Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Synthesis of network polymers composed of well-defined silyl ether macrocycles
Chihiro
Nakai†
a,
Sota
Amano†
a,
Koh
Sugamata
 bc,
Naoki
Watanabe
bc,
Naoki
Watanabe
 d,
Hiroaki
Imoto
d,
Hiroaki
Imoto
 a,
Kensuke
Naka
a,
Kensuke
Naka
 a and
Takahiro
Iwamoto
a and
Takahiro
Iwamoto
 *a
*a
aFaculty of Molecular Chemistry and Engineering, Kyoto Institute of Technology, Goshokaido-cho, Matsugasaki, Sakyo-ku, Kyoto 606-8585, Japan. E-mail: tiwamoto@kit.ac.jp
bDepartment of Chemistry, Institute of Pure and Applied Sciences, University of Tsukuba, 1-1-1 Tennodai, Tsukuba, Ibaraki 305-8571, Japan
cTsukuba Research Center for Energy Materials Sciences (TREMS), University of Tsukuba, 1-1-1 Tennodai, Tsukuba, Ibaraki 305-8571, Japan
dJNC Petrochemical Corporation, 5-1, Goikaigan, Ichihara, Chiba 290-8551, Japan
First published on 2nd March 2026
Abstract
We report a straightforward synthetic route to silyl ether-based network polymers, enabled by efficient macrocyclization of diols with dichlorosilacyclobutane (DCSB) followed by thermal ring-opening of the silacyclobutane units. This strategy allows well-defined macrocyclic motifs composed of silyl ether linkages to be embedded directly into the polymer backbone. The resulting films are optically transparent and exhibit excellent thermal stability. Mechanical characterization reveals relatively low hardness, reflecting the intrinsic flexibility of the silyl ether framework. Despite the hydrolytic sensitivity of the macrocyclic precursors, the resulting network films demonstrate high acid resistance, which is attributed to their hydrophobic surface properties.
Silyl ether-based polymers (SEPs) have gained considerable interest due to their fascinating properties distinct with conventional hydrocarbon materials, such as excellent thermal stability, flexibility, and optical transparency.1 Consequently, SEPs have found various applications including elastomers, thermosets, and other advanced polymer materials.2–4 Furthermore, although the Si–O bond is generally prone to hydrolysis and alcoholysis, such a labile nature of the Si–O bonds has been utilized for the development of degradable polymer and vitrimer with dynamic covalent networks.5–13 In addition to these advantageous features, the development of robust synthetic methodologies has greatly advanced SEP chemistry. Traditionally, SEPs have been synthesized by polycondensation of diols with dichlorosilanes, diaminosilanes, or dialkoxysilanes.14,15 More recently, hydrosilylation of bis(carbonyl) comonomers has emerged as a powerful alternative route.16 In addition, new strategies have been continuously established to expand the structural diversity and tunability of SEPs.
Dichlorosilacyclobutane is a commercially available silyl building block that possesses a distinctive dual and orthogonal reactivity. The Si–Cl groups readily undergo nucleophilic substitution to generate silyl ether linkages, whereas the silacyclobutane ring can undergo ring-opening reactions under thermal, photochemical, and catalytic conditions without compromising the integrity of the newly formed Si–O bonds.17–22 Combination of these independent reactivity modes provides a promising platform for the construction of unprecedented polymer architectures. However, silyl ethers derived from silacyclobutanes are generally highly susceptible to hydrolysis, necessitating handling under inert atmosphere. Consequently, to take advantage of the dual reactivity of dichlorosilacyclobutane for the synthesis of SEPs, there is a strong need for a highly efficient synthetic method without any complicated experimental operation like column purification.
Previously, we reported an efficient macrocyclization that provides silyl ether-based macrocycles from readily accessible diols and dichlorodimethylsilanes.23 In general, macrocyclization reactions suffer from intrinsic challenges, including the requirement for extremely dilute conditions (typically in the millimolar range) and inherently low macrocyclization efficiencies.24–26 In contrast, our method achieves remarkably high efficiency and selectivity, affording macrocycles featuring silyl ether units at the vertices. Building upon this macrocyclization strategy, we now present a facile and efficient synthetic approach for producing novel silyl ether-based polymer networks composed of well-defined macrocyclic units. The method involves macrocyclization of diols with dichlorosilacyclobutane through Si–O bond formation, followed by thermal ring-opening of the silacyclobutane moieties to produce a densely crosslinked network polymer in which discrete macrocyclic structures are preserved (Fig. 1). The preparation of polymer films and their bulk properties are also described.
 | ||
| Fig. 1 The synthetic strategy for silyl ether-based network polymer consisting of well-defined macrocyclic structures. | ||
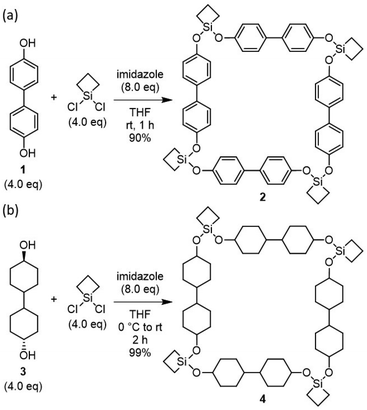
Our study began with syntheses of macrocyclic compounds using 4,4′-dihydroxybiphenyl (1) and [1,1′-bi(cyclohexane)]-4,4′-diol (3) as a linker (Scheme 1). Following the previously reported method, dichlorosilacyclobutane was treated with dihydroxybiphenyl in the presence of imidazole. The reaction afforded the desired product along with small amount of byproduct. After washing the crude product by acetonitrile, an analytically pure macrocycle was obtained in 90% yield. 1H, 13C, and 29Si NMR spectroscopic analyses and mass spectrometry supported the formation of macrocycle 2 with exclusive selectivity. A rapid and efficient synthesis was also achieved when used with diol 3. In this case, the starting diol was hardly dissolved in THF, while the reaction efficiently proceeded to provide soluble macrocyclic compound 4 almost quantitatively. The structure was further examined by single-crystal X-ray diffraction. Although the crystal quality was insufficient for full structural refinement (see SI), the analysis indicated that macrocycle 4 consists of four silacyclobutane units and four bicyclohexylene linkers, adopting an approximately square-shaped geometry in the solid state. This structural motif closely resembles that of the macrocycles reported in our previous work, whereas the conformations of the silyl ether units show slight variations that reflect their inherent flexibility.
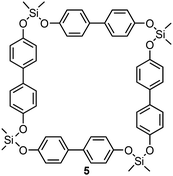
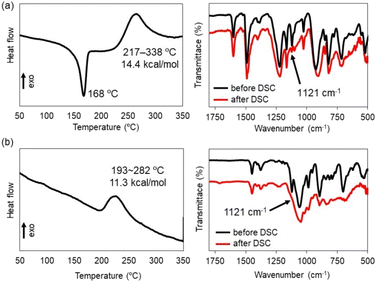
Polymerization via the ring-opening reaction of the silacyclobutane units was first assessed by thermogravimetric analyses (TGA) and differential scanning calorimetry (DSC). As shown in Fig. 2a, macrocycle 2 exhibited melting point at 168 °C and subsequently an exothermic peak ranging from 217 °C to 338 °C. The exothermic peak was no longer observed upon reheating the sample. After heating over the observed exothermic peak, the sample became insoluble in common organic solvents. Fourier transform infrared (FTIR) spectroscopy indicated that, upon heating, a characteristic peak at 1121 cm−1 assigned to the silacyclobutane ring significantly decreased.17 These observations implied that a polymeric structure was formed via ring-opening reaction of the silacyclobutane units. We have already reported that macrocycle 5 bearing Me2Si units instead of silacyclobutane unit at the vertices exhibited high thermal stability within 300 °C (Fig. 3). Furthermore, the peak of the reference compound 5 at around the characteristic peak of the silacyclobutan unit in the FT-IR spectrum remained essentially unchanged (see the SI). On the basis of these results, we concluded that the macrocyclic skeleton is maintained during the ring-opening reaction of the silacyclobutane unit. Note that other peaks particularly derived from linker units hardly changed upon the ring-opening reaction. This result also supported retention of the macrocyclic skeleton during the ring-opening reaction. Macrocycle 4 showed a similar thermal response (Fig. 2b). Although the exothermic enthalpy derived from the ring-opening reaction was slightly lower than that of macrocycle 2, FT-IR analysis of the obtained sample indicated that efficient consumption of the silacyclobutane units. Therefore, we concluded that a polymer network is formed also in a reaction of macrocycle 4.
 | ||
| Fig. 2 DSC analyses and FT-IR spectra of macrocycle 2 (a) and 4 (b). FT-IR analyses were performed by using the samples used in TGA analyses. | ||
Next, we investigated the effect of heating conditions on network formation (Fig. 4). Macrocycle 2 bearing biphenylene linkers was used for this study. When the sample was continuously heated at 10 °C min−1 up to 350 °C (condition A, red line in Fig. 4a), the resulting material was visually transparent. Strong birefringence was observed under polarized optical microscopy (POM) (Fig. 4b). Although birefringence suggests the presence of molecular orientation, no diffraction peaks were detected in the XRD analysis, indicating that the degree of orientation was limited and did not lead to long-range order. In contrast, when the sample was held isothermally at its melting point for 15 minutes (condition B, black line), the birefringence became significantly weaker. These observations suggest that rapid heating promotes network formation while partially retaining the molecular orientation inherited from the crystalline initial material, whereas isothermal annealing at the melting point relaxes these orientations prior to network formation.
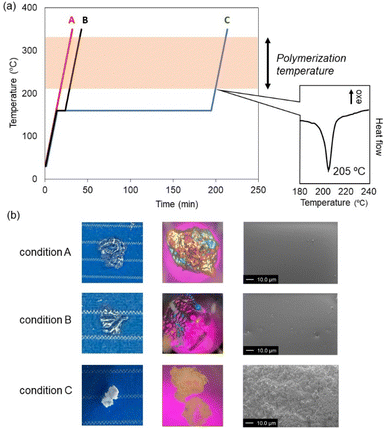 | ||
| Fig. 4 (a) Examination of heating conditions of the ring-opening reactions of macrocycle 2. (b) Appearance, POM, and SEM images of the samples obtained by DSC under conditions A–C. | ||
We next examined holding the sample at the melting point for 3 hours before the thermal polymerization (condition C, blue line). In this case, a distinct behavior was observed: a new endothermic peak appeared at 205 °C, different from the ring-opening process. After the subsequent heating for the thermal polymerization, exothermic peak derived from the ring-opening was observed. The cured material appeared turbid, and SEM imaging revealed the presence of particles with diameters of approximately 1.7 µm. Notably, such particles were absent in the materials obtained under conditions A and B. We have previously shown that a similar type of macrocyclic molecule can exhibit cold crystallization.27 Thus, we assumed that the newly observed endothermic peak should be attributed to the melting of crystals formed by cold crystallization during the holding for 3 h at 160 °C. Overall, these results demonstrate that the heating protocol strongly influences not only the orientation of the macrocyclic units but also the bulk morphology of the resulting polymer network.
With these results in mind, we next prepared thin films via drop-casting a solution of macrocycle 2 onto a glass substrate, followed by heating at 255 °C for 1 h (film A). The treatment temperature of 255 °C was chosen because it lies within the exothermic region associated with the ring-opening of the silacyclobutane units, allowing efficient network formation under melt conditions. The resulting film became insoluble in common organic solvent. Additionally, FT-IR spectrum was identical to that obtained after DSC analysis (see the SI), confirming successful polymerization. SEM images revealed smooth surfaces, and powder X-ray diffraction showed no distinct diffraction peaks, indicating that the films were amorphous. The transparent appearance and the negligible birefringence observed under POM suggested that the obtained films closely resembled those produced under condition B in Fig. 4a. The same procedure was applied to the preparation of thin film of macrocycle 4 (film B). The obtained film was insoluble and exhibited IR spectra consistent with those of the sample obtained by DSC. The films were also optically transparent, transmitting more than 97% of visible light. In both cases, the high optical transmittance is consistent with the absence of crystal aggregation, as supported by the XRD and SEM analyses (Fig. 5).
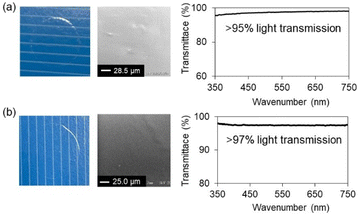 | ||
| Fig. 5 Appearance, SEM images, and UV-vis absorption spectra of the films obtained by thermal treatment of macrocycle 2 (a) and 4 (b). | ||
The thermal stability of the resulting films was evaluated by TGA under a nitrogen atmosphere. For both films A and B, no 5% weight loss (Td5) was observed below 517 and 481 °C, respectively, demonstrating their high thermal resistance (Fig. 6a). In contrast, the macrocyclic monomer 5 decomposed at approximately 350 °C (Td5 = 367 °C). Therefore, the excellent heat resistance of the films originates not only from the inherent stability of the silyl ether units but also from the formation of a high cross-linking network structure.
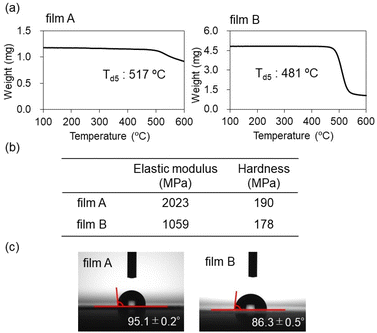 | ||
| Fig. 6 (a) TGA thermograms, (b) elastic modulus and instrumented indentation hardness, and (c) water contact angles of film A and B. | ||
The mechanical properties of the films were assessed by nanoindentation using a Shimadzu DUH-211 device equipped with a Triangular115 indenter (Fig. 6b). Despite the presence of a highly crosslinked network, the films exhibited relatively low hardness (190 MPa) and a modest elastic modulus (2023 MPa). This mechanically soft behavior is attributed to the flexibility of the silyl ether units within the network. A similar mechanical profile was observed for film B. Interestingly, films derived from macrocycle 4 displayed even lower mechanical rigidity, highlighting the impact of the linker structure on the mechanical properties of the resulting networks.
Although macrocycles 2, 4, and 5 are highly susceptible to hydrolysis in their isolated state, the corresponding films exhibited markedly enhanced resistance toward hydrolysis even under acidic conditions. After immersion in ethanol containing 1 M HCl for 24 h, the films showed a 34% weight loss (see SI for details). A 1H NMR spectrum of the soluble fraction obtained after acidic treatment indicated the film was decomposed to the corresponding diol species (see the SI). While the extent of decomposition was not negligible, this result clearly indicates a substantial increase in hydrolytic stability, as macrocycles 2 and 4 readily decompose under ambient conditions. A similar trend was observed for film B. In both films, the network structure remains largely preserved under neutral conditions. This enhanced durability is likely attributable to the dense cross-linked network and/or the hydrophobic surface, which may limit the accessibility of water and acid to the hydrolyzable silyl ether bonds. To assess the surface properties of the films, static water contact angle measurements were conducted (Fig. 6c). The measured contact angle for film A was 95.1 ± 0.2°, indicating an intrinsically hydrophobic surface. Film B also exhibited hydrophilic surface with water contact angle of 86.3 ± 0.5°. Interestingly, despite its more hydrophobic linker structure, film B displayed a lower contact angle than film A. This apparent discrepancy may arise from microscopic differences in surface topography. Gas sorption measurements showed negligible N2 and CO2 uptake, indicating the absence of permanent microporosity in the network structure (see SI). This structures might also inhibit access of water to the hydrolyzable silyl ether bonds.
Conclusions
In summary, we have demonstrated a facile synthetic route to silyl ether-based network polymers through efficient macrocyclization of diols with dichlorosilacyclobutane, followed by thermal ring-opening of the silacyclobutane units. This strategy enabled the direct incorporation of well-defined macrocyclic motifs into a polymer backbone, a structural feature rarely explored in conventional silyl ether polymer chemistry. The resulting crosslinked networks exhibited excellent thermal stability and optical transparency, together with remarkable resistance to hydrolytic degradation despite the intrinsic sensitivity of the macrocyclic precursors. Mechanical tests further revealed that the inherent flexibility of the silyl ether linkages imparts relatively low hardness and modulus, while the linker structure significantly influences the final material properties. Incorporating well-defined macrocyclic motifs into a polymer skeleton could unlock unexplored opportunities to design a novel silyl ether-based polymer. Thus, investigation of other diol structures is currently underway.Conflicts of interest
There are no conflicts to declare.Data availability
The data supporting this article have been included as part of the supplementary information (SI). Supplementary information is available. See DOI: https://doi.org/10.1039/d6py00035e.Acknowledgements
This work was supported by the Izumi Science and Technology Foundation, Proterial Materials Science Foundation, Shorai Foundation for Science and Technology, and Ogasawara Foundation for the Promotion of Science and Engineering. Diol 3 was kindly gifted by JNC.References
- E. Yilgör and I. Yilgör, Prog. Polym. Sci., 2013, 39, 1165–1195 CrossRef.
- S. Gao, Y. Liu, S. Feng and Z. Lu, J. Mater. Chem. A, 2019, 7, 17498–17504 RSC.
- Y. Zhang, Z. Zhu, Z. Bai, W. Jiang, F. Liu and J. Tang, RSC Adv., 2017, 7, 16616–16622 RSC.
- X. Chen, L. Fang, X. Chen, J. Zhou, J. Wang, J. Sun and Q. Fang, ACS Sustainable Chem. Eng., 2018, 6, 13518–13523 CrossRef CAS.
- C. M. Bunton, Z. M. Bassampour, J. M. Boothby, A. N. Smith, J. V. Rose, D. M. Nguyen, T. H. Ware, K. G. Csaky, A. R. Lippert, N. V. Tsarevsky and D. Y. Son, Macromolecules, 2020, 53, 9890–9900 CrossRef CAS.
- C. Cheng, A. Watts, M. A. Hillmyer and J. F. Hartwig, Angew. Chem., Int. Ed., 2016, 55, 11872–11876 CrossRef CAS PubMed.
- S. Vijjamarri, M. Hull, E. Kolodka and G. Du, ChemSusChem, 2018, 11, 2881–2888 CrossRef CAS PubMed.
- H. Fouilloux, M.-N. Rager, P. Ríos, S. Conejero and C. M. Thomas, Angew. Chem., Int. Ed., 2022, 61, e202113443 CrossRef CAS.
- M. C. Parrott, J. C. Luft, J. D. Byrne, J. H. Fain, M. E. Napier and J. M. DeSimone, J. Am. Chem. Soc., 2010, 132, 17928–17932 CrossRef CAS PubMed.
- P. Zheng and T. J. McCarthy, J. Am. Chem. Soc., 2012, 134, 2024–2027 CrossRef CAS PubMed.
- W. Schmolke, N. Perner and S. Seiffert, Macromolecules, 2015, 48, 8781–8788 CrossRef CAS.
- T. Debsharma, V. Amfilochiou, A. A. Wróblewska, I. D. Baere, W. V. Paepegem and F. E. D. Prez, J. Am. Chem. Soc., 2022, 144, 12280–12289 CrossRef CAS PubMed.
- K. E. L. Husted, C. M. Brown, P. Shieh, I. Kevlishvili, S. L. Kristufek, H. Zafar, J. V. Accardo, J. C. Cooper, R. S. Klausen, H. J. Kulik, J. S. Moore, N. R. Sottos, J. A. Kalow and J. A. Johnson, J. Am. Chem. Soc., 2023, 145, 1916–1923 CrossRef CAS PubMed.
- R. MacFarlane and E. S. Yankura, Synthesis of Regulated Structure Polyphenylether Siloxane Block Copolymers, United States Rubber Co Naugatuck CT Chemical Div., 1963 Search PubMed.
- J. E. Curry and J. D. Byrd, J. Appl. Polym. Sci., 1965, 9, 295–311 CrossRef CAS.
- S. Luleburgaz, U. Tunca and H. Durmaz, Polym. Chem., 2023, 14, 2949–2957 RSC.
- W. Yuan, X. Wei, Q. Peng, L. Fan, X. Li, H. Hu, Y. Huang, J. Ma and J. Yang, J. Appl. Polym. Sci., 2021, 138, e51376 CrossRef.
- X. Wu, O. Grinevich and D. C. Neckers, Chem. Mater., 1999, 11, 3687–3692 CrossRef CAS.
- G. Raabe and J. Michl, Chem. Rev., 1985, 85, 419–509 CrossRef CAS.
- H. Yamashita, M. Tanaka and K. Honda, J. Am. Chem. Soc., 1995, 117, 8873–8874 CrossRef CAS.
- R. Jain, A. P. J. Brunskill, J. B. Sheridan and R. A. Lalancette, J. Organomet. Chem., 2005, 690, 2272–2277 CrossRef CAS.
- K. Matsumoto and H. Yamaoka, Macromolecules, 1995, 28, 7029–7031 CrossRef CAS.
- T. Iwamoto, S. Amano, K. Maeda, N. Shibama, W. Sekiguchi, Y. Kazama, Y. Nakamura, K. Sugamata, H. Imoto, K. Naka and Y. Ishii, Chem. Commun., 2025, 61, 8180–8183 RSC.
- F. Garnes-Portolés and A. Leyva-Pérez, ACS Catal., 2023, 13, 9415–9426 CrossRef.
- V. Martí-Centelles, M. D. Pandey, M. I. Burguete and S. V. Luis, Chem. Rev., 2015, 115, 8736–8834 CrossRef PubMed.
- J. Blankenstein and J. Zhu, Eur. J. Org. Chem., 2005, 1949–1964 CrossRef CAS.
- T. Iwamoto, S. Amano, K. Maeda, N. Shibama, W. Sekiguchi, A. Imaizumi, A. Honda, H.-C. Chang, H. Houjou, H. Imoto, K. Naka and Y. Ishii, J. Phys. Chem. B, 2026, 130, 1724–1729 CrossRef CAS PubMed.
Footnote |
| † Equal contribution of both authors. |
| This journal is © The Royal Society of Chemistry 2026 |