Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Dynamic N→B coordination and anion-selective turn-on fluorescence in oxadiazole-functionalized organoboranes
Jonas D. W.
Schepper
c,
Andreas
Orthaber
 b and
Frank
Pammer
b and
Frank
Pammer
 *a
*a
aHelmholtz Institute Ulm, Karlsruhe Institute for Technology, Helmholtzstrasse 11, 89081 Ulm, Germany. E-mail: frank.pammer@kit.edu
bDepartment of Chemistry – Ångström Laboratories, Uppsala University, BOX 523, 75120 Uppsala, Sweden. E-mail: andreas.orthaber@kemi.uu.se
cInstitute of Organic Chemistry II and Advanced Materials, Ulm University, Albert-Einstein-Allee 11, 89081 Ulm, Germany
First published on 3rd March 2026
Abstract
A versatile route for the preparation of chemically and electronically diverse Mes2BPh-based boranes (Mes = mesityl, 2,4,6-trimethylphenyl) is presented that allows the conversion of tetrazolyl rings in a common borane precursor (2H) into boranes bearing variously substituted oxadiazolyl groups. A series of eight boranes (4a–4h) was prepared with functional groups on the 5-position of the oxadiazole ranging from electron donating (4a: 4-Me-phenyl, 4b: 4-MeO-phenyl, etc.) to strongly electron withdrawing (4d: 4-O2N-phenyl, 4e: 3,5-bis(CF3)-phenyl, 4h: CF3) and also including a bifunctionalized example bearing two Mes2B moieties (4g). A full characterization study of the optical, electrochemical and electronic properties, both experimentally and by DFT calculations, was carried out. Our investigation shows that the boranes exhibit dynamic equilibria between closed intramolecularly N→B-coordinated and open non-coordinated conformers, as indicated by variable temperature NMR, 11B NMR and anion binding studies with F− and CN−. The anion binding studies reveal substantial differences in the fluorescence response of the compounds ranging from differing degrees of quenching to fluorescence shifts (4g) and enhanced emission (4c) (4-OMe-phenyl). These results show that this synthetic strategy allows easy creation of a series of compounds with incrementally varied optical properties and Lewis acidities.
Introduction
The exploitation of intramolecular dynamic processes is of increasing interest in current chemical research.1,2 For instance, hemilabile coordination of chelating ligands to transition metals has been recognized as crucial for many catalytic processes3 and was exploited in the development of sensing applications.4One of the greatest strengths of organic materials is their large structural variety and the resulting broad range of physical and electronic properties. In this context, the introduction of main group elements into organic scaffolds is being intensively investigated to access new structural motifs and to exploit unusual electronic effects.5 The introduction of boron6–14 has attracted particular interest in this regard as it can be incorporated in either the tri-7,15–17 or tetracoordinate18–26 form, which gives rise to different electronic and chemical properties.
Tricoordinate boron centers in pendant groups17,27–32 or embedded within π-systems33–40 lead to a lowering of the Lowest Unoccupied Molecular Orbital (LUMO) and hence increased electron affinity, due to conjugation with their pz-orbital. Likewise, tricoordinate boranes with tailored Lewis acidity are of great interest as catalysts and chemical sensors.41–43 Compounds featuring intramolecular N,C2-chelated tetracoordinate boron (N→B-ladders)26,44–49 also exhibit increased electron affinity and have therefore been considered as electron-transporting (n-type) materials.18–26,50 Recently, N→B-ladders have attracted growing interest due to their promising results in organic light emitting devices,51 n-channel organic field effect transistors,52 and organic photovoltaic cells.23,24,53 The N→B-ladder motif has also been exploited for generating compounds with helical topology.54–56
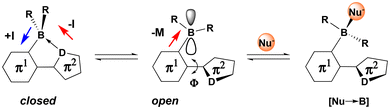
An intriguing feature of ladder boranes is that they can exist in dynamic equilibria between closed D→B-coordinated (D = N, O, P) and open non-coordinated conformers (Scheme 1). This behavior is observed, for instance, when the donor atom is part of a heterocycle with low basicity, such as a triazole57,58 or a tetrazole,59 and also in sterically congested compounds60 or when strained 4-,61,62 7-63 or 8-membered64,65 ring systems are formed. Similarly, reduced Lewis acidity at the boron center66 and weak Lewis bases such as ethers67 or carbonyl moieties68 facilitate dynamic coordination.
Labile D→B-coordination can have a strong influence on the electronic properties of a given π-system (Scheme 1). In the closed D→B-conformation, the π-system is planarized (Φ ≈ 0°), and conjugation along the backbone is more effective, while the tetracoordinate boron center acts as an electron-rich inductive donor (+I) towards the π1-ring systems. At the same time, the D→B-interaction exerts an electron withdrawing effect (−I) onto the donor atom (D) in the π2-ring. In the open conformation, the empty pz-orbital on boron exerts a strong electron withdrawing mesomeric effect (−M) on the π-system, while conjugation along the backbone is less efficient due to increased torsion (|Φ| ≫ 0°). Furthermore, in the open conformation, the boron center can act as a Lewis acid that can bind nucleophiles ([Nu→B]). This latter property of organoboranes has been exploited to develop e.g. chemical sensors for various anions69,70 and to develop fluorescent dyes with extreme Stokes’ shifts.67
A key limitation in the exploration of organoboranes is the limited choice of methods for their preparation. The most commonly used methods are step-wise metalation26,30,34 and electrophilic C–H-borylation.71,72 Hydroboration of suitable substrates can also yield a broad range of N→B-heterocycles.24,73–79
In this report, we demonstrate a versatile synthetic route to compounds capable of labile intramolecular N→B-coordination that builds on our previous work on this topic.
We developed strategies for the synthesis of N→B-heterocycles by building up the N-heterocyclic component through cycloaddition reactions. We found that 1,3-dipolar [3 + 2] azide–alkyne cycloaddition,57,58 [3 + 2] azide–nitrile cycloaddition,59 and cobalt-mediated [2 + 2 + 2] cycloaddition between nitriles and alkynes80 are highly efficient tools for the preparation of electronically and structurally diverse N→B-ladder boranes.
In a recent paper on the synthesis of tetrazole containing organoboranes (Scheme 2A),59 we reported the preparation of phenyltetrazole 2H, which proved to be a versatile starting material for the syntheses described herein: in this paper, we explored the scope of tetrazole/oxadiazole conversion for the preparation of a broad range of organoboranes with variable electronic properties (Schemes 2B and 4).
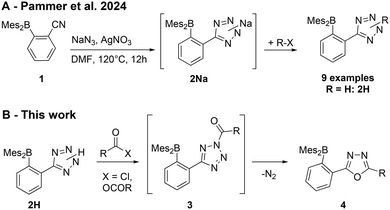 | ||
| Scheme 2 (A) Preparation of tetrazole-functionalized organoboranes81 and (B) their conversion into 1,3,4-oxadiazoles. | ||
The tetrazole ring in 2H can be acylated to give N-acyl-tetrazoles (3), which then rearrange into 2,5-disubstituted 1,3,4-oxadiazoles (4) with the concurrent loss of molecular nitrogen due to their thermal instability (Scheme 3).81,82 1,3,4-Oxadiazoles have a wide range of applications in pharmaceuticals83 and as semiconductors in organic light-emitting diodes (OLEDs).84,85 Examples of 1,3,4-oxadiazole-based ladder boranes have been reported86,87 that exhibit thermally activated delayed fluorescence (TADF)87 and can serve as blue-emitter materials in OLEDs with high electron mobilities.86
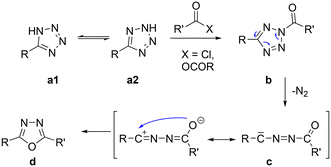 | ||
| Scheme 3 Mechanism for the conversion of tetrazoles into 1,3,4-oxadiazoles.81 | ||
A significant advance over previously reported N→B-ladders is the full conjugation across the heterocycle in the systems reported herein (Chart 1A, ‘type 4’). In triazole-57,58 (Chart 1B, ‘type 5’) and tetrazole-59 (Chart 1C, ‘type 6’) containing N→B-ladders, substituents could only be introduced on a saturated, conjugation-breaking ring nitrogen ( ). Unsaturated peripheral substituents therefore invariably experience only limited (cross-) conjugation in the π-system. In the case of tetrazoles (C), the synthesis only allowed the introduction of alkyl-substituents on the ring nitrogen, which preempts extension of the conjugated system. Due to a lack of boranes with directly comparable π-systems, the experimental data only weakly reflect the improved conjugation in type 4 vs. type 5/6, as will be discussed further on.
). Unsaturated peripheral substituents therefore invariably experience only limited (cross-) conjugation in the π-system. In the case of tetrazoles (C), the synthesis only allowed the introduction of alkyl-substituents on the ring nitrogen, which preempts extension of the conjugated system. Due to a lack of boranes with directly comparable π-systems, the experimental data only weakly reflect the improved conjugation in type 4 vs. type 5/6, as will be discussed further on.
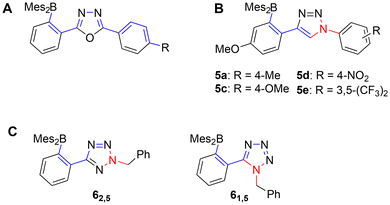 | ||
Chart 1 Conjugation across the heterocycle in different N→B-ladder boranes.  : Maximum conjugated pathway starting at boron. (A) Oxadiazoles in this work. (B) Triazoles adopted from ref. 57 (see also ref. 58). (C) Tetrazoles adopted from ref. 69. : Maximum conjugated pathway starting at boron. (A) Oxadiazoles in this work. (B) Triazoles adopted from ref. 57 (see also ref. 58). (C) Tetrazoles adopted from ref. 69.  : Conjugation breaking atoms/bonds. R: functional groups. : Conjugation breaking atoms/bonds. R: functional groups. | ||
Results and discussion
Synthesis and structure
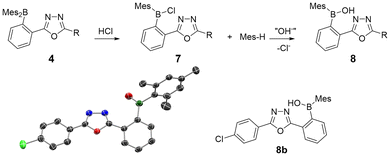
The oxadiazole synthesis was tested and optimized by acylating tetrazole 2H![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 59 with p-toluoyl chloride (→3a) at ambient temperature and subsequent conversion into 1,3,4-oxadiazole 4a (Table 1) at different temperatures. Upon addition of p-toluoyl chloride to a suspension of 2H in toluene, the reaction mixture immediately turned intensely yellow in color attributed to the formation of acyltetrazole 3a. Upon heating, foaming and gas evolution could be observed as deazotation set in and the yellow coloring steadily faded. When the deazotation reaction was carried out at 110 °C, the corresponding oxadiazole 4a could be isolated in a yield of 35% after purification by column chromatography (Table 1). Lower (90 °C) and higher (130 °C) reaction temperatures gave significantly lower yields of 15% and 13%, respectively (see Table 1). To increase the yield, different bases (NEt3 and K2CO3) were added to trap the liberated HCl. The use of NEt3 did not result in a substantial increase in yield, but the addition of K2CO3 allowed near quantitative isolation (95%) of 4a. A comparison of NMR experiments with and without the addition of K2CO3 (see Fig. S1A in the SI) showed that mesitylene (Mes-H) was formed after heating to 110 °C when no base was present, likely due to protolysis of the B-Mes bonds in 2H, 3 or 4 by HCl (see Fig. 1). In contrast, Mes-H-formation could not be observed when K2CO3 was added, since the base effectively captured the acid (see Fig. S1A in the SI). The protolysis would initially produce boryl-chloride 7 (Fig. 1) which could react further to the more chemically robust borinic acids (8) through reaction with trace moisture or OH− from the glass of the reaction vessel or during workup. This was confirmed in the synthesis of compound 4b, with the isolation of 8b (see Fig. 1) when no basic additive was used.
59 with p-toluoyl chloride (→3a) at ambient temperature and subsequent conversion into 1,3,4-oxadiazole 4a (Table 1) at different temperatures. Upon addition of p-toluoyl chloride to a suspension of 2H in toluene, the reaction mixture immediately turned intensely yellow in color attributed to the formation of acyltetrazole 3a. Upon heating, foaming and gas evolution could be observed as deazotation set in and the yellow coloring steadily faded. When the deazotation reaction was carried out at 110 °C, the corresponding oxadiazole 4a could be isolated in a yield of 35% after purification by column chromatography (Table 1). Lower (90 °C) and higher (130 °C) reaction temperatures gave significantly lower yields of 15% and 13%, respectively (see Table 1). To increase the yield, different bases (NEt3 and K2CO3) were added to trap the liberated HCl. The use of NEt3 did not result in a substantial increase in yield, but the addition of K2CO3 allowed near quantitative isolation (95%) of 4a. A comparison of NMR experiments with and without the addition of K2CO3 (see Fig. S1A in the SI) showed that mesitylene (Mes-H) was formed after heating to 110 °C when no base was present, likely due to protolysis of the B-Mes bonds in 2H, 3 or 4 by HCl (see Fig. 1). In contrast, Mes-H-formation could not be observed when K2CO3 was added, since the base effectively captured the acid (see Fig. S1A in the SI). The protolysis would initially produce boryl-chloride 7 (Fig. 1) which could react further to the more chemically robust borinic acids (8) through reaction with trace moisture or OH− from the glass of the reaction vessel or during workup. This was confirmed in the synthesis of compound 4b, with the isolation of 8b (see Fig. 1) when no basic additive was used.
With the reaction conditions optimized as described above, seven other 5-aryl-substituted 1,3,4-oxadiazoles could be synthesized using commercially available acid chlorides (see Scheme 4). Oxadiazoles 4a to 4f were isolated in very good yields of 84–99%. The diborylated compound 4g, which is accessible by using terephthalic acid dichloride as an acylating agent, could be isolated in only 36% yield. Both electron rich (4a, 4c) and electron poor (4d, 4e) aryl groups work equally well, with the exception of the 4-dimethylamino derivative (4i), which could not be generated.
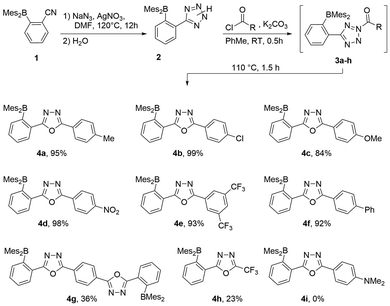 | ||
| Scheme 4 Synthesis of the borylated 1,3,4-oxadiazoles 4a to 4h starting from tetrazole 1. 4i did not form but is included in the computational survey. | ||
A 5-CF3-substituted oxadiazole (5h) was synthesized by acylation with trifluoroacetic anhydride.88 The corresponding acyltetrazole 3h is significantly less thermally stable with deazotation occurring at room temperature immediately after its formation. After purification by column chromatography, the CF3-functionalized oxadiazole 4h could be isolated in 23% yield.
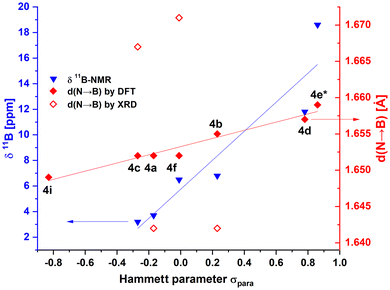
This is immediately apparent from the 11B NMR spectra of compounds 4a to 4f (Fig. 2: the chemical shifts of the 11B signals correlate linearly (R2 = 0.947) with the Hammett substituent parameters (σ) of the respective substituents on the phenyl ring, which can serve as measures for electron-withdrawing or electron-donating effects of the respective substituents.57,894g and 4h have been omitted from Fig. 3, since σ values cannot be given for these compounds. We included DFT data for 4i (Scheme 4), however, which could not be synthesized but was included in the computational survey.
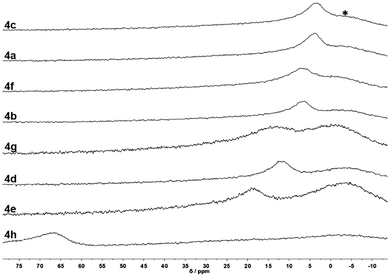 | ||
| Fig. 2 11B NMR spectra of the synthesized oxadiazoles. Recorded in either THF-d8 (4a–4f, 4h) or DCM (4g); * glass background signal. | ||
The more electron-rich oxadiazoles exhibit the most upfield 11B chemical shifts with signals at 3.7 ppm (4a) and 3.2 ppm (4c), which are within the range of four-coordinate boron centers of the form N→BAr3. Oxadiazoles with more neutral substituents exhibit slightly downfield 11B chemical shifts of 6.8 ppm (4b) and 6.5 ppm (4f), while for compounds with strongly electron-withdrawing groups 4d (–NO2) and 4e (m-CF3) clearly downfield-shifted 11B signals are observed at 11.8 ppm (4d) and 18.6 ppm (4e), respectively. The latter values fall into the range of N-alkylated phenyltetrazoles59 for which labile N→B-coordination was established. Oxadiazole 4g is only marginally soluble in THF but shows an 11B chemical shift of 13.1 ppm in DCM, in the same range as 4b and 4e. Compound 4h exhibits the most downfield 11B chemical shift of 67.1 ppm among the synthesized oxadiazoles. A value in this range suggests a purely three-coordinate boron center like Mes3B (79.0![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 90 ppm) or BPh3 (60.2
90 ppm) or BPh3 (60.2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 91 ppm).70 Therefore, N→B interactions do not occur in 4h.
91 ppm).70 Therefore, N→B interactions do not occur in 4h.
Single crystals suitable for X-ray diffraction analysis were obtained for oxadiazoles 4a, 4b, 4c, and 4f. The crystals were grown either by diffusion of n-pentane into THF solutions or by slow evaporation of THF solutions. The structures show that all four compounds adopt closed N→B-coordinated geometries in the solid state (see Fig. 4). As a result, the π systems are almost planarized with torsion angles between the oxadiazole and the PhBMes2 unit of 4.5° (4a), 5.1° (4b), 3.8° (4c) and 5.6° (4f). Curiously, unlike the 11B chemical shift, the experimental bond lengths do not correlate with the substituents on the phenyl ring. With respect to the N→B bond lengths, 4a (1.642(3) Å) and 4b (1.642(2) Å) form comparatively shorter bonds, while those in 4c (1.667(5) Å) and 4f (1.671(2) Å) are significantly longer. The bond lengths in 4a and 4b are comparable to those in N-benzyl-tetrazole-derivative 61,5 (1.643(8) Å, Chart 1),59 while those in 4c and 4f are far longer than the N→B-bonds in the structurally closely related triazole-based boranes 5a (1.632(2) Å), 5d (1.638(4) Å) and 5i (1.630(3) Å).57 This broad variation stands in contrast to the bond lengths in the computed structures, which correlate very well with the Hammett parameter σ (see “ ” in Fig. 3, R2 = 0.957). At first glance this correlation in itself might just be a reflection of a systemic error. However, for the structurally closely related type 5 boranes57 linear correlations (see Table 2, see also Fig. S58 in the SI) were found for both computed and experimental N→B-bond lengths, even though the calculations systematically overestimated the N–B-distance. We therefore tentatively attribute the large deviations to packing effects in the solid state.
” in Fig. 3, R2 = 0.957). At first glance this correlation in itself might just be a reflection of a systemic error. However, for the structurally closely related type 5 boranes57 linear correlations (see Table 2, see also Fig. S58 in the SI) were found for both computed and experimental N→B-bond lengths, even though the calculations systematically overestimated the N–B-distance. We therefore tentatively attribute the large deviations to packing effects in the solid state.
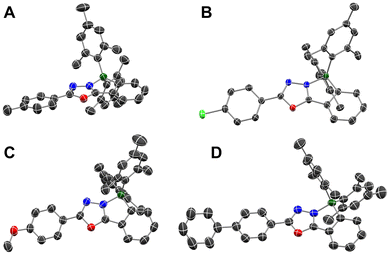 | ||
| Fig. 4 Crystal structures of oxadiazoles 4a (A), 4b (B), 4c (C) and 4f (D); ellipsoids are shown with 50% probability; hydrogen atoms have not been shown for clarity. | ||
| # | Compound | R on the Ph ring | σ | 11B-NMRb [ppm] | N→B bond lengths in oxadiazoles | N→B bond lengths in 1,2,3-triazolesr | ΔGopen/closed![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) e,f e,f |
FIAe,g [kJ mol−1] | ||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| XRDc [Å] | DFTd,e [Å] | XRDc [Å] | DFTd,e [Å] | Neutralf [kJ mol−1] | Anionsf [kJ mol−1] | Vs. open [kJ mol−1] | Vs. closed [kJ mol−1] | |||||
| a para-Hammett parameter, see ref. 894e: σ = 2 × σm(CF3) = 0.86. b Experimental 11B NMR shifts measured in THF-d8. c Experimental N→B-bond lengths derived from crystal structures. d N→B-bond lengths in the simulated structures. e Level of theory M06-2X/TZVP. f ΔG of the openvs.closed conformations in the neutral boranes and radical anions respectively. g Fluoride ion affinities vs. both open and closed conformers. The lower FIA corresponds to the more stable conformer. See the text for details. h 4g: Does not apply. 4h: 5-CF3-oxadiazolyl. See Scheme 4 for structures. i Recorded in CD2Cl2. j [open/open] vs. [closed/closed]. k [open/open] vs. [closed/open]. l 4g−1 [open/open] vs. [closed/closed]. m 4g−2 singlet, [open/open] vs. [closed/closed]. n FIA of 4g: [(F→B)/closed] vs. [open/open]. o FIA of 4g: [(F→B)/closed] vs. [closed/closed]. p Not synthesized. Only included in the DFT survey. q Ref. 43 reported 444 kJ mol−1 for this compound using BP86/SV(P). r Data for triazoles adopted from ref. 57. | ||||||||||||
| 1 | 4a | 4-Me | −0.17 | 3.7 | 1.642(3) | 1.652 | 1.632(2) | 1.668 | +4.9 | −8.3 | 307.4 | 302.5 |
| 2 | 4b | 4-Cl | 0.23 | 6.8 | 1.642(2) | 1.655 | 1.671 | −4.7 | −3.4 | 317.2 | 321.8 | |
| 3 | 4c | 4-OMe | −0.27 | 3.2 | 1.667(5) | 1.652 | 1.668 | +1.8 | −8.2 | 306.9 | 305.1 | |
| 4 | 4d | 4-NO2 | 0.78 | 11.8 | — | 1.657 | 1.638(4) | 1.678 | +4.4 | 45.3 | 363.9 | 359.5 |
| 5 | 4e | 3,5-(CF3)2 | 0.86 | 18.6 | — | 1.659 | 1.678 | −18.3 | 28.4 | 326.8 | 345.1 | |
| 6 | 4f | 4-Ph | −0.01 | 6.5 | closed | 1.652 | −7.0 | −7.7 | 313.0 | 314.3 | ||
| 7 | 4g | n.a.h | n.a. | 13.1i | — | 1.656 | −5.3j/−11.6k | −69.86l /+101.3m | 348.0n | 353.3o | ||
| 8 | 4h | CF3h | n.a. | 67.1 | — | 1.667 | −12.5 | −5.3 | 318.8 | 331.2 | ||
| 9 | 4i | 4-NMe2 | −0.83 | — | 1.649 | 1.630(3) | 1.676 | +7.4 | −13.7 | 301.5 | 294.1 | |
| 10 | Mes3Bp | 79.0 (ref. 90) | 287.7 | |||||||||
| 11 | Mes2BPhp | 79.5 (ref. 90) | 301.8 | |||||||||
| 12 | MesBPh2p | n.a. | 309.4 | |||||||||
| 13 | BPh3p | 60.2 (ref. 91) | 331.8 | |||||||||
| 14 | B(C6F5)3p | 471.4q | ||||||||||
The rotational barrier around the CMes–B bond could also be determined for compound 4a using variable temperature NMR spectroscopy (see Fig. S17 in the SI), whereby a coalescence temperature for the aromatic mesityl signal of 242 K was observed, corresponding to ΔGC = 47.7 ± 0.3 kJ mol−1, which is in a range similar to those for tetrazole-based N→B-ladders.59
Electrochemical properties
The electrochemical properties of the oxadiazoles were checked by cyclic voltammetry (CV) and square-wave voltammetry (SWV) (see section 2.5 in the SI). CV allowed classifying the irreversibility/reversibility of electrochemical reduction processes, while the first peak potentials observed in the SWV served to calculate the energies of the lowest unoccupied molecular orbitals (LUMOs) (Eredp; see Fig. S40 and S47 in the SI). The acceptor substituted compounds 4d, 4e, 4g and 4h exhibit higher electron affinities than the parent borane 1 (Eredp = −2.22 V vs. FcH/FcH+, ELUMO = −2.88 eV), with 4d showing the highest electron affinity at −1.38 V (ELUMO = −3.72 eV). Boranes with electron-rich (4a and 4c) or neutral (4b and 4f) substituents provide electron affinities that are lower than or comparable to that of borane 1, respectively.All oxadiazoles show only irreversible reductions (see Fig. S39), with the exceptions of 4e and 4g, which show quasi-reversible reductions at −1.99 V and −1.84 V (vs. FcH/FcH+), respectively. Only one reduction could be observed for compounds 4a, 4c, 4g and 4h. These reduction events are presumed to involve the empty p-orbital on boron.57,58 For the other oxadiazoles, between two (4b, 4e, 4f) and four reductions (4d) were detected. The two-fold reduction of 4b may be caused by electrochemical dechlorination92 and subsequent reduction of the defunctionalized borane, whereas the two-fold reduction of 4e and 4f is deemed to be a combination of reduction at the boron center and injection of an electron into the 5-aryl-oxadiazole π-system. The first reduction of 4d (−1.38 V vs. FcH/FcH+) is likely associated with a redox process centered on the nitro group since the potential is in the typical range of nitro-arenes.93 The origin of the other processes is unclear but may involve reduction at the boron center, injection of electrons into the 5-aryl-oxadiazole π-system or reductive degradation of the nitro group.
Optical and electronic properties
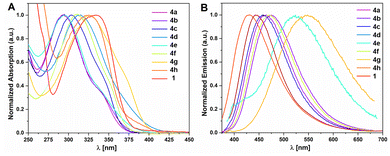
To characterize the optical properties of the boranes, absorption and fluorescence spectra were recorded in DCM solution (see Fig. 5A and B). All boranes exhibit absorption maxima between 292 and 325 nm accompanied by a shoulder band towards longer wavelengths. The underlying longest wavelength absorption band (λmax) that gives rise to the shoulder was derived for all compounds by Gaussian deconvolution of the experimental spectra (see Table 3 and Fig. S3–S10 in the SI). Its intensity relative to the main band varies significantly. We were able to conclude that λmax is associated with electronic transitions involving the empty pz-orbital on boron in open, non-N→B-coordinated conformers, as it is suppressed in the presence of strong nucleophiles (see below). An exception to this is 4d wherein the strongly accepting NO2 group rather than the BMes2 group dominates the optical properties. 4h shows the lowest energy λmax (385 nm/3.25 V) followed by 4g (369 nm/3.36 eV) and 4d (362 nm/3.43 eV), while the remaining boranes show values within ±0.05 eV of the parent borane 1 (346 nm, 3.58 eV), with the notable exception of the more electron rich 4a (338 nm/3.67 nm).| Compound |
E
redp![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) a,b [V] a,b [V] |
LUMOc [eV] | λ max [nm] | λ onset [nm] |
E
optg![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) e [eV] e [eV] |
λ em [nm] | Stokes shiftf [cm−1] | Φ [%] |
|---|---|---|---|---|---|---|---|---|
| a Peak-potential determined via square-wave-voltammetry in THF with 0.1 M [NnBu4][PF6], scan speed 0.1 V s−1. b Ir-/reversibility determined via cyclic voltammetry, scan speed 0.2 V s−1. c Relative to the LUMO energy of ferrocene (−5.1 eV).94 d Derived by deconvolution. See section 2.3 of the SI. e Derived from λonset. f Derived from λmax and λem. g Measured with an Ulbricht-integrating sphere. h Data adopted from ref. 57. i Data adopted from ref. 59. | ||||||||
| CN | −2.22r | −2.88 | 346 | 385 | 3.22 | 446 | 6480 | |
| 4a | −2.41i | −2.70 | 338 | 367 | 3.38 | 463 | 7988 | 34 |
| 4b | −2.25i | −2.85 | 342 | 372 | 3.33 | 475 | 8187 | 33 |
| −2.36i | ||||||||
| 4c | −2.43i | −2.67 | 345 | 366 | 3.39 | 459 | 7199 | 34 |
| 4d | −1.38qr | −3.72 | 362 | 380 | 3.26 | — | — | — |
| −1.63qr | ||||||||
| −1.90i | ||||||||
| −2.37i | ||||||||
| 4e | −1.96r | −3.14 | 348 | 378 | 3.28 | 521 | 9542 | n.d. |
| −2.56i | ||||||||
| 4f | −2.24i | −2.86 | 349 | 371 | 3.34 | 470 | 7377 | 35 |
| −2.81i | ||||||||
| 4g | −1.85r | −3.25 | 369 | 397 | 3.12 | 544 | 8718 | n.d. |
| 4h | −2.09i | −3.01 | 382 | 408 | 3.04 | 430 | 2922 | 4 |
| 5a | −2.64r | −2.46 | 330 | 360 | 3.44 | 380 | 3990 | 1 |
| 5c | −2.63r | −2.47 | 350 | 379 | 3.27 | 384 | 2530 | 2 |
| 5d | −1.52r | −3.58 | 360 | 417 | 2.97 | — | — | n.d. |
| 5e | −2.19r | −2.91 | 330 | 386 | 3.21 | — | — | n.d. |
| 6a | −2.58 | 314 | 352 | 3.52 | 444 | 9325 | 93 | |
| 6b | −2.66 | 337 | 353 | 3.51 | n.d. | — | — | |
With the exception of the NO2-functionalized oxadiazole 4d, all oxadiazoles synthesized here exhibit fluorescence with emission maxima between 430 nm (4h) and 544 nm (4g). Compounds 4a through 4g show quantum yields of 33–35% with large Stokes shifts between 6480 cm−1 (4a) and 6542 cm−1 (4e). 4h constitutes an exception with a Φ value of 4% and a much smaller Stokes shift of 2922 cm−1. The latter may be readily explained by the smaller molecular size of 4h, which consequently allows for less charge redistribution upon excitation.
Anion binding
Tricoordinate boron centers can bind various nucleophiles such as fluoride or cyanide anions.57,59,70,80 To investigate the influence of anion binding on the optical properties in more detail, absorption and emission spectra were recorded after the addition of an excess of tetrabutylammonium fluoride (TBAF) and tetrabutylammonium cyanide (TBACN) as fluoride and cyanide sources, respectively (see Fig. 6). With the exception of oxadiazole 4d, all oxadiazoles in the survey show a blue shift in the absorption that corresponds to a suppression of the λmax band. We attribute this to the binding of the anions to the boron center, because the empty pz-orbital contributes strongly to the LUMO of all open-conformers except 4d-open (see Fig. S51 to S53 in the SI). The LUMO is bound to participate in the lowest energy excitation. Consequently, binding of nucleophiles makes this electronic transition inaccessible. This is corroborated by experimental data on 4d: here, the main absorption band is suppressed in the presence of anions, but the onset remains unaffected. Orbital plots show that the pz-orbital contributes only to the LUMO+1 of 4d-open. Binding of nucleophiles is therefore expected to affect a higher energy transition, not the λmax-band.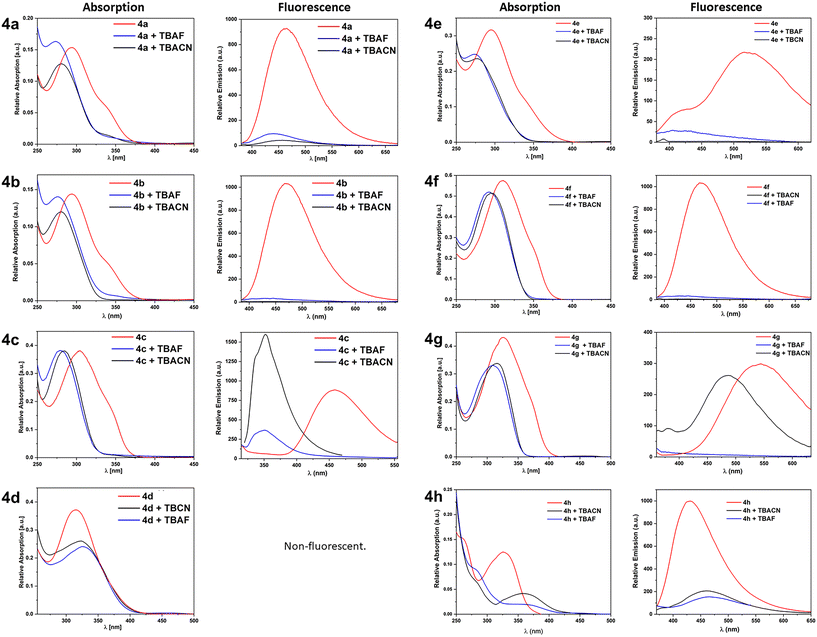 | ||
| Fig. 6 Normalized absorption and emission spectra of the synthesized oxadiazoles without and with the addition of an excess of TBAF or TBACN. Recorded in DCM. | ||
The nature of the anion does not seem to have a significant influence on the absorption properties of the oxadiazoles, as can be deduced from the very similar spectra of the fluoride- and cyanide-bound oxadiazoles.
The situation is somewhat different with regard to the fluorescence behavior. For most compounds (4a, 4b, 4e, 4f and 4h), the fluorescence intensity is partially (4a, 4e, and 4h) or fully suppressed regardless of the involved nucleophile (see Fig. 6).
In contrast, in the case of the OMe-substituted oxadiazole 4c, the addition of TBAF and TBACN leads to a blue shift of the emission from 459 nm to 351 nm (see Fig. 6). However, while TBAF effects a partial suppression of the emission, addition of TBACN leads to a significant increase in emission. The most interesting behavior has been observed for 4g: here, complete fluorescence quenching is observed upon addition of TBAF, whereas a blue shift of the emission from 544 nm to 486 nm occurs upon addition of TBACN (see Fig. 6). This means that oxadiazoles 4c and 4g are generally suitable to serve as fluorescence-based anion sensors. For 4c the fluorescence spectra of the pristine borane and of the F−1-saturated species (4c-F) form an isosbestic point at 395 nm, while the emission intensity after addition of TBACN is more intense at this wavelength. Consequently, selective detection at this wavelength allows discrimination between CN− and F−. A similar approach is possible for 4g. Below 504 nm the fluorescence of 4g-CN is more intense than that of 4g, while F−-addition quenches the emission altogether. At ca. 408 nm the residual fluorescence of 4g and 4g-F reaches barely above the background, while the emission from 4g-CN is significantly more intense.
Computational studies
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 80). Oxadiazoles with electron-donating substituents show shorter N→B bond lengths (4a: 1.652 Å, 4c: 1.652 Å, 4f: 1.652 Å) than compounds with electron-withdrawing substituents (4d: 1.657 Å, 4e: 1.659 Å, 4h: 1.667 Å). These results are consistent with the experimental 11B chemical shifts, which suggest a higher electron density at the boron center for the more electron-rich substituents.
80). Oxadiazoles with electron-donating substituents show shorter N→B bond lengths (4a: 1.652 Å, 4c: 1.652 Å, 4f: 1.652 Å) than compounds with electron-withdrawing substituents (4d: 1.657 Å, 4e: 1.659 Å, 4h: 1.667 Å). These results are consistent with the experimental 11B chemical shifts, which suggest a higher electron density at the boron center for the more electron-rich substituents.
Furthermore, we compared the energies of the open and closed N→B-coordinated conformers. The correlation of the Gibbs free energies indicates that the substituents on the oxadiazole regulate whether a closed N→B-coordination is favored over an open conformation (see Table 2). Boranes bearing strong acceptors (4d: ΔGo/c = −11.8 kJ mol−1, 4e: −18.3 kJ mol−1, 4h: −12.5 kJ mol−1) favor the open conformations. This may be attributed to the lower basicity of the oxadiazole-nitrogen due to the electron withdrawing functional groups. The reduced energetic gain from the N→B coordination cannot overcome the increased steric crowding around the tetrahedral boron center. Donor-substituted oxadiazoles (4a: ΔGo/c = +4.9 kJ mol−1, 4c: +1.8 kJ mol−1, 4i: +7.4 kJ mol−1) slightly favor the closed conformation. The effect is not large, however, and lies within a range of thermally accessible states at ambient temperature. The open and closed conformers of all compounds can therefore exist in dynamic equilibrium.
The conformers of the compounds introduced herein cover a slightly broader energetic range of about 25 kJ mol−1 (+7 kJ mol−1 to −18 kJ mol−1) – comparable to similarly labile N→B coordinating tetrazolyl-59 (+2 to −16 kJ mol−1) and triazolyl-functionalized57,58 (0 to −20 kJ mol−1) boranes. This may be due to the more direct conjugation to the functional group but could equally be caused by subtle differences in the coordination chemistry of oxadiazoles.
The situation is somewhat special in the diborylated 4g. The neutral borane slightly favors a closed/open (−5.3 kJ mol−1) or open/open (−11.6 kJ mol−1) conformation over a closed/closed arrangement. This trend is amplified in the radical anion 4g−1 (−69.9 kJ mol−1) which in the open/open−1 conformation benefits from stabilization by one empty pz orbital and delocalization across both oxadiazole rings in the wider π-system. In contrast, for the singlet dianion 4g−2 the open/open−2-conformation is strongly disfavored (+101.3 kJ mol−1). While the HOMOs of both the open/open−2 and the closed/closed−2-conformers involve delocalization across the whole π-system (see Fig. S52 in the SI), the closed/closed−2 conformation shows an evidently more effective conjugation across the planarized ring system.
Compared to other N→B-compounds, the range of energetic de-/stabilization of the closed conformers is broader for the oxadiazoles investigated herein compared to tetrazoles (2, Scheme 2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 59) but much weaker than in pyridine-based compounds.80
59) but much weaker than in pyridine-based compounds.80
The HOMO and HOMO−1 tend to be delocalized across the mesityl rings attached to boron, unless strong donors like –NMe2 (4i), –OMe (4c) or –Ph (4f) are present. Presumably, the very electron poor oxadiazole ring also lowers the frontier orbital energies of the π-system across the conjugated neighboring phenyl rings and thereby leaves the more electron rich mesityl rings to dominate the highest occupied orbitals. This effect is partially compensated for by donor–acceptor conjugation in 4i, 4c and 4f. Because of the very limited spatial overlap, the intensity of this transition – as indicated by the oscillator strength f – is expected to be very low (f = 0.036 (4i) to 0.125 (4d)).
The lowest energy transitions in closed conformers always involve a charge transfer from the mesityl groups towards π*-orbitals delocalized across the Ph-oxadiazole–Ph-π-system Apparently, the stronger +I-effect of the tetrahedral boron center makes the Mes groups much more electron rich. Only the strong donor –NMe2 leads to marginal conjugation across the Ph-oxadiazole–Ph-π-system in its HOMO.
The lowest energy transitions in most open conformers are 0.1 to 0.24 eV lower in energy than in closed ones, but electronic coupling seems more efficient in closed conformations, with f being between 2× (4e) and 34× (4i) higher than those in the corresponding open conformers. This corroborates the assumption that the longest-wavelength shoulder band in the absorption spectra originates from the open conformers.
| [Borane] + F−1 → [Borane–F]−1 | (1) |
Included in the survey were the oxadiazole-containing boranes 4a through 4i, along with Mes3B, Mes2BPh, MesBPh2, BPh3, and B(C6F5)3 to provide a frame of reference. For boranes 4a through 4i, the FIAs – expressed as −ΔG of the model reaction in kJ mol−1 –were computed for both open and closed forms. For the subsequent discussion the lowest FIA of the two is considered the most relevant because it represents the transition between the most stable neutral conformer and the [Borane–F]−1-complex.
The four reference boranes cover an FIA range from 287.7 kJ mol−1 for Mes3B to 331.8 kJ mol−1 for BPh3 and 471.4 kJ mol−1 for B(C6F5)3. From Mes3B to BPh3 the Lewis acidity increases because the Mes groups are successively replaced by the less sterically shielding and less electron rich phenyl rings. Still, BPh3 is rated as only moderately Lewis-acidic, while B(C6F5)3 is a benchmark for strong Lewis acids.
The FIAs of the synthesized boranes cover a range from 302.5 J mol−1 (4a) to 359.5 kJ mol−1 (4d). Borane 4i, which could not be accessed via the described route, gave a lower FIA of 294.1 kJ mol−1.
To put these results into perspective, while 4a–4h can be regarded as more sterically crowded than Mes2BPh, they all exhibit FIAs equal to (4a) or higher than that of this reference compound. The FIAs of the acceptor substituted 4d (359.5 kJ mol−1) and the bifunctional 4g (345.1 kJ mol−1) even exceed the FIA of BPh3. While even 4d and 4g must still be regarded as only moderately Lewis-acidic, these results indicate that the chemical transformation reported herein allows incrementally varying the Lewis acidity of a given borane precursor over a very broad range, even in the presence of other highly sterically shielding substituents on boron.
Conclusion
In summary, we reported the conversion of a tetrazolyl-functionalized borane precursor into a series of eight boranes bearing oxadiazolyl groups with chemically and electronically diverse substituents. The molecular structure allows for intramolecular N→B coordination and provides full conjugation between the boron center and the peripheral substituents.A full characterization study of the optical, electrochemical and electronic properties, both experimentally and by DFT calculations, showed that the borane exhibits dynamic N→B coordination with open and closed conformers of most boranes existing in dynamic equilibrium. 11B NMR indicates that only systems with the strongest acceptor (4h, 5-CF3-functionalized oxadiazole) exclusively adopt the open conformation.
Our most intriguing find is that the fluorescence behavior in particular varies drastically among the individual compounds in the presence of strong nucleophiles (F−, CN−). While differing degrees of fluorescence quenching are observed in most cases, 4c (4-OMe-phenyl) and 4g (bifunctional) exhibit fluorescence shifts and selective enhancement upon binding to CN−.
These results show that the synthetic strategy would offer a versatile synthetic route to libraries of tailored dyes, fluorescent sensors and Lewis acids.
Conflicts of interest
There are no conflicts of interest to declare.Data availability
The data that support the findings of this study are openly available in https://zenodo.org/ at https://zenodo.org/records/17866758.Supplementary information (SI): descriptions of the materials and instruments used, detailed synthetic procedures, and additional analytical data not provided in the main article. The latter part includes NMR (1H, 13C, 11B) spectra, optical spectra, electrochemical data, mass spectrometry data, and details on crystallographic data collection and computational results including optimized computed structures, orbital- and spin-density plots, and TDDFT results. See DOI: https://doi.org/10.1039/d5ob01979f.
The authors have cited additional references within the SI.95–102
CCDC 2514222–2514226 (4a, 4b, 4c, 4f and 8b) contain the supplementary crystallographic data for this paper.103a–e
Acknowledgements
JS and FP thank the German Science Foundation (DFG, German Research Foundation, project number FP 2217/4-1) and the German Chemical Industry Fund (FCI, Liebig scholarship to FP) for financial support. Prof. Orthaber thanks the Swedish Research Council (Vetenskapsrådet) for support.References
- W. Zhang and Y. Jin, Dynamic Covalent Chemistry: Principles, Reactions, and Applications, John Wiley & Sons, Hoboken, 2017 Search PubMed.
- Constitutional Dynamic Chemistry, ed. M. Barboiu, Springer Berlin Heidelberg, Berlin, Heidelberg, 2012, vol. 322 Search PubMed.
- A. J. M. Miller, Controlling ligand binding for tunable and switchable catalysis: cation-modulated hemilability in pincer-crown ether ligands, Dalton Trans., 2017, 46, 11987–12000 Search PubMed.
- S. E. Angell, C. W. Rogers, Y. Zhang, M. O. Wolf and W. E. Jones, Hemilabile coordination complexes for sensing applications, Coord. Chem. Rev., 2006, 250, 1829–1841 CrossRef CAS.
- T. Baumgartner and F. Jäkle, Main Group Strategies towards Functional Hybrid Materials, John Wiley & Sons, 2008 Search PubMed.
- A. Doshi and F. Jäkle, in Comprehensive Inorganic Chemistry II, Elsevier, 2013, pp. 861–891 Search PubMed.
- Y. Ren and F. Jäkle, Merging thiophene with boron: new building blocks for conjugated materials, Dalton Trans., 2016, 45, 13996–14007 RSC.
- F. Jäkle, Topics in Organometallic Chemistry, in Synthesis and Application of Organoboron Compounds. ed. E. Fernández and A. Whiting, Springer, Cham, 2015, vol. 49, pp. 297–325 Search PubMed.
- F. Jäkle, Lewis acidic organoboron polymers, Coord. Chem. Rev., 2006, 250, 1107–1121 CrossRef.
- F. Jäkle, Advances in the Synthesis of Organoborane Polymers for Optical, Electronic, and Sensory Applications, Chem. Rev., 2010, 110, 3985–4022 CrossRef PubMed.
- F. Jäkle, in Encyclopedia of Inorganic Chemistry, ed. R. B. King, Wiley & Sons Ltd, 2005 Search PubMed.
- C. D. Entwistle and T. B. Marder, Applications of Three-Coordinate Organoboron Compounds and Polymers in Optoelectronics, Chem. Mater., 2004, 16, 4574–4585 CrossRef CAS.
- C. D. Entwistle and T. B. Marder, Die Borchemie leuchtet: optische Eigenschaften von Molekülen und Polymeren, Angew. Chem., 2002, 114, 3051 CrossRef.
- D. Shimoyama and F. Jäkle, Controlling the aggregation and assembly of boron–containing molecular and polymeric materials, Aggregate, 2022, 3(2), e149 CrossRef CAS.
- E. A. Patrick and W. E. Piers, Twenty-five years of bis-pentafluorophenyl borane: a versatile reagent for catalyst and materials synthesis, Chem. Commun., 2020, 56, 841–853 RSC.
- E. von Grotthuss, A. John, T. Kaese and M. Wagner, Doping Polycyclic Aromatics with Boron for Superior Performance in Materials Science and Catalysis, Asian J. Org. Chem., 2018, 7, 37–53 CrossRef CAS.
- L. Ji, S. Griesbeck and T. B. Marder, Recent developments in and perspectives on three-coordinate boron materials: a bright future, Chem. Sci., 2017, 8, 846–863 RSC.
- X. Long, J. Yao, F. Cheng, C. Dou and Y. Xia, Double B←N bridged bipyridine-containing polymer acceptors with enhanced electron mobility for all-polymer solar cells, Mater. Chem. Front., 2019, 3, 70–77 RSC.
- X. Long, Z. Ding, C. Dou, J. Liu and L. Wang, A double B←N bridged bipyridine (BNBP)-based polymer electron acceptor: all-polymer solar cells with a high donor : acceptor blend ratio, Mater. Chem. Front., 2017, 1, 852–858 RSC.
- R. Zhao, C. Dou, Z. Xie, J. Liu and L. Wang, Polymer Acceptor Based on B←N Units with Enhanced Electron Mobility for Efficient All–Polymer Solar Cells, Angew. Chem., Int. Ed., 2016, 55, 5313–5317 CrossRef CAS PubMed.
- X. Long, Y. Gao, H. Tian, C. Dou, D. Yan, Y. Geng, J. Liu and L. Wang, Electron-transporting polymers based on a double B←N bridged bipyridine (BNBP) unit, Chem. Commun., 2017, 53, 1649–1652 RSC.
- X. Long, Z. Ding, C. Dou, J. Zhang, J. Liu and L. Wang, Polymer Acceptor Based on Double B←N Bridged Bipyridine (BNBP) Unit for High–Efficiency All–Polymer Solar Cells, Adv. Mater., 2016, 28, 6504–6508 CrossRef CAS PubMed.
- C. Dou, J. Liu and L. Wang, Conjugated polymers containing B←N unit as electron acceptors for all-polymer solar cells, Sci. China: Chem., 2017, 60, 450–459 CrossRef CAS.
- M. Grandl, J. Schepper, S. Maity, A. Peukert, E. Von Hauff and F. Pammer, N→B Ladder Polymers Prepared by Postfunctionalization: Tuning of Electron Affinity and Evaluation as Acceptors in All-Polymer Solar Cells, Macromolecules, 2019, 52, 1013–1024 Search PubMed.
- M. Vanga, A. Sahoo, R. A. Lalancette and F. Jäkle, Linear Extension of Anthracene via B←N Lewis Pair Formation: Effects on Optoelectronic Properties and Singlet O 2 Sensitization, Angew. Chem., 2022, 134, e202113075 CrossRef.
- A. Wakamiya, T. Taniguchi and S. Yamaguchi, Intramolecular B–N Coordination as a Scaffold for Electron–Transporting Materials: Synthesis and Properties of Boryl–Substituted Thienylthiazoles, Angew. Chem., 2006, 118, 3242–3245 CrossRef.
- D.-T. Yang, J. Radtke, S. K. Mellerup, K. Yuan, X. Wang, M. Wagner and S. Wang, One-Pot Synthesis of Brightly Fluorescent Mes2B-Functionalized Indolizine Derivatives via Cycloaddition Reactions, Org. Lett., 2015, 17, 2486–2489 CrossRef CAS PubMed.
- D. R. Levine, M. A. Siegler and J. D. Tovar, Thiophene-Fused Borepins As Directly Functionalizable Boron-Containing π-Electron Systems, J. Am. Chem. Soc., 2014, 136, 7132–7139 CrossRef CAS PubMed.
- C. Reus, S. Weidlich, M. Bolte, H.-W. Lerner and M. Wagner, C-Functionalized, Air- and Water-Stable 9,10-Dihydro-9,10-diboraanthracenes: Efficient Blue to Red Emitting Luminophores, J. Am. Chem. Soc., 2013, 135, 12892–12907 CrossRef CAS PubMed.
- A. Wakamiya, K. Mori and S. Yamaguchi, 3-Boryl–2,2′–bithiophene as a Versatile Core Skeleton for Full–Color Highly Emissive Organic Solids, Angew. Chem., Int. Ed., 2007, 46, 4273–4276 Search PubMed.
- F. Vidal, J. McQuade, R. Lalancette and F. Jäkle, ROMP-Boranes as Moisture-Tolerant and Recyclable Lewis Acid Organocatalysts, J. Am. Chem. Soc., 2020, 142, 14427–14431 CrossRef CAS PubMed.
- H. Lin, S. Patel and F. Jäkle, Tailored Triarylborane Polymers as Supported Catalysts and Luminescent Materials, Macromolecules, 2020, 53, 10601–10612 Search PubMed.
- T. Kushida and S. Yamaguchi, A Radical Anion of Structurally Constrained Triphenylborane, Organometallics, 2013, 32, 6654–6657 CrossRef CAS.
- X. Yin, J. Chen, R. A. Lalancette, T. B. Marder and F. Jäkle, Highly Electron–Deficient and Air–Stable Conjugated Thienylboranes, Angew. Chem., 2014, 126, 9919–9923 CrossRef.
- X. Yin, F. Guo, R. A. Lalancette and F. Jäkle, Luminescent Main-Chain Organoborane Polymers: Highly Robust, Electron-Deficient Poly(oligothiophene borane)s via Stille Coupling Polymerization, Macromolecules, 2016, 49, 537–546 CrossRef CAS.
- V. M. Hertz, N. Ando, M. Hirai, M. Bolte, H.-W. Lerner, S. Yamaguchi and M. Wagner, Steric Shielding vs Structural Constraint in a Boron-Containing Polycyclic Aromatic Hydrocarbon, Organometallics, 2017, 36, 2512–2519 CrossRef CAS.
- N. Baser-Kirazli, R. A. Lalancette and F. Jäkle, Tuning the Donor–π–Acceptor Character of Arylborane–Arylamine Macrocycles, Organometallics, 2021, 40, 520–528 CrossRef CAS.
- Z. Yang, K. Yang, X. Wei, W. Liu, R. Gao, F. Jäkle, Y.-L. Loo and Y. Ren, A Multiple Excited-State Engineering of Boron-Functionalized Diazapentacene Via a Tuning of the Molecular Orbital Coupling, J. Phys. Chem. Lett., 2021, 12, 9308–9314 CrossRef CAS PubMed.
- P. Li, D. Shimoyama, N. Zhang, Y. Jia, G. Hu, C. Li, X. Yin, N. Wang, F. Jäkle and P. Chen, A New Platform of B/N–Doped Cyclophanes: Access to a π–Conjugated Block–Type B 3 N 3 Macrocycle with Strong Dipole Moment and Unique Optoelectronic Properties, Angew. Chem., Int. Ed., 2022, 61, e202200612 CrossRef CAS PubMed.
- A. F. Alahmadi, X. Yin, R. A. Lalancette and F. Jäkle, Synthesis and Structure–Property Relationships in Regioisomeric Alternating Borane–Terthiophene Polymers, Chem. – Eur. J., 2022, 19(18), e202203619 CrossRef PubMed.
- I. B. Sivaev and V. I. Bregadze, Lewis acidity of boron compounds, Coord. Chem. Rev., 2014, 270–271, 75–88 CrossRef CAS.
- R. J. Mayer, N. Hampel and A. R. Ofial, Lewis Acidic Boranes, Lewis Bases, and Equilibrium Constants : A Reliable Scaffold for a Quantitative Lewis Acidity/Basicity Scale, Angew. Chem., Int. Ed., 2008, 47, 7659–7663 CrossRef PubMed.
- L. O. Müller, D. Himmel, J. Stauffer, G. Steinfeld, J. Slattery, G. Santiso-Quinones, V. Brecht and I. Krossing, Simple Access to the Non–Oxidizing Lewis Superacid PhF->Al(ORF)3 RF = C(CF3)3, Angew. Chem., Int. Ed., 2008, 47, 7659–7663 CrossRef PubMed.
- M. Fontani, F. Peters, W. Scherer, W. Wachter, M. Wagner and P. Zanello, Adducts of Ferrocenylboranes and Pyridine Bases: Generation of Charge-Transfer Complexes and Reversible Coordination Polymers, Eur. J. Inorg. Chem., 1998, 1998, 1453–1465 CrossRef.
- A. F. Alahmadi, J. Zuo and F. Jäkle, B-N Lewis pair-fused dipyridylfluorene copolymers incorporating electron-deficient benzothiadiazole comonomers, Polym. J., 2023, 55, 433–442 CrossRef CAS.
- J. Zuo, K. Liu, J. Harrell, L. Fang, P. Piotrowiak, D. Shimoyama, R. A. Lalancette and F. Jäkle, Near–IR Emissive B–N Lewis Pair–Functionalized Anthracenes via Selective LUMO Extension in Conjugated Dimer and Polymer, Angew. Chem., Int. Ed., 2024, 63, e202411855 CrossRef CAS PubMed.
- M. Vanga, A. Sahoo, R. A. Lalancette and F. Jäkle, Linear Extension of Anthracene via B←N Lewis Pair Formation: Effects on Optoelectronic Properties and Singlet O 2 Sensitization, Angew. Chem., Int. Ed., 2022, 61, e202113075 CrossRef CAS PubMed.
- Y. Shi, Y. Zeng, P. Kucheryavy, X. Yin, K. Zhang, G. Meng, J. Chen, Q. Zhu, N. Wang, X. Zheng, F. Jäkle and P. Chen, Dynamic B/N Lewis Pairs: Insights into the Structural Variations and Photochromism via Light–Induced Fluorescence to Phosphorescence Switching, Angew. Chem., Int. Ed., 2022, 61, e202213615 CrossRef CAS PubMed.
- R. P. Nandi, J. Zuo and F. Jaekle, B-N Fused Anthracene as Functional Linker for π-Extended Viologens: Near-IR Emission and Electrochromism, Angew. Chem., 2026, 138, e21634 CrossRef.
- J. Miao, Y. Wang, J. Liu and L. Wang, Organoboron molecules and polymers for organic solar cell applications, Chem. Soc. Rev., 2022, 51, 153–187 RSC.
- S. Wang, D. Yang, J. Lu, H. Shimogawa, S. Gong, X. Wang, S. K. Mellerup, A. Wakamiya, Y. Chang, C. Yang and Z. Lu, In Situ Solid–State Generation of (BN)2-Pyrenes and Electroluminescent Devices, Angew. Chem., Int. Ed., 2015, 54, 15074–15078 CrossRef CAS PubMed.
- R. Hecht, J. Kade, D. Schmidt and A. Nowak-Król, n–Channel Organic Semiconductors Derived from Air–Stable Four–Coordinate Boron Complexes of Substituted Thienylthiazoles, Chem. – Eur. J., 2017, 23, 11620–11628 CrossRef CAS PubMed.
- M. Liu, D. Zhou, Y. B. He, Y. Fu, X. Qin, C. Miao, H. Du, B. Li, Q. H. Yang, Z. Lin, T. S. Zhao and F. Kang, Novel gel polymer electrolyte for high-performance lithium-sulfur batteries, Nano Energy, 2016, 22, 278–289 CrossRef CAS.
- X. Zhang, F. Rauch, J. Niedens, R. B. da Silva, A. Friedrich, A. Nowak-Król, S. J. Garden and T. B. Marder, Electrophilic C–H Borylation of Aza[5]helicenes Leading to Bowl-Shaped Quasi-[7]Circulenes with Switchable Dynamics, J. Am. Chem. Soc., 2022, 144, 22316–22324 CrossRef CAS PubMed.
- D. Volland, J. Niedens, P. T. Geppert, M. J. Wildervanck, F. Full and A. Nowak-Król, Synthesis of a Blue–Emissive Azaborathia[9]helicene by Silicon–Boron Exchange from Unusual Atropisomeric Teraryls, Angew. Chem., Int. Ed., 2023, 62(30), e20230491 CrossRef PubMed.
- F. Full, Q. Wölflick, K. Radacki, H. Braunschweig and A. Nowak-Król, Enhanced Optical Properties of Azaborole Helicenes by Lateral and Helical Extension, Chem. – Eur. J., 2022, 28(62), e202202280 CrossRef CAS PubMed.
- R. Koch, Y. Sun, A. Orthaber, A. J. Pierik and F. Pammer, Turn-on fluorescence sensors based on dynamic intramolecular N→B-coordination, Org. Chem. Front., 2020, 7, 1437–1452 RSC.
- S. Schraff, Y. Sun and F. Pammer, Tuning of electronic properties via labile N→B-coordination in conjugated organoboranes, J. Mater. Chem. C, 2017, 5, 1730–1741 RSC.
- J. Schepper, A. Orthaber and F. Pammer, Tetrazole–Functionalized Organoboranes Exhibiting Dynamic Intramolecular N→B–Coordination and Cyanide–Selective Anion Binding, Chem. – Eur. J., 2024, 30, e202401466 CrossRef CAS PubMed.
- J. Chen, R. A. Lalancette and F. Jäkle, Chiral Organoborane Lewis Pairs Derived from Pyridylferrocene, Chem. – Eur. J., 2014, 20, 9120–9129 CrossRef CAS PubMed.
- S. Schwendemann, R. Fröhlich, G. Kehr and G. Erker, Intramolecular frustrated N/B Lewis pairs by enamine hydroboration, Chem. Sci., 2011, 2, 1842 RSC.
- G.-Q. Chen, F. Türkyilmaz, C. G. Daniliuc, C. Bannwarth, S. Grimme, G. Kehr and G. Erker, Enamine/butadienylborane cycloaddition in the frustrated Lewis pair regime, Org. Biomol. Chem., 2015, 13, 10477–10486 RSC.
- C. Zeng, K. Yuan, N. Wang, T. Peng, G. Wu and S. Wang, The opposite and amplifying effect of B ← N coordination on photophysical properties of regioisomers with an unsymmetrical backbone, Chem. Sci., 2019, 10, 1724–1734 RSC.
- Y. Cao, J. K. Nagle, M. O. Wolf and B. O. Patrick, Tunable Luminescence of Bithiophene-Based Flexible Lewis Pairs, J. Am. Chem. Soc., 2015, 137, 4888–4891 CrossRef CAS PubMed.
- Y. Cao, N. E. Arsenault, D. Hean and M. O. Wolf, Fluorescence Switching of Intramolecular Lewis Acid–Base Pairs on a Flexible Backbone, J. Org. Chem., 2019, 84, 5394–5403 CrossRef CAS PubMed.
- C. T. Hoang, I. Prokes, G. J. Clarkson, M. J. Rowland, J. H. R. Tucker, M. Shipman and T. R. Walsh, Study of boron–nitrogen dative bonds using azetidine inversion dynamics, Chem. Commun., 2013, 49, 2509 RSC.
- S. J. Cassidy, I. Brettell-Adams, L. E. McNamara, M. F. Smith, M. Bautista, H. Cao, M. Vasiliu, D. L. Gerlach, F. Qu, N. I. Hammer, D. A. Dixon and P. A. Rupar, Boranes with Ultra-High Stokes Shift Fluorescence, Organometallics, 2018, 37, 3732–3741 CrossRef CAS.
- Y. Shi, J. Wang, H. Li, G. Hu, X. Li, S. K. Mellerup, N. Wang, T. Peng and S. Wang, A simple multi-responsive system based on aldehyde functionalized amino-boranes, Chem. Sci., 2018, 9, 1902–1911 RSC.
- Z. M. Hudson and S. Wang, Impact of Donor–Acceptor Geometry and Metal Chelation on Photophysical Properties and Applications of Triarylboranes, Acc. Chem. Res., 2009, 42, 1584–1596 CrossRef CAS PubMed.
- C. R. Wade, A. E. J. J. Broomsgrove, S. Aldridge and F. P. Gabbaï, Fluoride ion complexation and sensing using organoboron compounds, Chem. Rev., 2010, 110, 3958–3984 CrossRef CAS PubMed.
- N. Ishida, T. Moriya, T. Goya and M. Murakami, Synthesis of Pyridine–Borane Complexes via Electrophilic Aromatic Borylation, J. Org. Chem., 2010, 75, 8709–8712 CrossRef CAS PubMed.
- S. A. Iqbal, J. Pahl, K. Yuan and M. J. Ingleson, Intramolecular (directed) electrophilic C–H borylation, Chem. Soc. Rev., 2020, 49, 4564–4591 RSC.
- M. Grandl, B. Rudolf, Y. Sun, D. F. Bechtel, A. J. Pierik and F. Pammer, Intramolecular N→B Coordination as a Stabilizing Scaffold for π-Conjugated Radical Anions with Tunable Redox Potentials, Organometallics, 2017, 36, 2527–2535 CrossRef CAS.
- M. Grandl, Y. Sun and F. Pammer, Generation of an N→B Ladder-type Structure by Regioselective Hydroboration of an Alkenyl-Functionalized Quaterpyridine, Chem. – Eur. J., 2016, 22, 3976–3980 CrossRef CAS PubMed.
- M. Grandl, T. Kaese, A. Krautsieder, Y. Sun and F. Pammer, Hydroboration as an Efficient Tool for the Preparation of Electronically and Structurally Diverse N→B-Heterocycles, Chem. – Eur. J., 2016, 22, 14373–14382 CrossRef CAS PubMed.
- M. Grandl, Y. Sun and F. Pammer, Electronic and structural properties of N→B-ladder boranes with high electron affinity, Org. Chem. Front., 2018, 5, 336–352 RSC.
- F. Pammer, J. Schepper, J. Glöckler, Y. Sun and A. Orthaber, Expansion of the scope of alkylboryl-bridged N→B-ladder boranes: New substituents and alternative substrates, Dalton Trans., 2019, 48, 10298–10312 RSC.
- M. Grandl and F. Pammer, Preparation of Head-to-Tail Regioregular 6-(1-Alkenyl)-Functionalized Poly(pyridine-2,5-diyl) and its Post-Functionalization via Hydroboration, Macromol. Chem. Phys., 2015, 216, 2249–2262 CrossRef CAS.
- K. Yuan, N. Suzuki, S. K. Mellerup, X. Wang, S. Yamaguchi and S. Wang, Pyridyl Directed Catalyst-Free trans -Hydroboration of Internal Alkynes, Org. Lett., 2016, 18, 720–723 CrossRef CAS PubMed.
- J. D. W. Schepper, A. Orthaber and F. Pammer, Preparation of Structurally and Electronically Diverse N→B-Ladder Boranes by [2 + 2 + 2] Cycloaddition, J. Org. Chem., 2021, 86, 14767–14776 CrossRef CAS PubMed.
- B. Reichart and C. O. Kappe, High-temperature continuous flow synthesis of 1,3,4-oxadiazoles via N-acylation of 5-substituted tetrazoles, Tetrahedron Lett., 2012, 53, 952–955 CrossRef CAS.
- R. Huisgen, J. Sauer and H. J. Sturm, Acylierung 5-substituierter Tetrazole zu 1.3.4-Oxdiazolen, Angew. Chem., 1958, 9, 272–273 CrossRef.
- V. K. Gour, S. Yahya and M. Shahar Yar, Unveiling the chemistry of 1,3,4-oxadiazoles and thiadiazols: A comprehensive review, Arch. Pharm., 2023, 357, 2300328 CrossRef PubMed.
- J. Lu, I. Persson, H. Lind, J. Palisaitis, M. Li, Y. Li, K. Chen, J. Zhou, S. Du, Z. Chai, Z. Huang, L. Hultman, P. Eklund, J. Rosen, Q. Huang and P. O. Å. Persson, Tin+1Cn MXenes with fully saturated and thermally stable Cl terminations, Nanoscale Adv., 2019, 1, 3680–3685 RSC.
- U. Mitschke and P. Bäuerle, The electroluminescence of organic materials, J. Mater. Chem., 2000, 10, 1471–1507 RSC.
- R. Deng, L. Li, M. Song, S. Zhao, L. Zhou and S. Yao, Synthesis and optoelectronic properties of oxadiazole coordinated boron complexes, CrystEngComm, 2016, 18, 4382–4387 RSC.
- D. Zhou, D. Liu, X. Gong, H. Ma, G. Qian, S. Gong, G. Xie, W. Zhu and Y. Wang, Solution-Processed Highly Efficient Bluish-Green Thermally Activated Delayed Fluorescence Emitter Bearing an Asymmetric Oxadiazole–Difluoroboron Double Acceptor, ACS Appl. Mater. Interfaces, 2019, 11, 24339–24348 CrossRef CAS PubMed.
- L. I. Vereshchagin, O. N. Verkhozina, F. A. Pokatilov and V. N. Kizhnyaev, Synthesis of 2-Substituted 5-Trifluoromethyl-1, 3, 4-oxadiazoles, Russ. J. Org. Chem., 2007, 43, 1575–1576 CrossRef CAS.
- C. Hansch, A. Leo and R. W. Taft, A Survey of Hammett Substituent Constants and Resonance and Field Parameters, Chem. Rev., 1991, 91, 165–195 CrossRef CAS.
- N. M. D. Brown, F. Davidson and J. W. Wilson, Dimesitylboryl compounds, J. Organomet. Chem., 1981, 209, 1–11 CrossRef CAS.
- C. D. Good and D. M. Ritter, Alkenylboranes. II. Improved Preparative Methods and New Observations on Methylvinylboranes, J. Am. Chem. Soc., 1962, 84, 1162–1166 CrossRef CAS.
- N. Hoshi, K. Sasaki, S. Hashimoto and Y. Hori, Electrochemical dechlorination of chlorobenzene with a mediator on various metal electrodes, J. Electroanal. Chem., 2004, 568, 267–271 CrossRef CAS.
- H. Jung, R. H. Heflich, P. P. Fu, A. U. Shaikh and P. E. Hartman, Nitro group orientation, reduction potential, and direct–acting mutagenicity of nitro–polycyclic aromatic hydrocarbons, Environ. Mol. Mutagen., 1991, 17, 169–180 CrossRef CAS PubMed.
- C. M. Cardona, W. Li, A. E. Kaifer, D. Stockdale and G. C. Bazan, Electrochemical Considerations for Determining Absolute Frontier Orbital Energy Levels of Conjugated Polymers for Solar Cell Applications, Adv. Mater., 2011, 23, 2367–2371 CrossRef CAS PubMed.
- W. L. Armarego and D. D. Perrin, Purification of laboratory chemicals, Butterworth-Heinemann, Oxford, 4th edn, 1997 Search PubMed.
- G. R. Fulmer, A. J. M. Miller, N. H. Sherden, H. E. Gottlieb, A. Nudelman, B. M. Stoltz, J. E. Bercaw and K. I. Goldberg, NMR Chemical Shifts of Trace Impurities: Common Laboratory Solvents, Organics, and Gases in Deuterated Solvents Relevant to the Organometallic Chemist, Organometallics, 2010, 29, 2176–2179 CrossRef CAS.
- S. Ando and T. Matsuura, Substituent shielding parameters of fluorine–19 NMR on polyfluoroaromatic compounds dissolved in dimethyl sulphoxide– d 6, Magn. Reson. Chem., 1995, 33, 639–645 CrossRef CAS.
- P. Mani, A. K. Singh and S. K. Awasthi, AgNO3 catalyzed synthesis of 5-substituted-1H-tetrazole via [3 + 2] cycloaddition of nitriles and sodium azide. Dedicated to the memory of late Dr. Tarkeshwar Gupta, Tetrahedron Lett., 2014, 55, 1879–1882 CrossRef CAS.
- I. V. Bliznets, A. A. Vasil'ev, S. V. Shorshnev, A. E. Stepanov and S. M. Lukyanov, Microwave-assisted synthesis of sterically hindered 3-(5-tetrazolyl) pyridines, Tetrahedron Lett., 2004, 45, 2571–2573 CrossRef CAS.
- B. Akhlaghinia and S. Rezazadeh, A Novel Approach for the Synthesis of 5-Substituted-1, J. Braz. Chem. Soc., 2012, 23, 2197–2203 CrossRef CAS.
- B. Du, X. Jiang and P. Sun, Palladium-Catalyzed Highly Selective ortho -Halogenation (I, Br, Cl) of Arylnitriles via sp 2 C–H Bond Activation Using Cyano as Directing Group, J. Org. Chem., 2013, 78, 2786–2791 CrossRef CAS PubMed.
- G. M. Sheldrick, A short history of SHELX, Acta Crystallogr., Sect. A: Found. Crystallogr., 2008, 64, 112–122 CrossRef CAS PubMed.
- (a) CCDC 2514222: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2qd7yb; (b) CCDC 2514223: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2qd7zc; (c) CCDC 2514224: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2qd80f; (d) CCDC 2514225: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2qd81g; (e) CCDC 2514226: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2qd82h.
| This journal is © The Royal Society of Chemistry 2026 |