Open Access Article
Open Access ArticleSolvated electron-driven hydroamination of olefins
Anietie W.
Williams
 a and
Kerry M.
Gilmore
a and
Kerry M.
Gilmore
 *ab
*ab
aDepartment of Chemistry, University of Connecticut, Storrs, Connecticut 06269, USA. E-mail: kerry.m.gilmore@uconn.edu
bInstitute of Materials Science, University of Connecticut, Storrs, Connecticut 06269, USA
First published on 16th March 2026
Abstract
We report a transition metal-free method for the intermolecular hydroamination of olefins using solvated electrons, generated in situ from granulated lithium under sonication in 2-methyltetrahydrofuran (2-MeTHF). This additive-free protocol enables rapid formation of mixed secondary and tertiary amines under ambient conditions and is broadly applicable to vinylarenes and alkyl amines. Selected allylarenes and conjugated dienes are also competent substrates, exhibiting distinct regioselectivity under the reaction conditions. Mechanistic studies support an SET and HAT pathway, with lithium amide intermediates acting as both reductants and nucleophiles. The method offers excellent atom economy, sustainability, and synthetic utility, exemplified by the selective synthesis of the pharmaceutical benzphetamine in high yield.
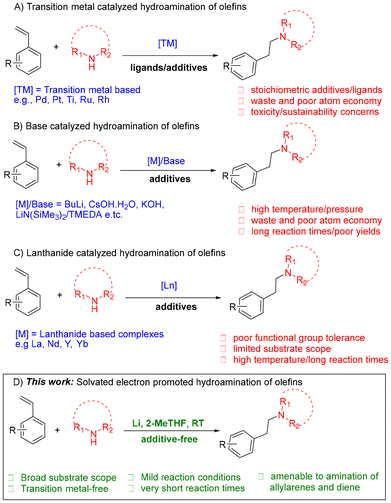
Efficient synthesis of nitrogen-containing compounds remains a critical goal in organic chemistry due to their central role in pharmaceuticals, agrochemicals, and materials science. Among the diverse C–N bond-forming strategies, hydroamination of olefins stands out as an inherently atom-economical method for generating amines in a single step from readily available substrates.1 In particular, the anti-Markovnikov addition of amines to vinylarenes and the Markovnikov addition of amines to allylarenes both yield β-arylethylamines—motifs frequently found in psychodysleptics, analgesics, anorectics, and antihistamines (Fig. 1). Structurally diverse examples such as verapamil, methamphetamine, and benzedrine underscore the broad pharmacological utility of these scaffolds. Despite its appeal, intermolecular hydroamination remains kinetically disfavored due to the low reactivity between the two reaction partners.2–5 Transition-metal-catalyzed variants have enabled key advances but often rely on rare, expensive metals (e.g., Rh, Ru, Pd) and suffer from low sustainability due to catalyst toxicity, recovery issues, and poor atom economy in the presence of stoichiometric additives1,3,6–17 (Scheme 1A). Base- and lanthanide-mediated protocols offer partial solutions, but still face limitations such as extreme reaction conditions (high temperature/pressure, often long reaction times) or low functional group tolerance.1,4,5,18–22 (Scheme 1B and C).
Recently, alkali–metal-based hydroaminations have emerged as promising, transition-metal-free alternatives. For example, the lithium amide protocol reported by Mulks et al. proceeds under ambient air and moisture but requires careful control of amide solubility and does not proceed using primary amines.23 In parallel, Tortajada and Hevia developed bimetallic alkali–metal ferrate complexes that enable room-temperature hydroamination of vinylarenes.23 Despite its innovation, this approach necessitates the multistep synthesis of air- and moisture-sensitive pre-catalysts and shows limited functional group tolerance – only a single haloarene substrate (3-fluorostyrene) afforded product, and in modest yield (32%).24 Moreover, both protocols are confined to vinylarenes and show poor performance with allylic substrates, leaving key classes of synthetically valuable olefins underexplored. Herein, we report how the use of solvated electrons (granulated lithium and sonication in the presence of excess amine in the green solvent, 2-MeTHF) enables efficient hydroamination of vinylarenes for synthesis of β-arylethylamines with defined scope across substituted vinylarenes. We also showcase the applicability of this protocol to the amination of allylarenes, conjugated dienes and the straightforward synthesis of the anorectic drug benzphetamine. The reaction is additive-free, operates with olefins as the limiting reagent, and proceeds within 20 minutes to 2 hours at ambient temperature – making it a practical option for rapid synthesis under mild conditions (Scheme 1D).
This strategy is inspired by our previous work on the amination of aryl fluorides via a proposed solvated electron-induced SRN1 mechanism that involves single electron transfers (SETs) between lithium-amido complexes and radical intermediates.25 Solvated electrons, especially from alkali metals, typically reduce unsaturated double and triple bonds, or induce their polymerization; thus limiting their use in hydroamination.26,27 Our goal, therefore, was to understand and influence this inherent chemoselectivity by identifying conditions that favor C–N bond formation. Recognizing the key influence of solvent and amine coordination on electron solvation and transfer pathways,28 we hypothesized that a suitable reaction medium could tip the balance in favor of hydroamination over alkene reduction.
To evaluate this hypothesis, we studied the hydroamination of 4-methylstyrene (1) with pyrrolidine (2) as a model reaction, using conditions from our aryl fluoride amination protocol.25 With 1.4 equivalents of lithium, 2.5 equivalents of pyrrolidine, and 0.5 equivalents of KI in 4 mL of THF, the desired phenylethylamine 3 was obtained in 64% yield after 0.5 h, along with a significant amount of reduced product 4 (36%, Table 1, entry 1). Reducing the lithium and pyrrolidine equivalents to 1 and 1.5, respectively, and omitting the additive KI, increased the selectivity for 3 to 77%, with 23% of 4 (entry 2). Employing triethylamine as the solvent decreased both conversion and selectivity (79% conversion, 53% yield of 3, entry 3). Diethyl ether provided excellent selectivity towards 3, but conversion was only 77% after 0.5 h (entry 4), although the reaction reached completion at 1 h. 1,4-Dioxane performed worse than other ethereal solvents (entry 5, 43% conversion with 91% yield of 3). Non-ethereal solvents like pentane and toluene also gave poor results, with an approximate ratio of 60![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 40 of 3 to 4 and conversions ≤80% (entries 6 and 7). Ethyl acetate and chloroform led to side reactions with lithium, resulting in no conversion of 1 (entries 8 and 9). Further solvent screening identified 2-methyltetrahydrofuran (2-MeTHF, entry 10) as the optimal medium, affording the desired tertiary amine in >99% yield with only trace formation of 4.
40 of 3 to 4 and conversions ≤80% (entries 6 and 7). Ethyl acetate and chloroform led to side reactions with lithium, resulting in no conversion of 1 (entries 8 and 9). Further solvent screening identified 2-methyltetrahydrofuran (2-MeTHF, entry 10) as the optimal medium, affording the desired tertiary amine in >99% yield with only trace formation of 4.
| Entry | Deviation from standard conditions | Lithium (x equiv.) | Amine (y equiv.) | Conv. (%) | Yield (%) | |
|---|---|---|---|---|---|---|
| 3 | 4 | |||||
| a Optimization reactions were carried out on a 1.0 mmol scale of 4-methylstyrene (1) at a total reaction concentration of 0.25 M in the indicated solvent (4 mL). Lithium (1.0–1.4 equiv.) and amine (1.5–2.5 equiv.) were added sequentially under an inert atmosphere and subjected to sonication for 30 min at room temperature. Conversions and isolated yields correspond to the formation of the hydroamination product (3) and the reduced product (4), respectively, after purification by preparative TLC. S.R. = side reaction, indicated by complete lithium consumption without substrate conversion. See SI for complete experimental details and stoichiometric ratios. | ||||||
| 1 | THF + 0.5 mmol KI | 1.4 | 2.5 | >99 | 64 | 36 |
| 2 | THF | 1 | 1.5 | >99 | 77 | 23 |
| 3 | Triethylamine | 1 | 1.5 | 79 | 53 | 47 |
| 4 | Et2O | 1 | 1.5 | 77 | 95 | 5 |
| 5 | 1,4-Dioxane | 1 | 1.5 | 43 | 91 | 9 |
| 6 | Pentane | 1 | 1.5 | 65 | 61 | 39 |
| 7 | Toluene | 1 | 1.5 | 80 | 63 | 37 |
| 8 | Ethyl acetate | 1 | 1.5 | S.R. | — | — |
| 9 | Chloroform | 1 | 1.5 | S.R. | — | — |
| 10 | 2-MeTHF | 1 | 1.5 | >99 | >99 | Trace |
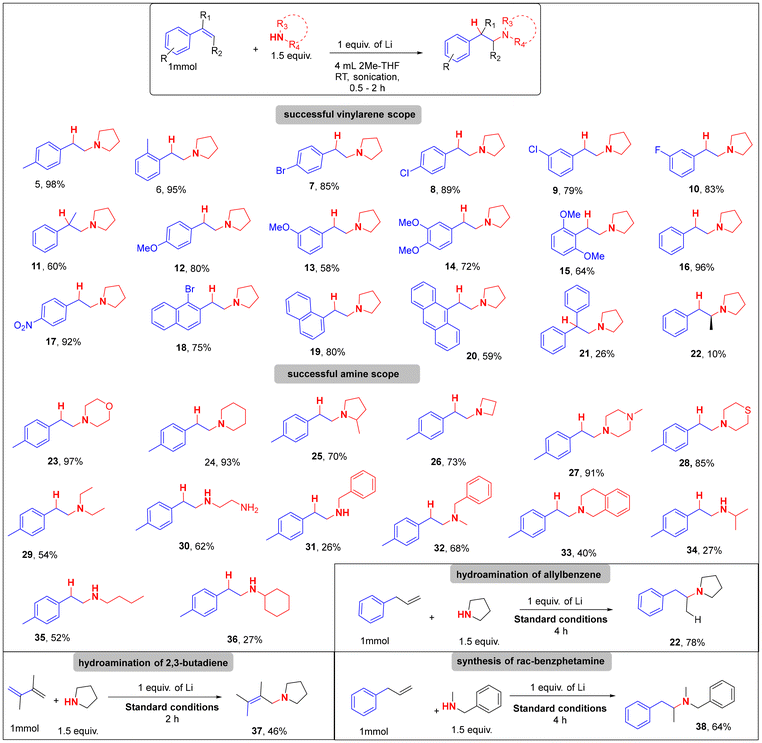
With optimized conditions in hand (1.0 equiv. Li, 1.5 equiv. amine, sonication, 2-MeTHF), we next evaluated the generality of the hydroamination protocol across a diverse array of olefins and amines (Table 2).29 The method demonstrated good compatibility with various vinylarenes, including mono- and di-substituted electron-donating groups (e.g., –OMe, –Me) in the ortho, meta, and para positions, affording the corresponding β-arylethylamines (5–6, 12–15) in moderate to excellent yields (58–98%). Notably, the sterically hindered 2,6-dimethoxystyrene yielded 15 in 64% yield, whereas 2,6-dimethylstyrene showed no reaction (see SI Fig. S3), indicating the potentially greater importance of electronic versus steric factors with respect to the alkene. All mono-ortho-substituted derivatives, however, performed well under the optimized conditions. Vinylarenes bearing electron-withdrawing groups (e.g., –Br, –F, –NO2, –Cl) were also efficiently converted. For instance, 4-bromo- and 4-chlorostyrenes yielded products 7 and 8 in 88% and 89% yield, respectively, while 3-chloro- and 3-fluorostyrenes afforded 9 and 10 in 79% and 87% yields. Interestingly, 3-fluorostyrene exhibited competing aminodefluorination when THF was used as solvent but gave exclusive hydroamination product in 2-MeTHF (see SI, Fig. S4), highlighting a significant role of solvent in the reaction's pathway. Importantly, several strongly electron-withdrawing substituents and electrophile-sensitive groups were not tolerated under these reducing conditions (see SI). α-Substituted styrenes showed variable outcomes; α-methylstyrene yielded 11 in 60% yield, while the more sterically encumbered α-phenylstyrene provided only 26% of 21, along with significant formation of the reduced side product 1,1-diphenylethane.
Encouragingly, fused aromatic olefins such as 1-vinyl- and 1-bromo-2-vinylnaphthalenes, as well as 9-vinylanthracene, underwent hydroamination smoothly, giving products 18–20 in 62–85% yields. In contrast, β-substituted styrenes proved more challenging. While cis-β-methylstyrene delivered 22 in only 10% yield after extended reaction time (6 h), the trans-isomer was completely unreactive. Similarly, trans-β-nitrostyrene yielded undefined polymeric material, and 1,2-dihydronaphthalene gave exclusively the reduced product (67%, see SI Fig. S3). These observations are consistent with literature reports on the steric and electronic difficulties β-substituted olefins pose in SET-initiated transformations, where hindered approach of the electron donor or radical intermediates can suppress productive reactivity.30–33 These findings further support the SET-based mechanistic proposal discussed below.
Following our exploration of the olefin scope, we next investigated the versatility of the amine component. A broad range of cyclic secondary amines was well-tolerated under the optimized conditions. Morpholine, piperidine, 2-methylpyrrolidine, azetidine, N-methylpiperazine, and thiomorpholine all delivered the corresponding hydroamination products (23–28) in excellent yields ranging from 70–97% (Table 2). Acyclic secondary amines, such as dibutylamine, were also compatible, affording product 29 in 54% yield. In contrast, primary amines generally gave lower yields despite high substrate conversion (80–100%, see SI Fig. S5). We attribute this to the additional N–H bond in primary amines increasing their susceptibility to competitive reduction by solvated electrons. For instance, benzylamine, isopropylamine, and cyclohexylamine afforded products 31, 34 and 36 in <30% yield, despite complete or near-complete conversion. Interestingly, butylamine showed a modestly improved yield (35, 45%), while ethylenediamine (EDA) provided product 30 in 62% yield within just 20 minutes. This rapid reactivity aligns with literature precedent citing EDA as a facilitator of solvated electron generation, suggesting a dual role as both nucleophile and redox mediator.34 Our method also tolerated benzylamines delivering both secondary and tertiary amines (31–33) in good to excellent yields.
Notably, transition-metal- and lanthanide-catalyzed hydroaminations are well known to perform poorly with unprotected primary amines, often showing no conversion or catalyst deactivation due to strong amine coordination and protonolysis under high-temperature conditions.1,35 In contrast, our protocol achieves measurable reactivity with these substrates under ambient, additive-free conditions, highlighting its broader substrate tolerance relative to previously reported systems. Anilines were unreactive under our optimized conditions, exhibiting partial conversion of the alkene but no detectable hydroamination product. Instead, reduction and dimerization pathways predominated (see SI, Section 5.5 and Fig. S6), consistent with the lower nucleophilicity and higher oxidation potential of anilines.
Next, we explore the applicability of our protocol with allylbenzenes and 1,3-dienes, which has been found to be challenging in transition-metal systems due to regioselectivity issues.1,7,36–40 Interestingly, allylbenzene underwent hydroamination with Markovnikov selectivity to afford product 22 in 78% yield. Under SET conditions, reduction of allylbenzene generates an allylic radical anion with delocalized charge and spin density across the alkene. Subsequent protonation occurs intermolecularly and preferentially at the terminal position, generating a more substituted carbon-centered radical that is intercepted in the C–N bond-forming step, leading to Markovnikov-selective hydroamination. 2,3-Dimethyl-1,3-butadiene afforded 37 (46% yield). While we did not exhaustively explore this class of substrates, we predict similar substrates will also work under these conditions. For example, we extend the applicability of this protocol to the high yielding (64%) synthesis of benzphetamine 38, a clinically used anorectic using allylbenzene and N-methylbenzylamine. The rapid reaction (≤30 min) under ambient conditions without additives or transition metals distinguishes this method from previous protocols.29,41
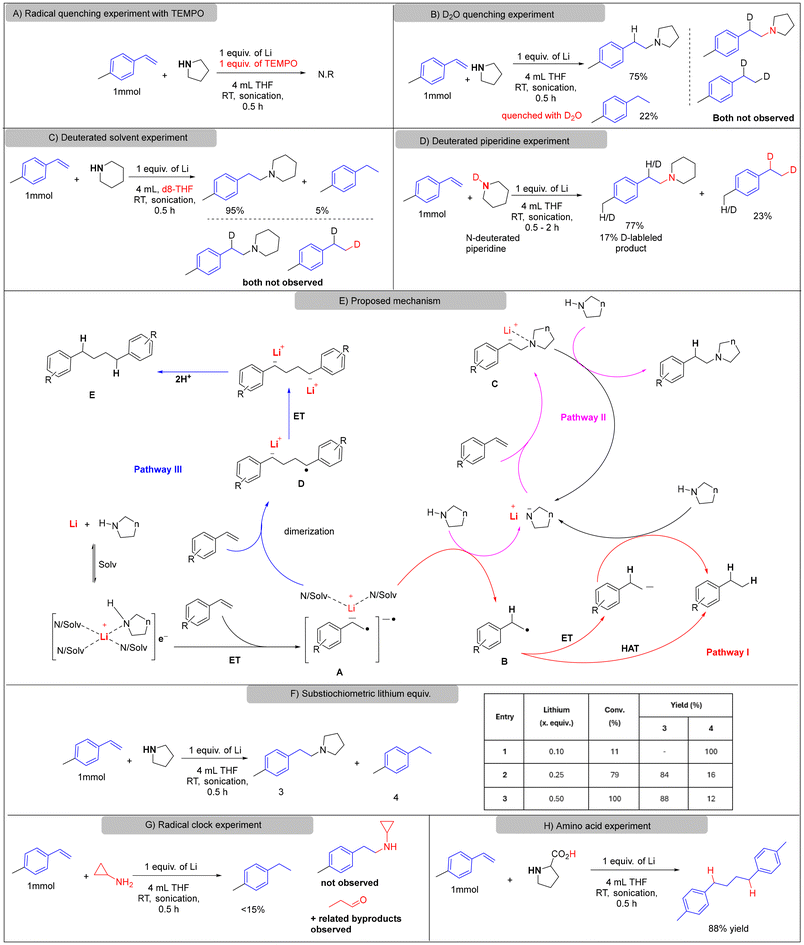
To elucidate the mechanism, we first performed radical inhibition experiments using 4-methylstyrene and piperidine as model substrates. The addition of TEMPO completely suppressed product formation, consistent with a radical-mediated process (Scheme 2A). D2O quenching and d8-THF experiments revealed no deuterium incorporation into either the hydroamination or reduction products (Scheme 2B and C), confirming that neither solvent nor water serve as the proton or hydrogen-atom source. In contrast, using N-deuterated piperidine led to deuterium incorporation in both products at the benzylic position, indicating that the amine functions as the hydrogen-atom donor in the hydrogen-atom transfer (HAT) step. A moderate kinetic isotope effect was observed when comparing the reaction rates of N–H versus N–D piperidine: the N–H reaction reached full conversion within 30 minutes, whereas the N–D analogue required 1 hour to reach completion (Scheme 2D; SI, Fig. S7–S16). This result implicates cleavage of the N–H bond as part of the rate-determining step.
These findings, together with literature precedent,9,23,27,34 are consistent with a SET/HAT/PT manifold (Scheme 2E). The very negative potential of Li+/Li (E° = −3.04 V vs. SHE; ≈−3.28 V vs. SCE),42,43 compared to reported styrene reduction potentials (ca. −2.58 V vs. SCE),44 supports the feasibility of initial SET from lithium-derived solvated electrons to the alkene to form radical anion A.25,34 Intermediate A may then follow one of three plausible pathways:
Pathway I: Proton transfer (PT) from the amine to form radical B, followed by hydrogen-atom transfer (HAT) from the amine N–H bond or a subsequent electron transfer (ET) and protonation step to furnish the reduced product 4; although this pathway may be attenuated in sterically hindered solvents due to solvation effects, it likely initiates the reaction.
Pathway II: PT concomitantly generates a lithium amide, which nucleophilically adds to the alkene to form aminated anion C, followed by protonation to deliver the hydroamination product;23 notably, base alone (e.g., NaH in the absence of Li0) did not promote hydroamination under otherwise comparable conditions (see SI), consistent with SET initiation being required.
Pathway III: Radical anion A couples with another alkene molecule to yield dimeric radical anion D, which undergoes ET and proton quenching to afford dimeric byproduct E.27 While the substoichiometric lithium experiments (Scheme 2F) are compatible with chain propagation and/or regeneration of the active reductant, we do not distinguish these possibilities here.
Radical inhibition with TEMPO completely suppressed product formation, confirming radical intermediacy. D2O and d8-THF quenching experiments showed no deuterium incorporation, indicating that neither water nor solvent acts as the hydrogen source. In contrast, reactions with N-deuterated piperidine afforded deuterium-labeled products and a measurable kinetic isotope effect, implicating the amine N–H bond in the rate-determining HAT step. The proposed mechanism involves three competing pathways: Pathway I, SET/PT/HAT sequence leading to reduction; Pathway II, SET/PT/nucleophilic addition affording hydroamination; and Pathway III, radical coupling yielding dimeric byproducts. Substoichiometric lithium experiments confirmed its catalytic role, while radical clock and amino-acid studies further validated the amine-mediated radical pathway.
The catalytic role of lithium was confirmed by varying its loading, with 0.50 equivalents achieving complete conversion (Scheme 2F). Moreover, radical clock experiments with cyclopropylamine (Scheme 2G) further support an amine-mediated radical HAT/ET process (Scheme 2G, SI, Fig. S17), while the high-yield formation of E from proline (88% yield, Scheme 2H, SI Fig. S18) corroborates the radical anion A as a key reactive intermediate.
In summary, we have developed a mild, transition metal-free protocol for the hydroamination of olefins using solvated electrons generated from lithium and sonication in 2-MeTHF. This method demonstrates good compatibility with substituted vinylarenes and cyclic secondary amines, along with rapid reaction times without the need for additives or external oxidants/reductants. Mechanistic investigations support a SET–HAT pathway in which lithium-amido complexes play dual roles as both HAT donors and nucleophiles. The selective synthesis of β-arylethylamines and the anti-Markovnikov hydroamination of allylarenes underscore the synthetic potential of this strategy. By leveraging a green solvent and avoiding precious metals, this protocol offers a sustainable platform for amine synthesis with significant relevance to medicinal and industrial chemistry.
Author contributions
A. W. W. carried out the experimental work and data analysis and prepared the manuscript and SI. K. G. supervised the research, provided conceptual guidance, and reviewed and edited the manuscript. Both authors contributed to the final review of the manuscript and approved the final version.Conflicts of interest
There are no conflicts to declare.Data availability
Data are available within the article and supplementary information (SI). Supplementary information: experimental details, including the optimization of the synthetic method, synthesis and characterization of all the starting materials and products reported in this study, and the mechanistic studies. NMR spectra of all the products, and isolation. See DOI: https://doi.org/10.1039/d5ob01936b.Acknowledgements
The authors thank Dr Luke Fulton for assistance with NMR spectroscopy and Dr Adam Graichen for support with high-resolution mass spectrometry. We also acknowledge the University of Connecticut for access to research facilities and instrumentation.References
- J. Escorihuela, A. Lledós and G. Ujaque, Anti-Markovnikov Intermolecular Hydroamination of Alkenes and Alkynes: A Mechanistic View, Chem. Rev., 2023, 123, 9139–9203 CrossRef CAS PubMed.
- J. Penafiel, L. Maron and S. Harder, Early main group metal catalysis: how important is the metal?, Angew. Chem., 2015, 127, 203–208 CrossRef.
- M. Beller, J. Seayad, A. Tillack and H. Jiao, Catalytic Markovnikov and anti–Markovnikov functionalization of alkenes and alkynes: recent developments and trends, Angew. Chem., Int. Ed., 2004, 43, 3368–3398 CrossRef CAS PubMed.
- L. Davin, A. Hernán-Gómez, C. McLaughlin, A. R. Kennedy, R. McLellan and E. Hevia, Alkali metal and stoichiometric effects in intermolecular hydroamination catalysed by lithium, sodium and potassium magnesiates, Dalton Trans., 2019, 48, 8122–8130 Search PubMed.
- A. A. Kissel, T. V. Mahrova, D. M. Lyubov, A. V. Cherkasov, G. K. Fukin, A. A. Trifonov, I. D. Rosal and L. Maron, Metallacyclic yttrium alkyl and hydrido complexes: synthesis, structures and catalytic activity in intermolecular olefin hydrophosphination and hydroamination, Dalton Trans., 2015, 44, 12137–12148 Search PubMed.
- L. Huang, M. Arndt, K. Gooßen, H. Heydt and L. J. Goossen, Late transition metal-catalyzed hydroamination and hydroamidation, Chem. Rev., 2015, 115, 2596–2697 CrossRef CAS PubMed.
- A. M. Johns, M. Utsunomiya, C. D. Incarvito and J. F. Hartwig, A highly active palladium catalyst for intermolecular hydroamination. Factors that control reactivity and additions of functionalized anilines to dienes and vinylarenes, J. Am. Chem. Soc., 2006, 128, 1828–1839 CrossRef CAS PubMed.
- P. A. Dub, M. Rodriguez-Zubiri, J.-C. Daran, J.-J. Brunet and R. Poli, Platinum-catalyzed ethylene hydroamination with aniline: Synthesis, characterization, and studies of intermediates, Organometallics, 2009, 28, 4764–4777 CrossRef CAS.
- J. Seayad, A. Tillack, C. G. Hartung and M. Beller, Base-Catalyzed Hydroamination of Olefins: An Environmentally Friendly Route to Amines, Adv. Synth. Catal., 2002, 344, 795–813 CrossRef CAS.
- K. Muniz, A. Lishchynskyi, J. Streuff, M. Nieger, E. C. Escudero-Adán and M. M. Belmonte, Metal–ligand bifunctional activation and transfer of N–H bonds, Chem. Commun., 2011, 47, 4911–4913 RSC.
- A. L. Casalnuovo, J. C. Calabrese and D. Milstein, Rational design in homogeneous catalysis. Iridium(I)-catalyzed addition of aniline to norbornylene via nitrogen-hydrogen activation, J. Am. Chem. Soc., 1988, 110, 6738–6744 CrossRef CAS.
- E. Khaskin, M. A. Iron, L. J. Shimon, J. Zhang and D. Milstein, N–H activation of amines and ammonia by Ru via metal–ligand cooperation, J. Am. Chem. Soc., 2010, 132, 8542–8543 CrossRef CAS PubMed.
- M. Utsunomiya, R. Kuwano, M. Kawatsura and J. F. Hartwig, Rhodium-catalyzed anti-Markovnikov hydroamination of vinylarenes, J. Am. Chem. Soc., 2003, 125, 5608–5609 CrossRef CAS PubMed.
- M. Kawatsura and J. F. Hartwig, Palladium-catalyzed intermolecular hydroamination of vinylarenes using arylamines, J. Am. Chem. Soc., 2000, 122, 9546–9547 CrossRef CAS.
- S. Park, J. Jeong, K. Fujita, A. Yamamoto and H. Yoshida, Anti-Markovnikov Hydroamination of Alkenes with Aqueous Ammonia by Metal-Loaded Titanium Oxide Photocatalyst, J. Am. Chem. Soc., 2020, 142, 12708–12714 CrossRef CAS PubMed.
- S. Ma, C. K. Hill, C. L. Olen and J. F. Hartwig, Ruthenium-Catalyzed Hydroamination of Unactivated Terminal Alkenes with Stoichiometric Amounts of Alkene and an Ammonia Surrogate by Sequential Oxidation and Reduction, J. Am. Chem. Soc., 2021, 143, 359–368 Search PubMed.
- S. Ma and J. F. Hartwig, Progression of Hydroamination Catalyzed by Late Transition-Metal Complexes from Activated to Unactivated Alkenes, Acc. Chem. Res., 2023, 56, 1565–1577 Search PubMed.
- A. L. Reznichenko, H. N. Nguyen and K. C. Hultzsch, Asymmetric intermolecular hydroamination of unactivated alkenes with simple amines, Angew. Chem., Int. Ed., 2010, 49, 8984–8987 Search PubMed.
- J.-S. Ryu, G. Y. Li and T. J. Marks, Organolathanide-catalyzed regioselective intermolecular hydroamination of alkenes, alkynes, vinylarenes, di-and trivinylarenes, and methylenecyclopropanes. Scope and mechanistic comparison to intramolecular cyclohydroaminations, J. Am. Chem. Soc., 2003, 125, 12584–12605 Search PubMed.
- C. Brinkmann, A. G. M. Barrett, M. S. Hill and P. A. Procopiou, Heavier Alkaline Earth Catalysts for the Intermolecular Hydroamination of Vinylarenes, Dienes, and Alkynes, J. Am. Chem. Soc., 2012, 134, 2193–2207 CrossRef CAS PubMed.
- S. Harder, From limestone to catalysis: application of calcium compounds as homogeneous catalysts, Chem. Rev., 2010, 110, 3852–3876 Search PubMed.
- H. Liu, S. Saha and M. S. Eisen, Recent advances in organo- lanthanides and actinides mediated hydroaminations, Coord. Chem. Rev., 2023, 493, 215284 Search PubMed.
- F. F. Mulks, L. J. Bole, L. Davin, A. Hernán-Gómez, A. Kennedy, J. García-Álvarez and E. Hevia, Ambient Moisture Accelerates Hydroamination Reactions of Vinylarenes with Alkali-Metal Amides under Air, Angew. Chem., Int. Ed., 2020, 59, 19021–19026 CrossRef CAS PubMed.
- A. Tortajada and E. Hevia, Room-Temperature Intermolecular Hydroamination of Vinylarenes Catalyzed by Alkali-Metal Ferrate Complexes, ACS Org. Inorg. Au, 2025, 5, 62–68 CrossRef CAS PubMed.
- A. W. Williams and K. M. Gilmore, Transition–Metal Free Amination and Hydrodefluorination of Aryl Fluorides Promoted by Solvated Electrons, Chem. – Eur. J., 2024, 30, e202403410 CrossRef CAS PubMed.
- U. Melamed and B.-A. Feit, A novel reduction of activated olefins by lithium amides, J. Chem. Soc., Perkin Trans. 1, 1980, 1267–1269 RSC.
- Y. Jiang and H. Yorimitsu, Taming Highly Unstable Radical Anions and 1,4-Organodilithiums by Flow Microreactors: Controlled Reductive Dimerization of Styrenes, JACS Au, 2022, 2, 2514–2521 CrossRef CAS PubMed.
- C. C. Lai and G. R. Freeman, Solvent effects on the reactivity of solvated electrons with organic solutes in methanol/water and ethanol/water mixed solvents, J. Phys. Chem., 1990, 94, 302–308 CrossRef CAS.
- A. Allen and R. Ely, Review: Synthetic Methods for Amphetamine. Link accessed 2026-3-5: http://www.nwafs.org/newsletters/SyntheticAmphetamine.pdf Search PubMed.
- M. Patel, B. Desai, A. Sheth, B. Z. Dholakiya and T. Naveen, Recent Advances in Mono- and Difunctionalization of Unactivated Olefins, Asian J. Org. Chem., 2021, 10, 3201–3232 CrossRef CAS.
- N. P. van Leest, F. J. de Zwart, M. Zhou and B. de Bruin, Controlling Radical-Type Single-Electron Elementary Steps in Catalysis with Redox-Active Ligands and Substrates, JACS Au, 2021, 1, 1101–1115 CrossRef CAS PubMed.
- W. Zhou, I. A. Dmitriev and P. Melchiorre, Reductive Cross-Coupling of Olefins via a Radical Pathway, J. Am. Chem. Soc., 2023, 145, 25098–25102 CrossRef CAS PubMed.
- N. P. van Leest, F. J. de Zwart, M. Zhou and B. de Bruin, Controlling Radical-Type Single-Electron Elementary Steps in Catalysis with Redox-Active Ligands and Substrates, JACS Au, 2021, 1, 1101–1115 CrossRef CAS PubMed.
- J. Burrows, S. Kamo and K. Koide, Scalable Birch reduction with lithium and ethylenediamine in tetrahydrofuran, Science, 2021, 374, 741–746 CrossRef CAS PubMed.
- J. F. Hartwig, S. Richards, D. Barañano and F. Paul, Influences on the Relative Rates for C–N Bond-Forming Reductive Elimination and β-Hydrogen Elimination of Amides. A Case Study on the Origins of Competing Reduction in the Palladium-Catalyzed Amination of Aryl Halides, J. Am. Chem. Soc., 1996, 118, 3626–3633 CrossRef CAS.
- A. M. Canfield, D. Rodina and S. M. Paradine, Dienes as Versatile Substrates for Transition Metal-Catalyzed Reactions, Angew. Chem., Int. Ed., 2024, 63, e202401550 CrossRef CAS PubMed.
- S. Ma and J. F. Hartwig, Progression of Hydroamination Catalyzed by Late Transition-Metal Complexes from Activated to Unactivated Alkenes, Acc. Chem. Res., 2023, 56, 1565–1577 CrossRef CAS PubMed.
- P. Pal, S. Show, S. Das, S. Bhandari, S. Nandy, Md. A. Sk and R. K. Nandi, Recent advancement on metal free hydroamination reaction of C–C multiple bonds, Tetrahedron, 2025, 179, 134625 CrossRef CAS.
- S. Pradhan, S. Das, G. Kumar and I. Chatterjee, Transition-Metal-Free Regioselective Intermolecular Hydroamination of Conjugated 1,3-Dienes with Heterocyclic Amines, Org. Lett., 2022, 24, 2452–2456 CrossRef CAS PubMed.
- X.-H. Yang, A. Lu and V. M. Dong, Intermolecular Hydroamination of 1,3-Dienes To Generate Homoallylic Amines, J. Am. Chem. Soc., 2017, 139, 14049–14052 CrossRef CAS PubMed.
- S. Dey and A. Sudalai, A concise enantioselective synthesis of (R)-selegiline, (S)-benzphetamine and formal synthesis of (R)-sitagliptin via electrophilic azidation of chiral imide enolates, Tetrahedron: Asymmetry, 2015, 26, 67–72 CrossRef CAS.
- Q. Wang, B. Liu, Y. Shen, J. Wu, Z. Zhao, C. Zhong and W. Hu, Confronting the Challenges in Lithium Anodes for Lithium Metal Batteries, Adv. Sci., 2021, 8, 2101111 CrossRef CAS PubMed.
- S. Xiong, L. Nyholm, A. Matic and C. Zhang, Lithium electrodeposition for energy storage: filling the gap between theory and experiment, Mater. Today Energy, 2022, 28, 101060 CrossRef CAS.
- S. Gambino, A. Gennaro, G. Filardo, G. Silvestri and E. Vianello, Electrochemical carboxylation of styrene, J. Electrochem. Soc., 1987, 134, 2172 CrossRef CAS.
| This journal is © The Royal Society of Chemistry 2026 |