Louis, MO, 2018.
- I. T. Horváth, Fluorous Biphase Chemistry, Acc. Chem. Res., 1998, 31(10), 641–650, DOI:10.1021/ar970342i.
- L. E. Kiss, I. Kövesdi and J. Rábai, An Improved Design of Fluorophilic Molecules: Prediction of the Ln P Fluorous Partition Coefficient, Fluorophilicity, Using 3D QSAR Descriptors and Neural Networks, J. Fluorine Chem., 2001, 108(1), 95–109, DOI:10.1016/S0022-1139(01)00342-6.
- S. M. Daniels, R. A. Saunders and J. A. Platts, Prediction of Fluorophilicity of Organic and Transition Metal Compounds Using Molecular Surface Areas, J. Fluorine Chem., 2004, 125, 1291–1298, DOI:10.1016/j.jfluchem.2004.03.014.
| This journal is © The Royal Society of Chemistry 2026 |