Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
3H-Phenothiazin-3-one: a photocatalyst for the mild oxidation of boronic acids under green LED irradiation
Constantinos T.
Constantinou
 a,
Petros L.
Gkizis
a,
Petros L.
Gkizis
 a,
Dragos F.
Flesariu
b,
Stamatis K.
Serviou
a,
Georgia A.
Zissimou
a,
Dragos F.
Flesariu
b,
Stamatis K.
Serviou
a,
Georgia A.
Zissimou
 b,
Panayiotis A.
Koutentis
b,
Panayiotis A.
Koutentis
 b and
Christoforos G.
Kokotos
b and
Christoforos G.
Kokotos
 *a
*a
aLaboratory of Organic Chemistry, Department of Chemistry, National and Kapodistrian University of Athens, Panepistimiopolis 15771, Athens, Greece. E-mail: ckokotos@chem.uoa.gr
bDepartment of Chemistry, University of Cyprus, P.O. Box 20537, 1678 Nicosia, Cyprus
First published on 9th March 2026
Abstract
3H-Phenothiazin-3-one is a small organic molecule with established biological properties, yet its potential as an organic photocatalyst remains largely unexplored. Herein, we report a green and efficient aerobic oxidation of boronic acids employing 3H-phenothiazin-3-one as the photocatalyst. The transformation proceeds under mild conditions, using green LED irradiation and atmospheric oxygen as the terminal oxidant. A broad range of boronic acids were successfully converted to the corresponding hydroxy derivatives, showcasing the excellent functional-group tolerance and synthetic utility of the developed photochemical protocol. Preliminary mechanistic investigations were undertaken to elucidate the reaction pathway and to identify the reactive oxygen species involved.
Introduction
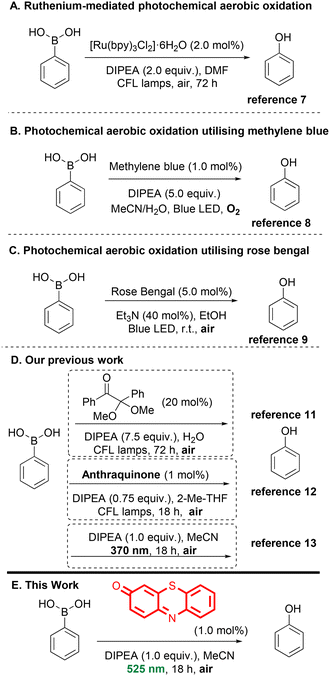
Boronic acids and esters are versatile intermediates in organic synthesis, capable of undergoing a wide range of functional group transformations. Among these, their conversion into phenols or alcohols is particularly valuable, providing a platform for further functionalization in pharmaceuticals, agrochemicals and materials science.1,2 In recent decades, sustainable and environmentally friendly methodologies have attracted significant attention.3 Visible light photochemistry using low-energy irradiation sources, such as LED lamps or sunlight, offers a green and mild alternative for promoting chemical reactions.3 This approach, recognized for more than a century,4 has gained renewed interest over the last two decades,5 due to its ability to perform transformations under milder and safer conditions, a feature highly appreciated in the pharmaceutical industry.The direct conversion of boronic acids or esters into hydroxy compounds is well-studied.6 Several photochemical protocols for the aerobic oxidation of boronic acids have been reported, using various photocatalysts. Jorgensen and co-workers employed a ruthenium-based catalyst in DMF (Scheme 1A).7 From a pharmaceutical industry perspective, the use of DMF and prolonged reaction time limits the practicality of this approach and raises environmental concerns, due to DMF's toxicity. In 2013, Scaiano and co-workers investigated methylene blue as an organic photocatalyst for the same transformation (Scheme 1B).8 Compared to the ruthenium complex [Ru(bpy)3Cl2]·6H2O, methylene blue offered superior mechanistic performance, although the substrate scope remained limited. Similarly, Zhang and co-workers utilized Rose Bengal as the photocatalyst (Scheme 1C),9 but this protocol required high catalyst loading (5 mol%). Our group has focused on developing eco-friendly, aerobic photochemical protocols.10 Using CFL lamps as the irradiation source, we identified 2,2-dimethoxyphenylacetophenone as a photoinitiator with N,N′-diisopropylethylamine (DIPEA) (7.5 equiv.) as sacrificial donor (Scheme 1D).11 Replacing 2,2-dimethoxyphenylacetophenone with anthraquinone allowed a 10-fold reduction in DIPEA loading (Scheme 1D),12 and further studies led to a catalyst-free UV-light protocol (370 nm) combined with a low amount of DIPEA under mild reaction conditions, further expanding the substrate scope.13
Despite these advances, milder reaction conditions using lower energy irradiation sources, such as green LEDs remain largely unexplored.14 A literature example of green LED-mediated oxidation of boronic acids employed a Zr-based MOF as the photocatalyst. Building on our prior studies of quinonimines as photocatalysts in aerobic oxidation,15 we investigated their application to the aerobic oxidation of boronic acids to the corresponding hydroxy derivatives. Quinonimines exhibit multiple biological and antioxidant activities,16 and molecules such as 3H-phenothiazin-3-one have demonstrated diverse biological effects, including antiparasitic activity,17 enzyme inhibition (e.g., succinoxidase, cytochrome oxidase, and cytochromes P450), and psychopharmacological effects.17–20 While phenothiazine derivatives, such as methylene blue21 or methylene violet,22 have been widely employed as photocatalysts, the structurally simpler 3H-phenothiazin-3-one remains underexplored in photochemical applications. Previous studies from the Koutentis group, described the synthesis of the quinonimines investigated in this work.16a,23,24 Building on these studies and our earlier investigations of the effect of irradiation wavelength on the aerobic oxidation process of boronic acids,13 we developed a mild, green and sustainable aerobic photooxidation protocol that converts boronic acids to hydroxy derivatives using 3H-phenothiazin-3-one as the photocatalyst under green light irradiation (Scheme 1E).
Results and discussion
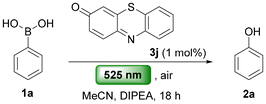
Previously, we had examined the effect of irradiation wavelength on the oxidation of boronic acid derivatives.13 Although that study provided a photocatalyst-free protocol, it required UV light (370 nm), which limits functional-group tolerance and promotes side reactions. To address these limitations, we sought to develop a milder photochemical method using green LED irradiation at a wavelength not absorbed by the substrate. As the transformation does not proceed in the absence of a photocatalyst,13 our effort focused on identifying a catalyst capable of promoting the reaction efficiently under these conditions. Optimisation commenced with phenylboronic acid (1a) as the model substrate and a small library of in-house quinonimines (3, 1 mol%), including benzotriazinones, phenazinones, phenoxazinones and phenothiazinones to identify the most active photocatalyst (Scheme 2) (see SI, Sect. S2). Reactions were carried out in acetonitrile with DIPEA as the reductant under 525 nm irradiation for 18 h. Quinonimine 3a delivered phenol 2a in low yield (7%), and modifications to the aromatic core (3b and 3c) offered no improvement. Notably, introduction of an anilino substituent at the α-position of the quinonimine moiety (3d) significantly enhanced the reaction efficiency, affording phenol 2a in 46% yield. Decorating the benzotriazinone scaffold with heteroaryl or trifluoromethyl groups led to poor yields (3e–3i), while derivative 3h, afforded 2a in only 4%. These unsatisfactory results prompted exploration of phenazine, phenothiazine, and phenoxazine derivatives. Phenothiazine 3j, N-phenylphenazinone 3l and phenoxazinone 3m proved highly effective affording phenol 2a in 90%, 79%, and 80% yields, respectively (Scheme 2). Considering the beneficial effect of an anilino substituent on the benzotriazinone scaffold (3avs. 3d, 7% → 46%), we next evaluated 2-phenylaminophenothiazinone 3k. In this case, the anilino substituent had a detrimental effect, and no product was observed, likely due to poor solubility in the reaction mixture.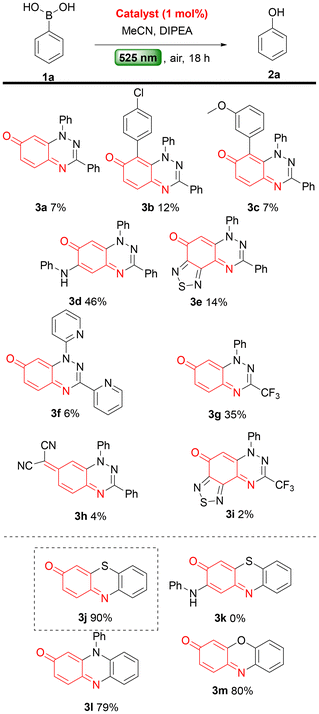 | ||
| Scheme 2 Quinonimine-based photocatalysts for the aerobic oxidation of phenylboronic acid (1a) under green LED irradiation (525 nm, 40 W). | ||
Upon identifying phenothiazinone 3j as the most efficient photocatalyst, we investigated solvent effects (Table 1) (see SI, Sect. S2). Polar aprotic solvents, such as dichloromethane, chloroform or ethyl acetate, afforded moderate yields (entries 2–4). Polar protic solvents (EtOH, i-PrOH) gave variable results, with i-PrOH completely suppressing reactivity (entries 5 and 6), while ethereal solvents (dioxane, THF) afforded moderate yields (entries 7 and 8). Reactions in non-polar solvents were sluggish (petroleum ether, cyclohexane; entries 9 and 10), likely due to poor solubility of the polar substrate and catalyst. Aromatic solvents, particularly toluene and xylene, performed best among the alternatives (entries 11–13), whereas benzene gave only moderate yield. The highly polar aprotic solvent dimethylsulfoxide (DMSO) completely suppressed reactivity (entry 14), likely due to quenching of the photocatalyst excited state or interference with electron-transfer processes. Although DMF and DMAc showed excellent performance by 1H NMR analysis, they were not pursued, due to their poor green credentials (entries 15 and 16). Water was also evaluated as a greener alternative, but low solubility of the organic substrate and catalyst resulted in only trace product (entry 17).
| Entry | Solvent | 2a (%) |
|---|---|---|
| a The reaction was performed with phenylboronic acid (1a) (24 mg, 0.20 mmol), DIPEA (26 mg, 0.20 mmol) and catalyst 3j (1.0 mol%) in solvent (1 mL), under green LED (Kessil PR160L, 525 nm, 40 W) irradiation for 18 h. Conversion was determined by 1H NMR. Yield of isolated product after column chromatography in parenthesis. | ||
| 1 | MeCN | 100 (90) |
| 2 | CH2Cl2 | 57 |
| 3 | CHCl3 | 29 |
| 4 | EtOAc | 60 |
| 5 | EtOH | 65 |
| 6 | i-PrOH | 0 |
| 7 | Dioxane | 55 |
| 8 | THF | 32 |
| 9 | Pet. Ether (40–60 °C) | 7 |
| 10 | Cyclohexane | 14 |
| 11 | Benzene | 65 |
| 12 | Toluene | 87 (58) |
| 13 | Xylene | 84 (79) |
| 14 | DMSO | 0 |
| 15 | DMF | 90 (61) |
| 16 | DMAc | 82 |
| 17 | H2O | 2 |
| 18 | H2O–MeCN (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) 1) |
85 (80) |
We next investigated the effect of the additive on the reaction outcome (Table 2). Having established DIPEA as the base, we first examined the effect of lowering its equivalents (see SI, Sect. S2). Decreasing the amount of DIPEA led to a gradual decline in yield, with 0.75 and 0.5 equivalents affording 63% and 50% of phenol 2a, respectively (entries 2 and 3). Use of primary amines, such as propylamine, were ineffective (5% yield, entry 4), while secondary amines, including morpholine or N-methylmorpholine, afforded poor yields (18% and 27%, entries 6 and 7). Tertiary amines, such as triethylamine, provided a moderate yield (76%, entry 5). The biomimetic reducing agent Hantzsch ester was also tested as a hydrogen donor, affording phenol 2a in 46% yield (entry 8). Finally, inorganic base K2CO3 proved completely ineffective, with no product observed (entry 9).
| Entry | Additive (equiv.) |
2a![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) a (%) a (%) |
|---|---|---|
| a The reaction was performed with phenylboronic acid (1a) (24 mg, 0.20 mmol) and catalyst 3j (1 mol%) in MeCN (1 mL), under green LED (Kessil PR160L, 525 nm, 40 W) irradiation for 18 h. b Yield of isolated product after column chromatography. | ||
| 1 | DIPEA (1) | 90 |
| 2 | DIPEA (0.75) | 63 |
| 3 | DIPEA (0.5) | 50 |
| 4 | Propylamine (1) | 5 |
| 5 | Et3N (1) | 76 |
| 6 | Morpholine (1) | 18 |
| 7 | N-Methylmorpholine (1) | 27 |
| 8 | Hantzsch ester (1) | 46 |
| 9 | K2CO3 (1) | 0 |
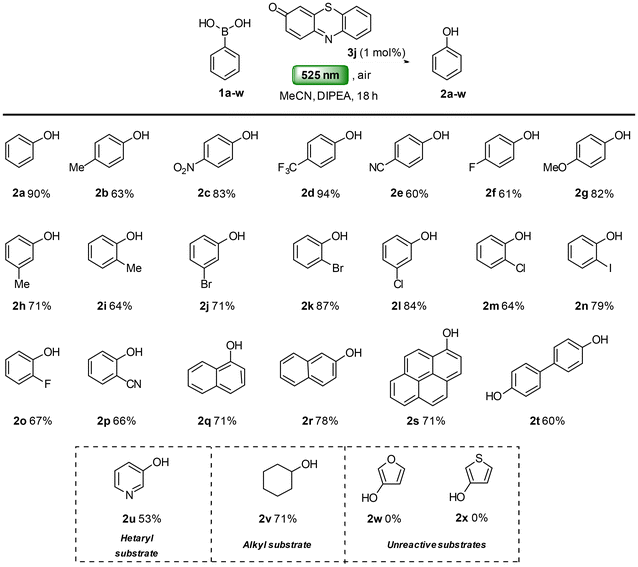
Having established the optimal reaction conditions, we next turned our attention to the substrate scope and functional group tolerance of the photooxidative protocol. A variety of boronic acids were subjected to the optimised conditions (Scheme 3). The reaction tolerates both electron-withdrawing and electron-releasing substituents at ortho-, meta- and para-positions of the phenyl boronic acid, affording the corresponding phenols in good to excellent yields (2a–p), indicating only a subtle electronic influence on the reaction efficiency. Minor variations in yields may be attributed to the possible sublimation during their isolation. The position of the substituent on the aryl ring did not impede the reaction, although slight differences in yield were observed (2h–r). Notably, in all cases, functional groups including methyl (2b, 2h, 2i), nitro (2c), trifluoromethyl (2d), cyano (2e, 2p), halogens (2f, 2j–o), and methoxy (2g) remained intact under the reaction conditions. Fused arylboronic acids were also well tolerated, giving the corresponding phenols in good to excellent yields (2q–s). More challenging substrates were then examined. 1,1′-Diphenyl-4,4′-bisboronic acid (1t), was converted to the corresponding 1,1′-diphenol (2t) in 60% yield. Heteroarylboronic acids (2w–x) were also tested: pyrid-3-ylboronic acid (1u) gave pyridin-3-ol (2u) in 53% yield without any N-oxidised side product. In contrast, fur-3-ylboronic acid (1w) and thien-3-ylboronic acid (1x) produced complex mixtures that were not further investigated. Finally, an alkylboronic acid, cyclohexylboronic acid (1v) gave cyclohexanol (2v) in 71% yield, with no overoxidized products, such as cyclohexanone, observed.
With phenothiazinone 3j established as an efficient photocatalyst for the aerobic photochemical oxidation of boronic acids, we next investigated the reaction mechanism. The involvement of radical intermediates was probed by conducting the reaction in the presence of TEMPO or BHT as radical scavengers (see SI, Table S4). In both cases, formation of the desired product was significantly suppressed, consistent with a radical pathway. Additional control experiments further demonstrated the necessity of all reaction components (Table 3).
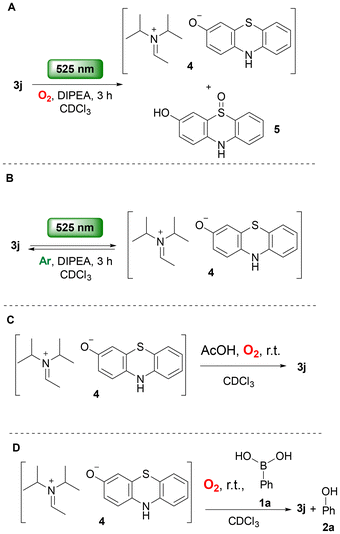
UV-Vis studies were conducted to probe the possible formation of an electron donor–acceptor (EDA) complex (see SI, Sect. S5), as the pivotal role of EDA complexes in photochemical reactions is well established.25 Mixing phenylboronic acid (1a) with DIPEA did not produce a bathochromic shift in the green-light region, and the addition of phenothiazinone 3j did not induce further spectral changes (SI, Fig. S6). However, upon irradiation of phenothiazinone 3j at 525 nm in the presence of DIPEA, a significant change in the UV-Vis spectrum was observed, suggesting that the excited catalyst interacts with DIPEA (SI, Fig. S5). Prior to irradiation, the mixture of catalyst 3j and DIPEA showed absorption bands at 367 and 500 nm, corresponding to the catalyst. After irradiation at 525 nm for 120 s, the original bands disappeared, and a single band at 323 nm appeared, characteristic of phenothiazines (SI, Fig. S5).26
To further probe the role of DIPEA and its interaction with the excited phenothiazinone catalyst 3j, a series of NMR experiments were conducted (see SI, Sect. S6). Irradiation of catalyst 3j or DIPEA individually produced no observable changes. However, upon irradiation of a mixture of catalyst 3j with DIPEA at 525 nm in CDCl3 for 3 h, consumption of catalyst 3j was observed, accompanied by the formation of 3-hydroxyphenothiazine salt 4 and 3-hydroxy-phenothiazine sulfoxide 5 (Scheme 4A). The formation of sulfoxide 5 can be rationalized by a photochemical aerobic sulfide oxidation. The aerobic oxidation of salt 4 to the correspondingsulfoxide 5 is well-precedented. Such oxidations of diaryl sulfides typically proceed via SET between the sulfide and molecular oxygen, generating superoxide radical anion.10c,27,28 Therefore, the formation of sulfoxide 5 provides evidence that superoxide anion is generated under our photochemical conditions.20 When the same irradiation experiment was carried out in the presence of phenylboronic acid (1a), no formation of by-product 5 was detected. Further evidence for the generation of superoxide anion came from the H2O2 test (see SI, Sect. S8). H2O2 was detected upon irradiation of phenothiazinone 3j with DIPEA at 525 nm, whereas irradiation of 3j in the absence of DIPEA did not produce H2O2. Additional experiments were conducted to gain insight into the fate of the 3-hydroxyphenothiazine salt 4. Irradiation of a mixture of phenothiazinone 3j with DIPEA at 525 nm under an oxygen-free atmosphere (argon) resulted exclusively in the formation of salt 4 (Scheme 4B). When the reaction mixture was subsequently stirred under air and treated with 1.0 equivalent of acetic acid to quench the salt, regeneration of catalyst 3j was observed (Scheme 4C). Furthermore, when phenylboronic acid (1a) was added instead of acetic acid, phenol (2a) was formed (Scheme 4D) (see SI, Sect. S6).
Cyclic voltammetry (CV) measurements of phenothiazinone 3j showed a reduction potential of Ered = −0.66 V vs. SCE (see SI, Sect. S9). The excited-state reduction potential of catalyst 3j was estimated at 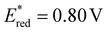 vs. SCE, indicating that the excited state is a strong oxidant capable of accepting an electron from DIPEA (Ered = 0.64 V vs. SCE).15 To evaluate possible side pathways, we assessed whether DIPEA could be oxidized by 1O2 to generate superoxide. This process is thermodynamically disfavoured: the reduction potential of 1O2 (Ered = −0.87 V vs. SCE)27 is more negative than the oxidation potential of DIPEA (Eox = 0.64 V vs. SCE). Consequently, oxidation of DIPEA by 1O2, to form superoxide is not supported.8 Additionally, the quantum yield (Φ) of the photochemical reaction was determined following the procedure by Yoon and co-workers.29 The value of Φ = 0.28 is consistent with a closed catalytic cycle rather than a radical chain mechanism.
vs. SCE, indicating that the excited state is a strong oxidant capable of accepting an electron from DIPEA (Ered = 0.64 V vs. SCE).15 To evaluate possible side pathways, we assessed whether DIPEA could be oxidized by 1O2 to generate superoxide. This process is thermodynamically disfavoured: the reduction potential of 1O2 (Ered = −0.87 V vs. SCE)27 is more negative than the oxidation potential of DIPEA (Eox = 0.64 V vs. SCE). Consequently, oxidation of DIPEA by 1O2, to form superoxide is not supported.8 Additionally, the quantum yield (Φ) of the photochemical reaction was determined following the procedure by Yoon and co-workers.29 The value of Φ = 0.28 is consistent with a closed catalytic cycle rather than a radical chain mechanism.
Summarizing our findings, a plausible mechanistic pathway is depicted in Scheme 5. Upon irradiation at 525 nm, phenothiazinone 3j is excited to its singlet state and via intersystem crossing is led to its triplet state 3j*. The excited photocatalyst then interacts with DIPEA via a SET event to produce the radical anion of photocatalyst 3j, which proceeds to be quenched by molecular oxygen and provides superoxide radical anion that oxidizes boronic acid 1a, leading to intermediate II (Scheme 5, Path A top). A HAT event leads to III, which upon rearrangement and hydrolysis leads to the desired product. Our mechanistic (NMR and UV-Vis) studies further support an alternative possible interaction between the excited state catalyst 3j* and DIPEA, leading to species V. Under aerobic conditions, V is unstable and undergoes oxidation, generating hydrogen peroxide, which can contribute to the oxidation of boronic acids (Scheme 5, Path B).
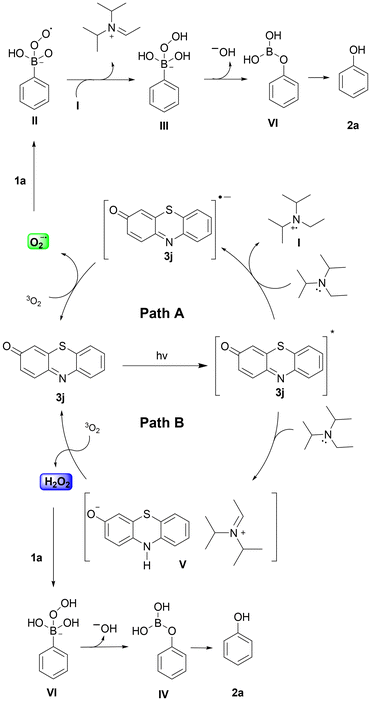 | ||
| Scheme 5 Proposed mechanistic pathway for the photochemical aerobic oxidation of phenylboronic acid (1a). | ||
Conclusions
The advent of photochemistry has opened new avenues for accessing unique reactivities. In particular, oxygen-mediated photochemical oxidation of boronic acids has attracted considerable attention. Herein, we report a mild and efficient photochemical protocol for the aerobic oxidation of boronic acids to their hydroxy derivatives, employing 3H-phenothiazin-3-one (3j) as the photocatalyst. This catalyst proved highly effective under green LED irradiation (525 nm), exhibiting excellent functional group tolerance across a broad range of substituted boronic acids.Author contributions
C. G. K. conceived the study, P. L. G., C. T. C., S. K. S. and C. G. K. designed the experiments and analyzed the data. D. F. F. and G. A. Z. performed the photocatalyst's synthesis. C. T. C., S. K. S. and P. L. G. performed the experiments. C. T. C. and P. L. G. prepared the draft of the manuscript. P. A. K. and C. G. K. performed the final editing. The manuscript was written through contributions of all authors.Conflicts of interest
There are no conflicts to declare.Data availability
The data that support the findings of this study are available in the supplementary information (SI). Supplementary information: experimental data, 1H and 13C NMR data, UV-Vis, optimisation and mechanistic studies. See DOI: https://doi.org/10.1039/d6ob00133e.Acknowledgements
The authors gratefully acknowledge the Hellenic Foundation for Research and Innovation (HFRI) for financial support through a grant, which is financed by “Greece 2.0 Basic Research Financing Action (Horizontal support of all Sciences) Sub-action 2 Funding Projects in Leading-Edge Sectors” (grant number 15949) and a grant, which is financed by 3rd Call for H.F.R.I. PhD Fellowships (fellowship number 18553). G.A.Z. and P.A.K. acknowledge funding from the European Regional Development Fund and the Republic of Cyprus through the Research and Innovation Foundation [POST-DOC/0524/0150]. The funders had no role in the study design, experimental execution, or data interpretation. D.F.F. and P.A.K. are grateful for funding from the Horizon Europe Framework Programme under the Marie Skłodowska-Curie Doctoral Networks (Grant Agreement 101119780; project duration: from Feb. 1, 2024, to Jan. 31, 2028).References
- (a) K. A. Scott, P. B. Cox and J. T. Njardarson, J. Med. Chem., 2022, 65, 7044–7072 CrossRef CAS PubMed; (b) M. A. Rahim, S. L. Kristufek, S. Pan, J. J. Richardson and F. Caruso, Angew. Chem., Int. Ed., 2019, 58, 1904–1927 CrossRef CAS PubMed.
- (a) J. H. P. Tyman, Synthetic and Natural Phenols, Elsevier, 1996, vol. 52 Search PubMed; (b) J.-J. Sui, D.-C. Xiong and X.-S. Ye, Chin. Chem. Lett., 2019, 30, 1533–1537 CrossRef CAS; (c) F. Yang, H. Zhang, X. Liu, B. Wang and L. Ackermann, Chin. J. Org. Chem., 2019, 39, 59–73 CrossRef CAS.
- For selected reviews, see: (a) C. Michelin and N. Hoffmann, Curr. Opin. Green Sustainable Chem., 2018, 10, 40–45 CrossRef; (b) S. Banerjee, S. Periyasamy, K. Muthukumaradoss, P. Deivasigamani and V. Saravanan, Front. Chem., 2025, 13, 1656935 CrossRef CAS PubMed.
- G. Ciamician, Science, 1912, 36, 385–394 CrossRef CAS PubMed.
- For selected reviews, see: (a) G. Goti, K. Manal, J. Sivaguru and L. Dell'Amico, Nat. Chem., 2024, 16, 684–692 CrossRef CAS PubMed; (b) P. L. Gkizis, Eur. J. Org. Chem., 2022, e202201139 CrossRef CAS; (c) P. L. Gkizis, I. Triandafillidi and C. G. Kokotos, Chem, 2023, 9, 3401–3414 CrossRef CAS; (d) F. Julia, ACS Catal., 2025, 15, 4665–4680 CrossRef CAS PubMed; (e) V. P. Demertzidou, E. Skolia and C. G. Kokotos, ChemCatChem, 2025, 17, e00760 CrossRef CAS.
- (a) L. Hao, G. Ding, D. A. Deming and Q. Zhang, Eur. J. Org. Chem., 2019, 7307–7321 CrossRef CAS; (b) L. Sun, D. Wang, Y. Li, B. Wu, Q. Li, C. Wang, S. Whang and B. Jiang, Chin. Chem. Lett., 2023, 34, 107490 CrossRef CAS.
- Y. Q. Zou, J. R. Chen, X. P. Liu, L. Q. Lu, R. L. Davis, K. A. Jorgensen and W. J. Xiao, Angew. Chem., Int. Ed., 2012, 51, 784–788 CrossRef CAS PubMed.
- S. P. Pitre, C. D. McTiernan, H. Ismaili and J. C. Scaiano, J. Am. Chem. Soc., 2013, 135, 13286–13289 CrossRef CAS PubMed.
- W.-Z. Weng, H. Liang and B. Zhang, Org. Lett., 2018, 20, 4979–4983 CrossRef CAS PubMed.
- For contributions in photochemical reactions, see: (a) E. Voutyritsa and C. G. Kokotos, Angew. Chem., Int. Ed., 2020, 59, 1735–1741 CrossRef CAS PubMed; (b) C. S. Batsika, C. Mantzourani, D. Gkikas, M. G. Kokotou, O. G. Mountanea, C. G. Kokotos, P. K. Politis and G. Kokotos, J. Med. Chem., 2021, 64, 5654–5666 CrossRef CAS PubMed; (c) E. Skolia, P. L. Gkizis, N. F. Nikitas and C. G. Kokotos, Green Chem., 2022, 24, 4108–4118 RSC; (d) O. G. Mountanea, E. Skolia and C. G. Kokotos, Chem. – Eur. J., 2024, 30, e202401588 CrossRef CAS PubMed; (e) N. A. Stini, E. T. Poursaitidis, N. F. Nikitas, M. Kartsinis, N. Spiliopoulou, P. Ananida-Dasenaki and C. G. Kokotos, Org. Biomol. Chem., 2023, 21, 1284–1289 RSC; (f) S. K. Serviou, P. L. Gkizis, D. P. Sánchez, S. Azar, A. H. G. David, C. Cabanetos and C. G. Kokotos, Eur. J. Org. Chem., 2025, e202500782 CrossRef CAS.
- I. K. Sideri, E. Voutyritsa and C. G. Kokotos, Synlett, 2018, 1324–1328 CAS.
- P. L. Gkizis, S. K. Serviou, A. Balaskas, C. T. Constantinou, I. Triandafillidi and C. G. Kokotos, Synlett, 2024, 330–336 CrossRef CAS.
- P. L. Gkizis, C. T. Constantinou and C. G. Kokotos, Eur. J. Org. Chem., 2023, e202300898 CrossRef CAS.
- T. Toyao, N. Ueno, K. Miyahara, Y. Matsui, T. H. Kim, Y. Horiuchi, H. Ikeda and M. Matsuoka, Chem. Commun., 2015, 51, 16103–16106 RSC.
- C. K. Spyropoulou, E. Skolia, D. F. Flesariu, G. A. Zissimou, P. L. Gkizis, I. Triandafillidi, M. Athanasiou, G. Itskos, P. A. Koutentis and C. G. Kokotos, Adv. Synth. Catal., 2023, 365, 2643–2650 CrossRef CAS.
- For selected examples, see: (a) P. A. Koutentis, H. Krassos and D. Lo Re, Org. Biomol. Chem., 2011, 9, 5228–5237 RSC; (b) L. A. J. Keane, S. I. Mirallai, M. Sweeney, M. P. Carty, G. A. Zissimou, A. A. Berezin, P. A. Koutentis and F. Aldabbagh, Molecules, 2018, 23, 574–582 CrossRef PubMed.
- A. Mackie and J. Raeburn, Br. J. Pharmacol. Chemother., 1952, 7, 215–518 CrossRef CAS PubMed.
- S. C. J. Oliver and W. I. Combe, Recl. Trav. Chim. Pays-Bas, 1950, 60, 526–534 CrossRef.
- J. B. Sutherland, J. P. Freeman, T. M. Heinze, J. D. Moody, I. A. Parhikov, A. J. Williams and D. Zhang, Xenobiotica, 2001, 31, 799–809 CrossRef CAS PubMed.
- K.-I. Hasegawa and Y. Ueno, Bull. Chem. Soc. Jpn., 1984, 57, 510–517 CrossRef CAS.
- R. I. Patel, A. Sharma, S. Sharma and A. Sharma, Org. Chem. Front., 2021, 8, 1694–1718 RSC.
- F. Ronzani, A. Trivella, P. Bordat, S. Blanc and S. Lacombea, J. Photochem. Photobiol., A, 2014, 284, 8–87 CrossRef CAS.
- G. A. Zissimou, A. Kourtellaris, M. Manoli and P. A. Koutentis, J. Org. Chem., 2018, 83, 9391–9402 CrossRef CAS PubMed.
- A. C. Savva, S. I. Mirallai, G. A. Zissimou, A. A. Berezin, M. Demetriades, A. Kourtellaris, C. P. Constantinides, C. Nicolaides, T. Trypiniotis and P. A. Koutentis, J. Org. Chem., 2017, 82, 7564–7575 CrossRef CAS PubMed.
- (a) W.-W. Liu, P. Xu, H.-X. Jiang, M.-L. Li, T.-Z. Hao, Y.-Q. Liu, S.-L. Zhu, K.-X. Zhang and X. Zhu, ACS Catal., 2024, 14, 10053–10059 CrossRef CAS; (b) A. K. Wortman and C. R. J. Stephenson, Chem, 2023, 9, 2390–2415 CrossRef CAS; (c) T. Tasnim, M. J. Ayodele and S. P. Pitre, J. Org. Chem., 2022, 87, 10555–10563 CrossRef CAS PubMed.
- M.-C. Chen, Y.-L. Lee, Z. X. Huang, D.-G. Chen and P.-T. Chou, Chem. – Eur. J., 2020, 26, 7124–7130 CrossRef CAS PubMed.
- S. M. Bonesi, I. Manet, M. Freccero, M. Fagnoni and A. Albini, Chem. – Eur. J., 2006, 12, 4844–4857 CrossRef CAS.
- E. Skolia, P. L. Gkizis and C. G. Kokotos, ChemPlusChem, 2022, 87, e202200008 CrossRef CAS PubMed.
- M. A. Cismesia and T. P. Yoon, Chem. Sci., 2015, 6, 5426–5434 RSC.
| This journal is © The Royal Society of Chemistry 2026 |