Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Palladium-catalyzed 1,4-migration for the regioselective C–H bond functionalization at C2-position of 3-arylthiophenes
Bo
Lan
,
Norman
Le Floch
and
Henri
Doucet
 *
*
Univ Rennes, ISCR-UMR 6226, F-35000 Rennes, France. E-mail: henri.doucet@univ-rennes.fr
First published on 6th March 2026
Abstract
The regioselective functionalization of the C–H bond at the C2-position of 3-substituted thiophenes is challenging, as both thienyl α-positions may be reactive, generally affording mixtures of C2-, C5- and C2,C5-(di)functionalized thiophenes. We established that using palladium 1,4-migration allows for the regioselective functionalization of only one of the two α-positions of 3-arylthiophenes. The oxidative addition of the 3-(2-bromoaryl)thiophenes to palladium followed by such palladium migration, regioselectively activates the thienyl C2-α-position. Next, C2-heteroarylated 3-arylthiophene derivatives can be obtained through palladium-catalyzed direct coupling with heteroarenes. The new C–C bond that this reaction generates comes from the functionalization of two C–H bonds. This thienyl heteroarylation method tolerates a variety of heteroarenes and several substituents on the 3-arylthiophene. In addition, an easily available air-stable catalyst and an inexpensive base were employed for this reaction.
Introduction
Thiophene derivatives bearing a heteroaryl substituent at the C2 position and an aryl group at the C3 position exhibit properties that make them highly attractive for applications in pharmaceutical chemistry,1–3 including as antitumor agents, as well as in organic electronics, such as solar cells.4 However, the preparation of such compounds remains a challenging process and necessitates a number of synthetic steps. They are currently generally prepared via palladium-catalyzed Suzuki and Stille cross-coupling reactions, typically from 3-aryl-2-halothiophenes.5–11 Consequently, the identification of more straightforward methods for their preparation is a significant area of current research.Catalytic reactions that functionalize specific C–H bonds of organic molecules often result in simple synthetic methods.12–20 For instance, the Pd-catalyzed C–H bond activation/functionalization of heteroarenes, reported by Ohta et al. in 1990,21,22 has become one of the most reliable and cost-effective methods for preparing heteroarylated arenes, including thiophenes.23–26 However, in several cases, due to the similar reactivity of two C–H bonds on some substrates, such Pd-catalyzed C–H bond functionalization led to the formation of mixtures of products.27 For example, the reaction of 3-phenylthiophene with 4-bromotoluene produced a mixture of C2-, C5-, and C2,C5-(di)arylated thiophenes in a 9![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 9
9![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 70 ratio, even when an equimolar amount of reactants was used (Scheme 1b).28 Consequently, the regioselective C2-(hetero)arylation of such 3-arylthiophenes via a C–H bond functionalization remains challenging. This is why synthetic chemists currently rely on more traditional palladium-catalyzed reactions, such as the Suzuki coupling, to synthesize C2-(hetero)arylated 3-arylthiophene derivatives.6–11
70 ratio, even when an equimolar amount of reactants was used (Scheme 1b).28 Consequently, the regioselective C2-(hetero)arylation of such 3-arylthiophenes via a C–H bond functionalization remains challenging. This is why synthetic chemists currently rely on more traditional palladium-catalyzed reactions, such as the Suzuki coupling, to synthesize C2-(hetero)arylated 3-arylthiophene derivatives.6–11
 | ||
| Scheme 1 Pd-catalyzed C2-(hetero)arylations of 3-arylthiophenes; direct arylation vs. classical cross-couplings. | ||
We recently reported that, during the palladium-catalyzed heteroarylation of 2-(2-bromoaryl)thiophenes with heteroarenes, partial palladium 1,4-migration29–33 occurred in some cases.34 The predominant formation of 2-arylthiophenes heteroarylated at the β-position of thiophene, such as 2′-aryl-2,3′-bithiophenes, was the result of this Pd-migration, providing a simple method to functionalize such thienyl β-C–H bonds. To the best of our knowledge, there has been no report of the C–H bond activation of the thienyl α-position via such Pd 1,4-migration. Using Pd 1,4-migration with 3-(2-bromoaryl)thiophenes would enable the regioselective preparation of 2-heteroaryl-3-arylthiophenes in only two steps. This Pd 1,4-migration method is different from the more traditional Pd-catalyzed direct arylation in that it should only activate the thienyl C2-position, not its C5-position (see Scheme 1). Therefore, we investigated the outcome of the reaction of the palladium-catalyzed coupling of 3-(2-bromoaryl)thiophenes with heteroarenes. Herein, we report on (1) the regioselectivity of the Pd-catalyzed direct arylations of 3-(2-bromoaryl)thiophenes, (2) the scope of the reaction using various heteroarenes and 3-(2-bromoaryl)thiophene derivatives (Scheme 1, bottom).
Results and discussion
First, we synthesized a series of 3-(2-bromoaryl)thiophenes via palladium-catalyzed Suzuki (Scheme 2). The expected products 1a–1p were obtained in good yields.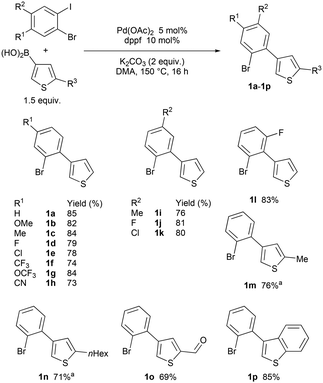 | ||
Scheme 2 Preparation of the 3-(2-bromoaryl)thiophenes 1a–1p. a![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) For procedure, see ref. 36. For procedure, see ref. 36. | ||
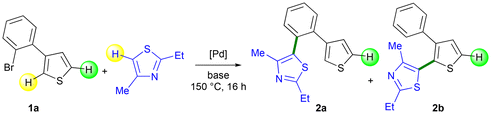
Next, we determined the selectivity of the palladium-catalyzed coupling reaction between 3-(2-bromophenyl)thiophene 1a and 2-ethyl-4-methylthiazole (Table 1).38–40 Based on our previously reported reaction conditions,34 using a 5 mol% Pd(OAc)2 catalyst and KOPiv as the base in DMA, the desired product 2b, arising from Pd-catalyzed 1,4-migration associated with direct arylation, was obtained in 78% selectivity (Table 1, entry 1). In contrast, product 2a, which arose from the non-migrated palladium intermediate, was obtained in 22% selectivity. The conversion of 1a was only 63% using this phosphine ligand-free Pd(OAc)2 catalyst. Conversely, complete conversion of 1a was observed in the presence of the more stable PdCl(C3H5)(dppb)37 catalyst (Table 1, entry 2). Furthermore, the ratio of products 2a and 2b increased to 5![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 95, and desired product 2b was isolated with a yield of 77%. Using acetate bases KOAc and NaOAc gave the product 2b with slightly lower selectivity and yield than when KOPiv base was employed (Table 1, entries 3 and 4). The Na2CO3 base also gave a good selectivity in the desired product 2b, but a very low conversion of 1a was observed (Table 1, entry 5). In contrast, the other carbonate bases, Cs2CO3 and K2CO3, led to poor selectivities in 2a and 2b products (Table 1, entries 6 and 7). With these two bases, the product 2a, resulting from the non-migrated palladium intermediate, was predominant with 60%–65% selectivity. The influence of a few solvents on the reaction outcome was also examined. Reactions performed in DMF and NMP gave the desired product 2b resulting from Pd 1,4-migration, albeit with slightly lower selectivity than in DMA (Table 1, entries 8 and 9). Conversely, the less polar solvents, xylene and diethyl carbonate (DEC) were ineffective (Table 1, entries 10 and 11). Finally, reducing the reaction temperature to 130 °C (from 150 °C) produced 2b in 70% yield due to a slightly lower selectivity of the reaction (Table 1, entry 12).
95, and desired product 2b was isolated with a yield of 77%. Using acetate bases KOAc and NaOAc gave the product 2b with slightly lower selectivity and yield than when KOPiv base was employed (Table 1, entries 3 and 4). The Na2CO3 base also gave a good selectivity in the desired product 2b, but a very low conversion of 1a was observed (Table 1, entry 5). In contrast, the other carbonate bases, Cs2CO3 and K2CO3, led to poor selectivities in 2a and 2b products (Table 1, entries 6 and 7). With these two bases, the product 2a, resulting from the non-migrated palladium intermediate, was predominant with 60%–65% selectivity. The influence of a few solvents on the reaction outcome was also examined. Reactions performed in DMF and NMP gave the desired product 2b resulting from Pd 1,4-migration, albeit with slightly lower selectivity than in DMA (Table 1, entries 8 and 9). Conversely, the less polar solvents, xylene and diethyl carbonate (DEC) were ineffective (Table 1, entries 10 and 11). Finally, reducing the reaction temperature to 130 °C (from 150 °C) produced 2b in 70% yield due to a slightly lower selectivity of the reaction (Table 1, entry 12).
| Entry | Catalyst | Solvent | Base | Conv. (%) | Ratio 2a![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2b 2b |
Yield in 2b (%) |
|---|---|---|---|---|---|---|
a [Pd] (0.05 equiv.), 2-ethyl-4-methylthiazole (2 equiv.), 3-(2-bromophenyl)thiophene 1a (1 equiv.), base (2 equiv.), 150 °C, 16 h, 2a![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2b ratios determined by 1H NMR and GC/MS analysis of the crude mixtures, isolated yields.
b 130 °C. 2b ratios determined by 1H NMR and GC/MS analysis of the crude mixtures, isolated yields.
b 130 °C.
|
||||||
| 1 | Pd(OAc)2 | DMA | KOPiv | 63 | 22![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 78 78 |
— |
| 2 | PdCl(C3H5)(dppb) | DMA | KOPiv | 100 | 5![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 95 95 |
77 |
| 3 | PdCl(C3H5)(dppb) | DMA | KOAc | 99 | 6![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 94 94 |
75 |
| 4 | PdCl(C3H5)(dppb) | DMA | CsOAc | 100 | 11![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 89 89 |
71 |
| 5 | PdCl(C3H5)(dppb) | DMA | Na2CO3 | 20 | 8![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 92 92 |
<5 |
| 6 | PdCl(C3H5)(dppb) | DMA | K2CO3 | 100 | 60![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 40 40 |
— |
| 7 | PdCl(C3H5)(dppb) | DMA | Cs2CO3 | 100 | 65![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 35 35 |
41 of 2a |
| 8 | PdCl(C3H5)(dppb) | DMF | KOPiv | 100 | 9![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 91 91 |
75 |
| 9 | PdCl(C3H5)(dppb) | NMP | KOPiv | 100 | 13![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 87 87 |
70 |
| 10 | PdCl(C3H5)(dppb) | Xylene | KOPiv | 100 | Not determined | <10 |
| 11 | PdCl(C3H5)(dppb) | DEC | KOPiv | 100 | Not determined | <10 |
| 12 | PdCl(C3H5)(dppb) | DMA | KOPiv | 100 | 11![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 89 89 |
70b |
Then, we investigated the influence of the substituents on the aryl group of the 3-(2-bromoaryl)thiophene in this reaction, using 2-ethyl-4-methylthiazole as the reaction partner (Scheme 3). Our study began with the examination of the influence of a set of 3-(2-bromoaryl)thiophene compounds with various substituents in the meta-position to the C–Br bond on the selectivity. The presence of a methyl substituent slightly increased the selectivity in the desired product 4b to 96%. Conversely, in the presence of fluorine and chlorine substituents, we obtained lower selectivities of 80% and 73%, respectively, for isomers 5b and 6b. These results suggest that the presence of an electron-withdrawing substituent on the aryl unit is less favourable to the palladium 1,4-migration. This trend was confirmed using a 3-arylthiophenes with CF3 or OCF3 substituents that were also in the meta-position relative to the C–Br bond, which gave isomers 7b and 8b with 79% and 76% selectivity, respectively. A nitrile substituent, which is strongly electron-withdrawing, on the aryl unit, was well tolerated, affording product 9b in 80% selectivity. Then, reactions involving 3-(2-bromoaryl)thiophene with substituents on the aryl in the para-position relative to the C–Br bond were performed. Methyl and fluoro substituents (Hammett σp constants of −0.17 and 0.06) furnished the target products 10b and 11b in 98% and 96% selectivities, respectively. A chloro para-substituent was also well tolerated and produced compound 12b in 93% selectivity. Therefore, the selectivity of this reaction appears to be influenced by the Hammett constants (σm or σp depending on the position of the aryl substituent relative to the C–Br bond), since low Hammett constants tend to favour Pd 1,4-migration in all cases (Scheme 4). However, kinetic studies would be required to gain more insight into the factors governing the Pd 1,4-migration process. In the presence of 3-(2-bromo-6-fluorophenyl)thiophene 1l, we also obtained a good selectivity of 87% in desired product 13b. It should be noted that reaction selectivity also depends on the heteroaryl coupling partner. For example, when we used 2-isobutylthiazole instead of 2-ethyl-4-methylthiazole, we observed a lower selectivity in the Pd-1,4-migration product, affording isomer 14b with only 77% selectivity.
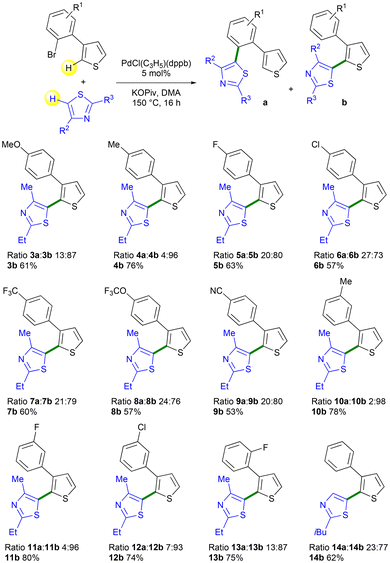 | ||
| Scheme 3 Pd-catalyzed direct C2-heteroarylations of the thienyl ring of a set of 3-(2-bromoaryl)thiophenes. | ||
 | ||
Scheme 4 Influence of the substituents on the aryl group of the 3-(2-bromoaryl)thiophenes: correlation of the a![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) b selectivity with meta Hammett constants. b selectivity with meta Hammett constants. | ||
The impact of the thienyl substituents of 3-(2-bromoaryl)thiophenes on the Pd 1,4-migration process was also examined (Scheme 5). The presence of a methyl substituent at the C5 position of the 3-arylthiophene had almost no effect on the selectivity or yield of the reaction. The desired products, 15b and 16b, were obtained with 94% and 75% selectivity, respectively, using 2-ethyl-4-methylthiazole and 2-isobutylthiazole. As expected, the reaction of a 5-hexyl-3-arylthiazole derivative with 2-ethyl-4-methylthiazole produced isomer 17b with a similar selectivity of 95%. A benzothiophene unit was also well tolerated. In the presence of 3-(2-bromophenyl)benzothiophene 1p, both 2-ethyl-4-methylthiazole and 2-isobutylthiazole gave the desired products 18b and 19b with very high selectivities of 99% and 98%, respectively. The presence of a formyl C2-substituent on the thienyl ring was also tolerated. Using 4-(2-bromophenyl)thiophene-2-carbaldehyde 1o and 2-ethyl-4-methylthiazole as the coupling partner, the target product 20b arising from Pd 1,4-migration was also obtained with a very high 99% selectivity and in 77% yield.
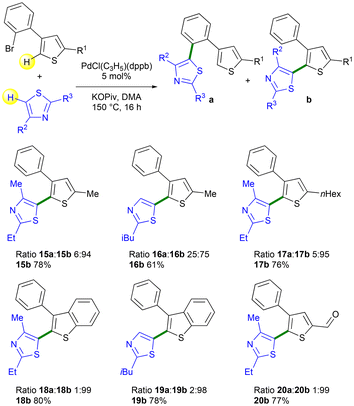 | ||
| Scheme 5 Pd-catalyzed direct C5-heteroarylations of the thienyl ring of 4-(2-bromophenyl)thiophene derivatives. | ||
Then, the heteroarylation of 3-(2-bromophenyl)thiophenes 1j–1m, using a variety of heteroarenes was explored (Scheme 6). Similar selectivity to that observed with 2-isobutylthiazole was achieved using 2-acetylthiazole, affording product 21b with 81% selectivity and 68% yield. Regioselectivities of 98% and 94% were obtained for isomers 22b and 23b, respectively, when 2-hexylthiophene was used with 1j and 1m as the reaction partners. With 2-chlorothiophene, the target product 24b was obtained with a lower selectivity of 68%. Notably, no cleavage of the thienyl C–Cl bond was observed in the course of this reaction, which allowed for subsequent transformations. The product 25b was obtained in 96% selectivity from 2-methyl-2-(thien-2-yl)-1,3-dioxolane and 4-(2-bromophenyl)-2-methylthiophene 1m. Using 2-butylfuran, products 26b and 27b were obtained with selectivities of 97%, but in quite low yield due to the formation of unidentified side products. Very high selectivity in isomer 29b was obtained when 1-methylpyrrole was used; however, unidentified side products were formed again. In the presence of 1l and 1m, imidazo[1,2-a]pyrazine afforded the target Pd 1,4-migration products 30b and 31b with 98% and 95% selectivity, respectively. Finally, a selectivity of 91% in product 32b was obtained using imidazo[1,2-a]pyrazine.
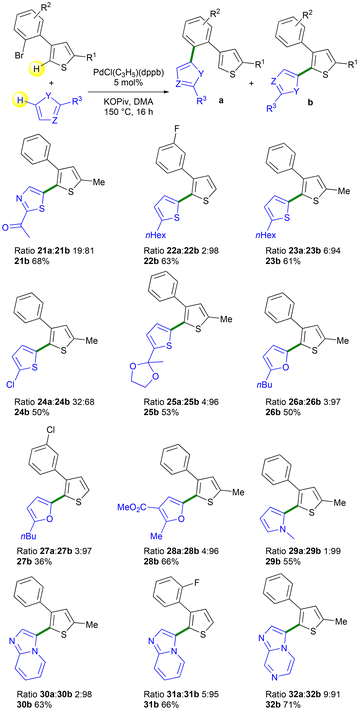 | ||
| Scheme 6 Pd-catalyzed direct C2- or C5-heteroarylations of the thienyl ring of 3-(2-bromoaryl)thiophenes 1j–1m using a set of heteroarenes. | ||
The synthesis of a 2,5-diheteroaryl-3-arylthiophene from the 5-(thien-2-yl)thiazole derivative 4b, prepared in Scheme 3, via Pd-catalyzed direct arylation, was also investigated (Scheme 7). We obtained the desired 2-heteroaryl-3,5-diarylthiophene derivative 33 in an 86% yield from 4b, 3-bromopyridine, 5 mol% PdCl(C3H5)(dppb) catalyst, and KOPiv base. This result demonstrates that our method allows for the introduction of two different heteroaryl substituents at positions C2 and C5 of the thienyl unit.
 | ||
| Scheme 7 Preparation of a tri(hetero)arylated thiophene derivative via successive Pd-catalyzed direct (hetero)arylations. | ||
To further illustrate the advantage of our method for synthesizing 2-heteroaryl-3-arylthiophenes, we tried to synthesize one of them using palladium-catalyzed coupling of 3-phenylthiophene and a 2-bromothiophene (Scheme 8). We found that preparing 2-thienyl-substituted 3-phenylthiophene 34b from 3-phenylthiophene and 1-(5-bromothien-2-yl)ethan-1-one was very challenging. A low conversion of 3-phenylthiophene was observed in this reaction. Moreover, we obtained a mixture between the C2 and C5 arylated 3-phenylthiophene in a 47![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 53 ratio, affording the desired isomer 34b in only 8% isolated yield (Scheme 8, top). In addition, it should be noted that the formation of a large amount of the side product 1,1′-([2,2′-bithiophene]-5,5′-diyl)bis(ethan-1-one) was also observed. In contrast, our method produced 35b in 96% selectivity and 56% yield from 3-(2-bromophenyl)thiophene 1a and 2-methyl-2-(thien-2-yl)-1,3-dioxolane (Scheme 8, bottom). Product 35b could be easily deprotected under acidic conditions into product 34b in 86% yield.
53 ratio, affording the desired isomer 34b in only 8% isolated yield (Scheme 8, top). In addition, it should be noted that the formation of a large amount of the side product 1,1′-([2,2′-bithiophene]-5,5′-diyl)bis(ethan-1-one) was also observed. In contrast, our method produced 35b in 96% selectivity and 56% yield from 3-(2-bromophenyl)thiophene 1a and 2-methyl-2-(thien-2-yl)-1,3-dioxolane (Scheme 8, bottom). Product 35b could be easily deprotected under acidic conditions into product 34b in 86% yield.
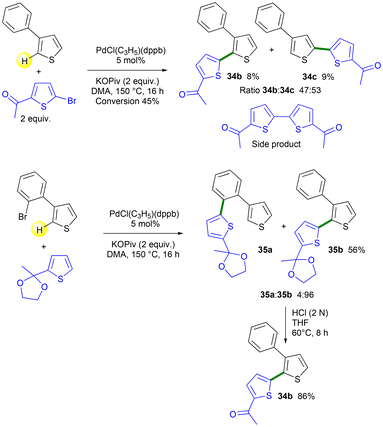 | ||
| Scheme 8 Use of 3-phenylthiophene vs. 3-(2-bromophenyl)thiophene 1a in Pd-catalyzed direct C2-heteroarylation. | ||
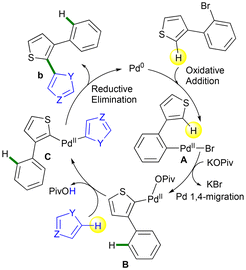
Mechanism for the access to b products likely proceed via a Pd-1,4-migration29–31 followed by a direct arylation41–44 as described in the Scheme 9. The first step of the catalytic cycle certainly involves the oxidative addition of the 3-(2-bromoaryl)thiophene to palladium to give the intermediate A. Then, a Br/OPiv ligand exchange and a Pd-1,4-migration29–31 occurs to give the intermediate B. From B, a concerted metallation deprotonation (CMD)41–44 of the heteroarene coupling partner, gives the intermediate C. Finally, reductive elimination regenerates a Pd(0) species and produces the C2-heteroarylated 3-arylthiophene derivative b. In the absence of computational studies, we assume that the reaction is driven by the irreversible formation of the C–C bond.
Conclusions
In summary, Pd 1,4-migration associated with Pd-catalyzed direct arylation allowed for the synthesis of a variety of C2-heteroarylated 3-arylthiophenes in only two steps from commercially available compounds. This method enables the regioselective C2-heteroarylation of the thienyl ring, as the bromo substituent on the 3-arylthiophene behaves as a traceless directing group; whereas the thienyl C5-position remained untouched. This coupling reaction creates a C–C bond between two heteroarenes through the functionalization of two C–H bonds. This reaction is compatible with a variety of heteroarenes, as well as several substituents on the aryl unit of 3-(2-bromoaryl)thiophene. Furthermore, these reactions use an air-stable palladium catalyst associated with an inexpensive base. Therefore, this method is undoubtedly one of the most efficient synthetic pathways for producing C2-heteroarylated 3-arylthiophenes.Experimental
General
[PdCl(C3H5)]2 (98%) was purchased from Aldrich. DMA (99+%) extra pure was purchased from ACROS. Dppb (1,4-bis(diphenylphosphino)butane) (98%), KOPiv (95+%), thien-3-ylboronic acids, 1,2-dihalobenzenes and 3-phenylthiophene were purchased from Fluorochem. These compounds were not purified before use. All reagents were weighed and handled in air. All reactions were carried out under an inert atmosphere with standard Schlenk techniques. 1H, 19F and 13C NMR spectra were recorded on a Bruker Avance III 400 MHz spectrometer. High-resolution mass spectra were measured on a Thermo Fisher Scientific Q-Exactive spectrometer. Melting points were determined with a Kofler hot bench system. 3-(2-Bromophenyl)thiophene351a and 4-(2-bromophenyl)-2-methylthiophene361m were prepared using reported procedures.Preparation of the PdCl(C3H5)(dppb) catalyst37
An oven-dried 40 mL Schlenk tube equipped with a magnetic stirring bar under argon atmosphere, was charged with [Pd(C3H5)Cl]2 (182 mg, 0.5 mmol) and dppb (426 mg, 1 mmol). 10 mL of anhydrous dichloromethane were added, then, the solution was stirred at room temperature for twenty minutes. The solvent was removed in vacuum. The yellow powder was used without purification. 31P NMR (162 MHz, CDCl3) δ 19.3 (s).General procedure for the preparation of 3-(2-bromophenyl)thiophenes 1b–1l, 1o and 1p
As a typical experiment, a mixture of 1,2-dihalobenzene derivative (3 mmol), thien-3-ylboronic acid derivative (4.5 mmol), K2CO3 (0.828 g, 6 mmol), Pd(OAc)2 (33.6 mg, 0.15 mmol) and dppf (166.2 mg, 0.30 mmol) in DMA (20 mL) was stirred at 150 °C for 16 h. After being allowed to cool to room temperature, the resulting mixture was extracted with Et2O (3 × 10 mL). The combined organic phase was dried over MgSO4, filtrated, and concentrated under reduced pressure. The residue was purified by column chromatography on silica gel to give the 3-(2-bromoaryl)thiophenes.1H NMR (400 MHz, CDCl3) δ 7.40–7.36 (m, 2H), 7.34 (d, J = 8.5 Hz, 1H), 7.31 (d, J = 3.3 Hz, 1H), 7.26 (d, J = 2.6 Hz, 1H), 6.93 (dd, J = 8.6, 2.6 Hz, 1H), 3.86 (s, 3H).
13C NMR (101 MHz, CDCl3) δ 159.3, 140.9, 131.7, 130.0, 129.1, 124.7, 123.5, 122.9, 118.5, 113.6, 55.6.
HRMS calcd for [M + H]+ C11H10BrOS 268.9630, found: 268.9630.
1H NMR (400 MHz, CDCl3) δ 7.53 (s, 1H), 7.41 (dd, J = 3.1, 1.3 Hz, 1H), 7.38 (dd, J = 4.9, 3.0 Hz, 1H), 7.35–7.27 (m, 2H), 7.17 (d, J = 7.8 Hz, 1H), 2.39 (s, 3H).
13C NMR (101 MHz, CDCl3) δ 141.1, 138.9, 134.6, 133.8, 131.0, 129.0, 128.2, 124.7, 123.7, 122.3, 20.7.
HRMS calcd for [M + H]+ C11H10BrS 252.9681, found: 252.9681.
1H NMR (400 MHz, CDCl3) δ 7.44 (dd, J = 8.3, 2.6 Hz, 1H), 7.42–7.38 (m, 3H), 7.28 (dd, J = 3.5, 3.0 Hz, 1H), 7.09 (ddd, J = 8.5, 7.9, 2.7 Hz, 1H).
19F NMR (376 MHz, CDCl3) δ −113.2.
13C NMR (101 MHz, CDCl3) δ 161.6 (d, J = 250.8 Hz), 140.1, 133.8 (d, J = 3.7 Hz), 132.1 (d, J = 8.4 Hz), 128.9, 125.0, 124.1, 122.7 (d, J = 9.6 Hz), 120.5 (d, J = 24.3 Hz), 114.6 (d, J = 21.0 Hz).
HRMS calcd for [M + H]+ C10H7BrFS 256.9430, found: 256.9429.
1H NMR (400 MHz, CDCl3) δ 7.70 (t, J = 1.2 Hz, 1H), 7.42 (dd, J = 3.0, 1.4 Hz, 1H), 7.41–7.38 (m, 1H), 7.35–7.32 (m, 2H), 7.30–7.25 (m, 1H).
13C NMR (101 MHz, CDCl3) δ 140.0, 136.1, 133.7, 132.9, 131.9, 128.7, 127.6, 125.1, 124.3, 122.9.
HRMS calcd for [M + H]+ C10H7BrClS 272.9135, found: 272.9135.
1H NMR (400 MHz, CDCl3) δ 7.96 (s, 1H), 7.62 (d, J = 8.2 Hz, 1H), 7.53 (d, J = 8.2 Hz, 1H), 7.50 (dd, J = 3.0, 1.3 Hz, 1H), 7.44 (dd, J = 5.0, 3.0 Hz, 1H), 7.33 (dd, J = 5.0, 1.3 Hz, 1H).
19F NMR (376 MHz, CDCl3) δ −62.6.
13C NMR (101 MHz, CDCl3) δ 141.1, 139.8, 131.5, 130.8 (q, J = 33.2 Hz), 130.4 (q, J = 3.9 Hz), 128.5, 125.4, 124.9, 124.2 (q, J = 3.6 Hz), 122.7, 120.3 (d, J = 272.5 Hz).
HRMS calcd for [M]+ C11H6BrF3S 305.9320, found: 305.9322.
1H NMR (400 MHz, CDCl3) (δ 7.62 s, 1H), 7.49–7.40 (m, 3H), 7.32 (dd, J = 5.0, 1.4 Hz, 1H), 7.27 (d, J = 7.4 Hz, 1H).
19F NMR (376 MHz, CDCl3) δ −57.9.
13C NMR (101 MHz, CDCl3) δ 148.3 (q, J = 2.0 Hz), 139.8, 136.5, 131.9, 128.7, 125.9, 125.2, 124.5, 122.9, 120.4 (d, J = 258.4 Hz), 119.9.
HRMS calcd for [M]+ C11H6BrF3OS 321.9269, found: 321.9271.
1H NMR (400 MHz, CDCl3) δ 7.94 (d, J = 1.7 Hz, 1H), 7.62 (dd, J = 8.0, 1.7 Hz, 1H), 7.53 (dd, J = 3.0, 1.4 Hz, 1H), 7.50 (d, J = 8.0 Hz, 1H), 7.42 (dd, J = 5.0, 3.0 Hz, 1H), 7.32 (dd, J = 5.0, 1.4 Hz, 1H).
13C NMR (101 MHz, CDCl3) δ 142.2, 139.3, 136.7, 131.7, 130.9, 128.4, 125.8, 125.6, 122.8, 117.4, 112.4.
HRMS calcd for [M + H]+ C11H7BrNS 263.9477, found: 263.9477.
1H NMR (400 MHz, CDCl3) δ 7.57 (d, J = 8.1 Hz, 1H), 7.44 (dd, J = 3.0, 1.3 Hz, 1H), 7.40 (dd, J = 5.0, 3.0 Hz, 1H), 7.33 (dd, J = 5.0, 1.3 Hz, 1H), 7.26 (s, 1H), 7.04 (ddd, J = 8.1, 2.2, 0.8 Hz, 1H), 2.38 (s, 3H).
13C NMR (101 MHz, CDCl3) δ 141.3, 137.3, 137.2, 133.1, 132.1, 129.6, 129.0, 124.7, 123.9, 119.2, 20.9.
HRMS calcd for [M + H]+ C11H10BrS 252.9681, found: 252.9681.
1H NMR (400 MHz, CDCl3) δ 7.63 (dd, J = 8.8, 5.4 Hz, 1H), 7.47 (dd, J = 3.0, 1.4 Hz, 1H), 7.41 (dd, J = 5.0, 3.0 Hz, 1H), 7.31 (dd, J = 4.9, 1.3 Hz, 1H), 7.15 (dd, J = 9.3, 3.1 Hz, 1H), 6.94 (ddd, J = 8.8, 7.8, 3.1 Hz, 1H).
19F NMR (376 MHz, CDCl3) δ −115.1.
13C NMR (101 MHz, CDCl3) δ 161.8 (d, J = 247.3 Hz), 140.1 (d, J = 1.5 Hz), 139.2 (d, J = 8.1 Hz), 134.6 (d, J = 8.1 Hz), 128.6, 125.2, 124.6, 118.2 (d, J = 23.0 Hz), 116.7 (d, J = 3.2 Hz), 115.8 (d, J = 22.3 Hz).
HRMS calcd for [M]+ C10H6BrFS 255.9352, found: 255.9353.
1H NMR (400 MHz, CDCl3) δ 7.60 (d, J = 8.5 Hz, 1H), 7.46 (dd, J = 3.0, 1.3 Hz, 1H), 7.43–7.37 (m, 2H), 7.30 (dd, J = 5.0, 1.3 Hz, 1H), 7.18 (dd, J = 8.5, 2.6 Hz, 1H).
13C NMR (101 MHz, CDCl3) δ 139.9, 139.0, 134.4, 133.3, 131.1, 128.7, 128.6, 125.2, 124.6, 120.5.
HRMS calcd for [M + H]+ C10H7BrClS 272.9135, found: 272.9135.
1H NMR (400 MHz, CDCl3) δ 7.52 (d, J = 7.9 Hz, 1H), 7.48–7.42 (m, 2H), 7.27–7.11 (m, 3H).
19F NMR (376 MHz, CDCl3) δ −109.0.
13C NMR (101 MHz, CDCl3) δ 160.4 (d, J = 249.9 Hz), 133.4, 129.6 (d, J = 9.1 Hz), 129.2 (d, J = 1.5 Hz), 128.8 (d, J = 3.5 Hz), 126.3 (d, J = 18.1 Hz), 125.9 (d, J = 1.9 Hz), 124.8, 124.5 (d, J = 2.8 Hz), 114.9 (d, J = 23.5 Hz).
HRMS calcd for [M + H]+ C10H7BrFS 256.9430, found: 256.9429.
1H NMR (400 MHz, CDCl3) δ 7.66 (dd, J = 8.0, 1.3 Hz, 1H), 7.41 (dd, J = 7.7, 1.8 Hz, 1H), 7.34 (td, J = 7.5, 1.3 Hz, 1H), 7.19 (d, J = 1.5 Hz, 1H), 7.17 (td, J = 7.5, 1.7 Hz, 1H), 6.99 (s, 1H), 2.87 (t, J = 7.6 Hz, 2H), 1.75 (quint., J = 7.6 Hz, 2H), 1.50–1.21 (m, 6H), 0.95 (t, J = 7.6 Hz, 3H).
13C NMR (101 MHz, CDCl3) δ 145.2, 140.6, 138.0, 133.3, 131.2, 128.5, 127.3, 125.9, 122.5, 121.5, 31.6, 30.1, 28.8, 22.6, 14.1.
HRMS calcd for [M + H]+ C16H20BrS 323.0464, found: 323.0464.
1H NMR (300 MHz, CDCl3) δ 10.00 (d, J = 1.3 Hz, 1H), 7.97 (d, J = 1.5 Hz, 1H), 7.83 (t, J = 1.4 Hz, 1H), 7.71 (d, J = 7.8 Hz, 1H), 7.47–7.36 (m, 2H), 7.30–7.22 (m, 1H).
13C NMR (75 MHz, CDCl3) δ 182.9, 143.4, 142.3, 137.5, 135.9, 133.5, 133.4, 131.0, 129.5, 127.7, 122.4.
HRMS calcd for [M + H]+ C11H8BrOS 266.9474, found: 266.9474.
1H NMR (400 MHz, CDCl3) δ 8.01 (d, J = 7.6 Hz, 1H), 7.84 (d, J = 7.6 Hz, 1H), 7.68–7.59 (m, 1H), 7.55–7.42 (m, 5H), 7.36 (ddd, J = 8.0, 6.5, 2.6 Hz, 1H).
13C NMR (101 MHz, CDCl3) δ 139.9, 138.5, 136.9, 136.7, 133.3, 132.1, 129.5, 127.4, 125.5, 124.6, 124.4, 124.2, 123.4, 122.9.
LRMS calcd for [M]+ C14H9BrS 290, found: 290.
General procedure for the preparation of products 2b–35b
As a typical experiment, the reaction of the 3-(2-bromoaryl)thiophene derivative 1a–1p (1 mmol), heteroarene (2 mmol) and KOPiv (0.280 g, 2 mmol) at 150 °C during 16 h in DMA (4 mL) in the presence of PdCl(C3H5)(dppb) (30.5 mg, 0.05 mmol) under argon affords the coupling product after evaporation of the solvent and purification by column chromatography on silica gel. The a![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) b ratios were determined by 1H NMR and GC/MS analysis of the crude mixtures.
b ratios were determined by 1H NMR and GC/MS analysis of the crude mixtures.
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 95 ratio and 2b was isolated in 77% (0.219 g) yield as a yellow oil.
95 ratio and 2b was isolated in 77% (0.219 g) yield as a yellow oil.
1H NMR (400 MHz, CDCl3) δ 7.43 (d, J = 5.3 Hz, 1H), 7.37–7.25 (m, 5H), 7.23 (d, J = 5.3 Hz, 1H), 2.98 (q, J = 7.6 Hz, 2H), 2.08 (s, 3H), 1.38 (t, J = 7.6 Hz, 3H).
13C NMR (101 MHz, CDCl3) δ 172.1, 150.1, 141.4, 136.0, 129.5, 128.5, 128.4, 127.5, 127.1, 126.1, 122.7, 27.0, 15.7, 14.1.
HRMS calcd for [M + H]+ C16H16NS2 286.0719, found: 286.0717.
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 35 ratio and 2a was isolated in 41% (0.117 g) yield as a yellow oil.
35 ratio and 2a was isolated in 41% (0.117 g) yield as a yellow oil.
1H NMR (400 MHz, CDCl3) δ 7.53 (d, J = 8.0 Hz, 1H), 7.47–7.34 (m, 3H), 7.21 (dd, J = 5.0, 3.0 Hz, 1H), 7.10 (dd, J = 2.9, 1.3 Hz, 1H), 6.86 (dd, J = 5.0, 1.3 Hz, 1H), 2.99 (q, J = 7.6 Hz, 2H), 2.02 (s, 3H), 1.38 (t, J = 7.6 Hz, 3H).
13C NMR (101 MHz, CDCl3) δ 171.2, 148.3, 141.3, 137.2, 132.1, 130.3, 129.9, 129.7, 128.6, 128.2, 127.2, 125.0, 123.0, 26.9, 15.3, 14.3.
HRMS calcd for [M + H]+ C16H16NS2 286.0719, found: 286.0719.
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 87 ratio and 3b was isolated in 61% (0.192 g) yield as a yellow oil.
87 ratio and 3b was isolated in 61% (0.192 g) yield as a yellow oil.
1H NMR (400 MHz, CDCl3) δ 7.41 (d, J = 5.2 Hz, 1H), 7.23 (d, J = 8.8 Hz, 2H), 7.19 (d, J = 5.3 Hz, 1H), 6.86 (d, J = 8.8 Hz, 2H), 3.82 (s, 3H), 2.98 (q, J = 7.6 Hz, 2H), 2.11 (s, 3H), 1.39 (t, J = 7.6 Hz, 3H).
13C NMR (101 MHz, CDCl3) δ 172.0, 158.7, 150.1, 141.0, 129.5, 129.4, 128.5, 126.5, 126.0, 122.9, 114.0, 55.2, 27.0, 15.7, 14.1.
HRMS calcd for [M + H]+ C17H18NOS2 316.0824, found: 316.0825.
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 96 ratio and 4b was isolated in 76% (0.227 g) yield as a yellow oil.
96 ratio and 4b was isolated in 76% (0.227 g) yield as a yellow oil.
1H NMR (400 MHz, CDCl3) δ 7.41 (d, J = 5.3 Hz, 1H), 7.27–7.16 (m, 3H), 7.12 (d, J = 8.4 Hz, 2H), 2.98 (q, J = 7.6 Hz, 2H), 2.36 (s, 3H), 2.09 (s, 3H), 1.39 (t, J = 7.6 Hz, 3H).
13C NMR (101 MHz, CDCl3) δ 172.0, 150.1, 141.4, 136.9, 133.0, 129.5, 129.2, 128.2, 127.0, 126.0, 122.8, 27.0, 21.2, 15.7, 14.1.
HRMS calcd for [M + H]+ C17H18NS2 300.0875, found: 300.0873.
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 80 ratio and 5b was isolated in 63% (0.191 g) yield as a yellow oil.
80 ratio and 5b was isolated in 63% (0.191 g) yield as a yellow oil.
1H NMR (400 MHz, CDCl3) δ 7.43 (d, J = 5.3 Hz, 1H), 7.26 (dd, J = 8.7, 5.4 Hz, 1H), 7.19 (d, J = 5.3 Hz, 1H), 7.01 (t, J = 8.7 Hz, 2H), 2.98 (q, J = 7.6 Hz, 2H), 2.10 (s, 3H), 1.38 (t, J = 7.6 Hz, 3H).
19F NMR (376 MHz, CDCl3) δ −114.8.
13C NMR (101 MHz, CDCl3) δ 172.3, 162.0 (d, J = 246.9 Hz), 150.2, 140.3, 132.0 (d, J = 3.4 Hz), 130.0 (d, J = 8.1 Hz), 129.3, 127.5, 126.3, 122.4, 115.5 (d, J = 21.4 Hz), 27.0, 15.7, 14.1.
HRMS calcd for [M + H]+ C16H15FNS2 304.0625, found: 304.0624.
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 73 ratio and 6b was isolated in 57% (0.182 g) yield as a yellow oil.
73 ratio and 6b was isolated in 57% (0.182 g) yield as a yellow oil.
1H NMR (400 MHz, CDCl3) δ 7.44 (d, J = 5.2 Hz, 1H), 7.29 (d, J = 8.5 Hz, 2H), 7.22 (d, J = 8.5 Hz, 2H), 7.19 (d, J = 5.2 Hz, 1H), 2.99 (q, J = 7.6 Hz, 2H), 2.10 (s, 3H), 1.39 (t, J = 7.6 Hz, 3H).
13C NMR (101 MHz, CDCl3) δ 172.4, 150.3, 140.1, 134.4, 133.1, 129.7, 129.1, 128.8, 127.9, 126.5, 122.3, 27.0, 15.7, 14.0.
HRMS calcd for [M + H]+ C16H15ClNS2 320.0329, found: 320.0329.
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 79 ratio and 7b was isolated in 60% (0.212 g) yield as a yellow solid: mp 116–118 °C.
79 ratio and 7b was isolated in 60% (0.212 g) yield as a yellow solid: mp 116–118 °C.
1H NMR (400 MHz, CDCl3) δ 7.58 (d, J = 8.5 Hz, 2H), 7.48 (d, J = 5.3 Hz, 1H), 7.41 (d, J = 8.5 Hz, 2H), 7.24 (d, J = 5.3 Hz, 1H), 3.00 (q, J = 7.6 Hz, 2H), 2.08 (s, 3H), 1.39 (t, J = 7.6 Hz, 3H).
19F NMR (376 MHz, CDCl3) δ −62.6.
13C NMR (101 MHz, CDCl3) δ 172.6, 150.4, 139.8, 139.5, 129.2 (q, J = 32.6 Hz), 129.1, 128.9, 128.6, 126.7, 125.5 (q, J = 3.9 Hz), 122.0, 121.2 (q, J = 272.0 Hz), 27.0, 15.7, 14.0.
HRMS calcd for [M + H]+ C17H15F3NS2 354.0593, found: 354.0591.
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 76 ratio and 8b was isolated in 57% (0.210 g) yield as a yellow oil.
76 ratio and 8b was isolated in 57% (0.210 g) yield as a yellow oil.
1H NMR (400 MHz, CDCl3) δ 7.45 (d, J = 5.2 Hz, 1H), 7.31 (d, J = 8.7 Hz, 2H), 7.20 (d, J = 5.3 Hz, 1H), 7.17 (d, J = 8.3 Hz, 2H), 2.99 (q, J = 7.6 Hz, 2H), 2.09 (s, 3H), 1.39 (t, J = 7.6 Hz, 3H).
19F NMR (376 MHz, CDCl3) δ −57.8.
13C NMR (101 MHz, CDCl3) δ 172.4, 150.3, 148.3 (q, J = 1.8 Hz), 139.8, 134.6, 129.7, 129.2, 128.1, 126.5, 122.2, 120.9, 120.5 (q, J = 257.3 Hz), 27.0, 15.7, 14.0.
HRMS calcd for [M + H]+ C17H15F3NOS 370.0542, found: 370.0542.
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 80 ratio and 9b was isolated in 53% (0.164 g) yield as a white solid: mp 118–120 °C.
80 ratio and 9b was isolated in 53% (0.164 g) yield as a white solid: mp 118–120 °C.
1H NMR (400 MHz, CDCl3) δ 7.61 (d, J = 8.5 Hz, 2H), 7.49 (d, J = 5.3 Hz, 1H), 7.40 (d, J = 8.6 Hz, 2H), 7.23 (d, J = 5.3 Hz, 1H), 2.99 (q, J = 7.6 Hz, 2H), 2.09 (s, 3H), 1.39 (t, J = 7.6 Hz, 3H).
13C NMR (101 MHz, CDCl3) δ 172.8, 150.5, 140.5, 139.3, 132.4, 129.5, 128.9, 128.8, 127.0, 121.7, 118.8, 110.8, 27.0, 15.7, 14.0.
HRMS calcd for [M + H]+ C17H15N2S2 311.0671, found: 311.0671.
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 98 ratio and 10b was isolated in 78% (0.233 g) yield as a yellow oil.
98 ratio and 10b was isolated in 78% (0.233 g) yield as a yellow oil.
1H NMR (400 MHz, CDCl3) δ 7.42 (d, J = 5.2 Hz, 1H), 7.21 (d, J = 5.3 Hz, 1H), 7.19 (t, J = 7.6 Hz, 1H), 7.13 (s, 1H), 7.11–7.02 (m, 2H), 2.98 (q, J = 7.6 Hz, 2H), 2.33 (s, 3H), 2.11 (s, 3H), 1.38 (t, J = 7.6 Hz, 3H).
13C NMR (101 MHz, CDCl3) δ 172.0, 150.1, 141.5, 138.0, 135.9, 129.5, 129.1, 128.4, 127.9, 127.3, 126.0, 125.5, 122.7, 27.0, 21.5, 15.8, 14.2.
HRMS calcd for [M + H]+ C17H18NS2 300.0875, found: 300.0873.
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 96 ratio and 11b was isolated in 80% (0.242 g) yield as a yellow oil.
96 ratio and 11b was isolated in 80% (0.242 g) yield as a yellow oil.
1H NMR (400 MHz, CDCl3) δ 7.44 (d, J = 5.3 Hz, 1H), 7.31–7.24 (m, 1H), 7.20 (d, J = 5.3 Hz, 1H), 7.06 (d, J = 7.7 Hz, 1H), 7.04–6.93 (m, 2H), 2.99 (q, J = 7.5 Hz, 2H), 2.10 (s, 3H), 1.39 (t, J = 7.6 Hz, 3H).
19F NMR (376 MHz, CDCl3) δ −112.9.
13C NMR (101 MHz, CDCl3) δ 172.4, 162.8 (d, J = 245.8 Hz), 150.3, 140.0 (d, J = 2.3 Hz), 138.1 (d, J = 8.1 Hz), 130.0 (d, J = 8.5 Hz), 129.2, 128.3, 126.4, 124.1 (d, J = 3.0 Hz), 122.1, 115.3 (d, J = 22.2 Hz), 114.1 (d, J = 21.1 Hz), 27.0, 15.7, 14.1.
HRMS calcd for [M + H]+ C16H15FNS2 304.0625, found: 304.0625.
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 93 ratio and 12b was isolated in 74% (0.237 g) yield as a yellow oil.
93 ratio and 12b was isolated in 74% (0.237 g) yield as a yellow oil.
1H NMR (400 MHz, CDCl3) δ 7.44 (d, J = 5.2 Hz, 1H), 7.31 (s, 1H), 7.30–7.22 (m, 2H), 7.20 (d, J = 5.3 Hz, 1H), 7.14 (dt, J = 6.7, 1.8 Hz, 1H), 2.99 (q, J = 7.6 Hz, 2H), 2.12 (s, 3H), 1.39 (t, J = 7.6 Hz, 3H).
13C NMR (101 MHz, CDCl3) δ 172.4, 150.4, 139.8, 137.7, 134.3, 129.7, 129.1, 128.4, 128.4, 127.3, 126.6, 126.5, 122.1, 27.0, 15.8, 14.1.
HRMS calcd for [M + H]+ C16H15ClNS2 320.0329, found: 320.0330.
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 87 ratio and 13b was isolated in 75% (0.227 g) yield as a yellow oil.
87 ratio and 13b was isolated in 75% (0.227 g) yield as a yellow oil.
1H NMR (400 MHz, CDCl3) δ 7.45 (d, J = 5.3 Hz, 1H), 7.34–7.25 (m, 1H), 7.21 (dd, J = 5.3, 2.0 Hz, 1H), 7.16 (td, J = 7.5, 1.9 Hz, 1H), 7.13–7.03 (m, 2H), 2.96 (q, J = 7.6 Hz, 2H), 2.10 (s, 3H), 1.36 (t, J = 7.6 Hz, 3H).
19F NMR (376 MHz, CDCl3) δ −114.7.
13C NMR (101 MHz, CDCl3) δ 171.9, 159.8 (d, J = 248.1 Hz), 150.0, 135.1, 131.4 (d, J = 3.4 Hz), 130.1 (d, J = 2.7 Hz), 129.9, 129.3 (d, J = 8.2 Hz), 125.8, 124.1 (d, J = 3.7 Hz), 123.8 (d, J = 15.0 Hz), 122.3, 116.0 (d, J = 22.3 Hz), 27.0, 15.7, 14.0.
HRMS calcd for [M + H]+ C16H15FNS2 304.0625, found: 304.0625.
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 77 ratio and 14b was isolated in 62% (0.185 g) yield as a yellow oil.
77 ratio and 14b was isolated in 62% (0.185 g) yield as a yellow oil.
1H NMR (400 MHz, CDCl3) δ 7.58 (s, 1H), 7.40–7.34 (m, 5H), 7.33 (d, J = 5.2 Hz, 1H), 7.12 (d, J = 5.3 Hz, 1H), 2.79 (d, J = 7.1 Hz, 2H), 2.15–1.93 (m, 1H), 0.98 (d, J = 6.6 Hz, 6H).
13C NMR (101 MHz, CDCl3) δ 170.5, 140.7, 140.3, 135.8, 130.4, 130.1, 129.2, 128.5, 127.9, 127.7, 124.7, 42.3, 29.7, 22.2.
HRMS calcd for [M + H]+ C17H18NS2 300.0875, found: 300.0873.
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 94 ratio and 15b was isolated in 78% (0.233 g) yield as a yellow oil.
94 ratio and 15b was isolated in 78% (0.233 g) yield as a yellow oil.
1H NMR (400 MHz, CDCl3) δ 7.35–7.25 (m, 6H), 2.97 (q, J = 7.6 Hz, 2H), 2.54 (d, J = 1.1 Hz, 3H), 2.07 (s, 3H), 1.38 (t, J = 7.6 Hz, 3H).
13C NMR (101 MHz, CDCl3) δ 171.8, 149.8, 141.3, 140.5, 136.2, 128.4, 128.3, 127.7, 127.0, 124.8, 123.1, 27.0, 15.7, 15.3, 14.1.
HRMS calcd for [M + H]+ C17H18NS2 300.0875, found: 300.0875.
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 75 ratio and 16b was isolated in 61% (0.191 g) yield as a yellow oil.
75 ratio and 16b was isolated in 61% (0.191 g) yield as a yellow oil.
1H NMR (400 MHz, CDCl3) δ 7.52 (s, 1H), 7.37–7.28 (m, 5H), 6.79 (q, J = 1.1 Hz, 1H), 2.77 (d, J = 7.2 Hz, 2H), 2.52 (d, J = 1.1 Hz, 3H), 2.15–1.93 (m, 1H), 0.97 (d, J = 6.7 Hz, 6H).
13C NMR (101 MHz, CDCl3) δ 170.0, 140.2, 139.2, 136.1, 130.4, 129.1, 128.8, 128.4, 127.5, 125.3, 42.3, 29.7, 22.2, 15.2.
HRMS calcd for [M + H]+ C18H20NS2 314.1032, found: 314.1031.
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 95 ratio and 17b was isolated in 76% (0.281 g) yield as a yellow oil.
95 ratio and 17b was isolated in 76% (0.281 g) yield as a yellow oil.
1H NMR (400 MHz, CDCl3) δ 7.40–7.14 (m, 5H), 6.91 (s, 1H), 2.97 (q, J = 7.5 Hz, 2H), 2.85 (t, J = 7.5 Hz, 2H), 2.07 (s, 3H), 1.88–1.70 (m, 2H), 1.52–1.30 (m, 6H), 1.37 (t, J = 7.6 Hz, 3H), 0.93 (t, J = 7.6 Hz, 3H).
13C NMR (101 MHz, CDCl3) δ 171.7, 149.7, 146.6, 140.9, 136.3, 128.4, 128.3, 126.9, 126.5, 124.5, 123.3, 31.6, 31.4, 30.2, 28.9, 27.0, 22.6, 15.7, 14.1.
HRMS calcd for [M + H]+ C22H28NS2 370.1658, found: 370.1655.
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 99 ratio and 18b was isolated in 80% (0.268 g) yield as a white solid: mp 112–114 °C.
99 ratio and 18b was isolated in 80% (0.268 g) yield as a white solid: mp 112–114 °C.
1H NMR (400 MHz, CDCl3) δ 7.88 (d, J = 7.0 Hz, 1H), 7.71 (d, J = 7.5 Hz, 1H), 7.46–7.32 (m, 7H), 2.96 (q, J = 7.6 Hz, 2H), 2.15 (s, 3H), 1.37 (t, J = 7.6 Hz, 3H).
13C NMR (101 MHz, CDCl3) δ 172.6, 150.5, 139.8, 139.6, 136.8, 134.8, 130.0, 129.2, 128.7, 127.6, 125.1, 124.7, 123.5, 122.7, 122.1, 27.0, 16.1, 14.1.
HRMS calcd for [M + H]+ C20H18NS2 336.0875, found: 336.0872.
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 98 ratio and 19b was isolated in 78% (0.272 g) yield as a yellow solid: mp 67–69 °C.
98 ratio and 19b was isolated in 78% (0.272 g) yield as a yellow solid: mp 67–69 °C.
1H NMR (300 MHz, CDCl3) δ 7.84 (d, J = 8.5 Hz, 1H), 7.70 (s, 1H), 7.57–7.47 (m, 3H), 7.47–7.27 (m, 5H), 2.77 (d, J = 7.2 Hz, 2H), 2.15–1.93 (m, 1H), 0.97 (d, J = 6.6 Hz, 6H).
13C NMR (75 MHz, CDCl3) δ 170.9, 141.0, 140.9, 138.2, 134.8, 134.7, 130.5, 130.4, 129.5, 129.0, 128.4, 125.2, 124.7, 123.4, 121.9, 42.2, 29.7, 22.2.
HRMS calcd for [M + H]+ C21H20NS2 350.1032, found: 350.1028.
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 99 ratio and 20b was isolated in 77% (0.241 g) yield as a yellow oil.
99 ratio and 20b was isolated in 77% (0.241 g) yield as a yellow oil.
1H NMR (400 MHz, CDCl3) δ 9.96 (s, 1H), 7.85 (s, 1H), 7.40–7.33 (m, 3H), 7.32–7.26 (m, 2H), 2.98 (q, J = 7.6 Hz, 2H), 2.13 (s, 3H), 1.38 (t, J = 7.6 Hz, 3H).
13C NMR (101 MHz, CDCl3) δ 182.7, 173.2, 151.2, 142.8, 142.3, 137.9, 137.8, 134.7, 128.8, 128.4, 128.0, 121.6, 27.0, 16.1, 14.0.
HRMS calcd for [M + H]+ C17H16NOS2 314.0668, found: 314.0666.
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 81 ratio and 21b was isolated in 68% (0.203 g) yield as a yellow solid: mp 116–118 °C.
81 ratio and 21b was isolated in 68% (0.203 g) yield as a yellow solid: mp 116–118 °C.
1H NMR (400 MHz, CDCl3) δ 7.75 (s, 1H), 7.40 (m, 3H), 7.36–7.30 (m, 2H), 6.81 (q, J = 1.1 Hz, 1H), 2.65 (s, 3H), 2.55 (d, J = 1.1 Hz, 3H).
13C NMR (101 MHz, CDCl3) δ 191.6, 164.8, 142.3, 141.9, 141.3, 139.6, 135.6, 129.6, 129.0, 128.9, 128.2, 124.5, 25.7, 15.3.
HRMS calcd for [M + H]+ C16H14NOS2 300.0511, found: 300.0510.
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 98 ratio and 22b was isolated in 63% (0.217 g) yield as a colourless oil.
98 ratio and 22b was isolated in 63% (0.217 g) yield as a colourless oil.
1H NMR (400 MHz, CDCl3) δ 7.39–7.30 (m, 1H), 7.26 (d, J = 5.2 Hz, 1H), 7.18 (d, J = 7.7 Hz, 1H), 7.10 (ddd, J = 10.0, 2.6, 1.6 Hz, 1H), 7.07 (d, J = 5.2 Hz, 1H), 7.02 (tdd, J = 8.5, 2.6, 1.0 Hz, 1H), 6.80 (d, J = 3.5 Hz, 1H), 6.63 (d, J = 3.3 Hz, 1H), 2.75 (t, J = 7.6 Hz, 2H), 1.64 (quint., J = 7.4 Hz, 2H), 1.42–1.17 (m, 6H), 0.90 (t, J = 7.4 Hz, 3H).
19F NMR (376 MHz, CDCl3) δ −113.4.
13C NMR (101 MHz, CDCl3) δ 162.7 (d, J = 245.6 Hz), 147.1, 138.6 (d, J = 8.1 Hz), 137.0 (d, J = 2.2 Hz), 132.9, 132.6, 130.1, 129.7 (d, J = 8.4 Hz), 126.7, 125.0 (d, J = 3.0 Hz), 124.3, 123.9, 116.2 (d, J = 21.9 Hz), 114.1 (d, J = 21.0 Hz), 31.5, 31.5, 30.1, 28.7, 22.6, 14.0.
HRMS calcd for [M + H]+ C20H22FS2 345.1142, found: 345.1143.
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 94 ratio and 23b was isolated in 61% (0.207 g) yield as a yellow oil.
94 ratio and 23b was isolated in 61% (0.207 g) yield as a yellow oil.
1H NMR (400 MHz, CDCl3) δ 7.44–7.27 (m, 5H), 6.76 (d, J = 1.2 Hz, 1H), 6.73 (d, J = 3.6 Hz, 1H), 6.59 (d, J = 3.6 Hz, 1H), 2.72 (t, J = 7.6 Hz, 2H), 2.51 (d, J = 1.2 Hz, 3H), 1.64 (quint., J = 7.4 Hz, 2H), 1.41–1.30 (m, 6H), 0.90 (t, J = 7.4 Hz, 3H).
13C NMR (101 MHz, CDCl3) δ 146.1, 138.2, 137.9, 136.7, 133.6, 129.7, 129.2, 128.8, 128.2, 127.1, 125.8, 124.0, 31.5, 31.4, 30.1, 28.7, 22.6, 15.2, 14.0.
HRMS calcd for [M + H]+ C21H25S2 341.1392, found: 341.1389.
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 68 ratio and 24b was isolated in 50% (0.145 g) yield as a yellow oil.
68 ratio and 24b was isolated in 50% (0.145 g) yield as a yellow oil.
1H NMR (400 MHz, CDCl3) δ 7.40–7.33 (m, 5H), 6.76 (q, J = 1.1 Hz, 1H), 6.74 (d, J = 3.9 Hz, 1H), 6.72 (d, J = 3.9 Hz, 1H), 2.51 (d, J = 1.1 Hz, 3H).
13C NMR (101 MHz, CDCl3) δ 139.3, 138.9, 136.1, 135.1, 129.4, 129.2, 128.9, 128.5, 128.2, 127.5, 126.1, 125.2, 15.2.
HRMS calcd for [M + H]+ C16H12ClS2 291.0064, found: 291.0064.
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 96 ratio and 25b was isolated in 53% (0.181 g) yield as a colourless oil.
96 ratio and 25b was isolated in 53% (0.181 g) yield as a colourless oil.
1H NMR (300 MHz, CDCl3) δ 7.43–7.30 (m, 5H), 6.84 (d, J = 3.6 Hz, 1H), 6.76 (q, J = 1.1 Hz, 1H), 6.73 (d, J = 3.7 Hz, 1H), 4.17–3.93 (m, 4H), 2.51 (d, J = 1.1 Hz, 3H), 1.75 (s, 3H).
13C NMR (75 MHz, CDCl3) δ 146.8, 138.8, 138.4, 136.5, 136.0, 129.1, 129.0, 128.9, 128.3, 127.2, 125.7, 124.2, 107.1, 64.9, 27.3, 15.2.
HRMS calcd for [M + H]+ C19H19O2S2 343.0821, found: 343.0822.
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 97 ratio and 26b was isolated in 50% (0.148 g) yield as a yellow oil.
97 ratio and 26b was isolated in 50% (0.148 g) yield as a yellow oil.
1H NMR (400 MHz, CDCl3) δ 7.45–7.24 (m, 5H), 6.73 (q, J = 1.1 Hz, 1H), 5.96 (d, J = 3.2 Hz, 1H), 5.90 (d, J = 3.2 Hz, 1H), 2.60 (t, J = 7.5 Hz, 2H), 2.52 (d, J = 1.1 Hz, 3H), 1.65–1.52 (m, 2H), 1.44–1.27 (m, 2H), 0.94 (t, J = 7.3 Hz, 3H).
13C NMR (101 MHz, CDCl3) δ 155.6, 146.9, 137.8, 137.4, 137.1, 128.9, 128.7, 128.2, 127.1, 126.4, 107.2, 106.5, 30.2, 27.7, 22.2, 15.2, 13.8.
HRMS calcd for [M + H]+ C19H21OS 297.1308, found: 297.1306.
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 97 ratio and 27b was isolated in 36% (0.114 g) yield as a colourless oil.
97 ratio and 27b was isolated in 36% (0.114 g) yield as a colourless oil.
1H NMR (400 MHz, CDCl3) δ 7.44 (s, 1H), 7.33–7.29 (m, 3H), 7.26 (d, J = 5.1 Hz, 1H), 7.03 (d, J = 5.2 Hz, 1H), 6.07 (d, J = 3.3 Hz, 1H), 5.95 (d, J = 3.3 Hz, 1H), 2.61 (t, J = 7.6 Hz, 2H), 1.67–1.51 (m, 2H), 1.46–1.30 (m, 2H), 0.94 (t, J = 7.3 Hz, 3H).
13C NMR (101 MHz, CDCl3) δ 156.4, 146.3, 138.7, 135.9, 134.1, 130.1, 129.6, 129.5, 129.1, 127.3, 127.2, 123.7, 108.2, 106.7, 30.2, 27.7, 22.2, 13.8.
HRMS calcd for [M + H]+ C18H18ClOS 317.0761, found: 317.0761.
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 96 ratio and 28b was isolated in 66% (0.206 g) yield as a yellow oil.
96 ratio and 28b was isolated in 66% (0.206 g) yield as a yellow oil.
1H NMR (400 MHz, CDCl3) δ 7.65–7.31 (m, 5H), 6.75 (q, J = 1.2 Hz, 1H), 6.29 (s, 1H), 3.79 (s, 3H), 2.56 (s, 3H), 2.53 (d, J = 1.1 Hz, 3H).
13C NMR (101 MHz, CDCl3) δ 164.3, 158.1, 146.7, 139.0, 139.0, 136.5, 128.9, 128.7, 128.4, 127.5, 124.6, 114.8, 106.9, 51.3, 15.2, 13.7.
HRMS calcd for [M + H]+ C18H17O3S 313.0893, found: 313.0892.
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 99 ratio and 29b was isolated in 55% (0.139 g) yield as a yellow oil.
99 ratio and 29b was isolated in 55% (0.139 g) yield as a yellow oil.
1H NMR (400 MHz, CDCl3) δ 7.31–7.24 (m, 3H), 7.22 (d, J = 7.5 Hz, 2H), 6.97 (q, J = 1.2 Hz, 1H), 6.74–6.54 (m, 1H), 6.27 (dd, J = 3.6, 1.8 Hz, 1H), 6.19 (dd, J = 3.6, 2.7 Hz, 1H), 3.08 (s, 3H), 2.54 (d, J = 1.1 Hz, 3H).
13C NMR (101 MHz, CDCl3) δ 139.5, 139.4, 136.8, 128.5, 127.7, 127.1, 126.7, 126.6, 125.7, 122.9, 111.1, 107.8, 34.0, 15.3.
HRMS calcd for [M + H]+ C16H16NS 254.0998, found: 254.0995.
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 98 ratio and 30b was isolated in 63% (0.183 g) yield as a white solid: mp 120–122 °C.
98 ratio and 30b was isolated in 63% (0.183 g) yield as a white solid: mp 120–122 °C.
1H NMR (400 MHz, CDCl3) δ 7.73 (s, 1H), 7.63 (d, J = 9.1 Hz, 1H), 7.59 (dd, J = 6.9, 1.2 Hz, 1H), 7.21–7.15 (m, 5H), 7.12 (dd, J = 9.1, 6.9 Hz, 1H), 7.07 (q, J = 1.1 Hz, 1H), 6.55 (t, J = 6.8 Hz, 1H), 2.59 (d, J = 1.1 Hz, 3H).
13C NMR (101 MHz, CDCl3) δ 146.0, 141.5, 140.8, 135.9, 134.8, 128.7, 127.9, 127.3, 127.2, 124.5, 124.2, 121.1, 118.2, 117.7, 112.1, 15.4.
HRMS calcd for [M + H]+ C18H15N2S 291.0550, found: 291.0548.
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 95 ratio and 31b was isolated in 66% (0.194 g) yield as a yellow solid: mp 167–169 °C.
95 ratio and 31b was isolated in 66% (0.194 g) yield as a yellow solid: mp 167–169 °C.
1H NMR (400 MHz, CDCl3) δ 7.77 (d, J = 6.9 Hz, 1H), 7.68 (s, 1H), 7.61 (d, J = 9.1 Hz, 1H), 7.56 (d, J = 5.3 Hz, 1H), 7.38 (dd, J = 5.3, 2.3 Hz, 1H), 7.25–7.12 (m, 2H), 7.11–6.99 (m, 2H), 6.89 (td, J = 7.5, 1.2 Hz, 1H), 6.63 (td, J = 6.8, 1.2 Hz, 1H).
19F NMR (376 MHz, CDCl3) δ −115.0.
13C NMR (101 MHz, CDCl3) δ 159.4 (d, J = 248.0 Hz), 146.2, 135.0, 130.5 (d, J = 3.3 Hz), 130.3 (d, J = 3.1 Hz), 129.4 (d, J = 8.2 Hz), 126.6, 126.1, 124.6, 124.3 (d, J = 3.6 Hz), 123.8, 123.4 (d, J = 14.7 Hz), 117.8, 117.5, 116.0 (d, J = 22.2 Hz), 112.3.
HRMS calcd for [M + H]+ C17H12FN2S 295.0700, found: 295.0699.
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 91 ratio and 32b was isolated in 71% (0.207 g) yield as a yellow oil.
91 ratio and 32b was isolated in 71% (0.207 g) yield as a yellow oil.
1H NMR (400 MHz, CDCl3) δ 9.09 (d, J = 1.5 Hz, 1H), 7.86 (s, 1H), 7.63 (d, J = 4.7 Hz, 1H), 7.48 (dd, J = 4.7, 1.5 Hz, 1H), 7.24–7.19 (m, 3H), 7.18–7.12 (m, 2H), 7.08 (q, J = 1.2 Hz, 1H), 2.61 (d, J = 1.1 Hz, 3H).
13C NMR (101 MHz, CDCl3) δ 143.8, 142.4, 141.7, 141.1, 136.5, 135.6, 129.5, 129.0, 128.2, 127.7, 127.2, 120.0, 119.3, 117.0, 15.4.
HRMS calcd for [M + H]+ C17H14N3S 292.0903, found: 292.0902.
1H NMR (400 MHz, CDCl3) δ 8.93 (s, 1H), 8.57 (d, J = 3.4 Hz, 1H), 7.90 (ddd, J = 8.0, 2.4, 1.6 Hz, 1H), 7.45 (s, 1H), 7.35 (dd, J = 7.2, 4.8 Hz, 1H), 7.22 (d, J = 8.2 Hz, 2H), 7.15 (d, J = 7.9 Hz, 2H), 2.99 (q, J = 7.6 Hz, 2H), 2.37 (s, 3H), 2.14 (s, 3H), 1.39 (t, J = 7.6 Hz, 3H).
13C NMR (101 MHz, CDCl3) δ 172.3, 150.4, 148.9, 146.8, 142.5, 140.3, 137.3, 132.7, 132.7, 129.9, 129.4, 128.2, 127.7, 126.5, 123.7, 122.4, 27.0, 21.2, 15.9, 14.1.
HRMS calcd for [M + H]+ C22H21N2S2 377.1141, found: 377.1138.
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 53 ratio. Product 34b was isolated in 8% (0.023 g) yield as a yellow solid: mp 127–129 °C, and product 34c was isolated in 9% (0.026 g) yield as a yellow solid: mp 121–123 °C.
53 ratio. Product 34b was isolated in 8% (0.023 g) yield as a yellow solid: mp 127–129 °C, and product 34c was isolated in 9% (0.026 g) yield as a yellow solid: mp 121–123 °C.
13C NMR (101 MHz, CDCl3) δ 190.3, 144.8, 143.4, 140.8, 135.8, 132.6, 131.1, 130.7, 129.2, 128.7, 127.9, 127.0, 125.6, 26.6.
HRMS calcd for [M + H]+ C16H13OS2 285.0402, found: 285.0403.
13C NMR (101 MHz, CDCl3) δ 190.3, 145.6, 143.5, 142.7, 137.0, 135.1, 133.3, 129.0, 127.7, 126.4, 124.7, 124.3, 121.2, 26.6.
HRMS calcd for [M + H]+ C16H13OS2 285.0402, found: 285.0403.
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 96 ratio and 35b was isolated in 56% (0.184 g) yield as a colourless oil.
96 ratio and 35b was isolated in 56% (0.184 g) yield as a colourless oil.
1H NMR (300 MHz, CDCl3) δ 7.41–7.32 (m, 5H), 7.27 (d, J = 5.2 Hz, 1H), 7.09 (d, J = 5.2 Hz, 1H), 6.86 (d, J = 3.7 Hz, 1H), 6.80 (d, J = 3.7 Hz, 1H), 4.08–3.91 (m, 4H), 1.76 (s, 3H).
13C NMR (75 MHz, CDCl3) δ 147.4, 139.0, 136.3, 135.6, 131.6, 130.6, 129.2, 128.4, 127.4, 126.2, 124.2, 124.1, 107.1, 64.9, 27.4.
HRMS calcd for [M + H]+ C18H17O2S2 329.0665, found: 329.0665.
Deprotection of 35b into 34b: A 2 N aqueous HCl solution (1 mL) was added to a solution of 35b (0.164 g, 0.5 mmol) in THF (1 mL). The resulting mixture was stirred at 60 °C for 8 h. The mixture was extracted with Et2O (3 × 10 mL). The combined organic phase was dried over MgSO4, filtrated, and concentrated under reduced pressure. The residue was purified by column chromatography on silica gel to give product 34b in 86% (0.122 g).
Author contributions
The manuscript was written through contributions of all authors. All authors have given approval to the final version of the manuscript.Conflicts of interest
There are no conflicts to declare.Data availability
The data underlying this study are available in the published article and its supplementary information (SI). Supplementary information: copies of NMR spectra. See DOI: https://doi.org/10.1039/d6ob00099a.Acknowledgements
We are grateful to the CSC for a grant to B. L. We thank CNRS and Rennes Metropole for providing financial support.References
- W. Liao, Z. Wang, Y. Han, Y. Qi, J. Liu, J. Xie, Y. Tian, Q. Lei, R. Chen, M. Sun, L. Tang, G. Gong and Y. Zhao, Design, synthesis and biological activity of novel 2,3,4,5-tetrasubstituted thiophene derivatives as PI3Ka inhibitors with potent antitumor activity, Eur. J. Med. Chem., 2020, 197, 112309 CrossRef CAS.
- K. K.-C. Liu, J. Zhu, G. L. Smith, M.-J. Yin, S. Bailey, J. H. Chen, Q. Hu, Q. Huang, C. Li, Q. J. Li, M. A. Marx, G. Paderes, P. F. Richardson, N. W. Sach, M. Walls, P. A. Wells and A. Zou, Highly Selective and Potent Thiophenes as PI3K Inhibitors with Oral Antitumor Activity, ACS Med. Chem. Lett., 2011, 2, 809–813 CrossRef CAS PubMed.
- X. Wu, Y. Wan, A. K. Mahalingam, A. M. S. Murugaiah, B. Plouffe, M. Botros, A. Karlén, M. Hallberg, N. Gallo-Payet and M. Alterman, Selective Angiotensin II AT2 Receptor Agonists: Arylbenzylimidazole Structure-Activity Relationships, J. Med. Chem., 2006, 49, 7160–7168 CrossRef CAS PubMed.
- W. Wang, S. Zhang, T. Zhang, Y. Yang, Y. Cui, Y. Yu, Y. Xiao, D. H. Ryu, C. E. Song and J. Hou, Molecule Design of Novel Electron Acceptor with Superior Chemical Stability for Photovoltaic Applications, Adv. Funct. Mater., 2023, 33, 2304752 CrossRef CAS.
- C. Messina, K. McKibbon, K. S.-L. Wong, P. Prevost, V. Petkov and P. Forgione, A Modular and Regioselective Synthesis of Di- and Triarylated Thiophenes: Strategies for Accessing Challenging Isomeric Motifs, J. Org. Chem., 2025, 90, 15595–15611 CrossRef CAS.
- S.-i. Sakurai, H. Goto and E. Yashima, Synthesis and Chiroptical Properties of Optically Active, Regioregular Oligothiophenes, Org. Lett., 2001, 3, 2379–2382 CrossRef PubMed.
- C. Zhao, Y. Zhang, C. Wang, L. Rothberg and M.-K. Ng, Synthesis of Homopolymer Containing Diphenyl End-Capped Oligothiophene Co-oligomer Unit in the Side Chain, Org. Lett., 2006, 8, 1585–1588 CrossRef CAS PubMed.
- R. Takita, C. Song and T. M. Swager, π-Dimer Formation in an Oligothiophene Tweezer Molecule, Org. Lett., 2008, 10, 5003–5005 CrossRef CAS.
- D. Schiefer, H. Komber, K. F. Mugwanga, S. Kunz, R. Hanselmann, G. Reiter and M. Sommer, Poly(3-(2,5-dioctylphenyl)thiophene) Synthesized by Direct Arylation Polycondensation: End Groups, Defects, and Crystallinity, Macromolecules, 2016, 49, 7230–7237 CrossRef CAS.
- C. A. Briehn, T. Kirschbaum and P. Baeuerle, Polymer-Supported Synthesis of Regioregular Head-to-Tail-Coupled Oligo(3-arylthiophene)s Utilizing a Traceless Silyl Linker, J. Org. Chem., 2000, 65, 352–359 CrossRef CAS PubMed.
- E. Naudin, N. El Mehdi, C. Soucy, L. Breau and D. Belanger, Poly(3-arylthiophenes): Syntheses of Monomers and Spectroscopic and Electrochemical Characterization of the Corresponding Polymers, Chem. Mater., 2001, 13, 634–642 CrossRef CAS.
- P. B. Arockiam, C. Bruneau and P. H. Dixneuf, Ruthenium(II)-catalyzed C–H bond activation and functionalization, Chem. Rev., 2012, 112, 5879–5918 Search PubMed.
- L. Theveau, C. Schneider, C. Fruit and C. Hoarau, Orthogonal palladium-catalyzed direct C–H bond arylation of heteroaromatics with aryl halides, ChemCatChem, 2016, 8, 3183–3194 CrossRef CAS.
- S. Ruiz, P. Villuendas and E. P. Urriolabeitia, Ru-catalysed C–H functionalisations as a tool for selective organic synthesis, Tetrahedron Lett., 2016, 57, 3413–3432 CrossRef CAS.
- T. Gensch, M. J. James, T. Dalton and F. Glorius, Increasing catalyst efficiency in C–H activation catalysis, Angew. Chem., Int. Ed., 2018, 57, 2296–2306 CrossRef CAS.
- J. Kalepu, P. Gandeepan, L. Ackermann and L. T. Pilarski, C4–H indole functionalisation: precedent and prospects, Chem. Sci., 2018, 9, 4203–4216 RSC.
- K. Hirano and M. Miura, A lesson for site-selective C–H functionalization on 2-pyridones: radical, organometallic, directing group and steric controls, Chem. Sci., 2018, 9, 22–32 RSC.
- S. Mao, H. Li, X. Shi, J.-F. Soulé and H. Doucet, Environmentally benign arylations of 5-membered ring heteroarenes by Pd-catalyzed C–H bonds activations, ChemCatChem, 2019, 11, 269–286 Search PubMed.
- S. Rej, Y. Ano and N. Chatani, Bidentate directing groups: An efficient tool in C–H bond functionalization chemistry for the expedient construction of C–C bond, Chem. Rev., 2020, 120, 1788–1887 Search PubMed.
- J. H. Docherty, T. M. Lister, G. Mcarthur, M. T. Findlay, P. Domingo-Legarda, J. Kenyon, S. Choudhary and I. Larrosa, Transition-Metal-Catalyzed C–H Bond Activation for the Formation of C–C Bonds in Complex Molecules, Chem. Rev., 2023, 123, 7692–7760 Search PubMed.
- Y. Akita, A. Inoue, K. Yamamoto, A. Ohta, T. Kurihara and M. Shimizu, Palladium-catalyzed coupling reaction of chloropyrazines with indole, Heterocycles, 1985, 23, 2327–2333 CrossRef CAS.
- A. Ohta, Y. Akita, T. Ohkuwa, M. Chiba, R. Fukunaga, A. Miyafuji, T. Nakata, N. Tani and Y. Aoyagi, Palladium-catalyzed arylation of furan, thiophene, benzo[b]furan and benzo[b]thiophene, Heterocycles, 1990, 31, 1951–1958 CrossRef CAS.
- K. Masui, A. Mori, K. Okano, K. Takamura, M. Kinoshita and T. Ikeda, Syntheses and properties of donor–acceptor-type 2,5-diarylthiophene and 2,5-diarylthiazole, Org. Lett., 2004, 6, 2011–2014 CrossRef CAS.
- E. David, S. Pellet-Rostaing and M. Lemaire, Heck-like coupling and Pictet–Spengler reaction for the synthesis of benzothieno[3,2-c]quinolines, Tetrahedron, 2007, 63, 8999–9006 CrossRef CAS.
- J. Roger, F. Požgan and H. Doucet, Ligand-less palladium-catalyzed direct 5-arylation of thiophenes at low catalyst loadings, Green Chem., 2009, 11, 425–432 RSC.
- B. Liégault, I. Petrov, S. I. Gorlesky and K. Fagnou, Modulating reactivity and diverting selectivity in palladium-catalyzed heteroaromatic direct arylation through the use of a chloride activating/blocking group, J. Org. Chem., 2010, 75, 1047–1060 CrossRef.
- H.-Y. Huang, A. Benzai, X. Shi and H. Doucet, Effective tools for the metal-catalyzed regiodivergent direct arylations of (hetero)arenes, Chem. Rec., 2021, 21, 343–356 CrossRef CAS PubMed.
- Y. Li, J. Wang, M. Huang, Z. Wang, Y. Wu and Y. Wu, Direct C–H Arylation of Thiophenes at Low Catalyst Loading of a Phosphine-Free Bis(alkoxo)palladium Complex, J. Org. Chem., 2014, 79, 2890–2897 CrossRef CAS PubMed.
- S. Ma and Z. Gu, 1,4-Migration of rhodium and palladium in catalytic organometallic reactions, Angew. Chem., Int. Ed., 2005, 44, 7512–7517 CrossRef CAS PubMed.
- X. Dong, H. Wang, H. Liu and F. Wang, Recent advances in transition metal migration involving reactions, Org. Chem. Front., 2020, 7, 3530–3556 RSC.
- M.-Y. Li, D. Wei, C.-G. Feng and G.-Q. Lin, Tandem Reactions involving 1,4-Palladium Migrations, Chem. – Asian J., 2022, 17, e202200456 CrossRef CAS PubMed.
- R. C. Larock, Y. D. Lu, A. C. Bain and C. E. Russell, Palladium-catalyzed coupling of aryl iodides, nonconjugated dienes and carbon nucleophiles by palladium migration, J. Org. Chem., 1991, 56, 4589–4590 CrossRef CAS.
- M. A. Campo, H. Zhang, T. Yao, A. Ibdah, R. D. McCulla, Q. Huang, J. Zhao, W. S. Jenks and R. C. Larock, Aryl to aryl palladium migration in the Heck and Suzuki coupling of o-halobiaryls, J. Am. Chem. Soc., 2007, 129, 6298–6307 CrossRef CAS PubMed.
- L. Liu, M. Cordier, T. Roisnel and H. Doucet, Double C–H bond functionalization for C–C coupling at the β-position of thiophenes using palladium-catalyzed 1,4-migration associated with direct arylation, Org. Chem. Front., 2023, 10, 1441–1455 RSC.
- M. A. Campo and R. C. Larock, Synthesis of Fluoren-9-ones by the Palladium-Catalyzed Cyclocarbonylation of o-Halobiaryls, J. Org. Chem., 2002, 67, 5616–5620 CrossRef CAS PubMed.
- W. Hagui, N. Besbes, E. Srasra, T. Roisnel, J.-F. Soule and H. Doucet, Short Synthesis of Sulfur Analogues of Polyaromatic Hydrocarbons through Three Palladium-Catalyzed C–H Bond Arylations, Org. Lett., 2016, 18, 4182–4185 CrossRef CAS PubMed.
- T. Cantat, E. Génin, C. Giroud, G. Meyer and A. Jutand, Structural and kinetic effects of chloride ions in the palladium catalyzed allylic substitutions, J. Organomet. Chem., 2003, 687, 365–376 CrossRef CAS.
- A. Mori, A. Sekiguchi, K. Masui, T. Shimada, M. Horie, K. Osakada, M. Kawamoto and T. Ikeda, Facile synthesis of 2,5-diarylthiazoles via palladium-catalyzed tandem C–H substitutions. Design of tunable light emission and liquid crystalline characteristics, J. Am. Chem. Soc., 2003, 125, 1700–1701 CrossRef CAS PubMed.
- A. Yokooji, T. Okazawa, T. Satoh, M. Miura and M. Nomura, Palladium-catalyzed direct arylation of thiazoles with aryl bromides, Tetrahedron, 2003, 59, 5685–5689 CrossRef CAS.
- J. J. Dong, J. Roger, C. Verrier, T. Martin, R. Le Goff, C. Hoarau and H. Doucet, Carbonates: eco-friendly solvents for palladium-catalysed direct arylation of heteroaromatics, Green Chem., 2010, 12, 2053–2063 RSC.
- D. L. Davies, S. M. A. Donald and S. A. Macgregor, Computational study of the mechanism of cyclometalation by palladium acetate, J. Am. Chem. Soc., 2005, 127, 13754–13755 CrossRef CAS PubMed.
- M. Lafrance and K. Fagnou, Palladium-catalyzed benzene arylation: Incorporation of catalytic pivalic acid as a proton shuttle and a key element in catalyst design, J. Am. Chem. Soc., 2006, 128, 16496–16497 CrossRef CAS.
- S. I. Gorelsky, Origins of regioselectivity of the palladium-catalyzed (aromatic)C-H bond metalation–deprotonation, Coord. Chem. Rev., 2013, 257, 153–164 CrossRef CAS.
- D. L. Davies, S. A. Macgregor and C. L. McMullin, Computational studies of carboxylate-assisted C-H activation and functionalization at group 8–10 transition metal centers, Chem. Rev., 2017, 117, 8649–8709 CrossRef CAS PubMed.
- M. Kienle, A. Unsinn and P. Knochel, Synthesis of Dibenzothiophenes and Related Classes of Heterocyclesby Using Functionalized Dithiocarbamates, Angew. Chem., Int. Ed., 2010, 49, 4751–4754 CrossRef CAS.
| This journal is © The Royal Society of Chemistry 2026 |